DOI:
10.1039/D5RA08856A
(Paper)
RSC Adv., 2026,
16, 11100-11104
Rational synthesis of pyrazolopyrimidines via cyclocondensation of ynones obtained from the Sonogashira reaction
Received
17th November 2025
, Accepted 28th January 2026
First published on 25th February 2026
Abstract
Pyrazolopyrimidines combine the structural and electronic properties of both pyrazole and pyrimidine, imparting unique characteristics that make them valuable in medicinal chemistry and drug discovery. We successfully developed an atom economic protocol for the synthesis of a series of pyrazolo-fused pyrimidines by employing various ynones and 3-aminopyrazole using K2CO3 as a base in ethanol. The reactions proceeded under milder conditions, furnishing the desired products in moderate to excellent yields.
Introduction
Heterocyclic molecules are fundamental to many scientific and industrial applications.1 Understanding and synthesizing heterocyclic compounds continue to be a significant area of research with profound implications in various fields. In particular, pyrazolo-fused pyrimidines display unique properties and a diverse range of biological activities due to the cumulative effect of pyrazole and pyrimidine moieties, making them indispensable in drug development.2–5 Pyrazolo-fused pyrimidine-based drugs are present in the market, like ocinaplon (A) for anxiety, zaleplon (B) and indiplon (E) as sedatives, presatovir (C) as an anti-infective agent and pyrazophos (D) as a fungicide (Fig. 1).6–17 Therefore, the scientific community has put substantial effort into the experimental research and synthesis of these compounds.18–20 Among the reported methods, protocols using environmentally benign conditions21,22 are in demand and require future synthesis.
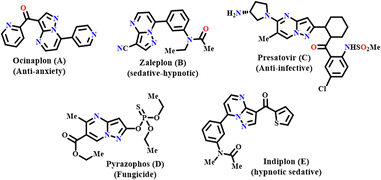 |
| | Fig. 1 Pyrazolo-fused pyrimidine-based marketed drugs. | |
A lot of interest has been shown in the efficient synthesis of these molecules by applying sustainable and environmentally friendly techniques, including atom-economic processes.23 In addition to adhering to the principles of green chemistry, these approaches have the potential to provide potent anticancer medicines.
Keeping the biological importance of pyrazolo-fused pyrimidines in view, conventional synthetic methods have been developed for their synthesis. Carbonyl compounds and aminopyrazole using Rh and Cu catalysts at 150 °C and 120 °C were employed for the synthesis of pyrazolopyrimidines in 2018 and 2023, respectively (Scheme 1a and c).24,25 In 2020, Yu and group reported the synthesis of pyrazolopyrimidines in the presence of FeNi3/gold nanoparticles, supported by magnetic ionic gelation under solvent-free conditions (Scheme 1b).26 Besides, many other reports have involved conventional methods.27–30 Previously, we also reported a transition-metal-free approach for the chemo-selective synthesis of pyrazolo-fused pyrimidines and their derivatives from acetophenones and aminopyrazole by employing acetic acid as a solvent at 80 °C for 24 h (Scheme 1d).31 Considering the importance of a greener approach, we attempted to develop a method that uses a greener solvent. In this process, we established a novel, kinetically efficient, transition-metal-free methodology for the synthesis of pyrazolo pyrimidines by employing aminopyrazole with various naphthalenes and thiophenes based on ynones in ethanol.
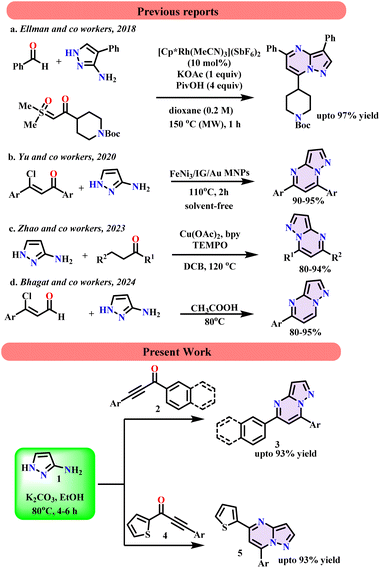 |
| | Scheme 1 Previous reports vs present work. | |
Results and discussion
Optimization of reaction conditions
We conducted a comprehensive investigation of the reactions between ynone (2h) and 3-amino pyrazole (1) to establish robust synthetic parameters for the efficient synthesis of pyrazolopyrimidines. Through systematic analysis, we explored the effects of base strength, solvent polarity, reaction temperature and reaction time on reaction efficiency. After careful modulation of these variables, we were successful in identifying the optimal reaction conditions that maximize our yield and minimise ecological footprint. We started with the optimisation of the reaction with ynone (1.0 eq.) and 3-aminopyrazole (1, 1.5 eq.) using KOH as a base in DMSO at 100 °C for 12 h (Table 1, entry 1). Contrary to our expectations, the reaction did not proceed to completion, and the desired product was obtained in a low yield of 12%. We further increased the amount of base and obtained an 18% yield (3h) (Table 1, entry 2). Re-evaluating, we carried out the reaction in K2CO3 in DMSO and DMF for 12 hours, but the anticipated increase in yield was not achieved, as the observed results fell short of our expectations (Table 1, entries 3 and 4). We further investigated the reaction in water at 80 °C for 12 hours (Table 1, entry 5), resulting in a slight increase in the yield of the desired product. Upon further optimising the reaction conditions by changing the solvent to ethanol, we successfully obtained a 56% yield for our desired product (Table 1, entry 6). Continuing our optimisation, by maintaining the reaction temperature at 80 °C, and increasing the amount of K2CO3, we successfully obtained 85% yield of our product (Table 1, entry 7). Again, modifying and performing the reaction at room temperature in the same solvent gave a similar yield; however, the reaction took 22 hours to complete (Table 1, entry 8). At 0 °C, no successful result was obtained (Table 1, entry 9).
Table 1 Optimization of reaction conditionsa,b
| S. no. |
Base |
Solvent |
Temp |
Time |
Yield |
| Reaction conditions: reactions were carried out using substituted ynones (2, 1 eq.) and 3-amino pyrazole (1, 1.5 eq.) in an appropriate solvent (2 mL). Isolated yield. |
| 1 |
KOH (1.0 mmol) |
DMSO |
100 |
12 |
12 |
| 2 |
KOH (2.0 mmol) |
DMSO |
100 |
12 |
18 |
| 3 |
K2CO3 (1.0 mmol) |
DMSO |
100 |
12 |
10 |
| 4 |
K2CO3 (1.0 mmol) |
DMF |
100 |
12 |
15 |
| 5 |
K2CO3 (1.0 mmol) |
H2O |
80 |
12 |
35 |
| 6 |
K2CO3 (1.0 mmol) |
EtOH |
80 |
10 |
57 |
| 7 |
K2CO3(2.0 mmol) |
EtOH |
80 |
6 |
85 |
| 8 |
K2CO3 (2.0 mmol) |
EtOH |
RT |
24 |
68 |
| 9 |
K2CO3 (2.0 mmol) |
EtOH |
0 |
24 |
— |
The substrate scope of the reaction was investigated with a wide range of ynones (2) and 3-amino pyrazole (1) in order to create transition metal-free chemo-selective C–N linkages after determining the ideal conditions (Scheme 2). The desired products were obtained in good-to-excellent yields with great functional group tolerance, such as –F, –Cl, –OMe, –NMe2, –CF3, and CN. Further, the reaction was compatible with thiophene-substituted ynones (4) (Scheme 3). We employed the method shown in Scheme 3 to produce equivalent pyrazolo-fused pyrimidines (5) in good yields.
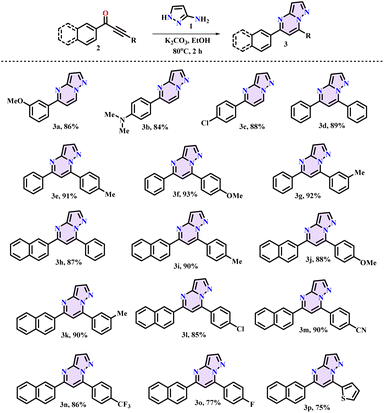 |
| | Scheme 2 Synthesis of benzene-/naphthalene-substituted pyrazolo pyrimidines (3a–3p). | |
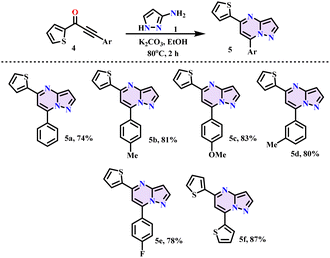 |
| | Scheme 3 Synthesis of thiophene-substituted pyrazolo pyrimidines (5a–5f). | |
Plausible mechanism
After successful analysis of the transition metal-free C–N bond formation process for the synthesis of pyrazolo-fused pyrimidines, a tenable mechanism was put forth (Scheme 4). In this plausible mechanism, the reaction started with the abstraction of an acidic proton in the presence of a base, which attacked the allylic carbon of the ynone (2) to form the allene intermediate (7). A similar intermediate was observed by Verma et al. in 2012.32 This intermediate underwent proton transfer, followed by the cyclization reaction (9), which led to the final pyrazolo fused pyrimidines (3) with the elimination of a water molecule. We synthesised the final products 3(a–p) and 5(a–f), and their structures were confirmed by the 1H and 13C NMR and HRMS spectral data. These results support the assertion that the proposed reaction mechanism proceeds through cyclocondensation, as shown in Scheme 4.
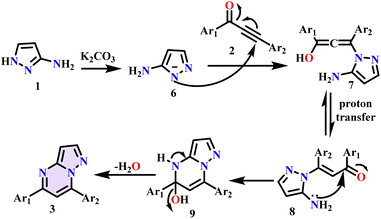 |
| | Scheme 4 Plausible mechanism for the synthesis of pyrazolo pyrimidines. | |
Calculation of green chemistry metrics for the scale-up reaction of product (3):
A green chemistry matrix is calculated based on the following parameters:
(1) E-factor or environmental factor.
(2) Atom economy (AE).
(3) Product mass intensity (PMI).
(4) Reaction mass efficiency (RME).
E-factor: E-factor is defined as the ratio of the mass of waste to the mass of the product.
| E-Factor = Amount of waste/Amount of product |
| Amount of reactants: ynone (1) = 1.0 mmol and 3-aminopyrazole (2) = 1.5 mmol |
| Total amount of reactants (1h + 2) = (256.30 mg mmol−1 × 1.0 mmol) + (83.09 mg mmol−1 × 1.5 mmol) = 256.30 mg + 124.64 mg = 380.94 mg = 0.381 g |
| Amount of product (3h) = 0.257 g |
| Amount of waste = 0.381 g − 0.257 g = 0.124 g |
| E-factor = 0.124/0.257 = 0.48 (Ideal value of E-factor is considered zero.) |
Process mass index (PMI): PMI is defined as the total mass used in a chemical process divided by the mass of the product.
| PMI = Σ(mass of stoichiometric reactants + solvent)/mass of product (3h) |
| PMI = (256.30 mg × 1.0) + (83.09 mg × 1.5)/257 = 1.48 |
OR
| PMI = E-factor + 1 = 0.48 + 1 = 1.48 |
Atomic economy (AE): AE of a chemical reaction is a measure of the efficiency of that reaction with regard to how many atoms from the starting materials reside within the product. The ideal value of the AE factor is 100% (i.e., all atoms from the starting materials reside in the product).
| Atom economy (AE) = MW of product ÷ Σ (MW of stoichiometric reactants) × 100 |
| Molecular weight of product (3h) = 321.38 g mol−1 |
| Molecular weight of stoichiometric reactants (1h + 2) = (256.30)(1.0) + (83.09)(1.5) = 380.94 g mol−1 |
| Atom economy (AE) = 321.38 × 100/380.94 = 84.4% |
Reaction mass efficiency (RME): Reaction mass efficiency is defined as the mass of the product divided by the sum of the total mass of the stoichiometric reactants. The value of the RME varies from 0 to 100%. A larger number of RME is considered better as it is the measure of “cleanness” of the reaction.
| Reaction mass efficiency = mass of product/Σ(mass of stoichiometric reactants) × 100 |
| Mass of product (3h) = 0.257 g |
| Total mass of reactants (1h + 2) = (256.30 mg mmol−1 × 1.0 mmol) + (83.09 mg mmol−1 × 1.5 mmol) = 256.30 mg + 124.64 mg = 380.94 mg = 0.381 g |
RME = (0.257 ÷ 0.381) × 100 = 67.4%. The main text of the article should appear here with headings as appropriate.
Conclusions
Pyrazolo-fused pyrimidine heterocycles have a variety of biological functions and possible therapeutic uses that emphasise their medicinal importance. Ynones obtained from Sonogashira coupling reactions were used to create a new, environmentally friendly, transition-metal-free synthesis technique that ensures atom economy.
Author contributions
Ms. Nikita Goel: writing – review & editing, writing –original draft, visualization, validation, conceptualization, Prof. Sunita Bhagat: visualization, supervision, investigation and Dr Pradeep Kumar and Dr Poonam Kumari: writing – review & editing.
Conflicts of interest
There are no conflicts to declare.
Data availability
Experimental details and spectral data for all new compounds have been included as part of the supplementary information (SI). Supplementary information: ref. 33–36 are cited in the SI. See DOI: https://doi.org/10.1039/d5ra08856a.
Acknowledgements
N. G. is thankful to CSIR-India, for fellowship. P. K., S. B. are thankful to SERB-CRG, India, for providing financial support (CRG/2021/007123) and P. K. is thankful to DST for WISE-PDF (DST/WISE-PDF/CS-136/2023). The authors are thankful to ARSD College, University of Delhi, and USIC, University of Delhi, for providing infrastructure and instrumentation facilities.
Notes and references
- S. R. Tiwari, P. Mishra and J. Abraham, Neratinib, a novel HER2-targeted tyrosine kinase inhibitor, Clin. Breast Cancer, 2016, 16(5), 344–348 CrossRef CAS PubMed.
- S. Son, A. Elkamhaey, A. R. Gul, A. A. Al-Karmalawy, R. Alnajjar, A. Abdeen, S. F. Ibrahim, S. O. Alsohammari, Q. A. Alshammari, W. J. Choi and T. J. Park, Development of new TAK-285 derivatives as potent EGFR/HER2 inhibitors possessing antiproliferative effects against 22RV1 and PC3 prostate carcinoma cell lines, J. Enzyme Inhib. Med. Chem., 2023, 38(1), 2202358 CrossRef PubMed.
- D. Pollok and S. R. Waldvogel, Electro-organic synthesis–a 21st century technique, Chem. Sci., 2020, 11, 12386–12400 RSC.
- R. A. Sheldon, I. Arends and U. Hanefeld, Green Chemistry and Catalysis, Wiley−VCH, Weinheim, 2007, vol. 21 Search PubMed.
- R. A. Sheldon, E factors, green chemistry and catalysis: an odyssey, Chem. Commun., 2008, 29, 3352–3365 RSC.
- S. Fustero, M. Sánchez-Roselló, P. Barrio and A. Simon-Fuentes, From 2000 to mid-2010: A fruitful decade for the synthesis of pyrazoles, Chem. Rev., 2011, 111, 6984–7034 CrossRef CAS PubMed.
- K. Karrouchi, S. Radi, Y. Ramli, J. Taoufik, Y. N. Mabkhot, F. A. Al-Aizari and M. H. Ansar, Synthesis and pharmacological activities of pyrazole derivatives: A review, Molecules, 2018, 23, 134 CrossRef PubMed.
- D. A. Schmidt, Recent advances in the chemistry of pyrazoles. Properties, biological activities, and syntheses, Curr. Org. Chem., 2011, 15, 1423 CrossRef.
- M. R. Bhosle, J. R. Mali, S. Pal, A. K. Srivastava and R. A. Mane, Synthesis and antihyperglycemic evaluation of new 2-hydrazolyl-4-thiazolidinone-5-carboxylic acids having pyrazolyl pharmacophores, Bioorg. Med. Chem. Lett., 2014, 24, 2651–2654 CrossRef CAS PubMed.
- M. A. A. El-Sayed, N. I. Abdel-Aziz, A. A. M. Abdel-Aziz, A. S. El-Azab and K. E. H. ElTahir, Synthesis, biological evaluation and molecular modeling study of pyrazole and pyrazoline derivatives as selective COX-2 inhibitors and anti-inflammatory agents, Bioorg. Med. Chem., 2012, 20, 3306–3316 CrossRef CAS PubMed.
- S. R. Cox, S. P. Lesman, J. F. Boucher, M. J. Krautmann, B. D. Hummel, M. Savides, S. Marsh, A. Fielder and M. R. Stegemann, The pharmacokinetics of mavacoxib, a long-acting COX-2 inhibitor, in young adult laboratory dogs, J. Vet. Pharmacol. Ther., 2010, 33, 461–470 CrossRef CAS PubMed.
- A. A. Gomez, A. Godoy and J. Portilla, Functional pyrazolo [1, 5-a] pyrimidines: Current approaches in synthetic transformations and uses as an antitumor scaffold, Molecules, 2021, 26, 2708 CrossRef PubMed.
- J. Ren, S. Ding, X. Li, R. Bi and Q. Zhao, An approach for the synthesis of pyrazolo [1, 5-a] pyrimidines via Cu (II)-catalyzed [3+ 3] annulation of saturated ketones with aminopyrazoles, J. Org. Chem., 2021, 86, 12762–12771 CrossRef CAS PubMed.
- S. B. Annes, K. Perumal, K. Anandhakumar, B. Shankar and S. Ramesh, Transition-metal-free dehydrogenation of benzyl alcohol for C–C and C–N bond formation for the synthesis of pyrazolo [3, 4-b] pyridine and pyrazoline derivatives, J. Org. Chem., 2023, 88, 6039–6057 CrossRef CAS PubMed.
- P. Kaswan, K. Pericherla, D. Purohit and A. Kumar, Synthesis of 5, 7-diarylpyrazolo [1, 5-a] pyrimidines via KOH mediated tandem reaction of 1H-pyrazol-3-amines and chalcones, Tetrahedron Lett., 2015, 56, 549–553 CrossRef CAS.
- S. B. Annes, K. Perumal, K. Anandhakumar, B. Shankar and S. Ramesh, Transition-metal-free dehydrogenation of benzyl alcohol for C–C and C–N bond formation for the synthesis of pyrazolo [3, 4-b] pyridine and pyrazoline derivatives, J. Org. Chem., 2023, 88, 6039–6057 CrossRef CAS PubMed.
- B. J. Newhouse, J. D. Hansen, J. Grina, M. Welch, G. Topalov, N. Littman, M. Callejo, M. Martinson, S. Galbraith, E. R. Laird and B. J. Brandhuber, Non-oxime pyrazole-based inhibitors of B-Raf kinase, Bioorg. Med. Chem. Lett., 2021, 21, 3488–3492 CrossRef PubMed.
- A. Rahmouni, S. Souiei, M. A. Belkacem, A. Romdhane, J. Bouajila and H. B. Jannet, Synthesis and biological evaluation of novel pyrazolopyrimidines derivatives as anticancer and anti-5-lipoxygenase agents, Bioorg. Chem., 2016, 66, 160–168 CrossRef CAS PubMed.
- N. H. Metwally, T. H. Koraa and S. M. H. Sanad, Green one-pot synthesis and in vitro antibacterial screening of pyrano[2,3-c] pyrazoles, 4H-chromenes and pyrazolo[1,5-a]pyrimidines using biocatalyzed pepsin, Synth. Commun., 2022, 52, 1139–1154 Search PubMed.
- M. Nayak, N. Rastogi and S. Batra, Copper-Catalyzed Cascade Reaction of 4-Iodopyrazole Derivatives with Amidines for the Synthesis of Pyrazolo[4,3-d]pyrimidine Derivatives, Eur. J. Org Chem., 2012, 1360–1366 CrossRef CAS.
- R. Nishanth, S. Jena, M. Mukherjee, B. Maiti and K. Chanda, Green synthesis of biologically active heterocycles of medicinal importance: a review, Environ. Chem., 2021, 19, 3315–3358 Search PubMed.
- P. Phukan and D. Sarma, Synthesis of medicinally relevant scaffolds - triazoles and pyrazoles in green solvent ionic liquids, Curr. Org. Chem., 2021, 25, 1523–1538 CrossRef CAS.
- T. Chatterjee, P. Pattanayak, A. N. Satyanarayana and N. Mukherjee, Recent advances in developing highly atom-economic C–H annulation reactions in water, Curr. Opin. Green Sustain. Chem., 2023, 41, 100826 Search PubMed.
- J. Ren, S. Ding, X. Li, R. Bi and Q. Zhao, An approach for the synthesis of pyrazolo [1, 5-a] pyrimidines via Cu (II)-catalyzed [3+ 3] annulation of saturated ketones with aminopyrazoles, J. Org. Chem., 2021, 86, 12762–12771 CrossRef CAS PubMed.
- G. L. Hoang, A. D. Streit and J. A. Ellman, Three-component coupling of aldehydes, aminopyrazoles, and sulfoxonium ylides via rhodium (III)-catalyzed imidoyl C–H activation: synthesis of pyrazolo [1, 5-a] pyrimidines, J. Org. Chem., 2018, 83, 15347–15360 CrossRef CAS PubMed.
- L. Yu, S. Xing, K. Zheng and S. M. Sadeghzadeh, Synthesis of pyrazolopyrimidines in mild conditions by gold nanoparticles supported on magnetic ionic gelation in aqueous solution, Appl. Organomet. Chem., 2020, 34, 5663–5672 CrossRef.
- A. E. Rashad, M. I. Hegab, R. E. Abdel-Megeid, J. A. Micky and F. M. Abdel-Megeid, Synthesis and antiviral evaluation of some new pyrazole and fused pyrazolopyrimidine derivatives, Bioorg. Med. Chem., 2008, 16, 7102–7106 CrossRef CAS PubMed.
- A. Rahmouni, A. Romdhane, A. B. Said, K. Majouli and H. B. Jannet, Synthesis and antiviral evaluation of some new pyrazole and fused pyrazolopyrimidine derivatives, Turk. J. Chem., 2014, 38, 210–221 CrossRef CAS.
- A. E. Rashad, A. H. Shamroukh, R. E. Abdel-Megeid and H. S. Ali, Synthesis and isomerization of some novel pyrazolopyrimidine and pyrazolotriazolopyrimidine derivatives, Molecules, 2014, 19, 5459–5469 CrossRef PubMed.
- S. Y. Ewieda, A. Sonousi, A. M. Kamal and M. K. Abdelhamid, Design, synthesis, and cytotoxicity screening of novel pyrazolopyrimidines over Renal Cell Carcinoma (UO-31 Cells) as p38α inhibitors, and apoptotic cells inducing activities, Eur. J. Med. Chem., 2025, 281, 117005 CrossRef CAS PubMed.
- N. Goel, P. Kumar and S. Bhagat, Transition-Metal-Free Chemo-Selective C-C/C-N Bond Formation Reaction for the Highly Efficient Synthesis of Pyrazoles, Pyrazolo fused Pyrimidines and Pyridines, Eur. J. Org Chem., 2024, 27, e202400492 CrossRef CAS.
- M. Joshi, M. Patel, R. Tiwari and A. K. Verma, Base-Mediated selective synthesis of diversely substituted N-Heterocyclic enamines and enaminones by the hydroamination of alkynes, J. Org. Chem., 2012, 77, 5633–5645 CrossRef CAS PubMed.
- C. Nájera, L. K. Sydnes and M. Yus, Conjugated ynones in organic synthesis, Chem. Rev., 2019, 119(20), 11110–11244 CrossRef PubMed.
- A. S. Karpov and T. J. Müller, New entry to a three-component pyrimidine synthesis by TMS− ynones via Sonogashira coupling, Org. Lett., 2003, 5(19), 3451–3454 CrossRef CAS PubMed.
- R. Chinchilla and C. Nájera, The Sonogashira reaction: a booming methodology in synthetic organic chemistry, Chem. Rev., 2007, 107(3), 874–922 CrossRef CAS PubMed.
-
(a) G. Albano and L. A. Aronica, Acyl sonogashira cross-coupling: State of the art and application to the synthesis of heterocyclic compounds, Catalysts, 2019, 10(1), 25 CrossRef;
(b) R. Chinchilla and C. Nájera, Recent advances in Sonogashira reactions, Chem. Soc. Rev., 2011, 40(10), 5084–5121 RSC.
|
| This journal is © The Royal Society of Chemistry 2026 |
Click here to see how this site uses Cookies. View our privacy policy here.  Open Access Article
Open Access Article *,
Nikita Goel,
Poonam Kumari and
Pradeep Kumar
*,
Nikita Goel,
Poonam Kumari and
Pradeep Kumar





