Open Access Article
Open Access ArticleTriphenylene as a versatile scaffold for advanced functional materials
Ehsan Ullah Mughal *a,
Nafeesa Naeem
*a,
Nafeesa Naeem a,
Ayza Jabeena,
Farwa Zainaba,
Amina Sadiqb and
Kenneth E. Maly
a,
Ayza Jabeena,
Farwa Zainaba,
Amina Sadiqb and
Kenneth E. Maly *c
*c
aDepartment of Chemistry, University of Gujrat, Gujrat-50700, Pakistan. E-mail: ehsan.ullah@uog.edu.pk
bDepartment of Chemistry, Govt. College Women University, Sialkot-51300, Pakistan
cDepartment of Chemistry and Biochemistry, Wilfrid Laurier University, Waterloo, Ontario N2L 3C5, Canada. E-mail: kmaly@wlu.ca
First published on 2nd February 2026
Abstract
Triphenylenes are a class of polycyclic aromatic hydrocarbon that have been attracting increasing attention owing to their widespread applications in areas such as liquid crystals, organic electronics, photovoltaics, light emitting diodes, and catalysts. The utility of triphenylenes stems from their flatness, rigidity, and aromatic nature. This review provides an exploration of triphenylene derivatives, with an emphasis on recent advancements in their synthesis, properties, and multifaceted applications. Highlighting their synthetic strategies, we discuss both classical methods and modern approaches, including metal-catalyzed reactions and photochemical techniques, which have enabled the development of a wide range of substituted triphenylenes, as well as their dimers, trimers, twinned molecules, and oligomers. The electronic structure of triphenylene, characterized by a delocalized π-electron system, underpins its remarkable charge transport properties. In terms of applications, triphenylene-based liquid crystals are particularly notable for forming columnar mesophases with highly ordered structures, facilitating advantageous macroscopic molecular orientation. These properties underscore its potential for next-generation functional materials across diverse domains, including organic electronics, photovoltaics, light-emitting diodes, and catalysis. By integrating insights into its properties and future potential, this review aims to provide a valuable resource for researchers investigating triphenylene and its derivatives.
1. Introduction
Triphenylene 1 (Fig. 1), also known as isochrysene, 1,2,3,4-dibenzonaphthalene, and 9,10-benzophenanthrene,1 is a polycyclic aromatic hydrocarbon that is of interest in organic chemistry and materials science. It is a planar, aromatic, symmetrical hydrocarbon having molecular formula C18H12. It was first isolated from the pyrolytic decomposition products of benzene by Schultz, who named the compound “triphenylene”.1 The most detailed review to date was authored by Buess and Lawson, published approximately six decades ago, which provided extensive insights into the compound's structure, synthetic routes, chemical reactivity, and potential applications.1 The X-ray crystal structure of triphenylene was first determined by Banerjee and Guha in 1937, revealing its planar molecular structure and orthorhombic crystal lattice.2 Triphenylene itself packs in a herringbone motif featuring offset π-stacking and C–H⋯π interactions,2 the intermolecular packing can be influenced by adding substituents to the ring.3 The stability of triphenylene compared to its isomers; chrysene, benzo[a]anthracene, benzo[c]phenanthrene, and tetracene can be explained in terms of the Clar sextet rule, which is used to evaluate the aromatic stability of polycyclic aromatic hydrocarbons (PAHs).4 According to this rule, stability increases with the number of distinct benzene-like aromatic sextets (fully delocalized π-electrons within a six-membered ring) that can be drawn in the structure without overlapping π-electrons.5 Triphenylene can accommodate three Clar sextets, each located in one of its outer benzene rings, without any overlap of π-electrons. The presence of additional Clar sextets in triphenylene results in greater aromatic stabilization compared to its isomers, highlighting its enhanced thermodynamic stability.6 It has twelve positions for substitution, peripheral sites including 2, 3, 6, 7, 10 and 11 are most common (Fig. 1). Triphenylene molecules exhibit exceptional stacking behavior, primarily governed by their planar aromatic structure and the nature of peripheral functional groups. These functional groups extend laterally from the core, forming lateral chains that influence intermolecular interactions and molecular organization. The type of intermolecular interactions induced by these substituents includes π–π stacking and van der Waals forces, both of which play a crucial role in the formation of well-ordered columnar assemblies. Alkoxy (–OR) groups, while electron-donating and often leading to electron-rich aromatic systems, can actually reduce the strength of π–π stacking interactions due to increased electronic repulsion between π-systems. However, in many liquid crystals, the observed columnar stacking is believed to arise from micro segregation between the rigid aromatic cores and the flexible alkyl or alkoxy chains, promoting phase-separated self-assembly and stabilizing columnar mesophases.7,8 | ||
| Fig. 1 (a) Structural representation of triphenylene scaffold 1 2D structure,9 (b) Herringbone packing arrangement of triphenylene molecules in the crystal lattice. Reproduced from ref. 9 with permission from International Union of Crystallography, copyright 1963. | ||
The lateral chains also improve solubility and influence molecular packing, directly impacting the material's charge transport and optoelectronic properties. These attributes make triphenylene a versatile scaffold for applications in organic electronics, photovoltaics, and advanced functional materials.
Triphenylene's geometry, comprised of one central benzene ring and three nonadjacent benzene rings,10 has led to a vast range of properties and uses such as luminescence,11 1D charge migration,12 1D energy migration,13 liquid crystalline nature,14,15 ferroelectric switching,16 self-assembling nature,17 organic light-emitting-diodes,18 liquid crystal display (LCD) application,19 catalysis,20 as chemo-sensors for the detection of ions21 and as transporting materials in solar cells.22 The polycyclic compound and its derivatives due to their various applications have been the major factor behind the progress of electronics, materials science, and nanotechnology.23,24 The crystalline structure of triphenylene that is well-defined supports its capability to create stable and highly conductive films, which makes it a strong candidate for the development of next-generation optoelectronics and organic electronics where efficient charge transport is the result of ordered molecular packing.25,26 The extended π-system and peripheral substituents also allow it to act as a molecular receptor, interacting with the guests.27,28
The transfer of charge between molecules has been enhanced thanks to the 2D planar geometry and also the π-conjugated system.29,30 Moreover, triphenylene's physicochemical properties are crucial for its practical applications. Triphenylene is usually characterized by low solubility in polar solvents, such as water31 because of its strong aromatic interactions and non-polar character; thus, it creates various difficulties especially in methods that require solution-based processing or monitoring the environment through aqueous sensors.32,33 The solubility, however, can be made to increase substantially by attaching various functional groups at the edge like alkyl chains, alkoxy groups, and other polar substituents.34
Triphenylene derivatives are extensively studied in the context of discotic liquid crystals (DLCs). The core structure of most DLCs is made up of three characteristics: flatness, rigidity, and an aromatic character.14,35 Flexible chains are then attached to the core, which act as its outer layers. A large number of symmetrical and unsymmetrical derivatives have been studied and these compounds frequently self-assemble to form columnar mesophases.36 In addition to this, they also focused on the “twinned” and “oligomeric” triphenylene structures to add more value and function.37,38 The connection of the discotic units with a flexible spacer of the right length leads to the forming of this columnar mesophase, while a rigid space results in the nematic phase.39,40
The methods outlined in previous reviews41,42 focus on synthetic methods applicable to triphenylene derivatives. Nevertheless, the current review consists of a new view of triphenylene and its functionalized derivatives, pointing out synthetic paths, structural characteristics, and wide applications. In this case, classical and modern synthetic methods are considered, and the latest developments are presented as a means to produce the exact functionalization and create derivatives with customized properties.
This review also highlights the applications of triphenylene derivatives. Applications in liquid crystals and organic electronics rely on the electronic properties and self-assembly of triphenylenes, while sensing applications rely on their photophysical properties. Furthermore, their rigid structure and 3-fold symmetry make these compounds well-suited as molecular scaffolds for other functional materials.
2. Syntheses of triphenylene derivatives
2.1. A historical approach
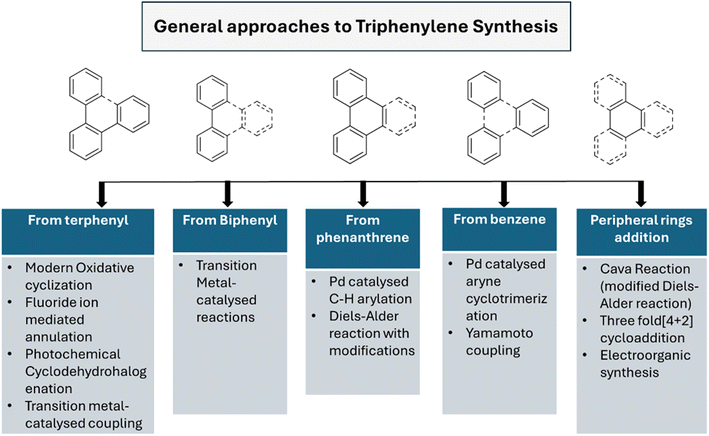
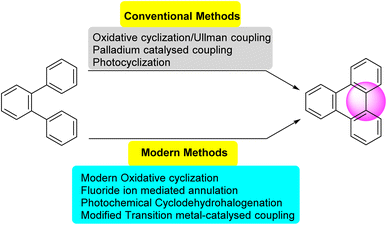
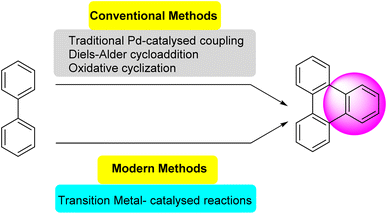
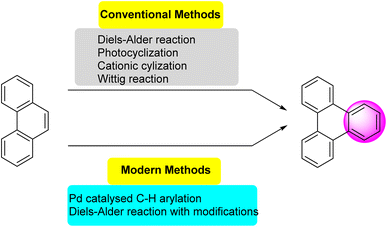
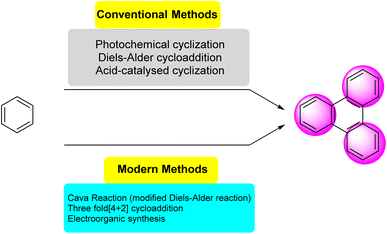
Several synthetic strategies were developed for triphenylene synthesis. The earliest synthesis was devised by Mannich, when cyclohexanone was treated with methyl alcoholic sulfuric acid, relatively small yield of dodecahydrotriphenylene was obtained which was then dehydrogenated with selenium to obtain triphenylene.43 Since its initial discovery, extensive research has been conducted on the structure and synthesis of triphenylene and its derivatives. The first review of triphenylenes focused on their synthesis, reactivity, and properties, was written by Buess and Lawson in 1960.1 Forty years later, Perez and Guitian surveyed multiple methods to synthesize substituted triphenylene and their benzo-derivatives.41 This review aims to consolidate and summarize the significant body of work in these areas, offering an overview of the advances made in understanding and utilizing this versatile class of compounds. Terphenyl is the most frequently used precursor for the synthesis of triphenylenes, using methods including oxidative cyclization,44 photocyclization,45 Cu-mediated Ullman coupling reaction or palladium catalyzed cross-coupling reaction employing aryl boronic acids.46 Starting from biphenyl, traditional methods include Diels Alder cycloaddition,47 oxidative cyclization,48 palladium-catalyzed coupling. These methods suffer from drawbacks such as over-oxidation, aggressive reagents and low solubility of oxidants; regioselectivity problems, harsh conditions and limited substrate scope.While some of the traditional synthetic approaches to triphenylene derivatives remain in use, the development of synthetic methods has led to new approaches to this class of compounds. As shown in Fig. 2, these methods generally involve constructing triphenylenes from simpler aromatic precursors, offering several different ways to access triphenylenes.
2.2. Modern approaches (2005–2024)
Recent approaches involve modern oxidative cyclization strategies with improved functional group compatibility and milder reaction conditions. Dehydrogenative cyclization and oxidative cyclodehydrogenation are synonymous terms that describe oxidative ring-closing accompanied by dehydrogenation. These reactions offer unique advantages depending on the desired application. Some other innovative synthetic approaches are fluoride ion-mediated annulation,53 oxidative biaryl coupling using Cerium Ammonium Nitrate (CAN),54 photochemical cyclodehydrohalogenation.55
One of the milder approaches to triphenylenes by Rathore and coworkers involves oxidative cyclodehydrogenation using DDQ (3) in the presence of methanesulfonic acid (Scheme 1).51 This method is a variant of the classical Scholl reaction, which promote C–C bond formation through oxidative cyclization of o-terphenyls and hexaarylbenzenes.56,57 In this method, o-terphenyl 2 in dichloromethane/methanesulfonic acid was treated with 1 equiv. of DDQ at 0 °C under argon. The reaction turned from green to dark brown within minutes, and after aqueous workup, triphenylene 5 was obtained in quantitative yield.51 The method is milder than traditional FeCl3-based protocols and allows easy recovery of the reduced DDQ byproduct via bicarbonate extraction, making it both efficient and practical.
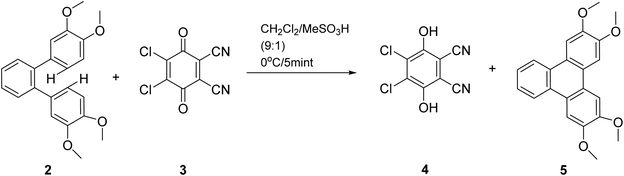 | ||
| Scheme 1 Oxidative cyclodehydrogenation using DDQ and methanesulfonic acid. Reproduced from ref. 51 with permission from American Chemical Society, copyright 2010. | ||
Several research groups extended oxidative cyclization strategies to complex macrocyclic frameworks. For example, cyclic nonaphenylenes 8 and dodecaphenylenes can be prepared by electron transfer oxidation of Lipshutz cuprates with duroquinone.58 Subsequent dehydrogenative cyclization by treating with Cu(OTf)2 and AlCl3 in CS2 with 73% yield or by using DDQ and CH3SO3H in dichloromethane can yield the corresponding triphenylene macrocycle 9 as illustrated in Scheme 2.59 These carbocyclic conjugated molecules 9 with long alkyl and alkoxy chains exhibit amphiphilic properties, phase separation, and supramolecular self-aggregation, allowing them to self-associate into oligomers in both solution and the solid state. Their shape-persistent π-frames make them highly suitable for designing advanced organic materials.
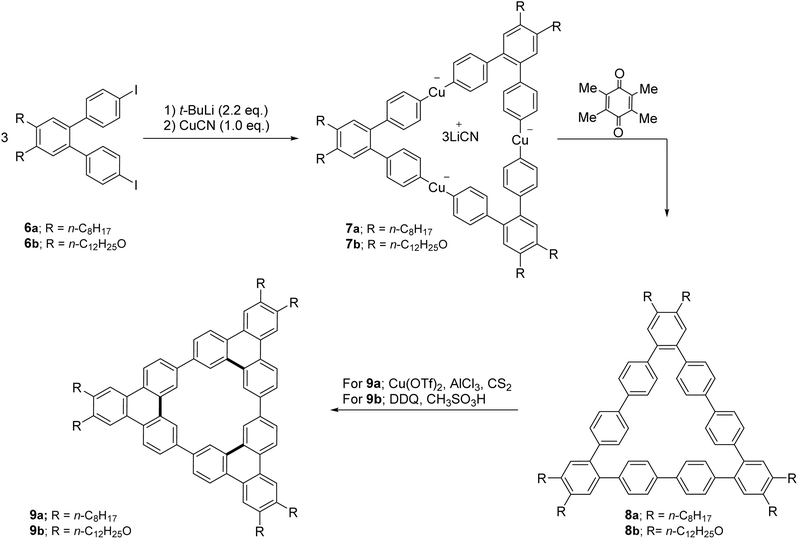 | ||
| Scheme 2 Synthesis of cyclic triphenylene trimers (9a–b) by dehydrogenative cyclization. Reproduced from ref. 59 with permission from John Wiley and Sons, copyright 2013. | ||
Oxidative biaryl coupling using ceric ammonium nitrate (CAN) is the facile, safer and mild method to synthesize substituted triphenylene and their derivatives as compared to strategies using other oxidants such as FeCl3, CuCl2 or Cu(OTf)2 with AlCl3 in CS2, MoCl5 with or without TiCl4 in CH2Cl2.60,61 Gupta and coworkers demonstrated Scholl reactions employing CAN which can act as an oxidant as well as Lewis acid can convert various o-terphenyls to triphenylene (Scheme 3).54 This CAN mediated Scholl reaction showed tolerance for a broad range of functional groups and heteroaromatic system gave good to excellent yield.
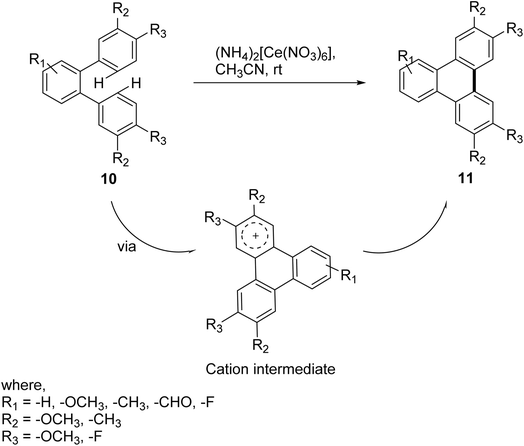 | ||
| Scheme 3 Oxidative biaryl coupling of o-terphenyl to form 11. Reproduced from ref. 54 with permission from Royal Society of Chemistry, copyright 2018. | ||
In contrast, Kumar and coworkers at first reported the cyclization of terphenyl to triphenylene without using any oxidizing agent.53 By using Suzuki–Miyaura coupling reaction, terphenyl derivatives 13 possessing tert-butyldimethylsilyl (OTBS) groups were synthesized (Scheme 4). Irreversible cyclization of terphenyl yields substituted triphenylene 14 induced by fluoride ion along with deprotection of OTBS groups.53 Mechanistically, the intermediate undergoes nucleophilic attack at the ortho-position of an adjacent aryl ring, promoting cyclization through an electrophilic rearrangement (Scheme 5). The reaction concludes with aromatization, yielding the triphenylene core with enhanced π-conjugation and planarity. This method provides a mild, oxidant-free approach for the selective formation of triphenylene derivatives with controlled substitution patterns. Thus methoxy-substituted o-terphenyl (13a, b) are successfully annulated to tetrahydroxy triphenylene derivatives (14a, b).
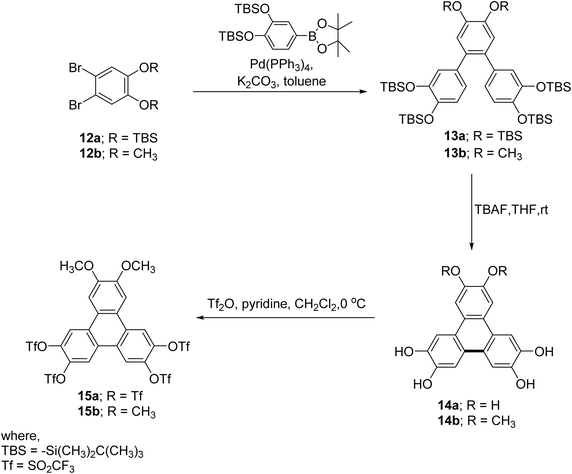 | ||
| Scheme 4 Tetrahydroxy triphenylene 14 synthesis. Reproduced from ref. 53 permission from Royal Society of Chemistry, copyright 2013. | ||
 | ||
| Scheme 5 Proposed mechanism for cyclization of terphenyl to triphenylene. Reproduced from ref. 53 permission from Royal Society of Chemistry, copyright 2013. | ||
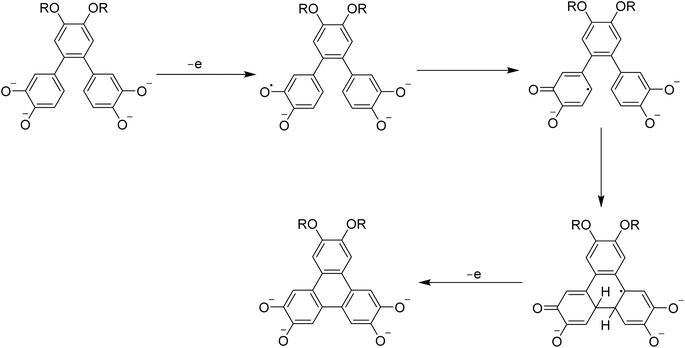
The fluoride-mediated triphenylene synthesis reported by Kumar was sensitive to substituents and was unsuccessful when methoxy groups were replaced by longer chains.62 These factors combined likely led to decreased reaction efficiency when scaled up. As an alternative approach, they reacted the o-terphenyl compound 16 with DDQ in a mixture of dichloromethane/TFA by 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to produce 17, followed by deprotection of the TBS groups using KF and HBr to furnish 2,3-bis(hexyloxy)-6,7,10,11-tetrahydroxy triphenylene 18 in good yield (Scheme 6).
1 to produce 17, followed by deprotection of the TBS groups using KF and HBr to furnish 2,3-bis(hexyloxy)-6,7,10,11-tetrahydroxy triphenylene 18 in good yield (Scheme 6).
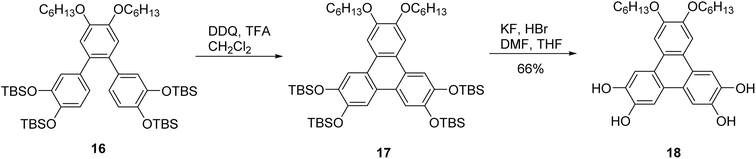 | ||
| Scheme 6 Hexyloxy substituted tetrahydroxytriphenylene 18 derivatives. Reproduced from ref. 62 with permission from Royal Society of Chemistry, copyright 2014. | ||
Photocyclization reactions have been widely employed in the synthesis of PAHs. In 1964, Mallory introduced the use of iodine as a catalyst for the photochemical cyclization of stilbenes leading to the synthesis of phenanthrene.63 This photocyclization approach has also been applied to the synthesis of triphenylenes.45,64 The reaction is thought to proceed by a photochemical electrocyclic ring-closing reaction, followed by dehydrogenation. As such, an oxidant is required for the reaction. Building upon this, alternative photocyclization strategies have emerged in which halogenated terphenyls undergo elimination of HX after photocyclization. These methods avoid the need for an oxidant to form the fused aromatic system. For example, photochemical dyclodehydrochlorination (CDHC) of chloro-substituted terphenyl gives triphenylene in good yield.65 Similarly, photocyclodehydrofluorination (PCDHF) involves photocyclization, followed by elimination of HF to produce triphenylenes. For example. Li and coworkers used fluorinated precursors and o-terphenyls in a PCDHF reaction to prepare fluorinated alkoxytriphenylene 24 which can be used as discotic liquid crystal (Scheme 7).55 Crucial intermediate o-terphenyl was prepared by fluorinated biphenyl 19 (2,3,4,5,6-pentafluoro-2′-iodo-4′,5′-dimethoxy-1′,1′-biphenyl) and phenylboronic acid 20, using Suzuki reaction. o-terphenyl underwent the PCDHF reaction to yield 1,2,3,4-tetrafluoro-6,7,10,11-tetraalkoxytriphenylene 22. Deprotection of alkoxy group was done by BBr3 and dichloromethane to yield 23. It was then treated with alkyl halide and potassium carbonate to yield 24 which subsequently underwent SNAr to produce 25 where fluorine atoms were replaced by alkoxy groups. This technique provided opportunity to control core fluorine and a alkoxy groups readily at different positions.55 Remarkably, metal-free synthetic strategies have recently emerged as viable alternatives for constructing fluorinated triphenylene derivatives. For instance, Zhou and co-workers reported an efficient SNAr-based, transition-metal-free approach to structurally related triphenylene systems, highlighting the growing potential of sustainable methodologies in this field.66
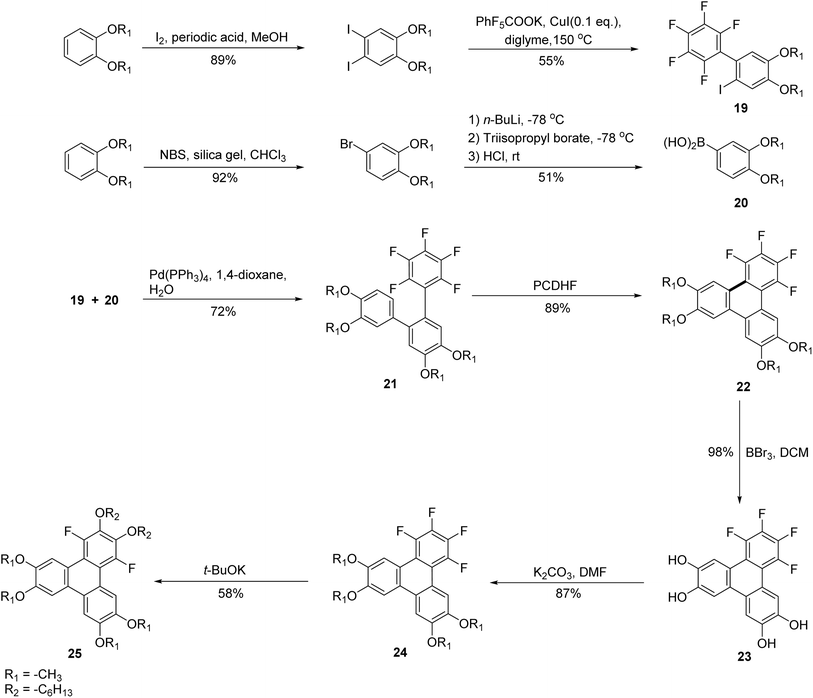 | ||
| Scheme 7 Synthesis of fluorinated hexakisalkoxytriphenylene 25 discotic liquid crystals. Reproduced from ref. 55 with permission from Royal Society of Chemistry under a Creative Commons Attribution-NonCommercial 3.0 Unported Licence., copyright 2022. | ||
Morin and colleagues demonstrated that phenylsulfonates can serve as efficient photolabile groups for generating aryl radicals under light irradiation.67 These radicals then undergo intramolecular cyclization to form a range of triphenylene derivatives. Notably, the phenylsulfonate group proved to be both versatile and stable, and it remained compatible with Suzuki cross-coupling reactions illustrated in Scheme 8. In Path-I to assess the efficiency of the phenylsulfonate group as a photocleavable moiety, a simple o-terphenyl was prepared. Starting from precursor 26, 27 was obtained via condensation with phenol under basic conditions. Subsequent Suzuki–Miyaura coupling with a boronic ester yielded compound 28 in 70%, confirming the phenylsulfonate group's stability under cross-coupling conditions, an essential feature for constructing larger PAHs. Path-II was adopted to overcome the steric hindrance caused by phenylsulfonate group by introducing this group after the Suzuki–Miyaura coupling. The o-terphenyl core 30 was synthesized via selective coupling in 88% yield, followed by the introduction of a fluorosulfonyl group using a modified Willis method (35% yield). Subsequent condensation with various p-substituted phenols (e.g., NH2, F, NO2, CN) gave good to excellent yields.67
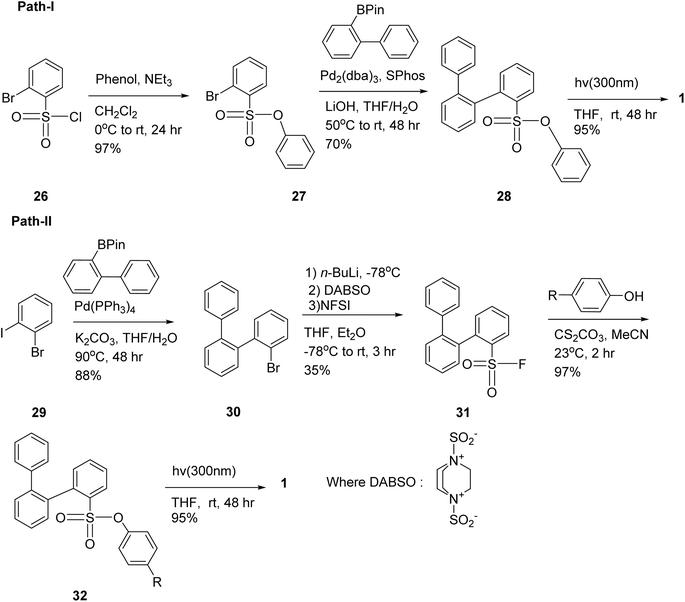 | ||
| Scheme 8 (Path-I): Synthetic pathway for the preparation of triphenylene from chlorosulfonyl derivatives. (Path-II): An alternative strategy involving the preparation of fluorosulfonyl derivatives as precursors. Reproduced from ref. 67 with permission from Royal Society of Chemistry under a Creative Commons Attribution-NonCommercial 3.0 Unported Licence., copyright 2024. | ||
An alternative strategy for triphenylene synthesis involves the use of transition metal-catalyzed cross-coupling reactions, followed by intramolecular oxidative cyclodehydrogenation to form the fused aromatic core. For instance, o-terphenylene derivative 35 was synthesized using a Pd(PPh3)4 catalyzed Suzuki reaction, highlighting the versatility of palladium catalysis in introducing various substituents.68 Ultimately, the intramolecular Scholl cyclodehydrogenation emerged as a pivotal step, enabling the synthesis of a diverse array of polar triphenylene derivatives with potential applications in materials science and organic electronics as illustrated in Scheme 9.
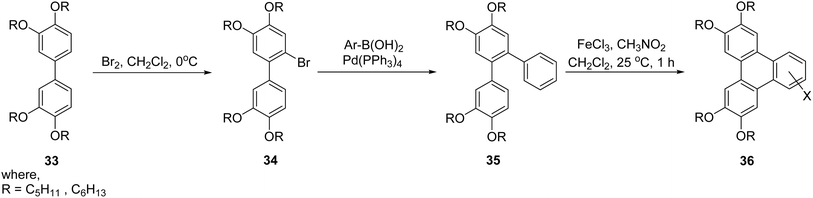 | ||
| Scheme 9 Synthesis of polar triphenylene 36 derivatives. Reproduced from ref. 68 with permission from Elsevier, copyright 2017. | ||
Recent advances in triphenylene synthesis have introduced several efficient methods. Palladium-catalyzed C–H arylation,69 oxidative coupling, and phenyl-addition/dehydrocyclization (PAC) are among the notable approaches.
Transition metal-catalyzed annulation strategies have emerged as important tools for the construction of aryl–aryl bonds, which can be used effectively for the preparation of triphenylene derivatives. For example, Liu and coworkers reported an aryne based palladium-catalyzed annulative π-extension involving a biphenyl derivative.70 The reaction involves formation of a benzyne intermediate, which reacts with an aryl palladium species, leading to cyclization. They reacted 2-iodo-4′-methylbiphenyl 37 with 2 equivalents of o-(trimethylsilyl)phenyl triflate 38, 5 mol% of Pd(dba)2, 5 mol% of P(o-tolyl)3, and 3 equivalents of CsF in a solvent mixture of 2 mL MeCN and 2 mL toluene at 110 °C for 24 hours, yielding the annulation product 39 in 38%. Optimization revealed that the controlled generation of benzyne from 38 using CsF is essential for the reaction's success (Scheme 10). Increasing the toluene content in the solvent mixture gradually improved the yield by slowing the benzyne formation.
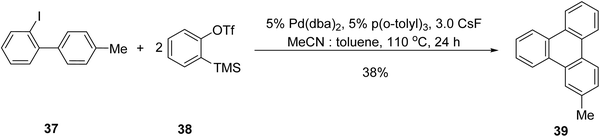 | ||
| Scheme 10 Pd-catalyzed annulation of benzyne. Reproduced from ref. 70 with permission from American Chemical Society, copyright 2005. | ||
In contrast, Pan and coworkers developed a non-aryne, direct arylation approach for synthesizing triphenylene through the palladium-catalyzed coupling of 2-iodobiphenyls 40 and iodobenzenes.69 This reaction entails palladium-catalyzed dual activations of C–H bonds and double palladium-catalyzed carbon–carbon bond formation (Scheme 11). A wide range of unsymmetrically functionalized triphenylenes (42) can be produced using this approach from readily available substrates as illustrated.
 | ||
| Scheme 11 Palladium catalyzed C–H arylation. Reproduced from ref. 69 with permission from American Chemical Society, copyright 2016. | ||
The catalytic cycle (Scheme 12) for the synthesis of triphenylene begins with the oxidative addition of 2-iodobiphenyl to Pd(0), forming complex A. This is followed by intramolecular C–H activation to produce palladacycle B. Subsequent oxidative addition of iodobenzene to B results in Pd(IV) complex C, which undergoes reductive elimination to form Pd(II) complex E. An alternative pathway may involve the transmetalation-type exchange of aryl ligands between B and a phenylpalladium(II) species, yielding dinuclear arylpalladium complex D, which upon reductive elimination also leads to complex E.69
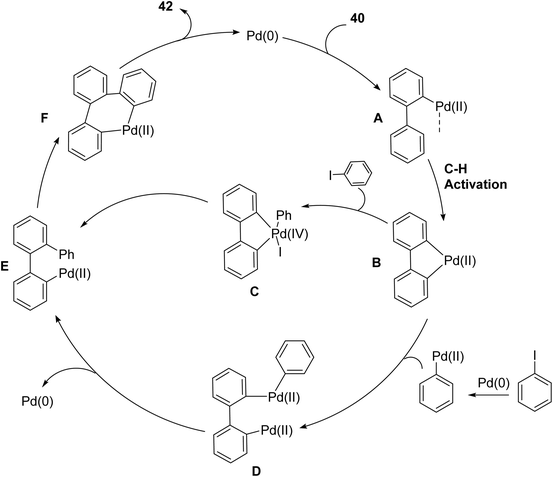 | ||
| Scheme 12 Proposed mechanism for Pd-catalyzed C–H arylation. Reproduced from ref. 69 with permission from American Chemical Society, copyright 2016. | ||
One of the drawbacks of some of these non-aryne based approaches catalyzed by palladium complexes is that they typically require laborious pre-functionalization steps.71,72 So, a new one-step mixture of functionalized triphenylene frameworks via Pd-catalyzed 2- or 4-fold C–H arylation of inactivated benzene derivatives and readily available arene substrate was described by Mathew and coworkers.73 This synthesis employs pivanilide, a Pd2(dba)3 catalytic system along with cyclic diaryliodonium salts 44 as p-extending agents, enabling site-selective inter- and intramolecular tandem arylation sequences (Scheme 13) with proposed 2-fold C–H arylation mechanism (Scheme 14). The resulting N-substituted triphenylene is of interest for using a field-effect transistor sensor for the fast, subtle, and reversible detection of alcohol vapor. The N-substituted triphenylenes synthesized are integrated into an alcohol field-effect transistor (FET) sensor, which demonstrates a swift and reversible response.
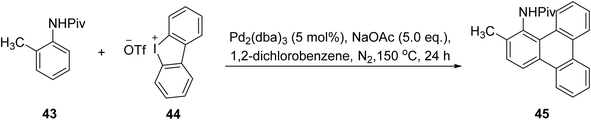 | ||
| Scheme 13 2-Fold C–H arylation for building triphenylene. Reproduced from ref. 73 with permission from John Wiley & Sons, copyright 2017. | ||
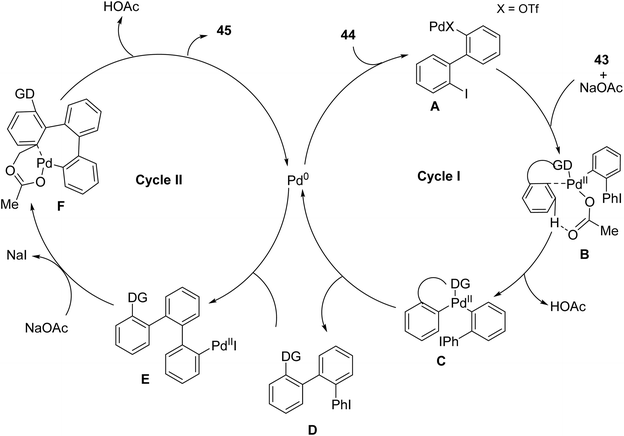 | ||
| Scheme 14 2-Fold C–H arylation mechanism. Reproduced from ref. 73 with permission from John Wiley & Sons, copyright 2017. | ||
The previously reported tandem arylation route using coordinating moiety-bearing arenes to synthesize triphenylene (Scheme 13) as a completely benzenoid PAH building unit suffered as few drawbacks. This route aided by preinstalled directing groups (DGs), restricts access to the parent framework.74 Lee et al. later introduced the inaugural non-directed, DGs free and one to two step Pd-catalyzed aromatic extension method for constructing triphenylene frameworks using basic arenes.75 Palladium-catalyzed 2-fold C–H arylation enables consecutive C–C linkage processes, leading to the formation of fully benzenoid triphenylene structures employing cyclic diaryliodonium salts in Scheme 15.
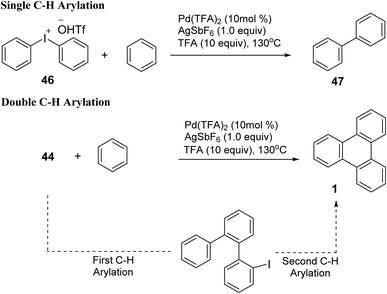 | ||
| Scheme 15 Single arylation followed by double arylation. Reproduced from ref. 75 with permission from American Chemical Society, copyright 2019. | ||
Yang and coworkers developed a regioselective approach to synthesize unsymmetrical triphenylene using o-bromobenzoic acid and aryl iodides via palladium/norbornene catalyzed coupling reaction.76 Moreover, they showed the synthesis of a number of triphenylenes through a palladium-catalyzed C–H arylation reaction between o-bromobenzoic acids 49 and 2-iodobiphenyls 48 without the need for norbornene and benzoquinone as mentioned in Scheme 16.
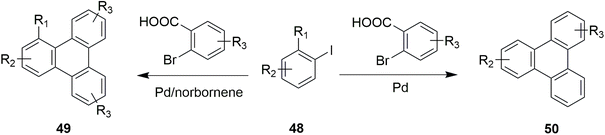 | ||
| Scheme 16 Palladium catalyzed C–H arylation for triphenylene synthesis. Reproduced from ref. 76 with permission from American Chemical Society, copyright 2018. | ||
In 2018, Xu and co-workers reported an efficient palladium-catalyzed Suzuki–Miyaura coupling strategy for the synthesis of functionalized triphenylene derivatives 53 using arylboronic acids 51 and 2,2′-dibromobiphenyl 52 precursors (Scheme 17).77 This approach combines cross-coupling with intramolecular C–H activation, enabling a cascade annulation process that leads directly to triphenylene frameworks. Elevated reaction temperatures were found to significantly enhance cyclization efficiency, thereby facilitating the formation of asymmetrically substituted triphenylenes in a single synthetic sequence. A wide range of p-substituted arylboronic acids, including those bearing methyl, fluoro, chloro, cyano, and ester functionalities, participated smoothly in the transformation, affording the desired products in high yields. In addition, boronic acids containing electron-withdrawing groups displayed pronounced regioselectivity during the annulation step. Hence, this methodology offers high atom efficiency, reduced synthetic cost, and excellent control over regioselectivity, making it a powerful strategy for constructing polycyclic aromatic systems through consecutive C–C bond formation and ring fusion.
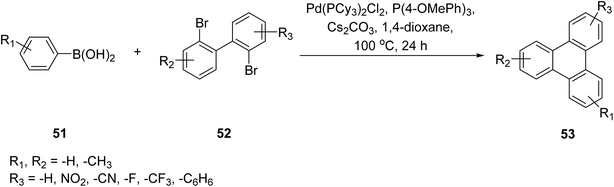 | ||
| Scheme 17 Construction of triphenylene frameworks via palladium-catalyzed Suzuki–Miyaura cross-coupling of arylboronic acids and 2,2′-dibromobiphenyl building blocks. Reproduced from ref. 77 with permission from Elsevier, copyright 2019. | ||
In 2018, Zhao and co-workers reported an efficient copper-catalyzed strategy for the synthesis of asymmetrically substituted triphenylene derivatives 56 via oxidative annulation of electron-rich biphenyl precursors 54, mainly 3,3′,4,4′-tetraalkoxybiphenyls, with diaryliodonium salts 55 that tolerate halogen substituents (Scheme 18).78 The transformation proceeds through successive C–H activation steps and exhibits broad functional-group compatibility, including aryl iodonium reagents bearing bromine, chlorine, and fluorine substituents. Aryl iodonium salts containing electron-donating groups proved particularly effective, delivering comparable yields while retaining halogen functionalities suitable for further post-synthetic cross-coupling modifications. Mechanistic investigations indicate that the high chemoselectivity originates from the preferential activation of electron-rich biphenyl substrates, as electron-deficient biphenyls and unsubstituted biphenyls failed to undergo cyclization under the same conditions. Although operationally straightforward and synthetically valuable, the methodology is currently limited to alkoxy-substituted biphenyl frameworks and catechol-derived aryl partners, which restricts its broader applicability to more diverse aromatic systems.
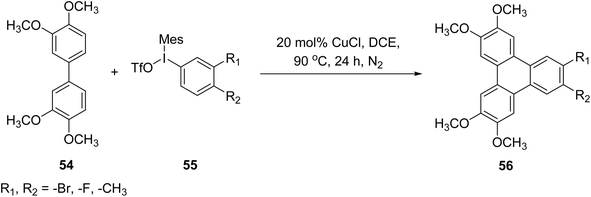 | ||
| Scheme 18 Copper-catalyzed C–H arylation route to unsymmetrical triphenylenes. Reproduced from ref. 78 with permission from American Chemical Society, copyright 2018. | ||
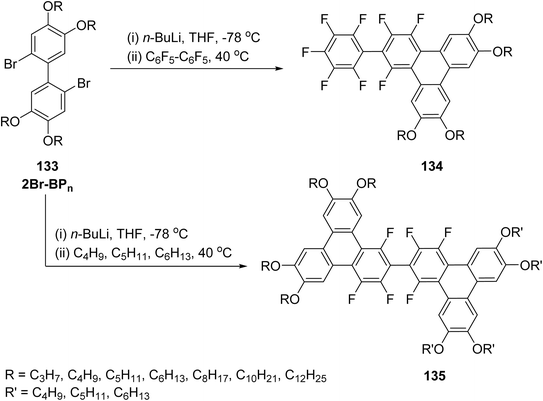
In addition to the classical oxidative trimerization, Yamamoto coupling, and the more recent photo-cyclo-de-hydrofluorination (PCDHF) protocols, the field has witnessed rapid progress in nucleophilic aromatic substitution of polyfluoroarenes (SNArF) as a powerful route to fluorinated triphenylene scaffolds and discotic mesogens. Polyfluorinated arenes such as hexafluorobenzene (C6F6), octafluoronaphthalene (C10F8), and decafluorobiphenyl (C12F10–C12F10) possess highly activated C–F bonds capable of undergoing regioselective substitution with organolithium nucleophiles, enabling controlled construction of π-extended and heavily fluorinated aromatic architectures.
In this work,79 the fluorinated precursors 59 (1,2,3,4-tetrafluoro-6,7,10,11-tetraalkoxytriphenylene) and 58 (9,10,11,12,13,14-hexafluoro-2,3,6,7-tetraalkoxybenzo[f]tetraphene), each functionalized with four racemic 3,7-dimethyloctyloxy chains, were subjected to reactions with a series of aryl-lithium reagents (ArLi; Ar = naphthalen-1-yl or -2-yl (NA/NB), benzothiophen-2-yl (BT), triphenylen-2-yl (TP), pyren-1-yl (PY), and tetraphenylethylenyl (TPE)). These transformations afforded the corresponding monosubstituted derivatives, 61 (trifluorotriphenylene-based) and 60 (pentafluorobenzo[f]tetraphene-based) (Scheme 19). Single-crystal X-ray diffraction analyses of three chain-free model derivatives revealed characteristic F⋯H contacts and antiparallel π–π stacking, providing structural evidence that supports and confirms the aryl substitution sites. The mesomorphic behaviour of these fluorinated pseudo-discotic systems was examined using polarized optical microscopy, differential scanning calorimetry, and small- and wide-angle X-ray scattering (SAXS/WAXS).
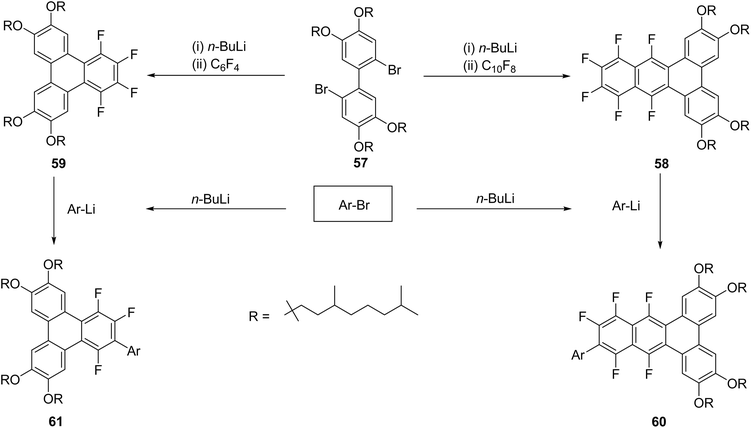 | ||
| Scheme 19 Synthesis of tri- and penta-fluorotriphenylenes. Reproduced from ref. 79 with permission from American Chemical Society, copyright 2025. | ||
One of the major contributions in this area is the development of F-discotic liquid crystal (F-DLC) frameworks obtained from SNArF reactions between polyfluoroarenes and biphenyl-dilithium or aryl-lithium reagents, as demonstrated by Zhang et al.66 The method provides efficient access to multi-fluorinated discogens displaying enhanced mesomorphic stability, strong π–π interactions, and tunable dielectric properties. In this study,66 a rapid synthesis of a series of 1,2,3,4-tetrafluoro-6,7,10,11-tetraalkoxytriphenylenes 65 (4F-TPn, n = 3–14) and 9,10,11,12,13,14-hexafluoro-2,3,6,7-tetraalkoxybenzo[f]tetraphenes 64 (6F-BTPn, n = 6–16) was achieved via SNFAr of perfluoroarenes, following a slightly modified literature procedure. The key precursors, 2,2′-dilithio-1,1′-biphenyl derivatives 62 with varied alkoxy chain lengths, were reacted with hexafluorobenzene (C6F6) and octafluoronaphthalene (C10F8) to afford the Janus-like 4F-TPn 65 and 6F-BTPn 64 compounds in good yields (Scheme 20). Starting from inexpensive and readily available 5-bromosalicylaldehyde, 4F-TPn 65 were synthesized in four steps, with overall yields ranging from 25–53%. Upon warming to room temperature, 1.2 equivalents of the respective perfluoroarene were added, yielding the target Janus compounds. The yields for this second, double-annulation step was notably lower (approximately 15% for 2F-TBTPn 67 and 40% for 4F-TBTC6 66) compared to the initial single SNFAr reactions (65–83% for 4F-TPn and 53–73% for 6F-BTPn), likely due to steric hindrance around the triphenylene core and the reduced solubility of the tribenzotetraphene products. Nonetheless, these yields are expected to be amenable to optimization in future studies.
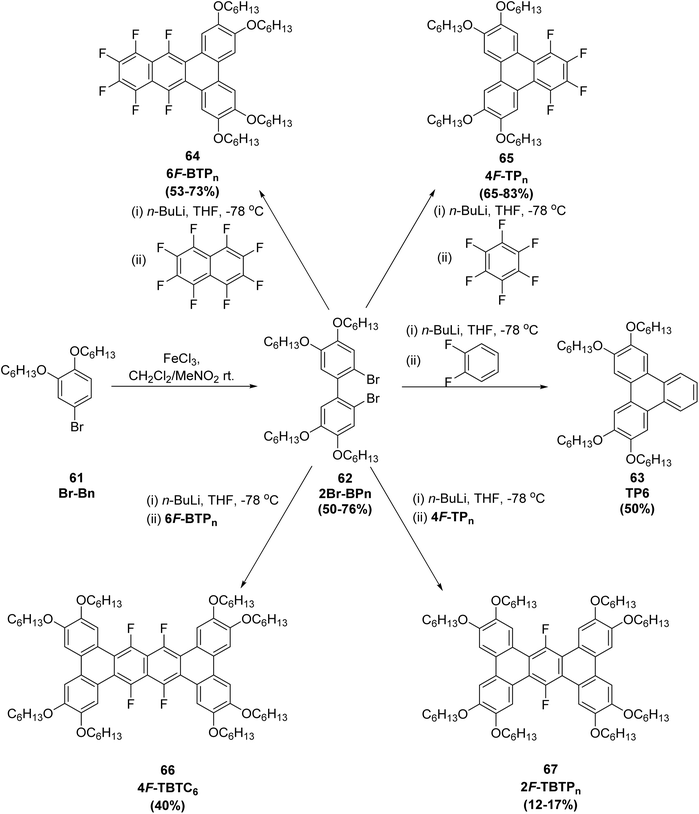 | ||
| Scheme 20 General synthetic route for Janus-type and π-extended discotic fluorinated polyaromatic hydrocarbons (F-PAHs). Reproduced from ref. 66 with permission from John Wiley & Sons, copyright 2023. | ||
Subsequent studies further expanded this chemistry to a variety of aryl-fluoroarene discotic systems (Ar-FDLCs), showing that selective substitution patterns could be achieved by adjusting the stoichiometry of the lithiated reagents and the electronic configuration of the polyfluoroarene substrates. These developments were articulated in ref. 80 and later refined to yield structurally diverse F-DLCs with controllable optical textures.81
Non-covalent interactions between arenes and fluoroarenes have emerged as a valuable tool for constructing both flexible and rigid supramolecular architectures, and they are now increasingly applied in the design of liquid-crystalline materials.82 These interactions arise from the strong electronegativity of fluorine, which significantly alters the electronic distribution and dipole moment of the fluorinated aromatic units. In this context, two families of fluorinated triphenylene derivatives equipped with four peripheral alkyl chains of different lengths were synthesized through SNArF of polyfluoroarenes.80 The first family consists of the homologous 2-phenyl-1,3,4-trifluorotriphenylenes 71 (PHn, R = CnH2n+1, n = 3–12), obtained in approximately 60–90% yield by reacting lipophilic 2,2′-dilithiobiphenyl reagents with pentafluorobiphenyl (C6F5–C6H5). Using analogous conditions, a second set of 2-aryl-1,3,4-trifluorotriphenylene derivatives bearing larger aromatic substituents—such as naphthyl (1- or 2-isomers), benzothiophen-2-yl, triphenylen-2-yl, pyren-1-yl, and tetraphenylethenyl groups—was prepared in moderate to high yields (Scheme 21). A key outcome of this study is the systematic observation of columnar mesophases across all fluorinated derivatives. Compound such as 71 consistently generate columnar hexagonal (Colhex) mesophases as well, although the breadth and stability of these phases strongly depend on the bulk and geometry of the appended aromatic units. These findings highlight the decisive role of aromatic fluorination and the strength of arene–fluoroarene interactions in promoting columnar ordering by favoring face-to-face assembly of electron-rich and electron-poor aromatic frameworks.80
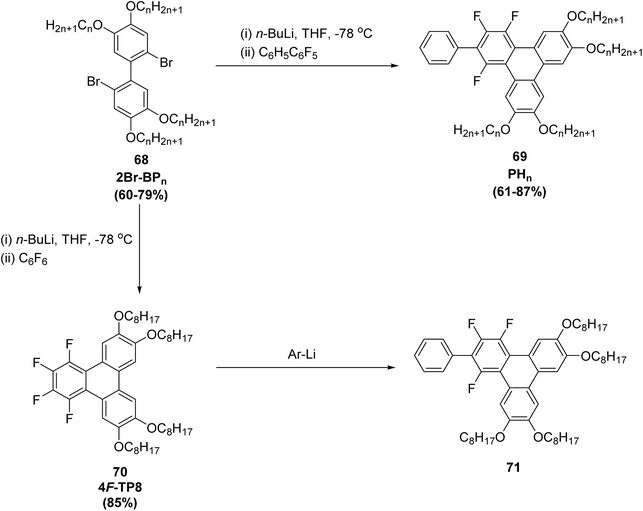 | ||
| Scheme 21 Synthesis of 2-aryl-1,3,4-trifluorotriphenylene derivatives. Reproduced from ref. 80 with permission from Royal Society of Chemistry, copyright 2023. | ||
Two positional isomeric families of triphenylene derivatives featuring “inverted” alkoxy-chain substitution patterns were prepared: 7,10-dialkoxy-1,2,3,4-tetrafluoro-6,11-dimethoxytriphenylenes and 6,11-dialkoxy-1,2,3,4-tetrafluoro-7,10-dimethoxytriphenylenes, designated as p-TPFn (81) and m-TPFn (76), respectively. In addition, the corresponding non-fluorinated isomers, 3,6-dibutoxy-2,7-dimethoxytriphenylene (p-TP4; 80) and 2,7-dibutoxy-3,6-dimethoxytriphenylene (m-TP4; 75), were synthesized (Scheme 22). All compounds were obtained in good overall yields through a three-step sequence based on transition-metal-free nucleophilic aromatic substitution of fluoroarenes. The reactions involved the coupling of suitably substituted 2,2′-dilithiobiphenyl intermediates with either perfluorobenzene (C6F6) to generate the fluorinated series p-TPFn (81) and m-TPFn (76), or o-difluorobenzene (C6H4F2) to afford the non-fluorinated analogues p-TP4 (80) and m-TP4 (75). Furthermore, the fluorinated derivatives adopt closely packed columnar stacking arrangements, an organization that is typically favorable for the formation of columnar liquid-crystalline phases.81
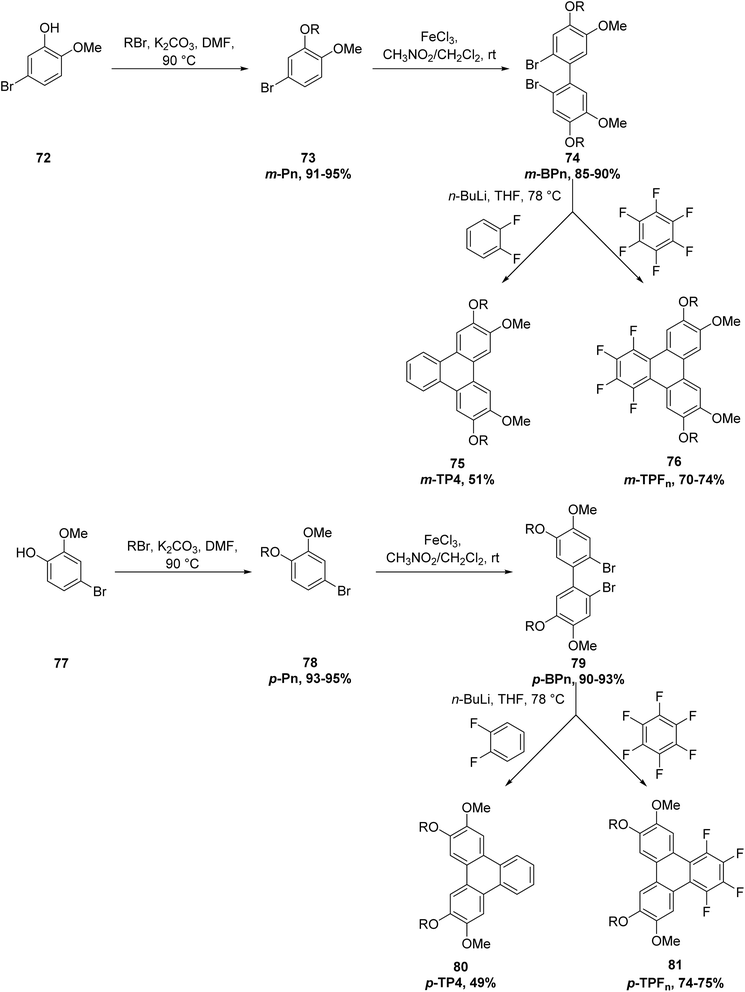 | ||
| Scheme 22 Synthetic route for the synthesis of Janus-like TP discogens, p-TPFn, m-TPFn, p-TP4 and m-TP4 (and intermediates); R = CnH2n+1, with n = 4, 6, 8, 10. Reproduced from ref. 81 with permission from John Wiley & Sons, copyright 2024. | ||
Guitian and coworkers devised a route to achieve triphenylene that passes through 1-triphenylenol, employing a Diels–Alder cycloaddition of 9,10-phenanthryne, which was derived from precursor 82, with furan to yield compound 83.86 Subsequent acidic treatment of 83 resulted in a quantitative yield of 1-triphenylenol 84. A subsequent one-pot synthesis using hexamethyldisilazane (HMDS) in refluxing THF, trapped by n-BuLi and Tf2O afforded triflate. Treatment of this triflate with 5 mol% of Pd2(dba)3 and dimethyl acetylene dicarboxylate (DMAD) yielded polycyclic arene 86, subsequent from reaction of one aryne and two alkynes, this method allows for subsequent transformations using arynes in Scheme 23.
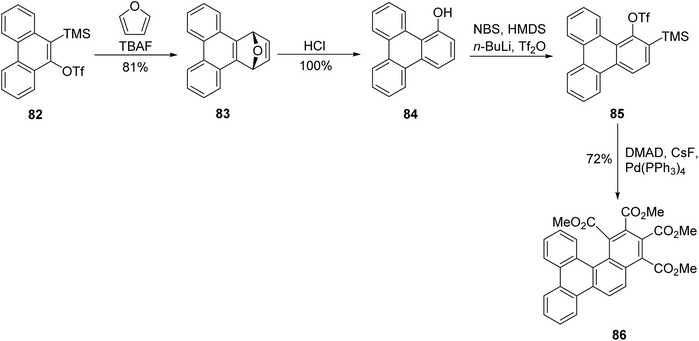 | ||
| Scheme 23 Trimerization of 3,4-phenanthryne to yield sterically congested trimer. Reproduced from ref. 86 with permission from American Chemical Society, copyright 2008. | ||
Junji and coworkers demonstrated that fluorinated phenanthrenes could be used to generate arynes via metalation and elimination of fluoride.87 Diels–Alder reactions of these arynes with isobenzofurans 88 yielded the respective cycloadducts, which underwent reductive aromatization in an SnCl2/HBr system, resulting in benzotriphenylenes 91. For example, in the presence of 88, treatment of bromo(fluoro)phenanthrene with butyllithium at −90 °C allowed for Li–Br exchange followed by LiF elimination, generating the phenanthryne 90. Upon warming to room temperature, 90 was obtained in 55% yield, along with fluorophenanthrene in 24% yield. In this process, phenanthryne underwent a Diels–Alder reaction with isobenzofuran 88. Reductive aromatization of this adduct to benzotriphenylene 91, took place under basic conditions from 90 (Scheme 24).
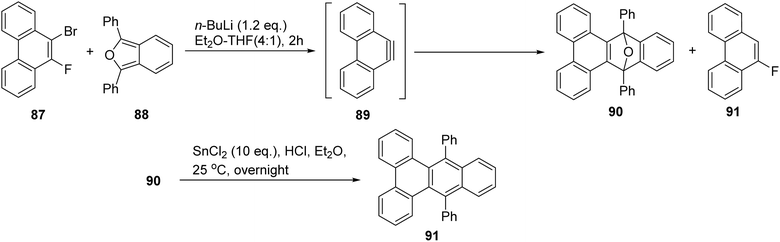 | ||
| Scheme 24 Diels Alder reaction between extended arynes and isobenzofuran. Reproduced from ref. 87 with permission from John Wiley & Sons, copyright 2020. | ||
Sterically congested large triphenylenes can be accessed via Diels Alder reaction of phencyclones with alkynes, as method demonstrated by Perez and coworkers.88 Cycloaddition of alkyne 93 with cyclopentadienone 92 could lead to the formation of iodotriphenylene 94, after cheletropic extrusion of CO (Scheme 25). Compound 94 was formed in a remarkable 73% yield, and was subsequently used to obtain sterically congested PAHs. Later, Müllen and coworkers extended the use of this method to prepare triangular triphenylene macrocycles (e.g. 98), the phencyclones were produced from the corresponding phenanthrene diones.89 The Diels Alder reaction of alkynes with phencyclone 97 was followed by a nickel-mediated coupling reaction (Scheme 26).
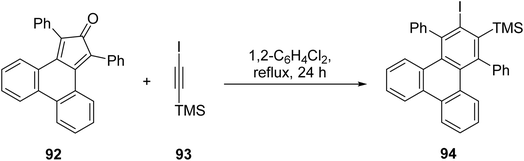 | ||
| Scheme 25 Synthesis of 1,4-diphenyl-2,3-triphenyene. Reproduced from ref. 88 with permission from Royal Society of Chemistry, copyright 2013. | ||
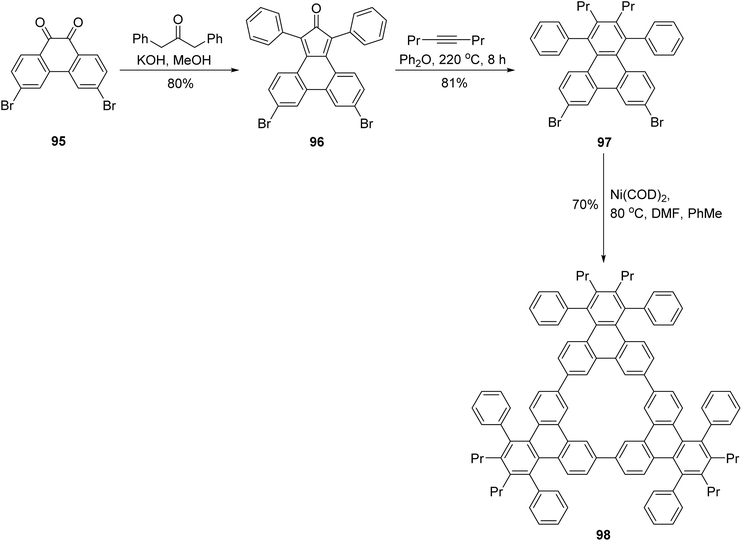 | ||
| Scheme 26 Synthesis of triphenylene molecular triangle. Reproduced from ref. 89 with permission from John Wiley & Sons, copyright 2011. | ||
Guitian and coworkers reported the production of aryne 101 ensued by treating triflate 99 with tetrabutylammonium fluoride (TBAF) in the presence of cyclopentadienone 92. Subsequent refluxing of this mixture in tetrachloroethane for 3 hours resulted in a 44% yield of benzotriphenylenol 100.90 This achievement is noteworthy given the presence of an unprotected hydroxyl group, which could potentially react with the extremely electrophilic aryne. It appears that the intermediate formed from the Diels–Alder reaction of aryne 101 and dienone undergoes an in situ transformation, ultimately yielding compound 100 through CO extrusion as described earlier (Scheme 27).
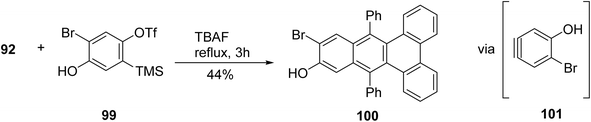 | ||
| Scheme 27 Diels Alder reaction between unprotected hydroxyl containing triflate and dienone. Reproduced from ref. 90 with permission from John Wiley & Sons, copyright 2011. | ||
Peña and coworkers performed the catalytic trimerization of arynes that involves carefully selecting the catalyst and the method for aryne generation 103 using mild reaction conditions. Treating tirmethylsilylaryl triflate 102 with 2 equivalents of anhydrous CsF in the presence of catalytic [Pd(PPh3)4] in acetonitrile at room temperature resulted in fluoride-induced elimination of TMS and triflate, followed by aryne trimerization, yielding 83% triphenylene derivatives 104 with yields up to 83% (Scheme 28).74
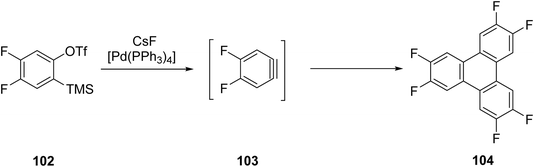 | ||
| Scheme 28 Earliest Palladium catalyzed cyclotrimerization of arynes. Reproduced from ref. 74 with permission from John Wiley & Sons, copyright 1998. | ||
Galow and colleagues extended Peña's aryne cyclotrimerization to access sterically hindered triphenylenes.94 They reported 1,4,5,8,9,12-hexamethyltriphenylene 107 synthesis by using o-trimethylsilylaryl triflate bearing multiple methyl groups. Addition of cesium fluoride in the presence of a palladium catalyst resulted in the formation of 106 (Scheme 29). X-ray crystallography indicates that it adopts a highly distorted C2 geometry, featuring a 53.30° end-to-end twist, due to increased steric interference from the methyl groups.
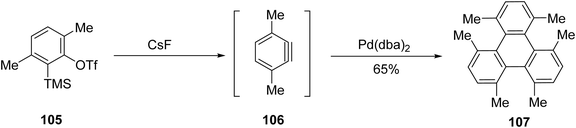 | ||
| Scheme 29 Synthesis of 1,4,5,8,9,12-hexamethyltriphenylene 107. Reproduced from ref. 94 with permission from American Chemical Society, copyright 2007. | ||
In a similar approach, Guitian and coworkers synthesized 1-methoxytriphenylene through a palladium-catalyzed [2 + 2 + 2] cycloaddition involving mixed arynes that is one molecule of 3-methoxybenzyne and two molecules of benzyne.86 This process yields seven potential products due to homo- and heterocyclotrimerization of the arynes. The distribution of these products appears to be influenced by the reagent ratio, the rate of aryne–palladium complex formation, and the subsequent transformation rates. Optimal conditions for obtaining a high yield of 111 involve using a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 molar ratio of aryl precursors 108 and benzene triflate with CsF in the presence of 10 mol% of Pd(PPh3)4 (Scheme 30).
4 molar ratio of aryl precursors 108 and benzene triflate with CsF in the presence of 10 mol% of Pd(PPh3)4 (Scheme 30).
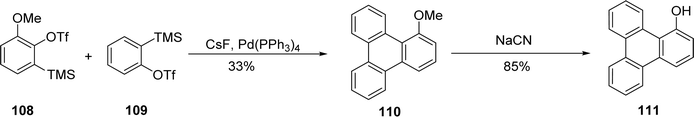 | ||
| Scheme 30 Cyclotrimerization of aryne with benzyne. Reproduced from ref. 86 with permission from American Chemical Society, copyright 2008. | ||
In contrast to Peña's work, Liu and coworkers devised Pd-catalyzed reaction to generate triphenylenes by reaction of two arynes with an iodobenzene.70 For example, reaction of ethyl 4-iodobenzoate 112 and triflate 113 afforded the target triphenylene 114 in a 50% yield, as illustrated in Scheme 31. The two possible pathways were proposed, in one, the Pd(0) species undergoes oxidative cyclization with the aryne to form a palladacycle, followed by C–H activation and reductive elimination. Alternatively, Pd(0) may first insert into the aryl iodide bond, giving an aryl–Pd intermediate that subsequently engages with the aryne. This reaction allowed access to low symmetry triphenylenes where one of the outer rings bears different substituents. However, the reaction necessitated the use of an excess of aryne, which resulted in aryne cyclotrimerization to give 115 as a byproduct.
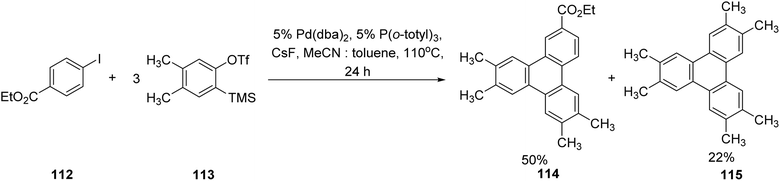 | ||
| Scheme 31 Triphenylene synthesis by Pd-catalyzed cyclotrimerization. Reproduced from ref. 70 with permission from American Chemical Society, copyright 2005. | ||
Since aryne cyclotrimerization often results in low yields and requires the synthesis of aryne precursors, an alternative approach consists of nickel-mediated Yamamoto coupling. Schroeder et al. showcased the nickel-mediated Yamamoto coupling of o-dibromoarenes 116 as a succinct and efficient method for synthesizing electron-deficient substituted triphenylene derivatives 117 which could not be prepared using Scholl reaction or palladium-catalyzed cyclotrimerization.95 Homocoupling of aryl bromides in the presence of bis(1,5-cyclooctadiene)nickel(0) forms new aryl–aryl bond thus cyclotrimerization occurs to form 3-fold symmetric triphenylene (Scheme 32). Yamamoto coupling results with various o-dibromoarenes are also given in Scheme 32. Remarkably, Rudiger and coworkers also demonstrated the use of Yamamoto coupling as a convenient alternative to aryne based methods by the synthesis of cyclotrinaphthylenes in a good yield.96 It is also effective in preparing the compounds, which are crucial for organic semiconductor applications due to their ability to form columnar liquid crystalline phases with extended π-stacked arrays.97
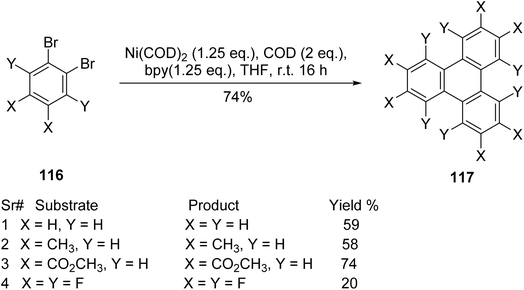 | ||
| Scheme 32 Cyclotrimerization of o-dibromobenzene by using Yamamoto coupling. Reproduced from ref. 95 with permission from Royal Society of Chemistry, under a Creative Commons Attribution-NonCommercial 3.0 Unported Licence, copyright 2021. | ||
Furthermore, Schroeder and coworkers also illustrated the applicability of this method in preparation of electron-deficient mesogens bearing imide groups.95 They synthesized triphenylene trisimide 121 and its thionated counterpart 122, enabling a comparative analysis of their mesomorphic properties. 4,5-Dibromo-o-xylene 118 was oxidized to anhydride 119 which reacts with octyl amine in the presence of Ni(OAc)2 to give imide (Scheme 33). Polarized optical microscopy and differential scanning calorimetry studies reveal that compound 120 exhibits a columnar mesophase with high clearing point.
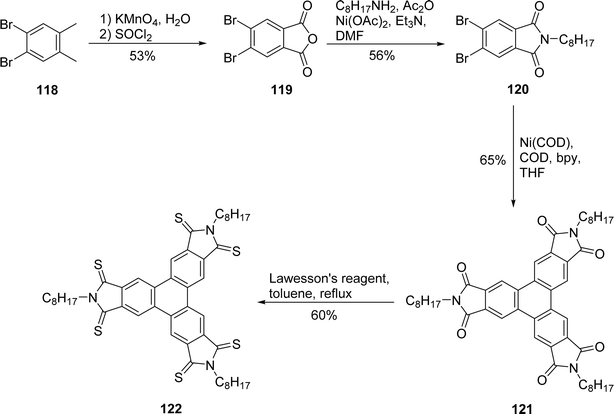 | ||
| Scheme 33 Synthesis of electron deficient triphenylene trisimide and its thionated analog. Reproduced from ref. 95 with permission from Royal Society of Chemistry, under a Creative Commons Attribution-NonCommercial 3.0 Unported Licence, copyright 2021. | ||
Electrochemical methods provide a metal-free and environmentally friendly route for the oxidative trimerization of catechol derivatives to synthesize triphenylene and its ketal-protected intermediates. Unlike conventional approaches that rely on high-valent metal salts (e.g., FeCl3, MoCl5), electrochemical oxidation avoids metal waste, reduces reagent costs, and minimizes side reactions such as ketal cleavage.98 The compound 2,3,6,7,10,11-hexahydroxytriphenylene 124, is widely utilized as a building block in supramolecular chemistry. Regenbrecht and coworkers demonstrated its efficient synthesis through a galvanostatic anodic oxidation process.99 This method involves the oxidation of catechol ketals 123, followed by acidic hydrolysis to yield the desired product with high purity and optical clarity. The electrolysis was feasible under practical galvanostatic conditions using platinum sheets as electrode materials and propylene carbonate (PC) and tetrabutylammonium tetrafluoroborate (TBABF4) as electrolyte in Scheme 34. Precipitation of non-polar triphenylene is favoured in the polar electrolyte. PC is inexpensive and not hazardous to environment so this procedure effectively improved access to triphenylene derivatives.
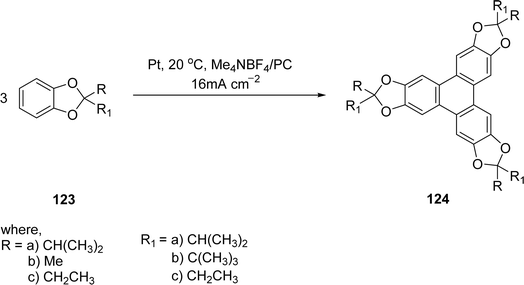 | ||
| Scheme 34 Anodic oxidation of catechol ketals to 2,3,6,7,10,11-hexahydroxytriphenylene Reproduced from ref. 99 with permission from Beilstein Institute for the Advancement of Chemical Sciences, a German non-profit foundation, copyright 2012. | ||
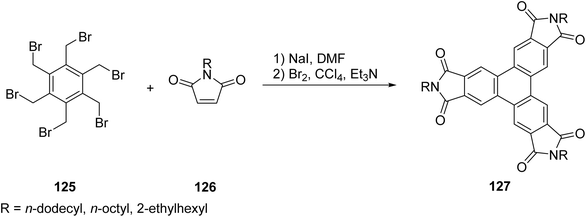 | ||
| Scheme 35 Synthesis of triphenylene derivatives having carboximides. Reproduced from ref. 100 with permission from American Chemical Society, copyright 2009. | ||
Osawa and coworkers used a similar approach to prepare triphenylene hexaesters.101 Initially, hexamethylbenzene 129 underwent benzylic bromination, yielding hexakis(bromomethyl)benzene 125 in quantitative yield. The subsequent Cava reaction of 125 with dimethyl acetylene-dicarboxylate (DMAD) in the presence of sodium iodide resulted in the formation of hexahydrotriphenylene with a modest yield of 14% (Scheme 36). This product was further oxidized with MnO2 to afford 129 in high yield (95%). The drawbacks of this approach are that the hexamethylbenzene starting material is relatively costly, and that the Cava reaction is low-yielding.
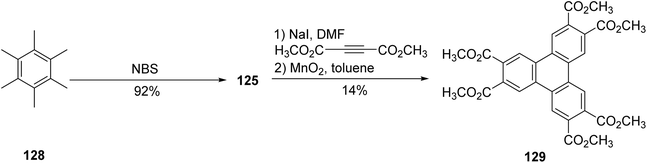 | ||
| Scheme 36 Preparation of triphenylene hexaester 129. Reproduced from ref. 101 with permission from John Wiley & Sons, copyright 2012. | ||
The SNArF approach has also been applied to the synthesis of fluorinated triphenylenes, including the preparation of 4F-substituted triphenylene (4F-TP) via the reaction of triphenylene bromide/lithium intermediates with Ar–F substrates, reported by Donnio et al. (2024) in ref. 102. This strategy enables late-stage fluorination and modular diversification of the triphenylene core, which is not readily achievable through classical oxidative cyclization or metal-mediated coupling routes. In this study,102 a series of aryl- and fluoroaryl-functionalized triphenylenes (Ar-TP; 131 and FAr-TP; 132) were prepared using two complementary methods: Suzuki–Miyaura cross-coupling for arylation and SNAr of perfluoroarenes for fluoroarylation (Scheme 37). Both strategies originate from the common precursor 2-bromo-3,6,7,10,11-pentakis(hexyloxy)triphenylene (Br-TP; 130). Some derivatives even display columnar order at room temperature and exhibit higher thermal stability than the benchmark 2,3,6,7,10,11-hexakis(hexyloxy)triphenylene. Structural variations significantly influence mesomorphic behavior. Increasing the size and rigidity of the side aryl substituents—from phenyl (P-TP) to naphthyl (N-TP), and further to biphenyl (BP-TP)—leads to higher clearing temperatures and broader mesophase stability. Fluorination of the peripheral aryl groups produces a similar effect: derivatives bearing a greater number of fluorine atoms show enhanced Colhex stability, consistent with stronger arene–fluoroarene interactions that reinforce columnar stacking.
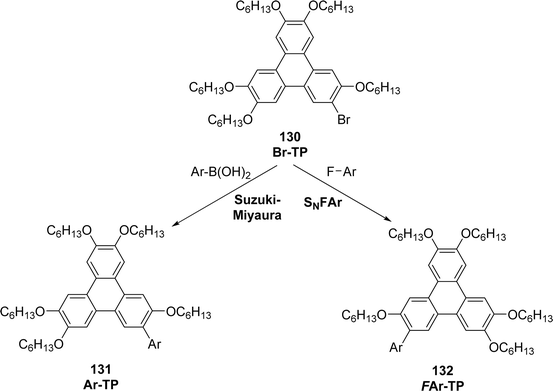 | ||
| Scheme 37 Synthesis of aryl- and fluoroaryl-functionalized triphenylenes. Reproduced from ref. 102 with permission from Elsevier under the terms of the Creative Commons CC-BY license, copyright 2024. | ||
Additional expansions of the methodology include Suzuki-type transformations on fluoroarene systems described in ref. 103, and the construction of Ar-FDLC materials designed for optoelectronic applications, as showcased in ref. 79, where fluoroarene-based mesogens were successfully integrated into LED architectures.
This work describes the synthesis of two structurally related families of π-conjugated aromatic derivatives constructed from a simple triphenylene scaffold and subsequently investigates their mesomorphic and optical behavior.103 This study aims to achieve two goals. The first is to elucidate how fluorination of the peripheral aryl ring influences both molecular self-assembly and photophysical behavior by systematically comparing Fn with their partially fluorinated PHn and non-fluorinated BTPn analogues—an essential step for tailoring structure–property relationships for targeted applications (Scheme 38). The second objective is to demonstrate that the terminal fluoroarene unit 135 serves as a versatile synthetic handle, enabling the construction of new π-extended architectures that would otherwise be difficult to obtain using conventional methods. Together, these aspects underscore the potential of this approach to contribute both to the development of advanced functional materials and to the expansion of synthetic methodologies in aromatic π-systems.
 | ||
| Scheme 38 Synthesis of fluorinated triphenylene. Reproduced from ref. 103 with permission from Beilstein Institute for the Advancement of Chemical Sciences, a German non-profit foundation, copyright 2024. | ||
Collectively, these reports establish SNArF-driven fluoroarene chemistry as a rapidly emerging platform for generating structurally diverse and functionally rich triphenylene derivatives. The combination of predictable regioselectivity, high functional-group tolerance, and compatibility with polyfluorinated π-systems has enabled access to new molecular designs that were previously synthetically inaccessible. These advances underscore the need for a comprehensive updated review that integrates Yamamoto coupling, PCDHF chemistry, and SNArF methodologies into a unified understanding of contemporary triphenylene synthesis.
3. Applications for triphenylene and its derivatives
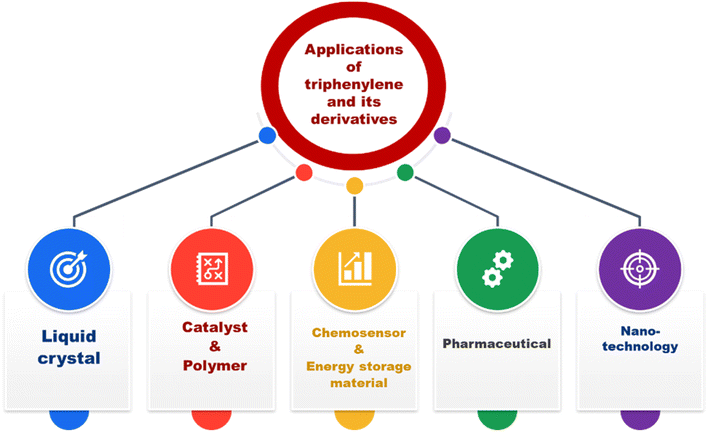
Triphenylene and its derivatives exhibit remarkable versatility, finding applications across various scientific and technological fields. In liquid crystal technology, their ability to form stable columnar mesophases through strong π–π interactions makes them valuable for semi conducting and light emitting properties and molecular self-assembly systems. As catalysts and polymers, triphenylene derivatives are used to create materials with enhanced mechanical and thermal properties, often serving as building blocks for high-performance polymers and catalytic systems in organic synthesis. In the field of chemosensing, triphenylene-derived materials are used as selective sensors capable of detecting ions, molecules, or environmental changes by taking full advantage of their adjustable photophysical characteristics and their strong bonding with the target analytes. Besides that, they are also used in energy storage applications, especially organic batteries and supercapacitors, due to their extended π-conjugation, which is responsible for the improved charge storage and transport abilities. Additionally, triphenylene derivatives are likely to be an attractive area of research in pharmaceuticals and nanotechnology, as their functionalized forms are particularly considered for drug delivery, bioimaging, and synthesis of nanoscaled materials for biomedical applications. Their unique structural and electronic properties position triphenylene as a cornerstone for developing innovative materials in both established and emerging fields (Fig. 8).3.1. Triphenylene derivatives as liquid crystals
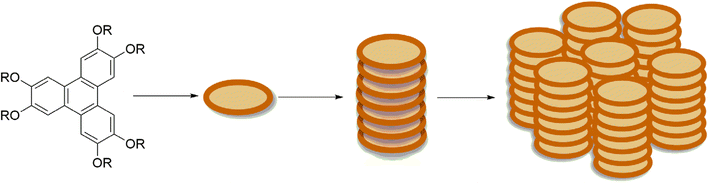
Triphenylenes are among the most common compounds used in liquid crystals applications. The triphenylenes possessing flexible peripheral chains can give rise to columnar liquid crystals, which consist of vertically arranged molecules (Fig. 9). Columnar liquid crystalline materials formed by triphenylenes have been thoroughly reviewed in different literature.8,14,36,104 In this section we aim to focus on structural and functional features of triphenylene relevant to their emerging applications.In optoelectronic and energy storage systems, columnar liquid crystals ordered molecule stacks can take on hexagonal, rectangular, or oblique configurations, enabling effective charge transfer. Tailoring the structure of triphenylenes allows control over their mesomorphic properties. Specifically, modification of side-chains, changing the linker between the side chains and the core, and incorporation of substituents on the core can all be used to control liquid crystalline properties. In addition, the incorporation of triphenylene units into dimeric and trimeric structures can be used to control properties.8
The design principles for discotic liquid crystals with varied molecular tails have been comprehensively reviewed.36 In these systems, modifying the side chains attached to the triphenylene core enables control over mesophase behavior.105 Zhao et al. undertook a study to investigate the effectiveness of using fluorinated chains on the self-assembling behaviour of triphenylene discotic liquid crystals to expand the knowledge concerning their mesomorphic behaviour and thermal stability (Fig. 10).106 Their studies showed that triphenylenes bearing fluorinated ester side chains 136, 137, 138 exhibited columnar hexagonal mesophases, and that the incorporation of fluorinated side chains increased the clearing points as compared to their non-fluorinated analogs.107
 | ||
| Fig. 10 Triphenylene discotic liquid crystals with fluorinated ester chains studied. Reproduced from ref. 107 with permission from Royal Society of Chemistry, copyright 2011. | ||
Beyond side chains, altering the core has proven impactful; for instance, triphenylene hexacarboxylic ester derivatives with electron-withdrawing ester groups influence the thermal stability, clearing temperatures, and overall mesophase behavior of discotic liquid crystals.108,109 In a related study, for example, Varshney synthesized compound 140A by nitration of hexaalkoxytriphenylene 139A and characterized its liquid crystalline properties. Most significantly, they possessed a wide thermal range in the hexagonal columnar liquid crystal phase. He also succeeded in showcasing structural isomerism by controlling the functionalization of specific sites on the triphenylene core to get different isomers. This work demonstrates that strategic core substitution both in terms of functional group identity and positional isomerism is a powerful tool for fine-tuning the thermal stability, mesophase range, and functional versatility of triphenylene-based discotic liquid crystals (Fig. 11).110
 | ||
| Fig. 11 Dinitro substituted hexa(alkoxy)triphenylene and monochlorohexa(alkoxy) triphenylene. Reproduced from ref. 110 with permission from Taylor & Francis, copyright 2024. | ||
Another approach to controlling the mesomorphic properties of triphenylenes is to link them together via groups capable of hydrogen bonding. For example, a novel liquid crystalline 1,3,5-benzenetrisamide bearing three triphenylene units has been synthesized, where intermolecular hydrogen bond-stabilizes columnar discotic liquid phase.111 This highly ordered compound exhibits a plastic columnar discotic hexagonal phase (ColhP), characterized by exceptional charge carrier mobility for triphenylene based liquid crystal (Fig. 12). The structural design promotes π–π stacking of triphenylene moieties, resulting in robust charge transport pathways. Further optimization of the molecular orientation within the π-stacked columns could enhance charge mobility, making this material highly promising for organic electronic applications.111
 | ||
| Fig. 12 (a) Structure of C3-symmetrical trisamide derivative 1 (b) image showing the central trisamide hydrogen-bond stabilized column (blue) surrounded by three triphenylene π-stacked coilumns (red). The hydrogen bond helical network is indicated by the blue arrow. Reproduced from ref. 111 with permission from American Chemical Society, copyright 2006. | ||
Early work by Sergeyev and co-workers8 demonstrated that benzene trisamide derivatives with peripheral triphenylene units could form unusual columnar mesophases, attributed to strong π–π stacking and hydrogen bonding within the trisamide linkers. Building on the concept of covalently linking triphenylene discotic units to a central core, El Mansoury and coworkers reported the synthesis of a series of novel star-shaped oligomers comprising triphenylene discogen units tied to a central benzene core.112 They showed that although a higher branching within the oligomers could generate liquid crystal properties within the system, longer chains were highly essential for the development of the desired columnar phase 141 (Fig. 13).112 More recently, Yang and coworkers focused on the development of novel discotic liquid crystal oligomers based on 1,3,5-triazine, featuring triphenylene dimers and trimers.113 The newly synthesized compound 142 (Fig. 14) exhibits broad mesophase ranges, combining the self-assembling capabilities of triphenylene with the structural versatility of the 1,3,5-triazine core. The presence of triphenylene units in both dimeric and trimeric forms improve the stability and functionality of the mesophases, thus making these materials quite advantageous for high-end applications in liquid crystal displays (LCDs) and optoelectronic devices. Through the analysis of their thermal and optical properties, the research brings out the noteworthy capability of these oligomers for the forthcoming liquid crystalline technologies.
 | ||
| Fig. 13 Model ‘star-shaped’ oligomers. Reproduced from ref. 112 with permission from Taylor & Francis, copyright 2012. | ||
 | ||
| Fig. 14 Chemical structure of 1,3,5-triazine-based triphenylene trimer. Reproduced from ref. 114 with permission from Taylor & Francis, copyright 2012. | ||
Modifying molecular structures through linkage groups such as in star-shaped and oligomeric triphenylene systems provides additional means to modulate mesophase type and stability.115 The diversity of the various synthetic strategies shows clearly that the molecular design can make the triphenylene-based discotic liquid crystals suitable for the optoelectronic and energy applications. Zhao and his co-workers started their novel work using the copper-catalyzed coupling cycloaddition reaction to create four different star-shaped oligomer structures.116 The oligomers were TP-discotic mesogens that played the key role in forming the right order of liquid crystal phases (Fig. 15). The oligomers have three lateral triphenylene units that are connected by methylene spacers to a central 1,3,5-tris(triazolyl)benzene core 143, where the length of the spacer is a critical factor in the mesophase formation. They further indicated that one non-mesomorphic oligomer could be used to form hydrogen-bonded supramolecular complexes with simple acids in the ratios of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 or 1
2 or 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 that led to the emergence of the columnar mesophases. Such an extension greatly enhanced their research, allowing them to obtain even deeper knowledge about the structural diversity and functional versatility of the materials involved in their study.116
3 that led to the emergence of the columnar mesophases. Such an extension greatly enhanced their research, allowing them to obtain even deeper knowledge about the structural diversity and functional versatility of the materials involved in their study.116
 | ||
| Fig. 15 Star-shaped triphenylene discotic oligomers. Reproduced from ref. 116 with permission from Royal Society of Chemistry, copyright 2015. | ||
Bi et al. synthesized and investigated several triphenylene esters with two ester substituents to improve the charge carrier mobility and structural ordering of discotic liquid crystals.117 These modifications promoted highly ordered columnar superlattice nanostructures through thermotropic self-assembly, which enhanced their electrical properties. Among these the 3,6-substituted triphenylene ester T5E36 144 (Fig. 16) created a unique helical hexagonal columnar superlattice structure composed of 91 right-handed helices. With bipolar charge carrier mobility of about 10−1 cm2 V−1 s−1, the helical superlattice nanostructure enhanced electrical characteristics and increased its appeal in organic electronics.
 | ||
| Fig. 16 Chemical structure of the T5E36. Reproduced from ref. 117 with permission from Royal Society of Chemistry, copyright 2019. | ||
Early studies demonstrated that covalently linked triphenylene oligomers and fused polycyclic frameworks could be accessed through controlled oxidative cyclization, annulative coupling strategies, and stepwise aryl–aryl bond formation, leading to rigid, shape-persistent architectures with extended conjugation. Triphenylene dimers represent a structurally distinctive class of π-conjugated systems, and when two triphenylene units are interconnected at the 3,6-positions via dipyrromethene linkers, they give rise to a novel family of macrocyclic architectures. These assemblies can be regarded as rigid, porphyrin-like expanded macrocycles in which structural constraints enforce near coplanarity despite the presence of a formally antiaromatic π-electron framework.118 Notably, the macrocyclization process results in an unexpected stabilization of the dipyrromethene chromophoric units, highlighting the beneficial electronic effects induced by rigidification and extended conjugation within the macrocyclic scaffold.119 Later on, quinoxalino[2′,3′:9,10]phenanthro[4,5-abc]phenazine (QPP) derivatives 145 are of interest as electron acceptors, fluorescent materials, and self-organizing organic semiconductors. Condensation of tetraketopyrene with electron-rich diamino terphenylene or triphenylene units yields donor–acceptor QPP systems that exhibit broad columnar mesophases, strong fluorescence across solution and solid states, and effective electron-accepting behavior. The study also reports the first synthesis of diamino-tetraalkoxytriphenylene, providing a useful new building block for functional material design (Fig. 17).120 These pioneering contributions established synthetic blueprints for assembling higher-order triphenylene-based systems while maintaining structural precision.
 | ||
| Fig. 17 Chemical structure of QPP-triphenylene derivative. Reproduced from ref. 120 with permission from American Chemical Society, copyright 2013. | ||
Subsequent efforts focused on electronic communication and photophysical modulation within fused or oligomeric triphenylene arrays. Investigations revealed that increasing molecular rigidity and conjugation length significantly influences exciton delocalization, fluorescence behavior, and charge carrier mobility.121 The incorporation of peripheral δ-methyl-branched alkyl chains together with aromatic moieties, including o-terphenyl or triphenylene units, onto a symmetry-lowered crown-ether core 146 (Fig. 18) results in the formation of hexagonal columnar mesophases that are stable at room temperature, with mesophase temperature ranges extending up to 147 K.122 These findings underscored the importance of controlled fusion patterns and molecular topology in dictating functional properties.
 | ||
| Fig. 18 Chemical structure of [15]crown-5 derivative. Reproduced from ref. 122 with permission from John Wiley & Sons, copyright 2016. | ||
More recent reports expanded this concept toward functional materials and device-relevant applications, including organic electronics and emissive systems. Several studies demonstrated that rigid triphenylene-containing oligomers exhibit enhanced thermal stability, improved mesophase organization, and superior optoelectronic performance compared to their monomeric analogues. Two new large N-heteroarene discotic systems based on triphenylene-type architectures were prepared using a modular cross-coupling/annulation strategy. Carbazole- and fluorene-fused ditriphenylene derivatives were synthesized via Suzuki–Miyaura coupling of suitable diboronic esters with alkylated 2-bromobiphenyl precursors, followed by FeCl3-mediated Scholl cyclization 147 (Fig. 19). Related fluorenone-fused ditriphenylenes were accessed through an inverted coupling sequence. Polarized optical microscopy revealed that several of these triphenylene-derived “butterfly” structures (carbazole (CTP), fluorenone (FTP) and fluorene (LTP)) exhibit mesomorphic behavior, self-assembling into hexagonal columnar phases characterized by fan-shaped textures, consistent with their rigid, π-extended discotic cores.123
 | ||
| Fig. 19 Chemical structure of CTP, FTP and LTP butterflies and precursors. Reproduced from ref. 123 with permission from John Wiley & Sons, copyright 2021. | ||
The work describes the synthesis of rigid π-bridged triphenylene dimers and highlights their liquid-crystalline behavior. Covalent linking of two triphenylene units produces enlarged, rigid aromatic cores that promote strong π–π interactions and efficient self-assembly. Several dimers exhibit stable hexagonal columnar mesophases over wide temperature ranges, showing enhanced mesophase stability compared with monomeric triphenylenes. This study demonstrates that π-bridging is an effective approach to modulate the self-organization and optical properties of triphenylene-based discotic liquid crystals.118 These attributes are particularly advantageous for applications requiring long-range molecular order and stability.
Deng et al., reported the design and synthesis of butterfly-shaped ditriphenylenothiophene derivatives, exploring how variations in the polyarene core topology influence their liquid crystalline behavior.124 The compounds self-assemble into columnar mesophases, with the core geometry strongly affecting the organization and stability of the mesophase. Optical studies reveal that the fluorescence properties are modulated by the molecular arrangement, while photoconductivity measurements demonstrate enhanced charge transport along the columnar stacks. Overall, this work highlights the critical role of polyarene core topology in tuning the self-assembly, optical, and electronic properties of triphenylene-based liquid crystals, offering insights for the design of functional discotic materials.124
Advanced synthetic methodologies have further enabled access to diverse fused triphenylene motifs, including ladder-type structures and extended polycyclic aromatic hydrocarbons 148, broadening the structural landscape of triphenylene-based materials in liquid crystals. This work reports the synthesis of triphenylene dimers connected via linear and bent alkyne–aryl–alkyne linkers (Fig. 20) and investigates their mesomorphic behavior.125 The rigid π-conjugated bridges promote strong π–π stacking, facilitating the formation of well-ordered columnar liquid-crystalline phases. The study highlights how linker geometry influences the stability, temperature range, and alignment of the mesophases. These structurally tunable triphenylene dimers demonstrate potential for applications in discotic liquid crystal devices, including organic electronics and optoelectronic materials, where controlled self-assembly and charge transport are critical.126 Such systems often display unique solid-state packing and enhanced intermolecular interactions, contributing to improved charge transport characteristics.125
 | ||
| Fig. 20 Chemical structure of alkyne–arylalkyne bridged triphenylene dimer. Reproduced from ref. 126 with permission from John Wiley & Sons, copyright 2022. | ||
Donnio et al. (2023) presented the development of star-shaped triphenylene–triazine architectures 149 as a new class of multi-stimuli-responsive discotic liquid crystals (Fig. 21).127 By integrating triphenylene discotic units with a triazine core, the resulting materials exhibit well-defined columnar liquid-crystalline phases driven by strong π–π stacking and peripheral chain organization. These systems respond sensitively to external stimuli such as temperature, light, and chemical environment, enabling reversible modulation of their mesomorphic and optical properties. Owing to their ordered columnar structures and tunable responsiveness, triphenylene–triazine discotic liquid crystals show significant potential for applications in organic electronics, optoelectronic devices, and smart functional materials.127
 | ||
| Fig. 21 Molecular structure of star-shaped triphenylene-phenyl-triazine derivative. Reproduced from ref. 127 with permission from the Chinese Chemical Society (CCS) and the Shanghai Institute of Organic Chemistry (SIOC), copyright 2023. | ||
Very recent contributions highlight the growing relevance of these rigid triphenylene architectures in next-generation functional materials, including luminescent systems and emerging device platforms. For example, tetraphenylethylene (TPE) and triphenylene (TP) units were combined within crossed-shaped tetrads (TPE-TP4; 150 and TPE-ThTP4; 151) via Suzuki–Miyaura coupling to develop multifunctional mesomorphic materials.128 TPE provides strong aggregation-induced emission (AIE), while TP promotes columnar self-assembly. TPE-TP4 (Fig. 22) exhibits crystalline behavior with reversible melting, whereas TPE-ThTP4 forms a rectangular multicolumnar (Colrec) phase over a broad temperature range (∼300 °C). Both compounds self-assemble into fibrous gels in various solvents and display strong fluorescence in solution and thin films (460–550 nm) with solvent-dependent quantum yields up to 84.5%. These findings highlight the potential of combining discotic mesogens with AIE-active units for advanced liquid crystal-based optoelectronic applications.128
 | ||
| Fig. 22 Star-like tetraphenylethylene-triphenylene tetrads 150 (TPE-TP4) and 151 (TPE-ThTP4). Reproduced from ref. 128 with permission from Elsevier, copyright 2025. | ||
A triskelion-shaped mesogen was constructed by linking a central hexabenzocoronene (HBC) core to three radial triphenylene (TP) units 152 (Fig. 23).129 The resulting π-extended oligomer exhibits a broad temperature columnar mesophase with self-sorted HBC and TP stacks arranged in a rectangular 2D lattice. Photophysical studies show combined absorption features from both HBC and TP, with emission primarily from HBC. The compound also displays ambipolar charge transport with a dominant electron mobility, highlighting its potential for liquid crystal-based electronic applications.129 These studies collectively demonstrate that oligomeric and fused triphenylene systems represent a fertile area for future exploration, particularly at the interface of molecular design, self-assembly, and materials performance.
 | ||
| Fig. 23 Chemical structure of HBC-3TP 152. Reproduced from ref. 129 with permission from Royal Society of Chemistry, copyright 2025. | ||
Taken together, these works emphasize that rigid and π-extended triphenylene-containing systems constitute a crucial and rapidly advancing subfield, complementing classical discotic liquid crystals and opening new avenues for high-performance organic materials.
Direct functionalization of the central aromatic core remains synthetically challenging due to steric and electronic constraints; however, new amidoalkylation protocols have enabled efficient C–H alkylation at internal triphenylene positions, producing C3-symmetric tris-alkylated derivatives in good yields and introducing versatile sites for further derivatization and tuning of electronic properties.130
C3-Symmetric triphenylene derivatives functionalized with amide groups at the 1-, 5-, and 9-positions 153 were designed and synthesized as modular building blocks for self-assembly (Fig. 24). The molecules formed supramolecular gels through a combination of hydrogen bonding, π–π stacking, and van der Waals interactions. Furthermore, by varying the length of the peripheral alkyl chains, these assemblies exhibited liquid crystalline mesophases. This study highlights a representative example of functional C3-symmetric triphenylene-based supramolecular materials.131
 | ||
| Fig. 24 Chemical structure of C3-symmetric triphenylene derivative functionalized with amide groups at the 1-, 5-, and 9-positions. Reproduced from ref. 131 with permission from Royal Society of Chemistry, copyright 2017. | ||
Dendronized triphenylene derivatives 154 were synthesized and shown to undergo hierarchical self-assembly into well-defined supramolecular architectures, including helical pyramidal columns and chiral spherical aggregates (Fig. 25).132 The formation of these structures is driven by a combination of π–π stacking, hydrogen bonding, and steric effects of the peripheral dendrons. These assemblies exhibit liquid crystalline behavior, with the ability to form columnar mesophases and chiral superstructures. This study demonstrates how functionalization of the triphenylene core with dendritic units enables precise control over molecular organization, offering potential applications in chiral optoelectronic materials and advanced liquid crystalline systems.132
 | ||
| Fig. 25 Dendronized triphenylene derivatives 154. Reproduced from ref. 132 with permission from American Chemical Society, copyright 2009. | ||
Mesogenic Rh(L4) complexes were investigated for their liquid crystalline behavior, revealing the formation of columnar mesophases in which RhI⋯RhI interactions and triphenylene-discotic cores segregate into distinct columns.133 The presence of the counterion A− = [Au(CN)2]− influences the packing and enhances charge transport, particularly hole mobility, within the mesophase. This study highlights how metal–metal interactions and discotic triphenylene units can be strategically combined to design liquid crystalline materials with tunable electronic properties.133
Coordination of metal ions to triphenylene-based Schiff base derivatives 155 significantly enhances hole mobility in their multicolumnar liquid crystalline mesophases (Fig. 26). The study demonstrates that metal–ligand interactions promote more ordered columnar stacking, improving charge transport while maintaining the mesomorphic behavior of the triphenylene cores. These findings underscore the potential of metal-functionalized triphenylene mesogens for high-performance organic electronic applications.134
 | ||
| Fig. 26 Chemical structure of triphenylene-based Schiff base metal complexes. Reproduced from ref. 134 with permission from American Chemical Society, copyright 2020. | ||
Triphenylene–bis(dithiolene)nickel complexes 156 form columnar liquid crystalline mesophases that exhibit soft photothermal properties (Fig. 27). The organized columnar arrangement of the triphenylene cores, combined with the nickel-dithiolene units, enables efficient light absorption and conversion to heat, highlighting their potential for photothermal applications in soft materials and optoelectronic devices.135
 | ||
| Fig. 27 Chemical structure of triphenylene-bis(dithiolene)nickel complexes. Reproduced from ref. 135 with permission from Royal Society of Chemistry, copyright 2024. | ||
Imidazolium-annulated triphenylene derivative 157 exhibits discotic columnar mesophases driven by the positive core charge of the imidazolium units (Fig. 28). The electrostatic interactions, combined with π–π stacking of the triphenylene cores, promote organized self-assembly into columnar structures, demonstrating the role of charged cores in modulating mesomorphic behavior and enabling potential applications in ionic liquid crystalline materials.136
 | ||
| Fig. 28 Chemical structure of imidazolium-annulated triphenylene derivative. Reproduced from ref. 136 with permission from Royal Society of Chemistry, copyright 2024. | ||
Collectively, these developments demonstrate that multifunctionalization strategies and metal incorporation expand the design space of triphenylene-based compounds well beyond simple discotic mesogens, enabling tailored self-assembly, enhanced electronic functionality, and new applications in organic electronics, sensing, and hybrid materials.
3.2. Triphenylene derivatives as catalysts
Triphenylene-based materials have emerged as catalysts due to their rigid, planar and π-conjugated structures which allow for strong π–π stacking interactions and charge transfer. These properties make them extremely suitable for photo-catalysis and electro-catalysis where light absorption and electron mobility are important. Their enlarged π-systems give rise to their catalytic potency by making the redox cycle easier, stabilizing the intermediate steps, and providing either electrons or accepting them. Besides, the molecular structures can be tailored to the characteristics required through functionalization which in turn leads to various applications in oxygen reduction, hydrogen generation, pollutant degradation, and other organic transformations. Their role in eco-friendly catalytic systems is further backed up by their durability, versatility and thermal stability.Triphenylene units, having an identity of their own, can endow strong π-conjugated systems for the accurate placement of catalytically active groups and tuning the electronic properties. For example, the rigid triphenylene structure plays a crucial role in controlling the geometry of a triphenylene-tris-NHC ligand synthesized and coordinated to Ir and Au centres,137 thus resulting in catalytic (yield/turnover) activity that is better than simple mono-metallic analogs. One potential direction for developing the next generation of catalysts could be the use of triphenylene in two-dimensional polymers. To illustrate, Zhao and co-workers have provided evidence that nitrogen-doped triphenylene-graphene monolayers are highly active as catalysts.138 These monolayers are capable of driving the reactions of water splitting, particularly the reactions of hydrogen and oxygen evolution, that under normal circumstances would require significant energy input, with almost no energy needed at all. Their catalytic efficiency is significantly improved in both acid and base environments, akin to the activity of ion exchange resins. Most importantly, the incorporation of nitrogen into the monolayers greatly facilitates these reactions and gives rise to catalytic activities similar to those of noble metals. Thus, this study significantly points out the attractiveness of the nitrogen-doped triphenylene-graphene monolayers as cheap non-precious metal catalysts, which leads to a practical and efficient supplying strategy of photocatalytic systems for water splitting and clean hydrogen fuel production near those metals.138
Only a handful of two-dimensional carbon-based substances are able to remove water entirely via photocatalysis although their carrier mobility is high and they have porous structures. According to a recent computational study, the triphenylene-acetylenic framework (compound 158 [GDY] & 159 [TDY]) possessing an electrical band structure favorable for water splitting was one of the promising eco-friendly, low-cost photocatalysts.139,140 The focus of this paper was on triphenyldiyne, a structure similar to graphene, where the replacement of a benzene ring with a triphenyl group increases the localization of the electronic states. The monolayer of TDY displays an impressive absorption coefficient over the range of visible light and has a direct bandgap of 2.70 eV. Its excellent carrier mobility opens up channels for rapid charge transfer making it possible to have very good redox capabilities to assist hydrogen evolution reactions (HER). In addition, the nickel doping lowers the overpotential needed for the oxygen evolution reaction (OER), thus raising the efficiency of the water-splitting process (Fig. 29).139
 | ||
| Fig. 29 Chemical structure of graphdiyne (GDY) and triphenyldiyne (TDY). Reproduced from ref. 139 with permission from Royal Society of Chemistry, copyright 2020. | ||
It is important to mention that although these characteristics were predicted by computation, there has been an experimental breakthrough in the production of triphenylene-cored graphdiyne analogues by Matsuoka and colleagues.141 A modified liquid/liquid interfacial polymerization technique was used to produce TP-GDY with hexaethynyltriphenylene monomer. The TP-GDY film produced had a free-standing structure, was smooth in texture, produced in large domains (>1 mm), and was analyzed through a range of microscopic, spectroscopic, and analytical methods.
The development of small-molecule catalysts made from triphenylene has shown that the aromatic core is a material with more applications.20 Peris and co-workers have turned triphenylene-based tris(NHC) ligands 162 into scaffolds for homogeneous catalysis after extensive exploration, making use of their rigid, planar, and π–rich aromatic core to boost the catalyst–substrate interactions via π–π stacking.20 In one of their studies, they developed a promising new type of AuI catalyst for organic reactions. This catalyst features a rigid, flat molecule with a triphenylene core at its center, “decorated” with three groups containing nitrogen and a heavy metal (palladium or gold). The peculiarity of this success is found in the geometry of the catalyst which has a 3D structure. The structure of tetra-substituted triphenylene core enable aromatic molecules-often a common component in the organic reactions to interact with the catalyst more effectively. This opens avenues for improved design of homogeneous catalysts, which are used in a majority of the processes in industries (Scheme 39).20
 | ||
| Scheme 39 Synthesis of D3h- symmetric trisazolium salt. Reproduced from ref. 20 with permission from Royal Society of Chemistry, copyright 2013. | ||
Building on same concept, a series of triphenylenes with N-heterocyclic carbene groups (163, 164) were reported as potential transition metal ligands for catalysis.142 These triphenylene based tris-NHC ligand exhibits unique topological features, enabling the coordination of three metal fragments within a pseudo-D3h symmetric environment, characteristic not observed in other tris-NHC ligands reported so far. The rigid triphenylene core facilitates supramolecular interactions between the ligand and aromatic substrates, resulting in catalysts (Palladium (Pd) and Gold (Au) complexes) that outperform their monometallic counterparts lacking a polycyclic aromatic framework (Fig. 30).
 | ||
| Fig. 30 Tris(N-heterocyclic carbene) ligands based on triphenylene for Pd(II) (163) and gold(I) (164) that shown catalytic advantages in benchmark processes. Reproduced from ref. 143 with permission from Royal Society of Chemistry, copyright 2013. | ||
Using the substrates containing aryl groups, three test reactions: Compound 163 was produced by hydroaminating phenylacetylene. Compound 164, a precursor to the tris(NHC) triphenylene complexes of Au(I)Cl and Pd(II), was produced by a Suzuki–Miyaura coupling and a Csp3–Csp2 (aryl) coupling. In fact, catalysts were marginally more efficient than their monometallic (benzimidazol-2-ylidene ligand) and unconjugated tris-metallic (triptycene ligand) counterparts, as seen by the 7–20% increase in yields of catalyzed processes over uncatalyzed ones.143
Two triphenylene-based tris(N-heterocyclic carbene)-gold–acetylide main-chain organometallic microporous polymers were obtained and characterized.144 They have spherical shapes and showed high activity in catalytic reduction of nitroarenes with NaBH4 and Strecker reaction for aα-aminonitriles synthesis. The activity may be due to Au nanoparticle formation. Gonell and coworkers reported the preparation of a planar triphenylene derived tris-NHC which can be utilized to prepare a range of Pd and Au ancillaries with better catalytic performance.145 Due to the symmetry of the ligand and the presence of three p-species in this tris-NHC complex, they expected similar molecular connection that would lead to the formation of interesting extended structure that can exhibit some exotic physical and chemical properties. Moreover, for the synthesis of triphenylene-based tris-NHC 167, which is favorable compared to the ligands used in the that three carbenes share an extended p-delocalized polyaromatic system. Presumably, it is capable of being used as a component molecular building block to synthesize electronically active materials (Scheme 40).145
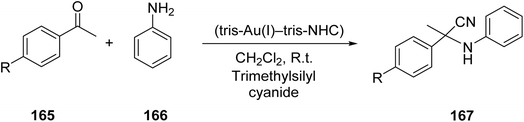 | ||
| Scheme 40 Three-component Strecker reaction of ketones using triphenylene catalyst. Reproduced from ref. 145 with permission from John Wiley & Sons, copyright 2014. | ||
Moreover, although 1D organometallic polymer derived from NHC and comprising a π-extended system has been reported, the corresponding π -extended 2D or 3D materials have yet been described. Recently, triphenylene-based tris-NHC–gold–acetylide metallopolymeric materials were synthesized via a straightforward mixing approach. These compounds demonstrated catalytic utility in the three-component Strecker reaction to afford α-aminonitriles, as well as in the reduction of nitroarenes to anilines (Scheme 41).145
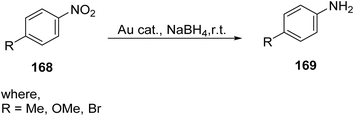 | ||
| Scheme 41 Reduction of nitroarenes to anilines. Reproduced from ref. 145 with permission from John Wiley & Sons, copyright 2014. | ||
3.3. Triphenylene derivatives as light emitting materials
Triphenylene is a π-conjugated, planar molecule that exhibits superior photostability and thermal stability. Triphenylenes have attracted attention as light-emitting materials due to their stiff planar structure, high temperature stability, and efficient fluorescence. It functions as a core for light-emitting components in OLEDs, particularly those that generate blue or blue-green light. One of the challenges of incorporating triphenylenes into light-emitting devices is that they tend to aggregate in the solid state, which causes fluorescence quenching. Therefore, one of the important design considerations for light-emitting triphenylenes is preventing aggregation.146For example, Saleh and coworkers studied the synthesis of new blue-emitting triphenylene-based polymers used in polymer light-emitting diodes (PLEDs).18 Various soluble triphenylene derivatives were polymerized using Suzuki–Miyaura and Yamamoto reactions to give polymer 170 and compound 171, which are blue emitters (Fig. 31). Thus, obtained polymers revealed a feature that polymers were emitting blue light. It is important to note that in solutions and thin film applications, the light-emitting properties remained constant. Nevertheless, the researchers noticed that the deposition of thin films altered some of its previously observed properties of light absorption and emission than those of the solutions. This indicates that TP-based polymers could be potential candidates for blue-PLEDs by remotely monitored light-emitting properties and behavior of the polymers inside thin film devices.18
 | ||
| Fig. 31 Polymers 170 synthesized via Suzuki–Miyaura coupling reaction. Polymers 171 synthesized via Yamamoto Coupling reaction. Reproduced from ref. 18 with permission from American Chemical Society, copyright 2010. | ||
Based on these studies, Saleh and colleagues extended their research to structurally modify the polymers by changing side chains in order to enhance OLED device performance.147 Three different conjugated triphenylene polymers were investigated by them for their potential use in OLEDs. Each polymer had a core structure of triphenylene units but side chains differed in that phenyl and alkyl groups are attached. The research effort was aimed at optimization device performance, specifically reducing the voltage required to turn on the OLED (onset voltage) and boosting its efficacy converting electricity into light (luminance efficiency). Poly(2-heptyl-3-(4-octylphenyl)-1,4-diphenyl-6,11-triphenylenyl-1,4-benzene) 172 showed the most promising properties, with both lower onset voltage and higher luminance efficiency (0.73 cd A−1), indicating that it is crucial to carefully select components within an OLED in order to achieve optimum performance (Fig. 32). This means that altering side chain structure of triphenylene polymers may be an effective approach to designing efficient OLEDs.147
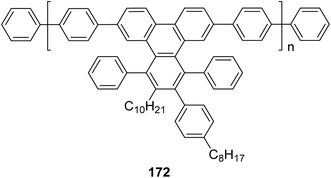 | ||
| Fig. 32 Structure of poly(2-heptyl-3-(4-octylphenyl)-1,4-diphenyl-6,11-triphenylenyl-1,4-benzene). Reproduced from ref. 147 with permission from John Wiley & Sons, copyright 2009. | ||
Two novel iridium complexes, YF3 (173) and YF4 (174), with a triphenylene unit have been developed by Wang and coworkers for potential use in OLEDs (Fig. 33).148 Here, the triphenylene units do not participate in the emission, rather they facilitate charge transport and improve device performance. When solid or dissolved, both YF3 and YF4 show strong blue-green emission. Interestingly, the film of YF4 complex annealed at 600 °C gave the best performance. It was through this annealing process that an extremely high hole mobility value of 0.001 cm2 V−1 s−1 necessary for efficient OLED operation was achieved. This highlights the effectiveness of ancillary ligand modification in enhancing electroluminescent device efficiency. It also indicates that integrating TP units into iridium complexes can lead to excellent OLEDs.
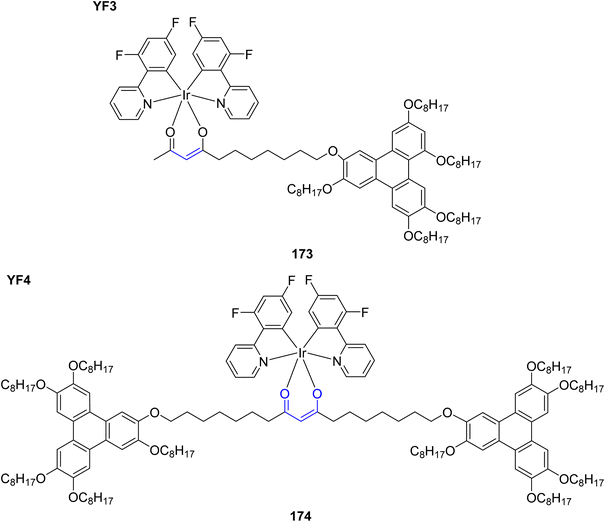 | ||
| Fig. 33 Chemical structure of YF3 & YF4. Reproduced from ref. 148 with permission from Royal Society of Chemistry, copyright 2017. | ||
Pal and coworkers have synthesized a new series of DLCs based on triphenylene and pentaalkynylbenzene units (175, Fig. 34).149 Long alkoxy chains promote the transfer of charge and solubility, while the central triphenylene unit increases the efficiency of photoluminescence. OLED devices thus have efficient blue emission and pure colour.149 These non-symmetric molecules emit blue light in solution and align well in specific ordered structures. The researchers used two of the DLCs as emitters in doped OLED devices. The device made with 175 (Fig. 34) exhibited the best performance, achieving good efficiency and color purity.149
 | ||
| Fig. 34 Chemical structure of target compounds 175. Reproduced from ref. 149 with permission from Royal Society of Chemistry, copyright 2019. | ||
In addition to light emitting materials, the semiconducting properties of triphenylene derivatives means they can also be explored as charge transport materials. Park and coworkers synthesized two new hole transport materials for OLEDs, substituted triphenylene derivatives with diarylamino groups 176 and 177 (Fig. 35) known for their thermal stability.150 These materials were compared to a commonly used material, N,N′-di(naphthalene-1-yl)-N,N′ diphenyl benzidine (NPB). The study found that both 176 and 177 performed well as hole transport layers in OLEDs due to their thermal stability. This makes them suitable for applications requiring high thermal stability and low power consumption.150
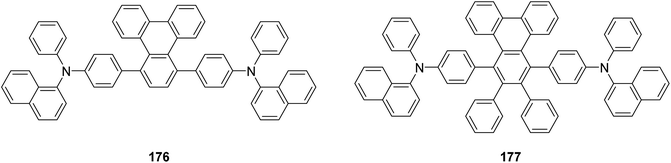 | ||
| Fig. 35 Chemical structures of substituted triphenylene derivatives with diarylamino groups. Reproduced from ref. 150 with permission from Elsevier, copyright 2011. | ||
In a subsequent study, the same group synthesized novel blue emitters. These emitters are built from triphenylene and diphenyl fluoranthene derivatives (Fig. 36).151 To evaluate the electroluminescence efficacy of the new emitters, these were embedded into multistack OLEDs. Using these emitters, publicize devices shown enhanced luminous, power, and external quantum efficiencies. Compound 178 showed higher luminous efficiency of 2.34 cd A−1, and CIE (0.17, 0.23). In contrast, Compound 179 exhibited deeper blue emission with CIE (0.16, 0.11). Thus, compound 178 offers better performance, while compound 179 achieves pure blue color. The function of triphenyl fluoranthene derivatives and triphenylene for blue emitters in OLED suggests that they have potential for applications.152
 | ||
| Fig. 36 Chemical structures of 1,4-bis(7,10 diphenylfluoranthen-9-yl)benzene and 1-(1,4-diphenyltriphenylen-2-yl)-3-(1,4-diphenyltriphenylen-3-yl) benzene. Reproduced from ref. 152 with permission from Elsevier, copyright 2015. | ||
3.4. Triphenylene derivatives as sensors
Triphenylenes are increasingly popular in sensor design because of their outstanding photostability, adjustable electrical characteristics, and stiff, π-conjugated architectures. Stable signal production and effective interaction with analytes are made possible by these features. Sensors may be designed for selective detection by adding functional groups to the triphenylene core, which makes them useful for chemical, biological, and environmental sensing applications.153,154Triphenylene derivatives 180 and 181 (Fig. 37) interact with electron-deficient nitroaromatics by means of their electron-rich π-systems, resulting in highly sensitive sensors for nitroaromatic compounds. The use of them in detecting traces of nitroaromatic explosives depends on the quenching of fluorescence that results from this interaction.153 Fluorescence titrations were performed using these compounds (5 µM) in a THF![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9.5
H2O (9.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5) combination with several nitroaromatic derivatives. Because of its extended conjugation, derivative 180, which contains carbazole moieties, has a strong emission intensity. Conversely, derivative 181, which contains triazole moieties, may form supramolecular aggregates both in solution and in bulk. Because of its ordered structure, derivative 181 shows a more sensitive reaction to nitroaromatic derivatives than derivative 180, even though derivative 181 has a higher emission intensity. Photoinduced electron transfer (PET) is used for detection, which quenches the fluorescence. Because of its ordered arrangement and aggregation behaviors, 181 offers greater sensitivity than 180, which displays strong emission due to reached conjugation. The study highlights how molecular organization enhances nitroaromatic selective detection. The current work thus highlights the significance of well-organized systems for the sensitive identification of nitroaromatics.153
0.5) combination with several nitroaromatic derivatives. Because of its extended conjugation, derivative 180, which contains carbazole moieties, has a strong emission intensity. Conversely, derivative 181, which contains triazole moieties, may form supramolecular aggregates both in solution and in bulk. Because of its ordered structure, derivative 181 shows a more sensitive reaction to nitroaromatic derivatives than derivative 180, even though derivative 181 has a higher emission intensity. Photoinduced electron transfer (PET) is used for detection, which quenches the fluorescence. Because of its ordered arrangement and aggregation behaviors, 181 offers greater sensitivity than 180, which displays strong emission due to reached conjugation. The study highlights how molecular organization enhances nitroaromatic selective detection. The current work thus highlights the significance of well-organized systems for the sensitive identification of nitroaromatics.153
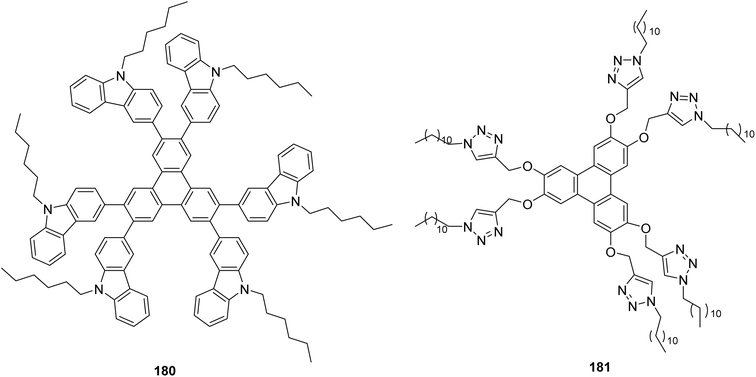 | ||
| Fig. 37 Chemical structure of carbazole substituted triphenylene and triazole modified triphenylene. Reproduced from ref. 153 with permission from Royal Society of Chemistry, copyright 2013. | ||
The Lewis acid coordination or hydrogen bonding is the basis for the majority of the known fluoride ion receptors. The lack of sensitivity toward fluoride ions at low concentrations severely limits their effectiveness as highly selective systems. Conversely, reaction-based receptors, such as chemo dosimeters, have elevated selectivity and sensitivity.155 Triphenylene derivative 182 (Fig. 38) with twelve OTBS groups has been produced in good yield.155 As tetrabutylammonium fluoride is used to deprotect OTBS groups, it experiences an unparalleled irreversible cyclization to super triphenylene 183 by the addition of triflic anhydride (Fig. 38), which enables it to function as a highly selective chemosensor for F-ions. This transformation leads to a marked enhancement in fluorescence, due to the formation of a rigid, planar π-conjugated system upon cyclization, which restores the emissive properties of the molecule. Additionally, it has been shown how useful TLC strips coated with derivative 182 are for quickly detecting fluoride ions in aqueous conditions.
 | ||
| Fig. 38 Chemical structures of super triphenylenes. Reproduced from ref. 155 with permission from Elsevier, copyright 2012. | ||
Competitive experiments were conducted in the presence of F− at 12 M mixed with Cl−, Br−, I−, OAc−, HSO4−, NO3, H2PO4, and CN− at 200 M to test the practical applicability of compound 182 as a F− selective sensor. No obvious change in fluorescence behavior was seen either is the presence or absence of the other anions besides F−. A TLC strip was prepared by immersing it in the 182 solution in THF in order to test its practical usefulness for fluoride detection in water. The strip was first air-dried and then dipped into a potassium fluoride solution where a color change was instantly seen which was an indication of the presence of fluoride ions.155
Given the ability of triazole groups to act as multi-dentate ligands for metal coordination, the inclusion of six triazole moieties in compound 184 prompted an investigation into its binding interactions with various metal ions (Fig. 39). This study aimed to explore the compound's coordination behavior, which could provide insights into its potential applications in metal-ion sensing, catalysis, and other coordination-driven processes. They added aliquots of various metal ions to DMSO and conducted UV-Vis and fluorescence studies to assess compound the binding capacity of compound 184 toward metal ions.156
 | ||
| Fig. 39 Structure of triazole modified triphenylene. Reproduced from ref. 156 with permission from Royal Society of Chemistry, copyright 2012. | ||
Since anions play a significant role in a variety of clinical, biological, and environmental systems, there is considerable interest in the development of chemo sensors for anion detection. UV-Vis and fluorescence spectroscopy have been used to examine the binding behavior of an in situ-prepared TP-based copper ensemble toward different anions. Among the many anions studied, the 184 ensemble reacts to CN− ions and is sensitive to cyanide ions, even when blood serum milieu and bovine serum albumin are present. Additionally, they used TLC strips coated in compound 132HF solution for the solid-state detection of copper and cyanide ions as a practical use of 184.156
An alternative approach to using triphenylenes as a sensor takes advantage of their tendency to self-assemble and aggregate in solution. For example, compounds 185 and 186 (Fig. 40) exhibit aggregation enhanced emission in aqueous media.157 In the presence of picric acid, this luminescence is quenched by energy transfer from the aggregates to picric acid. It is noteworthy that this system showed excellent sensitive to picric acid able to identify even minute amounts of explosive picric acid in aqueous media at nanomolar concentrations. Additionally, the derivative 185 solution-coated test strips have an up to 22.9 × 10−15 g cm−2 detection limit for picric acid, making it an affordable, portable, and user-friendly approach for on-site trace detection.158
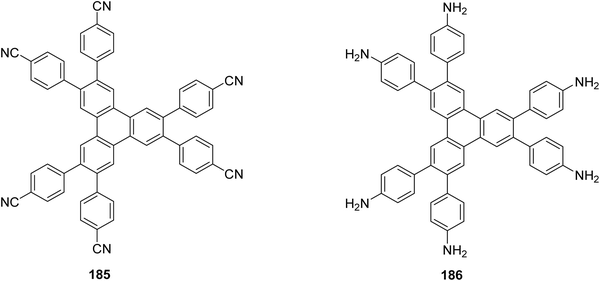 | ||
| Fig. 40 Chemical structure of triphenylene derivatives. Reproduced from ref. 158 with permission from Royal Society of Chemistry, copyright 2015. | ||
3.5. Triphenylene derivatives as COFS
Covalent organic frameworks (COFs) are a unique class of highly porous, crystalline materials composed entirely of light elements such as hydrogen, boron, carbon, nitrogen, and oxygen, all interconnected through strong covalent bonds.159 Due to its delocalized π-system, rigid planar structure, excellent stability, and tunable architecture, triphenylene is a valuable building block in the construction of both COFs and Metal–Organic Frameworks (MOFs).154,160 These characteristics of the triphenylene-based materials help in the formation of supramolecular interlayer stacking within the MOFs and COFs that eventually lead to an increase in their thermal and chemical stability as well as electrical conduction. Moreover, the resulting materials exhibit uniform nanoporous channels, contributing to high surface area and porosity, properties that are highly desirable for gas storage, molecular sieving, and ion exchange applications.160The first COFs were reported by Côté, Yaghi, and coworkers in 2005, where they produced microcrystalline networks by condensation of boronic acids with diols to form boronate ester linkages.161 Specifically, COF 188 was constructed through the condensation reaction of hexahydroxytriphenylene 187 with benzene-1,4-diboronic acid (Fig. 41). These frameworks represented the first examples of porous, crystalline organic materials assembled entirely through covalent bonds, exhibiting both high surface area and long-range order.161
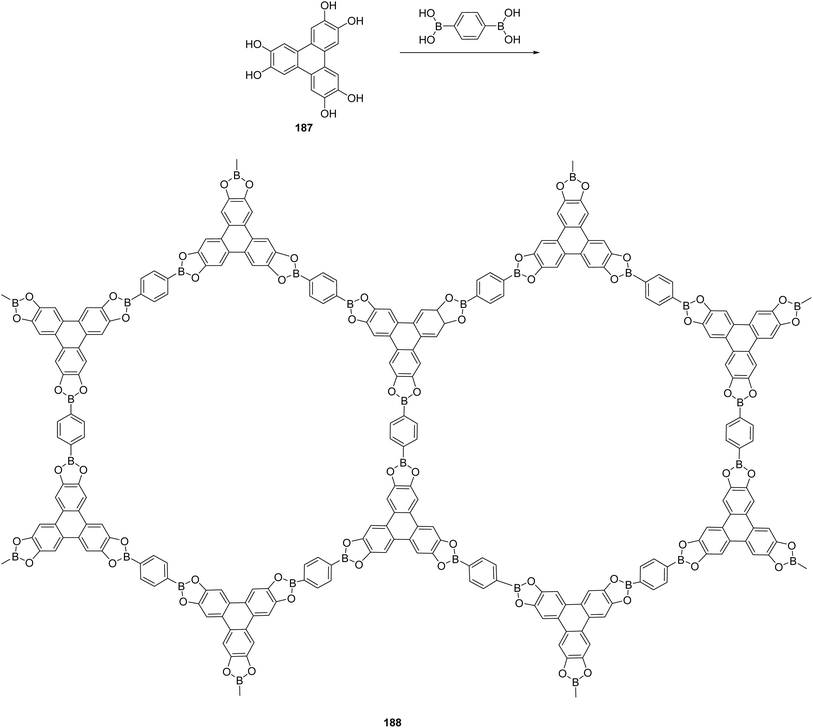 | ||
| Fig. 41 First COFs constructed through the condensation reaction of hexahydroxytriphenylene with benzene-1,4-diboronic acid. Reproduced from ref. 161 with permission from American Association for the Advancement of Science, copyright 2005. | ||
Bein and co-workers later studied thieno[2,3-b]thiophene-based COFs, where the incorporation of sulfur atoms into the central ring system contributed to both high surface area and improved crystallinity.162 These improved properties make the COFs more effective for photocatalysis. Interestingly, the study also revealed that non-planar aromatic systems can “lock” COF layers together. This finding suggests that this locking mechanism might be applicable to other structural cores as well.163 Jiang investigated luminescence and semiconducting behavior of a new class of materials called Triphenylene-Pyrene COFs, which are COFs with extended π-conjugated systems, formed by combining hexahydroxytriphenylene 187 and pyrene-2,7-diboronic acid 189, resulting in a unique belt-like hexagonal structure. This structure is particularly interesting because it efficiently captures light over a broad range of wavelengths. As a result, these COFs can transfer this energy throughout the material, leading to blue luminescence.164 The change in fluorescence intensities at 474 and 402 nm indicates electronic coupling, favoring energy transfer in the extended TP-COF structure. The material's stability was demonstrated through continuous on-off switching without important weakening of the electric current. The self-condensation of 189 also resulted in PPy-COF with a pore diameter of 1.73 nm and a surface area of (923 m2 g−1) (Fig. 42).165 PPy-COF displayed photoconducting behavior as well as p-type semiconducting behavior with electrical conductivity of the same order as that of triphenylene-pyrene COF.
 | ||
| Fig. 42 Chemical structure of pyrene-2,7-diboronic. Reproduced from ref. 165 with permission from John Wiley & Sons, copyright 2017. | ||
Chen and colleagues successfully synthesized uniform hollow spherical 2D COFs using 2,3,6,7,10,11-hexakis(4-aminophenyl)triphenylene as a building block (HAPTP).166 The resulting COF 192 is distinguished by its high crystallinity, uniform structure, and a remarkable pore volume reaching 1.947 cm3 g−1, positioning them as ideal nanocarriers for controlled drug delivery. HAPTP was synthesized by using a Suzuki cross-coupling reaction between 2,3,6,7,10,11-hexabromotriphenylene and boronic ester achieving quantitative yield. Morphology exposure by SEM and TEM revealed hollow spherical morphology resulting in high ibuprofen, a non-steroidal anti-inflammatory drug, loading capacity and sustained release as shown in Fig. 43.
 | ||
| Fig. 43 Schematic illustration of an IBU molecule in a triangular pore of HAPTP-TPA 2DCOFs. Reproduced from ref. 166 with permission from American Chemical Society, copyright 2023. | ||
Considering the low dispersibility and solubility of COFs, Chen and colleagues prepared a water-soluble nanocomposite COF@IR783 by assembling cyanin IR783 with a triphenylene COF.167 The COF itself was synthesized through a co–condensation reaction between 2,3,6,7,10,11-hexahydroxytriphenylene (HHTP) pointed at vertices and 5,15-bis(4-boronophenyl)porphyrin (Por) at center, resulting in a porous hexagonal framework.168 The resultant 2D COF with IR783, exhibited a nanoscale morphology (∼200 nm), aqueous dispersibility, and a negative charge (−36 mV), all of which are beneficial for extending blood circulation time and enhancing the enhanced permeability and retention (EPR) effect (Fig. 44). It was also used as a drug carrier for the prodrug cisaconityl-doxorubicin (CAD). The combination of photothermal therapy (PTT) and chemotherapy led to a significant reduction in the viability of 4T1 cells in vitro, with a cell survival rate of 19.8%.
 | ||
| Fig. 44 Schematic illustration of 2D CON fabrication and in vivo cancer therapy. Reproduced from ref. 168 with permission from American Chemical Society, under the terms of the CC BY 3.0 license, copyright 2019. | ||
Chen and coworkers presented COF created by stacking 2D covalent organic nanosheets (CONs) into long range ordered π-columnar structures and periodically ordered bicontinuous heterojunction networks.167–170 The electron and hole lifetimes of CONs can be extended, and effective carrier separation can be achieved because of their molecular hetero structure. In the meantime, type I PDT mechanism allows electrons to decrease O2 to O2−, which was advantageous for ROS formation. In this experiment, liquid ultrasonic exfoliation was used to directly manufacture CONs from bulk triphenylene-porphyrin COFs. Under 635 nm laser irradiation, intravenous injection of the resultant CONs into nude mice resulted in a considerable tumor ablation.168
Bein and colleagues developed a triphenylene-based COF by co-condensing thieno[3,2-b]thiophene-2,5-diyldiboronic acid (TTBA) with hexahydroxytriphenylene (HHTP).162 The resulting COF effectively incorporated [6,6]-phenyl-C61-butyric acid methyl ester (PCBM), as evidenced by BET surface area reduction and photoluminescence quenching. A photovoltaic device using ITO/TT-COF![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PCBM/Al achieved a power conversion efficiency of 0.053% with 0.62 V open-circuit voltage. Monte Carlo simulations suggested that increasing pore size could further enhance charge transfer. This COF highlights potential for use in organic photovoltaic applications.
PCBM/Al achieved a power conversion efficiency of 0.053% with 0.62 V open-circuit voltage. Monte Carlo simulations suggested that increasing pore size could further enhance charge transfer. This COF highlights potential for use in organic photovoltaic applications.
3.6. Triphenylene derivatives as pharmaceuticals
Triphenylene derivatives have gained increasing interest in the pharmaceutical field due to their diverse chemical structures and potential therapeutic properties. Their polycyclic aromatic nature enables them to interact effectively with biological systems, showing promising results in various pharmacological activities such as antimicrobial171 and anticancer172 effects. Modifications to the triphenylene core, including functionalization and substitution with different functional groups, further enhance their bioactivity and selectivity, making them suitable candidates for drug development and delivery systems.173 Additionally, the ability of these derivatives to form self-assembled structures adds another layer of versatility for use in advanced pharmaceutical applications.Triphenylene dimethyl succinimide (TDS) is a novel class of organic compounds that have potential for treatment of acquired immune deficiency syndrome (AIDS).174 Loret and coworkers designed ten derivatives of TDS (193 in Fig. 45) and verified their efficacy against Tat, which is a viral protein having key role in the HIV virus lifecycle, helping in virus replication and spreading in body.174 HIV infected cells secrete Tat which transactivate distant infected cells inducing immunodeficiency in non-infected T-cell and causing other issues like Kaposi's sarcoma. TDS molecules bind with Tat protein and inhibits activation of HIV-LTR in HeLa cells at µM concentration. Mickael and coworkers also synthesized a new family of antivirals having TP aromatic ring substituted with one carbon chain bearing the same succinimide group.175
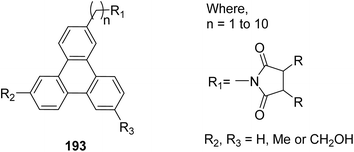 | ||
| Fig. 45 TDS derivatives synthesized 193. Reproduced from ref. 174 with permission from De Gruyter, copyright 2001. | ||
Naidek and coworkers synthesized 2,3,6,7,10,11-hexahydroxytriphenylene (HHTP), which exhibits catechol redox-properties, accessible higher oxidation state thus causing cytotoxic effects on 5 human cancer cell lines.176 They compared the cellular toxicity of HHTP with a standard chemotherapy drug cisplatin, which is widely used in the treatment of several human cancers. They treated human glioma cell lines with HHTP and cisplatin at concentrations ranging from 5 to 25 µmol L−1 for 48 hours finding that the in vitro cytotoxicity of HHTP was nearly identical to that of cisplatin across all tested drug concentrations. HHTP also exhibits effects comparable to those of other polyphenolic compounds, such as curcumin, apigenin, genistein, epigallocatechin, and epigallocatechin-3-gallate, which are known to make apoptosis in glioma and lung tumor cells and enhance the anti-neoplastic efficacy of chemotherapeutic drugs.177
Hayashida and coworkers prepared a water-soluble cyclophane hexamer (Fig. 46) via Williamson ether synthesis of tetraaza[6.1.6.1]paracyclophane bearing a bromoacetamide moiety with hexahydroxytriphenylene as core and as central platform for multivalent receptor.172,178 It can serve as molecular receptor to bind daunorubicin (DNR) and doxorubicin (DOX), which are anticancer drugs. An aqueous HEPES buffer was used to assess the binding interactions with these drugs, through fluorescence emission measurements. The association constant was found to be significantly high at 1.3 × 105 M−1, in contrast to the lower value of 3.3 × 102 M−1 observed with a single macrocycle, which can be attributed to the multivalent effect. The complex formation is primarily driven by a hydrophobic effect that facilitates the association between the triphenylene core and the amphiphilic guest, along with additional weak interactions.
 | ||
| Fig. 46 Water-soluble hexa-cyclophane triphenylene that detect daunorubicin, an anticancer drug. Reproduced from ref. 172 and 178 with permission from American Chemical Society, copyright 2016. | ||
Bibal created a triphenylene hexacarboxylate 194 that is soluble in water and intended to complex biological ammoniums like dopamine, serotonin, aceylcholine, L-dopa etc.179 Then, this simple receptor provided a hydrophobic centre and negatively charged groups to target substances that are amphiphilic, like neurotransmitters. The monitoring of acetylcholine complexation by infrared spectroscopy indicated that the guest's ammonium group was interacting with the host carboxylate groups (exchange with sodium) while the ester group of the guest was desolvated during complexation. The triphenylene hexacarboxylate was able to recognize D-glucosamine (Ka = 87 M−1) and acetylcholine (Ka = 94 M−1) with a moderate strength in phosphate buffered water. In phosphate buffered water, catecholamines that had an aromatic group attached to them were complexed with greater binding constants ranging from 200 M−1. Molecular dynamics revealed two types of host–guest interactions in water: π–π interactions and ionic interactions (between CO2− and +H3N groups). Remarkably, no contact was found between the host and analogous guests such L-dopa, D-glucose (lack of an ammonium group), and choline (more hydrophilic than acetylcholine). Thus, in phosphate-buffered water, the triphenylene hexacarboxylate showed a slight selectivity towards biological ammoniums.
3.7. Triphenylene derivatives in nanotechnology
Triphenylene derivatives have found significant applications in nanotechnology due to their rigid, planar structure, strong π–π stacking interactions which facilitates their integration with nanoparticles to develop functional nanocomposites for chemical sensing, drug delivery and optoelectronic applications.One of the notable applications involves the use of silver nanoparticles (AgNPs) functionalized with bis-acridinium lucigenine (LG) for surface-enhanced Raman scattering (SERS)-based detection of triphenylene.180 This allows the ability to locate organic contaminants with more accuracy. The acridinium planes are staggered, creating cavities for hydrophobic triphenylene. The orientation of LG changes with triphenylene concentration, with perpendicular orientation preferred. This technique can detect trace triphenylene concentrations, making it a potential organic pollutants chemical sensor. LG concentration did not significantly affect the Raman signal, but the limit of detection was approximately 10−7 M. When triphenylene interacted with LG, its structural shape remained unchanged, but when the concentration of triphenylene decreased, its orientation changed. This study investigated the interaction between triphenylene and LG in the formation of a LG/triphenylene complex on metal surfaces, using LG-functionalized Ag nanoparticles for triphenylene detection and analyzing SERS spectra to understand triphenylene's interaction with LG.180
Kumar and colleagues described triphenylene based discotic liquid crystals have been used to functionalize gold nanoparticles (GNPs), resulting in nanocomposites with enhanced electrical conductivity and stable mesophase behavior181 (Fig. 47). These were synthesized and uniformly dispersed within a columnar DLC matrix. Thermophysical characterization confirmed the incorporation of the nanoparticles into the matrix without disrupting the intrinsic mesophase organization, aside from minor shifts in transition temperatures. Most notably, doping the columnar matrix with these functionalized nanoparticles led to a remarkable enhancement in electrical conductivity, exceeding six orders of magnitude, under ambient conditions.
 | ||
| Fig. 47 There are three methods to create a 1D arrangement of NPs: (a) create molecules by combining gold nanoparticles stabilized by TPDn and triphenylene ligands (TPDn, n = 4,8, or 12); increase the amount of free space between the triphenylene moieties: (b) utilize a longer alkyl chain; (c) decrease the size of the core. Reproduced from ref. 181 with permission from Royal Society of Chemistry, copyright 2007. | ||
Shen and coworkers reported the self-assembly of gold nanoparticles (AuNPs) functionalized with newly synthesized hexaalkoxy-substituted triphenylene-based discotic liquid crystal ligands.182 A stripelike (2D linear) nanoparticle arrangement is attained in a 0.5 µm area due to strong π–π stacking. The degree of π–π interaction between adjacent triphenylene ligands significantly influences the resulting nanoparticle arrangement, allowing for tunable nanostructures. Hence, these functionalized ligands can guide the assembly of nanoparticles into well-defined architectures, making them highly promising for use in electronic and optical nanodevices.
Another triphenylene derivative, amphiphilic TPC10TEG 195 have shown remarkable self-assembling and self-healing properties, particularly beneficial for developing nanostructured functional materials (Fig. 48).183 Mesoporous silica/alumina hybrid nanocomposites were effectively created in the study by employing TPC10TEG as a template in sol–gel synthesis. After the nanocomposites were drop-casted onto anodic aluminum oxide membrane (AAO), the hexagonal configurations were verified by X-ray diffraction examination and electron microscopy confirmed successful infiltration into the AAO channels, highlighting their potential for use in nanoelectronic and optoelectronic applications. For example, a study created a high-performance self-powered photodetector using nickel hexahydroxytriphenylene (Ni-CAT) grown in a composite structure.184
 | ||
| Fig. 48 Chemical structure of the TPC10TEG. Reproduced from ref. 184 with permission from American Chemical Society, copyright 2023. | ||
Researchers have explored the photoconductivity of both undoped and nanostructured hexaalkoxytriphenylene series (HAT4, HAT5, and HAT6) doped with gold chloride.185 For HAT4 and HAT5, but not for HAT6, the photoconductivity improved by more than 10 times following doping with gold chloride. It is possible to explain the high photoconductivity in terms of core–core lengths, transition temperatures, and their side chains' length. The derivatives of hexa-alkoxytriphenylene doped with gold chloride exhibit photoconductivity in the range of 10−4–10−5 S m−1.which are five times more than the undoped value mixtures. This nanocomposite's strong NIR absorbance and photoconductivity make it a possible contender for use in organic solar cell technology.185
The increasing interest in chiral supramolecular nanostructures in p-conjugated supramolecular assemblies is a result of their possible uses in organic semiconductors. Using chiral limonene solution as the chiral source, a series of optically active triphenylene-based supramolecular gels were created.186 These gels eventually form a heterochiral packing structure or a homochiral P- or M-type stacking. A detailed investigation was conducted into the effects of supramolecular packing on homochiral or heterochiral assemblies on their mechanical and electrical properties. This could potentially establish a connection between a chiral supramolecular nanostructure and the mechanical strength and electrical performance of the final supramolecular physical gels. This work is important for the rational design of p-conjugated supramolecular nanostructure assemblies and for their possible use in optoelectronics and chiral organic electronics.186
Carbon nanotubes (CNTs), which are hollow cylinders composed completely of carbon, have been studied for their electrical, mechanical, and thermal properties.187 To address the challenge of poor dispersibility in discotic liquid crystals, single-walled CNTs (SWNTs) were functionalized with triphenylene-based discotic mesogens. Hydroxylated triphenylene derivatives were synthesized and chemically grafted onto acid-purified SWNTs. The resulting nanocomposites, studied via differential scanning calorimetry and X-ray diffraction, revealed that the SWNTs were embedded within the columnar phase of the liquid crystal matrix. This highlights the potential of integrating π-conjugated mesogens with carbon nanostructures for advanced nanocomposite materials with tailored optical and electronic properties.
Lintang and coworkers have explored the mixture of thin film nanocomposites using functional amphiphilic organic moieties in mesoporous silicon channels.183 Amphiphilic triphenylene derivatives have been utilized as self-assembling templates for the fabrication of mesostructured silica films. These derivatives enable the organization of silica precursors into well-defined nanostructures through their ability to form ordered assemblies in solution, providing a versatile approach for the development of functional silica-based materials with controlled porosity and morphology. However, these films depend on donor–acceptor systems and ethanol vapor-mediated maturing.
4. Structure–property relationship of triphenylene and its derivatives
Triphenylene, an incredibly symmetrically organized polycyclic aromatic hydrocarbon (PAH), has very strong and flat π-conjugated structure made up of four benzene rings fused together. The whole π-electron system being delocalized gives triphenylene very good optical, electronic, and thermal stability, which is the main reason for its widespread use in different areas like materials science and molecular electronics (Table 1). The flat molecular shape of triphenylene allows for very good π–π stacking interactions, which in turn helps with the movement of charge carriers and the transport of excitons – a necessity for high-performance optoelectronic devices. The triphenylene core can be significantly changed in terms of its physicochemical properties by substituents attached to its edges. When electron donating substituents like alkoxy, amino or alkyl groups are used, they create a higher electron density which, in turn, results in a smaller band gap and greater fluorescence quantum yield. On the other hand, if an electron withdrawing group is present (like cyano, carbonyl or nitro) the LUMO energy level will be lowered which will result in better electron transport and stabilization of the molecular orbitals for solar energy applications. The addition of flexible alkyl or alkoxy chains at the edge leads to the formation of columnar mesophase, a property of triphenylene-based discotic liquid crystals. These side chains help to dissolve and process the molecules while allowing molecular ordering that is beneficial for isotropic charge conduction over long distances. Meanwhile, the presence of a heteroatom or coordination with metal centers is another way to change the redox activity and catalytic potential through the alteration of the electronic distribution.| Structural modification | Influenced property | Representative application |
|---|---|---|
| Long alkyl or alkoxy substituents | Promote columnar mesophases; enhance solubility | Discotic liquid crystals, organic semiconductors |
| Electron-donating groups (–OR, –NR2) | Raise HOMO level; increase fluorescence efficiency | OLEDs, luminescent sensors |
| Electron-withdrawing groups (–CN, –NO2, –COOR) | Lower LUMO level; improve electron transport | Organic photovoltaics, n-type materials |
| Heteroatom incorporation (N, S, O) | Modify electronic delocalization; introduce redox activity | Catalysts, coordination networks, COFs |
| Metal–triphenylene complexes | Enable charge–transfer transitions; enhance photostability | Photocatalysis, electrocatalysis |
| π-Extended frameworks (dimers, trimers, oligomers) | Increase conjugation length; broaden absorption | Photovoltaic and optoelectronic materials |
| Functional groups for sensing (–COOH, –SH, –NH2) | Provide analyte recognition and binding sites | Chemical and biological sensors |
| Planar rigid cores with edge substitution | Improve molecular packing and mechanical integrity | COFs and nanostructured materials |
| Amphiphilic or hydrophilic side chains | Induce self-assembly in aqueous or hybrid media | Drug delivery, biofunctional materials |
Thus, the structure–property relationship in triphenylene derivatives is governed by three main factors: (i) the degree of π-conjugation, (ii) the nature and position of substituents, and (iii) molecular packing in the solid state. Fine-tuning these parameters allows the rational design of triphenylene-based materials with tailored optical, electronic, and self-assembly characteristics.
5. Future perspectives
The future of triphenylene-based chemistry is poised for significant expansion as advances in molecular design, synthetic methodology, and materials engineering continue to converge. The intrinsic rigidity, planarity, and π-conjugation of the triphenylene framework render it an exceptional platform for developing multifunctional materials. However, despite notable progress, substantial opportunities remain for enhancing its performance, scalability, and integration into emerging technologies.A primary direction for future research lies in the development of sustainable and atom-economical synthetic routes. Conventional methods for constructing the triphenylene core often rely on harsh reaction conditions, toxic reagents, or precious metal catalysts. To align with green chemistry principles, the focus should shift toward metal-free cyclization, photochemical, and electrochemical approaches, as well as flow-assisted synthesis for scalable production. These methodologies would not only reduce environmental impact but also improve reproducibility and yield, enabling the large-scale fabrication of substituted triphenylene derivatives with controlled functionalities.
Another promising avenue involves precise structure–property modulation through rational molecular engineering. Introducing heteroatoms, functional groups, or fused aromatic rings can fine-tune electronic structures, optimize HOMO–LUMO energy levels, and control molecular packing. The strategic design of asymmetric or donor–acceptor-type triphenylene systems could enable ambipolar charge transport and broaden absorption spectra, which are crucial for next-generation OLEDs, photovoltaic cells, and OFETs.
In the realm of functional materials, triphenylene-based covalent organic frameworks (COFs) and metal–organic frameworks (MOFs) present rapidly growing research frontiers. Their ordered π-stacked networks, chemical robustness, and large surface areas make them suitable for diverse applications, including gas adsorption, heterogeneous catalysis, ion conduction, and energy storage.
The incorporation of triphenylene derivatives into the fields of nanotechnology and hybrid materials is a very productive area of research. Nanoscale triphenylene-based architectures like nanoribbons, nanosheets, and 2D assemblies can exhibit outstanding properties regarding charge transport and light harvesting. The merger of such systems with graphene, transition-metal dichalcogenides, or conductive polymers could result in the invention of flexible, light-weight, and high-efficiency devices. In addition to material science, the biological and pharmaceutical possibilities of triphenylene derivatives are still mostly unexplored. Their flat aromatic cores and editable peripheries provide ways to create molecules that are bioactive and photoreactive for use in drug delivery, imaging, and photodynamic therapy.
6. Conclusions
Triphenylene derivatives represent an important class of polycyclic aromatic hydrocarbons that continue to gained considerable scientific attention owing to their rigid planar architecture and extensively delocalized π-conjugated system. These structural features impart remarkable thermal stability, high charge-carrier mobility, and the ability to form well-ordered self-assembled monolayers, establishing triphenylene-based systems as key building blocks in advanced organic materials. Their integration into OLEDs, OPVs, and OFETs has demonstrated their immense potential to enhance the efficiency and durability of next-generation electronic devices. The growing diversity of triphenylene derivatives, achieved through tailored functionalization and molecular engineering, has expanded their applicability across multiple disciplines, including nanotechnology, materials science, and pharmaceutical research. Progress in synthetic strategies—particularly the development of catalytic, photochemical, and green synthetic routes—has enabled the preparation of derivatives with fine-tuned electronic and optical properties suited for targeted applications. These advancements have positioned triphenylene-based materials as promising candidates for high-performance, sustainable, and versatile organic electronic systems. Beyond electronic applications, triphenylene and its derivatives have emerged as valuable components in nanostructured materials, catalysis, polymer science, and energy storage technologies. Their extended π-conjugation facilitates efficient charge transport and light absorption, characteristics essential for the advancement of next-generation solar energy conversion materials.Conflicts of interest
Authors have no conflicts of interest to declare.Data availability
No primary research results, software or code have been included and no new data were generated or analyzed as part of this review.Acknowledgements
The authors gratefully acknowledge the University of Gujrat, Pakistan, for providing the necessary academic support and research facilities to complete this review work.References
- C. M. Buess and D. D. Lawson, Chem. Rev., 1960, 60, 313–330 CrossRef CAS.
- S. Banerjee and A. Guha, Z. fur Krist.-Cryst. Mater., 1937, 96, 107–110 CrossRef CAS.
- G. R. Desiraju and A. Gavezzotti, Acta Crystallogr., Sect. B: Struct. Sci., 1989, 45, 473–482 CrossRef.
- K. P. Vijayalakshmi and C. H. Suresh, New J. Chem., 2010, 34, 2132–2138 RSC.
- E. Clar and R. Schoental, Polycyclic Hydrocarbons, Springer, 1964 Search PubMed.
- M. Solà, Front. Chem., 2013, 1, 66422 Search PubMed.
- C. Tschierske, J. Mater. Chem., 2001, 11, 2647–2671 RSC.
- S. Sergeyev, W. Pisula and Y. H. Geerts, Chem. Soc. Rev., 2007, 36, 1902–1929 RSC.
- F. Ahmed and J. Trotter, Acta Crystallogr., 1963, 16, 503–508 CrossRef CAS.
- R. Wehlitz, T. Hartman and D. L. Huber, J. Chem. Phys., 2021, 154(4), 44304–44315 CrossRef CAS PubMed.
- J. W. Levell, A. Ruseckas, J. B. Henry, Y. Wang, A. D. Stretton, A. R. Mount, T. H. Galow and I. D. Samuel, J. Phys. Chem. A, 2010, 114, 13291–13295 CrossRef CAS PubMed.
- A. Bayer, S. Zimmermann and J. Wendorff, Mol. Cryst. Liq. Cryst., 2003, 396, 1–22 CrossRef CAS.
- D. Markovitsi, I. Lécuyer, P. Lianos and J. Malthête, J. Chem. Soc., Faraday Trans., 1991, 87, 1785–1790 RSC.
- S. Kumar, Liq. Cryst., 2004, 31, 1037–1059 CrossRef CAS.
- M. Gupta and S. K. Pal, Langmuir, 2016, 32, 1120–1126 CrossRef CAS PubMed.
- A. Sugita, K. Suzuki and S. Tasaka, Phys. Rev. B, 2004, 69, 212201 CrossRef.
- J. De La Rie, M. Enache, Q. Wang, W. Lu, M. Kivala and M. Stöhr, J. Phys. Chem. C, 2022, 126, 9855–9861 Search PubMed.
- M. Saleh, M. Baumgarten, A. Mavrinskiy, T. Schäfer and K. Müllen, Macromolecules, 2010, 43, 137–143 CrossRef CAS.
- H. Kim, M. Park, S. Yang, D.-G. Kang, K.-U. Jeong and J.-H. Lee, Liq. Cryst., 2015, 42, 1779–1784 CrossRef CAS.
- S. Gonell, M. Poyatos and E. Peris, Angew. Chem., 2013, 125, 7147–7151 CrossRef.
- V. Bhalla, H. Arora and M. Kumar, Dalton Trans., 2013, 42, 4450–4455 Search PubMed.
- W. Chen, H. Zhang, H. Zheng, H. Li, F. Guo, G. Ni, M. Ma, C. Shi, R. Ghadari and L. Hu, Chem. Commun., 2020, 56, 1879–1882 Search PubMed.
- Z. Yang, J. Luo, W. Zhang, J. Lei, C. Liu and Y. Li, Org. Biomol. Chem., 2025, 23, 323–327 Search PubMed.
- K. Dou, W. Zhao, C. Wang, Y. Fan, C. He, L. Zhang and S. Pang, Chem. Eng. J., 2024, 501, 157677 Search PubMed.
- X. Kong, H. Gong, P. Liu, W. Yao, Z. Liu, G. Wang, S. Zhang and Z. He, New J. Chem., 2018, 42, 3211–3221 Search PubMed.
- C. J. Lu, Q. Xu, J. Feng and R. R. Liu, Angew. Chem., Int. Ed., 2023, 62, e202216863 CrossRef CAS PubMed.
- D. Sonet and B. Bibal, Tetrahedron Lett., 2019, 60, 872–884 CrossRef CAS.
- Z. Qi, Z. An, B. Huang, M. Wu, Q. Wu and D. Jiang, Org. Biomol. Chem., 2023, 21, 6419–6423 RSC.
- A. Mähringer, A. C. Jakowetz, J. M. Rotter, B. J. Bohn, J. K. Stolarczyk, J. Feldmann, T. Bein and D. D. Medina, ACS Nano, 2019, 13, 6711–6719 Search PubMed.
- Y. Xue, Y. Li, F. Zhong, Z. Bai, P. Wang, K. Li, F. Lei, Z. He, Y. Yang and W. Feng, Polym. Degrad. Stab., 2025, 111542 CrossRef CAS.
- A. Maczynski, M. Goral and B. Wisniewska-Goclowska, IUPAC-NIST Solubility Data Series. 81. Hydrocarbons with Water and Seawater—Revised and Updated. Part 11. C13–C36 Hydrocarbons with Water, 2006, vol. 35, p. 753 Search PubMed.
- Q. Ye, L. Li, J. Zhang, M. Teng, F. Wu and D. Han, Appl. Organomet. Chem., 2025, 39, e70374 Search PubMed.
- E. Wu, Y. Ma, Q. Tian, Z. Wang, Z. Song, S. Huo, F. Meng, Y. Xie and C. Pan, ACS Nano, 2025, 19, 35701–35711 CrossRef CAS PubMed.
- Z. Chen, S. Zhang, Y. Zhao, Z. Wu, M. Chen, Y. Zeng, Z. Gao, Y. Wei, Z. Li and Z. Liang, Laser Photonics Rev., 2025, 19, 2401926 CrossRef CAS.
- J. A. Rego, S. Kumar and H. Ringsdorf, Chem. Mater., 1996, 8, 1402–1409 CrossRef CAS.
- S. K. Pal, S. Setia, B. Avinash and S. Kumar, Liq. Cryst., 2013, 40, 1769–1816 CrossRef CAS.
- N. Boden, R. J. Bushby, A. N. Cammidge and P. S. Martin, J. Mater. Chem., 1995, 5, 1857–1860 RSC.
- G.-Y. Zhang, L.-L. Ma, E. Lin, Z.-Y. Wang, J.-T. Pan, J. Yang, M. Deng, Y. Wei, Y. Ye and N. Wang, Nat. Commun., 2025, 16, 9419 CrossRef CAS PubMed.
- S. Kumar and S. K. Varshney, Mol. Cryst. Liq. Cryst., 2002, 378, 59–64 Search PubMed.
- L. Zhang, C. Li and S. Pang, Coord. Chem. Rev., 2026, 546, 217081 CrossRef CAS.
- D. Pérez and E. Guitián, Chem. Soc. Rev., 2004, 33, 274–283 RSC.
- H. C. Cheng, J. L. Ma, P. H. Guo and J. C. Shi, Chem.–Asian J., 2025, 20, e00809 CrossRef CAS PubMed.
- C. Mannich, Ber. Dtsch. Chem. Ges., 1907, 40, 153–158 CrossRef.
- R. Freudenmann, B. Behnisch and M. Hanack, J. Mater. Chem., 2001, 11, 1618–1624 RSC.
- R. J. Bushby and C. Hardy, J. Chem. Soc., Perkin Trans. 1, 1986, 721–723 RSC.
- J. W. Goodby, M. Hird, K. J. Toyne and T. Watson, J. Chem. Soc. Chem. Commun., 1994, 1701–1702 RSC.
- M. Müller, J. Petersen, R. Strohmaier, C. Günther, N. Karl and K. Müllen, Angew Chem. Int. Ed. Engl., 1996, 35, 886–888 Search PubMed.
- N. Boden, R. J. Bushby and A. N. Cammidge, J. Am. Chem. Soc., 1995, 117, 924–927 CrossRef CAS.
- H. Naarmann, M. Hanack and R. Mattmer, Synthesis, 1994, 477–478 CrossRef CAS.
- S. R. Waldvogel, A. R. Wartini, P. H. Rasmussen and J. Rebek Jr, Tetrahedron Lett., 1999, 40, 3515–3518 Search PubMed.
- L. Zhai, R. Shukla, S. H. Wadumethrige and R. Rathore, J. Org. Chem., 2010, 75, 4748–4760 CrossRef CAS PubMed.
- S. M. Alhunayhin, R. J. Bushby, A. N. Cammidge and S. S. Samman, Liq. Cryst., 2024, 51, 1333–1344 Search PubMed.
- V. Bhalla, H. Singh and M. Kumar, Org. Lett., 2010, 12, 628–631 Search PubMed.
- V. Gupta, S. K. Pandey and R. P. Singh, Org. Biomol. Chem., 2018, 16, 7134–7138 RSC.
- Z. Li, M. Powers, K. Ivey, S. Adas, B. Ellman, S. D. Bunge and R. J. Twieg, Mater. Adv., 2022, 3, 534–546 Search PubMed.
- Y. Zhang, S. H. Pun and Q. Miao, Chem. Rev., 2022, 122, 14554–14593 CrossRef CAS PubMed.
- M. Grzybowski, K. Skonieczny, H. Butenschön and D. T. Gryko, Angew. Chem., Int. Ed., 2013, 52, 9900–9930 Search PubMed.
- M. J. Rahman, J. Yamakawa, A. Matsumoto, H. Enozawa, T. Nishinaga, K. Kamada and M. Iyoda, J. Org. Chem., 2008, 73, 5542–5548 Search PubMed.
- Z. Sun, Z. Zeng and J. Wu, Chem.–Asian J., 2013, 8, 2894–2904 Search PubMed.
- M. A. Alsharif, Q. A. Raja, N. A. Majeed, R. S. Jassas, A. A. Alsimaree, A. Sadiq, N. Naeem, E. U. Mughal, R. I. Alsantali and Z. Moussa, RSC Adv., 2021, 11, 29826–29858 Search PubMed.
- R. S. Jassas, E. U. Mughal, A. Sadiq, R. I. Alsantali, M. M. Al-Rooqi, N. Naeem, Z. Moussa and S. A. Ahmed, RSC Adv., 2021, 11, 32158–32202 Search PubMed.
- M. K. Smith, N. E. Powers-Riggs and B. H. Northrop, RSC Adv., 2014, 4, 38281–38292 RSC.
- F. B. Mallory, C. S. Wood and J. T. Gordon, J. Am. Chem. Soc., 1964, 86, 3094–3102 CrossRef CAS.
- T. Sato, S. Shimada and K. Hata, Bull. Chem. Soc. Jpn., 1971, 44, 2484–2490 CrossRef CAS.
- M. Daigle, A. Picard-Lafond, E. Soligo and J. F. Morin, Angew. Chem., 2016, 128, 2082–2087 Search PubMed.
- M. M. Zhou, J. He, H. M. Pan, Q. Zeng, H. Lin, K. Q. Zhao, P. Hu, B. Q. Wang and B. Donnio, Chem.–Eur. J., 2023, 29, e202301829 CrossRef CAS PubMed.
- S. Plaize and J.-F. Morin, RSC Adv., 2024, 14, 35227–35231 Search PubMed.
- K.-Q. Zhao, J.-Q. Du, X.-H. Long, M. Jing, B.-Q. Wang, P. Hu, H. Monobe, B. Henrich and B. Donnio, Dyes Pigm., 2017, 143, 252–260 Search PubMed.
- S. Pan, H. Jiang, Y. Zhang, D. Chen and Y. Zhang, Org. Lett., 2016, 18, 5192–5195 CrossRef CAS PubMed.
- Z. Liu, X. Zhang and R. C. Larock, J. Am. Chem. Soc., 2005, 127, 15716–15717 Search PubMed.
- M. Iwasaki, Y. Araki, S. Iino and Y. Nishihara, J. Org. Chem., 2015, 80, 9247–9263 Search PubMed.
- J. A. García-López and M. F. Greaney, Synthesis of biaryls using aryne intermediates, Chem. Soc. Rev., 2016, 45(24), 6766–6798 RSC.
- B. P. Mathew, H. J. Yang, J. Kim, J. B. Lee, Y. T. Kim, S. Lee, C. Y. Lee, W. Choe, K. Myung and J. U. Park, Angew. Chem., Int. Ed., 2017, 56, 5007–5011 Search PubMed.
- D. Peña, S. Escudero, D. Pérez, E. Guitián and L. Castedo, Angew. Chem., Int. Ed., 1998, 37, 2659–2661 Search PubMed.
- J. B. Lee, M. H. Jeon, J. K. Seo, G. von Helden, J.-U. Rohde, B. S. Zhao, J. Seo and S. Y. Hong, Org. Lett., 2019, 21, 7004–7008 CrossRef CAS PubMed.
- Y. Yang, B. Zhou, X. Zhu, G. Deng, Y. Liang and Y. Yang, Org. Lett., 2018, 20, 5402–5405 Search PubMed.
- J. Tu, G. Li, X. Zhao and F. Xu, Tetrahedron Lett., 2019, 60, 44–47 CrossRef CAS.
- Y. Wu, W. Zhang, Q. Peng, C.-K. Ran, B.-Q. Wang, P. Hu, K.-Q. Zhao, C. Feng and S.-K. Xiang, Org. Lett., 2018, 20, 2278–2281 CrossRef CAS PubMed.
- C.-M. Qin, C.-Y. Zeng, W.-H. Yu, P. Hu, B.-Q. Wang, K.-Q. Zhao and B. Donnio, Cryst. Growth Des., 2025, 25, 9524–9535 CrossRef CAS.
- H.-M. Pan, J. He, W.-H. Yu, P. Hu, B.-Q. Wang, K.-Q. Zhao and B. Donnio, J. Mater. Chem. C, 2023, 11, 14695–14704 RSC.
- K. L. Zhang, W. H. Yu, K. Q. Zhao, P. Hu, B. Q. Wang and B. Donnio, Chem.–Asian J., 2024, 19, e202301080 CrossRef CAS PubMed.
- R. E. Yardley, J. A. Paquette, H. Taing, H. M. Gaebler, S. H. Eichhorn, I. P. Hamilton and K. E. Maly, Org. Lett., 2019, 21, 10102–10105 CrossRef CAS PubMed.
- J. Moursounidis and D. Wege, Aust. J. Chem., 1988, 41, 235–249 CrossRef CAS.
- E. Fischer, J. Larsen, J. B. Christensen, M. Fourmigué, H. G. Madsen and N. Harrit, J. Org. Chem., 1996, 61, 6997–7005 CrossRef CAS PubMed.
- K. B. Sukumaran and R. G. Harvey, J. Org. Chem., 1981, 46, 2740–2745 Search PubMed.
- C. Romero, D. Pena, D. Perez and E. Guitian, J. Org. Chem., 2008, 73, 7996–8000 Search PubMed.
- K. Fuchibe, M. Abe, H. Idate and J. Ichikawa, Chem.–Asian J., 2020, 15, 1384–1392 Search PubMed.
- D. Rodriguez-Lojo, D. Perez, D. Pena and E. Guitian, Chem. Commun., 2013, 49, 6274–6276 Search PubMed.
- M. G. Schwab, T. Qin, W. Pisula, A. Mavrinskiy, X. Feng, M. Baumgarten, H. Kim, F. Laquai, S. Schuh and R. Trattnig, Chem.–Asian J., 2011, 6, 3001–3010 Search PubMed.
- J. M. Alonso, A. E. Díaz-Álvarez, A. Criado, D. Pérez, D. Peña and E. Guitián, Angew. Chem., 2011, 1, 177–181 Search PubMed.
- R. C. Borner, N. Boden, R. J. Bushby, R. Borner, A. N. Cammidge, R. Bushby, A. Cammidge and M. Jesudason, Liq. Cryst., 2006, 33, 1439–1448 CrossRef CAS.
- Z.-h. Zhou and T. Yamamoto, J. Organomet. Chem., 1991, 414, 119–127 CrossRef CAS.
- H. Shirai, N. Amano, Y. Hashimoto, E. Fukui, Y. Ishii and M. Ogawa, J. Org. Chem., 1991, 56, 2253–2256 CrossRef CAS.
- Y. Wang, A. D. Stretton, M. C. McConnell, P. A. Wood, S. Parsons, J. B. Henry, A. R. Mount and T. H. Galow, J. Am. Chem. Soc., 2007, 129, 13193–13200 CrossRef CAS PubMed.
- Z. W. Schroeder, J. LeDrew, V. M. Selmani and K. E. Maly, RSC Adv., 2021, 11, 39564–39569 RSC.
- E. C. Rüdiger, F. Rominger, L. Steuer and U. H. Bunz, J. Org. Chem., 2016, 81, 193–196 CrossRef PubMed.
- E. J. Foster, R. B. Jones, C. Lavigueur and V. E. Williams, J. Am. Chem. Soc., 2006, 128, 8569–8574 CrossRef CAS PubMed.
- S. R. Waldvogel and D. Mirk, Tetrahedron Lett., 2000, 41, 4769–4772 CrossRef CAS.
- C. Regenbrecht and S. R. Waldvogel, Beilstein J. Org. Chem., 2012, 8, 1721–1724 CrossRef CAS PubMed.
- J. Yin, H. Qu, K. Zhang, J. Luo, X. Zhang, C. Chi and J. Wu, Org. Lett., 2009, 11, 3028–3031 CrossRef CAS PubMed.
- T. Osawa, T. Kajitani, D. Hashizume, H. Ohsumi, S. Sasaki, M. Takata, Y. Koizumi, A. Saeki, S. Seki and T. Fukushima, Angew. Chem., Int. Ed., 2012, 51, 7990–7993 Search PubMed.
- J. He, Y. Chen, P. Hu, B.-Q. Wang, K.-Q. Zhao and B. Donnio, J. Mol. Liq., 2024, 414, 126218 CrossRef CAS.
- Y. Chen, J. He, H. Lin, H.-F. Wang, P. Hu, B.-Q. Wang, K.-Q. Zhao and B. Donnio, Beilstein J. Org. Chem., 2024, 20, 3263–3273 CrossRef CAS PubMed.
- T. Wöhrle, I. Wurzbach, J. Kirres, A. Kostidou, N. Kapernaum, J. Litterscheidt, J. C. Haenle, P. Staffeld, A. Baro and F. Giesselmann, Chem. Rev., 2016, 116, 1139–1241 Search PubMed.
- M. A. Alam, J. Motoyanagi, Y. Yamamoto, T. Fukushima, J. Kim, K. Kato, M. Takata, A. Saeki, S. Seki and S. Tagawa, J. Am. Chem. Soc., 2009, 131, 17722–17723 CrossRef CAS PubMed.
- Z. Ke-Qing, W. Bi-Qin, H. Ping, L. Quan and Z. Liang-Fu, Chin. J. Chem., 2005, 23, 767–774 CrossRef.
- H.-M. Chen, K.-Q. Zhao, L. Wang, P. Hu and B.-Q. Wang, Soft Mater., 2011, 9, 359–381 Search PubMed.
- P. Guragain, M. Powers, B. Ellman and R. J. Twieg, Mater. Adv., 2024, 5, 9032–9040 RSC.
- T.-R. Zhang, Y.-P. Fan, W.-H. Yu, Q.-G. Li, Y. Shi, S.-K. Xiang, K.-Q. Zhao, B.-Q. Wang and C. Feng, J. Mol. Liq., 2024, 408, 125399 Search PubMed.
- S. K. Varshney, Liq. Cryst., 2013, 40, 1477–1486 CrossRef CAS.
- I. Paraschiv, M. Giesbers, B. van Lagen, F. C. Grozema, R. D. Abellon, L. D. Siebbeles, A. T. Marcelis, H. Zuilhof and E. J. Sudhölter, Chem. Mater., 2006, 18, 968–974 CrossRef CAS.
- A. El Mansoury, R. J. Bushby and N. Karodia, Liq. Cryst., 2012, 39, 1222–1230 Search PubMed.
- L. Tao, Y. Xie, K.-X. Zhao, P. Hu, B.-Q. Wang, K.-Q. Zhao and X.-Y. Bai, New J. Chem., 2024, 48, 12006–12014 Search PubMed.
- F. Yang, J. Xie, H. Guo, B. Xu and C. Li, Liq. Cryst., 2012, 39, 1368–1374 CrossRef CAS.
- K. Zhang, Y. Bai, C. Feng, G. Ning, H. Ni, W. Yu, K. Zhao, B. Wang and P. Hu, New J. Chem., 2018, 42, 272–280 RSC.
- K.-Q. Zhao, X.-Y. Bai, B. Xiao, Y. Gao, P. Hu, B.-Q. Wang, Q.-D. Zeng, C. Wang, B. Heinrich and B. Donnio, J. Mater. Chem. C, 2015, 3, 11735–11746 RSC.
- J. Bi, H. Wu, Z. Zhang, A. Zhang, H. Yang, Y. Feng, Y. Fang, L. Zhang, Z. Wang and W. Qu, J. Mater. Chem. C, 2019, 7, 12463–12469 Search PubMed.
- H. Lin, K.-X. Zhao, M. Jing, X.-H. Long, K.-Q. Zhao, P. Hu, B.-Q. Wang, P. Lei, Q.-D. Zeng and B. Donnio, J. Mater. Chem. C, 2022, 10, 14453–14470 RSC.
- H. Gopee, X. Kong, Z. He, I. Chambrier, D. L. Hughes, G. J. Tizzard, S. J. Coles and A. N. Cammidge, J. Org. Chem., 2013, 78, 9505–9511 CrossRef CAS PubMed.
- S. Chen, F. S. Raad, M. Ahmida, B. R. Kaafarani and S. H. Eichhorn, Org. Lett., 2013, 15, 558–561 Search PubMed.
- P. Staffeld, M. Kaller, P. Ehni, M. Ebert, S. Laschat and F. Giesselmann, Crystals, 2019, 9, 74 Search PubMed.
- J. Kirres, F. Knecht, P. Seubert, A. Baro and S. Laschat, ChemPhysChem, 2016, 17, 1159–1165 CrossRef CAS PubMed.
- J. F. Hang, H. Lin, K. Q. Zhao, P. Hu, B. Q. Wang, H. Monobe, C. Zhu and B. Donnio, Eur. J. Org Chem., 2021, 2021, 1989–2002 CrossRef CAS.
- W.-J. Deng, S. Liu, H. Lin, K.-X. Zhao, X.-Y. Bai, K.-Q. Zhao, P. Hu, B.-Q. Wang, H. Monobe and B. Donnio, New J. Chem., 2022, 46, 7936–7949 RSC.
- Q. Zeng, S. Liu, H. Lin, K.-X. Zhao, X.-Y. Bai, K.-Q. Zhao, P. Hu, B.-Q. Wang and B. Donnio, Molecules, 2023, 28, 1721 CrossRef CAS PubMed.
- A. O. Alsahli and A. N. Cammidge, Eur. J. Org Chem., 2022, 2022, e202200990 CrossRef CAS.
- Z. Chongyang, H. Ping, W. Biqin, F. Wenyan, Z. Keqing, D. Bertrand and B. Donnio, Acta Chim. Sin., 2023, 81, 469 Search PubMed.
- H. Lin, K.-X. Zhao, B.-Q. Wang, P. Hu, K.-Q. Zhao and B. Donnio, Giant, 2025, 100362 CrossRef CAS.
- X. Wu, H. Lin, X.-Y. Bai, P. Hu, B.-Q. Wang, K.-Q. Zhao and B. Donnio, J. Mater. Chem. C, 2025, 13, 7678–7685 RSC.
- T. Lorenzetto, G. Berton, F. Castellini, G. Casson, A. Scarso and F. Fabris, Chem.–Eur. J., 2024, 30, e202402348 CrossRef CAS PubMed.
- Y. Li, Y.-X. Wang, X.-K. Ren and L. Chen, Mater. Chem. Front., 2017, 1, 2599–2605 Search PubMed.
- V. Percec, M. R. Imam, M. Peterca, D. A. Wilson, R. Graf, H. W. Spiess, V. S. Balagurusamy and P. A. Heiney, J. Am. Chem. Soc., 2009, 131, 7662–7677 CrossRef CAS PubMed.
- V. Conejo-Rodríguez, B. Donnio, B. Heinrich, R. Termine, A. Golemme and P. Espinet, J. Mater. Chem. C, 2023, 11, 1435–1447 Search PubMed.
- E. de Domingo, C. L. Folcia, J. Ortega, J. Etxebarria, R. Termine, A. Golemme, S. Coco and P. Espinet, Inorg. Chem., 2020, 59, 10482–10491 CrossRef CAS PubMed.
- S. Cobos, G. García, C. L. Folcia, J. Ortega, J. Etxebarria, G. López-Peña, D. H. Ortgies, E. M. Rodriguez and S. Coco, J. Mater. Chem. C, 2024, 12, 12332–12340 RSC.
- S. Chen, H. Taing, M. Ahmida, H. Y. He, A. Carr, H. M. Muchall and S. H. Eichhorn, Soft Matter, 2024, 20, 7854–7864 RSC.
- S. Ibáñez, M. Poyatos and E. Peris, Chem. Commun., 2017, 53, 3733–3736 RSC.
- Y. Fan, J. Wang, Z. Zhang, X. Ma and M. Zhao, ACS Appl. Nano Mater., 2023, 6, 13037–13047 Search PubMed.
- S. Qi, Y. Fan, W. Li and M. Zhao, Phys. Chem. Chem. Phys., 2020, 22, 20061–20068 Search PubMed.
- K. Yu, J. Zhang, Y. Hu, L. Wang, X. Zhang and B. Zhao, Catalysts, 2024, 14, 184 CrossRef CAS.
- R. Matsuoka, R. Toyoda, R. Shiotsuki, N. Fukui, K. Wada, H. Maeda, R. Sakamoto, S. Sasaki, H. Masunaga and K. Nagashio, ACS Appl. Mater. Interfaces, 2018, 11, 2730–2733 Search PubMed.
- E. Peris, Chem. Commun., 2016, 52, 5777–5787 RSC.
- S. Gonell, R. G. Alabau, M. Poyatos and E. Peris, Chem. Commun., 2013, 49, 7126–7128 RSC.
- T. Mandal, T. K. Dutta, S. Mohanty and J. Choudhury, Chem. Commun., 2021, 57, 10182–10185 Search PubMed.
- S. Gonell, M. Poyatos and E. Peris, Chem.–Eur. J., 2014, 20, 5746–5751 CrossRef CAS PubMed.
- K. E. Shelar, N. Le, K. M. Mukeba, S. Dey, B. Farajidizaji, S. Athukorale, C. U. Pittman, C. E. Webster, B. Donnadieu and E. Caldona, Mater. Chem. Front., 2022, 6, 1391–1404 RSC.
- M. Saleh, Y. S. Park, M. Baumgarten, J. J. Kim and K. Müllen, Macromol. Rapid Commun., 2009, 30, 1279–1283 CrossRef CAS PubMed.
- Y. Wang, M. Zhu, J. Deng, G. Xie and E. Baranoff, New J. Chem., 2017, 41, 1773–1780 RSC.
- I. Bala, W.-Y. Yang, S. P. Gupta, J. De, R. A. K. Yadav, D. P. Singh, D. K. Dubey, J.-H. Jou, R. Douali and S. K. Pal, J. Mater. Chem. C, 2019, 7, 5724–5738 RSC.
- J.-Y. Park, J. M. Kim, H. Lee, K.-Y. Ko, K. S. Yook, J. Y. Lee and Y. G. Baek, Thin Solid Films, 2011, 519, 5917–5923 CrossRef CAS.
- S. Kumar, D. Kumar, Y. Patil and S. Patil, J. Mater. Chem. C, 2016, 4, 193–200 RSC.
- S. N. Park, H. W. Lee, Y. S. Kim, J. Kim, S. E. Lee, H. W. Lee, Y. K. Kim and S. S. Yoon, Synth. Met., 2015, 206, 124–130 CrossRef CAS.
- V. Bhalla, H. Arora, H. Singh and M. Kumar, Dalton Trans., 2013, 42, 969–974 RSC.
- N. Contreras-Pereda, S. Pané, J. Puigmartí-Luis and D. Ruiz-Molina, Coord. Chem. Rev., 2022, 460, 214459 CrossRef CAS.
- V. Bhalla, H. Singh, H. Arora and M. Kumar, Sens. Actuators, B, 2012, 171, 1007–1012 CrossRef.
- V. Bhalla, H. Singh and M. Kumar, Dalton Trans., 2012, 41, 11413–11418 RSC.
- S. Kandel, V. Sathish, L. Mathivathanan, A. N. Morozov, A. M. Mebel and R. G. Raptis, New J. Chem., 2019, 43, 7251–7258 Search PubMed.
- H. Arora, V. Bhalla and M. Kumar, RSC Adv., 2015, 5, 32637–32642 Search PubMed.
- S.-Y. Ding and W. Wang, Chem. Soc. Rev., 2013, 42, 548–568 RSC.
- D. Sonet and B. Bibal, Triphenylene: A versatile molecular receptor, Tetrahedron Lett., 2019, 60(12), 872–884 CrossRef CAS.
- A. P. Cote, A. I. Benin, N. W. Ockwig, M. O'Keeffe, A. J. Matzger and O. M. Yaghi, science, 2005, 310, 1166–1170 CrossRef CAS PubMed.
- M. Dogru, M. Handloser, F. Auras, T. Kunz, D. Medina, A. Hartschuh, P. Knochel and T. Bein, Angew. Chem. Int. Ed, 2013, 52, 2920–2924 CrossRef CAS.
- F. Haase and B. V. Lotsch, Chem. Soc. Rev., 2020, 49, 8469–8500 Search PubMed.
- S. Wan, J. Guo, J. Kim, H. Ihee and D. Jiang, Angew. Chem. Int. Ed, 2008, 47, 8826–8830 CrossRef CAS PubMed.
- A. K. Mandal, J. Mahmood and J. B. Baek, ChemNanoMat, 2017, 3, 373–391 CrossRef CAS.
- C. Du, W. Na, M. Shao, S. Shang, Y. Liu and J. Chen, Chem. Mater., 2023, 35, 1395–1403 CrossRef CAS.
- K. Wang, Z. Zhang, L. Lin, K. Hao, J. Chen, H. Tian and X. Chen, ACS Appl. Mater. Interfaces, 2019, 11, 39503–39512 CrossRef CAS PubMed.
- K. Wang, Z. Zhang, L. Lin, J. Chen, K. Hao, H. Tian and X. Chen, Chem. Mater., 2019, 31, 3313–3323 CrossRef CAS.
- X. Ding, L. Chen, Y. Honsho, X. Feng, O. Saengsawang, J. Guo, A. Saeki, S. Seki, S. Irle and S. Nagase, J. Am. Chem. Soc., 2011, 133, 14510–14513 CrossRef CAS PubMed.
- M. Calik, F. Auras, L. M. Salonen, K. Bader, I. Grill, M. Handloser, D. D. Medina, M. Dogru, F. Löbermann and D. Trauner, J. Am. Chem. Soc., 2014, 136, 17802–17807 CrossRef CAS PubMed.
- K. Ashwini Kumar, K. L. Reddy, S. Vidhisha and S. Satyanarayana, Appl. Organomet. Chem., 2009, 23, 409–420 CrossRef.
- C. Du, W. Na, M. Shao, S. Shang, Y. Liu and J. Chen, Self-templated synthesis of triphenylene-based uniform hollow spherical two-dimensional covalent organic frameworks for drug delivery, Chem. Mater., 2023, 35(3), 1395–1403 CrossRef CAS.
- H. Srinivasa and S. Hariprasad, ChemistrySelect, 2022, 7, e202200783 Search PubMed.
- A. Ané, G. Prestat, G. Manh, M. Thiam, S. Josse, M. Pipelier, J. Lebreton, J. Pradère and D. Dubreuil, Pure Appl. Chem., 2001, 73, 1189–1196 Search PubMed.
- M. Montembault, G. Vo-Thanh, A. Deyine, V. Fargeas, M. Villiéras, A. Adjou, D. Dubreuil, D. Esquieu, C. Grégoire and S. Opi, Bioorg. Med. Chem. Lett., 2004, 14, 1543–1546 Search PubMed.
- K. P. Naidek, C. R. Zuconelli, O. M. Cruz, R. Ribeiro, S. M. Winnischofer and H. Winnischofer, Biochem. Cell Biol., 2016, 94, 205–211 Search PubMed.
- A. Das, N. L. Banik and S. K. Ray, Cancer, 2010, 116, 164–176 CrossRef CAS PubMed.
- O. Hayashida, T. Matsuo, K. Nakamura and S. Kusano, J. Org. Chem., 2016, 81, 4196–4201 CrossRef CAS PubMed.
- C. Givelet and B. Bibal, Org. Biomol. Chem., 2011, 9, 7457–7460 RSC.
- I. López-Tocón, J. C. Otero, J. F. Arenas, J. V. García-Ramos and S. Sánchez-Cortés, Langmuir, 2010, 26, 6977–6981 CrossRef.
- S. Kumar, S. K. Pal, P. S. Kumar and V. Lakshminarayanan, Soft Matter, 2007, 3, 896–900 RSC.
- Z. Shen, M. Yamada and M. Miyake, J. Am. Chem. Soc., 2007, 129, 14271–14280 CrossRef CAS PubMed.
- H. O. Lintang, M. A. Jalani, L. Yuliati and M. M. Salleh, Fabrication of mesoporous silica/alumina hybrid membrane film nanocomposites using template sol-gel synthesis of amphiphilic triphenylene, IOP Conf. Ser.: Mater. Sci. Eng., 2017, 202(1), 012003 CrossRef.
- Y. Wang, L. Liu, Y. Shi, S. Li, F. Sun, Q. Lu, Y. Shen, S. Feng and S. Qin, ACS Appl. Mater. Interfaces, 2023, 15, 18236–18243 CrossRef CAS PubMed.
- C. Kavitha, B. Avinash, S. Kumar and V. Lakshminarayanan, Mater. Chem. Phys., 2012, 133, 635–641 CrossRef CAS.
- H. Zhang, J. Cheng, Q. Zhou, Q. Zhang and G. Zou, Soft Matter, 2020, 16, 5203–5209 Search PubMed.
- S. Kumar and H. K. Bisoyi, Angew. Chem., Int. Ed. Engl., 2007, 46, 1501 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |