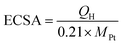Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Hydroxyl group's effects on the activity and durability of supported carbon–TiO2 for proton exchange membrane fuel cells
Su-Jin Janga,
Yi Kyeong Junga,
Jeong Han Leea,
Seok Hee Lee*a,
Tae Ho Shin *a and
Young Wook Lee
*a and
Young Wook Lee *b
*b
aKorea Institute of Ceramic Engineering & Technology, Jin-ju 52851, Republic of Korea. E-mail: lsh@kicet.re.kr; ths@kicet.re.kr
bDepartment of Education Chemistry and Research Institute of Advanced Chemistry Gyeongsang National University, Jinju 52828, Korea. E-mail: lyw2020@gnu.ac.kr
First published on 24th February 2026
Abstract
Pt catalysts used for the cathode in proton exchange membrane fuel cells (PEMFCs) are mostly supported on carbon materials. However, durability issues arise under operating conditions due to carbon corrosion, which is a critical degradation mechanism. To improve the support durability, metal oxide supports combined with carbon have been extensively investigated. In this study, oxygen vacancy and hydroxyl group modified TiO2 particles supporting Pt nanoparticles were developed for applications in the oxygen reduction reaction (ORR) and the membrane electrode assemblies (MEAs). The Pt catalyst supports were prepared using a microwave-assisted method, and their performance was compared according to the degree of TiO2 crystallinity with and without heat treatment. X-ray diffraction (XRD) measurements before and after heat treatment revealed differences in the crystallinity of TiO2, showing the presence of anatase TiO2 and Ti–OH species. In the uncalcined carbon–TiO2 composite, the presence of titanium hydroxide (Ti–OH) species was confirmed by X-ray photoelectron spectroscopy (XPS) and attenuated total reflection (ATR) spectroscopy. The prepared Pt nanoparticle/TiO2–carbon (TiO2–Pt/SC) and Pt nanoparticle/heat-treated TiO2–carbon (TiO2–Pt/SC–H) catalysts exhibited superior ORR activity compared to that of the calcined catalyst. The effect of Pt loading on the ORR performance was also examined, revealing enhanced activity in the presence of anatase TiO2, which is attributed to its strong metal–support interactions (SMSIs). In addition, MEA tests confirmed that these samples exhibited high activity and improved stability. The enhanced oxygen reduction kinetics are ascribed to water dissociation and the formation of surface-adsorbed hydroxyl moieties. We believe that the results described herein provide important implications for the development of durable TiO2–carbon hybrid supports for MEA applications.
Introduction
As environmental pollution caused by the extensive use of fossil fuels has become increasingly severe, research on green and sustainable energy technologies has intensified.1 Many countries and industries have made substantial investments in developing alternative and renewable energy sources capable of bridging the present and future energy demand-supply gap in a sustainable manner. These energy sources include solar, wind, hydropower, biomass, geothermal, and hydrogen energy, which encompasses fuel cell technologies. Among these, proton exchange membrane fuel cells (PEMFCs) have attracted significant attention due to their zero greenhouse gas emissions, high theoretical power density, and strong potential for sustainable energy systems.2,3 A PEMFC consists of a cathode, an anode, and a proton-conducting membrane, and extensive research has focused on catalyst and support materials to improve their electrocatalytic activity and stability. PEMFCs are particularly attractive for automotive applications, where the membrane electrode assembly (MEA), which generates electricity through electrochemical reactions, is the most critical component.4 Degradation of the MEA is a major cause for the performance loss of PEMFCs during long-term operations. Under PEMFC operating conditions, especially during startup and shutdown, the high electrode potential at cathodes can induce carbon oxidation and corrosion. This process leads to Pt dissolution, Pt particle agglomeration, and a significant decrease in the fuel cell performance and lifetime.2,5–7 As the degradation of Pt nanoparticles directly deteriorates the catalytic performance, improving the durability of the cathode catalyst is of critical importance. In particular, enhancing the binding energy between oxygen (O) or hydroxyl (OH) species and the catalyst surface has been suggested as an effective strategy.8 Moreover, improving the rate of the oxygen reduction reaction (ORR)—the rate-determining and slowest reaction in PEMFCs under all operating conditions—is essential.3 Loss of electrochemical surface area (ECSA), primarily caused by corrosion of the catalyst support and subsequent agglomeration of Pt nanoparticles, is widely recognized as a major challenge for catalyst durability.9 Numerous studies have reported that Pt dissolution and carbon support corrosion are responsible for the performance degradation of PEMFCs.8–10 Carbon materials are commonly used as catalyst supports due to their high electrical conductivity and large specific surface area.6,11 However, carbon corrosion and catalyst detachment severely impair fuel cell performance, highlighting the urgent need for corrosion-resistant support materials. To address this issue, hybrid catalyst supports composed of carbon and TiO2 have been proposed by tailoring the surface characteristics of carbon. Carbon corrosion at potentials around 0.9 V and Pt nanoparticle agglomeration lead to a decrease in ECSA and overall catalytic performance during fuel cell operation. Strong metal–support interaction (SMSI) between Pt and metal oxide supports has been shown to enhance the catalytic activity of Pt nanoparticles.12,13 Metal oxide supports can donate electrons to Pt nanoparticles, modifying the unfilled Pt d-band states and facilitating the adsorption, dissociation, and desorption of oxygen species. Among various metal oxides, titanium dioxide (TiO2) has been extensively investigated as a catalyst support for PEMFCs due to its high chemical stability and resistance to oxidation.7,9 The hypo-d-electron nature of TiO2 and related oxides (e.g., ZrO2 and HfO2) enables strong metal–support interactions, resulting in enhanced nanoparticle dispersion and improved catalytic activity.5,14,15 TiO2 exists in several crystalline phases, including anatase, brookite, and rutile. In the absence of heat treatment, TiO2 typically exhibits low crystallinity or an amorphous structure. Depending on the synthesis method and precursor, TiO2 can possess varying oxidation states and surface functional groups. The spillover of surface hydroxyl species (M–OH) from metal oxides and their interactions play a critical role in electrocatalytic processes, particularly in ORR performance. Consumption of adsorbed M–OH species on the metallic catalyst surface reduces their chemical potential, thereby disturbing the established equilibrium and enhancing the catalytic activity.14–16 Furthermore, TiO2 containing oxygen vacancies and Ti3+ species has been reported to improve the ORR performance by lowering the kinetic energy barrier, modifying the OH adsorption energy on Pt surfaces, and altering the TiO2 band structure.17In this study, we investigate the effect of TiO2 crystallinity in TiO2–carbon hybrid catalyst supports on stability and ORR activity, aiming to achieve performance comparable to that of conventional Pt/C catalysts. Low-crystallinity TiO2 and anatase TiO2 were deposited on carbon supports using a microwave-assisted synthesis method. Pt nanoparticles were uniformly dispersed on the prepared supports. The amorphous TiO2–Pt catalyst exhibited superior ORR activity, stability, and durability, as well as excellent MEA performance. The presence of oxygen vacancies and surface hydroxyl groups (OH) on TiO2 was found to play a crucial role in enhancing the electrochemical performance.
Experimental
Materials
SP carbon was obtained from VINATech Co., Ltd and titanium(IV) tetrachloride (TiCl4, 99%, Wako Chemicals, Japan), ethylene glycol (99.8%, Sigma-Aldrich), chloroplatinic acid hexahydrate (H2PtCl6·6H2O, Sigma-Aldrich), isopropyl alcohol (IPA, 99.5%, Sigma-Aldrich), sodium hydroxide (NaOH, 97%, Sigma-Aldrich), and perchloric acid (HClO4, 70%, Sigma-Aldrich) were purchased. All the chemicals were used as received without further purification.Synthesis of the carbon-TiO2 composite
The SP-TiO2 composites were obtained by a microwave-assisted hydrothermal reaction without any surfactants. 0.1 g SP carbon was dispersed in 40 mL deionized water in a Teflon beaker. 2 M TiCl4 (in deionized water) was added to 0.4 mL under vigorous stirring. The microwave system was operated at 200 W, and the sample temperature was kept at 80 °C for 30 min. After the microwave process, the resultant slurry was filtered, washed with deionized water and ethanol, and dried in a drying oven at 80 °C. This product is denoted as TiO2/SC. To increase the crystallinity of TiO2, TiO2/SC was heat-treated at 350 °C in N2 atmosphere, and this product is denoted as TiO2/SC–H.Synthesis of TiO2–Pt/SC and TiO2–Pt/SC–H
The synthesis method of TiO2/SC and TiO2/SC–H (60 mg) was dispersed in 150 mL of ethylene glycol and sonicated for 10 min, respectively. 84 mg of H2PtCl6 was added to this solution and vigorously stirred for 10 min. The pH of the solution was adjusted to 12 by adding 5 M NaOH, and the mixed solution was continuously stirred for 60 min. The suspension was heated at 160 °C for 3 h. Thereafter, the suspension was filtered and washed with ethanol and distilled water. The obtained TiO2–Pt/SC and TiO2–Pt/SC–H were dried overnight at 80 °C.Characterization
Phase analysis was performed using powder X-ray diffraction (XRD) (Bruker Inc., D8-Advanced, Germany) with Cu Kα (λ = 1.5406 Å) radiation. X-ray photoelectron spectroscopy (XPS) was performed using a Thermo VG Scientific Sigma Probe spectrometer using an Al Kα X-ray (λ = 1486.6 eV) as the excitation source. The XPS data were calibrated using the C 1s peak at 284.5 eV. The morphologies of the samples were observed using transmission electron microscopy (TEM) (JEOL, JEM-4010, Japan). The oxygen and hydroxyls (OHs) were investigated by Fourier transform infrared-attenuated total reflectance (FTIR-ATR) (PerkinElmer Frontier, FT-IR/FIR Spectrometer, USA). Spectra were collected at room temperature over the range of 4000–500 cm−1 (16 scans, 4 cm−1 resolution) using the same FT-IR/FIR spectrophotometer as previously cited (PerkinElmer Frontier, Waltham, MA, USA), equipped with a germanium crystal.Electrochemical performance
Cyclic voltammetry (CV) and oxygen reduction reaction (ORR) activity measurements were performed in a three-electrode cell system using a VSP potentiostat (Biologic, France) and RRDE-3A rotating ring disk electrode model. The electrocatalytic activity of the hybrid samples was evaluated using a three-electrode setup with the counter, reference and working electrodes of Pt wire, Ag/AgCl, and glassy carbon (5 mm diameter), respectively. The catalyst solution was prepared by mixing the catalyst (10.0 mg), distilled water (280 µL), isopropyl alcohol (200 µL), and a 5 wt% Nafion solution (20 µL) under sonication for 1 h. Then, the catalyst (3 µL) was deposited on the glassy carbon electrode, and the working electrode was dried. The measured potential vs. Ag/AgCl was converted to the reversible hydrogen electrode (RHE) scale according to the Nernst equation:Here, ERHE is the converted potential vs. RHE,
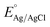 is 0.1976 V at 25 °C, and EAg/AgCl is the experimentally measured potential vs. Ag/AgCl reference. The prepared working electrode was washed with highly purified water, and then electrochemically cleaned by multiple potential cycles between 0.05 and 1.10 V vs. RHE in 0.1 M HClO4 at a scan rate of 50 mV s−1. CV data were measured by sweeping the potential between 0.05 and 1.10 V vs. RHE at a scan rate of 20 mV s−1. The ORR polarization curves of the catalysts loaded on rotating disk electrodes (diameter = 5 mm) were obtained using the linear sweep voltammetry technique in O2-saturated 0.1 M HClO4 solution at a scan rate of 10 mV s−1 with a rotating speed of 1600 rpm. Furthermore, to study the durability of the catalysts, an accelerated stability test was conducted by cycling the working electrode between 0.6 and 1.0 V vs. RHE at 500 mV s−1 in a N2-saturated electrolyte for 5000 cycles. The ORR activity measurement and CV were taken every 1000 cycles to observe the stability area. The ECSA of Pt can be calculated from the coulombic charges accumulated during hydrogen adsorption or desorption after correcting for the double-layer charging current from the CVs as follows:
is 0.1976 V at 25 °C, and EAg/AgCl is the experimentally measured potential vs. Ag/AgCl reference. The prepared working electrode was washed with highly purified water, and then electrochemically cleaned by multiple potential cycles between 0.05 and 1.10 V vs. RHE in 0.1 M HClO4 at a scan rate of 50 mV s−1. CV data were measured by sweeping the potential between 0.05 and 1.10 V vs. RHE at a scan rate of 20 mV s−1. The ORR polarization curves of the catalysts loaded on rotating disk electrodes (diameter = 5 mm) were obtained using the linear sweep voltammetry technique in O2-saturated 0.1 M HClO4 solution at a scan rate of 10 mV s−1 with a rotating speed of 1600 rpm. Furthermore, to study the durability of the catalysts, an accelerated stability test was conducted by cycling the working electrode between 0.6 and 1.0 V vs. RHE at 500 mV s−1 in a N2-saturated electrolyte for 5000 cycles. The ORR activity measurement and CV were taken every 1000 cycles to observe the stability area. The ECSA of Pt can be calculated from the coulombic charges accumulated during hydrogen adsorption or desorption after correcting for the double-layer charging current from the CVs as follows:Here, QH (mC) is the charge due to the hydrogen adsorption/desorption in the hydrogen region of the CV, and 0.21 mC cm−2 is the electrical charge associated with the monolayer adsorption of hydrogen on Pt. MPt is the mass of Pt loaded on the working electrode.
MEA performance
A commercially available gas diffusion layer (GDL, SGL-39BC, CNL) was used as the anode for the fuel cell experiments. The single cell was assembled by a commercial MEA (5 cm2 active area), GDL (SGL-39BC, CNL), Teflon gaskets, and Nafion 211 membrane, and operated for PEMFC. Catalyst inks were prepared by blending the sample with a Nafion (5 wt%) ionomer solution and isopropyl alcohol for 4 h. The catalyst ink was deposited on GDL with the Pt loading of 0.5 mg cm−2 on the cathode and the anode loading was 0.5 mg cm−2. Then, the single cell was installed at a fuel cell station (CNL Energy). The cell operation temperature was 70 °C and the relative humidity was 100%, while the stoichiometry of hydrogen and O2 was 105 and 330 cm3 min−1, respectively.Results & discussion
Scheme 1 schematically depicts the synthesis of the Pt catalysts for PEMFC. We supported the Pt catalysts on TiO2/SC via the microwave-assisted process. In this system, TiO2/SC has low crystallinity and mixed hydroxyl groups with anatase. After heat treatment at 350 °C, TiO2 has an anatase structure verifying oxygen content named as TiO2–Pt/SC–H. The support obtained by the above method was compared to that without additional heat treatment. As route 2, TiO2–SC was heated under nitrogen at 350 °C to obtain the anatase TiO2. The catalysts supported on the sample by routes 1 and 2 were synthesized via ethylene glycol reduction of the Pt precursor at 160 °C.As shown in Fig. 1a, the structure and morphology of TiO2–Pt/SC were investigated by transmission electron microscopy (TEM), revealing that the Pt nanoparticles (NPs) are homogeneously dispersed on the TiO2–carbon surface. In the corresponding high-resolution TEM (HR-TEM) images, lattice fringes with a spacing of 2.32 Å were observed, which can be assigned to the (111) crystal plane of Pt nanoparticles in TiO2–Pt/SC.18,19 The Pt nanoparticles supported on TiO2–Pt/SC exhibit a smaller particle size, which can be attributed to the more oxidized TiO2 surface being electron-deficient and the resulting lower diffusion rates of the Pt species.20–22 In contrast, the lattice fringes of TiO2 are not clearly observed in the HR-TEM images, likely due to its amorphous structure and disordered lattice, as shown in Fig. 1b.23 The average particle size of the Pt nanoparticles was determined to be 3.5 ± 1.4 nm (Fig. 1b). The compositional structure of TiO2–Pt/SC was further confirmed by elemental mapping using high-angle annular dark-field scanning transmission electron microscopy (HAADF-STEM) coupled with energy-dispersive X-ray spectroscopy (EDS), which clearly reveals the spatial distribution of Pt (red), C (green), and Ti (yellow), as shown in Fig. 1c–f. These results indicate that Pt nanoparticles are uniformly dispersed on the surfaces of both TiO2 and SP carbon, even though TiO2 partially covers the carbon surface (Fig. 1). For the TiO2–Pt/SC–H sample synthesized after heat treatment, the formation of TiO2 was confirmed by the observed lattice spacing and EDS elemental mapping obtained from TEM analysis. The average particle size of the Pt nanoparticles in TiO2–Pt/SC–H was determined to be 3.9 ± 1.3 nm (Fig. S1). In general, when TiO2 is synthesized in the presence of a metal species, hydroxyl (OH−) groups are removed.24 Accordingly, the formation mechanism of the synthesized catalysts can be described as follows: TiO2–Pt/SC and TiO2/SC–H are generated as a result of the removal of the surface OH groups induced by metal incorporation. These bands are associated with bridging OH species adsorbed on Ti atoms with different valence states. The hydroxyl species present on the TiO2 additive are likely to interact with interfacial water molecules through hydrogen bonding, inducing structural rearrangements in the interfacial water layer. Such structural changes effectively increase the local concentration of OH species near the electrode surface, thereby enhancing the supply of hydroxyl species to the Pt surface.25,26 The crystal structure of the TiO2–carbon composite was investigated by X-ray diffraction (XRD), which reveals the presence of both TiO2 and carbon phases. A diffraction peak observed at approximately 25° can be indexed to the (002) plane of carbon. In addition, the diffraction peaks of TiO2/SC and TiO2/SC–H located at 2θ values of 25.38°, 32.00°, and 38.11° correspond to the (101), (110), and (200) planes of anatase TiO2, respectively (JCPDS no. 21-1276).27,28 The broad diffraction feature observed in the 30°–40° range for TiO2/SC is attributed to the presence of the titanium hydroxide (Ti–OH) species (Fig. 1a).29 The weak crystallinity of TiO2 in TiO2/SC is consistent with the amorphous characteristics observed in the TEM images. As shown in Fig. S2a, diffraction peaks at 39.8°, 46.89°, and 68.38° can be assigned to the (111), (200), and (220) planes of the Pt nanoparticles, respectively (JCPDS no. 04-0802).30 After heat treatment, the crystallinity of TiO2 was significantly enhanced, and the formation of anatase TiO2 was further confirmed by lattice spacing measurements obtained from both XRD analysis and TEM images (Fig. S2a and b).
 | ||
| Fig. 1 (a) TEM image of TiO2–Pt/SC. (b) HR-TEM image of a Pt particle on TiO2–carbon. (c) HADDF-STEM image and (d–f) corresponding EDS elemental mapping images of TiO2–Pt/SC. | ||
Attenuated total reflectance (ATR) spectroscopy was performed to further investigate the oxygen species and surface hydroxyl groups on TiO2. The ATR spectra of Pt-loaded TiO2/SC and TiO2/SC–H were collected in the range of 500–4000 cm−1. The broad absorption band observed at 3400–3600 cm−1 is attributed to weakly bound surface hydroxyl groups with lateral interactions on TiO2–Pt/SC, and the broadening of this band indicates increased surface oxidation. The absorption band centered at approximately 3150 cm−1 is assigned to ordered hydroxyl (OH) species on the Ti surface (Fig. 2b).30,31 To further examine the surface oxygen states, X-ray photoelectron spectroscopy (XPS) was conducted to analyze the surface composition and chemical states of TiO2–Pt/SC and TiO2–Pt/SC–H. As shown in Fig. 2c and d, the O 1s spectra can be deconvoluted into three components: a peak at 531.0 eV corresponding to oxygen vacancies (V0) and surface hydroxyl groups (–OH), a peak at 529.5 eV assigned to lattice oxygen (O2−) in the Ti–O bonds of TiO2, and a peak at 527.8 eV attributed to weakly bound Ti–O species on the TiO2 surface.19,30 To elucidate the effect of carbon, additional measurements were performed on bulk TiO2 and heat-treated TiO2, and similar trends in the oxygen-related peaks were observed (Fig. S3). Overall, heat treatment induces the reoxidation of TiO2, leading to improved crystallinity and a reduction in the intensity of oxygen vacancy- and hydroxyl-related peaks. To investigate the influence of the oxygen-related surface species on the surface catalytic activity, the electrocatalytic performance of the synthesized catalysts was evaluated. Platinum nanoparticles are widely employed as catalysts for the oxygen reduction reaction (ORR) in fuel cells, and extensive efforts have been devoted to improving their catalytic activity and stability. In this study, the synthesized catalyst was applied to the ORR to evaluate its electrochemical performance. The ORR in fuel cells is strongly dependent on shifts in the d-band center of the catalyst. Strong metal–support interactions (SMSI) between TiO2 and Pt induce electron transfer from the support to Pt, leading to a downward shift of the Pt d-band center and, consequently, enhanced catalytic activity. This mechanism has been reported in theoretical studies of the ORR on Pt/C and TiO2–Pt systems, and such interactions are also known to improve catalyst durability.31,32
 | ||
| Fig. 2 (a) XRD patterns of SC, TiO2/SC and TiO2/SC–H. (b) ATR spectra of TiO2/SC and TiO2/SC–H on the Pt–support. XPS spectra with O 1s of (c) TiO2/SC–H and (d) TiO2/SC. | ||
Fig. 3a presents the cyclic voltammograms (CVs) of TiO2–Pt/SC and TiO2–Pt/SC–H recorded in a 0.1 M HClO4 electrolyte, which were used to determine the electrochemically active surface areas (ECSAs) of the corresponding catalysts. The influence of TiO2 on the electrocatalytic performance was evaluated through electrochemical measurements. The specific ECSAs, normalized to the Pt mass, were calculated by integrating the charge associated with the hydrogen adsorption/desorption region after correction for the double-layer contribution. The ECSAs of TiO2–Pt/SC, TiO2–Pt/SC–H, and Pt/C were determined to be 82.1, 71.3, and 66.8 m2 g−1, respectively. To evaluate the catalytic activity toward the oxygen reduction reaction (ORR), polarization curves were recorded in O2-saturated 0.1 M HClO4 solution at a scan rate of 10 mV s−1 and a rotation speed of 1600 rpm (Fig. 3b). A comparison of the ORR performance of TiO2–Pt/SC–H, TiO2–Pt/SC, and Pt/C revealed that TiO2–Pt/SC exhibited superior catalytic activity. In particular, the current density of TiO2–Pt/SC was approximately 0.2 mA cm−2 higher than that of TiO2–Pt/SC–H at 0.9 V, indicating a significant enhancement in the ORR electrocatalytic activity (Fig. 3b). This improvement can be attributed to the presence of TiO2 on the catalyst surface, which facilitates the dissociation of oxygen and water molecules and enhances oxygen adsorption on the support. The adsorbed oxygen species can subsequently migrate to adjacent Pt active sites via surface diffusion, thereby promoting the ORR.33,34 In addition, the enhanced ORR performance of TiO2–Pt/SC and TiO2–Pt/SC–H can be attributed to strong metal–support interactions (SMSI) between the Pt nanoparticles and the TiO2-containing support. These strong interactions lead to tight interfacial bonding, which effectively suppresses Pt migration and aggregation, thereby improving both catalytic stability and activity. The SMSI effect is likely associated with the relatively electron-rich nature of the Pt nanoparticles formed on the TiO2 support, enabling electron donation from Pt to the support via the Pt d-band centers. This electronic interaction weakens the Pt–O bond strength, which in turn facilitates ORR kinetics. As a result, TiO2–Pt/SC exhibited the highest ORR activity, outperforming previously reported catalysts (Table S1). Since electrochemical reactions of the catalysts primarily occur at the surface, X-ray photoelectron spectroscopy (XPS) was employed to analyze the surface binding energies of the metal species. The Ti 2p XPS spectrum exhibits characteristic doublet peaks at approximately 459.4 and 465.1 eV, corresponding to the Ti 2p3/2 and Ti 2p1/2 states, respectively (Fig. 3c). The spin–orbit splitting of 5.7 eV confirms the presence of Ti exclusively in the Ti4+ oxidation state. Compared with pristine TiO2, the Ti 2p peaks show a slight shift toward higher binding energies, suggesting a modification of the Ti chemical environment, likely arising from strong interactions with the carbon support. In contrast, the broader Ti 2p peaks observed for titanium powder are attributed to the coexistence of metallic Ti and sub-stoichiometric oxides associated with lower oxidation states (Ti2+ and Ti3+), which are present beneath the anatase layer covering the powder surface. As shown in Fig. 3d, the Pt 4f XPS spectra display asymmetric 4f7/2 and 4f5/2 spin–orbit components, a characteristic feature of metallic Pt (Pt0). The presence of higher binding energy components indicates a partial oxidation state of Pt.35 After heat treatment, an increase in the Pt binding energy is observed, which can be attributed to electron transfer from Pt to the oxide support. Furthermore, the noticeable positive shift of approximately 1.2 eV relative to TiO2–Pt/SC–H is ascribed to enhanced metal–support interactions. Analysis of the valence band XPS spectra reveals that the d-band center positions of TiO2–Pt/SC and TiO2–Pt/SC–H are located at −4.41 and −4.04 eV, respectively (Fig. S4). The downshift of the Pt 5d-band center is known to enhance the ORR activity by lowering the energy level of the antibonding orbitals associated with oxygen adsorption on Pt (Pt–O) relative to the Fermi level, thereby weakening the Pt–O bond strength.33 The metal–support interactions reduce the adsorption strength of oxygen-containing intermediates formed on the Pt surface during the rate-determining step of the oxygen reduction reaction (ORR), thereby accelerating the overall ORR kinetics. In addition, the reaction between the surface-bound Pt–OH species and oxygen constitutes a critical step in the ORR pathway, which is consistent with the enhanced ORR performance observed. The Pt–TiO2 nanocomposites exhibit superior electrocatalytic activity toward oxygen reduction, which can be attributed to the hypo-d-electron character of titanium oxide. This electronic feature facilitates the effective spillover of the adsorbed hydroxyl species from the hypo-d-electron oxide support to the Pt surface at the Pt–TiO2 interface. Since hypo-d-electron oxides possess ion-exchange membrane-like properties, a higher valence capacity promotes a more pronounced spillover effect.15 Furthermore, the enhanced electrocatalytic activity of the Pt catalyst is associated with the formation of crystalline TiO2. The stability of the catalysts was evaluated through repeated cyclic voltammetry (CV) measurements. Catalyst durability was further examined by conducting accelerated degradation tests (ADTs) via CV in the potential range of 1.0–1.5 V at a scan rate of 500 mV s−1. Fig. 4 presents the ORR polarization curves and CV profiles recorded before and after the durability tests. Both catalysts exhibited a decrease in the electrochemically active surface area (ECSA) after maintaining the potential at 1.23 V for 1 h, which is attributed to the Pt migration and particle growth during prolonged cycling. Notably, after 5000 cycles, the ECSA loss of TiO2–Pt/SC (Fig. 4a) was significantly smaller than that of TiO2–Pt/SC–H (Fig. 4d), demonstrating its superior durability.
The electrochemical activity of the TiO2–Pt/SC catalyst is slightly higher than that of the commercial Pt/C catalyst, which can be attributed to the similar electronic structures of Ti and Pt, as both elements belong to the d-block and possess comparable outer electronic configurations. The interaction between Ti and Pt is facilitated by the overlap between the occupied d orbitals of Pt and the vacant d orbitals of Ti, promoting strong interfacial interactions. These interactions effectively anchor the Pt nanoparticles, suppressing their migration and agglomeration. Moreover, the electronic interaction between TiO2 and Pt modifies the electronic structure of Pt, thereby enhancing the intrinsic catalytic activity.
The prepared TiO2–Pt/SC catalyst and commercial Pt/C were employed as cathode catalysts in PEMFCs, as schematically illustrated in Fig. 5a, with a catalyst loading of 0.5 mg cm−2. Fig. 5b and c present the current density–voltage (I–V) polarization curves of the membrane electrode assemblies (MEAs) recorded in an H2/O2 atmosphere before and after the accelerated stress test (AST). The effect of potential cycling on the overall cell performance was minimal. Nafion® 212 was used as the proton exchange membrane, and the effective electrode area was 5 cm2. Both gas humidification and cell operation were conducted at 70 °C. The AST was performed by holding the cell at 1.23 V for 24 h. After the AST, the open-circuit voltage (OCV) slightly decreased from 1.00 to 0.978 V. The polarization curves can be divided into three distinct regions: activation polarization, ohmic polarization, and concentration polarization. For the TiO2–Pt/SC catalyst, current densities of 237 mA cm−2 at 0.80 V and 1519 mA cm−2 at 0.60 V were achieved, whereas the Pt/C catalyst delivered 282 mA cm−2 at 0.80 V and 1522 mA cm−2 at 0.60 V. The maximum power densities (MPDs) of TiO2–Pt/SC and Pt/C were measured to be 1076 and 1050 mW cm−2, respectively. In the low current density region, the Pt/C catalyst exhibited higher electrochemical polarization, indicating slower electrochemical reaction kinetics. This behavior may be attributed to differences in the electrode wettability during the initial discharge stage. The faster electrode wettability of the Pt/C catalyst facilitates proton conduction, thereby reducing electrochemical polarization. The superior stability of TiO2–Pt/SC is attributed to strong metal–support interactions (SMSI) between the Pt nanoparticles and the carbon/TiO2 support, which effectively suppress Pt detachment and subsequent agglomeration.19,35 As shown in Fig. 5d, the ohmic resistance was calculated from the slope of the I–V curve within the linear range from 0.5 to 1.0 A cm−2; the value of TiO2–Pt/SC (0.146 Ω cm2) was similar to that of Pt/C (0.150 Ω cm2) before cycling. This similarity might imply that the carbon/TiO2 support is sufficiently conductive to be used as a catalyst support, comparable to the commercial carbon black in Pt/C. Therefore, we could infer that the poor electrical conductivity issues of the ceramic support could be mitigated by the carbon/TiO2 assembly. Furthermore, the ohmic resistance of TiO2–Pt/SC increased by only 2.74% after AST, which is a very small change, indicating the robustness of the TiO2/SC support. On the other hand, the resistance of Pt/C increased drastically from 0.150 to 0.173 Ω cm2, which is an increase of 15.3% (Fig. 5d and e). The stability of the synthesized TiO2–Pt/SC and Pd/C was evaluated through repeated cyclic voltammetry (CV) measurements.36,37 As a result, the PEMFC employing TiO2–Pt/SC as the cathode catalyst exhibited better performance than that using the commercial Pt/C catalyst. The current density and decay rate values measured before and after accelerated stress tests (ASTs) are summarized in Table S2 and Fig. S5. Notably, the TiO2–Pt/SC-based fuel cell maintained a high current density and stable cyclic voltammetry (CV) characteristics after accelerated durability testing.
Conclusion
In this work, a TiO2–carbon hybrid support for Pt catalysts was successfully synthesized via a simple microwave-assisted process. The crystallinity and phase composition of TiO2 were found to vary, depending on the presence or absence of post-synthesis heat treatment. In the absence of a highly crystalline anatase TiO2 matrix, TiO2–Pt/SC exhibited higher ORR activity than TiO2–Pt/SC–H, which can be attributed to the presence of surface hydroxyl (–OH) groups and their associated binding energies. Electrocatalytic ORR activity and durability evaluations demonstrated that TiO2–Pt/SC possesses superior resistance to carbon corrosion compared with TiO2–Pt/SC–H, owing to strong metal–support interactions (SMSI). After accelerated stress tests (ASTs), the electrochemically active surface area (ECSA) loss of TiO2–Pt/SC was limited to only 6.41%, whereas the commercial Pt/C catalyst exhibited a significantly higher loss of 29.86%. Furthermore, membrane electrode assembly (MEA) tests conducted under AST protocols revealed that TiO2–Pt/SC maintained excellent fuel cell performance, showing only a 2.42% decrease in maximum power density after holding at 1.23 V. Overall, this study provides a feasible strategy for the synthesis of carbon–metal oxide hybrid composite supports for Pt catalysts. Moreover, optimization of surface functional groups on the support offers a promising pathway for the development of high-performance cathode catalysts for proton exchange membrane fuel cells (PEMFCs).Conflicts of interest
There are no conflicts to declare.Data availability
The supplementary information (SI) file includes all data supporting the article. Supplementary information is available. See DOI: https://doi.org/10.1039/d5ra08718j.Acknowledgements
This work was supported by the Glocal University 30 Project Fund of Gyeongsang National University in 2025 and the Nano & Material Technology Development Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science and ICT (RS-2025-25454853).References
- P. Simon and Y. Gogotsi, Capacitive energy storage in nanostructured carbon–electrolyte systems, Acc. Chem. Res., 2013, 46, 1094 CrossRef PubMed.
- L. Du, Y. Shao, J. Sun, G. Yin, J. Liu and Y. Wang, Advanced catalyst supports for PEM fuel cell cathodes, Nano Energy, 2016, 29, 314 CrossRef.
- J. K. Nørskov, J. Rossmeisl, A. Logadottir, L. Lindqvist, J. R. Kitchin, T. Bligaard and H. Jonsson, Origin of the overpotential for oxygen reduction at a fuel-cell cathode, J. Phys. Chem. B, 2004, 108, 17886 CrossRef PubMed.
- M. K. Debe, Electrocatalyst approaches and challenges for automotive fuel cells, Nature, 2012, 486, 43 CrossRef PubMed.
- K. I. Ozoemena, and S. Chen, Nanomaterials for Fuel Cell Catalysis, Springer, 2016 Search PubMed.
- S. W. Lee, S. R. Choi, J. Jang, G.-G. Park, S. H. Yu and J.-Y. Park, Tolerance to carbon corrosion of various carbon structures as catalyst supports for polymer electrolyte membrane fuel cells, J. Mater. Chem. A, 2019, 7, 25056 Search PubMed.
- N. Abdullah and S. Kamarudin, Titanium dioxide in fuel cell technology: an overview, J. Power Sources, 2015, 278, 109 Search PubMed.
- Z. W. Seh, J. Kibsgaard, C. F. Dickens, I. Chorkendorff, J. K. Nørskov and T. F. Jaramillo, Combining theory and experiment in electrocatalysis: Insights into materials design, Science, 2017, 355, 4998 CrossRef PubMed.
- E. C. Barbosa, L. S. Parreira, I. C. de Freitas, L. R. Aveiro, D. C. de Oliveira, M. C. dos Santos and P. H. Camargo, Pt-decorated TiO2 materials supported on carbon: increasing activities and stabilities toward the ORR by tuning the Pt loading, ACS Appl. Energy Mater., 2019, 2, 5759 CrossRef.
- A. Kongkanand, W. Gu, and M. F. Mathias, Proton-exchange membrane fuel cells with low-Pt content, in In Fuel Cells and Hydrogen Production, Springer, New York, NY, 2019, p. 323 Search PubMed.
- S. Tauster, S. Fung, R. Baker and J. Horsley, Strong interactions in supported-metal catalysts, Science, 1981, 211, 1121 CrossRef PubMed.
- S. Tauster, S. Fung and R. L. Garten, Strong metal-support interactions. Group 8 noble metals supported on titanium dioxide, J. Am. Chem. Soc., 1978, 100, 170 Search PubMed.
- J. M. Jaksic, N. V. Krstajic, L. M. Vracar, S. G. Neophytides, D. Labou, P. Falaras and M. M. Jaksic, Spillover of primary oxides as a dynamic catalytic effect of interactive hypo-d-oxide supports, Electrochim. Acta, 2007, 53, 349 Search PubMed.
- J. M. Jaksic, G. D. Papakonstantinou, D. Labou, A. Siokou, and M. M. Jaksic, New and Future Developments in Catalysis: Chapter 8. Spillover Phenomena in Electrocatalysis for Oxygen and Hydrogen Electrode Reactions, Elsevier Inc. Chapters, 2013 Search PubMed.
- N. V. Krstajic, L. M. Vracar, V. R. Radmilovic, S. G. Neophytides, M. Labou, J. M. Jaksic, R. Tunold, P. Falaras and M. M. Jaksic, Advances in interactive supported electrocatalysts for hydrogen and oxygen electrode reactions, Surf. Sci., 2007, 601, 1949 CrossRef.
- A. Truppi, F. Petronella, T. Placido, V. Margiotta, G. Lasorella, L. Giotta, C. Giannini, T. Sibillano, S. Murgolo and G. Mascolo, Gram-scale synthesis of UV-vis light active plasmonic photocatalytic nanocomposite based on TiO2/Au nanorods for degradation of pollutants in water, Appl. Catal., B, 2019, 243, 604 CrossRef.
- K. Thamaphat, P. Limsuwan and B. Ngotawornchai, Phase characterization of TiO2 powder by XRD and TEM, J. Agric. Nat. Resour., 2008, 42, 357 Search PubMed.
- E. Lee, C. Park, D. W. Lee, G. Lee, H.-Y. Park, J. H. Jang, H.-J. Kim, Y.-E. Sung, Y. Tak and S. J. Yoo, Tunable synthesis of N, C-codoped Ti3+-enriched titanium oxide support for highly durable PEMFC cathode, ACS Catal., 2020, 10, 12080 CrossRef.
- R. P. Galhenage, H. Yan, S. A. Tenney, N. Park, G. Henkelman, P. Albrecht, D. R. Mullins and D. A. Chen, Understanding the nucleation and growth of metals on TiO2: Co compared to Au, Ni, and Pt, J. Phys. Chem. C, 2013, 117, 7191 CrossRef.
- C. T. Campbell, Ultrathin metal films and particles on oxide surfaces: structural, electronic and chemisorptive properties, Surf. Sci. Rep., 1997, 27, 1 CrossRef.
- A. Zielińska-Jurek, Z. Wei, M. Janczarek, I. Wysocka and E. Kowalska, Size-controlled synthesis of Pt particles on TiO2 surface: Physicochemical characteristic and photocatalytic activity, Catalysts, 2019, 9, 940 CrossRef.
- T. Xia, W. Zhang, Z. Wang, Y. Zhang, X. Song, J. Murowchick, V. Battaglia, G. Liu and X. Chen, Amorphous carbon-coated TiO2 nanocrystals for improved lithium-ion battery and photocatalytic performance, Nano Energy, 2014, 6, 109 CrossRef.
- R. Zanella, L. Delannoy and C. Louis, Mechanism of deposition of gold precursors onto TiO2 during the preparation by cation adsorption and deposition–precipitation with NaOH and urea, Appl. Catal., A, 2005, 291, 62 CrossRef.
- S. Ali, Z. Li, W. Ali, Z. Zhang, M. Wei, Y. Qu and L. Jing, Synthesis of Au-decorated three-phase-mixed TiO2/phosphate modified active carbon nanocomposites as easily-recycled efficient photocatalysts for degrading high-concentration 2, 4-DCP, RSC Adv., 2019, 9, 38414 RSC.
- D. Dambournet, I. Belharouak and K. Amine, Tailored preparation methods of TiO2 anatase, rutile, brookite: mechanism of formation and electrochemical properties, Chem. Mater., 2010, 22, 1173–1179 CrossRef.
- A. E. Nogueira, L. S. Ribeiro, L. F. Gorup, G. T. Silva, F. F. Silva, C. Ribeiro and E. R. Camargo, New approach of the oxidant peroxo method (OPM) route to obtain Ti (OH)4 nanoparticles with high photocatalytic activity under visible radiation, Int. J. Photoenergy, 2018, 1, 6098302 Search PubMed.
- S. Shanmugam and A. Gedanken, Carbon-Coated Anatase TiO2 Nanocomposite as a High-Performance Electrocatalyst Support, Small, 2007, 3, 1189 Search PubMed.
- T.-H. Wang, A. M. Navarrete-López, S. Li, D. A. Dixon and J. L. Gole, Hydrolysis of TiCl4: initial steps in the production of TiO2, J. Phys. Chem. A, 2010, 114, 7561 CrossRef PubMed.
- J. Li, H. Yu, Q. Sun, Y. Liu, Y. Cui and Y. Lu, Growth of TiO2 coating on wood surface using controlled hydrothermal method at low temperatures, Appl. Surf. Sci., 2010, 256, 5046 Search PubMed.
- Q. Sun, Y. Lu, H. Zhang, H. Zhao, H. Yu, J. Xu, Y. Fu, D. Yang and Y. Liu, Hydrothermal fabrication of rutile TiO2 submicrospheres on wood surface: An efficient method to prepare UV-protective wood, Mater. Chem. Phys., 2012, 133, 253 Search PubMed.
- X. L. S. Li, L. Li, X. Zhu, H. Tang and Y. Wang, Pt Nanoparticles Supported on N-Doped TiO2/C Composite Supports for Fuel Cell Catalysis, J. Alloys Compd., 2025, 184103 Search PubMed.
- Z. Wang, X. Jin, F. Chen, X. Kuang, J. Min, H. Duan and J. Chen, Oxygen vacancy induced interaction between Pt and TiO2 to improve the oxygen reduction performance, J. Colloid Interface Sci., 2023, 650, 901 CrossRef PubMed.
- E. I. Vovk, A. V. Kalinkin, M. Y. Smirnov, I. O. Klembovskii and V. I. Bukhtiyarov, XPS study of stability and reactivity of oxidized Pt nanoparticles supported on TiO2, J. Phys. Chem. C, 2017, 121, 17297–17304 CrossRef.
- M. Mavrikakis, B. Hammer and J. K. Nørskov, Effect of strain on the reactivity of metal surfaces, Phys. Rev. Lett., 1998, 81, 2819 CrossRef.
- D. L. Wood III, S. Y. Jung and T. V. Nguyen, Effect of direct liquid water injection and interdigitated flow field on the performance of proton exchange membrane fuel cells, Electrochim. Acta, 1998, 43, 3795 CrossRef.
- A. Yao-Lin, Z. Y. Du, H. J. Ze, X. T. Wang, Y. Zhang, H. Zhang and J. F. Li, Understanding the molecular mechanism of oxygen reduction reaction using in-situ Raman spectroscopy, Curr. Opin. Electrochem., 2023, 42, 101381 Search PubMed.
- Y. Xiong, Y. Ma, L. Zou, S. Han, H. Chen, S. Wang and H. Yang, N-doping induced tensile-strained Pt nanoparticles ensuring an excellent durability of the oxygen reduction reaction, J. Catal., 2020, 382, 247 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |