 Open Access Article
Open Access ArticleAn investigation on the electrochemical performance of Mn3O4-based aqueous symmetric supercapacitor devices
Chrisma Rose Babu ab,
E. I. Anila
ab,
E. I. Anila *a,
A. V. Avania,
Roshan Josec and
Xavier T. S
*a,
A. V. Avania,
Roshan Josec and
Xavier T. S d
d
aOptoelectronic and Nanomaterials' Research Laboratory, Department of Physics and Electronics, CHRIST University, Bengaluru, Karnataka-560029, India. E-mail: anila.ei@christuniversity.in
bCochin University of Science and Technology, Ernakulam, Kerala-682022, India
cDepartment of Physics, Marian College Kuttikkanam Autonomous, Idukki, Kerala-685531, India
dGovernment College for Women, Thiruvananthapuram, Kerala-695014, India
First published on 19th February 2026
Abstract
Manganese (II, III) oxide (Mn3O4) is one of the promising materials in the realm of high-performance supercapacitors. The high theoretical specific capacitance, low cost, non-toxicity, environmental compatibility, and natural abundance made it significant in the research field. A low-temperature hydrothermal synthesis method was adopted to prepare Mn3O4 (hausmannite) nanoparticles with a tetragonal spinel structure. The as-prepared nanoparticles were assessed for the structural, elemental, electrical, optical and nitrogen adsorption–desorption studies through XRD, FTIR, Raman spectroscopy, XPS, DC conductivity, UV-vis absorption and BET analyses. Morphological studies were done using FESEM and TEM and a mixture of nanorods and nanocubes were observed. The electrochemical performances of the as-prepared Mn3O4 nanoparticles were investigated by cyclic voltammetry (CV), galvanostatic charge/discharge (GCD) method and electrochemical impedance spectroscopy (EIS) in a three-electrode system. The present work reports the fabrication of a prototype aqueous symmetric supercapacitor device for the first time. The electrochemical studies were performed in 0.5 M Na2SO4 electrolyte on the separator with a potential window of 0 V to 1 V. A specific capacitance of 68 Fg−1 at a current density of 1 Ag−1 was observed from the constant charge/discharge method. It exhibited a cyclic stability of 72% with a coulombic efficiency of 100% after 1000 cycles. This underscores the noteworthy role of manganese oxide nanoparticles as electrode materials in supercapacitors.
1. Introduction
The increasing energy consumption has recently emerged as a severe focus to develop clean, sustainable, environmentally acceptable, and affordable energy resources. Considerable research has focused on this arena by promoting energy storage devices like batteries, supercapacitors and fuel cells. The demand for fast charge/discharge rates, cycle life, stability, power density and production costs consider supercapacitors as alternate energy storage devices for many applications in daily life. Supercapacitors are a power source for hybrid vehicles, electronic devices and grid systems. The performance of supercapacitors mainly depends on the properties of the electrode materials.1–3 The electric double-layer capacitor and pseudocapacitor are two significant kinds of supercapacitors. The charge storage mechanism of these supercapacitors is classified into two: faradaic and non-faradaic charge storage. Electric double-layer capacitors (EDLC) comprise carbon compounds that undergo non-faradaic charge storage, whereas pseudocapacitors consist of either metal oxide-based or conducting polymer-based electrode materials that undergo a faradaic charge storage mechanism.4,5Transition metal oxides play a vital role as electrode materials in pseudocapacitors because of their high energy and power density, which can store energy through quick faradaic redox reactions.6,7 Various transition metal oxides like RuO2,8 Co3O4,9 NiO,10 MoO3 (ref. 11) and manganese-based oxides are utilized for the fabrication of supercapacitor electrodes. Manganese oxides are considered promising electrode materials for their advantages: low cost, high power density, high specific capacitance, availability and environment-friendly nature. There are four different types of manganese oxides: MnO, MnO2, Mn2O3 and Mn3O4 due to the presence of three kinds of oxidation states of manganese such as Mn2+, Mn3+ and Mn4+. Mn3O4 was the most stable oxide form among them.12 Hausmannite nanostructures can be synthesized through the hydrothermal method,13 co-precipitation,14 sol–gel,15 solvothermal,16 microwave-assisted17 and chemical reduction,18 etc. Mn3O4 has a normal spinel structure at room temperature, in which Mn2+ ions are at the tetrahedral sites and Mn3+ ions are occupied at the octahedral sites.19 It possesses a theoretical specific capacitance of 1400 Fg−1, low electronic conductivity and a wide potential window in the aqueous electrolyte medium.20
Wang et al.21 synthesized Mn3O4 nanomaterials through a two-step hydrothermal method and produced a specific capacitance of 233.4 Fg−1 at a lower current density of 0.5 Ag−1. A one-step hydrothermal method was followed by Prasad et al. for developing a Mn3O4 supercapacitor electrode with a specific capacitance of 147.5 Fg−1 at a current density of 0.5 Ag−1.22 Jiang et al.13 found Mn3O4 nano-octahedrons through hydrothermal synthesis method to develop an electrode material that provides a specific capacitance of 244 Fg−1 at a scan rate of 5 mV s−1. In all these works, electrochemical properties in a three-electrode configuration. Fabrication of symmetric supercapacitor device with Mn3O4 synthesized by hydrothermal method was done for the first time in this work.
The above-discussed works have inspired the present work to synthesize hausmannite Mn3O4 nanoparticles in a cost-effective way, without any surfactants or further calcination process. The structural, elemental, morphological, electrical, optical and surface properties were observed through X-ray diffraction studies (XRD), Fourier transform infrared spectroscopy (FTIR), Raman spectroscopy, transmission electron microscopy (TEM), X-ray photoelectron spectroscopy (XPS), field emission scanning microscopy (FESEM), DC conductivity, UV-visible absorption spectroscopy and Branauer Emmett Teller analyses. The fabrication of the Mn3O4 electrode presents a specific capacitance value of 163 Fg−1 at a current density of 4 Ag−1, which was examined by the galvanostatic charge–discharge method. In this work, we report the fabrication of prototype aqueous symmetric supercapacitors for the first time with Mn3O4 electrodes prepared via simple hydrothermal method. The Mn3O4 symmetric supercapacitor device gained a specific capacitance of 68 Fg−1 at 1 Ag−1 with an energy density 9 W h kg−1.
2. Experimental section
2.1. Chemicals
The chemical reagents used in the present work are of analytical grade and used without additional refinement. Sodium hydroxide and manganese chloride tetrahydrate were purchased from S. D. Fine Chem Ltd and Loba Chemie PVT. Ltd, respectively, India.2.2. Preparation of Mn3O4 nanoparticles
For the synthesis of Mn3O4 nanoparticles, 1 M manganese chloride tetrahydrate (MnCl2·4H2O) and 2 M sodium hydroxide (NaOH) taken in 25 mL distilled water separately were allowed for continuous stirring. NaOH was added dropwise to the manganese chloride precursor solution, forming a pale brown colloidal solution. The solution was then transferred to the 100 mL Teflon-lined autoclave. The solution underwent thermal treatment for 24 hours at 120 °C. After filtration and double-washing with distilled water, a dark brown product was obtained. It was dried at 60 °C to obtain pure Mn3O4 nanoparticles.2.3. Material characterizations
X-ray diffraction studies were analyzed using the Rigaku Miniflex-XRD diffractometer with radiation of Cu Kα (1.5406 Å) working at 40 kV and 15 mA to determine the crystallinity of the as-prepared samples. By using Renishaw in Via Raman Microscope, the vibrational modes were identified through the Raman spectra. FTIR was observed using Shimadzu IR Spriti-L. Morphological analysis was done with Apero 2 FESEM (ThermoFisher Scientific). The elemental analysis was evaluated using a PHI-VERSAPROBE III X-ray photoelectron spectrometer. Altamira Instrument (model TB440A) was used for measuring specific surface area and pore size distribution through Brunaeur-Emmett-Teller (BET) and Barret-Joyner-Halenda (BJH) methods. DC conductivity studies were observed from Keithely 2450 SMU. The optical studies were done using a UV-1800 Shimadzu spectrophotometer. Electrochemical mechanisms were recorded using the CH instrument (CH660E).2.4. Electrode fabrication and electrochemical tests
The supercapacitor electrode material was fabricated using nickel foam as the current collector. The nickel foam was cleaned by sonicating with acid, alkaline solutions and distilled water for few minutes. The working electrode was made up of Mn3O4 nanoparticles (80 wt% −0.0008 g), activated carbon (10 wt% −0.0001 g) as the conductive agent, and polytetrafluoroethylene (10% PTFE −0.0001 g) as the binder, which was sonicated in 2 ml ethanol and drop-casted on the substrate. The substrate used in the present work was a nickel foam of an area of 1 cm2. The drop-casted electrodes were further dried at 60 °C for a few hours. The average mass loading was found to be 1 mg cm−2. A three-electrode setup was assisted in analyzing the electrochemical performance of the active electrode material using cyclic voltammetry curves, galvanostatic charge–discharge curves and electrochemical impedance studies (1 M Hz to 1 Hz) at room temperature. The electrochemical mechanisms were performed in a CH (CHI660E) electrochemical workstation. The specific capacitance value (Csp) can be observed from both cyclic voltammetry and charge–discharge analyses using the following equations:
 | (1) |
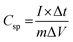 | (2) |
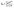 | (3) |
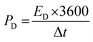 | (4) |
2.5. Preparation of Mn3O4//Mn3O4 symmetric supercapacitor device
The prototype aqueous symmetric supercapacitor device with the as-prepared Mn3O4 electrodes followed by sandwiching the two similar electrodes (1 cm2 nickel foam with a thickness of 1.5 mm) separated by a separator. The Whatman filter paper (90 mm pore size) was taken as the separator for the device filled with 0.5 M Na2SO4 electrolyte solution.3. Results and discussions
The crystal structure of the Mn3O4 nanoparticles were observed from the X-ray diffraction pattern as given in Fig. 1a. It corresponds to its normal spinel oxide with Mn2+ ions at the tetrahedral sites and Mn3+ at the octahedral sites (hausmannite). The predominant peaks were identified at 2θ values of 28.8°, 32.3°, 36.07° and 59.8°corresponding to (112), (103), (211) and (224) crystal planes. The other peaks with lower intensities correspond to the crystal planes of (101), (200), (004), (220), (105), (312), (303), (321), (224), (400), (206) and (413). No other impurity peaks were identified from the synthesized sample and the observed peaks are concordant with PDF card no: 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 001
001![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 963.24
963.24
 | ||
| Fig. 1 (a). X-ray diffraction pattern, (b). Raman spectrum and (c). FTIR spectrum of Mn3O4 nanoparticles. | ||
The average crystallite size, D, can be measured from the Scherrer formula,
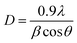 | (5) |
 | (6) |
The calculated lattice constants, a = b = 5.771 Å and c = 9.450 Å which differ from those of bulk (a = b = 5.769 Å and c = 9.460 Å) due to size defect and presence of point defects. Structural refinements of Fig. 1a were performed using Profex software with a tetragonal structure and space group I41/and (No. 141). The Rietveld-refined XRD pattern and the corresponding crystal structure are shown in Fig. S1 and S2. The reliability factors, atomic positions, and bond lengths between atoms are provided in Tables S1 and S2. The presence of the hausmannite phase is confirmed from the analysis.
The phase of the Mn3O4 nanoparticles can be confirmed by Raman spectroscopic analysis, a sensitive technique to identify the atomic vibrations and arrangements.26 Fig. 1b shows the Raman spectra of the Mn3O4 nanoparticles synthesized through the hydrothermal method. The spectrum was analyzed, and some major peaks were found in the 300–700 cm−1 range. The single broad peak at 658 cm−1 agrees with the spinel structure of Mn3O4, which corresponds to the stretching mode (Mn–O) of Mn3O4, i.e., A1g mode. This mode explains the atomic vibrations of divalent manganese ions at the tetrahedral site which may be associated with Jahn–Teller distortion under certain conditions.27 The other two major peaks were observed at 320 cm−1 and 373 cm−1 corresponding to the doubly degenerate T2g (O–Mn–O) and Eg (Mn–O–Mn) symmetry modes respectively.28 These observations confirm that no other secondary phase was formed which further agrees with the XRD results.
The chemical bonds present in the Mn3O4 nanoparticles were analyzed using Fourier transform infrared (FT-IR) spectroscopy as shown in Fig. 1c. The spectrum exhibits characteristic peaks at 611 cm−1 and 465 cm−1 which corresponds to the vibrations of Mn3+ ions at the octahedral sites. The absorption peak near 1095 cm−1 is attributed to Mn–OH vibrational modes. The peaks at 1625 cm−1 and 3392 cm−1 are associated with the presence of stretching and bending vibrations of water molecules adsorbed.16,29,30
The investigation structure and morphology of the Mn3O4 nanoparticles were analyzed from the transmission electron microscopy images. Fig. 2a and b showed the nanorods-like and distorted nanocube-like structures of Mn3O4 nanoparticles at different magnifications. The HR-TEM image revealed an interplanar spacing of 0.49 nm corresponding to the (101) crystal planes of Mn3O4 as given in Fig. 2c. The SAED (selected area electron diffraction) (Fig. 2d) pattern supports the fringe spacing observation and it exhibits the polycrystalline nature of Mn3O4. The surface morphology can be identified using field emission scanning electron microscopy (FESEM). The FESEM image shows the mixture of distorted cube-like and rod-like structures at different magnifications are shown in Fig. 3. At high magnifications, the rod-like structures were covered with cube-like particles. An average particle size from 40–85 nm was observed for the cube-like structures. This kind of morphology promotes active sites for the application of supercapacitors.
 | ||
| Fig. 2 (a and b). TEM images at different magnifications, (c). HR-TEM image and (d). SAED pattern of Mn3O4 nanoparticles. | ||
The oxidation states and elemental composition of the Mn3O4 nanoparticles were analyzed from the X-ray photoelectron spectroscopy as shown in Fig. 4. The XPS survey was observed in the binding energy range between 0 to 1100 eV as given in Fig. 4a. No other impurities were significantly identified from the survey spectrum. The detailed spectrum of Mn 2p is shown in Fig. 4b. The major peaks of Mn 2p1/2 and Mn 2p3/2 were identified at binding energies of 652.5 eV and 641.0 eV, respectively. The spin–orbit splitting width between them was found to be 11.5 eV. The peak components of Mn 2p1/2 was deconvoluted into Mn2+ and Mn3+ at binding energies of 654.4 eV and 652.25 eV. Also, Mn 2p3/2 was deconvoluted into Mn2+ with a binding energy of 642.9 eV and Mn3+ with a binding energy of 640.75 eV.31–34 The purity of the Mn3O4 compound can be confirmed by analyzing the O 1s spectrum given in Fig. 4c. The peaks at 530.2 eV, 532.3 eV and 533.9 eV correspond to the presence of Mn–O–Mn bond for oxide, Mn–O–H for hydroxide and O–H for residual water, respectively.22,35
The detailed curve-fitting of the Mn 2p XPS spectra confirms the presence of Mn2+ and Mn3+ oxidation states. The quantitative analysis determined the Mn2+ and Mn3+ fractions to be 36% and 64% based on Mn 2p3/2 peak area and 35% and 65% based on Mn 2p1/2 peak area, which resulted in the Mn2+/Mn3+ ratio as ∼0.54. The ratio confirms the mixed valence manganese oxide phase with a composition close to the stable hausmannite Mn3O4.36
The surface area and pore-size distributions of porous Mn3O4 nanoparticles were observed from the N2 adsorption–desorption isotherms given in Fig. 5a. Type IV isotherm can be analyzed from the distinct hysteresis loop at high pressure region. The BET surface area obtained for the as-synthesized Mn3O4 is 15.25 m2 g−1. The pore size distribution was observed through Barrett–Joyner–Halenda (BJH) method shown in Fig. 5b. The mesoporous nature of Mn3O4 nanoparticles was identified from the average pore diameter, 8.561 nm and it has an average pore volume of 0.033 cm3 g−1. The BET surface area provided effective reaction sites for the accessibility of the electrolyte ions to the active electrode material for fast charging-discharging process.
 | ||
| Fig. 5 (a). N2 adsorption–desorption isotherm and (b). Pore size distribution of Mn3O4 nanoparticles. | ||
The synthesized Mn3O4 nanoparticles were subjected to DC conductivity analysis using Keithley 2450 SMU conductivity meter. The conductivity studies were performed in a two-probe setup by pelletizing the obtained Mn3O4 powder. The pellet was found to be 12 mm in diameter and 36 mm in thickness. The electrical properties were analyzed based on the current vs. voltage characteristics. The conductivity obtained for the Mn3O4 nanoparticles was 9.38 × 10−6 S m−1 using the relation, conductivity is directly proportional to thickness of the pellet and inversely proportional to the product of area and electrical resistance.
The optical properties were observed from the UV-vis absorption analysis. The absorption spectrum was recorded in the range of 200–800 nm. The absorption peaks were found near 260 nm corresponds to the charge transfer transition at O2− to Mn3+. Fig. 6a demonstrates the change in the absorbance plot with respect to the wavelength of Mn3O4 nanoparticles. The optical energy band gap (Eg) of the material can be measured using the following equation:37
| (αhϑ)1/n = A(hϑ − Eg) | (7) |
The relation between phonon energy (hϑ), optical absorption coefficient (α), power factor of the transition mode (n) and proportionality constant or band tailing parameter (A). Here, Mn3O4 nanoparticles exhibit a direct band gap (n = ½) energy of 2.99 eV as given Fig. 6b. The higher band gap was obtained due to the presence of size defects and it caused a blue shift from the bulk (2.3 eV).38,39
The electrochemical performance of the electrode material can be evaluated using cyclic voltammetry (CV) curves, galvanostatic charge–discharge (GCD), and electrochemical impedance spectroscopic (EIS) techniques. The capacitive behavior of the nickel foam-supported Mn3O4 nanoparticles was directly studied in aqueous 0.5 M Na2SO4 electrolyte solution at room temperature in a three-electrode setup. A potential window of −0.5 V to 0.2 V was investigated for the CV and GCD analysis at different scan rates and current densities, respectively. The three-electrode setup consists of a working electrode (Mn3O4 drop-casted on Ni foam), a reference electrode (Ag/AgCl) and a counter electrode (Platinum).
The cyclic voltammograms of Mn3O4 nanoparticles were observed for different scan rates ranging from 1 mV s−1 to 1000 mV s−1 as shown in Fig. 7a. All the CV curves are identified to be quasi-rectangular shape which exhibits the capacitive kind behaviour in its nature. This property would enhance its electrochemically reversible nature along with faradaic redox reactions. The CV curves also exhibit an ideal electrochemical capacitive behaviour. The mechanism of charge storage during the charging and discharging can be expressed as follows:
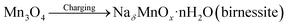 | (8) |
| NaδMnOx·nH2O + yH+ + zNa+ + (y + z)e− ↔ HyNaδ+zMnOx·nH2O | (9) |
 | ||
| Fig. 7 (a and b). Cyclic voltammogram and (c). relationship between specific capacitance and scan rate (mV s−1). | ||
The Na2+ ions in the electrolyte solution move towards the fabricated Mn3O4 electrode and occupy the available sites on the surface and on the inner portion also at lower scan rates. On the other hand, it will be difficult for the ions to occupy the available sites of the Mn3O4 electrode at higher scan rates.40 This is due to the partial movement of ions in the electrolyte solution. The formation of spinel Mn3O4 to layered birnessite can be observed from the first reaction represented by eqn (8), which is an irreversible and complex process. The second reaction eqn (9) confirms the reversibility of the product obtained.3 These two mechanisms occurred due to the adsorption of solvated cations which leads to the lowering of binding energy. The oxidation of Mn2+ to Mn3+ occurs during the cathodic sweep. This will result in a change in the oxidation number of manganese. Also, the reduction of Mn3+ to Mn2+ occurs during the anodic sweep; this, in turn, reduces the average oxidation number of manganese.41 Fig. 7b shows the variation of specific capacitance value with the scan rates. A decrease in the specific capacitance value can be observed on increasing the scan rate. This nature of the electrode material confirms the capacitive behaviour of the fabricated Mn3O4 electrode. From the CV curves, the specific capacitance value was found to be 436 Fg−1 at a scan rate of 1 mV s−1. The nature of charge storage mechanism of the electrode can be analyzed using Dunn's method. From the Power law,
| i = avb | (10) |
log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) i = log i = log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a + b a + b![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) v v
| (11) |
A b-value of 0.5 indicates the faradaic capacitive process and 1.0 corresponds to non-faradaic process. The b-value obtained for Mn3O4 electrode was 0.83, which indicates the pseudocapacitive behaviour (both faradaic and non-faradaic mechanisms coexists) of the Mn3O4 electrode (Fig. 7c). The contribution studies of capacitive or diffusion-controlled nature of electrodes were analyzed from the Dunn's method using the following equations,
| i = k1v + k2v1/2 | (12) |
| i/v1/2 = k1v1/2 + k2 | (13) |
The surface or capacitive-controlled contribution is represented by k1v and k2v1/2 gives the diffusion-controlled contribution.42 Fig. 7d shows the surface-controlled and diffusion-controlled behaviour of Mn3O4 electrode from the cyclic voltammetric analyses.
The charge–discharge mechanism of the Mn3O4 electrode was observed from the galvanostatic charge–discharge method, as shown in Fig. 8a. The charge–discharge curves of Mn3O4 were observed at different current densities in the potential range between −0.5 V to 0.2 V. Fig. 8b shows the variation of the specific capacitance value according to the current densities. A significant reduction in the capacitance with respect to the increase in the current density is due to the potential drop observed in the discharging curves because of the resistance of the electrode. It can also be due to the insufficient redox reactions at the electrode/electrolyte interface at higher current densities.43 The non-linear curves identified from the charge–discharge curves indicate the pseudocapacitive nature of the Mn3O4 electrode.41 The specific capacitance value obtained at a current density of 4 Ag−1 is found to be 163 Fg−1. The redox effect of the Mn3O4 electrode can be analyzed from the GCD curves.
Further investigation of the electrochemical properties was done with electrochemical impedance spectroscopy (EIS) and the equivalent fitted diagram, as given in Fig. 8c. The Nyquist plots describe the ohmic property (Z′) and capacitive property (–Z″). It consists of three regions: high-frequency, middle-frequency, and low-frequency regions. The equivalent circuit diagram consists of the solution resistance (Rs), charge-transfer resistance (Rct), the Warburg diffusion element (W) and the constant phase element (Q). The equivalent series resistance (Rs) is given by the x-intercept of the curve, 4.61 Ω. The interface or charge transfer resistance of the electrode material can be identified from the diameter of the semicircle region of the Nyquist plot which was found to be 0.25 Ω. This semicircle formation occurred due to the faradaic reactions at the electrode/electrolyte interface. The lower the diameter of the semicircle, the lesser the interface resistance and it enhances the utilization of the active electrode surface.21 The linear region obtained in the low-frequency region corresponds to the typical capacitive behavior of the Mn3O4 electrode.44 Fig. S3 (a–c) shows the cyclic voltammogram, charge–discharge curves and Nyquist plot of bare Ni foam under the same electrochemical conditions where the active material was studied. The electrochemical behaviour of the Ni foam is very poor and it could be studied to analyze the enhancement of the capacitive property of the active material (Mn3O4) after drop-casting. The specific capacitance of the Ni foam with an imaginary mass loading of 1 mg is 18.4 Fg−1 at a current density of 4 Ag−1, which is very low compared to the specific capacitance obtained for Mn3O4. The redox peaks were negligible from the CV and GCD curves, revealing the absence of Ni oxidation during the electrochemical processes. The Nyquist plot was also observed in the same electrochemical conditions of Mn3O4 and revealed the difference in the electrochemical behaviour of the active material.
3.1 Prototype aqueous symmetric device studies
The observations from the preliminary studies exhibited a better electrochemical performance of Mn3O4 electrode. A prototype symmetric supercapacitor device was fabricated with an average mass loading of 1 mg per electrode. The device was analyzed in 0.5 M Na2SO4 electrolyte solution in a potential window of 0–1 V. The CV curves obtained for the device was analyzed within a scan rate of 5 mV s−1 to 1000 mV s−1. The redox peaks in CV curves were not clearly evident and the curves are not changing drastically with the increase of scan rates. The reversibility of the faradaic reactions can be examined from Fig. 9a. The charge storage mechanism at lower scan rate allows Na+ ions in the electrolyte get inserted into the available pores on the surface of the electrode and also to the inner sides of Mn3O4. The effective interaction of ions and electrode would be reduced at higher scan rate which decreases the specific capacitance values. | ||
| Fig. 9 Prototype aqueous symmetric supercapacitor device studies with (a). CV, (b). GCD, (c). Ragone plot and (d). Cyclic stability and coulombic efficiency over 1000 cycles. | ||
Capacitive performance can be acquired from the galvanostatic charge–discharge curves (Fig. 9b). As the current density increases, the specific capacitance value decreases (Fig. 9c). The distortion in the linear nature of charge–discharge curves can be due to the high internal resistance between the electrode/electrolyte interface of the device. The discharging process exhibits a higher specific capacitance at lower current densities. The specific capacitance obtained at 1 Ag−1 is 68 Fg−1 with an energy density of 9.34 W h kg−1 and a power density of 500 W kg−1. The device showed a cyclic stability of 72% after 1 k cycles of charging and discharging (Fig. 9d). The coulombic efficiency of the Mn3O4//Mn3O4 device initiates at 92% and gradually it increases and remained consistent at 100% after 850th cycle up to 1000th cycle. Table 1 shows a comparison of symmetric prototype devices with respect to other reports of Mn3O4 and other materials. The energy density of our material is better compared to other reports of Mn3O4 symmetric devices except the work in which Mn3O4 prepared with cocosin through chemical precipitation method.
| Material in device form | Synthesis | Substrate | Current density (Ag−1 or mA cm−2)/Scan rate (mV s−1) | Csp (Fg−1) | ED (W h kg−1) | Ref. |
|---|---|---|---|---|---|---|
| NiO//NiO | Atomic layer deposition | ITO@PET | 10 µA cm−2 | 0.612 mF cm−2 | 0.0544 µ Wh cm−2 | 45 |
| MnO2//MnO2 | Hydrothermal | Nickel foam | 0.5 Ag−1 | 181 | 3.4 | 46 |
| Fe2O3@ACC//Fe2O3@ACC | Hydrothermal | Carbon cloth | 1 mA cm−2 | 1565 mF cm−2 | 0.0092 | 47 |
| MoO3//MoO3 | Electrodeposition | Carbon cloth | 1 Ag−1 | 141 | 78 | 48 |
| V2O5@AC//V2O5@AC | Hydrothermal | Carbon fabric | 2.77 mg cm−2 | 135 | 48.32 | 49 |
| Co3O4//Co3O4 | Hydrothermal | Nickel foam | 5 Ag−1 | 870.6 | 77.3 | 9 |
| ZnO//ZnO | Sol–gel | Cellulose nanofibers | 1 Ag−1 | 220 | 30.2 | 36 |
| Mn3O4//Mn3O4 | Chemical precipitation with cocosin | Nickel foil | 1 Ag−1 | 203.8 | 91.7 | 50 |
| Mn3O4//Mn3O4 | Successive ionic layer adsorption and reaction | Stainless steel | 2 mA cm−2 | 72 | 1.3 | 51 |
| Mn3O4//Mn3O4 | Microwave-assisted chemical route | Stainless steel coin | 1.27 mA cm−2 | 665.08 | 0.0789 | 52 |
| Mn3O4//Mn3O4 | Hydrothermal | Nickel foam | 1 Ag−1 | 68 | 9 | Present work |
The electrochemical impedance spectroscopy (EIS) studies observed the solution resistance and charge transfer resistance in a frequency range of 1 Hz to 1 M Hz (Fig. 10). The solution resistance (Rs) is found to be 5 Ω and a charge transfer resistance (Rct) is 0.35 Ω were identified from the fitted circuit diagram. The linear variation at the low-frequency region in the Nyquist plot determines the Warburg resistance, which is observed due to the ion diffusion. It is one of the significant properties of supercapacitor applications.53
4. Conclusion
Hausmannite Mn3O4 nanoparticles were synthesized at a low temperature by hydrothermal method. The obtained Mn3O4 nanoparticles showed a tetragonal spinel structure confirmed by the XRD and Raman spectrum analyses. Mn2+ and Mn3+ oxidation states were observed from the Mn 2p spectrum through XPS. The morphological analysis found a mixture of nanocubes and nanorods in the as-synthesized Mn3O4 nanoparticles. The optical band gap was observed near 2.99 eV. The investigation of the electrochemical performance was observed from the cyclic voltammetry curves, galvanostatic charging/discharging curves and impedance plots. A prototype aqueous symmetric supercapacitor was fabricated to study the device-level performance in a working potential window of 1 V. The device exhibited a specific capacitance of 63 Fg−1 at 1 Ag−1 with an energy density of 9 W h kg−1 and power density of 500 W kg−1.Author contributions
Chrisma Rose Babu: conceptualization, methodology, validation, formal analysis, investigation, writing – original draft, writing – review and editing. E. I. Anila: methodology, writing – original draft, writing – review and editing, supervision, validation. A. V. Avani: methodology, writing – review and editing. Roshan Jose: formal analysis and writing. Xavier T. S: formal analysis and data curation.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
The data supporting the findings of this study are available within the article and its supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5ra08702c.Acknowledgements
This research work was supported by Centre for Advanced Research and Development (CARD), CHRIST (Deemed to be University), Bangalore, Karnataka.References
- D. P. Dubal, D. S. Dhawale, R. R. Salunkhe, S. M. Pawar and C. D. Lokhande, Appl. Surf. Sci., 2010, 256, 4411–4416 Search PubMed.
- P. Suktha, N. Phattharasupakun, P. Dittanet and M. Sawangphruk, RSC Adv., 2017, 7, 9958–9963 Search PubMed.
- B. Gnana Sundara Raj, A. M. Asiri, J. J. Wu and S. Anandan, J. Alloys Compd., 2015, 636, 234–240 Search PubMed.
- R. B. Chrisma, R. I. Jafri and E. I. Anila, J. Mater. Sci., 2023, 58, 6124–6150 CrossRef CAS.
- T. Kansaard, T. Singha, S. Wannapop, A. Boonkitkoson, Y. Chuminjak, J. Sodtipinta and A. Somdee, J. Alloys Compd., 2025, 1010, 178185 CrossRef CAS.
- Z. S. Doroudkhani, J. Mazloom and M. M. Ghaziani, Sci. Rep., 2025, 15, 1–17 Search PubMed.
- B. J. Rani, M. Ravina, G. Ravi, S. Ravichandran, V. Ganesh and R. Yuvakkumar, Surf. Interfaces, 2018, 11, 28–36 Search PubMed.
- R. Ge and L. Chen, Adv. Compos. Hybrid Mater., 2022, 5, 696–703 CrossRef CAS.
- C. R. Babu, A. V Avani, T. S. Xavier, M. Tomy, S. Shaji and E. I. Anila, J. Energy Storage, 2024, 80, 110382 Search PubMed.
- X. Huang, Z. Yin, S. Wu, X. Qi, Q. He, Q. Zhang, Q. Yan, F. Boey and H. Zhang, Small, 2011, 7, 1876–1902 Search PubMed.
- L. Wang, L. Gao, J. Wang and Y. Shen, J. Mater. Sci., 2019, 54, 13685–13693 CrossRef CAS.
- E. Yu, Y. M. Kang and W. D. Yang, Electrochim. Acta, 2025, 513, 145520 CrossRef CAS.
- H. Jiang, T. Zhao, C. Yan, J. Ma and C. Li, Nanoscale, 2010, 2, 2195–2198 Search PubMed.
- S. Jamil, S. R. Khan, B. Sultana, M. Hashmi, M. Haroon and M. R. S. A. Janjua, J. Cluster Sci., 2018, 29, 1099–1106 Search PubMed.
- V. C. Bose, K. Maniammal, G. Madhu, C. L. Veenas, A. S. A. Raj and V. Biju, IOP Conf. Ser. Mater. Sci. Eng., 2015, 73, 012084 CrossRef.
- K. A. M. Ahmed and K. Huang, Arab. J. Chem., 2019, 12, 429–439 Search PubMed.
- M. Shahid, T. R. Katugampalage, M. Khalid, W. Ahmed, C. Kaewsaneha, P. Sreearunothai and P. Opaprakasit, Sci. Rep., 2022, 12, 1–16 Search PubMed.
- V. C. Bose and V. Biju, Phys. E, 2015, 66, 24–32 Search PubMed.
- B. G. S. Raj, R. N. R. Ramprasad, A. M. Asiri, J. J. Wu and S. Anandan, Electrochim. Acta, 2015, 156, 127–137 CrossRef CAS.
- D. P. Shaik, R. Pitcheri, Y. Qiu and O. M. Hussain, Ceram. Int., 2019, 45, 2226–2233 Search PubMed.
- Z. Wang, J. Fang, Y. Hao, C. Chen and D. Zhang, Mater. Sci. Semicond. Process., 2021, 130, 105823 CrossRef CAS.
- K. Prasad, T. V. M. Sreekanth, K. Yoo and J. Kim, Vacuum, 2024, 221, 112930 CrossRef CAS.
- C. R. Babu, E. I. Anila and A. V Avani, Fuel, 2026, 406, 137225 CrossRef CAS.
- H. Dhaouadi, A. Madani and F. Touati, Mater. Lett., 2010, 64, 2395–2398 CrossRef CAS.
- A. Rizky, A. Fadhillah, Irzaman and Irmansyah, J. Phys., Conf. Ser., 2021, 2019, 012070 CrossRef CAS.
- D. P. M. D. Shaik, P. Rosaiah and O. M. Hussain, Mater. Today Proc., 2016, 3, 64–73 CrossRef.
- T. Larbi, K. Doll and T. Manoubi, J. Alloys Compd., 2016, 688, 692–698 CrossRef CAS.
- Z. Y. Tian, P. Mountapmbeme Kouotou, N. Bahlawane and P. H. Tchoua Ngamou, J. Phys. Chem. C, 2013, 117, 6218–6224 CrossRef CAS.
- Y. Luo, W. Tan, S. L. Suib, G. Qiu and F. Liu, Chem. Geol., 2018, 487, 54–62 CrossRef CAS.
- M. Jarvin, S. A. Kumar, G. Vinodhkumar, E. Manikandan and S. S. R. Inbanathan, Mater. Lett., 2021, 305, 130750 CrossRef CAS.
- Z. Ren, W. Wu, L. Yu and Y. Yu, Environ. Sci. Pollut. Res., 2019, 26, 36776–36785 CrossRef CAS PubMed.
- B. Gnana Sundara Raj, A. M. Asiri, J. J. Wu and S. Anandan, J. Alloys Compd., 2015, 636, 234–240 CrossRef CAS.
- A. Moses Ezhil Raj, S. G. Victoria, V. B. Jothy, C. Ravidhas, J. Wollschläger, M. Suendorf, M. Neumann, M. Jayachandran and C. Sanjeeviraja, Appl. Surf. Sci., 2010, 256, 2920–2926 CrossRef CAS.
- V. C. Bose and V. Biju, Bull. Mater. Sci., 2015, 38, 865–873 CrossRef CAS.
- R. Aswathy, M. Ulaganathan and P. Ragupathy, J. Alloys Compd., 2018, 767, 141–150 Search PubMed.
- I. Rabani, J. Yoo, C. Bathula, S. Hussain and Y. S. Seo, J. Mater. Chem. A, 2021, 9, 11580–11594 Search PubMed.
- B. Jos, C. R. Babu, S. Shaji and E. I. Anila, J. Mater. Sci. Mater. Electron., 2024, 35, 233 Search PubMed.
- N. M. Hosny and A. Dahshan, Mater. Chem. Phys., 2012, 137, 637–643 CrossRef CAS.
- R. Vignesh, R. Sivakumar and C. Sanjeeviraja, Opt. Mater., 2021, 111, 110580 CrossRef CAS.
- F. Zhang, X. G. Zhang and L. Hao, Mater. Chem. Phys., 2011, 126, 853–858 CrossRef CAS.
- S. G. Sayyed, H. M. Pathan, A. V. Shaikh, S. F. Shaikh and A. M. Al-Enizi, J. Energy Storage, 2021, 33, 102076 CrossRef.
- Y. Shao, M. F. El-Kady, J. Sun, Y. Li, Q. Zhang, M. Zhu, H. Wang, B. Dunn and R. B. Kaner, Chem. Rev., 2018, 118, 9233–9280 CrossRef CAS PubMed.
- D. Li, F. Meng, X. Yan, L. Yang, H. Heng and Y. Zhu, Nanoscale Res. Lett., 2013, 8, 1–8 Search PubMed.
- A. A. Yadav, S. N. Jadhav, D. M. Chougule, P. D. Patil, U. J. Chavan and Y. D. Kolekar, Electrochim. Acta, 2016, 206, 134–142 CrossRef CAS.
- T. Kavinkumar, A. T. Sivagurunathan and D.-H. Kim, Appl. Surf. Sci., 2023, 616, 156453 CrossRef CAS.
- R. S. Kalubarme, H. S. Jadhav and C. J. Park, Electrochim. Acta, 2013, 87, 457–465 Search PubMed.
- J. Li, Y. Wang, W. Xu, Y. Wang, B. Zhang, S. Luo, X. Zhou, C. Zhang, X. Gu and C. Hu, Nano Energy, 2019, 57, 379–387 CrossRef CAS.
- N. Zhao, H. Fan, M. Zhang, J. Ma, Z. Du, B. Yan, H. Li and X. Jiang, Chem. Eng. J., 2020, 390, 124477 CrossRef CAS.
- K. Panigrahi, P. Howli and K. K. Chattopadhyay, Electrochim. Acta, 2020, 337, 135701 CrossRef CAS.
- J. Yesuraj, S. Vajravijayan, R. Yang, N. Nandhagopal, K. Gunasekaran, N. C. S. Selvam, P. J. Yoo and K. Kim, Langmuir, 2022, 38, 2928–2941 Search PubMed.
- D. P. Dubal, A. D. Jagadale and C. D. Lokhande, Electrochim. Acta, 2012, 80, 160–170 CrossRef CAS.
- V. C. Bose and V. Biju, Bull. Mater. Sci., 2015, 38, 865–873 Search PubMed.
- C. R. Babu, A. V. Avani and E. I. Anila, Int. J. Hydrogen Energy, 2025, 109, 242–253 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |





