 Open Access Article
Open Access ArticleSustainable metal recovery from waste printed circuit boards through citric acid leaching using response surface methodology and predictive modelling
Syed Suffia Iqbala,
Ovais Iqbal Shahb and
Deblina Dutta *a
*a
aDepartment of Environmental Science and Engineering, School of Engineering and Sciences, SRM University-AP, Amaravati, Andhra Pradesh-522240, India. E-mail: deblina.d@srmap.edu.in
bDepartment of Computer Science and Engineering, Indian Institute of Technology Jammu, Jammu-181221, India
First published on 19th February 2026
Abstract
Recycling printed circuit boards (PCBs) is crucial for reducing pollution, recovering valuable metals, and conserving resources. This study presents a green method for metal extraction from mobile phone PCBs using citric acid (C6H8O7). The dissolution of Ag, Al, and Zn was examined in relation to variables such as pulp density, time, citric acid concentration, and temperature. The findings demonstrate the leaching of Ag, Al, and Zn is approximately 93%, 91% and 92%, respectively, under conditions of 1 M citric acid, a pulp density of 70 g L−1, a temperature of 75 °C, and a mixing time of 120 minutes. Response surface methodology was employed to optimize the conventional hydrometallurgical method. Thus, utilizing citric acid as a green solvent not only enhances leaching efficiency but also offers a more sustainable alternative for the recovery of valuable metals from mobile PCBs. To further enhance prediction accuracy, multiple machine learning models were evaluated using process data. Among these, polynomial regression achieved the best performance (R2 = 1.000; RMSE ≈ 0), effectively capturing nonlinear relationships between parameters and silver recovery. This integration of experimental optimization and data driven modeling confirms citric acid as a sustainable lixiviant for efficient silver recovery from PCBs, reducing reliance on inorganic acids and environmental impact. It also demonstrates the effectiveness of polynomial modeling in minimizing experimental effort and enhancing leaching efficiency.
1. Introduction
Technological advancements have significantly driven societal transitions from traditional electrical and electronic equipment (EEE) to modern alternatives with enhanced, multifunctional capabilities.1 EEE that is no longer functional or useful is commonly referred to as electronic waste, or E-waste.2 The rising global demand for metals presents a major challenge, especially considering the declining quality of natural ore deposits. Remarkably, E-waste can contain up to 26 times more Cu and up to 50 times more Au than conventional ores and concentrates.3 Steady technological innovation and socioeconomic development have driven an unprecedented surge in waste printed circuit boards (PCBs), exposing the hidden environmental surge of digitalization and inadequate waste management. Among the various E-waste components, PCBs are especially noteworthy as they contain considerable quantities of valuable and base metals.4,5 PCBs represent a metal rich secondary resource; however the present study specifically investigates the selective leaching behavior of Ag, Al and Zn using citric acid,6 as Ag contributes to the economic values of PCBs while Al and Zn are common co-dissolving base metals that influence selectivity and hydrometallurgical processing. PCBs are integral components of most electronic devices and are widely studied in consumer electronics. They account for approximately 4–7% of the total E-waste stream.7,8Typical EEEs include household and communication devices such as computers, televisions, and mobile phones. Highlighting the economic potential of E-waste as a secondary resource, recent studies estimated its total value at about USD $57 billion.9 Discarded PCBs can be regarded as an “urban mine” for the extraction of valuable metals. However, conventional disposal practices such as open dumping, incineration, and landfilling lead to the generation of hazardous byproducts and toxic gaseous emissions, creating significant risks to human wellbeing, the environment, and the goal of sustainable development.10 Therefore, the proper management of E-waste based on sound knowledge of its generation, collection and proper disposal can play a key role in environmental protection and the conservation of natural resources. As a result, many researchers are now focusing on the recycling and reuse of E-waste. Among the various recovery techniques, chemical leaching has appeared as a key technique for extracting valuable metals from waste batteries and PCBs, thereby contributing to the prevention of environmental pollution.11 Established recycling methodologies include hydrometallurgy, pyrometallurgy and direct recycling approaches, which are actively employed in PCBs metal recovery processes.12,13
The implementation of hydrometallurgical approaches for the recovery of metals from discarded PCBs is significantly more advantageous than alternative methods, due to its superior selectivity, reduced financial expenditure, lower energy consumption, and elevated recovery efficiencies.14 In contrast, pyrometallurgical methods are associated with numerous disadvantages, including elevated gaseous emissions, significant capital expenditure, excessive energy utilization, and the financial burden related to the extraction of metals from slag. To optimize parameters for effective recovery and feasibility, the RSM model was employed across various scenarios. Compared with conventional optimization techniques, RSM requires fewer experimental runs, significantly reduces experimental time, and enables the identification of optimal conditions through response surface plots. In this study, a Central Composite Design (CCD) was applied to evaluate the interaction effects of key process variables. Within hydrometallurgical processing, the selection of an appropriate leaching agent plays a critical role in determining metal dissolution efficiency, process sustainability, and environmental impact.15 Conventional inorganic acids, although effective, often pose challenges related to toxicity, corrosiveness, and secondary pollution. Consequently, increasing attention has been directed towards organic acids as green alternatives due to their biodegradability, lower toxicity, and reduced environmental footprint. Among the various organic acids, citric acid (C6H8O7) has emerged as a particularly promising leaching agent because of its strong metal-chelating capability, wide availability, and cost-effectiveness. Previous studies have reported recovery efficiencies for Ag and other valuable metals using citric acid, especially when combined with oxidizing agents such as hydrogen peroxide, thereby supporting its selection in the present study.16,17
Despite growing interest, most hydrometallurgical studies on PCBs metal recovery still rely on strong mineral acids such as H2SO4, HNO3, or aqua regia, which generate toxic fumes and hazardous residues. Only limited research has explored biodegradable organic acids like citric acid as sustainable leaching agents, and many of these studies lack systematic optimization or predictive evaluation. Beyond organic acids, emerging solvent systems such as ionic liquids (ILs) and deep eutectic solvents (DESs) are being explored for metal recovery. ILs have shown strong selectivity and high recovery efficiencies for metals from E-waste.18 DESs are prepared by mixing a hydrogen-bond acceptor and a hydrogen-bond donor.19 They are increasingly explored due to their relatively low toxicity and potential biodegradability. Acidic DESs formulations often based on choline chloride combined with carboxylic acids have been reported to recover both base and precious metals from PCBs, with efficiencies commonly above 80% and in some cases reaching 99%. Overall, ILs and DESs are promising green alternatives for sustainable hydrometallurgical metal recovery and can complement organic acids such as citric acid in eco-friendly recycling approaches.20 Furthermore, although RSM and Machine Learning (ML) have been individually applied for process optimization and prediction, their integrated application remains underexplored.
This study presents an environmentally sustainable and data driven framework for the metal recovery from the waste mobile phone PCBs. RSM has been used to optimize key variables (pulp density, temperature, time and citric acid concentration) for Ag, Al and Zn recovery, and polynomial regression has been applied to predict recovery outcomes. Using citric acid as the leaching agent supports green chemistry principles by reducing environmental footprint of the overall process. ML models (polynomial regression, random forest, and XGBoost) were evaluated using R2, RMSE and cross validation with polynomial regression showing the best predictive performance. The framework supports green chemistry and circular economy goals while enabling scalable optimization to reduce time, cost and reagent use.
2. Experimental section
2.1. Material preparation
The mobile phones of different brands and models were collected from the households and from shops dealing with discarded/second-hand mobile phones. The devices were dismantled manually, and the PCBs were separated, cleaned to remove adhering dust/plastics, dried, and then mechanically ground, and their size was reduced using scissors and pliers. They were then cut into small pieces, followed by comminution and grinding to obtain a finely ground powder (≤500 µm) suitable for further analysis. The resulting powder samples were subsequently used for the leaching experiments.2.2. Characterization studies
A thorough characterization was performed to understand the material properties and behavior of the mobile phone PCBs. Studies for the characterization of the mobile phone PCBs were carried out using XRD (make: Malvern Panalytical Ltd; model: Empyrean; SRM University-AP, Andhra Pradesh, Guntur). ICP-OES (make: Thermo Fisher Scientific; model: ICAPPROX DUO; SRM University-AP, Andhra Pradesh, Guntur) was used for the analysis of the metal concentration. All experiments used analytical grade chemicals to ensure reliability.2.3. Leaching procedure
The metals were extracted from the PCBs using a three necked Pyrex glass reactor, fitted with a condenser to capture any gases produced during the leaching process. A hot plate with a magnetic stirrer (Accumax, India, Model-I Stir HP 320) was utilized to maintain consistent agitation to achieve the required temperature during the experiment. For preliminary leaching investigations, multiple leaching agents, including nitric acid, oxalic acid, citric acid, and glycine, were employed under controlled leaching conditions, specifically at a leachant concentration of 1 M, maintained at a temperature of 75 °C, a pulp density of 70 g L−1 of PCBs powder, for a duration of 120 minutes. Furthermore, the influence of time, pulp density, temperature, acid concentration, and various other process variables were systematically examined. After completion of the leaching experiment, the slurry was allowed to cool to room temperature, and the solid–liquid separation was carried out by filtration. The collected filtrate (leach liquor) was used for metal analysis. The leach liquor was analyzed using Inductively Coupled Plasma-Optical Emission Spectroscopy (ICP-OES) to determine the concentrations of dissolved metals.The leaching effectiveness of the experimental process was determined in eqn (1).21
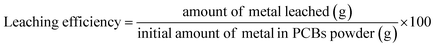 | (1) |
2.4. Experimental design and optimization
The design of experiments (DOE) constitutes a powerful methodology applicable across a multitude of experimental contexts.22 RSM represents a modeling technique within the DOE framework, employed to evaluate the significance of variations in parameters by integrating empirical quantitative data with mathematical and statistical models,23 thereby enhancing the optimization of parameters. The enhancement of the leaching procedure was demonstrated through the application of RSM. This methodology integrates both quantitative and qualitative analytical techniques for the examination of a process response, which is influenced by a multitude of parameters. The experimental design utilized in RSM diminishes the requisite number of trials in comparison to traditional methodologies, thereby facilitating the identification of the impact of various parameters on the response variable.24 In the present study, a three-level coded RSM based on a CCD was employed, where each variable temperature, pulp density, citric acid concentration and leaching time was evaluated at three coded levels (−1, 0, and +1) to optimize the leaching conditions for silver recovery from mobile phone PCBs. Minitab software was employed to perform experimental design, in which 31 experimental sets were executed to examine the impact of four independent variables on the response variable, specifically the metal leaching efficiency (dependent variable). All leaching experiments were performed in triplicate under identical conditions to ensure reproducibility.2.5. Machine learning for yield prediction
The extraction of precious metals from E-waste represents both an environmental necessity and a significant economic opportunity. Among these metals, Ag is a particularly important target due to its extensive use in electronic components and higher market value. This study focuses on predicting Ag recovery efficiency (%) from leaching experiments using ML techniques.To enhance empirical optimization and minimize experimental effort, ML frameworks were developed to forecast metals recovery efficiency as a function of these process variables.25 The modeling framework incorporated the four critical input variables pulp density, temperature, acid concentration, and leaching duration while the output variable represented the experimentally measured Ag recovery (%). The effectiveness of multiple regression-based ML models was assessed using four statistical indicators: coefficient of determination (R2), mean absolute error (MAE), root mean square error (RMSE), and cross-validated R2 (CV R2). These metrics were used to evaluate both the reliability and generalization performance of the predictive models.
3. Results and discussion
3.1. Characterization of PCBs
For characterization, XRD analysis was performed on the PCBs powder to identify its phase composition. The XRD pattern of the PCBs powder is shown in Fig. 1, and the diffraction peaks indicate additional reflections attributable to Al, Ag, and Zn containing phases.To determine the initial metal content required for leaching-efficiency calculations, the aqua regia was used for the digestion of 1 g of sample using solid liquid ratio of about 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 and leaching time of 2 h at 60 °C.26,27 Following the necessary dilution, the leached samples of the PCBs were analyzed by the ICP-OES. The initial metal concentrations measured in the PCBs powder were Al = 10.2 g kg−1, Zn = 31.9 g kg−1, Ag = 2.9 g kg−1, Cu = 387 g kg−1 and Fe = 34.3 g kg−1.
20 and leaching time of 2 h at 60 °C.26,27 Following the necessary dilution, the leached samples of the PCBs were analyzed by the ICP-OES. The initial metal concentrations measured in the PCBs powder were Al = 10.2 g kg−1, Zn = 31.9 g kg−1, Ag = 2.9 g kg−1, Cu = 387 g kg−1 and Fe = 34.3 g kg−1.
3.2. Selection of the leaching agent
The leaching agent or solvent is a crucial factor in metal extraction, as it largely governs the effectiveness and mechanism of the leaching process. In this study, hydrometallurgical processing was employed to extract Ag from the PCBs of mobile phones. The selection of an appropriate leaching agent is vital for achieving optimal metal recovery and relies on several factors, including the selectivity, reagent cost, physicochemical properties of the material, and regeneration potential.28 To identify the most effective leaching agent, experiments were conducted using nitric acid, citric acid, oxalic acid, and glycine under various conditions: 0.5 M leachant concentration, 50 g L−1 pulp density, 75 °C temperature, and 120 minutes duration. Analytical grade reagents were used throughout the experiments. The results (Fig. 2(a)) revealed that citric acid and nitric acid exhibited the highest metal dissolution efficiencies for Ag, Al, and Zn, achieving over 80–95%, while glycine and oxalic acid showed comparatively lower efficiencies. Among these, citric acid was identified as the most promising green solvent for metal recovery. Being a mild, triprotic organic acid, it effectively complexes with metal ions such as Ag+, Al3+, and Zn2+, promoting their dissolution while minimizing corrosion and environmental risks.Furthermore, the integration of citric acid and hydrogen peroxide (H2O2) enhanced leaching kinetics, achieving rapid extraction compared to bioleaching methods that typically require several days.29 Owing to its biodegradable and non-toxic nature, citric acid adheres to the fundamentals of green chemistry principles, making it an environmentally friendly and economically viable alternative to conventional inorganic acids. However, precious studies indicate that excessively high organic acid concentrations can reduce leaching efficiency.30,31
3.3. Effect of pulp density
Pulp density exerts a considerable influence on the efficiency of metal leaching from PCBs of mobile phones. To examine this effect, experiments were conducted by varying the pulp density between 40 g L−1 to 100 g L−1, while maintaining constant conditions of 1 M citric acid concentration, 75 °C temperature, and 120 minutes duration. As shown in Fig. 2(b), lower pulp density (40–60 g L−1) resulted in higher leaching efficiencies for Al, Zn, and Ag attaining 74.40%, 64.18%, and 63.99%, respectively. The dissolution of metals increased progressively to a pulp density of 70 g L−1, beyond which a decline was detected. At elevated pulp densities (up to 100 g L−1), the leaching efficiencies of Al, Zn, and Ag decreased to 77.29%, 70.60%, and 73.63%, respectively. This reduction in metal dissolution at higher pulp densities can be attributed to particle agglomeration in the PCBs powder, which hinders the mass transfers of reactants and limits the effective contact between the leachant and metals bearing phases. According to the experimental results, the optimum pulp density was established at 70 g L−1 for maximizing metal solubilization, as it provides sufficient solid liquid interaction without causing excessive particle aggregation. Similar trends, where pulp density exhibits an inverse relationship with metal extraction efficiency, have also been reported in previous studies.32 However, an increase in the pulp density resulted in a marked decrease in metal leaching efficiency.333.4. Effect of temperature
Temperature is a key factor influencing the solubilization of metallic components during the leaching stage. The influence of temperature was analyzed while maintaining constant reaction parameters, including a 1 M citric acid concentration, a pulp density of 70 g L−1, and a leaching duration of 120 minutes. Increasing the temperature from 45 °C to 70 °C enhanced the reaction rate, diffusivity, and mass transfer efficiency, thereby improving metal dissolution and overall leaching efficiency. As the system's temperature increased within the range of 40 °C to 60 °C, the dissolution of all metals improved, reaching approximately 70–80%. At temperatures above 70 °C, the dissolution of Ag reached up to 92.5%, however, a vigorous reaction was observed, and no further significant improvement in leaching efficiency occurred. This may be attributed to the partial degradation or volatilization of the solvent at elevated temperatures. Therefore, considering both leaching efficiency and energy consumption, a temperature of 75 °C was identified as the optimal condition for Al, Zn, and Ag leaching, as shown in Fig. 2(c). The solubility of metallic elements is profoundly affected by thermal conditions.34,35 Meanwhile, rising temperatures enhance molecular kinetic energy, resulting in more collisions and quicker reaction rates. This, in turn, facilitates the diffusion of acid ions and other reactants toward the reaction interface, as well as the transfer of dissolved products into the leaching solution, ultimately improving the overall leaching efficiency.36,373.5. Effect of time
Leaching time is an essential parameter governing the extent of metal extraction. The efficiency of metal recovery is strongly influenced by leaching time, as it directly influences the degree of metal dissolution and overall leaching performance. The effect of time was observed as the duration increased from 60 to 180 minutes, the dissolution of metals increased from 20% to 90% as exhibited in Fig. 2(d). During the initial phase of leaching, the extraction efficiency of all three metals (Al, Zn, and Ag) increased sharply, indicating that metal dissolution is strongly time-dependent at this stage due to the high concentration gradient and the availability of abundant reactive surface sites. As no substantial variation in the dissolution of metals was observed between the intervals of 120 to 180 minutes, it was thus determined that a duration of 120 minutes is optimal for achieving maximal metal dissolution. Extending the leaching time beyond 120 minutes would therefore not only be inefficient but may also increase operational costs without improving recovery efficiency. Leaching time has a significant influence on the dissolution of metals.343.6. Effect of citric acid concentration
The influence of citric acid concentration on metal leaching was examined over a series of 0.5–2 M, as illustrated in Fig. 2(e). The concentration of citric acid significantly influences the efficiency of the leaching process, however, raising the concentration beyond the optimum level yielded no significant enhancement in metal recovery from PCBs.38 Citric acid has been reported to extract numerous metals, including Cu, Ni, Sn, Zn, Fe, Pb, Au, Al, Ag, Pd.39,40 The optimum acid concentration was determined to be 1 M, which provided the highest metal dissolution efficiency under the given conditions, achieving more than 90% extraction for all three metals. Higher concentrations did not enhance leaching performance, while lower concentrations led to reduced metal recovery. Therefore, 1 M citric acid concentration is considered both effective and economically viable for the extraction process. When used as a leaching agent, citric acid facilitates metal dissolution through the acidification (proton-promoted dissolution), oxidant assisted dissolution in the presence of H2O2 and stabilization of the dissolved metal ions via citrate complexation.Citric acid (H3Cit) is a triprotic ligand and dissociates in water to form citrate species (HCit2− and Cit3−), supplying protons (H+) for acidity and citrate ions that act as complexing ligands.
| H3Cit ⇌ H+ + H2Cit− ⇌ 2H+ + HCit2− ⇌ 3H+ + Cit3− | (2) |
In presence of H2O2, the target metals (Ag, Al, and Zn) are oxidized and dissolve, producing their corresponding ions in the aqueous phase as metal ions (eqn (3)–(5)).
| 2Ag(s) + H2O2 + 2H+ → 2Ag+ + 2H2O | (3) |
| Zn(s) + H2O2 + 2H+ → Zn2+ + 2H2O | (4) |
| 2Al(s) + 3H2O2 + 6H+ → Zn2+ + 2H2O | (5) |
Citrate ions coordinate with the dissolved metal ions, stabilizing them in solution, enhancing their solubility, and preventing re-precipitation during leaching.
| Zn2+ + Cit3− ⇌ [ZnCit]− | (6) |
| Al3+ + Cit3− ⇌ [AlCit]0 | (7) |
| Ag+ + Cit3− ⇌ [AgCit]2− | (8) |
This accounts for the higher leaching observed with 1 M citric acid + 5% H2O2 as H2O2 promotes oxidation and dissolution to ionic species, while citrate complexes stabilize Ag+, Zn2+, and Al3+ in solution, thereby minimizing re-precipitation and favors continued dissolution. At higher reagent concentrations, the leaching efficiency decreased, consistent with previous reports.32,41
4. Leaching study using response surface methodology
The empirical data attained through RSM were evaluated using analysis of variance (ANOVA) to ascertain the importance of the independent variables and the overall regression model concerning the responsible variable. The statistical significance was evaluated through key parameters such as the coefficient of determination (R2), adjusted R2, F-statistics, p-values, and lack-of-fit tests. The ANOVA study additionally examined the interaction effects among the four independent variables and their individual contributions to the response variable. The high R2 value indicated the adequacy and reliability of the second order polynomial regression model. Subsequently, optimization was carried out using the empirical data derived from the DOE and the RSM, focusing on the percentage of Ag dissolution presented in Table 1. Based on the observed responses corresponding to the regression coefficients of the second-order polynomial equation, it was established that the dissolution behavior of Ag with respect to polynomial equation, temperature, acid concentration, pulp density, and time followed a quadratic relationship, as confirmed by the analytical software.| StdOrder | x1 | x2 | x3 | x4 | Yield (observed) | Yield (predicted) |
|---|---|---|---|---|---|---|
| a x1: pulp density, x2: temperature, x3: acid concentration, x4: time. | ||||||
| 1 | 40 | 45 | 0.5 | 60 | 45.38 | 50.38 |
| 2 | 100 | 45 | 0.5 | 60 | 48.13 | 48.45 |
| 3 | 40 | 105 | 0.5 | 60 | 50.32 | 48.81 |
| 4 | 100 | 105 | 0.5 | 60 | 53.83 | 50.02 |
| 5 | 40 | 45 | 1.5 | 60 | 55.97 | 53.67 |
| 6 | 100 | 45 | 1.5 | 60 | 59.15 | 58.10 |
| 7 | 40 | 105 | 1.5 | 60 | 60.32 | 58.07 |
| 8 | 100 | 105 | 1.5 | 60 | 63.65 | 65.64 |
| 9 | 40 | 45 | 1.5 | 180 | 68.53 | 67.71 |
| 10 | 100 | 45 | 0.5 | 180 | 69.39 | 69.47 |
| 11 | 40 | 105 | 0.5 | 180 | 65.12 | 63.99 |
| 12 | 100 | 105 | 0.5 | 180 | 65.42 | 68.89 |
| 13 | 40 | 45 | 1.5 | 180 | 62.34 | 63.97 |
| 14 | 100 | 45 | 1.5 | 180 | 69.41 | 72.09 |
| 15 | 40 | 105 | 1.5 | 180 | 65.37 | 66.22 |
| 16 | 100 | 105 | 1.5 | 180 | 84.65 | 77.48 |
| 17 | 40 | 75 | 1 | 120 | 83.45 | 83.98 |
| 18 | 100 | 75 | 1 | 120 | 85.17 | 88.65 |
| 19 | 70 | 45 | 1 | 120 | 86.43 | 80.90 |
| 20 | 70 | 105 | 1 | 120 | 73.27 | 82.81 |
| 21 | 70 | 75 | 0.5 | 120 | 70.64 | 69.03 |
| 22 | 70 | 75 | 1.5 | 120 | 69.36 | 74.97 |
| 23 | 70 | 75 | 1 | 60 | 70.52 | 74.11 |
| 24 | 70 | 75 | 1 | 180 | 88.28 | 88.69 |
| 25 | 70 | 75 | 1 | 120 | 90.18 | 86.71 |
| 26 | 70 | 75 | 1 | 120 | 92.32 | 86.71 |
| 27 | 70 | 75 | 1 | 120 | 90.64 | 86.71 |
| 28 | 70 | 75 | 1 | 120 | 87.26 | 86.71 |
| 29 | 70 | 75 | 1 | 120 | 83.26 | 86.71 |
| 30 | 70 | 75 | 1 | 120 | 85.62 | 86.71 |
| 31 | 70 | 75 | 1 | 120 | 89.73 | 86.71 |
Additionally, a second-order polynomial model (eqn (9)) was employed to predict the output response for Ag leaching.
| Ag recovery (%) = −40.2 − 0.093x1 + 0.717x2 + 115.8x3 + 0.521x4 − 0.00044x1 × x1 − 0.00540x2 × x2 − 58.8x3 × x3 − 0.001475x4 × x4 + 0.00087x1 × x2 + 0.1060x1 × x3 + 0.000512x1 × x4 + 0.0994x2 × x3 − 0.000299x2 × x4 − 0.0586x3 × x4 | (9) |
To examine the predictive accuracy of the model, an ANOVA was performed. The (R2) coefficient of the determination was utilized to assess the adequacy and reliability of the quadratic polynomial regression model. The p-value for each model coefficient was obtained through significance testing to determine their statistical relevance. The regression model's elevated F-values and low p-values (p < 0.001), together with a non-significant lack of fit, confirm its statistical significance and demonstrate that it accurately represents the experimental data.42
The ANOVA results for the quadratic response surface model of Ag dissolution are presented in Table 2. The overall model for Ag dissolution yielded a p-value of 0.000, which is beneath the statistical significance level (α = 0.05). This indicates a statistically significant relationship between the independent variables and the response, thus rejecting the null hypothesis. The full quadratic model, incorporating four factors, was found to significantly influence the percentage of Ag dissolution. Additionally, the linear terms of all variables showed p-values below 0.05, confirming their significant individual effects on Ag leaching. The model also shows no significant lack-of-fit, as indicated by a p-value of 0.078, which crosses the threshold of 0.05 suggesting the model fits the data well. A high R2 value of 0.93 reflects strong predictive ability and a strong consistency between predicted and actual values Ag dissolution values. Higher R2 values indicate a stronger association between the observed and predicted results.43,44
| Analysis of variance | |||||
|---|---|---|---|---|---|
| Source | DF | Adj SS | Adj MS | F-Value | p-Value |
| a DF = Degrees of Freedom; SS = Sum of Squares; MS = Mean Squares. | |||||
| Model | 14 | 5465.28 | 390.38 | 15.78 | 0 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Linear Linear |
4 | 1230.14 | 307.53 | 12.43 | 0 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Pulp density Pulp density |
1 | 98 | 98 | 3.96 | 0.064 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Temperature Temperature |
1 | 16.47 | 16.47 | 0.67 | 0.426 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Acid concentration Acid concentration |
1 | 158.78 | 158.78 | 6.42 | 0.022 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Time Time |
1 | 956.89 | 956.89 | 38.68 | 0 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Square Square |
4 | 4081.63 | 1020.41 | 41.25 | 0 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Pulp density × pulp density Pulp density × pulp density |
1 | 0.41 | 0.41 | 0.02 | 0.899 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Temperature × temperature Temperature × temperature |
1 | 61.28 | 61.28 | 2.48 | 0.135 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Acid concentration × acid concentration Acid concentration × acid concentration |
1 | 561.49 | 561.49 | 22.7 | 0 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Time × time Time × time |
1 | 73.15 | 73.15 | 2.96 | 0.105 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2-Way interaction 2-Way interaction |
6 | 153.51 | 25.59 | 1.03 | 0.439 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Pulp density × temperature Pulp density × temperature |
1 | 9.86 | 9.86 | 0.4 | 0.537 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Pulp density × acid concentration Pulp density × acid concentration |
1 | 40.45 | 40.45 | 1.64 | 0.219 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Pulp density × time Pulp density × time |
1 | 13.58 | 13.58 | 0.55 | 0.439 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Temperature × acid concentration Temperature × acid concentration |
1 | 35.58 | 35.58 | 1.44 | 0.248 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Temperature × time Temperature × time |
1 | 4.62 | 4.26 | 0.19 | 0.671 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Acid concentration × time Acid concentration × time |
1 | 49.42 | 49.42 | 2 | 0.177 |
| Error | 16 | 395.79 | 24.74 | ||
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Lack-of-fit Lack-of-fit |
10 | 335.02 | 33.5 | 3.31 | 0.078 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Pure error Pure error |
6 | 60.76 | 10.13 | ||
| Total | 30 | 5861.06 | |||
To get better understanding about the interaction effect of variables on the leaching of Ag 3-dimensional response plots were generated based on equations. Fig. 3 presents surface response plots illustrating the influence of various factor combinations on Ag dissolution at a given time. In Fig. 3(a), Ag dissolution increases with rising temperature and pulp density. Fig. 3(b) shows that both temperature and time positively affect the dissolution rate, with a maximum observed at 70 g L−1 pulp density; beyond this, the rate declines due to particle agglomeration. In Fig. 3(c), increasing both pulp density and time enhances the dissolution rate, while other parameters remain constant. Fig. 3(d) demonstrates that, at fixed temperature and time, increasing citric acid concentration and pulp density initially improves dissolution, but the rate declines at higher pulp densities. Similarly, Fig. 3(e) shows that a rise in both citric acid concentration and time leads to increased Ag dissolution. Lastly, Fig. 3(f) reveals that, at constant pulp density and time, higher citric acid concentration and temperature results in a higher dissolution rate. Each 3D response surface plot generated from the predicted model illustrates the interaction between two operational parameters. Fig. 3(a–f) describes the relationship between the percentage dissolution of Ag and key variables such as acid concentration, leaching time, temperature, and pulp density. The graphical representation indicates that the maximum dissolution of Ag occurs at 75 °C after 120 minutes of leaching.
Furthermore, the residuals displayed in the normal probability plot (Fig. 4) follow a linear trend, indicating normally distributed errors suggesting a normal distribution.45
5. Integration of machine learning (ML) for yield prediction
5.1. Dataset partitioning for experimental evaluation using ML
For the experimental evaluation, the dataset comprising a total of 31 samples was partitioned into training and testing subsets in an 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 proportion. Specifically, the train_test_split function from the scikit-learn library was utilized with a predetermined random seed (random_state = 42) to guarantee the consistency of results. This resulted in 80% of the samples being allocated for training and 20% for testing, enabling consistent model assessment while preventing data leakage between the two subsets.
20 proportion. Specifically, the train_test_split function from the scikit-learn library was utilized with a predetermined random seed (random_state = 42) to guarantee the consistency of results. This resulted in 80% of the samples being allocated for training and 20% for testing, enabling consistent model assessment while preventing data leakage between the two subsets.
5.2. Predicted results of test data and plot visualization
This research investigates the utilization of ML methodologies for forecasting Ag recovery efficiency (FITS1) from PCBs based on experimental parameters including temperature, pulp density, acid concentration, and leaching time. The dataset comprised 31 samples with no missing values, ensuring data consistency for model training and evaluation. A comprehensive comparative analysis was conducted employing a suite of seven distinct machine learning algorithms to develop predictive models for Ag recovery yield. The model selection encompassed a strategic range of techniques to ensure a robust evaluation. The model included ridge polynomial regression, polynomial regression, Lasso polynomial regression, random forest regressor, gradient boosting regressor, Multi-Layer Perceptron (MLP) neural network, XGBoost regressor. The predictive capability of the models was analyzed through R2, RMSE, and CV score evaluations. The summary is depicted in Table 3.| Model | R2 score | RMSE | CV score |
|---|---|---|---|
| Polynomial regression | 1.0 | 0.0028 | 0.9956 ± 0.0088 |
| Ridge polynomial | 0.1481 | 7.8353 | −1.0945 ± 2.4751 |
| Lasso polynomial | 0.8893 | 2.8245 | 0.2976 ± 0.7921 |
| Random forest | 0.2794 | 7.2065 | 0.6708 ± 0.2418 |
| Gradient boosting | −0.3309 | 9.7936 | 0.2984 ± 0.5337 |
| MLP neural network | 0.3905 | 6.6273 | −3.8266 ± 5.6796 |
| XGBoost | 0.082 | 8.1337 | 0.6814 ± 0.3389 |
Table 4 compares the predictive performance of four regression models polynomial regression, Lasso polynomial regression, MLP neural network, and random forest against actual values. It presents the predicted values and their corresponding residuals (the difference between observed and predicted values) for each model. The residuals indicate how accurately each model fits the data smaller residuals imply better prediction accuracy. The evaluation of the seven machine learning models revealed a clear hierarchy of predictive performance for Ag recovery yield. The polynomial regression model (degree = 2) demonstrated exceptional efficacy, achieving a near-perfect fit on the test data, as evidenced by a coefficient of determination (R2) of 1.000, a root mean squared error (RMSE) of 0.0028, and a robust (CV) R2 score of 0.9956 ± 0.0088. This performance suggests the model encapsulates the underlying dynamic process with remarkable precision. The Lasso polynomial regression model also exhibited strong performance, validating the significance of the polynomial feature space while confirming that a sparse model remains highly effective.46 In contrast, the more complex, non-linear algorithms including the random forest, XGBoost regressors, and gradient boosting, alongside the Multi-Layer Perceptron (MLP) neural network yielded comparatively lower predictive accuracy on this specific dataset.47 The superior performance of the polynomial model strongly indicates that the relationship between the investigated process parameters (pulp density, temperature, acid concentration, and time) and the Ag recovery efficiency is predominantly quadratic and linear in nature, with significant interaction effects. This finding is consistent with the principles of RSM commonly used in process optimization.
| Actual | Polynomial_regression_predicted | Polynomial_regression_residuals | Lasso_polynomial_predicted | Lasso_polynomial_residuals | MLP_reural_network_predicted | MLP_neural_network_residuals | Random_rorest_predicted | Random_forest_residuals |
|---|---|---|---|---|---|---|---|---|
| 86.71 | 86.7105 | −0.0005 | 84.3912 | 2.3187 | 86.8405 | −0.1305 | 86.4751 | 0.2349 |
| 77.48 | 77.4790 | 0.0009 | 82.4488 | −4.9688 | 80.2203 | −2.7403 | 70.0608 | 7.4192 |
| 88.69 | 88.6917 | −0.0017 | 86.0588 | 2.6311 | 78.0447 | 10.6452 | 71.5855 | 17.1045 |
| 88.65 | 88.6451 | 0.0048 | 85.1341 | 3.5158 | 78.8071 | 9.8428 | 85.2437 | 3.4063 |
| 67.71 | 67.7063 | 0.0036 | 67.8353 | −0.1253 | 75.2060 | −7.4960 | 65.6884 | 2.0216 |
| 69.47 | 69.4661 | 0.0038 | 68.4195 | 1.0504 | 75.2590 | −5.7890 | 69.1098 | 0.3602 |
| 86.71 | 86.7105 | −0.0005 | 84.3912 | 2.3187 | 86.8405 | −0.1305 | 86.4751 | 0.2349 |
The comparatively lower performance of the tree-based and neural network models suggests that their inherent capacity for modeling highly complex, non-linear patterns may not be necessary for this system, and they may be more susceptible to learning noise or require more extensive hyperparameter tuning on a dataset of this size.
This Fig. 5 depicts comparative performance evaluation of seven machine learning models for predicting Ag recovery yield, measured by R2, RMSE, MAE, and cross-validation R2 scores. The polynomial regression model (far left) demonstrates superior predictive accuracy with near-perfect R2 and lowest error values.48,49
 | ||
| Fig. 5 Comparison of regression models based on R2 and RMSE values and performance evaluation of regression models using MAE and cross-validated R2 scores. | ||
The predictive performance of polynomial regression, Lasso polynomial regression, and MLP neural network for estimating Ag recovery from the experimental dataset is compared in Fig. 6. Each plot shows the correlation between the actual and predicted Ag recovery percentages, with the red dashed line representing the line of perfect prediction (where the predicted values perfectly align with the actual values).
6. Conclusion
Based on the experimental findings, citric acid was found to be the most effective leaching agent for the recovery of Al, Zn, and Ag from PCBs compared to glycine and oxalic acid. Approximately 91.58% of silver was successfully leached in a single stage process. While traditional hydrometallurgical optimization requires multiple experimental trials, advanced techniques such as RSM significantly require multiple experimental trails, advanced techniques such as RSM significantly reduce both time and chemical consumption while enhancing leaching efficiency. Under optimized conditions i.e., 1 M citric acid, 70 g L−1 pulp density, 75 °C, and 120 minutes, the leaching efficiency exceeded 90% for the target metals. The obtained leaching solution will undergo further purification through solvent extraction or ion exchange, followed by metal recovery via electrowinning or precipitation. To support process intensification, ML models (polynomial regression, random forest, and XGBoost) were developed to predict metal recovery based on leaching parameters. Among these, polynomial regression demonstrated the highest predictive accuracy, confirming its effectiveness for forecasting metal yields with minimal error. These data-driven models not only validated the experimental outcomes but also provide a scalable framework for optimizing future leaching operations, thereby minimizing time, cost, and chemical usage. Regarding scale-up, the citric acid system is promising because citrate-based lixiviants are relatively non-toxic and biodegradable. However, large-scale operation requires careful control of reagent cost and process integration. Citric acid costs can be reduced by using the identified optimum concentration (1 M), selecting technical-grade citric acid, and recycling the leach liquor after downstream metal recovery. Citrate can also be regenerated and reused by recycling the spent lixiviant after metal removal with pH adjustment and make-up oxidant, while controlling impurity build-up through periodic bleed-and-replenish. However, reuse efficiency was not experimentally evaluated in this study. Future work will quantify multi-cycle reuse, acid consumption, and regeneration routes.Author contributions
Syed Suffia Iqbal: data curation, validation, visualization, methodology, conceptualization and writing original draft. Ovais Iqbal Shah: writing: reviewing and editing. Deblina Dutta: supervision; writing: reviewing and editing.Conflicts of interest
The authors state that they have no financial conflicts of interest or personal relationships that may have affected the research presented in this manuscript.Data availability
All data are available in the manuscript.Acknowledgements
We gratefully acknowledge the financial support from SRM University-AP under the SEED grant (Grant number: SRMAP/URG/SEED/2023-24/017).References
- J. Xiao, M. He, B. Zhan, H. Guo, J. Yang, Y. Zhang, X. Qi and J. Gu, Mater. Horiz., 2024, 11, 123456 Search PubMed.
- S. P. Tembhare, B. A. Bhanvase, D. P. Barai and S. J. Dhoble, Environ. Dev. Sustain., 2021, 23, 1–83 Search PubMed.
- A. Akcil, C. Erust, C. S. Gahan, M. Ozgun, M. Sahin and A. Tuncuk, Waste Manag., 2015, 45, 25–41 Search PubMed.
- H. Duan and X. N. Zhu, Energy Sources, Part A, 2022, 44, 1640–1659 Search PubMed.
- P. Lin, J. Werner, J. Groppo and X. Yang, Sustainability, 2022, 14, 13479 CrossRef CAS.
- L. Pietrelli, S. Ferro and M. Vocciante, Renewable Sustainable Energy Rev., 2019, 112, 317–323 CrossRef CAS.
- H. Li, J. Eksteen and E. Oraby, Resour., Conserv. Recycl., 2018, 139, 122–139 CrossRef.
- M. D. Rao, K. K. Singh, C. A. Morrison and J. B. Love, RSC Adv., 2020, 10, 4300–4309 RSC.
- M. Guo, R. Deng, M. Gao, C. Xu and Q. Zhang, Curr. Opin. Green Sustainable Chem., 2024, 100913 Search PubMed.
- S. S. Iqbal, P. P. Dagwar and D. Dutta, in Sustainable Waste Management: Challenges and Visions, Springer Nature Switzerland, Cham, 2025, pp. 37–58 Search PubMed.
- S. Kumari, R. Panda, R. Prasad, R. D. Alorro and M. K. Jha, Sustainability, 2024, 16, 418 Search PubMed.
- S. Sudarsan, M. Anandkumar and E. A. Trofimov, Int. J. Environ. Sci. Technol., 2024, 21, 1–20 Search PubMed.
- D. Dutta, S. Goel and S. Kumar, J. Environ. Chem. Eng., 2022, 10, 107269 Search PubMed.
- S. Sudarsan, M. Anandkumar and E. A. Trofimov, Int. J. Environ. Sci. Technol., 2025, 22, 1263–1282 Search PubMed.
- S. S. Iqbal, N. Ramadan, P. P. Dagwar, D. Dutta and D. M. Abdo, Sep. Purif. Technol., 2025, 133860 Search PubMed.
- U. Jadhav, C. Su and H. Hocheng, Environ. Sci. Pollut. Res., 2016, 23, 24384–24392 Search PubMed.
- N. Nagarajan and P. Panchatcharam, Heliyon, 2023, 9, e13742 CrossRef PubMed.
- E. A. Oke and J. H. Potgieter, Sep. Purif. Technol., 2025, 133712 CrossRef CAS.
- S. Suffia and D. Dutta, J. Mol. Liq., 2024, 394, 123738 CrossRef CAS.
- E. A. Oke and H. Potgieter, J. Mater. Cycles Waste Manage., 2024, 26, 1349–1368 CrossRef CAS.
- R. Rautela, B. R. Yadav and S. Kumar, J. Cleaner Prod., 2024, 447, 141602 CrossRef CAS.
- H. Nayebzadeh, N. Saghatoleslami and M. Tabasizadeh, J. Taiwan Inst. Chem. Eng., 2016, 68, 379–386 CrossRef CAS.
- G. E. Box and K. B. Wilson, in Breakthroughs in Statistics: Methodology and Distribution, Springer, New York, 1992, pp. 270–310 Search PubMed.
- M. D. Rao, K. K. Singh, C. A. Morrison and J. B. Love, Clean. Eng. Technol., 2021, 4, 100180 CrossRef.
- P. Mokarian, I. Bakhshayeshi, F. Taghikhah, Y. Boroumand, E. Erfani and A. Razmjou, Sep. Purif. Technol., 2022, 291, 120919 CrossRef CAS.
- R. Kumari and S. R. Samadder, J. Environ. Manage., 2023, 345, 118550 CrossRef CAS PubMed.
- F. Faraji, R. Golmohammadzadeh, F. Rashchi and N. Alimardani, J. Environ. Manage., 2018, 217, 775–787 CrossRef CAS PubMed.
- B. K. Biswal, B. Zhang, P. T. M. Tran, J. Zhang and R. Balasubramanian, Chem. Soc. Rev., 2024, 53, 5552–5592 RSC.
- F. Pourhossein, S. M. Mousavi and F. Beolchini, Resour., Conserv. Recycl., 2022, 182, 106306 CrossRef CAS.
- N. Nagarajan and P. Panchatcharam, Heliyon, 2023, 9, e13714 CrossRef PubMed.
- L. Li, J. Ge, F. Wu, R. Chen, S. Chen and B. Wu, J. Hazard. Mater., 2010, 176, 288–293 CrossRef CAS PubMed.
- S. Fatima, M. K. Khosa, A. Noor, S. Qayyum and M. El Oirdi, Sustainability, 2024, 16, 6817 CrossRef CAS.
- F. Meng, Q. Liu, R. Kim, J. Wang, G. Liu and A. Ghahreman, Hydrometallurgy, 2020, 191, 105160 CrossRef CAS.
- M. Aaltonen, C. Peng, B. P. Wilson and M. Lundström, Recycling, 2017, 2, 20 CrossRef.
- A. Javed and J. Singh, Environ. Sci. Pollut. Res., 2024, 31, 9886–9919 CrossRef CAS PubMed.
- W. Ding, S. Bao, Y. Zhang, B. Chen, X. Wan and J. Xiao, Miner. Process. Extr. Metall. Rev., 2024, 45, 495–508 CrossRef CAS.
- J. Ran, Y. Li, X. Wang, H. Duan, Y. Chen, S. Yin, S. Li, L. Zhang and J. Zhao, Environ. Chem. Lett., 2024, 22, 2055–2090 CrossRef CAS.
- J. Chen, S. Huang, S. BalaMurugan and G. S. Tamizharasi, Environ. Impact Assess. Rev., 2021, 87, 106498 CrossRef.
- S. Anwer, A. Panghal, I. Majid and S. Mallick, Int. J. Environ. Sci. Technol., 2022, 19, 9731–9740 CrossRef CAS.
- D. Dutta, R. Panda, A. Kumari, S. Goel and M. K. Jha, Sustainable Mater. Technol., 2018, 17, e00066 Search PubMed.
- R. Ozairy, S. O. Rastegar, R. Beigzadeh and T. Gu, J. Mater. Cycles Waste Manage., 2022, 24, 179–188 CrossRef CAS.
- J. M. Hartley, S. Scott, R. M. Rivera, P. Hunt, A. J. Lucio, P. Bird, R. Harris, G. R. T. Jenkin and A. P. Abbott, RSC Sustainability, 2023, 1, 107–116 Search PubMed.
- F. Faraji, A. Alizadeh, F. Rashchi and N. Mostoufi, Rev. Chem. Eng., 2022, 38, 113–148 CrossRef CAS.
- M. Arshadi and S. M. Mousavi, Bioresour. Technol., 2014, 174, 233–242 CrossRef CAS PubMed.
- V. H. Ha, J. C. Lee, T. H. Huynh, J. Jeong and B. D. Pandey, Hydrometallurgy, 2014, 149, 118–126 CrossRef CAS.
- M. Somasundaram, R. Saravanathamizhan, C. A. Basha, V. Nandakumar, S. N. Begum and T. Kannadasan, Powder Technol., 2014, 266, 1–6 CrossRef CAS.
- B. K. Körbahti and M. A. Rauf, J. Hazard. Mater., 2009, 161, 281–286 CrossRef PubMed.
- V. Flores and C. Leiva, Sensors, 2021, 21, 2119 CrossRef PubMed.
- A. Sharma, N. Patel and R. Gupta, European Advanced AI Journal, 2022, 11, 8 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |





