DOI:
10.1039/D5RA08490C
(Review Article)
RSC Adv., 2026,
16, 12663-12681
A review of commercial and laboratory-based microfluidic devices based on glass and/or silicon substrates
Received
4th November 2025
, Accepted 10th February 2026
First published on 6th March 2026
Abstract
Microfluidic devices are increasingly being sought after as powerful alternatives to traditional macroscopic benchtop biochemical analysis systems due to their lower cost, faster analysis times, and reduced reagent consumption. Advances in additive manufacturing and semiconductor fabrication have simplified device realization, enabling their integration into a wide range of biochemical applications. Microfluidic devices were initially fabricated using glass and silicon as substrate materials. However, as the demand for more complex structures has increased, new materials have been introduced. Among these are soft and thermoplastic polymers, which are well suited to techniques such as soft molding, injection molding, and hot embossing. Despite these developments, surface-treated glass and silicon are preferred for high-grade analyses due to their superior chemical, biological, thermal, and physical properties. In this review, we highlight recent progress in commercially and semi-commercially available microfluidic devices based on glass and silicon substrates. We summarize key fabrication methods and their applications and examine how these platforms are advancing toward integrated readout, actuation, and sensing capabilities for a complete lab-on-a-chip system.
Introduction
Microfluidics encompasses the precise manipulation of fluids within microscale and nanoscale channels, enabling controlled chemical and biological processes with minimal reagent volume. The early development of microfluidics was enabled by advances in silicon microfabrication, microelectromechanical systems (MEMS), and microelectronics which later expanded into various different materials and fabrication platforms. Microanalytical techniques such as gas-phasechromatography (GPC), high-performance liquid chromatography (HPLC), and capillary electrophoresis (CE) achieve high sensitivity using minute sample volumes. This drove the evolution toward compact and versatile microfluidic systems for chemical and biochemical applications.1,2
A growing need for flexible, low-cost, and rapid prototyping methods emerged3 as the applications of microfluidics further expanded to include chemical analysis, diagnostics, materials synthesis, single-cell studies, and microreactors. In response, polymeric materials, including polydimethylsiloxane (PDMS) and various thermoplastics, became increasingly prevalent.
Advances in microfabrication have enabled planarized microfluidic systems. It integrates complex microchannel networks for miniaturized LOC (lab on a chip) platforms.4 The development of multifunctional lab-on-a-chip systems has been facilitated with the help of hybrid fabrication techniques, where glass moulds are integrated with polymer casting or combining silicon microfabrication with soft materials helps incorporate embedded sensing, actuation and optical components.3 There are several easy and affordable alternative fabrication techniques, such as 3D printing,1 liquid molding,5 rapid manufacturing,6 and laser cutters/engravers,7 that can be used to lower fabrication costs and production time. They are successful in reducing costs and producing minuscule micro/nanoscale features.8 The variety of fabrication methods employed, materials, and variation of sizes of these microfluidic devices are shown in Fig. 1. It provides a clearer understanding of their versatility. The primary determinant of these devices' overall performance is the substrate on which they are fabricated. Initially, silicon was used as the substrate material in microfluidics, but today, a variety of materials, including glass, thermoplastics, hydrogels, and soft polymers, are used. Glass and silicon, in particular, offer a unique combination of chemical inertness, thermal stability, optical transparency, mechanical rigidity, and compatibility with semiconductor-grade microfabrication9 that is not generally achievable only using polymeric materials.10,11 These materials uniquely enable semiconductor-grade microfabrication, allowing precise, reproducible, and scalable device manufacturing that is not consistently achievable with polymer-based systems. Silicon-based materials have been the most standard microfluidic platform for decades due to their ability to simplify manufacturing, semiconducting properties and their capability of easy modification of the surface. The practical use of silicon limits opacity, which prevents its use in optical detection across the visible and UV spectra, so alternative materials are required for applications that account for situ imaging. Also, integration of functional components such as valves and pumps in silicon devices is impeded due to their fragility and elevated elastic moduli. Nevertheless, silicon microfluidic systems are effectively implemented in various biological applications, such as point-of-care diagnostics and organ-on-chip technology for drug screening.
 |
| | Fig. 1 Various fabrication processes, materials and length scales of widely used microfluidic devices. | |
In contrast, glass is chemically inert. It is thermally and electrically insulating, mechanically rigid, biocompatible, and amenable to surface functionalization. It allows glass-based microreactors suitable for chemical reactions under demanding conditions, including high temperatures, pressures, and corrosive environments. Glass microcapillaries afford micrometer scale resolution, enabling the precision synthesis of emulsions and nanoparticles.6,12 Glass offers substantial benefits compared to silicon with respect to optical transparency, production cost and the flexibility of integrating active elements, such as incorporating pumps and valves made from a variety of materials, where ultrathin glass sheets can act as active parts. Its transparency supports optical detection, while its stability allows for effective cleaning with heat or chemicals. Though manufacturing glass microfluidic devices can be both resource-intensive and costly, often requiring cleanroom environments. The widespread use of glass in laboratories and its compatibility with biological samples further encourage its adoption in microfluidic biochemical analysis. Glass is a wide material category, including standard substrates such as soda-lime, borosilicate, and fused quartz.5 Maskless lithography emerged as a cost and time efficient alternative to conventional photolithography for rapid prototyping. In these systems, a focused laser directly patterns the photoresist which eliminates the need for physical photomasks. It enables fast design iteration on both silicon and glass substrates. Similar laser-assisted or lithographic approaches are also used in soft lithography workflows to fabricate glass-based master moulds.
Many microfluidic applications require specific surface modification for wettability, solvent compatibility, and stable surface condition at elevated operating temperatures. For instance, polymerase chain reaction (PCR) and nanoparticle synthesis often demand materials that can hold out against temperatures exceeding 100 °C and prolonged exposure to organic solvents. In these kinds of scenarios, glass remains a preferable substrate as it is able to maintain chemical stability as well as structural integrity under harsh operating conditions.13 Hybrid silicon–glass devices are mostly employed to combine the exact channel definition achievable in silicon with the excellent optical transparency of glass. Silicon and glass substrates are bonded using various techniques; for example anodic bonding architectures produce microfluidic devices which are optically accessible. In these architectures, silicon substrates are commonly sealed with glass cover plates. This bonding strategy yields devices that are highly transparent with precise channel definition and robust mechanical integrity, which are well-suited for high-pressure microfluidic operation.
A major part of the industrial and semi commercial microfluidic platforms relies on silicon and glass substrates yet there is a notable absence of comprehensive systematic reviews that directly compare their performance, fabrication strategies and application tradeoffs.14 Moreover, prior literature reviews cut out the critical assessment of the impact of substrate selection on device reproducibility, functionality, and their adaptability for advanced scientific applications. This includes droplet microfluidics, high-temperature chemical reactions, and optofluidic system integration.15 The existing literature focuses on broad spectrum fabrication methods or on polymer-based microfluidic systems. This literature gap is significant as the recent advancements in microfluidic technologies are directed towards the domains that demand enhanced chemical durability, seamless integration with photonic or electronic components and optical transparency – areas where silicon and glass substrates provide significant margins.16 Thus, this review presents a structured report of commercially and semi-commercially available microfluidic platforms fabricated from glass, silicon, and hybrid materials. Firstly, we discuss the common microfabrication materials. Then the fabrication techniques are reviewed with emphasis on their practical advantages and limitations for microfluidic device implementation. The discussion then examines how material choice and fabrication strategy influence the key performance metrics, including dimensional precision, scalability, and system integration. Finally, the review surveys commercial manufacturing platforms and reported applications, encompassing both standalone microfluidic devices and integrated systems such as optofluidic and CMOS (Complementary Metal-Oxide-Semiconductor)-enabled microfluidic technologies, highlighting current trends toward increased integration, automation and reduced reagent and power consumption.
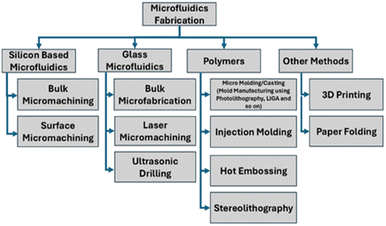
Fabrication process
The materials used for microfluidic devices determine the fabrication process; some of these processes are depicted in Fig. 2 for common materials.
 |
| | Fig. 2 Fabrication process based on different materials used for microfluidics devices. The diagram specifies bulk and surface micromachining for silicon and glass, laser-based etching for glass substrates, and alternative polymer-based techniques such as hot embossing and micro molding, showing diversity in methods and highlighting how material choice influences device design, resolution and scalability. Adapted with permission from ref. 17. Copyright 2019 Elsevier. | |
Conventional microfabrication of silicon and glass microfluidic devices relies on photolithography combined with wet or dry etching techniques. In this process, the pattern is transferred by spin-coating a silicon wafer with photoresist, then exposing it to ultraviolet light through a photomask and developing it. Using either wet chemical etching or dry plasma etching process, pattern transfer into the underlying silicon or glass substrate is achieved.
Depending on the etching method and targeted application, a typical microchannel depth produced using silicon etching techniques range from approximately, 10 to 300 µm. After the etching is completed, any hardmask layers and residual photoresist are removed. This is often followed by bonding to a cover plate to form enclosed microchannels. This sequence allows highly reproducible fabrication and compatible electronic integration or subsequent surface functionalization. Deep reactive ion etching (DRIE) is typically used for silicon substrates to attain channel depths ranging from tens to several hundreds of micrometres. Optimised DRIE processes yield near-vertical sidewalls and dimensional tolerances which are typically on the order of ±1–2 µm across a wafer.18 For optimised DRIE processes, with roughness strongly influenced by etch cycling parameters and post-processing conditions, sidewall surface roughness is typically on the order of hundreds down to tens of nanometers with much slower processes. Glass microfluidic devices, that are fabricated using methods like photolithography and wet chemical etching usually achieve channel depths between approximately, 10 and 100 µm. Channel sidewalls exhibit rounded profiles and lower aspect ratios than in silicon DRIE due to the isotropic nature of glass wet etching. However, following the appropriate chemical or thermal post-treatment, surface roughness can be imporoved to tens of nanometers. Reactive ion etching of glass to define the channels has also been employed. Other fabrication approaches such as laser-based micromachining techniques for glass based microfluidic devices are widely employed including femtosecond laser etching (LionIX19), femtosecond laser based selective etching (Femtika20), laser-induced deep etching (LDPE) from LPKF Laser & Electronics,21 and femtosecond laser processing by Posalux to achieve more DRIE-like anisotropy. To define inlet and outlet ports in glass substrates ultrasonic drilling is also commonly used in glass substrates.
Fig. 3 illustrates a soft lithography process where the device is fabricated using a two-step soft lithography process. It begins with photolithographic patterning of a master mold and followed by replica molding of polydimethylsiloxane (PDMS). This forms a microfluidic layer after which the PDMS prepolymer is subsequently poured onto the master mold. Then it is degassed under vacuum to remove all the trapped air bubbles. To achieve solidification, it is thermally cured for approximately two hours. To enhance the interfacial adhesion and bond robustness the PDMS replica is peeled from the mold and using oxygen plasma treatment it is irreversibly bonded to a flat substrate after curing by moderate thermal treatment typically 70–90 degree Celsius. Several commercial microfluidic technology providers offer partially or fully integrated platforms including PDMS based chips. For instance, to achieve precise flow regulation and study droplet generation, microfluidic devices from companies like Elveflow and Fluigent specialise in pressure-based flow control systems and microfluidic instrumentation that have been used.7,22,23 For pressure-driven and electrokinetic flow control, supporting flexible system-level integration, LabSmith offers modular microfluidic components.24 Darwin Microfluidics provides access to silicon- and glass-based microfabrication services and master moulds, enabling integration with external fluidic and sensing components.25,26 In addition, µFluidix and related platforms provide PDMS-based microfluidic devices and accessories, including droplet-based analytical systems such as droplet sequencing technologies.27 Owing to their low cost and design flexibility, polymer based microfluidic devices are widely used and are not limited to planar fabrication.28 Micromolding, casting, injection molding, hot embossing such replication-based techniques are mainly employed for thermoplastic materials.
 |
| | Fig. 3 Stepwise illustration of soft lithography process for microfluidic device fabrication. The diagram outlines photolithographic mold creation using silicon wafers, UV exposure for pattern transfer, PDMS casting into the mold, and subsequent plasma bonding to form enclosed microchannels, which enables rapid prototyping and fabrication of flexible microfluidic devices with high-resolution features. Adapted with permission from ref. 29 and 30. Copyright 2013 Humana Press. | |
Fig. 4 illustrates the injection process which involves injecting the material into preprepared molds that are then cast into the desired shape. In this instance, the molds are fabricated beforehand and equipped with aligners to ensure that the top and bottom have the proper alignment; otherwise, the device construction would be misaligned. After the PDMS material is injected within the mold, the newly formed device is encapsulated and held firmly to prevent any fluid from escaping before the PDMS is injected inside and allowed to set. After that, it is removed from the molds and bonded to the glass to cover the channels. Similar to injection molding, hot embossing employs a prefabricated mold that is pressed into a heated thermoplastic substrate to transfer microchannel features. Fig. 5 shows the hot embossing fabrication process, where a rigid mold usually fabricated from steel, nickel, or silicon is pressed into a thermoplastic substrate under controlled temperature and pressure to replicate all the features of the microchannel. In this procedure cyclic olefin polymer (COP) substrates are normally employed due to their chemical resistance and optical transparency. After embossing, the patterned COP substrate is bonded to a COP cover layer to enclose the microfluidic channels and form a sealed device.
 |
| | Fig. 4 A brief description of the injection molding process used to mass-produce thermoplastic microfluidic devices. The figure shows molten polymer being injected into prefabricated molds to replicate the microchannel features, followed by cooling and demolding, ideal for producing multilayered microfluidic systems with complex geometries. Adapted with permission from ref. 31. Copyright 2016 Royal Society of Chemistry. | |
 |
| | Fig. 5 Illustration of the hot embossing process of thermoplastic microfluidic device fabrication. A silicon-based mold with microchannel features is heated and pressed against a cyclic olefin polymer (COP) substrate to transfer the pattern. The bonded COP layers form enclosed channels, enabling durable and chemically resistant devices. Adapted with permission from ref. 32. Copyright 2008 IEEE NEMS. | |
Alternative approaches, such as additive manufacturing and paper-based microfluidics, have been explored for rapid prototyping and low-volume production of these devices. 3D printing is an additive fabrication technique where the structures are generated layer by layer from a digital CAD (computer-aided design) model using thermoplastic filaments or photosensitive resins. Though 3D printing offers design flexibility, it comes with a provision of lower feature resolution and higher surface roughness compared to ideal lithography-based methods. On the other hand, for low-cost diagnostic applications, paper-based microfluidics are primarily used, which relies on capillary-driven fluid transport within patterned porous substrates. Several key material properties relevant to microfluidic device performance are summarized in Table 1. Polymer-based materials, including thermoplastics and polydimethylsiloxane (PDMS), are widely used due to their low cost, ease of replication, and ability to form complex geometries. These include several transparent thermoplastic materials including polymethyl methacrylate (PMMA), polystyrene (PS), polycarbonate (PC), cyclic olefin polymer (COP), and cyclic olefin copolymer (COC), commercially known as Zeonor and Topas. Among these materials, Zeonor and Topas exhibit optical clarity and low autofluorescence, making them suitable for optofluidic applications. As summarized in Table 2, glass is a resilient alternative to polymers even at temperatures above 100 °C when exposed to organic solvents, as in the application of nanoparticle synthesis.33 Thermoplastics can perform similarly to glass in applications requiring intricate microchannel geometries. Although fabrication constraints such as mould complexity, material flow behaviour, thermal shrinkage, and achievable aspect ratios impose limitations in injection-moulded devices. As a result, instead of using traditional planar manufacturing techniques and bonding layers to finish the device, they are occasionally manufactured in complex 3D structures.
Table 1 Overview of the materials for microfluidic devices. Adapted with permission from ref. 28. Copyright 2013 Accounts of Chemical Research
| Mechanical properties |
Elastomers |
Glass |
Hydrogel |
Paper |
Silicon |
Thermoset |
Thermoplastics |
| Young's modulus (GPa) |
∼0.0005 |
50–90 |
Low |
0.0003–0.0025 |
130–180 |
2.0–2.7 |
1.4–4.1 |
| A common technique for fabrication |
Casting |
Photolithography, laser etching |
Casting, photopolymerization |
Photolithography, printing |
Photolithography, ultrasonic drilling |
Casting, photopolymerization |
Thermomolding |
| Thermal conductivity (W m−1 K−1) |
0.1 to 0.5 |
0.5 to 1.38 |
0.33 to 0.51 |
0.15 to 0.21 |
139 |
0.15 to 0.25 |
0.17 to 0.5 |
| Smallest channel dimension |
<1 um |
<100 nm |
∼10 um |
∼200 um |
<100 nm |
<100 nm |
∼100 nm |
| Channel profile |
3D |
Limited 3D |
3D |
2D |
Limited 3D |
Arbitrary 3D |
3D |
| Thermostability |
Medium |
Very high |
Low |
Medium |
Very high |
High |
Medium to high |
| Resistance to oxidizer |
Moderate |
Excellent |
Low |
Low |
Excellent |
Good |
Moderate to good |
| Solvent compatibility |
Low |
Very high |
Low |
Medium |
Very high |
High |
Medium to high |
| Hydrophobicity |
Hydrophobic |
Hydrophilic |
Hydrophilic |
Amiphilic |
Hydrophilic |
Hydrophobic |
Hydrophobic |
| Surface charge |
Not stable |
Very stable |
N/A |
N/A |
Very stable |
Stable |
Stable |
| Permeable to oxygen (Barrer) |
∼500 |
<0.01 |
>1 |
>1 |
<0.01 |
0.03–1 |
0.05–5 |
| Optical transparency |
High |
High |
Low to medium |
Low |
No |
High |
Medium to high |
| Autofluorescence |
Some/high |
Some |
— |
High |
No |
Some/high |
Some/high |
Table 2 Comparison between thermoplastics and glass. Adapted with permission from ref. 34. Copyright 2013 American Chemical Society
| Optics |
Standard thermoplastics |
Glass |
| Transparency |
Good |
Good |
| Autofluorescence |
Low (the right polymer choice is important) |
Low |
| Application in the UV region |
In near UV, special polymers are available |
Quartz glass needs to be chosen |
| Surface roughness |
Can be optically smooth |
Depending on technology |
| Thermal stability |
Depending on the polymer choice, 100 °C is possible |
Approximately 600 °C transition to the liquid phase |
| Stability against organic solvents |
Limited |
High |
| Stability against standard solvents in life sciences |
Polymers available |
High |
| Stability against basic solutions |
High |
Medium |
| Stability against acidic solutions |
High |
High |
| Unspecific binding of biological components |
Polymers with low unspecific binding are available, and surface functionalization to avoid the issue is available |
High, surface functionalization to avoid the issue available |
The chemical compatibility varies significantly depending on the polymer and the reagents employed despite the versatility that thermoplastics offers. Table 3 summarizes the compatibility of common thermoplastics with frequently used microfluidic reagents, highlighting that no single polymer is universally suitable for all applications. Material selection must be guided by application-specific requirements rather than fabrication convenience alone.
Table 3 Chemical compatibility of common thermoplastics (S denotes a chemical that is suitable for thermoplastics, whereas NS does not suit a specific thermoplastic, and X denotes data not available). Adapted with permission from ref. 35. Copyright Microfluidic ChipShop
| Chemicals |
Thermoplastic |
| Polymethylmethacrylate (PMMA) |
Polystyrene (PS) |
Cyclo-olefin polymer (Zeonor), Cyclo-olefin-copolymer (Topas) |
Polycarbonate (PC) |
| Acids and bases |
NS (concentrated) |
S (organic) |
X |
S (diluted acid) & NS (bases) |
| Alcohols |
NS |
X |
X |
S |
| Aldehydes |
S |
X |
X |
NS |
| Amines |
S |
X |
X |
NS |
| Aqueous solution |
S |
X |
S |
X |
| Aromatics |
NS |
X |
X |
NS |
| Butyl alcohol |
X |
S |
X |
|
| Esters |
NS |
NS |
X |
NS |
| Ethers |
X |
NS |
X |
X |
| Ethylene glycol |
X |
S |
X |
X |
| Halogenated hydrocarbons |
NS |
NS |
NS |
X |
| Halogenated organic reagents |
X |
NS |
X |
X |
| Hydrogen oxide |
X |
S |
X |
X |
| Ketones |
NS |
NS |
X |
NS |
| Mineral oil |
X |
S |
NS |
X |
| Nonpolar solvents |
X |
X |
NS |
X |
| Oils & fats |
S |
X |
NS |
S |
| Polar solvents |
X |
X |
S |
X |
| Silicone oils |
X |
X |
S |
X |
Here, we reviewed commercially available companies and the work done using those devices. They are sectioned on the basis of different commertially available techniques, labeled and emphasized with the fabrication technique they alone or the user has employed using their unit to provide an overview of the flexibility. Table 4 summarises the key fabrication metrics, including their feature resolution, surface roughness, achievable aspect ratio, and reproducibility, which address the critical fabrication considerations highlighted in this review.
Table 4 Summary of fabrication metrics for representative microfluidic manufacturing techniques across silicon, glass, PDMS, and thermoplastic substrates. The data are summarized from literature sources.9,18,19,26–28,32
| Substrate & fabrication method |
Feature resolution |
Surface roughness (Ra) |
Aspect ratio |
Processing rate |
Reproducibility |
Limitations |
| Silicon: DRIE (Bosch) |
250 nm minimum trench width |
≈13 nm sidewall roughness |
160![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (high-aspect structures shown) 1 (high-aspect structures shown) |
≈5–6.6 µm min−1 etch depth |
Sidewalls etch lag < 1.5% |
Scallops/roughness depend on recipe |
| Glass: photolithography + wet etching |
Minimum mask line transfer: 5 µm |
Ra ≈ 5 nm (HF-etched glass) |
Limited by isotropic etching (∼2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
Etch rate ∼3 µm min−1 reported |
Thickness control ±5 µm |
Rounded profiles due to isotropy |
| Glass: SLE (selective laser etching) |
— |
≈200 nm RMS |
Depth/width ratios up to ∼100–1000![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
820 µm h−1 etch volume removal |
— |
Slow serial process, equipment cost |
| Glass: laser ablation (direct) |
≥10 µm widths demonstrated |
178.7–1028.3 nm at different pulse conditions |
Up to ∼7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (by wall taper constraint) 1 (by wall taper constraint) |
|
— |
Rough walls: post-processing needed |
| PDMS: soft lithography |
Replica example channel 44 µm |
Ra ≈ 200 nm (replica) |
Achievable ∼7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
SLA mold step ∼<40 min |
Example spacing 117 ± 1.3 µm |
Swelling, solvent issues |
| Thermoplastics: hot embossing |
Embossed width 51.1 µm, depth 37.4 µm |
— |
AR up to ∼10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
∼1.5 mm s−1 embossing speed |
RSD 2.8–4.3% (dim accuracy) |
Tooling cost & mold fidelity |
Device performance is inherently design-dependent, and the selection of fabrication methods is often guided not only by cost and resolution but also by application-specific requirements such as pressure tolerance, chemical compatibility, optical access, and integration with sensing or actuation components. From a tooling and cost perspective, fabrication approaches differ substantially in their infrastructure requirements and scalability. Silicon-based processes such as deep reactive ion etching (DRIE) require access to photolithography equipment in combination with plasma etching systems and post-etch stripping processes, resulting in higher capital and operational costs compared to wet-etched glass processes, which can often be implemented using standard wet benches and photolithography tools. Selective laser etching (SLE) of glass similarly relies on wet chemical etching but additionally requires specialized ultrafast laser systems, which substantially increases equipment cost. In contrast, direct laser ablation can be performed using comparatively lower-cost laser systems, although achievable feature quality and surface roughness are typically inferior and often require post-processing. Soft lithography represents a lower cost alternative for rapid prototyping. It primarily relies on photolithographic mold fabrication followed by replica molding with fewer downstream processing steps. The cost of lithography tools widely varies with resolution, throughput, and substrate size. While high-end stepper-based systems are generally unnecessary for typical channel dimensions, benchtop or contact lithography systems offer enough precision for most microfluidic applications. Replication-based methods such as injection molding and hot embossing exhibit relatively low per-unit cost once molds are established, although initial tooling costs are high and design changes are less flexible. Additive manufacturing techniques, including 3D printing, present the lowest barrier to entry in terms of startup cost and design iteration, but their scalability and surface quality remain inferior to molding-based approaches for large-volume production. Polymer-based microfluidic devices are generally more suitable for low-cost disposable use, whereas glass- and silicon-based devices favour reusabilty due to their superior chemical and thermal durability, at the expense of higher replacement cost and longer fabrication turnaround in the event of device failure.28
Manufacturer of microfluidic devices for commercial use
Commercial microfluidic platforms differ substantially in material selection, fabrication strategy and the domain of their intended application. The following sections analyses how design choices can reflect targeted performance requirements. Pressure tolerance, chemical resistance, integration capability, and manufacturing scalability are analyzed.
Lamination, soft lithography, laser cutting, 3D printing
Aline provides various microfluidic devices employing a variety of materials, including silicon and glass. The Aline device has been utilized by researchers to separate cancer cells into individual cells.36 This device consists of seven layers, with polyethylene terephthalate (PET) having its channel laser cut. The layers were then aligned and fused via a lamination technique that used pressure and adhesive. The apparatus is depicted in Fig. 6(a), with the channels constricted to prevent cancer cells from aggregating. The technology was from Aline, which is based on the soft lithography process.33 They created molds via a silicon-based photolithography process and used PDMS for the devices. This PDMS-based apparatus is used to make nanowell arrays, which are then utilised in cell assays based on mammals. A different team manufactured a multilayer device with channels on PET laser etched and on PMMA drilled with CNC.37
 |
| | Fig. 6 Examples of microfluidic devices developed by Aline Inc., USA. (a) Multilayer PET device for single cancer cell analysis with laser-cut channels. (b) PMMA-PET multilayer reagent dispenser fabricated via CNC drilling and lamination. (c): Hybrid system for dry sample prevention using PMMA, PET and PDMS layers, which demonstrates the expertise in customized microfluidic solutions Aline has for biomedical research. Adapted with permission from ref. 36, 37 and 38. Copyright 2013, 2015 Royal Society of Chemistry, Copyright 2013 SLAS Technology. | |
The components were then joined via a lamination technique, as shown in Fig. 6(b). This apparatus served as a reagent dispenser for tests. Another team used a system comprising PMMA, PET, and PDMS to store hydrated samples in a dry state when needed, then retrieve them from the storage area.38 Fig. 6(c) shows the multilayer nature of the device. A summary of some of the articles utilizing Aline devices for microfluidic applications is displayed in the table above. From a design perspective, Aline's use of multilayer PET and PMMA architecture reflects a strategy optimized for rapid customization, low-cost fabrication, and application-specific functionality. Laser cutting, CNC drilling, and lamination enable fast design iteration and are well suited for biomedical workflows such as reagent dispensing and single-cell handling. However, compared to glass- or silicon-based platforms, these polymer systems may exhibit reduced chemical resistance and long-term dimensional stability, particularly under aggressive solvents or elevated temperatures. As a result, such platforms are best suited for short-to medium-term experimental use rather than high-pressure or chemically demanding application.
In many of the reported applications, the devices are effectively disposable, either due to biofouling, surface functionalization constraints, or cross-contamination risks, such that limited operational lifetime does not constitute a critical drawback.
As the targeted use case such as benchtop biological assays, proof-of-concept studies, and low-pressure microfluidic operations do not impose stringent requirements for chemical resistance or sustained high-pressure performance, the transitioning to alternative fabrication routes based on glass or silicon would offer marginal functional benefit while significantly increasing fabrication complexity, cost, and turnaround time.
| Company name |
Country |
Fabrication materials |
Fabrication process |
Applications |
| Aline |
California, US |
Polyethylene terephthalate (PET) |
Laser |
Single cancer cell analysis37 |
| PDMS & silicon wafer |
Soft lithography |
Cell assay39 |
| Glass, PET, PMMA, and polyurethane tape |
CNC drill on PMMA and laser etch on PET |
Reagent dispensing system for assays40 |
| PDMS, PMMA, and PET |
3D printing and LASER |
To store the sample in a dry state38 |
Wet etching
Wet etching of glass to produce microfluidics is widely used method. For example, Dolomite offers standard microfluidic structures for encapsulating Active Pharmaceutical Ingredient (API) in polylactic-co-glycolic acid (PGLA) particles using single or double emulsions, and porous media. The primary materials of choice for microfluidic devices are glass-based materials. Using one of their devices, researchers generated droplets for the purpose of RNA sequencing of human dental pulp stem cells and periodontal ligament stem cells.41 However, specific details regarding the fabrication of the glass-based devices were not provided. A different team subsequently produced Repaglinide (Rp) nanocrystals via a glass-based droplet generator, which helped the poor by assisting water-soluble drugs.42 Next, via mechanical phenotyping for cell sorting, a group employed a glass-based chip,39 which is depicted in Fig. 7(a). As shown in the figure, mechanical prototyping was carried out with the use of optics, with the laser pointing in the direction of the channel from both sides. The production of droplets at a cross junction, as well as the development of jetting to dripping and back to jetting, is shown in Fig. 7(b) and was subsequently investigated by another team of university researchers with similar interests.39 Similarly, various research groups used glass-based materials from Dolomite Microfluidics, as tabulated below. These methods allow researchers to conduct reproducible experiments without the expense and lead time of custom fabrication. The availability of off-the-shelf geometries and compatible instrumentation further reduces system integration effort and overall project cost. There are alternative fabrication routes which can be adapted based on the emphasized design priorities whether it is manufacturing scalability and disposability (thermoplastics), fast prototyping (PDMS), or maximum robustness (all-glass/silicon–glass) as each route optimizes a different constraint set.
| Company name |
Country |
Fabrication materials |
Fabrication process |
Applications |
| Dolomite microfluidics |
Royston, UK |
Glass |
NA |
RNA sequencing41 |
| Glass |
NA |
Repaglinide (Rp) nanocrystals were created42 |
| Glass |
Wet etching |
Single-cell sorting39 |
| Glass |
NA |
The flow characteristics of the formation of a droplet is studied40 |
 |
| | Fig. 7 Glass-based microfluidic devices fabricated by Dolomite Microfluidics, UK. (a) Chip used for mechanical phenotyping in single cell sorting with integrated optics. (b) Droplet generation at a microfluidic cross junction demonstrating jetting–dripping transitions, illustrating Dolomite's contribution to high-precision droplet generation and cell analysis. Adapted with permission from ref. 39 and 40. Copyright 2015 Royal Society of Chemistry, Copyright 2019 Springer Nature. | |
Femtosecond laser-based selective etching (SLE), multiphoton polymerization (MPP), 3D lasers lithography
Femtosecond laser-based selective etching (FLSE) microfluidics fabrication methods uses ultrafast laser pulses to precisely modify substrate at the microscale. Commercially available Femtika works in 3D microstructure manufacturing via femtosecond lasers. In biomedicine, photonics, micromechanics, and micro-optics, the fabrication of devices involves multiple steps. Initially, they employed the 3D laser lithography method; then, they performed precision cutting, microdrilling, and surface structuring/ablation for the functionalized surfaces. Next, there is selective glass etching (SLE) which is used in micromechanics, micro robotics, and microfluidics. Researchers have investigated the insertion of a mechanical valve inside microfluidic channels via the SLE technique43 as shown in Fig. 8(a), the SLE approach was used to produce three-dimensional microfluidic architectures within glass substrates which is challenging to achieve with conventional planar lithography. Though it comes at the expense of higher equipment cost and lower throughput relative to moulding-based approaches, these competencies particularly priced for integrated valves, organ-on-chip systems and optofluidic devics.
| Company name |
Country |
Fabrication materials |
Fabrication process |
Applications |
| Femtika |
Vilnius, Lithuania |
Glass |
Laser subtraction and printing |
Using additive manufacturing techniques to add mechanical valves43 |
| Amorphous UV-grade fused silica (UVFS)/glass |
Femtosecond laser-based selective laser etching |
Organ on a chip (liver)44 |
| PDMS and photopolymer SZ20280 photoresist as membrane |
Soft lithography and Femto laser-based SEI |
Organ on a chip with membrane45 |
| Glass |
Femtosecond laser-based selective laser etching |
Tesla Valve20 |
 |
| | Fig. 8 Microfluidic devices fabricated using femtosecond laser technologies by Femtika, Lithuania. (a) Integration of mechanical valves in glass channels using selective laser etching (SLE). (b) Liver-on-a-chip devices were created with combined laser glass microprocessing. (c) PDMS-based organ-on-a-chip with embedded nanomembranes fabricated via multiphoton polymerization. (d) Tesla valve structure etched into glass. These approaches highlight the versatility of femtosecond lasers for complex 3D microfluidics. Adapted with permission from ref. 20, 43, 44 and 45. Copyright 2022, 2020 MDPI, Copyright 2022 Hindawi (published by Wiley). | |
Soft lithography (PDMS), hot embossing (thermoplastics), and spark-assisted chemical engraving (SACE) for glass molds
Polymer based microfluidic devices can be fabricated using soft lithography, hot embossing, or other engraving methods that translated into commercial technologies. For example, FlowJEM Polymer Microfluidic Technology uses polymers as substrates for a wide range of device fabrication using thermoplastic materials and PDMS, and prebuilt designs such as Drop-seq and FlowSpec for different applications. As shown in Fig. 9a, to better understand the anticancer behavior of immune cells, a team of researchers created droplets containing single-cell RNA via Drop-seq technology.47 In this work, SU-8 patterned silicon master molds were employed within a soft lithography workflow to generate PDMS replicas, which subsequently served as embossing tools for thermoplastic microchannel fabrication. Building on this established approach, the authors demonstrated a one-step hot embossing strategy to produce three-dimensional thermoplastic microfluidic structures with integrated, controllable microfeatures. As shown in Fig. 9b, a similar fabrication philosophy was adopted by another group, who likewise utilized hot embossing following mold imprinting to construct thermoplastic microfluidic devices.48 In that case, glass molds were fabricated using Posalux's spark-assisted chemical engraving (SACE) technique prior to embossing.46 The fabrication methods, substrate materials, and representative applications of these devices are summarized in the table below.
| Company name |
Country |
Fabrication materials |
Fabrication process |
Applications |
| FlowJEM |
Ontario, Canada |
PDMS and silicon |
Soft lithography and photolithography for master mold |
Single-cell RNA-sequencing identifies anti-cancer immune phenotypes in the early lung metastatic niche during breast cancer47 |
| Thermoplastics (polycarbonate, cycloolefin copolymer and polystyrene) and imprint (glass) |
Thermoplastics and imprint templates were produced by FlowJEM |
Microfluidic analysis48 |
| Thermoplastics (polycarbonate, cycloolefin copolymer and polystyrene) and imprint (glass) |
Imprint production using sparks assisted chemical engraving (SAC) |
Different embossing material studies and conditions46 |
 |
| | Fig. 9 Polymer-based microfluidic devices fabricated by FlowJEM, Canada. The schematic shows (a) a PDMS-based single-cell RNA sequencing device created using soft lithography, (b) thermoplastic microstructures formed via hot embossing and spark-assisted chemical engraving (SACE). These platforms enable applications from single-cell analysis to microfluidic profile optimization. Adapted with permission from ref. 46. Copyright 2017 MDPI. | |
The reflection of this hybrid approach of soft lithography and thermoplastic embossing balances design flexibility with improved manufacturability, which is particularly attractive for academic prototyping transitioning toward small-scale production.
Silicon polymer and glass hybrid layered fabrication
Microfluidic devices can be fabricated using hybrid-layer soft lithography and etching to achieve precise and application-specific microstructures. Gesim manufactures microfluidic devices and bio-instruments. Researchers utilized the platform to investigate the effect of a narrowed downstream channel on the formation of non-Newtonian (xanthan-based) droplets at a T-junction.49 Owing to the interest of the chemical and food processing industries in non-Newtonian droplets, this study was conducted as seen in Fig. 10(a), they did it by using a silicon wafer to create the master mold and then a PDMS material for the soft lithography process.50 Another research group aimed to make channels through which the cells might be suspended in a homogenous acoustic fluid and subsequently separated according to the acoustic contrast.51 For this purpose, they used DRIE to build the channel in silicon, which was subsequently anodically bonded to the glass on both the top and bottom. A divergent team of scientists designed microfluidic channels with internal separation bars to divide the bubbles produced during the electrophoresis procedure.52 Here, a silicon wafer was used to construct the mold, which was then covered with a thermoplastic PC before the PDMS was poured into the mold to transfer the design. After the design is transferred, the silicon mold is removed, NaCl salt is added, an electrode-plated glass slide is affixed, and finally, the NaCl is removed to form the hollow channel inside the PDMS layer, as shown in Fig. 10(b). GESim's hybrid silicon–glass–polymer workflows demonstrate how traditional MEMS fabrication can be extended to accommodate multiphase and electrokinetic microfluidic applications.12
 |
| | Fig. 10 Illustration of microfluidic systems fabricated using master mold and multi-layer design by GESim, Germany. (a) Silicon-PDMS devices designed for studying non-Newtonian droplet flow at a T-junction. (b) An electrophoresis microchip featuring internal separation bars for bubble segregation, constructed using polycarbonate, PDMS, and glass layers, highlighting GESim's expertise in soft lithography and hybrid material integration. Adapted with permission from ref. 49 and 52. Copyright (1994 – Present) American Institute of Physics, Copyright 2011 Royal Society of Chemistry. | |
| Company name |
Country |
Fabrication materials |
Fabrication process |
Applications |
| GeSim |
Radeberg, Germany |
Silicon and PDMS |
Master mold on silicon using photolithography and then soft lithography using PDMS |
Study of the non-Newtonian droplet flow in the channel49 |
| Silicon and glass |
Silicon etched using DRIE |
Separation of cells using acoustic fluid51 |
| Three layers: polycarbonate, PDMS and glass |
Soft lithography |
Electrophoresis52 |
Micro milling (PMMA mold), soft lithography (PDMS), laser etching (ITO glass), lamination
Polymer micro-milling, soft lithography combined with glass laser etching can collectively enable rapid, scalable prototyping that can translate into commercial microfluidic fabrication. HiComp Microtech has microfluidic devices fabricated using polymer-based workflows.
In one study, PVA droplets were produced for microemboli modeling by crosslinking PVA with glutaraldehyde present in the oil phase at 38 °C, enabling droplet solidification.55 The device master mold was fabricated from PMMA using micromilling, followed by pattern transfer through PDMS soft lithography.
| Company name |
Country |
Fabrication materials |
Fabrication process |
Applications |
| HiComp microfluidic innovated/HiComp Microtech |
California, US/Suzhou, China |
PMMA and PDMS |
PMMA (master mold) etched using micromilling and PDMS through soft lithography |
Embolization treatment55 |
| PMMA and PET |
NA |
Immunoassay53 |
| PMMA, ITO glass, and adhesive tape |
Pulse laser to etch the ITO glass to form electrodes, and lamination microfabrication |
Dielectrophoretic54 |
| PDMS and glass |
Soft lithography |
Droplet generation and sorting56 |
As shown in Fig. 11a, in subsequent work, a PMMA-based microfluidic device incorporating an S-shaped channel, probe reaction zone, detection area, and waste reservoir was employed for immunoassay and fluorescence signal detection.53 The structured PMMA layer, supplied by HiComp Microtech, was sealed with a PET membrane to complete the device assembly. Fig. 11b shows, in another study, dielectrophoresis was used to recover cell-free plasma, utilizing an ITO-coated glass substrate with laser-patterned electrodes fabricated by HiComp Microtech.54 This platform combined a double-sided adhesive microfluidic layer with an acrylic top plate containing fluidic ports.
 |
| | Fig. 11 Microfluidic devices developed by HiComp Microtech, USA and China. (a) PMMA-based devices for immunoassays, featuring S-shaped channels and PET membranes for sample detection. (b) Dielectrophoretic microchip with ITO glass electrodes for cell-free plasma extraction, which demonstrates HiComp's expertise in MEMS and microfluidics. Adapted with permission from ref. 53 and 54. Copyright 2022 MDPI. | |
More recently, SU-8-patterned silicon master molds and PDMS soft lithography were used to fabricate flow-focusing devices for lentivirus droplet generation and subsequent target cell sorting.56
Silicon-glass hybrid microfluidics: wet etching, LPCVD deposition, femtosecond laser etching
Silicon and glass hybrid microfluidics usually utilizes silicon microfluidic structures with transparent glass substrates to enable optical accessibility lab-on-chip devices. LionIX International's work involves the integration of photonics in many applications, such as microfluidics/optofluidics. For example, to perform the immunoassay necessary to detect organic material on Mars, researchers have developped an optofluidic channel for the ExoMars expedition.57 As shown in Fig. 12(a), they were divided into two pieces. The fluidic channels and chambers are composed of fused silica in the top two layers. The bottom one is a silicon channel that contains the waveguides, and the Viton seal, which serves as a gasket seal, is used to attach the two. The silicon nitride waveguides were placed atop the oxidized silicon wafer and subsequently etched to create the desired structures. On the other hand, the polysilicon generated by (Low-Pressure Chemical Vapor Deposition) LPCVD is employed as a mask for the fused silica that is wet etched using the HF solution. Next, a different group employed LionIX's unique manufacturing method, TriPleX, as a waveguide.58 To find cancer cells in blood, this waveguide is combined with a disposable microfluidic cartridge. Here, an array of six asymmetric Mach–Zehnder Interferometer (aMZI) waveguides is employed to detect the change in the phase of the output signal. Another group synthesized gold nanoparticles (AuNPs) via droplet microreactors, which were discovered via ultraviolet/visible spectroscopy.59 They employed waveguides for this purpose, and Fig. 12(b) depicts the entire production process. The bottom half of the microfluidic channel was wet etched on the deposited silicon dioxide after the waveguide was first made with silicon nitride. The other silicon dioxide layer is then etched into the upper half, and the two layers are joined to create a fluidic channel. Another group used a commercially available (model D8-LIF from LionIX BV) lab-on-a-chip made from fused silica and integrated the waveguides that were manufactured by glass etched directly via a femtosecond laser.19
| Company name |
Country |
Fabrication materials |
Fabrication process |
Applications |
| LionIX International |
Enschede, Netherlands |
Fused silica, silicon, and silicon nitride |
Wet etched |
Immunoassay to detect the organic matter on Mars57 |
| Silicon and silicon nitride |
Microfluidic channels are created on silicon, which have deposition of silicon nitride by LPCVD |
Cancer cell detection58 |
| Fused silica and silicon nitride |
Wet etching, dry etching (RIE) and LPCVD |
Microreactors process analysis59 |
| Fused silica |
Femtosecond laser etching |
Lab on a chip19 |
 |
| | Fig. 12 Schematics of the optofluidic microfluidic platforms fabricated by LionIX International, Netherlands. (a) ExoMars lab-on-a-chip device integrating fused silica fluidic channels and silicon nitride waveguides for life detection assays. (b) Droplet microreactor with UV/Vi's spectroscopy for nanoparticle synthesis. Adapted with permission from ref. 57 and 59. Copyright 2013 Royal Society of Chemistry, Copyright 2011 SPIE Digital Library. | |
Microinjection molding and precision micromilling
Microinjection molding and precision micromilling can enable the rapid fabrication of microfluidic features on various substrates. Microfluidic ChipShop produces 3D microfluidic devices from thermoplastic polymers. As shown in Fig. 13(a), researchers sought to purify a DNA sample via magnetic particles in a microfluidic chip.60 The chips installed within the apparatus regulate both the device's temperature and the magnetic particles used for purification. The chip with the channel and the magnetic particles inside it is visible on the right side. A cyclic olefin polymer (COP) is used in the fabrication of the device. The HepaChip Microplate is the name of a microplate made by another group.61 This system has an automated dielectrophoretic system for separation and holds 24 of them in one device. Cyclic olefin polymer (COP) materials were used in the manufacturing of these chips, which are depicted in Fig. 13(b). The microinjection process method was used to produce the three-dimensional structure of the microfluidic device. Ultraprecision micromilling was used to build the mold, and with the use of a shadow mask, platinum and gold evaporated to form electrodes. A different group developed compounds with other compounds, such as benzoalphapyrene (BαP) and aflatoxin B1 (AFB1). Here, they used cyclic olefin polymer (COP) material to develop an organ-on-a-chip because of the flexibility of fabricating complex geometries, such as the vertical diffusion barriers needed to mimic an organ. A different team subsequently employed a microfluidic system with many well plates to analyze an organ-on-a-chip (kidney) to investigate the toxicity of drug metabolites in the kidney.62 The goal of this study was to investigate the toxicity, biotransformation, and interactions of these cells.63 Using Topas material and microinjection molding, they created a microfluidic device. As in the preceding group, the electrodes are transferred to the device via the shadow mask after being produced on the mold. Injection molding of thermoplastic materials such as cyclic olefin polymers (COP) enables scalable production of complex three-dimensional microfluidic devices, although it imposes design constraints related to mold complexity and tooling cost.
| Company name |
Country |
Fabrication materials |
Fabrication process |
Applications |
| Microfluidic chip shop |
Jena, Germany |
Cyclic olefin polymer (COP) |
NA |
DNA purification process60 |
| Cyclic olefin polymer (COP) |
Micro injection molding process |
Dielectrophoretic61 |
| Cyclic olefin polymer (COP) |
NA |
Organ on a chip62 |
| Cyclic olefin copolymer (COC) |
Microinjection molding process |
Cell analysis63 |
 |
| | Fig. 13 Thermoplastic-based microfluidic devices by Microfluidic chip shop, Germany. (a) DNA purification chips using magnetic particle separation and temperature regulation. (b) HepaChip microplate with automated dielectrophoretic systems for liver coculture models, which highlights the capabilities of precision micro milling and microinjection molding in commercial microfluidics. Adapted with permission from ref. 60 and 61. Copyright 2016 Elsevier, Copyright 2020 Royal Society of Chemistry. | |
UV-lithography mask, wet/dry etching, fusion bonding, UV-LIGA
Glass–glass and glass–silicon microfluidics that leverages wet and dry etching techniques can create high-precision microfluidic features for various microfluidic applications. Teledyne Micralyne, which specialises in MEMS and microfluidic device manufacturing using glass, silicon, and hybrid materials, has glass–glass, silicon–glass, silicon–silicon and SOI–glass microfabrication process for microfluidic device fabrication as shown in Fig. 14. The fabrication method follows the standard photolithography process, followed by stripping of both the photoresist and the metal mask, and the etched substrate aligned and thermally bonded to a cover glass to form enclosed microchannels. Researchers applied it to investigate the impact of the aspect ratio on the internal flow velocity in passive microfluidic mixers and valves via microparticle image velocimetry (micro-PIV).64 It was later used to compare the mixing efficiencies of various geometries under pulsatile versus steady flow conditions. Researchers employed a similar chip design to perform continuous dielectrophoretic separation of yeast cells using non-uniform electric fields.65
 |
| | Fig. 14 (a) Schematics of microfabrication capabilities at Teledyne Micralyne, including (i) multilevel silicon and glass processing, (ii) SOI processing for channel depth uniformity, (iii and iv) integration of glass or silicon ports, (v) electrode patterning for dielectrophoresis and (vi) embedded sensors, and full silicon processing. (b) Fabrication workflow of glass-based microfluidic devices, starting with a glass substrate, followed by etch mask deposition, photoresist coating, and patterning, dry/wet etching of channels, mask stripping, and final bonding to cover glass. | |
Silicon microfluidics and CMOS integration
Silicon and CMOS-integrated microfluidics can integrate microelectronics with fluid handling on-chip to enable compact lab-on-a-chip. Xfab specializes in semiconductor technology for analogue and mixed-signal applications. It includes the production of MEMS-based devices, where a complementary metal–oxide–semiconductor (CMOS) is used, and microfluidic device products integrated with CMOSs. CMOS is where the electronic components needed to control and sense the MEMS devices that are accommodated. This CMOS is preferred because it consumes less power and space than traditional electronic circuits do. Nearly all of the earlier microfluidic devices without an electrode contained only fluidic controlling and sensing components, whereas when an electrode is added, such as in Dielectrophoresis or electrophoresis, or even just for the purpose of sensing the fluid itself, a significant additional electricalcomponent is needed. A microfluidic device with CMOS integrated directly eliminates the need for extra electrical components, which is the desired future, and silicon and glass are easy to integrate with them. For this reason, we have focused on examining several publications that employ silicon or glass as the primary component of commercially purchased microfluidic devices. There are several papers that have used devices from Xfab. Italian scientists have employed a lab-on-a-chip microfluidic device based on CMOS technology to analyze DNA.66 The enzyme Telomerase, which maintains the length of Telomeres, was the one they were trying to measure. This silicon device features CMOS circuitry that combines temperature control systems, readout and apparent diffusion coefficient (ADC) circuitry, a telomerase biosensor, and other features. An additional study comes from the Xfab foundry, where researchers worked on adhesive wafer bonding using dry film resist (DFR).67 The DFR serves as both an adhesive to hold the top glass cap, which has fluidic channel ports, and the bottom silicon-based CMOS, as shown in Fig. 15(a). To provide the required height, the DFR material utilized in this instance is ORDYL SY330 resist, which is made via a lamination method and repeated three times to reach a height of 90 µm. A stretchable electrocardiogram (ECG) patch (SEP) was subsequently developed by a different group in Singapore.68 This featured tubes for the liquid metal (Galinstan), which melts at −19 °C, and a microfluidic structure based on PDMS. The SEP, which is integrated with the CMOS chip and connected to the patches via liquid metal-filled channels, is depicted in Fig. 15(b). The SEP is housed inside the PDMS device. This coarse liquid metal's benefit is that it expands with the PDMS, preventing any loss of connection between the electrode pads and the SEP. As shown in Fig. 15(b), the process of creating the mold occurs before the process of creating the PDMS channels, integrating the SEP and injecting Galinstan into the fluidic channels to work as the link between the SEP and the electrode patch. The integration of CMOS electronics with microfluidic channels shows a significant change from conventional lab-on-chip architectures, enabling compact, low-power systems for sensing applications where silicon-based platforms are used.
| Company name |
Country |
Fabrication materials |
Fabrication process |
Applications |
| Teledyne micralyne (micralyne technologies) |
Alberta, Canada |
Glass |
Wet etched and fusion-bonded |
Particle imaging velocimetry64 |
| Glass |
Wet etched and fusion-bonded |
Mixer flow in simulation and experiment69 |
| Glass |
Wet etched and fusion-bonded |
Cell separation70 |
| Glass |
Abrasive jet micromachining and wet etching |
The electro-osmotic flow limit of detection and separation efficiency71 |
| PEM-PMMA, glass and platinum electrodes |
UV-LIGA process |
Electrophoresis72 |
| Company name |
Country |
Fabrication materials |
Fabrication process |
Applications |
| Xfab |
Erfurt, Germany |
Silicon |
CMOS fabrication |
Lab on a chip66 |
| Silicon, ORDYL SY330 negative-type permanent dry-resist (DFR) and BF33 glass |
CMOS fabrication |
Cell sorting67 |
| Silicon and PDMS |
Soft lithography |
Wearable sensor68 |
 |
| | Fig. 15 Silicon and PDMS-based microfluidic devices developed by XFab, Germany. (a) CMOS integrated lab-on-a-chip device with adhesive wafer bonding for DNA analysis. (b) Stretchable electrocardiogram (ECG) patch featuring microfluidic liquid-metal interconnects and integrated sensors for wearable heart monitoring. These systems exemplify the integration of electronics and microfluidics for next-generation biomedical devices. Adapted with permission from ref. 67 and 68. Copyright 2020 IOP, Copyright 2019 Wiley. | |
Conclusion
This review examines the state of commercially and semi commercially available microfluidic device platforms which are fabricated using glass and silicon substrates. This work also highlights how the right material and fabrication strategy dictates the overall device performance, reliability and integration capability. While polymer-based systems dominate low-cost application and rapid prototyping, glass and silicon platforms remain indispensable for applications that require high chemical stability, optical fidelity, dimensional precision and compatibility with electronic and photonic components. Recently there has been efforts developing commercially viable microfluidic technology which can seamlessly integrate MEMS (microelectromechanical system) and CMOS (complementary metal–oxide–semiconductor) fabrication processes. Unlike traditional microfluidic devices that rely on external instrumentation for sensing, actuation, or data acquisition, these methods have the potential to embed on-chip temperature control, electrophoretic and dielectrophoretic actuation, integrated optical/electrical sensors, and signal processing circuits directly within the microfluidic substrate. Such monolithic integration allows for compact, low-power, and multifunctional lab-on-a-chip systems, paving the way for applications for true portable diagnostics, wearable biosensors, and real-time monitoring systems. Another emerging trend is the increasing adoption of hybrid material systems. Here glass and silicon substrates are combined with polymers, elastomers, or functional coatings to balance mechanical robustness, chemical resistance, and manufacturability. Such hybrid strategies enable the complementary advantages of different material classes to the designers. It is effective for organ-on-chip platforms, integrated sensors, and multifunctional lab-on-a-chip devices.
From an industrial perspective, the successful translation of microfluidic technologies from laboratory demonstrations to deployable products increasingly depends on process standardization, reproducibility, and system-level integration.73 Recent reviews emphasize the essential need for bridging the gap between prototyping and scalable manufacturing. Particularly for high-performance glass- and silicon-based platforms with integrated microelectronics. As microfluidics continues to mature, the integration of advanced materials, scalable fabrication workflows, and embedded electronics is expected to define the next generation of high-impact microfluidic technologies.
Conflicts of interest
Teledyne MEMS is one of the funders of this research.
Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.
References
- A. Agarwal, A. Salahuddin, H. Wang and M. J. Ahamed, Design and development of an efficient fluid mixing for 3D printed lab-on-a-chip, Microsyst. Technol., 2020, 26(8), 2465–2477, DOI:10.1007/s00542-020-04787-9.
- G. M. Whitesides, The origins and the future of microfluidics, Nature, 2006, 442(7101), 368–373, DOI:10.1038/nature05058.
- N. Shembekar, C. Chaipan, R. Utharala and C. A. Merten, Droplet-based microfluidics in drug discovery, transcriptomics and high-throughput molecular genetics, Lab Chip, 2016, 16(8), 1314–1331, 10.1039/C6LC00249H.
- P. Tirandazi and C. H. Hidrovo, An integrated gas-liquid droplet microfluidic platform for digital sampling and detection of airborne targets, Sens. Actuators, B, 2018, 267, 279–293, DOI:10.1016/j.snb.2018.03.057.
- L. Yang, L. Zhu, Z. Li and B. Lu, A liquid molding method for the fabrication of microfluidic devices based on a drop-on-demand generation of patterned substrates, Microsyst. Technol., 2017, 23(10), 4543–4551, DOI:10.1007/s00542-016-3242-3.
- L. Donvito, L. Galluccio, A. Lombardo, G. Morabito, A. Nicolosi and M. Reno, Experimental validation of a simple, low-cost, T-junction droplet generator fabricated through 3D printing, J. Micromech. Microeng., 2015, 25(3), 035013, DOI:10.1088/0960-1317/25/3/035013.
- A. Yang, et al., Pressure-controlled microfluidics for automated single-molecule sample preparation, HardwareX, 2023, 14, e00425, DOI:10.1016/j.ohx.2023.e00425.
- M. Jin, et al., Super-Hydrophobic PDMS Surface with Ultra-Low Adhesive Force, Macromol. Rapid Commun., 2005, 26(22), 1805–1809, DOI:10.1002/marc.200500458.
- S. Aralekallu, R. Boddula and V. Singh, Development of glass-based microfluidic devices: A review on its fabrication and biologic applications, Mater. Des., 2023, 225, 111517, DOI:10.1016/j.matdes.2022.111517.
- M. J. Ahamed, S. I. Gubarenko, R. Ben-Mrad and P. Sullivan, A Piezoactuated Droplet-Dispensing Microfluidic Chip, J. Microelectromech. Syst., 2010, 19(1), 110–119, DOI:10.1109/JMEMS.2009.2036866.
- Z. Mao, K. Yoshida and J. Kim, Developing O/O (oil-in-oil) droplet generators on a chip by using ECF (electro-conjugate fluid) micropumps, Sens. Actuators, B, 2019, 296, 126669, DOI:10.1016/j.snb.2019.126669.
- T. Tang, Y. Yuan, Y. Yalikun, Y. Hosokawa, M. Li and Y. Tanaka, Glass based micro total analysis systems: Materials, fabrication methods, and applications, Sens. Actuators, B, 2021, 339, 129859, DOI:10.1016/j.snb.2021.129859.
- E. M. Chan, A. P. Alivisatos and R. A. Mathies, High-Temperature Microfluidic Synthesis of CdSe Nanocrystals in Nanoliter Droplets, J. Am. Chem. Soc., 2005, 127(40), 13854–13861, DOI:10.1021/ja051381p.
- A.-G. Niculescu, C. Chircov, A. C. Bîrcă and A. M. Grumezescu, Fabrication and Applications of Microfluidic Devices: A Review, Int. J. Mol. Sci., 2021, 22(4), 2011, DOI:10.3390/ijms22042011.
- H. V. Nguyen, H. Q. Nguyen, V. D. Nguyen and T. S. Seo, A 3D printed screw-and-nut based droplet generator with facile and precise droplet size controllability, Sens. Actuators, B, 2019, 296, 126676, DOI:10.1016/j.snb.2019.126676.
- M. J. Ahamed, R. Ben-Mrad and P. Sullivan, A Drop-on-Demand-Based Electrostatically Actuated Microdispenser, J. Microelectromech. Syst., 2013, 22(1), 177–185, DOI:10.1109/JMEMS.2012.2221681.
- A. Francesko, V. F. Cardoso, and S. Lanceros-Méndez, Lab-on-a-chip technology and microfluidics, in Microfluidics for Pharmaceutical Applications, Elsevier, 2019, pp. 3–36, DOI:10.1016/B978-0-12-812659-2.00001-6.
- Z. Akbari, M. A. Raoufi, S. Mirjalali and B. Aghajanloo, A review on inertial microfluidic fabrication methods, Biomicrofluidics, 2023, 17(5), 051504, DOI:10.1063/5.0163970.
- R. M. Vazquez, et al., Integration of femtosecond laser written optical waveguides in a lab-on-chip, Lab Chip, 2009, 9(1), 91–96, 10.1039/B808360F.
- D. Andriukaitis, et al., Fabrication of Microfluidic Tesla Valve Employing Femtosecond Bursts, Micromachines, 2022, 13(8), 1180, DOI:10.3390/mi13081180.
- N. Sandström, et al., Live single cell imaging assays in glass microwells produced by laser-induced deep etching, Lab Chip, 2022, 22(11), 2107–2121, 10.1039/D2LC00090C.
- E. Kara, A. İhsan Kutlar and K. Güngör, Construction of a novel microfluidic experimental setup for testing recent glaucoma drainage devices, Curr. Dir. Biomed. Eng., 2019, 5(1), 219–222, DOI:10.1515/cdbme-2019-0056.
- M. Hébert, W. Baxter, J. P. Huissoon and C. L. Ren, A Quantitative study of the dynamic response of soft tubing for pressure-driven flow in a microfluidics context, Microfluid. Nanofluid., 2020, 24(12), 90, DOI:10.1007/s10404-020-02396-6.
- Y. Fintschenko, Education: a modular approach to microfluidics in the teaching laboratory, Lab Chip, 2011, 11(20), 3394, 10.1039/c1lc90069b.
- A. Verma, T. Mateo, J. Quintero Botero, N. Mohankumar and T. P. Fraccia, Microfluidics-Based Drying–Wetting Cycles to Investigate Phase Transitions of Small Molecules Solutions, Life, 2024, 14(4), 472, DOI:10.3390/life14040472.
- V. Girod, et al., A self-purifying microfluidic system for identifying drugs acting against adult schistosomes, R. Soc. Open Sci., 2022, 9(11), 220648, DOI:10.1098/rsos.220648.
- P. Hu, E. Fabyanic, D. Y. Kwon, S. Tang, Z. Zhou and H. Wu, Dissecting Cell-Type Composition and Activity-Dependent Transcriptional State in Mammalian Brains by Massively Parallel Single-Nucleus RNA-Seq, Mol. Cell, 2017, 68(5), 1006–1015, DOI:10.1016/j.molcel.2017.11.017.
- K. Ren, J. Zhou and H. Wu, Materials for Microfluidic Chip Fabrication, Acc. Chem. Res., 2013, 46(11), 2396–2406, DOI:10.1021/ar300314s.
- G. Jenkins and C. D. Mansfield, Microfluidic Diagnostics: Methods and Protocols, in Methods in Molecular Biology, vol. 949. Totowa, NJ, Humana Press, 2013, DOI:10.1007/978-1-62703-134-9.
- Microfluidic Diagnostics: Methods and Protocols, in Methods in Molecular Biology, ed. G. Jenkins and C. D. Mansfield, vol. 949, Totowa, NJ, Humana Press, 2013, DOI:10.1007/978-1-62703-134-9.
- C. Szydzik, et al., Fabrication of complex PDMS microfluidic structures and embedded functional substrates by one-step injection moulding, RSC Adv., 2016, 6(91), 87988–87994, 10.1039/C6RA20688C.
- Y.-H. Shih et al., Microfluidic chip fabrication for silicon mold insert by micro hot embossing, in 2008 3rd IEEE International Conference on Nano/Micro Engineered and Molecular Systems, Sanya: IEEE, 2008, pp. 690–693, DOI:10.1109/NEMS.2008.4484423.
- K. Woodruff, L. M. Fidalgo, S. Gobaa, M. P. Lutolf and S. J. Maerkl, Live mammalian cell arrays, Nat. Methods, 2013, 10(6), 550–552, DOI:10.1038/nmeth.2473.
- Glass versus polymers, Microfluidic ChipShop, 2013, accessed: Sep. 06, 2024, Online, Available: https://www.microfluidic-chipshop.com/microfluidics/materials-in-microfluidics/glass-versus-polymers/ Search PubMed.
- Polymers in Microfluidics, Microfluidic ChipShop, accessed: Sep. 06, 2024, Online, Available: https://www.microfluidic-chipshop.com/microfluidics/materials-in-microfluidics/polymers-in-microfluidics/ Search PubMed.
- X. Qiu, J. De Jesus, M. Pennell, M. Troiani and J. B. Haun, Microfluidic device for mechanical dissociation of cancer cell aggregates into single cells, Lab Chip, 2015, 15(1), 339–350, 10.1039/C4LC01126K.
- J. Ly, M. Masterman-Smith, R. Ramakrishnan, J. Sun, B. Kokubun and R. M. Van Dam, Automated Reagent-Dispensing System for Microfluidic Cell Biology Assays, SLAS Technol., 2013, 18(6), 530–541, DOI:10.1177/2211068213504758.
- S. Begolo, F. Shen and R. F. Ismagilov, A microfluidic device for dry sample preservation in remote settings, Lab Chip, 2013, 13(22), 4331, 10.1039/c3lc50747e.
- C. Faigle, F. Lautenschläger, G. Whyte, P. Homewood, E. Martín-Badosa and J. Guck, A monolithic glass chip for active single-cell sorting based on mechanical phenotyping, Lab Chip, 2015, 15(5), 1267–1275, 10.1039/C4LC01196A.
- N. M. Kovalchuk, M. Sagisaka, K. Steponavicius, D. Vigolo and M. J. H. Simmons, Drop formation in microfluidic cross-junction: jetting to dripping to jetting transition, Microfluid. Nanofluid., 2019, 23(8), 103, DOI:10.1007/s10404-019-2269-z.
- S. Lee, et al., Single-Cell RNA Sequencing Analysis of Human Dental Pulp Stem Cell and Human Periodontal Ligament Stem Cell, J. Endod., 2022, 48(2), 240–248, DOI:10.1016/j.joen.2021.11.005.
- M. Ahmad, et al., Formulation and Optimization of Repaglinide Nanoparticles Using Microfluidics for Enhanced Bioavailability and Management of Diabetes, Biomedicines, 2023, 11(4), 1064, DOI:10.3390/biomedicines11041064.
- L. Hernandez-Cedillo, et al., Peculiarities of Integrating Mechanical Valves in Microfluidic Channels Using Direct Laser Writing, Appl. Bionics Biomech., 2022, 2022, 1–8, DOI:10.1155/2022/9411024.
- A. Butkutė, et al., Combined Femtosecond Laser Glass Microprocessing for Liver-on-Chip Device Fabrication, Materials, 2023, 16(6), 2174, DOI:10.3390/ma16062174.
- L. Bakhchova, et al., Femtosecond Laser-Based Integration of Nano-Membranes into Organ-on-a-Chip Systems, Materials, 2020, 13(14), 3076, DOI:10.3390/ma13143076.
- L. Hof, X. Guo, M. Seo, R. Wüthrich and J. Greener, Glass Imprint Templates by Spark Assisted Chemical Engraving for Microfabrication by Hot Embossing, Micromachines, 2017, 8(1), 29, DOI:10.3390/mi8010029.
- S. M. Orbach, et al., Single-cell RNA-sequencing identifies anti-cancer immune phenotypes in the early lung metastatic niche during breast cancer, Clin. Exp. Metastasis, 2022, 39(6), 865–881, DOI:10.1007/s10585-022-10185-4.
- M. Debono, et al., One-Step Fabrication of Microchannels with Integrated Three Dimensional Features by Hot Intrusion Embossing, Sensors, 2016, 16(12), 2023, DOI:10.3390/s16122023.
- P. Kumari and A. Atta, Non-Newtonian droplet breakup in a T-junction microdevice containing constriction induced asymmetric parallel branches, Phys. Fluids, 2023, 35(2), 022004, DOI:10.1063/5.0135186.
- E. Chiarello, L. Derzsi, M. Pierno, G. Mistura and E. Piccin, Generation of Oil Droplets in a Non-Newtonian Liquid Using a Microfluidic T-Junction, Micromachines, 2015, 6(12), 1825–1835, DOI:10.3390/mi6121458.
- M. Rezayati Charan, F. Berg and P. Augustsson, Acoustofluidic Three-Dimensional Motion of Suspended Cells at Near-Zero Acoustic Contrast in Homogeneous Media, Phys. Rev. Appl., 2023, 19(1), 014046, DOI:10.1103/PhysRevApplied.19.014046.
- S. Köhler, C. Weilbeer, S. Howitz, H. Becker, V. Beushausen and D. Belder, PDMS free-flow electrophoresis chips with integrated partitioning bars for bubble segregation, Lab Chip, 2011, 11(2), 309–314, 10.1039/C0LC00347F.
- X. Wang, D. Lu, Q. Huang and J. Yang, Microfluidics-Based Time-Resolved Fluorescence Immunoassay for the On-Site Detection of Aflatoxins B1 Zearalenone and Deoxynivalenol in Cereals, Foods, 2022, 11(9), 1319, DOI:10.3390/foods11091319.
- F. Yang, et al., Extraction of Cell-Free Whole Blood Plasma Using a Dielectrophoresis-Based Microfluidic Device, Biotechnol. J., 2019, 14(3), 1800181, DOI:10.1002/biot.201800181.
- Y. Luo, et al., Shape-Anisotropic Microembolics Generated by Microfluidic Synthesis for Transarterial Embolization Treatment, Adv. Healthcare Mater., 2022, 11(10), 2102281, DOI:10.1002/adhm.202102281.
- M. Lyu, et al., Generation and Screening of Antigen-Specific Nanobodies from Mammalian Cells Expressing the BCR Repertoire Library Using Droplet-Based Microfluidics, Anal. Chem., 2022, 94(22), 7970–7980, DOI:10.1021/acs.analchem.2c00865.
- A. Prak, H. Leeuwis, R. G. Heideman, A. Leinse, and G. Borst, Integration of optical waveguides and microfluidics in a miniaturized antibody micro-array system for life detection in the NASA/ESA ExoMars mission, ed. Presented at the SPIE MOEMS-MEMS, S. Garcia-Blanco and R. Ramesham, San Francisco, California, 2011, p. 79280L, DOI:10.1117/12.879309.
- M. Chatzipetrou, et al., A Miniature Bio-Photonics Companion Diagnostics Platform for Reliable Cancer Treatment Monitoring in Blood Fluids, Sensors, 2021, 21(6), 2230, DOI:10.3390/s21062230.
- J. Yue, F. H. Falke, J. C. Schouten and T. A. Nijhuis, Microreactors with integrated UV/Vis spectroscopic detection for online process analysis under segmented flow, Lab Chip, 2013, 13(24), 4855, 10.1039/c3lc50876e.
- S. Julich, et al., Evaluation of a microfluidic chip system for preparation of bacterial DNA from swabs, air, and surface water samples, Biologicals, 2016, 44(6), 574–580, DOI:10.1016/j.biologicals.2016.06.013.
- M. Busche, et al., HepaChip-MP – a twenty-four chamber microplate for a continuously perfused liver coculture model, Lab Chip, 2020, 20(16), 2911–2926, 10.1039/D0LC00357C.
- J. Theobald, et al., Liver-Kidney-on-Chip To Study Toxicity of Drug Metabolites, ACS Biomater. Sci. Eng., 2018, 4(1), 78–89, DOI:10.1021/acsbiomaterials.7b00417.
- A. L. Nair, et al., Parallelizable Microfluidic Platform to Model and Assess In Vitro Cellular Barriers: Technology and Application to Study the Interaction of 3D Tumor Spheroids with Cellular Barriers, Biosensors, 2021, 11(9), 314, DOI:10.3390/bios11090314.
- P. Chiarot, P. Sullivan, and R. Ben Mrad, Micro Particle Imaging Velocimetry Measurements in High Aspect Ratio Passive Microfluidic Components, in ASME 3rd International Conference on Microchannels and Minichannels, Part B Cont'd, Toronto, Ontario, Canada, ASMEDC, 2005, pp. 481–486, DOI:10.1115/ICMM2005-75137.
- Y. Li, C. Dalton, H. J. Crabtree, G. Nilsson and K. V. I. S. Kaler, Continuous dielectrophoretic cell separation microfluidic device, Lab Chip, 2007, 7(2), 239–248, 10.1039/B613344D.
- S. Sonedda, C. Napoli, and M. Barbaro, A CMOS Lab-on-a-Chip for Fully Automated Telomerase Activity Detection, in 2019 IEEE Biomedical Circuits and Systems Conference (BioCAS), Nara, Japan, IEEE, 2019, pp. 1–4, DOI:10.1109/BIOCAS.2019.8918729.
- W. J. Karl, M. Schikowski, J.-E. Thon and R. Knechtel, Adhesive wafer bonding for CMOS based lab-on-a-chip devices, Jpn. J. Appl. Phys., 2020, 59(SB), SBBD04, DOI:10.7567/1347-4065/ab591b.
- Y. Li, et al., A Stretchable-Hybrid Low-Power Monolithic ECG Patch with Microfluidic Liquid-Metal Interconnects and Stretchable Carbon-Black Nanocomposite Electrodes for Wearable Heart Monitoring, Adv. Electron. Mater., 2019, 5(2), 1800463, DOI:10.1002/aelm.201800463.
- P. Chiarot, R. B. Mrad, and P. Sullivan, A Study of Passive Microfluidic Mixers, in 2004 International Conference on MEMS, NANO and Smart Systems (ICMENS’04), Banff, AB, Canada, IEEE, 2004, pp. 287–293, DOI:10.1109/ICMENS.2004.1508963.
- Y. Li and K. V. I. S. Kaler, A microfluidic channel for cell separation, in Proceedings of the 25th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (IEEE Cat. No.03CH37439), Cancun, Mexico, IEEE, 2003, pp. 3403–3406, DOI:10.1109/IEMBS.2003.1280876.
- A. Ghobeity, H. J. Crabtree, M. Papini and J. K. Spelt, Characterisation and comparison of microfluidic chips formed using abrasive jet micromachining and wet etching, J. Micromech. Microeng., 2012, 22(2), 025014, DOI:10.1088/0960-1317/22/2/025014.
- C. A. Currie, et al., Comparing polyelectrolyte multilayer-coated PMMA microfluidic devices and glass microchips for electrophoretic separations, Electrophoresis, 2009, 30(24), 4245–4250, DOI:10.1002/elps.200900403.
- Y. Ma, et al., Transformation gap from research findings to large-scale commercialized products in microfluidic field, Mater. Today Bio, 2024, 29, 101373, DOI:10.1016/j.mtbio.2024.101373.
|
| This journal is © The Royal Society of Chemistry 2026 |
Click here to see how this site uses Cookies. View our privacy policy here.  Open Access Article
Open Access Article a,
Gnanesh Nagesh
a,
Gnanesh Nagesh a,
Tyler Harrisonb,
Dean Spicerb,
David S.-K. Ting
a,
Tyler Harrisonb,
Dean Spicerb,
David S.-K. Ting a and
Mohammed Jalal Ahamed
a and
Mohammed Jalal Ahamed *a
*a





![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (high-aspect structures shown)
1 (high-aspect structures shown)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)
1)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (by wall taper constraint)
1 (by wall taper constraint)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1









