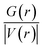Open Access Article
Open Access ArticleChemical tailoring of heteroatom (P, S, Si) doping of COF-PEDOT for adsorption of paracetamol: perspective from DFT studies
Innocent E. Emeng *a,
Uduak Lukeb,
Nguuma I. Gberc,
Onyinyechi V. Ugochukwu
*a,
Uduak Lukeb,
Nguuma I. Gberc,
Onyinyechi V. Ugochukwu d,
Ambika Sundaravadivelue,
Gopinath Sampathkumarf and
Musa Rundedg
d,
Ambika Sundaravadivelue,
Gopinath Sampathkumarf and
Musa Rundedg
aDepartment of Human Physiology, University of Calabar, Calabar, Nigeria. E-mail: innocentekoro@gmail.com
bDepartment of Biochemistry, University of Uyo, Uyo, Nigeria
cDepartment of Microbiology, University of Calabar, Calabar, Nigeria
dDepartment of Chemistry, National Open University of Nigeria, Abuja, Nigeria
eDepartment of Chemistry, M. Kumarasamy College of Engineering (Autonomous), Karur - 639113, India
fDepartment of Chemistry, Chettinad College of Engineering & Technology, Karur - 639114, India
gDepartment of Research Analytics, Saveetha Dental College and Hospitals, Saveetha Institute of Medical and Technical Sciences, Saveetha University, Chennai, India
First published on 4th February 2026
Abstract
The growing occurrence of pharmaceutical contaminants in aquatic systems has intensified the demand for advanced nanostructured materials capable of selective adsorption and removal of such pollutants. Paracetamol, a commonly used analgesic and antipyretic drug, is frequently detected in wastewater and poses severe ecological and health risks when accumulated in the water bodies and the soil. In this study, density functional theory (DFT) calculations were employed to investigate the structural, electronic, and adsorption behaviors of phosphorus (P), sulfur (S), and silicon (Si) doped COF-PEDOT frameworks for paracetamol adsorption. All geometries were optimized using the PBE0-D3/6-311G(d) basis set. The optimized structures exhibited minimal distortion after adsorption, indicating stable interactions between the adsorbate and the doped surfaces. Density of states (DOS) analysis revealed that heteroatom incorporation enhanced the electronic activity and reactivity of the complexes, while frontier molecular orbital (FMO) analysis showed a notable narrowing of the energy gap, confirming improved electron transfer capability. The ionization potential values (5.29–5.44 eV) remained within the range of moderately stable adsorbents. Natural bond orbital (NBO) analysis indicated that phosphorus doping produced the highest orbital stabilization energies, suggesting stronger donor–acceptor interactions. Adsorption energy calculations yielded negative values for all systems, confirming exothermic and thermodynamically favorable adsorption processes. Furthermore, quantum theory of atoms in molecules (QTAIM) and non-covalent interaction (NCI) analyses demonstrated the presence of weak but stable van der Waals and hydrogen-bond interactions governing paracetamol adsorption. The results demonstrate that tailored heteroatom doping can effectively tune the electronic and adsorption characteristics of COF-PEDOT frameworks. The P-, S-, and Si-doped systems exhibit enhanced sensitivity, stability, and reversibility, making them promising candidates for the selective adsorption of paracetamol from pharmaceutical contaminants in aquatic environments.
1 Introduction
Pharmaceutical contaminants have increasingly become a serious environmental concern due to their persistent release into aquatic ecosystems through various routes. Improper disposal of expired or unused drugs, hospital effluents, pharmaceutical manufacturing wastewater, and domestic sewage contribute significantly to the accumulation of these pollutants in surface and groundwater systems.1 Paracetamol is a widely used pharmaceutical that is often detected in wastewater due to its incomplete removal in conventional treatment processes. Its persistence poses environmental and ecological risks, thereby creating a need for advanced materials with improved adsorption efficiency and selectivity. Paracetamol enters aquatic environments mainly through wastewater treatment plants that are often inefficient in removing such micro-pollutants.2 Prolonged exposure to paracetamol-contaminated water has been reported to induce oxidative stress, endocrine disruption, and reproductive impairments in aquatic organisms. The prolonged exposure can alter antioxidant defense systems, leading to elevated reactive oxygen species (ROS), and cellular damage in fish and invertebrates. Paracetamol has also been shown to interfere with hormone regulation, affecting growth, metabolism, and developmental processes.3 Also, chronic accumulation of paracetamol in aquatic environments may impair fertility, and disrupt early life stages, thereby posing long-term risks to aquatic biodiversity and ecosystem stability in the long run. The persistence of paracetamol in water bodies also raises concerns for human health, as contaminated surface and groundwater can serve as sources of drinking water. Long-term human exposure to this contamination, even at trace levels, may contribute to cumulative toxicity, liver and kidney stress, and potential endocrine-related effects, emphasizing the need for effective removal strategies to protect both environmental and public health.3 Moreover, its biotransformation products can accumulate in sediments and enter the food chain, posing indirect risks to human health such as liver dysfunction, carcinogenic effects, and antimicrobial resistance development.4 On a global scale, pharmaceutical pollution is now recognized as a “planetary boundary threat,” with studies indicating contamination in more than 70% of sampled rivers worldwide.5 The collective ecological and health burden resulting from these contaminants further highlights the urgent need for advanced adsorption materials capable of effectively detecting and removing paracetamol and related pharmaceuticals from aquatic environments.Recent research has focused on nanostructured materials as efficient adsorbents for pharmaceutical wastewater treatment. Among them, covalent organic frameworks (COFs) have attracted significant attention owing to their high surface area, chemical tunability, and porous architectures that enable selective adsorption of organic pollutants.6 For instance, Niculescu, et al. (2023) reported the successful removal of paracetamol using a β-ketoenamine-linked COF with a maximum adsorption capacity of 284 mg g−1, though the regeneration efficiency was limited after multiple cycles.7 Similarly, Geng, Z. (2025) designed a sulfur-doped COF composite that exhibited enhanced adsorption performance toward non-steroidal anti-inflammatory drugs (NSAIDs) but showed poor stability under variable pH conditions.8 Many other studies have integrated conductive polymers such as poly(3,4-ethylenedioxythiophene) (PEDOT) with COFs to form COF-PEDOT hybrids, improving electronic conductivity and adsorption kinetics performed by Cao, et al.,.9 Despite these advances, the adsorption efficiency of these systems is often hindered by weak interactions between pollutant molecules and the adsorbent surface, limited recyclability, and insufficient understanding of the adsorption mechanisms at the molecular level.10,11 Experimental work on COFs and PEDOT composites supports the feasibility of using hybrid conductive materials for pharmaceutical capture and sensing.33 Tunable COF chemistries have been shown to affect paracetamol adsorption and degradation, while PEDOT-based films exhibit enhanced adsorbate interaction and electrochemical responsiveness.34 These experimental demonstrations motivated this work's DFT-driven approach to map how heteroatom doping (P, S, Si) tunes adsorption energetics and electronic response, thereby providing molecular-level guidance to synthesis and performance optimization.
Despite the reported adsorption capabilities of existing materials, key limitations remain in understanding how electronic structure alteration governs paracetamol binding at the molecular level. This study addresses this gap by providing a first-principles framework for the rational design of high-performance COF-PEDOT adsorbents through systematic heteroatom (P, S, and Si) doping. Using density functional theory (DFT), it can be explained how targeted chemical tailoring alters charge distribution, frontier orbital alignment, and interfacial interaction strength with paracetamol. The comparative evaluation of multiple dopants enables identification of the most effective electronic environment for adsorption, offering mechanistic insight rather than empirical observation. By establishing direct structure–property–adsorption relationships, this work advances the design principles of COF-based nanomaterials and provides a transferable strategy for developing efficient, selective, and environmentally sustainable adsorbents for pharmaceutical wastewater remediation of paracetamol.
2 Method
The research was conducted using computational simulations via the density functional theory (DFT) as executed in the Gaussian 09 suite. All geometry optimizations were performed using the hybrid PBE0 functional with Grimme's D3 dispersion correction and a 6-311G(d) basis set (PBE0-D3/6-311G(d)), which ensures accurate treatment of the electronic structure and non-covalent interactions that are relevant to adsorption phenomena.12 DFT was employed to study paracetamol adsorption on P-, S-, and Si-doped COF-PEDOT. This method accurately captures electronic interactions and adsorption energies, offering insights beyond those from semi-empirical or classical methods used in previous reports. The COF-PEDOT framework was selected as the model nanomaterial due to its combined structural rigidity, extended π-conjugation, and intrinsic electrical conductivity, which are desirable properties for adsorption and sensing applications. Conductive polymer–COF hybrids such as PEDOT-functionalized frameworks have been shown to exhibit enhanced charge transport and stronger electronic coupling with aromatic pharmaceutical molecules, resulting in improved adsorption sensitivity and signal transduction (Zhang et al., J. Mater. Chem. C, 2021, 9, 11234–11243). Heteroatom doping was introduced to deliberately tune the electronic structure and adsorption behavior of COF-PEDOT. Phosphorus was selected because P-doping introduces electron-rich sites and localized charge polarization, which can enhance donor–acceptor interactions with oxygen-containing molecules such as paracetamol (Wang et al., RSC Adv., 2020, 10, 27645–27653). Sulphur was chosen due to its high polarizability and ability to participate in non-covalent interactions, including hydrogen bonding and π–sulphur interactions, which have been reported to improve adsorption strength in sensing materials (Liu et al., New J. Chem., 2019, 43, 17821–17829). Silicon doping was included to evaluate the effect of a less electronegative heteroatom, as Si incorporation has been shown to modulate band structure and surface reactivity without significantly disrupting the framework geometry (Chen et al., Phys. Chem. Chem. Phys., 2021, 23, 14678–14686). A low dopant concentration corresponding to single-atom substitution was employed to preserve the structural integrity of the COF-PEDOT framework while enabling direct comparison of electronic and adsorption properties across all systems, consistent with previous DFT studies on heteroatom-doped porous materials (Sun et al., RSC Adv., 2018, 8, 38967–38975). Dopant atoms were positioned at chemically active sites within the framework to maximize electronic perturbation and interaction with the adsorbate. Paracetamol was optimized independently at the same level of theory to ensure consistency. Adsorption energies, frontier molecular orbital properties, NBO charge transfer, and QTAIM/NCI analyses were subsequently used to elucidate the nature and strength of interactions between paracetamol and pristine and doped COF-PEDOT systems. These analyses provide direct insight into how heteroatom doping influences binding strength and electronic response upon adsorption. The optimized Cartesian coordinates of all structures are provided in the SI to ensure reproducibility.3 Results and discussion
3.1 Geometry optimization
Geometry optimization provides fundamental insights into how paracetamol (PCL) interacts with COF-PEDOT and heteroatom-doped (P, S, Si) COF-PEDOT frameworks. Structural responses such as shifts in bond lengths and bond angles reflect the degree of electronic rearrangement and the nature of interaction occurring upon adsorption.13,14 Across all systems, the optimized structures before and after PCL adsorption showed only minor geometric perturbations, indicating that adsorption does not distort the COF-PEDOT backbone but still induces measurable electronic adjustments at the active sites. In pristine COF-PEDOT, the characteristic C–S bond (S130–C127) retained its length at 1.75 Å before and after adsorption, signifying structural strength of the thiophene unit. The B–O bonds (e.g., O69–B75), which define the COF linker environment, also remained within the expected 1.36–1.39 Å range, with negligible deviation after adsorption.15 These results suggest that adsorption primarily occurs through weak noncovalent forces without altering the main COF framework. Phosphorus doping introduced more pronounced local changes. The P–O bond (P159–O32) increased slightly from 1.67 Å to 1.68 Å following the adsorption of paracetamol there was an angle shift from 98.44° to 99.32°. These distortions imply enhanced electron redistribution around the phosphorus site, consistent with P acting as an electron-rich doping center capable of interacting with the phenolic –OH and amide groups of PCL.16. In the S-doped system, subtle deviations were also observed, such as the B74–O68–C72 angle increasing by approximately 1°. Sulfur's polarizability allows greater flexibility in structural response, enabling moderate charge rearrangement during adsorption. For Si-doped COF-PEDOT, slight reductions in the Si–O bond length (1.86 Å → 1.85 Å) and bond angles reflect silicon's tendency to form flexible, partially polar bonds. This mild structural adjustment supports weaker binding relative to P- and S-doped systems. Overall, geometry analysis suggests the following trend in local structural responsiveness during adsorption: P-doped > S-doped > Si-doped > pristine COF-PEDOT. This ranking correlates strongly with later electronic property analysis and explains why phosphorus-doped materials demonstrate the most favorable adsorption characteristics (Table 1 and Fig. 1).| Compound before adsorption | Bonding atoms | Bond angle atoms | Bond-length (Å) | Bond-angle (Å) | Compound-after adsorption | Bond-length (Å) | Bond-angle (Å) |
|---|---|---|---|---|---|---|---|
| COF-PEDOT | S130–C127 | S130–C127–C137 | 1.75 | 122.400 | PCL-COF-PEDOT | 1.75 | 122.49 |
| O69–B75 | O69–B75–O71 | 1.38 | 118.91 | 1.38 | 118.86 | ||
| C116–C115 | C116–C115–S113 | 1.37 | 109.62 | 1.37 | 109.60 | ||
| P@COF-PEDOT | P159–O32 | O32–P159–C93 | 1.67 | 98.44 | PCL-P@COF-PEDOT | 1.68 | 99.32 |
| S129–C125 | S129–C125![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C127 C127 |
1.75 | 109.62 | 1.75 | 109.66 | ||
| O1–B9 | O1–B9–C7 | 1.38 | 118.72 | 1.38 | 118.83 | ||
| P159–O32 | P159–O32–B2 | 1.67 | 125.70 | 1.68 | 125.12 | ||
| S@COF-PEDOT | S112–C114 | S112–C114–C125 | 1.75 | 121.92 | PCL-S@COF-PEDOT | 1.70 | 121.79 |
| B43–O38 | O38–B43–C36 | 1.38 | 120.65 | 1.38 | 119.93 | ||
C130![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C131 C131 |
C130![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C131–O132 C131–O132 |
1.33 | 123.95 | 1.33 | 123.96 | ||
| O68–B74 | C72–B74–O68 | 1.38 | 121.56 | 1.38 | 122.63 | ||
| Si@COF-PEDOT | C93–Si159 | C93–Si159–O32 | 1.86 | 110.14 | PCL-Si@COF-PEDOT | 1.85 | 109.83 |
| O40–B43 | O40–B43–C36 | 1.37 | 120.68 | 1.37 | 119.71 | ||
| S142–C136 | S142–C136–C126 | 1.75 | 121.78 | 1.75 | 121.55 | ||
| B30–O67 | B30–O67–Si159 | 1.37 | 122.80 | 1.36 | 123.04 |
 | ||
| Fig. 1 (a) Structural representations of the studied compounds before adsorption of paracetamol. (b) Structural representations of the studied compounds after adsorption. | ||
3.2 Electronic properties
 | (1) |
| Compound | Donor (i) | Acceptor (j) | E(2) kcal mol−1 | E(j) − E(i) a. u. | F(i, j) a. u. | Transition |
|---|---|---|---|---|---|---|
| PCL-P@COF-PEDOT | LP-O67 | σ*B30–O67 | 277.56 | 0.780 | 0.418 | LP → σ* |
| LP-O66 | σ*LP-B37 | 263.59 | 0.590 | 0.357 | LP → σ* | |
| LP-O38 | σ*LP-B37 | 258.97 | 0.580 | 0.351 | LP → σ* | |
| PCL-S@COF-PEDOT | LP-O11 | σ*LP-B30 | 148.98 | 0.60 | 0.384 | LP → σ* |
| LP-O32 | σ*LP-B2 | 217.96 | 0.85 | 0.548 | LP → σ* | |
| LP-O66 | σ*LP-B37 | 132.05 | 0.59 | 0.357 | LP → σ* | |
| PCL-Si@COF-PEDOT | LP-O66 | σ*LP-B37 | 132.47 | 0.59 | 0.358 | LP → σ* |
| LP-O33 | σ*LP-B5 | 131.22 | 0.58 | 0.355 | LP → σ* | |
| LP-O6 | σ*LP-B43 | 121.14 | 0.59 | 0.343 | LP → σ* |
3.2.3.1 Frontier molecular orbital (FMO) and global reactivity descriptor analysis. To obtain a comprehensive understanding of the electronic reactivity and adsorption behavior of COF-PEDOT and its heteroatom-doped derivatives toward paracetamol, frontier molecular orbital (FMO) analysis was combined with global quantum reactivity descriptors derived from HOMO and LUMO energies. These parameters collectively provide insight into stability, charge-transfer propensity, and adsorption-induced electronic modifications.22 The HOMO energy represents the electron-donating ability of the adsorbent. As shown in Table 3, HOMO values for all systems fall within the range of −5.29 to −5.44 eV, indicating good intrinsic electronic stability. Upon PCL adsorption, a general downward shift in HOMO energies is observed, exemplified by pristine COF-PEDOT, which shifts from −5.29 eV to −5.44 eV after adsorption. This decrease signifies enhanced electronic stabilization and partial electron donation from the framework to the adsorbate, a behavior commonly associated with physisorption accompanied by mild charge redistribution. The LUMO energies, which describe the electron-accepting capacity of the systems, remain relatively invariant (−2.06 to −2.23 eV) before and after adsorption. This limited variation suggests that the ability of the frameworks to accept electrons is largely preserved upon PCL binding. Such stability in LUMO levels indicates that adsorption does not significantly perturb the conduction characteristics of the COF-PEDOT backbone, which is desirable for reusable sensing and adsorption materials. The HOMO–LUMO energy gap (ΔE) is a critical indicator of chemical reactivity and charge-transfer efficiency. Smaller ΔE values correspond to higher polarizability and enhanced electronic communication between the adsorbent and adsorbate. Prior to adsorption, ΔE values range from 3.11 to 3.32 eV. Following PCL adsorption, most systems exhibit a reduction in ΔE, particularly in doped frameworks. For instance, P@COF-PEDOT shows a decrease from 3.32 eV to 3.10 eV after adsorption, reflecting increased reactivity and improved charge-transfer interactions induced by PCL binding. The global softness (Σ) and chemical hardness (η) further support this trend. Chemical hardness, defined as half of the energy gap, reflects resistance to charge transfer, whereas softness is its inverse measure and indicates the ease of electron cloud deformation. As presented in Table 3, η values decrease slightly upon PCL adsorption (e.g., from 1.66 to 1.55 eV for P-doped systems), while Σ shows a corresponding marginal increase. This complementary behavior confirms that adsorption enhances the electronic responsiveness of the systems, facilitating charge exchange at the interface. The negative chemical potential (−µ), which approximates the electronegativity of the system, provides insight into the tendency of the framework to attract electrons. The relatively consistent −µ values (−3.68 to −3.81 eV) indicate that both pristine and doped COF-PEDOT systems maintain a strong driving force for electron exchange. Slightly more negative values observed after PCL adsorption suggest stabilization of the electronic system and increased affinity for charge redistribution during adsorption. The electrophilicity index (ω) quantifies the ability of a system to accept electrons. Higher ω values are associated with stronger electrophilic character and improved interaction with nucleophilic species. In Table 3, ω values show a modest increase after PCL adsorption, particularly for doped systems such as PCL-P@COF-PEDOT (ω = 4.61 eV). This enhancement implies that adsorption strengthens the electrophilic nature of the framework, thereby favoring stronger adsorbate–adsorbent interactions.23 Finally, the Fermi level (EFL) reflects the position of the electronic equilibrium level and plays a crucial role in charge-transfer processes. The observed shift in EFL toward more negative values after adsorption (e.g., from 3.73 to 3.81 eV for pristine COF-PEDOT) suggests electron transfer from the adsorbent to the adsorbate. Such shifts are indicative of adsorption-induced electronic perturbations and further confirm the occurrence of interfacial charge redistribution. Since the work function is defined as the energy difference between the vacuum level and the Fermi level, this shift indicates a corresponding change in the work function upon paracetamol adsorption, which directly influences surface reactivity and adsorption strength.
| Compound | HOMO (eV) | LUMO (eV) | Energy gap (eV) | Σ | H | (−µ) | ω | EFL |
|---|---|---|---|---|---|---|---|---|
| COF-PEDOT | −5.29 | −2.17 | 3.12 | 0.32 | 1.56 | −3.73 | 4.46 | 3.73 |
| P@COF-PEDOT | −5.40 | −2.08 | 3.32 | 0.30 | 1.66 | −3.74 | 4.21 | 3.74 |
| S@COF-PEDOT | −5.33 | −2.22 | 3.11 | 0.32 | 1.56 | −3.78 | 4.59 | 3.78 |
| Si@COF-PEDOT | −5.30 | −2.06 | 3.24 | 0.31 | 1.62 | −3.68 | 4.18 | 3.68 |
| PCL-COF-PEDOT | −5.44 | −2.18 | 3.26 | 0.31 | 1.63 | −3.81 | 4.46 | 3.81 |
| PCL-P@COF-PEDOT | −5.33 | −2.23 | 3.10 | 0.32 | 1.55 | −3.78 | 4.61 | 3.78 |
| PCL-S@COF-PEDOT | −5.37 | −2.21 | 3.17 | 0.32 | 1.58 | −3.79 | 4.54 | 3.79 |
| PCL-Si@COF-PEDOT | −5.44 | −2.18 | 3.26 | 0.31 | 1.63 | −3.81 | 4.46 | 3.81 |
3.3 Adsorption studies
Previous studies have emphasized that adsorption energy is a key parameter in evaluating the interaction strength between adsorbent materials and pollutant molecules such as pharmaceuticals.24 In environmental remediation, particularly for paracetamol removal, adsorption energy provides critical insight into how effectively an adsorbent surface can capture and retain target molecules while maintaining structural stability and potential reusability.25 Materials exhibiting moderately strong adsorption energies are often preferred, as they ensure a balance between high adsorption capacity and facile desorption or regeneration, which are essential for sustainable wastewater treatment operations.26In this work, adsorption energies were considered for paracetamol interacting with the various heteroatom-doped PCL@COF-PEDOT systems, with dopants including phosphorus, sulfur, and silicon. From the adsorption studies (see Table 4), it was observed that all the systems yielded negative adsorption energies, this confirms that the interaction was exothermic, hence it is thermodynamically favorable. Among the three complexes, PCL-P@COF-PEDOT and PCL-S@COF-PEDOT exhibited the same adsorption energies of −1.170 eV. This similar negative adsorption energies suggest that both dopants form moderately strong non-covalent interactions with paracetamol, making them capable of sensing paracetamol molecule effectively, while allowing for reversibility. On the other hand, PCL-Si@COF-PEDOT had a lower adsorption energy of −0.980 eV, this lower adsorption energy can be attributed to silicon atomic radius, which limit the orbital overlap and ultimately leads to a reduced charge transfer with paracetamol. The finding from the adsorption studies showed that the introduction of heteroatoms enhanced the sensitivity of COF-PEDOT for paracetamol. Although this study is purely theoretical, the DFT predictions in this work are consistent with experimental trends reported for COF- and PEDOT-based adsorbents. Tunable COFs have been shown experimentally to control adsorption and degradation of paracetamol through tailored framework chemistry.35 PEDOT-containing composites have also demonstrated strong polymer–drug interactions and favorable electrochemical adsorption behavior in practical films, supporting the concept that conductive polymer integration improves adsorbate affinity.36 Furthermore, COF adsorbents have been used successfully to capture pharmaceutical pollutants in aqueous media, providing real-world context for the adsorption energies we report.34 These studies suggest that our predicted trend (P ≈ S > Si in adsorption strength) can guide targeted synthesis of doped COF-PEDOT composites for paracetamol removal and sensing in wastewater treatment.
| System | E (complex) | E (adsorbent) | E (adsorbate) | E (a.u) | E (eV) |
|---|---|---|---|---|---|
| PCL-P@COF-PEDOT | −7873.527 | −7397.752 | −475.732 | −0.043 | −1.170 |
| PCL-S@COF-PEDOT | −7930.232 | −7454.457 | −475.732 | −0.043 | −1.170 |
| PCL-Si@COF-PEDOT | −7821.685 | −7345.917 | −475.732 | −0.036 | −0.980 |
3.4 Visual studies
| Systems | Bond | CP | ρ(r) | ∇2ρ(r) | G(r) | V(r) | H(r) | EFL | λ1 | λ2 | λ3 | λ1/λ3 | ε | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PCl-COF-PEDOT | H60–H166 | 235 | 0.0034 | 0.0157 | 0.0025 | −0.0012 | 0.0013 | 2.083 | 0.0074 | −0.0031 | 0.0221 | −0.0023 | 1.3478 | 0.0508 |
| C170–H49 | 241 | 0.0061 | 0.0341 | 0.0062 | −0.0038 | 0.0023 | 1.631 | 0.0090 | −0.0072 | 0.0471 | −0.0058 | 1.2414 | 0.2354 | |
| PCl-P@COF-PEDOT | H52–C174 | 324 | 0.0014 | 0.0052 | 0.0009 | −0.0006 | 0.0004 | 1.500 | 0.0029 | −0.0007 | 0.0015 | 0.0044 | −1.636 | −1.496 |
| C105–C181 | 369 | 0.0071 | 0.0198 | 0.0047 | −0.0044 | 0.0025 | 1.760 | 0.0252 | −0.0042 | −0.0010 | 0.0251 | −0.167 | 3.0615 | |
| PCl-S@COF-PEDOT | S129–H52 | 78 | 0.0003 | 0.0018 | 0.0003 | −0.0001 | 0.0001 | 3.000 | 0.0002 | 0.0011 | −0.0001 | 0.0008 | 1.375 | −1.181 |
| O175–H78 | 248 | 0.0151 | 0.0690 | 0.0153 | −0.0134 | 0.0019 | 1.141 | 0.0291 | −0.0207 | 0.1107 | −0.021 | 0.985 | 0.0124 | |
| PCl-Si@COF-PEDOT | O45–O78 | 261 | 0.0112 | 0.0529 | 0.0110 | −0.0088 | 0.0022 | 1.250 | 0.0212 | 0.0695 | −0.0077 | −0.0087 | −7.988 | 0.1239 |
| O143–S122 | 244 | 0.0101 | 0.0484 | 0.0099 | −0.0077 | 0.0021 | 1.285 | 0.1882 | 0.0622 | −0.0061 | −0.0077 | −8.077 | 0.2498 |
4 Future perspectives and challenges
The theoretical insights obtained in this study provided a clear guidance for the experimental design of heteroatom-doped COF-PEDOT materials for paracetamol adsorption. The predicted influence of P, S, and Si dopants on electronic structure and adsorption strength suggests that controlled heteroatom incorporation can be used to optimize adsorption performance. These materials show potential for practical applications in wastewater treatment filters, membranes, and sensing platforms. However, challenges remain in achieving precise dopant control, ensuring long-term stability in aqueous environments, and validating adsorption capacity and regeneration through experiments. Addressing these aspects will be essential for translating the present DFT predictions into real-world applications.5 Conclusions
This study investigated the chemical modification of covalent organic framework poly(3,4-ethylenedioxythiophene) (COF-PEDOT) composites through heteroatom (P, S, and Si) doping to enhance their adsorption capability toward paracetamol in environmental systems. Using first-principles density functional theory (DFT) calculations, the structural, electronic, and adsorption properties of the doped systems were thoroughly analyzed. Geometry optimization revealed only minor angular distortions following paracetamol adsorption, indicating that the complexes remained structurally stable throughout the interaction process.Electronic structure analyses, including density of states (DOS) and frontier molecular orbital (FMO) evaluations, provided further insights into the reactivity trends of the doped frameworks. The DOS profiles showed that heteroatom incorporation enhanced the electronic activity of COF-PEDOT, with the Si-doped system exhibiting the most pronounced Fermi-level shift (−5.9 eV), suggesting stronger orbital delocalization and improved adsorption affinity. Similarly, FMO analysis indicated that heteroatom doping effectively narrowed the HOMO–LUMO energy gaps, signifying increased chemical reactivity and stability. The ionization potentials of all materials ranged between 5.29 eV and 5.44 eV, consistent with literature-reported values (4.5–6.0 eV) for moderately stable adsorbents. Natural bond orbital (NBO) results further demonstrated that phosphorus doping produced the highest orbital stabilization energies, reflecting stronger donor–acceptor interactions. Adsorption energy calculations revealed negative values for all systems, confirming that the adsorption of paracetamol was exothermic and thermodynamically favorable. Among the three doped structures, PCL-P@COF-PEDOT and PCL-S@COF-PEDOT exhibited comparable adsorption energies (−1.170 eV), suggesting moderately strong and reversible noncovalent interactions suitable for reusable adsorbent applications. Conversely, the Si-doped complex (PCL-Si@COF-PEDOT) showed a relatively lower adsorption energy (−0.980 eV), likely due to silicon's larger atomic radius, which limits orbital overlap and charge transfer efficiency. Topological (QTAIM) and non-covalent interaction (NCI) analyses confirmed the presence of weak but stable van der Waals interactions across all systems, indicating that adsorption primarily occurs through physisorption rather than chemisorption.
Although the present study focuses on the adsorption mechanisms of paracetamol on P, S, and Si-doped COF-PEDOT via DFT calculations, repeatability and cycling studies are important to assess the practical stability and reusability of these materials. Future experimental investigations are encouraged to perform adsorption–desorption cycling tests and replicate adsorption measurements to validate the computational predictions and ensure consistent performance. Collectively, these findings establish that heteroatom doping effectively tunes the electronic configuration and adsorption characteristics of COF-PEDOT frameworks. The incorporation of phosphorus, sulfur, and silicon atoms enhances both sensitivity and reversibility, making the doped COF-PEDOT materials promising candidates for the selective adsorption of paracetamol from pharmaceutical contaminants in aquatic environments.
Author contributions
Innocent E. Emeng conceptualized the research, drafted the initial manuscript, contributed in the adsorption studies and approved the final version of the manuscript. Uduak Luke and Nguuma I. Gber carried out the computational modeling, data validation and preparation of figures and tables. Onyinyechi V. Ugochukwu contributed to the interpretation of the electronic structure. Ambika Sundaravadivelu assisted in literature review. Gopinath Sampathkumar contributed in editing for intellectual content. Musa Runde supervised the entire project and provided critical revisions.Conflicts of interest
There are no conflicts of interest to declare.Data availability
Additional computational data are available upon request from the corresponding author.All data supporting the findings of this study are included within the manuscript and its supplementary information (SI). Supplementary information: optimized Cartesian coordinates of all structures obtained at the DFT level used in this work. See DOI: https://doi.org/10.1039/d5ra08291a.
References
- N. Zaidi, M. A. Mir, S. K. Chang, N. Abdelli, S. M. Hasnain, M. A. Ali Khan and K. Andrews, Pharmaceuticals and personal care products as emerging contaminants: environmental fate, detection, and mitigation strategies, Int. J. Environ. Anal. Chem., 2025, 1–29 Search PubMed.
- I. U. Islam, A. N. Qurashi, A. Adnan, A. Ali, S. Malik, F. Younas and E. Yabalak, Bioremediation and adsorption: strategies for managing pharmaceutical pollution in aquatic environment, Water, Air, Soil Pollut., 2025, 236(9), 579 CrossRef CAS.
- W. K. Wardhani, E. S. Soedjono, H. S. Titah and M. A. Mardyanto, Pharmaceutical emerging micropollutants potential in septic tanks: its fate and transport study in Indonesia – a literature review, Environ. Qual. Manage., 2024, 34(1), e22176 CrossRef.
- P. Loffler, B. I. Escher, C. Baduel, M. P. Virta and F. Y. Lai, Antimicrobial transformation products in the aquatic environment: global occurrence, ecotoxicological risks, and potential of antibiotic resistance, Environ. Sci. Technol., 2023, 57(26), 9474–9494 CrossRef PubMed.
- H. P. H. Arp, A. Gredelj, J. Gluge, M. Scheringer and I. T. Cousins, The global threat from the irreversible accumulation of trifluoroacetic acid (TFA), Environ. Sci. Technol., 2024, 58(45), 19925–19935 Search PubMed.
- K. Patra, S. Dey, C. Solanki, A. Sengupta and V. K. Mittal, Harnessing advanced porous materials, covalent organic frameworks, and porous organic polymers as next-generation porous frameworks for targeted removal of emerging water contaminants, ACS Appl. Eng. Mater., 2025, 3(5), 1130–1165 CrossRef CAS.
- A. G. Niculescu, B. Mihaiescu, D. E. Mihaiescu, T. Hadibarata and A. M. Grumezescu, An updated overview of magnetic composites for water decontamination, Polymers, 2024, 16(5), 709 Search PubMed.
- Z. Geng, A highly sensitive fluorescent detection method utilizing B and S co-doped graphene quantum dots for ibuprofen analysis, Alexandria Eng. J., 2025, 112, 17–25 CrossRef.
- C. Cao, Q. Chen, R. Suizu and K. Awaga, Critical transition to a highly conductive state through PEDOT oligomer percolation in redox-active COFs, J. Mater. Chem. C, 2024, 12(9), 3072–3076 RSC.
- D. Bakowies and O. A. von Lilienfeld, Density functional geometries and zero-point energies in ab initio thermochemical treatments of compounds with first-row atoms (H, C, N, O, F), J. Chem. Theory Comput., 2021, 17(8), 4872–4890 Search PubMed.
- Z. Ullah, H. J. Kim, Y. S. Mary, N. Belboukhari, K. Sekkoum, A. Kraimi and H. W. Kwon, Unlocking the potential of ovalene: a dual-purpose sensor and drug enhancer, J. Mol. Liq., 2023, 377, 121540 CrossRef CAS.
- G. Sanyal, M. Kandasamy, B. Mondal, A. Seetharaman and B. Chakraborty, Pd-anchored VSe2 for glucose sensing: prediction from first principle simulations, Surf. Interfaces, 2024, 54, 105235 CrossRef CAS.
- K. W. Qadir, M. D. Mohammadi and H. Y. Abdullah, Modelling gas adsorption onto Al12(Zn)N12 surfaces: a theoretical study of CH4, CO, CO2, H2O, N2, NH3, NO, NO2, O2, and SO2 interactions, Comput. Theor. Chem., 2025, 1244, 115063 CrossRef CAS.
- R. Zhao, B. Li, S. Chen, B. Zhang, J. Chen, J. Sun and X. Ma, Intertwined role of mechanism identification by DFT-XAFS and engineering considerations in the evolution of P adsorbents, Sci. Total Environ., 2024, 946, 174159 CrossRef CAS PubMed.
- R. Rahimi and M. Solimannejad, A novel pentagonal BCN monolayer for sensing and drug delivery of nitrosourea and hydroxyurea anticancer drugs: a DFT outlook, Mater. Sci. Semicond. Process., 2024, 173, 108109 CrossRef CAS.
- I. S. Oliveira, M. S. Garcia, N. M. Cassani, A. L. Oliveira, L. C. Freitas, V. K. Bertolini and C. Abbehausen, Exploring antiviral and antiparasitic activity of gold N-heterocyclic carbenes with thiolate ligands, Dalton Trans., 2024, 53(47), 18963–18973 RSC.
- T. Takeshita and D. Kinoshita, Evaluation of darrow red–organosilane composite as a photosensitizer for application in dye-sensitized zinc oxide photocatalysts: DFT and TD-DFT studies, J. Mol. Model., 2022, 28(12), 407 Search PubMed.
- H. Gu, F. Wang, S. Chen, J. Lan, J. Wang, C. Pei and J. Gong, Suppressing Jahn-Teller distortion of MnO2 via B-Ni dual single-atoms integration for methane catalytic combustion, Nat. Commun., 2025, 16(1), 1008 CrossRef CAS PubMed.
- S. Suthar and K. C. Mondal, Unveiling the anomaly of reduction of carborane-bis-silylene-stabilised silylone/germylone leading to unusual oxidation of Si0/Ge0 to SiI/GeI with EDA-NOCV analyses, Chem.–Eur. J., 2024, 30(10), e202303355 CrossRef CAS PubMed.
- W. A. Mahdi, A. Alhowyan and A. J. Obaidullah, A DFT insight into the potential of cycloparaphenylenes as efficient sensors for detecting paracetamol, Sci. Rep., 2025, 15(1), 8150 Search PubMed.
- W. Dong, J. Xing, Q. Chen, Y. Huang, M. Wu, P. Yi and B. Xing, Hydrogen bonds between the oxygen-containing functional groups of biochar and organic contaminants significantly enhance sorption affinity, Chem. Eng. J., 2024, 499, 156654 CrossRef CAS.
- A. U. Rahman, M. K. Rokunuzzaman, D. M. Saaduzzaman, M. S. Rahman, M. Amin, S. M. Hasan and M. K. U. Sikder, Designing nitride-derived fullerenes X20N20 (X = B, Al, and Ga) for the effective delivery of the cardiovascular drug felodipine: a DFT study, Nanoscale, 2025, 17(42), 24755–24772 RSC.
- Y. Liang, L. Feng, X. Liu, Y. Zhao, Q. Chen, Z. Sui and N. Wang, Enhanced selective adsorption of NSAIDs by covalent organic frameworks via functional group tuning, Chem. Eng. J., 2021, 404, 127095 CrossRef CAS.
- L. Wu, Z. Dai, H. Fu, M. Shen, L. Cha, Y. Lin and B. Lu, Multiple electron transfers enable high-capacity cathode through stable anionic redox, Adv. Mater., 2025, 37(9), 2416298 CrossRef CAS PubMed.
- A. Zamani, F. F. Sead, I. Kaur, A. Kubaev, S. T. Hamedani and H. Majedi, DFT analysis of dimethyl fumarate interactions with B12N12 and B24 nanoclusters for enhanced anticancer drug delivery, Comput. Theor. Chem., 2025, 1247, 115168 Search PubMed.
- R. K. Sharma, A. Srivastava, U. Punia, R. Bansal, P. Prajapat, G. Gupta and S. K. Srivastava, Multifunctioning graphene oxide capping layer for highly efficient and stable PEDOT:PSS–silicon hybrid solar cells, Sustainable Energy Fuels, 2024, 8(20), 4799–4812 RSC.
- S. A. Halim, A. B. El-Meligy, A. M. El-Nahas and S. H. El-Demerdash, DFT study and natural bond orbital (NBO) population analysis of 2-(2-hydroxyphenyl)-1-azaazulene tautomers and their mercapto analogues, Sci. Rep., 2024, 14(1), 219 CrossRef CAS PubMed.
- H. Louis, L. P. Ifediora, O. C. Enudi, T. O. Unimuke, F. C. Asogwa and Y. L. Moshood, Evaluation of the excited state dynamics, photophysical properties, and the influence of donor substitution in a donor-π-acceptor system, J. Mol. Model., 2021, 27(10), 284 CrossRef CAS PubMed.
- A. Mahmood, A. Irfan, F. Ahmad and M. R. S. A. Janjua, Quantum chemical analysis and molecular dynamics simulations to study the impact of electron-deficient substituents on electronic behavior of small molecule acceptors, Comput. Theor. Chem., 2021, 1204, 113387 CrossRef CAS.
- H. A. Rizwan, M. U. Khan, A. Anwar, S. Alharthi and M. A. Amin, First theoretical framework of Al9N9 and B9N9 nanorings for unveiling their unique detection and sensing potential for SF6 decomposition gases (H2S, SO2, SOF2, and SO2F2): toward real-time gas sensing in high-voltage power systems, RSC Adv., 2025, 15(25), 20020–20039 RSC.
- S. Noreen, S. H. Sumrra, A. U. Hassan, M. Afzaal, A. Y. E. Elnaggar, I. H. E. Azab and M. H. Mahmoud, A DFT and molecular correlational analysis on newly designed silicon-carbide quantum dots with extended acceptors for their photovoltaic performance, Silicon, 2025, 1–13 Search PubMed.
- Q. Wang, S. Lian, C. Guo, X. Gao, Y. Dou, C. Song and J. Lin, The chemical adsorption effect of surface-enhanced Raman spectroscopy of nitrobenzene and aniline using density functional theory, Spectrochim. Acta, Part A, 2022, 279, 121428 CrossRef CAS PubMed.
- X. Liu, D. Huang, C. Lai, G. Zeng, L. Qin, H. Wang and S. Chen, Recent advances in covalent organic frameworks (COFs) as a smart sensing material, Chem. Soc. Rev., 2019, 48(20), 5266–5302 RSC.
- A. Mellah, S. P. Fernandes, R. Rodríguez, J. Otero, J. Paz, J. Cruces and L. M. Salonen, Adsorption of pharmaceutical pollutants from water using covalent organic frameworks, Chem.–Eur. J., 2018, 24(42), 10601–10605 CrossRef CAS PubMed.
- F. Liu, Z. Ma, Y. Deng, M. Wang, P. Zhou, W. Liu and D. Ma, Tunable covalent organic frameworks with different heterocyclic nitrogen locations for efficient Cr(VI) reduction, Escherichia coli disinfection, and paracetamol degradation under visible-light irradiation, Environ. Sci. Technol., 2021, 55(8), 5371–5381 CrossRef CAS PubMed.
- J. H. Viteri, N. Cotolan, L. Barbu-Tudoran and G. L. Turdean, A paracetamol-poly(3,4-ethylenedioxythiophene) composite film for drug release studies, Mater. Today Commun., 2023, 34, 105084 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |