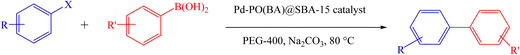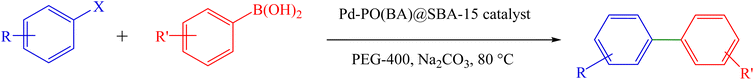Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
A palladium complex of 2,2′-(propane-1,3-diylbis(oxy))dibenzenaminium chloride on SBA-15 as a returnable, environmental and selective nanostructured-catalyst in the Suzuki C–C coupling reaction
Shirin Mohammadi,
Mohsen Nikoorazm *,
Bahman Tahmasbi* and
Yunes Abbasi Tyula
*,
Bahman Tahmasbi* and
Yunes Abbasi Tyula
Department of Chemistry, Faculty of Science, Ilam University, P. O. Box 69315516, Ilam, Iran. E-mail: m.nikorazm@ilam.ac.ir; b.tahmasbi@ilam.ac.ir
First published on 27th April 2026
Abstract
In this work, mesoporous SBA-15 was synthesized by a simple procedure using P123 and TEOS in HCl acidic conditions, followed by calcination at 550 °C. 2,2′-(Propane-1,3-diylbis(oxy))dibenzenaminium chloride ligand (PO(BA)) as ligand was synthesized from 2-nitrophenol and 1,3-dibromopropane, following reduction of nitro groups. The step-by-step synthesis of the PO(BA) ligand was confirmed by 1H NMR and 13C NMR. 3-Iodopropyltriethoxysilane (IPTES) was synthesized from 3-chloropropyltriethoxysilane (CPTES) in dry acetone. Then, the mesoporous SBA-15 surface was modified by IPTES and next it was functionalized by PO(BA). Finally, immobilized PO(BA) on SBA-15 became complex with palladium acetate (Pd-PO(BA)@SBA-15). The prepared Pd-PO(BA)@SBA-15 was characterized by SEM, ICP, TGA/DSC, EDS, BET/BJH, and WDX techniques. TGA and BET/BJH methods showed high thermal stability of this catalyst up to 230 °C and a high surface area for this catalyst. Then, the catalytic usage of Pd-PO(BA)@SBA-15 was investigated in the selective carbon–carbon bond formation. Various aryl halides (including aryl iodides (Ar–I) and aryl bromides (Ar–Br), having electron-donating or electron-withdrawing functional groups) and some derivatives of phenylboronic acid (such as phenylboronic acid, 4-methoxyphenylboronic acid and 4-formylphenylboronic acid) were investigated and all biphenyl products were obtained with high yields and TOF values. NMR spectroscopy was used to determine the synthesized biphenyl products. Pd-PO(BA)@SBA-15 catalyst was shown to be reusable without significant loss in its performance in the Suzuki–Miyaura cross-coupling reaction.
Introduction
The C–C bond formation, which plays a usual and great role in the synthesis of organic materials to forming preparation of hydrocarbons, UV screens, conductive polymers, herbicides, pharmaceuticals, natural products, polymerization processes, ligands, advanced materials, insecticides, agrochemicals, etc.1 Homologous of C–C coupling reactions (include coupling of two similar reactants to synthesis of a symmetric molecule) and cross-coupling of C–C reactions (include coupling of two quite different reactants to form a new molecule) are two general categories for C–C bond formation.2 The Suzuki reaction is an important catalytic C–C coupling reaction, including the coupling of an arylboronic acid with an aryl halide,2,3 which can produce various drugs, e.g., valsartan, lapatinib, losartan, atazanavir, diflunisal, and clonazepam.2 The Suzuki reaction is usually performed in the presence of homogeneous and heterogeneous Pd-catalyts.3b,4 Given the sensitive ligands and high price of Pd-catalysts, heterogeneous-based palladium catalysts are suitable over palladium-containing homogeneous catalysts due to the recoverability and reusability of heterogeneous catalysts.3b,5 Therefore, the heterogeneous Pd-catalysts, especially immobilized palladium on the surface of various supports, have attracted much attention in the field of catalysis.3b In this regard, biochar, CMK-3, graphene oxide(GO), polymers, mesoporous/amorphous silica materials, boehmite nanoparticles, MOFs (metal–organic frameworks), magnetic nanoparticles, zeolites, carbon nanomaterials, etc. Were used as support for immobilization of practical catalysts.6 Among mesoporous silica materials, SBA-15 is an ideal nanostructure for immobilization of catalyst active species, which has excellent stability, great specific surface area, including modifiable silanol groups, and customizable pore size.7 SBA-15 is classified as a mesoporous material in the IUPAC classification (type IV isotherm with H1 hysteresis).8 Therefore, supported catalysts on SBA-15 have high catalytic activity (like homogeneous catalysts), and also these catalysts are recyclable (like heterogeneous catalysts). Therefore, in this work, SBA-15 was synthesized and then modified by IPTES, and then functionalized with PO(BA) for immobilization of a palladium complex (Pd-PO(BA)@SBA-15) as an efficient, returnable and practical nanocatalyst in the Suzuki C–C cross-coupling reaction.Experimental
Synthesis of SBA-15
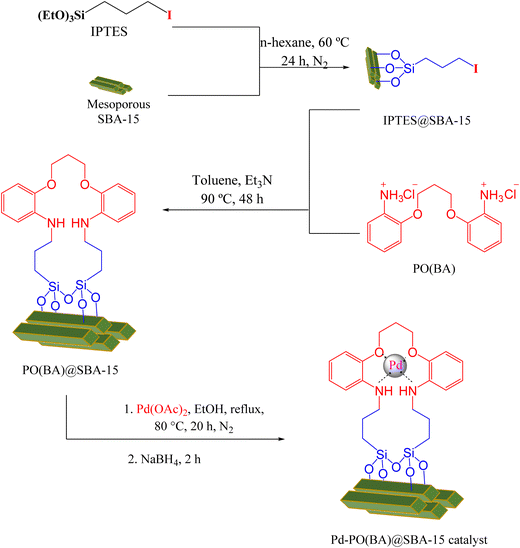
As the first step in the catalyst synthesis, mesoporous SBA-15 was prepared as a catalyst support. The synthesis process of SBA-15 was carried out according to the previous report. This process includes: pluronic 123 (4 g) was dissolved in 130 ml of deionized water and 20 ml HCl 37% and stirred at 40 °C for 90 min. Then, tetraethylorthosilicate (8.5 g) was added dropwise and stirred at room temperature until it became a clear solution. Next, the solution was stirred at 40 °C for 24 h. Then, it was kept in an autoclave at 100 °C for 48 h. The white precipitate obtained was recovered by filtration, washed with H2O and dried at room temperature. Finally, the template was dried and calcined at 550 °C with 2 °C min−1 for 5 h. The obtained white powder is SBA-15.Synthesis of 3-iodopropyltriethoxysilane (IPTES)
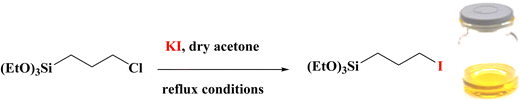
KI (5 mmol) was added in dry acetone (25 ml), and then 3-chloropropyltriethoxysilane (5 mmol) was injected dropwise under stirring. The mixture was mixed under N2 atmosphere at 50 °C for 24 h (Scheme 1). Finally, the mixture was filtered, and then, the acetone solvent was removed by evaporation. The remaining liquid is 3-iodopropyltriethoxysilane (IPTES).Synthesis of 2,2′-(propane-1,3-diylbis(oxy))dibenzenaminium chloride (PO(BA))
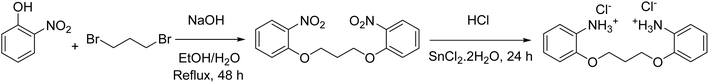
First, 5.65 g (40 mmol) of 2-nitrophenol was completely dissolved in a flask containing 35 ml of ethanol. Then, 1.6 g (40 mmol) of NaOH was dissolved in 10 ml of water and added to the above solution. In this case, the color of the resulting solution changed from light yellow to red. In the next step, 4.04 g (20 mmol) of 1,3-dibromopropane was dissolved in 20 ml of ethanol and added dropwise to the above solution. Then, the solution was refluxed for 48 h. After about 24 h from the start of reflux, a yellow precipitate began to form on the wall of the container. At the end of the reflux time, a light yellow precipitate had formed in the flask, which was filtered and washed with ethanol and recrystallized in ethanol solvent for further purification (Precipitate weight 5.33 g, yield 41.75% after recrystallization and melting point 110 °C). At this stage, 1,3-bis(2-nitrophenoxy)propane was synthesized, which was identified and confirmed by 1H NMR and 13C NMR (Scheme 2). 1H NMR (250 MHz, δ ppm DMSO-d6): 7.84 (d, J = 7.5 Hz, 2H), 7.62 (t, J = 7.5 Hz, 2H), 7.34 (d, J = 7.5 Hz, 2H), 7.08 (t, J = 7.5 Hz, 2H), 4.31 (t, J = 7.5 Hz, 4H), 2.20 (quin, J = 7.5 Hz, 2H) ppm. 13C NMR (100 MHz, DMSO-d6): 151.6, 139.9, 134.9, 125.4, 121.0, 115.4, 65.9, 28.5 ppm.To synthesize PO(BA), the method reported by Cannon and colleagues9 and optimization of their method for the reduction of the nitro group were used. In a 250 ml flask, 2.06 g of 1,3-bis(2-nitrophenoxy)propane was dissolved in 100 ml of hydrochloric acid (5 N) at a temperature of 40–50 °C using a magnetic stirrer. Then, 10 g of SnCl2·2H2O was added gradually to the above solution and the solution was refluxed. After 24 hours, the color of the solution changed from light yellow to white. The reaction was stopped at this stage and the reaction mixture was filtered, and the amine salt was obtained as a white precipitate (Scheme 2). The synthesized 2,2′-(propane-1,3-diylbis(oxy))dibenzenaminium chloride (PO(BA)) was identified and confirmed by 1H NMR and 13C NMR. 1H NMR (250 MHz, δ ppm DMSO-d6): 7.42–7.38 (m, 2H), 7.36–7.32 (m, 2H), 7.22 (d, J = 7.5 Hz, 2H), 7.00 (t, J = 7.5 Hz, 2H), 4.33 (t, J = 5 Hz, 4H), 3.80 (br, 6H), 2.23 (s, 2H) ppm. 13C NMR (100 MHz, DMSO-d6): 151.8, 129.7, 124.2, 123.4, 121.0, 113.6, 65.5, 28.7 ppm.
Synthesis of Pd-PO(BA)@SBA-15 catalyst
Mesoporous SBA-15 (1 g) was added to n-hexane (25 ml), and 1.5 ml of IPTES was injected, and further it was stirred at 60 °C for 24 h under N2 atmosphere. The modified SBA-15 (IPTES@SBA-15) was isolated with simple filtration. Then, IPTES@SBA-15 (1.2 g) and 1.5 mmol OF PO(BA) ligand were dispersed in toluene (30 ml). Then, 9 mmol of triethylamine (Et3N) was injected dropwise under stirring. The mixture was stirred for 48 h at 90 °C under reflux conditions. The PO(BA)@SBA-15 (above functionalized SBA-15) was isolated with simple filtration, washed with DMSO and further with EtOH. At the end step, PO(BA)@SBA-15 (0.5 g) was mixed with palladium acetate (0.25 mmol) in ethanol for 20 h under reflux conditions and N2 atmosphere. After that, 0.3 mmol (0.018 g) of NaBH4 was added, and stirring was continued for 2 h. The immobilized Pd-PO(BA) on the surface of SBA-15 (Pd-PO(BA)@SBA-15 catalyst) was isolated by filtration and washing with H2O and further ethanol (Scheme 3).Procedure for the synthesizing of biphenyl catalyzed by Pd-PO(BA)@SBA-15
A mixture of arylboronic acid (1 mmol), aryl halide (1 mmol), Na2CO3 (0.31 g, 3 mmol), and Pd-PO(BA)@SBA-15 catalyst (12 mg) was mixed by a magnetic stirrer at 80 °C in PEG-400 (Scheme 4). The reaction progress was monitored by TLC analysis. After completion, the Pd-PO(BA)@SBA-15 catalyst was removed by filtration after the addition of water and ethyl acetate (EtOAc), and the remaining mixture after filtration was moved to a separator funnel containing water. Then the biphenyls were extracted in EtOAc.NMR spectral data
13C NMR (100 MHz, CDCl3): 147.6, 147.1, 138.8, 129.2, 128.9, 127.8, 127.4, 124.1 ppm.
13C NMR (100 MHz, CDCl3): 159.1, 140.8, 133.8, 128.7, 128.2, 126.8, 126.7, 114.2, 55.4 ppm.
13C NMR (100 MHz, CDCl3): 160.4, 147.2, 146.5, 131.0, 128.6, 127.1, 124.1, 114.6, 55.4 ppm.
13C NMR (100 MHz, CDCl3): 141.3, 128.8, 127.3, 127.2 ppm.
13C NMR (100 MHz, CDCl3): 191.7, 147.7, 146.0, 144.5, 136.2, 130.5, 128.2, 128.1, 124.3 ppm.
13C NMR (100 MHz, CDCl3): 140.0, 139.7, 133.4, 128.9, 128.9, 128.4, 127.6, 127.0 ppm.
13C NMR (100 MHz, CDCl3): 141.2, 138.4, 137.0, 129.5, 128.7, 127.0, 127.0, 21.1 ppm.
13C NMR (100 MHz, CDCl3): 145.7, 139.1, 132.6, 129.1, 128.7, 127.7, 127.2, 119.0, 110.9 ppm.
Results and discussion
Characterization of Pd-PO(BA)@SBA-15
The distribution of all elements was studied by WDX analysis (Fig. 3). WDX analysis of Pd-PO(BA)@SBA-15 indicates a homogeneous distribution of Si, O, Pd, C, N, and Cl species.
Considering that palladium specie has the main active catalytic species in Pd-PO(BA)@SBA-15, the exact loaded palladium on the surface of modified SBA-15 was calculated by the ICP analysis, which was found to be 0.358 mmol g−1.
The synthesizing conditions of biphenyls through Suzuki C–C coupling reaction in the presence of Pd-PO(BA)@SBA-15 were optimized in the coupling of phenylboronic acid and iodobenzene (C6H5–I) under different conditions, including the amount of Pd-PO(BA)@SBA-15 catalyst (Table 2, entries 1–3), solvent type (Table 2, entries 3–7), reaction temperature (Table 2, entries 10 and 11), and base type (Table 2, entries 7–10). The obtained experimental data are listed in Table 2, indicating 12 mg of Pd-PO(BA)@SBA-15 catalyst using Na2CO3 (sodium carbonate) as a base in PEG-400 as a solvent at 80 °C for optimal reaction conditions (Table 2, entry 3). As outlined in Table 2, 12 mg of Pd-PO(BA)@SBA-15 catalyst was selected for the coupling of phenylboronic acid with aryl halides, because lower amounts of Pd-PO(BA)@SBA-15 catalyst is not enough for completion of C–C bond formation (Table 2, entries 1 and 2) and the amount catalyst increasing of Pd-PO(BA)@SBA-15 not significant affect in results of the C–C coupling reaction in the presence of this catalyst. Among various inorganic and organic solvents such as PEG-400 (polyethylene glycol 400), EtOH, DMF (dimethylformamide), DMSO (dimethyl sulfoxide), and H2O, PEG-400 as a suitable solvent provided the best environmental conditions for the coupling of phenylboronic acid with C6H5–I in the presence of Pd-PO(BA)@SBA-15 catalyst (Table 2, entries 3–7). So, PEG-400 was selected as a green solvent over many traditional organic solvents due to its interesting advantages, e.g. non-toxicity, bio-degradability, non-flammability, readily recyclability, thermal stability, abundant availability, and cost-effectiveness.11 Among various solid and liquid bases e.g. Et3N (triethylamine), Na2CO3 (sodium carbonate), NaOEt (sodium ethoxide), and NaOH (sodium hydroxide), Na2CO3 as a suitable base provided the best conditions for the coupling of phenylboronic acid with C6H5–I in the presence of Pd-PO(BA)@SBA-15 catalyst at 80 °C.
| Entry | Catalyst (mg) | Solvent | Base | Temperature (°C) | Time (min) | Yield (%) |
|---|---|---|---|---|---|---|
| 1 | 10 | PEG-400 | Na2CO3 | 80 | 15 | 94 |
| 2 | 8 | PEG-400 | Na2CO3 | 80 | 30 | 89 |
| 3 | 12 | PEG-400 | Na2CO3 | 80 | 10 | 97 |
| 4 | 12 | EtOH | Na2CO3 | 80 | 40 | 85 |
| 5 | 12 | DMSO | Na2CO3 | 80 | 25 | 85 |
| 6 | 12 | DMF | Na2CO3 | 80 | 70 | 80 |
| 7 | 12 | H2O | Na2CO3 | 80 | 60 | 79 |
| 8 | 12 | PEG-400 | NaOH | 80 | 40 | 84 |
| 9 | 12 | PEG-400 | NaOEt | 80 | 55 | 80 |
| 10 | 12 | PEG-400 | Et3N | 80 | 160 | 77 |
| 11 | 12 | PEG-400 | Na2CO3 | 60 | 45 | 80 |
To extend the scope of Pd-PO(BA)@SBA-15 performance, we studied a large number of aryl halides in the coupling reaction with phenylboronic acid (Table 3, entries 1–5). The obtained experimental results are outlined in Table 3, showing the high yield of the isolated biphenyl products and low reaction times in the coupling reaction of phenylboronic acid with aryl halides having electron-donating functional groups or electron-withdrawing functional groups. For example, Cl, OH, NO2, and Me functional groups on the aromatic ring of aryl halides were investigated.
| Entry | R | R′ | X | Product | Time (min) | Yield (%) | TOF (h−1) | Melting point (°C) |
|---|---|---|---|---|---|---|---|---|
| 1 | H | H | I |  |
10 | 97 | 1353.5 | 64–67 (ref. 14) |
| 2 | 4-NO2 | H | Br | 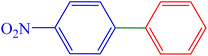 |
40 | 88 | 306.9 | 112–116 (ref. 14) |
| 3 | 4-Cl | H | Br | 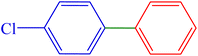 |
65 | 86 | 184.6 | 72–74 (ref. 14) |
| 4 | 4-CN | H | Br | 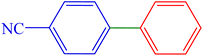 |
160 | 84 | 73.2 | 83–85 (ref. 14) |
| 5 | 4-Me | H | I | 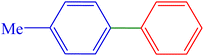 |
210 | 89 | 59.1 | 46–48 (ref. 14) |
| 6 | H | 4-OMe | I | 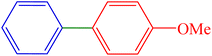 |
80 | 84 | 146.5 | 92–95 (ref. 15) |
| 7 | H | 4-CHO | I | 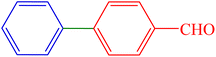 |
60 | 87 | 202.3 | 55–58 (ref. 14) |
| 8 | 4-NO2 | 4-CHO | Br | 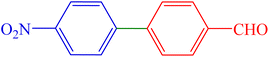 |
135 | 89 | 91.9 | 127–129 (ref. 10) |
| 9 | 4-NO2 | 4-OMe | Br | 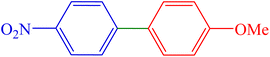 |
50 | 90 | 251.2 | 103–106 (ref. 10) |
To extend the scope of Pd-PO(BA)@SBA-15 performance, except aryl halide derivatives, some derivatives of phenylboronic acid -having electron-donating functional groups or electron-withdrawing functional groups-were also investigated in the coupling reaction with aryl halides in the presence of this catalyst. For example, 4-methoxyphenylboronic acid was investigated in the Suzuki C–C coupling reaction with aryl iodides (Ar–I) and aryl bromides (Ar–Br) derivatives in the presence of Pd-PO(BA)@SBA-15 catalyst (Table 3, entries 6 and 9). Also, 4-formylphenylboronic acid was investigated in the Suzuki C–C coupling reaction with aryl iodides (Ar–I) and aryl bromides (Ar–Br) derivatives in the presence of Pd-PO(BA)@SBA-15 catalyst (Table 3, entries 7 and 8). All products were formed with high yield in the presence of Pd-PO(BA)@SBA-15 catalyst.
TOF (Turnover Frequency) and TON (Turnover Number) are important factors in determining the performance of catalysts.12 Therefore, TON is defined as moles of product/moles of catalyst, and TOF is defined as TON/reaction time (h).13 All biphenyl products were formed with high TOF and TON values in the presence of Pd-PO(BA)@SBA-15 catalyst, indicating the practicality of this catalyst.
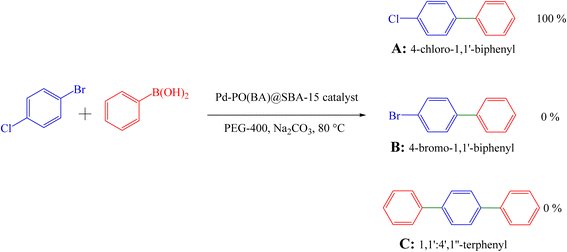
Also, 1-chloro-4-bromobenzene was investigated for the formation of the biphenyl (Table 3, entry 3), indicating good selectivity of Pd-PO(BA)@SBA-15 catalyst in the Suzuki C–C coupling reaction (Scheme 5). The selectivity of Pd-PO(BA)@SBA-15 catalyst in the coupling reaction of phenylboronic acid with iodobenzene has been confirmed using the melting point. Melting points for possible products A (4-chloro-1,1′-biphenyl), B (4-bromo-1,1′-biphenyl) and C (1,1′![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4′,1″-terphenyl) are reported as 71–73 °C, 89–92 °C and 212–213 °C, respectively. As shown in entry 3 (Table 3), the obtained melting point for the synthesized product in the presence of Pd-PO(BA)@SBA-15 catalyst is 72–74 °C, corresponding with product A.
4′,1″-terphenyl) are reported as 71–73 °C, 89–92 °C and 212–213 °C, respectively. As shown in entry 3 (Table 3), the obtained melting point for the synthesized product in the presence of Pd-PO(BA)@SBA-15 catalyst is 72–74 °C, corresponding with product A.
The homoselectivity of Pd-PO(BA)@SBA-15 in the coupling reaction of phenylboronic acid with iodobenzene toward the synthesis of A, B, or C has been investigated using 13C NMR spectroscopy. In the 13C NMR spectrum of C, six peaks for carbons should be observed. But in the 13C NMR spectra of A and B, eight peaks for carbons should be observed. As can be seen in the 13C NMR spectrum of the final synthesized product, eight peaks have been observed, as follows: 13C NMR (100 MHz, CDCl3): 140.0, 139.7, 133.4, 128.9, 128.9, 128.4, 127.6, 127.0 ppm. So product C is not formed.
Therefore, based on the melting point and NMR data, only 4-chloro-1,1′-biphenyl is selectively formed in the presence of Pd-PO(BA)@SBA-15 catalyst.
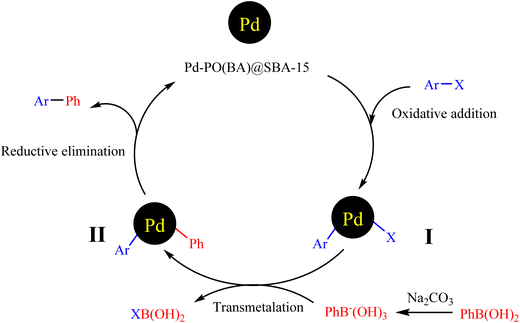
Scheme 6 investigates a mechanism for the C–C bond formation through the Suzuki coupling reaction in the presence of Pd-PO(BA)@SBA-15 catalyst.1b First, the oxidative addition of aryl halide with Pd-complex on the surface of SBA-15 forms intermediate I. Then, an acid/base reaction occurs between aryl boronic acid and sodium carbonate. Then, intermediate II was formed by a transmetalation step. Finally, intermediate II with a reductive elimination is converted to the biphenyl product and regeneration of the catalyst, which returns in the catalytic cycle of the reaction.
| Entry | Catalyst | Reaction conditions | Time (min) | Yield (%) | Ref. |
|---|---|---|---|---|---|
| 1 | Ru-dithizone@biochar-Ni MNPs | H2O, Na2CO3, 80 °C | 90 | 96 | 16 |
| 2 | Polymer anchored Pd(II) | DMF/H2O, K2CO3, 80 °C | 300 | 96 | 17 |
| 3 | PANI-Pd | 1,4-Dioxane![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (1 H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), K2CO3, 95 °C 1), K2CO3, 95 °C |
240 | 91 | 18 |
| 4 | ZnFe2O4@SiO2@CPTMS@PYA-Pd | DMSO, K2CO3, 95 °C | 100 | 96 | 4 |
| 5 | NHC–Pd(II) complex | THF, CsCO3, 80 °C | 12 h | 88 | 19 |
| 6 | Pd(II)–NHC complex | DMF, CsCO3, 100 °C | 24 h | 99 | 20 |
| 7 | Cu–ninhydrin@GO–Ni MNPs | H2O, Na2CO3, 80 °C | 50 | 97 | 21 |
| 8 | CA/Pd(0) | H2O, K2CO3, 100 °C | 120 | 94 | 22 |
| 9 | Pd NP | H2O, K2CO3, 100 °C | 12h | 95 | 23 |
| 10 | Pd-PO(BA)@SBA-15 | PEG-400, Na2CO3, 80 °C | 10 | 97 | This work |
Conclusion
In summary, we synthesized the SBA-15 as a support, then its surface was modified using the IPTES ligand. Then the modified SBA-15 was functionalized by 2,2′-(propane-1,3-diylbis(oxy))dibenzenaminium chloride ligand (PO(BA)) for immobilization of palladium as an efficient nanocatalyst for organic reactions such as the Suzuki C–C coupling reaction. The synthesized Pd-PO(BA)@SBA-15 catalyst was characterized by SEM, ICP, TGA/DSC EDS, BET/BJH, and WDX techniques. TGA analysis showed thermal stability of this catalyst up to 230 °C. BET/BJH methods showed a high surface area for this catalyst, indicating suitable application as a practical catalyst. Pd-PO(BA)@SBA-15 was investigated as a friendly environmental catalyst in the synthesis of biphenyl derivatives. All products were successfully formed with high yields and high TOF values in very fast reaction rates in PEG-400 as a green and available solvent. Pd-PO(BA)@SBA-15 exhibited a good selectivity in the synthesis of biphenyl derivatives. In this work, various aryl halide (Ar–X) derivatives (having electron-donating functional or electron-withdrawing functional groups) were successfully coupled with various aryl boronic acid derivatives (such as phenylboronic acid, 4-methoxyphenylboronic acid and 4-formylphenylboronic acid). Also, Pd-PO(BA)@SBA-15 was reused 5 times without reducing its activity.Conflicts of interest
There are no conflict of declare.Data availability
All data generated or analyzed during this study are included in this published article and supplementary information (SI). Supplementary information: NMR data. See DOI: https://doi.org/10.1039/d5ra08145a.Acknowledgements
Authors thank the research facilities of Ilam University, Ilam, Iran, for financial support of this research project.References
- (a) R. Martin and S. L. Buchwald, Palladium-catalyzed Suzuki–Miyaura cross-coupling reactions employing dialkylbiaryl phosphine ligands, Acc. Chem. Res., 2008, 41(11), 1461–1473 CrossRef CAS PubMed; (b) M. C. D'Alterio, È. Casals-Cruañas, N. V. Tzouras, G. Talarico, S. P. Nolan and A. Poater, Mechanistic aspects of the palladium-catalyzed Suzuki–Miyaura cross-coupling reaction, Chem–Eur J., 2021, 27(54), 13481–13493 Search PubMed; (c) S. Nadri, M. Joshaghani and E. Rafiee, Biphenyl-based phosphine: A well-defined, air-stable, and efficient ligand for the Mizoroki–Heck reaction, Appl. Catal., A, 2009, 362(1–2), 163–168 Search PubMed; (d) S. Pasa, Y. S. Ocak, H. Temel and T. Kilicoglu, Synthesis, characterization and catalytic behavior in the Suzuki reaction of Schiff base and its complexes and the optical properties of nickel complex used in the fabrication of a photodiode, Inorg. Chim. Acta, 2013, 405, 493–504 CrossRef CAS.
- M. Farhang, A. R. Akbarzadeh, M. Rabbani and A. M. Ghadiri, A retrospective-prospective review of Suzuki–Miyaura reaction: From cross-coupling reaction to pharmaceutical industry applications, Polyhedron, 2022, 227, 116124 CrossRef CAS.
- (a) T. Niwa, Y. Uetake, M. Isoda, T. Takimoto, M. Nakaoka, D. Hashizume, H. Sakurai and T. Hosoya, Lewis acid-mediated Suzuki–Miyaura cross-coupling reaction, Nat. Catal., 2021, 4(12), 1080–1088 CrossRef CAS; (b) A. K. King, A. Brar, G. Li and M. Findlater, Homogeneous and recyclable palladium catalysts: Application in Suzuki–Miyaura cross-coupling reactions, Organometallics, 2023, 42(17), 2353–2358 CrossRef CAS.
- S. Safari, A. Ghorbani-Choghamarani, A. Monem and M. Rezaeivala, Preparation and identification of new magnetic heterogeneous nanostructural palladium catalyst, and its catalytic study in Suzuki and Stille reactions, Sci. Rep., 2025, 15(1), 1–21 Search PubMed.
- (a) A. M. Trzeciak and J. J. Ziółkowski, Structural and mechanistic studies of Pd-catalyzed CC bond formation: The case of carbonylation and Heck reaction, Coord. Chem. Rev., 2005, 249(21–22), 2308–2322 CrossRef CAS; (b) C. Yue, Q. Xing, P. Sun, Z. Zhao, H. Lv and F. Li, Enhancing stability by trapping palladium inside N-heterocyclic carbene-functionalized hypercrosslinked polymers for heterogeneous CC bond formations, Nat. Commun., 2021, 12(1), 1875 CrossRef CAS PubMed.
- (a) Sheetal, P. K. Gupta and R. K. Rawal, A Review of Design Strategies and Catalytic Applications of Polymer-Bound Palladium Systems for C– C Bond Formation via Heck and Suzuki Reactions, ChemistrySelect, 2025, 10(22), e00428 CrossRef CAS; (b) M. Akkoç, N. Buğday, S. Altın, N. Kiraz, S. Yaşar and İ. Özdemir, N-heterocyclic carbene Pd (II) complex supported on Fe3O4@ SiO2: Highly active, reusable and magnetically separable catalyst for Suzuki-Miyaura cross-coupling reactions in aqueous media, J. Organomet. Chem., 2021, 943, 121823 CrossRef; (c) B. Tahmasbi, M. Darabi, P. Moradi, Y. A. Tyula and M. Nikoorazm, Gadolinium Schiff-base complex on nanocomposite of graphene oxide magnetic nanoparticles as a robust, reusable and chemoselective nanocatalyst in the C–C coupling reactions, Polyhedron, 2024, 258, 117038 CrossRef CAS; (d) N. Emad-Abbas, J. Naji, P. Moradi and T. Kikhavani, 3-(Sulfamic acid)-propyltriethoxysilane on biochar nanoparticles as a practical, biocompatible, recyclable and chemoselective nanocatalyst in organic reactions, RSC Adv., 2024, 14(31), 22147–22158 RSC; (e) Y. Abbasi Tyula, P. Moradi and M. Nikoorazm, A New Neodymium Complex on Boehmite Nanoparticles with 1, 3-Bis (pyridine-3-ylmethyl) thiourea as a Practical and Reusable Nanocatalyst for the Chemoselective Synthesis of Tetrazoles, ChemistrySelect, 2023, 8(24), e202301674 CrossRef; (f) M. Ghasemirad, M. Norouzi and P. Moradi, A novel Cu complex coated on hercynite magnetic nanoparticles as an efficient and recoverable nanocatalyst for the selective synthesis of tetrazoles, J. Nanopart. Res., 2024, 26(1), 14 CrossRef CAS; (g) B. Tahmasbi, P. Moradi and M. Darabi, A new neodymium complex on renewable magnetic biochar nanoparticles as an environmentally friendly, recyclable and efficient nanocatalyst in the homoselective synthesis of tetrazoles, Nanoscale Adv., 2024, 6(7), 1932 RSC; (h) M. Norouzi, P. Moradi and M. Khanmoradi, Aluminium-based ionic liquid grafted on biochar as a heterogeneous catalyst for the selective synthesis of tetrazole and 2, 3-dihydroquinazolin 4 (1 H)-one derivatives, RSC Adv., 2023, 13(50), 35569–35582 RSC; (i) M. Norouzi and P. Moradi, A new copper complex on functionalized magnetic biochar nano-sized materials as sustainable heterogeneous catalysts for C–O bond formation in the natural deep eutectic solvent, Biomass Convers. Biorefinery, 2025, 15, 2465–2477 CrossRef CAS; (j) A. Ghorbani-Choghamarani, M. Nikoorazm, H. Goudarziafshar and B. Tahmasbi, An Efficient and New Method on the Oxidative Coupling of Thiols under Mild and Heterogeneous Conditions, Bull. Korean Chem. Soc., 2009, 30(6), 1388–1390 CrossRef CAS; (k) A. Zeinivand-Lorestani, A. Taheri and B. Tahmasbi, Chemoselective Oxidation of Sulfides and Oxidative Coupling of Thiols by a New Polyoxometalate and Heterogeneous Cobalt and Molybdenum Hybrid Nanocatalyst Stabilized on Modified Mesoporous CMK-3, Appl. Organomet. Chem., 2025, 39(4), e7997 CrossRef CAS.
- (a) R. Huirache-Acuña, R. Nava, C. L. Peza-Ledesma, J. Lara-Romero, G. Alonso-Núñez, B. Pawelec and E. M. Rivera-Muñoz, SBA-15 mesoporous silica as catalytic support for hydrodesulfurization catalysts, Materials, 2013, 6(9), 4139–4167 CrossRef PubMed; (b) H. Wang and C.-j. Liu, Preparation and characterization of SBA-15 supported Pd catalyst for CO oxidation, Appl. Catal., B, 2011, 106(3–4), 672–680 CrossRef CAS; (c) G. Du, S. Lim, M. Pinault, C. Wang, F. Fang, L. Pfefferle and G. L. Haller, Synthesis, characterization, and catalytic performance of highly dispersed vanadium grafted SBA-15 catalyst, J. Catal., 2008, 253(1), 74–90 CrossRef CAS.
- (a) P. I. Ravikovitch and A. V. Neimark, Characterization of nanoporous materials from adsorption and desorption isotherms, Colloids Surf., A, 2001, 187, 11–21 CrossRef; (b) M. Kruk, M. Jaroniec, S. H. Joo and R. Ryoo, Characterization of regular and plugged SBA-15 silicas by using adsorption and inverse carbon replication and explanation of the plug formation mechanism, J. Phys. Chem. B, 2003, 107(10), 2205–2213 CrossRef CAS.
- R. Cannon, B. Chiswell and L. M. Venanzi, Some complexes of cobalt (II), nickel (II), and palladium (II), with multidentate ligands and the ligand field strength of co-ordinated ethers and sulphides, J. Chem. Soc. Inorg. Phys. Theor., 1967, 1277–1281 RSC.
- A. Darabi, M. Nikoorazm, B. Tahmasbi and A. Ghorbani-Choghamarani, Palladium (0) complex of 1-(1H-benzo [d] imidazole-2-yl) guanidine on SBA-15 as an effective, selective, green, and robust catalyst for the Suzuki-Miyaura coupling reaction, Inorg. Chem. Commun., 2025, 180, 115083 CrossRef CAS.
- (a) N. Sharma, P. Sharma and S. Bhagat, Eco-friendly reactions in PEG-400: a highly efficient and green approach for stereoselective access to multisubstituted 3, 4-dihydro-2 (1 H)-quinazolines using 2-aminobenzylamines, RSC Adv., 2018, 8(16), 8721–8731 RSC; (b) A. Hasaninejad and M. Beyrati, Eco-friendly polyethylene glycol (PEG-400): a green reaction medium for one-pot, four-component synthesis of novel asymmetrical bis-spirooxindole derivatives at room temperature, RSC Adv., 2018, 8(4), 1934–1939 RSC.
- (a) S. Chehab, Y. Merroun, T. Ghailane, R. Ghailane, S. Boukhris and A. Souizi, Mono-ammonium phosphate fertilizer modified by cadmium: an efficient and recyclable catalyst for 2, 3-dihydroquinazolin-4 (1H)-one derivatives synthesis, Polycyclic Aromat. Compd., 2022, 42(9), 6199–6214 CrossRef CAS; (b) Y. Merroun, S. Chehab, T. Ghaılane, S. Boukhrıs, R. Ghaılane, N. Habbadı, A. Hassıkou, B. Lakhrıssı and A. Souızı, An effective method to synthesize 2, 3-dihydroquinazolin-4 (1H)-One using phosphate fertilizers (MAP, DAP and TSP) as green heterogeneous catalysts, J. Turk. Chem. Soc., Sect. A, 2018, 5(1), 303–316 Search PubMed.
- Y. Merroun, S. Chehab, A. El Hallaoui, R. Ghailane, S. Boukhris and A. Souizi, Tin pyrophosphate (SnP2O7): as a novel heterogeneous and highly efficient catalyst for the one pot-three component synthesis of Tetrahydrobenzo [b] pyran and Dihydropyran [c] chromene derivatives, Iran. J. Chem. Chem. Eng., 2023, 42(7), 2115–2130 CAS.
- P. Moradi, M. Hajjami and F. Valizadeh-Kakhki, Biochar as heterogeneous support for immobilization of Pd as efficient and reusable biocatalyst in C–C coupling reactions, Appl. Organomet. Chem., 2019, 33(11), e5205 CrossRef CAS.
- Y.-Y. Peng, J. Liu, X. Lei and Z. Yin, Room-temperature highly efficient Suzuki–Miyaura reactions in water in the presence of Stilbazo, Green Chem., 2010, 12(6), 1072–1075 RSC.
- P. Moradi and M. Hajjami, Stabilization of ruthenium on biochar-nickel magnetic nanoparticles as a heterogeneous, practical, selective, and reusable nanocatalyst for the Suzuki C–C coupling reaction in water, RSC Adv., 2022, 12(21), 13523–13534 RSC.
- S. M. Islam, A. S. Roy, P. Mondal and N. Salam, Selective hydrogenation and Suzuki cross-coupling reactions of various organic substrates using a reusable polymer-anchored palladium (II) Schiff base complex, Appl. Organomet. Chem., 2012, 26(11), 625–634 CrossRef CAS.
- H. A. Patel, A. L. Patel and A. V. Bedekar, Polyaniline-anchored palladium catalyst-mediated Mizoroki–Heck and Suzuki–Miyaura reactions and one-pot Wittig–Heck and Wittig–Suzuki reactions, Appl. Organomet. Chem., 2015, 29(1), 1–6 CrossRef CAS.
- T. Chen, J. Gao and M. Shi, A novel tridentate NHC–Pd (II) complex and its application in the Suzuki and Heck-type cross-coupling reactions, Tetrahedron, 2006, 62(26), 6289–6294 CrossRef CAS.
- Q. Xu, W.-L. Duan, Z.-Y. Lei, Z.-B. Zhu and M. Shi, A novel cis-chelated Pd (II)–NHC complex for catalyzing Suzuki and Heck-type cross-coupling reactions, Tetrahedron, 2005, 61(47), 11225–11229 CrossRef CAS.
- P. Moradi and M. Hajjami, Magnetization of graphene oxide nanosheets using nickel magnetic nanoparticles as a novel support for the fabrication of copper as a practical, selective, and reusable nanocatalyst in C–C and C–O coupling reactions, RSC Adv., 2021, 11(42), 25867–25879 RSC.
- V. W. Faria, D. G. Oliveira, M. H. Kurz, F. F. Gonçalves, C. W. Scheeren and G. R. Rosa, Palladium nanoparticles supported in a polymeric membrane: an efficient phosphine-free “green” catalyst for Suzuki–Miyaura reactions in water, RSC Adv., 2014, 4(26), 13446–13452 RSC.
- M. Nasrollahzadeh, S. M. Sajadi and M. Maham, Green synthesis of palladium nanoparticles using Hippophae rhamnoides Linn leaf extract and their catalytic activity for the Suzuki–Miyaura coupling in water, J. Mol. Catal. A: Chem., 2015, 396, 297–303 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |