Open Access Article
Open Access ArticleA portable paper-based and smartphone-assisted colorimetric sensor for copper oxychloride
Muhammad Adnan Samia,
Daim Asif Rajaabc,
Imdad Ali ae,
Farid Ahmedd,
Mahmood Fazala,
Muhammad Raza Shah
ae,
Farid Ahmedd,
Mahmood Fazala,
Muhammad Raza Shah *a and
Muhammad Imran Malik
*a and
Muhammad Imran Malik *ab
*ab
aH.E.J. Research Institute of Chemistry, International Center for Chemical and Biological Sciences (ICCBS), University of Karachi, Karachi 75270, Pakistan. E-mail: raza.shah@iccs.edu; mimran.malik@iccs.edu
bThird World Center for Science and Technology, International Center for Chemical and Biological Sciences (ICCBS), University of Karachi, Karachi 75270, Pakistan
cSchool of Science, Minzu University of China, Beijing 100081, China
dInstitute for Advanced Study, Shenzhen University, Shenzhen 518060, China
eDepartment of Chemistry, University of Selcuk, Konya, 42031, Turkey
First published on 25th February 2026
Abstract
Copper oxychloride (CuOxy) is a copper-containing fungicide and bactericide used to combat fungal diseases in fruits, vegetables, and ornamental crops. The maximum residue limit (MRL) for CuOxy is crop- and region-specific. The MRL for CuOxy is set in a range of 5–30 mg kg−1 for different sources by the European Food Safety Authority (EFSA). Herein, we report a novel colorimetric sensing platform based on coumarin triazole-functionalized silver nanoparticles (CT-AgNPs) for rapid, selective, and highly sensitive detection of CuOxy. The CT-AgNPs-based sensor exhibits excellent sensitivity, with a limit of detection (LoD) of 0.007 µM, a limit of quantification (LoQ) of 0.023 µM, and a linear detection range (LDR) of 0.1–100 µM. The system is efficiently integrated with AI-assisted smartphone detection and paper-based analytical devices (PADs) for real-time, on-site detection of CuOxy. Under optimized conditions, the sensor demonstrated outstanding selectivity, with no substantial interference from other environmental contaminants. The sensor's real-world applicability was verified through testing on actual samples, including tap and river water, along with different fruits and vegetables. To our knowledge, this is the first report of a CuOxy sensor combining both smartphone integration and paper-based analysis. The approach demonstrated high recovery rates in various environmental samples, validating its capability for quantitative CuOxy monitoring and offering a promising tool for ensuring food safety and environmental health.
1 Introduction
Water is an important and crucial ingredient for life. More than 70% of the planet Earth is water, of which 68% is saline, and 3% is freshwater. Life on earth primarily depends on freshwater, which anthropogenic activities have polluted; the three most dominant contributors are human settlements, industries, and agriculture.1–3 Many emerging pollutants, including antibiotics, cosmetics, and pesticides, are continuously polluting the freshwater reservoirs of the world. This increasing contamination renders the emergence of severe public health issues.4,5Pesticides are one of the major classes of emerging pollutants classified as herbicides, rodenticides, and fungicides. As per a recent estimate, approximately 33% of crops are damaged by pests, having a direct impact on their yield.6,7 Therefore, the use of pesticides is essential for improved production of cultivable crops. Thus, the annual worldwide use of pesticides has exceeded 3.5 million tons.8 Due to excessive use, significant amounts of different pesticides are released into environmental compartments, such as soil and water. Typically, the pesticides detected in water are in low concentrations, i.e., from low ng L−1 to a few µg L−1. Nonetheless, these are still harmful to human life and aquatic organisms. Owing to the public health concern, government agencies offer guidelines regarding the minimization of the use of harmful pollutants, synthesizing less harmful pesticides, and decreasing maximum concentration limits of these pesticides in freshwater.9–14 Thus, fast, robust, and economic analytical methodologies for quantitative detection of pesticides in real samples are highly sought-after.15,16
Copper oxychloride (CuOxy) is a copper-containing fungicide and bactericide used to combat fungal diseases in fruits, vegetables, and ornamental crops. The maximum residue limit (MRL) for CuOxy is crop- and region-specific. The MRL for CuOxy is set in a range of 5–30 mg kg−1 for different sources by the European Food Safety Authority (EFSA). For quantitative determination of CuOxy, various analytical methods have been reported such as flow injection chemiluminescence (FIA),17 gas chromatography-mass spectrometry (GC-MS),18 reverse phase high performance liquid chromatography with ultraviolet detection (RP-HPLC-UV),19 liquid chromatography-mass spectrometry (LC-MS),20 electrothermal atomic absorption spectroscopy (ETAAS),21 liquid chromatography-mass spectrometry/atomic absorption spectroscopy (LCMS/AAS),22 and diffuse reflectance-near infrared spectroscopy (DIR-NIR).22 However, most of these methods are associated with certain caveats, such as pre-treatment of the sample, requiring well-trained technicians, high cost, long protocols, low sensitivity, and limited selectivity. To overcome these limitations, the development of facile, fast, efficient, and accurate CuOxy detection methods in environmental samples is a challenging task. Such methods are needed for real-time, accurate, on-the-spot, and visual monitoring in the context of food safety and human health.23–25
With the rapid progress of nanotechnology, nanoparticles have been extensively explored for their diverse research applications and unique physicochemical attributes.26–30 Metal nanoparticles (MNPs) are particularly important because they can undergo surface electron transitions from the valence band to the conduction band when interacting with electromagnetic radiation. This unique behavior confers them outstanding catalytic and optical properties, supporting their use in a broad range of applications, including catalysis and contaminant removal,31 sensing,17,32 and drug delivery.33–42 Especially, silver (Ag) and gold (Au) nanoparticles (NPs) have drawn considerable interest in sensor technologies because of their distinctive optical characteristics. Their surface plasmon resonance (SPR) bands, typically observed between 200 and 750 nm, shift noticeably in response to external species, reflecting changes in the surrounding environment of NPs. Despite these advantages, metal nanoparticles face the challenge of rapid aggregation, which reduces their stability and functional performance. To counteract this issue, diverse capping agents are utilized, ranging from natural biomolecules such as proteins, polysaccharides, and flavonoids to synthetic stabilizers like organic molecules and polymers.37,40,43–46 Silver nanoparticles stabilized with triazole moiety (ligands) have been widely used as a probe colorimetric sensor for the detection of heavy metal ions,47,48 drugs, and pesticides.49 Stabilized silver nanoparticles (AgNPs) are widely used in sensing applications, as their aggregation or stabilization is triggered by interactions between surface functional groups and the analyte, a process that largely depends on the type of stabilizing agent and the nature of the analyte.3
Developing on-the-spot assays for environmental contaminants like pesticides and drugs is a key challenge for analytical chemists. The combination of colorimetric assays and portable instruments has significantly advanced the real-time analysis. These assays indicate contaminant levels through color changes, enhanced by smartphones with high-resolution cameras and AI-inspired applications like color meter, which measure RGB values to correlate color intensity with concentration. Colorimetric assays can be converted into paper-based analytical devices (PADs), where color changes are analyzed with AI-inspired applications like ImageJ to determine grayscale values. Various assays, including enzyme assays, fluorescence-based chemical sensing, multimodal analysis, polymer and metal nanoparticle-based methods, facilitate the development of such on-the-spot sensors.28,50–55
In this context, coumarin-triazole-based ligands provide a highly versatile framework for the stabilization and functionalization of silver nanoparticles (AgNPs), while simultaneously enabling selective metal-ion detection.56 The triazole moiety, enriched with multiple nitrogen donor atoms, exhibits strong affinity toward the AgNP surface, leading to enhanced nanoparticle stability, controlled growth, and long-term colloidal robustness.57 Moreover, synergistic coordination between metal cations and triazole nitrogen atoms, together with coumarin-derived donor sites, facilitates regulated interparticle aggregation and modulates the surface electronic properties of AgNPs.58
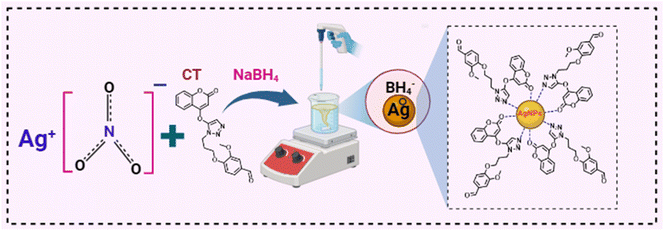
In this study, we report the synthesis of coumarin triazole-based conjugated silver nanoparticles (CT-AgNPs) and their application as a sensing material. CT-AgNPs had a distinct pale yellow color and demonstrated a stable LSPR absorbance peak at 408 nm. Upon CuOxy introduction, the color of CT-AgNPs immediately turned light pink due to aggregation, accompanied by significant hypochromic and bathochromic shifts with a visible spectral broadening. This visual color change was seamlessly integrated with smartphone-asisted PADs for on-site detection of CuOxy. Furthermore, the CT-AgNPs were successfully applied for colorimetric, smartphone, and paper-based CuOxy detection in real environmental samples such as river and tap water, as well as food samples including guava, orange, turnip, and spinach. The developed CuOxy sensor demonstrated excellent sensitivity and selectivity for the on-site detection of CuOxy in environmental and agricultural samples, eliminating the need for a dedicated laboratory setup.
2 Experimental section
2.1 Materials and instrumentation
Vanillin, propargyl bromide, potassium carbonate, sodium azide, copper sulphate, sodium ascorbate, and ethyl 2-(2-(2-chloroacetamido) thiazole-4-yl) acetate were purchased from Merck (Germany). Various pesticides used for interference study, sodium borohydride (NaBH4) and silver nitrate (AgNO3), were acquired from Sigma Aldrich (USA). All the solutions were prepared in HPLC-grade solvents and DI water. All the sensing experiments were performed in DI water at room temperature.All glassware was rigorously cleaned using 10% nitric acid, thoroughly rinsed with DI water, and then oven-dried to eliminate residual contaminants.
A Hanna Instruments HI2211 pH meter, equipped with a glass electrode and an Ag/AgCl reference electrode, was used to measure the pH values. UV-Visible absorbance spectra (300–800 nm) were obtained using a Shimadzu UV-1800 spectrophotometer (Japan) with measurements performed in quartz cuvettes having a 1 cm optical path length. The structural characterization of the synthesized compound was carried out using proton nuclear magnetic resonance (1H-NMR) spectroscopy on a Bruker AV-300 spectrometer, with tetramethylsilane (TMS) serving as the internal reference. Spectral data were analyzed using Topspin software (version 3.6.1). The molecular mass of CT was identified using a JEOL-600H mass spectrometer. To examine the structural properties of CT-AgNPs, Powder X-ray diffraction (PXRD) analysis was conducted using a Bruker D8 Advance powder diffractometer. The instrument was operated with Cu-Kα radiation (λ = 1.5406 Å) as the X-ray source. The measurements were taken at 40 kV and 25 mA, with scanning performed over a 2θ range of 20° to 90° at an incremental step size of 0.05° per min. FT-IR characterization was performed using a Bruker Vector 22 spectrometer, equipped with a deuterated triglycine sulfate (DTGS) detector. Spectral data were collected in the mid-IR range of 4000 to 500 cm−1 using the KBr pellet technique. Each spectrum was acquired at a resolution of 0.1 cm−1, averaging ten scans per sample. The size distribution and surface charge (zeta potential) of CT-AgNPs were evaluated with a Nano-ZSP zeta sizer (Malvern Instruments) at 25 °C. Measurements were conducted at a fixed scattering angle of 90°, employing a disposable cuvette for particle sizing and a specialized zeta potential cell for analysis.
Topographical characterization of CT-AgNPs, both before and after the drug incorporation, was carried out using an Agilent 5500 Atomic Force Microscope (AFM) operating in tapping mode. The system was equipped with a triangular silicon nitride cantilever (Veeco MLCT-AUHW), featuring a spring constant of 0.1 N m−1. For sample preparation, a drop of the NP suspension was placed onto a silicon wafer and allowed to dry at room temperature for 24 h. Surface morphology analysis via field emission scanning electron microscopy (FE-SEM) was performed using a Thermo Fisher Scientific Apero 2C LoVac SEM. Before imaging, the samples were gold-coated using a Quorum Technologies SC7620 mini sputter coater, with argon employed as the sputtering gas, resulting in an approximate coating thickness of 153 Å. Samples were prepared by drop-casting onto silicon wafers and dried under ambient conditions for 24 h before examination.
2.2 Synthesis of nanosensor
The synthesis procedure (Scheme S1-SI) and structural characterization of the Coumarin triazole are provided in the SI. The synthesized CT was thoroughly analyzed using various techniques, including fast atom bombardment mass spectrometry (FAB-MS) (Fig. S1-SI), and proton nuclear magnetic resonance (1H-NMR) spectroscopy (Fig. S2-SI).A 0.1 mM solution of CT was prepared by dissolving 2.17 mg of CT in 1 mL of DMSO, followed by dilution to a final volume of 50 mL with deionized (DI) water. Separately, aqueous solutions of 0.1 mM AgNO3 and 5.0 mM NaBH4 were prepared using DI water.
For the fabrication of nanoparticles, the CT, AgNO3, and NaBH4 solutions were mixed in a volumetric ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.1, respectively. Initially, a 0.1 mM AgNO3 solution was transferred to a reaction flask and stirred magnetically at room temperature for 30 minutes. Subsequently, the 5.0 mM NaBH4 solution was added dropwise using a micropipette over a period of 15 min under continuous stirring. The reaction mixture was then stirred for an additional 1 h to ensure complete reduction of Ag+ ions.
0.1, respectively. Initially, a 0.1 mM AgNO3 solution was transferred to a reaction flask and stirred magnetically at room temperature for 30 minutes. Subsequently, the 5.0 mM NaBH4 solution was added dropwise using a micropipette over a period of 15 min under continuous stirring. The reaction mixture was then stirred for an additional 1 h to ensure complete reduction of Ag+ ions.
The formation of CT-functionalized silver nanoparticles (CT-AgNPs) was confirmed by the appearance of a characteristic yellow coloration. Furthermore, the stability of the synthesized CT-AgNPs was systematically evaluated under various environmental conditions, including different pH levels, temperatures, and electrolyte concentrations. The resulting CT-AgNPs were characterized using a combination of spectroscopic and microscopic methods, including UV-Vis absorption spectroscopy, field emission scanning electron microscopy (FE-SEM), dynamic light scattering (DLS), atomic force microscopy (AFM), and powder X-ray diffraction (PXRD).
2.3 Detection procedure for CuOxy by colorimetric, smartphone, and paper-based sensors
2.4 Detection of CuOxy in real samples
CT-AgNPs were employed for the detection of copper oxychloride (CuOxy) in six different real samples. Laboratory tap water, diluted two-fold with deionized (DI) water, was used according to the established protocol. In addition, four food matrices—guava, orange, turnip, and spinach—were procured from a local Metro vegetable market in Karachi. Environmental water samples were collected from the University of Karachi and the Sindh River.For sample preparation, 20 g of each finely chopped fruit or vegetable was homogenized with 100 mL of DI water using a blender. The volume was then adjusted to 200 mL, followed by filtration through a 0.45 µm polytetrafluoroethylene (PTFE) syringe filter. Each real sample was spiked with CuOxy to obtain a final concentration of 100 µM.
The prepared samples were subsequently analyzed using three different colorimetric approaches, following the protocol described in Section 2.3, and the results were further validated by UPLC analysis. Each measurement was performed three times to ensure precision in quantification based on the previously established calibration curve.
Quantification was achieved using a calibration curve generated through linear regression analysis, and CuOxy recovery percentages were determined using eqn (1).
 | (1) |
2.5 UPLC analysis of CuOxy in different samples
The efficacy of the proposed CuOxy detection technique was validated using a variety of real samples and benchmarked against an ultra-performance liquid chromatography (UPLC) method.17 Chromatographic separation was achieved on an Octadecyl Silica column (250 × 6.0 mm, JH08S04-2506WT) connected to a diode array detector (DAD). The absorbance at the wavelength of 254 nm was taken as a standard and used for quantification.The mobile phase consisted of 30% Milli-Q water and 70% acetonitrile (ACN) delivered at a flow rate of 1.0 mL min−1, with an injection volume of 1.0 µL. All analyses were conducted under isocratic conditions, and the column temperature was maintained at 30 °C.
Standard solutions of CuOxy were prepared in Milli-Q water, with the pH adjusted to acidic conditions using few drops of acetic acid. A calibration curve was constructed by plotting CuOxy concentrations in the range of 40–140 µM against the corresponding peak area values. Quantification of CuOxy in real water and food samples was carried out using this calibration curve. To evaluate the accuracy and recovery of the method, real samples were spiked with CuOxy at a concentration of 100 µM and subsequently analyzed by UPLC.
3 Results and discussion
Details regarding the synthesis and characterization of coumarin triazole (CT) are provided in the SI. The text herein focuses on the fabrication and analytical characterization of CT-AgNPs, along with their assessment as a colorimetric sensing platform.3.1 Synthesis and characterization of CT-AgNPs
The synthesis of CT-AgNPs was accomplished through the chemical reduction method of AgNO3 using NaBH4 as a reducing agent, while coumarin triazole (CT) as a stabilizer, Scheme 1. Firstly, different concentrations of CT were used alongwith the 0.1 mM AgNO3, and 0.1 mM provided the maximum absorption and was taken as the optimum for CT, Fig. S3-SI. To optimize the stability of CT-AgNPs, different volume ratios of 0.1 mM CT and 0.1 mM AgNO3 solutions ranging from 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 to 1
3 to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 were tested. Initially colorless CT solution turned yellow upon the formation of CT-AgNPs, as illustrated in Fig. 1A. The appearance of a vivid yellow color along with a pronounced SPR peak at 408 nm confirms the successful synthesis of CT-AgNPs, while CT had a characteristic absorption peak at 314 nm (Fig. 1A). The most intense SPR signal was recorded at a CT to AgNO3 solutions ratio of 1
15 were tested. Initially colorless CT solution turned yellow upon the formation of CT-AgNPs, as illustrated in Fig. 1A. The appearance of a vivid yellow color along with a pronounced SPR peak at 408 nm confirms the successful synthesis of CT-AgNPs, while CT had a characteristic absorption peak at 314 nm (Fig. 1A). The most intense SPR signal was recorded at a CT to AgNO3 solutions ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10, Fig. 1B. Therefore, the optimized ratio (1
10, Fig. 1B. Therefore, the optimized ratio (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10) was selected for the subsequent experiments involving CT-AgNPs.
10) was selected for the subsequent experiments involving CT-AgNPs.
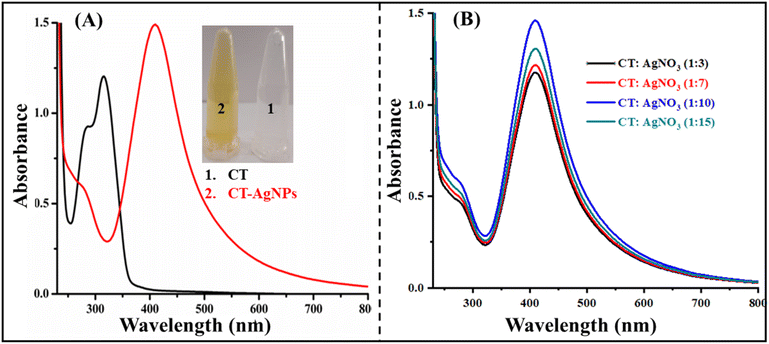 | ||
| Fig. 1 (A) UV-Visible spectra of CT and CT-AgNPs (1–10); (B) optimization of the ratio CT and AgNO3 for CT-AgNPs synthesis through UV-Visible analysis. | ||
The stability of CT-AgNPs was systematically investigated under various environmental parameters, as shown in Fig. 2. The stability of CT-AgNPs was monitored over 30 days under controlled storage conditions. The CT-AgNPs were kept at room temperature in sealed vials wrapped in aluminium foil to prevent light exposure. As shown in Fig. 2A, the SPR peak at ∼420 nm remained sharp and unchanged throughout the study, with no noticeable peak shift or decrease in intensity. This indicates that the CT-AgNPs maintained excellent colloidal stability with no significant aggregation or degradation under these storage conditions. Elevated electrolyte concentrations frequently promote nanoparticle aggregation.36 CT-AgNPs were exposed to different NaCl concentrations from 0.1 mM to 1000 mM to evaluate ionic strength tolerance. A noticeable decrease in SPR intensity and red shift at higher salt concentrations (250 mM) indicated partial aggregation, suggesting reduced stability under strong ionic conditions,59 Fig. 2B. However, the CT-AgNPs remain quite stable under practical electrolyte concentrations of real samples. In addition, the thermal stability of CT-AgNPs was evaluated by subjecting the CT-AgNPs solution to a boiling temperature (100 °C). An increase in absorbance intensity was observed upon heating, indicating that the CT-AgNPs retained their stability after high-temperature treatment (Fig. 2C). The pH of the synthesized CT-AgNPs was 7. The stability of CT-AgNPs was further assessed across a pH range from 1 to 12. The CT-AgNPs maintained stability in the pH range of 4–10, whereas extreme acidic and basic conditions led to broadened and diminished SPR bands, indicating aggregation (Fig. 2D). As a result, pH 7 was selected for subsequent experimental procedures because it corresponds to the native pH of the synthesized CT-AgNPs, maintains nanoparticle stability within the pH window of 4–10, and matches the pH of real environmental samples. These findings confirm the suitability of CT-AgNPs for applications under physiological and moderately varied environmental conditions.
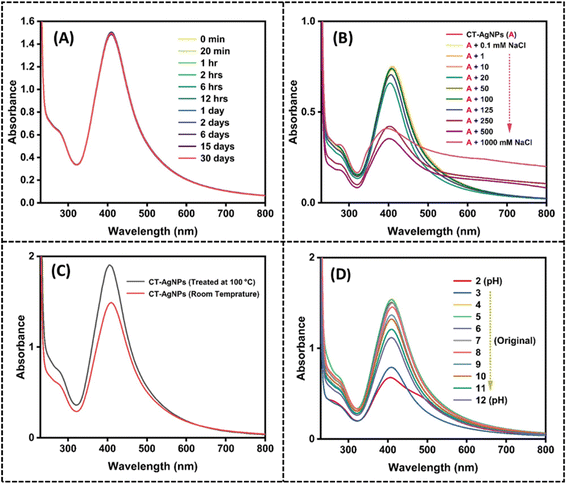 | ||
| Fig. 2 Stability of CT-AgNPs through UV-Visible analysis as a function of (A) storage time, (B) ionic strength, (C) temperature, (D) pH. | ||
The average size, surface charge, and surface morphology of CT-AgNPs were investigated using DLS, AFM, and SEM. The DLS data revealed that the average particle size of CT-AgNPs was found to be 40.89 nm with a polydispersity index of 0.38, Fig. 3A. Additionally, zeta potential measurements of CT-AgNPs were found to be −28.7 mV, which shows the excellent stability of AgNPs (Fig. 3B). Moreover, the results obtained from DLS were further endorsed by AFM and SEM. The AFM topographical image a show a spherical morphology of CT-AgNPs, Fig. 3C and E. Fig. 3D depicts the histogram as obtained by AFM that revealed the average size of 34.8 nm. FE-SEM analysis demonstrated that the average particle size of CT-AgNPs was 38 nm with well-defined spherical morphology (Fig. 3F). The size of the CT-AgNPs predominantly remains within range of 30–40 nm. PXRD analysis was conducted to investigate the crystalline nature of silver within the CT-AgNPs. The resulting diffraction pattern displayed Bragg reflections at 2θ angles of 39.04°, 48.1°, 67.3°, and 77.2°, which are indicative of the (111), (200), (220), and (311) crystallographic planes, respectively, Fig. 3G.60 The diffraction pattern confirms that the AgNPs possess a face-centered cubic (fcc) crystal structure. Additionally, the obtained XRD results are in good agreement with previously reported data for silver nanoparticles.61,62
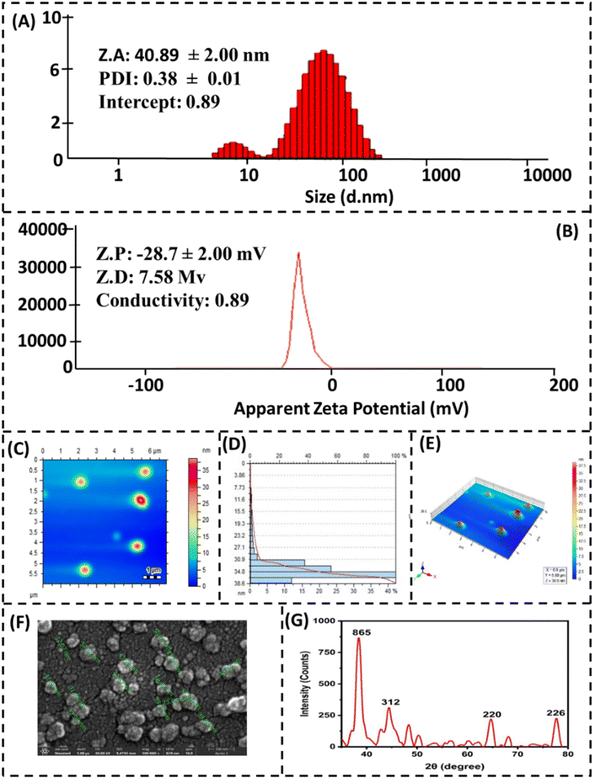 | ||
| Fig. 3 Characterization of CT-AgNPs, (A) zeta size, (B) zeta potential, (C) AFM, (D) histogram obtained by AFM, (E) three dimensional image by AFM, (F) FE-SEM, (G) XRD. | ||
3.2 CT-AgNPs as a colorimetric sensor
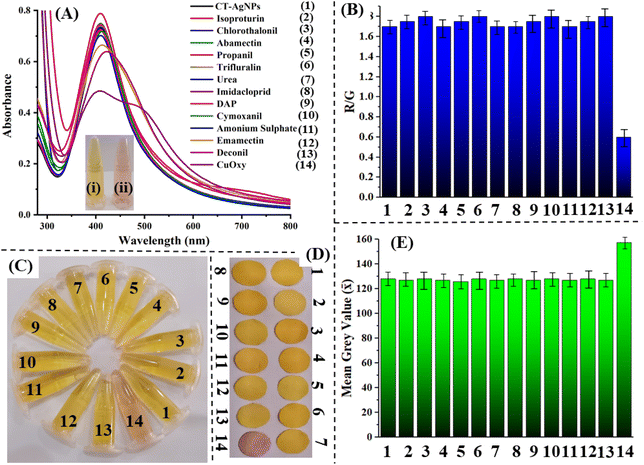
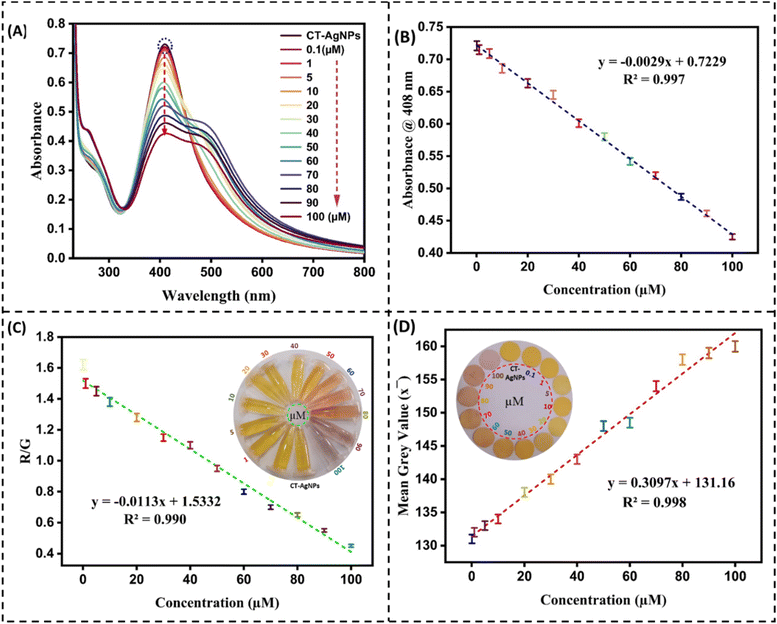
In this study, the influence of the addition of various drugs and pesticides/fungicides on the SPR band behavior of CT-AgNPs was evaluated. Twelve structurally diverse pesticides/fungicides, namely isoproturon, chlorothalonil, abamectin, propanil, trifluralin, urea, imidacloprid, diammonium phosphate (DAP), cymoxanil, ammonium sulfate, emamectin, and deconil, were introduced into the CT-AgNPs solution. The addition of these compounds produced no noticeable color alteration, indicating absence of any interaction with the CT-AgNPs. In contrast, introducing CuOxy to the CT-AgNPs solution led to a prompt visual change from pale yellow to light pink. This unique interaction was confirmed through UV-Vis spectroscopic analysis, where all other analytes caused negligible variations in the SPR band, while CuOxy significantly decreased the absorbance intensity and induced a bathochromic (red) shift, with the SPR peak moving to approximately 545 nm and becoming broader, Fig. 4A. The inset shows the color change in CT-AgNPs after the introduction of CuOxy in them. These findings strongly suggest that the CT-AgNPs exhibit selective binding and specific optical response only to CuOxy, establishing their potential as a targeted colorimetric sensor.A rapid and on-site method for detecting and quantifying CuOxy was implemented using a standard Android smartphone. CT-AgNPs were placed in a transparent Eppendorf and positioned inside a fume hood under consistent LED lighting, Fig. 4C. To evaluate the selectivity of the CT-AgNPs-based detection system, 100 µM solutions of the previously mentioned analytes were introduced individually, and any resulting color changes were monitored. Digital images of each sample were captured using the smartphone camera after analyte addition. A noticeable color change in the CT-AgNPs was observed exclusively upon the addition of CuOxy, while other analytes produced negligible effects. Subsequently, the RGB intensities of the solutions were measured using a smartphone color analysis application. A bar graph was generated based on the red-to-green (R/G) ratios of CT-AgNPs before and after analyte addition. The R/G ratio remained approximately 1.75 for all analytes except CuOxy, which exhibited a decrease to 0.6, Fig. 4B.
In addition, a paper-based colorimetric sensor incorporating CT-AgNPs was fabricated and integrated with a smartphone for visual detection. Specific zones on the sensor paper were allocated to different analytes at a fixed concentration of 100 µM. No noticeable color alteration was observed in any of the zones except for the one containing CuOxy, which changed from yellow to a light pink color. This visible shift was captured using a smartphone camera. The resulting images were analyzed using FIJI software to assess color histograms and calculate gray values. The initial mean gray value of the CT-AgNPs was recorded as 128. Following the application of various analytes, no significant deviation in this value was detected. However, upon exposure to CuOxy, the mean grey value increased to 158, highlighting the system's sensitivity and specificity. This confirms the sensor's effectiveness for CuOxy detection, Fig. 4D and E. It is pertinent to mention here that the smartphone/paper-based sensor operates as a single-use device, similar to pH indicator strips; therefore, reusing the same strip for repeated measurements is not feasible. Reproducibility was evaluated by performing the assay in triplicate using freshly prepared strips, and the results are presented with error bars to demonstrate measurement consistency.
The changes in the average size, surface charge, and surface morphology of CT-AgNPs after the addition of CuOxy were evaluated using the aforementioned techniques. DLS revealed an increase in the size of CT-AgNPs to 215 nm upon CuOxy incorporation, Fig. 5A. This increase was accompanied by a minor rise in the polydispersity index from 0.38 to 0.42, suggesting a broader particle size distribution and supporting the aggregation behavior. Additionally, a shift in zeta potential from −28.7 mV to −19.6 mV was observed after CuOxy exposure, Fig. 5B, indicating reduced colloidal stability. Furthermore, the observations gathered from DLS were confirmed by AFM and FE-SEM analyses, which also showed an increase in particle size, Fig. 5C and D.
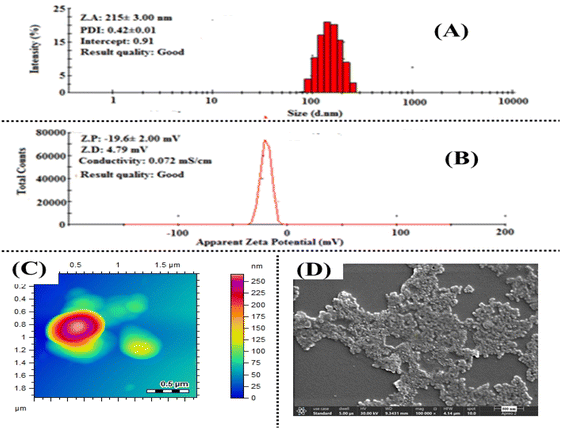 | ||
| Fig. 5 Characterization of CT-AgNPs/CuOxy complex (A) average size through DLS, (B) zeta potential, (C) AFM, (D) FE-SEM. | ||
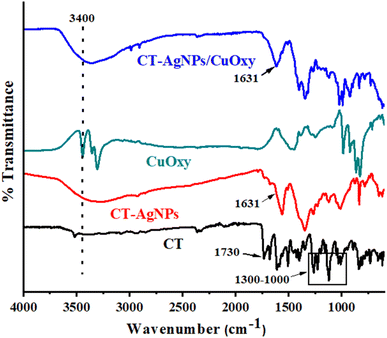
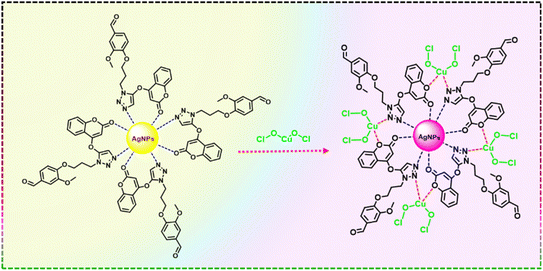
FT-IR spectroscopy was used to examine the functional group interactions that stabilize AgNPs with CT and their response to CuOxy (Fig. 6). The FT-IR spectrum of pure CT shows a prominent absorption band at 1730 cm−1, reflecting the stretching vibration of the carbonyl (–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) group in the coumarin part. Additionally, several bands in the 1300–1000 cm−1 range are assigned to C–O and C–N stretching vibrations, characteristic of the coumarin triazole backbone. When CT-AgNPs form, notable spectral changes occur. The intensity of the carbonyl band at 1730 cm−1 decreases substantially, and modifications are also seen in the C–O and C–N stretching bands in the 1300–1000 cm−1 region. These observations reveal that the carbonyl, ether, and amine groups of CT are directly involved in binding and capping AgNPs, confirming their role in stabilising the nanoparticles. The FT-IR spectrum of CuOxy shows characteristic vibrational features, while in the CT-AgNPs/CuOxy composite, a broad absorption band around 3400 cm−1 becomes evident. This band is linked to O–H stretching vibrations, indicating hydrogen bonding between CuOxy and surface-bound CT ligands. Notably, the band at 1631 cm−1, related to AgNP formation, remains unchanged in both the spectra of CT-AgNPs and the CT-AgNPs/CuOxy complex. This suggests that CuOxy does not directly interact with the AgNP core; instead, it interacts mainly through the terminal functional groups of the CT ligands attached to the nanoparticle surface. These ligand-mediated interactions maintain the structural integrity of the AgNPs and facilitate effective recognition and sensing of CuOxy. Based on these FT-IR findings, a plausible mechanism for the CuOxy detection is proposed and depicted in Scheme 2.
O) group in the coumarin part. Additionally, several bands in the 1300–1000 cm−1 range are assigned to C–O and C–N stretching vibrations, characteristic of the coumarin triazole backbone. When CT-AgNPs form, notable spectral changes occur. The intensity of the carbonyl band at 1730 cm−1 decreases substantially, and modifications are also seen in the C–O and C–N stretching bands in the 1300–1000 cm−1 region. These observations reveal that the carbonyl, ether, and amine groups of CT are directly involved in binding and capping AgNPs, confirming their role in stabilising the nanoparticles. The FT-IR spectrum of CuOxy shows characteristic vibrational features, while in the CT-AgNPs/CuOxy composite, a broad absorption band around 3400 cm−1 becomes evident. This band is linked to O–H stretching vibrations, indicating hydrogen bonding between CuOxy and surface-bound CT ligands. Notably, the band at 1631 cm−1, related to AgNP formation, remains unchanged in both the spectra of CT-AgNPs and the CT-AgNPs/CuOxy complex. This suggests that CuOxy does not directly interact with the AgNP core; instead, it interacts mainly through the terminal functional groups of the CT ligands attached to the nanoparticle surface. These ligand-mediated interactions maintain the structural integrity of the AgNPs and facilitate effective recognition and sensing of CuOxy. Based on these FT-IR findings, a plausible mechanism for the CuOxy detection is proposed and depicted in Scheme 2.
3.3 Analytical figures of merit
The primary objective of this study is to synthesize a sensing material with selectivity, sensitivity, and specificity for CuOxy. The presence of CuOxy in CT-AgNPs caused a noticeable colour change, which was confirmed by a hypochromic shift and the band broadening in the SPR band (Fig. 4A). However, it is essential to assess the sensitivity of CT-AgNPs in the context of quantification. Hence, CT-AgNPs were exposed to different concentrations of CuOxy. When 0.1 µM CuOxy was added, the SPR band of CT-AgNPs underwent a slight hypochromic shift at 408 nm, which consistently decreased till the addition of 100 µM CuOxy, Fig. 7A. A direct linear correlation between the quantity of added CuOxy and the change in the intensity of the SPR band at 408 nm in the concentration range of 0.1 to 100 µM is established (Fig. 7B). The newly developed method for sensing CuOxy had a LOD of 0.007 µM and a LOQ of 0.023 µM, calculated as 3.3σ/S and 10σ/S, respectively, in a linear dynamic range of 0.1–100 µM.63The analytical performance of the PADs and smartphone-based CuOxy sensing devices developed using CT-AgNPs was also thoroughly investigated. The sensitivity of the proposed detection technique was assessed using a smartphone by varying the CuOxy concentration in the range of 0.1 to 100 µM in an Eppendorf containing CT-AgNPs solution (Fig. 7C, inset). At each concentration, images of the Eppendorf tubes were recorded and analyzed using a color-picking application. The corresponding R, G, and B values were extracted and transferred to a computer for linear fitting. Among the different parameters tested, the R/G ratio exhibited the best linear correlation with CuOxy concentration over the range of 0.1–100 µM, Fig. 7C. The corresponding LoD and LoQ were calculated to be 0.009 µM and 0.03 µM, respectively.
In addition, 5.0 µL of CuOxy solution with concentrations ranging from 0.1 to 100 µM was precisely applied to the detection zone of the paper-based sensor using a micropipette (Section 2.3.3). The subsequent color variation in the detection zone was documented with a smartphone camera. A clear trend was observed, as higher CuOxy concentrations produced a progressively lighter pink coloration. To quantitatively analyse this change, the captured images were processed in FIJI for colour histogram and grey value analysis. A calibration curve was plotted with the mean grey value as a function of CuOxy concentration, Fig. 7D. The visual changes in the detection zone as a function of added CuOxy concentration are shown in the Fig. 7D-inset. This confirms a strong correlation between mean gray value and CuOxy concentration, with a calculated LoD of 0.01 µM and LoQ of 0.033 µM.
The Bland–Altman plot was employed to evaluate the degree of agreement between two measurement methods. In this study, Bland–Altman analysis was applied to compare copper oxychloride concentrations determined by the spectrophotometric method with those obtained using two alternative approaches: a portable paper-based analytical device and a smartphone-based colorimetric method.
The results from the paper-based analytical device showed good agreement with the UV-Visible spectrophotometric reference method (Fig. S4A-SI). The mean difference (bias) between the two methods was −0.96 (pink line), indicating a slight underestimation of copper oxychloride concentration by the paper-based device relative to the reference method. Similarly, Bland–Altman analysis comparing the smartphone-based colorimetric method with the UV-Visible spectrophotometer (Fig. S4B-SI) revealed a mean bias of −0.291 (pink line), suggesting minimal systematic error between the test and reference methods.
In both Fig. S4A and S4AB-SI, the 95% limits of agreement (blue and green dotted lines) ranged from −9.69 to 7.72 and −8.97 to 8.39, respectively, indicating that the two alternative methods differed from the reference method by approximately ±8–9 concentration units across the measurement range. Overall, the Bland–Altman validation demonstrates that the developed paper-based analytical device and the smartphone-based colorimetric approach provide colorimetric detection performance comparable to that of the spectrophotometric method, supporting their suitability as reliable, low-cost alternatives for copper oxychloride quantification.
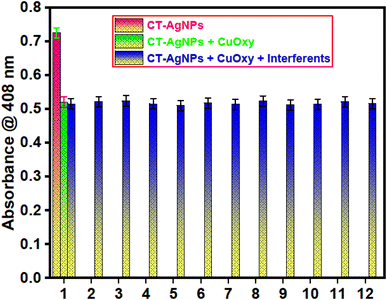
A key feature of any nanosensor is its ability to distinguish a target analyte even when other potentially interfering substances, such as similar pesticides or fungicides, are present. To assess the selectivity of the developed approach, a thorough series of experiments was conducted.64 In this context, equal amounts of similar concentrations (70 µM) of other fungicides and pesticides, including isoproturon, chlorothalonil, abamectin, propanil, trifluralin, urea, imidacloprid, DAP, cymoxanil, ammonium sulphate, emamectin, and deconil, were introduced alongside CuOxy. The absorbance response of the sensor showed only minor deviations compared to the standard CuOxy detection, indicating its high specificity in the presence of potential interferents, Fig. 8. The recoveries of CuOxy in the presence of interfering compounds remained in a range of 98.13 to 104.72%, Table S1-SI. Furthermore, to determine the optimal ratio between CT-AgNPs and CuOxy, a Job's plot was constructed, revealing a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometric interaction (Fig. S5-SI). This analysis describes stoichiometric behaviour but does not imply a specific coordination structure at the molecular level. It should be noted that the reported 1
1 stoichiometric interaction (Fig. S5-SI). This analysis describes stoichiometric behaviour but does not imply a specific coordination structure at the molecular level. It should be noted that the reported 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometric relationship reflects the experimentally optimized ratio that yielded the maximum colorimetric response under the present conditions. A more rigorous determination of binding constants and detailed molecular interaction mechanisms was beyond the scope of this study and will require further in-depth investigation using advanced physicochemical and spectroscopic approaches.
1 stoichiometric relationship reflects the experimentally optimized ratio that yielded the maximum colorimetric response under the present conditions. A more rigorous determination of binding constants and detailed molecular interaction mechanisms was beyond the scope of this study and will require further in-depth investigation using advanced physicochemical and spectroscopic approaches.
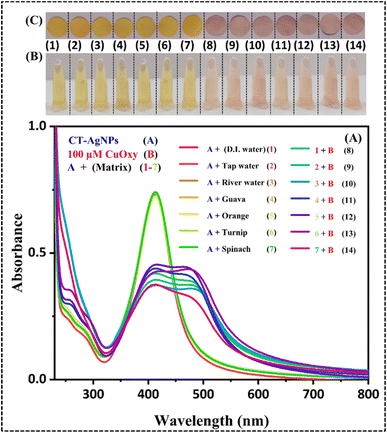
Initially, all experiments were conducted in DI water. To verify whether the CT-AgNPs-based CuOxy sensor is suitable for real-world applications, the same sensing method was applied to environmental water samples, including tap water (TW) and river water (RW), as well as food samples, such as guava, orange, turnip, and spinach (Fig. 9). The food samples show maximum absorption below 400 nm, thus not interfering with the UV-Vis peak of CT-AgNPs at 408 nm (Fig. S6-SI). Upon addition of CuOxy in different matrices making a final concentration of 100 µM and their addition to CT-AgNPs resulted in change of the typical yellow colour of CT-AgNPs to pink. This visible transformation was further confirmed by the characteristic hypochromic shift in the SPR band of CT-AgNPs, as shown in the UV-Vis spectra (Fig. 9A). The SPR band behaviour remained reasonably aligned with the standard calibration curve in all cases, validating the sensor's consistent performance in real samples, even in the presence of naturally occurring interferents.
In addition to the UV-Vis spectrohpotometric, smartphone-assisted solution-based and smartphone-assisted paper-based sensing platforms were also employed to detect CuOxy in the same real samples. The detection zone on the paper sensor and in the vial, after application of CuOxy-spiked water and food samples, showed a clear change in colour from yellow to pink in all matrices (Fig. 9B and C). These changes were captured using a smartphone, and the images were analysed using colourimetric tools to extract quantitative information. The results demonstrated excellent consistency with the standard analytical performance, with recoveries in a range of 93.41–113.66%, 94.97–108.13%, 95.94–112.85% for UV-Vis spectrophotometric, smartphone-assisted solution-based, and smartphone-assisted paper-based methods in the same order (Table S2-SI), thereby confirming the feasibility, reliability, and sensitivity of the CT-AgNPs-based UV-Vis, smartphone-based solution, and smartphone-based paper sensor for CuOxy detection in environmental and food samples. These results indicate that the nanoprobe is very specific for detecting CuOxy, and components in real water samples and food matrices do not interfere with the detection.
Moreover, the efficacy of the proposed CuOxy detection technique was also validated and benchmarked against a reported ultra-performance liquid chromatography (UPLC) method.17 The method was developed using DI water as sample solvent having concentrations of CuOxy in a range of 40–140 µM, Fig. S7-SI. The calibration curve of the CuOxy concentration as a function of the absorption peak area at 2.8 mL is depicted in Fig. S8-SI. Lastly, the spiked samples analyzed by the developed methods in this study were analyzed by the reported UPLC method vis-à-vis. Fig. S9A-SI depicts the elugrams of blank matrices at the optimized UPLC conditions, while Fig. S9B-SI depcts the spiked real samples with 100 µM CuOxy chloride. The absence of a peak at 2.8 mL in all blank smaples confirm that CuOxy was not used in their production. The same samples were spiked with 100 µM of CuOxy and analyzed by the UPLC method, as was done for the developed sensor methods in this study. The peak at 2.8 mL clearly shows the presence of CuoXY compared to blank samples. The recoveries of the CuOxy in real samples by this method remained in a range of 90.24–106.95%, Table S2-SI. The UPLC results validate the outcome of different approaches of the sensors developed in this study.
The comparative analysis of the current study with previously reported CuOxy sensors is presented in Table 1, which clearly highlights that the detection limit achieved by the current sensor is significantly lower than most existing methods.
| Method | LDR | LDR (nM)a | LoD | LoD (nM)a | Samples | Remarks | Ref. |
|---|---|---|---|---|---|---|---|
| a Converted to nanomolar for better comparison. | |||||||
| FIA | 0.001 to 10 µg L−1 | 4.68 × 10−3–46.8 | 2.62 × 10−4 µg L−1 | 1.23 × 10−3 | Peach, guava, banana, apple, and sapodilla | Expensive instrumentation, intricate preparation, and skilled operation | 17 |
| RP-HPLC-UV | 10–50 mg L−1 | 4.68 × 107–2.34 × 108 | 0.003 mg kg−1 | 14 | Coconut water | High-cost instrumentation, lengthy protocols | 19 |
| ETAAS | 0.19–20 µg L−1 | 0.88–9.3 × 10−8 | 3.8 ng | 1.78 ×104 | Olive tree | High-cost instrumentation, lengthy protocols, and requires trained operators | 21 |
| LCMS/AAS | 0.2 to 20 mg L−1 | 1.1 × 103–1.1 × 105 | 0.07 mg L−1 | 328 | Grapes | High-cost instrumentation, lengthy protocols, expensive standards, tedious sample preparation, and optimization problems | 22 |
| CT-AgNPs | 0.1–100 µM | 100–100000 | 0.007 0.009 0.01 | 7.0, 9.0, and 10 | Tap water, river water, guava, orange, spinach, and turnip | Economical, simple, no sample pre-treatment, highly selective and sensitive, enables on-site visual and quantitative water analysis via paper- and smartphone-based methods | This study |
4 Conclusions
In this study, CT-capped silver nanoparticles (CT-AgNPs) were synthesized and utilized as an efficient colourimetric sensor for the quick and sensitive detection of copper oxychloride (CuOxy). The developed sensor demonstrated a remarkably low limit of detection (LoD) of 0.007 µM and a limit of quantification (LoQ) of 0.023 µM, with a linear dynamic range (LDR) spanning from 0.1 to 100 µM under optimized conditions. Integration of the sensor with smartphone-based analysis enabled in-solution detection, and it was also embedded into paper-based analytical platforms for on-the-spot applications. The LoD and LoQ for smartphone in-solution analysis were 0.009 µM and 0.03 µM, respectively. Similarly, the LoD and LoQ of the paper-based assay were 0.01 µM and 0.03 µM. The CT-AgNPs sensor exhibited high selectivity for CuOxy even in the presence of various other pesticides and fungicides. To validate its real-world applicability, the sensor was tested in actual water samples and food samples, confirming its capability as a rapid, on-site, non-destructive, and reliable detection system for CuOxy in environmental settings.Conflicts of interest
Authors declare no conflicting financial interests.Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5ra08077k.
Acknowledgements
The authors received no specific financial support for research, authorship, or publication of this particular article.References
- D.-P. Häder, A. T. Banaszak, V. E. Villafañe, M. A. Narvarte, R. A. González and E. W. Helbling, Sci. Total Environ., 2020, 713, 136586 CrossRef PubMed.
- D. Ghosh, S. Chaudhary, S. Sarkar and P. Singh, Water Resour. Manage. Rural Dev, 2024, 15–28 Search PubMed.
- I. Ali, S. Khan, Z. Ali Shah, F. Ahmed, I. Shah, A. Hameed, R. Ullah and M. Raza Shah, Chem. Select., 2024, 9, e202304484 CAS.
- N. Morin-Crini, E. Lichtfouse, G. Liu, V. Balaram, A. R. L. Ribeiro, Z. Lu, F. Stock, E. Carmona, M. R. Teixeira and L. A. Picos-Corrales, Environ. Chem. Lett., 2022, 20, 2311–2338 CrossRef CAS.
- R. K. Mishra, S. S. Mentha, Y. Misra and N. Dwivedi, Water-Energy Nexus, 2023, 6, 74–95 Search PubMed.
- M. Syafrudin, R. A. Kristanti, A. Yuniarto, T. Hadibarata, J. Rhee, W. A. Al-Onazi, T. S. Algarni, A. H. Almarri and A. M. Al-Mohaimeed, Int. J. Environ. Res. Public Health., 2021, 18, 468 Search PubMed.
- C. Panis, L. Z. P. Candiotto, S. C. Gaboardi, S. Gurzenda, J. Cruz, M. Castro and B. Lemos, Environ. Int., 2022, 165, 107321 CrossRef CAS PubMed.
- Z. Zhang, X. Yan, K. C. Jones, C. Jiao, C. Sun, Y. Liu, Y. Zhu, Q. Zhang, L. Zhai and Z. Shen, npj Clean Water, 2022, 5, 59 CrossRef.
- R. Painuli, S. Raghav, P. C. Jha, M. Athar and D. Kumar, Inorg. Nano-Metal Chem., 2023, 53, 239–256 CrossRef CAS.
- P. Kudsk, L. N. Jørgensen and J. E. Ørum, Land Use Policy, 2018, 70, 384–393 CrossRef.
- C. Corvalan, S. Hales, A. J. McMichael and C. Butler, Ecosystems and Human Well-Being: Health Synthesis, World health organization, 2005 Search PubMed.
- L. Rani, K. Thapa, N. Kanojia, N. Sharma, S. Singh, A. S. Grewal, A. L. Srivastav and J. Kaushal, J. Clean. Prod., 2021, 283, 124657 CrossRef CAS.
- S. Kumari, K. S. Sharma, M. Nemiwal, S. Khan and D. Kumar, Int. J. Phytorem., 2022, 24, 808–821 Search PubMed.
- N. A. Rajpurohit, K. Bhakar, M. Nemiwal and D. Kumar, Arabian J. Geosci., 2022, 15, 1–16 Search PubMed.
- P. Marriage, W. Saidak and F. Von Stryk, Weed Res., 1975, 15, 373–379 CrossRef CAS.
- D. J. Lapworth, B. Lopez, V. Laabs, R. Kozel, R. Wolter, R. Ward, E. V. Amelin, T. Besien, J. Claessens and F. Delloye, Environ. Res. Lett., 2019, 14, 035004 CrossRef.
- K. U. N. Jaffar, K. A. U. Rehman, A. Muhammad, U. Sami, K. Naqeebullah, H. Abdul, A. Manzoor and S. Afsar, J. Liq. Chromatogr. Relat. Technol., 2025, 48, 37–47 CrossRef CAS.
- H. C. Menezes, B. P. Paulo, M. J. N. Paiva and Z. L. Cardeal, J. Anal. Methods Chem., 2016, 2016, 7058709 Search PubMed.
- R. Paranthaman and S. Kumaravel, J. Chromatogr. Sep. Tech., 2013, 4, 1000208 Search PubMed.
- H. Karasali, K. M. Kasiotis, K. Machera and A. Ambrus, J. Agric. Food Chem., 2014, 62, 11347–11352 CrossRef CAS PubMed.
- M. E. Soares, J. A. Pereira and M. L. Bastos, J. Agric. Food Chem., 2006, 54, 3923–3928 CrossRef CAS PubMed.
- Y. Verma, T. Bharat, K. Pratap, H. Sandip and Tp Shabeer, J. Environ. Sci. Health, Part B, 2022, 57, 670–679 CrossRef CAS PubMed.
- C. Yuan, L. Sun, Z. Wang, X. Hong, H. He, X. Lu and Z. Chen, Langmuir, 2025, 41, 11046–11055 CrossRef CAS PubMed.
- M. Yuan, X. Yuan, Y. Jin, Y. Shi, Z. Zou and X. Xiong, Microchem. J., 2025, 210, 112969 Search PubMed.
- J. Huang, H. Zhao, X. Chen, T. Lin, L. Hou and S. Zhao, Spectrochim. Acta, Part A, 2025, 333, 125894 Search PubMed.
- M. Mascini, S. Gaggiotti, F. Della Pelle, J. Wang, J. M. Pingarrón and D. Compagnone, Biosens. Bioelectron., 2019, 123, 124–130 CrossRef CAS PubMed.
- A. Masri, A. Anwar, D. Ahmed, R. B. Siddiqui, M. Raza Shah and N. A. Khan, Antibiotics, 2018, 7, 100 CrossRef CAS PubMed.
- S. Panicker, A. Prabhu, B. Sundarrajan, B. P. Quadros and N. K. Mani, Anal. Methods, 2024, 16, 6264–6270 RSC.
- S. Sarwar, D. A. Raja, D. Hussain, M. R. Shah and M. I. Malik, in Handbook of Nanomaterials, ed. M. I. Malik, D. Hussain, M. R. Shah and D.-S. Guo, Elsevier, vol. 1, 2024, pp. 1–26, DOI:10.1016/B978-0-323-95511-9.00012-3.
- X. Fu, H. Lin, J. Qi, F. Li, Y. Chen, B. Li and L. Chen, Sens. Actuators, B, 2022, 362, 131830 CrossRef CAS.
- J. Khatoon, M. R. Shah, M. I. Malik, I. A. T. Khan, S. Khurshid and R. Naz, RSC Adv., 2019, 9, 2978–2996 Search PubMed.
- N. ul Ain, Z. Aslam, M. Yousuf, W. A. Waseem, S. Bano, I. Anis, F. Ahmed, S. Faizi, M. I. Malik and M. R. Shah, New J. Chem., 2019, 43, 1972–1979 RSC.
- D. A. Raja, M. R. Shah and M. I. Malik, Anal. Chim. Acta, 2022, 1223, 340216 CrossRef CAS PubMed.
- D. A. Raja, S. G. Musharraf, M. R. Shah, A. Jabbar, M. I. Bhanger and M. I. Malik, J. Ind. Eng. Chem., 2020, 87, 180–186 CrossRef CAS.
- D. A. Raja, F. Munir, M. R. Shah, M. I. Bhanger and M. I. Malik, R. Soc. Open Sci., 2021, 8, 210185 CrossRef CAS PubMed.
- D. A. Raja, S. Rahim, M. R. Shah, M. I. Bhanger and M. I. Malik, J. Mol. Liq., 2023, 372, 121200 CrossRef CAS.
- A. Aijaz, D. A. Raja, F.-A. Khan, J. Barek and M. I. Malik, Chemosensors, 2023, 11, 91 Search PubMed.
- A. Talha, D. A. Raja, D. Hussain and M. I. Malik, Microchim. Acta, 2024, 191, 164 Search PubMed.
- D. A. Raja, A. Bano, M. Zehra, I. Vohra, S. Ahmed, A. Ahmed, M. I. Bhanger and M. I. Malik, Colloids Surf., A, 2024, 695, 134264 Search PubMed.
- I. Ali, D. A. Raja, F. Ahmed, M. Fazal, A. Hameed, M. R. Shah and M. I. Malik, J. Mol. Struct., 2024, 1315, 138971 Search PubMed.
- N. Iqbal, B. Amber, R. D. Asif, R. Ali, I. Rabia, A. Rafia, S. Imran, A. Ayaz, M. S. Ghulam and M. I. and Malik, Drug Dev. Ind. Pharm., 2024, 50, 789–800 Search PubMed.
- M. Z. Malik, D. A. Raja, D. Hussain and M. I. Malik, J. Environ. Chem. Eng., 2024, 12, 113626 Search PubMed.
- S. K. Sonawane, A. Ahmad and S. Chinnathambi, ACS Omega, 2019, 4, 12833–12840 Search PubMed.
- O. Bulut and M. D. Yilmaz, ACS Appl. Nano Mater., 2022, 5, 13761–13767 CrossRef CAS.
- M. I. Malik, B. Trathnigg and C. O. Kappe, J. Chromatogr., A, 2009, 1216, 1167–1173 CrossRef CAS PubMed.
- R. Abdul-Karim, A. Hameed and M. I. Malik, Eur. Polym. J., 2018, 105, 95–106 CrossRef CAS.
- I. Ali, S. Khan, C. A. T. Toloza, Z. A. Shah, Z. Iqbal, R. Ullah and M. R. Shah, J. Mol. Str., 2025, 1323, 140587 Search PubMed.
- T. Qadri, S. Khan, I. Begum, S. Ahmed, Z. A. Shah, I. Ali, F. Ahmed, M. Hussain, Z. Hussain, S. Rahim and M. R. Shah, J. Mol. Str., 2022, 1267, 133589 CrossRef CAS.
- S. Khan, I. Ali, A. Ali, C. A. T. Toloza, N. Ahmad and M. R. Shah, Opt. Mater., 2025, 164, 117088 Search PubMed.
- Y. Manmana, S. Kinugasa, Y. Hiruta and D. Citterio, Anal. Chem., 2025, 97, 1500–1506 CrossRef CAS PubMed.
- H. Wang, L. Dong, G. Ma, S. Qiu, G. Shan, L. Zhao, Y. Sun, A. Cui, R. Zhang and X. Liu, Anal. Chim. Acta, 2025, 1336, 343489 Search PubMed.
- D. Lin, B. Li, L. Fu, J. Qi, C. Xia, Y. Zhang, J. Chen, J. Choo and L. Chen, Microsyst. Nanoeng., 2022, 8, 53 CrossRef CAS PubMed.
- B. Chettri, R. S. Fernandes, S. Jha and N. Dey, Spectrochim. Acta, Part A Mol. Biomol. Spectrosc., 2024, 308, 123620 Search PubMed.
- A. S. Mohitkar, N. Dey and S. Jayanty, RSC Sustain., 2024, 2, 3946–3958 RSC.
- A. Pal and N. Dey, J. Fluoresc., 2025, 35, 1335–1344 Search PubMed.
- U. Salma, M. Z. Alam, S. Ahmad, M. Mohasin and S. A. Khan, Inorg. Chim. Acta, 2025, 582, 122600 CrossRef CAS.
- A. L. Mohamed, A. G. Hassabo, S. Shaarawy and A. Hebeish, Carbohydr. Polym., 2017, 178, 251–259 Search PubMed.
- B. M. Geetha, K. N. Brinda, G. Achar, J. G. Małecki, M. Alwarsamy, V. S. Betageri and S. Budagumpi, J. Mol. Liq., 2020, 301, 112352 Search PubMed.
- S. Rahim, S. Perveen, S. Ahmed, M. R. Shah and M. I. Malik, R. Soc. Open Sci., 2020, 7, 201097 CrossRef CAS PubMed.
- Y. Meng, Nanomaterials, 2015, 5, 1124–1135 CrossRef CAS PubMed.
- M. Sadeghi-Kiakhani, E. Hashemi, M.-M. Norouzi, P. Soleimani and V. Babaahmadi, J. Ind. Eng. Chem., 2025, 145, 732–744 Search PubMed.
- C. A. Z. Souto, F. G. de Souza, K. Pal, J. T. de Seixas Filho, H. K. Toma, N. Asthana, A. A. Khan and S. Fatima, J. Mol. Struct., 2025, 1321, 139961 CrossRef.
- C. Saenjum, N. Pattapong, T. Aunsakol, T. Pattananandecha, S. Apichai, H. Murakami, K. Grudpan and N. Teshima, J. Food Compos. Anal., 2022, 105, 104215 CrossRef CAS.
- S.-J. Kim, S.-J. Choi, J.-S. Jang, H.-J. Cho and I.-D. Kim, Acc. Chem. Res., 2017, 50, 1587–1596 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |