Open Access Article
Open Access ArticleEngineering the electronic structure towards enhanced performances of CO2 adsorption and conversion of a Ti-doped MoS2 monolayer: a DFT study
Junbo Wanga,
Qiankai Zhang b,
Qi Tanga,
Yin Zhanga,
Jun Zhou
b,
Qi Tanga,
Yin Zhanga,
Jun Zhou b,
Yang Wang*c and
Kai Wub
b,
Yang Wang*c and
Kai Wub
aFoshan Power Supply Bureau of Guangdong Power Grid, Foshan 528000, China
bInstitute of Science and Education Development, Xi'an Jiaotong University, 28 Xianning West Road, Xi'an 710049, China
cSchool of Electronics and Information, Xi'an Polytechnic University, 19 Jinhua South Road, Xi'an 710048, China. E-mail: wangyang@xpu.edu.cn
First published on 5th February 2026
Abstract
The active site of MoS2, predominantly located at the crystalline edge, limits selectivity for the CO2 reduction reaction (CO2RR). Single-Atom Catalysts (SACs), have emerged as a promising avenue to enhance the catalytic performance of MoS2, owing to their high catalytic selectivity on the basal plane and tunable activity in various chemical reactions. In this regard, transition metals from the 8B and 1B groups (Cr, Cu, Sc, Ti, V, and Ni) were investigated as dopants on the basal plane for the first time, employing first-principles calculations based on a 4 × 4 × 1 supercell of the MoS2 monolayer. The Ti/MoS2 catalyst was identified as the most stable among the SACs, attributed to its optimal formation energy. Various Ti-doped models were analyzed, encompassing energy band structure, density of states, charge differential density, Bader charge, and Gibbs free energy. Our findings indicate that Ti induces diminished electron binding, thereby weakening C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O with lower energy, consequently enhancing the availability of surface sites and facilitating catalytic reactions. In our investigation of possible reaction pathways, the preferred CO2RR pathway was identified as the reverse water gas conversion (RWGS), with the rate-limiting step being CO2 hydrogenation into carboxyl (*COOH). The Ti modification model on the MoS2 basal surface demonstrated exceptional catalytic performance, reducing the rate-limiting step to 0.177 eV, which is 17 times lower than that of pure MoS2. These calculational results provide valuable theoretical insights for designing highly efficient SACs on MoS2-based functional materials.
O with lower energy, consequently enhancing the availability of surface sites and facilitating catalytic reactions. In our investigation of possible reaction pathways, the preferred CO2RR pathway was identified as the reverse water gas conversion (RWGS), with the rate-limiting step being CO2 hydrogenation into carboxyl (*COOH). The Ti modification model on the MoS2 basal surface demonstrated exceptional catalytic performance, reducing the rate-limiting step to 0.177 eV, which is 17 times lower than that of pure MoS2. These calculational results provide valuable theoretical insights for designing highly efficient SACs on MoS2-based functional materials.
1 Introduction
The escalating emission of greenhouse gases (GHGs) has led to significant detrimental effects on Earth's climate systems, driving urgent demand for efficient CO2 capture and conversion technologies.1 This urgency has catalyzed the development of advanced carbon capture and utilization (CCU) technologies, among which the CO2 reduction reaction (CO2RR) stands out for its dual capacity to mitigate emissions and generate value-added carbon-based products (e.g., CH4, CH3OH).2 Central to CO2RR efficiency is the intricate interplay between CO2 adsorption energetics and intermediate desorption kinetics on catalyst surfaces—a dynamic process governed by the modulation of electronic structure and atomic-scale site specificity.3–5Two-dimensional (2D) transition metal dichalcogenides (TMDs), particularly molybdenum disulfide (MoS2), have emerged as versatile platforms for CO2RR due to their tunable band structures (1.2–1.9 eV), layer-dependent conductivity, and propensity for defect engineering.6–9 Unlike graphene's homogeneous π-electron system, the anisotropic d-orbital configuration of MoS2 enables selective activation of CO2's degenerate molecular orbitals.10 However, intrinsic limitations persist: the basal plane exhibits weak CO2 physisorption (Eads < 0.3 eV), while catalytically active edge sites constitute <5% surface coverage in conventional synthesis.11,12
To overcome the limitations of pristine MoS2 in CO2 reduction, chemical doping has emerged as an effective strategy to reconfigure its electronic structure and unlock new catalytic functionalities. Noble metals (e.g., Au, Pt, Pd, Ag) reduce the bandgap and facilitate charge transfer, while transition metals (TMs) such as Cu, Fe, and Ni introduce localized electronic states near the Fermi level, enabling direct interaction with CO2's antibonding orbitals and modulating the d-band center.13–15 These dopants not only enhance CO2 chemisorption but also reshape intermediate binding energies. For instance, Cu doping increases CO2 adsorption by up to 200%, attributed to synergistic surface area expansion and charge redistribution.16 Fe dopants, via Fe 3d–C 2p orbital hybridization, weaken the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond, achieving ΔEads ≈ −1.2 eV.17 Furthermore, Ni-doped MoS2 demonstrates altered spin polarization and charge transfer dynamics that influence catalytic selectivity.18 In situ spectroscopic studies reveal that even minor shifts in charge density (Δρ < 0.1 e− Å−3) at active sites can lead to over 50% changes in product selectivity, highlighting the catalytic sensitivity to local electronic environments.19 In addition, a recent systematic investigation demonstrated that substitutional doping at both Mo and S sites strongly modulates the work function of MoS2, with Ti and Nb identified as the most effective dopants for enhancing electronic responsiveness.20
O bond, achieving ΔEads ≈ −1.2 eV.17 Furthermore, Ni-doped MoS2 demonstrates altered spin polarization and charge transfer dynamics that influence catalytic selectivity.18 In situ spectroscopic studies reveal that even minor shifts in charge density (Δρ < 0.1 e− Å−3) at active sites can lead to over 50% changes in product selectivity, highlighting the catalytic sensitivity to local electronic environments.19 In addition, a recent systematic investigation demonstrated that substitutional doping at both Mo and S sites strongly modulates the work function of MoS2, with Ti and Nb identified as the most effective dopants for enhancing electronic responsiveness.20
Despite these advances, balancing CO2 adsorption with controlled CO desorption remains a persistent challenge that limits catalyst performance and product selectivity. While reduced energy barriers for CO2 activation are often desirable, excessively facile *CO desorption can hinder further reduction steps, resulting in accumulation of CO as a by-product. Lv et al.21 showed that doped MoS2 structures with enhanced surface charge densities lower reaction barriers, yet compromise *CO retention required for deeper reduction. Similarly, vertical Nb-doping reported by Abbasi et al. promotes CO desorption but restricts C–C coupling pathways.22 Conversely, strategies aimed at increasing *CO desorption barriers—such as transition metal doping designed by Li et al.—successfully suppress CO evolution, enabling longer cascade reactions.23 These findings underscore the importance of fine-tuning *CO binding energies to balance catalytic activity and selectivity. However, most existing studies focus on isolated descriptors (e.g., adsorption energy or conversion), without a holistic view of how dopant characteristics simultaneously influence both CO2 capture and conversion steps.
To address this critical gap, we employ first-principles density functional theory (DFT) to systematically evaluate a set of transition metal dopants (Cr, Cu, Sc, Ti, V, Ni) in MoS2, targeting dual-functionality in CO2 reduction. Among these, Ti emerges as the most promising candidate, offering the lowest formation energy and a favorable electronic environment for CO2 activation. We conduct a comprehensive comparison of three distinct Ti doping configurations—sulfur substitution, molybdenum substitution, and interlayer intercalation24,25—revealing clear structure–function relationships among dopant positioning, Bader charge transfer, and activation barriers. Furthermore, our detailed reaction pathway analysis elucidates the CO2-to-CO conversion mechanism on Ti/MoS2, identifying rate-limiting steps and quantifying *CO desorption energetics. Our study offers a unified design framework that links dopant physicochemical properties with catalytic performance, providing critical insights for the development of next-generation MoS2-based CO2RR catalysts with optimized capture–conversion synergy.
2 Computional approach
In order to ensure the simulation was sufficiently large that an adsorbed gas molecule was not affected by nearby gas molecules, a periodic (4 × 4 × 1) supercell of the MoS2 monolayer was modelled. The MoS2 monolayer was composed of 16 Mo and 32 S atoms.26 It should be noted that to avoid interlayer interactions, a vacuum layer of 15 Å was constructed in the direction from the interface to the isolated slab boundary condition.We conducted all calculations using CASTEP on the DFT theory plane-wave pseudopotential method for exploring the electronic and structural properties.27 Electronic interaction energies were computed employing the generalized gradient approximation (GGA) functional and the Perdew–Burke–Ernzerhof (PBE) functional.28 Here, the cutoff energy was set to be 500 eV, and the k-point grids used were 6 × 6 × 1.29 We set the convergence criteria for the optimization of energy, remaining stress, and force to be 0.01 meV per atom, 0.05 GPa, and 0.01 eV Å−1, respectively.30 The long-range dispersion correction (DFT-D2) method, proposed by Grimme, was selected to modify the effects of the van der Waals interactions in all the constructed models.31,32 The total energy was converged to <10 meV. All calculations were performed using the same relaxation criteria.
The validity of the calculation can be established by comparing the formation energy of the M-doping model. The formation energy (Ef) served as a metric to evaluate the feasibility of dopant integration and the stability of the doped system within the lattice, as outlined in eqn (1):33
 | (1) |
In terms of adsorption energy, the greater the absolute value of adsorption energy on the surface, the better the adsorption performance of CO2. The formula is as follows:
| Eads = Egas+surface − Egas − Esurface | (2) |
3 Results and discussion
3.1 Properties of Ti-doped MoS2 monolayer
To identify suitable dopants for MoS2 in CO2 hydrogenation, we focused on 3d transition metals (Cr, Cu, Sc, Ti, V, and Ni) due to their propensity to modulate the electronic structure of the host catalyst. The practical feasibility of doping hinges critically on the thermodynamic stability of the doped structure. Therefore, we first computed the formation energy (Ef) for each candidate, as presented in Table 1. Among them, Ti doping exhibits the most negative formation energy, indicating its most stable incorporation into the MoS2 lattice.| Dopant | Formation energy (eV) | Dopant | Formation energy (eV) |
|---|---|---|---|
| Cr | −3.45 | Ti | −3.93 |
| Cu | −0.29 | V | −3.88 |
| Sc | −2.54 | Ni | −2.27 |
Although V and Cr also show reasonably low formation energies, the difference from Ti remains sufficient to suggest that Ti is the most thermodynamically favorable dopant under comparable conditions. For this reason—and to allow a focused mechanistic analysis—we selected Ti as the representative dopant for detailed electronic, adsorption, and reaction-pathway investigations. This computational selection aligns with and provides a theoretical foundation for experimental observations of Ti's enhanced catalytic performance.34–36 The comparative study of other dopants with similar stability (e.g., V, Cr) is considered an interesting direction for future work. Fig. 1(a) depicts the relaxed structure of MoS2 obtained in this study, while Fig. 1(b–d) illustrates the Ti-doped and decorated structures. Overall, the influence of doping and decorating on the MoS2 structure was observed to be minimal. Doping agents remain centered between neighboring atoms, and the positions of Mo and S atoms within the structure exhibit minor changes. The calculated Mo–S bond length and Mo–Mo distance in pristine MoS2 were determined to be 2.400 Å and 3.167 Å, respectively, and were consistent with theoretical data (2.420 Å and 3.190 Å).37,38 Additionally, the angle of the S–Mo–S bonds in the optimized pure MoS2 was calculated to be 82.548°, showing reasonable agreement with theoretical and experimental data.39–41 In this regard, the calculation method and parameters used for modeling MoS2 were appropriate. Table 2 reveals that the Ef values for all Ti-doped MoS2 surfaces are negative, indicating an exothermic process. We considered all potential sites on the basal plane. In this context, 1Ti-MoS2 denotes MoS2 with a Ti atom substituting for a Mo atom, 2Ti-MoS2 represents MoS2 with a Ti atom replacing an S atom, and 3Ti-MoS2 signifies MoS2 with a single Ti atom modifying the surface. Among them, given the comparable ionic radii of Ti4+ and Mo6+, the incorporation of Ti dopant into the lattice induced subtle deviations in the lattice parameters; nevertheless, the observed alterations were deemed negligible in magnitude. In contrast, 2Ti-MoS2 and 3Ti-MoS2 are challenging to synthesize compared to 1Ti-MoS2, owing to differences in ionic radius and doping site. However, overall, they still provide indications of thermodynamic stability.
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O is the angle between the Ti atom and its two nearest neighbors)
O is the angle between the Ti atom and its two nearest neighbors)
| Structure | Ef, eV | ΔTi−X, Å | θX=Ti=X, ° |
|---|---|---|---|
| MoS2 | * | * | * |
| 1Ti-MoS2 | −9.15 | 2.415 | 83.718 |
| 2Ti-MoS2 | −3.93 | 2.611 | 73.609 |
| 3Ti-MoS2 | −3.81 | 2.319 | 89.510 |
It is important to clarify the purpose of studying these less-favorable configurations: they are not presented as the most readily synthesizable structures, but as critical models to elucidate the structure–property relationship. The comparative analysis of 1Ti, 2Ti, and 3Ti-MoS2 allows us to isolate the effect of Ti coordination environment—from fully coordinated lattice substitution to under-coordinated surface sites—on the electronic structure and catalytic activity.
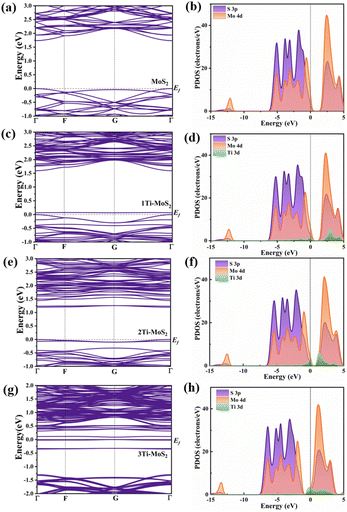
To unveil the electronic structure changes in doped MoS2 on the basal plane, DFT calculations were conducted for pure MoS2 and three distinct doping systems at various sites. Fig. 2 shows the band structure and partial density of states (PDOS) of all the models. The results indicate that the band gap of pure MoS2 is 1.735 eV, consistent and reliable compared to previous calculations.42
Additionally, Fig. 2(b) illustrates that the Mo 4d and S 3p states constitute the conduction and valence bands of pure MoS2, respectively. In the case of 1Ti-MoS2, the Ti 3d state exhibits a slight peak change in the band gap, as depicted in Fig. 2(c and d). Despite Ti replacing Mo, the system undergoes insignificant changes due to the similar atomic radius and the same number of valence electrons. The Mo 4d state and Ti 3d state exhibit a consistent trend above the Fermi level and effectively overlap with the S 3p state, demonstrating a stable bonding effect. In comparison to 1Ti-MoS2, the Ti state effect in 2Ti-MoS2 is more pronounced. Due to the substitution at the S-site, Ti introduces additional electrons in the model, represented as the donor level.
As depicted in Fig. 2(e), noticeable isolated energy levels appear. Fig. 2(f) corresponds to the contribution of the Ti 3d state, where influences the bottom of the conduction band, concurrently reducing the overall band gap to 1.196 eV, a value similar to the other theoretically calculated one.43 In contrast to the empty band in 1Ti-MoS2, these filled electrons can more effectively facilitate electron participation in surface CO2 adsorption and catalytic conversion. Among these, the peak coincidence between Ti 3d and the other two states in PDOS is reduced, suggesting that alternative sites in 2Ti-MoS2 may weaken the bonding ability with surrounding atoms, potentially leading to lattice distortion in the model. Besides, from Fig. 2(g and h), Ti doping on the surface significantly influences the energy band, introducing multiple impurity levels within the band gap. This facilitates easier electron transition to surface reactions. The PDOS diagram also illustrates that doping causes a leftward shift in the overall band edge, with Ti 3d states comprising the majority of the impurity levels.24 In contrast to the previous calculation model, the Ti 3d state in PDOS and its binding ability with the surrounding S atoms is not as strong as in 1Ti-MoS2.
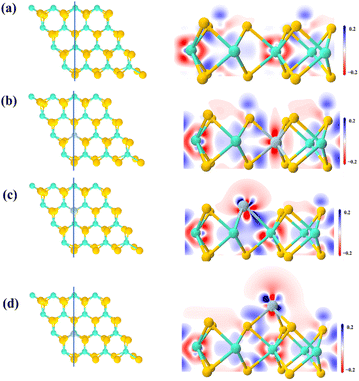
Combining the band structure and PDOS analysis, it can be concluded that the introduction of impurity energy levels into the model through Ti doping facilitates the migration of carriers and improves catalytic activity. Moreover, with the different doping sites, the electron density differences and Bader charge were calculated to explore the influence of doping on charge transfer and interaction. The difference in electron density between pure MoS2, 1Ti-MoS2, 2Ti-MoS2 and 3Ti-MoS2 is represented in Fig. 3. From Fig. 3(a), it can be clearly seen that Mo and S gain and lose electrons in the model. In line with the electronic structure results in Fig. 2(c and d), there is no noticeable change in the gain and loss of electrons after Ti substitutes for Mo atoms, and the electrons remain relatively stable. Similarly, in the 2Ti-MoS2 model, the relatively large atomic radius of Ti and the additional electron contribution result in a slightly distorted optimized model, leading to the appearance of impurity levels. The substitution of Ti at the S site introduces extra electrons on the surface, enhancing their involvement in reactions. This phenomenon is likely to be advantageous for CO2 adsorption and subsequent catalytic conversion reactions.
In the 3Ti-MoS2 model, the charge density difference reveals substantial electron accumulation around the Ti sites, indicating that Ti introduces additional surface-active electrons capable of strengthening CO2 adsorption and facilitating its subsequent activation. Meanwhile, the structural deformation after Ti incorporation remains minimal, allowing the reaction to occur on a geometrically stable surface. Combined with the reduced band gap and enhanced charge redistribution observed in the 2Ti-MoS2 and 3Ti-MoS2 models, these features collectively contribute to more efficient catalytic behavior. Notably, these trends are consistent with the recent systematic doping study,20 which also identified Ti as one of the most effective dopants for tuning the electronic structure and reactivity of MoS2.
In this regard, we meticulously calculated the Bader net atomic charge of each atom in all structures in order to determine an atom's charge state quantitatively by comparing its calculated charge with its own valence charge. Table 3 outlines the range of variation in Bader charge for different doping configurations. Within pure MoS2, the S atom exhibits an acquired electron state, while the Mo atom experiences electron loss. In 1Ti-MoS2, some S atoms gain additional electrons, indicating a relative equilibrium between Ti doping and surrounding S electrons at the Mo site. Contrastingly, in 2Ti-MoS2, the Ti contribution to the electron decreases compared to 1Ti-MoS2, resulting in an increase in electron donation from Mo. This phenomenon correlates with the observed change in Ti 3d states depicted in Fig. 2(f), where the overlap between Ti 3d and other energy levels near the Fermi level diminishes, thereby weakening electron transfer. In 3Ti-MoS2, the Ti atom loses 0.96e of electrons. However, unlike 1Ti-MoS2, electron gains and losses predominantly occur at the surface rather than achieving charge equilibrium with surrounding S atoms. Consequently, Mo atoms, which originally lost electrons, now gain electrons within the system. This phenomenon likely activates the surface active region, thereby facilitating the adsorption and conversion of gas molecules.
| Atom type | Bader net atomic charge | |||
|---|---|---|---|---|
| MoS2 | 1Ti-MoS2 | 2Ti-MoS2 | 3Ti-MoS2 | |
| S | (−0.30) – (−0.35) | (−0.23) – (−0.54) | (−0.45) – (−0.68) | (−0.03) – (−1.00) |
| Mo | (0.65) – (0.66) | (0.51) – (1.10) | (0.75) – (1.12) | (−0.84) – (1.05) |
| Ti | * | 1.00 | 0.62 | 0.96 |
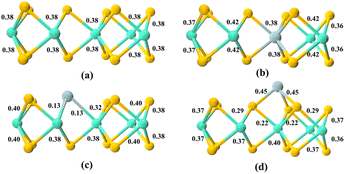
Besides, the bonding between different atoms inside MoS2 before and after doping was investigated using Mulliken bond population calculations.44 These calculations further quantify the extent of electron sharing between two atoms, providing deeper insights into the bonding characteristics.45 It should be noted, however, that negative values for Mulliken bond populations are possible, indicating that the bond type is ionic and relatively easy to break if the absolute value is low.46 Although Mulliken bond populations do not have a precise threshold value for distinguishing between ionic and covalent bonds, a bond with a Mulliken bond population greater than 0.1 is typically considered covalent, according to the literature.47–49 Fig. 4 shows the calculated Mulliken bond populations for all models. Through calculations, it is observed that Mo and S exhibit distinct covalent bond characteristics. Similarly, in 1Ti-MoS2 and 3Ti-MoS2, Ti and S also form covalent bonds. However, the covalent bond characteristics in 2Ti-MoS2 are significantly weakened. This result aligns with the lower overlap of Ti and Mo orbitals in the PDOS.
3.2 CO2 adsorption on the Ti-doped MoS2 monolayer
In order to analyze the optimal adsorption site of CO2 on the surface, the geometric structure of the CO2 was first optimized to its most stable configuration. The calculated C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond length was 1.172 Å, and the O
O bond length was 1.172 Å, and the O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond angle was found to be 180°, which is in good agreement with the experimental data and previously derived theoretical values.50
O bond angle was found to be 180°, which is in good agreement with the experimental data and previously derived theoretical values.50
Then the adsorption energy of CO2 at different sites on the surface after optimization was calculated. The molecular height is defined as the minimum distance from any atom in the CO2 molecule from any atom in the MoS2. Four potential locations for CO2 adsorption on the MoS2 surface were identified: above a Mo atom (Mo site), above an S atom (S site), above a Ti atom (Ti site), above the center of a hexagon composed of three Mo atoms and three S atoms (Hollow site), and above a Mo–S bond (Bridge site). With a view to establish which of these are the most stable configuration for the CO2 adsorption, two adsorption orientations (horizontal-1 and vertical-2) of the CO2 molecule were also considered. Fig. 5 illustrates the CO2 adsorption energy for all potential adsorption sites. Certain adsorption sites, not depicted in the figure, result from CO2 relocating to other best adsorption sites during calculations. For simplicity, we only present the optimal adsorption sites.
Analyzing changes in adsorption energy at different sites reveals that pure MoS2 exhibits the maximum adsorption energy of −0.011 eV for horizontal placed CO2 at the bridge site. Meanwhile, MOS2 and 1Ti-MOS2, lacking effective Ti sites on the surface, exhibit minimal changes in adsorption energy. In some cases, the CO2 adsorption energy at certain sites is positive, significantly impeding the adsorption process and hindering the catalytic conversion of CO2. Furthermore, the adsorption energy of 2Ti-MoS2 and 3Ti-MoS2 increases at multiple sites, with vertically placed CO2 displaying higher adsorption energy, indicating that the activation of oxygen atoms may be the key to CO2 adsorption.
Fig. 6 depicts the optimal structure of the four models with the best adsorption energy. In Fig. 6(a), the relaxed structure of a CO2 molecule adsorbed on pristine MoS2 is shown. The calculated distance between CO2 and MoS2 is 3.544 Å. The length of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond in the adsorbed CO2 measures 1.172 Å, similar to the C
O bond in the adsorbed CO2 measures 1.172 Å, similar to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond length in free CO2, indicating that MoS2 does not significantly affect the adsorbed CO2.
O bond length in free CO2, indicating that MoS2 does not significantly affect the adsorbed CO2.
This suggests a weak interaction between the CO2 and MoS2. Fig. 6(b–d) clearly shows a gradual decrease in the physical adsorption distance of CO2, with the minimum adsorption distance between 3Ti-MoS2 and CO2 measuring 1.878 Å. This aligns with our predictions in Fig. 2 and 3, showing that 2Ti-MoS2 and 3Ti-MoS2 exhibit stronger adsorption capacity due to the additional electron cloud.
By further calculating adsorption energy, total charge, and the absolute values of the change in bond length/angle, we identified the adsorption changes of CO2 on the surface, as presented in Table 4. Table 4 reveals that the Eads value for CO2 on pristine MoS2 is 0.04 eV, consistent with the theoretically calculated value,12 further indicating the limited CO2 adsorption capacity of MoS2. Additionally, the adsorption energy of 3Ti-MoS2 is as high as −1.251 eV, suggesting a potential strong chemisorption reaction on the surface. The bond length of CO2 in MoS2 and 1Ti-MoS2 remains unchanged, indicating ineffective activation of the surface C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O in the system. The absolute values of the change in the length of the C
O in the system. The absolute values of the change in the length of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond in CO2 for 2Ti-MoS2 exhibit a change of 0.013 Å, while the change for 3Ti-MoS2 is twice that amount. Besides, similar conclusions are observed in the change of bond angle. The extra electrons and surplus active sites provided by Ti on the surface lead to significant distortion of C
O bond in CO2 for 2Ti-MoS2 exhibit a change of 0.013 Å, while the change for 3Ti-MoS2 is twice that amount. Besides, similar conclusions are observed in the change of bond angle. The extra electrons and surplus active sites provided by Ti on the surface lead to significant distortion of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O in 3Ti-MoS2 (ΔθO
O in 3Ti-MoS2 (ΔθO![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O=49.770°), enhancing surface catalytic performance. Considering the changes in C
O=49.770°), enhancing surface catalytic performance. Considering the changes in C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond length and bond angle, both 2Ti-MoS2 and 3Ti-MoS2 demonstrate remarkable catalytic performance, with the activation performance of 3Ti-MoS2 on CO2 being more than 2 times higher than that of 2Ti-MoS2.
O bond length and bond angle, both 2Ti-MoS2 and 3Ti-MoS2 demonstrate remarkable catalytic performance, with the activation performance of 3Ti-MoS2 on CO2 being more than 2 times higher than that of 2Ti-MoS2.
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond in CO2, respectively. θO
O bond in CO2, respectively. θO![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O is the O
O is the O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond angle. ΔθO
O bond angle. ΔθO![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O is the absolute value of the change in the O
O is the absolute value of the change in the O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O angle from when the molecule is free)
O angle from when the molecule is free)
| Structure | Eads, eV | Δd1C–O, Å | Δd2C–O, Å | θO![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, ° O, ° |
ΔθO![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, ° O, ° |
|---|---|---|---|---|---|
| MoS2 | −0.011 | 0 | 0 | 179.971 | 0.029 |
| 1Ti-MoS2 | −0.016 | 0 | 0 | 179.535 | 0.465 |
| 2Ti-MoS2 | −0.500 | 0.013 | 0.013 | 176.228 | 3.772 |
| 3Ti-MoS2 | −1.251 | 0.029 | 0.199 | 130.230 | 49.770 |
In order to further investigate the electron effect of different doped configuration on CO2 adsorption, we also calculate the energy band and density state of all models. As shown in Fig. 7, comparing the structure of unadsorbed CO2 model, it is found that the energy band and PDOS of pure MOS2/CO2 and 1Ti-MOS2/CO2 do not change significantly. The PDOS before and after CO2 adsorption largely overlap, particularly in the region near the Fermi level. Consequently, the interaction between CO2 and 1Ti-MoS2 resembles the interaction between CO2 and the pristine MoS2.
Thus, we can infer that the interaction between CO2 molecules and 1Ti-MoS2 monolayers is weak, consistent with the calculated adsorption energy. The bandgaps of 2Ti-MOS2/CO2 and 3Ti-MOS2/CO2 undergo slight variations within a certain range after CO2 adsorption, but the electronic transition types remain essentially unchanged.
Besides, upon comparing the PDOS of different models in Fig. 7, it is evident that as the Ti doping site approaches the surface, the overall PDOS energy gradually shifts to the left. This characteristic is consistent with the unadsorbed structure.
Moreover, detailed comparisons of the changes in CO2 1s state, CO2 2p state, and Ti 3d state are depicted in Fig. 8. It is more evident from Fig. 8(a) that the orbitals of CO2 and Ti do not significantly overlap, and the doping of Ti at the Mo site also does not affect the adsorption effect of CO2. In Fig. 8(b), partial energy overlap is observed in the region above the 2–3 eV, and a faint overlap near −6 eV. These overlaps in state density confirm the increased adsorption energy of CO2 on 2Ti-MoS2. Simultaneously, Fig. 8(c) reveals a substantial decrease in the overall density of states compared to the previous model. This reduction in the electron state density of CO2 suggests that the original C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O electron binding is influenced by Ti-doping. The diminished electron binding can weaken the double bond energy with lower energy, thereby increasing the availability of surface sites and facilitating the catalytic reaction. Furthermore, the overlapping range of the Ti 3d state and CO2 electronic state near the Fermi level increases. This undoubtedly enhances the binding performance of Ti and CO2, aligning with the calculated maximum adsorption energy.
O electron binding is influenced by Ti-doping. The diminished electron binding can weaken the double bond energy with lower energy, thereby increasing the availability of surface sites and facilitating the catalytic reaction. Furthermore, the overlapping range of the Ti 3d state and CO2 electronic state near the Fermi level increases. This undoubtedly enhances the binding performance of Ti and CO2, aligning with the calculated maximum adsorption energy.
 | ||
| Fig. 8 The PDOS curves of (a) 1Ti-MoS2; (b) 2Ti-MoS2; (c) 3Ti-MoS2. The dash lines are Fermi level. The energy is referenced to the Fermi level (Ef), which is indicated by the dashed line at 0 eV. | ||
In view of the above results, we also used electron differential charge density and Bader charge calculation to verify the charge transfer between CO2 and the catalyst, as shown in Fig. 9 and Table 5. Table 5 specifically presents the Bader net atomic charge of CO2 and its closest atom. Where Snear represents the S atom closest to CO2. Consistent with the previously calculated total charge results, the Bader net atomic charge sum of C and O in the remaining structures, except for the 3Ti-MoS2 structure, remained relatively stable. This is also evident in Fig. 9, where only the CO2 molecules in 2Ti-MoS2 exhibit a slight change in adsorption among the first three catalysts. This, coupled with the electron density difference, substantiates the fact that its adsorption energy was heightened, leading to improved catalytic performance.
| Atom type | Bader net atomic charge | |||
|---|---|---|---|---|
| MoS2 | 1Ti-MoS2 | 2Ti-MoS2 | 3Ti-MoS2 | |
| Snear | −0.50 | −0.38 | — | — |
| Ti | — | 1.02 | 0.79 | 1.12 |
| C | 1.45 | 1.89 | 1.82 | 0.69 |
| O1 | −0.71 | −0.96 | −0.92 | −0.67 |
| O2 | −0.78 | −0.96 | −0.94 | −0.90 |
Despite Ti losing 1.02e, the gain and loss of these electrons remain stably coordinated with the surrounding S, considering its doping site is at the Mo site. As mentioned earlier, the optimal adsorption configuration of 2Ti-MoS2 involves the vertical adsorption of Ti and O2. Table 5 reveals that Ti loses 0.79e electrons, while O2 gains a slightly larger number of electrons. This enhances the adsorption energy and involves a small amount of charge transfer. Furthermore, the Bader net atomic charge calculation for 3Ti-MoS2 revealed that Ti lost more electrons, and C transferred fewer electrons to O. These findings confirm that Ti doping at this site significantly activates C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, reducing energy in the adsorption process. In alignment with Fig. 9(d), an electron cloud structure formed between Ti and CO2.
O, reducing energy in the adsorption process. In alignment with Fig. 9(d), an electron cloud structure formed between Ti and CO2.
In addition, Mulliken bond populations of the adsorption model are also calculated, as shown in the Fig. 10. According to the calculation results, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O presents a typical covalent double bond, and the calculated result is the same as the charge transfer observed in the MoS2 and 1Ti-MoS2 models, indicating that the bond type does not change. In the 2Ti-MoS2 model, Ti and Mo tend to form ionic bonds, whereas in the 3Ti-MoS2 model, both C and O atoms form covalent bonds with surface Ti atoms. These findings align well with the charge analysis results and suggest that Ti incorporation significantly enhances surface reactivity toward CO2 adsorption. Such dopant-induced electronic reconstruction effectively activates the otherwise inert basal plane of MoS2, providing a general strategy for designing 2D catalysts with tunable intrinsic activity beyond conventional edge or defect engineering.
O presents a typical covalent double bond, and the calculated result is the same as the charge transfer observed in the MoS2 and 1Ti-MoS2 models, indicating that the bond type does not change. In the 2Ti-MoS2 model, Ti and Mo tend to form ionic bonds, whereas in the 3Ti-MoS2 model, both C and O atoms form covalent bonds with surface Ti atoms. These findings align well with the charge analysis results and suggest that Ti incorporation significantly enhances surface reactivity toward CO2 adsorption. Such dopant-induced electronic reconstruction effectively activates the otherwise inert basal plane of MoS2, providing a general strategy for designing 2D catalysts with tunable intrinsic activity beyond conventional edge or defect engineering.
 | ||
| Fig. 10 The Mulliken bond populations for the: (a) MoS2/CO2; (b) 1Ti-MoS2/CO2; (c) 2Ti-MoS2/CO2; (d) 3Ti-MoS2/CO2. | ||
3.3 The reaction mechanism of CO2RR
Considering the importance of CO in CO2RR over MoS2, as observed in other experimental results,25 we conducted further studies on the hydrogenation of intermediates *COOH and *CO to elucidate the underlying mechanisms. In order to comprehend the reaction mechanisms for the reduction of CO2RR over MoS2 and Ti/MoS2, Fig. 9 illustrates the potential reaction pathways of CO2RR based on previous DFT calculations.22 Additionally, for the optimal reaction pathway, the following steps take place:| CO2 + * → *CO2 | (3) |
| *CO2 + H* → *COOH | (4) |
| *COOH + H* → *CO + H2O | (5) |
Reaction step I involves the adsorption of CO2, activating the CO2 molecule. Step II encompasses the hydrogenation process of *CO2, and the proposed reaction step III involves the hydrogenation of carboxyl (*COOH), leading to the reaction CO2 → CO. As depicted in Fig. 9, the chemisorption of CO2 on the surface lays the groundwork for subsequent reactions. Particularly evident in Fig. 11(b), the transition of CO2 from the surface to the chemisorbed state reveals that the free energy barrier is lowest for 3Ti-MoS2. This finding suggests that 3Ti-MoS2 facilitates further adsorption reactions. Following CO2 chemisorption, *CO2 can undergo hydrogenation to form *COOH. However, the hydrogenation of *CO2 to *COOH on pristine MoS2 is found to be kinetically unfavorable, attributed to the high barrier energy of 3.046 eV, as shown in Fig. 11(b).
 | ||
| Fig. 11 The reaction mechanism for CO2RR into CO on 3Ti-MoS2. (a) The structures of the reaction intermediates; (b) the energy profile for different catalytic. | ||
Our analysis of the energy profiles indicates that the *CO2 → *COOH hydrogenation step represents the rate-determining step (RDS) for pristine MoS2, 1Ti-MoS2, and 2Ti-MoS2 due to its high activation barrier. In contrast, the most active 3Ti-MoS2 exhibits a markedly reduced barrier of only 0.177 eV for this step, effectively eliminating this kinetic limitation. As a result, the CO desorption step becomes the new RDS, with a relative barrier of 1.017 eV. This shift of the RDS clearly reflects a fundamental change in catalytic behavior induced by surface-exposed Ti dopants, highlighting the superior CO2-to-CO conversion capability of 3Ti-MoS2.
The introduction of the Ti coordination site on the basal plane significantly reduces the Gibbs free energy of the *COOH intermediate. It can be obviously found that Ti can effectively reduce the energy of the determination step under three different doping modes.
Compared with the electronic structure of Ti/MoS2 as discussed before, the Ti doping on MoS2 changes the band gap and charge, which favors strong adsorption of CO2, resulting in superior catalytic CO2RR performance toward CO. Notably, 3Ti-MoS2 exhibits exceptional catalytic performance with its *CO2 to *COOH energy reduced to 0.177 eV, 17 times lower than that of pure MoS2. Ultimately, the introduction of Ti enhances the desorption of CO, as evidenced by the calculated energy barrier for the CO desorption step on the surface. Both pure MoS2 and 1Ti-MoS2 surfaces exhibit intensified CO desorption processes, whereas 2Ti-MoS2 and 3Ti-MoS2 demonstrate notable energy barriers for the reaction. Notably, the energy barrier for CO desorption on the 3Ti-MoS2 surface is particularly significant, reaching 1.017 eV. This suggests that the presence of *CO on this surface is conducive to further conversion processes. These theoretical trends are consistent with experimental observations20,21,51 that MoS2-based catalysts primarily yield CO as the dominant product in CO2 reduction reactions, where moderate CO binding and reduced COOH activation barriers favor CO evolution.
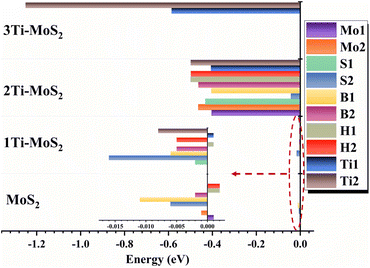
Based on the energy calculations described above, a comparison is made among the formation energy, adsorption energy, adsorption barrier, determination step, and desorption barrier of various models, as illustrated in Fig. 12. It is worth noting that the formation energy of pure MoS2 is merely used as a reference value of 0 eV and holds no practical significance in this context.
Besides, the outstanding performance of the 3Ti-MoS2 model pinpoints the surface-exposed Ti as the most potent active site design for CO2RR, providing a valuable guiding principle for future catalyst development. The systematic theoretical analysis of the different doping sites offers valuable insight into future research on the TMD material, particularly in terms of assessing their overall CO2RR performance. These results highlight a clear structure–property relationship between dopant configuration, charge redistribution, and catalytic activity, providing general design principles for developing efficient MoS2-based single-atom catalysts that feature near-Fermi impurity levels and moderate charge transfer to optimize both adsorption and conversion steps.
Looking forward, the insights from this study offer clear pathways for experimental realization and application. The predicted thermodynamic stability of Ti in the MoS2 lattice guides its synthesis via controlled hydrothermal or chemical vapor deposition methods.52 Furthermore, the exceptionally low reaction barrier and high selectivity predicted for CO production position Ti/MoS2 as a highly promising catalyst for CO2 electrolyzers, motivating future work on fabricating and testing gas diffusion electrodes for high-current-density CO2-to-CO conversion.
4 Conclusions
In conclusion, first principles quantum theory was employed to computationally screen M1/MoS2 (M1 = Cr, Cu, Sc, Ti, V, and Ni) with the first series of transition metals. Ti/MoS2 exerted a significant impact on the energy band structure, inducing changes in the band gap and electronic transition energy through the introduction of impurity energy levels. Notably, the catalytic advantages of 3Ti-MoS2 in CO2 conversion were demonstrated through PDOS and electron density difference analysis. 3Ti-MoS2 emerged as a potential material for CO2 sensing due to its favorable adsorption and response characteristics. Furthermore, by calculating the reaction mechanism for CO2RR into CO, we observed exceptional catalytic performance in 3Ti-MoS2, with its *CO2 to *COOH energy reduced to 0.177 eV, 17 times lower than that of pure MoS2. These results highlight a clear structure–property relationship between dopant configuration, charge redistribution, and catalytic activity. The insights obtained here provide general design principles for developing efficient MoS2-based single-atom catalysts, emphasizing dopants that introduce near-Fermi impurity levels and moderate charge transfer to optimize both adsorption and conversion steps.Author contributions
Junbo Wang: conceptualization, investigation, methodology, visualization, software, writing–original draft. Qiankai Zhang: formal analysis, methodology, visualization, software, writing–original draft. Qi Tang: investigation, visualization, software, writing–original draft. Yin Zhang: methodology, visualization. Jun Zhou: writing–review & editing, project administration, supervision. Yang Wang: writing–review & editing, project administration, supervision. Kai Wu: supervision.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting the findings of this study are available within the article. No additional data were generated or analyzed in the presented research.Supplementary information (SI): the optimized crystal structure data (in .cif format) supporting the computational findings of this study. The data is organized into three composite files: (1) ‘All-Models_Clean-Structures’ containing pristine and doped MoS2 base models; (2) ‘All-Models_with-CO2’ containing structures after CO2 adsorption; and (3) ‘All-Models_Step-Intermediates’ containing the key reaction intermediate structures for all models. See DOI: https://doi.org/10.1039/d5ra08046k.
Acknowledgements
This research was funded by the Science and Technology Project of China Southern Power Grid Co., Ltd (Grant No. GDKJXM20231333), entitled “Key Technology for Power-to-Gas Based on Coupled Co-electrolysis of CO2and H2O”.References
- M. Asadi, B. Kumar, A. Behranginia, B. A. Rosen, A. Baskin, N. Repnin, D. Pisasale, P. Phillips, W. Zhu and R. Haasch, et al., Robust carbon dioxide reduction on molybdenum disulphide edges, Nat. Commun., 2014, 5, 4470 CrossRef CAS PubMed.
- S. Budinis, S. Krevor, N. M. Dowell, N. Brandon and A. Hawkes, An assessment of CCS costs, barriers and potential, Energy Strategy Rev., 2018, 22, 61–81 Search PubMed.
- L. Luo, M. Wang, Y. Cui, Z. Chen, J. Wu and Y. Cao, et al., Surface iron species in palladium-iron intermetallic nanocrystals that promote and stabilize CO2 methanation, Angew. Chem., Int. Ed., 2020, 59, 14434–14442 Search PubMed.
- Y. Yang, J. Liu, F. Liu and D. Wu, Reaction mechanism of CO2 methanation over Rh/TiO2 catalyst, Fuel, 2020, 276, 118093 CrossRef CAS.
- M. Qiu, H. Tao, Y. Li, Y. Li, K. Ding and X. Huang, et al., Toward improving CO2 dissociation and conversion to methanol via CO-hydrogenation on Cu (100) surface by introducing embedded Co nanoclusters as promoters: a DFT study, Appl. Surf. Sci., 2018, 427, 837–847 CrossRef CAS.
- Z. Y. Sun, T. Ma, H. C. Tao, Q. Fan and B. X. Han, Fundamentals and challenges of electrochemical CO2 reduction using two-dimensional materials, Chem, 2017, 3, 560–587 Search PubMed.
- M. Chhowalla, H. S. Shin, G. Eda, L. J. Li, K. P. Loh and H. Zhang, The chemistry of two-dimensional layered transition metal dichalcogenide nanosheets, Nat. Chem., 2013, 5, 263–275 CrossRef PubMed.
- X. Chia, A. Adriano, P. Lazar, Z. Sofer, J. Luxa and M. Pumera, Layered platinum dichalcogenides (PtS2, PtSe2, and PtTe2) electrocatalysis: monotonic dependence on the chalcogen size, Adv. Funct. Mater., 2016, 26, 4306–4318 CrossRef CAS.
- H. Li, C. Tsai, A. L. Koh, L. Cai, A. W. Contryman, A. H. Fragapane, J. Zhao, H. S. Han, H. C. Manoharan, F. Abild-Pedersen, J. K. Norskov and X. Zheng, Activating and optimizing MoS2 basal planes for hydrogen evolution through the formation of strained sulphur vacancies, Nat. Mater., 2016, 15, 48–53 CrossRef CAS PubMed.
- J. Liu, T. X. Liang, R. Tu, W. S. Lai and Y. J. Liu, Redistribution of π and σ electrons in boron-doped graphene from DFT investigation, Appl. Surf. Sci., 2019, 481, 344–352 CrossRef CAS.
- N. Yu, L. Wang, M. Li, X. Sun, T. Hou and Y. Li, Molybdenum disulfide as a highly efficient adsorbent for non-polar gases, Phys. Chem. Chem. Phys., 2015, 17, 11700–11704 RSC.
- X. Mao, L. Wang, Y. Xu and Y. Li, Modulating the MoS2 edge structures by doping transition metals for electrocatalytic CO2 reduction, J. Phys. Chem. C, 2020, 124, 10523–10529 CrossRef CAS.
- Z. Y. Li, D. A. Zhou and J. Xiangke, Highly selective adsorption on monolayer MoS2 doped with Pt, Ag, Au and Pd and effect of strain engineering: A DFT study, Sens. Actuators, A, 2021, 332, 113176 Search PubMed.
- J. Q. Xu, X. Li, W. Liu, Y. Sun, Z. Ju, T. Yao, C. Wang, H. Ju, J. Zhu and S. Wei, et al., Carbon dioxide electroreduction into syngas boosted by a partially delocalized charge in molybdenum sulfide selenide alloy monolayers, Angew. Chem., Int. Ed., 2017, 56, 9121–9125 Search PubMed.
- S. Park, J. Park, H. Abroshan, L. Zhang, J. K. Kim, J. M. Zhang, J. H. Guo, S. Siahrostami and X. L. Zheng, Enhancing catalytic activity of MoS2 basal plane S-vacancy by Co cluster addition, ACS Energy Lett., 2018, 3, 2685–2693 CrossRef CAS.
- G. D. Shi, L. Yu, X. Ba, X. S. Zhang, J. Q. Zhou and Y. Yu, Copper nanoparticle interspersed MoS2 nanoflowers with enhanced efficiency for CO2 electrochemical reduction to fuel, Dalton Trans., 2017, 46, 10569–10577 Search PubMed.
- C. C. Wu, W. J. Yang, J. Y. Wang, R. Kannaiyan and I. D. Gates, CO2 adsorption and dissociation on single and double iron atomic molybdenum disulfide catalysts: A DFT study, Fuel, 2021, 305, 121547 Search PubMed.
- Q. Yu, Theoretical studies of non-noble metal single-atom catalyst Ni1/MoS2: Electronic structure and electrocatalytic CO2 reduction, Sci. China Mater., 2022, 66, 1079–1088 Search PubMed.
- B. Moss, Q. Wang and K. T. Butle, et al., Linking in situ charge accumulation to electronic structure in doped SrTiO3 reveals design principles for hydrogen-evolving photocatalysts, Nat. Mater., 2021, 20, 511–517 CrossRef CAS PubMed.
- V. S. Vaiss and L. T. Costa, Theoretical Systematic Investigation as a Strategic Tool for the Design of More Efficient Pure and Doped MoS2 Catalysts for CO2 Electroreduction, Chem. Phys., 2025, 591, 112597 CrossRef CAS.
- K. L. Lv, W. Q. Suo, M. D. Shao, Y. Zhu, X. P. Wang, J. J. Feng, M. W. Fang and Y. Zhu, Nitrogen doped MoS2 and nitrogen doped carbon dots composite catalyst for electroreduction CO2 to CO with high Faradaic efficiency, Nano Energy, 2019, 63, 103834 CrossRef CAS.
- P. Abbasi, M. Asadi, C. Liu, S. Sharifi-Asl, B. Sayahpour, A. Behranginia, P. Zapol, R. Shahbazian-Yassar, L. A. Curtiss and A. Salehi-Khojin, Tailoring the edge structure of molybdenum disulfide toward electrocatalytic reduction of carbon dioxide, ACS Nano, 2017, 11, 453–460 Search PubMed.
- J. W. Li, W. M. Jia, C. Liu, S. S. Lv, J. T. Wang and Z. C. Li, Static and dynamic evolution of CO adsorption on γ-U (1 0 0) surface with different levels of Mo doping using DFT and AIMD calculations, Nucl. Sci. Tech., 2023, 34, 142–154 CrossRef.
- S. L. Yang, X. T. Wang, G. Lei, H. X. Xu, Z. Wang, J. Xiong and H. S. Gu, A DFT study on the outstanding hydrogen storage performance of the Ti-decorated MoS2 monolayer, Surf. Interfaces, 2021, 26, 101329 CrossRef CAS.
- Y. L. Xie, X. Li, Y. Wang, B. W. Li, L. Yang, N. Zhao, M. F. Liu, X. Z. Wang, Y. Yu and J. M. Liu, Reaction mechanisms for reduction of CO2 to CO on monolayer MoS2, Appl. Surf. Sci., 2020, 499, 143964 Search PubMed.
- Y. P. Miao, H. W. Bao, W. Fan, Y. Li and F. Ma, The adsorption behaviors of pristine MoS2 and N-MoS2 monolayer: A first-principles calculation, Surf. Interfaces, 2021, 27, 101580 CrossRef CAS.
- S. J. Clark, M. D. Segall, C. J. Pickard, P. J. Hasnip, M. J. Probert, K. Refson and M. C. Payne, First principles methods using CASTEP, Z. Kristallogr., 2005, 220, 567–570 Search PubMed.
- J. P. Perdew, K. Burke and M. Ernzerhof, Generalized gradient approximation made simple, Phys. Rev. Lett., 1996, 77, 3865–3868 CrossRef CAS PubMed.
- H. Cui and P. F. Jia, Doping effect of small Rhn (n = 1–4) clusters on the geometric and electronic behaviors of MoS2 monolayer: A first-principles study, Appl. Surf. Sci., 2020, 526, 146659 Search PubMed.
- C. P. Huelmo, M. G. Menezes, R. B. Capaz and P. A. Denis, Structural and magnetic properties of a defective graphene buffer layer grown on SiC(0001): a DFT study, Phys. Chem. Chem. Phys., 2020, 22, 19963–19972 Search PubMed.
- S. Grimme, Semiempirical GGA-type density functional constructed with a long-range dispersion correction, J. Comput. Chem., 2006, 27, 1787–1799 Search PubMed.
- C. Li, J. B. Li, F. M. Wu, S. S. Li, J. B. Xia and L. W. Wang, High capacity hydrogen storage in Ca decorated graphyne: A first-principles study, J. Phys. Chem. C, 2011, 115, 23221–23225 Search PubMed.
- C. Freysoldt, B. Grabowski and T. Hickel, et al., First-principles calculations for point defects in solids, Rev. Mod. Phys., 2014, 86, 253–305 Search PubMed.
- C. Z. He, R. Wang, D. Xiang, X. Y. Li, L. Fu, Z. Y. Jian, J. R. Huo and S. Li, Charge-regulated CO2 capture capacity of metal atom embedded graphyne: a first-principles study, Appl. Surf. Sci., 2020, 515, 145392 Search PubMed.
- H. P. Zhan, A. J. Du, Q. B. Shi, Y. F. Zhou, Y. P. Zhang and Y. H. Tang, Adsorption behavior of CO2 on pristine and doped phosphorenes: A dispersion corrected DFT study, J. CO2 Util., 2018, 24, 463–470 Search PubMed.
- K. Ghosh, N. K. Mridha, A. A. Khan, N. Baildya, T. Dutta and K. Biswas, CO2 activation on transition metal decorated graphene quantum dots: An insight from first principles, Phys. E, 2022, 135, 114993 Search PubMed.
- M. Y. Wang, W. Wang, M. Ji and X. L. Cheng, Adsorption of phenol and hydrazine upon pristine and X-decorated (X = Sc, Ti, Cr and Mn) MoS2 monolayer, Appl. Surf. Sci., 2018, 439, 350–363 CrossRef CAS.
- C. Ataca and S. Ciraci, Functionalization of single-layer MoS2 honeycomb structures, J. Phys. Chem. C, 2011, 115, 13303–13311 Search PubMed.
- A. Abbasi, A. Abdelrasoul and J. J. Sardroodi, Adsorption of CO and NO molecules on Al, P and Si embedded MoS2 nanosheets investigated by DFT calculations, Adsorption, 2019, 25, 1493–1506 CrossRef.
- A. Abbasi and J. J. Sardroodi, Theoretical study of the adsorption of NOx on TiO2/MoS2 nanocomposites: a comparison between undoped and N-doped nanocomposites, J. Nanostruct. Chem., 2016, 6, 309–327 CrossRef CAS.
- R. Y. Zhang, D. Fu, J. M. Ni, C. B. Sun and S. X. Song, Adsorption for SO2 gas molecules on B, N, P and Al doped MoS2: the DFT study, Chem. Phys. Lett., 2018, 712, 51–58 Search PubMed.
- W. Huang, H. Da and G. Liang, Thermoelectric performance of MX2 (M=Mo, W; X=S, Se) monolayers, J. Appl. Phys., 2013, 113, 104304 CrossRef.
- M. J. Szary, Adsorption of ethylene oxide on doped monolayers of MoS2: A DFT study, Mater. Sci. Eng., B, 2021, 272, 115341 Search PubMed.
- R. S. Mulliken, Electronic population analysis on LCAO–MO molecular wave functions. II. Overlap populations, bond orders, and covalent bond energies, J. Chem. Phys., 1955, 23, 1841–1846 Search PubMed.
- Y. Wen, L. Wang, H. Liu and L. Song, Ab initio study of the elastic and mechanical properties of B19TiAl, Crystals, 2017, 7, 39 Search PubMed.
- M. L. Ali, M. Khan, M. A. Al Asad, M. Z. Rahaman, M. Hasan, M. Chowdhury, M. K. Hossain, J. K. Modak, S. Ezzine and M. Amami, Highly efficient and stable lead-free cesium copper halide perovskites for optoelectronic applications: A DFT based study, Heliyon, 2023, 9, e18816 Search PubMed.
- Y. Li, R. Wang, G. Zhu, G. Li and Y. Cao, A DFT study on the effect of lattice defects on the electronic structures and floatability of spodumene, Phys. B, 2024, 676, 415657 Search PubMed.
- P. Barman, M. F. Rahman, M. R. Islam, M. Hasan, M. Chowdhury, M. K. Hossain, J. K. Modak, S. Ezzine and M. Amami, Lead-free novel perovskite Ba3AsI3: First-principles insights into its electrical, optical, and mechanical properties, Heliyon, 2023, 9, e21675 CrossRef CAS PubMed.
- X. Wang, W. Liu, H. Duan, B. Wang, C. Han and D. Wei, The adsorption mechanism of calcium ion on quartz (101) surface: A DFT study, Powder Technol., 2018, 329, 158–166 CrossRef CAS.
- W. W. Cheng, Y. S. Xue, X. M. Luo and Y. Xu, A rare three-dimensional POM-based inorganic metal polymer bonded by CO2 with high catalytic performance for CO2 cycloaddition, Chem. Commun., 2018, 54, 1185–1188 Search PubMed.
- Y. Luo, Y. Xie, J. Zhou, Y. Li, Y. Yu and Z. Ren, Robust and Selective Electrochemical Reduction of CO2: The Case of Integrated 3D TiO2@MoS2 Architectures and Ti–S Bonding Effects, J. Mater. Chem. A, 2018, 6, 4706–4713 Search PubMed.
- J. Radhakrishnan and K. Biswas, Facile Synthesis of Ti Doped MoS2 and Its Superior Adsorption Properties, Mater. Lett., 2020, 280, 128522 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |