Open Access Article
Open Access ArticleVisible-light induced trifluoromethylation/cyclization cascade to access CF3-containing pyrazolones by EDA complex
Zilin Liu†
,
Mingxi Hu†,
Shuo Gao,
Pifeng Wei,
Mengmeng Zhao*,
Yunqiang Sun* and
Zhen-Hua Zhang *
*
School of Chemistry and Chemical Engineering, Linyi University, Linyi 276000, P. R. China. E-mail: zhaomengmeng@lyu.edu.cn; sunyunqiang@lyu.edu.cn; zhangzhenhua@iccas.ac.cn
First published on 9th March 2026
Abstract
The incorporation of trifluoromethyl groups into heterocycles often significantly enhances their bioactivity, thereby driving demand for efficient synthetic methods. Herein, a novel visible-light-driven, metal-free strategy is developed for the synthesis of trifluoromethylated pyrazolones via a radical addition/cyclization cascade. Under 390–400 nm irradiation, the reaction proceeds through an electron donor–acceptor (EDA) complex between Togni's reagent II and DABCO to generate CF3 radicals, without the need for an external photocatalyst. A wide range of N-methacryloylhydrazones are converted into the corresponding products with good to excellent yields. This method is operationally simple and scalable, and exhibits high functional group tolerance, thus providing a sustainable route to functionalized heterocycles.
Introduction
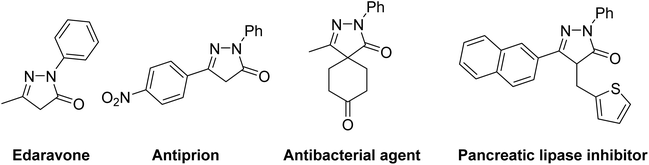
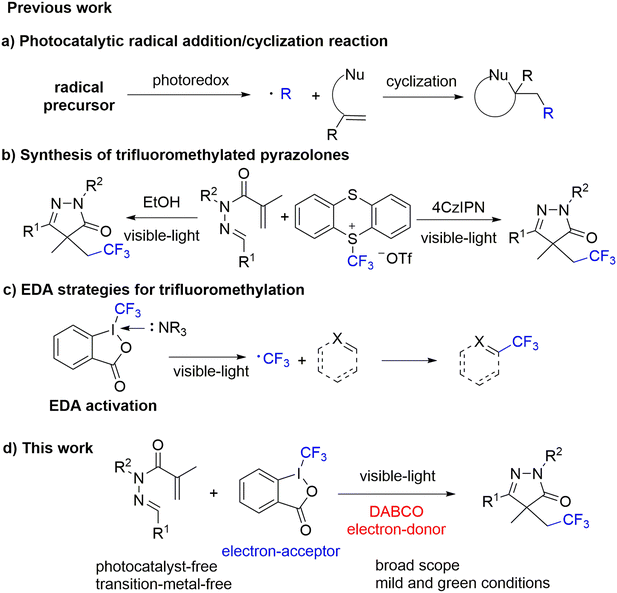
Pyrazolone compounds, as an important class of five-membered heterocycles, have attracted widespread interest in pharmaceutical research and development owing to their diverse medicinal properties.1,2 In particular, the 2-pyrazolin-5-one scaffold serves as a key structural motif in numerous bioactive molecules, and its derivatives are widely employed as intermediates for synthesizing functionalized pyrazoles (Fig. 1).2 This versatility has motivated sustained efforts over recent decades to develop synthetic routes to construct the 2-pyrazolin-5-one cores. Conventional methods primarily rely on the Knorr condensation between β-ketoesters and substituted hydrazines.3 Alternative strategies, such as palladium-catalyzed cyclocarbonylation of 1,2-diaza-1,3-butadienes,4 palladium-catalyzed carbonylative coupling of a-chloroketones with hydrazines,5 and gold-catalyzed tandem reactions of N-propioloyl hydrazones,6 have also been explored. However, these approaches suffer from inherent limitations, including dependence on expensive noble metal catalysts, requirement for high-temperature conditions, and a narrow substrate scope. Therefore, the development of efficient and generalizable synthetic methods for 2-pyrazolin-5-ones remains an ongoing challenge in modern synthetic chemistry.In recent years, photoredox catalysis has emerged as an exceptionally versatile tool in modern synthetic chemistry due to its high efficiency and sustainability.7 Visible-light-induced radical addition/cyclization cascade reactions are of great importance for the synthesis of nitrogen-containing heterocycles, owing to the unique redox properties of photocatalysts and their outstanding capability to generate diverse radical species (Scheme 1a).8 Through this reaction strategy, various alkenes can undergo radical addition followed by subsequent intramolecular radical cascade cyclization, enabling the construction of diversely functionalized heterocyclic compounds under mild conditions.9 However, most of these processes rely on noble Ru-/Ir-complexes or organic dyes as photocatalysts to drive the photoreactions, which imposes both economic and environmental burdens. In 2024, Murarka developed Ru-photoredox-catalyzed arylative and arylsulfonylative radical cascade reactions of N′-arylidene-N-acryloylhydrazides to access desired functionalized pyrazolones.10a Recently, Yu disclosed the visible-light-induced sulfamoylation/5-endo-trig cyclization between N′-arylidene-N-acryloylhydrazides and sulfamoyl chlorides using 4CzIPN as a photocatalyst.10b Therefore, the development of external catalyst-free visible-light-induced photocatalytic systems is of significant importance. Since 2000, the utilization of electron donor–acceptor (EDA) complexes, which formed electronically excited states in absence of organic and metal photocatalysts, has attracted significant interest.11 In 2024, Murarka established photo-induced radical cascade between alkyl NHPI esters and N-acryloyl aldehyde hydrazones via the EDA complex, providing a route to alkylated 2-pyrazoline-5-ones with high efficiency.12
The introduction of a trifluoromethyl group (–CF3) into organic compounds significantly enhances their lipophilicity and metabolic stability.13 This structural modification not only alters the electronic properties and three-dimensional conformation of the molecules, thereby improving the potency and selectivity of drug candidates, but also markedly affects key physicochemical properties such as boiling point and solubility.14 Radical trifluoromethylation reactions have emerged as a crucial approach for CF3 group incorporation, owing to their mild reaction conditions and excellent compatibility with diverse functional groups.15 A widely adopted strategy involves generating CF3 radicals from trifluoromethylation reagents, which subsequently undergo addition reactions with unsaturated systems such as alkenes or arenes, representing a mainstream methodology for CF3 group installation.16 Recently, Pan reported a photocatalytic trifluoromethylation/cyclization reaction of N-methacryloylhydrazone with trifluoromethyl thianthrenium triflate to afford trifluoromethylated pyrazolones in moderate to excellent yields (Scheme 1b).17 Almost simultaneously, He developed a self-catalyzed phototandem trifluoromethylation/cyclization of N-methacryloylhydrazones with trifluoromethyl thianthrenium triflate in EtOH (Scheme 1b).18 Building upon prior research, we aim to extend the applications of EDA complexes to more demanding and structurally complex chemical transformations. Although significant progress has been achieved in the trifluoromethylation of unsaturated bond using Togni's reagent II (Scheme 1c), the direct incorporation of the trifluoromethyl group into heterocyclic frameworks remains a substantial challenge in modern organic synthesis.19 Inspired by these elegant precedents, we present a visible-light-driven radical addition/cyclization cascade of N-methacryloylhydrazones with Togni's reagent II by an EDA complexes, which affords a range of trifluoromethylated pyrazolones derivatives (Scheme 1d).
Results and discussion
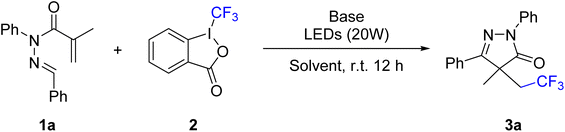
Initially, (E)-N′-benzylidene-N-phenylmethacrylohydrazide 1a was selected as the model substrate and Togni's reagent II 2 as the trifluoromethyl radical source to explore the cyclization reaction under visible-light conditions (Table 1). When the reaction was carried out in MeCN in the presence of 1,4-diazabicyclo[2.2.2]octane (DABCO) under irradiation with a 420–430 nm LED for 12 hours, the CF3-substituted pyrazolone 3a was obtained in 67% yield (entry 1). The influence of irradiation wavelength was then evaluated using LEDs of different emission ranges (entries 2–5). The highest reaction efficiency was achieved with a purple 390–400 nm LED. Screening of various bases, including Cs2CO3, DBU, and t-BuOK, showed that these alternatives were less effective (entries 6–8). Replacing MeCN with other solvents, such as THF, MeOH, EtOH, DMSO, or DMF, did not significantly improve the product yield (entries 9–13). Reducing the amount of DABCO to 0.2 mmol or 0.04 mmol led to diminished yields of 68% and 41%, respectively (entries 14 and 15). Control experiments confirmed that both light irradiation and DABCO are essential for the trifluoromethylation/cyclization process (entries 16 and 17). Finally, conducting the reaction under an air atmosphere resulted in a significantly reduced product yield (entry 18).| Entry | Base | LEDs | Solvent | Yieldb (%) |
|---|---|---|---|---|
| a Reaction conditions: 1a (0.2 mmol), 2 (0.4 mmol), base (0.4 mmol), in dry solvent (2.0 mL) under N2 at room temperature with the irradiation of LED lamps for 12 h.b Isolated yield based on 1a.c DABCO (0.2 mmol) was added.d DABCO (0.04 mmol) was added.e Under air. DABCO, 1,4-diazocyclic[2.2.2]octane; DBU, 8-diazabicyclo[5.4.0]undec-7-ene; THF, tetrahydrofuran; DMSO, dimethyl sulfoxide; DMF, N,N-dimethylformamide. | ||||
| 1 | DABCO | 420–430 nm | MeCN | 67 |
| 2 | DABCO | 390–400 nm | MeCN | 91 |
| 3 | DABCO | 380–390 nm | MeCN | 75 |
| 4 | DABCO | 400–800 nm | MeCN | 47 |
| 5 | DABCO | 520–530 nm | MeCN | Trace |
| 6 | Cs2CO3 | 390–400 nm | MeCN | 52 |
| 7 | DBU | 390–400 nm | MeCN | 35 |
| 8 | t-BuOK | 390–400 nm | MeCN | 21 |
| 9 | DABCO | 390–400 nm | THF | 78 |
| 10 | DABCO | 390–400 nm | MeOH | 56 |
| 11 | DABCO | 390–400 nm | EtOH | 82 |
| 12 | DABCO | 390–400 nm | DMSO | 37 |
| 13 | DABCO | 390–400 nm | DMF | 46 |
| 14c | DABCO | 390–400 nm | MeCN | 68 |
| 15d | DABCO | 390–400 nm | MeCN | 41 |
| 16 | — | 390–400 nm | MeCN | Trace |
| 17 | DABCO | Dark | MeCN | 0 |
| 18e | DABCO | 390–400 nm | MeCN | 18 |
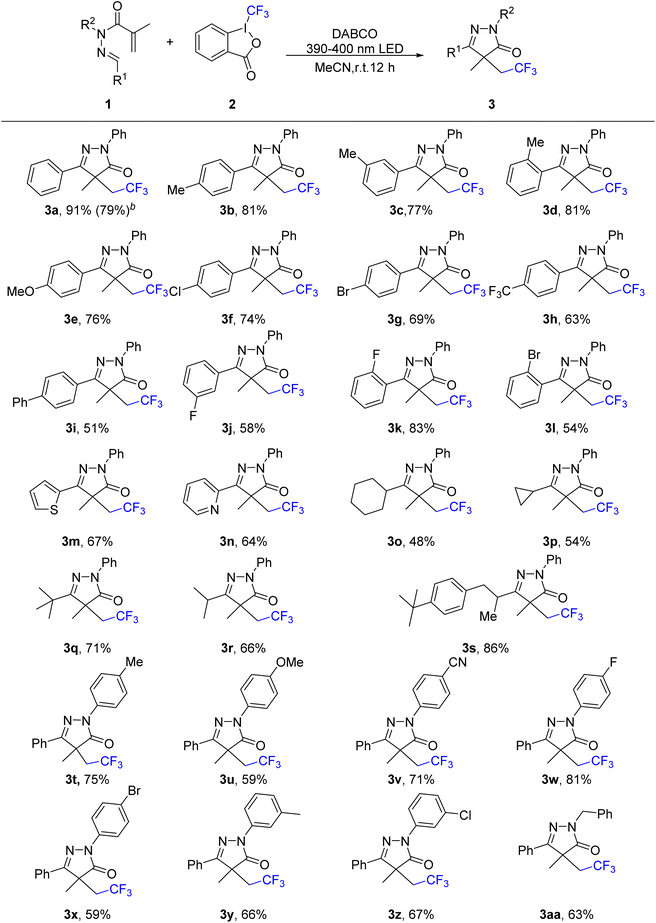
With the optimized conditions established (Table 1, entry 2), we next evaluated the substrate scope of the photoinduced trifluoromethylation/cyclization tandem reaction (Scheme 2). As expected, the current metal-free photocatalytic system exhibited broad applicability across a diverse range of N-methacryloyl hydrazones using Togni's reagent II. Aryl aldehyde-derived hydrazones bearing a methyl group at the ortho-, meta-, or para-position of the benzene ring underwent the cascade reaction smoothly, affording products 3b–3d in good to excellent yields. Notably, both electron-donating (methoxy and phenyl) and electron-withdrawing (bromo, chloro, fluoro, and trifluoromethyl) substituents on the phenyl ring were well tolerated, delivering products 3e–3l in 51–83% yields. Replacement of the benzene ring with thiophene or pyridine heterocycles also proceeded efficiently, affording the corresponding trifluoromethylated pyrazolones 3m and 3n in 67% and 64% yield, respectively. Furthermore, N-methacryloyl hydrazones derived from alkyl aldehydes containing cycloalkyl (cyclopropyl and cyclohexyl) or alkyl (tert-butyl and isopropyl) groups were also compatible, yielding the corresponding products 3o–3r. The hydrazone derived from Lily aldehyde also afforded product 3s in 86% yield. Subsequently, we investigated the reactivity of arylhydrazine-derived hydrazones under the optimized conditions. This method proved effective, delivering the target products 3t–3z in moderate to good yields, irrespective of whether the aryl ring bore electron-donating or electron-withdrawing substituents. In addition, the benzyl hydrazine-derived substrate was also compatible under the standard conditions, affording the corresponding product 3aa in 63% yield. Finally, to demonstrate the utility of this photoinduced cascade method, the reaction between 1a and 2 can scale-up to 2 mmol with the isolation of 3a in 79% yield.
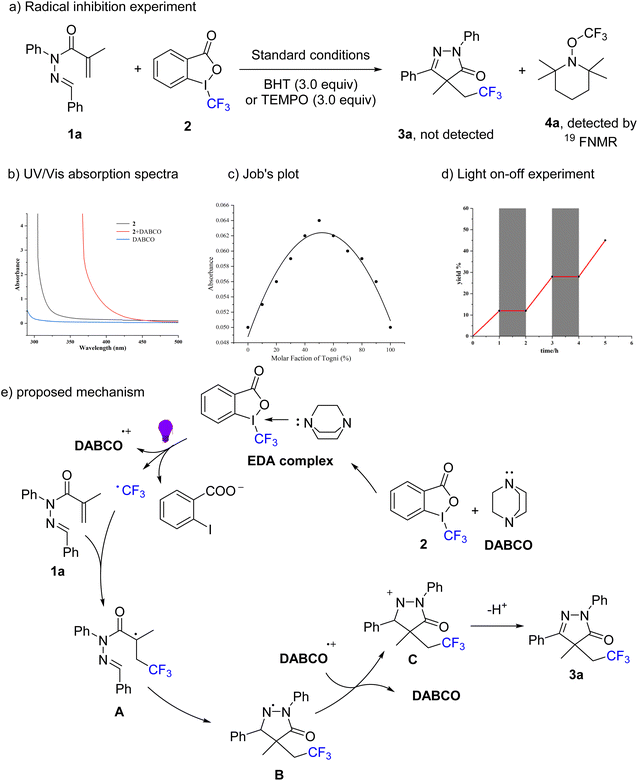
To gain deeper mechanistic insights into the visible-light-driven trifluoromethylation/cyclization cascade, a series of control experiments were carried out. In first, the addition of 3.0 equivalents of the radical scavengers 2,6-di-tert-butyl-4-methylphenol (BHT) or 2,2,6,6-tetramethylpiperidin-1-oxyl (TEMPO) under standard conditions completely inhibited the reaction (Scheme 3a). The formation of the radical adduct 4a was detected by 19F NMR spectroscopy, providing strong evidence for the involvement of radical intermediates in this transformation. Since the reaction proceeded efficiently in the absence of any exogenous photocatalyst, we sought to investigate the potential involvement of an EDA complex as the initiating species in the radical mechanism. Consistent with the findings of previous studies, a distinct bathochromic shift was detected in the UV-vis absorption spectrum when Togni's reagent II 2 and DABCO were mixed at a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio in acetonitrile solution, indicating the formation of an EDA complex between the two reactants (Scheme 3b). Furthermore, subsequent analysis via the Job's plot method confirmed the formation of a 1
1 molar ratio in acetonitrile solution, indicating the formation of an EDA complex between the two reactants (Scheme 3b). Furthermore, subsequent analysis via the Job's plot method confirmed the formation of a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 EDA complex between Togni's reagent II 2 and DABCO (Scheme 3c). The possibility of a radical chain mechanism operating in this EDA complex system was ruled out by a light on–off experiment conducted under standard conditions between N-methacryloyl hydrazone 1a and 2 (Scheme 3d).
1 EDA complex between Togni's reagent II 2 and DABCO (Scheme 3c). The possibility of a radical chain mechanism operating in this EDA complex system was ruled out by a light on–off experiment conducted under standard conditions between N-methacryloyl hydrazone 1a and 2 (Scheme 3d).
According to the aforementioned mechanistic studies and previous reports,17–19 a plausible reaction mechanism was illustrated in Scheme 3e. Initially, Togni's reagent II 2 formed an EDA complex with DABCO, which underwent dissociation under purple light irradiation to generate a trifluoromethyl radical, a DABCO radical cation, and an 2-iodobenzoate anion. Subsequently, the electrophilic CF3 radical attacked the acryl double bond in 1a to afford the radical intermediate A. Then, this key intermediate preferred to add the more electron-rich C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N double bond of the hydrazone via 5-endo-trig cyclization, generating the N-centered radical intermediate B. Next, the SET oxidation of intermediate B by the DABCO radical cation concurrently regenerated DABCO and produced cationic intermediate C. Ultimately, this cationic intermediate then underwent deprotonation to furnish the target product 3a.
N double bond of the hydrazone via 5-endo-trig cyclization, generating the N-centered radical intermediate B. Next, the SET oxidation of intermediate B by the DABCO radical cation concurrently regenerated DABCO and produced cationic intermediate C. Ultimately, this cationic intermediate then underwent deprotonation to furnish the target product 3a.
Conclusions
In summary, we have developed a novel visible-light-promoted, metal-free radical addition/cyclization cascade strategy for the efficient construction of trifluoromethylated pyrazolone derivatives. Under optimized conditions using commercially available Togni's reagent II and DABCO as the radical source under 390–400 nm light irradiation, a variety of N-methacryloylhydrazones were smoothly transformed into the desired products in good to excellent yields. Mechanistic studies revealed that a key EDA complex formed between the trifluoromethylation reagent and DABCO facilitates efficient radical generation without the need for an external photocatalyst. This method is operationally simple, readily scalable, and exhibits broad substrate scope with good functional group tolerance, offering a green and sustainable approach to the functionalization of heterocyclic compounds.Author contributions
Zilin Liu: investigation, data curation, and methodology. Mingxi Hu: investigation and data curation. Shuo Gao: data curation. Pifeng Wei: data curation. Mengmeng Zhao: supervision and writing – review & editing. Yunqiang Sun: writing – original draft. Zhen-Hua Zhang: writing – review & editing, supervision, funding acquisition, and conceptualization.Conflicts of interest
There are no conflicts to declare.Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5ra08028b.
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (No. 21804066), Shandong Provincial Natural Science Foundation (No. ZR2017BB065, ZR2020QB038 and ZR2024MB041), and the College Students' Innovation and Entrepreneurship Training Program by Linyi University (No. X202410452251 and X2025104520566).Notes and references
- (a) Z. Zhao, X. Dai, C. Li, X. Wang, J. Tian, Y. Feng, J. Xie, C. Ma, Z. Nie, P. Fan, M. Qian, X. He, S. Wu, Y. Zhang and X. Zheng, Eur. J. Med. Chem., 2020, 186, 111893 CrossRef CAS PubMed; (b) R. Verma, S. K. Verma, K. P. Rakesh, Y. R. Girish, M. Ashrafizadeh, K. S. Sharath Kumar and K. S. Rangappa, Eur. J. Med. Chem., 2021, 212, 113134 CrossRef CAS PubMed.
- (a) H. Yoshida, H. Yanai, Y. Namiki, K. Fukatsu-Sasaki, N. Furutani and N. Tada, CNS Drug Rev., 2006, 12, 9–20 CrossRef CAS PubMed; (b) A. Kimata, H. Nakagawa, R. Ohyama, T. Fukuuchi, S. Ohta, T. Suzuki and N. Miyata, J. Med. Chem., 2007, 50, 5053–5056 CrossRef CAS PubMed; (c) V. Hadi, Y.-H. Koh, T. W. Sanchez, D. Barrios, N. Neamati and K. W. Jung, Bioorg. Med. Chem. Lett., 2010, 20, 6854–6857 CrossRef CAS PubMed; (d) J. Zhang, Y. Yang, X.-K. Qian, P.-F. Song, Y.-S. Zhao, X.-Q. Guan, L.-W. Zou, X. Bao and H. Wang, ChemMedChem, 2021, 16, 1600–1604 CrossRef CAS PubMed.
- (a) M. M. Mojtahedi, M. R. Jalali, M. S. Abaee and M. Bolourtchian, Heterocycl. Commun., 2006, 12, 225–228 CrossRef CAS; (b) M. Desroses, M.-C. Jacques-Cordonnier, S. Llona-Minguez, S. Jacques, T. Koolmeister, T. Helleday and M. Scobie, Eur. J. Org Chem., 2013, 2013, 5879–5885 CrossRef CAS; (c) J. L. Howard, W. Nicholson, Y. Sagatov and D. L. Browne, Beilstein J. Org. Chem., 2017, 13, 1950–1956 CrossRef CAS PubMed.
- R. K. Boeckman, J. E. Reed and P. Ge, Org. Lett., 2001, 3, 3651–3653 CrossRef CAS PubMed.
- M. Capua, C. Granito, S. Perrone, A. Salomone and L. Troisi, Tetrahedron Lett., 2016, 57, 3363–3367 CrossRef CAS.
- Z.-C. Ding, H.-T. Tang, R.-H. Li, L.-C. Ju and Z.-P. Zhan, J. Org. Chem., 2015, 80, 9307–9313 CrossRef CAS PubMed.
- (a) C. K. Prier, D. A. Rankic and D. W. C. MacMillan, Chem. Rev., 2013, 113, 5322–5363 CrossRef CAS PubMed; (b) Y. Chen, L.-Q. Lu, D.-G. Yu, C.-J. Zhu and W.-J. Xiao, Sci. China: Chem., 2019, 62, 24–57 CrossRef CAS; (c) N. Holmberg-Douglas and D. A. Nicewicz, Chem. Rev., 2022, 122, 1925–2016 CrossRef CAS PubMed; (d) Y.-Z. Cheng, Z. Feng, X. Zhang and S.-L. You, Chem. Soc. Rev., 2022, 51, 2145–2170 RSC; (e) P. Bellotti, H.-M. Huang, T. Faber and F. Glorius, Chem. Rev., 2023, 123, 4237–4325 CrossRef CAS PubMed; (f) S. Khalid, M. Bilal, N. Rasool and M. Imran, Chin. Chem. Lett., 2024, 35, 109498 CrossRef CAS.
- (a) M. P. Plesniak, H.-M. Huang and D. J. Procter, Nat. Rev. Chem., 2017, 1, 0077 CrossRef; (b) D. Menigaux, P. Belmont and E. Brachet, Eur. J. Org Chem., 2017, 15, 2008–2055 CrossRef; (c) J. Liao, X. Yang, L. Ouyang, Y. Lai, J. Huang and R. Luo, Org. Chem. Front., 2021, 8, 1345–1363 RSC.
- (a) J. Singh and A. Sharma, Adv. Synth. Catal., 2021, 363, 4284–4308 CrossRef CAS; (b) M.-J. Luo, Q. Xiao and J.-H. Li, Chem. Soc. Rev., 2022, 51, 7206–7237 RSC; (c) Y. Zhou, S. Xu, X. Zhang, L. Zhou, H. Zheng and G. Zhu, Chem. Commun., 2024, 60, 10098–10111 RSC; (d) A. Das, S. Gouthamana and K. R. J. Thomas, Green Chem., 2024, 26, 1223–1280 RSC.
- (a) K. R. Thombare, S. K. Parida, P. Meher and S. Murarka, Chem. Commun., 2024, 60, 13907–13910 RSC; (b) S. Yu, Y. Cheng, C. Pan and J.-T. Yu, Chem. Commun., 2025, 61, 1196–1199 RSC.
- (a) C. G. S. Lima, T. d. M. Lima, M. Duarte, I. D. Jurberg and M. W. Paixão, ACS Catal., 2016, 6, 1389–1407 CrossRef CAS; (b) G. E. M. Crisenza, D. Mazzarella and P. Melchiorre, J. Am. Chem. Soc., 2020, 142, 5461–5476 CrossRef CAS PubMed; (c) Y. Yuan, S. Majumder, M. Yang and S. Guo, Tetrahedron Lett., 2020, 61, 151506 CrossRef CAS; (d) J. D. Lasso, D. J. Castillo-Pazos, J. M. Salgado, C. Ruchlin, L. Lefebvre, D. Farajat, D. F. Perepichka and C.-J. Li, J. Am. Chem. Soc., 2024, 146, 2583–2592 CrossRef CAS PubMed; (e) X.-L. Huang, D.-L. Zhang, Q. Li, Z.-B. Xie, Z.-G. Le and Z.-Q. Zhu, Org. Lett., 2024, 26, 3727–3732 CrossRef CAS PubMed.
- S. P. Panda, R. Dash, S. K. Hota and S. Murarka, Org. Lett., 2024, 26, 3667–3672 CrossRef CAS PubMed.
- (a) N. A. Meanwell, J. Med. Chem., 2018, 61, 5822–5880 CrossRef CAS PubMed; (b) B. M. Johnson, Y. Z. Shu, X. Zhuo and N. A. Meanwell, J. Med. Chem., 2020, 63, 6315–6386 CrossRef CAS PubMed.
- (a) O. A. Tomashenko and V. V. Grushin, Chem. Rev., 2011, 111, 4475–4521 CrossRef CAS PubMed; (b) T. Furuya, A. S. Kamlet and T. Ritter, Nature, 2011, 473, 470–477 CrossRef CAS PubMed; (c) T. Liang, C. N. Neumann and T. Ritter, Angew. Chem., Int. Ed., 2013, 52, 8214–8264 CrossRef CAS PubMed; (d) Y. Zhou, J. Wang, Z. Gu, S. Wang, W. Zhu, J. L. Aceña, V. A. Soloshonok, K. Izawa and H. Liu, Chem. Rev., 2016, 116, 422–518 CrossRef CAS PubMed.
- (a) T. Chatterjee, N. Iqbal, Y. You and E. J. Cho, Acc. Chem. Res., 2016, 49, 2284–2294 CrossRef CAS PubMed; (b) S. Barata-Vallejo, M. V. Cooke and A. Postigo, ACS Catal., 2018, 8, 7287–7307 CrossRef CAS; (c) H. Xiao, Z. Zhang, Y. Fang, L. Zhu and C. Li, Chem. Soc. Rev., 2021, 50, 6308–6319 RSC.
- (a) D. A. Nagib and D. W. C. MacMillan, Nature, 2011, 480, 224–228 CrossRef CAS PubMed; (b) C. Hyeon Ka, S. Kim and E. Jin Cho, Chem. Rec., 2023, 23, e202300036 CrossRef CAS PubMed; (c) Y. Ouyang and F.-L. Qing, J. Org. Chem., 2024, 89, 2815–2824 CrossRef CAS PubMed.
- C. Pan, Y. Gong and M. Zeng, J. Org. Chem., 2025, 90, 9457–9467 CrossRef CAS PubMed.
- C.-H. Liu, Z.-W. Zhang, Z.-J. Zhao, J. Jiang, Z. Yang, Z.-L. Wang and W.-M. He, J. Org. Chem., 2025, 90, 11982–11989 CrossRef CAS PubMed.
- (a) Y. Cheng and S. Yu, Org. Lett., 2016, 18, 2962–2965 CrossRef CAS PubMed; (b) H. Jiang, Y. He, Y. Cheng and S. Yu, Org. Lett., 2017, 19, 1240–1243 CrossRef CAS PubMed; (c) H. Liu, X. Fan, J. Hu, T. Ma, F. Wang, J. Yang and D. Li, J. Org. Chem., 2022, 87, 12877–12889 CrossRef CAS PubMed; (d) Y. Li, J. Hou, P. Zhang, P. Dai, Y.-C. Gu, Q. Xia and W. Zhang, Chem.–Eur. J., 2024, 30, e202400237 CrossRef CAS PubMed; (e) J. Wang, Y. Liu, J. Xu, P. Li, X. Xu, S. Zhang, J. Yang and X. Xu, Org. Chem. Front., 2025, 12, 1156–1161 RSC.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |