Open Access Article
Open Access ArticleFerroptosis and immunity: rewiring the tumor microenvironment for therapy
Gaowa Ailuna,
Hongmei Gub,
Peter Uchenna Amadic,
XiuJuan Chena,
Manzar Abbas de,
Ya Tuoa and
Da-wei Zhang*b
de,
Ya Tuoa and
Da-wei Zhang*b
aThe Affiliated Hospital of Inner Mongolia Medical University, Hohhot 010010, Inner Mongolia, China
bDepartment of Pediatrics, Department of Biochemistry (Adjunct) Group on Molecular and Cell Biology of Lipids Cardiovascular Research Center, Women and Children's Health Research Institute, Faculty of Health Sciences, University of Alberta, Alberta, Canada. E-mail: dzhang@ualberta.ca
cDepartment of Pediatrics, Faculty of Medicine & Dentistry, College of Health Sciences, University of Alberta, Alberta, Canada
dInner Mongolia Saikexing Institute of Breeding and Reproductive Biotechnology in Domestic Animals, Hohhot, China
eNational Center of Technology Innovation for Dairy, Hohhot, 010020, China
First published on 20th February 2026
Abstract
Ferroptosis, a distinct form of programmed cell death driven by iron-dependent lipid peroxidation, has emerged as a critical player in cancer biology, influencing both tumor progression and therapeutic responses. This review explores the multifaceted role of ferroptosis within the tumor microenvironment (TME), focusing on its dual potential to promote anti-tumor immunity and immune evasion. Key molecular mechanisms regulating ferroptosis, including the roles of GPX4, SLC7A11, and ACSL4, are examined alongside their interplay with immune cells such as CD8+ T cells, dendritic cells, and macrophages. The review addresses the immunosuppressive effects of ferroptosis-induced signals such as prostaglandin E2 and damage-associated molecular patterns that foster tumor growth. Furthermore, therapeutic strategies leveraging ferroptosis to overcome resistance in cancer treatment, including its integration with immunotherapy and radiotherapy, are discussed. This study underscores the potential of targeting ferroptosis to enhance cancer therapy while emphasizing the need for further research to optimize its application in immunotherapy.
1 Background
The human immune system is comprised of leukocytes, along with organs and tissues of the lymphatic system, including the thymus, spleen, tonsils, lymph nodes, lymphatic vessels, and bone marrow.1 Its primary function is to identify and eliminate various threats to maintain homeostasis. To fully comprehend the role of immunotherapy as a cornerstone of cancer treatment, it is essential to understand the complex interactions between cancer cells and the immune system.2 Unlike chemotherapy, which exerts cytotoxic effects directly on cancer cells, immunotherapies leverage the host immune system to target and eliminate tumor cells. Edward Jenner's development of the first successful vaccine against smallpox in 1796 is widely regarded as the foundation of immunology.3 Later, in 1891, Doctor William Coley, often referred to as the father of immunotherapy, sought alternative treatments for sarcoma following the tragic death of an adolescent patient.4 The intricate networks of the immune system predominantly suppress cancer cells. However, tumors have evolved various mechanisms to evade anti-tumor immunity. A tumor is not merely a collection of malignant cells but represents a heterogeneous ensemble comprising infiltrating and resident cells, secreted bioactive factors, and extracellular matrix (ECM).5 Tumor cells derive significant molecular cellular and structural remodeling of the surrounding host tissue to facilitate tumorigenesis and progression. The tumor microenvironment (TME) is a dynamic evolving entity with its composition varying across different tumor types.6 Nonetheless, essential components of the TME typically include immune cells, stromal cells, vasculature, and the ECM. During the early stage of tumor development, intricate and reciprocal interactions emerge between malignant cells and the TME constituents, supporting cancer cell survival, local invasion, and metastatic dissemination.7 To adapt to hypoxic and acidic conditions, the TME activates an angiogenic program, ensuring the restoration of oxygen and nutrient supply while facilitating the clearance of metabolic byproducts. Tumors are infiltrated by a diverse repertoire of innate and adaptive immune cells that exhibit either pro-tumorigenic or anti-tumorigenic activity.8 Specific immune cells, such as regulatory T-cells and myeloid-derived suppressor cells, promote tumor growth and progression, whereas others, including cytotoxic T lymphocytes and natural killer cells, mediate the recognition and destruction of malignant cells.9 Growing research on the TME has expanded our understanding of its role in tumor progression and identified novel therapeutic targets within the microenvironment, offering promising avenues for cancer treatment strategies.Ferroptosis is implicated in various biological processes and diseases, particularly cancer. Its relationship with cancer is complex, as it can act as a tumor suppressor through mechanisms such as p53-mediated LPO product accumulation.10 Although this tumor suppressor gene is often mutated in cancer, factors like ACSL4 and HIF1α, which regulate lipid metabolism and hypoxic responses, can increase cancer cell sensitivity to ferroptosis and promote metastasis.11 While ferroptosis has shown promise in limiting tumor growth and enhancing cancer therapies, recent studies suggest it may also have tumor-promoting roles by influencing the tumor immune microenvironment. Ferroptosis plays a dual role in cancer biology, acting as a tumor-suppressing mechanism and a potential promoter of tumor progression through its effects on the TME.11
Many recent studies have emphasized that ferroptosis function as a context-dependent immunogenic cell death that can either enhance immune checkpoint inhibitor efficacy by releasing damage-associated molecular patterns or suppress antitumor immunity through immune cell ferroptotic vulnerability and inflammatory lipid byproducts.12,13 These reviews focused on divers perspectives including its molecular regulation, involvement in cancer and inflammatory disease, and broader role in immunity and metabolism. More recent articles have further explored ferroptosis in context of CD8+ T-cell functional, lipid metabolism, ACSL regulation and redox-immune cross talk.14–16 In contrast to these studies, which largely focus on either the intrinsic mechanism of ferroptosis or its general immunological consequences, the present review carefully center on ferroptosis-induced immunomodulatory signal within the tumor microenvironment, including postglandin E2, lipid peroxidation products, and damage-associated molecular patterns, and how these signal actively rewire immune surveillance and therapeutic respossivness. By integrating ferroptosis biology with tumor-associated immune suppression and immunotherapy outcomes, this review aims to provide a therapy-oriented and microenvironment-focused synthesis that complements, rather than duplicates existing literature.
2 Ferroptosis: mechanisms, metabolic regulation, and biomedical implications
2.1 Molecular pathways of ferroptosis
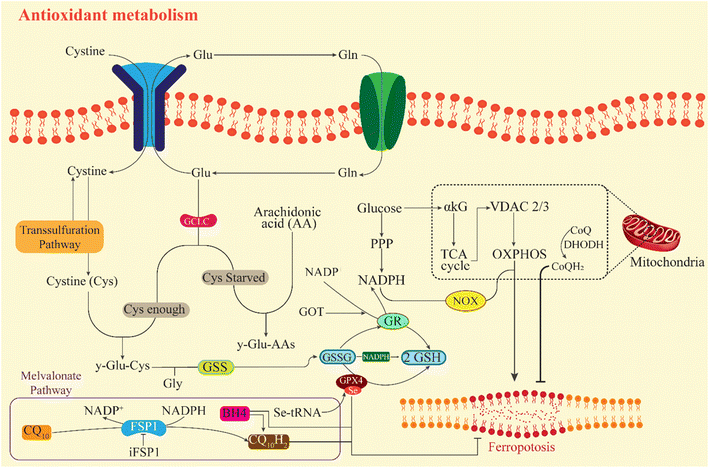
Ferroptosis, first defined in 2012, is a unique form of programmed cell death driven by iron-dependent lipid peroxidation (LPO) and is mechanistically distinct from apoptosis, necrosis, and autophagy.17 It is characterized by specific mitochondrial alterations, including reduced volume, condensed membrane, loss or absence of cristae, and ruptured outer membranes.18 The process is primarily mediated by iron accumulation, which promotes excessive production of ROS through the Fenton reaction and enhances lipoxygenase activity. Additionally, the depletion of intercellular glutathione disrupts the function of glutathione peroxidase 4 (GPX4), a critical antioxidant enzyme.19 This inactivation leads to uncontrolled free radical activity, resulting in cell membrane rupture and the execution of ferroptosis. This ferroptotic process is primarily driven by lipid peroxidation triggered by an accumulation of reactive oxygen species (ROS) that overwhelms the antioxidative defense provided by glutathione (GSH) and GPX4.20 Iron, lipids, and ROS are essential for maintaining cellular function and homeostasis, but their critical roles come with inherent risks. While these factors are indispensable for sustaining normal physiological processes, their dysregulation can lead to severe consequences, including pyroptotic cell death.21 This duality underscores their role as a double-edged sword in cellular metabolism. Central to this imbalance is the formation of ROS, which begins with the conversion of molecular oxygen (O2) into superoxide radicals (O2−) through enzymatic activity involving NADPH oxidases (NOX), NADPH-cytochrome P450 reductase (POR), or the mitochondrial electron transport chain.22 Superoxide (O2−) is subsequently converted into hydrogen peroxide by the enzyme superoxide dismutase (SOD). In the presence of ferrous iron (Fe2+), (H2O2) undergoes the Fenton reaction, generating highly reactive hydroxyl radicals (HO). These radicals then abstract hydrogen atoms from polyunsaturated fatty acids (PUFAs), initiating the process of lipid peroxidation.23 Lipid peroxidation progresses through a cyclical chain reaction and its termination. Hydroxyl radicals (HO) or lipid radicals (RO) catalyze the conversion of lipids (R) into lipid radicals (RO), which rapidly react with oxygen to form lipid peroxide radicals (ROO).24 These ROO generate new RO and lipid hydroperoxides (ROOH) in positive feedback. The antioxidant enzyme GPX4 mitigates this reaction by reducing ROOH to inert lipid molecules (R), thereby terminating the peroxidation cycle (Fig. 1).25 Ferroptosis occurs when lipid peroxidation reaches a critical level, lipid peroxide radicals, ROO, decompose to form malondialdehyde, MDA, and 4-hydroxy-2-nonenal (4-HNE), which subsequently create covalent adducts with macromolecules, including proteins, DNA, and lipids.26 These adducts can inactivate or cross-link proteins, destabilizing cellular structures and promoting cell membrane rupture, ultimately triggering ferroptosis.Furthermore, recent experimental in vivo and in vitro studies provided evidence that FSP1/CoQ10 axis functions as a metabolically regulated defense mechanism against ferroptosis that work independently of GPX4, leading to the removal of lipid peroxides at the plasma membrane. In an experimental analysis of sepsis-related acute kidney injury, ferroptosis was strongly increased due to suppression of FSP1/CoQ10 pathways, causing lower level FSP1, loss of cellular CoQ10 and buildup of ferrous iron and lipid peroxidation products.27 Activating this pathways, such as with dexmedetomidine treatment or increased Nrf2 expression, restore FSP1 and CoQ10 level, raises glutathione, lowers oxidative stress, and clearly reduces ferroptotic damage.27 In contrast, blocking Nrf2 removes these protective effects, proving that the FSP1/CoQ10 system directly suppress ferroptosis. Further support for the role of FSP1/CoQ10 in ferroptosis defense comes from a recent study of vanadium-induced liver injury.28 The study demonstrated that disruption of glucose metabolism weakens the NADH-dependent FSP1/CoQ10 pathways, which is required for the reduction of ubiquinone to ubiquinol, thereby enhancing ferroptosis process. Specifically, a reduced NADH/NAD+ ratio limits FSP1 activity which decreases CoQ10 and ubiquinol levels, causes failure of peroxide detoxification, promotes excessive Fe2+ accumulation and further enhances ferroptosis.28 Such literature data reinforce the impairment of FSP1/CoQ10 axis with the promotion of ferroptotic cell death by allowing uncontrolled lipid peroxidation at cellular membrane and associated inflammatory responses.27,28
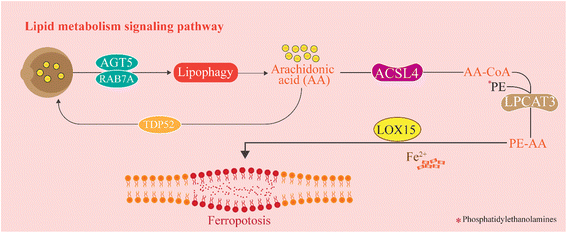
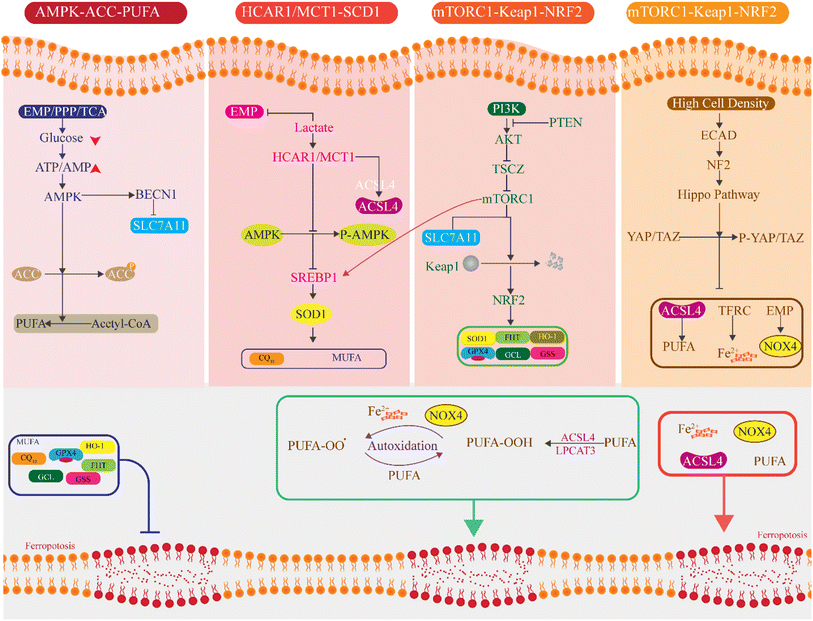
Lipid biosynthesis is essential for cellular survival, whereas lipid peroxidation is a critical ferroptosis biomarker. The process of lipids peroxidation and ferroptosis are facilitated by key enzymes, including Acyl-CoA Synthetase Long-Chain Family Member 4 (ACSL4), Lys phosphatidylcholine acetyltransferase 3 (LPCAT3) and Arachidonic Acid 15-Lipoxygenase (ALOX15).29 Quantitative lipidomic analyses have identified Arachidonic Acid and Adrenic Acid (ADA), both integral components of phosphatidylethanolamines (PEs), as pivotal substrates driving ferroptosis.30 Recent findings reveal that enhancing TPD52-mediated lipid storage or inhibiting lipid degradation pathways reliant on ATG5 and RAB7A can mitigate RSL3-induced lipid peroxidation and ferroptosis, emphasizing the critical balance between lipid synthesis, storage, and degradation in ferroptosis regulation.31 Several factors, including the Hippo signaling pathway, radiotherapy, and a lactic acid-enriched microenvironment, influence the expression of ACSL4. LPCAT3 is pivotal in lipid metabolism, primarily localized in the endoplasmic reticulum and expressed in metabolic tissues such as the liver, adipose tissue, and pancreas.32 LPCAT3 catalyzes the incorporation of the acyl group into phosphatidylcholine (PC) to produce PE-CoA, which is further converted into PE-AA. A deficiency in LPCAT3 disrupts this process, reducing AA levels in cellular membranes and accumulating cytoplasmic lipid droplets. This deficiency is associated with mice's lethality and intestinal epithelial damage, underscoring LPCAT3's critical role in cell viability.33 Interestingly, the saturation levels of ether lipids have merged as determinants of ferroptosis susceptibility. During the differentiation of cardiomyocytes and neurons, elevated polyunsaturated ether phospholipids (PUFA-ePLs) correlate with ferroptosis sensitivity.34 The selective downregulation of highly unsaturated PUFA-ePL levels may act as a cellular defense mechanism against ferroptosis, offering a promising avenue for therapeutic intervention.
Lipoxygenases (LOXs) are pivotal in oxidizing polyunsaturated fatty acids (PUFAs) into their hydroperoxide derivatives. In mammalian cells, linoleic acid and AA are the most abundant PUFA substrates for LOXs. PUFAs are particularly susceptible to peroxidation during ferroptosis.35 Pre-treatment of cells with deuterium-labeled PUFAs, DPUFAs, at the peroxidation site prevents PUFA oxidation and blocks ferroptosis. Additionally, acetyl-coenzyme A carboxylase (ACC) a key enzyme in fatty acid synthesis, facilitates the production of PUFAs from acyl-CoA.36 PE-AA, a polyunsaturated fatty acid, undergoes catalytic oxidation by LOX15 and Fe2+ to produce oxidized phospholipids (ox-PE), contributing to ferroptosis.37 Mutations in iPLA2β, a critical protein involved in phospholipid remodeling, impart the ability to hydrolyze oxidized phospholipids such as ox-PE, leading to the accumulation of oxidized lipids in dopaminergic neurons.38 This underscores the role of lipid metabolism in maintaining cellular homeostasis and the potential consequences of its dysregulation. Evidence suggests that antioxidant metabolism, iron homeostasis, and lipid metabolism collectively play vital roles in regulating ferroptosis and cellular hypertrophy. Imbalances in any of these pathways can induce ferroptosis.39 However, the precise mechanisms through which lipid peroxidation differentially regulates ferroptotic and non-ferroptotic cell death remain incomplete and warrant further investigation (Fig. 2).
2.2 Iron metabolism in ferroptosis
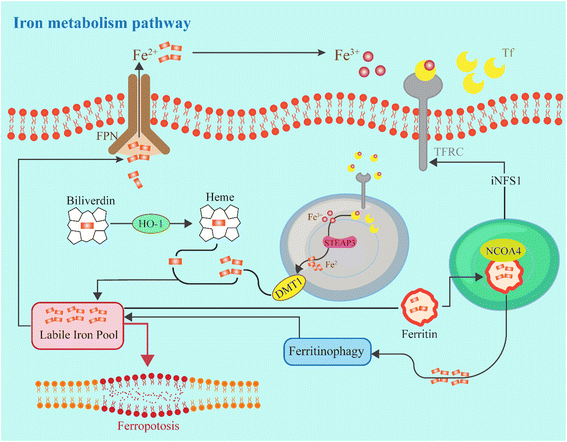
Iron is classified as one of the “bio elements” that can produce reactive oxygen species (ROS) in an oxygen-rich environment through a process known as the Fenton reaction. In the bloodstream, iron in the form of Fe3+ binds to transferrin (TF) to facilitate its transport to cells.40 This transportation is mediated by endocytosis through the transferrin receptor (TFRC). Once the Fe3+ enters the cells, it is reduced to Fe2+ by the action of a metal reductase called Six-transmembrane epithelial antigen of the prostate 3 (STEAP3), which acts on the ferrosomes.41 The release of Fe2+ from ferrosomes into the labile iron pool (LIP) is governed by the divalent metal transporter (DMT1). Recent research has shown that transferrin plays a crucial role in protecting the liver by inhibiting liver damage, fibrosis, and cirrhosis through its regulation of ferroptosis.42 Additionally, the pathway of iron homeostasis, driven by sterol regulatory element binding protein-2 (SREBP2), contributes to cancer progression, drug resistance, and metastasis, highlighting the vital importance of iron homeostasis for the survival of organs.43 Most of the iron in the body is stored in ferritin and heme. Ferritin has two subunits: FTL (light chain) and FTH1 (heavy chain). The nuclear receptor co-activator 4 (NCOA4) interacts directly with FTH1 to form a ferritin complex that targets lysosomes for a process called “ferritinophagy”.44 This process promotes ferritin degradation and increases intracellular free iron, especially during periods of iron deficiency. In addition to ferritin, Fe2+ can also be released from heme through the action of heme oxygenase-1 (HO-1), which leads to its accumulation in cardiomyocytes and can induce ferroptosis.45 NRF2 and BAY may activate this process. Ferroportin (FPN1) is the sole cellular efflux channel for iron, and its degradation occurs when it binds to hepcidin secreted by the liver, resulting in reduced cellular iron output.46 However, many cells do not possess effective mechanisms for iron export, which can lead to increased LIP levels when excess iron exceeds the storage capacity. Consequently, regardless of the pathway, an elevated level of intracellular free iron enhances the induction of ferroptosis driven by LIP levels (Fig. 3).2.3 Comparison of ferroptosis with other cell death modalities (apoptosis, necroptosis, pyroptosis)
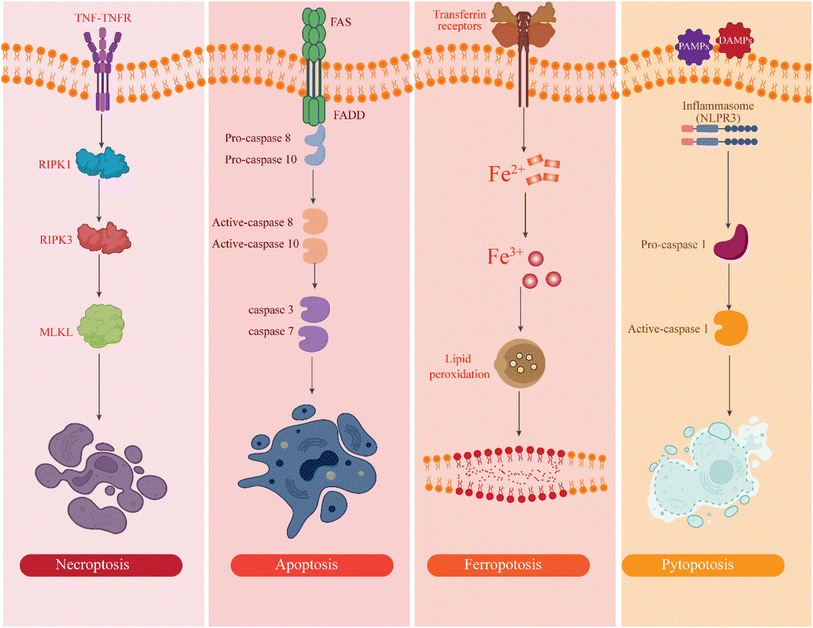
Programmed cell death (PCD) plays a crucial role in various physiological processes, including embryogenesis, tissue homeostasis, immune responses, and the prevention of hyperproliferative diseases such as cancer.47 Several forms of PCD have been identified, including apoptosis, pyroptosis, parthanatos, necroptosis, and ferroptosis, each with unique mechanisms and characteristics. Apoptosis is a highly regulated form of cell death during development, tissue homeostasis, and response to infection or pathogenesis. It is an evolutionarily conserved process, and its regulation is primarily governed by the activation of caspases, a family of proteases that orchestrate cellular disassembly.48 Pyroptosis is a form of programmed cell death (PCD) closely associated with inflammation. It is driven by the activation of inflammatory caspases, such as caspase-1 or caspase-11, which cleave gastrin proteins to form membrane pores.49 This pore formation leads to cell swelling, membrane rupture, and the release of intracellular contents, including pro-inflammatory cytokines such as IL-1β and IL-18.50 Necroptosis is a form of programmed necrosis regulated by receptor-interacting protein kinases (RIPKs), specifically RIP1 and RIP3.51 It involves a complex signaling cascade that leads to mitochondrial dysfunction, ATP depletion, calcium overload, and the opening of mitochondrial permeability transition pores. These events culminate in increased cell volume, organelle swelling, and plasma membrane rupture, resulting in cellular contents' leakage and subsequent inflammation.52 Ferroptosis is a unique form of programmed cell death (PCD) characterized by the accumulation of lipid peroxides, particularly those derived from polyunsaturated fatty acids (PUFAs) and cellular membranes.53 It is iron-dependent and is driven by an imbalance between the production of reactive oxygen species (ROS) and the cell's antioxidant defense systems.Unlike apoptosis and necroptosis, ferroptosis does not involve caspase activation or receptor-interacting protein kinase signaling.54 Instead, ferroptosis is induced by excessive ROS accumulation caused by iron overload, which leads to lipid peroxidation and loss of membrane integrity.55 This unique form of cell death is distinguished from apoptosis, necroptosis, and pyroptosis by its specific biochemical markers, including increased lipid peroxidation and altered mitochondrial morphology. Unlike apoptosis, which does not trigger inflammation, ferroptosis can also cause inflammatory responses due to releasing oxidized lipids and other cellular debris.56 Ferroptosis is distinct from apoptosis, necroptosis, and pyroptosis regarding morphological features. Cells undergoing ferroptosis do not display the classical characteristics of apoptosis, such as chromatin condensation and membrane blebbing, or necroptosis, such as swelling and rupture of organelles and plasma membrane.57 Instead, cells undergoing ferroptosis show damaged mitochondria and disrupted membrane integrity without the characteristic features of other forms of cell death. Ferroptosis is primarily regulated by iron-dependent lipid peroxidation, a process that involves the generation of reactive oxygen species (ROS) and breaking redox homeostasis.58 In contrast, apoptosis involves caspase activation, pyroptosis is driven by inflammatory activation and gastrin cleavage, and necroptosis is regulated by RIP1, RIP3, and MLKL signaling.58 Ferroptosis has emerged as a key player in cancer biology due to its potential to target cancer cells with specific metabolic vulnerabilities. Ferroptosis is triggered by compounds such as elastin and RSL3, which induce ROS production and inhibit GPX4 activity, leading to lipid peroxidation and cell death.59 Cancer cells often exhibit altered redox states and iron metabolism, making them susceptible to ferroptosis. Like other regulated cell death modalities, ferrotopsis has been increasingly classified as a stress-induced immunogenic cell death, characterized by lipid peroxidation-driven membrane damage that promotes antigen release and immune activation.16,60,61 However, recent comparative analysis of non-apoptotic cell death pathways now distinguish ferroptosis from apoptosis and pyroptosis by its reliance on lipid-derived immunomodulators rather than proteinaceous DAMPs, resulting in qualitatively different immune activation patterns that are highly dependent on local lipid metabolism62 (Fig. 4).
2.4 Natural ferroptosis inducer (fin): key regulatory genes
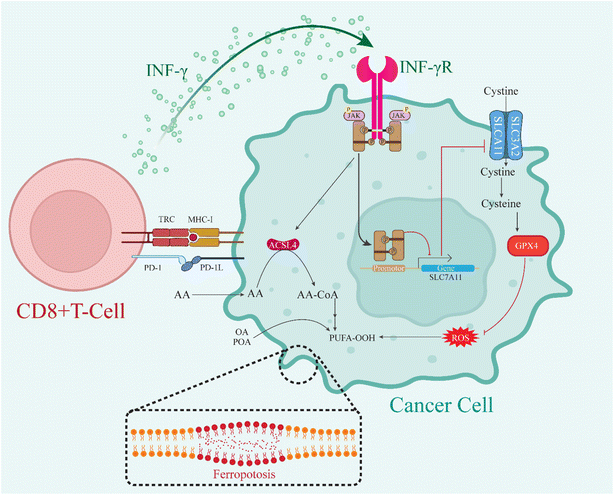
A recent study published in Cancer Cell identified a novel natural ferroptosis inducer (FIN) in the form of CD8+ T cell-derived interferon-γ (IFNγ) in combination with polyunsaturated fatty acids (PUFAs).63 This combination triggers tumor ferroptosis and enhances anti-tumor immunity in an ACSL4-dependent manner. The study suggests that ACSL4, which catalyzes the ligation of PUFAs to coenzyme A to form PUFA-CoAs, is essential for ferroptosis.64 Ferroptosis can be inhibited by disrupting enzymes like ACSL4 involved in PUFA-PL biosynthesis.65 One defense mechanism is the GPX4-dependent antioxidant system, where GPX4 uses glutathione to detoxify lipid peroxides and suppress ferroptosis. Additionally, the system Xc, which consists of SLC7A11 and SLC3A2 subunits, imports cystine for glutathione synthesis and ferroptosis defense.66 Interestingly, natural ferroptosis inducers such as IFNγ secreted by CD8+ T cells can sensitize tumor cells to ferroptosis, mainly by suppressing the expression of SLC7A11. However, IFNγ alone does not trigger potent ferroptosis. The authors of this study found that combining IFNγ with arachidonic acid (AA), a 20-carbon PUFA, leads to potent ferroptosis in various cancer cell lines in an ACSL4-dependent manner.67 This synergy occurs because IFNγ activates the JAK-STAT1-IRF1 signaling axis, upregulating ACSL4 expression. Lipidomic analysis revealed that AA, in combination with IFNγ, increases ACSL4 levels and promotes the incorporation of AA into phospholipids (PLs).68 This increase in PUFA-PL synthesis and decreased SLC7A11 expression due to IFNγ treatment significantly induce ferroptosis in tumor cells. In addition to AA, the study found that monounsaturated fatty acids (MUFAs) such as oleic acid (C18:1) also synergize with IFNγ to induce ferroptosis in an ACSL4-dependent manner.69 This effect was unique, as MUFAs generally suppressed ferroptosis induced by synthetic fins like RSL3. However, the role of oleic acid in promoting ferroptosis, particularly in the context of IFNγ treatment, remains unclear and warrants further investigation.70 Regarding immunological findings, the study showed that ACSL4 deletion did not impact tumor growth in immunodeficient mice but significantly promoted tumor growth in immunocompetent mice.63 Moreover, a combination of low-dose AA and anti-PD-L1 treatment more effectively reduced tumor growth than either treatment alone, suggesting that AA treatment and immune checkpoint inhibition may be a promising therapeutic strategy for cancer.71 The study's findings indicate that IFNγ derived from CD8+ T cells in combination with AA represents a novel natural fin that could be harnessed in cancer therapy. The authors also note that future clinical trials should focus on identifying patient populations suitable for this combination therapy, potentially guided by ACSL4 expression.72 ALOX5, ALOX12, ALOX15, ALOX15B, ALOXE3, and POR (cytochrome P450 oxidoreductase) are isoforms of arachidonate lipoxygenase (ALOX) that mediate lipid peroxidation, a critical process leading to ferroptosis (Fig. 4).73 Current immune-focused studies further emphasize that while IFN-γ-driven ferrotopsis operated within a narrow therapeutics windo, as identical IFN-γ-lipid-peroxidation signaling axes simultaneously sensitize tumor cells and destabilize CD8+ T-cell membrane integrity, imposing a previously underappreciated constraint on ferroptosis-based immunotherapy.16,74–772.5 Diagnostic and biomarker implications of ferroptosis
Ferroptosis is implicated in various pathological conditions characterized by iron overload, including cancer, where it plays a dual role in tumor progression. On one hand, ferroptosis induced by small-molecule compounds demonstrates tumor-suppressive effects in preclinical models.78 For example, in a genetically engineered mouse model of pancreatic cancer, treatment with cyst(e) kinase induces ferroptotic morphological changes, including mitochondrial abnormalities and the formation of lipid droplets which may serve as reservoirs for polyunsaturated fatty acids (PUFAs) required for lipid peroxidation.79 Immunohistochemical staining for 4-hydroxynonenal ($-HNE) further confirms elevated lipid peroxidation levels in pancreatic tumors following cyst(e) kinase therapy. On the other hand, ferroptosis can contribute to tumor progression by promoting an immunosuppressive microenvironment by releasing damage-associated molecular patterns (DAMPs) by ferroptosis cells.80 For example, conditional depletion of Gpx4 in the pancreas or administration of a high-iron diet triggers the release of mutant KRASG12D protein and nuclear DNA, stimulating macrophage-driven tumor-promoting response. Moreover, the expression of KRASG12D in macrophags negatively correlates with pancreatic cancer patients' survival outcomes.81 These findings highlight the complex role of ferroptosis in cancer and suggest that monitoring intracellular and extracellular markers of ferroptosis may aid in diagnosing and treating ferroptosis-associated pathologies.PTGS2 (prostaglandin-endoperoxide synthase 2/COX-2) is a key enzyme involved in metabolizing arachidonic acid (AA) into prostaglandins.82 It is the most upregulated gene among oxidative stress-associated genes in ferroptosis, particularly in response to elastin or RSL3. PTGS2 mRNA is a pharmacodynamic marker in ferroptotic tissues in both in vitro and in vivo models, including mouse studies.83 However, PTGS2 does not directly contribute to ferroptotic cell death, as inhibitors like indomethacin fail to impact the ferroptotic process. Despite its widespread use as a biomarker, its role is limited to monitoring lipid peroxidation during ferroptosis. CHAC1 (ChaC glutathione-specific gamma-glutamylcyclotransferase 1) promotes ferroptosis by degrading intracellular glutathione (GSH) into 5-oxoproline and cysteinyl–glycine dipeptide.84 This degradation reduces cellular GSH levels, a critical antioxidant that protects against ferroptosis. CHAC1 is upregulated by activating the endoplasmic reticulum (ER) stress pathway, specifically via the EIF2A-ATF4 axis. It plays a significant role in ferroptosis induced by system Xc− non-inhibitors, such as elastin and artesunate and serves as a reliable pharmacodynamic marker for ferroptosis.85
Ferroptosis is closely linked to dysregulated iron metabolism, and several markers reflect this association. FTH1 (ferritin heavy chain 1) a protein involved in iron storage, is upregulated during ferroptosis to manage cellular iron levels.70 SLC40A1 (solute carrier family 40 member 1) regulates iron export and is similarly elevated in ferroptotic conditions. HMOX1 (Heme oxygenase 1) which degrades heme and releases iron and carbon monoxide, also plays a regulatory role in ferroptosis.86 MT1G (Metallothionein 1G) has also been identified as an NFE2L2 target gene associated with ferroptosis resistance, particularly in sorafenib-induced ferroptosis in hepatocellular carcinoma cells.87 Several markers reflect oxidative stress and antioxidant responses during ferroptosis. NQO1(NAD(P)H quinone dehydrogenase 1) is a detoxifying enzyme that is regulated during oxidative stress, providing protection during ferroptosis.88 TXNRD1 (thioredoxin reductase 1) also contributes to antioxidant defense mechanisms and is consistently elevated during ferroptotic conditions.89 Furthermore, members of the aldo-keto reductase (AKR) family, including AKR1C1, AKR1C2, and AKR1C3, are involved in detoxification and oxidative stress responses, highlighting their role in ferroptosis.90 The detection of intracellular or mitochondrial ferric iron (Fe2+) is critical in ferroptosis studies. Biochemical assay kits are commonly used to quantify iron levels, while Prussian blue staining detects iron deposits in tissues.91 Advanced probes such as FerroFarRed and Phen Green SK offer high sensitivity for monitoring iron levels during ferroptosis, providing valuable tools for studying the role of iron in this regulated cell death process.92
Markers of glutathione metabolism and transport are pivotal in ferroptosis regulation. SLC7A11, (Solute carrier family 7 member 11) is a key component of the system Xc-transporter, which imports cystine for GSH synthesis.93 Its activity is crucial in maintaining cellular redox balance. CBS (cystathionine β-synthase) also plays a role in GSH metabolism, contributing to ferroptosis regulation.94 Additionally, ABCC1/MRP1 (ATP-binding cassette subfamily C member 1) is involved in GSH transport and has been implicated in ferroptosis-related processes.95 NFE2L2 (Nuclear Factor Erythroid 2-like 2/NRF2) is a master regulator of oxidative stress responses, activating genes involved in detoxification, antioxidant defense, and iron metabolism. Key NFE2L2 target genes include FTH1, SLC7A11, HMOX1, and MT1G, which are essential in ferroptosis regulation.96 While NFE2L2 activation protects cells during oxidative stress, excessive upregulation of its target genes may indicate increased oxidative damage during ferroptosis. However, distinguishing its role in ferroptosis from other forms of regulated cell death requires further investigation (Fig. 5).
3 Ferroptosis in cancer
3.1 Ferroptosis-induced tumor promotion
Ferroptosis, a regulated form of cell death driven by iron-dependent lipid peroxidation, plays a crucial role in cancer biology. The regulation of ferroptosis has been implicated in various pathological conditions, including cancer, neurodegenerative disorders, tissue damage, inflammation, and infections.97 The metabolic adaptability of cancer cells provides valuable insights into the mechanisms through which metabolic reprogramming contributes to cancer persistence. In certain instances, this metabolic plasticity is associated with an acquired susceptibility to ferroptosis. Consequently, targeting ferroptosis has emerged as a promising strategy to augment the effectiveness of immunotherapy, underscoring its physiological relevance in tumor suppression and immune surveillance.53 However, the therapeutic potential of ferroptosis in oncology remains underexplored and warrants further investigation. A deeper understanding of the molecular mechanisms underlying ferroptosis and its role in cancer biology is essential to unlock its therapeutic potential.98 In the tumor immune microenvironment, the induction of tumor cell death to reduce tumor burden is central to current ferroptosis-based cancer therapies. However, emerging evidence suggests that ferroptosis in tumor cells can paradoxically promote a tumor-supportive immune microenvironment, contributing to tumor genesis and progression.19Research has demonstrated that ferroptosis induced by a high iron diet or glutathione peroxidase 4 (GPX4) depletion in mice led to the release of 8-hydroxyguanosine (8-OHG), activating the TME173-STING-dependent DNA sensor pathway.99 This activation resulted in macrophage infiltration and promoted pancreatitis and Kras-driven pancreatic carcinogenesis. Furthermore, during the ferroptosis of KRAS pancreatic cancer cells, the KRAS G12D protein was found in exosomes taken up by macrophages via the advanced glycosylation and product-specific receptor (AGER).100 This process facilitated M2 macrophage polarization and tumor growth through the STAT3-dependent fatty acid oxidation pathway. AGER also mediated inflammation and macrophages triggered by high mobility group box 1 (HMGB1),54 a damage-associated molecular pattern molecule released by ferroptotic tumor cells. HMGB1 enhanced pro-tumor inflammation via NF-κB and inflammation pathways.101 Although its role in tumor immunity appears dualistic, treatment with deferiprone, vitamin E, and anti-HMGB1 antibodies reduced tumor-associated macrophages and conferred protection against pancreatic cancer in mouse models.99
3.2 Immunosuppressive effects of ferroptosis in tumor microenvironments
Ferroptosis in tumor cells elevates prostaglandin-endoperoxide synthase 2 (PTGS2) expression, resulting in increased prostaglandin E2 (PGE2) levels.92 Bi et al. found that PGE2 promoted M2 macrophage phenotypes while suppressing M1 macrophage polarization in lipopolysaccharide-treated bone marrow mononuclear cells (BMMCs), thus enhancing the immunosuppressive environment in melanoma models.99 While direct evidence linking ferroptosis tumor cells to the infiltration and polarization of regulatory T cells (Tregs) and myeloid-derived suppressor cells (MDSCs) is limited, studies indicate that these cells resist ferroptosis. Tregs and tumors exhibit minimal lipid peroxidation (LPO) due to up-regulated GPX4 expression, which shields them from ferroptosis.102 Similarly, tumor-infiltrating MDSCs are protected from ferroptosis by expressing high levels of enoyl-acyl carrier protein reductase (ENR).3.3 Ferroptosis-mediated immune cells dysregulation in the tumor microenvironment
Recent studies on immune-centric underscore that ferrotopsis susceptibility varies substantially across immune cell subsets, governed by difference in lipid composition, metabolic demand, and antioxidant capacity.103–106 This heterogeneity critically shapes whether ferrotopsis promotes antitumor immunity or reinforce immune suppression within the tumor microenvironment.107–110 CD8+ T cells play a pivotal role in tumor immune responses, including ferroptosis induction in tumor cells through interferon-gamma (IFN-γ).68 However, the lipid-rich tumor microenvironment (TME) drives CD8+ T cells to increase fatty acid and cholesterol uptake via CD36. Overexpression of CD36 induces LPO and ferroptosis in CD8+ T cells, reducing their anti-tumor activity by decreasing IFN-γ and tumor necrosis factor-alpha (TNF-α) levels, which accelerates tumor progression.111 Similarly, natural killer (NK) cells exposed to the TME display morphological and molecular features consistent with ferroptosis, further compromising innate immune responses. Dendritic cells (DCs), which are vital for antigen presentation and activation of cytotoxic T lymphocytes (CTLs), also impaired by ferroptosis.112 Tumor-associated DCs exhibit elevated lipid levels, which hinder their antigen-presenting ability. The lipid peroxidation byproduct 4-hydroxynonenal (4-HNE) induces endoplasmic reticulum stress, leading to DC dysfunction.113 Additionally, peroxisome proliferator-activated receptor gamma (PPAR-γ) mediates ferroptosis in DCs by regulating lipid metabolism, reducing their ability to secrete TNF and IL-6, express MHC class I, and activate CD8+ T cells.114 Beyond direct effects on immune cells, ferroptosis tumor cells further contribute to immune suppression by promoting the polarization of M2 macrophages, Tregs, and MDSCs, which inhibit the cytotoxic functions of CD8+ T cells and NK cells.115 Damage-associated molecular patterns (DAMPs) like HMGB1 released by ferroptotic cells can induce apoptosis in DCs or convert them to an immunosuppressive phenotype, reducing T-cell activation. Additionally, increased PGE2 levels can inhibit the recruitment and activation of CD103+ DCs, further impairing CTL and NK cell functions (Fig. 6 and 4).1163.4 ROS in ferroptosis
Mitochondria metabolism is the primary source of cellular reactive oxygen species (ROS), playing a significant role in ferroptosis.117 The breakdown of glutamine, a precursor for glutathione (GSH), regulates ferroptosis by supplying α-ketoglutarate (α-KG) through the tricarboxylic acid (TCA) cycle in mitochondria.118 Disruption of the mitochondria TCA cycle, knockout of voltage-dependent anion channel 2 (VDAC2), or inhibition of the electron transport chain (ETC) reduces the accumulation of lipid peroxides. It suppresses ferroptosis, highlighting the involvement of mitochondria ROS dysregulation in this process. Although blocking mitochondria activities strongly inhibits ferroptosis, it can still occur independently of mitochondria under certain conditions.95 For instance, pharmacological inhibition of glutathione peroxidase 4 (GPX4) triggers ferroptosis even without mitochondria function. However, in nutrient-deprived environments, mitochondria metabolism accelerates the depletion of GSH, leading to enhanced lipid ROS production in ferroptosis.119 This suggests that mitochondria have roles upstream and downstream in the ferroptosis pathway. Despite these insights, the precise mechanisms by which mitochondria contribute to ferroptosis remain to be fully elucidated (Fig. 6).4 Immunogenicity and immune suppression in ferroptosis cancer cells
4.1 Immunogenicity of ferroptotic cancer cells
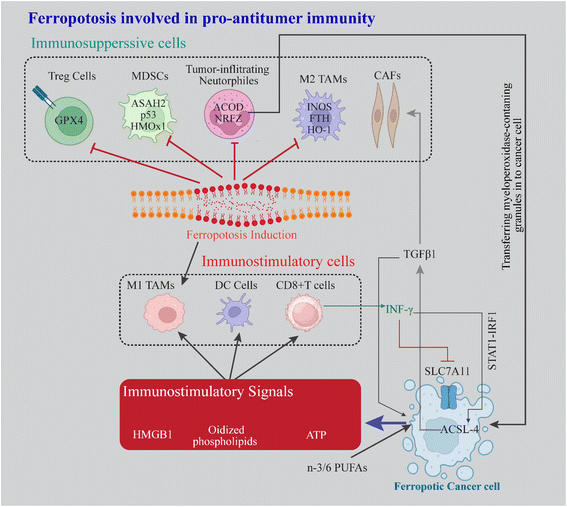
Ferroptotic cancer cells play a dual role in modulating the tumor immune microenvironment, exerting both immunostimulatory and immunosuppressive effects that influence anti-tumor immunity and the efficacy of immunotherapies. Ferroptotic cancer cells release DAMPs such as HMGB1, calreticulin, DNA, and ATP.120 These signals act as “find me” and “eat me” cues, facilitating immune cell recruitment to the site of dying tumor cells. Recently, a membrane-oxidized phospholipid, 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine (POPC), was identified as a critical “eat me” signal that promotes macrophage-mediated phagocytosis of ferroptotic cells.121 The immunogenic properties of ferroptotic cancer cells enhance tumor-specific immune responses. They improve the efficacy of immune checkpoint inhibitors (ICIs) and anti-programmed cell death protein 1 (PD-1)/PD-L1 therapies. Early ferroptotic cancer cells can also stimulate dendritic cell (DC) maturation, triggering vaccination-like effects that further support anti-tumor immunity.122 Current syntheses of ferrotopsis-associated immunogenic stress responses highlight that oxidized phospholipids and ferrotopsis-associated DAMPs can behave as in situ adjuvants that convert immunologically “cold” tumors to “hot” tumors, although this effect is highly dependent on the immune composition of the tumor micro-environment.13,15,65,123–1254.2 Immunosuppressive signals from ferroptotic cancer cells
While ferroptotic cancer cells can stimulate immune responses, they also release factors that suppress anti-tumor immunity.126 One, oxidized DNA damage and TAM polarization: ferroptotic tumor cells release 8-hydroxy-2′-deoxyguanosine (8-OHdG), an oxidized DNA damage product, which activates the STING-dependent DNA sensor pathway and tumor-associated macrophages (TAMs).127 This activation promotes TAM infiltration and M2 polarization, fostering an immunosuppressive environment and enhancing pancreatic tumor genesis. Two, gastrin protein and exosomes: ferroptosis tumor cells release exosomes containing the oncogenic gastrin protein, which TAMs uptake, leading to M2 polarization and promoting tumor growth.128 Three, prostaglandin E2 (PGE2): ferroptotic cancer cells release PGE2, an immunosuppressive molecule that inhibits the cytotoxic activities of natural killer (NK) cells, DCs, and cytotoxic T cells. Four lipid-derived products: ferroptosis cells release various oxidized lipid species, including 15-hydroperoxy eicosatetraenoic acid phosphatidylethanolamine (15-HPETE), 15-HpETE, oxidized, truncated lipids (5-HPETE), and 4-hydroxynonenal (4-HNE).129 These lipid derivatives may impair DC maturation, antigen cross-presentation, and anti-tumor immune responses. High levels of 15-HPETE can also trigger ferroptosis in surrounding epithelial and immune cells, further promoting immune suppression.12 The complex crosstalk between tumor and immune cells creates a delicate balance between immune elimination and evasion.130 This balance directly influences the outcomes of immune checkpoint inhibitors (ICIs) and other immunotherapies. When ferroptosis shifts toward immune suppression, it may diminish the efficacy of ICIs and lead to acquired immune tolerance.131Ferroptosis-based crosstalk in the tumor microenvironment highlights the importance of understanding the dual roles of ferroptosis and immunotherapy.132 While ferroptosis inducers can enhance tumor cell death, they may also exacerbate immune suppression.133 Similarly, ferroptosis inhibitors may improve T cell survival but could inadvertently protect tumor cells. Further research is needed to explore the context-dependent effects of ferroptosis modulation on anti-tumor immunity and to optimize strategies for integrating ferroptosis-targeted therapies with immunotherapy.134 Understanding the complex interactions between ferroptosis, immune cells, and tumor cells will be crucial for developing effective combination therapies to overcome immune evasion and promote durable anti-tumor responses.
5 Ferroptosis and T cell-mediated antitumor immunity
5.1 GPX4 and T cell ferroptosis
Glutathione peroxidase 4 (GPX4) is a key regulator of ferroptosis in T cells. Previous studies demonstrated that T cells lacking GPX4 rapidly accumulated LPO after activation, leading to ferroptosis.135 This resulted in impaired expansion and an inability to prevent infection in an in vivo infection model. However, this study did not examine the impact of GPX4 deletion on tumor-infiltrating T cells. Recent findings revealed significant LPO in CD8+ T cells derived from tumors, identifying ferroptosis as a metabolic vulnerability of tumor-specific CD8+ T cells.136 In co-culture experiments, activated CD8+ T cells were more sensitive to GPX4 inhibitors than cancer cells. GPX4 inhibition reduced the specific killing rates and numbers of CD8+ T cells without affecting the survival of cancer cells. Similarly, conventional CD4+ T cells exhibited comparable sensitivity to GPX4 inhibitors upon activation.1375.2 Mechanisms of ferroptosis in T cells
Several factors influence the susceptibility of activated CD8+ T cells to ferroptosis. Acyl-coenzyme A synthetase long-chain family member 4 (ACSL4) has been identified as a critical determinant of ferroptosis sensitivity. ACSL4 deficiency in CD8+ T cells reduced ferroptosis induced by GPX4 inhibitors.138 Fatty acid translocase CD36, which mediates fatty acid uptake, was also shown to play a role. CD36-expressing CD8+ T cells underwent ferroptosis in the lipid-rich TME, exhibiting reduced interferon-gamma (IFN-γ) production and impaired cytotoxic functions.68 CD36 expression was essential for fatty acid or oxidized lipid-induced ferroptosis, which impaired the anti-tumor activity in these T cells (Fig. 4). Overexpression of GPX4 in CD8+ T cells protected them from ferroptosis and restored cytotoxic cytokine production in vivo. GPX4-overexpressing CD8+ T cells infiltrated tumors in more significant numbers, resulting in enhanced tumor control.1395.3 Ferroptosis modulation in T cell-mediated anti-tumor immunity
The effects of ferroptosis inducers and inhibitors on T cell-mediated anti-tumor immunity have been explored. GPX4 inhibitors selectively killed CD8+ T cells in vitro, promoting cancer cell survival.140 Conversely, the ferroptosis inhibitor ferrostatin-1 rescued T cell-mediated cytotoxicity against cancer cells. Tumor-bearing mice receiving ferrostatin-1-treated CD8+ T cells exhibited reduced tumor burden and improved survival.141 In contrast, GPX4 inhibitor-treated CD8+ T cells had diminished anti-tumor effects. These findings suggest that ferroptosis inhibitors may enhance anti-tumor immunity, although the data primarily stems from in vitro studies and in vivo models with localized administration.5.4 Conflicting evidence and methodological differences
Contradictory evidence has emerged regarding the relative sensitivity of tumor cells and CD8+ T cells to ferroptosis. One study suggested that tumor cells are more sensitive to ferroptosis than activated CD8+ T cells and that ferroptosis inducers do not impair the anti-tumor activities of CD8+ T cells.142 This study utilized systemically administered inhibitors and cysteine to induce ferroptosis, which may have allowed CD8+ T cells to survive by relying on alternative systems and sources.132 Additionally, systemic administration of the ferroptosis inhibitor prostatin-1 reversed the effects of immunotherapy by preventing IFN-γ-induced ferroptosis in tumor cells, with no direct impact on tumor growth.143 The divergent outcomes highlight the need for further research into the role of ferroptosis inhibitors in various tumor models and their effects on immunotherapy. The sensitivity differences between tumor cells and T cells must be carefully examined to optimize therapeutic strategies that balance tumor suppression and immune cell-protection (Fig. 7).1446 Enhancing cancer treatment through immunotherapy, ferroptosis and radiotherapy
Immunotherapy is a promising cancer treatment strategy that works by stimulating or enhancing the immune response against tumors. CD8+ T cells activated through immunotherapy primarily induce tumor cell death via two key mechanisms:145 the perforin and granzyme pathways, and Fas/FasL interactions. Key molecular pathways, such as Janus kinase (JAK), signal transducer and activator of transcription 1 (STAT1), mediate interferon-gamma (IFN-γ) signaling and play a crucial role in regulating immune responses.146 Recent findings indicate that IFN-γ produced by CD8+ T cells enhances STAT1 binding to the transcription start site of SLC7A11, inhibiting its transcription.65 This downregulation of SLC7A11 by STAT1 is critical because a lack of STAT1 in tumor cells negates the IFN-γ-mediated suppression of SLC7A11, reversing lipid peroxidation and ferroptosis induced by RSL3. Notably, ferroptosis inducers like Erastin and RSL3 selectively promote ferroptosis in cancer cells, sparing human and mouse CD8+ T cells.147 This differential susceptibility suggests distinct ferroptosis pathways in tumor cells and T cells. Combining immune checkpoint inhibitors with ferroptosis inducers has been shown to elicit robust immune responses and promote tumor ferroptosis. Several recent reviews on immunogenic cell stress highlights that ferrotopsis induction alone in not sufficient for durable immune activation and must be integrated with modalities that preserve immune effector cell viability, optimize antigen presentation, and avoid excessive inflammatory toxicity.13,103,115,148Radiotherapy, historically regarded as a cornerstone of tumor treatment, remains limited by radiation resistance, which often results in tumor recurrence and metastasis.149 To overcome these challenges, combining radiotherapy with specific chemical agents, including ferroptosis inducers, has shown promise in enhancing treatment sensitivity. Research has revealed a cross-talk between ferroptosis and radiotherapy.150 For instance, mutations in the KEAP1 gene induced by radiotherapy upregulate SLC7A11 expression, conferring resistance to both ferroptosis and radiotherapy.151 Conversely, combining radiotherapy with immunotherapy enhances tumor cell susceptibility to ferroptosis, where cytoplasmic radiation rather than nuclear radiation synergizes effectively with ferroptosis inducers by promoting sugar uptake and lipid peroxidation instead of DNA damage.2
Effector T cells also interact with radiotherapy to enhance tumor clearance by ferroptosis. CD8+ T cells activated by immunotherapy increase lipid peroxidation and ferroptosis through IFN-γ, directly sensitizing tumor cells to radiation.152 Moreover, radiotherapy upregulates the ATM gene, which inhibits SLC7A11 expression, reducing cystine uptake, depleting glutathione, increasing lipid peroxidation, and driving ferroptosis in tumor cells.153 Radiotherapy generates reactive oxygen species (ROS) and decreases ferroptosis markers like 4-HNE. While combining radiotherapy with ferroptosis inducers, such as those targeting SLC7A11 or GPX4, significantly improves efficacy and reverses radiation resistance.154 It is essential to consider that ferroptosis is also associated with adverse radiation-induced effects, such as pulmonary fibrosis and the death of granulocyte-macrophage hematopoietic progenitor cells. This highlights the potential of combining radiotherapy, ferroptosis inducers, and immune checkpoint inhibitors as a novel strategy to overcome radiotherapy resistance.155 However, further research is needed to fully understand the complex interactions between ferroptosis, radiotherapy, and immunotherapy, and to develop effective combination therapies that can overcome radiation resistance and promote durable anti-tumor responses.
7 Impact of ferroptosis on different immune cells
7.1 Macrophages: polarization and ferroptosis interplay
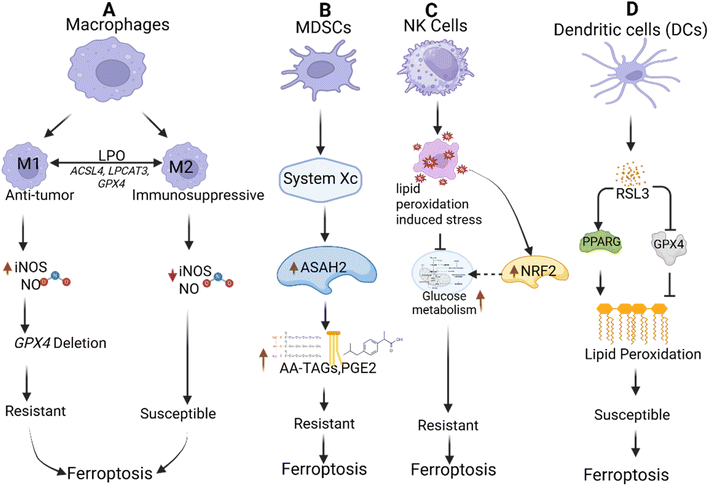
Associated macrophages, tumor-associated macrophages (TAMs) in the tumor microenvironment (TME), play critical roles in cancer progression and immunity.153 Resting macrophages can polarize into two distinct activation states: the M1 phenotype, which exhibits pro-inflammatory and anti-tumor properties, and the M2 phenotype, which is immunosuppressive and promotes tumor growth.156 TAMs in the TME predominantly adopt the M2-like phenotype, suppressing anti-tumor immunity. Although M1 and M2 macrophages have similar levels of lipid peroxidation (LPO) and expression of ferroptosis-related proteins, such as ACSL4, lysophosphatidylcholine acyltransferase 3 (LPCAT3), and GPX4, M1 macrophages are more resistant to ferroptosis when GPX4 is deleted.157 This resistance is attributed to high levels of inducible nitric oxide synthase (iNOS) and nitric oxide (NO) production in M1 macrophages, which can react with lipid radicals and lipid peroxidation intermediates, substituting for GPX4 as an anti-ferroptotic defense. In contrast, M2 and resting macrophages have lower iNOS expression and NO production, making them more susceptible to ferroptosis.158 In vivo studies have confirmed the role of iNOS/NO in macrophage survival in the TME. Deleting iNOS in bone marrow-derived macrophages significantly reduced the population of M1 TAMs. Still, it did not affect M2 TAMs, suggesting that M1 TAMs lacking iNOS cannot adapt to the pro-ferroptosis conditions of the TME, especially when phagocytosis of ferroptotic cells was accumulated LPO.159 Targeting TAMs to eliminate the M2 phenotype or repolarize them into M1 macrophages has emerged as a promising approach in cancer immunotherapy.160 For example, ferroptosis inducers can selectively inhibit the survival of M2 TAMs without affecting M1 TAMs, and pro-ferroptotic nanoparticles have been shown to efficiently repolarize M2 TAMs into the anti-tumor M1 phenotype.161 However, challenges remain in leveraging ferroptosis inducers therapeutically. GPX4 is crucial for macrophages to initiate innate immune responses, and the impact of ferroptosis inducers on the viability and functions of macrophages in the TME must be thoroughly investigated.162 Future studies should focus on understanding the effects of ferroptosis-inducing strategies on macrophage populations and their potential to reverse the immunosuppressive TME while enhancing anti-tumor immunity.7.2 MDSCs exhibit resistance to ferroptosis
Myeloid-derived suppressor cells (MDSCs), which are present in most tumors, play a significant role in suppressing the immune response within the tumor microenvironment (TME).9 Research indicates that tumor-infiltrating MDSCs are protected from ferroptosis due to their high expression of system Xc- and the enzyme neutral ceramidase and acid ceramidase, also known as AC or ASAH2. ASAH2 destabilizes the p53 protein, which is often a key regulator of cell death, thereby enhancing the resistance of MDSCs to ferroptosis.163 In addition, liquid chromatography-mass spectrometry (LC-MS) analysis has revealed that a specific subset of MDSCs in the TME164 selectively accumulates arachidonate (AA)-esterified triglycerides, AA-TAGs, oxidized AA-TAGs, and prostaglandin E2 (PGE2), which is a primary oxidative metabolite of free AA.165 Interestingly, these MDSCs do not accumulate AA-phospholipids (AA-PLs) or their associated lipid peroxidation products. This finding suggests that the metabolic pathway converting AA into AA-PLs and initiating lipid peroxidation may be suppressed in tumor-infiltrating MDSCs, further contributing to their ability to resist ferroptosis.166 These insights underline how metabolic adaptations within MDSCs enable them to evade ferroptosis and maintain their immunosuppressive function in the TME, highlighting potential therapeutic targets to disrupt this resistance and restore anti-tumor immune activity.167 The discovery of these metabolic adaptations in MDSCs provides a new perspective on the mechanisms underlying their immunosuppressive functions. It suggests that targeting these pathways may be a promising strategy for enhancing anti-tumor immunity.7.3 Neutrophils: role of inflammation and ferroptosis
Natural killer (NK) cells play a pivotal role in anti-tumor immunity, and impairments in their function are associated with increased tumorogenesis and tumor growth.168 Recent research has revealed that tumor-associated NK cells exhibit elevated expression of proteins related to ferroptosis, lipid peroxidation, and oxidative damage alongside morphological features characteristic of ferroptotic cells.169 This lipid peroxidation-induced oxidative stress suppresses glucose metabolism in NK cells, leading to their dysfunction within the tumor microenvironment (TME). Activation of nuclear factor E2-related factor 2 (NRF2), a key regulator of ferroptosis resistance, restores glucose metabolism and enhances the anti-tumor activity of NK cells in vivo.170 However, the impact of ferroptosis inducers or inhibitors on the survival and function of tumor-associated NK cells remains unexplored.7.4 Dendritic cells (DCs)
Dendritic cells (DCs), as professional antigen-presenting cells, are crucial for activating naive T cells and maintaining T cell-dependent immunity.171 Although direct evidence is limited, studies suggest that ferroptosis may influence DC function. Tumor-associated DCs often display impaired antigen-processing capabilities due to lipid accumulation, which makes them more vulnerable to ferroptosis.172 Increased levels of 4-hydroxynonenal (4-HNE) protein adducts, a lipid peroxidation byproduct, have been observed in tumor-associated DCs. This accumulation may activate X-box binding protein 1 (XBP1), contributing to DC dysfunction.173 During their maturation, bone marrow-derived naive DCs exhibit high levels of lipoxygenase (ELOX12/15), which generates oxidized phospholipids that can hinder their maturation process.174 Consequently, targeting ferroptosis regulators like GPX4 or system Xc-could impair the maturation and normal functioning of DCs in the TME.Tumor-infiltrating B cells primarily originate from memory B cells and exhibit diverse subpopulations with contrasting roles in tumor immunity.175 Although no studies have directly investigated the relationship between ferroptosis and tumor-infiltrating B cells, existing research highlights the importance of GPX4 and B cell survival.176 Specifically, GPX4 is critical for the survival of B1 and marginal zone (MZ) B cells, which express higher levels of CD33 and uptake more lipids than follicular B2 cells. Deleting GPX4 induces lipid peroxidation and ferroptosis in B1 and MZ B cells but not in follicular B2 cells.177 This suggests that pro-ferroptotic stimulation might impact the survival of specific B cell subsets and their roles in tumor immunity, offering a potential area for future research.
In summary, ferroptosis exerts a dual influence on tumor immunity. On the one hand, inducing ferroptosis can compromise the survival and functionality of anti-tumor immune cells, including CD8+ T cells, NK cells, and DCs, potentially weakening immune responses against tumors.178 On the other hand, immunosuppressive cells, such as M2 macrophages, TAMs, and regulatory T cells (Tregs), rely on GPX4 and other factors to evade ferroptosis and sustain their suppressive functions. Inducing ferroptosis in these cells may selectively disrupt their activity, reversing their immunosuppressive effects and promoting anti-tumor immunity.136 Further research is needed to refine strategies that harness ferroptosis modulation to target tumor-promoting cells while preserving anti-tumor immune function selectively (Fig. 8).
8 Ferroptosis and antitumor treatment
Ferroptosis, a regulated form of cell death driven by the accumulation of reactive oxygen species (ROS), has emerged as a promising therapeutic target for addressing cancer, particularly in overcoming resistance to conventional chemotherapy.156 Studies have demonstrated the critical role of ferroptosis in treating various cancers, including non-small cell lung cancer, breast cancer, pancreatic cancer, hepatocellular carcinoma (HCC), glioblastoma, colorectal cancer (CRC), ovarian cancer, and more.179 Ferroptosis-inducing strategies primarily target three mechanisms: inhibiting the Xc-glutathione-GPX4 axis to increase ROS accumulation, regulating the p62-Keap1-NRF2 pathway to enhance antioxidant responses, and activating iron metabolism pathways to trigger ferroptosis through lysosomal and transferrin activity.180In lung cancer, drugs like elastin and sulfasalazine inhibit the Xc-system to reduce cystine uptake and increase ROS levels, while cisplatin and dihydroartemisinin regulate iron homeostasis to enhance ferroptosis.181 Combination therapies, such as elastin with cisplatin or EGFR-TKIs, show potential for improved patient outcomes. Glioblastoma, a highly aggressive brain tumor, responds to sulfasalazine, which induces ferroptosis by inhibiting cystine uptake, and temozolomide, which activates the NRF2-ATF4 pathway to regulate antioxidant defenses.31 Aspirin and sorafenib, when combined, further enhance ferroptosis by depleting GSH and increasing ROS levels, even in drug-resistant glioblastoma cells. Furthermore, the HCC, a common and rapidly increasing primary liver cancer, is exceptionally responsive to ferroptosis-inducing drugs, like sorafenib, which modulates iron metabolism but faces resistance due to intracellular iron depletion by deferoxamine.182 Targeting metallothionein 1G or retinoblastoma protein loss enhances ferroptosis-inducing effects. In colorectal cancer (CRC), sulfasalazine and RSL3 promote ROS accumulation, sensitizing cancer cells to cisplatin.183 Pancreatic cancer, characterized by high mortality and chemoresistance, responds to sulfasalazine, artesunate, and combination therapies, like sulfasalazine with piperyl, which significantly increase ROS and ferroptosis.182 Similarly, ovarian cancer cells show ferroptosis sensitivity due to altered transferrin receptor and iron efflux pump expression, making drugs like artesunate effective.184 Similarly, breast cancer therapies targeting CDO1 overexpression and ROS accumulation, including small molecule drugs and combinations, demonstrate promise for treatment-resistant cases.185 Many traditional drugs, such as sulfasalazine, initially used for colitis and arthritis, have been repurposed to target ferroptosis pathways, highlighting the expanding potential of these therapies. Additionally, nanomedicines are emerging as precision tools for inducing ferroptosis, though their clinical applications remain limited.186
Overall, ferroptosis-targeting therapies offer a novel approach to treating aggressive and resistant cancers. Drug efficacy depends on tumor-specific characteristics, such as iron and ROS dependency, and the expression of ferroptosis regulatory genes.185 By leveraging drug repurposing, combination therapies, and advancements in nanomedicine, researchers aim to develop highly effective and tailored treatments for various malignancies.187 This growing body of research underscores the transformative potential of ferroptosis in oncology, paving the way for innovative clinical applications.
8.1 Targeting tumor metabolism and ferroptosis
Tumor cells rely heavily on altered metabolic processes to sustain their rapid growth and survival. One hallmark of cancer cell metabolism is the Warburg effect, where tumor cells exhibit increased glucose uptake and metabolic activity, making them highly dependent on antioxidant mechanisms.188 This dependency renders them particularly vulnerable to oxidative stress. Tumor cells also display glucose and glutamine addiction, consuming these nutrients at much higher rates than normal cells.189 Targeting these metabolic dependencies offers a promising strategy for selective cancer treatment by disrupting their energy supply and redox balance.190 Cancer cells with high SLC7A11 expression support cystine uptake and redox homeostasis and are particularly sensitive to metabolic stress. These cells depend on the glucose pentose phosphate pathway (glucose PPP).190 Limiting glucose availability through glucose transporter inhibitors depletes NADPH, disrupts the redox system, and leads to intracellular cystine accumulation, ultimately triggering ferroptosis. Similarly, glutamine-starved cancer cells with high SLC7A11 expression become more susceptible to oxidative stress and ferroptosis.191 Similarly, another metabolic vulnerability lies in aspartate metabolism. Aspartic acid undergoes transamination with GOT1 (aspartate aminotransferase) to produce oxaloacetate, which participates in reactions that generate NADPH, essential for maintaining redox balance during cancer cell proliferation.192 Knockdown of GOT1 disrupts mitochondrial oxidative phosphorylation, prevents mitochondrial metabolism, and increases the labile iron pool in cancer cells, making them more sensitive to ferroptosis. This disruption induces ferroptosis by impairing mitochondrial function and redox stability.193Hypoxia within the tumor microenvironment exacerbates metabolic stress, increasing ROS levels and contributing to T-cell dysfunction and exhaustion.194 Reducing ROS in T cells and alleviating tumor hypoxia can restore T cell function and synergize with immunotherapy to combat cancer effectively. For instance, the combination of tryptophan depletion and immune checkpoint blockade has enhanced T cell-mediated anti-tumor immunity while simultaneously inducing ferroptosis in tumor cells.163 In summary, targeting the unique metabolic features of cancer cells provides multiple therapeutic avenues, including: (1) Blocking SLC7A11 activity to inhibit cysteine absorption, induce lipid peroxidation, and trigger ferroptosis.195 (2) Inhibiting glucose uptake in SLC7A11-expressing cancer cells, leading to nutrient depletion, NADPH loss, and ferroptosis. (3) Targeting glutamine addiction in cancer cells by using glutaminase inhibitors to block their growth and survival.196 By disrupting critical metabolic pathways, these strategies offer promising approaches for selective cancer therapy, especially when combined with immunotherapy and other ferroptosis-inducing agents.
8.2 Addressing drug resistance in cancer therapy through ferroptosis and differentiation plasticity
Drug resistance and cancer through ferroptosis and differentiation plasticity. Drug resistance and cancer vary significantly across different cell lines and are influenced by their growth and differentiation states.197 For example, melanoma, which can be divided into four subtypes, shows varying sensitivities to ferroptosis induction. Targeting the differentiation plasticity of these subtypes offers a promising approach to improving treatment efficacy, particularly in combination with immunotherapy.198 Studies have shown that tumor necrosis factor-alpha (TNF-α) and interferon-gamma (IFN-γ) induce dedifferentiation in melanoma cells, rendering them more sensitive to ferroptosis inducers such as elastin or RSL3. In contrast, the melanocyte-like subtypes exhibit the highest resistance to these treatments, highlighting the variability in drug responses based on cell differentiation status.199 A hyper-mesenchymal state observed in many cancer cell lines is closely associated with resistance to various treatment modalities. This treatment-resistant state relies on a lipid peroxidation pathway characterized by increased lipid peroxidation activity and the synthesis of polyunsaturated lipids.200 Selective targeting of epithelial cancer cells in high mesenchymal states can be achieved using two types of compounds. The first includes ferroptosis inducers such as RSL3, ML210, and ML162. The second involves statins, which inhibit 3-hydroxy-3-methylglutaryl-coenzyme A reductase (HMGCR).201 Statins disrupt the mevalonate pathway and co-Q10 synthesis, selectively inducing cell death in high mesenchymal cancer cells. Thus, targeting the differentiation plasticity of cancer cells and modulating their states to improve treatment specificity offers a potential therapeutic avenue.202An iron-mediated lipid peroxidation-dependent cell death mechanism has shown promising associations with chemotherapy and radiotherapy. However, its connection to cancer immunotherapy remains largely speculative. Current evidence is based on limited studies, and prognostic analysis of ferroptosis-related genes is often unreliable due to the multi-functionality of these genes beyond ferroptosis. Despite this, ferroptosis holds potential as a complementary approach when used in combination with other treatment strategies, especially given the limited systemic anti-tumor response rates of single-modality therapies. Considering cancer cells' metabolic and differentiation characteristics, a combination of ferroptosis-based approaches could significantly enhance cancer treatment outcomes. These approaches include targeting differentiation plasticity, leveraging ferroptosis inducers, and combining them with immunotherapy, chemotherapy, and radiotherapy. While further research is needed to understand the role of ferroptosis in immunotherapy fully, its integration into multimodal treatment strategies offers a promising path forward for addressing drug resistance and improving the efficacy of cancer treatments.
9 Future perspectives and challenges
Despite sufficient advances in defining ferroptosis as an iron-dependent, lipid peroxidation-driven form of regulated cell death, its conversion into effective cancer therapies, especially within immune effective complex tumor microenvironments, left as a overwhelming challenge. As described throughout this review, ferroptosis does not work to actively suppress tumor formation but instead functions as an environment based metabolic regulator, capable of changing tumor–immune interactions in both beneficial and destructive manner. The fate of ferroptosis-based cancer therapy therefore depends not on bulk induction of oxidative cell death, but relies on governing lipid peroxidation signals at exact moment and specific site of the tumor microenvironment. The primary obstacle arises from the varied sensitivity to ferroptosis observed between malignant cells and the immune cells tasked with fighting them. While many malignant cells exhibit inherent sensitivities driven by high iron level, disordered lipid metabolism, and compromised antioxidant defenses, while immune cells, particularly CD8+ T cells, NK cells, and dendritic cells-often share or even exceed this vulnerability. Lipid-rich tumor microenvironments promote fatty acid uptake through CD36, causing the lipid peroxidation and effectively destroying cytotoxic lymphocytes through ferroptotic collapse. Consequently, ferroptosis induction may effectively check antitumor immunity by inhibiting immune effector cell integrity, cytokine synthesis, and antigen presentation. To surpass this hurdle, strategies must be there that activate ferroptosis in tumor cell while protecting the redox balance of immune cells specifically through cell-type-restricted delivery systems, immune-protective antioxidant buffering, or temporal sequencing with immunotherapy. Beyond its apoptotic nature, ferroptosis significantly reshape the immunological landscape via lipid-dependent signaling molecules, which constitute an overlooked hurdle to clinical efficacy. Ferroptotic tumor cells secretes oxidized phospholipids, prostaglandin E2, 4-hydroxynonenal, and truncated lipid entities that actively reshape the immune microenvironment. These lipid signals promote M2 macrophage polarization, deactivates dendritic cell maturation and cross-presentation, and inhibit cytotoxic T cell recruitment and function. Importantly, these immunosuppressive effects may prevail even when ferroptotic cell death is absent, suggesting that lipid peroxidation-driven signaling, are the primary drivers of immune dysfunction rather than actual cell death. Future therapeutic studies must therefore focus on decoupling fatal lipid peroxidation from immunosuppressive lipid signaling, like by combining ferroptosis inducers with inhibitors of COX-2, specific lipoxygenases, or oxidized lipid scavenging pathways. Although ferroptosis meets several criteria of immunogenic cell death, its immunostimulatory ability appears fundamentally different from apoptosis or pyroptosis. Unlike apoptosis or pyroptosis that rely on proteinaceous signals, ferroptosis predominantly generates lipid-based damage-associated molecular patterns, resulting in immune activation that is highly dependent on lipid metabolism, immune composition, and redox balance within the tumor microenvironment. Such metabolic nuances may explain the inconsistent synergy among ferroptosis induction and immune checkpoint blockade across different tumor models. It is essential to identify the unique ferroptosis-specific immunogenic signatures, including lipid-derived adjuvants capable of licensing dendritic cells and sustaining cytotoxic T cell responses while bypassing the pitfalls of immune tolerance.From a translational stand point, incorporating ferroptosis into combined cancer treatment introduces additional complexity. Radiotherapy and immunotherapy create a ferroptotic window by IFN-y-STAT1-mediated repression of SLC7A11 and enhanced oxidative stress, yet adaptive resistance frequently emerges via NRF2 activation, FSP1/CoQ10 upregulation, or metabolic rewiring of lipid saturation states. Moreover, ferroptosis induction may impose selective pressure that favors tumor cell populations with mesenchymal differentiation, enhanced iron sequestration, or altered phospholipid composition. Addressing these resistance challenges will require longitudinal metabolic and lipidomic profiling, as well as rational combination strategies that predict compensatory antioxidant and metabolic pathways. A further challenge to clinical translation is the deficiency of robust, ferroptosis-specific biomarkers abled enough to distinguish productive tumor ferroptosis from detrimental immune ferroptosis or nonspecific oxidative stress. Commonly employed markers such as PTGS2, CHAC1, or lipid peroxidation byproducts deficient in specificity and may not accurately reflect ferroptotic flux in vivo. Future progress will based on the discovery of new integrated biomarker platforms joining lipidomics, iron metabolism signatures, and immune phenotyping to guide patient stratification, monitor therapeutic responses, and optimize treatment timing.
Collectively, the evidence synthesized in this study argues against viewing ferroptosis as a universally beneficial cytotoxic modality. Instead, ferroptosis should be conceptualized as a dynamic tunable immuno–metabolic axis whose therapeutic value is determined by cellular context, immune composition, and metabolic state. Advances in nanotechnology, metabolic targeting, and immune engineering offer promising opportunities to selectively manipulate ferroptosis within tumors while preserving immune competence. Ultimately, the successful clinical deployment of ferroptosis-based therapies will require precision modulation rather than maximal induction, guided by a nuanced understanding of tumor–immune–lipid crosstalk and its dynamic evolution during therapy.
10 Conclusion
Ferroptosis represents a promising yet complex avenue in cancer therapy, with its dualistic role as a tumor suppressor and promoter within the tumor microenvironment. At the same time, ferroptosis induction can enhance anti-tumor immunity and immunosuppressive effects. Careful modulation is necessary to avoid fostering tumor progression. Advancements in understanding the molecular pathways of ferroptosis, such as the role of lipid metabolism and iron homeostasis, have revealed new therapeutic targets. Strategies combine ferroptosis inducers with immunotherapies and radiotherapy, which holds the potential to overcome treatment resistance and improve patient outcomes. However, challenges remain, particularly in addressing the context-dependent effects of ferroptosis and minimizing adverse immune consequences. Collectively, recent studies converge on the concept that ferroptosis represent a double-edged immunological regulator rather than a universally beneficial cytotoxic mechanisms. Precision modulation, instead of indiscriminate induction will be necessary to elaborate ferroptosis for cancer immunotherapy while avoiding immune exhaustion, inflammatory toxicity and treatment resistance. Future research should prioritize unraveling the intricate interactions between ferroptosis immune cells and tumor cells, paving the way for innovative, multi-model cancer therapies exploiting ferroptosis full therapeutic potential.Author contributions
Conceptualization, G. A., and D. Z.; software, G. A., H. G., P. U. A., and M. A.; resources, G. A. and M. A.; data curation, G.A., H.G., P. U.A., X.C., M. A., and Y. T.; writing – original draft preparation, G. A., and M. A.; writing – review and editing, D. Z.; supervision, D. Z.; project administration, G. A., and M. A.; funding acquisition, G. A. All authors have read and agreed to the published version of the manuscript.Conflicts of interest
The authors declare no conflicts of interest.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Funding
This study was supported by Doctoral Fund Project of the Affiliated Hospital of Inner Mongolia Medical University (NYFY BS 202121), Natural Science Foundation Project of Inner Mongolia Autonomous Region (2020LH03017), Inner Mongolia Autonomous Region Medical Association Clinical Medicine Research and Clinical New Technology Promotion Project (YSXH2024KYF033), Science and Technology Project of Inner Mongolia Autonomous Region (2025YFHH0267), and Research on the Bio-synthesis of a Long-acting, Highly Active Follicle-Stimulating Hormone (FSH) for Enhanced Ovulation in Dairy Cows, Supported by National Technology Innovation Center of Dairy (2025-QNJJ-010).Acknowledgements
We are grateful to kind administration of Affiliated Hospital of Inner Mongolia Medical University, Hohhot 010010, Inner Mongolia, China and University of Alberta, Alberta, Canada for providing us such a prestigious platform to this study.References
- A. A. Akhand and N. Ahsan, Cells and Organs of the Immune System. Immunology for Dentistry, 2023, pp. 1–12 Search PubMed.
- N. R. Raghani, et al., Revolutionizing cancer treatment: comprehensive insights into immunotherapeutic strategies, Med. Oncol., 2024, 41(2), 51 CrossRef PubMed.
- P. M. Vaishali and N. Boopathy, Edward Jenner: The Pioneer of Vaccination and His Enduring Legacy in Modern Medicine, Cureus, 2024, 16(9), e68805 Search PubMed.
- E. Nakata, et al., Immunotherapy for sarcomas, Jpn. J. Clin. Oncol., 2021, 51(4), 523–537 CrossRef PubMed.
- R. Siddhartha and M. Garg, Interplay Between Extracellular Matrix Remodeling and Angiogenesis in Tumor Ecosystem, Mol. Cancer Ther., 2023, 22(3), 291–305 CrossRef CAS PubMed.
- N. M. Anderson and M. C. Simon, The tumor microenvironment, Curr. Biol., 2020, 30(16), R921–R925 CrossRef CAS PubMed.
- X. Shi, et al., Mechanism insights and therapeutic intervention of tumor metastasis: latest developments and perspectives, Signal Transduct. Targeted Ther., 2024, 9(1), 192 CrossRef CAS PubMed.
- D. Tarin, Cell and tissue interactions in carcinogenesis and metastasis and their clinical significance, in Seminars in Cancer Biology, Elsevier, 2011 Search PubMed.
- M. Haist, et al., The functional crosstalk between myeloid-derived suppressor cells and regulatory T cells within the immunosuppressive tumor microenvironment, Cancers, 2021, 13(2), 210 CrossRef CAS PubMed.
- D. Shin, J. Lee and J.-L. Roh, Pioneering the future of cancer therapy: Deciphering the p53-ferroptosis nexus for precision medicine, Cancer Lett., 2024, 216645 CrossRef CAS PubMed.
- M. Luo, et al., Targeting lipid metabolism for ferroptotic cancer therapy, Apoptosis, 2023, 28(1), 81–107 CrossRef CAS PubMed.
- S. Deng, et al., Ferroptosis suppressive genes correlate with immunosuppression in glioblastoma, World Neurosurg., 2021, 152, e436–e448 CrossRef PubMed.
- L. Galluzzi, et al., Targeting immunogenic cell stress and death for cancer therapy, Nat. Rev. Drug Discov., 2024, 23(6), 445–460 CrossRef CAS PubMed.
- Y. Liang, et al., Ferroptosis: CD8+ T cells' blade to destroy tumor cells or poison for self-destruction, Cell Death Discov., 2025, 11(1), 128 CrossRef CAS PubMed.
- Y. Zheng, et al., The crosstalk between ferroptosis and anti-tumor immunity in the tumor microenvironment: molecular mechanisms and therapeutic controversy, Cancer Commun., 2023, 43(10), 1071–1096 CrossRef PubMed.
- J. Hu, et al., Mechanism and role of regulated cell death in tumor immunity and immunotherapy, Cancer Commun., 2025, 45(11), 1456–1495 CrossRef PubMed.
- Y. Yu, et al., Ferroptosis: a cell death connecting oxidative stress, inflammation and cardiovascular diseases, Cell Death Discov., 2021, 7(1), 193 Search PubMed.
- R. U. Macías-Rodríguez, et al., Reclassifying hepatic cell death during liver damage: ferroptosis—a novel form of non-apoptotic cell death?, Int. J. Mol. Sci., 2020, 21(5), 1651 CrossRef PubMed.
- X. Chen, et al., Ferroptosis and cardiovascular disease: role of free radical-induced lipid peroxidation, Free Radic. Res., 2021, 55(4), 405–415 Search PubMed.
- B. Hassannia, S. Van Coillie and T. Vanden Berghe, Ferroptosis: biological rust of lipid membranes, Antioxid. Redox Signaling, 2021, 35(6), 487–509 CrossRef CAS PubMed.
- Z. Shen, et al., Emerging strategies of cancer therapy based on ferroptosis, Adv. Mater., 2018, 30(12), 1704007 CrossRef PubMed.
- C. M. C. Andrés, et al., Superoxide anion chemistry—Its role at the core of the innate immunity, Int. J. Mol. Sci., 2023, 24(3), 1841 CrossRef PubMed.
- S. Onur Yaman and A. Ayhanci, Lipid peroxidation, 2021 Search PubMed.
- L. Valgimigli, Lipid peroxidation and antioxidant protection, Biomolecules, 2023, 13(9), 1291 Search PubMed.
- N. T. Moldogazieva, et al., Lipid peroxidation: Reactive carbonyl species, protein/DNA adducts, and signaling switches in oxidative stress and cancer, Biochem. Biophys. Res. Commun., 2023, 687, 149167 CrossRef CAS PubMed.
- R. Djorgbenoo, Trapping Effects of Lipid Peroxidation Products-Malonaldehyde and 4-Hydroxynonenal-By Dietary Flavonoids, North Carolina Agricultural and Technical State University, 2023 Search PubMed.
- J. Huang, et al., Dexmedetomidine inhibits ferroptosis and attenuates sepsis-induced acute kidney injury via activating the Nrf2/SLC7A11/FSP1/CoQ10 pathway, Redox Rep., 2024, 29(1), 2430929 CrossRef PubMed.
- H. Chen, et al., Dual-pathway mechanism of vanadium-induced hepatotoxicity in ducks: Synergistic crosstalk between glucose homeostasis disruption and NADH/FSP1/COQ10 axis-driven ferroptosis, Int. J. Biol. Sci., 2026, 22(1), 43–59 CrossRef PubMed.
- B. Jia, et al., ACSL4-Mediated ferroptosis and its potential role in central nervous system diseases and injuries, Int. J. Mol. Sci., 2023, 24(12), 10021 CrossRef CAS PubMed.
- Y. Dai, et al., Inhibition of ACSL4 ameliorates tubular ferroptotic cell death and protects against fibrotic kidney disease, Commun. Biol., 2023, 6(1), 907 CrossRef CAS PubMed.
- L. Zhao, et al., Ferroptosis in cancer and cancer immunotherapy, Cancer Commun., 2022, 42(2), 88–116 CrossRef PubMed.
- J. Wang, et al., Research progress, challenges and perspectives of phospholipids metabolism in the LXR-LPCAT3 signaling pathway and its relation to NAFLD, Int. J. Mol. Med., 2024, 53(4), 1–13 Search PubMed.
- Z. Ma, et al., Effects of Deoxynivalenol and Its Acetylated Derivatives on Lipid Metabolism in Human Normal Hepatocytes, Toxins, 2024, 16(7), 294 CrossRef CAS PubMed.
- Y. Zou, et al., Plasticity of ether lipids promotes ferroptosis susceptibility and evasion, Nature, 2020, 585(7826), 603–608 CrossRef CAS PubMed.
- M. S. Mortensen, J. Ruiz and J. L. Watts, Polyunsaturated fatty acids drive lipid peroxidation during ferroptosis, Cells, 2023, 12(5), 804 CrossRef CAS PubMed.
- J.-Y. Lee, et al., Lipid metabolism and ferroptosis, Biology, 2021, 10(3), 184 CrossRef CAS PubMed.
- J. Rodencal and S. J. Dixon, A tale of two lipids: Lipid unsaturation commands ferroptosis sensitivity, Proteomics, 2023, 23(6), 2100308 Search PubMed.
- U. Afnan and et al., Nanotherapeutic approaches for managing phospholipase-mediated neurodegenerative and metabolic diseases, in Phospholipases in Physiology and Pathology, Elsevier, 2023, p. 129–157 Search PubMed.
- Y. Chen, et al., Targeting iron metabolism and ferroptosis as novel therapeutic approaches in cardiovascular diseases, Nutrients, 2023, 15(3), 591 CrossRef PubMed.
- S. Maitra and D. Dutta, Iron-Siderophore and Tumorigenesis: Molecular Insight and Therapeutic Targets 1, in Metal Toxicology Handbook, CRC Press, 2020, p. 303–309 Search PubMed.
- V. Venkataramani, Iron homeostasis and metabolism: two sides of a coin, Ferroptosis: Mechanism and Diseases, 2021, pp. 25–40 Search PubMed.
- J. Chen, et al., The multifaceted role of ferroptosis in liver disease, Cell Death Differ., 2022, 29(3), 467–480 CrossRef CAS PubMed.
- K. Wu and F. Lin, Lipid metabolism as a potential target of liver cancer, J. Hepatocell. Carcinoma, 2024, 327–346 Search PubMed.
- N. Santana-Codina and J. D. Mancias, The role of NCOA4-mediated ferritinophagy in health and disease, Pharmaceuticals, 2018, 11(4), 114 Search PubMed.
- S. Shi, et al., Role of oxidative stress and inflammation-related signaling pathways in doxorubicin-induced cardiomyopathy, Cell Commun. Signal., 2023, 21(1), 61 Search PubMed.
- C. Huo, et al., The impacts of iron overload and ferroptosis on intestinal mucosal homeostasis and inflammation, Int. J. Mol. Sci., 2022, 23(22), 14195 Search PubMed.
- J. Hu, et al., Regulation of programmed cell death by Brd4, Cell Death Dis., 2022, 13(12), 1059 CrossRef CAS PubMed.
- J. Quarleri, C. Cevallos and M. V. Delpino, Apoptosis in infectious diseases as a mechanism of immune evasion and survival, Adv. Protein Chem. Struct. Biol., 2021, 125, 1–24 Search PubMed.
- S.-K. Hsu, et al., Inflammation-related pyroptosis, a novel programmed cell death pathway, and its crosstalk with immune therapy in cancer treatment, Theranostics, 2021, 11(18), 8813 CrossRef CAS PubMed.
- M. Zhu, et al., Caspase-Linked Programmed Cell Death in Prostate Cancer: From Apoptosis, Necroptosis, and Pyroptosis to PANoptosis, Biomolecules, 2023, 13(12), 1715 CrossRef CAS PubMed.
- J. Seo, et al., Necroptosis molecular mechanisms: Recent findings regarding novel necroptosis regulators, Exp. Mol. Med., 2021, 53(6), 1007–1017 CrossRef CAS PubMed.
- H. S. Chaouhan, et al., Necroptosis: a pathogenic negotiator in human diseases, Int. J. Mol. Sci., 2022, 23(21), 12714 CrossRef CAS PubMed.
- L. Rochette, et al., Lipid peroxidation and iron metabolism: two corner stones in the homeostasis control of ferroptosis, Int. J. Mol. Sci., 2022, 24(1), 449 CrossRef PubMed.
- V. Vella, et al., Insulin/IGF axis and the Receptor for Advanced Glycation End Products: role in meta-inflammation and potential in cancer therapy, Endocr. Rev., 2023, 44(4), 693–723 CrossRef CAS PubMed.
- Z. Xiao, et al., Programmed cell death and lipid metabolism of macrophages in NAFLD, Front. Immunol., 2023, 14, 1118449 CrossRef CAS PubMed.
- W. Ma, et al., Mechanisms of ferroptosis and targeted therapeutic approaches in urological malignancies, Cell Death Discov., 2024, 10(1), 432 CrossRef CAS PubMed.
- J. Yang, et al., Targeting cell death: pyroptosis, ferroptosis, apoptosis and necroptosis in osteoarthritis, Front. Cell Dev. Biol., 2022, 9, 789948 CrossRef PubMed.
- W. Gao, et al., Autophagy, ferroptosis, pyroptosis, and necroptosis in tumor immunotherapy, Signal Transduct. Targeted Ther., 2022, 7(1), 196 CrossRef PubMed.
- M. Qu, et al., Necroptosis, pyroptosis, ferroptosis in sepsis and treatment, Shock, 2022, 57(6), 161–171 CrossRef CAS PubMed.
- F. Chen, et al., Ferroptosis: principles and significance in health and disease, J. Hematol. Oncol., 2024, 17(1), 41 Search PubMed.
- Y. Cheng, et al., Role of lipid metabolism in hepatocellular carcinoma, Discov. Oncol., 2024, 15(1), 206 Search PubMed.
- C. I. Coclitu, C. S. Constantinescu and R. Tanasescu, Neuroprotective strategies in multiple sclerosis: a status update and emerging paradigms, Expert Rev. Neurother., 2025, 25(7), 791–817 Search PubMed.
- B. Gan, ACSL4, PUFA, and ferroptosis: new arsenal in anti-tumor immunity, Signal Transduct. Targeted Ther., 2022, 7(1), 128 CrossRef PubMed.
- Y. Wang, et al., Targeting epigenetic and posttranslational modifications regulating ferroptosis for the treatment of diseases, Signal Transduct. Targeted Ther., 2023, 8(1), 449 CrossRef CAS PubMed.
- Z. Lin, S. Zou and K. Wen, The crosstalk of CD8+ T cells and ferroptosis in cancer, Front. Immunol., 2024, 14, 1255443 CrossRef PubMed.
- P. Koppula, L. Zhuang and B. Gan, Cystine transporter SLC7A11/xCT in cancer: ferroptosis, nutrient dependency, and cancer therapy, Protein Cell, 2021, 12(8), 599–620 CrossRef CAS PubMed.
- C. Punziano, et al., Antioxidant systems as modulators of ferroptosis: focus on transcription factors, Antioxidants, 2024, 13(3), 298 CrossRef CAS PubMed.
- P. Liao, et al., CD8+ T cells and fatty acids orchestrate tumor ferroptosis and immunity via ACSL4, Cancer Cell, 2022, 40(4), 365–378 CrossRef CAS PubMed.
- X. Yang, et al., Ferroptosis as a new tool for tumor suppression through lipid peroxidation, Commun. Biol., 2024, 7(1), 1475 CrossRef PubMed.
- L. Zeng, et al., Advances in research on immunocyte iron metabolism, ferroptosis, and their regulatory roles in autoimmune and autoinflammatory diseases, Cell Death Dis., 2024, 15(7), 481 CrossRef PubMed.
- Y. Chen, et al., PD-1/PD-L1 immune checkpoint blockade-based combinational treatment: Immunotherapeutic amplification strategies against colorectal cancer, Int. Immunopharmacol., 2021, 96, 107607 CrossRef CAS PubMed.
- S. Mishra, V. Shelke and A. B. Gaikwad, Acyl-CoA Synthetase Long-Chain Isoenzymes in Kidney Diseases: Mechanistic Insights and Therapeutic Implications, Cell Biochem. Funct., 2024, 42(7), e4114 Search PubMed.
- E. Dai, et al., A guideline on the molecular ecosystem regulating ferroptosis, Nat. Cell Biol., 2024, 1–11 CAS.
- M. N. Park, et al., SH003 as a Redox-Immune Modulating Phytomedicine: A Ferroptosis Induction, Exosomal Crosstalk, and Translational Oncology Perspective, Cancers, 2025, 17(21), 3519 CrossRef CAS PubMed.
- Y. Chen, et al., Metabolic response to androgen deprivation therapy of prostate cancer, Cancers, 2024, 16(11), 1991 CrossRef CAS PubMed.
- Y.-h. Duan, et al., Reflections on the complex mechanisms of endometriosis from the perspective of ferroptosis, Pathol., Res. Pract., 2024, 259, 155353 CrossRef CAS PubMed.
- Q. Zhou, et al., Ferroptosis in cancer: from molecular mechanisms to therapeutic strategies, Signal Transduct. Targeted Ther., 2024, 9(1), 55 Search PubMed.
- X. Tong, et al., Predicting diagnostic gene biomarkers associated with immune checkpoints, N6-methyladenosine, and ferroptosis in patients with acute myocardial infarction, Front. Cardiovasc. Med., 2022, 9, 836067 CrossRef CAS PubMed.
- E. Ficiarà, et al., Alteration of iron concentration in Alzheimer's disease as a possible diagnostic biomarker unveiling ferroptosis, Int. J. Mol. Sci., 2021, 22(9), 4479 CrossRef PubMed.
- H. Wang, et al., Integrated analysis of ferroptosis-related biomarker signatures to improve the diagnosis and prognosis prediction of ovarian cancer, Front. Cell Dev. Biol., 2022, 9, 807862 CrossRef PubMed.
- M. Li, et al., Novel diagnostic biomarkers related to oxidative stress and macrophage ferroptosis in atherosclerosis, Oxid. Med. Cell. Longev., 2022, 2022(1), 8917947 CrossRef PubMed.
- F. Zhong, et al., The therapeutic and biomarker significance of ferroptosis in chronic myeloid leukemia, Front. Immunol., 2024, 15, 1402669 CrossRef CAS PubMed.
- X. Chen, et al., Characteristics and biomarkers of ferroptosis, Front. Cell Dev. Biol., 2021, 9, 637162 CrossRef PubMed.
- Y. Hu, et al., Identification of ferroptosis-associated biomarkers for the potential diagnosis and treatment of postmenopausal osteoporosis, Front. Endocrinol., 2022, 13, 986384 CrossRef PubMed.
- T. Zhang, et al., Glutathione-degrading enzymes in the complex landscape of tumors, Int. J. Oncol., 2024, 65(1), 1–22 Search PubMed.
- V. Consoli, et al., Heme oxygenase-1 signaling and redox homeostasis in physiopathological conditions, Biomolecules, 2021, 11(4), 589 CrossRef CAS PubMed.
- D. Tang and R. Kang, NFE2L2 and ferroptosis resistance in cancer therapy, Cancer Drug Resist., 2024, 7, 41 CAS.
- F. Alves, et al., In defence of ferroptosis, Signal Transduct. Targeted Ther., 2025, 10(1), 2 CrossRef CAS PubMed.
- R. Gencheva, Mechanisms of Action of Thioredoxin Reductase Inhibitors in the Context of Cancer, Karolinska Institutet, 2024 Search PubMed.
- Y. Wang, et al., Bioinformatics analysis of ferroptosis-related gene AKR1C3 as a potential biomarker of asthma and its identification in BEAS-2B cells, Comput. Biol. Med., 2023, 158, 106740 CrossRef CAS PubMed.
- Y. Chen, et al., Iron accumulation in ovarian microenvironment damages the local redox balance and oocyte quality in aging mice, Redox Biol., 2024, 73, 103195 CrossRef CAS PubMed.
- R. Wu, D. Tang, and R. Kang, Fluorogenic Probes for Intracellular Iron Detection, in Ferroptosis: Methods and Protocols, Springer, 2023, pp. 1–8 Search PubMed.
- X. Fang, T. Zhang and Z. Chen, Solute carrier family 7 member 11 (SLC7A11) is a potential prognostic biomarker in uterine corpus endometrial carcinoma, Int. J. Gen. Med., 2023, 481–497 Search PubMed.
- J. Liu, et al., ATF3-CBS signaling axis coordinates ferroptosis and tumorigenesis in colorectal cancer, Redox Biol., 2024, 71, 103118 Search PubMed.
- X. Chen, et al., Ferroptosis: machinery and regulation, Autophagy, 2021, 17(9), 2054–2081 CrossRef CAS PubMed.
- T. L. Duarte, N. P. Talbot and H. Drakesmith, NRF2 and hypoxia-inducible factors: key players in the redox control of systemic iron homeostasis, Antioxidants Redox Signal., 2021, 35(6), 433–452 CrossRef CAS PubMed.
- B. Wang, et al., ROS-induced lipid peroxidation modulates cell death outcome: mechanisms behind apoptosis, autophagy, and ferroptosis, Arch. Toxicol., 2023, 97(6), 1439–1451 CrossRef CAS PubMed.
- J. Liu, R. Kang and D. Tang, Signaling pathways and defense mechanisms of ferroptosis, FEBS J., 2022, 289(22), 7038–7050 CrossRef CAS PubMed.
- Q. Bi, et al., Ferroptosis-mediated formation of tumor-promoting immune microenvironment, Front. Oncol., 2022, 12, 868639 CrossRef CAS PubMed.
- E. Dai, et al., Autophagy-dependent ferroptosis drives tumor-associated macrophage polarization via release and uptake of oncogenic KRAS protein, Autophagy, 2020, 16(11), 2069–2083 CrossRef CAS PubMed.
- R. Chen, et al., The redox protein high-mobility group box 1 in cell death and cancer, Antioxidants Redox Signal., 2023, 39(7–9), 569–590 CrossRef CAS PubMed.
- S. Yang, et al., Maximizing Arsenic Trioxide's Anticancer Potential: Targeted Nanocarriers for Solid Tumor Therapy, Colloids Surf., B, 2024, 114014 Search PubMed.
- N. Jang, et al., Regulation of ferroptosis in cancer and immune cells, Immune Netw., 2025, 25(1), e6 CrossRef PubMed.
- Y. Sun, et al., Nanomedicine-mediated regulated cell death in cancer immunotherapy, J. Controlled Release, 2023, 364, 174–194 CrossRef CAS PubMed.
- D. An, et al., The role of lipid metabolism in cancer radioresistance, Clin. Transl. Oncol., 2023, 25(8), 2332–2349 CrossRef CAS PubMed.
- Y. Setayeshpour, Y. Lee and J.-T. Chi, Environmental Determinants of Ferroptosis in Cancer, Cancers, 2023, 15(15), 3861 Search PubMed.
- G. Carbone, et al., Endometriosis: cell death and cell signaling machinery, Endocrinology, 2023, 164(6), bqad057 Search PubMed.
- C. Peleman, S. Francque and T. Vanden Berghe, Emerging role of ferroptosis in metabolic dysfunction-associated steatotic liver disease: revisiting hepatic lipid peroxidation, EBioMedicine, 2024, 102, 105088 CrossRef CAS PubMed.
- A.-S. Amoah, et al., Lipoxygenases at the Intersection of Infection and Carcinogenesis, Int. J. Mol. Sci., 2024, 25(7), 3961 CrossRef CAS PubMed.
- F. I. Yapici, C. M. Bebber and S. von Karstedt, A guide to ferroptosis in cancer, Mol. Oncol., 2024, 18(6), 1378–1396 CrossRef PubMed.
- B. Xiang, et al., The epitranscriptional factor PCIF1 orchestrates CD8+ T cell ferroptosis and activation to control antitumor immunity, Nat. Immunol., 2025, 1–13 Search PubMed.
- L. Han, et al., PPARG-mediated ferroptosis in dendritic cells limits antitumor immunity, Biochem. Biophys. Res. Commun., 2021, 576, 33–39 CrossRef CAS PubMed.
- J. Yang, et al., Glioblastoma-derived exosomes promote lipid accumulation and induce ferroptosis in dendritic cells via the NRF2/GPX4 pathway, Front. Immunol., 2024, 15, 1439191 CrossRef CAS PubMed.
- K. T. Giuliani, et al., Hypoxic human proximal tubular epithelial cells undergo ferroptosis and elicit an NLRP3 inflammasome response in CD1c+ dendritic cells, Cell Death Dis., 2022, 13(8), 739 CrossRef CAS PubMed.
- L. Shi, et al., Emerging roles of ferroptosis in the tumor immune landscape: from danger signals to anti-tumor immunity, FEBS J., 2022, 289(13), 3655–3665 CrossRef CAS PubMed.
- S. Hu, et al., Engineered exosome-like nanovesicles suppress tumor growth by reprogramming tumor microenvironment and promoting tumor ferroptosis, Acta Biomater., 2021, 135, 567–581 CrossRef CAS PubMed.
- G. Napolitano, G. Fasciolo and P. Venditti, Mitochondrial management of reactive oxygen species, Antioxidants, 2021, 10(11), 1824 CrossRef CAS PubMed.
- B. Gan, Mitochondrial regulation of ferroptosis, J. Cell Biol., 2021, 220(9), e202105043 CrossRef CAS PubMed.
- X. Yao, et al., Emerging roles of energy metabolism in ferroptosis regulation of tumor cells, Advanced Science, 2021, 8(22), 2100997 CrossRef CAS PubMed.
- M. J. Lamberti, et al., Damage-associated molecular patterns modulation by microRNA: relevance on immunogenic cell death and cancer treatment outcome, Cancers, 2021, 13(11), 2566 CrossRef CAS PubMed.
- A. Murao, et al., Release mechanisms of major DAMPs, Apoptosis, 2021, 26(3), 152–162 CrossRef CAS PubMed.
- Z. Zheng, et al., Mechanisms and applications of radiation-induced oxidative stress in regulating cancer immunotherapy, Front. Immunol., 2023, 14, 1247268 CrossRef CAS PubMed.
- J. Dou, et al., Ferroptosis interaction with inflammatory microenvironments: mechanism, biology, and treatment, Biomed. Pharmacother., 2022, 155, 113711 CrossRef CAS PubMed.
- C. Gong, et al., Ferroptosis in tumor immunity and therapy, J. Cell. Mol. Med., 2022, 26(22), 5565–5579 CrossRef CAS PubMed.
- Y. Lv, et al., Ferroptosis: From regulation of lipid peroxidation to the treatment of diseases, Cell Biol. Toxicol., 2023, 39(3), 827–851 CrossRef PubMed.
- S. Du, F. Zeng and G. Deng, Tumor neutrophils ferroptosis: a targetable immunosuppressive mechanism for cancer immunotherapy, Signal Transduct. Targeted Ther., 2023, 8(1), 77 CrossRef PubMed.
- D. Tang, G. Kroemer and R. Kang, Ferroptosis in immunostimulation and immunosuppression, Immunol. Rev., 2024, 321(1), 199–210 CrossRef CAS PubMed.
- R. Kim, et al., Ferroptosis of tumour neutrophils causes immune suppression in cancer, Nature, 2022, 612(7939), 338–346 Search PubMed.
- Q. Yang, et al., A nanoformulation for immunosuppression reversal and broad-spectrum self-amplifying antitumor ferroptosis-immunotherapy, Biomaterials, 2023, 292, 121936 CrossRef CAS PubMed.
- Y. Lu, X. Xie and L. Luo, Ferroptosis crosstalk in anti-tumor immunotherapy: Molecular mechanisms, tumor microenvironment, application prospects, Apoptosis, 2024, 1–30 Search PubMed.
- Y. R. Murciano-Goroff, A. B. Warner and J. D. Wolchok, The future of cancer immunotherapy: microenvironment-targeting combinations, Cell Res., 2020, 30(6), 507–519 Search PubMed.
- Z. Mao, et al., The Mutual Regulatory Role of Ferroptosis and Immunotherapy in Anti-tumor Therapy, Apoptosis, 2024, 1–18 Search PubMed.
- H. Zhang, E. Zhang and H. Hu, Role of ferroptosis in non-alcoholic fatty liver disease and its implications for therapeutic strategies, Biomedicines, 2021, 9(11), 1660 CrossRef CAS PubMed.
- D. Tang, et al., Ferroptosis: molecular mechanisms and health implications, Cell Res., 2021, 31(2), 107–125 Search PubMed.
- Y. Yao, et al., Selenium–GPX4 axis protects follicular helper T cells from ferroptosis, Nat. Immunol., 2021, 22(9), 1127–1139 CrossRef CAS PubMed.
- C. Xu, S. Sun, T. Johnson, R. Qi, S. Zhang, J. Zhang and K. Yang, The glutathione peroxidase Gpx4 prevents lipid peroxidation and ferroptosis to sustain Treg cell activation and suppression of antitumor immunity, Cell Rep., 2021, 35(11), 109235. Search PubMed.
- S. P. Sok, et al., Gpx4 Regulates Invariant NKT Cell Homeostasis and Function by Preventing Lipid Peroxidation and Ferroptosis, J. Immunol., 2024, 213(7), 941–951 Search PubMed.
- G. Qu, et al., GPX4 is a key ferroptosis biomarker and correlated with immune cell populations and immune checkpoints in childhood sepsis, Sci. Rep., 2023, 13(1), 11358 Search PubMed.
- H. Li, et al., GPX4 aggravates experimental autoimmune encephalomyelitis by inhibiting the functions of CD4+ T cells, Biochem. Biophys. Res. Commun., 2023, 642, 57–65 Search PubMed.
- J. Luoqian, et al., Ferroptosis promotes T-cell activation-induced neurodegeneration in multiple sclerosis, Cell. Mol. Immunol., 2022, 19(8), 913–924 CrossRef CAS PubMed.
- Q. Hu, et al., GPX4 and vitamin E cooperatively protect hematopoietic stem and progenitor cells from lipid peroxidation and ferroptosis, Cell Death Dis., 2021, 12(7), 706 CrossRef CAS PubMed.
- S. Luo, et al., Vitamin E and GPX4 cooperatively protect treg cells from ferroptosis and alleviate intestinal inflammatory damage in necrotizing enterocolitis, Redox Biol., 2024, 75, 103303 CrossRef CAS PubMed.
- S. Chen, J. Fan, P. Xie, J. Ahn, M. Fernandez, L. K. Billingham, J. Miska, J. D. Wu, D. A. Wainwright, D. Fang, J. A. Sosman, Y. Wan, Y. Zhang, N. S. Chandel and B. Zhang, CD8+ T cells sustain antitumor response by mediating crosstalk between adenosine A2A receptor and glutathione/GPX4, J. Clin. Investig., 2024, 134(8), e170071 Search PubMed.
- I. Ingold, et al., Selenium utilization by GPX4 is required to prevent hydroperoxide-induced ferroptosis, Cell, 2018, 172(3), 409–422 CrossRef CAS PubMed.
- Q. Wang, Y. Qin and B. Li, CD8+ T cell exhaustion and cancer immunotherapy, Cancer Lett., 2023, 559, 216043 CrossRef CAS PubMed.
- X. Li, et al., GPX4 restricts ferroptosis of NKp46+ ILC3s to control intestinal inflammation, Cell Death Dis., 2024, 15(9), 687 CrossRef CAS PubMed.
- H. Li, et al., USP8-governed GPX4 homeostasis orchestrates ferroptosis and cancer immunotherapy, Proc. Natl. Acad. Sci. U. S. A., 2024, 121(16), e2315541121 CrossRef CAS PubMed.
- Y. Sun, et al., Lipid metabolism in ferroptosis and ferroptosis-based cancer therapy, Front. Oncol., 2022, 12, 941618 CrossRef CAS PubMed.
- S. Pereira, et al., The normal, the radiosensitive, and the ataxic in the era of precision radiotherapy: A narrative review, Cancers, 2022, 14(24), 6252 CrossRef CAS PubMed.
- L. Mi and H. Zhang, Myriad factors and pathways influencing tumor radiotherapy resistance, Open Life Sci., 2024, 19(1), 20220992 CrossRef CAS PubMed.
- G. Dagar, et al., The future of cancer treatment: Combining radiotherapy with immunotherapy, Front. Mol. Biosci., 2024, 11, 1409300 CrossRef CAS PubMed.
- J. Khalifa, et al., Radiotherapy in the era of immunotherapy with a focus on non-small-cell lung cancer: time to revisit ancient dogmas?, Front. Oncol., 2021, 11, 662236 CrossRef CAS PubMed.
- S. Dallavalasa, et al., The role of tumor associated macrophages (TAMs) in cancer progression, chemoresistance, angiogenesis and metastasis-current status, Curr. Med. Chem., 2021, 28(39), 8203–8236 CAS.
- M. Levis, et al., Modern stereotactic radiotherapy for brain metastases from lung cancer: current trends and future perspectives based on integrated translational approaches, Cancers, 2023, 15(18), 4622 CrossRef CAS PubMed.
- M. Elbanna, et al., Clinical and preclinical outcomes of combining targeted therapy with radiotherapy, Front. Oncol., 2021, 11, 749496 CrossRef CAS PubMed.
- X. Xu, et al., Impact of ferroptosis-related risk genes on macrophage M1/M2 polarization and prognosis in glioblastoma, Front. Cell. Neurosci., 2024, 17, 1294029 CrossRef PubMed.
- P. Hou, et al., Macrophage polarization and metabolism in atherosclerosis, Cell Death Dis., 2023, 14(10), 691 CrossRef PubMed.
- M. Yang, et al., Ferroptosis of macrophages facilitates bone loss in apical periodontitis via NRF2/FSP1/ROS pathway, Free Radic. Biol. Med., 2023, 208, 334–347 CrossRef CAS PubMed.
- D. Liu, et al., Cardiovascular Disease: Mitochondrial dynamics and mitophagy crosstalk mechanisms with novel programmed cell death and macrophage polarisation, Pharmacol. Res., 2024, 107258 CrossRef CAS PubMed.
- S. Wang, et al., Targeting M2-like tumor-associated macrophages is a potential therapeutic approach to overcome antitumor drug resistance, npj Precis. Oncol., 2024, 8(1), 31 CrossRef PubMed.
- H. Bai, L. Feng and F. Schmid, Macrophage-based cancer immunotherapy: challenges and opportunities, Exp. Cell Res., 2024, 114198 CrossRef CAS PubMed.
- X. Cao and et al., Targeting tumor-associated macrophages for cancer immunotherapy, in International Review of Cell and Molecular Biology, Elsevier, 2022, pp. 61–108 Search PubMed.
- S. He, L. Zheng and C. Qi, Myeloid-derived suppressor cells (MDSCs) in the tumor microenvironment and their targeting in cancer therapy, Mol. Cancer, 2025, 24(1), 1–24 CrossRef PubMed.
- V. Chagovets, et al., Specific changes in amino acid profiles in monocytes of patients with breast, lung, colorectal and ovarian cancers, Front. Immunol., 2024, 14, 1332043 CrossRef PubMed.
- M. Dysthe and R. Parihar, Myeloid-derived suppressor cells in the tumor microenvironment, Tumor Microenvironment: Hematopoietic Cells–Part A, 2020, pp. 117–140 Search PubMed.
- Y. Mi, et al., The emerging role of myeloid-derived suppressor cells in the glioma immune suppressive microenvironment, Front. Immunol., 2020, 11, 737 CrossRef CAS PubMed.
- K. Li, et al., Myeloid-derived suppressor cells as immunosuppressive regulators and therapeutic targets in cancer, Signal Transduct. Targeted Ther., 2021, 6(1), 362 CrossRef PubMed.
- C. Lu, et al., The role of innate immune cells in the tumor microenvironment and research progress in anti-tumor therapy, Front. Immunol., 2023, 13, 1039260 CrossRef PubMed.
- L. Quatrini, et al., The immune checkpoint PD-1 in natural killer cells: expression, function and targeting in tumour immunotherapy, Cancers, 2020, 12(11), 3285 CrossRef CAS PubMed.
- Y. Yang, et al., Metabolic hallmarks of natural killer cells in the tumor microenvironment and implications in cancer immunotherapy, Oncogene, 2023, 42(1), 1–10 CrossRef CAS PubMed.
- S. Ness, S. Lin and J. R. Gordon, Regulatory dendritic cells, T cell tolerance, and dendritic cell therapy for immunologic disease, Front. Immunol., 2021, 12, 633436 CrossRef CAS PubMed.
- R. Kedmi and D. R. Littman, Antigen-presenting cells as specialized drivers of intestinal T cell functions, Immunity, 2024, 57(10), 2269–2279 CrossRef CAS PubMed.
- G. Yang, et al., The transcription factor XBP1 in dendritic cells promotes the TH2 cell response in airway allergy, Sci. Signal., 2023, 16(791), eabm9454 CrossRef CAS PubMed.
- I. Rastogi, et al., Role of B cells as antigen presenting cells, Front. Immunol., 2022, 13, 954936 CrossRef CAS PubMed.
- D. Xue, et al., Tumor-infiltrating B cells: their dual mechanistic roles in the tumor microenvironment, Biomed. Pharmacother., 2024, 179, 117436 CrossRef CAS PubMed.
- X. Wu, et al., B cells and tertiary lymphoid structures in tumors: immunity cycle, clinical impact, and therapeutic applications, Theranostics, 2025, 15(2), 605 CrossRef CAS PubMed.
- J. Ma, et al., A blueprint for tumor-infiltrating B cells across human cancers, Science, 2024, 384(6695), eadj4857 CrossRef CAS PubMed.
- N. K. Wolf, D. U. Kissiov and D. H. Raulet, Roles of natural killer cells in immunity to cancer, and applications to immunotherapy, Nat. Rev. Immunol., 2023, 23(2), 90–105 CrossRef CAS PubMed.
- A. Ajoolabady, et al., Ferroptosis in hepatocellular carcinoma: mechanisms and targeted therapy, Br. J. Cancer, 2023, 128(2), 190–205 CrossRef PubMed.
- X. Tong, et al., Targeting cell death pathways for cancer therapy: recent developments in necroptosis, pyroptosis, ferroptosis, and cuproptosis research, J. Hematol. Oncol., 2022, 15(1), 174 CrossRef PubMed.
- Y. Long, et al., Targeting xCT with sulfasalazine suppresses triple-negative breast cancer growth via inducing autophagy and coordinating cell cycle and proliferation, Anti-Cancer Drugs, 2024, 35(9), 830–843 CrossRef CAS PubMed.
- D. Tang, G. Kroemer and R. Kang, Ferroptosis in hepatocellular carcinoma: from bench to bedside, Hepatology, 2024, 80(3), 721–739 CrossRef PubMed.
- R. Zhang, R. Kang and D. Tang, Ferroptosis in gastrointestinal cancer: from mechanisms to implications, Cancer Lett., 2023, 561, 216147 CrossRef CAS PubMed.
- E. Ozkan and F. Bakar-Ates, Ferroptosis: a trusted ally in combating drug resistance in cancer, Curr. Med. Chem., 2022, 29(1), 41–55 Search PubMed.
- L. Zhu, et al., Ferroptosis-driven nanotherapeutics to reverse drug resistance in tumor microenvironment, ACS Appl. Bio Mater., 2022, 5(6), 2481–2506 CrossRef CAS PubMed.
- R. Chen, et al., Targeting ferroptosis as a potential strategy to overcome the resistance of cisplatin in oral squamous cell carcinoma, Front. Pharmacol, 2024, 15, 1402514 CrossRef CAS PubMed.
- D. Wang, et al., Regulatory pathways and drugs associated with ferroptosis in tumors, Cell Death Dis., 2022, 13(6), 544 CrossRef CAS PubMed.
- Y. Yu, et al., Exploring the mystery of tumor metabolism: Warburg effect and mitochondrial metabolism fighting side by side, Cell. Signal., 2024, 120, 111239 CrossRef CAS PubMed.
- A. Singkarin and et al., Glucose Addiction of Cholangiocarcinoma: Opportunities for Therapeutic Development, Hepatoma Research, 2025, vol. 11 Search PubMed.
- M. Tufail, C.-H. Jiang and N. Li, Altered metabolism in cancer: Insights into energy pathways and therapeutic targets, Mol. Cancer, 2024, 23(1), 203 CrossRef CAS PubMed.
- J. Zheng and M. Conrad, Ferroptosis: when metabolism meets cell death, Physiol. Rev., 2024, 105(2), 651–706 Search PubMed.
- Q. Wei, et al., Plasma proteomics implicate glutamic oxaloacetic transaminases as potential markers for acute myocardial infarction, J. Proteonomics, 2024, 308, 105286 CrossRef CAS PubMed.
- G. H. Siwo, A. G. Singal and A. K. Waljee, Pan-cancer molecular signatures connecting aspartate transaminase (AST) to cancer prognosis, metabolic and immune signatures, bioRxiv, 2024, preprint DOI:10.1101/2024.03.01.582939.
- N. E. Scharping, et al., Mitochondrial stress induced by continuous stimulation under hypoxia rapidly drives T cell exhaustion, Nat. Immunol., 2021, 22(2), 205–215 Search PubMed.
- R. Azizi, H. H. Ahmed, R. Abdul Kareem, W. M. Taher Waam, M. Alwan, M. J. Jawad, A. K. Hamad and S. Darzi, SLC7A11 Inhibitors Represent a Promising Therapeutic Target by Facilitating the Induction of Ferroptosis in Breast Cancer, Int. J. Mol. Cell. Med., 2025, 14(1), 496–516 CAS.
- M. Jiang, H. Fang and H. Tian, Metabolism of cancer cells and immune cells in the initiation, progression, and metastasis of cancer, Theranostics, 2025, 15(1), 155 CrossRef CAS PubMed.
- M. Wu, et al., Cancer stem cell regulated phenotypic plasticity protects metastasized cancer cells from ferroptosis, Nat. Commun., 2022, 13(1), 1371 CrossRef CAS PubMed.
- R. Vishwa, et al., Lipid metabolism and its implications in tumor cell plasticity and drug resistance: what we learned thus far?, Cancer Metastasis Rev., 2024, 43(1), 293–319 CrossRef PubMed.
- Z.-D. Shi, et al., Tumor cell plasticity in targeted therapy-induced resistance: mechanisms and new strategies, Signal Transduct. Targeted Ther., 2023, 8(1), 113 CrossRef PubMed.
- P. C. McDonald and S. Dedhar, Persister cell plasticity in tumour drug resistance, in Seminars in Cell & Developmental Biology, Elsevier, 2024 Search PubMed.
- A. Schwab, et al., Zeb1 mediates EMT/plasticity-associated ferroptosis sensitivity in cancer cells by regulating lipogenic enzyme expression and phospholipid composition, Nat. Cell Biol., 2024, 26(9), 1470–1481 CrossRef CAS PubMed.
- W. Tan, et al., Role of non-coding RNA in lineage plasticity of prostate cancer, Cancer Gene Ther., 2024, 1–10 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |