Open Access Article
Open Access ArticleRational design and synthesis of new acetamide–indole–benzo[d]imidazole–carboxylic acid hybrids as dual PTP1B/α-glucosidase inhibitors
Navid Dastyafteha,
Seyedeh Niloufar Ghafourib,
Marjan Mollazadehc,
Nazanin Motamedi Shakibb,
Mohammad Halimid,
Samanesadat Hosseinie,
Somayeh Mojtabavi f,
Mohammad Ali Faramarzi
f,
Mohammad Ali Faramarzi f,
Bagher Larijanic,
Massoud Amanlou
f,
Bagher Larijanic,
Massoud Amanlou g,
Maryam Mohammadi-Khanaposhtani
g,
Maryam Mohammadi-Khanaposhtani *h and
Mohammad Mahdavi
*h and
Mohammad Mahdavi *c
*c
aCenter for Research of Endemic Parasites of Iran, Tehran University of Medical Sciences, Tehran, Iran
bSchool of Chemistry, College of Science, University of Tehran, Tehran, Iran
cEndocrinology and Metabolism Research Center, Endocrinology and Metabolism Clinical Sciences Institute, Tehran University of Medical Sciences, Tehran, Iran. E-mail: momahdavi@tums.ac.ir
dDepartment of Biology, Babol Branch, Islamic Azad University, Babol, Iran
eDepartment of Pharmaceutical Chemistry, School of Pharmacy, Shahid Beheshti University of Medical Sciences, Tehran, Iran
fDepartment of Pharmaceutical Biotechnology, Faculty of Pharmacy and Biotechnology Research Center, Tehran University of Medical Sciences, Tehran, Iran
gDepartment of Medicinal Chemistry, Faculty of Pharmacy, Tehran University of Medical Sciences, Tehran, Iran
hCellular and Molecular Biology Research Center, Health Research Institute, Babol University of Medical Sciences, Babol, Iran. E-mail: maryammoha@gmail.com
First published on 26th January 2026
Abstract
A series of novel acetamide–indole–benzo[d]imidazole–carboxylic acid hybrids (8a–n) was designed based on the structural scaffolds of known protein tyrosine phosphatase 1B (PTP1B) and α-glucosidase inhibitors. Evaluation against PTP1B revealed that six compounds (8d, 8f–h, 8j, and 8l) exhibited superior inhibitory activity compared to the standard inhibitor suramin, while one derivative (8a) demonstrated comparable potency. The remaining compounds showed reduced efficacy relative to suramin. In contrast, only two compounds (8j and 8k) displayed marginally superior α-glucosidase inhibition compared to acarbose, while all other derivatives were less potent than the standard. Based on these results, subsequent investigations focused on the PTP1B inhibitory potential of this series. Kinetic analysis of the most potent compound, 8l, confirmed a competitive inhibition mechanism against PTP1B. Molecular docking studies of the most active compounds yielded binding modes consistent with the in vitro activity, and molecular dynamics simulations further verified the stable binding of compound 8l within the PTP1B active site, supporting its potential as a lead PTP1B inhibitor.
Introduction
Diabetes mellitus, specifically type 2, and obesity are two important metabolic disorders that require multi-target therapeutic strategies for effective management.1,2 Two important targets for treating these disorders are protein tyrosine phosphatase 1B (PTP1B) and α-glucosidase.3 PTP1B is primarily a hepatic enzyme that negatively regulates both insulin and leptin signaling, and α-glucosidase is an intestinal enzyme for digestive carbohydrates.4,5 The inhibition of PTP1B enhances systemic insulin sensitivity and modulates energy balance, while α-glucosidase inhibition locally reduces glucose absorption.6,7Multi-target drug design is a valuable goal in modern medicinal chemistry for the treatment of complex disorders like diabetes and obesity.8 The multifaceted pathogenesis of these metabolic diseases provides a strong rationale for developing multi-target ligands to overcome the limitations of conventional single-target therapies.9 In this regard, the design of lead compounds targeting both PTP1B and α-glucosidase is a valuable pursuit for medicinal chemists.3 In this endeavor, the molecular hybridization of effective pharmacophores presents a particularly powerful and beneficial strategy.10
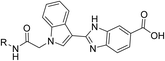
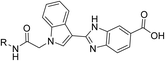
Based on this rationale, we initiated an investigation into novel compounds with the objective of developing dual-target inhibitors capable of simultaneously modulating both PTP1B and α-glucosidase enzymatic activities. Our survey led to the identification of promising molecular scaffolds such as compounds A and B, which contain key pharmacophores including acetamide, indole, benzo[d]imidazole, or carboxylic acid moieties within their structures (Fig. 1).11,12 Notably, with the exception of the carboxylic acid group, other latter pharmacophores are also consistently present in potent α-glucosidase inhibitors such as compounds C and D (Fig. 1).13,14 It should also be noted that each of these representative compounds (A–D) was reported as the most potent inhibitor within its respective structural classes. Guided by these observations, we rationally designed a new series of acetamide–indole–benzo[d]imidazole–carboxylic acid hybrids and synthesized fourteen derivatives 8a–n through conventional organic synthesis methodologies. Notably, our integrated acetamide–indole–benzo[d]imidazole–carboxylic acid hybrid represents a previously unreported scaffold in dual PTP1B/α-glucosidase inhibition, thereby exploring novel chemical space for multi-target anti-diabetic drug discovery. These synthesized compounds were evaluated for their inhibitory activity against both PTP1B and α-glucosidase.
 | ||
| Fig. 1 Design strategy for new acetamide–indole–benzo[d]imidazole–carboxylic acid hybrids as new dual PTP1B/α-glucosidase inhibitors. | ||
Materials and methods
Synthesis of indole-3-carboxaldehyde (2)
Phosphorus oxychloride (340 mmol) was added dropwise to DMF (50 mL), while cooled at 0–5 °C, and stirred in an ice bath for 1 h. Then, indole (1, 170 mmol dissolved in 15 mL of DMF) was added to the ice-cold mixture and the reaction was continued at 25 °C for 2 h. After that, the mixture was poured into ice water (600 mL) and NaOH was added to adjust the pH to 8.0 was added to mixture. The crude product was filtered off and washed with water to obtain indole-3-carboxaldehyde (2).15General synthesis for 2-chloroacetamide derivatives 5a–n
A mixture of aniline derivatives 3a–n (2 mmol) and chloroacetyl chloride (4) (2 mmol) in DMF (10 mL) was stirred at room temperature for 24 h. The reaction mixture was then diluted with cold water and poured onto ice. The resulting precipitate was filtered, washed with cold water, and dried to afford pure 2-chloroacetamide derivatives 5a–n.16General synthesis for acetamide–indole–carboxylic acid derivatives 6a–n
A mixture of indole-3-carboxaldehyde (2) (1 mmol), 2-chloroacetamide derivatives 5a–n (1.2 mmol) in dry acetone (10 mL) and anhydrous K2CO3 (1 mmol) was stirred at room temperature for 4 h, filtered, and the resulting solid was recrystallized from ethanol to give the final products 6a–n.17General synthesis for acetamide–indole–benzo[d]imidazole–carboxylic acid derivatives 8a–n
Compounds 6a–n (1 mmol) and 3,4-diaminobenzoic acid (7, 1.2 mmol) along with a catalytic amount of Na2S2O5 were dissolved in DMF (10 mL) and the mixture was stirred at 150 °C for 24 h. Upon completion of the reaction (as monitored by TLC), H2O (20 mL) was added to the reaction mixture, it was filtered, and recrystallized to give the target compounds 8a–n.18PTP1B inhibition assay
The inhibitory activity of the newly synthesized compounds 8a–n against human recombinant PTP1B was assessed using a spectrophotometric assay in a microplate format. Each test compound was dissolved in PTP1B assay buffer, which consisted of 100 mM HEPES (pH 7.2), 2 mM EDTA, 2 mM DTT, and 0.1% nonylphenyl polyethylene glycol, with a final volume of 45 µL per well. Following a 10-minute pre-incubation at 30 °C, the enzymatic reaction was initiated by adding 5 µL of enzyme (final concentration: 3.5 ng mL−1) and 50 µL of the phosphopeptide substrate IR5 (0.75 µM). After 25 minutes of incubation at 30 °C, the reaction was terminated by the addition of 10 µL of a red reagent. The released orthophosphate, resulting from the dephosphorylation of IR5 by PTP1B, was quantified by measuring the absorbance at 620 nm after a 20-minute incubation. The known PTP1B inhibitor suramin was used as a positive control. For the IC50 determinations, all experiments were performed in triplicate (n = 3). The IC50 values were calculated by nonlinear regression using logarithmic dose–response curve fitting.α-Glucosidase inhibition assay
The inhibitory activity of the synthesized acetamide–indole–benzimidazole–carboxylic acid derivatives (8a–n) was evaluated against Saccharomyces cerevisiae α-glucosidase (EC 3.2.1.20). The enzyme was prepared in potassium phosphate buffer (PPB, 50 mM, pH 6.8), while the test compounds were dissolved in DMSO, achieving a final concentration of 10% in the assay mixture. The reaction was conducted in a 96-well plate, where a mixture of 135 µL of PPB, 20 µL of the enzyme solution, and 20 µL of a test compound at varying concentrations was pre-incubated for 10 minutes at 37 °C. The enzymatic reaction was then initiated by adding 25 µL of the substrate, p-nitrophenyl glucopyranoside (4 mM). After an additional 25 minutes of incubation at 37 °C, the absorbance was measured at 405 nm using a BioTek Gen5 PowerWave XS2 microplate reader. The release of p-nitrophenol was quantified to determine the inhibitory potential of the compounds. All experiments were performed in triplicate (n = 3) for the determination of IC50 values. These values were calculated by nonlinear regression using logarithmic dose–response curve fitting.Kinetic study against PTP1B
The inhibition type of the most active PTP1B inhibitor was investigated at different substrate concentrations (IR5, 0.075–0.750 µM) in the absence and presence of the inhibitor at various concentrations (0.6, 1.1, and 2.2 µM). Lineweaver–Burk plots were generated to determine the inhibition type, and the Michaelis–Menten constant (Km) was determined from the relationship between the reciprocal of substrate concentration (1/[S]) and the reciprocal of the enzyme reaction rate (1/V) across different inhibitor concentrations.Docking study in the active site of PTP1B
Docking studies against PTP1B (pdb code: 2QBS) were performed by Autodock Tools 1.5.6.19 The 3D structures of the selected compounds (the standard inhibitor, the co-crystallized ligand, and the most potent compounds) were constructed by MarvinSketch 5.10.4, and then the pdbqt format of these ligands were prepare by Autodock Tools 1.5.6. The enzyme structure was also prepared in pdbqt format using the same software. The docking grid box was set to 60 × 60 × 60 Å, centered at X = 49.28, Y = 12.106, Z = 4.338. Each docked system was carried out by 20 runs of the Autodock search (by the Lamarckian genetic algorithm). Finally, according to obtain BE values, the best-docked pose of each inhibitor was selected and analyzed by Discovery Studio 2019 Client (Accelrys, Inc., San Diego, CA).Molecular dynamics on PTP1B–ligand complexes
The molecular dynamics (MD) simulations of PTP1B–ligand complexes were performed using GROMACS 5.1.2 on Ubuntu 18.04.5 LTS.20 Topology and force field parameters for the ligands were generated using the SwissParam server, which provides parameter files compatible with the CHARMM all-atom force field for small organic molecules.21 The protein topology was generated using the pdb2gmx command in GROMACS with the CHARMM27 all-atom force field (CHARMM22 plus CMAP for proteins). The ligand topology files (.itp) generated by SwissParam were incorporated into the protein topology (.top) file, and the corresponding GROMACS-formatted (.gro) files of the protein and ligands were merged using Notepad++.The protein–ligand complex was centered within a cubic simulation box, ensuring a 1.0 nm clearance from the box edges. Solvation was carried out using the SPC216 water model, and 5 water molecules were replaced by Na+ ions to neutralize the net negative charge of the system. Energy minimization was conducted using the steepest descent algorithm for up to 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 steps, reducing the maximum force to below 10.0 kJ mol−1. The system underwent equilibration in two stages. NVT equilibration was performed at 300 K for 500 ps using the V-rescale thermostat with a coupling constant of 0.1 ps. This was followed by NPT equilibration for 1000 ps using the Berendsen pressure coupling algorithm with a coupling constant of 5.0 ps. Long-range electrostatic interactions were treated using the Particle-Mesh Ewald (PME) method with a cutoff of 1.0 nm for electrostatics. van der Waals interactions were calculated using a 1.2 nm cutoff. After these preparatory steps, a 20 ns MD simulation was performed to investigate the stability and dynamic behavior of the protein–ligand complex.
000 steps, reducing the maximum force to below 10.0 kJ mol−1. The system underwent equilibration in two stages. NVT equilibration was performed at 300 K for 500 ps using the V-rescale thermostat with a coupling constant of 0.1 ps. This was followed by NPT equilibration for 1000 ps using the Berendsen pressure coupling algorithm with a coupling constant of 5.0 ps. Long-range electrostatic interactions were treated using the Particle-Mesh Ewald (PME) method with a cutoff of 1.0 nm for electrostatics. van der Waals interactions were calculated using a 1.2 nm cutoff. After these preparatory steps, a 20 ns MD simulation was performed to investigate the stability and dynamic behavior of the protein–ligand complex.
In silico ADMET, drug-likeness, and bioavailability studies
The ADMET properties of the selected compounds were predicted in silico using the pkCSM online server.22 Furthermore, drug-likeness and oral bioavailability of these compounds were assessed with the admetSAR and SwissADME web tools, respectively.23,24Result and discussion
Chemistry
The synthetic pathway employed to access the new acetamide–indole–benzo[d]imidazole–carboxylic acid derivatives 8a–n is outlined in Scheme 1.15–18 The synthesis started with the Vilsmeier formylation of indole (1), providing indole-3-carboxaldehyde (2). Concurrently, a series of 2-chloroacetamide derivatives 5a–n were prepared by reacting various aniline derivatives 3a–n with chloroacetyl chloride (4). The subsequent step involved the reaction of indole-3-carboxaldehyde (2) with compounds 5a–n to give the acetamide–indole–carboxylic acid derivatives 6a–n. The final target compounds 8a–n were efficiently constructed via a reaction between compounds 6a–n and 3,4-diaminobenzoic acid (7). | ||
| Scheme 1 Synthetic procedure for acetamide–indole–benzo[d]imidazole–carboxylic acid derivatives 8a–n. | ||
Anti-PTP1B assay of new compounds 8a–n
The inhibitory activities of the acetamide–indole–benzo[d]imidazole–carboxylic acid derivatives 8a–n against human recombinant PTP1B were evaluated, and the corresponding IC50 values are summarized in Table 1. The results indicate a significant range of potency, with IC50 values varying from the micromolar to the inactive range (IC50 > 10 µM). Notably, compound 8l emerged as the most potent PTP1B inhibitor in this series (IC50 = 2.2 µM), displaying an approximately 4.5-fold increase in potency compared to the standard inhibitor, suramin (IC50 = 10.0 µM).| Compound | R | IC50 (µM) | Compound | R | IC50 (µM) |
|---|---|---|---|---|---|
| 8a | -Ph | 10.0 ± 0.2 | 8h | 2-F-Ph- | 3.1 ± 0.1 |
| 8b | 2-Me-Ph- | >10 | 8i | 3-Cl-Ph- | >10 |
| 8c | 3-Me-Ph- | >10 | 8j | 4-Cl-Ph- | 6.7 ± 0.3 |
| 8d | 4-Me-Ph- | 8.1 ± 0.2 | 8k | 4-Br-Ph- | >10 |
| 8e | 2,3-diMe-Ph- | >10 | 8l | 4-NO2-Ph- | 2.2 ± 0 |
| 8f | 2,4-diMe-Ph- | 3.8 ± 0.1 | 8m | –CH2-Ph | >10 |
| 8g | 4-Et-Ph- | 5.9 ± 0.1 | 8n | –CH2–CH2-Ph | >10 |
| Suramin | — | 10.0 ± 0.2 | Suramin | — | 10.0 ± 0.2 |
As can be seen in Scheme 1 and Table 1, most of the newly synthesized compounds feature an acetamide moiety substituted with various R groups. These R groups are phenyl derivatives in compounds 8a–l, a benzyl derivative in compound 8m, and a phenethyl derivative in compound 8n. As expected, the structure–activity relationship (SAR) analysis reveals that the nature of this R group is a critical determinant for the observed PTP1B inhibitory activity. SAR analysis of compounds 8a–n is schematically depicted in Fig. 2.
SAR analysis of compounds 8a–n showed that all active PTP1B inhibitors belong to the N-phenylacetamide series. The most potent compound was the 4-nitro derivative 8l (IC50 = 2.2 µM). As shown in Fig. 2, inhibitory potency for 4-substituted derivatives followed the order: NO2 > Et (8g, IC50 = 5.9 µM) > Cl (8j, IC50 = 6.7 µM) > Me (8d, IC50 = 8.1 µM) > Br (8k, inactive). All these substituted compounds, with the exception of 4-bromo derivative, were more active than the un-substituted analogue 8a (IC50 = 10.0 µM), underscoring the importance of the 4-position. This trend is further reinforced by the activity of the 2,4-dimethyl derivative 8f (IC50 = 3.8 µM), the third most potent compound. Here, the favorable 4-methyl group appears to offset the steric hindrance from the 2-methyl, while the mono-2-methyl analogue was inactive (8b). On the other hand, the inactivity of the 4-bromo analogue (8k) suggests that its larger atomic radius and potential steric crowding may impair optimal binding within the enzyme's active site. The second potent compound was 2-fluoro derivative 8h while other 2-substituted analogs like compound 8b and compound 8e (2,3-diMe) were inactive (IC50 > 10 µM), underscoring that the steric tolerance is highly specific to the very small fluorine atom. Both 3-substituted compounds, 3-methyl derivative 8b and 3-chloro derivative 8i were also inactive against PTP1B. In summary, the observed SAR in N-phenylacetamide for the synthesized derivatives demonstrates that, in addition to whether the substituents are electron-donating or electron-withdrawing, their precise positions on the phenyl ring critically govern the inhibitory potency.
Table 1 and Fig. 2 also demonstrate that replacing the rigid phenyl ring of compound 8a with flexible alkyl chains bearing a terminal phenyl group, as in the cases of compounds 8m (–CH2-Ph) and 8n (–CH2–CH2-Ph), completely abolishes activity.
Anti-α-glucosidase assay of new compounds 8a–n
Given the presence of known pharmacophores effective against α-glucosidase and the enzyme's key role in diabetes treatment, the newly synthesized compounds (8a–n) were evaluated for this activity. The obtained inhibition percentages are listed in Table 2. The data reveal that all new compounds were less potent than acarbose, except for the 4-bromo (compound 8k, 60% inhibition) and 4-chloro (compound 8j, 54% inhibition) derivatives.| Compound | R | % inhibition | IC50 (µM) | Compound | R | % inhibition | IC50 (µM) |
|---|---|---|---|---|---|---|---|
| 8a | -Ph | 1 ± 0.84 | >750 | 8h | 2-F-Ph- | 0 | >750 |
| 8b | 2-Me-Ph- | 5 ± 0.63 | >750 | 8i | 3-Cl-Ph- | 3 ± 0.27 | >750 |
| 8c | 3-Me-Ph- | 19 ± 0.44 | >750 | 8j | 4-Cl-Ph- | 54 ± 0.19 | 659.98 ± 1.26 |
| 8d | 4-Me-Ph- | 34 ± 0.57 | >750 | 8k | 4-Br-Ph- | 60 ± 0.32 | 551.43 ± 0.95 |
| 8e | 2,3-diMe-Ph- | 0 | >750 | 8l | 4-NO2-Ph- | 40 ± 0.21 | >750 |
| 8f | 2,4-diMe-Ph- | 6 ± 0.58 | >750 | 8m | –CH2-Ph | 0 | >750 |
| 8g | 4-Et-Ph- | 28 ± 0.42 | >750 | 8n | –CH2–CH2-Ph | 40 ± 0.73 | >750 |
| Acarbose | — | 50 ± 0.12 | 750.3 ± 0.67 | Acarbose | — | 50 ± 0.12 | 750.3 ± 0.67 |
Analysis of the structure–activity relationship (SAR) for compounds 8a–l (R = phenyl derivatives) indicates significant steric hindrance at the 2-position of the phenyl ring. Any substitution at this position, as in compounds 8b (2-Me), 8e (2,3-diMe), 8f (2,4-diMe), and 8h (2-F), leads to inactivity or a significant loss of potency. In contrast, 4-substituted derivatives consistently exhibited moderate to good activity, with the 4-Br (compound 8k, 60% inhibition, IC50 = 551.43 µM), 4-Cl (compound 8j, 54% inhibition, IC50 = 659.98 µM), and 4-NO2 (compound 8l, 40% inhibition) groups being particularly favorable.
The data further demonstrate that both the N-phenylacetamide (compound 8a) and N-benzylacetamide (compound 8m) derivatives were inactive against α-glucosidase. Conversely, the N-phenethylacetamide derivative (compound 8n, 40% inhibition) exhibited moderate activity against this enzyme.
Comparison of SAR between anti-PTP1B and anti-α-glucosidase assays
As shown in Table 1, the most potent compound against PTP1B is the 4-nitro derivative 8l. Interestingly, this same compound is also the third most potent against α-glucosidase. The top two inhibitors of α-glucosidase are the 4-bromo and 4-chloro derivatives, 8k and 8j, respectively. Notably, while the 4-chloro derivative (8j) exhibits clear activity against PTP1B, its bromo analogue (8k) shows no significant inhibition of this enzyme. Overall, this comparative analysis suggests that electron-withdrawing groups enhance inhibitory activity against both enzymes, although with distinct selectivity profiles influenced by the specific halogen substituent.Kinetic study against PTP1B
The kinetic analysis of compound 8l against PTP1B was performed and its inhibition mechanism was determined by the characteristic Lineweaver–Burk plot pattern (Fig. 3a). This plot pattern indicates that 8l binds directly to the enzyme's active site and competes with the substrate. The inhibition constant (Ki) was determined to be 2.2 µM (Fig. 3b). The competitive nature of compound 8l, along with its high inhibitory potency, suggests that it effectively inhibits PTP1B catalytic activity by blocking substrate access to the active site.Docking study in the active site of PTP1B
Molecular docking was performed to explore the binding modes of the newly synthesized potent compounds within the active site of PTP1B. The crystal structure of PTP1B (PDB ID: 2QBS) was obtained from the Protein Data Bank (PDB) for this purpose. The co-crystallized ligand in this PTP1B structure is 4-bromo-3-(carboxymethoxy)-5-[3-(cyclohexylamino)phenyl]thiophene-2-carboxylic acid (listed as compound 024 in the PDB). For docking validation, this native ligand was re-docked. Subsequently, our novel compounds and the standard inhibitor, suramin, were docked into the active site.Fig. 4a illustrates the superimposed conformer of the native co-crystallized ligand (compound 024), the standard inhibitor suramin, and the most potent synthesized compound (8l). Interaction modes of these compounds are shown in Fig. 4b and c.
 | ||
| Fig. 4 (a) Superimposed conformer of compounds 024 (cyan), suramin (blue), and 8l (pink). Interaction modes of compounds 024 (b), suramin (c), and 8l (d) in the PTP1B's active site. | ||
As can be seen in Fig. 4b, compound 024 established six hydrogen bonds with residues Ser215, Cys214, Arg220, Phe181, Gln265, and Gln261. This ligand formed an unfavorable interaction with Arg220. Compound 024 also established a π–sigma interaction with Ala216 and several hydrophobic interactions with Val48, Ilu218, Ala216, and Met257. The binding energy (BE) value of compound 024 in the active site of PTP1B was −10.89 kcal mol−1.
Suramin established five classical hydrogen bonds with residues Ser27, Arg253, Gly258, Asp47, and Ala263 (Fig. 4c) and two non-classical hydrogen bonds with Lys119 and Gly182. Suramin also formed the following π-interactions: a π–cation with Arg23, a π–sigma with Thr262, two π–π interactions with Phe181, and two π–alkyl interactions with Ala216 and Tye45. BE value of suramin in the active site of PTP1B was −7.07 kcal mol−1.
2D-interaction mode of the most potent compound 8l was shown in Fig. 4d, as can be seen in this figure, compound 8l established six conventional hydrogen bonds with Cys214, Arg220, Arg253 (two interactions), Gln265, and Gln261 and a non-classical hydrogen bond with Gly219. This compound also formed π–π interactions Met257 (π–sulfur), Phe181 (π–π), Tyr45 (π–π), Val48 (a π–sigma and a π–alkyl), Ile218 (π–sigma), and Ala216 (π–alkyl). The BE value of compound 8l in the active site of PTP1B was −11.72 kcal mol−1.
The comparative analysis of binding energies highlights compound 8l as the most potent binder, with a BE value lower than those of the native ligand 024 and the reference inhibitor suramin, indicating its enhanced inhibitory efficacy.
As can be seen in Table 1, the second and third most potent compounds against PTP1B were compounds 8h and 8f, respectively. The interaction modes of these compounds are shown in Fig. 5. Compound 8h formed six hydrogen bonds with residues Cys214, Asp47, Arg220, Arg46, Gln265, and Phe181. This compound established the following π interactions with the PTP1B active site: three π–π interactions with Phe181 (two interaction) and Tyr45, three π–alkyl interactions with Val48 (two interaction) and Ala216, a π–cation interaction with Arg46, and a π–sigma with Ala216. The BE of this compound was −9.92 kcal mol−1. Compound 8f formed five conventional hydrogen bonds Glu114, Ser215, Arg220, Arg23, and Gln261 and a non-classical hydrogen bond with Arg23. This compound also formed one π–sigma with Ala216 and two π–π interactions with Phe181. Four hydrophobic interaction were also observed between compound 8f and residues Ile218 (two interactions), Met257, and Ala216. The BE value of this compound was −9.6 kcal mol−1.
Computational and experimental data were in strong concordance, as the BE rankings (suramin > 8f > 8h > 8l) predicted by the docking study directly correlated with the potency order observed in the in vitro assays (8l > 8h > 8f > suramin).
Molecular dynamics
The interaction between a substrate and the active site of an enzyme is a highly dynamic process, typically occurring within nanoseconds. To gain meaningful insights into the conformational stability and adaptability of receptor–ligand complexes, it is essential to investigate their behavior in a biologically realistic context that includes explicit water molecules and ions. In this study, molecular dynamics (MD) simulations were employed using the docking structures of suramin (a well-known reference inhibitor), compound 024 (the ligand observed in the co-crystal structure), and compound 8l (the most effective PTP1B inhibitor identified in vitro). These simulations, conducted in a fully solvated environment, aimed to assess the stability, conformational flexibility, and molecular interactions between the target protein and the selected ligands over the course of the simulation.In this study, a two-stage MD simulation approach was implemented to evaluate the stability of various ligands bound to the active site of PTP1B. In the first stage, each ligand–enzyme complex underwent a 10 nanoseconds simulation, which demonstrated that the ligands retained stable associations within the binding pocket. To gain deeper insight into their dynamic properties, the simulations were prolonged by an additional 10 nanoseconds. Throughout this extended phase, suramin, 024, and 8l consistently exhibited conformational stability within the active site. The resulting trajectory data were then thoroughly analyzed to elucidate the molecular interactions and structural dynamics characterizing each ligand–enzyme complex.
To evaluate the conformational stability of the protein–ligand complexes, root-mean-square deviation (RMSD) and radius of gyration (Rg) analyses were systematically conducted across all simulation trajectories. These parameters were monitored over time, and corresponding plots were generated to visualize structural deviations and variations in molecular compactness throughout the simulation period. Furthermore, root-mean-square fluctuation (RMSF) analysis was carried out on the backbone atoms of PTP1B to assess residue-level flexibility. Together, these evaluations provided valuable insights into the structural integrity of the enzyme and the dynamic behavior of the ligands within the active site, emphasizing the nature of their interactions and the adaptability of the complexes under simulated physiological conditions.
Fig. 6 presents the results of the RMSD analysis, demonstrating the structural stability of the PTP1B–ligand complexes throughout the simulation period. The backbone RMSD values of PTP1B in complex with suramin, 024, and 8l exhibited only minor fluctuations, consistently remaining below 0.25 Å, indicative of the enzyme's preserved structural integrity. The mean RMSD values for PTP1B were calculated to be 1.36 Å in the suramin complex and 1.11 Å in the 024 complex, while the value for the 8l complex was 1.51 Å. The ligands also maintained stable binding conformations, with RMSD values below 0.25 Å during the entire simulation. Specifically, the average RMSD values recorded were 1.45 Å for suramin, 0.87 Å for 024, and 1.20 for 8l. These results collectively support the conclusion that both the enzyme and ligands retained significant structural stability under the simulated conditions.
Protein stability during MD simulations is often assessed by examining structural compactness, typically measured by the radius of gyration (Rg). In the case of PTP1B, Rg values were computed throughout the simulation trajectory to evaluate changes in the protein's compactness, as depicted in Fig. 6. The average Rg for the suramin-bound complex was 19.10 Å, whereas the corresponding values for the 024 and 8l complexes were 19.07 Å and 19.19 Å, respectively. Across all simulations, the Rg values exhibited minimal variation, fluctuating within a narrow range of 18.73 to 19.41 Å. This consistency suggests that PTP1B retained a stable and compact structure throughout the simulation period, reflecting its conformational integrity under the modeled conditions.
The structural and dynamic evaluation of PTP1B offers valuable insight into the enzyme's flexibility and ligand-binding properties (Fig. 7). PTP1B is a single-domain protein characterized by a clearly defined catalytic core, which consists of a central β-sheet flanked by α-helices and flexible loop regions. The catalytic cleft, which functions as the primary ligand-binding site, is located between critical structural elements, notably the WPD loop and the P-loop, both essential for enzymatic function. The WPD loop, encompassing residues 177 to 183 and including the catalytically significant Asp180, plays a central role in modulating enzymatic activity. Meanwhile, the P-loop, which facilitates phosphate binding, includes key residues such as Arg46, Lys119, Ser117, and those within positions 44 to 49. Ligands like compound 024 occupy this cleft, forming stabilizing interactions with conserved amino acids and inducing localized structural adjustments. Peaks observed in the RMSF analysis align with flexible segments near the active site, particularly the WPD and P-loops. These regions display moderate mobility, consistent with their functional roles in ligand accommodation and catalysis. The similarity in RMSF profiles across all three ligand-bound systems suggests that the observed dynamic behavior is driven by their common binding location rather than ligand-specific effects. This finding underscores the structural resilience and functional adaptability of the PTP1B catalytic site in interacting with structurally diverse ligands.
In silico ADMET profiling and evaluation of drug-likeness
In this section, we employed the online pkCSM platform to perform a comprehensive in silico prediction of the absorption, distribution, metabolism, excretion, and toxicity (ADMET) properties of our most potent newly synthesized PTP1B inhibitor, compound 8l, in direct comparison with suramin (Table 3).| Property | Model name | Suramin | 8l | Unit |
|---|---|---|---|---|
| Absorption | Water solubility | −2.892 | −2.892 | Numeric (log mol L−1) |
| Caco2 permeability | −3.531 | −1.013 | Numeric (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Papp in 10−6 cm s−1) Papp in 10−6 cm s−1) |
|
| Intestinal absorption (human) | 0 | 73.764 | Numeric (% absorbed) | |
| Skin permeability | −2.735 | −2.735 | Numeric (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Kp) Kp) |
|
| P-glycoprotein substrate | No | Yes | Categorical (yes/no) | |
| P-glycoprotein I inhibitor | No | No | Categorical (yes/no) | |
| P-glycoprotein II inhibitor | No | Yes | Categorical (yes/no) | |
| Distribution | VDss (human) | −0.017 | 0.006 | Numeric (log L kg−1) |
| Fraction unbound (human) | 0.378 | 0.382 | Numeric (Fu) | |
| BBB permeability | −3.905 | −1.185 | Numeric (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) BB) BB) |
|
| CNS permeability | −5.037 | −2.309 | Numeric (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PS) PS) |
|
| Metabolism | CYP2D6 substrate | No | No | Categorical (yes/no) |
| CYP3A4 substrate | No | No | Categorical (yes/no) | |
| CYP1A2 inhibitor | No | Yes | Categorical (yes/no) | |
| CYP2C19 inhibitor | No | No | Categorical (yes/no) | |
| CYP2C9 inhibitor | No | Yes | Categorical (yes/no) | |
| CYP2D6 inhibitor | No | No | Categorical (yes/no) | |
| CYP3A4 inhibitor | No | No | Categorical (yes/no) | |
| Excretion | Total clearance | −4.274 | 0.538 | Numeric (log mL min−1 kg−1) |
| Renal OCT2 substrate | No | No | Categorical (yes/no) | |
| Toxicity | AMES toxicity | No | No | Categorical (yes/no) |
| Max. tolerated dose (human) | 0.438 | 0.439 | Numeric (log mg per kg per day) | |
| hERG I inhibitor | No | No | Categorical (yes/no) | |
| hERG II inhibitor | Yes | No | Categorical (yes/no) | |
| Oral rat acute toxicity (LD50) | 2.482 | 2.482 | Numeric (mol kg−1) | |
| Oral rat chronic toxicity (LOAEL) | 7.327 | 3.123 | Numeric (log mg per kg_bw per day) | |
| Hepatotoxicity | No | Yes | Categorical (yes/no) | |
| Skin sensitisation | No | No | Categorical (yes/no) | |
| T. pyriformis toxicity | 0.285 | 0.285 | Numeric (log µg L−1) | |
| Minnow toxicity | 5.545 | 1.114 | Numeric (log mM) |
The predicted absorption profile of compound 8l suggests potential improvements over suramin. Both compounds are predicted to have identical, moderately low water solubility (−2.892![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log mol L−1), which could pose formulation challenges. A notable predicted advantage for compound 8l is its markedly higher Caco-2 permeability (−1.013
log mol L−1), which could pose formulation challenges. A notable predicted advantage for compound 8l is its markedly higher Caco-2 permeability (−1.013![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log
log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Papp vs. −3.531 for suramin), indicating a greater potential for oral absorption. This is aligned with the predicted human intestinal absorption, where compound 8l is forecasted to be 73.764% absorbed, compared to 0% for suramin. This difference suggests that compound 8l might have better oral bioavailability compared to suramin, which is known to be poorly absorbed orally and administered intravenously in the clinic. It should be noted that compound 8l is predicted to be a potential substrate for P-glycoprotein (P-gp), which could actively efflux it from cells, potentially counteracting its high permeability. Furthermore, compound 8l is identified as a potential P-gp II inhibitor, indicating a possible risk of drug–drug interactions (DDIs) with co-administered P-gp substrates.
Papp vs. −3.531 for suramin), indicating a greater potential for oral absorption. This is aligned with the predicted human intestinal absorption, where compound 8l is forecasted to be 73.764% absorbed, compared to 0% for suramin. This difference suggests that compound 8l might have better oral bioavailability compared to suramin, which is known to be poorly absorbed orally and administered intravenously in the clinic. It should be noted that compound 8l is predicted to be a potential substrate for P-glycoprotein (P-gp), which could actively efflux it from cells, potentially counteracting its high permeability. Furthermore, compound 8l is identified as a potential P-gp II inhibitor, indicating a possible risk of drug–drug interactions (DDIs) with co-administered P-gp substrates.
The predicted volume of distribution (VDss) for compound 8l (0.006![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log L kg−1) is slightly higher than that of suramin (−0.017
log L kg−1) is slightly higher than that of suramin (−0.017![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log L kg−1), which could suggest a somewhat broader tissue distribution for the new compound. The fraction unbound (Fu) in plasma is nearly identical (∼0.38), implying a similar extent of plasma protein binding for both. A particularly promising prediction is the significantly enhanced Blood–Brain Barrier (BBB) permeability of compound 8l (−1.185
log L kg−1), which could suggest a somewhat broader tissue distribution for the new compound. The fraction unbound (Fu) in plasma is nearly identical (∼0.38), implying a similar extent of plasma protein binding for both. A particularly promising prediction is the significantly enhanced Blood–Brain Barrier (BBB) permeability of compound 8l (−1.185![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log
log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) BB) compared to suramin (−3.905
BB) compared to suramin (−3.905![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log
log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) BB). However, it is important to highlight that BBB permeability predictions from models like pkCSM are statistical and do not equate to established CNS bioavailability. Similarly, the predicted CNS permeability (log
BB). However, it is important to highlight that BBB permeability predictions from models like pkCSM are statistical and do not equate to established CNS bioavailability. Similarly, the predicted CNS permeability (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PS) of compound 8l (−2.309) is substantially higher than that of suramin (−5.037).
PS) of compound 8l (−2.309) is substantially higher than that of suramin (−5.037).
In terms of metabolism, both compound 8l and suramin are predicted not to be substrates for the major cytochrome P450 enzymes CYP2D6 and CYP3A4, which could reduce the likelihood of metabolism-mediated DDIs for both compounds. However, a key metabolic difference lies in the predicted inhibition profile. Compound 8l is predicted to potentially inhibit CYP1A2 and CYP2C9, while suramin is not.
The predicted total clearance of compound 8l (0.538![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log mL min−1 kg−1) is significantly higher than that of suramin (−4.274
log mL min−1 kg−1) is significantly higher than that of suramin (−4.274![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log mL min−1 kg−1), indicating a potentially faster elimination rate for the new compound. Neither compound is predicted to be a substrate for the renal organic cation transporter 2 (OCT2), minimizing potential concerns for specific renal transporter-mediated interactions or accumulations.
log mL min−1 kg−1), indicating a potentially faster elimination rate for the new compound. Neither compound is predicted to be a substrate for the renal organic cation transporter 2 (OCT2), minimizing potential concerns for specific renal transporter-mediated interactions or accumulations.
The toxicity predictions present a mixed profile for compound 8l. On the positive side, compound 8l is predicted to be negative for AMES toxicity and skin sensitization, similar to suramin. Notably, compound 8l is not predicted to be an inhibitor of the hERG II channel, whereas suramin is a predicted hERG II inhibitor. This suggests that compound 8l may carry a lower potential risk of cardiotoxicity (QT interval prolongation) compared to suramin. However, a significant concern is the prediction of hepatotoxicity for compound 8l, which was not flagged for suramin. This potential liability requires further experimental validation in subsequent in vitro and in vivo studies. The predicted chronic toxicity potential, as indicated by the Lower Oral Rat Chronic Toxicity (LOAEL) value for compound 8l (3.123![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log mg per kg_bw per day) compared to suramin (7.327
log mg per kg_bw per day) compared to suramin (7.327![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log mg per kg_bw per day), also warrants careful long-term toxicity assessment. The predicted reduced minnow toxicity value for compound 8l (1.114
log mg per kg_bw per day), also warrants careful long-term toxicity assessment. The predicted reduced minnow toxicity value for compound 8l (1.114![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log mM) compared to suramin (5.545
log mM) compared to suramin (5.545![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log mM) indicates a potentially lower environmental aquatic toxicity.
log mM) indicates a potentially lower environmental aquatic toxicity.
Drug-likeness study on compound 8l and suramin was performed by admetSAR online software. The obtained data were illustrated in Fig. 8. As can be seen in this figure, compound 8l, shows a higher presence within the drug-likeness acceptable region compared to suramin.
The bioavailability radar plots of suramin and compound 8l are shown in Fig. 9. The radar plot for compound 8l appears more balanced and lies predominantly within the desirable range (pink area), whereas the plot for suramin shows significant deviations outside the ideal zone. This graphical analysis confirms that compound 8l possesses a more favorable drug-like profile compared to suramin.
Conclusion
In conclusion, a novel series of acetamide–indole–benzo[d]imidazole–carboxylic acid hybrids (8a–n) was successfully designed and synthesized as potential dual-target inhibitors of PTP1B and α-glucosidase. While the majority of compounds exhibited promising PTP1B inhibitory activity, with six derivatives outperforming the standard inhibitor suramin, only two compounds showed marginally improved α-glucosidase inhibition compared to acarbose. Compound 8l emerged as the most potent PTP1B inhibitor, acting through a competitive mechanism, as confirmed by kinetic analysis. Molecular docking and dynamics simulations supported its stable binding within the PTP1B active site, aligning well with the in vitro results. Although the dual inhibitory profile was only partially achieved, the series represents a significant step toward the development of novel PTP1B-targeted therapeutics. Further structural optimization based on the acetamide–indole–benzo[d]imidazole scaffold may yield improved dual-target agents for the management of diabetes and related metabolic disorders.Author contributions
MM and MM-K conceived, designed, and supervised this study. ND, SNG, MM (Marjan Mollazadeh), SH, and NMS synthesized and interpreted analytical data of compounds. In vitro experiments were performed by SM, MAF, and BL. In silico studies were performed and written by MH and MA. The manuscript was drafted by MM-K. MM reviewed and edited the drafts.Conflicts of interest
The authors declare no conflict of interest.Data availability
The data supporting this article have been included as part of the Supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5ra07935g.Acknowledgements
This research work was financially supported by the National Institute for Medical Research Development (NIMAD; Grant Number: 4000259).References
- A. Artasensi, A. Pedretti, G. Vistoli and L. Fumagalli, Molecules, 2020, 25, 1987 CrossRef CAS PubMed.
- Y. Kong, H. Yang, R. Nie, X. Zhang, H. Zhang and X. Nian, Eur. J. Med. Res., 2025, 30, 477 CrossRef PubMed.
- M. Genovese, I. Nesi, A. Caselli and P. Paoli, Molecules, 2021, 26, 4818 CrossRef CAS PubMed.
- M. Teimouri, H. Hosseini, Z. ArabSadeghabadi, R. Babaei-Khorzoughi, S. Gorgani-Firuzjaee and R. Meshkani, J. Physiol. Biochem., 2022, 78, 307–322 Search PubMed.
- E. Di Stefano, T. Oliviero and C. C. Udenigwe, Curr. Opin. Food Sci., 2018, 20, 7–12 CrossRef.
- S. Rocha, I. Santos, M. L. Corvo, E. Fernandes and M. Freitas, Food Funct., 2025, 16, 5231–5277 RSC.
- B. Göke, C. Herrmann, R. Göke, H. C. Fehmann, P. Berghöfer, G. Richter and R. Arnold, Eur. J. Clin. Invest., 1994, 24, 25–30 CrossRef PubMed.
- X. H. Makhoba, C. Viegas, R. A. Mosa, F. P. Viegas and O. J. Pooe, Drug Des., Dev. Ther., 2020, 14, 3235–3249 CrossRef CAS PubMed.
- F. F. Lillich, J. D. Imig and E. Proschak, Front. Pharmacol., 2021, 11, 554961 Search PubMed.
- P. de Sena Murteira Pinheiro, L. S. Franco, T. L. Montagnoli and C. A. M. Fraga, Expert Opin. Drug Discovery, 2024, 19, 451–470 Search PubMed.
- M. I. Campos-Almazan, M. Flores-Ramos, A. Hernández-Campos, R. Castillo, E. Sierra-Campos, K. Torgeson, W. Peti, M. Valdez-Solana, J. Oria-Hernández, S. T. Mendez and A. Castillo-Villanueva, Bioorg. Med. Chem., 2021, 48, 116418 Search PubMed.
- J. Liu, Y. Chen, J. Y. Li, C. Luo, J. Li, K. X. Chen, X. W. Li and Y. W. Guo, Mar. Drugs, 2018, 16, 97 Search PubMed.
- N. Asemanipoor, M. Mohammadi-Khanaposhtani, S. Moradi, M. Vahidi, M. Asadi, M. A. Faramarzi, M. Mahdavi, M. Biglar, B. Larijani, H. Hamedifar and M. H. Hajimiri, Bioorg. Chem., 2020, 95, 103482 CrossRef CAS PubMed.
- M. H. Sayahi, S. Zareei, M. Halimi, M. Alikhani, A. Moazzam, M. Mohammadi-Khanaposhtani, S. Mojtabavi, M. A. Faramarzi, H. Rastegar, P. Taslimi and E. H. Ibrahim, Sci. Rep., 2024, 14, 15791 CrossRef CAS PubMed.
- A. M. Taherkhani, M. H. Sayahi, B. Hassani, N. Dastyafteh, M. Mohammadi-Khanaposhtani, E. Rafiei, M. Meshkani, S. Safapoor, M. M. Tehrani, B. Larijani and M. Mahdavi, J. Mol. Struct., 2025, 1336, 142089 CrossRef CAS.
- A. S. Shokouhi Asl, M. H. Sayahi, M. H. Hashempur, C. Irajie, A. H. Alaeddini, S. N. Ghafouri, M. Noori, N. Dastyafteh, J. Mottaghipisheh, M. Asadi and B. Larijani, Sci. Rep., 2025, 15, 655 CrossRef CAS PubMed.
- M. K. Ghomi, N. Dastyafteh, M. N. Montazer, M. Noori, S. Mojtabavi, M. A. Faramarzi, S. M. Hashemi and M. Mahdavi, Sci. Rep., 2023, 13, 501 CrossRef PubMed.
- N. Dastyafteh, M. Negahdaripour, M. H. Sayahi, M. Emami, Y. Ghasemi, E. Safaei, H. Azizian, Z. P. Jahromi, M. Asadi, M. R. Mohajeri-Tehrani and F. Zare, RSC Adv., 2024, 14, 35323–35335 RSC.
- W. S. Liu, R. R. Wang, H. Yue, Z. H. Zheng, X. H. Lu, S. Q. Wang, W. L. Dong and R. L. Wang, J. Biomol. Struct. Dyn., 2020, 38, 3814–3824 CrossRef CAS PubMed.
- M. J. Abraham, T. Murtola, R. Schulz, S. Páll, J. C. Smith, B. Hess and E. Lindahl, SoftwareX, 2015, 1, 19–25 CrossRef.
- V. Zoete, M. A. Cuendet, A. Grosdidier and O. Michielin, J. Comput. Chem., 2011, 32, 2359–2368 CrossRef CAS PubMed.
- D. E. Pires, T. L. Blundell and D. B. Ascher, J. Med. Chem., 2015, 58, 4066–4072 Search PubMed.
- H. Yang, C. Lou, L. Sun, J. Li, Y. Cai, Z. Wang, W. Li, G. Liu and Y. Tang, Bioinformatics, 2019, 35, 1067–1069 Search PubMed.
- A. Daina, O. Michielin and V. Zoete, Sci. Rep., 2017, 7, 42717 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2026 |