 Open Access Article
Open Access ArticleImproved electrical and bioactivities of lead-free BNT–SBT ceramics by STZ additive and post-sintering processes
Kamonporn Saenkama,
Waraporn Boontakama,
Phanrawee Sripraphaa,
Pichitchai Butnoib,
Kamonpan Pengpata,
Chamnan Randornce and
Gobwute Rujijanagul *ade
*ade
aDepartment of Physics and Materials Science, Faculty of Science, Chiang Mai University, Chiang Mai 50200, Thailand. E-mail: gobwute.ruji@cmu.ac.th
bDepartment of Metallurgical Technology, Faculty of Technical Education, Rajamangala University of Technology Krungthep, Bangkok 10120, Thailand
cDepartment of Chemistry, Faculty of Science, Chiang Mai University, Chiang Mai, 50200, Thailand
dScience and Technology Research Institute, Chiang Mai University, Chiang Mai 50200, Thailand
eMaterials Science Research Center, Faculty of Science, Chiang Mai University, Chiang Mai 50200, Thailand
First published on 19th January 2026
Abstract
This study examined the effects of Sr(Ti0.85Zr0.15)O3 (STZ) additive and post-sintering treatments on the electrical and biological properties of lead-free (1 − x)[0.7(Bi0.5Na0.5)TiO3–0.3(Sr0.7Bi0.2)TiO3]–xSr(Ti0.85Zr0.15)O3 or ((1 − x)(BNT–SBT)–xSTZ) ceramics synthesized by solid-state reaction. All compositions showed coexistence of rhombohedral and tetragonal phases, with increased STZ promoting the rhombohedral phase. The x = 0.15 composition exhibited favorable results, featuring a broad temperature coefficient of capacitance (TCC) stability range (±15% from 38–310 °C), a 43% increase in energy storage density, 92.90% energy efficiency, strong breakdown strength (95 kV cm−1), and excellent thermal stability, with only 1.48% energy density variation between 25 and 125 °C. Its initial electrostrain of 0.06% was notably low. Post-sintering aging enhanced electrostrain to 0.33% representing a 450% increase and significantly improving electromechanical response. Cytotoxicity testing confirmed excellent cell viability, and bioactivity in simulated body fluid, initially moderate, was notably enhanced by β-tricalcium phosphate surface coating. These results highlight the biomedical potential of the optimized x = 0.15 ceramic composition.
1. Introduction
Due to increasing environmental concerns over the toxicity and volatility of lead oxide found in many lead-based piezoelectric ceramics, recent research has shifted toward the development of lead-free alternatives. Materials such as BaTiO3 (BT), (Bi0.5K0.5)TiO3 (BKT), K0.5Na0.5NbO3 (KNN), and Bi0.5Na0.5TiO3 (BNT) have gained considerable attention for their promising electrical properties.1–3 Among these, BNT-based ceramics stand out for their excellent ferroelectric behavior,4,5 typically exhibiting high maximum polarization (∼43 µC cm−2), attributed to the hybridization between Bi 6s and O 2p orbitals.2,6,7 However, BNT ceramics also suffer from a large coercive field (∼73 kV cm−1)2,6,7 and high electrical conductivity, which hinder effective polarization during the poling process and limit their practical applications.8,9 To improve the properties of BNT, various modifications and complex solid solutions have been explored. Furthermore, modified BNT-based systems such as BNT–BKT (BNKT),1–3 BNT–Ba(Al0.5Sb0.5)O3,10 BNT–BKT–SrTiO3,11 BNT–BT–KNN,12 BNT–BKT–Ba(Fe0.5Nb0.5)O3,13 and BNT–BT–(Sr0.7Bi0.2)TiO3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 14 and BNKT–Ba(Nb0.01Ti0.99)O3–BiFeO3
14 and BNKT–Ba(Nb0.01Ti0.99)O3–BiFeO3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 have demonstrated excellent strain behavior, making them strong candidates for electromechanical applications.1,2
15 have demonstrated excellent strain behavior, making them strong candidates for electromechanical applications.1,2
For energy storage applications, the induced relaxor state in BNT-based materials has been shown to significantly improve their performance.3,16 Therefore, various composition design strategies have been employed by incorporating materials such as NaTaO3,17 BiTi0.5Zn0.5O3,18 Bi(Zn2/3Nb1/3)O3,19 Sr0.7Bi0.2TiO3,20 NaNbO3,21 and AgNbO3,22 and Sr(Ti0.85Zr0.15)O3.23 Additionally, BNT–(Sr0.7Bi0.2)TiO3 (or BNT–SBT) based ceramics also exhibit high remanent polarization.24 Furthermore, recent studies suggest the BNT–SBT based ceramics also offer a promising energy storage density.4,20,25 Therefore, modifying BNT–SBT ceramics with other materials may further enhance their overall properties.
Recently, Sr(ZrxTi1−x)O3 ceramics have attracted considerable research interest due to their notable dielectric properties.26 This ceramic system is developed by substituting Ti4+ with Zr4+ at the B-site of the ABO3 perovskite structure.27–29 The larger ionic radius of Zr4+ induces lattice distortion, which enhances the dielectric characteristics of the ceramic. Moreover, partial substitution of Zr4+ (up to 5 mol%) significantly improves dielectric breakdown strength (up to 14.4 kV cm−1), making these ceramics a promising candidate for high-voltage capacitor applications.27 Specifically, the composition of STZ has been employed as a modifier in BNT-based ceramics to reduce remanent polarization (Pr) and enhance breakdown strength. These modifications contribute to improved energy storage density, higher efficiency, and overall enhancement of dielectric performance.23
Since BNT–SBT ceramics exhibit promising electrical properties, and the incorporation of STZ has been shown to enhance the performance of BNT-based ceramics, it is worthwhile to investigate the BNT–SBT system modified with STZ. In this study, BNT–SBT–STZ ceramics were fabricated, and their properties were investigated, including phase formation, microstructure, dielectric behavior, energy density properties, and electrostrain performance. However, it is important to note that while promoting a relaxor state can effectively enhance certain electrical properties, such as energy storage density, a higher degree of relaxor behavior often leads to a reduction in electrostrain performance. To overcome this trade-off, many post-sintering treatments have been proposed to optimize the material properties.1,2 Therefore, in this study, the samples were subjected to an aging treatment after sintering. Additionally, with the growing interest in piezoelectric materials for medical applications,30,31 several lead-free piezoceramics have been developed to explore their potential for use in the human body, offering both biocompatibility and favorable electrical properties.30–34 Therefore, the biological properties of the fabricated ceramics were also evaluated in this study. To enhance their bioactivity, the ceramics were coated with a bioactive material, and their bioactive performance was subsequently evaluated.
2. Experimental procedure
The (1 − x)[0.7(Bi0.5Na0.5)TiO3–0.3(Sr0.7Bi0.2)TiO3]–xSr(Ti0.85Zr0.15)O3 or ((1 − x)[BNT–SBT]–xSTZ) ceramics with x = 0, 0.05, 0.10, 0.15, and 0.20 were synthesized via the conventional solid-state route. The high-purity reagent-grade powders used in this work were BiO3 (99.9%, average particle size 10 µm, Sigma-Aldrich), NaCO3 (99.5%, average particle size 9 µm, Sigma-Aldrich), TiO2 (99%, average particle size 2–3 µm, Sigma-Aldrich), SrCO3 (98%, average particle size 2 µm, Sigma-Aldrich), and ZrO2 (99%, average particle size 5 µm, Sigma-Aldrich). The raw powders were dried at 120 °C for 24 h to remove moisture. The powders were then weighed according to the stoichiometric formula of (1 − x)[BNT–SBT]–xSTZ, ball-milled in 99.9% ethanol for 24 h, dried, and subsequently calcined at 850 °C for 3 h in air. After adding 4 wt% polyvinyl alcohol (PVA) as a binder, the powders were uniaxially pressed into 10 mm pellets and sintered at 1150 °C for 3 h (heating rate: 5 °C min−1) in closed alumina crucibles. Selected samples were subjected to post-sintering aging at 120 °C in air for two weeks. Additionally, selected samples were coated with beta-tricalcium phosphate (β-TCP) using a dip-coating technique. For the dip-coating process, a slurry consisting of β-TCP powder, PVA binder, deionized water, and starch was prepared and thoroughly mixed until homogeneous. This slurry was applied to ceramic samples with x = 0.15. The samples were dipped and withdrawn at a constant speed, then dried at 120 °C for 24 h and calcined at 1000 °C for 1 h at a heating rate of 10 °C min−1.Bulk density was measured using Archimedes' method. Phase identification was performed using X-ray diffraction (XRD, RIGAKU, SMARTLAB), and Raman spectroscopy (T6400 JY, Horiba Jobin Yvon) was used to confirm structural features. Microstructure was examined using a scanning electron microscopy (SEM, JEOL JSM-6335F). Silver electrodes were applied to both sides of the samples and fired at 700 °C for 15 min. Dielectric properties were measured from 25 °C to 500 °C using an LCR meter (HP 4192A) at 1–1000 kHz. Polarization (P) and electrostrain (S) loops as a function of applied field (E) were recorded using a Radiant Precision High Voltage Interface at 25–150 °C, with E = 50–95 kV cm−1 at 1 Hz. From the measurements, energy storage density properties were calculated from the P–E hysteresis loop.
In vitro cytotoxicity was assessed using the MTT assay on human skin fibroblast cells obtained from Manose Health and Beauty Research Center (Thailand). The cells were cultured in DMEM with 10% FBS and 1% penicillin/streptomycin at 37 °C, 5% CO2, and 100% humidity. Cultures were refreshed weekly, and the medium was replaced twice per week. Cells were treated with autoclaved bioceramic powders at concentrations of 0.1–1000 µg mL−1 for 24 h. Sodium lauryl sulfate (SLS) was used as a positive control. Absorbance was measured at 510 nm using a microplate reader. The bioactivity of the samples was also evaluated by immersing them in a simulated body fluid (SBF). Prior to testing, the samples were thoroughly cleaned to remove surface contaminants and then dried in an oven overnight to eliminate residual moisture. Each sample was immersed in 10 mL of phosphate-buffered saline (PBS, Sigma, Germany) with a pH of 7.4 and incubated in a temperature-controlled chamber at 37 ± 0.5 °C for 15 and 30 days to simulate biological conditions. The formation of apatite layers on the sample surfaces was subsequently examined using the scanning electron microscope.
3. Results and discussion
3.1 Densification and phase formation
To optimize densification, ceramics with varying STZ content were sintered at 1000–1175 °C, as shown in Fig. 1. All samples showed increased density with increasing temperature, reaching a maximum at 1150 °C and then decreasing at higher sintering temperatures. The decrease in density above 1150 °C is likely due to ion volatilization, particularly of Na+ and Bi3+.35,36 Although 1150 °C was the optimal sintering temperature for all compositions, increasing STZ content reduced the maximum achievable density (inset of Fig. 1). Therefore, the densest ceramics, corresponding to the optimal sintering temperature for each composition, were selected for further investigation. | ||
| Fig. 1 Density of ceramics as a function of sintering temperature and composition. (Inset: relationship between optimal density and STZ content, x.) | ||
X-ray diffraction (XRD) patterns of the ceramics (2θ = 20–70°, Fig. 2(a)) revealed a perovskite structure with coexisting rhombohedral (R) and tetragonal (T) phases. This was evidenced by peak splitting near 2θ = 39–41° ((111)R/(1![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 1)R) and 45–48° ((200)T/(002)T), as shown in Fig. S1 of the SI.8,11,37 With increasing STZ content, the (111)R peak showed a slight change in shape, and the Δ2θ(200) (Δ2θ(200) = (2θ(200) − 2θ(002))) value decreased, indicating reduced tetragonality (Fig. S2(a) in the SI). Furthermore, the relative tetragonality, expressed as the c/a ratio and calculated from the d-spacings of the (200) and (002) reflection planes, also decreased with increasing STZ content (Fig. S2(b) in the SI). These observations suggest that higher STZ concentrations diminish the tetragonal character of the material. Additionally, the XRD peaks shifted toward lower angles, indicating lattice expansion arising from the combined substitution of larger Zr4+ at the B-site and Sr2+ at the A-site. This distortion was further enhanced by the increased incorporation of Sr2+, whose ionic radius is also larger than that of the host A-site ions.20,23,38 This increased the average ionic radii at both A- and B-sites (rA-site and rB-site), as shown in Fig. S3(a) in the SI. To evaluate the degree of lattice distortion, the tolerance factor (t) was calculated and found to increase with x, ranging from 0.717 to 0.9782 (Fig. S3(b) in the SI). This range falls within that typical of stable perovskite structures (t = 0.880–1.090).39 However, an increase in the t reflects reduced symmetry distortion and a shift toward a more cubic structure, resulting in decreased tetragonality.
1)R) and 45–48° ((200)T/(002)T), as shown in Fig. S1 of the SI.8,11,37 With increasing STZ content, the (111)R peak showed a slight change in shape, and the Δ2θ(200) (Δ2θ(200) = (2θ(200) − 2θ(002))) value decreased, indicating reduced tetragonality (Fig. S2(a) in the SI). Furthermore, the relative tetragonality, expressed as the c/a ratio and calculated from the d-spacings of the (200) and (002) reflection planes, also decreased with increasing STZ content (Fig. S2(b) in the SI). These observations suggest that higher STZ concentrations diminish the tetragonal character of the material. Additionally, the XRD peaks shifted toward lower angles, indicating lattice expansion arising from the combined substitution of larger Zr4+ at the B-site and Sr2+ at the A-site. This distortion was further enhanced by the increased incorporation of Sr2+, whose ionic radius is also larger than that of the host A-site ions.20,23,38 This increased the average ionic radii at both A- and B-sites (rA-site and rB-site), as shown in Fig. S3(a) in the SI. To evaluate the degree of lattice distortion, the tolerance factor (t) was calculated and found to increase with x, ranging from 0.717 to 0.9782 (Fig. S3(b) in the SI). This range falls within that typical of stable perovskite structures (t = 0.880–1.090).39 However, an increase in the t reflects reduced symmetry distortion and a shift toward a more cubic structure, resulting in decreased tetragonality.
To quantify the phase fractions of rhombohedral (R) and tetragonal (T) phases in the samples, Rietveld refinement was performed using the GSAS-II software suite. The refinement results are illustrated in Fig. 2(b–f), providing visual confirmation of the phase evolution. Detailed numerical data from the refinement are compiled in Table S1 of the SI. All samples exhibited a mixture of phase composed of rhombohedral (R3c) and tetragonal (P4bm) phases. Notably, the proportion of the rhombohedral R3c phase increased with increasing STZ content. Conversely, the fraction of the tetragonal P4bm phase showed a corresponding decline, indicating a compositional shift driven by STZ incorporation. This trend aligns with the observed reduction in the c/a ratio.
The Raman spectra (from 100–1000 cm−1) of the ceramics were measured at room temperature (RT) and are presented in Fig. 3. The spectral deconvolution, performed using Lorentzian functions, identified seven modes (P1–P7), as described in Table S2 of the SI. The acquired spectra were divided into four distinct regions based on their Raman shift characteristics. The band below 200 cm−1 is closely correlated with the A-site cation vibrations of the perovskite structure, involving Na, Bi, and Sr ions.40,41 The band between 200 and 400 cm−1 is related to the vibration of the Ti–O bond. This band exhibited a shift toward lower wavenumbers, which is likely attributed to distortions in the crystal lattice.42,43 The band in the 430–700 cm−1 region corresponds to the vibrational modes of the TiO6 octahedral units. This band became increasingly broadened as the x values rose, which may be linked to a higher degree of cation disorder. The high-frequency region above 700 cm−1 is associated with overlapping A1 (longitudinal optical) and E (longitudinal optical) vibrational modes.44,45 Therefore, the Raman results support the conclusions drawn from the XRD analysis.
3.2 Microstructure
The surface morphologies of the as-sintered ceramics were examined using scanning electron microscopy (SEM), as shown in Fig. 4. The corresponding fractured cross-sections are presented in Fig. S7. The average grain size and its distribution were quantified using the mean linear intercept method, as shown in the insets of Fig. 4. All compositions exhibited equiaxed grains with slightly rounded edges. Increasing the STZ content led to a systematic reduction in grain size, decreasing from 1.69 µm to 0.93 µm as x increased from 0.00 to 0.20. This grain refinement is likely associated with the incorporation of dopant ions into the BNT–SBT lattice, which introduces local lattice distortions and internal stresses that hinder grain boundary mobility during sintering.46 In addition, according to the lattice strain energy expression (ΔGstrain), differences in ionic radii between the substituent and host ions can generate substantial strain energy, further suppressing grain growth. This strain energy can be quantified using the following expression:47,48| ΔGstrain = 4πYNA[(r0/2)(rd − r0)2 + (1/3)(rd − r0)3], | (1) |
Transmission electron microscopy (TEM) was performed on the x = 0.00 and x = 0.15 samples. The corresponding bright-field TEM images are presented in Fig. 5(a and b). Nanodomains in the x = 0.00 samples exhibited an average width of ∼1.0 nm (highlighted by red boxes), while the x = 0.15 samples showed an average width of ∼0.6 nm. The presence of nanodomains in both compositions suggests that the incorporation of SBT and STZ disrupts the long-range ferroelectric order in the studied ceramics, thereby promoting nanodomain formation. This disruption can induce an ergodic relaxor state, which is typically characterized by a slim P–E hysteresis loop, as discussed in Section 3.3.2.19,49–51 For the x = 0.00 samples, the measured d-spacing was 0.2822 nm, corresponding to the (012) plane reflection, with weak ½(ooo) superlattice reflections observed in the selected area electron diffraction (SAED) pattern (Fig. 5(c), inset), indicating a rhombohedral phase.21,25 In contrast, the x = 0.15 sample exhibited d-spacings of 0.2770 nm and 0.2085 nm, corresponding to the (110) and (021) plane reflections, respectively, with no clear superlattice reflections observed in the SAED pattern (Fig. 5(d), inset). These observations are consistent with the XRD results.
3.3 Electrical properties
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ) of the poled ceramics across various frequencies and temperatures. All samples exhibited two distinct dielectric anomalies in the εr–T curves, consistent with the behavior typically observed in BNT-based ceramics.52,53 The first anomaly, marked by a dielectric peak at Tm (the temperature of maximum permittivity), corresponds to the relaxation of tetragonal polar nanoregions (PNRs) that emerge from rhombohedral PNRs.8,54,55 With increasing STZ content, the temperature of the dielectric maximum (Tm) decreased from 268 °C at x = 0.00 to 222 °C at x = 0.20 (Fig. 7(a)). Moreover, the peak became broader and flatter as the STZ content (x) increased. The second anomaly, denoted as Ts, appeared below 100 °C and has been proposed to be attributed to the thermal evolution of discrete PNRs.55,56
δ) of the poled ceramics across various frequencies and temperatures. All samples exhibited two distinct dielectric anomalies in the εr–T curves, consistent with the behavior typically observed in BNT-based ceramics.52,53 The first anomaly, marked by a dielectric peak at Tm (the temperature of maximum permittivity), corresponds to the relaxation of tetragonal polar nanoregions (PNRs) that emerge from rhombohedral PNRs.8,54,55 With increasing STZ content, the temperature of the dielectric maximum (Tm) decreased from 268 °C at x = 0.00 to 222 °C at x = 0.20 (Fig. 7(a)). Moreover, the peak became broader and flatter as the STZ content (x) increased. The second anomaly, denoted as Ts, appeared below 100 °C and has been proposed to be attributed to the thermal evolution of discrete PNRs.55,56
The Ts decreased from 106 °C to 66 °C as x increased from 0.00 to 0.20 (Fig. 7(a)). The decrease in transition temperatures may be associated with the weakening of ionic bond strength caused by the STZ additive, which lowers the energy required for structural rearrangements and phase transitions.57,58 Consequently, the reduced bond strength decreases the energy barrier for structural transformations, allowing phase transitions to occur at lower temperatures.57 Additionally, this behavior correlates with the increase in the tolerance factor (t) at higher STZ contents, further supporting the lattice distortion induced by the additive.
In this study, the Burns temperature (TB) was determined from the 1/εr versus T plot, as shown in Fig. S4 of the SI. TB represents the temperature at which the dielectric constant begins to deviate from the Curie–Weiss law. As the STZ content increased, TB exhibited a downward trend similar to that of Tm and Ts, decreasing from 394 °C at x = 0.00 to 326 °C at x = 0.20 (inset of Fig. 7(a)). This decrease in TB with increasing STZ concentration reflects a progressive reduction in the onset temperature of polarization in the relaxor state.52 Additionally, the incorporation of STZ increased the dielectric constant at room temperature (εr,room), which may be attributed to the downward shift of the Ts shoulder peak (Fig. 7(a)). However, the additive led to a reduction in the maximum dielectric constant (εr,max) at Tm (Fig. 6(f)), likely due to the broader of the εr–T curves observed with increasing STZ content.
It should be noted that as the STZ content increased, the dielectric constant (εr) near Tm decreased, and a plateau-like behavior became evident, suggesting improved thermal stability of the dielectric properties. To quantify this behavior, the temperature coefficient of capacitance (TCC) was calculated using the equation:48,59
| TCC (%) = (Δε/εb) × 100 (%), | (2) |
It should be noted that, although the x = 0.20 composition exhibited a higher rhombohedral phase fraction at room temperature, it still showed a narrower TCC stability window. This behavior can be understood from the relaxor nature of the system. In BNT-based relaxors, the temperature stability of the dielectric constant is governed not by the static phase fraction at room temperature but by the thermal evolution of polar nanoregions (PNRs). Compositions within the phase coexistence region (x = 0.10–0.15) contain both rhombohedral and tetragonal local distortions, which create a flatter free energy landscape and enhance the thermal stability of PNR correlations. This structural competition suppresses abrupt temperature-driven fluctuations, allowing PNRs to respond more gradually to heating and thereby maintaining a stable permittivity over a broader temperature range. In contrast, the x = 0.20 composition lies outside the coexistence region, where a dominant rhombohedral distortion leads to softer lattice dynamics and reduced PNR stability. As a result, thermal depolarization occurs at lower temperatures, giving rise to a narrower TCC stability window. This explanation has been incorporated into the revised manuscript.
 | ||
| Fig. 8 (a–e) The P–E hysteresis loops and I–E loops of the ceramics for various STZ compositions, (f) variation of Pr and Pmax, (g) Ec, and (h) (Pr/Pmax)A as a function of x. | ||
Furthermore, temperature-dependent P–E hysteresis loops, as presented in Fig. S5 of the SI revealed that samples with x = 0.00–0.15 maintained slim, pinched shapes. In contrast, the pinched feature became less pronounced in the x = 0.20 samples, which exhibited only slim, non-pinched loops. Furthermore, as the temperature increased from 25 °C to 150 °C, all loops became progressively slimmer, indicating enhanced ergodic relaxor behavior due to thermal activation.61
In the current work, the energy storage density (Wrec) and energy storage efficiencies (η) of the studied ceramics were determined from the P–E hysteresis loops by using the following equations:62–64
 | (3) |
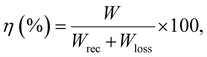 | (4) |
To assess the thermal stability of Wrec, its temperature dependence was evaluated using the following equation,62,65 and the results are presented in Fig. 9(c).
 | (5) |
 | (6) |
The effect of the applied electric field on Wrec and η values (at 125 °C) was also investigated in this study (Fig. 9(e) and (f)). Notably, all samples exhibited the highest η value at 125 °C; therefore, the analysis focused on this temperature. As the electric field increased, Wrec showed a clear upward trend (Fig. 9(e)), while η exhibited only slight variations (Fig. 9(f)). The Wrec and η values measured at 125 °C, along with the ceramics' breakdown strength (Eb) as a function of x, are shown in Fig. 9(h). The Wrec value increased with increasing x, reaching a maximum of 1.01 J cm−3 for the x = 0.15 samples. Furthermore, η increased from 85.80% for the x = 0.00 samples to 91.25% for the x = 0.15 samples. It is important to highlight that beyond x = 0.05, η remained relatively stable, varying only slightly between 91.25% and 92.90%. These results indicate that STZ enhances both Wrec and η, with minimal variation in η for x between 0.05 and 0.20 (at 125 °C). In addition, the trend of Wrec value closely aligns with that of the Eb value, as illustrated in Fig. 9(g) and (h). This correlation is consistent with the well-established understanding that a higher Eb typically leads to a higher Wrec, as elevated Eb values are often associated with finer grain sizes.3,68 However, the lower Wrec observed in the x = 0.20 sample can be attributed to the dominant influence of porosity. It is well known that increased porosity (i.e., lower density) in ceramics tends to reduce Eb, thereby resulting in a lower Wrec.2,3,69–71
In this work, the enhanced electrical properties of the studied ceramics arise from STZ-induced modifications to the coexistence and the resulting evolution of lattice and domain structures. XRD Rietveld analysis shows that increasing STZ progressively shifts the phase assemblage toward a more rhombohedral character while reducing tetragonality, indicating MPB tuning that facilitates easier polarization rotation. Complementary Raman softening and band broadening further reveal increased lattice distortion and cation disorder, consistent with a softened lattice and the suppression of long-range ferroelectric order. TEM observations confirm that macroscopic ferroelectric domains are fragmented into finer nanodomains (from ∼1.0 nm to ∼0.6 nm), demonstrating the development of polar nanoregions (PNRs) that govern the relaxor behavior. This structural transformation results in broadened dielectric anomalies, reduced Pr and Ec, and slimmer P–E loops, which collectively promote high energy-storage efficiency and excellent thermal stability. Additionally, STZ-induced grain refinement enhances dielectric breakdown strength, further increasing the recoverable energy density (Wrec).
Among all investigated compositions, x = 0.15 STZ demonstrated the optimum in dielectric and energy-storage performance, owing to its optimal synergy of phase structure, microstructure, dielectric relaxation, and electrical strength. XRD-Rietveld and Raman analyses revealed that this composition achieved the most balanced rhombohedral–tetragonal coexistence together with higher degree of local structural disorder (Fig. 2 and 3), fostering a pronounced relaxor state. TEM observations confirmed the presence of nanodomains (∼0.6 nm), which enable highly reversible polarization switching and yield slim P–E loops with low remanent polarization (Pr) and minimal energy loss (Fig. 8). SEM analysis further showed that x = 0.15 possessed the smaller and more uniform grains, resulting in the highest Eb (Fig. 9), which directly enhances the recoverable energy density (Wrec). Consequently, this composition delivered the maximum Wrec (≈1.01 J cm−3) with a high efficiency of 92.9% (Fig. 9). In addition, it exhibited one of the broadest temperature-stable dielectric windows (TCC ≤ ±15%), ensuring a reliable dielectric constant across a wide operating range (Fig. 7). Taken together, these structural and electrical merits establish x = 0.15 as the optimal STZ concentration.
 increased from 100 pm V−1 in the unaged samples to 550 pm V−1 in the aged samples, also reflecting a 450% improvement. In addition, the degree of strain hysteresis (H) was calculated using the equation:
increased from 100 pm V−1 in the unaged samples to 550 pm V−1 in the aged samples, also reflecting a 450% improvement. In addition, the degree of strain hysteresis (H) was calculated using the equation:  , where ΔSEmax/2 represents the strain deviation during the application and removal of the electric field, measured at half of the maximum electric field.72,73 The H value significantly improved after aging, changing from 29.50% in the unaged samples to 18.09% in the aged samples, which represents an improvement of 63.07%.
, where ΔSEmax/2 represents the strain deviation during the application and removal of the electric field, measured at half of the maximum electric field.72,73 The H value significantly improved after aging, changing from 29.50% in the unaged samples to 18.09% in the aged samples, which represents an improvement of 63.07%.
Many authors have proposed that the significant strain enhancement observed after post-sintering treatments arises from several mechanisms, most notably the generation and redistribution of oxygen vacancies. These vacancies can form defect-dipole complexes that interact with polar nanoregions, facilitating field-induced ordering and thereby increasing the strain response.74,75 To verify this, electron paramagnetic resonance (EPR) spectroscopy was conducted, and the resulting data are presented in Fig. 10(c). The aged samples exhibited a strong resonance signal at approximately g = 2.06, which is consistent with the typical EPR signal of oxygen vacancies, while the unaged sample showed no detectable signal.74 This EPR signal in the aged samples may indicate the presence of oxygen vacancies. However, the samples were aged at a relatively low temperature (120 °C), which may not be high enough to thermally generate additional oxygen vacancies beyond those already present from sintering. Therefore, X-ray photoelectron spectroscopy technique (XPS) was employed to confirm the EPR result, and the obtained results are displayed in Fig. S11 in SI. The obtained XPS data revealed that there was no significant change in concentration of oxygen vacancies. Therefore, aging may not affect to produce the oxygen vacancy concentration. This evidence can be explained as the following. Normally, oxygen vacancies are expected to form during the high-temperature sintering of BNT-based ceramics because volatilization of Bi and Na components, creating A-site vacancies, which are compensated by the formation of oxygen vacancies to maintain charge neutrality. Furthermore, heterovalent substitution, such as Sr2+ replacing Bi3+/Na+, creates charge imbalance and leads to oxygen vacancy formation during sintering. However, the oxygen vacancies produced during sintering are usually very mobile and can hop rapidly between neighboring oxygen lattice sites. Because they do not remain at one position long enough, they cannot form a stable paramagnetic center that EPR can detect. In many cases, the associated electrons are also electronically delocalized, meaning they spread over a wider region of the lattice instead of being trapped at a single vacancy. Another possibility is that the vacancies become part of charge-neutral defect complexes, for example, a combination of an A-site vacancy and an oxygen vacancy, and such complexes do not contain any unpaired electron.76 As a result, the oxygen vacancies present after sintering do not generate an EPR signal. Because none of these configurations produces a localized magnetic moment, the vacancies remain EPR-silent in the unaged state.76 During aging at 120 °C in air, the pre-existing oxygen vacancies can move slightly and become linked with nearby cations. When this happens, the vacancy can trap electrons and form occupied in-gap states. These localized electrons create paramagnetic centers that can be detected by EPR.77 Because oxygen re-incorporation is negligible at 120 °C, aging stabilizes oxygen vacancies, causing EPR signals to appear only after aging.
Although the aging treatment at 120 °C does not generate additional oxygen vacancies, it enables the pre-existing vacancies formed during sintering to migrate, associate with cation defects, and align into stable defect-dipole configurations.78 In the studied ceramics with a dominant ergodic relaxor phase, the oriented defect dipoles interact strongly with the dynamic polar nanoregions (PNRs). Their alignment generates local internal bias fields that partially steer the orientation of adjacent PNRs, modify the local energy landscape, and promote specific polarization directions. When an electric field is applied, this interaction promotes the field-induced transformation of the ergodic relaxor into a temporary ferroelectric-like state. Importantly, once the field is removed, the defect-dipole-induced internal bias acts as a restoring force, driving the PNRs to collapse back into the ergodic relaxor phase in a highly reversible manner.79,80 This may improve reversibility of the electric field driven relaxor to ferroelectric transition leads to a substantial increase in recoverable electrostrain. Therefore, the large electrostrain enhancement from 0.06% to 0.33% originates from vacancy reorganization and dipole alignment, not from an increase in vacancy concentration.
3.4 Bioactivities
Recently, there has been growing attention toward the application of piezoelectric materials, particularly lead-free piezoceramics, in the medical field. As a result, understanding their biological characteristics has become increasingly significant.30,81,82 In this study, we investigated the bioactivity of the x = 0.15 samples to evaluate their potential for biomedical use.
 | (7) |
The results showed that the x = 0.15 samples maintained cell viability above 95% across all tested concentrations (0.1–1000 µg ml−1), as illustrated in Fig. 11(b). Compared to previous studies, these results show noticeably higher values (Table S3 in the SI).84–88 This also indicates that the samples are non-toxic and may support cell growth, which is essential for their intended biomedical applications.
However, the delayed appearance of apatite in the x = 0.15 composition (compared with the coated samples) can be attributed to its lower initial surface reactivity. The incorporation of the STZ component may reduce the surface energy of the ceramics, thereby slowing the early stages of apatite nucleation,91 weakening interactions with ionic species in SBF and slowing the formation of Ca–P nuclei. Furthermore, Zr4+ substitution alters the local BO6 octahedral environment and may suppress dissolution-driven cation release, limiting the early development of a negatively charged, Ca2+-attracting surface. The generation of surface –OH groups also proceeds more gradually at this composition, reducing Ca2+ adsorption during the early stage of immersion. Collectively, these factors delay the surface activation process, resulting in apatite becoming detectable only after extended soaking (30 days).
To enhance apatite formation, samples with x = 0.15 were coated with β-tricalcium phosphate (β-TCP) using a dip-coating technique, as β-TCP is a well-known bioactive material. The coated surface exhibited a rounded grain morphology with an average grain size of 0.88 µm and a coating thickness of approximately 176 µm (Fig. 12(d)). After 15 days of SBF immersion, apatite particles with an average size of ∼0.08 µm were observed on the coated samples, although they did not fully cover the surface (Fig. 12(e)). After 30 days of immersion, the coated samples exhibited extensive apatite formation, characterized by larger flake-like precipitates approximately 0.17 µm in width and less than 0.03 µm in thickness (Fig. 12(f)). These precipitates covered a greater portion of the surface and formed larger clusters compared to those observed on the 15-day samples. Compared to the uncoated samples, the β-TCP coating significantly enhanced apatite formation, likely due to the release of Ca2+ and PO43− ions, which are essential for apatite development and contribute to the formation of a continuous apatite layer.92
4. Conclusions
The (1 − x)[BNT–SBT]–xSTZ ceramics were synthesized via a conventional solid-state reaction route. All compositions demonstrated coexistence of rhombohedral and tetragonal phases, with a progressive reduction in tetragonality as the STZ content increased. Notably, the x = 0.15 composition exhibited optimum dielectric and energy storage characteristics, including the highest TCC, optimized Wrec, η, and enhanced Eb. This composition also showed excellent thermal stability of Wrec, despite a relatively low initial strain response. Following aging treatment, the x = 0.15 samples exhibited a remarkable 450% increase in Smax and values, emphasizing the role of aging in boosting electromechanical performance. This enhancement likely originates from the reorganization of pre-existing oxygen vacancies into aligned defect dipoles rather than from an increase in vacancy concentration. The MTT assay results demonstrated good cell viability for the x = 0.15 composition, indicating favorable biocompatibility. Furthermore, SBF immersion tests indicated moderate bioactivity for the x = 0.15 samples. Consequently, bioactivity was markedly enhanced following the application of β-TCP surface coating, highlighting its beneficial role in promoting the biological response. These results collectively underscore the promising potential of the x = 0.15 ceramics for future biomedical applications.
values, emphasizing the role of aging in boosting electromechanical performance. This enhancement likely originates from the reorganization of pre-existing oxygen vacancies into aligned defect dipoles rather than from an increase in vacancy concentration. The MTT assay results demonstrated good cell viability for the x = 0.15 composition, indicating favorable biocompatibility. Furthermore, SBF immersion tests indicated moderate bioactivity for the x = 0.15 samples. Consequently, bioactivity was markedly enhanced following the application of β-TCP surface coating, highlighting its beneficial role in promoting the biological response. These results collectively underscore the promising potential of the x = 0.15 ceramics for future biomedical applications.
Author contributions
Kamonporn Saenkam: writing – original draft, validation, methodology, investigation. Waraporn Boontakam: methodology, investigation. Phanrawee Sriprapha: methodology, investigation. Pichitchai Butnoi: methodology, investigation. Kamonpan Pengpat: supervision, conceptualization, methodology, formal analysis. Chamnan Randorn: supervision, conceptualization, methodology, formal analysis. Gobwute Rujijanagul: writing – review & editing, conceptualization, supervision, formal analysis, methodology, investigation, supervision project administration.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5ra07450a.Acknowledgements
This project was supported by the Materials Science Research Center (Fundamental Fund 2025, Chiang Mai University), Chiang Mai University. We also acknowledge the Office of Research Administration, the Department of Physics and Materials Science, Faculty of Science, the Multidisciplinary Research Institute, Chiang Mai University for their contributions, and Science and Technology Research Grant (STRG) Thailand Toray Science Foundation (the 31st Science and Technology Research Grant, 2024).References
- J. Hao, W. Li, J. Zhai and H. Chen, Mater. Sci. Eng., R, 2019, 135, 1–57 CrossRef.
- X. Zhou, G. Xue, H. Luo, C. R. Bowen and D. Zhang, Prog. Mater. Sci., 2021, 122, 100836 Search PubMed.
- G. Wang, Z. Lu, Y. Li, L. Li, H. Ji, A. Feteira, D. Zhou, D. Wang, S. Zhang and I. M. Reaney, Chem. Rev., 2021, 121, 6124–6172 CrossRef CAS PubMed.
- X. Zhang, D. Hu, Z. Pan, X. Lv, Z. He, F. Yang, P. Li, J. Liu and J. Zhai, Chem. Eng. J., 2021, 406, 126818 CrossRef CAS.
- L. Li, P. Fan, M. Wang, N. Takesue, D. Salamon, A. N. Vtyurin, Y. Zhang, H. Tan, B. Nan, Y. Lu, L. Liu and H. Zhang, J. Phys. D: Appl. Phys., 2021, 54, 293001 Search PubMed.
- P. Jaita, K. Saenkam and G. Rujijanagul, RSC Adv., 2023, 13, 3743 RSC.
- B. Jaffe, W. R. Cook Jr. and H. Jaffe, Piezoelectric Ceramics, Academic Press, London and New York, 1971 Search PubMed.
- R. F. Ge, Z. H. Zhao, S. F. Duan, X. Y. Kang, Y. K. Lv, D. S. Yin and Y. Dai, J. Alloys Compd., 2017, 724, 1000–1006 Search PubMed.
- P. Fan, Y. Zhang, B. Xie, Y. Zhu, W. Ma, C. Wang, B. Yang, J. Xu, J. Xiao and H. Zhang, Ceram. Int., 2018, 44, 3211–3217 CrossRef CAS.
- Y. Hiruma, H. Nagata and T. Takenaka, Appl. Phys. Lett., 2009, 95, 052903 CrossRef.
- K. Wang, A. Hussain, W. Jo and J. Rödel, J. Am. Ceram. Soc., 2012, 95, 1–7 CrossRef.
- S. T. Zhang, A. B. Kounga and E. Aulbach, Appl. Phys. Lett., 2007, 91, 112906 CrossRef.
- P. Jaita, R. Sanjoom and G. Rujijanagul, Mater. Res. Bull., 2023, 167, 112417 CrossRef CAS.
- J. Shi, H. Fan, X. Liu and A. J. Bell, J. Am. Ceram. Soc., 2014, 97, 848–853 Search PubMed.
- S. Manotham, P. Jaita, C. Randorn, G. Rujijanagul and D. P. Cann, J. Alloys Compd., 2019, 808, 151655 Search PubMed.
- V. Veerapandiyan, F. Benes, T. Gindel and M. Deluca, Materials, 2020, 13, 5742 Search PubMed.
- X. Zhou, H. Qi, Z. Yan, G. Xue, H. Luo and D. Zhang, ACS Appl. Mater. Interfaces, 2019, 11, 43107–43115 Search PubMed.
- X. Liu, H. Du, X. Liu, J. Shi and H. Fan, Ceram. Int., 2016, 42, 17876–17879 CrossRef CAS.
- X. Zhou, H. Qi, Z. Yan, G. Xue, H. Luo and D. Zhang, J. Eur. Ceram. Soc., 2019, 39, 4053–4059 Search PubMed.
- X. Qiao, D. Wu, F. Zhang, M. Niu, B. Chen, X. Zhao, P. Liang, L. Wei, X. Chao and Z. Yang, J. Eur. Ceram. Soc., 2019, 39, 4778–4784 CrossRef CAS.
- H. Qi and R. Zuo, J. Mater. Chem. A, 2019, 7, 3971–3978 RSC.
- W. Ma, Y. Zhu, M. A. Marwat, P. Fan, B. Xie, D. Salamon, Z. G. Ye and H. Zhang, J. Mater. Chem. C, 2019, 7, 281 RSC.
- D. Li, Y. Lin, M. Zhang and H. Yang, Chem. Eng. J., 2020, 392, 123729 CrossRef CAS.
- C. Ang and Z. Yu, Appl. Phys. Lett., 2009, 95, 232908 CrossRef.
- D. Wang, B. Chu, P. Li, W. Han, Y. Kong, P. Fu, Y. Li, J. Hao and W. Li, Ceram. Int., 2023, 49, 37486–37493 Search PubMed.
- S. Parida, S. K. Rout, V. Subramanian, P. K. Barhai, N. Gupta and V. R. Gupta, J. Alloys Compd., 2012, 528, 126–134 CrossRef CAS.
- J. R. Quiñones-Gurrola, J. C. Rendón-Angeles, Z. Matamoros-Veloza, J. López-Cuevas, R. Pérez-Garibay and K. Yanagisawa, Nanomaterials., 2023, 13, 2195 CrossRef.
- C. Ostos, M. L. Martínez-Sarrión, L. Mestres, E. Delgado and P. Prieto, J. Solid State Chem., 2009, 182, 2620–2625 Search PubMed.
- C. Ostos, L. Mestres, M. L. Martínez-Sarrión, J. E. Garcı, A. Albareda and R. Perez, Solid State Sci., 2009, 11, 1016–1022 CrossRef CAS.
- A. Das and D. Pamu, Mater. Sci. Eng., C, 2019, 101, 539–563 CrossRef CAS.
- N. A. Kamel, Biophys. Rev., 2022, 14, 717–733 Search PubMed.
- A. K. Dubey, R. Kinoshita and K. Kakimoto, RSC Adv., 2015, 5, 19638–19646 RSC.
- S. Phromyoo, N. Lertcumfu, P. Jaita, P. Jarupoom, K. Pengpat and G. Rujijanagul, Ceram. Int., 2018, 44, 2661–2667 Search PubMed.
- N. Lertcumfu, P. Jarupoom and G. Rujijanagul, Ceram. Int., 2013, 39, S373–S377 Search PubMed.
- A. J. Moulson and J. M. Herbert, Electroceramics: Materials, Properties, Applications, John Wiley & Sons Ltd, 2nd edn, 2003 Search PubMed.
- J. U. Rahman, A. Hussain, A. Maqbool, R. A. Malik, M. S. Kim and M. H. Kim, J. Korean Phys. Soc., 2015, 66, 1072–1076 Search PubMed.
- M. R. Bafandeh, H. S. Han and J. S. Lee, J. Electroceram., 2021, 47, 89–99 CrossRef CAS.
- F. Akram, M. Sheeraz, A. Hussain, I. W. Kim, T. H. Kim and C. W. Ahn, Ceram. Int., 2021, 47, 23488–23496 CrossRef CAS.
- J. Wu, H. Zhang, N. Meng, V. Koval, A. Mahajan, Z. Gao, D. Zhang and H. Yan, Mater. Des., 2021, 198, 109344 CrossRef CAS.
- J. Wang, C. Zhou, Q. Li, W. Zeng, J. Xu, G. Chen, C. Yuan and G. Rao, J. Mater. Sci., 2018, 53, 8844–8854 CrossRef CAS.
- S. Manotham, P. Jaita, P. Butnoi, N. Lertcumfu and G. Rujijanagul, J. Alloys Compd., 2022, 897, 163021 CrossRef CAS.
- L. Li, C. Zhou, D. Yu, Y. Zheng, Y. Du, L. Ma, J. Zhao and G. Rao, J. Electron. Mater., 2023, 52, 3488–3498 Search PubMed.
- Q. Xu, X. Yan, Z. Zhu and H. Liu, Process. Appl. Ceram., 2023, 17, 400–409 CrossRef CAS.
- B. Yan, H. Fan, A. K. Yadav, C. Wang, Z. Du, M. Li, W. Wang, W. Dong and S. Wang, J. Mater. Sci., 2020, 55, 14728–14739 Search PubMed.
- R. A. Malik, A. Hussain, A. Zaman, A. Maqbool, J. U. Rahman, T. K. Song, W. J. Kimc and M. H. Kim, RSC Adv., 2015, 5, 96953–96964 Search PubMed.
- G. M. Rashwan, A. A. Ebnalwaled, E. M. Saad and M. A. Shahat, J. Sol-Gel Sci. Technol., 2024, 109, 707–719 Search PubMed.
- X. Gong, C. Zhang, D. Su, W. Xiao, F. Cen, Y. Yang, S. Jiang, J. Wang, K. Li and G. Zhang, J. Materiomics, 2024, 10, 1196–1205 CrossRef.
- N. Qin, X. Chen, L. Liu, L. Tian and Z. Liu, J. Mater. Sci.: Mater. Electron., 2025, 36, 262 Search PubMed.
- P. Jaita, S. Manotham and G. Rujijanagul, RSC Adv., 2020, 10, 32078 Search PubMed.
- C. Che, Y. Bao, Z. Hu, Q. Feng, M. Xie, B. Zhou, J. Yang, H. Nie, Z. Gao and G. Wang, J. Mater. Chem. C, 2024, 12, 16732 Search PubMed.
- Q. Xu, J. Xie, Z. C. He, L. Zhang, M. H. Cao, X. D. Huang, M. T. Lanagan, H. Hao, Z. H. Yao and H. X. Liu, J. Eur. Ceram. Soc., 2017, 37, 99–106 Search PubMed.
- S. K. Gupta, R. M. Quade, B. Gibbons, P. Mardilovich and D. P. Cann, J. Appl. Phys., 2020, 127, 074104 CrossRef.
- Q. Wei, M. Zhu, T. Qin, Z. Guo, M. Zheng, Y. Hou, H. Liu and J. Chen, RSC Adv., 2018, 8, 12269–12275 RSC.
- G. Liu, J. Dong, L. Zhang, Y. Yan, R. Jing and L. Jin, J. Materiomics, 2020, 6, 677–691 Search PubMed.
- M. Wang, Q. Feng, Y. Wei, N. Luo, C. Yuan, C. Zhou, T. Fujita, J. Xu and G. Chen, J. Phys. Chem. Solids, 2021, 157, 110209 CrossRef CAS.
- X. S. Qiao, X. M. Chen, H. L. Lian, W. T. Chen, J. P. Zhou and P. Liu, J. Am. Ceram. Soc., 2016, 99, 198–205 CrossRef CAS.
- J. H. Kim, D. H. Kim, T. H. Lee, T. G. Lee, J. H. Lee, B. Y. Kim, S. Nahm, C. Y. Kang and J. Ryu, J. Am. Ceram. Soc., 2016, 99, 1–8 CrossRef.
- K. Singh, S. Acharya and D. V. Atkare, Ferroelectrics, 2005, 315, 91–110 CrossRef CAS.
- W. Zhu and Z. Y. Shen, J. Mater. Sci., 2024, 59, 2998–3008 CrossRef CAS.
- X. Qiao, F. Zhang, D. Wu, B. Chen, X. Zhao, Z. Peng, X. Ren, P. Liang, X. Chao and Z. Yang, Chem. Eng. J., 2020, 388, 124158 CrossRef CAS.
- R. A. Malik, A. Hussain, A. Maqbool, A. Zaman, C. W. Ahn, J. U. Rahman, T. K. Song, W. J. Kim and M. H. Kim, J. Am. Ceram. Soc., 2015, 98, 3842–3848 Search PubMed.
- R. A. Malik, A. Hussain, A. Maqbool, A. Zaman, T. K. Song, W. J. Kim and M. H. Kim, J. Alloys Compd., 2016, 682, 302–310 Search PubMed.
- Q. Zheng, B. Xie, Y. Tian, Q. Wang, H. Luo, Z. Liu and H. Zhang, J. Materiomics, 2024, 10, 845–856 Search PubMed.
- Q. Zheng, B. Xie, Q. Wang, F. Xue, K. Guo, Z. Liu, P. Mao, W. Cao, H. Luo and H. Zhang, Chem. Eng. J., 2024, 483, 149154 Search PubMed.
- C. Ma, H. Du, J. Liu, L. Kang, X. Du, X. Xi and H. Ran, Ceram. Int., 2021, 47, 25029–25036 Search PubMed.
- Q. Xua, T. Lia, H. Haoa, S. Zhangb, Z. Wanga, M. Caoa, Z. Yaoa and H. Liua, J. Eur. Ceram. Soc., 2015, 35, 545–553 CrossRef.
- Y. Wan, N. Hou, P. Ren, M. Ma, K. Song, F. Yan, X. Lu and G. Zhao, J. Alloys Compd., 2021, 888, 161591 CrossRef CAS.
- Z. Li, B. Xie, Z. Liu, K. Guo, K. Li, H. Zhang and H. Luo, J. Mater. Chem. A, 2025, 13, 9339 Search PubMed.
- B. Zheng, Y. Lin, H. Yang, H. Jing, H. Nan, Y. Wang, F. Z. Yao, M. Wang and Q. Yuan, Adv. Sci., 2025, 12, 2409814 CrossRef CAS.
- B. Zheng, Q. Yuan, Y. Lin, D. Li, H. Yang, Z. Hong, Y. Ma, Y. Ma, J. Guo and J. Wang, Adv. Energy Mater., 2025, e04126 Search PubMed.
- M. Zhang, H. Yang, Y. Lin, Q. Yuan and H. Du, Energy Storage Mater., 2022, 45, 861–868 CrossRef.
- H. Xie, Y. Zhao, J. Xu, L. Yang, C. Zhou, H. Zhang, X. Zhang, W. Qiu and H. Wang, J. Alloys Compd., 2018, 743, 73–82 CrossRef CAS.
- P. Jarupoom, P. Jaita, D. R. Sweatman, A. Watcharapasorn and G. Rujijanagul, Mater. Sci. Eng., B, 2022, 277, 115579 CrossRef CAS.
- H. Luo, H. Liu, H. Huang, Y. Song, M. G. Tucker, Z. Sun, Y. Yao, B. Gao, Y. Ren, M. Tang, H. Qi, S. Deng, S. Zhang and J. Chen, Sci. Adv., 2023, 9, eade7078 CrossRef CAS PubMed.
- J. Lin, F. Lv, Z. Hong, B. Liu, Y. Wu and Y. Huang, Adv. Funct. Mater., 2024, 34, 2313879 CrossRef CAS.
- R. A. Eichel, Phys. Chem. Chem. Phys., 2011, 13, 368–384 Search PubMed.
- M. Tyunina, Materials, 2020, 13, 5596 CrossRef CAS.
- L. X. Zhang and X. Ren, Phys. Rev. B: Condens. Matter Mater. Phys., 2005, 71, 174108 CrossRef.
- Z. H. Zhao, Y. Dai and F. Huang, Sustainable Mater. Technol., 2019, 17, e00092 CrossRef.
- Y. Wang, P. Wang, L. Liu, Y. Wang, Y. Zhao, W. Tian, X. Liu, F. Zhu and J. Shi, Materials, 2023, 16, 4008 CrossRef CAS.
- F. Ali, W. Raza, X. Li, H. Gul and K. H. Kim, Nano Energy, 2019, 57, 879–902 CrossRef CAS.
- J. Jacob, N. More, K. Kalia and G. Kapusetti, Inflammation Regener., 2018, 38, 2 CrossRef PubMed.
- M. A. El-Morsy, N. S. Awwad, H. A. Ibrahium, W. Alharbi, M. Y. Alshahrani and A. A. Menazea, J. Saudi Chem. Soc., 2022, 26, 101463 CrossRef CAS.
- M. Safari-Gezaz, M. Parhizkar and E. Asghari, Sci. Rep., 2025, 15, 149 CrossRef CAS.
- P. Sriprapha, K. Chokethawai, C. Randorn, N. Chandet, K. Thongkorn, K. Saenkam, W. Boontakam and G. Rujijanagul, Mater. Today Sustain., 2024, 26, 100710 Search PubMed.
- A. I. Nicoară, V. Cocoş, C. Chircov, R. D. Truşcă and A. Hudiță, Ceram. Int., 2025, 51, 22327–22339 CrossRef.
- E. A. Mahdy, K. M. Sahbal, M. Mabrouk, H. H. Beherei and Y. K. Abdel-Monem, Ceram. Int., 2021, 47, 6251–6261 CrossRef CAS.
- A. Zerboni, R. Bengalli, G. Baeri, L. Fiandra, T. Catelani and P. Mantecca, Nanomaterials, 2019, 9, 1302 CrossRef CAS PubMed.
- F. Vietanti, Y. J. Lee and Y. J. Chou, Open Ceram., 2025, 22, 100801 CrossRef CAS.
- S. Ferraris, S. Yamaguchi, N. Barbani, M. Cazzola, C. Cristallini, M. Miola, E. Vernè and S. Spriano, Acta Biomater., 2020, 102, 468–480 CrossRef CAS.
- M. RaeisianAsl, S. Jouybar, S. S. Tafreshi and L. Naji, Mater. Today Sustain., 2025, 29, 101072 Search PubMed.
- Q. Zhang, J. Chen, J. Feng, Y. Cao, C. Deng and X. Zhang, Biomaterials, 2003, 24, 4741–4748 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |










