Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Decoding the interplay between protic ionic liquids and drug: spectrophotometric, electrochemical, and DFT exploration of mitoxantrone
Arti Sharmaa,
Ravinder Sharma *ab,
Ritua,
Pamita Awasthi
*ab,
Ritua,
Pamita Awasthi a and
Indra Bahadur
a and
Indra Bahadur *c
*c
aDepartment of Chemistry, National Institute of Technology, Hamirpur-177005, India. E-mail: sharmaravinder444@gmail.com
bDepartment of Chemistry, Dr B R Ambedkar National Institute of Technology, Jalandhar-144011, India
cDepartment of Chemistry, North-West University, Mafikeng Campus, Mmabatho 2735, South Africa. E-mail: bahadur.indra@nwu.ac.za
First published on 19th February 2026
Abstract
Thus far, ILs have attracted growing interest in pharmaceutical applications due to their systematic variation of physicochemical properties that can be tailored to overcome the relevant formulation hurdles associated with poorly soluble and unstable drugs. Beyond solvents, ILs could serve as media for stabilization or even as co-excipients to provide a versatile matrix for enhanced drug performance with decreased side effects. Although most anthracycline antibiotics, such as daunomycin and adriamycin, are cardiotoxic, others, such as mitoxantrone (MTX), are effective and less cardiotoxic. Because it has a planar tricyclic chromophore, MTX, with its protonated side chains, could potentially be involved in dynamic noncovalent interactions, particularly at the level of nucleic acids. In this work, we investigated the molecular interactions of MTX with three protic ILs, namely, ethanol ammonium acetate (EAAc), ethanol ammonium butyrate (EABu), and ethanol ammonium hexanoate (EAHx), using a combination of various techniques including spectroscopic, electrochemical and computational methods. In the UV-visible spectrum, the hypochromic effects observed with the addition of ILs indicate changes in the electronic environment around MTX. The corresponding fluorescence measurements demonstrated enhanced emission intensity with the formation of stable MTX–IL aggregates. Electrochemical studies unambiguously demonstrated an apparent effect of ionic liquid's alkyl chain length on binding affinity, with EAHx displaying the strongest interaction, as evidenced by the higher binding constants and lower Gibbs free energy changes. The experimental results were further supported by density functional theory calculations, indicating an enhancement in the thermodynamic stability and electrostatic complementarity of the MTX–EAHx complex. This combined evidence highlights protic ionic liquids as potential excipients for enhancing the solubility, stability and delivery efficiency of mitoxantrone, with potential for the formulation of safer and more efficient IL-assisted formulations against cancer.
1. Introduction
A key challenge in the pharmaceutical industry is improving the performance of active pharmaceutical ingredients. Many APIs exhibit low water solubility, poor bioavailability, low permeation through the gastrointestinal tract lining, and poor stability in physiological conditions. Such inherent restrictions generally diminish the therapeutic responses, and accordingly, this would demand the therapeutically administered dose to be increased, which would eventually lead to an increased risk of systemic toxicity.1–3 As a result of this, strategies to address these issues have been the aim of the most recent works in contemporary drug formulation research, and considerable resources are being made available for developing methods that enhance the solubility, stability, and targeted capabilities of poorly soluble/hydrophobic drugs. In this context, ionic liquids (ILs) have emerged as promising functional materials capable of overcoming several of these long-standing obstacles in drug development.4 Ionic liquids are low-melting organic salts made up of bulky and asymmetric ions having adjustable polarity, high chemical and thermal stability, and low volatility.5 By altering cation–anion combinations, their physicochemical characteristics can be precisely controlled, enabling the logical development of solvent systems customized for particular drug molecules.6–9 ILs have multiple applications in the pharmaceutical industry, including the ability to function as stabilizers, solvents, co-solvents, and even bioactive ingredients in formulations. A paradigm shift away from traditional solid-state formulations and toward liquid or quasi-liquid therapeutic systems with improved pharmacological performance has resulted from their capacity to improve the solubility, permeability, and stability of drugs.10–13 Protic ionic liquids (PILs), which are formed by proton transfer between a Brønsted acid and a base, are particularly appealing among various IL classes because of their biocompatibility, capacity to form hydrogen bonds, and modifiable polarity. Strong and targeted interactions with pharmaceutical molecules are made possible by their dual hydrogen-bond donor–acceptor nature, which makes them attractive options for enhancing drug solvation and stabilization.14–20 PILs offer a molecular-level mechanism to improve drug delivery performance by efficiently modifying solute–solvent interactions, aggregation behavior, and redox stability.Anthraquinones are aromatic compounds with a variety of biological activities, such as anticancer, antiproliferative, and antioxidant properties, and are a class of drugs that especially benefit from such solvation control.20–28 However, anthraquinone-based drugs frequently have low bioavailability and decreased therapeutic efficacy due to poor water solubility and aggregation. The polarity and hydrogen-bonding properties of their surroundings greatly influence their photophysical and redox behaviors.29–36 Protic ionic liquids provide a tunable medium that can improve the pharmacokinetic characteristics of anthraquinone derivatives by influencing solubility and molecular interactions through hydrogen bonding and electrostatic effects.37–46 A clinically used anthraquinone derivative, mitoxantrone (MTX), is a potent anticancer medication used to treat lymphoma, breast cancer, and leukemia. MTX has real-world formulation problems that make it hard to deliver effectively, even though it is more effective and less harmful to the heart than traditional anthracyclines like daunomycin and adriamycin. The pharmacological performance and therapeutic effectiveness of MTX can be improved by using ionic liquid-assisted strategies to overcome these problems. The present study investigates the intermolecular interactions between mitoxantrone (MTX) and three protic ionic liquids (PILs), ethanol ammonium acetate (EAAc), ethanol ammonium butyrate (EABu), and ethanol ammonium hexanoate (EAHx) in aqueous media. These PILs were selected because they share a common cationic structure, but with different anionic chain lengths, which enable the successive examination of the impact of anion size and hydrophobicity on the solvation and stabilization of MTX. These interactions were studied using a combination of spectroscopic, electrochemical and computational methods. Ultraviolet-visible and fluorescence spectroscopy have provided insight into the variations in the electronic and photophysical behavior of MTX, while cyclic voltammetry has shown redox stability changes in MTX due to the PILs. Density functional theory calculations also confirmed these experimental results, suggesting the thermodynamic and electronic factors controlling the interaction of MTX with PILs. Altogether, this work establishes the impact of protic ILs on the stability and binding of MTX. In this work, the binding of PIL with MTX has been examined, which has not been previously reported in the literature: although publications are available on imidazolium-based ILs, there have been no reports on a PIL. Herein, we report the interactions between MTX, a well-established standard drug, and PIL, along with the resulting structural changes. Computational studies have also been performed to support the experimental results.
2. Materials and methods
2.1 Materials
The reagents used were carefully selected and prepared to guarantee the reliability and reproducibility of the experiment. Acetic acid, butyric acid, hexanoic acid, TEMPO and MTX (commercial grade) were employed without further purification; ethanolamine was purchased separately. Before each experiment, chemicals were dried in a vacuum for 24 hours. Complete details of the chemicals used have been reported in Table 1.| Chemicals | CAS no. | Source | Purityb |
|---|---|---|---|
| Mass fraction | |||
| a Based on spectroscopic analysis (IR and NMR).b As provided by the supplier. | |||
| Butyric acid | 107-92-6 | TCI Pvt. Ltd | ≥0.99 |
| Acetic acid | 64-19-7 | TCI Pvt. Ltd | ≥0.99 |
| Hexanoic acid | 142-62-1 | TCI Pvt. Ltd | ≥0.99 |
| Ethanolamine | 141-43-5 | Loba Chemie Pvt. Ltd | ≥0.99 |
| Ethanol ammonium acetate (EAAc) | 54300-24-2 | Synthesized | ≥0.96a |
| Ethanol ammonium butyrate (EABu) | 56409-18-8 | Synthesized | ≥0.96a |
| Ethanol ammonium hexanoate (EAHx) | — | Synthesized | ≥0.96a |
| Mitoxantrone (MTX) | 65271-80-9 | TCI Pvt. Ltd | ≥0.95 |
2.2 Synthesis and characterization of ethanol ammonium acetate (EAAc), ethanol ammonium butyrate (EABu), and ethanol ammonium hexanoate (EAHx) protic ionic liquid (PIL)
Protic ionic liquids (PILs) were synthesised via controlled acid–base neutralisation, following previously reported procedures.12,29,46–52 Briefly, ethanolamine was dissolved in methanol and placed in a double-neck round-bottom flask equipped with a dropping funnel. The respective acids for each PIL were added slowly: acetic acid for ethanol ammonium acetate (EAAc), butyric acid for ethanol ammonium butyrate (EABu), and hexanoic acid for ethanol ammonium hexanoate (EAHx). The neutralization reaction was performed at a low temperature of approximately 278.15 K to maintain reactivity control and minimize side reactions. To promote uniformity and efficient proton transfer, continuous magnetic stirring was maintained. To ensure complete reaction and PIL stabilisation, the resulting viscous liquid was stirred for 24 hours at room temperature (about 25 °C). The residual solvent and volatile impurities were removed by rotary evaporation carried out at 50 ± 2 °C for 4 hours under reduced pressure (∼200 mbar), followed by further drying in high vacuum (10−3 mbar) for more than 36 hours to remove trace moisture and residual amine. Karl Fischer titration (Metrohm KF 870 Titrino) showed that the moisture content was less than 200 ppm. This was taken into account when determining the molality of the aqueous PIL solutions. To confirm the synthesis of protic ionic liquids, deuterated dimethyl sulfoxide (DMSO-d6) served as the solvent in Fourier-transform infrared spectroscopy (FT-IR, PerkinElmer) and proton nuclear magnetic resonance spectroscopy (1H NMR, Bruker Avance 500 MHz). The FT-IR spectra showed a unique broad band around 1600 cm−1 that was made up of carbonyl (![[double bond splayed left]](https://www.rsc.org/images/entities/char_e009.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) stretching and N–H plane bending vibrations. In addition, the 2400–3500 cm−1 range showed O–H stretches and unique N–H stretching vibrations of the ammonium groups. This aligns with our expectations for the PILs (Fig. S1–S3). The 1H NMR spectra and their peaks can be found in Fig. S4–S6 of the SI.
O) stretching and N–H plane bending vibrations. In addition, the 2400–3500 cm−1 range showed O–H stretches and unique N–H stretching vibrations of the ammonium groups. This aligns with our expectations for the PILs (Fig. S1–S3). The 1H NMR spectra and their peaks can be found in Fig. S4–S6 of the SI.
2.3 Absorption spectroscopy
A cutting-edge Shimadzu T80+ UV-visible spectrophotometer, equipped with high-precision quartz cuvette cells featuring a 1 cm path length, was used to conduct UV-visible spectroscopy and investigate the dynamic interactions between mitoxantrone (MTX) and protic ionic liquids (ILs). To examine the binding behavior of the three customized ionic liquids, ethanol ammonium acetate (EAAc), ethanol ammonium butyrate (EABu), and ethanol ammonium hexanoate (EAHx), absorption spectra of MTX were obtained both in their presence and absence. To create a precise baseline and guarantee measurement accuracy, double-distilled water was used as the blank. All spectroscopic measurements were performed in triplicate, and the spectra shown represent averaged data for reproducibility. UV-visible absorption spectra were recorded in the range of 190–700 nm. Stock solutions of mitoxantrone (MTX, 20 µM) and the protic ionic liquids EAAc, EABu, and EAHx were prepared with double-distilled water. In the absorbance studies, each ionic liquid was added systematically in a concentration range from 0.001 to 0.009 M in the presence of MTX, which was kept constant at a concentration of 20 µM. This approach allows for the examination of the effects of the ionic liquid concentration on the spectroscopic properties of MTX and provides useful information on the strength and nature of MTX–IL interactions.2.4 Fluorescence emission spectroscopy
Fluorescence emission spectra were recorded to examine the interactions between mitoxantrone (MTX) and the protic ionic liquids ethanol ammonium acetate (EAAc), ethanol ammonium butyrate (EABu), and ethanol ammonium hexanoate (EAHx). The fluorescence experiments were carried out on an RF-5301 PC fluorescence spectrophotometer using a 1 cm quartz cuvette. Baseline corrections were made for double-distilled water as the blank. The excitation wavelength for MTX was fixed at 610 nm for all experiments, as mentioned in previous literature. Fresh stock solutions of MTX (20 µM) and PIL (0.05 M) were prepared in double-distilled water to avoid inconsistency and impurities. The concentrations of EAAc, EABu, and EAHx were varied systematically from 0.001 M to 0.007 M during fluorescence experiments. MTX concentration was kept constant at 20 µM while varying the ionic liquid concentration to study the effects of the ionic liquids on the fluorescence behavior of MTX. The binding interactions were quantified by evaluating important parameters such as the binding constant (Kb) and the Stern–Volmer constant (Ksv). This experimental setup allowed for a proper determination of the strength and nature of MTX–IL interactions and provided molecular-level information useful in the design of ionic-liquid-based drug-delivery systems.2.5 Cyclic voltammetry
Cyclic voltammograms were recorded with a Metrohm Autolab PGSTAT302N potentiostat to study the redox activity of mitoxantrone (MTX) and its stability in the presence of protic ionic liquids (PILs). Cyclic voltammetry measurements were conducted at 25 °C in a standard three-electrode system using a glassy carbon working electrode, a platinum counter electrode, and an Ag/AgCl reference electrode. Before each measurement, the glassy carbon electrode was cleaned with double-distilled water and carefully wiped dry using lint-free tissue to achieve a clean, reproducible electrode surface.2.6 Density functional theory
Computational analysis was performed to investigate the molecular properties and interactions of mitoxantrone (MTX) with protic ionic liquids (ILs). Molecular structures were visualised using GaussView 6.0, and density functional theory (DFT) calculations were carried out with Gaussian 16 W. Geometry optimisations were performed for MTX, ethanol ammonium acetate (EAAc), ethanol ammonium butyrate (EABu), ethanol ammonium hexanoate (EAHx), and their corresponding MTX–IL complexes (MTX + EAAc, MTX + EABu, and MTX + EAHx). All calculations employed the B3LYP hybrid functional with the 6-31G basis set, providing a balanced description of the electronic structure, stability, and interaction energetics of the systems studied.3. Results and discussion
3.1. Analysis of the UV-visible spectrum
The interactions between the drug and ionic liquids (ILs) have been studied using the UV-visible absorption technique. The relationships between MTX and the ILs EAAc, EABu, and EAHx have been the subject of several experiments, as shown in Fig. 1.In an aqueous medium, mitoxantrone (MTX) exhibits two different absorption bands at λabs = 608 nm and λabs = 658 nm.52 The conjugated anthraquinone ring system is involved in π–π* transitions, which are responsible for the strong peak at 658 nm. The n–π* transitions, which involve non-bonding electrons on hydroxyl and amino groups, are responsible for the peak at 608 nm.31–33 The UV-visible absorption spectra of mitoxantrone (MTX) (20 µM) with varying concentrations of ionic liquids (ILs), ethanol ammonium acetate (EAAc), ethanol ammonium butyrate (EABu), and ethanol ammonium hexanoate (EAHx) (0.001–0.009 µM) have been recorded in water. With an increase in the concentrations of the ionic liquids (ILs), ethanol ammonium acetate (EAAc), ethanol ammonium butyrate (EABu), and ethanol ammonium hexanoate (EAHx), there was a decrease in the intensity of the absorbance of the mitoxantrone (MTX) peak at 658 nm (hypochromic shift) as shown in Fig. 1. Eqn (1) and (2) have been used to determine the binding constant (Kb) and free energy of binding (ΔGb):
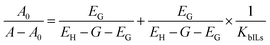 | (1) |
ΔG = −RT![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Kb Kb
| (2) |
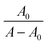 vs.
vs. 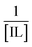 is linear, and the binding constant (Kb) has been calculated from the ratio of intercept to slope, as shown in Fig. 2. At 298.15 K, the binding constant (Kb) values of the mitoxantrone (MTX) complexes with ethanol ammonium acetate (EAAc), ethanol ammonium butyrate (EABu), and ethanol ammonium hexanoate (EAHx) were determined to be 307.51 M−1, 660.86 M−1, and 698.67 M−1, respectively. The observed rise in Kb with the length of the anion chain (acetate < butyrate < hexanoate) suggests that the MTX–IL complexes are stabilized primarily by hydrophobic contacts. Compared to acetate and butyrate, the hexanoate anion (CH3CH2CH2CH2CH2COO−) has a longer alkyl chain, which improves hydrophobic and van der Waals interactions with the aromatic sections of MTX. Because the hydrophobic chains aid in keeping water molecules out of the binding interface and lowering the system's free energy, these interactions result in stronger complex formation and better binding affinity. Furthermore, the oxygen and nitrogen atoms of MTX (such as carbonyl or hydroxyl groups) can interact electrostatically or establish hydrogen bonds with the ethanol ammonium cation (NH4+) in these ionic liquids. Better spatial accommodation and interaction with MTX are made possible by the longer and more flexible hexanoate anion in EAHx, which further stabilizes the complex. Therefore, the higher (Kb) value and greater stability of MTX–EAHx are due to both hydrophobic and electrostatic interactions. Calculated binding parameters have been compared with the literature data available33 and are shown in Table 2.
is linear, and the binding constant (Kb) has been calculated from the ratio of intercept to slope, as shown in Fig. 2. At 298.15 K, the binding constant (Kb) values of the mitoxantrone (MTX) complexes with ethanol ammonium acetate (EAAc), ethanol ammonium butyrate (EABu), and ethanol ammonium hexanoate (EAHx) were determined to be 307.51 M−1, 660.86 M−1, and 698.67 M−1, respectively. The observed rise in Kb with the length of the anion chain (acetate < butyrate < hexanoate) suggests that the MTX–IL complexes are stabilized primarily by hydrophobic contacts. Compared to acetate and butyrate, the hexanoate anion (CH3CH2CH2CH2CH2COO−) has a longer alkyl chain, which improves hydrophobic and van der Waals interactions with the aromatic sections of MTX. Because the hydrophobic chains aid in keeping water molecules out of the binding interface and lowering the system's free energy, these interactions result in stronger complex formation and better binding affinity. Furthermore, the oxygen and nitrogen atoms of MTX (such as carbonyl or hydroxyl groups) can interact electrostatically or establish hydrogen bonds with the ethanol ammonium cation (NH4+) in these ionic liquids. Better spatial accommodation and interaction with MTX are made possible by the longer and more flexible hexanoate anion in EAHx, which further stabilizes the complex. Therefore, the higher (Kb) value and greater stability of MTX–EAHx are due to both hydrophobic and electrostatic interactions. Calculated binding parameters have been compared with the literature data available33 and are shown in Table 2.
| Techniques | Systems | Literature | Kb (L mol−1) | R2 | ΔG (kJ mol−1) |
|---|---|---|---|---|---|
| UV-visible | Mitoxantrone + ethanol ammonium acetate (MTX + EAAc) | FLZ-[C10mim][Cl] | 3.07 × 102 (expt.) | 0.99 | −14.17 (expt.) |
| 4.05 × 104 (ref. 33) | −26.28 (ref. 33) | ||||
| Mitoxantrone + ethanol ammonium butyrate (MTX + EABu) | FLZ-[C6mim][Cl] | 6.60 × 102 (expt.) | 0.98 | −16.08 (expt.) | |
| 6.78 × 103 (ref. 33) | −24.32 (ref. 33) | ||||
| Fluorescence | Mitoxantrone + ethanol ammonium acetate (MTX + EAAc) | FLZ-[C10mim][Cl] | 3.31 (expt.) | 0.97 | −2.96 (expt.) |
| FLZ-[C6mim][Cl] | 3.30 × 104 (ref. 33) | −25.65 (ref. 33) | |||
| 1.45 × 104 (ref. 33) | −23.74 (ref. 33) | ||||
| Mitoxantrone + ethanol ammonium butyrate (MTX + EABu) | CIP-[C10mim][BF4] | 5.62 (expt.) | 0.96 | −4.28 (expt.) | |
| CIP-[C10mim][Br] | 48.62 (ref. 56) | −9.62 (ref. 56) | |||
| CIP-[C10mim][Cl] | 33.11 (ref. 56) | −8.67 (ref. 56) | |||
| 25.56 (ref. 56) | −8.03 (ref. 56) | ||||
| Cyclic voltammetry | Mitoxantrone + ethanol ammonium acetate (MTX + EAAc) | DH + C14mimBr | 5.57 × 102 (expt.) | 0.98 | −15.67 (expt.) |
| DH + TTAB | 26.54 × 104 (ref. 35) | −30.95 (ref. 35) | |||
| AC + C14mimBr | 7.45 × 104 (ref. 35) | −27.81 (ref. 35) | |||
| AC + TTAB | 0.43 × 104 (ref. 35) | −20.73 (ref. 35) | |||
| 0.19 × 104 (ref. 35) | −18.70 (ref. 35) | ||||
| Mitoxantrone + ethanol ammonium butyrate (MTX + EABu) | LVF-[Bmim][Cl] | 7.61 × 102 (expt.) | 0.95 | −16.44 (expt.) | |
| LVF-[Dmim][Cl] | 3.70 (ref. 30) | −3.24 (ref. 30) | |||
| 34.50 (ref. 30) | −8.54 (ref. 30) |
Because of the bigger, bulkier hexanoate ion, the interaction between mitoxantrone (MTX) and ethanol ammonium hexanoate (EAHx) (698.67 M−1) may entail more substantial non-covalent interactions (e.g., hydrophobic, electrostatic, and π–π stacking), which could result in stronger complexation. The smaller and more polar butyrate and acetate anions of ethanol ammonium butyrate (EABu) (660.86 M−1) and ethanol ammonium acetate (EAAc) (307.51 M−1), on the other hand, are likely to result in weaker interactions, which are reflected in smaller binding constants and less obvious spectral shifts. The free energy of binding (ΔG) was calculated to be −14.1 kJ mol−1 for mitoxantrone + ethanol ammonium acetate (MTX + EAAc), −16.1 kJ mol−1 for mitoxantrone + ethanol ammonium butyrate (MTX + EABu), and −16.3 kJ mol−1 for mitoxantrone + ethanol ammonium hexanoate (MTX + EAHx) using eqn (2). The process was spontaneous, as evidenced by the negative value of the change in the free energy of binding. The greater binding seen with EAHx suggests that this complex can provide a more stable and lipophilic drug–ionic liquid combination from the standpoint of drug delivery. Drug encapsulation, membrane permeability, and prolonged release all benefit from such increased lipophilicity. Longer hydrophobic chains in ionic liquids are known to improve the drug's bioavailability and controlled release characteristics by making it more compatible with lipid-based carriers (such as liposomes, micelles, or nanoparticles). The MTX–EAHx complex is therefore a viable system for targeted and long-term drug delivery applications because of its greater binding constant, which may result in enhanced stability, regulated release, and effective cellular uptake.
3.2. Fluorescence spectral studies
Mitoxantrone (MTX) exhibits a fluorescence peak at approximately 702 nm when exposed at 610 nm (Fig. 3). The addition of the ionic liquids (ILs), namely, ethanol ammonium acetate (EAAc), ethanol ammonium butyrate (EABu), and ethanol ammonium hexanoate (EAHx), enhanced mitoxantrone's fluorescence emission intensity. The complex formation between ionic liquids (ILs), ethanol ammonium acetate (EAAc), ethanol ammonium butyrate (EABu), ethanol ammonium hexanoate (EAHx), and mitoxantrone (MTX) was indicated by the rise in the emission spectra of mitoxantrone (MTX) upon the addition of ionic liquids (ILs),53–55 as represented in Fig. 3.A gradual increase in fluorescence intensity was observed upon the addition of ionic liquid, indicating binding-induced fluorescence enhancement. The relative fluorescence intensity (F/F0) increased in a concentration-dependent manner. These plots show the concentration-dependent enhancement, and the corresponding variations in the fluorescence emission spectra were investigated.
F/F0 plots of (MTX + EAAc), (MTX + EABu), and (MTX + EAHx) complexes against the concentration of ILs are presented in Fig. 4.
Kb was calculated using eqn (3):
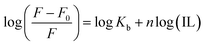 | (3) |
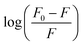 vs. log(IL) plot is presented in Fig. 5, where the slope provides the number of binding sites (n), and the intercept can be used to determine the binding constant (Kb). EAHx had a higher Kb (10.00 M−1) value than EABu (5.62 M−1) and EAAc (3.31 M−1), indicating a greater binding affinity of EAHx with MTX as compared to EABu and EAAc. Compared to EABu (butyrate) and EAAc (acetate), EAHx has a longer alkyl chain (hexanoate). The extended chain improves molecular association by increasing hydrophobic contacts with MTX's aromatic sections. By stabilizing the MTX–EAHx complex, these hydrophobic forces increase Kb. The large hexanoate anion strengthens binding by offering a larger surface area for van der Waals and potential π–π stacking interactions with the planar anthracenedione rings of MTX. Because EAHx is more hydrophobic, it tends to keep water molecules out of the MTX binding area, which lessens solvent interference and promotes a stronger drug–IL connection. The values of ΔG for the complexation between (MTX) and ILs have been calculated using eqn (4).
vs. log(IL) plot is presented in Fig. 5, where the slope provides the number of binding sites (n), and the intercept can be used to determine the binding constant (Kb). EAHx had a higher Kb (10.00 M−1) value than EABu (5.62 M−1) and EAAc (3.31 M−1), indicating a greater binding affinity of EAHx with MTX as compared to EABu and EAAc. Compared to EABu (butyrate) and EAAc (acetate), EAHx has a longer alkyl chain (hexanoate). The extended chain improves molecular association by increasing hydrophobic contacts with MTX's aromatic sections. By stabilizing the MTX–EAHx complex, these hydrophobic forces increase Kb. The large hexanoate anion strengthens binding by offering a larger surface area for van der Waals and potential π–π stacking interactions with the planar anthracenedione rings of MTX. Because EAHx is more hydrophobic, it tends to keep water molecules out of the MTX binding area, which lessens solvent interference and promotes a stronger drug–IL connection. The values of ΔG for the complexation between (MTX) and ILs have been calculated using eqn (4).
ΔG = −RT![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Kb Kb
| (4) |
The negative value of (ΔG) indicates the spontaneous complex formation process between the ionic liquids (ILs), ethanol ammonium acetate (EAAc), ethanol ammonium butyrate (EABu) and ethanol ammonium hexanoate (EAHx), and mitoxantrone (MTX). The mitoxantrone + ethanol ammonium hexanoate (MTX + EAHx) complex (−10.00 kJ mol−1) is more favorable than the mitoxantrone + ethanol ammonium butyrate (MTX + EABu) (−4.28 kJ mol−1) and mitoxantrone + ethanol ammonium acetate (MTX + EAAc) (−2.96 kJ mol−1) complexes due to the lower (ΔG) values. It can be concluded that the MTX-IL complex formation is largely influenced by hydrophobic and electrostatic forces. A stronger interaction between mitoxantrone and the longer-chain ionic liquid, ethanol ammonium hexanoate, is shown by the higher binding constant (Kb) of the MTX–EAHx complex (10.00 M−1) in comparison to MTX–EABu (5.62 M−1), and MTX–EAAc (3.31 M−1). This greater binding implies that MTX is better stabilized and encapsulated in the EAHx environment, preventing degradation and increasing its solubility. Such moderate-to-strong binding is advantageous for producing controlled or sustained drug release in the context of drug delivery because it permits progressive release at the target site while maintaining the drug's stability throughout transport. Therefore, in comparison to the shorter-chain equivalents, EABu and EAAc, EAHx seems to offer an ideal milieu for efficient MTX administration. Therefore, the fluorescence data indicate that EAHx provides a better milieu for MTX binding and stabilization than EAAc or EABu, making it a feasible ionic liquid for MTX-based drug delivery systems. The computed binding parameters, as displayed in Table 2, have been compared with the data from the literature.30,33,56
3.3. Cyclic voltammetry studies
Mitoxantrone (MTX), a synthetic anthracenedione anticancer drug, exhibits tautomeric flexibility due to proton and electron redistribution within its quinone core and aminoalkyl side chains. This structural adaptability plays an important role in governing the chemical reactivity and electrochemical behaviour of the molecule.57,58 In cyclic voltammetry (CV), MTX displays a well-defined reversible oxidation peak at lower positive potentials, which is attributed to the oxidation of the 5,8-hydroxyl groups on the anthracenedione framework, indicating a stable and thermodynamically favourable redox process. In addition to this primary response, a second oxidation peak is observed at more positive potentials. Previous studies have associated this feature with the presence of a less abundant electroactive tautomer formed through intramolecular rearrangement involving the side-chain nitrogen and the quinone system. Due to its different electronic structure, this tautomer has a higher oxidation potential and hence gives the secondary voltammetric signal. The influence of the solvent medium on this tautomeric equilibrium was explored by performing cyclic voltammetry measurements in the presence of protic ionic liquids (PILs), namely ethanol, ammonium acetate, butyrate, and hexanoate. Due to their marked hydrogen-bonding capability and ionic nature, PILs strongly alter the solvation environment of MTX, especially at heteroatoms involved in the proton-transfer process. Notably, CV experiments conducted in ethanol ammonium acetate showed a pronounced decrease in the intensity of the second oxidation peak, suggesting suppression of the electroactive tautomer. These observations demonstrate that PILs can modulate the tautomeric distribution of MTX and, consequently, its redox behaviour, highlighting the sensitivity of MTX electrochemistry to solvent–solute interactions.59,60Cyclic voltammograms of pure mitoxantrone (MTX), and mitoxantrone (MTX) with varied concentrations of ethanol ammonium acetate (EAAc) and ethanol ammonium butyrate (EABu), ethanol ammonium hexanoate (EAHx), are shown in Fig. 6. It was found that when ethanol ammonium acetate (EAAc), ethanol ammonium butyrate (EABu), and ethanol ammonium hexanoate (EAHx) concentrations are added incrementally, the peak current (ip) of mitoxantrone (MTX) increases. The development of the electrochemically active mitoxantrone (MTX) and ionic liquids (ILs) complex may be the cause of this notable rise in peak current values.
According to the Langmuir eqn (5), the increase in the peak current value (Δip) of mitoxantrone (MTX) in the presence of different concentrations of ionic liquids, namely, ethanol ammonium acetate (EAAc), ethanol ammonium butyrate (EABu), and ethanol ammonium hexanoate (EAHx), indicates a good linear relationship between the reciprocal of peak current and concentration:
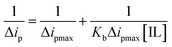 | (5) |
3.4. Density functional theory
To determine the strength of contacts in the MTX + IL complexes and the viability of electron transfer between the drug and IL donor–acceptor, the energy difference (ΔE) has been calculated (Fig. 9). The calculated HOMO–LUMO energy gaps for mitoxantrone (MTX), ethanol ammonium acetate (EAAc), ethanol ammonium butyrate (EABu), ethanol ammonium hexanoate (EAHx), mitoxantrone + ethanol ammonium acetate (MTX + EAAc), mitoxantrone + ethanol ammonium butyrate (MTX + EABu), and mitoxantrone + ethanol ammonium hexanoate (MTX + EAHx) are 0.09658, 0.239, 0.144, 0.125, 0.09526, 0.09748, and 0.101 Ha, respectively.
| η = (ELUMO − EHOMO)/2 | (7) |
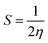 | (8) |
| ω = µ2/2η | (9) |
| χ = −(EHOMO + ELUMO)/2 | (10) |
| IP = −(ELUMO) | (11) |
| µ = (EHOMO + ELUMO)/2 | (12) |
| EA = −ELUMO | (13) |
| Parameters | Mitoxantrone (MTX) | EAAc | EABu | EAHx | MTX + EAAc | MTX + EABu | MTX + EAHx |
|---|---|---|---|---|---|---|---|
| LUMO (Ha) | −0.086 | −0.012 | −0.017 | −0.018 | −0.083 | −0.063 | −0.039 |
| HOMO (Ha) | −0.182 | −0.251 | −0.161 | −0.140 | −0.178 | −0.161 | −0.140 |
| LUMO − HOMO (Ha) | 0.096 | 0.239 | 0.144 | 0.122 | 0.095 | 0.097 | 0.101 |
| LUMO + HOMO (Ha) | −0.268 | −0.263 | −0.178 | −0.158 | −0.261 | −0.224 | −0.179 |
| Ionization potential (I) (Ha) | 0.182 | 0.251 | 0.161 | 0.140 | 0.178 | 0.161 | 0.140 |
| Electron affinity (A) (Ha) | 0.086 | 0.012 | 0.017 | 0.018 | 0.083 | 0.063 | 0.039 |
| Chemical potential (µ) (Ha) | −0.134 | −0.131 | −0.089 | −0.079 | −0.131 | −0.112 | −0.0895 |
| Electronegativity (χ) (Ha) | 0.134 | 0.131 | 0.089 | 0.079 | 0.131 | 0.112 | 0.0895 |
| Hardness (η) (Ha) | 0.048 | 0.119 | 0.072 | 0.061 | 0.048 | 0.049 | 0.050 |
| Softness (S) (Ha−1) | 10.350 | 4.184 | 6.944 | 8.196 | 10.497 | 10.258 | 9.90 |
| Electrophilicity index (ω) (Ha) | 0.186 | 0.072 | 0.055 | 0.051 | 0.179 | 0.129 | 0.079 |
| Thermodynamic properties | Mitoxantrone (MTX) | Ethanol ammonium acetate (EAAc) | Ethanol ammonium butyrate (EABu) | Ethanol ammonium hexanoate (EAHx) | Mitoxantrone + ethanol ammonium butyrate (MTX + EAAc) | Mitoxantrone + ethanol ammonium butyrate (MTX + EABu) | Mitoxantrone + ethanol ammonium butyrate (MTX + EAHx) |
|---|---|---|---|---|---|---|---|
| εo (Ha) | −1525.068 | −438.074 | −516.685 | −595.291 | −1963.292 | −2041.962 | −2120.610 |
| Ezpe (Ha) | 0.494 | 0.139 | 0.197 | 0.220 | 0.634 | 0.692 | 0.755 |
| εtot (Ha) | 0.526 | 0.149 | 0.209 | 0.240 | 0.679 | 0.739 | 0.771 |
| Hcorr (Ha) | 0.528 | 0.149 | 0.210 | 0.241 | 0.679 | 0.739 | 0.771 |
| Gcorr (Ha) | 0.427 | 0.101 | 0.156 | 0.184 | 0.545 | 0.601 | 0.640 |
| εo+ εzpe (Ha) | −1524.573 | −437.935 | −516.488 | −595.071 | −1962.658 | −2041.270 | −2119.855 |
| εo+ εtot (Ha) | −1524.541 | −437.925 | −516.476 | −595.051 | −1962.613 | −2041.223 | −2119.839 |
| εo + Hcorr (Ha) | −1524.540 | −437.924 | −516.475 | −595.050 | −1962.613 | −2041.223 | −2119.839 |
| εo + Gcorr (Ha) | −1524.640 | −437.972 | −516.529 | −595.107 | −1962.747 | −2041.361 | −2119.970 |
| Ethermal (kcal mol−1) | 330.505 | 93.523 | 131.207 | 153.112 | 425.854 | 463.540 | 490.144 |
| Heat capacity (Cv) cal mol−1 K−1) | 120.011 | 32.733 | 42.101 | 52.099 | 158.645 | 167.367 | 188.211 |
| Enthalpy change (ΔH) (kcal mol−1) | ̶ | ̶ | ̶ | ̶ | −92.870 | −124.870 | −155.196 |
| Free energy change (ΔG) (kcal mol−1) | ̶ | ̶ | ̶ | −84.713 | −120.48 | −138.991 |
Following complexation, mitoxantrone + ethanol ammonium acetate (MTX + EAAc) (−1963.292 Ha), mitoxantrone + ethanol ammonium butyrate (MTX + EABu) (−2041.962 Ha), and mitoxantrone + ethanol ammonium hexanoate (MTX + EAHx) (−2120.610 Ha) exhibited a notable drop in energy, showing that contact improves stability as indicated in Table 4. Interestingly, there was a greater drop in energy with mitoxantrone and ethanol ammonium hexanoate (MTX + EAHx) than mitoxantrone + ethanol ammonium butyrate (MTX + EABu), and mitoxantrone + ethanol ammonium acetate (MTX + EAAc), suggesting that mitoxantrone and ethanol ammonium hexanoate (MTX + EABu) have a stronger interaction. When compared to the electronic energies of the unbound molecules, the total energy increased with the addition of zero-point energy (Ezpe). Hence, it was concluded that complexation resulted in a more thermodynamically stable system supported by the zero-point energy-corrected values (εo + Ezpe), which showed that the complexes mitoxantrone + ethanol ammonium acetate (MTX + EAAc), mitoxantrone + ethanol ammonium butyrate (MTX + EABu), and mitoxantrone + ethanol ammonium hexanoate (MTX + EAHx) formed more stable states than their isolated components.
The sum of the electronic, vibrational, rotational, and translational contributions to the system's overall internal energy is known as the thermal energy (Ethermal). Ethanol ammonium acetate (EAAc) has the lowest thermal energy (93.523 kcal mol−1) among the isolated compounds, while mitoxantrone (MTX) has the highest (330.505 kcal mol−1), as shown in Table 4. Thermal energy increased upon complex formation, rising from 425.854 kcal mol−1 for mitoxantrone + ethanol ammonium acetate (MTX + EAAc) to 463.540 kcal mol−1 for mitoxantrone + ethanol ammonium butyrate (MTX + EABu), and reaching 490.144 kcal mol−1 for mitoxantrone + ethanol ammonium hexanoate (MTX + EAHx). The increase in the degrees of freedom brought about by the creation of larger molecular systems resulted in the rise in thermal energy, leading to more complicated interactions and higher total energy. The production of bigger and more flexible molecular structures led to the generation of more disordered systems, as indicated by the increase in entropy values upon the interaction of mitoxantrone (MTX) with ethanol ammonium acetate (EAAc), ethanol ammonium butyrate (EABu), and ethanol ammonium hexanoate (EAHx). Compared to mitoxantrone + ethanol ammonium butyrate (MTX + EABu) and mitoxantrone + ethanol ammonium acetate (MTX + EAAc), the entropy growth for mitoxantrone + ethanol ammonium hexanoate (MTX + EAHx) was more pronounced due to its larger size and complexity, offering more vibrational modes and rotational freedom.30,33,38
| System | −V (V) | +V (V) |
|---|---|---|
| Mitoxantrone (MTX) | −0.108 | +0.108 |
| Ethanol ammonium acetate (EAAc) | −0.082 | +0.082 |
| Ethanol ammonium butyrate (EABu) | −0.067 | +0.067 |
| Ethanol ammonium hexanoate (EAHx) | −0.061 | +0.061 |
| Mitoxantrone + ethanol ammonium acetate (MTX + EAAc) | −0.092 | +0.092 |
| Mitoxantrone + ethanol ammonium butyrate (MTX + EABu) | −0.117 | +0.117 |
| Mitoxantrone + ethanol ammonium hexanoate (MTX + EAHx) | −0.131 | +0.131 |
These findings are highly relevant to drug formulation since enhanced electrostatic stabilisation and improved solvation can mitigate premature degradation, aggregation, and precipitation, which are key factors limiting the bioavailability of salt-based anticancer drugs. Therefore, the more favored and beneficial interaction of MTX with EABu represents improved molecular stability, solubility, and controlled release potential, which are key to effective drug delivery. These results, which are supported by favorable DFT calculations and thermodynamic parameters, show that protic ionic liquids, especially EABu, can act as stabilizing co-solvents and may provide a suitable method for developing more stable, efficient, and less toxic ionic-liquid-based anticancer formulations.
4. Conclusions
Ionic liquids may serve as a promising platform in pharmaceutical formulations to enhance drug aqueous solubility, stability, and overall performance, particularly for salt-form drugs that exhibit low bioavailability and a tendency to aggregate in biological media. Mitoxantrone (MTX) is an example of such an anthracenedione anticancer drug, which, unfortunately, has poor aqueous solubility and a tendency to self-aggregate under physiological conditions. Herein, we have explored, at the molecular level, the interactions of MTX with three protic ionic liquids, namely, ethanol ammonium acetate (EAAc), butyrate (EABu) and hexanoate (EAHx), to investigate the effects of anionic chain length on drug solvation, stability and redox behavior. By employing a combined experimental (UV-visible absorption, fluorescence spectroscopy and cyclic voltammetry) and computational (DFT) approach, the study has demonstrated the spontaneous complexation of MTX with ILs, as well as thermodynamically favorable processes. Spectroscopic and electrochemical measurements show that fluorescence emission intensity, hypochromism, and redox reversibility increase with the length of the alkyl substituent and correlate with changes in ΔE. Results among the investigated systems, EAHx exhibited the most significant changes in binding and free energy (ΔG), indicating stronger hydrophobic, hydrogen-bonding, and electrostatic interactions with MTX. Evidence of these trends was also provided by the DFT-optimised structures and electrostatic potential maps, which showed extended charge spread and more non-covalent interactions in the MTX–EAHx complex. In general, the data presented clearly indicate a structure–property relationship for MTX–PIL systems with enhanced anionic chain length being directly related to molecular complex binding and stability. These findings, supported by molecular-level insights, provide evidence for the claimed benefits of protic ILs as functional excipients and suggest that higher molecular weight PILs may permit improved stability, solubility, and drug release performances for MTX with potential implications in the rational design of innovative ionic-liquid-based formulations as anticancer agents. The authors will consider conducting mechanistic or comparative studies in future work to demonstrate why the PIL nature or properties are necessary, as well as how the PIL characteristics offer advantages compared to the precursor materials from which they were derived, and why these properties lead to the observed experimental outcomes.Conflicts of interest
There are no conflicts to declare.Data availability
The authors declare that the data supporting the findings of this study are available within the article.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5ra05841d.
Acknowledgements
The authors gratefully acknowledge the Department of Chemistry, National Institute of Technology Hamirpur (H. P.), for providing the laboratory facilities necessary to carry out this research. The Department of Chemistry at Dr B. R. Ambedkar National Institute of Technology, Jalandhar, is also acknowledged for its support in the synthesis of ionic liquids. Ms Arti Sharma thanks the Ministry of Education (MoE), Government of India, New Delhi, for the award of the research fellowship. I. Bahadur acknowledges SASOL, South Africa, for the financial support.References
- M. R. Chowdhury, R. M. Moshikur, R. Wakabayashi, Y. Tahara, N. Kamiya, M. Moniruzzaman and M. Goto, Mol. Pharm., 2018, 15(6), 2484–2488 CrossRef CAS PubMed.
- L. Zhang, J. Liu, T. Tian, Y. Gao, X. Ji, Z. Li and Y. Luan, Chem. Phys. Chem., 2013, 14(15), 3454–3457 CrossRef CAS PubMed.
- H. D. Williams, Y. Sahbaz, L. Ford, T. H. Nguyen, P. J. Scammells and C. J. Porter, Chem. Commun., 2014, 50(14), 1688–1690 RSC.
- K. D. Amirchand, V. Singh and N. Islam, J. Chem. Eng. Data, 2022, 67(10), 2920–2930 CrossRef CAS.
- A. Akinlua, M. A. Jochmann and T. C. Schmidt, Ind. Eng. Chem. Res., 2015, 54(51), 12960–12965 Search PubMed.
- R. M. Dias, F. H. Sosa and M. C. da Costa, Polym. Bull., 2020, 77, 3637–3656 CrossRef CAS.
- A. McGrogan, J. Lafferty, L. O'Neill, L. Brown, J. M. Young, P. Goodrich, M. J. Muldoon, L. Moura, S. Youngs, T. Hughes, S. Gartner, T. G. A. Youngs, J. D. Holbrey and M. Swadźba-Kwaśny, J. Phys. Chem. B, 2024, 128(13), 3220–3235 Search PubMed.
- T. L. Greaves, A. Weerawardena, C. Fong, I. Krodkiewska and C. J. Drummond, J. Phys. Chem. B, 2006, 110(45), 22479–22487 CrossRef CAS PubMed.
- C. L. B. Reis, L. M. A. e Silva, T. H. S. Rodrigues, A. K. N. Félix, R. S. de Santiago-Aguiar, K. M. Canuto and M. V. P. Rocha, Bioresour. Technol., 2017, 224, 694–701 CrossRef CAS PubMed.
- A. Krishnan, K. P. Gopinath, D. V. N. Vo, R. Malolan, V. M. Nagarajan and J. Arun, Chem. Lett., 2020, 18, 2031–2054 CrossRef CAS.
- C. L. B. Reis, T. A. Campelo, C. C. Frota, A. P. Ayala, L. M. A. Silva, M. V. P. Rocha and R. S. de Santiago-Aguiar, Eur. J. Pharm. Biopharm., 2024, 201, 114345 CrossRef CAS PubMed.
- K. D. Amirchand and V. Singh, J. Mol. Liq., 2022, 354, 118845 CrossRef CAS.
- A. Akinlua, M. A. Jochmann and T. C. Schmidt, Eng. Chem. Res., 2015, 54(51), 12960–12965 CrossRef CAS.
- M. Moniruzzaman, M. Tamura, Y. Tahara, N. Kamiya and M. Goto, Int. J. Pharm., 2010, 400(1–2), 243–250 CrossRef CAS PubMed.
- S. S. Y. Tan and D. R. MacFarlane, Ionic Liq., 2010, 311–339 Search PubMed.
- E. D. Bates, R. D. Mayton, I. Ntai and J. H. Davis, J. Am. Chem. Soc., 2002, 124(6), 926–927 CrossRef CAS PubMed.
- A. Lewandowski and A. Świderska-Mocek, J. Power Sources, 2009, 194(2), 601–609 Search PubMed.
- Y. Fan, W. Li, S. Zhang, S. Sun and L. Yang, Ind. Crops Prod., 2020, 152, 112563 CrossRef CAS.
- K. Dong, S. Zhang and J. Wang, Chem. Commun., 2016, 52(41), 6744–6764 RSC.
- I. A. Sedov and T. I. Magsumov, J. Chem. Thermodyn., 2022, 170, 106779 CrossRef CAS.
- M. V. Oliveira, B. T. Vidal, C. M. Melo, R. D. C. de Miranda, C. M. Soares, J. A Coutinho, S. P. Ventura, S. mattedi and Á. S. Lima, Chemosphere, 2016, 147, 460–466 CrossRef CAS PubMed.
- L. Chen, G. E. Mullen, M. Le Roch, C. G. Cassity, N. Gouault, H. Y. Fadamiro, R. E. Barletta, R. A. O'Brien, R. E. Sykora, A. C. Stenson, K. N. West, H. E. Horne, J. M. Hendrich, K. R. Xiang and J. H. Davis Jr, Angew. Chem., Int. Ed., 2014, 53(44), 11762–11765 CrossRef CAS PubMed.
- M. G. Freire, A. F. M. Claudio, J. M. Araujo, J. A. Coutinho, I. M. Marrucho, J. N. C. Lopes and L. P. N. Rebelo, Chem. Soc. Rev., 2012, 41(14), 4966–4995 RSC.
- K. Huang, M. Zhang, Y. Xu, Y. T. Wu, X. B. Hu and Y. Xu, AIChE J., 2014, 60(12), 4232–4240 CrossRef CAS.
- M. S. Malik, R. I. Alsantali, R. S. Jassas, A. A. Alsimaree, R. Syed, M. A. Alsharif, K. Kalpana, M. Morad, I. I. Althagafi and S. A. Ahmed, RSC Adv., 2021, 11(57), 35806–35827 RSC.
- M. J. A. Martínez and P. B. Benito, Stud. Nat. Prod. Chem., 2005, 30, 303–366 CrossRef.
- P. Mehnati, H. Khoshsima, M. S. Zakerhamidi, R. Kian and M. Sahrai, J. Mol. Struct., 2024, 1310, 138317 CrossRef CAS.
- S. N. Khan, B. Islam, R. Yennamalli, A. Sultan, N. Subbarao and A. U. Khan, Eur. J. Pharm. Sci., 2008, 35(5), 371–382 CrossRef CAS PubMed.
- K. D. Amirchand and V. Singh, J. Chem. Thermodyn., 2023, 184, 107069 CrossRef CAS.
- M. A. Siddiquee, J. Saraswat, M. ud din Parray, P. Singh, S. Bargujar and R. Patel, Spectrochim. Acta, Part A, 2023, 285, 121803 CrossRef PubMed.
- A. Sharma and P. Awasthi, J. Chem. Sci., 2022, 134(4), 97 CrossRef CAS.
- U. Farooq, R. Patel and A. Ali, J. Solution Chem., 2008, 47, 568–585 CrossRef.
- Y. Kumar, F. A. Wani, S. Ahmedi, A. Shamsi, M. Nadeem, N. Manzoor, M. R. Kamli, M. A. Malik, M. A. Rizwi and R. Patel, J. Mol. Liq., 2024, 401, 124631 CrossRef CAS.
- R. K. Mahajan, J. Chawla and M. S. Bakshi, Colloids Surf., A, 2004, 237(1–3), 119–124 CrossRef CAS.
- S. Mahajan, R. Sharma and R. K. Mahajan, Langmuir, 2012, 28(50), 17238–17246 CrossRef CAS PubMed.
- S. Huang, F. Zhu, Q. Xiao, Q. Zhou, W. Su, H. Qiu, B. Hu, J. Sheng and C. Huang, RSC Adv., 2014, 4(68), 36286–36300 RSC.
- A. Kumar, K. Kumari, S. Singh, I. Bahadur and P. Singh, J. Mol. Liq., 2021, 325, 115159 CrossRef CAS.
- R. Sharma, P. Awasthi, N. Kumari, M. Vatsal, A. Sharma, I. Bahadur, M. Kabanda and F. Mohammad, J. Mol. Liq., 2024, 416, 126457 CrossRef CAS.
- R. Sharma, P. Awasthi, M. Vatsal, V. Devi, A. Sharma, R. Dogra, I. Bahadur, F. Mohammad and A. A. Soleiman, J. Chem. Eng. Data, 2025, 70(3), 1250–1276 CrossRef CAS.
- P. Awasthi, A. Sharma and M. Vatsal, J. Mol. Struct., 2021, 1223, 129293 CrossRef CAS.
- M. Matczuk, A. R. Timerbaev, B. K. Keppler and L. Ruzik, J. Mol. Liq., 2024, 399, 124403 CrossRef CAS.
- M. K. Shukla, H. Tiwari, R. Verma, W. L. Dong, S. Azizov, B. Kumar, S. Pandey and D. Kumar, Pharm., 2023, 15, 702 CAS.
- A. M. Curreri, S. Mitragotri and E. E. L. Tanner, Adv. Sci., 2021, 8(17), 2004819 CrossRef CAS PubMed.
- M. Ting, L. Hu, Y. Wang, S. Wei, S. He, Q. Huang and Z. Wang, RSC Adv., 2025, 15, 14632–14642 RSC.
- K. Yuan, S. Chen, X. Chen, S. Yao, X. Wang, H. Song and M. Zhu, J. Mol. Liq., 2020, 314, 113342 CrossRef CAS.
- K. D. Amirchand and V. Singh, J. Chem. Eng. Data, 2022, 354, 118845 CAS.
- D. Singh, V. Singh and R. L. Gardas, J. Solution Chem., 2015, 44(3–4), 634–651 CrossRef CAS.
- T. L. Greaves, A. Weerawardena, C. Fong, I. Krodkiewska and C. J. Drummond, J. Phys. Chem. B, 2006, 110(45), 22479–22487 CrossRef CAS PubMed.
- M. Iglesias, A. Torres, R. Gonzalez-Olmos and D. Salvatierra, J. Chem. Thermodyn., 2008, 40, 119–133 CrossRef CAS.
- I. Cota, R. Gonzalez-Olmos, M. Iglesias and F. Medina, J. Phys. Chem. B, 2007, 111, 12468–12477 CrossRef CAS PubMed.
- V. H. Alvarez, N. Dosil, R. Gonzalez-Cabaleiro, S. Mattedi, M. Martin-Pastor, M. Iglesias and J. M. Navaza, J. Chem. Eng. Data, 2010, 55(2), 625–632 CrossRef CAS.
- J.-P. Belieres and C. A. Angell, J. Phys. Chem. B, 2007, 111(18), 4926–4937 CrossRef CAS PubMed.
- N. Li, Y. Ma, C. Yang, L. Guo and X. Yang, Biophys. Chem., 2005, 116(3), 199–205 Search PubMed.
- A. Pal and A. Yadav, J. Mol. Liq., 2016, 222, 471–479 Search PubMed.
- Y. Shu, M. Liu, S. Chen, X. Chen and J. Wang, J. Phys. Chem. B, 2011, 115(42), 12306–12314 CrossRef CAS PubMed.
- S. Sharma, M. K. Banjare, N. Singh, J. Korábečný, K. Kuča and K. K. Ghosh, RSC Adv., 2020, 10(64), 38873–38883 RSC.
- A. Siddiquee, Z. Parray, A. Anand, S. Tasneem, N. Hasan, W. M. Alamier, A. A. Ageeli, F. A. Wani, P. Singh and R. Patel, ACS Omega, 2023, 8(45), 42699–42710 CrossRef CAS PubMed.
- A. O. Brett, T. R. A. Macedo, D. Raimundo, M. H. Marques and S. H. P. Serrano, Anal. Chim. Acta, 1999, 385(1–3), 401–408 Search PubMed.
- A. Erdem and M. Ozsoz, Turk. J. Chem., 2001, 25(4), 469–476 Search PubMed.
- C. Zhao, G. Burrell, A. A. Torriero, F. Separovic, N. F. Dunlop, D. R. MacFarlane and A. M. Bond, J. Phys. Chem. B, 2008, 112(23), 6923–6936 CrossRef CAS PubMed.
- J. S. Murray and P. Politzer, Wiley Interdiscip. Rev.:Comput. Mol. Sci., 2011, 1(2), 153–163 CAS.
- P. Politzer, P. Lane, M. C. Concha, Y. Ma and J. S. Murray, J. Mol. Model., 2007, 13(3), 305–311 CrossRef CAS PubMed.
- R. Zulfiqar, D. Ali, M. A. Ali, A. Yousuf, M. Sarfraz, A. Rauf and M. Arshad, ChemistrySelect, 2025, 10(18), e202405487 CrossRef CAS.
- Y. Huang, D. Ouyang and Y. Ji, AIChE J., 2022, 68(6), e17672 CrossRef CAS.
- A. Siddiquee, Z. Parray, A. Anand, S. Tasneem, N. Hasan, W. M. Alamier and R. Patel, ACS Omega, 2023, 8(45), 42699–42710 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |