Open Access Article
Open Access ArticleA new dioxo-molybdenum complex derived from an amide-based imine derivative for the fluorescence recognition of Zr(IV) with phosphatase activity
Amit Kumar Dea,
Tandrim Shayma,
Subhasis Ghosha,
Sunanda Dograb,
Angshuman Roy Choudhury *b and
Debasis Das
*b and
Debasis Das *a
*a
aDepartment of Chemistry, The University of Burdwan, Burdwan-713104, WB, India. E-mail: ddas100in@yahoo.com
bDepartment of Chemical Sciences, Indian Institute of Science Education and Research (IISER), Knowledge City, Sector 81, S.A.S. Nagar, Manauli P.O., Mohali, Punjab 140306, India
First published on 27th February 2026
Abstract
An amide-imine conjugate, namely, 2,4-dihydroxy-benzoic acid-(2-hydroxy-benzylidene)-hydrazide [DBASL], was prepared from 2,4-dihydroxy benzoic acid hydrazide (DBA) and salicylaldehyde and further used for the synthesis of the corresponding Mo(VI) complex (DBASLM). The structure of DBASLM was established using different spectroscopic techniques and confirmed by single-crystal X-ray diffraction (SC-XRD) analysis. Interestingly, DBASLM selectively recognizes ZrO2+ via ‘turn-on’ sky-blue emission, with notably strong binding constant of 1.3 × 105 M−1. The proposed sensing mechanism involves the displacement of Mo(VI) from DBASLM by ZrO2+. DFT studies also corroborate the interactions and allow the exploration of the associated orbital energy parameters. Silica-immobilized DBASLM is very effective for the solid-phase extractive separation/recovery of ZrO2+. A DBASLM-impregnated paper strip allows the low-cost, facile and instant optical detection of ZrO2+ in real samples. DBASLM promotes phospho-ester bond hydrolysis in p-nitrophenyl phosphate (p-NPP), which is one of the vital biological processes that play a key role in the destruction of poisonous organo-phosphorus substrates such as pesticides and insecticides used in the agricultural industry.
Introduction
Zirconium (Zr) has pervasive applications in different fields, such as metallurgy, materials science, aerospace, atomic energy, medicine, and petrochemicals.1 Its valuable attributes include corrosion resistance and enhancement of mechanical strength of alloys that function over a wide range of temperatures.2–4 Zr materials have also been used extensively in artificial internal organs in the last few years5–7 as they offer low toxicity with excellent bio-compatibility.8,9 However, the risks involved in the use of Zr compounds depend on various factors such as the concentration, duration of exposure, and chemical form.10–12 Frequent contact with high-concentration Zr compounds may result in health issues such as pulmonary granuloma, skin irritation, carcinogenesis, and hypersensitivity pneumonitis.13–19 The National Institute for Occupational Safety and Health (NIOSH) has proposed the maximum exposure limit for Zr compounds as 5 mg L−1 for an 8 h workday, while it is 10 mg L−1 for short-term exposure.8Given that Zr(IV) sensors are relatively scarce, ample opportunity exists to develop probes for the rapid and inexpensive detection of Zr(IV). Among the various techniques and methods for its detection, which depend on several factors including concentration range, sample matrix, sensitivity and selectivity, the fluorescence method is superior and therefore in high demand.20
On the other hand, phospho-ester bond cleavage, one of the vital biological processes, plays a key role in the destruction of poisonous organo-phosphorus substrates such as pesticides and insecticides used in the agricultural industry.21–27 Eventually, they accumulate in humans via the food chain, causing harmful effects.22,23,28–30
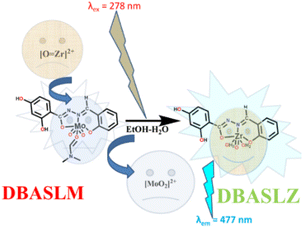
Considering the above-mentioned two facts, we have attempted to prepare materials that can serve both purposes, which are rarely found in the literature. Recently, a few probes have been reported that can selectively detect and determine Zr(IV) in acid media.31–33 Herein, we report a fluorescence probe (DBASLM) that can selectively detect Zr(IV) at neutral pH in the form of ZrO2+ via ‘turn-on’ sky-blue fluorescence (λex = 278 nm and λem = 477 nm) having the lowest limit of detection (LOD) of 23.1 nM and binding constant of 1.3 × 105 M−1. In addition, DBASLM efficiently cleaves the phospho-ester bond in poisonous organo-phosphorus substrates (p-nitrophenyl phosphate, p-NPP).
Experimental
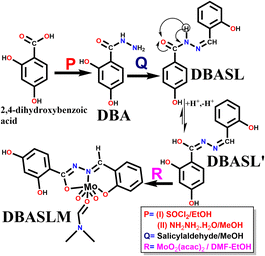
Synthesis (Scheme 1)
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O); 1050, ν (N–N) (Fig. S1d, SI).
O); 1050, ν (N–N) (Fig. S1d, SI).
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N); 1621, ν(C
N); 1621, ν(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N); 1084, ν(N–N) (Fig. S2d, SI).
N); 1084, ν(N–N) (Fig. S2d, SI).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMF, 2
DMF, 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, v/v) and DBASL (0.312 g, 1.15 mmol, 10 mL EtOH) was stirred for 2 h and left undisturbed for slow evaporation to yield orange crystals of DBASLM in 58% yield, molecular formula C20H24MoN4O8 (M.W. = 544.37). Anal. found (%), C, 43.91; H, 4.70 and N, 11.11; calcd (%) C, 44.06; H, 4.57 and N, 10.28. ESI-MS (m/z): [M + H]+, 474.0879 [including one DMF present in its crystal lattice] (Fig. S3a, SI). FT-IR (KBr, cm−1): 3404, ν(O–H); 3082, ν(C–H, aromatic); 1651, ν(–CH
3, v/v) and DBASL (0.312 g, 1.15 mmol, 10 mL EtOH) was stirred for 2 h and left undisturbed for slow evaporation to yield orange crystals of DBASLM in 58% yield, molecular formula C20H24MoN4O8 (M.W. = 544.37). Anal. found (%), C, 43.91; H, 4.70 and N, 11.11; calcd (%) C, 44.06; H, 4.57 and N, 10.28. ESI-MS (m/z): [M + H]+, 474.0879 [including one DMF present in its crystal lattice] (Fig. S3a, SI). FT-IR (KBr, cm−1): 3404, ν(O–H); 3082, ν(C–H, aromatic); 1651, ν(–CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N); 1610, ν(C
N); 1610, ν(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N)-imide; 1070, ν(N–N) (Fig. S3b, SI). The structure of DBASLM is confirmed by SC-XRD analysis.
N)-imide; 1070, ν(N–N) (Fig. S3b, SI). The structure of DBASLM is confirmed by SC-XRD analysis.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O, 1
H2O, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, v/v) solution of DBASLM (1 M) under stirring conditions. The resultant mixture was left undisturbed for slow evaporation of the solvent to get yellow-green DBASLZ in 48% yield, molecular formula C14H14N2O7Zr (M.W. = 413.49). Anal. found (%), C, 40.37; H, 3.46 and N, 6.82; calcd (%) C, 40.67; H, 3.41 and N, 6.77. ESI-MS (m/z): [M + H]+, 414.0481 (Fig. S4a, SI). FT-IR (KBr, cm−1): 3406, ν(O–H); 3078, ν(C–H, aromatic); 1645, ν(–CH
2, v/v) solution of DBASLM (1 M) under stirring conditions. The resultant mixture was left undisturbed for slow evaporation of the solvent to get yellow-green DBASLZ in 48% yield, molecular formula C14H14N2O7Zr (M.W. = 413.49). Anal. found (%), C, 40.37; H, 3.46 and N, 6.82; calcd (%) C, 40.67; H, 3.41 and N, 6.77. ESI-MS (m/z): [M + H]+, 414.0481 (Fig. S4a, SI). FT-IR (KBr, cm−1): 3406, ν(O–H); 3078, ν(C–H, aromatic); 1645, ν(–CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N); 1608, ν(C
N); 1608, ν(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N)-imide; 1065, ν(N–N) (Fig. S4b, SI).
N)-imide; 1065, ν(N–N) (Fig. S4b, SI).Results and discussion
Single-crystal X-ray diffraction analysis
The molecular structure of DBASLM (CCDC No. 2448883) has been authenticated by single-crystal X-ray diffraction analysis and presented in Fig. 1 and its packing diagram in Fig. S5a (SI). Its crystallographic parameters [triclinic, space group: P1, volume (Å3): 1132.43(7); Z = 2] are presented in Table S1 (SI). Selected bond lengths and bond angles are presented in Table S2 (SI). The geometry of DBASLM is distorted octahedral (Fig. 1). The bond lengths of O3–C1 [1.327(3) Å] for the amide C–O bond and O7–C10 [1.360(3) Å] for the phenol C–O bond are close and signify single-bond character (Fig. S5b, SI). The partial double-bond character is reflected by the bond lengths of N1–C1 [1.307(3) Å], N2–C8 [1.288(3) Å], N1–N2 [1.392(3) Å], C1–C2 [1.465(4) Å] and C8–C9 [1.448(4) Å]. Two Mo![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O groups, viz. Mo–O4 [1.693(2) Å] and Mo–O6 [1.711(2) Å] are in the cis-geometry and indicate double-bond character. On the other hand, the bond lengths of Mo1–O3 [2.007(2) Å], Mo–O7 [1.932(2) Å], and Mo–O5 [2.322(2) Å] indicate Mo–O single-bond character. The solvent DMF coordinated to Mo(VI) via the carbonyl oxygen (–O5). The bond angles of O3–Mo–O7 [149.35(6)°], O4–Mo–O5 [171.59(9)°] and O6–Mo–N2 [162.6(1)°] indicate that DBASLM exhibits a distorted octahedral geometry.
O groups, viz. Mo–O4 [1.693(2) Å] and Mo–O6 [1.711(2) Å] are in the cis-geometry and indicate double-bond character. On the other hand, the bond lengths of Mo1–O3 [2.007(2) Å], Mo–O7 [1.932(2) Å], and Mo–O5 [2.322(2) Å] indicate Mo–O single-bond character. The solvent DMF coordinated to Mo(VI) via the carbonyl oxygen (–O5). The bond angles of O3–Mo–O7 [149.35(6)°], O4–Mo–O5 [171.59(9)°] and O6–Mo–N2 [162.6(1)°] indicate that DBASLM exhibits a distorted octahedral geometry.
 | ||
| Fig. 1 Thermal ellipsoid plot of the asymmetric unit of DBASLM, where H atoms are excluded for clarity. | ||
Spectroscopic studies
DBASL interacts with common cations such as Zn2+, Cd2+, Al3+, ZrO2+, La3+ and Y3+ to produce fluorescence (Fig. S6a, SI). The interaction of DBASLM with common cations, including rare-earth ions, viz., K+, Na+, Fe2+, Mg2+, Mn2+, Ni2+, Zn2+, Ca2+, Cd2+, Co2+, Cu2+, Al3+, Cr3+, Fe3+, ZrO2+, Ce4+, Dy3+, Gd3+, La3+, Nd3+, Sm3+ and Y3+ (as nitrate salts), have been studied in different common media, namely, MeOH, EtOH, propanol, DMSO and THF (Fig. S6b–n, SI, respectively). It is found that DBASLM selectively recognizes ZrO2+ in EtOH–H2O (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7, v/v). DBASLM exhibits a higher emission intensity at λex = 278 nm in the presence of ZrO2+ in an ethanol–water system (Fig. S6o–q, SI). It is interesting to note that the emission intensity of the system decreases with an increasing water percentage in aqueous ethanol (Fig. S7a, SI). Interference from Y3+ may be eliminated using Na2EDTA (Fig. S8a and b, SI). Moreover, the emission intensity of the probe in the presence and absence of ZrO2+ varies with pH (Fig. S9, SI). The optimum sensing efficiency has been observed at around pH 7, which was maintained in all experiments using PBS buffer.
7, v/v). DBASLM exhibits a higher emission intensity at λex = 278 nm in the presence of ZrO2+ in an ethanol–water system (Fig. S6o–q, SI). It is interesting to note that the emission intensity of the system decreases with an increasing water percentage in aqueous ethanol (Fig. S7a, SI). Interference from Y3+ may be eliminated using Na2EDTA (Fig. S8a and b, SI). Moreover, the emission intensity of the probe in the presence and absence of ZrO2+ varies with pH (Fig. S9, SI). The optimum sensing efficiency has been observed at around pH 7, which was maintained in all experiments using PBS buffer.
Absorption spectroscopic studies
Studies on the spectroscopic interaction of common cations (mentioned supra) with DBASLM were performed in EtOH–H2O (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7, v/v) media. The parent ligand, DBASL, absorbs at 335 nm, which is almost the same in different solvents (Fig. S9a, SI). Upon the addition of Mo(VI) to DBASL, its absorbance increases significantly with a minor blue shift to 327 nm (Fig. 2a), reflecting the notable interaction between DBASL and Mo(VI).
7, v/v) media. The parent ligand, DBASL, absorbs at 335 nm, which is almost the same in different solvents (Fig. S9a, SI). Upon the addition of Mo(VI) to DBASL, its absorbance increases significantly with a minor blue shift to 327 nm (Fig. 2a), reflecting the notable interaction between DBASL and Mo(VI).
 | ||
Fig. 2 Changes in the absorption spectra of DBASL and DBASLM (50 µM) upon the addition of (a) common cations [1500 µM] and (b) ZrO2+ [0–1000 µM] in EtOH–H2O (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7, v/v, PBS buffer, pH 7.0). 7, v/v, PBS buffer, pH 7.0). | ||
On the other hand, upon the gradual addition of ZrO2+ to DBASLM, its absorbance at 327 nm decreases notably, while a new peak appears at 391 nm, along with an isosbestic point at 353 nm (Fig. 2b). This is attributed to the displacement of Mo(VI) from DBASLM by ZrO2+.
Emission spectroscopic studies
The addition of ZrO2+ to DBASLM results in an intense sky-blue emission at 477 nm (λex = 278 nm, Fig. 3a) (Fig. S7b, SI). Although most other common cations (mentioned supra) remain silent towards DBASLM, Y3+ enhances its fluorescence to some extent (Fig. S10, SI), and also interferes to some extent in a competitive environment (Fig. S11, SI). The emission intensity of DBASLM is gradually enhanced upon the addition of ZrO2+ (Fig. 3b). | ||
Fig. 3 Changes in the emission spectra of DBASLM (50 µM) upon the addition of (a) common cations (supra) [1500 µM] and (b) ZrO2+ [0–3000 µM] in EtOH–H2O (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7, v/v, PBS buffer). 7, v/v, PBS buffer). | ||
Job's plot discloses a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometry (mole ratio) for the [DBASLM–ZrO2+] adduct (Fig. S12a, SI), which is also supported by mass spectra. The binding constants for DBASLM and DBASL for ZrO2+ were determined using the Benesi–Hildebrand equation (considering 1
1 stoichiometry (mole ratio) for the [DBASLM–ZrO2+] adduct (Fig. S12a, SI), which is also supported by mass spectra. The binding constants for DBASLM and DBASL for ZrO2+ were determined using the Benesi–Hildebrand equation (considering 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 binding, Fig. S12b and d, SI, respectively). The corresponding LOD values for ZrO2+ were calculated using Fig. S13b and d, respectively (SI) and presented in Table 1. The plot of emission intensity of DBASLM vs. ZrO2+ is linear up to 23.1 nM ZrO2+ (Fig. S14, SI) and useful for the determination of an unknown ZrO2+ concentration. The effect of solvents reveals that its emission intensity is higher in polar protic coordinating solvents such as MeOH, EtOH, and propanol, while a lower intensity is observed in polar aprotic coordinating solvents such as DMSO and THF at relatively shorter wavelengths. On the other hand, in non-coordinating solvents such as CCl4, CHCl3, and CH2Cl2, no emission is observed (Fig. S6c, SI). These observations are in line with the literature that the emission intensities and excitation wavelengths are both influenced by the solvent polarity.
1 binding, Fig. S12b and d, SI, respectively). The corresponding LOD values for ZrO2+ were calculated using Fig. S13b and d, respectively (SI) and presented in Table 1. The plot of emission intensity of DBASLM vs. ZrO2+ is linear up to 23.1 nM ZrO2+ (Fig. S14, SI) and useful for the determination of an unknown ZrO2+ concentration. The effect of solvents reveals that its emission intensity is higher in polar protic coordinating solvents such as MeOH, EtOH, and propanol, while a lower intensity is observed in polar aprotic coordinating solvents such as DMSO and THF at relatively shorter wavelengths. On the other hand, in non-coordinating solvents such as CCl4, CHCl3, and CH2Cl2, no emission is observed (Fig. S6c, SI). These observations are in line with the literature that the emission intensities and excitation wavelengths are both influenced by the solvent polarity.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7, v/v)
7, v/v)
| Probe | Binding constant (M−1) | LOD (nM) |
|---|---|---|
| DBASLM | 1.3 × 105 | 23.1 |
| DBASL ligand | 1.5 × 105 | 21.9 |
Sensing mechanism
The weak emissions of DBASL are attributed to the photo-induced electron transfer (PET) process that arises from its amide –N to –CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N-portions. Mo(VI) inhibits the PET and –CH
N-portions. Mo(VI) inhibits the PET and –CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N-isomerisation, although the anticipated fluorescence enhancement is absent, which depends on several factors such as the nature of the metal ion and its oxidation state and electronic energy levels. Interestingly, only in the presence of ZrO2+, both the parent DBASL and the probe (DBASLM) experience a fluorescence enhancement (Fig. S7b, SI). One reason for this may be the comparatively lower charge of Zr(IV) over Mo(VI), which shows CHEF in the case of DBASL, while the other one is the selective replacement of Mo(VI) in DBASLM by ZrO2+, which may have several reasons for its better fit. It should be noted that almost similar absorption and emission profiles (Fig. 2a and S7b, SI) were observed when ZrO(NO3)2 was added separately to DBASL and DBASLM solutions. The ESI-MS (m/z) spectrum and single-crystal X-ray structure support the formation of DBASLM. In brief, the addition of ZrO2+ turns the fluorescence of DBASLM ON, which is attributed to the metal displacement process, where Mo(VI) is replaced by ZrO2+ due to the formation of strong Zr–O and Zr–N bonds,31,34,36,37 resulting in [DBASL–Zr(IV)] (DBASLZ, Scheme 2).38,39 This is reflected in the corresponding ESI-MS spectrum, having (m/z) of [M + H]+ = 414.0481. Job's plot also suggests the formation of a 1
N-isomerisation, although the anticipated fluorescence enhancement is absent, which depends on several factors such as the nature of the metal ion and its oxidation state and electronic energy levels. Interestingly, only in the presence of ZrO2+, both the parent DBASL and the probe (DBASLM) experience a fluorescence enhancement (Fig. S7b, SI). One reason for this may be the comparatively lower charge of Zr(IV) over Mo(VI), which shows CHEF in the case of DBASL, while the other one is the selective replacement of Mo(VI) in DBASLM by ZrO2+, which may have several reasons for its better fit. It should be noted that almost similar absorption and emission profiles (Fig. 2a and S7b, SI) were observed when ZrO(NO3)2 was added separately to DBASL and DBASLM solutions. The ESI-MS (m/z) spectrum and single-crystal X-ray structure support the formation of DBASLM. In brief, the addition of ZrO2+ turns the fluorescence of DBASLM ON, which is attributed to the metal displacement process, where Mo(VI) is replaced by ZrO2+ due to the formation of strong Zr–O and Zr–N bonds,31,34,36,37 resulting in [DBASL–Zr(IV)] (DBASLZ, Scheme 2).38,39 This is reflected in the corresponding ESI-MS spectrum, having (m/z) of [M + H]+ = 414.0481. Job's plot also suggests the formation of a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 complex (Fig. S12a, SI). The sensing protocol is further substantiated by 1HNMR spectroscopy and DFT studies.
1 complex (Fig. S12a, SI). The sensing protocol is further substantiated by 1HNMR spectroscopy and DFT studies.
1HNMR spectral studies
The 1HNMR spectral studies reveal the mode of binding of DBASLM with ZrO2+. Upon the addition of MoO2(acac)2 (1.0 equiv.) to DBASL, the protons of the phenol –OH in the salicylaldehyde moiety (c, 11.280 ppm) and imidic (d, 10.321 ppm) acid moiety disappear.37 The imine proton (e, 8.655 ppm) also shifted downfield to 8.661 ppm. On the other hand, when ZrO2+ (1.0 equiv.) was added to the DBASLM complex, the peaks for the phenol –OH proton (c, 11.280 ppm) and imidic acid (d, 10.321 ppm) were still absent. The imine proton (e, 8.655 ppm) also shifted downfield to 8.723 ppm (Fig. S4c, SI).34,35DFT studies
The theoretical energy parameters of the corresponding frontier molecular orbitals in the process of probe-analyte interaction were computed by density functional theory (DFT) studies.40 DFT was performed with DBASL, DBASLM, and DBASLZ molecule/complex/adduct using the TD-SCF/DFT/B3LYP/6-31G(d) (for C, H, N and O) and LANL2DZ (for Mo and Zr) basis sets.41 The energy gaps between the highest occupied molecular orbitals (HOMO) and lowest unoccupied molecular orbitals (LUMO) for DBASL, DBASLM and DBASLZ are important parameters (Fig. 4). The energy difference between the HOMO–LUMO for DBASL is 0.07777 eV, where the HOMO–LUMO energy gap for DBASLM decreased by 0.01924 eV. The corresponding value for DBASLM is 0.05853 eV, indicating the stability of the complex. The related energy difference for the DBASLZ adduct is 0.04813 eV. Here, a slight decrease in the energy gap for DBASLM by 0.01040 eV and DBASL by 0.02964 eV is observed. Moreover, the HOMO and LUMO energies also decreased in the same order from DBASL to DBASLZ via DBASLM. Therefore, the addition of ZrO2+ to DBASLM lowers the HOMO–LUMO energy gap and is associated with a red shift, demonstrating that the experimental observations match the theoretical values.37Solid-phase extraction
For the immobilization of DBASLM on silica (100–200 mesh), 1.8 g DBASLM and 8.5 g silica (100–200 mesh) were mixed and refluxed for 4 h in methanol.42,43 After removal of the solvent, DBASLM immobilized on silica was obtained.42,43 A glass column (10 cm × 1 cm) was filled with the DBASLM-immobilized silica to a height of approximately 7 cm. A real water sample spiked with ZrO(NO3)2 was passed through the column at a flow rate of 1 mL min−1. Upon exposure to UV light, the colour of the silica bed changed to sky blue, indicating the sorption of ZrO2+ (Fig. 5). The sorbed ZrO2+ could be eluted by a DCM–ethyl acetate (3/7, v/v) mixture. After evaporating the solvent, the residue was used to prepare a solution in EtOH–H2O and the concentration of ZrO2+ was measured following the developed method (Table 2). Comparison of the present probe with existing pioneering ZrO2+ selective probes are presented in Table 3.| Test | ZrO2+ added (10−2 M) | Found (10−2 M) | Recovery (%) |
|---|---|---|---|
| 1 | 8.85 | 6.43 | 72.90 ± 1.08 |
| 2 | 10.60 | 7.42 | 70.00 ± 1.25 |
| 3 | 12.80 | 8.48 | 66.25 ± 1.35 |
| Sl. No. | Probe | Method | pH | LOD (nM) | Ref. |
|---|---|---|---|---|---|
| 1 | Phosphorylated and pyrene-labeled oligonucleotides | Fluorescence | 7.4 | 200 | 46 |
| 2 | Histidine-functionalized gold nanoparticles | Colorimetric and chrominance | 7.0 | 2620 and 6250 | 47 |
| 3 |  |
Colorimetric | 6.5 | 82.23 | 31 |
| 4 |  |
Colorimetric and fluorescence | Non-aqueous medium | 50 and 0.90 | 34 |
| 5 |  |
Potentiometric | 4.8 | 18 | 48 |
| 6 |  |
PVC membrane-based electrode | 1.8–4.2 | 600 | 49 |
| 7 |  |
Fluorescence | 7.0 | 23.1 | Present work |
Visualization of ZrO2+ on a paper strip soaked with DBASLM
The paper strip method for the visualization of ZrO2+ utilizing DBASLM is cost-effective and instanteneous.44,45 A drop of ZrO2+ solution was placed on a colourless paper strip soaked with DBASLM and air dried. Sky-blue emission was observed upon the irradiation of the above-mentioned paper strip with UV light, indicating the presence of ZrO2+ (Fig. 6).Studies on p-nitrophenyl-phosphate (p-NPP) activity
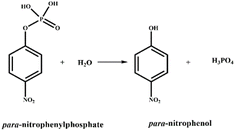
Phosphoester bond hydrolysis of p-nitrophenyl phosphate (p-NPP) is a very slow process, but its reaction rate (Scheme 3) may be enhanced using a catalyst.30,50,51 In the present case, a catalytic amount of DBASLM (0.4 mmol, 10 mol% relative to p-NPP) was reacted with p-NPP (4.0 mmol) in MeOH–H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4, v/v, pH 7, PBS buffer) and an increase in absorbance at 410 nm was observed with time due to the formation of yellow p-nitrophenol (Fig. 7).50 The proposed mechanism is supported by the QTOF mass spectra with the m/z value of 139.0286 (calcd m/z: 139.11) (Fig. S15, SI).52 The absorbance vs. time plot revealed the progress of the reaction (Fig. S16, SI).53 The hydrolysis rate constants derived from Fig. S17 (SI) at 25 °C are presented in Table 4.
4, v/v, pH 7, PBS buffer) and an increase in absorbance at 410 nm was observed with time due to the formation of yellow p-nitrophenol (Fig. 7).50 The proposed mechanism is supported by the QTOF mass spectra with the m/z value of 139.0286 (calcd m/z: 139.11) (Fig. S15, SI).52 The absorbance vs. time plot revealed the progress of the reaction (Fig. S16, SI).53 The hydrolysis rate constants derived from Fig. S17 (SI) at 25 °C are presented in Table 4.
| Substrate | Catalyst | Rate constant (s−1) |
|---|---|---|
| p-NPP (0.02–10.0 mmol) | Blank | 1.86 × 10−5 |
| p-NPP (0.02–10.0 mmol) | DBASL (0.01 mmol) | 4.00 × 10−5 |
| p-NPP (0.02–10.0 mmol) | DBASLM (0.01 mmol) | 1.84 × 10−4 |
Conclusion
An amide-imine (DBASL)-derived Mo(IV) complex, structurally authenticated by single-crystal X-ray diffraction analysis, has been employed for the selective recognition of ZrO2+ [Zr(IV)] via its sky-blue (λem = 477 nm and λex = 278 nm) emission in aqueous EtOH media. Its sensing is attributed to the displacement of Mo(VI) from DBASLM by ZrO2+, resulting in sky-blue fluorescence. The limit of detection for DBASLM for ZrO2+ is 23.1 nM. Silica-immobilized DBASLM is effective for the solid-phase extractive recovery of ZrO2+. A DBASLM-impregnated paper strip is also useful for the optical detection of ZrO2+. DBASLM efficiently catalyses the hydrolysis of the phospho-ester bond in p-NPP.Conflicts of interest
We have no conflicts of interest to disclose.Data availability
Supplementary information (SI): mass spectra, 1HNMR, 13CNMR, FTIR, binding constant and LOD plots, Job's plot, interference plots and crystal parameters. See DOI: https://doi.org/10.1039/d5ra04804d.Acknowledgements
We thank Dr Saurabh Das, Professor, Dept. of Chemistry, Jadavpur University, for the FT-IR facility. Sunanda thanks UGC, India, for research fellowship, and ARC thanks Department of Chemical Sciences, IISER Mohali, for the XtaLAB mini X-ray diffractometer facility.Notes and references
- Y. Ma, S. Stopic, X. Wang, K. Forsberg and B. Friedrich, Metals, 2020, 10, 1099–1112 Search PubMed.
- A. S. Amin, J. Taibah Univ. Sci., 2015, 9, 227–236 CrossRef.
- S. Ganesh, P. Velavendan, N. K. Pandey, U. K. Mudali and R. Natarajan, Int. J. Adv. Chem., 2015, 1, 53–59 Search PubMed.
- T.-L. Yau and V. E. Annamalai, Reference module in materials science and materials engineering, Elsevier, 2016 Search PubMed.
- D. S. Nakonieczny, A. Ziębowicz, Z. K. Paszenda and C. Krawczyk, Biocybern. Biomed. Eng., 2017, 37, 229–245 CrossRef.
- G. Singh, S. Khurana, A. Devi, A. Singh, D. R. Batish and A. Sharma, Int. J. Biol. Macromol., 2024, 261, 129689 CrossRef CAS PubMed.
- M. Piconi and G. Maccauro, Biomaterials, 1999, 20, 1–25 CrossRef PubMed.
- J. B. Asha and P. Suresh, ACS Sustainable Chem. Eng., 2020, 8, 14301–14311 CrossRef CAS.
- A. G. Evanoff and G. Chumanov, Chem. Rev., 2006, 106, 430–448 Search PubMed.
- S. Ghosh, A. Sharma and G. Talukder, Biol. Trace Elem. Res., 1992, 35, 247–271 CrossRef CAS PubMed.
- D. B. N. Lee, M. Roberts, C. G. Bluchel and R. A. Odell, ASAIO J., 2010, 56, 550–556 Search PubMed.
- R. A. Bapat, H. J. Yang, T. V. Chaubal, S. Dharmadhikari, A. M. Abdulla, S. Arora, S. Rawal and P. Kesharwani, RSC Adv., 2022, 12, 12773–12793 Search PubMed.
- L. Romeo, A. Cazzadori, L. Bontempini and S. Martini, Med. Lav., 1994, 85, 219–222 Search PubMed.
- E. Bingham, B. Cohrssen and C. H. Powell, Patty's toxicology, John Wiley & Sons, New York, 5th edn, 2001, pp. 45–46 Search PubMed.
- K. K. Liippo, S. L. Anttila, O. Taikina-Aho, E.-L. Ruokonen, S. T. Toivonen and T. Tuomi, Am. Rev. Respir. Dis., 1993, 148, 1089–1092 CrossRef CAS PubMed.
- J. E. Abraham and D. H. Hunt, Arch. Environ. Health, 1964, 8, 627–632 Search PubMed.
- IPCS International Programme on Chemical Safety, Zirconium and its Compounds, UK PID 90, WHO/PCS/INC/95.90.
- P. A. Curtis, Contact Dermat., 1980, 6, 319–322 Search PubMed.
- P. L. Currance, B. Clements, and A. C. Bronstein, Emergency care for hazardous materials exposure, Mosby, Elsevier, St. Louis, MO, 3rd edn, 2007 Search PubMed.
- D.-E. Zacharioudaki, I. Fitilis and M. Kotti, Molecules, 2022, 27, 4801 Search PubMed.
- L. Y. Kuo, S. Kuhn and D. Ly, Inorg. Chem., 1995, 34, 5341 CrossRef CAS.
- H. Morales-Rojas and R. A. Moss, Chem. Rev., 2002, 102, 2497 CrossRef CAS PubMed.
- J. Fanzo, R. Remans, and P. Sanchez, in The chemical element: chemistry's contribution to our global future, ed. J. Garcia-Martinez and E. Serrano-Torregrosa, Wiley-VCH Verlag, Weinheim, 2011 Search PubMed.
- A. N. Bigley and F. M. Raushel, Chem. Biol. Interact., 2019, 308, 80–88 CrossRef CAS PubMed.
- K. Lai, N. J. Stolowich and J. R. Wild, Arch. Biochem. Biophys., 1995, 318, 59–64 CrossRef CAS PubMed.
- K. Alejo-González, E. Hanson-Viana and R. Vazquez-Duhalt, J. Pestic. Sci., 2018, 43, 1–9 Search PubMed.
- E. Dyguda-Kazimierowicz, S. Roszak and W. A. Sokalski, J. Phys. Chem. B, 2014, 118, 7277–7289 CrossRef CAS PubMed.
- I. Mali, C. Shah, B. H. Raghunandan, A. S. Prajapati, D. H. Patel, U. Trivedi and R. B. Subramanian, J. Environ. Sci., 2023, 127, 234–250 Search PubMed.
- S. Ray and S. T. Shaju, Environ. Anal. Health Toxicol., 2023, 38, e2023017 CrossRef PubMed.
- C. M. Tomé, M. C. Oliveira, M. Pillinger, I. S. Gonçalves and M. Abrantes, Dalton Trans., 2013, 42, 3901–3907 RSC.
- A. A. Sari, A. O. Babalghith, R. El-Sayed and A. S. Amin, Results Chem., 2024, 7, 101513 Search PubMed.
- U. D. Uysal, A. A. Huseyinli and T. Güray, J. Sci. Ind. Res., 2011, 70, 45–50 Search PubMed.
- T. A. Lasheen, G. Hussein, Y. Khawassek and M. Cheira, Anal. Chem. Indian J., 2013, 12, 368–376 CAS.
- G. Singh, Heena, S. Khurana, Mithun, B. S. Gill, D. Baliyan, Vikas, N. Dege and E. Tarcan, Appl. Organomet. Chem., 2025, 39, e7983 CrossRef CAS.
- D. Marsh, BioMed Res. Int., 2015, 2015, 746980 Search PubMed.
- W. B. Blumenthal, J. Chem. Educ., 1949, 26, 472 CrossRef CAS.
- S. Chatterjee, S. Ta, S. Khanra and D. Das, RSC Adv., 2022, 12, 33293–33303 RSC.
- T. Shyam, S. Lohar and D. Das, Inorg. Chim. Acta, 2025, 583, 122689 CrossRef CAS.
- J. Cheng, X. Zhou and H. Xiang, Analyst, 2015, 140, 7082 Search PubMed.
- A. Ashraf, J. M. Herbert, S. Muhammad, B. A. Farooqi, U. Farooq, M. Salman and K. Ayub, ACS Omega, 2022, 7, 2260–2274 CrossRef CAS PubMed.
- Y. Yang, M. N. Weaver and K. M. Merz Jr, J. Phys. Chem. A, 2009, 113, 9843–9851 CrossRef CAS PubMed.
- L. D. Nguyen, T. X. H. Dao, C. T. G. Hua and P. H. Tran, J. Mol. Liq., 2024, 396, 123859 CrossRef.
- K. Szentmihályi, S. Klébert, Z. May, E. Bódis, M. Mohai, L. Trif, T. Feczkó and Z. Károly, Pharmaceutics, 2022, 14, 2332 CrossRef PubMed.
- T. Pan, T.-Y. Gao, X.-H. Fan, M.-L. Sa, X.-J. Yang, J.-N. Xu, X. Xu, M. Ma, R. Wang, Y. Zhang, W. Ye, Y.-P. Shi, H.-X. Zhang and Z.-C. Zeng, Talanta, 2025, 281, 126754 Search PubMed.
- T. Shyam, S. Ghosh, S. Maji, P. Mandal and D. Das, New J. Chem., 2023, 47, 14315–14322 RSC.
- H.-M. Meng, T. Fu, X.-B. Zhang, N.-N. Wang, W. Tan, G.-L. Shen and R.-Q. Yu, Anal. Chem., 2012, 84, 2124–2128 Search PubMed.
- Z. Liu, Y. Yan, J. Li, W. Zhou, H. Gao and R. Lu, Anal. Sci., 2024, 40, 1269–1278 Search PubMed.
- M. B. Gholivand, A. Babakhanian and M. Joshaghani, Anal. Chim. Acta, 2007, 584, 302–307 Search PubMed.
- H. A. Arida, Talanta, 2008, 76, 40–43, DOI:10.1016/j.talanta.2008.01.061.
- S. T. Frey, B. M. Hutchins, B. J. Anderson, T. K. Schreiber and M. E. Hagerman, Langmuir, 2003, 19, 2188–2192 CrossRef CAS.
- Y. Zhang, F. Wang, X. Liu, Y. Zhang and J. Liu, Chem. Rev., 2018, 118, 11618–11705 Search PubMed.
- Q. Zhang, J. Shu, Y. Zhang, Z. Xu, J. Yue, X. Liu, B. Xu, Z. Chen and W. Jiang, Dalton Trans., 2020, 49, 10261 Search PubMed.
- Z. Liang, K. Liu, Y. Li, Y. Liu, C. Jiang, T. Zhang and W. Chen, Environ. Sci.: Nano, 2025, 12, 1364 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |