Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Pd@CN-COF: a pool for palladium(0)-catalyzed reactions
Marcela Horáková a,
Milan Klikar
a,
Milan Klikar a,
Jakub Halamek
a,
Jakub Halamek b,
Roman Bulánek
b,
Roman Bulánek b,
Jhonatan Rodriguez-Pereira
b,
Jhonatan Rodriguez-Pereira c,
Veronika Čičmancová
c,
Veronika Čičmancová c,
Jan Podlesný
c,
Jan Podlesný d,
Eva Kolíbalová
d,
Eva Kolíbalová e,
Jan Michalička
e,
Jan Michalička e,
Jan Bartáček
e,
Jan Bartáček a,
Patrik Pařík
a,
Patrik Pařík a,
Jan M. Macak
a,
Jan M. Macak ce and
Filip Bureš
ce and
Filip Bureš *a
*a
aInstitute of Organic Chemistry and Technology, Faculty of Chemical Technology, University of Pardubice, Studentská 573, Pardubice, 53210, Czechia. E-mail: filip.bures@upce.cz; Web: https://www.bures.upce.cz
bDepartment of Physical Chemistry, Faculty of Chemical Technology, University of Pardubice, Studentská 573, Pardubice, 53210, Czechia
cCenter of Materials and Nanotechnologies – CEMNAT, Faculty of Chemical Technology, University of Pardubice, nám. Čs. legií 565, Pardubice, 53002, Czechia
dInstitute of Technology and Business in České Budějovice, Okružní 517/10, České Budějovice, 37001, Czechia
eCentral European Institute of Technology, Brno University of Technology, Purkyňova 123, Brno, 61200, Czechia
First published on 2nd March 2026
Abstract
A new cyano-substituted olefin-linked COF was synthesized and utilized as a support for palladium nanoparticles. The resulting Pd@CN-COF was demonstrated as a versatile heterogeneous catalyst in cross-coupling reactions, cyanation, and allylic substitution with the productivity exceeding the commercial homogeneous precatalyst [Pd(PPh3)4]. The catalytic activity of Pd@CN-COF was further evaluated in synthetically valuable transformations towards industrially, agrochemically, and pharmacologically relevant products.
Introduction
Ordered porous materials, especially inorganic zeolites/mesoporous silica, hybrid metal organic frameworks (MOFs), and covalent organic frameworks (COFs), play a pivotal role in modern materials science and catalysis.1 The latter class of 2D or 3D porous organic polymers is characterized by a highly ordered and periodic structure achieved through the rational selection of building units and tailor-made synthesis.2 Inspired by the well-established materials such as graphite and boron nitride, the first boron-based COFs were prepared by Yaghi et al.3 This breakthrough achievement has initiated intense research4 lasting two decades and resulting in the current extensive library of available COFs designed towards manifold applications.5 Whereas the nanoscale pores in COF scaffolds provide an ideal environment for gas storage,6 the ordered columnar arrangement enabling charge carrier transport makes COFs promising candidates for energy-storage7 and optoelectronic8 applications. Furthermore, the COF's high surface area is advantageous for chemical sensing9 and catalysis.10The COF-mediated heterogeneous catalysis has proven to be a versatile platform for various chemical transformations, stemming from the robust and insoluble character of COF scaffolds, ensuring stability and facile recyclability of the catalyst.10 Moreover, the structure of organic COFs can be easily tailored to achieve the desired catalytic performance. In principle, the catalytic activity can originate from either the COF (functionalized backbone and pore walls)11 or the COF pores may accommodate an external catalytic species such as single atoms,12 molecules,13 metal complexes,14 and nanoparticles.15 The first example of Pd(OAc)2 coordinated between the COF layers reported by Wang et al. revealed the application potential of COFs as ligands in the Suzuki–Miyaura cross-coupling.16 Since then, COF-supported heterogeneous catalysts have been employed in a wide range of catalytic processes, including photocatalysis17 and electrocatalysis.18 The current research interest focused on the COF-supported (noble) metal nanoparticles stems from their exceptional properties and catalytic performance.19 Principally, the COFs are used to stabilize nanoparticles via functionalization of their surface, which inherently affects their interaction with the given substrate and thus allows modulation of the catalytic performance.20
Targeting robust Pd-catalysis towards valuable industrial and pharmacological products21 and inspired by the pioneering work of Wang et al.16 and the recent study by Yang et al.22 (Fig. 1), we report herein the design, synthesis, and properties of a new cyano-substituted olefin-linked COF, CN-COF, and its application as a support for the in situ generated palladium nanoparticles (Pd NPs). The resulting Pd@CN-COF is demonstrated to act as a versatile Pd precatalyst in seven, synthetically valuable, palladium-catalyzed transformations, including various cross-coupling reactions, catalytic cyanation, and allylic substitution.
Results and discussion
CN-COF preparation and characterization
The synthesis of CN-COF was performed through a Knoevenagel condensation of 1,3,5-tris(4-formylbiphenyl)-benzene (TFBB) and (benzene-1,3,5-triyl)triacetonitrile (BTAN) as outlined in Fig. 2a. Upon multiple optimization experiments using different solvents and bases (Table S1), the optimal solvothermal conditions (ortho-dichlorobenzene (o-DCB)/n-butanol (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) and aq. NaOH (5 M)) afforded CN-COF as a yellow fluffy solid with high crystallinity, bright solid-state fluorescence, and in a reproducible way (∼90% yield). The model compound MC was obtained by reacting TFBB with phenylacetonitrile in the presence of KOH; see the SI for more details. The constitution and the connectivity of the novel CN-COF were confirmed by FT-IR, Raman, and solid-state 13C CP/MAS NMR spectroscopies. As compared to TFBB, the FT-IR spectrum of CN-COF (Fig. 2b) shows a significant decrease in the intensity of the carbonyl stretching band (∼1700 cm−1), indicating a high degree of polymerization between the building units. This is further supported by a shift of the nitrile stretching band from 2248 (BTAN) to 2211/2213 cm−1 (MC/CN-COF), which is attributed to the conjugation of the nitrile groups with the newly formed olefinic linkages. Due to the low intensity of these FT-IR signals, we have unambiguously confirmed the presence of cyano groups also by Raman spectroscopy (Fig. S11). A very similar peak pattern was observed in the 13C CP/MAS NMR spectra of CN-COF and MC, even when compared to 13C NMR spectrum of MC in the solution (Fig. S3). The crystal structure of CN-COF was investigated by the powder XRD analysis (PXRD). The experimentally obtained diffractogram was optimized by the Le Bail refinement (Fig. S14 and S15) and further compared with the patterns calculated for the AA and AB stacking modes (Fig. 2c). The comparison revealed a close agreement between the observed diffraction lines at low angles (< ∼12°), the (100), (110), (200), and (210), of both model data sets and the experiment. At higher angles, a very broad (002) diffraction line centered at approximately 24° was experimentally observed, which corresponds to the interlayer distance of hexagonal layers. The experimentally obtained diffractogram matches well with the predicted PXRD profile of the AA stacking mode. On the contrary, the presence of no other observably large peaks at angles above ∼12° (corresponding to shorter d-spacings) points to the lack of a clear presence of a well-ordered AB stacking mode, whose predicted PXRD profile contains clearly visible (101) (∼14°), (201) (∼15.5°), and (211) (∼17°) reflections. The surface morphology of CN-COF studied by SEM revealed cauliflower-like aggregates (Fig. 2d), while HR-TEM analysis confirmed a layered structure (Fig. S17). A parallel STEM-EDX analysis (Fig. S18) points to a minimal amount of silicon and oxygen, indicating that contamination with silicates, eventually formed by a reaction of NaOH with the glass tube, is not significant.
1) and aq. NaOH (5 M)) afforded CN-COF as a yellow fluffy solid with high crystallinity, bright solid-state fluorescence, and in a reproducible way (∼90% yield). The model compound MC was obtained by reacting TFBB with phenylacetonitrile in the presence of KOH; see the SI for more details. The constitution and the connectivity of the novel CN-COF were confirmed by FT-IR, Raman, and solid-state 13C CP/MAS NMR spectroscopies. As compared to TFBB, the FT-IR spectrum of CN-COF (Fig. 2b) shows a significant decrease in the intensity of the carbonyl stretching band (∼1700 cm−1), indicating a high degree of polymerization between the building units. This is further supported by a shift of the nitrile stretching band from 2248 (BTAN) to 2211/2213 cm−1 (MC/CN-COF), which is attributed to the conjugation of the nitrile groups with the newly formed olefinic linkages. Due to the low intensity of these FT-IR signals, we have unambiguously confirmed the presence of cyano groups also by Raman spectroscopy (Fig. S11). A very similar peak pattern was observed in the 13C CP/MAS NMR spectra of CN-COF and MC, even when compared to 13C NMR spectrum of MC in the solution (Fig. S3). The crystal structure of CN-COF was investigated by the powder XRD analysis (PXRD). The experimentally obtained diffractogram was optimized by the Le Bail refinement (Fig. S14 and S15) and further compared with the patterns calculated for the AA and AB stacking modes (Fig. 2c). The comparison revealed a close agreement between the observed diffraction lines at low angles (< ∼12°), the (100), (110), (200), and (210), of both model data sets and the experiment. At higher angles, a very broad (002) diffraction line centered at approximately 24° was experimentally observed, which corresponds to the interlayer distance of hexagonal layers. The experimentally obtained diffractogram matches well with the predicted PXRD profile of the AA stacking mode. On the contrary, the presence of no other observably large peaks at angles above ∼12° (corresponding to shorter d-spacings) points to the lack of a clear presence of a well-ordered AB stacking mode, whose predicted PXRD profile contains clearly visible (101) (∼14°), (201) (∼15.5°), and (211) (∼17°) reflections. The surface morphology of CN-COF studied by SEM revealed cauliflower-like aggregates (Fig. 2d), while HR-TEM analysis confirmed a layered structure (Fig. S17). A parallel STEM-EDX analysis (Fig. S18) points to a minimal amount of silicon and oxygen, indicating that contamination with silicates, eventually formed by a reaction of NaOH with the glass tube, is not significant.
Pd@CN-COF synthesis and characterization
Having verified the CN-COF structure and employing a suspension impregnation method carried out with Pd(OAc)2/EtOH, Pd@CN-COF was prepared (Fig. 2a). The formed Pd NPs were trapped on the COF surface, while the process is accompanied by a visible color change from yellow to green. The stabilizing effect of the nitrile groups is presumed to prevent migration and agglomeration of the nanoparticles. This is ascribed to the out-of-plane twisting of the nitrile groups, as illustrated by computed structural models (Fig. S12), which makes them accessible for Pd coordination. In addition, the calculated electron density distribution (Fig. S13) reveals density concentrated over the nitrile groups, further supporting their role as coordination sites for Pd NPs. Provided no COF was present in the reaction mixture, only Pd black formation was observed. The Pd content in Pd@CN-COF was determined by ICP-MS to be around 5 wt%. When comparing the FT-IR spectra of Pd(OAc)2, CN-COF, and Pd@CN-COF (Fig. 2e), the latter does not contain the characteristic bands at 1597 and 1417 cm−1, confirming a successful reduction of Pd(OAc)2 to Pd NPs. The incorporation of Pd NPs into the COF structure was further confirmed by the characteristic diffraction lines of Pd(0) (cubic (fcc), Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m (225); COD: 9008478) in the PXRD pattern of Pd@CN-COF (Fig. 2f) without affecting the crystallinity of the COF support. The SEM images shown in Fig. 2g confirm a spot-deposition of Pd NPs on the COF surface. The average size of Pd NPs (∼3.7 nm) was estimated using the Scherrer equation based on the main (111) reflection (∼40°) in the PXRD pattern of Pd@CN-COF. This value is consistent with the SEM observations, which reveals the presence of metal nanoparticles with an average size of 5.7 ± 1.3 nm (Fig. S16).
m (225); COD: 9008478) in the PXRD pattern of Pd@CN-COF (Fig. 2f) without affecting the crystallinity of the COF support. The SEM images shown in Fig. 2g confirm a spot-deposition of Pd NPs on the COF surface. The average size of Pd NPs (∼3.7 nm) was estimated using the Scherrer equation based on the main (111) reflection (∼40°) in the PXRD pattern of Pd@CN-COF. This value is consistent with the SEM observations, which reveals the presence of metal nanoparticles with an average size of 5.7 ± 1.3 nm (Fig. S16).
The surface chemical composition of CN-COF and Pd@CN-COF, analyzed by XPS, indicates the presence of C, O, N, Na, and Pd elements (Fig. S19a); see the SI for further details. The C 1s high-resolution spectra (Fig. S19b) revealed the expected signals of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, C–(C,H), C–N, C–O, C
C, C–(C,H), C–N, C–O, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, COOH, and π–π* shake-up satellite. The N 1s spectra (Fig. S19c) in both cases point to one chemical species (C
O, COOH, and π–π* shake-up satellite. The N 1s spectra (Fig. S19c) in both cases point to one chemical species (C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N), while the signal of Pd@CN-COF is shifted by 0.2 eV towards lower binding energy as a result of the nitrile groups interacting with Pd atoms. The Pd 3d XPS spectrum of Pd@CN-COF (Fig. S19d) confirmed the presence of Pd(0) at 335.3/340.6 eV23 along with Pd(II) at 336.8/342.1 eV23a,24 and Pd(IV) at 338.4/343.7 eV,25 most likely related to the corresponding surface oxides and hydroxides.
N), while the signal of Pd@CN-COF is shifted by 0.2 eV towards lower binding energy as a result of the nitrile groups interacting with Pd atoms. The Pd 3d XPS spectrum of Pd@CN-COF (Fig. S19d) confirmed the presence of Pd(0) at 335.3/340.6 eV23 along with Pd(II) at 336.8/342.1 eV23a,24 and Pd(IV) at 338.4/343.7 eV,25 most likely related to the corresponding surface oxides and hydroxides.
The TGA profile of CN-COF, supported by the DSC analysis (Fig. S20), exhibits a mass loss below 150 °C, which can be attributed to the evaporation of residual adsorbed solvents. The material then remained thermally stable up to ∼400 °C (Ti = 373 °C), after which gradual decomposition occurred. The thermal profile of Pd@CN-COF (Fig. S21) shows initial evaporation of solvents followed by a continuous weight loss. The DSC indicates an accelerated decomposition above ∼400 °C.
The parent CN-COF and the Pd@CN-COF catalyst exhibit porosity at the border between micro- and mesopores with the specific surface areas of 258 and 358 m2 g−1 and the total pore volumes of 0.186 and 0.225 cm3 g−1. This observation contrasts with the common expectation that impregnation with metal nanoparticles leads to a decrease in the surface area. To verify the trend, repeated measurements of the pristine support and the Pd catalyst were performed, consistently confirming this anomalous behavior. Considering these findings, we suggest that the COF framework undergoes solvent-induced swelling during palladium surface-deposition, which may result in an increase in the accessible surface area. An analogous trend was reported by Platero-Prats et al.,26 who observed surface areas of 550 and 664 m2 g−1 for the COF and the in situ impregnated material, respectively. The palladium dispersion and the average size of Pd NPs in Pd@CN-COF were estimated using specific CO adsorption on Pd. The dispersion, according to the considered CO/Pd stoichiometry, reaches values of 18–36%, while the size of Pd NPs was 3.2–6.4 nm, in a very good agreement with the estimations from the PXRD and SEM image analysis; see the SI for further details.
Pd@CN-COF initiated transformations
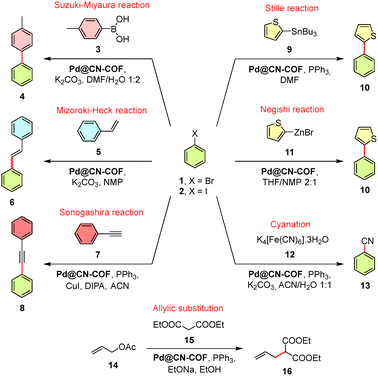
Encouraged by the recent reports on COF-based Pd catalysis27 and considering [Pd(PPh3)4] as a prototype Pd(0) precatalyst, we have further evaluated the catalytic performance of Pd@CN-COF in Pd-initiated cross-coupling reactions, cyanation, and allylic substitution (Table 1). The initial screening of solvents revealed highly polar solvents such as DMF, NMP, and ACN to be the most effective medium. Applying Pd@CN-COF (0.5 mol%) in a model Suzuki–Miyaura cross-coupling reaction between model bromo/iodobenzene 1/2 and tolylboronic acid 3 afforded 4-methylbiphenyl 4 nearly quantitatively at 25 °C and within 0.5 h. When compared with the performance of previously reported Pd(0)@COF catalysts in the Suzuki–Miyaura reaction (Table S6), Pd@CN-COF demonstrated an outstanding catalytic activity among them, as reflected by a rapid conversion at room temperature. A comparable performance was observed for Pd7/cyano-COF-1 (>99% within 1 h at 25 °C),22 suggesting that cyano-substituted COFs represent an effective support for catalytically active Pd(0), in contrast to imine-, enamine-, or hydrazone-based COFs. For the latter, elevated temperatures and longer reaction times are usually required and generally an uncomplete conversion is achieved. This can be attributed to a diffusion of reagents in/out of the COF's pores, whereas the surface-exposed catalytic sites of Pd@CN-COF are readily accessible for oxidative addition. Moreover, coordination of Pd NPS with the π-system and cyano groups of CN-COF renders the opposite face of Pd inaccessible, thereby enforcing the cis-orientation of the Pd(II) diaryl complex and facilitating reductive elimination. Notably, this also contrasts with [Pd(PPh3)4] precatalyst, which required a reaction time of 3 h and failed completely in converting the iodobenzene 2. The poor reactivity of aryl iodides in the Suzuki–Miyaura reactions employing the Pd/PPh3 catalytic systems at lower temperatures has previously been addressed by Bissember et al.28 The suggested poor turnover of the trans-intermediate resulting from the initial oxidative addition of 2 was confirmed through the Pd@CN-COF-initiated reaction carried out in the presence of PPh3, which resulted in a lower conversion (78%) and a significantly extended reaction time (24 h; Table S5). However, in other reactions conducted at elevated temperature, Sonogashira and cyanation in particular, a generally higher reactivity of 2 over 1 was observed. When comparing the reaction times and the achieved conversions across Table 1, both heterogeneous and homogeneous catalysis with Pd@CN-COF and [Pd(PPh3)4] are comparable. The lower reactivity of 1 in the Mizoroki–Heck and Stille reactions can be enhanced by increasing the catalysis (to 2 mol%) or by raising the reaction temperature. In contrast to the Suzuki–Miyaura reaction, the further screening also indicated an enhanced catalytic performance of Pd@CN-COF in the presence of an additional P-ligand (PPh3) in certain types of reactions. While the Mizoroki–Heck (Table S7) and Negishi (Table S10, X = I) reactions provided comparable results with or without PPh3, the presence of the P-ligand proved to be crucial in the Stille reaction (Table S9, X = Br) and the cyanation (Table S11). In the Sonogashira (Table S8) and Tsuji–Trost (Table S12) reactions, the additional PPh3 enabled full conversions within the same reaction time as those observed with [Pd(PPh3)4]. In the Negishi reaction of 1 (Table S10), a slight decrease in conversion was observed in the presence of PPh3. Regarding byproducts, the Pd@CN-COF-initiated cross-coupling reactions showed minor homocoupling (<5%), none dehalogenation, prevailingly (Z)-6 product in the Mizoroki–Heck reaction, and eventually two-fold allylation affording 17 (<14%, Table S12). These features are analogous to [Pd(PPh3)4].| Reaction | X | Catalysis (mol%) | T (°C) | PPh3 | Pd@CN-COF | [Pd(PPh3)4] | ||
|---|---|---|---|---|---|---|---|---|
| t (h) | Conversion (%) | t (h) | Conversion (%) | |||||
| a All reactions were carried out on a 0.2 mmol scale under an argon atmosphere. The conversions were determined by GC/MS analysis. | ||||||||
| Suzuki–Miyaura | Br | 0.5 | 25 | ✗ | 0.5 | >99 | 3 | 95 |
| I | 0.5 | 25 | ✗ | 0.5 | 97 | 24 | 2 | |
| Mizoroki–Heck | Br | 2 | 120 | ✗ | 24 | 78 | 3 | 95 |
| I | 0.5 | 120 | ✗ | 4 | 96 | 3 | 96 | |
| Sonogashira | Br | 0.5 | 80 | ✓ | 24 | 0 | 24 | 0 |
| I | 0.5 | 80 | ✓ | 2.5 | 97 | 2.5 | 95 | |
| Stille | Br | 2 | 80 | ✓ | 24 | 86 | 2 | 95 |
| I | 0.5 | 80 | ✓ | 4 | 95 | 4 | 95 | |
| Negishi | Br | 0.5 | 65 | ✗ | 24 | 60 | 24 | 61 |
| I | 0.5 | 65 | ✗ | 24 | 87 | 24 | 90 | |
| Cyanation | Br | 0.5 | 80 | ✓ | 24 | 0 | 24 | 0 |
| I | 0.5 | 80 | ✓ | 10 | >99 | 6 | >99 | |
| Allylic substitution | — | 0.5 | 50 | ✓ | 1 | 72 | 0.5 | 72 |
The reusability of Pd@CN-COF was examined in the Suzuki–Miyaura reaction, which revealed stable conversion over three cycles (99 to 94%) and a subsequent decrease in the catalytic activity to 61%. The Pd@CN-COF productivity reaches cumulative TON values up to ca. 890 with the cycle-TOF up to 396 h−1, which can be compared with TON 190 and TOF 63.3 h−1 obtained for the homogeneous [Pd(PPh3)4] (Table S15 and Fig. S43, S44). Based on the performed analysis of the used catalyst (Fig. S26–S32), this is attributed to the Pd surface site deactivation caused by adsorbed organic residues, particularly DMF, as evidenced by the infrared spectrum of the recycled Pd@CN-COF (Fig. S26). This is further supported by XPS analysis of the used catalyst, which revealed an increased signal of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and a marked reduction of Pd compared to the fresh catalyst (Fig. S30c and d). A similar ability of DMF to reduce Pd(II) towards Pd NPs has been reported by Obora et al.29 Characterization of the used catalyst by adsorption of dinitrogen and CO revealed a decrease in the specific surface area (from 358 to 204 m2 g−1) and the total pore volume (from 0.225 to 0.12 cm3 g−1), although the pore structure remained unchanged. The CO adsorption data indicates a reduced amount of CO interacting with Pd atoms (from 1.884 to 0.234 cm3 g−1), which would suggest an increase in nanoparticle size to 11–21 nm. Despite this, both PXRD and SEM analyses (Fig. S27 and S29) confirmed that the Pd NPs retained their original size, suggesting that no significant sintering has occurred. The apparent increase in nanoparticle size derived from the CO adsorption is caused by a partial blockage of the Pd surface sites by adsorbed DMF. The ICP-MS analysis reveals 4.14 wt% of Pd in the four-times recycled catalyst, indicating a minor leaching of the palladium from the initial 4.91 wt%. This finding is consistent with the Pd concentration detected in the filtrate after the first catalytic cycle, corresponding to a loss of 0.14 wt%. In the Suzuki–Miyaura reaction, repeatedly adding the reactants to the reaction mixture, while maintaining a steady amount of the catalyst (0.5 mol%), resulted in an initial quantitative conversion over 45 minutes, after which the subsequent conversion stabilized between 89–85%. This feature makes Pd@CN-COF a promising catalytic system for the use under continuous-flow conditions.
O and a marked reduction of Pd compared to the fresh catalyst (Fig. S30c and d). A similar ability of DMF to reduce Pd(II) towards Pd NPs has been reported by Obora et al.29 Characterization of the used catalyst by adsorption of dinitrogen and CO revealed a decrease in the specific surface area (from 358 to 204 m2 g−1) and the total pore volume (from 0.225 to 0.12 cm3 g−1), although the pore structure remained unchanged. The CO adsorption data indicates a reduced amount of CO interacting with Pd atoms (from 1.884 to 0.234 cm3 g−1), which would suggest an increase in nanoparticle size to 11–21 nm. Despite this, both PXRD and SEM analyses (Fig. S27 and S29) confirmed that the Pd NPs retained their original size, suggesting that no significant sintering has occurred. The apparent increase in nanoparticle size derived from the CO adsorption is caused by a partial blockage of the Pd surface sites by adsorbed DMF. The ICP-MS analysis reveals 4.14 wt% of Pd in the four-times recycled catalyst, indicating a minor leaching of the palladium from the initial 4.91 wt%. This finding is consistent with the Pd concentration detected in the filtrate after the first catalytic cycle, corresponding to a loss of 0.14 wt%. In the Suzuki–Miyaura reaction, repeatedly adding the reactants to the reaction mixture, while maintaining a steady amount of the catalyst (0.5 mol%), resulted in an initial quantitative conversion over 45 minutes, after which the subsequent conversion stabilized between 89–85%. This feature makes Pd@CN-COF a promising catalytic system for the use under continuous-flow conditions.
Practical applications of Pd@CN-COF
Having verified the catalytic behavior of our catalyst, we further address the application of Pd@CN-COF in the construction a key C–C bond of agrochemically, industrially, and pharmacologically relevant products 20, 23, 25, 27, and 32 (Fig. 3). Intermediate 20 is easily accessible via the Suzuki–Miyaura reaction of 18 and 19, representing the first step of the industrial synthesis of boscalid,30 a broad-spectrum carboxamide fungicide. The biphenyl 20 can be prepared nearly quantitatively and within only 2 h (Fig. 3a). Starting from iodides 21/24, propargyl alcohol 22 or ethynylbenzene 7, the catalytic performance of Pd@CN-COF in the Sonogashira cross-coupling reactions towards 23 and 25 is further demonstrated (Fig. 3b and c). Whereas 23 represents a synthetic precursor of justicidone, a dioxo-lignan derivative investigated for its potential biological activity,31 the product 25 is a part of ponatinib, a tyrosine kinase inhibitor used to treat some variety of leukemia.32 | ||
| Fig. 3 Application of Pd@CN-COF across selected valuable transformations including Suzuki–Miyaura (a), Sonogashira (b/c), Stille (d) and Heck/Suzuki–Miyauara (e) reactions. | ||
Both products can be obtained in very good yields of 90 and 91% by using Pd@CN-COF catalyst. Owing to their exceptional electronic and optical properties, thiophene-based π-conjugated systems with alternating D–A–D structure (D/A ∼ electron donor/acceptor) are attractive organic semiconductors.33 4,7-Disubstituted benzo[c][1,2,5]thiadiazole (BTDA) is a prototypical example, which can be obtained from 26 via a two-fold Stille cross-coupling reaction. The product 27 was isolated smoothly with a 89% yield (Fig. 3d). Encouraged by these positive results, we carried out a one-pot, two-step synthesis of the heterocyclic product 32, a core of a selective molecular sensor of Cu(II) and Hg(II) ions.34 The Mizoroki–Heck reaction adapted to the starting 1-methylvinazene 28, preventing hydrolysis of the cyano groups by replacing K2CO3 with DIPEA,35 afforded intermediate 30. Instead of its isolation, it was subsequently reacted with 2-iodothiophene 31 to form 32, which was isolated with an unoptimized overall yield of 33% (Fig. 3e).
Conclusions
In summary, we have designed and prepared a new cyano-substituted olefin-linked COF and subsequently used it as a support for the in situ generated Pd nanoparticles. In contrast to the thermally and chemically labile [Pd(PPh3)4], the heterogeneous Pd@CN-COF is easy to prepare, proved to be bench stable, and provides comparable or better catalytic performance and thus may compete with the commercially available homogeneous Pd precatalysts.Author contributions
Marcela Horáková: investigation, methodology, formal analysis, writing – original draft. Milan Klikar: investigation, methodology, supervision. Jakub Halamek: investigation. Roman Bulánek: investigation, writing – original draft. Jhonatan Rodriguez-Pereira: investigation. Veronika Čičmancová: investigation. Jan Podlesný: investigation. Eva Kolíbalová: investigation. Jan Michalička: investigation. Jan Bartáček: investigation. Patrik Pařík: investigation. Jan M. Macak: investigation. Filip Bureš: writing – review & editing, supervision, project administration, funding acquisition, conceptualization.Conflicts of interest
There are no conflicts to declare.Data availability
The data underlying this study are openly available in Figshare at https://doi.org/10.6084/m9.figshare.30691823.Supplementary information (SI): syntheses and characterizations, NMR/MS/IR spectra, simulations, PXRD, microscopy images, XPS, thermal properties, volumetry, and catalysis (PDF), simulated AA stacking of CN-COF (CIF), simulated AB stacking of CN-COF (CIF). See DOI: https://doi.org/10.1039/d5qm00889a.
Acknowledgements
This work has been funded by a grant from the Programme Johannes Amos Comenius under the Ministry of Education, Youth and Sports of the Czech Republic [no. CZ.02.01.01/00/23_021/0008593]. The financial support from the Ministry of Education, Youth and Sports of the Czech Republic for the CEMNAT and CzechNanoLab large research infrastructures (LM2023037, LM2023051) are gratefully acknowledged for XPS, SEM and HRTEM accesses, respectively. The authors also acknowledge Dr. Matthew A. Addicoat (Nottingham Trent University) for his guidance and support in performing structural simulations.References
- G. S. Day, H. F. Drake, H.-C. Zhou and M. R. Ryder, Evolution of porous materials from ancient remedies to modern frameworks, Commun. Chem., 2021, 4, 114 CrossRef CAS PubMed; A. Thomas, Much ado about nothing – a decade of porous materials research, Nat. Commun., 2020, 11, 4985 CrossRef PubMed.
- P. J. Waller, F. Gándara and O. M. Yaghi, Chemistry of Covalent Organic Frameworks, Acc. Chem. Res., 2015, 48, 3053–3063 CrossRef CAS PubMed.
- A. P. Côté, A. I. Benin, N. W. Ockwig, M. O’Keeffe, A. J. Matzger and O. M. Yaghi, Porous, Crystalline, Covalent Organic Frameworks, Science, 2005, 310, 1166–1170 CrossRef PubMed.
- K. Geng, T. He, R. Liu, S. Dalapati, K. T. Tan, Z. Li, S. Tao, Y. Gong, Q. Jiang and D. Jiang, Covalent Organic Frameworks: Design, Synthesis, and Functions, Chem. Rev., 2020, 120, 8814–8933 CrossRef CAS PubMed; M. S. Lohse and T. Bein, Covalent Organic Frameworks: Structures, Synthesis, and Applications, Adv. Funct. Mater., 2018, 28, 1705553 CrossRef.
- R. Liu, K. T. Tan, Y. Gong, Y. Chen, Z. Li, S. Xie, T. He, Z. Lu, H. Yang and D. Jiang, Covalent organic frameworks: an ideal platform for designing ordered materials and advanced applications, Chem. Soc. Rev., 2021, 50, 120–242 RSC.
- Y. Yin, Y. Zhang, X. Zhou, B. Gui, W. Wang, W. Jiang, Y.-B. Zhang, J. Sun and C. Wang, Ultrahigh–surface area covalent organic frameworks for methane adsorption, Science, 2024, 386, 693–696 CrossRef CAS PubMed; H. Furukawa and O. M. Yaghi, Storage of Hydrogen, Methane, and Carbon Dioxide in Highly Porous Covalent Organic Frameworks for Clean Energy Applications, J. Am. Chem. Soc., 2009, 131, 8875–8883 CrossRef PubMed.
- S. Kandambeth, V. S. Kale, O. Shekhah, H. N. Alshareef and M. Eddaoudi, 2D Covalent-Organic Framework Electrodes for Supercapacitors and Rechargeable Metal-Ion Batteries, Adv. Energy Mater., 2021, 12, 2100177 CrossRef.
- N. Keller and T. Bein, Optoelectronic processes in covalent organic frameworks, Chem. Soc. Rev., 2021, 50, 1813–1845 RSC.
- Z. Meng and K. A. Mirica, Covalent organic frameworks as multifunctional materials for chemical detection, Chem. Soc. Rev., 2021, 50, 13498–13558 RSC.
- Z. Alsudairy, N. Brown, A. Campbell, A. Ambus, B. Brown, K. Smith-Petty and X. Li, Covalent organic frameworks in heterogeneous catalysis: recent advances and future perspective, Mater. Chem. Front., 2023, 7, 3298–3331 RSC.
- A. Basak, S. Karak and R. Banerjee, Covalent Organic Frameworks as Porous Pigments for Photocatalytic Metal-Free C–H Borylation, J. Am. Chem. Soc., 2023, 145, 7592–7599 CrossRef CAS PubMed; F. Meng, S. Bi, Z. Sun, D. Wu and F. Zhang, 2,4,6-Trimethylpyridine-Derived Vinylene-Linked Covalent Organic Frameworks for Confined Catalytic Esterification, Angew. Chem., Int. Ed., 2022, 61, e202210447 CrossRef PubMed; B. Hou, S. Yang, K. Yang, X. Han, X. Tang, Y. Liu, J. Jiang and Y. Cui, Confinement-Driven Enantioselectivity in 3D Porous Chiral Covalent Organic Frameworks, Angew. Chem., Int. Ed., 2021, 60, 6086–6093 CrossRef PubMed.
- J.-M. Seo, H.-J. Noh, J.-P. Jeon, H. Kim, G.-F. Han, S. K. Kwak, H. Y. Jeong, L. Wang, F. Li and J.-B. Baek, Conductive and Ultrastable Covalent Organic Framework/Carbon Hybrid as an Ideal Electrocatalytic Platform, J. Am. Chem. Soc., 2022, 144, 19973–19980 CrossRef CAS PubMed.
- Y. Zheng, S. Zhang, J. Guo, R. Shi, J. Yu, K. Li, N. Li, Z. Zhang and Y. Chen, Green and Scalable Fabrication of High-Performance Biocatalysts Using Covalent Organic Frameworks as Enzyme Carriers, Angew. Chem., Int. Ed., 2022, 61, e202208744 CrossRef CAS PubMed; H. Lyu, C. S. Diercks, C. Zhu and O. M. Yaghi, Porous Crystalline Olefin-Linked Covalent Organic Frameworks, J. Am. Chem. Soc., 2019, 141, 6848–6852 CrossRef PubMed.
- Z. Fu, X. Wang, A. M. Gardner, X. Wang, S. Y. Chong, G. Neri, A. J. Cowan, L. Liu, X. Li, A. Vogel, R. Clowes, M. Bilton, L. Chen, R. S. Sprick and A. I. Cooper, A stable covalent organic framework for photocatalytic carbon dioxide reduction, Chem. Sci., 2020, 11, 543–550 RSC.
- C. Chandran, H. D. Singh, L. S. Leo, P. Shekhar, D. Rase, D. Chakraborty, C. P. Vinod and R. Vaidhyanathan, A covalent organic framework with electrodeposited copper nanoparticles – a desirable catalyst for the Ullmann coupling reaction, J. Mater. Chem. A, 2022, 10, 15647–15656 RSC; P. Pachfule, S. Kandambeth, D. D. Díaz and R. Banerjee, Highly stable covalent organic framework–Au nanoparticles hybrids for enhanced activity for nitrophenol reduction, Chem. Commun., 2014, 50, 3169–3172 RSC; P. Pachfule, M. K. Panda, S. Kandambeth, S. M. Shivaprasad, D. D. Díaz and R. Banerjee, Multifunctional and robust covalent organic framework-nanoparticle hybrids, J. Mater. Chem. A, 2014, 2, 7944–7952 Search PubMed.
- S.-Y. Ding, J. Gao, Q. Wang, Y. Zhang, W.-G. Song, C.-Y. Su and W. Wang, Construction of Covalent Organic Framework for Catalysis: Pd/COF-LZU1 in Suzuki–Miyaura Coupling Reaction, J. Am. Chem. Soc., 2011, 133, 19816–19822 Search PubMed.
- B. Mishra, A. Alam, A. Chakraborty, B. Kumbhakar, S. Ghosh, P. Pachfule and A. Thomas, Covalent Organic Frameworks for Photocatalysis, Adv. Mater., 2024, 2413118 Search PubMed.
- J.-D. Feng, W.-D. Zhang and Z.-G. Gu, Covalent Organic Frameworks for Electrocatalysis: Design, Applications, and Perspectives, ChemPlusChem, 2024, 89, e202400069 CrossRef CAS PubMed.
- T. S. Rodrigues, A. G. M. da Silva and P. H. C. Camargo, Nanocatalysis by noble metal nanoparticles: controlled synthesis for the optimization and understanding of activities, J. Mater. Chem. A, 2019, 7, 5857–5874 RSC.
- L. Liu and A. Corma, Metal Catalysts for Heterogeneous Catalysis: From Single Atoms to Nanoclusters and Nanoparticles, Chem. Rev., 2018, 118, 4981–5079 CrossRef CAS PubMed.
- J. Rayadurgam, S. Sana, M. Sasikumar and Q. Gu, Palladium catalyzed C–C and C–N bond forming reactions: an update on the synthesis of pharmaceuticals from 2015–2020, Org. Chem. Front., 2021, 8, 384–414 RSC; P. Devendar, R.-Y. Qu, W.-M. Kang, B. He and G.-F. Yang, Palladium-Catalyzed Cross-Coupling Reactions: A Powerful Tool for the Synthesis of Agrochemicals, J. Agric. Food Chem., 2018, 66, 8914–8934 CrossRef CAS PubMed.
- R. Tao, B. Feng, C. Wang, H. Zhang, R. Chen, K. Liu, J. Li, X. Deng, J. Li and Y. Yang, Feasible preparation of preferentially oriented (111) Pd nanocrystals supported on cyano-COFs for ultrahigh activity in C–C cross-couplings, J. Catal., 2024, 432, 115399 CrossRef CAS.
- (a) B. Bawab, S. M. Thalluri, J. Rodriguez-Pereira, H. Sopha, R. Zazpe and J. M. Macak, Anodic TiO2 nanotube layers decorated by Pd nanoparticles using ALD: an efficient electrocatalyst for methanol oxidation, Electrochim. Acta, 2022, 429, 141044 CrossRef CAS; (b) C. M. Schott, J. Holl, R. Zazpe, M. Kopp, O. Man, S. M. Thalluri, J. Rodriguez-Pereira, P. M. Schneider, K.-T. Song, E. Keles, P. Peljo, J. J. Jasielec, E. L. Gubanova, J. M. Macak and A. S. Bandarenka, Revealing Catalytic Properties of Palladium/Gold Systems toward Hydrogen Evolution, Oxidation, and Absorption with Scanning Electrochemical Microscopy, ACS Catal., 2025, 15, 9035–9046 CrossRef CAS PubMed.
- T. L. Barr, Recent advances in X-ray photoelectron spectroscopy studies of oxides, J. Vac. Sci. Technol., A, 1991, 9, 1793–1805 CrossRef CAS.
- B. Bawab, S. M. Thalluri, E. Kolíbalová, R. Zazpe, L. Jelínek, J. Rodriguez-Pereira and J. M. Macak, Synergistic effect of Pd single atoms and nanoparticles deposited on carbon supports by ALD boosts alkaline hydrogen evolution reaction, Chem. Eng. J., 2024, 482, 148959 CrossRef CAS.
- I. Romero-Muñiz, A. Mavrandonakis, P. Albacete, A. Vega, V. Briois, F. Zamora and A. E. Platero-Prats, Unveiling the Local Structure of Palladium Loaded into Imine-Linked Layered Covalent Organic Frameworks for Cross-Coupling Catalysis, Angew. Chem., Int. Ed., 2020, 59, 13013–13020 CrossRef.
- H. Salemi, M. Debruyne, V. Van Speybroeck, P. Van Der Voort, M. D’hooghe and C. V. Stevens, Covalent organic framework supported palladium catalysts, J. Mater. Chem. A, 2022, 10, 20707–20729 RSC.
- C. C. Ho, A. Olding, J. A. Smith and A. C. Bissember, Nuances in Fundamental Suzuki–Miyaura Cross-Couplings Employing [Pd(PPh3)4]: Poor Reactivity of Aryl Iodides at Lower Temperatures, Organometallics, 2018, 37, 1745–1750 CrossRef CAS.
- J. Ishida, M. Nakatsuji, T. Nagata, H. Kawasaki, T. Suzuki and Y. Obora, Synthesis and Characterization of N,N-Dimethylformamide-Protected Palladium Nanoparticles and Their Use in the Suzuki–Miyaura Cross-Coupling Reaction, ACS Omega, 2020, 5, 9598–9604 CrossRef CAS.
- I. Volovych, M. Neumann, M. Schmidt, G. Buchner, J.-Y. Yang, J. Wölk, T. Sottmann, R. Strey, R. Schomäcker and M. Schwarze, A novel process concept for the three step Boscalid® synthesis, RSC Adv., 2016, 6, 58279–58287 RSC.
- C. J. Boluda, H. López, J. A. Pérez and J. M. Trujillo, First Total Synthesis of Justicidone, a p-Quinone-Lignan Derivative from Justicia hyssopifolia, Chem. Pharm. Bull., 2005, 53, 930–933 CrossRef CAS PubMed.
- L. Han, Y. Yu, P. Deng, S. Wang, J. Hu, S. Wang, J. Zheng, J. Jiang, Y. Dang, R. Long and Z. Gan, Design, synthesis, and biological evaluation of Ponatinib-based N-Phenylpyrimidine-2-amine derivatives as novel fibroblast growth factor receptor 4 (FGFR4) selective inhibitors, Eur. J. Med. Chem., 2025, 284, 117206 CrossRef CAS.
- F. Zhang, D. Wu, Y. Xu and X. Feng, Thiophene-based conjugated oligomers for organic solar cells, J. Mater. Chem., 2011, 21, 17590–17600 RSC.
- N. Dey, J. Kulhánek, F. Bureš and S. Bhattacharya, Simultaneous Detection of Cu2+ and Hg2+ via Two Mutually Independent Sensing Pathways of Biimidazole Push–Pull Dye, J. Org. Chem., 2019, 84, 1787–1796 CrossRef CAS PubMed.
- J. Kulhánek, F. Bureš, O. Pytela, T. Mikysek and J. Ludvík, Imidazole as a Donor/Acceptor Unit in Charge-Transfer Chromophores with Extended π-Linkers, Chem. – Asian J., 2011, 6, 1604–1612 CrossRef.
| This journal is © the Partner Organisations 2026 |