Open Access Article
Open Access ArticleRedox-responsive Fe(II)/Fe(III) MRI probes with pentadentate or hexadentate macrocyclic ligands
Deepak Krishnan Balaji a,
Joseph A. Spernyak
a,
Joseph A. Spernyak b,
Matthew R. Crawley
b,
Matthew R. Crawley a and
Janet R. Morrow
a and
Janet R. Morrow *a
*a
aDepartment of Chemistry, University at Buffalo, The State University of New York, Amherst, New York 14260, USA. E-mail: Jmorrow@buffalo.edu
bDepartment of Cell Stress Biology, Roswell Park Comprehensive Cancer Center, Buffalo, New York 14263, USA
First published on 5th May 2026
Abstract
Fe(II) complexes of TACN (1,4,7-triazacyclononane) containing two 6-methyl-2-picolyl pendant groups are studied as redox-responsive agents for monitoring peroxide produced in inflammation. One Fe(II) complex has a pentadentate ligand with a sixth coordination site for binding water as confirmed by X-ray crystallography ([FeII(MPB)(CF3SO3)](CF3SO3)) and by 17O NMR studies in solution. The other complex is formed with a hexadentate ligand (FeII(MPH)) and also has an inner-sphere water ligand in solution as shown by variable temperature 17O NMR studies. FeII(MPB) is inert towards oxidation under ambient levels of O2 in aqueous solution at pH 7.4 over 24 hours whereas FeII(MPH) is resistant to oxidation over 4 hours. FeII(MPB) oxidizes rapidly (≤3 min) upon addition of one equivalent of peroxide or in the presence of 0.1 U mL−1 glucose oxidase (GOX). With one equivalent of peroxide in buffered solutions, the major product is FeIII(MPB), most likely as a buffer or hydroxide complex, but with a 10-fold excess of peroxide, a Fe(III) phenolate complex is produced as supported by electronic absorbance spectroscopy and analysis of products by high resolution mass spectrometry. The r1 relaxivity for the FeIII(MPB) (r1 = 0.71 mM−1 s−1) is increased by 14-fold over the FeII complex at 1.4 T, 34 °C and pH 7.4. Oxidation of FeII(MPH) by addition of one equivalent of peroxide or by GOX is complete within a few minutes and produces a 7-fold increase in relaxivity (r1 = 0.58 mM−1s−1). Changes in relaxivity are reversed by addition of ascorbate. FeII(MPB) is one of the first reported examples of a Fe(II) macrocyclic MRI probe that is unreactive to O2 but reacts rapidly with micromolar concentrations of peroxide for increased selectivity in monitoring inflammation.
Introduction
Contrast agents are essential tools in modern magnetic resonance imaging (MRI) diagnostic protocols.1 Clinically used gadolinium-based MRI contrast agents are administered intravenously and extravasate into tissue in a non-specific manner to produce a dynamically monitored signal for the visualization of pathophysiological states and anatomic details for tumor detection, angiography, blood–brain barrier evaluation and organ perfusion.2 In molecular MRI, contrast agents or probes provide further information towards diagnosis in the presence of biochemical markers or specific physiological environments.3 Such probes may either be attached to a targeting agent or contain a responsive element that leads to activation of the probe under aberrant physiological conditions.3 Such responsive probes would facilitate mapping tissue changes that are a result of molecular processes characteristic of disease states and would add to the information normally gained from a contrast agent. Responsive or activatable MRI probes have been reported for wide-ranging applications such as enzyme reactions,4,5 pH changes,3,4 redox status6–8 or metal ion concentrations.9,10One sought-after type of activatable MRI probe responds to the oxidizing conditions found in inflammation7,8,11 as postulated to be important in many disease states.12–15 For example, the capability to image reactive oxygen species (ROS) may enable discrimination of inflammation versus tumor growth during cancer treatment as a biochemical signature.14,16,17 Of the ROS, peroxide is the most likely biochemical marker to react with an MRI probe as it is present in micromolar concentrations in the extracellular environment18 and is generated by myeloid immune cells that are recruited to the site of inflammation.19 Other ROS such as hydroxyl or superoxide radicals are likely to be too reactive for registration by probes under physiological conditions.20
Probes based on Gd(III) T1 agents were among the earliest examples of redox-responsive probes. These Gd(III) complexes contained ligands that react under oxidizing conditions or with thiol-reactive ligands to give increased signal.21,22 More recently, redox-activated MRI probes containing Gd(III) complexes tagged with groups to produce signal by 19F NMR/MRI have been reported.23 An alternative approach is to use a redox-active metal that switches oxidation states. These are generally transition metal probes6,8,9,24 or certain lanthanides such as Eu(II)/Eu(III) complexes.25–27 The tunability of the redox potential of the metal center through coordination chemistry makes them ideal for development as responsive MRI probes. The effect of the probe on water proton resonances6–8 or on 19F NMR/MRI signal of fluorine-containing tags can be monitored.28,29 Switchable paramagnetic chemical exchange saturation transfer (paraCEST) probes have also been reported.30,31
Of the early transition metal ions, iron probes are of interest to capitalize on the abundance of iron in the body and the tunable redox range of the Fe(II)/Fe(III) couple in coordination complexes.8,32 There are two common spin states for each member of the Fe(II)/Fe(III) redox pair. High-spin Fe(II) is a shift agent for frequency-encoded methods such as paramagnetic shift or paraCEST agents and is not an effective relaxation agent,8,33,34 whereas low spin Fe(II) is diamagnetic and thus MRI silent. High-spin Fe(III) is a T1 agent32,35,36 whereas low spin Fe(III) is a paramagnetic shift agent.37 The most commonly-studied redox pair for MRI applications is Fe(II) (high-spin or low spin) to high-spin Fe(III) complexes.6,8 For this responsive pair, the divalent Fe(II) is the form of probe that is injected and, upon oxidation, gives rise to increased signal in T1 weighted imaging.38 One of the major challenges in this endeavor is to obtain complexes that oxidize in the presence of peroxide and not O2, given that O2 is present in blood and organs. Thus, it is desirable that the redox potential for the Fe(II)/Fe(III) couple is positive versus NHE (>200 mV) to favor the Fe(II) state under normal physiological conditions, but to produce Fe(III) under oxidizing conditions of inflammation. Of the handful of reported redox-activated iron agents, macrocyclic complexes are activated only at high concentrations of peroxide (10–100 mM) that are not biologically relevant39–41 and recent examples of closed coordination complexes with multiple metal centers also require excess millimolar concentrations of peroxide.42,43 In contrast, iron probes containing polyaminocarboxylate pendants and an available coordination site react readily with enzymatically generated peroxide.38,44 These linear polyaminocarboxylate-based probes have been studied in vivo to map inflammatory processes.44–47 However, the Fe(II) form of the probes oxidizes slowly in the presence of O2 over several hours.
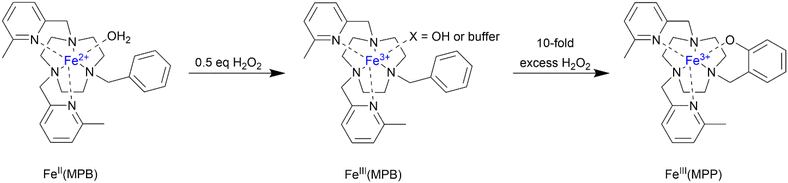
Here we report our efforts to prepare Fe(II)/Fe(III) redox activated probes that do not react readily with O2 in aerated solutions but react rapidly with micromolar amounts of peroxide. The key probe is an Fe(II) complex with a high redox potential (680 mV versus NHE) and an available coordination site for reacting with peroxide in the first step of oxidation. The iron complexes studied here (Scheme 1) are inspired by peroxide oxidation studies of Fe(II) complexes containing five nitrogen donor atoms, based mostly on linear48,49 and some macrocyclic chelates.50
 | ||
| Scheme 1 Iron complexes studied as redox probes with general labels designating all species in solution. X = OH2, OH, buffer, or peroxide. | ||
Results and discussion
Synthesis and characterization of ligands and Fe(II) complexes
Two complexes were prepared with the goal of examining the effect of macrocyclic ligands with different denticity on the oxidation and reduction of an activatable MRI probe. The MPB ligand is pentadentate and forms a Fe(II) complex with an available coordination site for a sixth ligand. The MPH ligand is hexadentate and preserves two pendant 6-methyl-2-picolyl(methylpyridine) groups but has a third pendant with an oxygen donor to tune the redox potential to more negative values compared to Fe(II) complexes with all nitrogen donors.51 The methyl group on the pyridine pendant serves to maintain a high-spin state for Fe(II)51 and Fe(III). Analogous iron complexes of TACN with 2-picolyl pendants that lack the methyl group adjacent to the nitrogen donor give low or intermediate spin-states for Fe(II) or Fe(III) complexes.52–54 Our goal is to produce increased signal from a low relaxivity probe containing high-spin Fe(II) to a higher relaxivity probe containing high-spin Fe(III).38The MPB ligand was prepared from benzyl TACN by addition of 6-methyl-2-picolyl pendants by reductive amination methods.51,55 To prepare MPH, the benzyl group of MPB was removed by catalytic hydrogenation followed by alkylation with S-propylene oxide. The Fe(II) complexes were synthesized upon addition of Fe(CF3SO3)2 and isolated as the triflate salts. Both complexes are water soluble up to 5 mM at neutral pH and had effective magnetic moments (Fig. S2 and S3) of 5.3 and 5.1, respectively that are consistent with high-spin Fe(II) complexes.
X-ray crystallography studies showed a hexacoordinate [FeII(MPB)(CF3SO3)](CF3SO3) complex with three bound nitrogens of the TACN macrocycle, two 6-methyl-2-picolyl nitrogens and the sixth coordination site occupied by triflate counterion (Fig. 1). The [FeII(MPB)(CF3SO3)](CF3SO3) complex crystallizes in the triclinic crystal system and the centrosymmetric space group P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) . The twist angle (θ) of the complex was determined as the dihedral angle formed between the centroids of two mean planes and two atoms, each selected from one of the planes. One plane was defined by the two coordinated pyridyl nitrogen atoms and the bound oxygen of the triflate, while the other was defined by the three coordinated nitrogen atoms of the macrocycle.
. The twist angle (θ) of the complex was determined as the dihedral angle formed between the centroids of two mean planes and two atoms, each selected from one of the planes. One plane was defined by the two coordinated pyridyl nitrogen atoms and the bound oxygen of the triflate, while the other was defined by the three coordinated nitrogen atoms of the macrocycle.
 | ||
| Fig. 1 Molecular structure of [FeII(MPB)(CF3SO3)]+ cation – Δ(λλλ) as ORTEP diagram (hydrogen, counter anion and solvent molecule omitted for clarity). | ||
Based on the twist angle (35.7°) and direction (clockwise) of rotation of mean planes, the overall chirality of the complex is determined as Δ and based on the helicity of the macrocyclic backbone the chirality is determined as λλλ. The asymmetric unit of [FeII(MPB)(CF3SO3)]+ contains one full isomer Δ(λλλ) of this complex (Fig. 1). Given that the space group is centrosymmetric, the crystal structure also contains the inversion isomer Λ(δδδ) (Fig. S1). Additionally, the twist angle is consistent with a distorted octahedral geometry of the complex.
The 1H NMR spectrum (Fig. 2B) of FeII(MPB) shows 26 distinct proton resonances, consistent with a single diastereomeric form in solution. The chemical shifts of the proton resonances range from 193 ppm to −67 ppm.
The narrow and intense resonance at −39 ppm is assigned as one of the methyl resonances of a pyridine pendant based on its integration and T2 value (1.2 ms). The second methyl resonance is likely at 29 ppm as it has a similar integration value and T2 value (2.3 ms). The 1H NMR spectrum (Fig. 2A) of FeII(MPH) shows 26 distinct proton resonances, consistent with a single diastereomeric form in solution. The proton chemical shifts span a slightly larger range from 230 ppm to −60 ppm. The methyl groups are assigned by their integration and T2 values (0.8 ms) and have chemical shifts (37 ppm and −38 ppm) similar to those of FeII(MPB).
The Fe(II) complexes exhibit electronic absorbance bands at 360 nm that are assigned to MLCT bands which are lacking in the spectrum of the ligand alone (Fig. S6). Monitoring these bands upon incubation shows that both complexes are inert over several hours in the presence of 25 mM carbonate and 0.5 mM phosphate at pH 7.4 and 37 °C (Fig. S7 & S9). The complexes remained intact upon incubation with an equivalent of Zn(II) over several hours (Fig. S8 & S10). In addition, the oxidation of the two complexes was monitored by UV-vis spectroscopy at pH 7.4 in buffered aerated solutions. FeII(MPB) showed a negligible change in absorbance at 320 nm over 24 hours in the presence of ambient concentrations of oxygen in buffered solutions at neutral pH (Fig. 3). In contrast, FeII(MPH) showed a gradual change in absorbance that was attributed to oxidation with up to 20% oxidation by 24 hours.
 | ||
| Fig. 3 Inertness of Fe(II) complexes to oxidation by oxygen monitored by UV-Vis absorbance at 320 nm. Conditions: 0.20 mM complex, 20 mM HEPES pH 7.4. | ||
Solution speciation of the Fe(II) complexes was studied by monitoring electronic absorbance spectra as a function of pH. A pH titration of FeII(MPB) showed the disappearance of the peak at 360 nm and an increase in a new absorbance peak at 425 nm as the pH was increased from 4.5 to 11. A plot of the peak intensity at 360 nm as a function of pH (Fig. 4B & Fig. S11) gave a pKa value of 9.8 whereas a plot of the peak intensity at 400 nm as a function of pH gave a similar pKa value of 9.9. These data suggest that the FeII(MPB)(OH2) complex deprotonates, but only at high pH. The group that deprotonates with this high pKa is most likely the Fe(II) bound water to form the FeII(MPB)(OH) complex. An analogous pH titration to determine the pKa of the hydroxyl of FeII(MPH) was not feasible as the complex oxidized to the Fe(III) form at basic pH values. However, the cyclic voltammetry of the complex in water as a function of pH as discussed below suggests that the pKa is ≈9 for the FeII(MPH) complex, most likely due to deprotonation of the hydroxyl group. A pH titration of the oxidized complex, FeIII(MPH), showed changes in the UV-vis bands that were consistent with a pKa of close to 6. This pKa value is similar to that of other Fe(III) complexes with hydroxyl groups,56 suggesting deprotonation of this pendant (Fig. S12 & 13).
Variable-temperature 17O NMR spectra recorded over the temperature range 25 to 75 °C are presented in (Fig. 4C) and in SI (Fig. S16). In this temperature range at pH 5 and 7.45 mM, the FeII(MPB) complex produces broadening and a paramagnetically induced shift of the bulk water 17O resonance, consistent with behavior expected for complexes with an inner-sphere bound water molecule. The resonance is broader at lower temperature and sharpens as the temperature increases. A plot of ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1/T2R versus temperature (Fig. 4D) showed the upward curve indicating the temperature dependence of the transverse relaxivity is dominated by the changes in the water exchange rate.57 A transition from the slow-exchange regime at low temperatures to the fast exchange regime at higher temperatures relative to the NMR timescale was observed. These data were fit to Swift Connick equations to determine the activation parameters (Table S4) including enthalpy, entropy and an exchange rate constant of 2.4 × 106 s−1 at 25 °C. Plots of the reduced chemical shift as a function of temperature (Fig. S14) gave kex values about 3-fold larger and global fitting efforts (Fig. S15) gave relatively poor fits. We attribute these discrepancies to relatively low concentrations of complex (7.45 mM) that were used due to solubility restrictions whereas these studies are typically carried out with 20–40 mM complex.58 Variable temperature 17O NMR studies with FeII(MPH) at pH 4.0 (Fig. S16 and S17) also were consistent with an inner-sphere water with a kex of 3.1 × 106 s−1. An inner-sphere water in this complex may result from the formation of a seven-coordinate complex or by displacement of the hydroxypropyl pendant as the temperature is raised in the 17O NMR experiment. At 25 °C, we propose that the hydroxypropyl pendant is bound based on the 1H NMR spectrum and electrochemical properties (see below) which differ from those of FeII(MPB). If the hydroxypropyl pendant was not bound, the 1H NMR and the redox potential of FeII(MPH) would be expected to be very similar to that of FeII(MPB). The kex rate constants for water exchange in FeII(MPB) and FeII(MPH) are similar to those reported for polyaminocarboxylate complexes of Fe(II) which range from 1.1 to 5.5 × 106 s−1.58–60
1/T2R versus temperature (Fig. 4D) showed the upward curve indicating the temperature dependence of the transverse relaxivity is dominated by the changes in the water exchange rate.57 A transition from the slow-exchange regime at low temperatures to the fast exchange regime at higher temperatures relative to the NMR timescale was observed. These data were fit to Swift Connick equations to determine the activation parameters (Table S4) including enthalpy, entropy and an exchange rate constant of 2.4 × 106 s−1 at 25 °C. Plots of the reduced chemical shift as a function of temperature (Fig. S14) gave kex values about 3-fold larger and global fitting efforts (Fig. S15) gave relatively poor fits. We attribute these discrepancies to relatively low concentrations of complex (7.45 mM) that were used due to solubility restrictions whereas these studies are typically carried out with 20–40 mM complex.58 Variable temperature 17O NMR studies with FeII(MPH) at pH 4.0 (Fig. S16 and S17) also were consistent with an inner-sphere water with a kex of 3.1 × 106 s−1. An inner-sphere water in this complex may result from the formation of a seven-coordinate complex or by displacement of the hydroxypropyl pendant as the temperature is raised in the 17O NMR experiment. At 25 °C, we propose that the hydroxypropyl pendant is bound based on the 1H NMR spectrum and electrochemical properties (see below) which differ from those of FeII(MPB). If the hydroxypropyl pendant was not bound, the 1H NMR and the redox potential of FeII(MPH) would be expected to be very similar to that of FeII(MPB). The kex rate constants for water exchange in FeII(MPB) and FeII(MPH) are similar to those reported for polyaminocarboxylate complexes of Fe(II) which range from 1.1 to 5.5 × 106 s−1.58–60
Cyclic voltammetry measurements of the two Fe(II) complexes in water were studied at neutral pH (Fig. 5) and then at several different pH values (Fig. 5 & S19). At pH 7.6, both FeII(MPB) and FeII(MPH) have quasi-reversible cyclic voltammetry waves with E1/2 of 680 mV and 490 mV, respectively. For Fe(MPB), the wave was consistent with an electrochemically-reversible and chemically-irreversible (ErCi) mechanism, in which a quasi-reversible electron transfer step is followed by an irreversible homogeneous chemical reaction of the electrochemically generated product.61 The FeII(MPB)(H2O) complex undergoes one-electron oxidation to give the FeIII(MPB)(H2O) product. The peak currents associated with this process increase linearly with the square root of the scan rate (Fig. S20), indicating that mass transport is governed by diffusion control as shown by fitting to the Randles–Sevcik equation. The wave with a redox potential of 675 ± 25 mV versus NHE and the peak separation between oxidation and reduction waves is substantially larger than the 59 mV expected for a Nernstian one-electron couple and increases systematically with scan rate, consistent with a quasi-reversible redox event.
The pH dependence of the cyclic voltammograms was studied to gain insight into speciation of the two complexes. For Fe(MPB), the wave remained virtually unchanged from pH 6 to 7.6, except for the new reduction peak that appears after the initially observed reduction peak. The 1st reduction (cathodic) peak current is consistently smaller in magnitude than the corresponding oxidation (anodic) peak current with current ratio Ipc/Ipa of 0.53. This current ratio increases at higher scan rates, which is characteristic of an ErCi mechanism. The first oxidation product FeIII(MPB)(H2O), undergoes deprotonation of bound water to give a hydroxide complex, FeIII(MPB)(OH), which is reduced to form FeII(MPB)(OH), alongside the major reduction product FeII(MPB)(H2O). FeII(MPH) also shows an ErCi type mechanism (Fig. 5B & S21) with quasi-reversible wave at E1/2 = 542 mV versus NHE at pH 4.7 which shifts slightly to 490 mV at neutral pH. A new wave at lower potential appears by pH 9.3 and continues to grow in at pH 10.3. This quasi-reversible wave is assigned to the deprotonated hydroxypropyl species (Fe(MPH-H+)). The lowered value is consistent with stabilization of the Fe(III) oxidation state by the anionic alkoxide pendant.56
Studies at low concentrations of peroxide
Reports of redox-responsive iron probes focus on the effect of oxidants or reductants on the MRI signal and kinetics of oxidation or reduction38,39,44 yet further characterization of products would be beneficial.40,41 Here we study the oxidation process and refer to the rich literature on reactions of Fe(II) with peroxide.48,49,62,63 Many studies of the species formed upon addition of peroxide to Fe(II) complexes are done in methanol or acetonitrile, but there are limited studies in aqueous solution.The oxidation of FeII(MPB) and FeII(MPH) by peroxide was initially studied by electronic absorption spectroscopy. For both complexes, the rate of oxidation with one equivalent of peroxide at neutral pH in buffered solutions was too rapid to be measured by conventional methods (<3 minutes). Under these conditions, the change in absorbance signifying oxidation to Fe(III) was a new absorbance band at 320 nm for FeIII(MPB) (Fig. S24 & S25) and at 300 nm for FeIII(MPH). Complete oxidation of the complexes was observed with one equivalent of peroxide in solutions containing 200 µM complex, concentrations that are relevant for contrast agents1 and peroxide in the blood stream.64
The kinetics of oxidation of FeII(MPB) and FeII(MPH) by the enzyme glucose oxidase (GOX) was studied next. GOX produces a steady stream of peroxide in the presence of oxygen and glucose, making it feasible to measure the rate constant for oxidation. As shown in Fig. 6A & S26, both complexes oxidized rapidly in the presence of 0.1 units per mL of enzyme and the reaction rate constant was proportional to the amount of GOX added to solutions (Fig. 6). The initial rates for FeII(MPB) and FeII(MPH) with 0.1 units per mL of GOX were 1.5 × 10−4 mM s−1 and 2.6 × 10−4 mM s−1, respectively, suggesting slightly higher reactivity for FeII(MPH), consistent with its lower redox potential. Interestingly, oxidation of the complexes by GOX could be monitored until completion and the data could be fit to an exponential decay curve for a pseudo-first order process due to the large amount of peroxide in excess of complex generated by GOX (Fig. S27 and S33). Notably, only the new peaks at 320 and 300 nm were observed upon oxidation of FeII(MPB) and FeII(MPH) by GOX, respectively. However, samples of FeII(MPB) left to stand for a day in the GOX solution showed a new band at 500 nm, consistent with a buildup of peroxide and the formation of a new species produced by hydroxylation of the benzyl group which is discussed in the next section.
To test the reversibility of the probe towards redox processes, FeIII(MPB) and FeIII(MPH) were generated by treatment of the Fe(II) complexes with half an equivalent of peroxide. Subsequently, two equivalents of ascorbate were added to fully regenerate the Fe(II) complexes as shown by UV-vis spectroscopy (Fig. S39 & S40). Notably, the original electronic absorbance spectra of both FeII complexes were regenerated nearly instantaneously upon addition of ascorbate.65
The solution chemistry of the Fe(III) complexes was further studied to probe water interactions and resistance to dissociation. Variable temperature 17O NMR of FeIII(MPB) in aqueous solutions showed very little line broadening of the 17O water resonance (Fig. S41). A plot of line broadening (r2O) as a function of temperature (Fig. S42) in comparison to FeIII(CDTA) and FeIII(DTPA) reveals that the FeIII(MPB) complex resembles Fe(DTPA), which does not possess inner-sphere water, except for a slight increase in line broadening at higher temperatures. The minimal effect of FeIII(MPB) on the water 17O resonance suggests that the complex does not have an inner-sphere water ligand that exchanges on the NMR time scale. This is attributed to the FeIII(MPB)(X) species with X as a tightly bound water or hydroxide ligand. This result is consistent with other six-coordinate Fe(III) complexes of TACN that exchange inner-sphere water slowly on the NMR time scale.56,66,67
Transferrin transchelation of iron was studied for both complexes by monitoring the increase in the absorbance at 465 nm which corresponds to the Fe-transferrin band, with an extinction coefficient of 4950 M−1 cm−1. These studies (Fig. S43) revealed that FeIII(MPB) undergoes 30% transchelation to apo-transferrin over five hours with a first order rate constant of 2.2 × 10−3 min−1 and half-life of 315 minutes, whereas FeIII(MPH) undergoes less than 10% transchelation to apo-transferrin over five hours. Thus, FeIII(MPH) is highly inert to loss of iron to transferrin, consistent with it being more robust as it contains a hexadentate ligand whereas FeIII(MPB) is inert, but less so. Further aspects of solution chemistry were captured by relaxivity changes over time as discussed below.
Studies with excess peroxide
Upon addition of an excess of peroxide to FeII(MPB) in buffered solution at neutral pH, a second band at 500 nm appeared (Fig. 7). By contrast, FeII(MPH) showed no absorbance band in that region of the spectrum, regardless of peroxide concentration (Fig. S25). Based on the following studies, we assign the species that forms upon treatment of FeII(MPB) under conditions of excess peroxide as FeIII(MPP) (Scheme 2) and the band at 500 nm as a LMCT band for a phenolate complex. At 2 equivalents of peroxide in buffered solution, approximately 15% of the species is FeIII(MPP), indicating that it is a minor component. However, the complex forms more readily in the absence of buffer, so further studies to identify this species were carried out in the absence of buffer. | ||
| Fig. 7 Electronic spectra tracking the oxidation of FeII(MPB) with various equivalents of peroxide. Conditions: 0.20 mM complex, 0.10 to 10 mM H2O2, 0.1 M NaCl and 20 mM HEPES pH 7.4. | ||
To probe solution speciation under conditions of excess peroxide addition to FeII(MPB), high resolution mass spectrometry was performed on the product solutions at pH 7.4 (Fig. S44 to S47). These data show that with two equivalents of peroxide in solutions lacking buffer the phenolate complex FeIII(MPP) is formed as a minor component (13% relative abundance). However, with ten equivalents of peroxide the FeIII(MPP) complex is one of the most abundant species in solution. Complexes assigned as end-on (FeIII(MPB)(OOH)) and side-on (FeIII(MPB)(OO)) peroxo are identified as minor species (38% and 13% relative abundance) with respect to FeIII(MPP). Notably, end-on and side on peroxide complexes of analogous Fe(III) complexes with five nitrogen donor groups form upon treatment with peroxide and with base.49,62,68 The Fe(III) peroxo complex then may undergo heterolytic O–O cleavage to produce a high valent Fe(IV) oxo complex.48,63 Such high valent oxoiron(IV) complexes have been shown to oxidize non-coordinating benzyl groups to phenolate ligands.69
Further solution studies were carried out with FeII(MPB) and peroxide in solutions lacking buffer, conditions where FeIII(MPP) more readily forms. UV-vis spectra are shown for FeII(MPB) treated with two equivalents of peroxide and titrated as a function of pH (Fig. S48). Under these conditions, the phenol complex FeIII(MPP) is present as a major species as shown by the absorbance band at 500 nm at neutral to basic pH. As the pH becomes more acidic, a new band appears at 600 nm which is assigned to the LMCT band of the protonated FeIII(MPP) complex. The intensity of this peak increases upon titration from pH 6.5 to pH 4.0 (Fig. S49). Plots of these data give an apparent pKa value of 5.7 for the deprotonation of the phenol. In a second set of experiments, cyclic voltammetry studies of aqueous solutions of FeII(MPB) with KCl, no buffer and one equivalent of peroxide were studied (Fig. S50). A new quasi-reversible wave was observed at more negative values than the original complex with an E1/2 of 375 mV. This species is assigned as a protonated phenol complex and is observed at acidic pH values (pH 4) in unbuffered solutions.
Probe characteristics
The water proton relaxivity of the Fe(II) complexes and their corresponding Fe(III) analogs were measured (Table S12) at 1.4 T and 34 °C on an NMR spectrometer and at 7.0 T and 37 °C on an MRI scanner. As anticipated, the Fe(II) complexes showed low relaxivities of 0.05 and 0.09 mM−1 s−1 respectively at 1.4 T. This low relaxivity is consistent with Fe(II) complexes being paramagnetic shift agents rather than relaxivity agents as highlighted by the NMR spectra in Fig. 2. The relaxivity increased by 14-fold and 7-fold respectively for FeIII(MPB) and FeIII(MPH) upon treatment with two equivalents of peroxide. Plots of R1 as a function of iron complex (Fig. S52) gave relaxivities of 0.71 mM−1 s−1 and 0.86 mM−1s−1 for FeIII(MPB) and 0.58 mM−1 s−1 and 0.63 mM−1s−1 for FeIII(MPH), at 1.4 T and 7 T respectively (Table S12). Addition of serum albumin did not alter the relaxivity of FeIII(MPB) but increased the relaxivity of FeIII(MPH) slightly to 0.73 mM−1 s−1. The relaxivity values for both complexes are characteristic of closed-coordinate Fe(III) high-spin complexes which function through second-sphere water interactions. This suggests that FeIII(MPB) either has a slowly exchanging water ligand, or a hydroxide ligand. Relaxivity values tracked as a function of equivalents of peroxide added to FeII(MPB) showed a decrease at high peroxide conditions (>100), with nearly constant values from one equivalent to 50 equivalents of peroxide (Table S13). This is consistent with the initial formation of FeIII(MPB) complex at low peroxide followed by the formation of the phenol complex at high concentrations of peroxide.MRI phantoms (Fig. 8, S54 & S56) show that the R1 and R2 of the FeII complexes increase as a function of concentration and upon oxidation by peroxide. (Note that R1 and R2 are the rate constants associated with T1 and T2 processes.) The left side of the array corresponds to the Fe(II) complex and the right side of the array corresponds to the Fe(III) complex. The most highly concentrated Fe(III) sample shows the brightest contrast in both the maps. This is consistent with the enhancement of R1 relaxation rate constant upon oxidation of the Fe(II) complexes to give rise to higher relaxivity Fe(III) species. A turn-on ratio of 11 and 7 for r1 relaxivity of FeIII to FeII is observed at 7 T for FeII(MPB) and FeII(MPH) respectively.
 | ||
| Fig. 8 R1 (s−1) map of FeII(MPB) (A) and FeII(MPH) (B) before and after oxidation. Conditions: 0 to 0.75 mM complex, 2 eq. H2O2, 20 mM HEPES pH 7.4, 0.1 M NaCl, 310 K, 7 T. | ||
The reversibility of the response of the MRI probes towards oxidation and reduction was studied (Fig. 9). One equivalent of peroxide was added followed by addition of one equivalent of ascorbate to solutions containing 500 µM probe. After the first cycle, the relaxivity values were similar to the original Fe(II) values. However, the second oxidation produced slightly lower relaxivity values than the first oxidation. The lack of complete reversibility is attributed, in part, to the characteristics of probe solution chemistry over time. Notably, the relaxivity of FeIII(MPB) and FeIII(MPH) decreases slightly when monitored over several hours in the presence and absence of human serum albumin (Tables S15 & S16, Fig. S57 & 58). In the absence of human serum albumin, the relaxivities of both the complexes decreased by 25 percent over four hours. In the presence of human serum albumin, FeIII(MPB) decreased by 33% and FeIII(MPH) decreased by 11%. Addition of an equivalent of peroxide partially restored the relaxivity of FeIII(MPB), but not FeIII(MPH) in solutions containing 500 µM complex at neutral pH (Fig. S59). UV-vis spectroscopy experiments (Fig. S60 and S61) at lower concentrations (200 µM) showed full restoration of electronic absorbance bands of FeIII(MPB) and FeIII(MPH), upon addition of half an equivalent of peroxide at 4 hours, suggesting that the complexes are slowly reduced in solution over several hours.
Conclusions
Consideration of the coordination chemistry of peroxide with Fe(II) complexes is instrumental in the design of a redox-responsive MRI probe that is selective for peroxide over O2. Although the relatively high oxidation potential of the iron center (680 mV) indicates that outer-sphere oxidation by peroxide (H2O2/OH˙−, E°′ = 390 mV) should be unfavorable, exposure to one equivalent of peroxide nonetheless yields the FeIII(MPB) complex, most likely through an inner-sphere redox processes. Oxidation is rapid, similar to other Fe(II) probes that contain available sites for inner-sphere oxidation with peroxide.38 The rapid reaction of the FeII(MPH) complex with peroxide is consistent with the availability of a coordination site for binding an incoming ligand either through formation of a seven-coordinate complex or by displacement of the hydroxypropyl pendant as well as the lower redox potential of the probe. Moreover, the interaction of the hydroxypropyl group with peroxide has been proposed to increase the rate of oxidation, as shown for analogous Co(II) complexes.70The selectivity of the FeII(MPB) probe for oxidation by peroxide over oxygen is promising and the turn-on ratio is similar to that of other iron-based probes.38,40 However, improved overall relaxivity of the Fe(III) complex is desirable and may be accomplished by optimizing relaxivity parameters such as correlation times through larger assemblies and by modulation of water exchange71 as well efforts towards improving water solubility.
Finally, the oxidation of the aromatic ring to a phenolate group might be prevented by addition of ortho-substituents72 or the non-coordinating pendent may be changed to a less easily oxidized group. These modifications are underway towards rapid-response, high selectivity peroxide-responsive iron-based MRI probes.
Author contributions
DKB: investigation, analysis, validation, writing; JAS: investigation, data curation, software; MRC: investigation and analysis; JRM: conceptualization, writing, funding acquisition.Conflicts of interest
JRM is the cofounder of Ferric Contrast, Inc., a company that develops iron-based contrast agents.Data availability
The datasets supporting this article have been uploaded as part of the supplementary information (SI). Supplementary information: materials and methods, synthetic procedures, spectroscopic characterization, crystallographic data, NMR data, electrochemistry, MRI phantoms. See DOI: https://doi.org/10.1039/d6qi00660d.Additional data will be provided by the authors upon reasonable request.
CCDC 2539019 contains the supplementary crystallographic data for this paper.73
Acknowledgements
JRM thanks the National Science Foundation (CHE-2400128) for support of this research. This work utilized a Rigaku XtaLAB Synergy-S diffractometer that was purchased with funding from NSF (CHE-2216151), and the Bruker 500 MHz NMR (NSF CHE-2018160), ICP-MS and FTMS (NSF CHE-0959565). JAS is grateful for the Roswell Park's Comprehensive Cancer Center Support Grant: P30CA016056. The 7T MRI scanner was purchased through funds from NIH award S10OD030397.References
- J. Wahsner, E. M. Gale, A. Rodriguez-Rodriguez and P. Caravan, Chemistry of MRI Contrast Agents: Current Challenges and New Frontiers, Chem. Rev., 2019, 119, 957–1057 CrossRef CAS PubMed.
- C. Westbrook and J. Talbot, MRI in Practice, Wiley-Blackwell, Hoboken, NJ, 5th edn, 2018 Search PubMed.
- S. Shuvaev, E. Akam and P. Caravan, Molecular MR Contrast Agents, Invest. Radiol., 2021, 56, 20–34 CrossRef CAS PubMed.
- L. M. De Leon-Rodriguez, A. J. Lubag, C. R. Malloy, G. V. Martinez, R. J. Gillies and A. D. Sherry, Responsive MRI agents for sensing metabolism in vivo, Acc. Chem. Res., 2009, 42, 948–957 CrossRef CAS.
- H. Li and T. J. Meade, Molecular magnetic resonance imaging with Gd(III)-based contrast agents: challenges and key advances, J. Am. Chem. Soc., 2019, 141, 17025–17041 CrossRef CAS PubMed.
- S. M. Pinto, V. Tome, M. J. F. Calvete, M. M. C. A. Castro, E. Toth and C. F. G. C. Geraldes, Metal-based redox-responsive MRI contrast agents, Coord. Chem. Rev., 2019, 390, 1–31 CrossRef CAS.
- S. S. Xue, Y. Pan, W. Pan, S. Liu, N. Li and B. Tang, Bioimaging agents based on redox-active transition metal complexes, Chem. Sci., 2022, 13, 9468–9484 RSC.
- J. R. Morrow, J. J. Raymond, M. S. I. Chowdhury and P. R. Sahoo, Redox-Responsive MRI Probes Based on First-Row Transition-Metal Complexes, Inorg. Chem., 2022, 61, 14487–14499 CrossRef CAS PubMed.
- C. S. Bonnet and E. Toth, Metal-based environment-sensitive MRI contrast agents, Curr. Opin. Chem. Biol., 2021, 61, 154–169 CrossRef CAS PubMed.
- P. Yue and G. Angelovski, How to Develop Bioresponsive MRI Probes Based on Paramagnetic Gd(III) for Applications, Anal. Sens., 2023, 3, 2629–2742 Search PubMed.
- S. Karbalaei and C. R. Goldsmith, Recent advances in the preclinical development of responsive MRI contrast agents capable of detecting hydrogen peroxide, J. Inorg. Biochem., 2022, 230, 111763 CrossRef CAS PubMed.
- J. D. Hayes, A. T. Dinkova-Kostova and K. D. Tew, Oxidative Stress in Cancer, Cancer Cell, 2020, 38, 167–197 CrossRef CAS.
- M. Mittal, M. R. Siddiqui, K. Tran, S. P. Reddy and A. B. Malik, Reactive Oxygen Species in Inflammation and Tissue Injury, Antioxid. Redox Signaling, 2014, 20, 1126–1167 CrossRef CAS PubMed.
- C. Nathan and A. Cunningham-Bussel, Beyond oxidative stress: an immunologist's guide to reactive oxygen species, Nat. Rev. Immunol., 2013, 13, 349–361 CrossRef CAS PubMed.
- A. El-Kenawi and B. Ruffell, Inflammation, ROS, and Mutagenesis, Cancer Cell, 2017, 32, 727–729 CrossRef CAS PubMed.
- M. Abdesselem, N. Pétri, R. Kuhner, F. Mousseau, V. Rouffiac, T. Gacoin, C. Laplace-Builhé, A. Alexandrou and C. I. Bouzigues, Real-time in vivo ROS monitoring with luminescent nanoparticles reveals skin inflammation dynamics, Biomed. Opt. Express, 2023, 14, 5392–5404 CrossRef CAS PubMed.
- W. H. Yu, Y. M. Tu, Z. Long, J. Z. Liu, D. Q. Kong, J. Peng, H. Wu, G. Zheng, J. Z. Zhao, Y. H. Chen, R. Liu, W. L. Li and C. X. Hai, Reactive Oxygen Species Bridge the Gap between Chronic Inflammation and Tumor Development, Oxid. Med. Cell. Longevity, 2022, 2022, 2606928 CrossRef PubMed.
- H. Sies, V. V. Belousov, N. S. Chandel, M. J. Davies, D. P. Jones, G. E. Mann, M. P. Murphy, M. Yamamoto and C. Winterbourn, Defining roles of specific reactive oxygen species (ROS) in cell biology and physiology, Nat. Rev. Mol. Cell Biol., 2022, 23, 499–515 CrossRef CAS PubMed.
- Y. Feng, C. Santoriello, M. Mione, A. Hurlstone and P. Martin, Live Imaging of Innate Immune Cell Sensing of Transformed Cells in Zebrafish Larvae: Parallels between Tumor Initiation and Wound Inflammation, PLos Biol., 2010, 14, e1002377 CrossRef PubMed.
- W. H. Koppenol and R. H. Hider, Iron and redox cycling. Do's and don'ts, Free Radical Biol. Med., 2019, 133, 3–10 CrossRef CAS PubMed.
- P. B. Tsitovich, P. J. Burns, A. M. Mckay and J. R. Morrow, Redox-activated MRI contrast agents based on lanthanide and transition metal ions, J. Inorg. Biochem., 2014, 133, 143–154 CrossRef CAS PubMed.
- Q. N. Do, J. S. Ratnakar, Z. Kovacs and A. D. Sherry, Redox- and Hypoxia-Responsive MRI Contrast Agents, ChemMedChem, 2014, 9, 1116–1129 CrossRef CAS PubMed.
- A. Li, X. X. Tang, X. Q. Gong, H. M. Chen, H. Y. Lin and J. H. Gao, A fluorinated bihydrazide conjugate for activatable sensing and imaging of hypochlorous acid by 19-F NMR/MRI, Chem. Commun., 2019, 55, 12455–12458 RSC.
- Y.-X. Chen, A. Ge, X. Guo, M. Yu and J. Tao, Spin State Modulation Strategies for Transition Metal-Based MRI Contrast Agents, Chem. Biomed. Imaging, 2026, 4, 274–303 CrossRef CAS PubMed.
- E. C. Lewandowski, C. B. Arban, M. P. Deal, A. L. Batchev and M. J. Allen, Europium(II/III) coordination chemistry toward applications, Chem. Commun., 2024, 60, 10655–10671 RSC.
- L. A. Basal and M. J. Allen, Synthesis, Characterization, and Handling of Eu Containing Complexes for Molecular Imaging Applications, Front. Chem., 2018, 6, 65 CrossRef PubMed.
- S. A. A. S. Subasinghe, C. J. Ortiz, J. Romero, C. L. Ward, A. G. Sertage, L. Kurenbekova, J. T. Yustein, R. G. Pautler and M. J. Allen, Toward quantification of hypoxia using fluorinated EuII/III-containing ratiometric probes, Proc. Natl. Acad. Sci. U. S. A., 2023, 120(15), e2220891120 CrossRef CAS PubMed.
- S. M. A. Pinto, A. R. R. Ferreira, D. S. S. Teixeira, S. C. C. Nunes, A. L. M. Batista de Carvalho, J. M. S. Almeida, Z. Garda, A. Pallier, A. A. C. C. Pais, C. M. A. Brett, E. Toth, M. P. M. Marques, M. M. Pereira and C. F. G. C. Geraldes, Fluorinated Mn(III)/(II)-Porphyrin with Redox-Responsive (1) H and (19) F Relaxation Properties, Chem. – Eur. J., 2023, 29, e202301442 CrossRef CAS PubMed.
- D. Xie, M. Yu, R. T. Kadakia and E. L. Que, F-19 Magnetic Resonance Activity-Based Sensing Using Paramagnetic Metals, Acc. Chem. Res., 2020, 53, 2–10 CrossRef CAS PubMed.
- P. B. Tsitovich, J. A. Spernyak and J. R. Morrow, A redox-activated MRI contrast agent that switches between paramagnetic and diamagnetic states, Angew. Chem., Int. Ed., 2013, 52, 13997–14000 CrossRef CAS PubMed.
- A. M. Funk, V. C. Jordan, A. D. Sherry, S. J. Ratnakar and Z. Kovacs, Oxidative Conversion of a Europium(II)-Based T-1 Agent into a Europium(III)-Based paraCEST Agent that can be Detected In Vivo by Magnetic Resonance Imaging, Angew. Chem., Int. Ed., 2016, 55, 5024–5027 CrossRef CAS PubMed.
- E. A. Kras, E. M. Snyder, G. E. Sokolow and J. R. Morrow, Distinct Coordination Chemistry of Fe(III)-Based MRI Probes, Acc. Chem. Res., 2022, 55, 1435–1444 CrossRef CAS PubMed.
- J. R. Morrow and J. J. Raymond, in Metal ions in life sciences (Lanthanides and Other Transition Metal Ion Complexes and Nanoparticles in Magnetic Resonance Imaging), Metal Ions in Life Sciences 27, ed. C. F. G. C. Geraldes, A. Sigel, H. Sigel, E. Fresinger and R. K. O. Sigel, Taylor & Francis, 2023 Search PubMed.
- A. Gupta, P. Caravan, W. S. Price, C. Platas-Iglesias and E. M. Gale, Applications for transition-metal chemistry in contrast-enhanced magnetic resonance imaging, Inorg. Chem., 2020, 59, 6648–6678 CrossRef CAS PubMed.
- Z. Baranyai, F. Carniato, A. Nucera, D. Horvath, L. Tei, C. Platas-Iglesias and M. Botta, Defining the conditions for the development of the emerging class of Fe(III)-based MRI contrast agents, Chem. Sci., 2021, 12, 11138 RSC.
- N. Kuznik and M. Wyskocka, Iron(III) Contrast Agent Candidates for MRI: a Survey of the Structure-Effect Relationship in the Last 15 Years of Studies, Eur. J. Inorg. Chem., 2016, 4, 445–458 CrossRef.
- P. B. Tsitovich, F. Gendron, A. Y. Nazarenko, B. N. Livesay, A. P. Lopez, M. P. Shores, J. Autschbach and J. R. Morrow, Low-Spin Fe(III) Macrocyclic Complexes of Imidazole-Appended 1,4,7-Triazacyclononane as Paramagnetic Probes, Inorg. Chem., 2018, 57, 8364–8374 CrossRef CAS PubMed.
- H. Wang, V. C. Jordan, I. A. Ramsay, M. Sojoodi, B. C. Fuchs, K. K. Tanabe, P. Caravan and E. M. Gale, Molecular magnetic resonance imaging using a redox-active iron complex, J. Am. Chem. Soc., 2019, 141, 5916–5925 CrossRef CAS PubMed.
- R. T. Kadakia, R. T. Ryan, D. J. Cooke and E. L. Que, An Fe complex for (19)F magnetic resonance-based reversible redox sensing and multicolor imaging, Chem. Sci., 2023, 14, 5099–5105 RSC.
- S. Karbalaei, A. Franke, A. Jordan, C. Rose, P. R. Pokkuluri, R. J. Beyers, A. Zahl, I. Ivanovic-Burmazovic and C. R. Goldsmith, A Highly Water- and Air-Stable Iron-Containing MRI Contrast Agent Sensor for H2O2, Chem. – Eur. J., 2022, 28, e202201179 CrossRef CAS.
- S. Karbalaei, A. Franke, A. Zahl, P. R. Pokkuluri, R. J. Beyers, I. Ivanovic-Burmazovic and C. R. Goldsmith, An Fe(ii) complex detects hydrogen peroxide with 1H and 19F magnetic resonance imaging responses, Chem. Commun., 2025, 61, 15898–15901 RSC.
- A. A. Kitos, R. Castañeda, Z. J. Comeau, N. Mavragani, N. D. Calvert, A. Kirby, F. M. Martinez-Santiesteban, P. J. Pallister, T. J. Scholl, M. Murugesu, A. J. Shuhendler and J. L. Brusso, Cluster-based redox-responsive super-atomic MRI contrast agents, Chem, 2025, 11, 102330 CAS.
- K. K. Huang, M. M. Li, X. Zhang, X. Linghu, Y. B. Wang, W. C. Li, P. J. Wu, J. Q. Dong, S. P. Yang and J. J. Jiao, Magnetically switchable coordination cages: a class of supramolecular contrast agents for tumor-responsive magnetic resonance imaging, Sci. China. Chem., 2026 DOI:10.1007/s11426-025-3181-8.
- Z. Garda, F. Szeremeta, C. N. Tóth, S. Bunda, C. Pifferi, R. Clémençon, S. Même, G. Tircsó and É. Tóth, Relaxation-Based In Vivo Discrimination of Oxidized and Reduced States of a Redox-Switchable 19F MRI Probe, J. Am. Chem. Soc., 2025, 147, 18017–18024 CrossRef CAS PubMed.
- V. C. Jordan, M. Sojoodi, S. Shroff, P. G. Pagan, S. C. Barrett, J. Wellen, K. K. Tanabe, R. T. Chung, P. Caravan and E. M. Gale, Molecular magnetic resonance imaging of liver inflammation using an oxidatively activated probe, JHEP Rep., 2023, 5(10), 100850 CrossRef PubMed.
- R. M. King, M. J. Gounis, E. J. Schmidt, A. Leporati, E. M. Gale and A. J. r. Bogdanov, Molecular Magnetic Resonance Imaging of Aneurysmal Inflammation Using a Redox Active Iron Complex, Invest. Radiol., 2023, 58, 656–662 CrossRef CAS.
- I. A. Rosales, I. Y. Zhou, I. Ay, M. Sojoodi, M. E. Sise and E. M. Gale, Imaging kidney inflammation using an oxidatively activated MRI probe, Kidney Int., 2024, 106, 671–678 CrossRef CAS PubMed.
- J. Chen, A. S. Sardjan, C. M. de Roo, M. Di Berto Mancini, A. Draksharapu, D. Angelone, R. Hage, M. Swart and W. R. Browne, Generation of [(N4Py)Fe(IV)=O]2+ through Heterolytic O-O Bond Cleavage in [(N4Py)Fe(II)(OOH)], Inorg. Chem., 2025, 64, 9408–9417 CrossRef CAS.
- G. Roelfes, V. Vrajmasu, K. Chen, R. Y. Ho, J. U. Rohde, C. Zondervan, R. M. La Crois, E. P. Schudde, M. Lutz, A. L. Spek, R. Hage, B. L. Feringa, E. Munck and L. Que Jr., End-on and side-on peroxo derivatives of non-heme iron complexes with pentadentate ligands: models for putative intermediates in biological iron/dioxygen chemistry, Inorg. Chem., 2003, 42, 2639–2653 CrossRef CAS PubMed.
- X. P. Zhang, D. L. Zhang, D. H. Busch and R. van Eldik, Catalytic action of the iron(II) complexes of 8-methyl-1,4-bis(2-pyridylmethyl)-1,4,8-triazacycloundecane and 1-methyl-5,9-bis(2-pyridylmethyl)-1,5,9 triazacyclododecane, J. Chem. Soc., Dalton Trans., 1999, 2751–2758 RSC.
- P. B. Tsitovich, J. M. Cox, J. B. Benedict and J. R. Morrow, Six-coordinate Iron(II) and Cobalt(II) paraSHIFT Agents for Measuring Temperature by Magnetic Resonance Spectroscopy, Inorg. Chem., 2016, 55, 700–716 CrossRef CAS PubMed.
- J. Wang, C. Gondrand, F. Touti and J. Hasserodt, A pair of highly biotolerated diamagnetic and paramagnetic iron(II) complexes displaying electroneutrality, Dalton Trans., 2015, 44, 15391–15395 RSC.
- J. Salaam, T. Fogeron, G. Pilet, R. Bolbos, C. Bucher, L. Khrouz and J. Hasserodt, Unprecedented Relaxivity Gap in pH-Responsive Fe(III) -Based MRI Probes, Angew. Chem., Int. Ed., 2023, 62, e202212782 CrossRef CAS PubMed.
- L. Christiansen, D. N. Hendrickson, H. Toftlund, S. R. Wilson and C. L. Xie, Synthesis and Structure of Metal-Complexes of Triaza Macrocycles with 3 Pendant Pyridylmethyl Arms, Inorg. Chem., 1986, 25, 2813–2818 CrossRef CAS.
- P. B. Tsitovich, T. Y. Tittiris, J. M. Cox, J. B. Benedict and J. R. Morrow, Fe(II) and Co(II) methylated CYCLEN complexes as paraSHIFT agents with large temperature dependent shifts, Dalton Trans., 2018, 47, 916–924 RSC.
- E. M. Snyder, D. Asik, S. M. Abozeid, A. Burgio, G. Bateman, S. G. Turowski, J. A. Spernyak and J. R. Morrow, A Class of FeIII Macrocyclic Complexes with Alcohol Donor Groups as Effective T1 MRI Contrast Agents, Angew. Chem., 2020, 132, 2435–2440 CrossRef.
- A. Brausam, J. Maigut, R. Meier, P. A. Szilágyi, H.-J. r. Buschmann, W. Massa, Z. Homonnay and R. van Eldik, Detailed spectroscopic, thermodynamic, and kinetic studies on the protolytic equilibria of FeIII(cydta) and the activation of hydrogen peroxide, Inorg. Chem., 2009, 48, 7864–7884 CrossRef CAS PubMed.
- J. Maigut, R. Meier, A. Zahl and R. van Eldik, Elucidation of the solution structure and water-exchange mechanism of paramagnetic [Fe (edta)(HO)], Inorg. Chem., 2007, 46, 5361–5371 CrossRef CAS.
- J. Maigut, R. Meier, A. Zahl and R. van Eldik, Effect of chelate dynamics on water exchange reactions of paramagnetic aminopolycarboxylate complexes, Inorg. Chem., 2008, 47, 5702–5719 CrossRef CAS PubMed.
- J. Maigut, R. Meier, A. Zahl and R. van Eldik, Triggering Water Exchange Mechanisms via Chelate Architecture. Shielding of Transition Metal Centers by Aminopolycarboxylate Spectator Ligands, J. Am. Chem. Soc., 2008, 130, 14556–14569 CrossRef CAS PubMed.
- N. Elgrishi, K. J. Rountree, B. D. McCarthy, E. S. Rountree, T. T. Eisenhart and J. L. Dempsey, A Practical Beginner's Guide to Cyclic Voltammetry, J. Chem. Educ., 2018, 95, 197–206 CrossRef CAS.
- A. Bohn, C. Chinaux-Chaix, K. Cheaib, R. Guillot, C. Herrero, K. Senechal-David, J. N. Rebilly and F. Banse, Base-controlled mechanistic divergence between iron(iv)-oxo and iron(iii)-hydroperoxo in the H2O2 activation by a nonheme iron(ii) complex, Dalton Trans., 2019, 48, 17045–17051 RSC.
- F. F. Li, J. England and L. Que, Near-Stoichiometric Conversion of H2O2 to Fe(IV)=O at a Nonheme Iron(II) Center. Insights into the O-O Bond Cleavage Step, J. Am. Chem. Soc., 2010, 132, 2134–2135 CrossRef CAS PubMed.
- H. J. Forman, A. Bernardo and K. J. A. Davies, What is the concentration of hydrogen peroxide in blood and plasma?, Arch. Biochem. Biophys., 2016, 603, 48–53 CrossRef CAS PubMed.
- F. I. Adam, P. L. Bounds, R. Kissner and W. H. Koppenol, Redox Properties and Activity of Iron-Citrate Complexes: Evidence for Redox Cycling, Chem. Res. Toxicol., 2015, 28, 604–614 Search PubMed.
- D. Asik, R. Smolinski, S. M. Abozeid, T. B. Mitchell, S. G. Turowski, J. A. Spernyak and J. R. Morrow, Modulating the properties of Fe(III) macrocyclic MRI contrast agents by appending sulfonate or hydroxyl groups, Molecules, 2020, 25, 2291 Search PubMed.
- D. Asik, S. M. Abozeid, S. G. Turowski, J. A. Spernyak and J. R. Morrow, Dinuclear Fe(III) Hydroxypropyl-Appended Macrocyclic Complexes as MRI Probes, Inorg. Chem., 2021, 60, 8651–8664 CrossRef CAS PubMed.
- K. B. Jensen, C. J. McKenzie, L. P. Nielsen, J. Z. Pedersen and H. M. Svendsen, Deprotonation of low-spin mononuclear iron(III)-hydroperoxide complexes give transient blue species assigned to high-spin iron(III)-peroxide complexes, Chem. Commun., 1999, 1313–1314 RSC.
- Y. Sheng, C. S. Abelson, J. Prakash, A. Draksharapu, V. Young and L. Que, Unmasking Steps in Intramolecular Aromatic Hydroxylation by a Synthetic Nonheme Oxoiron(IV) Complex, Angew. Chem., Int. Ed., 2021, 60, 20991–20998 CrossRef CAS PubMed.
- K. M. Scott, B. L. Davenport, S. Vasylevskyi and E. L. Que, Improved Redox-Responsive Cobalt(II) 19F Magnetic Resonance Imaging Agents through Addition of Hydrogen Bond Donors, Inorg. Chem., 2025, 64, 11130–11138 CrossRef CAS PubMed.
- L. Palagi, D. L. Longo, E. Toth, C. C. Quattrocchi, A. J. van der Molen, S. Aime and E. Gianolio, Molecular and supramolecular routes to enhance Gadolinium-based contrast agents relaxivity: How far are we from the theoretical optimalvalue?, Eur. J. Med. Chem., 2025, 292, 117668 CrossRef CAS PubMed.
- N. Pal, J. Xiong, M. Jahja, S. Mahri, V. J. r. Young, Y. S. Guo, M. Swart and L. J. r. Que, A 5,000-fold increase in the HAT reactivity of a nonheme Fe=O complex simply by replacing two pyridines of the pentadentate N4Py ligand with pyrazoles, Proc. Natl. Acad. Sci. U. S. A., 2025, 122, e2414962122 CrossRef CAS PubMed.
- CCDC 2539019: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2r71vx.
| This journal is © the Partner Organisations 2026 |