 Open Access Article
Open Access ArticleSolid-state NMR and theoretical studies illuminate lanthanum borohydride C–H borylation catalysts confined within a zeolite
Jinlei Cuia,
Yuting Li a,
Da-Jiang Liu
a,
Da-Jiang Liu a,
Marco Maisa,
Jie Zhang
a,
Marco Maisa,
Jie Zhang a,
Long Qi*a,
Aaron D. Sadow
a,
Long Qi*a,
Aaron D. Sadow *ab and
Takeshi Kobayashi
*ab and
Takeshi Kobayashi *a
*a
aU.S. DOE Ames National Laboratory, Iowa State University, Ames, Iowa 50011, USA. E-mail: lqi@ameslab.gov; sadow@iastate.edu; takeshi@iastate.edu
bDepartment of Chemistry, Iowa State University, Ames, Iowa 50011, USA
First published on 20th March 2026
Abstract
Zeolite-supported single-site lanthanum borohydride catalyzes C–H borylation of hydrocarbons, while the related silica-supported complex is inactive under comparable conditions. The identical composition of support-La(BH4)2(THF)2 sites in the two materials implies that the support bestows underlying structural features onto La that are required for bond activation catalysis, yet the nature of these effects, which could include confinement effects and/or electronic modulation of the site itself, remains to be identified. We used solid-state nuclear magnetic resonance (SSNMR) spectroscopy and molecular dynamics simulations with machine-learning potentials (ML-MD) to analyze the electronic and steric effects imparted by the faujasite support on the precatalyst structure to correlate with catalytic activity. ML-MD simulations show that THF dissociates from La under the influence of confinement, leading to coordinatively unsaturated sites in the zeolite pores. Then, the La complex grafts on Brønsted acid sites (LaBAS) or isolated silanols (LaSiO) or remains physisorbed in the zeolite pores. Catalytic studies comparing compounds supported on faujasite zeolites containing or lacking BAS and/or silanols show that only the former complexes lead to active sites, ruling out confinement as the sole requirement for catalysis. The DFT calculations and ML-MD simulations also reveal that the surface-lanthanum coordination number is two (bidentate) for LaBAS, with the metal center forming long, flexible bonds to two oxygen atoms bridging Si and Al, but only one oxygen atom (monodentate) for LaSiO. The structure–activity relationship identifies confined, BAS-grafted species as active sites and provides important guidance for the design of enhanced atom-efficient catalysts.
Introduction
Surface organometallic chemistry (SOMC)—the molecular grafting of organometallic complexes onto inorganic oxide surfaces—enables the preparation of well–defined single–site heterogeneous catalysts with similar precision to homogeneous systems.1–5 Some transformations using SOMC catalysts are faster or show higher selectivity than those using homogeneous analogues, and in some cases the SOMC approach leads to reactions that are otherwise inaccessible. For example, the silica-supported (![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) SiO)3Zr–H catalyzes hydrogenolysis of carbon–carbon bonds in hydrocarbons,6 which is not observed with homogeneous catalysts.7 Alternatively, SOMC species may be less active than their homogeneous analogues.8
SiO)3Zr–H catalyzes hydrogenolysis of carbon–carbon bonds in hydrocarbons,6 which is not observed with homogeneous catalysts.7 Alternatively, SOMC species may be less active than their homogeneous analogues.8
Because the metal oxide support acts as a ligand for the SOMC species, the organometallic surface bonding influences activity. For example, zirconium hydride supported on silica–alumina, which also provides surface hydroxyls as grafting sites, is more active for hydrogenolysis.9 Similarly, WMe6 supported on silica–alumina is more active in alkane metathesis than the original silica-supported material.10 Organozirconium supported on acidic sulfated alumina is even more active in alkane hydrogenolysis than silica or silica–alumina supported analogues.11
The immobilization of organometallic molecules onto these surfaces also results in a different coordination environment from that of the molecular precursors because of ligand substitution and/or desorption and bond formation with the support. This change in the coordination environment may be one of the reasons why SOMC shows inhibited, enhanced, or new modes of reactivity. Characterization of the coordination geometry of organometallic surface sites, including their interactions with supports, is key to understanding the structures that affect reactivity and to leveraging synthetic control over structure to create more active and more selective catalysts.
Solid-state (SS)NMR spectroscopy, distinguished by its sensitivity to local environments, has become an indispensable tool for probing species at surfaces and interfaces through direct measurement of the metals.12–15 For example, Gauvin et al. analyzed the structure of a scandium amide complex supported on silica using 45Sc NMR, revealing that the amide group is substituted by a ![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) SiO– moiety, thereby grafting the complex to the surface, and that the scandium center interacts with neighboring siloxane oxygen atoms.16 NMR measurements of the central metal in supported organometallic complexes are often limited by low sensitivity due to the low concentration of the complexes and the intrinsically broad NMR spectra of the metal centers. Nevertheless, Copéret et al. used dynamic nuclear polarization (DNP)-enhanced SSNMR and high-field NMR to analyze the structure of a molybdenum dioxo complex supported on silica. They found that the structural distortion arising from a sharp
SiO– moiety, thereby grafting the complex to the surface, and that the scandium center interacts with neighboring siloxane oxygen atoms.16 NMR measurements of the central metal in supported organometallic complexes are often limited by low sensitivity due to the low concentration of the complexes and the intrinsically broad NMR spectra of the metal centers. Nevertheless, Copéret et al. used dynamic nuclear polarization (DNP)-enhanced SSNMR and high-field NMR to analyze the structure of a molybdenum dioxo complex supported on silica. They found that the structural distortion arising from a sharp ![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) SiO–Mo(O)2–OSi
SiO–Mo(O)2–OSi![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) bond angle lowers the Mo LUMO, thereby facilitating conversion to an active species.17 Analysis of supported organometallic complexes is also possible through the SSNMR measurement of organic ligands. For example, Marks et al. revealed that HfNp4 (Np = neopentyl) is chemisorbed onto sulfated alumina as HfNp2, and that activation with molecular hydrogen produces Hf(Np)H and HfH2 species that are active for the hydrogenolysis of polyolefins.11 In the latter approach, quantifying dipolar coupling between nuclei also provides information related to their internuclear distances.18 These data can be used to establish the coordination geometry of surface species, including the proximity of substituents on ligands to the central metal, to the support surface, and to other ligands. For dynamic species, however, the measurement of internuclear distances can be overestimated by molecular motions that average dipolar interactions.19 To overcome this complication, the motionally averaged dipolar interactions can be estimated from atomic trajectories predicted by molecular dynamics (MD) simulations,20 permitting the application of NMR distance measurements to dynamic species.
bond angle lowers the Mo LUMO, thereby facilitating conversion to an active species.17 Analysis of supported organometallic complexes is also possible through the SSNMR measurement of organic ligands. For example, Marks et al. revealed that HfNp4 (Np = neopentyl) is chemisorbed onto sulfated alumina as HfNp2, and that activation with molecular hydrogen produces Hf(Np)H and HfH2 species that are active for the hydrogenolysis of polyolefins.11 In the latter approach, quantifying dipolar coupling between nuclei also provides information related to their internuclear distances.18 These data can be used to establish the coordination geometry of surface species, including the proximity of substituents on ligands to the central metal, to the support surface, and to other ligands. For dynamic species, however, the measurement of internuclear distances can be overestimated by molecular motions that average dipolar interactions.19 To overcome this complication, the motionally averaged dipolar interactions can be estimated from atomic trajectories predicted by molecular dynamics (MD) simulations,20 permitting the application of NMR distance measurements to dynamic species.
The acidic sites in crystalline, high surface area zeolites are promising locations for creating highly reactive d0 or fnd0 SOMC catalysts, given the trend sulfated alumina > SiO2–Al2O3 > SiO2 in zirconium hydride-catalyzed hydrogenolysis.11,21 In an example of an organometallic species activated by grafting to a support, rare earth borohydride complexes Ln(BH4)3(THF)3 (Ln = La and Sc) supported on the faujasite zeolite HY30 (Si/Al = 30) catalyze the C–H borylation of unactivated hydrocarbons using pinacol borane (HBpin). Similar conditions using the homogeneous molecular precursor complex or silica- or alumina-supported analogues lead to no discernible borylation activity.22 Furthermore, the yield and turnovers obtained by the HY30-supported La catalyst increased 5-fold when the zeolite support reacts with Ph3SiCl, which is larger in dimensions (10.0 × 10.0 × 7.9 Å, Fig. S3) than the HY30 micropore aperture (7.4 Å), to exclusively passivate protic sites on the external surface of the zeolite prior to the grafting reaction of La(BH4)3(THF)3. The composition of the grafted complexes, formulated as support-La(BH4)2(THF)2, was determined by elemental analysis, before and after the reaction of the materials with AlMe3, which desorbs THF as THF·AlMe3 as well as BOxHy species.23 Intriguingly, the zeolite-supported lanthanum borohydride material manifests a 11B nuclear magnetic resonance (NMR) signal at −16.4 ppm in addition to a signal at −23.1 ppm, which is comparable to the chemical shift of the molecular or silica-supported lanthanum borohydride complexes (–22 ppm). 11B NMR signals in this region are generally attributed to four-coordinated anionic borate species.24,25 All of these 11B NMR signals are assigned to BH4−; however, the difference in chemical shifts suggested a distinct coordination and/or electronic environment of the effective precatalyst. The increased intensity of this unique 11B signal in more active materials led to the interpretation that the active species forms via reaction of the La complex with ![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) Si–O(H)Al
Si–O(H)Al![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) (also known as the Brønsted acid sites or BAS). The BAS form at sites where a four-coordinate anionic aluminum substitutes for a four-coordinate framework silicon (
(also known as the Brønsted acid sites or BAS). The BAS form at sites where a four-coordinate anionic aluminum substitutes for a four-coordinate framework silicon (![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) SiO)4Al–, and a positive charge (e.g., H+ in BAS) satisfies the valence of the fourth oxygen. Because the BAS result from framework silicon substitution, they are located primarily inside the zeolite micropores. Capping the protic sites on the external surface with Ph3Si should increase grafting at the BAS; however, this modification also leads to increased confinement of the lanthanum borohydride species, and this effect could contribute to generating the active species.
SiO)4Al–, and a positive charge (e.g., H+ in BAS) satisfies the valence of the fourth oxygen. Because the BAS result from framework silicon substitution, they are located primarily inside the zeolite micropores. Capping the protic sites on the external surface with Ph3Si should increase grafting at the BAS; however, this modification also leads to increased confinement of the lanthanum borohydride species, and this effect could contribute to generating the active species.
A metal-centered SSNMR approach was used to analyze the bonding of scandium borohydride grafted in HY30, which also gave an active catalyst for benzene borylation. 45Sc–27Al transfer of population in double-resonance (TRAPDOR) experiments on scandium borohydride-grafted HY30 showed that scandium is grafted within 2.7 to 3.1 Å of an aluminum center, i.e., at BAS.26 In addition, scandium borohydride grafted at BAS (ScBAS) is monodentate at the ![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) SiO(
SiO(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Al)OSi
Al)OSi![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) site and dynamic, hopping back and forth between the two O atoms, whereas species grafted on silica are rigid and form a bidentate Sc–surface coordination via one silanoate and one siloxane. As a result, the dynamic ScBAS in the zeolite pores are more electrophilic than those on silica. This increased electrophilicity could be responsible for the catalytic activity of this zeolite-grafted scandium complex. The grafting reactions at BAS or silanols, the coordination sphere, and the dynamics of lanthanum borohydride, however, could be unique for the largest trivalent lanthanide (1.16 Å 8-coordinate radius) compared to the smallest rare earth Sc (0.87 Å 8-coordinate radius),27 especially confined in the micropores.
site and dynamic, hopping back and forth between the two O atoms, whereas species grafted on silica are rigid and form a bidentate Sc–surface coordination via one silanoate and one siloxane. As a result, the dynamic ScBAS in the zeolite pores are more electrophilic than those on silica. This increased electrophilicity could be responsible for the catalytic activity of this zeolite-grafted scandium complex. The grafting reactions at BAS or silanols, the coordination sphere, and the dynamics of lanthanum borohydride, however, could be unique for the largest trivalent lanthanide (1.16 Å 8-coordinate radius) compared to the smallest rare earth Sc (0.87 Å 8-coordinate radius),27 especially confined in the micropores.
In this study, we established the three-dimensional (3D) structure of the lanthanum borohydride complex grafted at the zeolite BAS (LaBAS), employing a combination of SSNMR and theoretical calculations. We further augmented the selective grafting of La precursors (LaBAS vs. LaSiO) by reacting the zeolite support with silanes of different sizes prior to reactions to graft La(BH4)3(THF)3. The correlations between SSNMR signals and the catalytic activity in response to selected silanes further show that LaBAS is the precatalyst site, leading to the formation of active species in benzene borylation, while distance measurements reveal distinct coordination chemistry of these sites.
Results and discussion
Solid-state NMR characterization of the zeolite-supported lanthanum borohydride precatalyst
The 11B direct polarization magic-angle spinning (DPMAS) NMR spectrum of the material La(BH4)2(THF)2/Ph3Si-HY30 (La/Ph3Si-HY30), synthesized via reaction of La(BH4)3(THF)3 and triphenylsilyl-protected HY30 (Ph3Si-HY30), reveals two distinct signals situated at −16.4 ppm (B1) and −23.1 ppm (B2), as well as a broad signal from −5 to 15 ppm (Fig. 1a). The former two signals are discernible along the chemical shift axis of the 2D 11B multiple quantum (MQ)MAS spectrum (Fig. 1b, diagonal line), indicating distinct magnetic environments of the BH4-containing species, rather than arising from the second-order perturbed quadrupolar powder pattern. The absence of off-diagonal signals in the 2D 11B–11B SQ/SQ correlation spectrum (Fig. 1c) precludes the possibility that these species reside in the same complex or that there is an exchange between two sites with a correlation time longer than the NMR timescale. The broad signal spanning between −5 ppm and +15 ppm is attributed to BOxHy species resulting from the reaction between hydroxy groups and BH3, generated as a byproduct of La(BH4)3(THF)3 grafting. The −16.4 ppm signal (B1) is detected when the complex is grafted onto the aluminosilicate zeolite but not onto silica as La(BH4)2(THF)2/SiO2 (La/SiO2; shown as a grey line in Fig. 1a), suggesting that the B1 species is attributed to BH4 ligands in LaBAS. The resonance frequency of the B2 species (−23.1 ppm) closely aligns with that of the BH4 in La/SiO2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 28 or in La(BH4)3(THF)3,22 suggesting that the B2 signal may arise from multiple species on/in the zeolite, including physisorbed La(BH4)3(THF)3 and/or silanol-grafted
28 or in La(BH4)3(THF)3,22 suggesting that the B2 signal may arise from multiple species on/in the zeolite, including physisorbed La(BH4)3(THF)3 and/or silanol-grafted ![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) Si–O–La(BH4)2.
Si–O–La(BH4)2.
These assignments are corroborated by 11B{27Al} TRAPDOR29 experiments, where only the 11B NMR signal of B1 is attenuated by recoupling 11B–27Al dipolar interaction (Fig. 2), demonstrating that the B1 species exclusively resides in close proximity to aluminum. This experiment unambiguously proves that B1 is correctly assigned to BH4 ligands close to the conjugate base of the BAS. Analysis of the TRAPDOR curve quantified the 11B–27Al dipolar coupling as 100 Hz (Fig. 2). The quantitative determination of the internuclear distance based on this dipolar interaction will be discussed below after considering the dynamics of the surface species.20
To investigate the other 11B NMR signals, physisorbed species were generated on purpose by contacting La(BH4)3(THF)3 with silane-modified (via reaction with Ph3Si-Cl or Me3Si-NMe2) dealuminated zeolite (DA-HY30, Si/Al = ∼121) and HY250 (Si/Al = 250). Hydroxy site titration reveals that the OH loadings in these zeolite supports are low (Table S2). Tiny signals at 3630 and 3550 cm−1 in DRIFTS spectra are associated with BAS, indicating that both HY250 and DA-HY30 have few BAS (Fig. S2). A small signal at 3595 cm−1 in the DRIFTS spectrum of DA-HY30 is assigned to extra-framework bridging OH,30,31 resulting from the dealumination process. The modified zeolitic supports Ph3Si-DA-HY30, Ph3Si-HY250, and Me3Si-HY250 are also characterized by DNP-enhanced 29Si{1H} and 13C{1H} CPMAS NMR experiments (Fig. 3a and b). The 29Si NMR signals between −10 ppm and −20 ppm are assigned to organosilyl groups covalently bonded to the zeolite surface. The DNP-enhanced 13C CPMAS spectra (Fig. 3b) confirm the presence of capping agents, and more importantly, their DNP-enhancement (εon/off), defined as the ratio of signal intensity obtained with and without microwave irradiation, provides insight into the distribution of the capping agent inside and outside the pore. Since the DNP polarizing agent TEKPol is too large to penetrate the zeolite pores, the enhancement of the NMR signal from the internal surface is typically much lower than that observed for the solvent.32 Therefore, the substantially lower DNP enhancement observed for Me3Si-HY250 (εon/off ∼ 12) compared to that of tetrachloroethene (TCE) solvent (εon/off ∼ 80) indicates that a significant portion of Me3Si passivated the hydroxy groups within the zeolite pores. In contrast, the comparable DNP enhancements in Ph3Si-DA-HY30 and Ph3Si-HY250 (εon/off = 68 and 70, respectively) to that of TCE prove our original assumption that Ph3Si is predominantly located on the external surface.
The grafting locations of Me3Si and Ph3Si are corroborated by IR measurements (Fig. S2a). Approximately 60% of the original intensity of the SiOH peak in the DRIFTS spectrum of DA-HY30 (normalized to the framework band at 1860 cm−1) remains in the spectrum of Ph3Si-DA-HY30, which is consistent with the selective capping of Ph3SiCl on the external surface of zeolite. In contrast, the SiOH peak decreased by more than 85% in the normalized spectrum of Me3Si-HY250 compared to the signal from HY250. We interpret this difference as evidence that the smaller Me3SiCl (4.9 × 4.9 × 4.3 Å, Fig. S3), compared to Ph3SiCl, is able to react with OH groups on the zeolite external surface and within the zeolite pores. Similar external and internal capping was also confirmed by the >65% decrease in intensity of peaks associated with SiOH as well as BAS sites in the normalized spectrum of HY30 treated with Me3SiCl (using the same 1.0 mmol g−1 loading as Ph3SiCl; Fig. S2c). Because not all BAS peaks are quenched in the DRIFTS of this Me3Si-HY30, the experiment reveals that BAS in certain supercages are still open for grafting with this degree of silylation.
The assignments of LaSiO and physisorbed La(BH4)3(THF)n were supported by the 11B DPMAS spectra of the corresponding samples (Fig. 3c). In particular, a prominent signal at −23 ppm appears in the spectrum of La/Ph3Si-DA-HY30. In this sample, the external surface of the zeolite is capped by Ph3Si (as mentioned above), while silanols are formed within the micropores from the hydrolysis of the BAS. From this, LaSiO in the BAS-free supercages may be identified by a −23 ppm 11B NMR signal. In La/Me3Si-HY250, all accessible hydroxy groups are passivated (∼0.1 mmol g−1) by reacting excess loading of Me3Si-NMe2 with HY250 before physisorption of La(BH4)3(THF)3. The spectrum of this sample also shows a 11B NMR signal at −23 ppm, indicating that the physisorbed La complex gives rise to the 11B NMR signal at this position. The 11B NMR signals from −5 to 15 ppm, assigned to BH3-grafted products, are much less intense in silanol- and BAS-free materials. In contrast, the reaction of Ln(BH4)3(THF)3 and amorphous silica gives a large signal from −5 to 15 pm.28 Likely, only a small amount of free BH3 is generated because La(BH4)3(THF)3 barely grafts in these samples, and instead the majority of the −23 ppm signal in both samples comes from physisorbed La(BH4)3(THF)3. In addition, the signal at −16.4 ppm diminishes for La/DA-HY30, La/Ph3Si-DA-HY30, and La/Ph3Si-HY250, which have low loadings of BAS sites. Thus, this signal is appropriately assigned to LaBAS.
Physisorption of lanthanum borohydrides within the zeolite micropores is also illustrated by the comparison of La(BH4)3(THF)3 and (coordinated) THF concentrations, measured by solution-phase 1H NMR spectroscopy, before and after the addition of Me3Si-SiO2 (lacking micropores) or Me3Si-HY250 (Fig. S7). The intensity of the 1H NMR signals assigned to THF and BH4 is equivalent before and after the addition of nonporous, silylated silica Me3Si-SiO2 to the solution, indicating that the La(BH4)3(THF)3 complex remains in the liquid phase rather than adsorbing onto the Me3Si-SiO2. In contrast, the intensities of the THF and BH4 signals decrease by 11.8% (∼0.0022 mmol) and 16.3% (∼0.0030 mmol), respectively, after Me3Si-HY250 (∼0.027 g) is added, suggesting that La(BH4)3(THF)3 (∼0.0010 mmol) is physically trapped in the zeolite pores (and thus invisible to solution NMR measurement). The smaller decrease in the integrated intensity of the THF signals compared to that of the BH4 corresponds to ca. 27% of THF (0.0008 mmol) in La(BH4)3(THF)3 (0.0010 mmol), implying that one THF molecule dissociates when La(BH4)3(THF)3 enters the micropores.
Structural understanding of the zeolite-supported lanthanum borohydride precatalyst from theory
11B NMR parameters were calculated for surface models of the aforementioned grafted lanthanum borohydride species including LaBAS, LaSiO, the La(BH4)3(THF)2 trapped in the zeolite pores, the La(BH4)3(THF)3 precursor, and La/SiO2. The LaBAS, LaSiO and physisorbed species were modeled within micropores of a full-size periodic zeolite framework model to accurately replicate a confined environment, rather than being placed on an open surface exposed to free space. Because standard DFT methods typically fail to accurately describe the long-range van der Waals forces originating from dynamic electron correlation, the DFT-D3 Grimme dispersion correlation was included in the calculations.33,34The solvent-exclusion size of the La(BH4)3(THF)3 precursor (∼11.0 × 8.9 × 7.9 Å) prevents this species from easily diffusing into the ∼7.4 Å micropores of HY30. On the other hand, the TRAPDOR experiment on La/Ph3Si-HY30 (Fig. 2) clearly demonstrates that the La complex is in close proximity to Al and thus inside the micropores of the zeolite. To reconcile these two conflicting facts, we considered the possibility that THF dissociates as the precursor enters the pores because it is the least unfavorable ligand to dissociate (Scheme 1) and because THF apparently dissociates upon physisorption (as shown above by an increase in the THF/BH4 ratio in solution upon the addition of Me3Si-HY250).
 | ||
| Scheme 1 Ligand dissociation to enable pore adsorption. The energies shown are the total electronic energy. | ||
The calculated total electronic energy of La(BH4)3(THF)2 and THF in free space is higher by 48 kJ mol−1 than La(BH4)3(THF)3. Allowing the resulting La(BH4)3(THF)2 to infiltrate into the zeolite micropores, however, stabilizes the system by 121 kJ mol−1 (i.e., 73 kJ mol−1 vs. La(BH4)3(THF)3). We also evaluated the Gibbs free energy change associated with this series of processes. First, the enthalpy and entropy of La(BH4)3(THF)3, La(BH4)3(THF)2, and THF at 300 K were determined, from which the Gibbs energy change for THF dissociation (ΔG1) was estimated to be +24 kJ mol−1. The large size of the model system including the zeolite framework prohibited the calculations of thermal corrections to the calculated adsorption energy. Therefore, the enthalpy change for adsorption of La(BH4)3(THF)2 at 300 K was approximated by the total electronic energy change (−121 kJ mol−1), as adsorption is expected to have a negligible effect on the rigid zeolite framework and the difference in thermal contribution between 0 K (for DFT electronic energy calculations) and 300 K should be small. The entropy loss upon adsorption of La(BH4)3(THF)2 was estimated to be 267 J mol−1 K−1 by assuming complete loss of translational and rotational degrees of freedom within the zeolite pores. Given this value, the Gibbs free energy change for adsorption (ΔG2) was calculated as ΔG2 = −121 kJ mol−1 – 300 K × (−267 J mol−1 K−1) = −41 kJ mol−1. Accordingly, the overall Gibbs free energy change for the entire process was estimated as ΔG1 + ΔG2 = −17 kJ mol−1. Because the translational and rotational motions of La(BH4)3(THF)2 are unlikely to be completely quenched upon adsorption in practice, the actual entropy loss is expected to be smaller than this estimate, implying that the process in which La(BH4)3(THF)3 releases one THF ligand and diffuses into the zeolite pores is even more thermodynamically favorable.
We attribute the stabilization of the low coordinate lanthanum species to the effect of confinement (Scheme 1). Two key points emerge. First, the confinement effect is critical for lanthanum borohydride to enter and diffuse with the micropores, in terms of its size and geometry. Without confinement stabilizing the smaller La(BH4)3(THF)2 intermediate, the lanthanum species would be too large to enter the micropore. Second, confinement creates a thermodynamic driving force for La(BH4)3(THF)3 to dissociate THF, enter the micropores, and form a stable, encapsulated structure. Without this effect, the energy penalty of 48 kJ mol−1 to dissociate THF would make adsorption of La(BH4)3(THF)2 into the pores unfavorable, and diffusion of the La complex into the pores would be driven solely by the chemical potential gradient.
The computed NMR parameters for 11B of {![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) SiO(
SiO(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Al)OSi
Al)OSi![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) }La(BH4)2(THF)2 (Fig. 4a) showed an excellent agreement with those of B1 obtained experimentally (Table 1). In the modeled {κ2-
}La(BH4)2(THF)2 (Fig. 4a) showed an excellent agreement with those of B1 obtained experimentally (Table 1). In the modeled {κ2-![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) SiO(
SiO(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Al)OSi
Al)OSi![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) }La(BH4)2(THF)2 low energy structure, the two distances from the La to the oxygen of two Si–O–Al bridges (denoted as OBAS) are equal at approximately 2.7 Å. This distance is longer than the common metal–support distances of 2.1 Å.35 Thus, immobilization of the La complex by reaction with a BAS results in a bidentate coordination of the surface to La. Notably, optimization of initial structures with monodentate surface-La configurations, in which La is positioned directly above one of the OBAS, also led to the singular bidentate low-energy structure.
}La(BH4)2(THF)2 low energy structure, the two distances from the La to the oxygen of two Si–O–Al bridges (denoted as OBAS) are equal at approximately 2.7 Å. This distance is longer than the common metal–support distances of 2.1 Å.35 Thus, immobilization of the La complex by reaction with a BAS results in a bidentate coordination of the surface to La. Notably, optimization of initial structures with monodentate surface-La configurations, in which La is positioned directly above one of the OBAS, also led to the singular bidentate low-energy structure.
| Species | δ | δiso (ppm) | CQ (MHz) | ηQ | PQ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a (MHz) a (MHz) |
δQIS![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a (ppm) a (ppm) |
|
|---|---|---|---|---|---|---|---|
a δQIS (ppm) = 3/17 × [4S(S + 1) − 3]/[4S(2S − 1)]2·(PQ2/ν02) × 106, 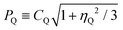 , where S is the spin quantum number of the nucleus, PQ is the second order quadrupolar effect, ν0 is the Larmor frequency of the nucleus, CQ is the quadrupolar coupling constant, and ηQ is the quadrupolar asymmetry parameter.b δ = δiso + δQIS. , where S is the spin quantum number of the nucleus, PQ is the second order quadrupolar effect, ν0 is the Larmor frequency of the nucleus, CQ is the quadrupolar coupling constant, and ηQ is the quadrupolar asymmetry parameter.b δ = δiso + δQIS. |
|||||||
| B1 | Exp. | −16.4 | −14.6 | 1.4 | −1.8 | ||
| LaBAS (Fig. 4a) | Calc. | −17.0b | −15.4 | 1.34 | 0.18 | 1.35 | −1.62 |
| B2 | Exp. | −23.1 | −21.8 | 1.2 | −1.3 | ||
| LaSiO (Fig. 4b) | Calc. | −22.9b | −21.4 | 1.27 | 0.20 | 1.28 | −1.46 |
| La, physisorbed (Fig. 4c) | Calc. | −22.5b | −21.1 | 1.25 | 0.11 | 1.25 | −1.40 |
| La on silica (Fig. 4d) | Calc. | −23.7b | −22.7 | 1.06 | 0.16 | 1.06 | −1.00 |
| La(BH4)3(THF)3 (precursor) | Exp. | −22.2 | −20.8 | 1.49 | −1.29 | ||
| Calc. | −24.3b | −22.9 | 1.23 | 0.14 | 1.23 | −1.36 | |
The 11B NMR parameters for B2 are also in good agreement with those of models where the La complex is grafted by reaction with silanols, as ![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) Si–O–La, either on the zeolite or on an amorphous silica surface (Fig. 4b and d, respectively; Table 1). In the models for both surfaces, the O–La distances are 2.3 Å, while those between the La and the nearest siloxane oxygen were all greater than 4.8 Å, indicating that bonding to silanoate results in monodentate surface-La coordination (i.e., monopodal structures), unlike {κ2-
Si–O–La, either on the zeolite or on an amorphous silica surface (Fig. 4b and d, respectively; Table 1). In the models for both surfaces, the O–La distances are 2.3 Å, while those between the La and the nearest siloxane oxygen were all greater than 4.8 Å, indicating that bonding to silanoate results in monodentate surface-La coordination (i.e., monopodal structures), unlike {κ2-![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) SiO(
SiO(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Al)OSi
Al)OSi![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) }La(BH4)2(THF)2. In the zeolite model, a lattice defect generates a pair of hydroxy groups. The La grafted to one of the hydroxy groups generated by the lattice defect is 7.9 Å away from the neighboring hydroxy, and the boron atoms of the two BH4 units are 6.7 Å and 12.3 Å away from the exposed hydroxy group. MD simulations at 300 K, described below, show that the shortest distances between these pairs are 6.6 Å, 6.2 Å, and 10.6 Å, respectively, which are too far to react to create a bipodal (
}La(BH4)2(THF)2. In the zeolite model, a lattice defect generates a pair of hydroxy groups. The La grafted to one of the hydroxy groups generated by the lattice defect is 7.9 Å away from the neighboring hydroxy, and the boron atoms of the two BH4 units are 6.7 Å and 12.3 Å away from the exposed hydroxy group. MD simulations at 300 K, described below, show that the shortest distances between these pairs are 6.6 Å, 6.2 Å, and 10.6 Å, respectively, which are too far to react to create a bipodal (![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) SiO)2La(BH4) site. The 11B NMR shift of a hypothetical dipodal structure of silanol-grafted lanthanum borohydride species, (
SiO)2La(BH4) site. The 11B NMR shift of a hypothetical dipodal structure of silanol-grafted lanthanum borohydride species, (![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) SiO–)2La(BH4)(THF)2, was also calculated. The computed 11B resonance frequency significantly shifted toward the lower frequency side ∼−31 ppm (Fig. S3), and thus this dipodal structure would be precluded. The computed 11B NMR shift for La(BH4)3(THF)2 trapped in the zeolite pores, −22.5 ppm, is also in good agreement with that of B2 (Fig. 4c and Table 1), suggesting the presence of physisorbed species.
SiO–)2La(BH4)(THF)2, was also calculated. The computed 11B resonance frequency significantly shifted toward the lower frequency side ∼−31 ppm (Fig. S3), and thus this dipodal structure would be precluded. The computed 11B NMR shift for La(BH4)3(THF)2 trapped in the zeolite pores, −22.5 ppm, is also in good agreement with that of B2 (Fig. 4c and Table 1), suggesting the presence of physisorbed species.
The long Osurface–La distances in the faujasite-micropore-confined {κ2-![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) SiO(
SiO(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Al)OSi
Al)OSi![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) }La(BH4)2(THF)2 and the electron-deficient
}La(BH4)2(THF)2 and the electron-deficient ![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) Si–O–La(BH4)2 structures illustrated in Fig. 4a and b suggest labile ligand coordination. Therefore, the surface coordination was examined in greater detail using MD simulations with machine-learning (ML) potentials (Fig. 5a). Specifically, ML-MD simulations were performed on the models shown in Fig. 4a and b, with atomic positions recorded every 10 fs over a 1 ns simulation time. During the 1 ns ML-MD simulation at 300 K, the two La–OBAS distances remain nearly equivalent, at around 2.7 Å, indicating that the surface coordination is bidentate, but still fluctuates by ±0.3 Å. The two La–OBAS remain equidistance, but slightly elongated, as the simulation temperature is increased to 500 K (Fig. 5b), indicating that the bidentate structure is maintained. For comparison, the La–Osilanol distance in LaSiO is shorter than the La–OBAS distance, with smaller fluctuations (2.3 ± 0.2 Å), and these values remain almost unchanged even with increasing temperature. These observations indicate that the La–Osilanol bond exhibits a more covalent nature. The determination that LaBAS maintains its bidentate configuration contrasts with the structure and dynamics of the lighter Sc congener, which adopts a monodentate configuration and hops between Al-adjacent oxygen sites. Most likely, the smaller size of Sc, which leads to a shorter Ln–OSi distance compared to La, leads to an unfavorable bidentate coordination geometry. These longer La–O distances also reduce steric pressure between the ligands and the pore walls, which, on the other hand, limits Sc–surface coordination. We also note that the number of ligands in the Sc(BH4)3(THF)2 precursor is five, whereas six ligands surround La(BH4)3(THF)3. While La could reasonably access higher coordination numbers than Sc, bidentate surface coordination in Sc(BH4)2(THF)2/SiO2 compared to monodentate coordination in Sc(BH4)2(THF)2/Ph3Si-HY30, suggests that the shape of the surface around a grafted metal compound greatly influences the coordination mode.
Si–O–La(BH4)2 structures illustrated in Fig. 4a and b suggest labile ligand coordination. Therefore, the surface coordination was examined in greater detail using MD simulations with machine-learning (ML) potentials (Fig. 5a). Specifically, ML-MD simulations were performed on the models shown in Fig. 4a and b, with atomic positions recorded every 10 fs over a 1 ns simulation time. During the 1 ns ML-MD simulation at 300 K, the two La–OBAS distances remain nearly equivalent, at around 2.7 Å, indicating that the surface coordination is bidentate, but still fluctuates by ±0.3 Å. The two La–OBAS remain equidistance, but slightly elongated, as the simulation temperature is increased to 500 K (Fig. 5b), indicating that the bidentate structure is maintained. For comparison, the La–Osilanol distance in LaSiO is shorter than the La–OBAS distance, with smaller fluctuations (2.3 ± 0.2 Å), and these values remain almost unchanged even with increasing temperature. These observations indicate that the La–Osilanol bond exhibits a more covalent nature. The determination that LaBAS maintains its bidentate configuration contrasts with the structure and dynamics of the lighter Sc congener, which adopts a monodentate configuration and hops between Al-adjacent oxygen sites. Most likely, the smaller size of Sc, which leads to a shorter Ln–OSi distance compared to La, leads to an unfavorable bidentate coordination geometry. These longer La–O distances also reduce steric pressure between the ligands and the pore walls, which, on the other hand, limits Sc–surface coordination. We also note that the number of ligands in the Sc(BH4)3(THF)2 precursor is five, whereas six ligands surround La(BH4)3(THF)3. While La could reasonably access higher coordination numbers than Sc, bidentate surface coordination in Sc(BH4)2(THF)2/SiO2 compared to monodentate coordination in Sc(BH4)2(THF)2/Ph3Si-HY30, suggests that the shape of the surface around a grafted metal compound greatly influences the coordination mode.
In contrast to the limited mobility of the La, the ligands exhibited highly dynamic behavior. While the two La⋯B distances of 2.6 Å remain essentially constant (Fig. 6a), one BH4 changes its position vigorously relative to the La center (Fig. 6b). Such dynamics likely reduced the 11B–27Al dipolar interaction, making 11B{27Al} TRAPDOR experiments at room temperature difficult. Rather surprisingly, one THF repeatedly dissociates from and re-coordinates to the LaBAS center (Fig. 6a), indicating that one La-bound THF is labilized within the zeolite micropore. An obvious correlation exists between the La–OTHF distance and the position of BH4 relative to the La, suggesting that the dissociation of THF induces the migration of BH4 in the coordination sphere, or vice versa. Furthermore, one THF molecule is also dynamic and labile in the zeolite-confined ![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) Si–O–La(BH4)2(THF)2 site model (LaSiO), ruling out the bidentate surface coordination model at the BAS conjugate base as the cause of THF dissociation. Unstable transients or intermediates from bulky species have been stabilized within the micropores of zeolites by the interaction with the inner surface.36–38 In the cases of LaBAS and LaSiO, the confinement effect may have enabled the formation of an undercoordinated state, albeit temporarily.
Si–O–La(BH4)2(THF)2 site model (LaSiO), ruling out the bidentate surface coordination model at the BAS conjugate base as the cause of THF dissociation. Unstable transients or intermediates from bulky species have been stabilized within the micropores of zeolites by the interaction with the inner surface.36–38 In the cases of LaBAS and LaSiO, the confinement effect may have enabled the formation of an undercoordinated state, albeit temporarily.
 | ||
| Fig. 6 La–B and La–OTHF distances (a) and azimuthal angles θ and polar angle φ representing La → B vectors (b) in the LaBAS and LaSiO site models predicted by the ML-MD simulations at 300 K. | ||
Significant dynamics were observed for BH4 also at LaSiO (Fig. 6b), but the dissociation of THF is not correlated to the BH4 dynamics, unlike the behavior of the LaBAS. The BH4 dynamics at the LaSiO may result from its less crowded coordination sphere than at LaBAS, from the flexibility of the Si–O–La moiety, or from rotation around the monodentate Osurface–La bond.
We further scrutinized the consistency between the proposed structure (Fig. 4a) and the TRAPDOR analysis (Fig. 2) using the results of the MD simulations. Dipolar coupling constants are generally inversely proportional to the cube of the internuclear distance.39 The observable effect of the dipolar interaction additionally depends on the orientation of the internuclear vector with respect to the external magnetic field and is therefore averaged out by molecular motion.40 Consequently, in dynamic systems such as surface species, relating experimentally observed dipolar coupling to internuclear distances must take into account the time-dependent fluctuations of the internuclear vector. To address this, we calculated the motionally averaged B–Al dipolar coupling by deriving time-dependent internuclear vectors from atomic trajectories obtained via MD simulations.20 At each time step i of the simulation of the LaBAS model from Fig. 4a, the Cartesian dipolar coupling tensor in the laboratory frame, DLAB, was computed based on the internuclear vector (Fig. 7a), and a rolling average of the tensor
Catalytic activity of zeolite-supported lanthanum borohydrides
We then sought to identify which of the surface species identified above are precatalyst sites for benzene borylation. As extensively established by SSNMR and ML-MD simulations above, potential sites include BAS or silanol grafted species that are confined in supercages of the zeolite micropores or on external or mesopore-exposed surfaces. Here, we again note that our previous studies revealed that blocking Ln reactions on the outer surface of the zeolite led to increased activity and implied that immobilization inside pores was responsible. The activity of the sites themselves could result from confinement and/or grafting at BAS, and both of these factors were affected by our previous protocol and both influence lanthanum borohydride coordination chemistry. To distinguish these effects, lanthanum borohydride was grafted on faujasite supports containing BAS and external and internal silanols (La/HY30), low, medium and high loadings of Al/BAS and internal silanols (La/Ph3Si-HY15, La/Ph3Si-HY30, and La/Ph3Si-HY50), external and internal silanols (La/DA-HY30), only internal silanols (La/Ph3Si-DA-HY30), and grafting-site free (La/Me3Si-HY250, i.e., physiosorbed La) and these materials were heated in benzene with HBpin under the reaction conditions given in Table 2.| La loading (mmol g−1) | HBpin | PhBpin | PhBpin | PhBpin | Initial rate constant | ||
|---|---|---|---|---|---|---|---|
| Conversion (%) | Yield (%) | Turnovers | Selectivity (%) | HBpin conversion (M h−1) | PhBpin formation (mM h−1) | ||
| a Conditions: 0.025 g precatalysts, 0.5 mL benzene, 0.15 mL HBpin (∼1.0 mmol), 120 °C, 12 h.b Not applicable. | |||||||
| La/HY30 | 0.09 | 99 | 1.6 | 10 | 1.8 | 0.27 | 7.6 |
| La/Ph3Si-HY30 | 0.07 | 95 | 7.6 | 45 | 7.9 | 0.16 | 9.4 |
| La/Ph3Si-HY15 | 0.10 | 99 | 2.1 | 8 | 2.1 | 0.20 | 4.1 |
| La/Ph3Si-HY50 | 0.06 | 12 | 0.97 | 6 | 8.1 | 0.03 | 1.2 |
| La/Ph2MeSi-HY30 | 0.06 | 98 | 6.0 | 42 | 6.1 | 0.15 | 7.9 |
| La/Me3Si-HY30 | 0.07 | 77 | 4.4 | 26 | 5.7 | 0.08 | 5.2 |
| La/SiO2 | 0.12 | <1 | ∼0 | n.a.b | n.a.b | n.a.b | n.a.b |
| La/DA-HY30 | 0.10 | 4.3 | ∼0 | n.a.b | n.a.b | n.a.b | n.a.b |
| La/Ph3Si-DA-HY30 | 0.07 | 3.7 | ∼0 | n.a.b | n.a.b | n.a.b | n.a.b |
| La/Ph3Si-HY250 | 0.08 | 5.4 | ∼0 | n.a.b | n.a.b | n.a.b | n.a.b |
| La/Me3Si-HY250 | 0.03 | 2.2 | ∼0 | n.a.b | n.a.b | n.a.b | n.a.b |
Our previous studies22,23 showed a higher yield of PhBpin from La/Ph3Si-HY30 (7.6%) compared to La/HY30 (1.6%), corresponding to a 5-fold increase in turnovers. Grafting La(BH4)3(THF)3 on Ph3Si-HY30 resulted in a slightly lower La loading (0.07 mmolLa g−1) in La/Ph3Si-HY30 compared to 0.09 mmolLa g−1 in La/HY30. The capping of the external surface of HY30 (0.86 mmol OH per g) with Ph3SiCl consumed ca. 0.4 mmol OH per g, leaving 0.46 mmol OH per g. Furthermore, the hydroxy species on the external surface are primarily silanols. Thus, the ∼50% decrease in OH upon external capping compared to only a ∼20% decrease in lanthanum loading suggested a higher ratio of lanthanum borohydride sites grafted inside the micropores to form LaBAS sites in La/Ph3Si-HY30 compared to La/HY30. We previously interpreted these results to indicate that the lanthanum grafted at BAS, now structurally characterized as LaBAS, leads to active sites for benzene borylation.
In addition to more LaBAS correlating with higher activity, there is an additional effect of Ph3SiCl capping on the catalysis. There are fewer accessible uncapped BAS in La/Ph3Si-HY30 compared to La/HY30 because more BAS are converted into LaBAS. HBpin decomposition is catalyzed by free BAS. Thus, the BAS-catalyzed side reaction is slower in La/Ph3Si-HY30 than La/HY30. Both effects lead to higher yields and higher selectivity in the capped catalyst. In this context, we point out that activity and selectivity for PhBpin depend on the HBpin conversion rate vs. the PhBpin formation rate.
In this context, we investigated BAS loading on catalysis by grafting La(BH4)3(THF)3 on Ph3Si-capped HY zeolites with high (Si/Al = 15 in La/Ph3Si-HY15) or low (Si/Al = 50 in La/Ph3Si-HY50) aluminum content. The trend in Al content in HY15, HY30, and HY50 parallels the non-quantitative relative intensity of the framework-normalized BAS signals in DRIFTS (Fig. S2e and S2f). The trend in HBpin conversion is La/Ph3Si-HY15 (0.20 M h−1) > La/Ph3Si-HY30 (0.16 M h−1) > La/Ph3Si-HY50 (0.03 M h−1). We attribute the faster HBpin conversion to higher BAS and higher LaBAS. In contrast, the selectivity for PhBpin is high for La/Ph3Si-HY50 (8.1%) and low for La/Ph3Si-HY15 (2.1%). As a result, the yield of PhBpin was lower in these samples than in La/Ph3Si-HY15. BAS provides a site for forming active species while at the same time causing the degradation of HBpin,23 and thus, a monotonic correlation was not observed between the Si/Al ratio and the PhBpin yield.
We also tested different sized capping agents at similar loading to establish the external grafting of Ph3Si compared to smaller silyl groups. The idea that Ph3SiCl, primarily grafted on the external surface of HY30, should affect catalysis differently from other silane capping locations was tested by reaction with the slightly smaller chlorosilane Ph2MeSiCl or the much smaller Me3SiCl, at similar loadings as in Ph3Si-HY30 (and in contrast to experiments of Me3Si-HY250, where Me3SiCl capped all accessible OHs). The size of Ph2MeSiCl (9.5 × 7.9 × 4.6 Å, Fig. S3) approaches that of the pore diameter, while Me3SiCl is smaller than the pore diameter as noted above. Similar OH loadings in Ph2MeSi-HY30 and Me3Si-HY30 of 0.42 and 0.39 mmol g−1, respectively, indicate a similar degree of capping. The La loadings in the catalyst samples La/Ph2MeSi-HY30 (0.06 mmol La per g) and La/Me3Si-HY30 (0.07 mmol g−1) were also similar to La/Ph3Si-HY30. 11B SSNMR spectra of these materials all include the B1 signal (Fig. S4) assigned to LaBAS. The catalytic activities of La/Ph2MeSi-HY30 (42 turnovers) and La/Ph3Si-HY30 (45 turnovers) were comparable. These experiments support the idea that SiOH defects on the external surface and in mesopores of HY30 are effectively capped by Ph3SiCl since the smaller Ph2MeSiCl appears to graft similarly. La/Me3Si-HY30, in contrast, gave only 26 turnovers. Since Me3SiCl can cap both BAS in micropores and silanols on the external surface, lower HBpin conversion and fewer turnovers for PhBpin formation indicate that fewer BAS are accessible to HBpin and a smaller fraction of La sites are active.
The silanol-rich and BAS-poor zeolite DA-HY30 was studied to probe the C–H borylation activity of silanol-grafted La sites on and confined within the zeolite. 11B SSNMR spectra of La/DA-HY30 and its externally silylated derivative, La/Ph3Si-DA-HY30, did not contain detectable signals at −16.4 ppm. A catalytic test of this material produced HBpin, with <5% conversion; PhBpin was not detected in the reaction mixture by 11B NMR or GC-MS. This material shows that silanol-grafted ![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) SiO–La(BH4)2(THF)n species, inside and outside micropores of the zeolite, are not catalytically active for benzene borylation or HBpin decomposition under these conditions.
SiO–La(BH4)2(THF)n species, inside and outside micropores of the zeolite, are not catalytically active for benzene borylation or HBpin decomposition under these conditions.
The selective external silylation of DA-HY30, to favor micropore- and silanol-grafted lanthanum species, also decreased the La loading from 0.10 mmolLa g−1 in La/DA-HY30 to 0.07 mmolLa g−1 in La/Ph3Si-DA-HY30. Unlike the La/Ph3Si-HY30-improved PhBpin yield compared to La/HY30, La/Ph3Si-DA-HY30 also gave <5% HBpin conversion and PhBpin was not detected. Thus, confined, silanol-grafted lanthanum borohydride is also insufficient to generate active sites. For the same reasons, 0.08 mmolLa g−1 La/Ph3Si-HY250 is catalytically inactive under these conditions, giving <5% HBpin decomposition. Capping HY250 with Me3SiCl further reduced accessible silanols and BAS for grafting. The La loading in La/Me3Si-HY250 was only 0.03 mmolLa g−1, with La mostly in the form of physisorbed species inside the faujasite micropores, and catalytic activity was not observed. These results indicate that confinement of Osurface–La(BH4)2(THF)2 or even physisorbed La(BH4)3(THF)2 is not sufficient to generate active sites, and LaBAS is an essential species for C–H borylation under these conditions.
Conclusion
This study reveals that confinement effects and surface coordination act synergistically to create uniquely active LaBAS for benzene borylation. First, confinement generates the thermodynamic and geometric conditions required for lanthanum borohydride to enter the micropores of the faujasite zeolite. Without confinement to create the driving force for THF to dissociate from La(BH4)3(THF)3, coupled to the formation of an appropriately sized La(BH4)3(THF)2 intermediate, lanthanum borohydride could not physisorb into the micropores and graft on internal silanols or BAS. Second, lanthanum coordinates to the conjugate base of BAS and to surface silanoates via bidentate or monodentate modes, respectively.We demonstrated that the confined silanoate-coordinated LaSiO species are inactive in benzene borylation, even though their coordination number is lower than the bidentate surface-coordinated LaBAS species. This surface coordination mode is important to form the active catalyst, but not essential, since the smaller Sc center bonds to BAS in a monodentate fashion and also leads to catalytically active species. In fact, scandium forms an inactive, bidentate surface coordinated species on silica. On the other hand, the O–La distance of 2.7 Å in LaBAS determined by a combination of SSNMR and ML-MD simulations is longer than the 2.3 Å O–La distance in LaSiO. That is, the weak surface coordination in LaBAS leads to an undercoordinated species and is likely a significant contributor to its reactivity. The high catalytic activity associated with these sites also correlates with the dynamic coordination of THF to lanthanum under pore confinement.
Experimental
Catalyst preparation
Structural characterization
Conflicts of interest
There are no conflicts to declare.Data availability
The data that support the findings of this study are available at Datashare, an open-access repository at Iowa State University at https://doi.org/10.25380/iastate.31258684.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5qi02586a.
Acknowledgements
This research was supported by the U.S. Department of Energy, Office of Science, Office of Basic Energy Sciences, Chemical Sciences, Geosciences, and Biosciences Division, Catalysis Science program. MD simulations (D.-J. Liu) were supported under the Computational and Theoretical (CTC) project. Ames National Laboratory is operated for the U.S. Department of Energy by Iowa State University under Contract No. DEAC02-07CH11358.References
- M. K. Samantaray, V. D’Elia, E. Pump, L. Falivene, M. Harb, S. O. Chikh, L. Cavallo and J.-M. Basset, The Comparison between Single Atom Catalysis and Surface Organometallic Catalysis, Chem. Rev., 2020, 120, 734–813 CrossRef CAS PubMed.
- M. K. Samantaray, E. Pump, A. Bendjeriou-Sedjerari, V. D'Elia, J. D. A. Pelletier, M. Guidotti, R. Psaro and J.-M. Basset, Surface Organometallic Chemistry in Heterogeneous Catalysis, Chem. Soc. Rev., 2018, 47, 8403–8437 RSC.
- C. Copéret, A. Comas-Vives, M. P. Conley, D. P. Estes, A. Fedorov, V. Mougel, H. Nagae, F. Núñez-Zarur and P. A. Zhizhko, Surface Organometallic and Coordination Chemistry toward Single-Site Heterogeneous Catalysts: Strategies, Methods, Structures, and Activities, Chem. Rev., 2016, 116, 323–421 CrossRef PubMed.
- C. Copéret, M. Chabanas, R. P. Saint-Arroman and J.-M. Basset, Homogeneous and Heterogeneous Catalysis: Bridging the Gap through Surface Organometallic Chemistry, Angew. Chem., Int. Ed., 2003, 42, 156–181 CrossRef PubMed.
- B. C. Gates, Mononuclear Metal Complex Catalysts on Supports: Foundations in Organometallic and Surface Chemistry and Insights into Structure, Reactivity, and Catalysis, Chem. Sci., 2024, 15, 16821–16843 RSC.
- C. Lecuyer, F. Quignard, A. Choplin, D. Olivier and J. M. Basset, Surface Organometallic Chemistry on Oxides - Selective Catalytic Low-Temperature Hydrogenolysis of Alkanes by a Highly Electrophilic Zirconium Hydride Complex Supported on Silica, Angew. Chem., Int. Ed., 1991, 30, 1660–1661 CrossRef.
- J. Corker, F. Lefebvre, C. Lecuyer, V. Dufaud, F. Quignard, A. Choplin, J. Evans and J. M. Basset, Catalytic Cleavage of the C-H and C-C Bonds of Alkanes by Surface Organometallic Chemistry: An Exafs and Ir Characterization of a Zr-H Catalyst, Science, 1996, 271, 966–969 CrossRef CAS.
- N. Eedugurala, Z. Wang, K. Yan, K. C. Boteju, U. Chaudhary, T. Kobayashi, A. Ellern, I. I. Slowing, M. Pruski and A. D. Sadow, B-Sih-Containing Tris(Silazido) Rare-Earth Complexes as Homogeneous and Grafted Single-Site Catalyst Precursors for Hydroamination, Organometallics, 2017, 36, 1142–1153 CrossRef CAS.
- V. Dufaud and J.-M. Basset, Catalytic Hydrogenolysis at Low Temperature and Pressure of Polyethylene and Polypropylene to Diesels or Lower Alkanes by a Zirconium Hydride Supported on Silica-Alumina: A Step toward Polyolefin Degradation by the Microscopic Reverse of Ziegler–Natta Polymerization, Angew. Chem., Int. Ed., 1998, 37, 806–810 CrossRef CAS PubMed.
- M. K. Samantaray, R. Dey, E. Abou-Hamad, A. Hamieh and J.-M. Basset, Effect of Support on Metathesis of n-Decane: Drastic Improvement in Alkane Metathesis with WMe5 Linked to Silica–Alumina, Chem. – Eur. J., 2015, 21, 6100–6106 CrossRef CAS PubMed.
- W. C. Edenfield, A. H. Mason, Q. H. Lai, A. Agarwal, T. Kobayashi, Y. Kratish and T. J. Marks, Rapid Polyolefin Plastic Hydrogenolysis Mediated by Single-Site Heterogeneous Electrophilic/Cationic Organo-Group Iv Catalysts, ACS Catal., 2023, 14, 554–565 CrossRef.
- R. Lang, X. Du, Y. Huang, X. Jiang, Q. Zhang, Y. Guo, K. Liu, B. Qiao, A. Wang and T. Zhang, Single-Atom Catalysts Based on the Metal–Oxide Interaction, Chem. Rev., 2020, 120, 11986–12043 CrossRef CAS PubMed.
- J. Li, M. F. Stephanopoulos and Y. Xia, Introduction: Heterogeneous Single-Atom Catalysis, Chem. Rev., 2020, 120, 11699–11702 CrossRef CAS PubMed.
- S. K. Kaiser, Z. Chen, D. F. Akl, S. Mitchell and J. Pérez-Ramírez, Single-Atom Catalysts across the Periodic Table, Chem. Rev., 2020, 120, 11703–11809 CrossRef CAS PubMed.
- A. Q. Wang, J. Li and T. Zhang, Heterogeneous Single-Atom Catalysis, Nat. Rev. Chem., 2018, 2, 65–81 CrossRef CAS.
- T. Vancompernolle, X. Trivelli, L. Delevoye, F. Pourpoint and R. M. Gauvin, On the Use of Solid-State 45Sc NMR for Structural Investigations of Molecular and Silica-Supported Scandium Amide Catalysts, Dalton Trans., 2017, 46, 13176–13179 RSC.
- Z. J. Berkson, R. Zhu, C. Ehinger, L. Lätsch, S. P. Schmid, D. Nater, S. Pollitt, O. V. Safonova, S. Björgvinsdóttir and A. B. Barnes, et al., Active Site Descriptors from 95Mo NMR Signatures of Silica-Supported Mo-Based Olefin Metathesis Catalysts, J. Am. Chem. Soc., 2023, 145, 12651–12662 CrossRef CAS.
- R. Jabbour, M. Renom-Carrasco, K. W. Chan, L. Völker, P. Berruyer, Z. R. Wang, C. M. Widdifield, M. Lelli, D. Gajan and C. Copéret, et al., Multiple Surface Site Three-Dimensional Structure Determination of a Supported Molecular Catalyst, J. Am. Chem. Soc., 2022, 144, 10270–10281 CrossRef CAS PubMed.
- Y. Ishii, T. Terao and S. Hayashi, Theory and Simulation of Vibrational Effects on Structural Measurements by Solid-State Nuclear Magnetic Resonance, J. Chem. Phys., 1997, 107, 2760–2774 CrossRef CAS.
- T. Kobayashi, D. J. Liu and F. A. Perras, Spatial Arrangement of Dynamic Surface Species from Solid-State NMR and Machine Learning-Accelerated MD Simulations, Chem. Commun., 2022, 58, 13939–13942 RSC.
- V. R. Dufaud and J. M. Basset, Catalytic Hydrogenolysis at Low Temperature and Pressure of Polyethylene and Polypropylene to Diesels or Lower Alkanes by a Zirconium Hydride Supported on Silica-Alumina: A Step toward Polyolefin Degradation by the Microscopic Reverse of Ziegler-Natta Polymerization, Angew. Chem., Int. Ed., 1998, 37, 806–810 CrossRef CAS PubMed.
- Y. Li, U. Kanbur, J. Cui, G. Wang, T. Kobayashi, A. D. Sadow and L. Qi, Supported Lanthanum Borohydride Catalyzes CH Borylation inside Zeolite Micropores, Angew. Chem., Int. Ed., 2022, 61, e202117394 CrossRef CAS PubMed.
- Y. Li, T. Kobayashi, D. M. Meira, L. An, L. Qi and A. D. Sadow, Trimethylaluminum Activates Zeolite-Confined Lanthanum Borohydrides to Enhance Catalytic C–H Borylation, J. Am. Chem. Soc., 2025, 147, 12574–12586 CrossRef CAS PubMed.
- Z. Lodziana, P. Blonski, Y. G. Yan, D. Rentsch and A. Remhof, NMR Chemical Shifts of 11B in Metal Borohydrides from First-Principle Calculations, J. Phys. Chem. C, 2014, 118, 6594–6603 Search PubMed.
- S. Hermanek, B-11 NMR-Spectra of Boranes, Main-Group Heteroboranes, and Substituted Derivatives - Factors Influencing Chemical-Shifts of Skeletal Atoms, Chem. Rev., 1992, 92, 325–362 Search PubMed.
- S. A. Southern, Y. Li, D.-J. Liu, A. D. Sadow, L. Qi and F. A. Perras, Enhanced Activity from Coordinatively Unsaturated and Dynamic Zeolite-Bound Organoscandium Species, ACS Catal., 2024, 14, 9440–9451 CrossRef CAS.
- R. D. Shannon, Revised Effective Ionic-Radii and Systematic Studies of Interatomic Distances in Halides and Chalcogenides, Acta Crystallogr., Sect. A, 1976, 32, 751–767 CrossRef.
- N. Ajellal, G. Durieux, L. Delevoye, G. Tricot, C. Dujardin, C. M. Thomas and R. M. Gauvin, Polymerization of Racemic B-Butyrolactone Using Supported Catalysts: A Simple Access to Isotactic Polymers, Chem. Commun., 2010, 46, 1032–1034 RSC.
- E. R. H. Vaneck, R. Janssen, W. Maas and W. S. Veeman, A Novel Application of Nuclear Spin-Echo Double-Resonance to Aluminophosphates and Aluminosilicates, Chem. Phys. Lett., 1990, 174, 428–432 CrossRef CAS.
- S. M. T. Almutairi, B. Mezari, G. A. Filonenko, P. Magusin, M. S. Rigutto, E. A. Pidko and E. J. M. Hensen, Influence of Extraframework Aluminum on the Bronsted Acidity and Catalytic Reactivity of Faujasite Zeolite, ChemCatChem, 2013, 5, 452–466 CrossRef CAS.
- T. W. Beutel, A. M. Willard, C. Lee, M. S. Martinez and R. Dugan, Probing External Bronsted Acid Sites in Large Pore Zeolites with Infrared Spectroscopy of Adsorbed 2,4,6-Tri-Tert-Butylpyridine, J. Phys. Chem. C, 2021, 125, 8518–8532 CrossRef CAS.
- L. Lakiss, C. Kouvatas, J. P. Gilson, V. Valtchev, S. Mintova, C. Fernandez, R. Bedard, S. Abdo and J. Bricker, Atomic-Insight into Zeolite Catalyst Forming-an Advanced NMR Study, J. Phys. Chem. C, 2021, 125, 20028–20034 CrossRef CAS.
- S. Grimme, J. Antony, S. Ehrlich and H. Krieg, A Consistent and Accurate Ab Initio Parametrization of Density Functional Dispersion Correction (DFT-D) for the 94 Elements H-Pu, J. Chem. Phys., 2010, 132, 154104 CrossRef PubMed.
- S. Grimme, S. Ehrlich and L. Goerigk, Effect of the Damping Function in Dispersion Corrected Density Functional Theory, J. Comput. Chem., 2011, 32, 1456–1465 CrossRef CAS PubMed.
- O. Alexeev and B. C. Gates, EXAFS Characterization of Supported Metal-Complex and Metal-Cluster Catalysts Made from Organometallic Precursors, Top. Catal., 2000, 10, 273–293 CrossRef CAS.
- E. G. Derouane, Zeolites as Solid Solvents, J. Mol. Catal. A: Chem., 1998, 134, 29–45 CrossRef CAS.
- G. Noh, Z. C. Shi, S. I. Zones and E. Iglesia, Isomerization and B-Scission Reactions of Alkanes on Bifunctional Metal Acid Catalysts: Consequences of Confinement and Diffusional Constraints on Reactivity and Selectivity, J. Catal., 2018, 368, 389–410 CrossRef CAS.
- S. P. Crossley, D. E. Resasco and G. L. Haller, Clarifying the Multiple Roles of Confinement in Zeolites: From Stabilization of Transition States to Modification of Internal Diffusion Rates, J. Catal., 2019, 372, 382–387 CrossRef CAS.
- M. Ernst, S. Bush, A. C. Kolbert and A. Pines, Second-Order Recoupling of Chemical-Shielding and Dipolar-Coupling Tensors under Spin Decoupling in Solid-State NMR, J. Chem. Phys., 1996, 105, 3387–3397 CrossRef.
- P. Schanda, M. Huber, J. Boisbouvier, B. H. Meier and M. Ernst, Solid-State NMR Measurements of Asymmetric Dipolar Couplings Provide Insight into Protein Side-Chain Motion, Angew. Chem., Int. Ed., 2011, 50, 11005–11009 CrossRef CAS PubMed.
- J. N. Louwen, S. Simko, K. Stanciakova, R. E. Bulo, B. M. Weckhuysen and E. T. C. Vogt, Role of Rare Earth Ions in the Prevention of Dealumination of Zeolite Y for Fluid Cracking Catalysts, J. Phys. Chem. C, 2020, 124, 4626–4636 CrossRef CAS.
- A. Comas-Vives, Amorphous SiO2 Surface Models: Energetics of the Dehydroxylation Process, Strain, Ab Initio Atomistic Thermodynamics and IR Spectroscopic Signatures, Phys. Chem. Chem. Phys., 2016, 18, 7475–7482 RSC.
- P. Giannozzi, S. Baroni, N. Bonini, M. Calandra, R. Car, C. Cavazzoni, D. Ceresoli, G. L. Chiarotti, M. Cococcioni and I. Dabo, et al., Quantum Espresso: A Modular and Open-Source Software Project for Quantum Simulations of Materials, J. Phys.: Condens. Matter, 2009, 21, 395502 CrossRef PubMed.
- P. Giannozzi, O. Andreussi, T. Brumme, O. Bunau, M. B. Nardelli, M. Calandra, R. Car, C. Cavazzoni, D. Ceresoli and M. Cococcioni, et al., Advanced Capabilities for Materials Modelling with Quantum Espresso, J. Phys.: Condens. Matter, 2017, 29, 465901 CrossRef CAS PubMed.
- J. P. Perdew, K. Burke and M. Ernzerhof, Generalized Gradient Approximation Made Simple, Phys. Rev. Lett., 1996, 77, 3865–3868 CrossRef CAS PubMed.
- P. Gianluca, M. Antimo, C. E. Ivano, M. Nicolas, P. Elsa and M. Nichola, A Standard Solid State Pseudopotential (SSSP) Library Optimized for Precision and Efficiency, https://www.materialscloud.org/discover/sssp/table/efficiency.
- C. J. Pickard and F. Mauri, All-Electron Magnetic Response with Pseudopotentials: NMR Chemical Shifts, Phys. Rev. B: Condens. Matter Mater. Phys., 2001, 63, 245101 CrossRef.
- D. Ceresoli, Pseudopotentials, https://sites.google.com/site/dceresoli/pseudopotentials.
- H. J. Monkhorst and J. D. Pack, Special Points for Brillouin-Zone Integrations, Phys. Rev. B: Solid State, 1976, 13, 5188–5192 CrossRef.
- G. Kresse and J. Furthmuller, Efficient Iterative Schemes for Ab Initio Total-Energy Calculations Using a Plane-Wave Basis Set, Phys. Rev. B: Condens. Matter Mater. Phys., 1996, 54, 11169–11186 CrossRef CAS PubMed.
- G. Kresse and J. Furthmuller, Efficiency of Ab initio Total Energy Calculations for Metals and Semiconductors Using a Plane-Wave Basis Set, Comput. Mater. Sci., 1996, 6, 15–50 CrossRef CAS.
- L. F. Zhang, J. Q. Han, H. Wang, R. Car and E. Weinan, Deep Potential Molecular Dynamics: A Scalable Model with the Accuracy of Quantum Mechanics, Phys. Rev. Lett., 2018, 120, 143001 CrossRef CAS PubMed.
- D. J. Liu and J. W. Evans, Fluorine Spillover for Ceria- vs Silica-Supported Palladium Nanoparticles: A MD Study Using Machine Learning Potentials, J. Chem. Phys., 2023, 159, 024101 CrossRef CAS PubMed.
- A. H. Larsen, J. J. Mortensen, J. Blomqvist, I. E. Castelli, R. Christensen, M. Dulak, J. Friis, M. N. Groves, B. Hammer and C. Hargus, et al., The Atomic Simulation Environment-A Python Library for Working with Atoms, J. Phys.: Condens. Matter, 2017, 29, 273002 CrossRef PubMed.
| This journal is © the Partner Organisations 2026 |







