Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Lewis superacids for catalytic reductions of stronger element–oxygen double bonds with hydrosilanes
Daniel
Franz
 ,
Thomas R.
Frost
,
Thomas R.
Frost
 ,
Sebastian
Stigler
,
Sebastian
Stigler
 and
Shigeyoshi
Inoue
and
Shigeyoshi
Inoue
 *
*
TUM School of Natural Sciences, Department of Chemistry, Catalysis Research Center and Institute of Silicon Chemistry, Technical University of Munich, Garching bei München, Germany. E-mail: s.inoue@tum.de
First published on 15th January 2026
Abstract
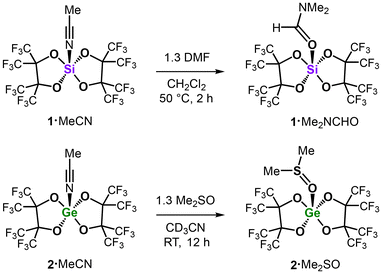
The main-group Lewis superacid complexes (pinF)2Si·MeCN (1·MeCN) and (pinF)2Ge·MeCN (2·MeCN) were successfully applied as promoters in the catalytic reduction of phosphine oxides (e.g., Me3PO, Bu3PO, and Ph3PO), a sulfoxide (i.e., Me2SO), and an amide (i.e., Me2NCHO) to furnish the respective phosphines, dimethyl sulfide, and trimethylamine using silanes (e.g., PhSiH3 and (EtO)3SiH) as hydrogen sources (pinF = perfluoropinacolato). These substrates target difficult to reduce representatives of oxo compounds in comparison with, for example, the ketones or aldehydes often targeted in such types of catalytic reductions. As benchmark promoters, we also studied B(C6F5)3 and HNTf2 as reference (soft) Lewis superacid and Brønsted superacid, respectively (Tf = SO2CF3). Among the combinations of (pre)catalyst, substrate, and reducing agent investigated, the silicon complex 1·MeCN turned out to be the most versatile system, being the by far most potent (DMSO) or just slightly underperforming (R3PO and DMF) promoter. Moreover, the hitherto undescribed Lewis acid base adducts 1·Me2NCHO and 2·Me2SO were synthesized, isolated, and structurally investigated using NMR spectroscopy and single-crystal XRD analysis.
Introduction
In the wake of Stephan's groundbreaking report on catalytic dihydrogenation with frustrated Lewis pairs and Power's pioneering article on the resemblance of low valent main-group elements with transition metals, the study of s- and p-block complexes for homogeneous catalysis markedly intensified.1 Stephan's, as well as Power's approaches, exploited the high reactivity profile of low-coordinate main group metal(loid) atoms. Similarly, the pronounced Lewis acidity of silyl cations derives not only from their coulombic attraction but also from their low-coordinated state. Consequently, silyl cations have evolved after Lambert's seminal finding in 1993 into a vast field of catalytic applications.2 As a more recent development, the use of strongly electron-withdrawing ligands was found to confer outstanding Lewis acidity to higher-coordinated and uncharged silicon complexes, as well, and the respective compounds were successfully applied in molecular catalysis.3 Notably, higher-coordinated cationic silicon complexes with outstanding Lewis acidity have also been reported.4For the classification of Lewis acids, Krossing coined the term ‘Lewis superacid’ as a category of complexes with a larger fluoride ion affinity (FIA) in the gas phase than antimony pentafluoride.5 Some controversy exists about the limitation of this threshold to a theoretical FIA, or whether experiment and theory need to coincide. More recently, Greb extended this concept to the definition of ‘soft Lewis superacids’, that is, molecular Lewis acids that have a larger hydride ion affinity (HIA) than B(C6F5)3 in the gas phase.3b Perhalogenated species of the chelate fashioned catecholato ligand and its derivatives have been established as particularly suitable ligands for various Si and Ge complexes that meet the criteria for Lewis superacidity (selected species A–C, Fig. 1).6 In 2021, we reported the silicon complex 1·MeCN, which bears the bidentate and strongly electron-withdrawing perfluoropinacolato ligand and abstracts fluoride from AgSbF6 in acetonitrile solution (Fig. 1).7 Also, the heavier germanium congener 2·MeCN was described, which exhibited larger FIA and HIA values (Fig. 1).8 These compounds promoted catalytic conversions such as hydrodefluorination and hydrosilylation (i.e., reduction) of double bonds, as well as polyether degradation.7–9 In a detailed fashion we had investigated the catalytic hydrosilylation (i.e., reduction) of ketones and aldehydes with 1·MeCN.7 In this work, we focus on more difficult to reduce element oxygen double bonds as found in phosphine oxides, sulfoxides, and amides using 1·MeCN and 2·MeCN as promoters. Moreover, we compare the catalytic activities of these with the ubiquitous benchmark Lewis acid B(C6F5)3 and the strong Brønsted acid HNTf2 (Tf = SO2CF3).
Results and discussion
Phosphine oxide reductions
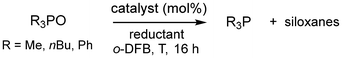
Phosphines are used in many synthetic applications because the formation of the strong phosphorus–oxygen double bond often drives a reaction, as seen in the Wittig and Mitsunobu reactions.10 The reduction of phosphine oxides back to the corresponding phosphines is attractive in the light of waste material recycling. Catalytic amounts of titanium(IV) alkoxy compounds were reported by Lawrence to catalyze the reduction of tertiary phosphine oxides by silanes.11 Beller used copper halides and copper(II) triflate to facilitate the reduction of secondary and tertiary phosphine oxides with organosilanes.12 Notably, non-catalytic methods for the reduction of tertiary phosphine oxides using highly reactive silanes (e.g., PhSiH3, Cl3SiH, and Si2Cl6) were reported about 60 years ago and commonly required harsh reaction conditions.13 Other procedures that work without a catalyst typically implement hydroboranes or aluminum hydrides and the reader is referred to the respective reviews for details.14 Prominent examples for phosphine oxide reduction by main-group promoters rely on potent Lewis acids of boron (e.g., B(C6F5)3, (2-Cl-C6H4)2BOH) or highly Lewis acidic phosphonium cations (Fig. 2, rows 1 and 2).15 More recently, Greb described the implementation of the silicon Lewis superacid B in the reduction of Et3PO and Ph3PO at 100 °C in toluene using 3 eq. of PhSiH3 as a reducing agent (Fig. 2, row 3).6c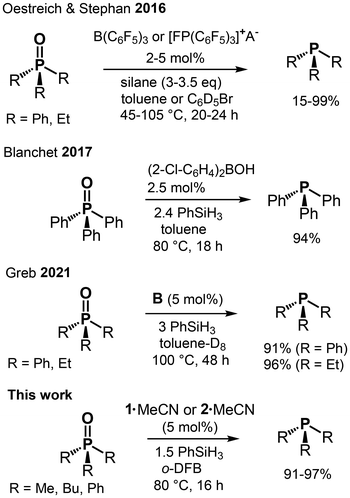 | ||
| Fig. 2 Selected catalytic tertiary phosphine oxide reductions to phosphine using main group complexes (A− = B(C6F5)4−; silane = PhSiH3, (EtO)3SiH; o-DFB = ortho-difluorobenzene). | ||
Notably, the experimental assessment of Lewis acidity is commonly conducted by the use of phosphine oxides and 31P NMR analysis, that is, the Gutmann–Beckett method.16 Accordingly, many Lewis superacids have been probed for complexation of Et3PO, but the further conversion of these complexes, if formed, has often not been investigated.
In the light of Greb's result, we set out to probe the catalytic activity of 1·MeCN and 2·MeCN for phosphine oxide reduction using Bu3PO, Me3PO, and Ph3PO as substrates. The ubiquitous Et3PO was left out due to its higher pricing and lower convenience (i.e., waxy nature of the solid) which renders it less suitable for large-scale applications. As expected, the use of PhSiH3 resulted in the production of phosphine and the results of our catalytic conversions are shown in Table 1. The conversion of Bu3PO with 1.5 eq. of PhSiH3 and 1·MeCN or 2·MeCN as a promoter (5 mol%) furnished the respective phosphine in a near-quantitative fashion (97% or 96%) after 16 h in ortho-difluorobenzene (o-DFB) at 80 °C (Table 1, entries 1 and 2). Decreasing the catalyst load to 1 mol% 2·MeCN resulted in a markedly lower yield (75%), which might possibly be compensated by a longer reaction time (Table 1, entry 3). The alternative reducing agents Et3SiH (5.5 eq.) and pinacolborane (HBpin, 4.5 eq.) were also tested and resulted in practically no formation of phosphine (Et3SiH) and high phosphine yield (HBpin, 89%), respectively, which agrees with the expected deoxyhydrogenation activity of these compounds (Table 1, entries 4 and 5). In the context of applying the germanium complex 2·MeCN, we had described its reaction to a germylene species of the type pinFGe (pinF = ((CF3)2CO)2) upon reaction with Et3SiH.8 Though this reactivity is mostly favored in the absence of electron–pair donors, it may gain relevance at elevated temperature and in the presence of excess hydrosilane. Thus, we tested the germylene adduct (pinFGe)2·(1,4-dioxane) ((3)2·diox) as a promoter in catalytic phosphine oxide reduction, as well, after we had synthesized it independently as described in the literature.8 In fact, the use of (3)2·diox (5 mol% loading in Ge) afforded markedly lower yield (52%) of Bu3P in combination with PhSiH3 (1.5 eq.) in comparison with the reactions with 1·MeCN and 2·MeCN (Table 1, entries 6, 7, cf. entries 1 and 2). We assume that the reduced Lewis acidity of the germylene, as compared to the germane, accounts for the decreased activity of the former. Accordingly, the partial conversion of 2·donor into 3·donor might hamper the catalytic performance of 2·MeCN in the course of the reaction and come into effect for longer reaction times or lower catalyst loadings, as we had observed (Table 1, entry 3). For comparing our Lewis acids with a strong Brønsted acid, we used HN(SO2CF3)2 as a (pre)catalyst, and the reduction of Bu3PO proceeded similarly to that of our Si and Ge promoters (Table 1, entry 8). One must note that in the absence of any promoter, the phosphine oxide was also reduced to a non-negligible degree (20%, Table 1, entry 9), which agrees with the literature reports.17 We also probed Me3PO as the substrate to find complete consumption of the oxide and formation of Me3P (91%, the non-quantitative detected yield is attributed to partial loss of volatile Me3P to the headspace) after just 5 h at 80 °C and, thus, considerably shorter than the ca. 16 h required for the reduction of the bulkier Bu3PO under very similar conditions (Table 1, entry 10 vs. 1). In stark contrast, Ph3PO was more difficult to reduce as we found only 69% of the respective phosphine after the full 16 h of reaction period (Table 1, entry 11). For comparison, Oestreich and Stephan reported near-quantitative conversion of Ph3PO with the use of B(C6F5)3 (5 mol%) in toluene at 105 °C over 20 h.15a
| R | Cat. (mol%) | Red. (eq.) | T [°C] | Yielda [%] | |
|---|---|---|---|---|---|
| (3)2·diox = (pinFGe)2·(1,4-dioxane), pinF = perfluoropinacolato.a Yield determined using 31P{1H} NMR by the addition of tris(2,4-di-tert-butylphenyl) phosphite as an internal standard (after the conversion).b Yield determined using 1H NMR by the addition of 4,4′-di-tert-butyl-biphenyl as an internal standard (at conversion start). | |||||
| 1 | nBu | 1·MeCN (5) | PhSiH3 (1.5) | 80 | 97 |
| 2 | nBu | 2·MeCN (5) | PhSiH3 (1.5) | 80 | 96 |
| 3 | nBu | 2·MeCN (1) | PhSiH3 (1.5) | 80 | 75 |
| 4 | nBu | 2·MeCN (5) | Et3SiH (5.5) | 110 | <1 |
| 5 | nBu | 2·MeCN (5) | HBpin (4.5) | 80 | 89 |
| 6 | nBu | (3)2·diox (2.5) | PhSiH3 (1.5) | 80 | 52 |
| 7 | nBu | (3)2·diox (2.5) | Et3SiH (5) | 120 | <1 |
| 8 | nBu | HNTf2 (5) | PhSiH3 (1.5) | 80 | 99 |
| 9 | nBu | None | PhSiH3 (1.5) | 80 | 20 |
| 10 | Me | 1·MeCN (5) | PhSiH3 (1.5) | 80 | 91b |
| 11 | Ph | 2·MeCN (5) | PhSiH3 (1.5) | 80 | 69 |
Dimethylsulfoxide reductions
With our successful reduction of the P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O double bond as a starting point, we set out to apply similar conditions to the reduction of S
O double bond as a starting point, we set out to apply similar conditions to the reduction of S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O double bonds. Me2SO (DMSO) marks one of the most widespread sulfoxides due to its application as a polar-aprotic solvent. Generally, sulfur possesses a higher electronegativity and a smaller atomic radius in comparison with phosphorus. These attributes will affect bond polarization and π-interaction with oxygen and, in consequence, grant a lesser degree of zwitterionic character to the S
O double bonds. Me2SO (DMSO) marks one of the most widespread sulfoxides due to its application as a polar-aprotic solvent. Generally, sulfur possesses a higher electronegativity and a smaller atomic radius in comparison with phosphorus. These attributes will affect bond polarization and π-interaction with oxygen and, in consequence, grant a lesser degree of zwitterionic character to the S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O double bond as compared to the P
O double bond as compared to the P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O double bond. The generally higher electron affinity of sulfur over phosphorus should facilitate the reduction of formal oxidation state S(+IV) to S(+II) as compared to the reduction of P(+V) to P(+III). The conversion of sulfoxides to sulfides is a vast field employing transition metal catalysis,18 main group catalysis,15b,19 electrochemical procedures,20 photochemical,21 and catalyst-free methods (Fig. 3).22 DMSO is one of the most fundamental sulfoxides and, due to its occurrence in the biosphere, the DMSO/DMS redox system plays an important role in biochemistry, as well as environmental and food analytics. For example, reductions include the use of molybdenum-containing enzyme DMSO reductase23 or rhodium(III) and molecular hydrogen.24 Trace analysis of DMSO in natural water after its reduction to DMS with NaBH4 has been described.25 The occurrence of DMS in beer brewing processes is notable, as well.26
O double bond. The generally higher electron affinity of sulfur over phosphorus should facilitate the reduction of formal oxidation state S(+IV) to S(+II) as compared to the reduction of P(+V) to P(+III). The conversion of sulfoxides to sulfides is a vast field employing transition metal catalysis,18 main group catalysis,15b,19 electrochemical procedures,20 photochemical,21 and catalyst-free methods (Fig. 3).22 DMSO is one of the most fundamental sulfoxides and, due to its occurrence in the biosphere, the DMSO/DMS redox system plays an important role in biochemistry, as well as environmental and food analytics. For example, reductions include the use of molybdenum-containing enzyme DMSO reductase23 or rhodium(III) and molecular hydrogen.24 Trace analysis of DMSO in natural water after its reduction to DMS with NaBH4 has been described.25 The occurrence of DMS in beer brewing processes is notable, as well.26
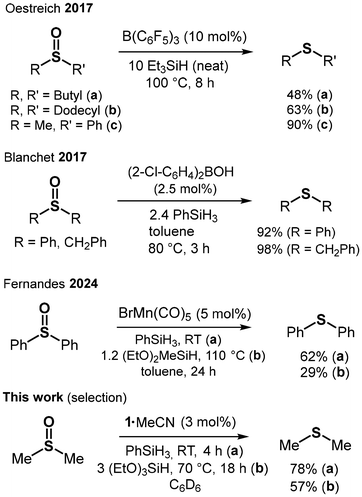 | ||
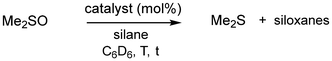
| Fig. 3 Selected catalytic reductions of sulfoxides to sulfides using hydrosilane reducing agents (RT = room temperature). | ||
We converted DMSO with PhSiH3 (1.5 eq.) under the addition of 1·MeCN as a catalyst (5 mol%) in C6D6 at room temperature (RT). As a result, we observed full consumption of the DMSO and major conversion to Me2S (DMS, 73%) within 2 hours (using 1H and 13C NMR analysis with 4,4′-di-tert-butyl-biphenyl as the internal standard, Table 2, entry 1). This contrasts the increased temperature (80 °C) and longer reaction time (5 h) required for the reduction of Me3PO with PhSiH3. The discrepancy between the converted DMSO (>99%) and the determined DMS yield (73%) we attribute to a larger relevance of side-reactions for the conversion of this substrate as compared to the phosphine oxide reductions. As expected, decreasing the load of 1·MeCN and the equivalents of PhSiH3 resulted in lower conversion rates (Table 2, entries 2, 3 and 4). For the phosphine oxide reduction, the use of 2·MeCN as a (pre)catalyst had afforded just marginally less efficient conversions of the substrates. Remarkably, the catalytic DMSO reduction with PhSiH3 showed a substantially diminished performance at RT when using this germanium complex as a promoter (Table 2, entries 5 and 6). Using catalytic amounts of the preformed DMSO complex 2·Me2SO (see the supplementary information for its isolation) instead of 2·MeCN had a negligible effect on the conversion rate (Table 2, entry 7). It is reasonable to assume that a ‘softer’ sulfur atom has a higher affinity to the Ge center in 2 as it has to the ‘harder’ Si center in 1. Thus, 2·MeCN will suffer more strongly from ‘catalyst poisoning’ by the sterically unhindered product Me2S. We also employed both the reference acids B(C6F5)3 and HNTf2 as (pre)catalysts, which in resemblance to 2·MeCN, turned out to perform substantially worse than the silicon Lewis acid 1·MeCN (Table 2, entries 8 and 9). Notably, when no promoter was applied, 14% DMSO was consumed to yield 8% DMS at 100 °C over 24 h (Table 2, entry 10). Moreover, we investigated the use of Et3SiH and (EtO)3SiH as alternative reducing agents. These are less atom economical but commonly cheaper and more suitable for process upscaling than PhSiH3. The Et3SiH reducing agent (4 eq.) proved ineffective in our hands: very harsh conditions (100 °C, 24 h) provided only 6% yield in DMS (28% conversion of DMSO) when applying the potent 1·MeCN as a (pre)catalyst (Table 2, entry 11). In contrast, (EtO)3SiH (4 eq.) afforded conversion to DMS in a moderate yield (52%) at RT in C6D6 using 1·MeCN (5 mol%), though on a notably longer timescale (72 h, Table 2, entry 12). Changing the solvent (C6D6 for CDCl3) and lowering the equivalents of hydrides had only a minor impact on the reaction outcome (Table 2, entry 13). Conducting the catalysis at 70 °C in C6D6 boosted the conversion rate with only a negligible impact on product distribution (Table 2, entry 14). In resemblance to our finding with PhSiH3, three equivalents of hydride were required for an effective reduction: the use of only 2 eq. (EtO)3SiH resulted in incomplete DMSO conversion (70%) to afford 39% DMS at 70 °C (3 mol% load with 1·MeCN) even at prolonged reaction times (Table 2, entry 15). The high performance of 1·MeCN as a promoter was verified by decreasing the load down to 1 mol%, which delivered almost 2/3 of the DMS yield after the same reaction time as with 5 mol% load (Table 2, entry 16 vs. entry 12). With (EtO)3SiH as a reducing agent, the use of germanium complex 2·MeCN as a (pre)catalyst was, yet again, less effective than using the silicon Lewis acid (Table 2, entries 17 and 18). The Brønsted superacid promoter HN(Tf)2 was also markedly less effective than 1·MeCN (Table 2, entries 19 and 20). Notably, we observed the separation of oil and solid from the reaction solution. We found the combination of (EtO)3SiH with B(C6F5)3 as a (pre)catalyst least suitable: with a load of 5 mol% and use of 4 eq. silane we could not observe DMS formation even after allowing the experiment to run a couple of days beyond the 72 h time mark (Table 2, entry 21). The outcome that the boron Lewis acid does not promote the DMSO reduction in combination with (EtO)3SiH (at RT) but performs moderately when brought together with PhSiH3 needs to be considered in the light of the respective 11B NMR data: the 11B NMR spectrum of the triethoxysilane conversion reveals a singlet at −0.4 ppm (h1/2 = 230 Hz), which we ascribe to [EtOB(C6F6)3]−, though Me2SB(C6F5)3 is a conceivable species, as well.27 The 11B analysis of the PhSiH3 reaction similarly shows a major singlet at −0.2 ppm (h1/2 = 240 Hz) but also reveals a doublet at −24.7 ppm (J = 80 Hz) of minor intensity (see Fig. S21). The doublet can be assigned to the [HB(C6F5)3]− anion, which was reported for catalytic conversions using combinations of hydrosilane and tris(pentafluorophenyl)borane and, of course, for catalytic dihydrogenations with frustrated Lewis pairs containing this borane Lewis acid.28 The borohydride anion may indicate the formation of highly Lewis acidic silyl cation or the borohydride itself may act as a hydride transfer reagent. Finally, it is of note that without the application of a catalyst, no relevant consumption of DMSO was indicated by the 1H NMR analysis at 100 °C for 24 h using 5 eq. of (EtO)3SiH (Table 2, entry 22).
| Cat. (mol%) | Silane (eq.) | T | t | Yielda [%] | |
|---|---|---|---|---|---|
| a Yield of DMS was determined by addition of 1,4-di-tert-butyl-biphenyl as internal standard. b DMSO was fully consumed and yield was increased after 12 h. c Monitoring for additional 6 h resulted only in negligible change. d DMSO was fully consumed and 78% yield achieved after 13 h at 70 °C. e An oil separated from the mixture. f After a total of 96 h, DMSO was fully consumed and 58% yield was achieved. g CDCl3 was used as the solvent instead of C6D6. h No further conversion by additional heating for 12 h. i CDCl3 was used as the solvent and a solid separated after few hours. | |||||
| 1 | 1·MeCN (5) | PhSiH3 (1.5) | RT | 2 h | 73 |
| 2 | 1·MeCN (3) | PhSiH3 (1) | RT | 4 h | 78 |
| 3 | 1·MeCN (1) | PhSiH3 (1) | RT | 4 h | 53b |
| 4 | 1·MeCN (3) | PhSiH3 (2/3) | RT | 24 h | 66c |
| 5 | 2·MeCN (5) | PhSiH3 (1.5) | RT | 3 h | 9 |
| 6 | 2·MeCN (3) | PhSiH3 (1) | RT | 4 h | 6 |
| 7 | 2·DMSO (3) | PhSiH3 (1) | RT | 4 h | 12d |
| 8 | B(C6F5)3 (5) | PhSiH3 (1) | RT | 18 h | 13 |
| 9 | HNTf2 (5) | PhSiH3 (1.5) | RT | 2 h | 12e |
| 10 | None | PhSiH3 (3) | 100 °C | 24 h | 8 |
| 11 | 1·MeCN (5) | Et3SiH (4) | 100 °C | 24 h | 6 |
| 12 | 1·MeCN (5) | (EtO)3SiH (4) | RT | 72 h | 52f |
| 13 | 1·MeCN (5) | (EtO)3SiH (3) | RT | 72 h | 40g |
| 14 | 1·MeCN (3) | (EtO)3SiH (3) | 70 °C | 18 h | 57 |
| 15 | 1·MeCN (3) | (EtO)3SiH (2) | 70 °C | 36 h | 39h |
| 16 | 1·MeCN (1) | (EtO)3SiH (4) | RT | 72 h | 31 |
| 17 | 2·MeCN (5) | (EtO)3SiH (4) | RT | 72 h | 26 |
| 18 | 2·MeCN (3) | (EtO)3SiH (3) | 70 °C | 18 h | 12 |
| 19 | HNTf2 (5) | (EtO)3SiH (4) | RT | 72 h | 10e |
| 20 | HNTf2 (5) | (EtO)3SiH (3) | RT | 72 h | 11i |
| 21 | B(C6F5)3 (5) | (EtO)3SiH (4) | RT | 72 h | <1 |
| 22 | None | (EtO)3SiH (5) | 100 °C | 24 h | <1 |
Dimethylformamide reductions
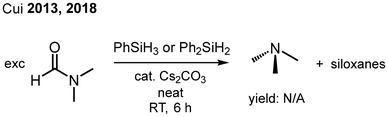
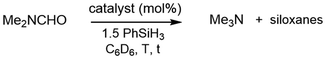
DMF is difficult to reduce due to the delocalization of the nitrogen lone pair into the amide system, which renders the carbonyl carbon atom less electrophilic. It is used in large quantities as a solvent in the synthesis of peptides.29 DMF has a high boiling point (153 °C) and is commonly known for its hepatotoxicity.30 This combination of properties makes complete removal of the solvent from products most desirable, yet difficult. The ability to reduce DMF to volatile trimethylamine (boiling point 3 °C) would be beneficial for clean-up of reactions where large quantities of DMF waste are produced. Several methods for the catalytic reduction of DMF to Me3N by hydrosilanes using transition metal compounds as promoters have been described.31 Cui has reported the use of Cs2CO3 as a suitable catalyst for the reduction of DMF (and other amides) using phenylsilanes (Fig. 4).32Given the potency of the Lewis acids 1·MeCN and 2·MeCN in the catalytic reduction of R3PO and Me2SO with silanes, we tested these systems for the transformation of DMF to Me3N using PhSiH3, as well. Promoting the reaction with 1·MeCN (5 mol%) in toluene-D8 furnished trimethylamine in a near-quantitative fashion (99%) at 110 °C over 12 h (Table 3, entry 1). Under milder reaction conditions (80 °C), a smaller ratio of Me3N was detected (71%) even after longer processing (24 h), which underlines the stability of this amide bond (Table 3, entry 2). The Ge complex 2·MeCN exhibited decreased catalytic activity (61%, Table 3, entry 3). The benchmark soft lewis superacid B(C6F5)3 performed similarly to 1·MeCN after 22 h (Table 3, entry 4). The 11B NMR spectra of the process revealed a signal at −24.7 ppm, which can be assigned to HB(C6F5)3, and this observation resembles the DMSO reduction with B(C6F5)3 and PhSiH3 described above. In the absence of a Lewis acid catalyst, no conversion of DMF was observed in toluene after 48 hours at 110 °C using 1.5 equivalents of PhSiH3 (Table 3, entry 5). To our knowledge, this is the first report of an uncharged tetrel Lewis superacid to successfully catalyze the reduction of an amide to an amine.
| Cat. (mol%) | Solvent | T [°C] | t | Yielda [%] | |
|---|---|---|---|---|---|
| a Yield determined using 1H NMR with 4,4-di-tert-butyl-biphenyl as an internal standard. b Near-quantitative consumption of DMF. | |||||
| 1 | 1·MeCN (5) | C7D8 | 110 | 12 h | 99 |
| 2 | 1·MeCN (5) | C6D6 | 80 | 24 h | 71 |
| 3 | 2·MeCN (5) | C6D6 | 80 | 24 h | 61 |
| 4 | B(C6F5)3 (5) | C6D6 | 80 | 22 h | 70b |
| 5 | None | o-DFB | 110 | 48 h | 0 |
Synthesis and isolation of 1·Me2NCHO and 2·Me2SO
In order to independently synthesize possible intermediates of our catalytic conversions, we reacted 1·MeCN and 2·MeCN with 1.3 eq. of DMF and DMSO, respectively (Scheme 1). The analytically pure Lewis acid base complexes 1·Me2NCHO and 2·Me2SO were isolated in good yields (89% and 84%) and characterized by NMR spectroscopy, combustion analysis (CHNS), mass spectrometry, and single-crystal XRD (SC-XRD) study.As a notable characteristic in the 1H NMR spectrum of 1·Me2NCHO the signals of the Si-coordinated DMF are notably shifted to lower fields (δ = 8.32, 3.33, and 3.18 ppm) as compared to “free” DMF in CD3CN.33 Suitable crystals of 1·Me2NCHO for SC-XRD analysis were obtained from a saturated CH2Cl2/MeCN (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) solution at −35 °C. The study reveals a silicon center that is coordinated in a trigonal bipyramidal fashion with the DMF ligand assuming an equatorial position (Fig. 5, top). The C
1) solution at −35 °C. The study reveals a silicon center that is coordinated in a trigonal bipyramidal fashion with the DMF ligand assuming an equatorial position (Fig. 5, top). The C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond length in 1·Me2NCHO amounts to C1–O1 = 1.296(4) Å, which is elongated as compared to DMF in the solid state (note: crystalline DMF forms a hydrogen bonding network with a mean C–O of 1.23 Å).34 This suggests a weakening in the C
O bond length in 1·Me2NCHO amounts to C1–O1 = 1.296(4) Å, which is elongated as compared to DMF in the solid state (note: crystalline DMF forms a hydrogen bonding network with a mean C–O of 1.23 Å).34 This suggests a weakening in the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond and may facilitate the hydride-induced reductive cleavage.
O bond and may facilitate the hydride-induced reductive cleavage.
In the 1H NMR spectrum of 2·Me2SO, the singlet for the CH3 groups of coordinated DMSO at 3.10 ppm (J1H13C = 144 Hz) is notable, which is markedly shifted to lower field as compared to the value for “free” Me2SO in CD3CN (2.50 ppm).33
Crystals of 2·Me2SO, suitable for SC-XRD analysis, were crystallized directly from the reaction mixture at −25 °C and the molecular structure of 2·Me2SO in the solid state marks a trigonal bipyramidal coordinate Ge center (sum of the equatorial bond angles ≈ 360°, Fig. 5, bottom). The bond to the oxygen atom of the (equatorial) DMSO ligand amounts to a length of Ge1–O1 = 1.820(1) Å. This distance is in between the values for the Ge–O distances involving the perfluoropinacol ligands, which show longer bonds for the axial positions (1.836(1) Å, 1.854(1) Å) and shorter bonds for the equatorial positions (1.801(1) Å, 1.802(1) Å). Notably, the S1–O1 distance of 1.596(1) Å is elongated relative to the respective bond lengths in Greb's octahedral coordinate (catCl)2Ge·(Me2SO)2 (Ge–ODMSO = 1.561(1) Å, symmetry equivalent).6b Interestingly, Greb's SC-XRD study also involves an uncoordinated lattice DMSO with a shorter S–O bond length of 1.502(1) Å that may serve as a reference for “free” Me2SO in the condensed phase. These data indicate that the S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond strength in 2·Me2SO is slightly lower than in (catCl)2Ge·(Me2SO)2. We assume that the six-coordinate Ge center in Greb's compound bearing two Me2SO donor ligands draws less electron density from each sulfoxide than the five-coordinate Ge center in 2·Me2SO, which bears only one Me2SO donor ligand to compensate its electron deficiency. Future studies might investigate into a correlation between the S–O distance in sulfoxide adducts of potent Lewis acids and their catalytic activity in sulfoxide reduction.
O bond strength in 2·Me2SO is slightly lower than in (catCl)2Ge·(Me2SO)2. We assume that the six-coordinate Ge center in Greb's compound bearing two Me2SO donor ligands draws less electron density from each sulfoxide than the five-coordinate Ge center in 2·Me2SO, which bears only one Me2SO donor ligand to compensate its electron deficiency. Future studies might investigate into a correlation between the S–O distance in sulfoxide adducts of potent Lewis acids and their catalytic activity in sulfoxide reduction.
To test the relative affinity of the DMSO ligand to 2, we added DMF (2 eq.) to a CD3CN solution of 2·Me2SO in an NMR sample tube, and 1H NMR analysis revealed a marked upfield shift of the CH3 signal of the DMSO protons. Notably, only one signal (set) for the DMSO (2.83 ppm), as well as the DMF (8.03, 3.00, 2.87 ppm) was observed neither of which corresponded to the respective 1H chemical shift of the “free” sulfoxide or amide, respectively (δ(1H) = 2.50 or 7.92, 2.89, and 2.77 ppm in CD3CN),33 but was consistently shifted to lower field. This suggests that DMSO and DMF are in a dynamic competitive equilibrium toward coordination to the Ge center in 2 (participation of CD3CN cannot be fully excluded), which is quick on the NMR timescale (at RT). The addition of Bu3PO (1.1 eq.) to this mixture resulted in a (further) upfield shift of the DMSO and DMF signals in the 1H NMR spectrum to values that match the “free” oxo compounds. In the 31P{1H} NMR analysis, one broad resonance at 87.2 ppm was found, which refers to the 31P signal observed, as also, upon mixing 2·MeCN and this phosphine oxide in CD3CN (without DMSO and DMF). We presume that Bu3PO majorly extrudes both DMSO and DMF from the Ge complex to furnish the more stable 2·Bu3PO. In addition, we converted 2·Me2SO with PhSiH3 (1.5 eq.) in CD3CN, which resulted in the anticipated formation of Me2S but also complete decomposition of 2 (and not refurnishing of 2·Do, Do = SMe2, CD3CN and Me2SO, as expected from a “true” catalyst) as concluded from 1H and 19F NMR analysis (full conversion after 9 h at 70 °C). This also shows that the Lewis acid-catalyzed reduction of DMSO to DMS with PhSiH3 can be conducted in MeCN, though conversion rates might be lower.
Considerations in the catalysis mechanism
We have shown using NMR spectroscopy and SC-XRD study that 1 and 2 form strong coordination compounds with Bu3PO, Me2SO, and Me2NCHO. Complex 2·MeCN was reported to react with hydrosilane to the germylene species 3 and silylated perfluoropinacol and we showed that the latter exhibits weaker catalytic activity.8 In contrast, we found 1·MeCN not to react with PhSiH3 at RT (1H, 19F NMR monitored for 4 h in CD3CN; at 70 °C traces of new species noted after 2 h). Tilley proposed a Lewis acid catalysis mechanism for the reduction of aldehydes with Et3SiH using bis(perfluorocatecholato)silane as catalyst.3a The catalytic cycle marks the initial interaction of the Lewis acid with the carbonyl group of the substrate. An alternative mechanism, which comprises an initial interaction between the Lewis acid and the hydrosilane (to effect weakening of the Si–H bond), was suggested by Oestreich and Stephan for the phosphine oxide reduction with silanes promoted by B(C6F5)3 or electrophilic phosphonium cations.15a We reason that for the stronger adducts 1·Do (Do = R3PO, Me2SO, and Me2NCHO) the direct interaction of the Si center with an SiH group will be even more diminished than for 1·MeCN. Accordingly, we conclude that the catalytic reductions of the E![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O double bonds (E = P, S, C) presented in this study proceed similar to Tilley's Lewis acid catalysis mechanism when 1·MeCN is used as the promoter. For 2·MeCN, the situation is more ambiguous due to its pronounced hydride affinity. Consistent to Tilley's mechanism we suggest that the respective complexes 1·Do or 2·Do (Do = R3PO, Me2SO, Me2NCHO) mark the actual catalysts which renders the MeCN adducts to assume the role of precatalysts. It is remarkable that 1·MeCN seems to outperform the more Lewis acidic B in catalytic reduction of Ph3PO (Fig. 1, 2, and Table 1). Notably, strong Lewis acids of perhalogenated bis(catecholato) tetrelanes form hexacoordinate complexes with many Lewis bases, and, in stark contrast, the many bis(perfluoropinacol) tetrelanes which we have structurally characterized, so far, are limited to five-fold coordination. This demonstrates the pronounced impact of the ligand system (catecholate vs. pincacolate) on catalyst activity.
O double bonds (E = P, S, C) presented in this study proceed similar to Tilley's Lewis acid catalysis mechanism when 1·MeCN is used as the promoter. For 2·MeCN, the situation is more ambiguous due to its pronounced hydride affinity. Consistent to Tilley's mechanism we suggest that the respective complexes 1·Do or 2·Do (Do = R3PO, Me2SO, Me2NCHO) mark the actual catalysts which renders the MeCN adducts to assume the role of precatalysts. It is remarkable that 1·MeCN seems to outperform the more Lewis acidic B in catalytic reduction of Ph3PO (Fig. 1, 2, and Table 1). Notably, strong Lewis acids of perhalogenated bis(catecholato) tetrelanes form hexacoordinate complexes with many Lewis bases, and, in stark contrast, the many bis(perfluoropinacol) tetrelanes which we have structurally characterized, so far, are limited to five-fold coordination. This demonstrates the pronounced impact of the ligand system (catecholate vs. pincacolate) on catalyst activity.
Experimental
The relevant experimental work was conducted under an argon atmosphere using standard Schlenk techniques and a glovebox equipment. A general and representative procedure for the phosphine oxide reduction is as follows: an NMR sample tube was charged with the catalyst, and the phosphine oxide and the solids were dissolved in o-DFB. The reducing agent was added, and the reaction mixture was heated to the respective temperature for 16 hours. Tris(2,4-di-tert-butylphenyl) phosphite was added as an internal standard to determine the yield via intensity ratios in the 31P{1H} NMR spectrum. More detailed experimental data are given in the SI of this article.Conclusions
The main group Lewis acids 1·MeCN and 2·MeCN were successfully applied as precatalysts in the reduction of phosphine oxides (e.g. Me3PO, Bu3PO, and Ph3PO), a sulfoxide (i.e. Me2SO), and an amide (i.e. Me2NCHO) to afford the respective phosphines, dimethyl sulfide, and trimethylamine using PhSiH3 or (EtO)3SiH. These substrates mark generally more stable element oxygen double bonds in comparison with, for example, the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O double bonds in ketones or aldehydes often targeted for demonstrating the catalytic activity of Lewis acids. As benchmarks, we also studied B(C6F5)3 and HNTf2 as reference (soft) Lewis superacid and Brønsted superacid, respectively. Among all the investigated combinations of (pre)catalyst, substrate, and reducing agent, we pronounce the silicon complex 1·MeCN as the most versatile system, being the by far most potent (DMSO) or just slightly underperforming (R3PO, DMF) promoter. For the methylated substrates, we sort the ease of catalytic reduction using 1·MeCN and PhSiH3 in the order Me2SO > Me3PO > Me2NCHO (most facile to most difficult). Moreover, the hitherto undescribed Lewis acid base adducts 1·Me2NCHO and 2·Me2SO were synthesized, isolated, and structurally investigated using multinuclear NMR spectroscopy and single-crystal XRD analysis. After probing the reactivity of 1·MeCN, 2·MeCN, and 2·Me2SO with DMF, DMSO, phosphine oxide, and PhSiH3, we conclude that a Lewis acid catalysis mechanism prevails as had been proposed by Tilley for silane Lewis acids. The MeCN complexes act as precatalysts to in situ form the catalytically active species 1·Do or 2·Do (Do = Me3PO, Me2SO, Me2NCHO). Future studies should focus on extending the scope of sulfoxides and amides, as well as suitable reducing agents. Other substrates such as esters should be investigated, and the water tolerance of the system needs to be examined.
O double bonds in ketones or aldehydes often targeted for demonstrating the catalytic activity of Lewis acids. As benchmarks, we also studied B(C6F5)3 and HNTf2 as reference (soft) Lewis superacid and Brønsted superacid, respectively. Among all the investigated combinations of (pre)catalyst, substrate, and reducing agent, we pronounce the silicon complex 1·MeCN as the most versatile system, being the by far most potent (DMSO) or just slightly underperforming (R3PO, DMF) promoter. For the methylated substrates, we sort the ease of catalytic reduction using 1·MeCN and PhSiH3 in the order Me2SO > Me3PO > Me2NCHO (most facile to most difficult). Moreover, the hitherto undescribed Lewis acid base adducts 1·Me2NCHO and 2·Me2SO were synthesized, isolated, and structurally investigated using multinuclear NMR spectroscopy and single-crystal XRD analysis. After probing the reactivity of 1·MeCN, 2·MeCN, and 2·Me2SO with DMF, DMSO, phosphine oxide, and PhSiH3, we conclude that a Lewis acid catalysis mechanism prevails as had been proposed by Tilley for silane Lewis acids. The MeCN complexes act as precatalysts to in situ form the catalytically active species 1·Do or 2·Do (Do = Me3PO, Me2SO, Me2NCHO). Future studies should focus on extending the scope of sulfoxides and amides, as well as suitable reducing agents. Other substrates such as esters should be investigated, and the water tolerance of the system needs to be examined.
Author contributions
S. I. conceived and guided the study. D. F. and T. F. conceived and conducted the specific experiments. S. S. collected, solved, and refined the SC-XRD data. All authors have co-written the manuscript.Conflicts of interest
The authors declare no conflict of interest.Data availability
Supporting data for this article have been included as part of the supplementary information (SI). Supplementary information: detailed experimental and crystallographic data. See DOI: https://doi.org/10.1039/d5qi02493e.Further raw data are available upon reasonable request from the corresponding author.
CCDC 2492264 and 2492265 contain the supplementary crystallographic data for this paper.35a,b
Acknowledgements
The authors are grateful to the European Research Council (ALLOWE101001591) for financial support and thank Tobias Weng for LIFDI-MS analysis.References
-
(a) D. W. Stephan, Diverse Uses of the Reaction of Frustrated Lewis Pair (FLP) with Hydrogen, J. Am. Chem. Soc., 2021, 143, 20002–20014 Search PubMed
; (b) P. P. Power, An Update on Multiple Bonding between Heavier Main Group Elements: The Importance of Pauli Repulsion, Charge-Shift Character, and London Dispersion Force Effects, Organometallics, 2020, 39, 4127–4138 CrossRef CAS
; (c) P. P. Power, Main-group elements as transition metals, Nature, 2010, 463, 171–177 CrossRef CAS PubMed
; (d) G. C. Welch, R. R. S. Juan, J. D. Masuda and D. W. Stephan, Reversible, Metal-Free Hydrogen Activation, Science, 2006, 314, 1124–1126 Search PubMed
.
-
(a) H. F. T. Klare, L. Albers, L. Süsse, S. Keess, T. Müller and M. Oestreich, Silylium Ions: From Elusive Reactive Intermediates to Potent Catalysts, Chem. Rev., 2021, 121, 5889–5985 Search PubMed
; (b) J. B. Lambert, S. Zhang, C. L. Stern and J. C. Huffman, Crystal Structure of a Silyl Cation with No Coordination to Anion and Distant Coordination to Solvent, Science, 1993, 260, 1917–1918 CrossRef CAS PubMed
.
-
(a) A. L. Liberman-Martin, R. G. Bergman and T. D. Tilley, Lewis Acidity of Bis(perfluorocatecholato)silane: Aldehyde Hydrosilylation Catalyzed by a Neutral Silicon Compound, J. Am. Chem. Soc., 2015, 137, 5328–5331 CrossRef CAS
; (b) L. Greb, Lewis Superacids: Classifications, Candidates, and Applications, Chem. – Eur. J., 2018, 24, 17881–17896 CrossRef CAS PubMed
; (c) P. Erdmann and L. Greb, Multidimensional Lewis Acidity: A Consistent Data Set of Chloride, Hydride, Methide, Water and Ammonia Affinities for 183 p-Block Element Lewis Acids, ChemPhysChem, 2021, 22, 935–943 CrossRef CAS PubMed
; (d) A. Hermannsdorfer and M. Driess, Silicon Tetrakis(trifluoromethanesulfonate): A Simple Neutral Silane Acting as a Soft and Hard Lewis Superacid, Angew. Chem., Int. Ed., 2021, 60, 13656–13660 CrossRef CAS PubMed
; (e) A. Y. Timoshkin, The Field of Main Group Lewis Acids and Lewis Superacids: Important Basics and Recent Developments, Chem. – Eur. J., 2024, 30, e202302457 CrossRef CAS PubMed
; (f) F. Hanusch, D. Franz and S. Inoue, Uncharged Lewis superacidic silicon complexes—perfluoropinacolato-stabilized systems for homogenous catalysis, Chem. Lett., 2024, 53, upae232 CrossRef CAS
.
- A. Hermannsdorfer and M. Driess, Isolable Silicon-Based Polycations with Lewis Superacidity, Angew. Chem., Int. Ed., 2020, 59, 23132–23136 Search PubMed
.
- L. O. Müller, D. Himmel, J. Stauffer, G. Steinfeld, J. Slattery, G. Santiso-Quiñones, V. Brecht and I. Krossing, Simple Access to the Non-Oxidizing Lewis Superacid PhF→Al(ORF)3 (RF=C(CF3)3), Angew. Chem., Int. Ed., 2008, 47, 7659–7663 CrossRef PubMed
.
-
(a) R. Maskey, M. Schädler, C. Legler and L. Greb, Bis(perchlorocatecholato)silane—A Neutral Silicon Lewis Super Acid, Angew. Chem., Int. Ed., 2018, 57, 1717–1720 CrossRef CAS PubMed
; (b) D. Roth, H. Wadepohl and L. Greb, Bis(perchlorocatecholato)germane: Hard and Soft Lewis Superacid with Unlimited Water Stability, Angew. Chem., Int. Ed., 2020, 59, 20930–20934 CrossRef CAS PubMed
; (c) T. Thorwart, D. Roth and L. Greb, Bis(pertrifluoromethylcatecholato)silane: Extreme Lewis Acidity Broadens the Catalytic Portfolio of Silicon, Chem. – Eur. J., 2021, 27, 10422–10427 Search PubMed
; (d) T. Thorwart, D. Hartmann and L. Greb, Dihydrogen Activation with a Neutral, Intermolecular Silicon(IV)-Amine Frustrated Lewis Pair, Chem. – Eur. J., 2022, 28, e202202273 CrossRef CAS PubMed
.
- F. S. Tschernuth, T. Thorwart, L. Greb, F. Hanusch and S. Inoue, Bis(perfluoropinacolato)silane: A Neutral Silane Lewis Superacid Activates Si−F Bonds, Angew. Chem., Int. Ed., 2021, 60, 25799–25803 Search PubMed
.
- F. S. Tschernuth, A. Kostenko, S. Stigler, A. Gradenegger and S. Inoue, A neutral germanium-centred hard and soft lewis superacid and its unique reactivity towards hydrosilanes, Dalton Trans., 2024, 53, 74–81 Search PubMed
.
-
(a) F. S. Tschernuth, L. Bichlmaier, S. Stigler and S. Inoue, Tuning the Lewis Acidity of Neutral Silanes Using Perfluorinated Aryl- and Alkoxy Substituents, Eur. J. Inorg. Chem., 2023, 26, e202300388 Search PubMed
; (b) F. S. Tschernuth, L. Bichlmaier and S. Inoue, Catalytic Degradation of Aliphatic Ethers using the Lewis Superacidic Bis(perfluoropinacolato)silane, ChemCatChem, 2023, 15, e202300281 CrossRef CAS
.
-
(a) P. A. Byrne and D. G. Gilheany, The modern interpretation of the Wittig reaction mechanism, Chem. Soc. Rev., 2013, 42, 6670–6696 RSC
; (b) B. E. Maryanoff and A. B. Reitz, The Wittig olefination reaction and modifications involving phosphoryl-stabilized carbanions. Stereochemistry, mechanism, and selected synthetic aspects, Chem. Rev., 1989, 89, 863–927 CrossRef CAS
; (c) K. C. K. Swamy, N. N. B. Kumar, E. Balaraman and K. V. P. P. Kumar, Mitsunobu and Related Reactions: Advances and Applications, Chem. Rev., 2009, 109, 2551–2651 CrossRef CAS PubMed
; (d) S. Fletcher, The Mitsunobu reaction in the 21st century, Org. Chem. Front., 2015, 2, 739–752 RSC
.
- T. Coumbe, N. J. Lawrence and F. Muhammad, Titanium(IV) catalysis in the reduction of phosphine oxides, Tetrahedron Lett., 1994, 35, 625–628 CrossRef CAS
.
- Y. Li, S. Das, S. Zhou, K. Junge and M. Beller, General and Selective Copper-Catalyzed Reduction of Tertiary and Secondary Phosphine Oxides: Convenient Synthesis of Phosphines, J. Am. Chem. Soc., 2012, 134, 9727–9732 CrossRef CAS PubMed
.
-
(a) H. Fritzsche, U. Hasserodt and F. Korte, Reduktion organischer Verbindungen des fünfwertigen Phosphors zu Phosphinen, I. Reduktion tertiärer Phosphinoxyde zu tertiären Phosphinen mit Silanen, Chem. Ber., 1964, 97, 1988–1993 CrossRef CAS
; (b) L. Horner and W. D. Balzer, Phosphororganische verbindungen IXL zum sterischen verlauf der desoxygenierung von tertiären phosphinoxyden zu tertiären phosphinen mit trichlorsilan, Tetrahedron Lett., 1965, 6, 1157–1162 Search PubMed
; (c) K. Naumann, G. Zon and K. Mislow, Use of hexachlorodisilane as a reducing agent. Stereospecific deoxygenation of acyclic phosphine oxides, J. Am. Chem. Soc., 1969, 91, 7012–7023 CrossRef CAS
.
-
(a) D. Hérault, D. H. Nguyen, D. Nuel and G. Buono, Reduction of secondary and tertiary phosphine oxides to phosphines, Chem. Soc. Rev., 2015, 44, 2508–2528 Search PubMed
; (b) E. Podyacheva, E. Kuchuk and D. Chusov, Reduction of phosphine oxides to phosphines, Tetrahedron Lett., 2019, 60, 575–582 CrossRef CAS
.
-
(a) M. Mehta, I. Garcia de la Arada, M. Perez, D. Porwal, M. Oestreich and D. W. Stephan, Metal-Free Phosphine Oxide Reductions Catalyzed by B(C6F5)3 and Electrophilic Fluorophosphonium Cations, Organometallics, 2016, 35, 1030–1035 CrossRef CAS
; (b) A. Chardon, O. Maubert, J. Rouden and J. Blanchet, Metal-Free Reduction of Phosphine Oxides, Sulfoxides, and N-Oxides with Hydrosilanes using a Borinic Acid Precatalyst, ChemCatChem, 2017, 9, 4460–4464 Search PubMed
.
- P. Erdmann and L. Greb, What Distinguishes the Strength and the Effect of a Lewis Acid: Analysis of the Gutmann–Beckett Method, Angew. Chem., Int. Ed., 2022, 61, e202114550 CrossRef CAS PubMed
.
-
(a) O. M. Demchuk, R. Jasiński and K. M. Pietrusiewicz, New Insights into the Mechanism of Reduction of Tertiary Phosphine Oxides by Means of Phenylsilane, Heteroat. Chem., 2015, 26, 441–448 CrossRef CAS
; (b) K. L. Marsi, Phenylsilane reduction of phosphine oxides with complete stereospecificity, J. Org. Chem., 1974, 39, 265–267 Search PubMed
.
-
(a) Y. Takahashi, T. Mitsudome, T. Mizugaki, K. Jitsukawa and K. Kaneda, Highly Efficient Deoxygenation of Sulfoxides Using Hydroxyapatite-supported Ruthenium Nanoparticles, Chem. Lett., 2014, 43, 420–422 Search PubMed
; (b) D. L. Lourenço and A. C. Fernandes, Reduction of sulfoxides catalyzed by the commercially available manganese complex MnBr(CO)5, Org. Biomol. Chem., 2024, 22, 3746–3751 RSC
; (c) S. Enthaler and M. Weidauer, Reduction of Sulfoxides to Sulfides in the Presence of Copper Catalysts, Catal. Lett., 2011, 141, 833–838 CrossRef CAS
.
- D. Porwal and M. Oestreich, B(C6F5)3-Catalyzed Reduction of Sulfoxides and Sulfones to Sulfides with Hydrosilanes, Synthesis, 2017, 4698–4702 Search PubMed
.
-
(a) C. Edinger and S. R. Waldvogel, Electrochemical Deoxygenation of Aromatic Amides and Sulfoxides, Eur. J. Org. Chem., 2014, 5144–5148 Search PubMed
; (b) Z. Kong, C. Pan, M. Li, L. Wen and W. Guo, Scalable electrochemical reduction of sulfoxides to sulfides, Green Chem., 2021, 23, 2773–2777 RSC
.
- J. Zhao, Z. Luo, Y. Liu, S. Chen, J. He, J. Xu, W. Hu, Z. Huang and W. Xiong, Visible light induced deoxygenation of sulfoxides with isopropanol, Org. Chem. Front., 2023, 10, 5254–5259 RSC
.
-
(a) M. Madesclaire, Reduction of sulfoxides to thioethers, Tetrahedron, 1988, 44, 6537–6580 CrossRef CAS
; (b) F. Takahashi, K. Nogi and H. Yorimitsu, B2cat2-Mediated Reduction of Sulfoxides to Sulfides, Eur. J. Org. Chem., 2020, 3009–3012 CrossRef CAS
.
- H. Schindelin, C. Kisker, J. Hilton, K. V. Rajagopalan and D. C. Rees, Crystal Structure of DMSO Reductase: Redox-Linked Changes in Molybdopterin Coordination, Science, 1996, 272, 1615–1621 CrossRef CAS PubMed
.
- B. R. James, F. T. T. Ng and G. L. Rempel, Catalytic reduction of dimethylsulfoxide by molecular hydrogen using rhodium(III) complexes, Can. J. Chem., 1969, 47, 4521–4526 CrossRef CAS
.
- R. Simó, J. O. Grimalt and J. Albaigés, Sequential Method for the Field Determination of Nanomolar Concentrations of Dimethyl Sulfoxide in Natural Waters, Anal. Chem., 1996, 68, 1493–1498 Search PubMed
.
- B. J. Anness and C. W. Bamforth, DIMETHYL SULPHIDE—A REVIEW, J. Inst. Brew., 1982, 88, 244–252 CrossRef CAS
.
-
(a) S. Kronig, E. Theuergarten, D. Holschumacher, T. Bannenberg, C. G. Daniliuc, P. G. Jones and M. Tamm, Dihydrogen Activation by Frustrated Carbene-Borane Lewis Pairs: An Experimental and Theoretical Study of Carbene Variation, Inorg. Chem., 2011, 50, 7344–7359 CrossRef CAS PubMed
; (b) J.-M. Denis, H. Forintos, H. Szelke, L. Toupet, T.-N. Pham, P.-J. Madec and A.-C. Gaumont, B(C6F5)3-catalyzed formation of B–P bonds by dehydrocoupling of phosphine–boranes, Chem. Commun., 2003, 54–55 Search PubMed
.
-
(a) D. W. Stephan, Frustrated Lewis Pairs: From Concept to Catalysis, Acc. Chem. Res., 2015, 48, 306–316 Search PubMed
; (b) A. Berkefeld, W. E. Piers and M. Parvez, Tandem Frustrated Lewis Pair/Tris(pentafluorophenyl)borane-Catalyzed Deoxygenative Hydrosilylation of Carbon Dioxide, J. Am. Chem. Soc., 2010, 132, 10660–10661 Search PubMed
.
- Y. E. Jad, G. A. Acosta, S. N. Khattab, B. G. de la Torre, T. Govender, H. G. Kruger, A. El-Faham and F. Albericio, Peptide synthesis beyond DMF: THF and ACN as excellent and friendlier alternatives, Org. Biomol. Chem., 2015, 13, 2393–2398 Search PubMed
.
-
(a) H. Liu, M.-J. Li, X.-N. Zhang, S. Wang, L.-X. Li, F.-F. Guo and T. Zeng, N,N-dimethylformamide-induced acute liver damage is driven by the activation of NLRP3 inflammasome in liver macrophages of mice, Ecotoxicol. Environ. Saf., 2022, 238, 113609 Search PubMed
; (b) Y. Lei, S. Xiao, S. Chen, H. Zhang, H. Li and Y. Lu, N,N–dimethylformamide–induced acute hepatic failure: A case report and literature review, Exp. Ther. Med., 2017, 14, 5659–5663 CAS
.
-
(a) V. P. Taori and M. R. Buchmeiser, Tandem-reduction of DMF with silanes via necklace-type transition over Pt(0) nanoparticles: deciphering the dual Si–H effect as an extension of steric effects, Chem. Commun., 2014, 50, 14820–14823 Search PubMed
; (b) J. L. Martinez, H. K. Sharma, R. Arias-Ugarte and K. H. Pannell, Platinum-Catalyzed Reduction of DMF by 1,1,3,3-Tetramethyldisiloxane, HMeSi2OSiMe2H: New Intermediates HSiMe2OSiMe2OCH2NMe2 and HSiMe2(OSiMe2)3OCH2NMe2 and Their Further Chemical Reactivity, Organometallics, 2014, 33, 2964–2967 Search PubMed
; (c) R. Arias-Ugarte, H. K. Sharma, A. L. C. Morris and K. H. Pannell, Metal-Catalyzed Reduction of HCONR′2, R′ = Me (DMF), Et (DEF), by Silanes to Produce R′2NMe and Disiloxanes: A Mechanism Unraveled, J. Am. Chem. Soc., 2012, 134, 848–851 Search PubMed
; (d) H. K. Sharma, R. Arias-Ugarte, D. Tomlinson, R. Gappa, A. J. Metta-Magaña, H. Ito and K. H. Pannell, (Me3N)Mo(CO)5-Catalyzed Reduction of DMF by Disiloxane and Disilane Moieties: Fate of the Silicon-Containing Fragments, Organometallics, 2013, 32, 3788–3794 Search PubMed
.
-
(a) Y. Li and C. Cui, Cesium Carbonate-Catalyzed Oxidation of Substituted Phenylsilanes for the Efficient Synthesis of Polyhedral Oligomeric Silsesquioxanes, Inorg. Chem., 2018, 57, 13477–13485 Search PubMed
; (b) W. Xie, M. Zhao and C. Cui, Cesium Carbonate-Catalyzed Reduction of Amides with Hydrosilanes, Organometallics, 2013, 32, 7440–7444 Search PubMed
.
- G. R. Fulmer, A. J. M. Miller, N. H. Sherden, H. E. Gottlieb, A. Nudelman, B. M. Stoltz, J. E. Bercaw and K. I. Goldberg, NMR Chemical Shifts of Trace Impurities: Common Laboratory Solvents, Organics, and Gases in Deuterated Solvents Relevant to the Organometallic Chemist, Organometallics, 2010, 29, 2176–2179 CrossRef CAS
.
- H. Borrmann, I. Persson, M. Sandström and C. M. V. Stålhandske, The crystal and liquid structures of N,N-dimethylthioformamide and N,N-dimethylformamide showing a stronger hydrogen bonding effect for C–H⋯S than of C–H⋯O, J. Chem. Soc., Perkin Trans. 2, 2000, 393–402 RSC
.
-
(a)
CCDC 2492264: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pndmd
; (b) CCDC 2492265: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pndnf
.
| This journal is © the Partner Organisations 2026 |