Thermoplastic solid-state polymer electrolytes based on linear B–A–B tri-block copolymers
Xiaohang
Wang
ab,
Yingxue
Wang
b,
Shengxiao
Zhang
*a,
Zhengfeng
Ma
*bc and
Keling
Hu
 *bc
*bc
aSchool of Chemistry and Chemical Engineering, Ludong University, Yantai, Shandong 264025, China. E-mail: lduzsx@ldu.edu.cn
bShandong Laboratory of Advanced Materials and Green Manufacturing at Yantai, Yantai, Shandong 264006, P. R. China. E-mail: hukeling@amgm.ac.cn
cState Key Laboratory of Solid Lubrication, Lanzhou Institute of Chemical Physics, Chinese Academy of Sciences, Lanzhou, Gansu 730000, P. R. China. E-mail: mazhf@licp.cas.cn
First published on 20th March 2026
Abstract
Conventional liquid electrolytes in lithium-ion batteries suffer from safety concerns such as leakage, flammability, and lithium dendrite growth. To address these issues, microphase-separable linear B–A–B tri-block copolymers (PMMAn-b-PPEGMAm-b-PMMAn) were designed and synthesized via organocatalyzed living radical polymerization, and subsequently, solid polymer electrolytes (SPEs) were prepared by incorporating lithium bis(trifluoromethanesulfonyl)imide (LiTFSI) salt, where PMMA and PPEGMA are poly(methyl methacrylate) and poly(poly(ethylene glycol) methyl ether methacrylate), respectively. The metal-free polymerization strategy eliminates potential catalyst residues that may deteriorate electrochemical performance. The well-defined tri-block architecture enables microphase separation between the rigid PMMA and flexible PPEGMA segments, forming mechanically reinforced nanodomains and continuous Li+-conducting pathways, respectively. Atomic force microscopy (AFM) and scanning electron microscopy (SEM) analyses confirmed the formation of nanoscale phase-separated structures, while differential scanning calorimetry (DSC) and X-ray diffraction (XRD) results demonstrated the fully amorphous nature of the LiTFSI salt-containing solid polymer electrolyte (SPE) membranes. The optimized composition, PMMA30-b-PPEGMA19-b-PMMA30, exhibited a tensile strength of 38 MPa and an ionic conductivity of 6.08 × 10−6 S cm−1 at 70 °C. The temperature-dependent conductivity followed a non-Arrhenius behavior consistent with segmental-motion-assisted ion transport. Linear sweep voltammetry further demonstrated a wide electrochemical stability window up to approximately 5.4 V vs. Li/Li+. This well-defined linear B–A–B tri-block topological architecture enabled feasible microphase separation between PMMA and PPEGMA segments and provided an elegant combination of mechanical robustness and efficient Li+ conduction when an SPE is formed by the incorporation of LiTFSI salt into the block copolymer matrix.
Introduction
The increasing demand for renewable energy storage and electric transportation has accelerated the development of high-performance lithium-ion batteries (LIBs). Although liquid electrolytes offer high ionic conductivity, their flammability, leakage risk, and poor thermal stability raise significant safety concerns.1–4 In addition, uncontrolled lithium deposition in liquid systems often leads to dendritic growth, which can pierce the separator and trigger short circuits.5–8Solid polymer electrolytes (SPEs) provide safer and alternative candidates by combining mechanical toughness and efficient ionic conduction in LIBs.9–11 Among them, poly(ethylene oxide) (PEO) is the most widely studied, yet its strong crystallization at ambient temperature restricts chain mobility and limits lithium-ion (Li+) conductivity.1,12–14 Reducing the molecular weight of PEO can suppress crystallinity but compromises the tensile behaviour and solubility of Li+ salt, resulting in a weak mechanical framework and insufficient Li+ concentration.15–19 Thus, achieving both high conductivity and mechanical robustness remains a primary challenge.
Block copolymers offer promising candidates to overcome this trade-off between Li+ conductivity and mechanical robustness.16,20–22 By integrating rigid and flexible segments into a single macromolecular architecture, they allow independent tuning of mechanical properties and Li+ transportation.17,23 In particular, the rigid poly(methyl methacrylate) (PMMA) provides mechanical strength and modulus, while polyether-containing blocks, such as poly(poly(ethylene glycol) methyl ether methacrylate) (PPEGMA), create Li+-conducting pathways.13 A well-controlled linear tri-block topology is therefore desirable for constructing thermoplastic SPEs.
Recent studies have shown that the chemistry of the hard/soft blocks and the resulting microscopic morphology critically determine the trade-off between mechanical integrity and ion transport in methacrylate-based block-copolymer SPEs. For example, Bergfelt et al. reported the poly(benzyl methacrylate)-b-poly(oligo(ethylene glycol) methyl ether methacrylate)-b-poly(benzyl methacrylate) (PBnMA-b-POEGMA-b-PBnMA) tri-block copolymers blended with LiTFSI, where the addition of LiTFSI could trigger and tune microphase separation as evidenced by small-angle X-ray scattering analysis, leading to enhanced ionic conductivity and enabling a Li|SPE|LiFePO4 half-cell operation at 60 °C. In that system, replacing PMMA with PBnMA as the glassy mechanical block simultaneously changes block polarity, segmental dynamics, and salt–polymer interactions, which makes it difficult to decouple “architecture effects” (e.g., domain continuity and phase contrast) from “chemistry effects” originating from the specific hard-block structure and its interfacial affinity toward the ion-conducting domains.24
More recently, atom transfer radical polymerization (ATRP)-derived MMA/PPEGMA systems were formulated as dry SPEs, but the reported materials rely on comb-like (brush) PPEGMA rather than a linear B–A–B mid-block, implying distinct ion-coordination environments and phase connectivity compared with true linear tri-blocks. Therefore, establishing a structurally well-defined linear tri-block architecture is important for elucidating the intrinsic relationship between polymer topology, microphase separation, and ion-transport behavior.25
The organocatalyzed living radical polymerization technique, that is, reversible complexation-mediated polymerization (RCMP) developed by Goto et al.,26 enabled the metal-free synthesis of polymers with precise molecular weight (MW) control and narrow molecular weight distribution, also termed dispersity (Đ). RCMP is attractive because of no use of special capping agents or metal catalysts, ease of setup, applicability to a broad scope of monomers (e.g., methacrylates, acrylates, styrene, acrylonitrile, and even sterically crowded itaconates),27 and a diversity of polymer designs (e.g., linear, star, and brush).28–31
This technique avoids metallic residues that may interfere with electrochemical stability and provides synthetic flexibility for designing thermoplastic elastomer type SPEs based on block copolymers with various topologies (linear, star, or bottle-brush).32 In this study, a series of linear B–A–B tri-block copolymers, that is, PMMAn-b-PPEGMAm-b-PMMAn, with tunable block lengths and ratios were synthesized by RCMP. The two-terminal rigid PMMA blocks serve as a mechanical support and framework, whereas the middle PPEGMA block forms continuous Li+ conducting channels. Through systematic structural optimization (block ratio, MW), the relationship between polymer architecture and electrochemical performance was established.
Results and discussion
A linear B–A–B tri-block architecture, that is, PMMAn-b-PPEGMAm-b-PMMAn, was adopted because PMMA end blocks provide physical crosslinks enabling self-standing thermoplastic SPE membranes and suppressing creep without chemical crosslinkers or fillers, while a corresponding di-block copolymer typically lacks effective bridge formation between hard domains at comparable compositions. PEGMA with a number-average molecular weight (Mn, 300) was selected to balance salt solvation and segmental mobility while maintaining sufficient flexibility of the PPEGMA segments for Li+ conduction. The relatively short PEG side chains help maintain a low glass transition temperature (Tg) and facilitate Li+ coordination with ether oxygen sites, whereas longer PEGMA side chains (e.g., Mn = 500 or 950) may induce PEG-like crystallization and increase chain entanglement, which could hinder microphase separation and Li+ transport pathways. Based on this rationale, a series of PMMAn-b-PPEGMAm-b-PMMAn tri-block copolymers with controlled n/m values were synthesized and subsequently correlated with their thermal, morphological, and ionic transport properties.In this study, a series of macroinitiators (PPEGMA-II) with different MWs were first synthesized by polymerizing poly(ethylene glycol) methyl ether methacrylate (PEGMA, Mn = 300) using ethylene glycol bis(2-iodo-2-phenylacetate) (EPh-II) as the initiator and S-butyrylthiocholine iodide (S-BChI) as the catalyst via the organocatalyzed RCMP technique. The polymerization was carried out at 50 °C under a nitrogen atmosphere for different times to afford PPEGMA-II. Subsequently, using PPEGMA-II as the macroinitiator, a block extension reaction with methyl methacrylate (MMA) was performed at 60 °C to obtain the linear tri-block copolymers PMMAn–PPEGMAm–PMMAn (Scheme 1). The detailed synthetic procedures are provided in the SI of this article.
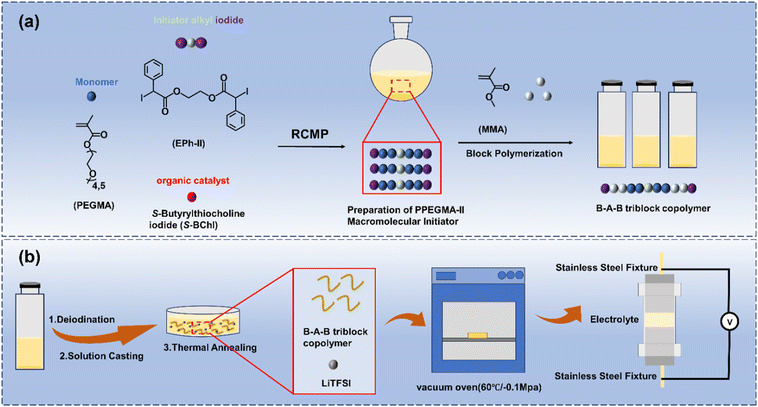 | ||
| Scheme 1 (a) The synthesis of PMMAn-b-PPEGMAm-b-PMMAn linear tri-block copolymers. (b) The preparation of a solid polymer electrolyte membrane from this linear tri-block copolymer. | ||
The gel-permeation chromatography (GPC) elution traces of PPEGMA-II and its related linear B–A–B tri-block copolymers are shown in Fig. 1a–c. It can be seen from the figures that all samples present symmetrical narrow peaks, with slight shoulder peaks, indicating the excellent block efficiency of PPEGMA-II and the successful block copolymerization with MMA. As the polymerization proceeded, the chain length of hard segments (PMMA) increased gradually, and the MWs of the final block copolymers increased accordingly. This is specifically reflected in the obvious left shift of the GPC curves by comparison before and after block polymerization, and polymers with larger MWs were eluted out first.33–35
Fig. 1d presents the proton nuclear magnetic resonance (1H NMR) spectrum of a representative tri-block copolymer, PMMA30–PPEGMA19–PMMA30, that was obtained by the copolymerization of PPEGMA-II (Mn = 6300; Đ = 1.19) and MMA by RCMP. As shown in Fig. 1d, the −OCH3 resonance peak at 3.40 ppm, the –OCH2CH2O– resonance peak at 3.67 ppm, and the −COOCH2− resonance peak at 4.08 ppm on PEGMA units together confirmed the existence of PPEGMA segments. In addition, the block ratio of the copolymer could be determined from the 1H NMR spectrum by integrating the characteristic peaks at 3.62 ppm, which is attributed to the –COOCH3 units in PMMA segments, and 4.08 ppm corresponding to the –COOCH2– units in PPEGMA segments. It is worth noting that the characteristic peak of vinyl protons in the PEGMA monomer disappeared in the range of 5.0–7.0 ppm, and a new methylene signal appeared at 0.8–2.0 ppm.36 These phenomena all indicated the successful synthesis of the PMMAm–PPEGMAm–PMMAm tri-block copolymer. The detailed integration regions, integration values, and step-by-step derivation process used to calculate the block ratios are provided in the SI (Fig. S1). This calculation is based on the stoichiometric evolution of the RCMP process, which provides reliable estimations of block lengths consistent with the molecular weight trends determined by GPC. The complete example of block length calculation is presented in Table 1.
| Entry | m | n | Block copolymer |
|---|---|---|---|
| S1 | 19 | 30 | PMMA30–PPEGMA19–PMMA30 |
| S2 | 19 | 39 | PMMA39–PPEGMA19–PMMA39 |
| S3 | 31.5 | 55 | PMMA55–PPEGMA31.5–PMMA55 |
| S4 | 31.5 | 59 | PMMA59–PPEGMA31.5–PMMA59 |
| S5 | 49 | 68 | PMMA68–PPEGMA49–PMMA68 |
| S6 | 49 | 87 | PMMA87–PPEGMA49–PMMA87 |
| S7 | 49 | 98 | PMMA98–PPEGMA49–PMMA98 |
The Fourier transform infrared (FTIR) spectra of the copolymers are exhibited in Fig. 1e. Their characteristic absorption peaks mainly correspond to three functional groups as explained in the following: C–H stretching vibrations in the range of 2800–3000 cm−1 (derived from methyl and methylene groups in PMMA and PPEGMA blocks), strong carbonyl stretching vibrations at around 1700–1750 cm−1 (from the ester groups of PMMA and PPEGMA blocks), and C–O–C stretching vibrations at 1000–1300 cm−1 (corresponding to the ether oxygen bonds in PPEGMA segments). The above characteristic evidence collectively confirmed the chemical structures of the linear B–A–B tri-block copolymers.
Fig. 2a–c show the surface morphology and elemental composition of the SPE membranes determined by field-emission scanning electron microscopy (FE-SEM) combined with energy dispersive X-ray spectroscopy (EDX) analysis, revealing a corrugated and rough surface morphology for the PMMA30–PPEGMA19–PMMA30 membrane. This topography originated from the microphase separation between the rigid PMMA segments and the flexible PPEGMA segments during membrane casting and thermal annealing.37 The PMMA domains tended to form ordered aggregates due to the chain rigidity, while the polyether-containing PPEGMA segments present complex molecular rearrangements driven by the chain flexibility. From the point of view of interfacial interactions, this textured morphology could enhance the electrode–electrolyte contact in solid-state batteries. Compared to smooth surfaces, the increased effective contact area minimized interfacial voids and gaps inherent in planar-to-planar contact interfaces. Crucially, reduced interfacial gaps eliminated additional ion-transport barriers (elongated migration paths, increased resistance), thereby facilitating the interfacial ion transfer and improving overall ionic conduction efficiency.38,39 EDX analysis demonstrated homogeneous distribution of carbon and oxygen across the membrane surface (Fig. 2c). At the molecular level, this uniformity arose from the following aspects: balanced elemental composition (C-rich PMMA via –COOCH3 groups; O-enriched PPEGMA via ether linkages); strong intermolecular interactions (van der Waals forces in PMMA; coordination of ether oxygens in PPEGMA); and nanoscale mixing during solvent casting. The uniform C/O distribution confirmed well-mixed block copolymer segments, ensuring continuous ion-conduction pathways through PPEGMA nanodomains. This structural uniformity prevented localized ion conduction bottlenecks or deficient regions, establishing an optimal chemical environment for efficient ion transport.
The corrugated morphology (Fig. 2a and b) optimized physical electrode contact, while homogeneous elemental distribution (EDX) guaranteed uniform ionic channels (Fig. 2c). Together, they demonstrated the structural advantages of the linear B–A–B tri-block copolymers for high-performance SPEs.
The 2D atomic force microscopy (AFM) height image is presented in Fig. 2d, revealing nanoscale surface topography. Driven by inherent chemical dissimilarities, the PMMA blocks (rigid, hydrophobic) and PPEGMA blocks (flexible, lithiophilic polyether moieties) underwent microphase separation during thermal annealing of the SPE membrane.40 This process generated distinct “protrusion–depression” nanostructures, that is, the PPEGMA-rich nanodomain appeared elevated (likely due to ether–oxygen interactions with the probing environment), while the PMMA-rich regions form depressions or plateaus. The quantified surface roughness (Ra = 3.01 nm) reflected the degree of microphase separation and bulk self-assembly, providing direct evidence of block architecture-directed morphological control. The 3D perspective shown in Fig. 2e elucidated correlations between nanoscale features and segmental dynamics. Local entanglement and ordering of the flexible polyether chains in PPEGMA segments produced characteristic “peak–valley” structures. Conversely, the rigid PMMA imposed confinement effects, yielding defined topographic features. This optimized nanoarchitecture critically enhanced electrolyte functionality. Primarily, the uniform surface undulations facilitated efficient Li+ transport along PPEGMA-conducting channels, while the PMMA mechanical integrity resisted lithium dendrite penetration. Collectively, these AFM analyses established a morphological foundation for structure–electrochemical performance correlations.
The thermal stability of a polymer depends on its backbone structure, bond strength and segmental mobility.41 Hard segments with rigid backbones generally show higher degradation temperatures, while soft and flexible segments degrade earlier. In block copolymers, the relative composition and phase arrangement further regulate heat resistance, and lithium salt coordination may introduce an additional effect.
These aspects govern the characteristic degradation features observed in thermogravimetric analysis (TGA). The thermal stability and segment decomposition behaviour of the block copolymers were measured by TGA, as presented in Fig. 3a. In the TGA curves, all samples present a slight mass loss (approximately 1%–4%) in the low-temperature region (<200 °C), which was mainly attributed to the evaporation of adsorbed moisture during storage or preparation. In the main decomposition stage (150–470 °C), the thermal decomposition behaviour was closely related to the segmental composition of the block copolymers. The PPEGMA segment, containing a large amount of ether oxygen bonds with relatively weak intermolecular forces, decomposed first in the range of 150–300 °C. The cleavage of ether oxygen bonds released small-molecule gases (such as low-carbon hydrocarbons and ether cleavage products), corresponding to a gradual mass loss in this temperature interval. When the temperature rose to 300–470 °C, the PMMA segments started to thermally decompose. The ester groups (–COO–) in PMMA underwent bond cleavage and elimination reactions at high temperatures, resulting in a significant mass loss of approximately 80%–90%.
 | ||
| Fig. 3 (a) TGA curves of the LiTFSI salt-free block copolymers under N2 flow. (b) DSC analysis of the LiTFSI salt-free block copolymers. | ||
For samples with different block lengths and ratios, the rigidity of the molecular chain increased with the length of PMMA blocks. Longer PMMA blocks could enhance the thermal stability of the molecular chain through intermolecular entanglement and van der Waals forces, increasing the initial decomposition temperature (Td) to approximately 250–300 °C. Overall, samples with higher PMMA content showed better thermal stability because a higher proportion of rigid PMMA segments could delay the thermal decomposition process. In contrast, samples with higher PPEGMA content exhibited faster decomposition rates, as the flexible PPEGMA segments tended to decompose at lower temperatures, resulting in a steeper decline in the TGA curves. These differences in thermal decomposition behaviour provided a basis for applications in high-temperature Li+ battery scenarios, where block copolymers with high PMMA content are more suitable for conditions requiring strict thermal stability.
Differential scanning calorimetry (DSC) thermograms of the LiTFSI salt-free tri-block copolymers are shown in Fig. 3b. All samples exhibited two distinct glass transition temperatures (Tg), with a low-temperature Tg (−40 to 0 °C) assigned to the PPEGMA-rich domains and a high-temperature Tg (+76 to +102 °C) corresponding to the PMMA-rich domains. The presence of two distinct Tg values, together with the single unimodal GPC traces, indicates that the PMMA and PPEGMA segments retain differentiated segmental dynamics and are at least partially microphase-separated on the DSC energy/time scale. This interpretation is further supported by the AFM and SEM observations, which reveal PMMA nanodomains dispersed within a soft PPEGMA matrix (vide infra), confirming that the observed behaviour arises from a well-defined block copolymer structure rather than from a simple physical mixture of two independent polymers.
Upon incorporation of LiTFSI, no additional thermal transitions were detected in the DSC thermograms, and the characteristic glass transition temperatures (Tg) of the block copolymer domains were largely preserved, indicating good miscibility of LiTFSI within the polymer host matrix. The second-heating DSC traces of the corresponding SPE membranes (Fig. S2) still exhibit two Tg values, corresponding to the PPEGMA-rich and PMMA-rich domains, respectively. However, the low-temperature Tg becomes more sample-dependent (Tg = −39 to −9 °C for S1–S7), reflecting the competition between Li+–ether coordination, which restricts chain mobility, and salt-induced plasticization within the PPEGMA-rich conducting phase. In contrast, the high-temperature Tg associated with the PMMA-rich domains remains nearly invariant for most samples, suggesting limited salt uptake or plasticization in the PMMA-rich phase and indicating that LiTFSI predominantly resides in the PPEGMA-rich domains.
Notably, no additional melting endotherms attributable to PEO-like crystallization were observed in the investigated temperature window after salt incorporation, implying that the electrolytes remain largely amorphous under these conditions.
The comparison of XRD patterns of the PMMAn–PPEGMAm–PMMAn tri-block copolymers before and after LiTFSI addition is shown in Fig. 4a and b. All pristine samples exhibited a broad amorphous halo centered at 2θ ≈ 35°–45°, originating from the atactic PMMA-rich matrix. The absence of sharp reflections confirmed that both PMMA and PPEGMA segments were fully amorphous, and no long-range ordered microdomains were present. This fully non-crystalline nature was consistent with the flexible polyether backbone of PPEGMA and the inherently amorphous characteristics of PMMA.
Upon incorporation of LiTFSI, no new crystalline peaks emerged, indicating that the salt was molecularly dispersed within the polymer and did not undergo phase separation. This behavior was essential for SPEs, as crystalline salt domains would impede ion mobility. Instead of introducing new reflections, salt addition subtly modified the shape and width of the amorphous halo. A slight broadening and intensity enhancement were observed for samples with longer PPEGMA blocks, reflecting increased disruption of local chain packing caused by Li+ coordination with ether oxygens. This coordination locally expanded the PPEGMA-rich domains and reduced short-range associations, consistent with improved segmental mobility.
In contrast, samples containing larger PMMA fractions showed only minimal changes in the halo profile, suggesting that the rigid PMMA domains dominated the scattering response and were less affected by salt incorporation. The overall amorphous character was maintained across all compositions, demonstrating that the tri-block architecture effectively suppressed crystallization of both the polyether segment and the lithium salt.
This comparative analysis revealed that LiTFSI primarily alters short-range packing within the PPEGMA domains while preserving the amorphous nature of the copolymer. Such behaviour was advantageous for ion transport, as homogeneous salt dispersion and increased free volume within polyether-rich regions promoted the formation of continuous ion-conducting pathways. These structural observations were fully consistent with the improved ionic conductivity observed in samples containing longer PPEGMA blocks.
The mechanical properties of SPE membranes are presented in Fig. 4c in the form of stress–strain curves. The tensile curves revealed the “rigidity–flexibility balance” of the SPE membranes. Within a wide strain range of 0–1200%, the membranes exhibited excellent deformability and stretchability, presenting the characteristics of elastomers. The rigid PMMA segments provided initial high modulus, leading to a rapid increase in stress at small strains (0–100%), endowing the material with certain deformation resistance. In contrast, the flexible PPEGMA segments functioned at large strains (beyond 200% strain) by stretching and sliding, imparting excellent ductility with elongation at break exceeding 1000%. For example, the SPE membrane from the PMMA30-b-PPEGMA19-b-PMMA30 tri-block copolymer showed a gentle stress rise curve and a final strength of 38 MPa, reflecting the balanced contribution of rigid PMMA domains and flexible PPEGMA chains, that is, PMMA and PPEGMA segments synergistically balanced the strength and toughness of the SPE membrane.
In summary, multi-dimensional analyses via TGA, DSC, XRD, and mechanical property tests revealed the thermal stability, segment movement, crystalline characteristics, amorphous structure, and mechanical behaviour of the SPEs based on thermoplastic elastomeric linear B–A–B tri-block copolymers. These properties are interrelated and synergistic with electrochemical performance (ionic conductivity, interface stability), providing comprehensive theoretical support and performance basis from thermal, structural, and mechanical aspects for the application of this material in solid-state lithium-ion batteries.
During charge–discharge cycles of solid-state batteries, electrodes undergo volume changes due to Li+ intercalation/extraction. The SPE needs good ductility to adapt to the deformation and avoid cracking. Meanwhile, moderate toughness can resist lithium dendrite penetration. The “rigidity–flexibility balance” enabled the SPE membranes to maintain interfacial stability (reducing poor interfacial contact caused by deformation) and inhibited lithium dendrites (via physical blocking by rigid segments), which, combined with electrochemical performance, collectively affects the overall performance of solid-state batteries.
The Li+ transport pathways in the PMMAn–PPEGMAm–PMMAn tri-block SPE membrane are shown in Scheme 2. Li+ ions migrate mainly through the PPEGMA-rich domains, where ether oxygen atoms provide repeated coordination sites that support a stepwise intra-chain hopping process. The carbonyl groups in the PMMA segments may also act as transient coordination sites and assist inter-domain transfer, although this contribution requires further verification.42–44
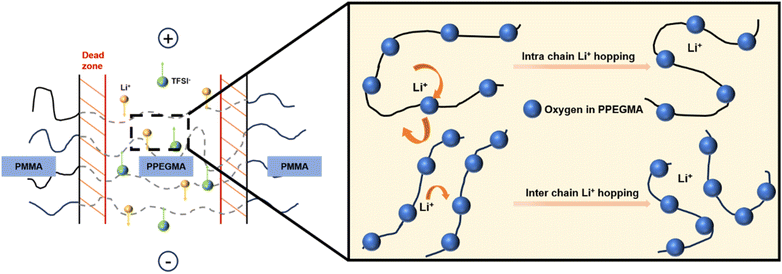 | ||
| Scheme 2 Li+ transport mechanism in the SPE membrane made from the linear PMMAn-b-PPEGMAm-b-PMMAn tri-block copolymer (Table S2, SI). | ||
Fig. 5 summarizes the temperature- and frequency-dependent ionic transport behaviour of the PMMAn-b-PPEGMAm-b-PMMAn SPE membranes. As shown in Fig. 5a, the ionic conductivity increases monotonically with increasing temperature for all compositions when plotted as log(σ) versus 1000/T, with the corresponding temperature (K) shown on the secondary X-axis for clarity. Notably, the log(σ) − 1000/T plots exhibit a slight upward curvature rather than a strictly linear Arrhenius dependence, suggesting non-Arrhenius transport characteristics.45,46 Such a curvature is commonly observed in polymer electrolytes where ion mobility is coupled to polymer segmental relaxation. The temperature dependence was therefore analysed using the Vogel–Tammann–Fulcher (VTF) formalism; the fitting procedure and extracted parameters are provided in the SI (Table S3).
Although the ionic conductivity of 6.08 × 10−6 S cm−1 at 70 °C is lower than that of liquid or gel electrolytes, it is comparable to previously reported PMMA-based or methacrylate-based block copolymer SPEs, which typically exhibit ionic conductivities in the range of 10−6–10−5 S cm−1 at elevated temperatures.11,47 This moderate conductivity reflects the intrinsic trade-off between mechanical robustness and ionic transport in block-copolymer SPE systems. In the present tri-block design, the rigid PMMA end blocks provide mechanical reinforcement and dimensional stability, while the PPEGMA mid-block forms the Li+-conducting phase, achieving a balance between mechanical robustness and ionic conductivity. Compared with many reported systems that exhibit higher conductivity but limited mechanical strength, the present tri-block architecture provides tensile strength up to 38 MPa and elongation at break exceeding 1000% (Fig. 4c). Such architecture prioritizes mechanical integrity and thermal stability while maintaining reasonable ionic transport capability.
To validate the conductivity extraction and clarify the frequency response, Fig. 5b presents the frequency-dependent AC conductivity, σac, for the representative membrane PMMA30-b-PPEGMA19-b-PMMA30 measured from 20 to 70 °C. The σac curves show a characteristic two-regime behaviour, that is, at higher frequencies, σac increases with frequency due to polarization relaxation and localized ion dynamics, whereas at lower frequencies, a near frequency-independent plateau is observed. The systematic upward shift of the plateau with increasing temperature confirms thermally enhanced ion transport, consistent with the segmental-motion-assisted conduction mechanism inferred from Fig. 5a.
The electrochemical stability window of the SPE was evaluated by linear sweep voltammetry (LSV) using a Li|SPE|stainless-steel cell at 70 °C (Fig. 6). The current density remains low and nearly constant over the investigated potential range below ∼5.4 V vs. Li/Li+, indicating negligible oxidative decomposition of the electrolyte. A pronounced increase in current is observed at higher potentials, which is attributed to the onset of oxidative decomposition of the polymer/salt matrix. Using the tangent-intersection method, the oxidative stability limit of PMMA30-b-PPEGMA19-b-PMMA30 is determined to be approximately 5.4 V vs. Li/Li+.
This relatively high oxidative stability can be ascribed to the presence of PMMA hard segments, whose carbonyl-containing framework provides improved resistance against oxidative degradation compared with typical PEO-based matrices. The stability window suggests that the present tri-block SPE is compatible with commonly used cathode materials operating below 4.3–4.5 V, while further evaluation under practical cell configurations is required for high-voltage cathodes.
Conclusions
In this work, the well-defined linear B–A–B tri-block copolymers, PMMAn-b-PPEGMAm-b-PMMAn, were successfully synthesized via the organocatalyzed and metal-free RCMP technique and employed as thermoplastic SPEs. The precisely controlled tri-block architecture enables microphase separation between the rigid PMMA and flexible PPEGMA segments, forming a mechanically reinforced framework and continuous Li+ conducting pathways.The resulting SPE membranes exhibit excellent mechanical robustness, with tensile strength up to 38 MPa and elongation at break exceeding 1000%, while maintaining an amorphous structure confirmed by XRD and DSC. Temperature-dependent conductivity analysis reveals non-Arrhenius behavior consistent with segmental-motion-assisted Li+ transport. For the optimized composition PMMA30-b-PPEGMA19-b-PMMA30, an ionic conductivity of 6.08 × 10−6 S cm−1 at 70 °C was achieved. Linear sweep voltammetry further demonstrates a wide electrochemical stability window up to approximately 5.4 V vs. Li/Li+, indicating compatibility with typical high-voltage cathode systems.
Overall, this study highlights that a rational B–A–B tri-block design offers an effective strategy to balance mechanical integrity and ionic transport in SPEs. The combination of thermoplastic processability, mechanical strength, and electrochemical stability provides valuable guidance for the development of structurally robust and high-performance SPE materials for lithium-based energy-storage applications.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: polymer synthesis and characterization methods, electrochemical property measurement and analysis, Tables S1–S3 and Fig. S1 and S2. See DOI: https://doi.org/10.1039/d6py00227g.Acknowledgements
This work was financially supported by the Research Funding of Shandong Provincial Laboratory of Advanced Materials and Green Manufacturing (Grant No: SYS202203), the Gansu Province Science and Technology Plan (Grant No: 24ZDWA003), the Major Science and Technology Projects of Gansu Province (Grant No: 22ZD6GA002), the Strategic Priority Research Program of the Chinese Academy of Sciences (Grant No: XDA0410301), the West Light Foundation of the Chinese Academy of Sciences, the LingChuang Research Project (Grant No: CNNC-LCKY-202285), the Science Fund of Shandong Laboratory of Advanced Materials and Green Manufacturing at Yantai (AMGM2024F29, E3R07SXM23), the Project of Synthesis and Characterization of High-Throughput Ion Permeation Membrane Resins (Grant No: E5R07QY002), and the Key Project of Yantai Natural Science Foundation (Grant No: ZR2025Z060).References
- K. Xu, Chem. Rev., 2004, 104, 4303–4418 CrossRef CAS PubMed.
- P. Arora and Z. Zhang, Chem. Rev., 2004, 104, 4419–4462 CrossRef CAS PubMed.
- X. Feng, M. Ouyang, X. Liu, L. Lu, Y. Xia and X. He, Energy Storage Mater., 2018, 10, 246–267 CrossRef.
- M.-K. Tran, A. Mevawalla, A. Aziz, S. Panchal, Y. Xie and M. Fowler, Processes, 2022, 10, 1192 CrossRef CAS.
- C. Monroe and J. Newman, J. Electrochem. Soc., 2005, 152, A396 CrossRef CAS.
- J. B. Goodenough and K.-S. Park, J. Am. Chem. Soc., 2013, 135, 1167–1176 CrossRef CAS PubMed.
- K. N. Wood, E. Kazyak, A. F. Chadwick, K.-H. Chen, J.-G. Zhang, K. Thornton and N. P. Dasgupta, ACS Cent. Sci., 2016, 2, 790–801 CrossRef CAS PubMed.
- H. An, Q. Liu, J. An, S. Liang, X. Wang, Z. Xu, Y. Tong, H. Huo, N. Sun, Y. Wang, Y. Shi and J. Wang, Energy Storage Mater., 2021, 43, 358–364 CrossRef.
- J. M. Tarascon and M. Armand, Nature, 2001, 414, 359–367 Search PubMed.
- X. Judez, H. Zhang, C. Li, G. G. Eshetu, Y. Zhang, J. A. González-Marcos, M. Armand and L. M. Rodriguez-Martinez, J. Phys. Chem. Lett., 2017, 8, 3473–3477 Search PubMed.
- D. Zhou, D. Shanmukaraj, A. Tkacheva, M. Armand and G. Wang, Chem, 2019, 5, 2326–2352 CAS.
- D. Mecerreyes, Prog. Polym. Sci., 2011, 36, 1629–1648 CrossRef CAS.
- J. L. Olmedo-Martínez, L. Meabe, A. Basterretxea, D. Mecerreyes and A. J. Müller, Polymers, 2019, 11, 452 CrossRef PubMed.
- V. St-Onge, M. Cui, S. Rochon, J.-C. Daigle and J. P. Claverie, Commun. Mater., 2021, 2, 83 CrossRef CAS.
- S. Lascaud, M. Perrier, A. Vallee, S. Besner, J. Prud'homme and M. Armand, Macromolecules, 1994, 27, 7469–7477 CrossRef CAS.
- M. Singh, O. Odusanya, G. M. Wilmes, H. B. Eitouni, E. D. Gomez, A. J. Patel, V. L. Chen, M. J. Park, P. Fragouli, H. Iatrou, N. Hadjichristidis, D. Cookson and N. P. Balsara, Macromolecules, 2007, 40, 4578–4585 CrossRef CAS.
- A. Panday, S. Mullin, E. D. Gomez, N. Wanakule, V. L. Chen, A. Hexemer, J. Pople and N. P. Balsara, Macromolecules, 2009, 42, 4632–4637 CrossRef CAS.
- K. Timachova, H. Watanabe and N. P. Balsara, Macromolecules, 2015, 48, 7882–7888 CrossRef CAS.
- D. Devaux, D. Glé, T. N. T. Phan, D. Gigmes, E. Giroud, M. Deschamps, R. Denoyel and R. Bouchet, Chem. Mater., 2015, 27, 4682–4692 CrossRef CAS.
- R. Bouchet, S. Maria, R. Meziane, A. Aboulaich, L. Lienafa, J.-P. Bonnet, T. N. T. Phan, D. Bertin, D. Gigmes, D. Devaux, R. Denoyel and M. Armand, Nat. Mater., 2013, 12, 452–457 CrossRef CAS PubMed.
- Z. Shao, H. Nederstedt and P. Jannasch, ACS Appl. Energy Mater., 2021, 4, 2570–2577 CrossRef CAS.
- T. Wang, L. Zhong, M. Xiao, D. Han, S. Wang, Z. Huang, S. Huang, L. Sun and Y. Meng, Prog. Polym. Sci., 2023, 146, 101743 CrossRef CAS.
- M. Chintapalli, K. Timachova, K. R. Olson, S. J. Mecham, D. Devaux, J. M. DeSimone and N. P. Balsara, Macromolecules, 2016, 49, 3508–3515 CrossRef CAS.
- A. Bergfelt, L. Rubatat, D. Brandell and T. Bowden, Solid State Ionics, 2018, 321, 55–61 CrossRef CAS.
- A. Szabo, D. Ershov, A. Abraham, E. Kiss, G. Szarka, I. Felhosi, B. Gyarmati, A. Domjan, B. Ivan and R. Kun, ACS Appl. Energy Mater., 2024, 7, 12036–12047 CrossRef CAS PubMed.
- A. Goto, T. Suzuki, H. Ohfuji, M. Tanishima, T. Fukuda, Y. Tsujii and H. Kaji, Macromolecules, 2011, 44, 8709–8715 CrossRef CAS.
- Z.-H. Chen, X.-Y. Wang and Y. Tang, Polym. Chem., 2022, 13, 2402–2419 RSC.
- W. Wang, L. Bai, H. Chen, H. Xu, Y. Niu, Q. Tao and Z. Cheng, RSC Adv., 2016, 6, 97455–97462 RSC.
- M. Huo, G. S. Tong, C. Y. Zhang and X. Y. Zhu, Macromolecules, 2020, 53, 9345–9352 CrossRef CAS.
- W. Mao, J. Sarkar, B. Peng and A. Goto, Polym. Chem., 2021, 12, 5770–5780 RSC.
- J. Sarkar, Y. F. Lim and A. Goto, Macromol. Chem. Phys., 2021, 222, 2100349 CrossRef CAS.
- K. Hu, S. Liu, Z. Zhao and Y. Zhang, Macromol. Rapid Commun., 2023, 44, 2300005 CrossRef CAS PubMed.
- D. Keddie, Chem. Soc. Rev., 2014, 43, 496–505 RSC.
- H. Feng, X. Lu, W. Wang, N. G. Kang and J. W. Mays, Polymers, 2017, 9, 494 CrossRef PubMed.
- K.-H. Wang, C.-H. Liu, D.-H. Tan, M.-P. Nieh and W.-F. Su, ACS Appl. Mater. Interfaces, 2024, 16, 6674–6686 CrossRef CAS PubMed.
- L. Yu, Y. Zhang, J. Wang, H. Gan, S. Li, X. Xie and Z. Xue, Macromolecules, 2021, 54, 874–887 CrossRef CAS.
- Y. Kambe, C. G. Arges, S. Patel, M. P. Stoykovish and P. F. Nealey, Electron. Sci. Interface, 2017, 26, 61 CrossRef CAS.
- L. Xu, S. Tang, Y. Cheng, K. Wang, J. Liang, C. Liu, Y.-C. Cao, F. Wei and L. Mai, Joule, 2018, 2, 1991–2015 CrossRef CAS.
- M. Liu, A. Song, X. Zhang, J. Wang, Y. Fan, G. Wang, H. Tian, Z. Ma and G. Shao, Nano Energy, 2025, 136, 110749 CrossRef CAS.
- S. Ma, Y. Hou, J. Hao, C. Lin, J. Zhao and X. Sui, Polymers, 2022, 14, 4568 CrossRef CAS PubMed.
- M. Guaita, O. Chiantore and L. Costa, Polym. Degrad. Stab., 1985, 12, 315–332 CrossRef CAS.
- M. Armand and J. M. Tarascon, Nature, 2008, 451, 652–657 CrossRef CAS PubMed.
- J. Fu, Z. Li, X. Zhou and X. Guo, Mater. Adv., 2022, 3, 3809–3819 RSC.
- J. Chattopadhyay, T. S. Pathak and D. M. F. Santos, Polymers, 2023, 15, 3907 CrossRef CAS PubMed.
- P. G. Bruce and C. A. Vincent, J. Chem. Soc., Faraday Trans., 1993, 89, 3187–3203 RSC.
- L. Ding, J. Shi, C. Yang and S. Dong, Polym. J., 1997, 29, 410–416 CrossRef CAS.
- A. Bergfelt, L. Rubatat, R. Mogensen, D. Brandell and T. Bowden, Polymer, 2017, 131, 234–242 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |


![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000
000 


