Open Access Article
Open Access ArticleElucidating the molecular design principles of N-alkylated nylons for LCST-type phase separation through a systematic polymer library
Akari
Sugano
a,
Keitaro
Matsuoka
 *ab and
Kazuki
Sada
*ab and
Kazuki
Sada
 *ab
*ab
aGraduate School of Chemical Sciences and Engineering, Hokkaido University, Kita 10, Nishi 8, Sapporo 060-0810, Japan
bDepartment of Chemistry, Faculty of Science, Hokkaido University, Kita 10, Nishi 8, Sapporo 060-0810, Japan. E-mail: sadatcm@sci.hokudai.ac.jp; ma2oka@sci.hokudai.ac.jp
First published on 8th April 2026
Abstract
Hydrophilic polymers showing LCST-type phase separation are useful functional polymer materials. In this study, we expanded the N-alkylated nylon library by varying the main-chain carbon numbers (p and q) and the N-alkyl side chains (R = ethyl, n-propyl, and isopropyl), and further designed branched and copolymer structures to systematically evaluate their effects on water solubility and LCST-type phase separation. We found that structural variations in the N-alkyl side chains, including linear and branched isomers with identical hydrophilic/hydrophobic balance, significantly influence water solubility, demonstrating that structural features permitting hydration of the amide groups are essential for molecular design of LCST-type phase separation. Moreover, backbone rigidity was identified as another important factor governing LCST-type phase separation. These findings provide a deeper understanding of the molecular design required for LCST-type phase separation and highlight N-alkylated nylons as a unique class of hydrophilic polymers exhibiting LCST-type phase separation.
1. Introduction
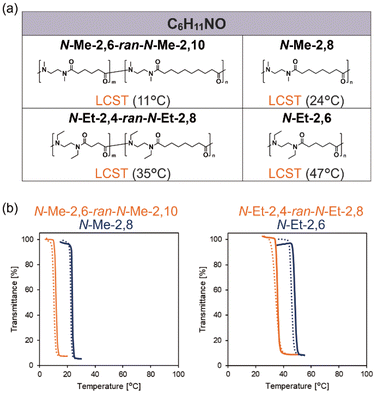
Hydrophilic polymers constitute a crucial class of polymer materials with widespread applications ranging from industrial and consumer products1–4 to biomaterials.5–9 They possess hydrophilic groups such as amide, hydroxyl, ether, carboxyl, phosphoester, and sulfoxide in their backbone or side chain, which hydrate through hydrogen bonding with water. At the same time, polymer structures inevitably incorporate hydrophobic groups such as alkyl chain, aromatic ring, and other less polar functionality such as ester and sulfur, which contribute to hydrophobic hydration, defined as the process of cage formation and structuring of water molecules around hydrophobic domains.10 Both groups allow for the expression of various functions depending on the extent of hydrophilicity and hydrophobicity. Among them, lower critical solution temperature (LCST)-type thermo-responsive polymers, represented by poly(N-isopropylacrylamide) (PNIPAM),11,12 have extensively been investigated for many years since they exhibit dramatic changes in water solubility upon heating or cooling around a temperature known as the cloud point.3,13–18 LCST-type phase separation arises from the conformational change of the polymer chains between the coil and globule state induced by hydration/dehydration. Owing to their promising potential for biomedical and smart material applications,19–22 LCST-type phase separation has been extensively explored in diverse polymer classes bearing hydrophilic functionalities, particularly amide-containing polymers such as polyacrylamides,11,12,23 poly(vinyl amide)s,24–26 poly(2-oxazoline)s,7,15,16,27,28 and polypeptoids.7,29–34 In these studies, adjusting hydrophilic/hydrophobic balance35 is essential for the molecular design of LCST-type phase separation, which is mainly demonstrated by tailoring the size of alkyl side chains.Recently, we developed N-methylated nylons as a new class of hydrophilic polymers.36 By tailoring the main-chain carbon numbers (p and q), we achieved continuous control over the aqueous solubility of N-methylated nylons. This approach explored the investigation of a wider range of the alkyl chain size than those of the similar studies of poly(2-oxazoline)s/poly(2-oxazine)s14 and poly(α-/β-peptoid)s.27 Through systematic synthesis and evaluation of these constitutional isomers with varying p and q, we identified N-methylated nylons with p + q = 10 as an optimal hydrophilic/hydrophobic balance for LCST-type phase separation. These polymers have a molecular formula of C6H11NO per amide unit, which is consistent with many LCST-type amide-containing polymers including PNIPAM.3 However, studies on poly(α-peptoid)s with different N-alkyl side chain demonstrated that C5H9NO is suitable for LCST-type phase separation.30 This suggests that factors beyond simple hydrophilic/hydrophobic balance may contribute to LCST-type phase separation, motivating the expansion of polymer libraries to systematically examine the effects of alkyl side-chain structure.
In this study, we construct an expanded library of N-alkylated nylons by independently tuned three structural variables, p and q in the main chain and R in the side chain. N-Alkylated nylons synthesized via A2 + B2 type polycondensation possess oppositely oriented amide groups, reflecting the inherent asymmetry between p and q.37–39 This molecular design strategy enables highly flexible arrangements of alkyl groups, resulting in a wide variety of constitutional isomers with an identical hydrophilic/hydrophobic balance. Therefore, comparison of the newly synthesized N-alkylated nylons with previously reported N-methylated nylons36 reveals more precise molecular design principle for LCST-type phase separation that extend beyond hydrophilic/hydrophobic balance. Accordingly, we primarily focus on N-alkylated nylons bearing N-ethyl and n-propyl side chains, as well as N-isopropyl substituent. In addition, the molecular design is extended to incorporate branched alkyl groups and copolymer systems containing randomly distributed larger and smaller hydrophobic alkyl group. Through a comprehensive evaluation of their LCST-type phase behavior, we aim to elucidate the structure–property relationships of N-alkylated nylons and establish their unique class as hydrophilic polymers exhibiting LCST-type phase separation (Fig. 1).
2. Results and discussion
We investigated N-ethylated and N-propylated nylons within the same molecular formula range as in our previous study.36 In total, fourteen N-ethylated nylons, N-Et-p,q (p = 2–4, q = 3–8), and four N-propylated nylons, N-nPr-p,q (p = 2, q = 3–6) were examined. These polymers have molecular formulas ranging from C5H9NO to C7H13NO per amide group, forming a library that allows direct comparison of their aqueous solubility with that of N-methylated nylons. All the N-alkylated nylons were synthesized via the polycondensation of corresponding diacid chlorides and diamines in the presence of triethylamine in dichloromethane at low temperature. The resulting polymers were characterized by 1H and 13C NMR analysis (SI section 3). The splitting of the proton signals for N–CH2 and O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CCH2 in both NMR spectra at room temperature revealed the existence of cis–trans conformations of the tertiary amide groups in solution. 1H–13C HMQC NMR measurements also supported the presence of multiple conformers. Size-exclusion chromatography (SEC) analysis revealed that N-alkylated nylons with number average molecular weights ranging from several thousand to tens of thousands were successfully obtained.
CCH2 in both NMR spectra at room temperature revealed the existence of cis–trans conformations of the tertiary amide groups in solution. 1H–13C HMQC NMR measurements also supported the presence of multiple conformers. Size-exclusion chromatography (SEC) analysis revealed that N-alkylated nylons with number average molecular weights ranging from several thousand to tens of thousands were successfully obtained.
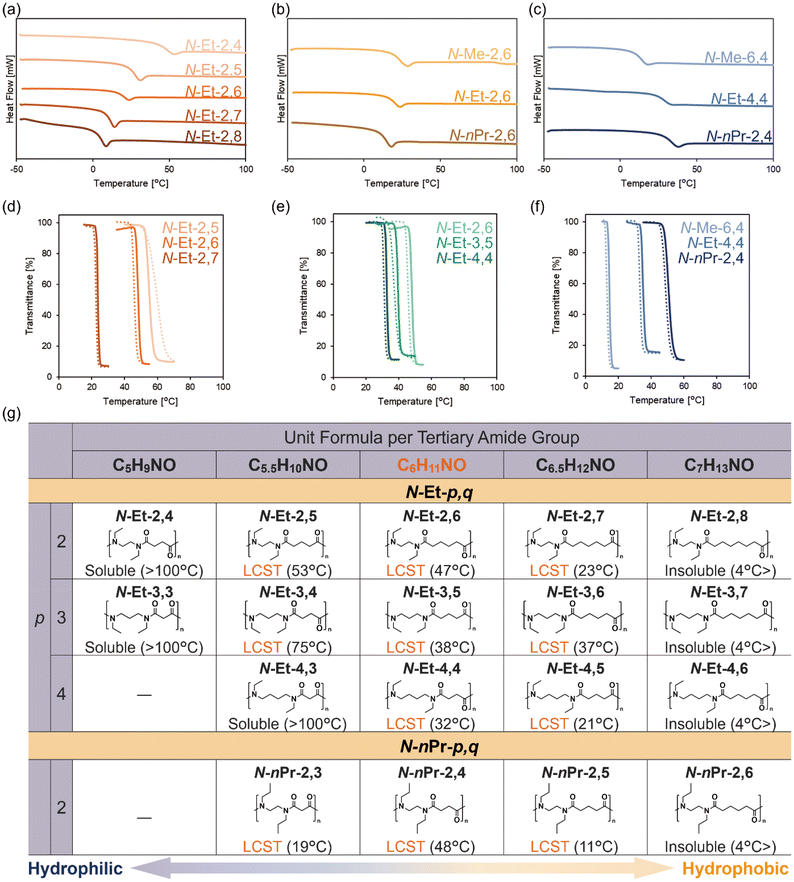
Thermal properties of the two series were investigated using differential scanning calorimetry (DSC). While conventional nylons with a secondary amide typically exhibit relatively high glass transition temperature (Tg) and high melting point (Tm),40 most of the synthesized N-alkylated nylons showed low Tg at around room temperature and no melting in higher temperature. Comparing the N-Et-2,q series, it was revealed that Tg decreases as the number of carbons in the main chain (q) increases (Fig. 2a), which is consistent with the similar trend observed in N-methylated nylons. Furthermore, when comparing Tg values among polymers with the same backbone structure N-R-2,6 (R = Me, Et, nPr), the order was found to be N-nPr-2,6 < N-Et-2,6 < N-Me-2,6 (Fig. 2b). This trend is attributed to increased mobility of the longer side chains, which suppresses intermolecular association of polymer chains. In contrast, a comparison of constitutional isomers N-Me-6,4, N-Et-4,4, and N-nPr-2,4 revealed a Tg order of N-Me-6,4 < N-Et-4,4 < N-nPr-2,4. This result indicates that the flexibility derived from the length of the alkyl chain in the backbone is a more dominant factor in determining Tg than the mobility of the side chain (Fig. 2c).
Solubility and thermo-responsive behavior in water were evaluated. Aqueous solutions of N-R-p,q (10 mg mL−1) were prepared and equilibrated at 4 °C for several days prior to measurement. Temperature-dependent transmittance change at 800 nm was measured using UV-vis spectroscopy, and the cloud point was defined as the temperature at which the transmittance decreased to 90% during heating. We initially investigated the N-Et-2,q (q = 4–8) series. Within 4–100 °C, N-Et-2,4 remained soluble (Tcp > 100 °C), whereas N-Et-2,8 was insoluble (Tcp < 4 °C). The intermediate polymers (N-Et-2,5, N-Et-2,6, and N-Et-2,7) exhibited LCST-type phase separation, with cloud points decreasing as the carbon number increased (Fig. 2d). The hysteresis between heating and cooling cycles is relatively smaller for N-Et-2,6, which has an optimal hydrophilic/hydrophobic balance, than for N-Et-2,5 and N-Et-2,7. Dynamic light scattering (DLS) measurements revealed a sharp increase in particle size at high temperatures, indicating aggregation of polymer chains and the emergence of LCST-type phase separation (SI section 5 and 6). Similar solubility trends were observed for the N-Et-3,q, N-Et-4,q, and N-nPr-2,q series, indicating that increasing alkyl chain length enhances hydrophobicity and lowers cloud point. N-Et-2,6, N-Et-3,5, and N-Et-4,4, which are constitutional isomers to each other with C6H11NO per amide unit among N-Et-p,q, exhibited LCST-type phase separation with small hysteresis within the relatively narrow range of 34–47 °C, close to body temperature (Fig. 2e). Among constitutional isomers (C6H11NO) bearing different N-substituents, N-Et-4,4 and N-nPr-2,4, with the previously reported N-Me-6,4,36 the cloud points increased in the order N-Me-6,4 < N-Et-4,4 < N-nPr-2,4 (Fig. 2f). This trend is likely due to the increasing number of carbon atoms between the tertiary amide groups, which results in larger hydrophobic domains and inhibits hydration. Variable-temperature 1H NMR measurements in D2O for N-Et-4,4 were performed, with all signals remaining observable above Tcp and showing only slight downfield shifts. These observations are consistent with partial dehydration of the polymer chains, leading to aggregation of aliphatic segments and subsequent coacervation (SI section 7). In addition, we examined the effect of polymer concentration (10–1.3 mg mL−1) for a series of N-R-p,q polymers with C6H11NO per amide unit (SI section 8). For all polymers, the cloud point gradually increased with decreasing concentration, consistent with typical LCST-type phase separation behavior. As an exception of these trends, N-nPr-2,3 exhibited LCST-type phase separation at a low cloud point despite the smaller alkyl size per amide unit. This behavior may be attributed to increased backbone rigidity, which reduces chain mobility and consequently lowers water solubility.
A summary of all eighteen synthesized N-alkylated nylons is provided in Fig. 2g. N-Alkylated nylons enable molecular designs in which hydrophobic alkyl groups can be freely positioned on both the main chain (p,q) and side chain (R) relative to the hydrophilic amide units. This feature provides a polymer library composed of multiple constitutional isomers with continuously tunable hydrophilicity. Consistent with our previous findings,36 these results demonstrate that a molecular formula of C6H11NO per amide unit provides the optimal hydrophilic/hydrophobic balance for LCST-type phase separation in water. In addition, backbone flexibility is another important factor in determining the water solubility of N-alkylated nylons, this structural feature also observed in poly(α-peptoid)s. Poly(α-peptoid)s possess relatively rigid backbones due to the short spacing between amide groups, which would lower their solubility in water. Consequently, inducing LCST-type phase separation in these polymers required increasing backbone hydrophilicity by reducing the number of methylene groups, corresponding to a per-amide molecular formula of C5H9NO.
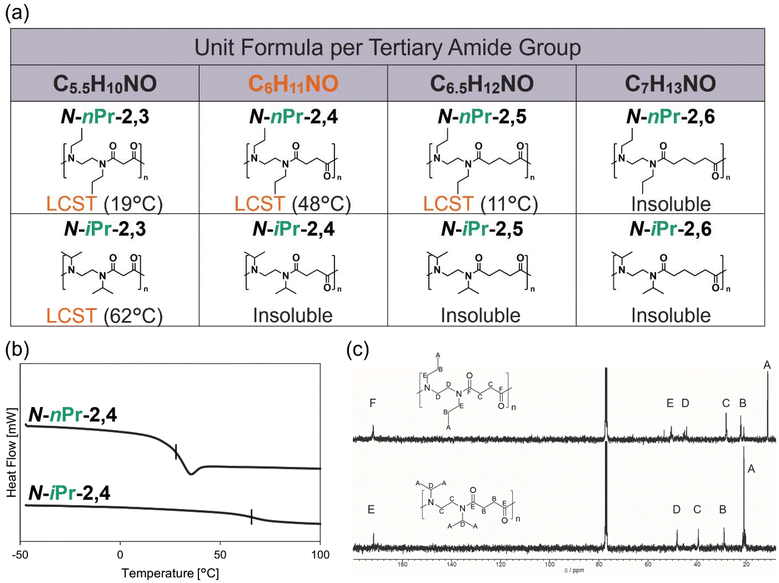
Because N-alkylated nylons examined thus far were limited to linear alkyl substituents, we next investigated N-isopropylated nylons bearing branched alkyl groups. N-Isopropylated nylons, N-iPr-2,q (q = 3–6) was synthesized via polycondensation, affording molecular weights comparable to those of the corresponding linear analogues (Fig. 3a). To evaluate water solubility, aqueous solutions of N-iPr-2,q (q = 3–6) were prepared at 10 mg mL−1, and N-iPr-2,3 exhibited LCST-type phase separation in water, consistent with the behavior of other N-alkylated nylons. In contrast, N-iPr-2,4 has the same molecular formula per amide unit (C6H11NO) as N-nPr-2,4, which was previously identified as optimal for LCST-type phase separation, yet it was insoluble in water over 4–100 °C. Further extension of the alkyl chain to N-iPr-2,5 and N-iPr-2,6 also remained insoluble in water. DSC measurements confirmed that N-iPr-2,4 exhibited a significantly higher Tg than N-nPr-2,4, indicating increased backbone rigidity (Fig. 3b). In addition, 13C NMR spectra of N-iPr-2,4 in CDCl3 showed no splitting of the carbon signals, suggesting slower cis/trans isomerization around the tertiary amide group and reduced conformers in the polymer chain in solution (Fig. 3c). The two neighboring bulky isopropyl groups increase the surface area of the hydrophobic domains, and the shorter alkyl group between the tertiary amide groups decreases the flexibility of the backbone. These two factors may decrease the water solubility of N-alkylated nylons. Because the isopropyl side chain is more hydrophilic than n-propyl group and enhances aqueous solubility in poly(α-peptoid)s,30 polyacrylamides,3 and poly(2-oxazoline)s,27,28 its markedly different influence here suggests a distinct interaction with the polymer backbone. Furthermore, in most thermo-responsive polymer classes, increasing the number of carbon atoms enhances hydrophobicity and leads to water insolubility.3,27,30 For N-alkylated nylons, water insolubility can be induced solely by the branching pattern of the alkyl side chain without changing the carbon number. This finding highlights a unique phenomenon arising from the structural characteristics of N-alkylated nylons. Thus, structural features permitting hydration of the amide groups are essential for the molecular design of LCST-type phase separation.
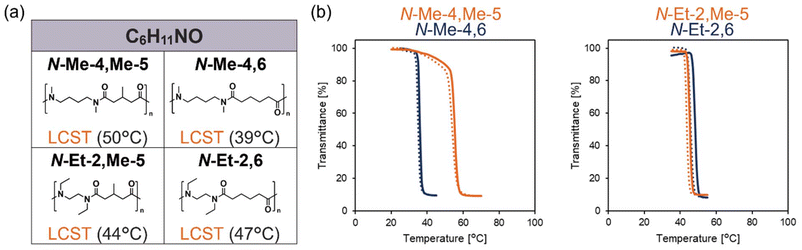
To further investigate the effects of branched alkyl group, we prepared N-alkylated nylons that contain a branched side chain between the tertiary amide groups. In the previous report on poly(2-oxazoline) and poly(2-oxazine) within the constitutional isomers, poly(2-alkyl-4-methyl-2-oxazoline)s with a methyl side chain on the backbone exhibits lower hydrophilicity than poly(2-alkyl-2-oxazine)s.28 Therefore, using the symmetric dicarboxylic acid monomer 3-methylglutaric acid (Me-5), we synthesized two polymers with a methyl side chain on the backbone, N-Me-4,Me-5 and N-Et-2,Me-5 (Mn = 6.1 × 103, 6.4 × 103). This design avoids head-to-head/tail-to-tail orientation in the backbone. These two polymers are the isomers with C6H11NO per amide group. The 13C NMR spectra showed peak splitting, indicating the presence of multiple conformers in solution, similar to those observed for N-alkylated nylons with a linear alkyl group. Both N-Me-4,Me-5 and N-Et-2,Me-5 exhibited LCST-type phase separation in water (10 mg mL−1) (Fig. 4a and b). Compared with their linear alkyl analogues, these branched polymers exhibited comparable or even enhanced solubility. This trend contrasts with that observed in the poly(2-oxazoline)/poly(2-oxazine) series. In poly(2-oxazoline)/poly(2-oxazine), introduction of a 4-methyl substituent in poly(2-oxazoline)s markedly increases backbone rigidity. In contrast, because N-alkylated nylons have longer spacing between amide groups, branching within the main chain does not substantially increase backbone rigidity and thus does not effectively reduce water solubility. In this case as well, unique behavior arising from the intrinsic structural feature of long amide–amide spacing in N-alkylated nylons was observed.
Finally, to further validate the conditions for LCST-type phase separation, we synthesized two 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 random copolymers with an average molecular formula of C6H11NO per amide unit. Random copolymerization, an established strategy widely used in the literature, enables a molecular design in which larger and smaller hydrophobic segments are randomly distributed along the polymer chain.35,41 One copolymer consisted of water-soluble N-Me-2,6 units and water-insoluble N-Me-2,10 units, resulting in an overall hydrophilic/hydrophobic balance equivalent to that of LCST-type thermo-responsive N-Me-2,8. Similarly, for the N-ethyl series, we synthesized a random copolymer of N-Et-2,4 and N-Et-2,8, resulting in a polymer with a composition equivalent to that of N-Et-2,6. The synthesis was performed by mixing the two corresponding dicarboxylic acid chlorides in a 1
1 random copolymers with an average molecular formula of C6H11NO per amide unit. Random copolymerization, an established strategy widely used in the literature, enables a molecular design in which larger and smaller hydrophobic segments are randomly distributed along the polymer chain.35,41 One copolymer consisted of water-soluble N-Me-2,6 units and water-insoluble N-Me-2,10 units, resulting in an overall hydrophilic/hydrophobic balance equivalent to that of LCST-type thermo-responsive N-Me-2,8. Similarly, for the N-ethyl series, we synthesized a random copolymer of N-Et-2,4 and N-Et-2,8, resulting in a polymer with a composition equivalent to that of N-Et-2,6. The synthesis was performed by mixing the two corresponding dicarboxylic acid chlorides in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio and adding the mixed dicarboxylic acid chlorides dropwise to the reaction solution (Scheme 1). 1H NMR analysis confirmed that the copolymers were synthesized with the intended 1
1 molar ratio and adding the mixed dicarboxylic acid chlorides dropwise to the reaction solution (Scheme 1). 1H NMR analysis confirmed that the copolymers were synthesized with the intended 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio, based on the integration values of the signals (Table 1). Both copolymers still showed LCST-type phase separation (10 mg mL−1) when its average composition per amide unit was C6H11NO, identical to that of the corresponding homopolymer (Fig. 5a and b). However, the copolymers showed lower cloud points than the corresponding homopolymers. Similarly, studies on poly(α-peptoid)s have demonstrated that random copolymers containing both water-soluble and water-insoluble units exhibit lower cloud point than the corresponding homopolymer with the same hydrophilic/hydrophobic balance.41 These findings indicate that water-insoluble units bearing larger hydrophobic groups influence the overall water solubility. Larger hydrophobic domains, for example those formed by contiguous water-insoluble units, suppress hydrophobic hydration, leading to decreased water solubility.
1 ratio, based on the integration values of the signals (Table 1). Both copolymers still showed LCST-type phase separation (10 mg mL−1) when its average composition per amide unit was C6H11NO, identical to that of the corresponding homopolymer (Fig. 5a and b). However, the copolymers showed lower cloud points than the corresponding homopolymers. Similarly, studies on poly(α-peptoid)s have demonstrated that random copolymers containing both water-soluble and water-insoluble units exhibit lower cloud point than the corresponding homopolymer with the same hydrophilic/hydrophobic balance.41 These findings indicate that water-insoluble units bearing larger hydrophobic groups influence the overall water solubility. Larger hydrophobic domains, for example those formed by contiguous water-insoluble units, suppress hydrophobic hydration, leading to decreased water solubility.
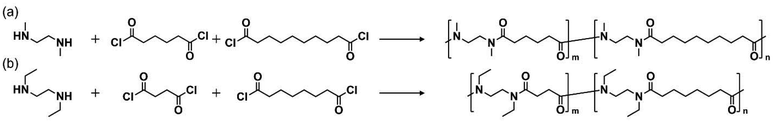 | ||
Scheme 1 Polymerization scheme for the synthesis of N-alkylated nylon 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 random copolymer. (a) N-Me-2,6-ran-N-Me-2,10. (b) N-Et-2,4-ran-N-Et-2,8. 1 random copolymer. (a) N-Me-2,6-ran-N-Me-2,10. (b) N-Et-2,4-ran-N-Et-2,8. | ||
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 random copolymer
1 random copolymer
m![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) n n |
M n | Đ | Solubility | ||
|---|---|---|---|---|---|
| Feed | Observed | ||||
| N-Me-2,6-ran-N-Me-2,10 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.03 1.03 |
1.9 × 104 | 1.6 | LCST |
| N-Et-2,4-ran-N-Et-2,8 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.04 1.04 |
5.5 × 103 | 1.5 | LCST |
We summarized a comprehensive list of all polymers synthesized in this study (Table 2). Comparison of N-alkylated nylons with other amide-containing polymers, including polyacrylamides, poly(2-oxazoline)s, and poly(α-peptoid)s, revealed that differences in water solubility originate from their structural features. These results suggest that hydration of the amide carbonyl groups is required for LCST-type phase separation. Regarding the hydrophilic–hydrophobic balance, polymers with sufficiently flexible backbones, such as N–alkylated nylons, polyacrylamides, and poly(2-oxazoline)s, exhibit LCST-type phase separation at an optimal composition of C6H11NO per amide unit. In contrast, poly(α-peptoid)s, which possess a more rigid backbone, show reduced water solubility and thereby require a more hydrophilic composition (C5H9NO) to exhibit LCST-type phase separation. Notably, the effect of backbone rigidity is also observed in N-alkylated nylons, polyacrylamides, and poly(2-oxazoline)s. Molecular designs that significantly increase rigidity, such as the introduction of methyl groups into the backbone, lead to a decrease in the cloud point. These findings demonstrate that systematic comparison of functional group arrangements in the repeating units of amide-containing polymers enables a clear understanding of the factors governing LCST-type phase separation and its associated cloud points.
| R | p,q |
M
n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a [g mol−1] a [g mol−1] |
Đ |
T
g![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b [°C] b [°C] |
T
cp![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c [°C] c [°C] |
|---|---|---|---|---|---|
| a Determined by SEC measurement. b Determined by DSC. c Determined from UV-vis spectroscopy at 10 mg mL−1. | |||||
| Et | 2,4 | 2.3 × 103 | 1.6 | 45 | >100 |
| 2,5 | 5.0 × 103 | 2.4 | 25 | 53 | |
| 2,6 | 5.1 × 103 | 1.7 | 18 | 47 | |
| 2,7 | 6.9 × 103 | 2.0 | 9 | 23 | |
| 2,8 | 1.4 × 104 | 1.7 | 3 | 4> | |
| 3,3 | 1.8 × 103 | 2.4 | 68 | >100 | |
| 3,4 | 1.9 × 103 | 2.1 | 46 | 75 | |
| 3,5 | 7.6 × 103 | 1.5 | 12 | 38 | |
| 3,6 | 4.0 × 103 | 1.6 | 6 | 37 | |
| 3,7 | 7.2 × 103 | 1.6 | 0 | 4> | |
| 4,3 | 3.3 × 103 | 2.3 | 54 | >100 | |
| 4,4 | 8.0 × 103 | 2.0 | 27 | 34 | |
| 4,5 | 1.0 × 104 | 1.7 | 6 | 21 | |
| 4,6 | 7.1 × 103 | 1.5 | −2 | 4> | |
| nPr | 2,3 | 1.4 × 103 | 1.7 | 55 | 19 |
| 2,4 | 2.1 × 103 | 1.1 | 29 | 48 | |
| 2,5 | 5.7 × 103 | 1.7 | 17 | 11 | |
| 2,6 | 7.4 × 103 | 1.7 | 12 | 4> | |
| iPr | 2,3 | 1.2 × 103 | 1.4 | 119 | 62 |
| 2,4 | 1.3 × 103 | 1.5 | 67 | 4> | |
| 2,5 | 4.9 × 103 | 1.5 | 55 | 4> | |
| 2,6 | 3.8 × 103 | 2.4 | 51 | 4> | |
| Me | 4,Me-5 | 6.4 × 103 | 1.8 | 23 | 50 |
| Et | 2,Me-5 | 6.1 × 103 | 1.6 | 36 | 44 |
| Me | 2,6-ran-2,10 | 1.9 × 104 | 1.6 | — | 11 |
| Et | 2,4-ran-2,8 | 5.5 × 103 | 1.5 | — | 35 |
3. Conclusion
In this study, we systematically investigated N-alkylated nylons bearing ethyl, n-propyl, and isopropyl groups as a N-alkyl side chains and significantly expanded the polymer library. In addition to linear alkyl groups, the molecular design was extended to branched alkyl structures as well as copolymers. By independently tuning the alkyl groups in both the main and side chains, this highly flexible molecular design allows for precise control over water solubility across a wide range of constitutional isomers. Systematic evaluation suggested that LCST-type phase separation in water is governed primarily by the water accessibility of the amide carbonyl groups. In addition to the optimal balance between hydrophilic and hydrophobic groups, we found that polymer chain rigidity is also an important molecular design guideline for achieving LCST-type thermo-responsiveness. Importantly, the orientation and long spacing between amide groups—structural characteristics intrinsic to N-alkylated nylons—result in solubility–structure relationships distinct from those of other polymer classes. These findings reinforce their position as a unique class of hydrophilic polymers exhibiting LCST-type phase separation. Future studies incorporating parameters such as molecular weight will provide a more comprehensive understanding of the hydration structure and precise control of the cloud points. With appropriate modification of substituents on both the main and side chains, N-alkylated nylons represent a promising class of hydrophilic polymers for applications including functional micelles and vesicles, microparticles, and hydrogels.Author contributions
The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript.Conflicts of interest
The authors declare no competing financial interest.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: experimental details, characterizations, and a summary of the survey on the research state of N-alkylated nylons to date. See DOI: https://doi.org/10.1039/d6py00191b.Acknowledgements
We acknowledge Prof. Y. Kageyama in HU for DSC measurement and Prof. K. Konishi in HU for DLS measurement.This work was supported in part by JSPS KAKENHI Grant Number JP21H01980 for K. S. and Promotion for Young Research Talent and Network from Northern Advancement Center for Science & Technology (NOASTEC) of Hokkaido Japan and the Kurita Water and Environment Foundation for K. M.
References
- P. A. Williams, Handbook of Industrial Water Soluble Polymers, Wiley, 2007, DOI:10.1002/9780470988701.
- N. Bayliss and B. V. K. J. Schmidt, Prog. Polym. Sci., 2023, 147, 101753, DOI:10.1016/j.progpolymsci.2023.101753.
- V. Aseyev, H. Tenhu and F. M. Winnik, Adv. Polym. Sci., 2010, 242, 29–89, DOI:10.1007/12_2010_57.
- M. S. Kamal, I. A. Hussein, A. S. Sultan and N. von Solms, Renewable Sustainable Energy Rev., 2016, 60, 206–225, DOI:10.1016/j.rser.2016.01.092.
- C. Weber, R. Hoogenboom and U. S. Schubert, Prog. Polym. Sci., 2012, 37, 686–714, DOI:10.1016/j.progpolymsci.2011.10.002.
- D. Roy, W. L. A. Brooks and B. S. Sumerlin, Chem. Soc. Rev., 2013, 42, 7214–7243, 10.1039/C3CS35499G.
- R. Hoogenboom and H. Schlaad, Polym. Chem., 2016, 8, 24–40, 10.1039/C6PY01320A.
- C. Zhao, Z. Ma and X. X. Zhu, Prog. Polym. Sci., 2019, 90, 269–291, DOI:10.1016/j.progpolymsci.2019.01.001.
- S. Nishimura and M. Tanaka, Bull. Chem. Soc. Jpn., 2023, 96, 1052–1070, DOI:10.1246/bcsj.20230168.
- W. Blokzijl and J. B. F. N. Engberts, Angew. Chem., Int. Ed. Engl., 1993, 32, 1545–1579, DOI:10.1002/anie.199315451.
- J. S. Scarpa, D. D. Mueller and I. M. Klotz, J. Am. Chem. Soc., 1967, 89, 6024–6030, DOI:10.1021/ja01000a006.
- A. Halperin, M. Kröger and F. M. Winnik, Angew. Chem., Int. Ed., 2015, 54, 15342–15367, DOI:10.1002/anie.201506663.
- C. Zhao, Z. Ma and X. X. Zhu, Prog. Polym. Sci., 2019, 90, 269–291, DOI:10.1016/j.progpolymsci.2019.01.001.
- M. A. C. Stuart, W. T. S. Huck, J. Genzer, M. Müller, C. Ober, M. Stamm, G. B. Sukhorukov, I. Szleifer, V. V. Tsukruk, M. Urban, F. Winnik, S. Zauscher, I. Luzinov and S. Minko, Nat. Mater., 2010, 9, 101–113, DOI:10.1038/nmat2614.
- O. Sedlacek and R. Hoogenboom, Adv. Ther., 2020, 3, 1900168, DOI:10.1002/adtp.201900168.
- A. Zahoranová and R. Luxenhofer, Adv. Healthcare Mater., 2021, 10, 2001382, DOI:10.1002/adhm.202001382.
- J.-F. Lutz, Ö. Akdemir and A. Hoth, J. Am. Chem. Soc., 2006, 128, 13046–13047, DOI:10.1021/ja065324n.
- J.-F. Lutz, J. Polym. Sci., Part A: Polym. Chem., 2008, 46, 3459–3470, DOI:10.1002/pola.22706.
- Y. Deng, T. Zou, X. Tao, V. Semetey, S. Trepout, S. Macro, J. Ling and M. H. Li, Biomacromolecules, 2015, 16, 3265–3274, DOI:10.1021/acs.biomac.5b00930.
- C. Pelosi, M. R. Tiné and F. R. Wurm, Eur. Polym. J., 2020, 141, 110079, DOI:10.1016/j.eurpolymj.2020.110079.
- M. A. Cruz, D. L. Morris, J. P. Swanson, M. Kundu, S. G. Mankoci, T. C. Leeper and A. Joy, ACS Macro Lett., 2018, 7, 477–481, DOI:10.1021/acsmacrolett.8b00118.
- D. Işik, E. Quaas and D. Klinger, Polym. Chem., 2020, 11, 7662–7676, 10.1039/D0PY01321H.
- K. Kolouchová, V. Lobaz, H. Beneš, V. R. de La Rosa, D. Babuka, P. Švec, P. Černoch, M. Hrubý, R. Hoogenboom, P. Štěpánek and O. Groborz, Polym. Chem., 2021, 12, 5077–5084, 10.1039/D1PY00843A.
- M. Akashi, S. Nakano and A. Kishida, J. Polym. Sci., Part A: Polym. Chem., 1996, 34, 301–303, DOI:10.1002/(SICI)1099-0518(19960130)34:2<301::AID-POLA16>3.0.CO;2-U.
- A. Laukkanen, L. Valtola, F. M. Winnik and H. Tenhu, Macromolecules, 2004, 37, 2268–2274, DOI:10.1021/ma035124l.
- Y. Maeda, T. Nakamura and I. Ikeda, Macromolecules, 2002, 35, 217–222, DOI:10.1021/ma011034.
- M. Glassner, M. Vergaelen and R. Hoogenboom, Polym. Int., 2018, 67, 32–45, DOI:10.1002/pi.5457.
- K. Mint, J. P. Morrow, N. M. Warne, X. He, D. Pizzi, S. Zainal, O. Shah, G. K. Pierens, N. L. Fletcher, C. A. Bell, K. J. Thurecht and K. Kempe, Polym. Chem., 2024, 15, 2662–2676, 10.1039/D4PY00332B.
- C. Fetsch, A. Grossmann, L. Holz, J. F. Nawroth and R. Luxenhofer, Macromolecules, 2011, 44, 6746–6758, DOI:10.1021/ma201015y.
- J. W. Robinson, C. Secker, S. Weidner and H. Schlaad, Macromolecules, 2013, 46, 580–587, DOI:10.1021/ma302412v.
- N. Gangloff, J. Ulbricht, T. Lorson, H. Schlaad and R. Luxenhofer, Chem. Rev., 2016, 116, 1753–1802, DOI:10.1021/acs.chemrev.5b00201.
- Z. Li, B. Cai, W. Yang and C.-L. Chen, Chem. Rev., 2021, 121, 14031–14087, DOI:10.1021/acs.chemrev.1c00024.
- M. Badreldin, P. Salas-Ambrosio, E. Garanger, S. Lecommandoux, S. Harrisson and C. Bonduelle, Prog. Polym. Sci., 2023, 147, 101752, DOI:10.1016/j.progpolymsci.2023.101752.
- D. Zhang, S. H. Lahasky, L. Guo, C.-U. Lee and M. Lavan, Macromolecules, 2012, 45, 5833–5841, DOI:10.1021/ma202319g.
- J.-F. Lutz and A. Hoth, Macromolecules, 2006, 39, 893–896, DOI:10.1021/ma0517042.
- A. Sugano, N. Inaba, K. Matsuoka and K. Sada, Macromolecules, 2012, 45, 5833–5841, DOI:10.1021/acs.macromol.5c01313.
- W. H. Carothers, US2130523A, 1938.
- W. H. Carothers, US2190770A, 1940.
- R. Hill and E. E. Walker, J. Polym. Sci., 1948, 3, 609–630, DOI:10.1002/pol.1948.120030501.
- A. Xenopoulos and B. Wunderlich, J. Polym. Sci., Part B: Polym. Phys., 1990, 28, 2271–2290, DOI:10.1002/polb.1990.090281209.
- X. Tao, J. Du, Y. Wang and J. Ling, Polym. Chem., 2015, 6, 3164–3170, 10.1039/c5py00191a.
| This journal is © The Royal Society of Chemistry 2026 |