Exploring thiol–ene and thiol–methacrylate reactions in the production of renewable materials from methacrylated eugenol
Gabriel Iago
dos Santos
 ,
Caroline
Gaglieri
,
Caroline
Gaglieri
 ,
Fernanda
Barreto dos Santos
,
Fernanda
Barreto dos Santos
 ,
Sabrina
Moises Moreno
,
Sabrina
Moises Moreno
 and
Gilbert
Bannach
and
Gilbert
Bannach
 *
*
São Paulo State University (UNESP), School of Sciences, Bauru, Brazil. E-mail: gilbert.bannach@unesp.br
First published on 20th March 2026
Abstract
Methacrylated eugenol (ME) is a promising biobased monomer for sustainable polymer networks due to its reactive carbon–carbon double bond and aromatic structure, which can enhance stiffness and thermal stability. Despite increasing interest in eugenol-derived polymers, systematic studies evaluating the relative contributions of thiol–ene and thiol–methacrylate reactions in ME-based systems, as well as their impact on material properties, remain scarce. In this work, eugenol was successfully methacrylated to obtain a versatile monomer containing two reactive sites, enabling polymerization via free-radical homopolymerization and thiol–ene/thiol–methacrylate pathways. Photocurable resins were formulated using ME, a type I photoinitiator, and pentaerythritol tetrakis(3-mercaptopropionate), with varying –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C/–SH molar ratios, to elucidate the theoretical extent of each polymerization mechanism. The resins exhibited high reactivity under UV irradiation, allowing the formation of renewable thermosets within seconds. Increasing the thiol content significantly promoted an increase in thiol–ene and thiol–methacrylate reactions, resulting in higher double-bond conversion, whereas lower thiol concentrations favoured homopolymerization and reduced overall conversion. These differences in reaction pathways strongly influenced the physicochemical properties of the resulting materials, yielding thermosets with tunable thermal stability, mechanical properties, and degradation profiles. Furthermore, the optimized formulation demonstrated suitable reactivity characteristics for vat photopolymerization 3D printing. Three-dimensional objects were successfully fabricated, exhibiting shape-memory behaviour and confirming the applicability of ME-based resins in additive manufacturing. Overall, this study establishes methacrylated eugenol as an active functional monomer rather than a simple reactive diluent, highlighting its potential for the development of sustainable, high-performance, and smart polymeric materials.
C/–SH molar ratios, to elucidate the theoretical extent of each polymerization mechanism. The resins exhibited high reactivity under UV irradiation, allowing the formation of renewable thermosets within seconds. Increasing the thiol content significantly promoted an increase in thiol–ene and thiol–methacrylate reactions, resulting in higher double-bond conversion, whereas lower thiol concentrations favoured homopolymerization and reduced overall conversion. These differences in reaction pathways strongly influenced the physicochemical properties of the resulting materials, yielding thermosets with tunable thermal stability, mechanical properties, and degradation profiles. Furthermore, the optimized formulation demonstrated suitable reactivity characteristics for vat photopolymerization 3D printing. Three-dimensional objects were successfully fabricated, exhibiting shape-memory behaviour and confirming the applicability of ME-based resins in additive manufacturing. Overall, this study establishes methacrylated eugenol as an active functional monomer rather than a simple reactive diluent, highlighting its potential for the development of sustainable, high-performance, and smart polymeric materials.
Introduction
Thermosetting polymers represent a significant class of materials widely used in industrial applications due to their exceptional thermal and mechanical properties and chemical stability, which originates from their permanent, three-dimensional cross-linked networks formed by covalent bonds.1 Thermosetting accounts for 18% of polymer production with an annual worldwide production higher than 65 million tons.1,2 However, the production of these materials is predominantly based on raw materials of fossil origin, which cannot be reused due to their irreversible cross-linking configurations, entails environmental impacts, and contributes to dependence on non-renewable resources.3 In this scenario, the use of biobased chemicals derived from renewable resources, such as plant-derived compounds, lipidic biomasses, and microorganisms, has attracted attention as a possible alternative to non-renewable resources. Many thermosetting materials have been produced from biobased feedstocks such as vegetable oils, organic acids, terpenes, and terpenoids.4,5 This strategy is also in line with the objectives of Green Chemistry, which seeks more sustainable processes with less environmental impact and greater efficiency in the use of resources. Therefore, when renewable feedstocks are used for polymer production, at least principles 4 (design of safe chemicals) and 7 (use of renewable feedstocks) are achieved, resulting in materials and processes with high sustainability metrics.6 Furthermore, the production of polymers with renewable origins contributes to the achievement of the Sustainable Development Goals (SDGs), especially those related to the promotion of sustainable technological innovations (9) and sustainable consumption and production (12).7,8Eugenol is a natural phenolic compound obtained mainly from various plants, including clove buds, cinnamon bark, and nutmeg, via the depolymerization or pyrolysis of lignin. Its relatively low cost makes eugenol a promising building block for polymer production.9 However, in the absence of chemical modification, eugenol is unsuitable for free-radical polymerization, as its allylic double bond exhibits low reactivity and the phenolic hydroxyl group acts as a radical scavenger, thereby inhibiting chain propagation and promoting premature termination reactions. Nevertheless, when a methacrylic group is incorporated into the eugenol structure by using the phenolic hydroxyl group, a reactive monomer for fast free radical chain polymerization (FRP) is obtained.10–12 This methacrylated eugenol (ME) can be obtained via a solvent-free and one-pot Steglich esterification reaction using methacrylic anhydride.10,11 Besides the renewable and sustainable character, the presence of a more reactive vinyl double bond in ME makes it suitable for photopolymerization reactions, which require less energy than thermal curing, since they can be performed at room temperature. Recent works demonstrate that methacrylated eugenol has been used primarily as a reactive diluent in photocurable formulations, with the aim of reducing the viscosity of resins and increasing the biobased content in the final material.13–15
Alongside conventional radical polymerizations of methacrylates, thiol–ene and thiol–acrylate type reactions stand out as efficient “click chemistry” routes for obtaining highly controlled polymer networks.16,17 The thiol–ene reaction occurs via radical addition between a thiol group (–SH) and a carbon–carbon double bond (–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C), promoting a predominantly additive step polymerization (ASP) mechanism, characterized by high functional conversion, low sensitivity to oxygen inhibition, and the formation of more homogeneous networks.18,19 Complementing this, thiol–acrylate polymerization combines the high reactivity of acrylate or methacrylate monomers with thiol-mediated chain transfer reactions, leading to hybrid polymerization systems in which chain polymerization and additive step polymerization mechanisms coexist. This dual character promotes the formation of more homogeneous polymer networks with reduced shrinkage stress and improved control over crosslink density.20
C), promoting a predominantly additive step polymerization (ASP) mechanism, characterized by high functional conversion, low sensitivity to oxygen inhibition, and the formation of more homogeneous networks.18,19 Complementing this, thiol–acrylate polymerization combines the high reactivity of acrylate or methacrylate monomers with thiol-mediated chain transfer reactions, leading to hybrid polymerization systems in which chain polymerization and additive step polymerization mechanisms coexist. This dual character promotes the formation of more homogeneous polymer networks with reduced shrinkage stress and improved control over crosslink density.20
Thiol–ene/meth(acrylate) reactions have been widely explored to modulate the thermal, mechanical, and structural properties of polymers, as they tend to produce networks with a more uniform cross-linking distribution, lower internal stress, and more predictable properties when compared to networks obtained exclusively by FRP of acrylates or methacrylates.16,21 In general, thiol–ene and thiol–acrylate systems usually employ two distinct types of monomers: a multifunctional thiol-containing compound and a second monomer bearing unsaturated groups (ene, acrylate, or methacrylate), which enables the adjustment of the resulting polymer properties.18,20 In this context, eugenol stands out as a versatile renewable building block, since its chemical structure contains both an allylic double bond and a methacrylate functionality, allowing it to participate in thiol–ene as well as thiol–acrylate reactions and, therefore, to be used for both purposes in the design of polymer networks.
In this scenario, methacrylated eugenol has high potential to act as the unsaturated component in thiol–ene and thiol–acrylate systems, since it has a reactive double bond and an aromatic structure capable of influencing the stiffness and thermal stability of the networks formed. Although there are studies involving eugenol derivatives in thiol–ene reactions to obtain biobased polymers,22–28 there are, to date, no systematic reports investigating the extent of thiol–ene and thiol–acrylate reactions specifically in ME, nor the direct influence of these reactions on the thermal and structural properties of the materials obtained from this monomer. Understanding these aspects is fundamental to positioning ME not only as a reactive diluent but also as an active functional monomer in the development of sustainable polymer networks with adjustable thermal and mechanical properties. Thus, in this work, eugenol was methacrylated to develop photocurable resins composed of this monomer, the type I photoinitiator 2,4,6-trimethylbenzoyl diphenylphosphine oxide, and the thiol pentaerythritol tetrakis(3-mercaptopropionate). Resin formulations with different –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C/–SH ratios were prepared to elucidate the relative extents of homopolymerization (driven by FRP) and the thiol–ene/methacrylate reaction (driven by ASP), and to investigate how these processes influence the thermal and mechanical properties of the resulting thermosets. In addition, the suitability of the resins for vat photopolymerization 3D printing was evaluated, and the optimized formulation was successfully used to fabricate 3D-printed objects with shape-memory behaviour, demonstrating the versatility of methacrylated eugenol beyond its role as a simple reactive diluent.
C/–SH ratios were prepared to elucidate the relative extents of homopolymerization (driven by FRP) and the thiol–ene/methacrylate reaction (driven by ASP), and to investigate how these processes influence the thermal and mechanical properties of the resulting thermosets. In addition, the suitability of the resins for vat photopolymerization 3D printing was evaluated, and the optimized formulation was successfully used to fabricate 3D-printed objects with shape-memory behaviour, demonstrating the versatility of methacrylated eugenol beyond its role as a simple reactive diluent.
Experimental
Materials
Eugenol (EU, 98%), methacrylic anhydride (MA, 94%, inhibited with 2000 ppm Topanol A), 4-dimethylaminopyridine (DMAP, 99%), pentaerythritol tetrakis(3-mercaptopropionate) (PETMP, 99%), dichloromethane (DCM, 99%), and 2,4,6-trimethylbenzoyl diphenylphosphine oxide (TPO, 97%), sodium hydroxide (NaOH, 97%), and hydrochloric acid (HCl, 37%) were purchased from Sigma-Aldrich and used as received without further purification. Sodium bicarbonate (NaHCO3, 99.7%) and anhydrous magnesium sulfate (MgSO4, 98%) were obtained from Dinâmica Química and used as received without further purification.Methacrylation of eugenol
The synthesis of methacrylated eugenol (ME) was carried out according to the literature,10 with only minor modifications. Briefly, the following components were added in sequence to a round-bottom flask: 100 g of EU, followed by 1.57 g of DMAP (2 mol% relative to the number of moles of MA), and 103.3 g of MA (1.1 molar equivalents based on eugenol). The reaction mixture was maintained under stirring at 45 °C for 24 h. After completion of the reaction, 100 mL of DCM was added to the crude product, and the system was washed several times and sequentially with different solutions: a saturated aqueous NaHCO3 solution, 1.0 mol L−1 NaOH aqueous solution, 0.5 mol L−1 NaOH aqueous solution, 1.0 mol L−1 HCl aqueous solution, and water, in order to remove traces of unreacted methacrylic anhydride and methacrylic acid. After phase separation, the organic phase was dried over MgSO4 and subsequently subjected to evaporation under reduced pressure to remove the solvent, yielding an orange product identified as methacrylated eugenol (ME). Yield: 98%. The synthetic route is summarized in Fig. 1. | ||
| Fig. 1 Summarized schemes for the methacrylation of eugenol, resin formulation, photopolymerization of the photocurable resins, and vat photopolymerization 3D printing. | ||
Resin formulation
In order to explore the structural features of ME for light-activated FRP and ASP, four resin formulations were prepared (Fig. 1): one containing only ME and three others containing different amounts of the polythiol PETMP. For the polythiol-containing resins, the proportions of S–H groups were varied while keeping the number of moles of methacrylate and allylic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C groups from ME constant, as shown in Table 1. In addition, a type I photoinitiator (TPO) was added to all systems at 3 wt% relative to the resin mass. All formulations were stirred for 20 min at room temperature to ensure homogenization.
C groups from ME constant, as shown in Table 1. In addition, a type I photoinitiator (TPO) was added to all systems at 3 wt% relative to the resin mass. All formulations were stirred for 20 min at room temperature to ensure homogenization.
| Resin name | C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C C![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) SH ratiosa SH ratiosa |
TPO amount/wt%b | Polymer name |
|---|---|---|---|
a C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C groups correspond to both methacrylic and allylic double bonds.
b Amount of TPO in relation to the resin mass (mixture containing ME + PETMP or ME only). C groups correspond to both methacrylic and allylic double bonds.
b Amount of TPO in relation to the resin mass (mixture containing ME + PETMP or ME only).
|
|||
| MER | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 0 |
3.0 | PMER |
| METH-0.25 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.25 0.25 |
3.0 | PMETH-0.25 |
| METH-0.5 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5 0.5 |
3.0 | PMETH-0.5 |
| METH-1 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
3.0 | PMETH-1 |
Production of thermoset materials and application in 3D printing
To obtain the polymeric materials via photopolymerization, a given amount of each resin was poured into a Teflon® mold and subsequently irradiated with ultraviolet light using a UV lamp (365 nm), model PR160 from Kessil (Emax = 399.0 mW cm−2), at an irradiation distance of approximately 2 mm between the lamp emission surface and the resin surface. The irradiation time for each system was 120 s. After evaluating the reactivity of each resin during the photopolymerization process, as well as their viscosities, the most suitable formulation was then applied in an Elegoo Mars 2 Pro DLP 3D printer equipped with a 405 nm light source (Emax = 3.8 mW cm−2) as shown in Fig. 1. The printed parts were detached from the build platform and subsequently washed in isopropyl alcohol, followed by a 30 min UV post-curing step (405 nm) using an Elegoo Wash & Cure Mercury Plus system. The printing parameters were adjusted using the Chitubox slicing software and are presented in Table 2.| Printing parameters | Settings |
|---|---|
| Layer height | 0.05 mm |
| Base layer amount | 25 |
| Exposure time for base layers | 70 s |
| Exposure time for the remaining layers | 60 s |
| Elevation after printing | 5 mm |
Characterization
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching band of the methacrylate and allylic groups at 1638 cm−1 (A1638). It should be noted that these double bonds give rise to overlapping bands, as discussed later in this work. All spectra were normalized by the area of the methylene C–H stretching band at 2937 cm−1 (A2937). Finally, the MC (%) values were calculated according to eqn (1), considering the time prior to UV exposure (t = 0) and the corresponding UV exposure time (t = x). All samples were analysed in triplicate.
C stretching band of the methacrylate and allylic groups at 1638 cm−1 (A1638). It should be noted that these double bonds give rise to overlapping bands, as discussed later in this work. All spectra were normalized by the area of the methylene C–H stretching band at 2937 cm−1 (A2937). Finally, the MC (%) values were calculated according to eqn (1), considering the time prior to UV exposure (t = 0) and the corresponding UV exposure time (t = x). All samples were analysed in triplicate.
The polymerization rate (Rp) was calculated using eqn (2), as reported in the literature,31 where ΔA is the difference between two areas related to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C band (methacrylate and allylic) at 1638 cm−1, A0 is the area of this band at time zero, and Δt is the time interval between the two analyses. All spectra were normalized as described above.
C band (methacrylate and allylic) at 1638 cm−1, A0 is the area of this band at time zero, and Δt is the time interval between the two analyses. All spectra were normalized as described above.
 | (1) |
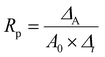 | (2) |
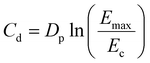 | (3) |
 | (4) |
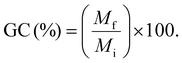 | (5) |
DSC measurements were performed using a DSC1 Stare system (Mettler-Toledo). Samples with masses of approximately 5 mg were placed in sealed aluminium crucibles with a perforated lid (40 μL). A heating rate of 20 °C min−1 and a dry air atmosphere with a flow rate of 50 mL min−1 were employed. The analyses were conducted over a temperature range from −30 °C to 150 °C.
For photo-DSC experiments, the same instrument was operated in isothermal mode at 25 °C, under a dry air atmosphere with a flow rate of 50 mL min−1. Approximately 5 mg of each sample was placed in open aluminium crucibles and irradiated using a Kessil KSPR160 lamp (365 nm) at 100% intensity (Emax = 399 mW cm−2). Two isothermal cycles of 120 s were performed for each sample, and the total enthalpy associated with photopolymerization (ΔHT) was determined by subtracting the second isothermal curve from the first, as reported in the literature.29,30
The polymerization rate (Rp) was calculated according to eqn (6) at the maximum of the exothermic polymerization peak, considering the heat flow under isothermal conditions at 25 °C (dH/dt).29 In addition, the time required for the reaction to reach the maximum energy release (peak time, PT) was obtained from the photo-DSC curves.
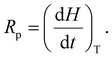 | (6) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ values. The glass transition temperature (Tg) was determined from the temperature at which the maximum loss factor (tan
δ values. The glass transition temperature (Tg) was determined from the temperature at which the maximum loss factor (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ) was observed.34
δ) was observed.34
The crosslink density (ν) of the materials was calculated using eqn (7),34 where T is the temperature in kelvin in the rubbery region (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ + 40 °C), E′ is the storage modulus in the rubbery region (determined at temperature T), and R is the gas constant (8.314 J mol−1 K−1).
δ + 40 °C), E′ is the storage modulus in the rubbery region (determined at temperature T), and R is the gas constant (8.314 J mol−1 K−1).
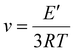 | (7) |
The tensile test was performed in the same equipment at 25 °C.
It should be noted that, due to the brittle nature of the PME sample, it could not be analysed by DMA.
Results and discussion
1H NMR and MIR analysis of the precursors and the product
The methacrylation of eugenol (EU) was carried out via a Steglich esterification reaction using methacrylic anhydride in the presence of the catalyst DMAP. The structures of methacrylated eugenol (ME) and its precursor EU were confirmed by 1H NMR, as shown in Fig. 2. In the EU spectrum (Fig. 2a), characteristic signals of the compound10,11 are observed, such as the singlet at a chemical shift of 8.73 ppm, which corresponds to the phenolic hydrogen and disappears completely after the reaction, as evidenced in the ME spectrum shown in Fig. 2b. In addition, new signals appear in the ME spectrum at 6.28 and 5.91 ppm, attributed to vinylic hydrogens, as well as a signal at 2.02 ppm, associated with the terminal methyl hydrogens of the methacrylic groups. | ||
| Fig. 2 1H-NMR spectra of (a) eugenol and (b) methacrylated eugenol. MIR spectra of (c) eugenol, (d) methacrylic anhydride, and (e) methacrylated eugenol. | ||
Fig. 2c–e present the MIR spectra of EU, methacrylic anhydride (MA), and ME, highlighting the relevant absorption bands. When comparing the spectrum of the product with those of the precursors, the characteristic carbonyl bands associated with the asymmetric (1780 cm−1) and symmetric (1720 cm−1) stretching of anhydride groups are no longer observed. Instead, a band at 1736 cm−1 appears, which is assigned to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching of ester groups.35 The disappearance of the O–H stretching band of the phenolic groups is also observed, indicating that the esterification reaction was successfully achieved. Furthermore, the magnified view of the normalized spectra (Fig. S1) shows that the intensity of the C
O stretching of ester groups.35 The disappearance of the O–H stretching band of the phenolic groups is also observed, indicating that the esterification reaction was successfully achieved. Furthermore, the magnified view of the normalized spectra (Fig. S1) shows that the intensity of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching band at 1638 cm−1 in ME increased compared to that of EU. Therefore, considering that the spectra were normalized, this increase suggests the overlap of methacrylic and allylic double-bond bands, which will be exploited in different polymerization reactions, as discussed later in this work.
C stretching band at 1638 cm−1 in ME increased compared to that of EU. Therefore, considering that the spectra were normalized, this increase suggests the overlap of methacrylic and allylic double-bond bands, which will be exploited in different polymerization reactions, as discussed later in this work.
Viscosity, biobased carbon content, and photopolymerization study
The viscosity of resins becomes a critical parameter when they are intended for specific applications such as vat photopolymerization 3D printing. In this context, viscosity is particularly important due to the need for resin flow from the internal cavities of the printed object and for resin replenishment into the vacant regions of the vat after the build platform is lifted. These conditions can directly affect the resolution of the printed objects. According to Guggenbiller et al.,36 the viscosity of resins used in stereolithography and digital light processing (DLP) 3D printing should remain within the range of 0.1 to 10 Pa s.The linear relationship between shear stress and shear rate observed for the analysed samples (Fig. S2) allowed the determination of the dynamic viscosity (μ) from the slope of the linear fit. As shown in Fig. S3 and Table 3, an increase in viscosity is initially observed after the methacrylation of eugenol, which can be attributed to the presence of carbonyl groups, leading to enhanced intermolecular interactions.37 Furthermore, an increase in the amount of PETMP resulted in higher viscosity values, as expected given the inherently high viscosity of this thiol compound. Although all resins exhibited viscosities below the recommended range, the METH-1 system presented the value closest to the lower recommended limit (μ = 74.0 ± 0.2 mPa s) for vat photopolymerization 3D printing.
| Sample | Viscosity/mPa s | BCC/% |
|---|---|---|
| EU | 8.3 ± 0.1 | 100 |
| ME | 16.2 ± 0.1 | 69 |
| METH-0.25 | 28.8 ± 0.1 | 73 |
| METH-0.5 | 43.8 ± 0.1 | 75 |
| METH-1 | 74.0 ± 0.2 | 79 |
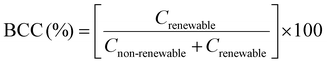
Regarding the biobased carbon content (BCC) (Table 3), all formulated resins exhibited values above 68%, which is associated with the renewable nature of the carbon atoms present in the monomers used. Eugenol is a constituent of several natural resources.9 Meanwhile, PETMP is a multifunctional thiol that has been considered potentially renewable in several studies,38–41 as it can be sourced from renewable feedstocks, as demonstrated by a retrosynthetic analysis conducted by Larsen et al.42 Therefore, the values achieved in this work allow all formulations to be classified as biobased materials, since a minimum biobased content of 25% is recommended for such classification.43–45
The reactivity of the resins was initially investigated in terms of double-bond conversion (MC) and the polymerization rate (Rp), as shown in Fig. 3. A clear reduction in the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C band at 1638 cm−1 (Fig. 3a–d) is observed during the photopolymerization process, which becomes more pronounced with increasing PETMP content in the formulation. This behaviour is more clearly visualized in the plot in Fig. 3e, where a significant increase in conversion values is observed after 20 s of reaction.
C band at 1638 cm−1 (Fig. 3a–d) is observed during the photopolymerization process, which becomes more pronounced with increasing PETMP content in the formulation. This behaviour is more clearly visualized in the plot in Fig. 3e, where a significant increase in conversion values is observed after 20 s of reaction.
In addition, at 10 s of irradiation, the MER and METH-0.25 resins exhibited similar and lower conversion values (56% ± 1 and 60% ± 1, respectively) compared to the METH-0.5 and METH-1 resins, which also showed comparable but higher conversion values (73% ± 3 and 74% ± 1, respectively). This trend is also reflected in the Rp plot shown in Fig. 3f. In this case, the MER resin exhibited the lowest polymerization rate (Rp = 0.056 s−1), whereas METH-1 showed the highest value (Rp = 0.074 s−1). For all systems, the reaction stabilized after 30 s, indicating that polymerization proceeds more intensely during the initial seconds of UV exposure, which is interesting for applications that demand fast polymerization, such as DLP 3D printing.
Therefore, based on these data and the results discussed later in this work, it is suggested that increasing the PETMP content promotes enhanced chain mobility, since the reactive sites of the thiol are spatially separated, leading to larger distances between crosslinking points, as already reported in the literature.33,46 These conditions allow greater diffusion of reactive species during polymerization, due to the longer time required for the system to reach the gel point or increase the viscosity, ultimately enabling higher C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C conversion values to be achieved.46,47
C conversion values to be achieved.46,47
Given the presence of both methacrylic and allylic alkene functionalities in ME, and in the presence of thiol groups, three polymerization pathways may occur simultaneously (Fig. 3g): additive step polymerization (ASP) of the thiol–ene type and thiol–methacrylate type, and free-radical chain polymerization (FRP).24,48,49 When acrylate or methacrylate groups react in the presence of thiols, both ASP and FRP mechanisms can coexist. In this context, a methacrylic radical may either propagate through addition to another methacrylate double bond (FRP) or undergo chain transfer by abstracting a hydrogen atom from a thiol group. This hydrogen abstraction generates a thiyl radical, which can subsequently propagate either by abstracting hydrogen from another thiol group or by adding to a methacrylate double bond.50
In this context, the extent of methacrylate homopolymerization and thiol–ene/methacrylate-type ASP reactions in each resin was evaluated based on the conversion of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C and S–H groups after photopolymerization (120 s of UV exposure). To determine the theoretical percentages of double bonds reacted via homopolymerization (C
C and S–H groups after photopolymerization (120 s of UV exposure). To determine the theoretical percentages of double bonds reacted via homopolymerization (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CHP) and ASP (C
CHP) and ASP (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CASP), the total amount (in moles) of alkenes and thiol groups present before and after the reaction was considered. The initial amounts of these groups were calculated considering the mass fraction of the ME monomer in each formulation (Table S1), while the reacted amounts were estimated from the corresponding conversion percentages (Table 4).
CASP), the total amount (in moles) of alkenes and thiol groups present before and after the reaction was considered. The initial amounts of these groups were calculated considering the mass fraction of the ME monomer in each formulation (Table S1), while the reacted amounts were estimated from the corresponding conversion percentages (Table 4).
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds involved in homopolymerization (C
C bonds involved in homopolymerization (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CHP) and in thiol–ene/methacrylated reactions (C
CHP) and in thiol–ene/methacrylated reactions (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CASP)
CASP)
For the calculation of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CASP, the amount of thiol groups reacted was first determined from their conversion and initial quantity. Assuming that each thiol group reacts stoichiometrically with one C
CASP, the amount of thiol groups reacted was first determined from their conversion and initial quantity. Assuming that each thiol group reacts stoichiometrically with one C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double bond, and that the formation of disulfides as a side reaction is negligible, the amount of thiol groups consumed directly corresponds to the amount of double bonds involved in ASP. Therefore, based on the total number of double bonds in the resin, the percentage of double bonds reacted via ASP was calculated according to eqn (8):
C double bond, and that the formation of disulfides as a side reaction is negligible, the amount of thiol groups consumed directly corresponds to the amount of double bonds involved in ASP. Therefore, based on the total number of double bonds in the resin, the percentage of double bonds reacted via ASP was calculated according to eqn (8):
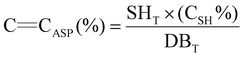 | (8) |
The fraction of double bonds consumed by homopolymerization (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CHP) was calculated using eqn (9), defined as the difference between the total number of reacted double bonds (DBR) and the number of double bonds consumed via ASP (DBASP), relative to the total number of double bonds (DBT).
CHP) was calculated using eqn (9), defined as the difference between the total number of reacted double bonds (DBR) and the number of double bonds consumed via ASP (DBASP), relative to the total number of double bonds (DBT).
The equations for determining these variables are shown in the SI (eqn (S1)–(S4)).
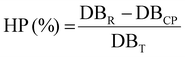 | (9) |
The conversion of thiol groups was determined by monitoring the area of the band at 2573 cm−1 during photopolymerization, which is attributed to S–H bond stretching, as shown in the magnified spectra of the METH-1 system in Fig. 3h and in the plot of S–H group conversion versus time presented in Fig. 3i. The magnified spectra of the METH-0.5 and METH-0.25 systems are shown in Fig. S4.
The conversion percentages of alkenes and thiol groups, as well as the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CHP and C
CHP and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CASP values for each resin, are presented in Table 4. It can be observed that the S–H conversion (SHConv.) values are similar for all resins. However, since the thiol content differs among the formulations, the C
CASP values for each resin, are presented in Table 4. It can be observed that the S–H conversion (SHConv.) values are similar for all resins. However, since the thiol content differs among the formulations, the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CHP and C
CHP and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CASP values exhibit a clear trend: homopolymerization reactions decrease with increasing thiol content, while thiol–ene/methacrylate reactions increase. This behaviour has already been reported by Li et al.20 They stated that the increase in thiol content led to a decrease in the mutual inhibition effect between ASP and FRP.20 Thus, based on the Cramer and Bowman findings for thiol–ene and thiol–acrylate reactions,50 we suggested that as the concentration of thiol groups in the system increases, a faster propagation of thiyl radicals is achieved, leading to an increase in the ASP reactions.
CASP values exhibit a clear trend: homopolymerization reactions decrease with increasing thiol content, while thiol–ene/methacrylate reactions increase. This behaviour has already been reported by Li et al.20 They stated that the increase in thiol content led to a decrease in the mutual inhibition effect between ASP and FRP.20 Thus, based on the Cramer and Bowman findings for thiol–ene and thiol–acrylate reactions,50 we suggested that as the concentration of thiol groups in the system increases, a faster propagation of thiyl radicals is achieved, leading to an increase in the ASP reactions.
In addition, the equimolar thiol![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ene system (METH-1) exhibits an S–H conversion of 75%, indicating the occurrence of thiol–methacrylate reactions in conjunction with thiol–ene, since values close to 50% would be expected if only allylic alkenes (thiol–ene) were involved in the reaction. This behaviour is expected, as the rate of addition of thiyl radicals to methacrylate double bonds is comparable to the rate of hydrogen abstraction from thiol groups by methacrylate radicals. Consequently, each hydrogen abstraction event that terminates a methacrylate radical simultaneously generates a thiyl radical capable of initiating the addition to another methacrylate double bond.50 Furthermore, in the MER resin, homopolymerization reactions involving allylic double bonds are also observed, as indicated by an MC value of 61% ± 1.
ene system (METH-1) exhibits an S–H conversion of 75%, indicating the occurrence of thiol–methacrylate reactions in conjunction with thiol–ene, since values close to 50% would be expected if only allylic alkenes (thiol–ene) were involved in the reaction. This behaviour is expected, as the rate of addition of thiyl radicals to methacrylate double bonds is comparable to the rate of hydrogen abstraction from thiol groups by methacrylate radicals. Consequently, each hydrogen abstraction event that terminates a methacrylate radical simultaneously generates a thiyl radical capable of initiating the addition to another methacrylate double bond.50 Furthermore, in the MER resin, homopolymerization reactions involving allylic double bonds are also observed, as indicated by an MC value of 61% ± 1.
All formulations were subjected to photo-DSC analysis to investigate the total enthalpy involved in photopolymerization (ΔHT), the polymerization rate (Rp) at the exothermic peak, and the time of the exothermic peak (PT). The results obtained for each resin are presented in Fig. S5 and Table S2. The same trend observed by MIR is also evident in this technique, namely that the MER resin exhibited the lowest ΔHT value, whereas the METH-1 resin showed the highest value. The differences observed in ΔHT values are associated with the gelation point, which is reached more rapidly with decreasing or the absence of the polythiol in the monomeric mixture, leading to incomplete polymerization conversion and, consequently, reduced energy release during the process.
When comparing the Rp values of the MER and METH-0.25 resins, an increase of 3 s−1 is observed upon incorporation of the polythiol into the formulation. This result indicates that the addition of this monomer at the corresponding proportion promotes higher conversion without significantly retarding the polymerization process, given that the homopolymerization rate is approximately 1.5 times higher than the rate of hydrogen abstraction from thiol groups in the ASP mechanism.49 On the other hand, the opposite effect is observed in the resins with higher PETMP contents (METH-0.5 and METH-1), as the difference in Rp values is 0.1 s−1, indicating a retardation process with increasing PETMP proportion.
TG-DTA, DSC, DMA, gel content, and degradation study
The TG/DTG-DTA curves of the materials are shown in Fig. 4a, while the data related to the temperature ranges (θ), mass loss values (Δm), peak temperatures (Tp), temperatures of the maximum degradation rate (TMDR), and maximum degradation rates (MDR) associated with each mass-loss stage are presented in Table S3. The thermal stability temperature (Tstability) of the polymers was significantly affected by the incorporation of the polythiol into the formulations. In this regard, the PMER material exhibited the lowest Tstability (144.0 °C), whereas the materials derived from PETMP-containing formulations showed an increase in this parameter with increasing polythiol content. Accordingly, Tstability followed the order: PMETH-1 > PMETH-0.5 > PMETH-0.25 > PMER. This trend may be attributed to the monomer conversion (MC), since higher conversion values can result in a greater extent of crosslinking and, consequently, enhanced resistance to structural degradation, thus requiring higher temperatures for degradation to occur.51 | ||
| Fig. 4 (a) TG/DTG-DTA, (b) DSC, (c) gel content, (d) relaxation, and (e) stress–strain results for the polymers produced. (f) Hydrolysis test of polymers under basic and acidic conditions. | ||
The mass-loss steps of the materials were also altered. In the case of the PMER, PMETH-0.25, and PMETH-0.5 samples, three steps were observed according to the TG curves. The first mass-loss stage was associated with the evaporation of the ME monomer, as evidenced by the decrease in mass variation with increasing monomer-to-polymer conversion; notably, this stage was no longer detectable in the PMETH-1 sample, which exhibited an MC of 99%. To support this hypothesis, a TG-DTA curve of ME was obtained (Fig. S6 and Table S4), showing that the compound evaporates above 78 °C. Although this temperature is significantly lower than the temperatures of the corresponding events in the polymer samples, it should be considered that the residual monomer within the polymer is trapped inside the polymer network. To validate this assumption, the PMETH-0.5 sample was heated to 180 °C, and the resulting condensate was collected and analysed by MIR spectroscopy. As shown in Fig. S7, the spectral profile closely resembles that of ME (Fig. 2e), corroborating the assignment of the first mass-loss step to ME evaporation.
The second and third mass-loss stages are associated with degradation of the polymer matrix, followed by decomposition and oxidation of the carbonaceous residue, as they correspond to exothermic events observed in the DTA curves. It is worth noting that the first and second mass-loss stages of the PMETH-1 sample are attributed to these same events, since this sample does not exhibit the step associated with ME evaporation. The absence of this stage indicates that no free thiol chains are present in the polymer structure, implying that at least one reactive site per structure has been involved in crosslinking.
During the polymeric matrix degradation step, analysis of the DTG curves reveals the presence of complex and overlapping thermal events in all samples after polythiol addition, with this separation being more pronounced in the sample with the highest polythiol content (PMETH-1). This behaviour suggests that degradation of the polymer matrix may occur over distinct temperature ranges for structures formed via ASP and homopolymerization. This hypothesis is supported by comparison between the PMER sample (absence of polythiol, with degradation attributed exclusively to homopolymerization) and the PMETH-1 sample (highest polythiol content and a high fraction of thiol–ene/methacrylate), considering the TMDR values associated with this mass-loss step. The PMER sample exhibits a TMDR of 430.0 °C, corresponding to degradation of the polymeric matrix resulting from homopolymerization, whereas the PMETH-1 sample shows a significantly lower TMDR (362.0 °C), followed by a second event with a TMDR value similar to that of PMER (431.0 °C). Thus, it is suggested that the lower-TMDR event can be attributed to degradation of the polymer matrix formed by thiol–ene/methacrylate reactions, while the higher-temperature event (∼430 °C) is associated with structures predominantly formed via homopolymerization.
The DSC curves of the polymers are shown in Fig. 4b and display the thermal events observed during the heating cycles (−30 to 150 °C). The highest glass transition temperature (Tg) was observed for the PMER sample, which can be attributed to the fact that ME is a small molecule containing an aromatic ring in its structure, enabling π–π stacking interactions. These interactions increase material rigidity and restrict molecular mobility, resulting in higher Tg values.52–54
Upon addition of the polythiol, a significant decrease in Tg was observed for the polymers. The PMETH-0.25 and PMETH-0.5 materials exhibited similar Tg values when compared to the variation observed for PMETH-1. This behaviour may be attributed to two combined factors. The first relates to the structural characteristics of PETMP, which is a large monomer containing long segments with weak interactions that keep crosslinking points farther apart, thereby allowing greater chain mobility, as already reported by Xue et al.22 The second factor is associated with a reduction in π–π stacking interactions, as the aromatic rings of the ME-derived segments become more spatially separated, as illustrated in Fig. S8.55 This effect is more pronounced in the PMETH-1 sample, given that thiol–ene/methacrylate reactions are dominant relative to homopolymerization reactions.
Gel content (GC) is a parameter that reflects the amount of crosslinked chains in the polymer network (degree of crosslinking), based on the insolubility of these chains in a given solvent, which, as a rule, must be capable of dissolving the precursor monomers. In this case, the data presented in Table 5 and Fig. 4c show the same trend observed for the monomer conversion (MC), namely, an increase in polythiol content led to an increase in GC. This behaviour indicates an increase in the degree of crosslinking of the materials, which is expected considering that PETMP provides four crosslinking points, whereas ME provides only two, one of which is less reactive (allylic double bond).20,22 This factor, together with the presence of the residual monomer due to the low MC, explains the lower GC value observed for PMER.
| PMER | PMETH-0.25 | PMETH-0.5 | PMETH-1 | |
|---|---|---|---|---|
| a The analysis could not be performed because the sample was brittle. b Maximum elongation and tensile strength were determined at the point of maximum force, as the samples did not break under the experimental conditions. c The sample ruptured under the experimental conditions. | ||||
| T stability/°C | 144.0 | 178.5 | 190.6 | 214.5 |
| T g/°C | 86.4 | 57.8 | 53.7 | 24.0 |
| GC/% | 64.2 ± 0.6 | 85.8 ± 0.6 | 91.8 ± 0.7 | 97.8 ± 0.5 |
| Tensile strength/MPa | —a | 3.8b | 3.5b | 0.7c |
| Strain/% | —a | 0.5b | 1.1b | 19.6c |
| Young modulus/MPa | —a | 910 | 430 | 100 |
tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ δ |
—a | 63.2 | 61.5 | 22.4 |
| ν/mol m−3 | —a | 1900 | 2274 | 3027 |
The DMA curves obtained from the relaxation tests are shown in Fig. 4d, and the corresponding data are summarized in Table 5. The tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ values are consistent with those obtained by DSC and follow the same trend previously observed for the PETMP-containing polymers. From these curves, the crosslink density values were determined. Accordingly, the PMETH-0.25 sample exhibited the lowest value (ν = 1900 mol m−3), whereas increasing the polythiol content in the formulation significantly increased this parameter, with the PMETH-1 sample presenting the highest value (ν = 3027 mol m−3). These results corroborate the increased extent of ASP reactions in the polythiol-containing systems, since PETMP has four crosslinking sites and the increase in its concentration enhances the availability of such sites, thereby leading to a higher crosslink density in the materials.20 Besides, the ν values obtained in this work are comparable to those of other eugenol-based photocurable resins.56
δ values are consistent with those obtained by DSC and follow the same trend previously observed for the PETMP-containing polymers. From these curves, the crosslink density values were determined. Accordingly, the PMETH-0.25 sample exhibited the lowest value (ν = 1900 mol m−3), whereas increasing the polythiol content in the formulation significantly increased this parameter, with the PMETH-1 sample presenting the highest value (ν = 3027 mol m−3). These results corroborate the increased extent of ASP reactions in the polythiol-containing systems, since PETMP has four crosslinking sites and the increase in its concentration enhances the availability of such sites, thereby leading to a higher crosslink density in the materials.20 Besides, the ν values obtained in this work are comparable to those of other eugenol-based photocurable resins.56
The stress–strain curves (Fig. 4e) show that the increase in thiol amount led to a decrease in the tensile strength and Young modulus, and an increase in the strain. The highest strain presented for PMETH-1 indicates the highest flexibility for this material, which can be attributed to the plasticizer effect of the unreacted thiol on the polymer network, as previously reported for Cramer and Bowman.50 Meanwhile, PMETH-0.25 and PMETH-0.5 are rigid materials.26 It is suggested that the behaviour shown by the materials analysed can be associated with the high impact of π–π stacking interactions on the mechanical properties. As the decrease in thiol content in the formulation led to an increase in homopolymerization, an approximation of the aromatic rings from ME is expected. This event can lead to an increase in π–π stacking interactions, resulting in an increase in the tensile strength exhibited by the materials, as already reported in the literature.57
A hydrolysis test was carried out on the samples containing polythiol in their formulations in order to evaluate the effect of varying polythiol content on the hydrolysis process under acidic and basic conditions. For this purpose, aqueous solutions of HCl and NaOH were used, both at a molar concentration of 1.0 mol L−1. As shown in Fig. 4f, at the initial time (t = 0 h), no changes were observed in the materials under either condition. After 24 h of immersion, no visual changes were observed for any material under acidic conditions, whereas under basic conditions, the PMETH-1 material completely degraded, and the PMETH-0.25 and PMETH-0.5 systems exhibited a slight change in the colour of the solution, indicating the onset of possible degradation. Therefore, it is evident that a higher polythiol content leads to a faster degradation process. This behaviour is directly related to the structural characteristics of PETMP, which contains four carbonyl groups susceptible to nucleophilic attack by the base, thereby increasing the number of hydrolysable sites and promoting more effective degradation. Thus, based on these initial tests, in terms of degradation efficiency for preventing environmental accumulation,58 the design of PMETH-1 is suitable, whereas in terms of resistance under severe conditions, materials with lower polythiol contents may provide a longer service life.
3D printing and shape memory testing
Since the resins containing polythiol exhibited high MC values and polymerization rates, they show potential for applications that require rapid polymerization. One such process is vat photopolymerization 3D printing, which enables the production of objects with specific and complex geometries. Prior to application, working curves of the METH-0.25, METH-0.5, and METH-1 resins were developed in order to determine the most suitable formulation. As illustrated in Fig. 5a and Table S5, the Ec values of the resins are similar, with the METH-0.25 resin presenting the lowest value, meaning that it requires less energy to reach the gel point. This occurs because this resin contains the lowest amount of polythiol and therefore reacts predominantly via homopolymerization, which, as previously mentioned, has a higher rate compared to ASP reactions via thiol–ene or thiol–methacrylate. Although a higher Ec value was expected for the METH-1 resin, it exhibited an intermediate value, which may reflect the simultaneous occurrence of different ASP and FRP reactions.20,33 | ||
| Fig. 5 (a) Working curves, (b) objects printed with METH-1 resin, and (c) qualitative shape memory test. | ||
The Dp values were higher for METH-0.5 and METH-1 resins, which can be attributed to the coloration of these resins. The addition of the polythiol, which is transparent, reduces the orange coloration of ME and, therefore, leads to different absorption capacities at the printer wavelength (405 nm).59
Based on the data obtained in this work, the resin METH-1 was considered the most suitable for 3D printing applications, since, in addition to having a low Ec value compared to other renewable methacrylated monomers,30 it exhibited high MC values, a viscosity closer to the recommended value, and a highly sustainable character, as it has a high BCC.
Based on the working curve data and to ensure the interfacial adhesion in the building plate, the printing parameters were determined as shown in Table 2. Fig. 5b shows the objects printed using the METH-1 resin. It can be observed that for smaller objects (object 3), an overcuring process inside the cavities is observed. The occurrence of overcuring can be attributed to the low viscosity of the resin, which facilitates the diffusion of reactive species into deeper regions of the layer, leading to undesired curing along the x/y axes during printing.60,61 For larger objects (objects 1 and 2), more well-defined shapes are obtained. Moreover, this represents an initial application proposal that can be further developed through the use of different photoinitiators and photosensitizers, which may allow a reduction in exposure time and thus optimize the printing process. Overall, these results demonstrate the applicability of this green resin in vat photopolymerization 3D printing processes.
For potential application as a shape memory material, an object was printed using the METH-1 resin and then subjected to a qualitative test, as shown in Fig. 5c. After being heated above Tg, the material was subjected to an external force to acquire a temporary shape, then cooled and reheated. The 3D-printed material exhibited a recovery time of 150 seconds for complete recovery of the permanent shape, with no visible damage or changes in physical appearance. This potential shape memory behaviour is attributed to the structural characteristics that form the polymeric network of PMETH-1. In this case, the long PETMP chains contribute flexible segments that allow rotational and conformational motions after heating above Tg, while the crosslinking points resulting from homopolymerization and thiol–ene/methacrylate act as “shape recovery” sites, leading to the “memory” effect.62 Based on this preliminary study, PMETH-1 indicates potential to be used as a renewable thiol–ene/methacrylate photopolymer for applications such as thermally actuated flexible active devices in fields of biomedicine and robotics.49,63 This finding opens new windows for quantitative studies of shape memory performance in order to contribute to smart renewable materials production.
Conclusions
The methacrylation of eugenol was successfully achieved, enabling the synthesis of a renewable and versatile monomer containing two reactive sites. This dual functionality allows polymerization to proceed via both free-radical chain polymerization (homopolymerization) and additive step polymerization (thiol–ene and thiol–methacrylate type). These two mechanisms were systematically investigated through the development of photocurable resin formulations with high biobased carbon content in the presence or absence of a polythiol and a type I photoinitiator.In addition to their bio-based content above 68%, the formulated resins exhibited high photoreactivity under UV irradiation, as the maximum value of Rp is reached after 10 s, enabling high and stable conversion values after 30 s. The relative extents of homopolymerization and thiol–ene/methacrylate reactions were evaluated in resins with different C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C and S–H molar ratios. Therefore, it was verified that increasing the thiol content led to a pronounced enhancement of ASP reactions and to higher C
C and S–H molar ratios. Therefore, it was verified that increasing the thiol content led to a pronounced enhancement of ASP reactions and to higher C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C conversion values, whereas lower thiol concentrations favoured homopolymerization and resulted in reduced conversion efficiency. Accordingly, the resin formulated with a stoichiometric alkene-to-thiol ratio polymerized predominantly through ASP, demonstrating that methacrylated eugenol is a robust and renewable building block for the production of networks resulting from thiol–ene/methacrylate reactions.
C conversion values, whereas lower thiol concentrations favoured homopolymerization and resulted in reduced conversion efficiency. Accordingly, the resin formulated with a stoichiometric alkene-to-thiol ratio polymerized predominantly through ASP, demonstrating that methacrylated eugenol is a robust and renewable building block for the production of networks resulting from thiol–ene/methacrylate reactions.
The influence of the polymerization pathway on the physicochemical properties of the resulting materials was also investigated. An increase in thiol content produced degradable thermosets with high thermal stability and lower glass transition temperatures. Moreover, owing to the dual polymerizable functionalities of methacrylated eugenol and the controlled extent of ASP reactions achieved using a tetrafunctional thiol, flexible materials with high gel content and crosslinking density were obtained within 120 s of UV exposure. Variations in the relative extents of homopolymerization and thiol–ene/methacrylated-type ASP reactions further resulted in thermosets with distinct tensile strengths and Young's modulus values, highlighting the versatility of methacrylated eugenol for tailoring material properties.
Finally, considering the advantageous characteristics of the METH-1 resin, including the highest Rp at 10 s, conversion values above 99% after 30 s, low critical energy, and a viscosity compatible with vat photopolymerization 3D printing, the formulation was successfully processed using a DLP 3D printer. Without additional optimization, the resin enabled the fabrication of renewable structures with complex geometries and potential shape memory behaviour. These results represent not only a meaningful advance toward greener additive manufacturing but also open new opportunities for employing this renewable monomer in the development of potential smart materials, including 4D-printed systems.
Author contributions
G. I. S.: conceptualization, data curation, formal analysis, funding acquisition, investigation, methodology, visualization, and writing – original draft. C. G.: formal analysis, investigation, methodology, and writing – original draft. F. B. S.: formal analysis, investigation, methodology, and writing – original draft. S. M. M.: formal analysis, investigation, methodology, and writing – original draft. G. B.: data curation, funding acquisition, supervision, and writing – review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: figures, tables, MIR spectra, and equations. See DOI: https://doi.org/10.1039/d6py00140h.Any raw data files in another format will be made available on request.
Acknowledgements
This study was financed, in part, by the São Paulo Research Foundation (FAPESP), Brazil, process numbers: 2024/14279-1; 2024/00779-2; 2025/25333-0, the National Council for Scientific and Technological Development – CNPq (grant 303968/2024-9), and the Brazilian Federal Agency for Support and Evaluation of Graduate Education – CAPES (grant 88887.146236/2025-00). The authors gratefully acknowledge Professor Éder Tadeu Gomes Cavalheiro for providing access to his laboratory and DMA equipment. The authors acknowledge the Multi-User Equipment Center of the Faculty of Sciences (CEMFC) and FAPESP (Proc. 09/14628-6 and 24/02935-1) for the MIR analyses.References
- Y. Liu, Z. Yu, B. Wang, P. Li, J. Zhu and S. Ma, Green Chem., 2022, 24, 5691–5708 RSC.
- B. Wang, Y. Wang, S. Du, J. Zhu and S. Ma, Mater. Horiz., 2023, 10, 41–51 RSC.
- M. A. Rashid, W. Liu, Y. Wei and Q. Jiang, Eur. Polym. J., 2022, 173, 111263 CrossRef.
- R. T. Alarcon, G. I. dos Santos, C. Gaglieri, A. de Moura, É.TG. Cavalheiro and G. Bannach, Chem. Commun., 2024, 60, 14557–14572 RSC.
- X. L. Zhao, P. X. Tian, Y. D. Li and J. B. Zeng, Green Chem., 2022, 24, 4363–4387 RSC.
- P. Anastas and N. Eghbali, Chem. Soc. Rev., 2009, 39, 301–312 RSC.
- A. Fleming, R. M. Wise, H. Hansen and L. Sams, Mar. Policy, 2017, 86, 94–103 CrossRef.
- THE 17 GOALS|Sustainable Development, https://sdgs.un.org/goals, (accessed 23 January 2026).
- S. Molina-Gutiérrez, A. Manseri, V. Ladmiral, R. Bongiovanni, S. Caillol and P. Lacroix-Desmazes, Macromol. Chem. Phys., 2019, 220, 1900179 CrossRef.
- Y. Zhang, Y. Li, L. Wang, Z. Gao and M. R. Kessler, ACS Sustainable Chem. Eng., 2017, 5, 8876–8883 CrossRef CAS.
- Y. Zhang, Y. Li, V. K. Thakur, Z. Gao, J. Gu and M. R. Kessler, Polym. Int., 2018, 67, 544–549 CrossRef CAS.
- A. Vittore, O. Santoro, M. Candida, S. Vaghi, S. Pragliola, M. Mella and L. Izzo, Polymer, 2025, 324, 128228 CrossRef CAS.
- H. Lai, J. Zhang and P. Xiao, ACS Sustainable Chem. Eng., 2023, 11, 16365–16406 CrossRef CAS.
- S. Brooks, D. Merckle and A. C. Weems, ACS Sustainable Chem. Eng., 2023, 11, 10252–10263 CrossRef CAS.
- Q. He, L. Hu, Y. Huang, T. Huang, Z. Zhu, Y. Li, Y. Hu and Z. Yang, Prog. Org. Coat., 2025, 200, 109079 CrossRef CAS.
- C. Schmidleitner, M. U. Kriehuber, R. Korotkov, S. Schlögl and E. Rossegger, Polym. Chem., 2025, 16, 963–971 RSC.
- C. E. Hoyle and C. N. Bowman, Angew. Chem., Int. Ed., 2010, 49, 1540–1573 CrossRef CAS PubMed.
- H. Okamura, M. Yamagaki and K. Nakata, Polymers, 2018, 11, 5 CrossRef PubMed.
- O. Ozukanar, E. Çakmakçi, G. Sagdic, U. S. Gunay, H. Durmaz and V. Kumbaraci, J. Polym. Environ., 2023, 31(7), 3259–3271 CrossRef CAS.
- X. Li, N. Ren, Y. Xiao, X. Li, M. Lang and X. Zhu, Macromolecules, 2024, 57, 9867–9876 CrossRef CAS.
- G. B. Coloma, C. E. Anderson, P. A. Vargas, K. Coyle, T. S. Kristufek, K. L. Mul, T. H. B. Nguyen, M. G. Siebecker and S. L. Kristufek, ACS Appl. Polym. Mater., 2025, 7, 16250–16261 CrossRef CAS.
- J. Xue, X. Yang, Y. Ke, Z. Yan, X. Dong, Y. Luo and C. Zhang, Ind. Crops Prod., 2021, 171, 113956 CrossRef CAS.
- R. Morales-Cerrada, S. Molina-Gutierrez, P. Lacroix-Desmazes and S. Caillol, Biomacromolecules, 2021, 22, 3625–3648 CrossRef CAS PubMed.
- P. Jia, M. E. Lamm, Y. Sha, Y. Ma, L. B. Kurnaz and Y. Zhou, Chem. Eng. J., 2023, 454, 140051 CrossRef CAS.
- O. Ozukanar, E. Çakmakçi, O. Daglar, H. Durmaz and V. Kumbaraci, Eur. Polym. J., 2023, 195, 112203 CrossRef CAS.
- J. Dai, Y. Jiang, X. Liu, J. Wang and J. Zhu, RSC Adv., 2016, 6, 17857–17866 RSC.
- M. Ge, Y. Li, L. Wu, R. Liu and J. T. Miao, Macromolecules, 2024, 57, 2394–2402 CrossRef CAS.
- T. Yoshimura, T. Shimasaki, N. Teramoto and M. Shibata, Eur. Polym. J., 2015, 67, 397–408 CrossRef CAS.
- G. I. dos Santos, C. Gaglieri, R. T. Alarcon, A. de Moura, F. B. dos Santos and G. Bannach, ACS Sustainable Chem. Eng., 2025, 13, 9771–9782 CrossRef CAS.
- R. T. Alarcon, A. Cellai, M. Porcarello, B. Sölle, E. Rossegger, C. C. Schmitt and M. Sangermano, ACS Sustainable Chem. Eng., 2025, 13, 16136–16153 CrossRef CAS.
- G. I. dos Santos, C. Gaglieri, R. T. Alarcon, R. Magri, M. Hartlieb and G. Bannach, J. Polym. Environ., 2024, 32, 4748–4762 CrossRef CAS.
- U. Shaukat, A. Thalhamer, E. Rossegger and S. Schlögl, Addit. Manuf., 2024, 79, 103930 CAS.
- R. T. Alarcon, A. Cellai, C. C. Schmitt, É. T. G. Cavalheiro and M. Sangermano, Eur. Polym. J., 2025, 235, 114068 CrossRef CAS.
- J. Dong, B. Liu, H. Ding, J. Shi, N. Liu, B. Dai and I. Kim, Polym. Chem., 2020, 11, 7524–7532 RSC.
- D. L. Pavia, G. M. Lampman, G. S. Kriz and J. R. Vyvyan, in Introduction to Spectroscopy, ed. D. L. Pavia, G. M. Lampman, G. S. Kriz and J. R. Vyvyan, Cengage Learning, 5th edn, 2014, vol. 1, pp. 15–126 Search PubMed.
- G. Guggenbiller, S. Brooks, O. King, E. Constant, D. Merckle and A. C. Weems, ACS Appl. Polym. Mater., 2023, 5, 3201–3229 CrossRef CAS.
- N. E. Rothfuss and M. D. Petters, Environ. Sci. Technol., 2016, 51, 271–279 CrossRef PubMed.
- M. K. Porwal, M. M. Hausladen, C. J. Ellison and T. M. Reineke, Green Chem., 2023, 25, 1488–1502 RSC.
- D. Guzmán, X. Ramis, X. Fernández-Francos, S. De la Flor and A. Serra, Prog. Org. Coat., 2018, 114, 259–267 CrossRef.
- D. Guzmán, X. Ramis, X. Fernández-Francos, S. De la Flor and A. Serra, Eur. Polym. J., 2017, 93, 530–544 CrossRef.
- M. Moreno, I. Armentano, E. Fortunati, S. Mattioli, L. Torre, G. Lligadas, J. C. Ronda, M. Galià and V. Cádiz, Eur. Polym. J., 2016, 79, 109–120 CrossRef CAS.
- D. B. Larsen, R. Sønderbæk-Jørgensen, J. Duus and A. E. Daugaard, Eur. Polym. J., 2018, 102, 1–8 CrossRef CAS.
- J. Dai, S. Ma, Y. Wu, J. Zhu and X. Liu, Prog. Org. Coat., 2015, 87, 197–203 CrossRef CAS.
- J. S. Terry and A. C. Taylor, J. Appl. Polym. Sci., 2021, 138, 50417 CrossRef CAS.
- F. K. Adom and J. B. Dunn, Biofuels Bioprod. Biorefin., 2017, 11, 258–268 CrossRef CAS.
- J. S. Young, A. R. Kannurpatti and C. N. Bowman, Macromol. Chem. Phys., 1998, 199, 1043–1049 CrossRef CAS.
- X. Li, N. Ren, Y. Xiao, X. Li, M. Lang and X. Zhu, Macromolecules, 2024, 57, 9867–9876 CrossRef CAS.
- L. Shahzadi, F. Maya, M. C. Breadmore and S. C. Thickett, ACS Appl. Polym. Mater., 2022, 4, 3896–3907 CrossRef CAS.
- U. Shaukat, B. Sölle, E. Rossegger, S. Rana and S. Schlögl, Polymers, 2022, 14, 5377 CrossRef CAS PubMed.
- N. B. Cramer and C. N. Bowman, J. Polym. Sci., Part A:Polym. Chem., 2001, 39, 3311–3319 CrossRef CAS.
- G. F. Levchik, K. Si, S. V. Levchik, G. Camino and C. A. Wilkie, Polym. Degrad. Stab., 1999, 65, 395–403 CrossRef CAS.
- P. G. Cortes, R. Araya-Hermosilla, K. Wrighton-Araneda, D. Cortés-Arriagada, F. Picchioni, F. Yan, P. Rudolf, R. K. Bose and F. Quero, Mater. Today Chem., 2023, 34, 101771 CrossRef.
- A. Rios de Anda, P. Sotta, T. Modjinou, V. Langlois, D. L. Versace and E. Renard, Macromolecules, 2020, 53, 2187–2197 CrossRef CAS.
- M. Di Consiglio, E. Sturabotti, B. Brugnoli, A. Piozzi, L. M. Migneco and I. Francolini, Polym. Chem., 2022, 14, 432–442 RSC.
- R. Xie, A. R. Weisen, Y. Lee, M. A. Aplan, A. M. Fenton, A. E. Masucci, F. Kempe, M. Sommer, C. W. Pester, R. H. Colby and E. D. Gomez, Nat. Commun., 2020, 11, 893 CrossRef CAS PubMed.
- J. Zheng, X. Huang, P. Liu, W. Wang and H. Fan, RSC Appl. Polym., 2025, 3, 1585–1595 RSC.
- B. Huang, S. Ai, J. Tan, B. Zhou, Y. Pan, Z. Zheng, X. Ding and J. Deng, ACS Appl. Eng. Mater., 2025, 3, 2176–2189 CrossRef CAS.
- F. M. Kerton, Curr. Opin. Green Sustainable Chem., 2025, 51, 100996 CrossRef CAS.
- C. Vazquez-Martel, L. Becker, W. V. Liebig, P. Elsner and E. Blasco, ACS Sustainable Chem. Eng., 2021, 9, 16840–16848 CrossRef CAS.
- M.-S. Ko and D.-S. Shim, Def. Technol., 2026 DOI:10.1016/j.dt.2026.01.010.
- M. Par, P. Burrer, K. Prskalo, S. Schmid, A. L. Schubiger, D. Marovic, Z. Tarle, T. Attin and T. T. Tauböck, Polymers, 2022, 14, 3296 CrossRef CAS PubMed.
- Y. Xia, Y. He, F. Zhang, Y. Liu and J. Leng, Adv. Mater., 2021, 33, 2000713 CrossRef CAS PubMed.
- V. Bijalwan, S. Schlögl and S. Rana, RSC Adv., 2025, 15, 49714–49727 RSC.
| This journal is © The Royal Society of Chemistry 2026 |

