Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Degrafting reaction of polymer brushes: the determining role of molecular weight and grafting density
Riccardo
Chiarcos
 *a,
Diego
Antonioli
a,
Luca
Gabutti
a,
Elo
Marsengo
a,
Michele
Perego
*a,
Diego
Antonioli
a,
Luca
Gabutti
a,
Elo
Marsengo
a,
Michele
Perego
 b and
Michele
Laus
a
b and
Michele
Laus
a
aDepartment of Science and Technological Innovation (DISIT), Università del Piemonte Orientale (UPO), Alessandria 15121, Italy. E-mail: riccardo.chiarcos@uniupo.it
bInstitute for Microelectronics and Microsystems (IMM), National Research Council of Italy (CNR), Agrate Brianza 20864, Italy
First published on 7th April 2026
Abstract
The chemical stability of polymer brushes is mandatory to ensure their applicability in a wide range of environments. However, it is well-known that, in the presence of water, polymer chains detach from the substrate due to the hydrolytic cleavage of the anchoring bonds at the polymer–substrate interface. Furthermore, this degrafting reaction is promoted by the tension generated on the labile anchoring bonds by the swelling of the brush. It is now widely demonstrated that the brush tension increases, speeding up the degrafting reaction, as the number of chains per unit area (grafting density) increases. In contrast, the role of the molecular weight of the grafted polymer in the degrafting reaction is much less investigated. In this work, polymer brushes made up of polystyrenes with different molecular weights were obtained by the grafting to approach. Chain degrafting was then induced by a THF/water mixture and the progress of the degrafting reaction was followed by thickness measurements on the dry brushes. The reaction rate constant of the degrafting process was calculated as a function of both the grafting density and the molecular weight of the polymer, confirming that both parameters have a significant role. Furthermore, the degrafting reaction was also evaluated in brushes containing long and short chains in different percentages. A reciprocal influence was observed between the two components, with the overall degrafting process being precisely dictated by the predominant one.
Introduction
Adaptable and programmable surfaces are highly required in all those applications in which the interaction between a material and the surrounding environment is critical.1 Among the tools of surface engineering, polymer brush technology is certainly the most promising.2–4 In fact, polymer brushes, which consist of polymer chains chemically anchored by one end to a substrate, are currently evaluated for a broad range of applications such as sensing,5–7 electronics,8–12 membranes,13,14 adhesives,15,16 energy devices,17 lubricants,18–21 biocompatible coatings22,23 and bioscience.3,24–26Polymer brushes are generally obtained through two distinct approaches: grafting from and grafting to.4,27 In grafting from, the substrate is initially reacted with a functional initiator and then the polymer chains are grown from the substrate dipped in a monomer solution.28 Although several polymerization techniques are suitable for the grafting from approach, the most common is undoubtedly the Surface Initiated-Atom Transfer Radical Polymerization (SI-ATRP), which allows several monomers to be polymerized and leads to polymers with controlled molecular weights and relatively narrow weight distributions.29–34 The grafting from approach is certainly the most suited for the synthesis of brushes with high grafting densities (Σ, number of chains per unit area) and thicknesses (H). However, the need to detach the grafted chains, also taking into account their very low amounts, makes their characterization non-trivial. Conversely, in the grafting to process, polymers with functional end-groups are synthesized, fully characterized and then chemically attached to the substrate.35 The drawback of this approach is the limited grafting density that can be achieved due to the self-limiting nature of the grafting to reaction.36 In fact, it has been widely demonstrated that the maximum obtainable values of both H and Σ of a brush obtained by grafting to depend on the molecular weight of the grafted polymer.37–41 Interestingly, this relationship between the maximum grafting density achievable and the molecular weight of the polymer was recently exploited for silicon doping applications in microelectronics.12,42,43
If grafting reactions are the tools that enable brush synthesis, the reverse process, generally called degrafting, deserves no less attention.27 For example, the self-limiting nature of the grafting to process was recently reconsidered, evaluating the competitive role of the degrafting reaction that brings the system to a dynamic equilibrium condition.44–46 However, most of the attention addressed to the degrafting process is focused on evaluating the stability of polymer brushes, eventually in the application environment.27,47,48 In particular, it was highlighted that the presence of water plays a decisive role in the detachment of the brush by breaking the hydrolysable bonds between polymers and the substrate.27,49 When silicon oxide is used as the substrate, the most common case, the hydrolysis reaction occurs on both siloxane and ester/amide bonds that are typically contained in the linker between the polymer and the substrate.50–52 Moreover, since Si–O bonds are the anchoring points of the SI-ATRP initiators on the substrate, improved brush stability was achieved by replacing them with non-hydrolysable Si–C bonds.51,53 Hydrolysable linkages of the Si–O type are also obtained at present in brushes prepared by grafting to reactions of hydroxy-terminated polymers to silicon oxide.44,46
Although hydrolysis resistance of the brush remains challenging, the use of hydrophobic SI-ATRP initiators or the inclusion of protective hydrophobic polymer blocks can markedly reduce the water access to the hydrolysable groups and, consequently, the degrafting process.54,55
While degrafting reactions were recently observed also for brushes in humid environments,56,57 polymer detachment takes place principally when polymer brushes are exposed to water containing solvents.27 Under these conditions, the brush tends to absorb the solvent for osmosis, thus swelling and stretching the polymer chains perpendicularly to the substrate.58 Furthermore, the chain stretching produces a tension (f) on the chain anchoring point that was estimated as:59–61
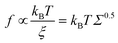 | (1) |
Beyond the grafting density, the affinity between the polymer and solvent also plays a crucial role in determining the brush degrafting tendency.63 For example, Wang et al. treated poly(tert-butyl methacrylate) brushes with different organic solvents containing a defined amount of water and observed that the degrafting kinetic constant k increases in parallel with the polymer swelling ratio, which becomes more pronounced when the solvent has more affinity towards the polymer brush.63 The more affinity the solvent has for the brush, the greater the quantity that enters the brush, thus increasing the chain stretching and, consequently, the tension f at the interface. Furthermore, for polyelectrolyte brushes in aqueous solutions, both pH and salts can influence the brush swelling and consequently the degrafting kinetics.47,48,50,52,54,65 Therefore, the same brush can be stable or not in different environments.
A further parameter that seems to influence the brush degrafting is the molecular weight of the grafted polymer.50 Although the polymer molecular weight does not appear to be a relevant parameter in determining f, according to eqn (1), there is a fair amount of evidence that brushes consisting of long polymer chains are keen to degraft faster than brushes made of short chains.48,50,54,64 In contrast, there is also a limited number of studies reporting slight or no influence of the molecular weight on k.52,63 In this context, a systematic study was performed by Melzak et al.64 to evaluate the degrafting of poly(N,N-dimethylacrylamide) brushes with different molecular weights and grafting densities in methanol solutions. In this study, a clear dependence of the degrafting rate constant on the molecular weight was observed with the higher molecular weight brushes undergoing degrafting faster than the lower molecular weight brushes. However, the interpretation of some experimental results in that paper is inconsistent. In particular, the degrafting of the polymer with the highest molecular weight (3.63 × 105 g mol−1) presents a marked decrease in k from 1.27 to 0.024 min−1 when Σ decreases from 0.79 to 0.31 chains per nm2. This decrease was attributed to reduced interactions between vicinal grafted chains and, consequently, to a reduced tension at the anchoring points. However, when the degrafting of fresh brushes with the same molecular weight but an initial Σ of 0.11 chains per nm2 was evaluated, a k value of 0.41 min−1 was calculated. This value is much higher than 0.024 min−1 obtained in the first experiment for Σ lower than 0.31 chains per nm2. Furthermore, the Σ level beyond which the interaction between vicinal chains reduces is higher for the polymer with a molecular weight of 3.63 × 105 g mol−1 than for smaller polymers with molecular weights of 7 × 104 and 4.3 × 104 g mol−1. This finding is unreasonable, as the volume occupied by a polymer increases with the molecular weight. These unsolved points, in combination with the incompleteness of the theory leading to eqn (1), make necessary further investigations of the molecular weight effect on the degrafting reaction of polymer brushes. Furthermore, a comprehensive study of degrafting in widely disperse systems containing polymers with different molecular weights is still lacking, despite their common use in applications.
In the present work, the degrafting of polystyrene brushes obtained by grafting to was investigated in a tetrahydrofuran (THF) solution containing water. The use of polystyrene as a model polymer is motivated by the fact that it contains neither cleavable nor ionisable groups in the main chain, which would add further variables to the study of the degrafting process. The clarification of the influence of both the molecular weight and the grafting density on the degrafting rate constant was the main focus of this work and was thus evaluated during the entire degrafting process. Furthermore, bimodal brushes containing different amounts of short and long polymers were also investigated with the aim to evaluate degrafting in disperse systems. The degrafting rate constants of the short and long components of the brush were determined and evaluated as a function of the brush composition.
Results and discussion
A hydroxy-terminated deuterated polystyrene with a number average molecular weight (Mn) of 5.2 kg mol−1 (dispersity index Đ = 1.12) and two hydroxy-terminated hydrogenated polystyrenes with Mn of 13 kg mol−1 (Đ = 1.06) and 43.2 kg mol−1 (Đ = 1.16) were synthesized by the ARGET-ATRP (Activator ReGenerated by Electron Transfer-Atom Transfer Radical Polymerization) technique.41,42,66 More details on the synthesis and characterization are published elsewhere.45 The three samples are indicated as PSd85.2-OH, PS13-OH and PS43.2-OH, respectively.Degrafting reaction in single polymer brushes
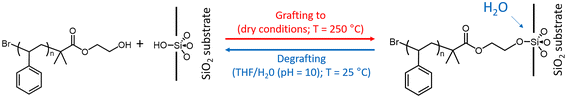
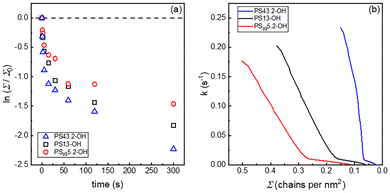
Polymer brushes consisting of either PSd85.2-OH, PS13-OH or PS43.2-OH were obtained by the grafting to approach on silicon wafer substrates covered by an ∼2 nm thick layer of native oxide (SiO2). The scheme of the reaction is represented in Fig. 1 and consists of a condensation reaction between the hydroxyl end-group of the polystyrene and the surface silanols.46 In detail, a thin layer (30 nm) of the functional polymer was deposited by spin-coating on the substrate and the grafting to reaction was performed at 250 °C for 900 s under melt conditions and under an inert atmosphere. The reaction conditions were selected to achieve the maximum values of H and Σ, in accordance with the self-limiting nature of the process.45,46 The unreacted chains were removed by toluene washes and the thickness of the dry brush was evaluated by ellipsometry. A representation of the overall process is included in the SI (see Fig. S1). The average thickness of the obtained brushes (H0) was 4.3, 7.6 and 14.5 nm when PSd85.2-OH, PS13-OH and PS43.2-OH were used, respectively. The grafting densities of the brushes were also calculated using eqn (2),67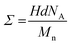 | (2) |
The degrafting reaction was performed at room temperature by immersing the polymer brushes in a mixture of 75% THF and 25% of a pH = 10 buffer solution. This mixture turned out to be much more effective than either simple deionized water or pH = 10 buffer solution alone, as clearly seen in Fig. S2. The addition of THF makes the mixture significantly more affine to polystyrene, thus promoting the swelling of the brush and the access of the mixture at the polymer/substrate interface. Consistent with this, THF and water mixtures with similar percentages were already used for studying degrafting reactions.63 In the present work, a pH = 10 buffer solution was used instead of pure water to speed up the reaction. The expected reaction is essentially the reverse of the grafting to reaction (see Fig. 1) and consists of the hydrolysis of the siloxane bond anchoring the polymer at the substrate.
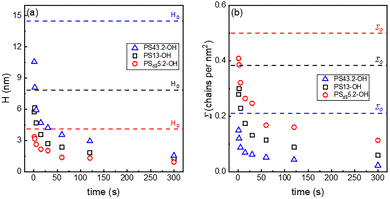
The hydrolysis of the polymer chains from the brush layer was monitored by recording the thickness of the dried brush after different times of exposure to the degrafting solution. The obtained H values are reported in Fig. 2(a). Similarly, the relative Σ values were calculated using eqn (2) and are reported in Fig. 2(b). Obviously, H and Σ present similar trends with a steep initial decrease followed by a slower decrease for long times.
It was already argued that the hydrolysis–degrafting reactions can be reasonably described by pseudo-first order kinetics, assuming that water is constant throughout the entire course of the reaction.65 In this perspective approach, the reaction rate constant (k) is estimated as the slope of the curve obtained by plotting ln(Σ/Σ0) as a function of the degrafting time, as reported in Fig. 3(a). The most immediate evidence is that the slope of the degrafting curve is not constant but decreases as the reaction proceeds, thus leading to a parallel decrease in k. This phenomenon was already described in the literature54,64 and is promoted by the tension f that the swollen and stretched chains induce on the anchoring points.34 Therefore, since f decreases as Σ decreases, as indicated in eqn (1), k is expected to decrease as the degrafting proceeds.
 | ||
| Fig. 3 Ln(Σ/Σ0) vs. degrafting time (a) and k vs. Σ (b) for the polymer brushes consisting of PSd85.2-OH (red), PS13-OH (black) and PS43.2-OH (blue). | ||
The value of k at each degrafting time was obtained by fitting the data reported in Fig. 3(a) and making the derivative of the obtained curve. Furthermore, to establish a direct relationship between k and Σ during the reaction course, at each degrafting time, the corresponding grafting density was assigned by fitting the data reported in Fig. 2(b). Finally, the values of k and Σ corresponding to the same degrafting time were associated, obtaining the curves reported in Fig. 3(b). More details on the fitting method are reported in the SI. The dependence of k on Σ is confirmed across the entire grafting density range. In more detail, k decreases as Σ decreases steeply at first and then, once a critical value of Σ (Σc) is reached, more gradually, thus suggesting the presence of two different regimes in which the degrafting occurs.
Furthermore, for each value of Σ, k is systematically higher for the polymer with a higher Mn, thus confirming that the detachment from the substrate is faster when the molecular weight is higher. This fact is particularly evident for Σ > Σc, whereas the difference being less pronounced. Finally, the value of Σc depends on the molecular weight of the polymer, with a Σc of approximately 0.27, 0.17 and 0.07 chains per nm2 for PSd85.2-OH, PS13-OH and PS43.2-OH, respectively.
A reasonable interpretation of the data presented in Fig. 3(b) is that for Σ < Σc the grafted chains are far enough and do not significantly interact with each other. Consequently, there is a negligible tension f at the anchoring points. In contrast, for Σ > Σc, inter-chain interactions take place more strongly and generate the tension f which promotes the hydrolytic reaction, thus resulting in an increase in k, as predicted using eqn (1). In this study, Σc is lower for the polymer with a higher molecular weight because of its larger volume, in good accordance with the experimental evidence. A first-order estimation of the grafting density at which the polymer chains begin to overlap is given by 1/πRg2, where Rg represents the radius of gyration of the polymer within the brush and πRg2 is the area occupied by a single chain. As a first approximation, Rg can be calculated using eqn (3),45
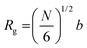 | (3) |
In any case, it should be stressed that eqn (1) is largely incomplete because it does not include a specific dependence of f on the molecular weight of the grafted polymer chain. Further research is certainly necessary to identify all the parameters involved in the degrafting process. However, what is clear is that the role of Mn cannot be ignored when polymer brushes obtained with classical grafting to approaches are subjected to degrafting.
The following section will be dedicated to the study of degrafting in disperse brushes, where long and short polymers are grafted together.
Degrafting reaction in bimodal polymer brushes
Bimodal polymer brushes containing both PSd85.2-OH and PS13-OH were prepared by grafting to. In particular, three polymer brushes containing PSd85.2-OH at percentages of 90, 50 and 20%, indicated as Brush90, Brush50 and Brush20, were the target brushes.It is well known that, during the grafting to reactions, polymers with lower molecular weights react preferentially with the substrate.68–70 This leads to an enrichment of short chains in the brush compared to the composition of the initial polymer mixture. In detail, when a mixture of polymer 1 and polymer 2 is grafted, the ratio between the grafting densities of the two polymers is calculated as follows:36
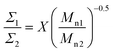 | (4) |
The grafting procedure was the same as described in the previous section. The thickness (H0) of the brushes was determined by ellipsometry, while the brush composition was investigated by TGA-GC-MS (ThermoGravimetric Analysis-Gas Chromatography-Mass Spectrometry) analysis. The TGA-GC-MS procedure is now a consolidated approach for determining the composition of polymer brushes containing different polymer species.45,71–73 In this case, since the two components of the brush are polystyrenes differing in their Mn only, the contrast was produced by using a deuterated and a hydrogenated polystyrene sample. Briefly, in the TGA oven the brush was heated to promote the degradation of the polymers via an unzipping process that generates principally monomers.71 The monomers are conveyed in an inert gas flow to a GC and analyzed in a mass detector. The intensity of the signals after proper calibration relative to deuterated styrene, for PSd85.2-OH, and hydrogenated styrene, for PS13-OH, were combined with the measurement of H0 to determine the grafting density of PSd85.2-OH (ΣD,0), the grafting density of PS13-OH (ΣH,0) and the total grafting density of the brush (ΣTOT,0 = ΣD,0 + ΣH,0).45 More details on the measurements and calculations are contained in the Experimental section. The characteristics of the obtained brushes are reported in Table 1.
| Sample | % PSd85.2-OH target | H 0 (nm) | Σ D,0 (chains per nm2) | Σ H,0 (chains per nm2) | Σ TOT,0 (chains nm−2) | % PSd85.2-OH measured |
|---|---|---|---|---|---|---|
| Brush90/10 | 90 | 4.7 | 0.44 | 0.05 | 0.49 | 88 |
| Brush50/50 | 50 | 6.1 | 0.24 | 0.20 | 0.44 | 54 |
| Brush20/80 | 20 | 7.4 | 0.07 | 0.33 | 0.40 | 19 |
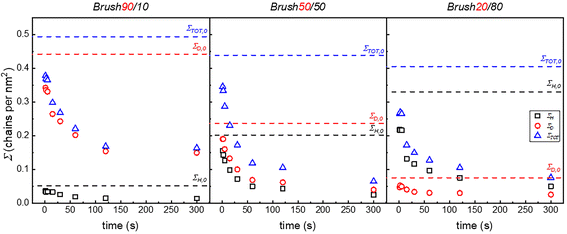
The degrafting procedure was the same as described in the previous section for single polymer brushes. The values of H measured after different degrafting times are reported in Fig. S6. The composition of the brushes was also determined with TGA-GC-MS analysis and ΣD, ΣH and ΣTOT are reported in Fig. 4. Both the partial and the total grafting densities follow the same trend that was already reported for the single polymer brushes, with a rapid decrease at short degrafting times, followed by a slower decrease at longer times. The same behavior was observed for Brush90, Brush50 and Brush20.
To further explore the degrafting kinetics, ln(ΣD/ΣD,0) and ln(ΣH/ΣH,0) were reported as a function of the degrafting time for each typology of brush (see Fig. S7–S9). The degrafting rate constants of both the deuterated and hydrogenated components were then calculated as described in the previous section and are reported in Fig. 5 as a function of ΣTOT. The presence of two regimes of degrafting is confirmed, even for disperse brushes. In fact, beyond a certain critical value of ΣTOT (ΣTOT,c), the dependence of k on ΣTOT is definitely more marked than in the case in which ΣTOT is lower than ΣTOT,c. Interestingly, for each brush composition, ΣTOT,c of the two components has essentially the same value. Furthermore, the value ΣTOT,c decreases as the percentage of short chains (PSd85.2-OH) decreases from 90 to 20%. In more detail, in Brush90, the value of ΣTOT,c is approximately the same as Σc of the single PSd85.2-OH brush. In the same way, in Brush20, ΣTOT,c and Σc of the single PS13-OH brush are similar. Finally, in Brush50, the value of ΣTOT,c is between the Σc of the single PSd85.2-OH brush and the one of the single PS13-OH brush.
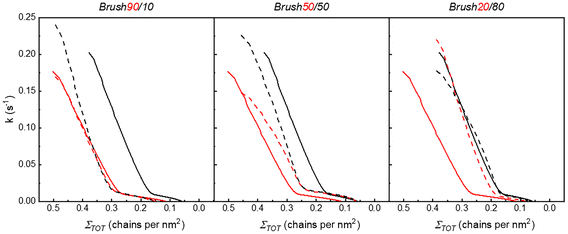 | ||
| Fig. 5 k vs. Σ TOT for the degrafting reaction of PSd85.2-OH (dashed red line) and PS13-OH (dashed black line) in Brush90, Brush50 and Brush20. The curves representing k vs. Σ for the degrafting reaction in the single polymer brushes (see Fig. 3(b)) are also included as references, with a full red line for PSd85.2-OH and a full black line for PS13-OH. | ||
In general, the overall curves representing k vs. ΣTOT of both the components of the bimodal brushes are included between two limits, which consist of the curves obtained for the two single polymer brushes. In more detail, the curves of the bimodal brush are shifted toward the limit curve relative to the more abundant component of the brush. It is finally important to observe that, for ΣTOT > ΣTOT,c, the k of PS13-OH is systematically higher than the k of PSd85.2-OH, indicating a faster degrafting of the longest component of the brush. The preferential detachment of the longest components of disperse brushes is in good accordance with the literature.64
The main conclusion emerging from the data presented in Fig. 5 regards the presence of mutual interactions between short and long chains. For example, a single ΣTOT,c for PSd85.2-OH and PS13-OH in each brush is observed. This means that the value of ΣTOT at which the overall brush experiences tension is determined by mixed contacts between short and long polymers. Furthermore, for Brush20, the k of PSd85.2-OH increases up to the value typical of PS13-OH. On the other side, in Brush90, the k of PS13-OH decreases to the value typical of PSd85.2-OH. Therefore, in both cases, the minor component of the brush is forced by the interactions with the major components to assume a tension state typical of the latter, thus indicating interdependent dynamics.
The above pieces of evidence further confirm that the degree of tension, and consequently k, of a polymer in a brush is not only determined by the molecular weight and the grafting density but also by the brush composition and dispersity, with the overall degrafting kinetics complying with one of the major components.
Conclusions
The degrafting reaction of polymer brushes constituted by two polystyrene samples with different molecular weights and related blends with different compositions was evaluated in a THF/water mixture. In all cases, a clear dependence of the degrafting rate constant on the grafting density of the brush was observed. In particular, the degrafting rate constant decreases linearly as the grafting density of the brush decreases with a sharp slope change corresponding to a critical grafting density value. For grafting densities lower than the critical value, the degrafting rate constant decreases slowly as the grafting density decreases. In contrast, when the grafting density is higher than the critical value, the degrafting rate constant decreases steeply. This behavior suggests the presence of two distinct regimes corresponding to tensioned and relaxed polymer chains in the brush. Furthermore, the critical grafting density value depends on the molecular weight of the polymer forming the brush. In addition, and in contrast to theoretical prediction, in the range of grafting densities investigated, the degrafting of long polymer chains is faster than the degrafting of short ones. When brushes are prepared from blends of the two polystyrene samples with low and high molecular weights, the degrafting reaction of both components results in a similar product to the one of the major species in the blend.This evidence suggests a mutual influence between the polymer chains in the brush, thus underscoring the cooperative nature of the degrafting mechanism. The overall picture provided by this work increases the understanding of the polymer brush stability, thus making it possible to produce adaptable surfaces able to change their structure and composition in a controlled way when applied in specific environments. Furthermore, these data indicate the potential to create brush layers with customized responsiveness to external stimuli associated with specific environments, rendering them suitable for the fabrication of sensors to monitor detrimental environmental modifications and expanding the scope of applications for polymer brush layers.
Experimental section
Materials
The deuterated (PSd85.2-OH) and hydrogenated (PS13-OH) polystyrenes were synthesized by ARGET-ATRP using 2-hydroxyethyl-2-bromoisobutyrate (HEBIB) as the initiator to introduce a hydroxyl functionality in the polymer end-group. All the details of the synthesis and characterization were already published.45 All the reagents were purchased from Merck and used as received. P-type Czochralski-grown Si(100) wafers (a nominal resistivity of 1–5 Ω cm) were purchased from Siltronix.PS43.2-OH synthesis and characterization
The sample was synthesized by a two-step procedure. Firstly, a hydroxy-terminated polystyrene with Mn = 25.4 kg mol−1 (Đ = 1.12) was synthesized by ARGET-ATRP using 2-hydroxyethyl-2-bromoisobutyrate (HEBIB) as the initiator. More details of the synthesis and polymer characterization are reported in the literature.42 The molecular weight of this polymer was then increased by using it as a macroinitiator for a second ARGET-ATRP step.41 In detail, 1.5 g of the macroinitiator (59 µmol) was dissolved in a mixture consisting of 2 mL of styrene (monomer, 18 mmol, 300 equivalents), 526.3 µg of CuBr2 (catalyst, 2.36 µmol, 0.04 equivalents), 0.6 µL of tris[2-(dimethylamino)ethyl]amine (ligand, 2.36 µmol, 0.04 equivalents) and 2 mL of anisole (solvent). The mixture was transferred to a Schlenk flask and degassed by two freeze–thaw cycles. After that, a solution consisting of 9.5 mg of tin(II) 2-ethylhexanoate (reducing agent, 23.6 µmol, 0.4 equivalents), 6 µL of tris[2-(dimethylamino)ethyl]amine (ligand, 23.6 µmol, 0.4 equivalents) and 1 mL of anisole was added to the mixture and a further freeze–thaw cycle was performed. The reaction was carried out at 90 °C in a thermostatic oil bath for 17 h. The polymer was then purified by two successive precipitations carried out with a THF polymer solution in cold methanol. The Mn and Đ values of PS43.2-OH were determined by Size Exclusion Chromatography (SEC). The analysis was performed with a 590 Waters chromatograph equipped with Waters HSPgel HR3 and HR4 columns and a refractive index detector. The eluent was THF, while the flow rate was 0.3 mL min−1. The instrument was calibrated with polystyrene standards with molecular weights ranging from 1 to 100 kg mol−1. The PS43.2-OH chromatograph is reported in Fig. S10.Grafting procedure
All the polymer brushes were obtained with the same procedure.68,69 Silicon wafers covered with an ∼2 nm thick layer of native oxide were cut into slices of 1 cm × 1 cm, treated with piranha solution (H2SO4/H2O2 3/1 volume ratio) for 40 min at 80 °C, rinsed with deionized water and dried under a nitrogen flow. The wafers were then washed with 2-propanol for 15 min in a sonication bath and dried under a nitrogen flow. An ∼30 nm thick layer of the polymer was deposited onto the wafer by spin-coating (3000 rpm, 30 s). The polymer solutions used for the spin-coating process are reported in Table 2.| Brush | PSd85.2-OH (mg) | PS13-OH (mg) | Toluene (mL) |
|---|---|---|---|
| Single PSd85.2-OH | 9.0 | 1 | |
| Single PS13-OH | 9.0 | 1 | |
| Brush90 | 6.2 | 2.8 | 1 |
| Brush50 | 1.8 | 7.2 | 1 |
| Brush20 | 0.5 | 8.5 | 1 |
The grafting to reaction of the deposited polymers was performed on a SAWATEC HP-150 hot plate placed in a MBRAUN LABstar glovebox (inert atmosphere, H2O, O2 < 1 ppm). The reaction was carried out at 250 °C for 900 s. The unreacted chains were removed by toluene washes carried out in a sonication bath for 5 min. The wafers were finally dried under a nitrogen flow.
Degrafting procedure
The sample was immersed in a mixture containing 75% THF and 25% buffer solution pH = 10 (boric acid/potassium chloride/sodium hydroxide). The reaction was carried out at room temperature for different times. The samples were then rinsed with deionized water, dried under a nitrogen flow, rinsed with fresh THF for 5 min and dried again under a nitrogen flow.Brush characterization
The brush thickness was measured using a FS-1 multiwavelength ellipsometer (Film Sense). The error of the measurement was seen to be less than 0.1 nm.The composition of the bimodal brush was evaluated by TGA-GC-MS analysis. The TGA instrument was a Mettler Toledo TGA/DSC 3+ model, while the GC-MS apparatus was a Finnigan GC Trace 1300 MS ISQ LT instrument equipped with a Phenomenex DB5-5MS capillary column of 30 m of length, 0.25 mm of inner diameter and 0.25 μm of thickness. The sample was annealed in the TGA oven from 25 to 1000 °C with a heating ramp of 20 °C min−1 under a helium flow (50 mL min−1). The degradation products were collected and introduced in the GC-MS instrument. The signals revealed in the MS were acquired in EI+ mode in the Selected Ion Monitoring (SIM) mode, with specific focus on the species with m/z of 104 and 112, relative to styrene and deuterated styrene monomers respectively. More details on this hyphenated technique, including the temperatures of the transfer lines and the various segments, are published elsewhere.71,73
The areas of the signals relative to the species with m/z of 104 and 112, indicated as ASTY and ASTYd8, were demonstrated to be proportional to the molar amount of styrene and deuterated styrene in the analyzed brush.45 The mass fraction of styrene (FPS13-OH) in the brush is then calculated using eqn (5).45
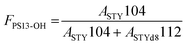 | (5) |
Finally, the grafting density of the deuterated PSd85.2-OH polymer (ΣD) and the hydrogenated PS13-OH polymer (ΣH) and the total grafting density (ΣTOT) were calculated using the following equations:45
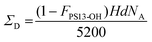 | (6) |
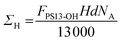 | (7) |
| ΣTOT = ΣD + ΣH | (8) |
Conflicts of interest
There are no conflicts to declare.Data availability
The data that support this work are available in the main text and in the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5py01210d.Acknowledgements
We acknowledge Professor Giuseppe Milano for the fruitful exchange of opinions that took place during the research work and the analysis of the data.References
- A. S. Goldmann, N. R. B. Boase, L. Michalek, J. P. Blinco, A. Welle and C. Barner-Kowollik, Adv. Mater., 2019, 31, 1–21 CrossRef.
- S. V. Orski, K. H. Fries, S. K. Sontag and J. Locklin, J. Mater. Chem., 2011, 21, 14135–14149 RSC.
- W. L. Chen, R. Cordero, H. Tran and C. K. Ober, Macromolecules, 2017, 50, 4089–4113 CrossRef CAS.
- W. J. Brittain and S. Minko, J. Polym. Sci., Part A: Polym. Chem., 2007, 45, 3505–3512 CrossRef CAS.
- H. Liu, Y. Li, K. Sun, J. Fan, P. Zhang, J. Meng, S. Wang and L. Jiang, J. Am. Chem. Soc., 2013, 135, 7603–7609 CrossRef CAS.
- N. Fortin and H. Klok, ACS Appl. Mater. Interfaces, 2015, 7, 4631–4640 CrossRef CAS.
- L. A. Smook, G. C. Ritsema van Eck and S. de Beer, ACS Appl. Polym. Mater., 2021, 3, 2336–2340 CrossRef CAS PubMed.
- E. Han, K. O. Stuen, M. Leolukman, C. C. Liu, P. F. Nealey and P. Gopalan, Macromolecules, 2009, 42, 4896–4901 CrossRef CAS.
- F. Ferrarese Lupi, T. J. Giammaria, F. G. Volpe, F. Lotto, G. Seguini, B. Pivac, M. Laus and M. Perego, ACS Appl. Mater. Interfaces, 2014, 6, 21389–21396 CrossRef CAS.
- S. Wang, Z. Wang, J. Li, L. Li and W. Hu, Mater. Chem. Front., 2020, 4, 692–714 RSC.
- S. Wang, Z. Wang, Y. Huang, Y. Hu, L. Yuan, S. Guo, L. Zheng, M. Chen, C. Yang, Y. Zheng, J. Qi, L. Yu, H. Li, W. Wang, D. Ji, X. Chen, J. Li, L. Li and W. Hu, ACS Appl. Mater. Interfaces, 2021, 13, 17852–17860 CrossRef CAS PubMed.
- R. Chiarcos, M. Laus and M. Perego, Eur. Polym. J., 2024, 208, 112849 CrossRef CAS.
- J. J. Keating IV, J. Imbrogno and G. Belfort, ACS Appl. Mater. Interfaces, 2016, 8, 28383–28399 CrossRef.
- E. Nur Durmaz, S. Sahin, E. Virga, S. de Beer, L. C. P. M. de Smet and W. M. de Vos, ACS Appl. Polym. Mater., 2021, 3, 4347–4374 CrossRef PubMed.
- K. B. Buhl, A. H. Agergaard, M. Lillethorup, J. P. Nikolajsen, S. U. Pedersen and K. Daasbjerg, Polymers, 2020, 12, 1475 CrossRef CAS.
- Y. Yu, M. B. Pérez, C. Cao and S. de Beer, Eur. Polym. J., 2021, 147, 110298 CrossRef CAS.
- J. M. Giussi, M. Lorena Cortez, W. A. Marmisolle and O. Azzaroni, Chem. Soc. Rev., 2019, 48, 814–849 Search PubMed.
- T. Kreer, Soft Matter, 2016, 12, 3479–3501 Search PubMed.
- J. Klein, E. Kumacheva, D. Mahalu, D. Perahla and L. J. Fetterst, Nature, 1994, 370, 634–636 Search PubMed.
- S. De Beer, E. Kutnyanszky, P. M. Schon, G. J. Vancso and M. H. Muser, Nat. Commun., 2014, 5, 3781 CrossRef PubMed.
- W. Yan, S. N. Ramakrishna, N. D. Spencer and E. M. Benetti, Langmuir, 2019, 35, 11255–11264 CrossRef CAS.
- K. Ishihara, Langmuir, 2019, 35, 1778–1787 Search PubMed.
- C. Yoshikawa, S. Hattori, C.-F. Huang, H. Kobayashi and M. Tanaka, J. Mater. Chem. B, 2021, 9, 5794–5804 RSC.
- M. Welch, A. Rastogi and C. Ober, Soft Matter, 2011, 7, 297–302 RSC.
- L. Li, S. Chen, J. Zheng, B. D. Ratner and S. Jiang, J. Phys. Chem. B, 2005, 109, 2934–2941 CrossRef CAS.
- M. Kim, S. K. Schmitt, J. W. Choi, J. D. Krutty and P. Gopalan, Polymers, 2015, 7, 1346–1378 CrossRef CAS.
- Z. Ding, C. Chen, Y. Yu and S. de Beer, J. Mater. Chem. B, 2022, 10, 2430–2443 RSC.
- J. O. Zoppe, N. C. Ataman, P. Mocny, J. Wang, J. Moraes and H. A. Klok, Chem. Rev., 2017, 117, 1105–1318 CrossRef CAS.
- S. Edmondson, V. L. Osborne and W. T. S. Huck, Chem. Soc. Rev., 2004, 33, 14–22 RSC.
- J. Pyun, T. Kowalewski and K. Matyjaszewski, Macromol. Rapid Commun., 2003, 24, 1043–1059 CrossRef CAS.
- B. Zhao and W. J. Brittain, Prog. Polym. Sci., 2000, 25, 677–710 CrossRef CAS.
- R. Barbey, L. Lavanant, D. Paripovic, N. Schuwer, C. Sugnaux, S. Tugulu and H. Klok, Chem. Rev., 2009, 109, 5437–5527 CrossRef CAS PubMed.
- C. M. Hui, J. Pietrasik, M. Schmitt, C. Mahoney, J. Choi, M. R. Bockstaller and K. Matyjaszewski, Chem. Mater., 2014, 26, 745–762 CrossRef CAS.
- H. Klok and J. Genzer, ACS Macro Lett., 2015, 4, 636–639 CrossRef CAS PubMed.
- B. Zdyrko and I. Luzinov, Macromol. Rapid Commun., 2011, 32, 859–869 CrossRef CAS PubMed.
- R. Chiarcos, M. Perego and M. Laus, Macromol. Chem. Phys., 2023, 224, 2200400 CrossRef CAS.
- L. J. Norton, V. Smigolova, M. U. Pralle, A. Hubenko, K. H. Dai, E. J. Kramer, S. Hahn, C. Berglund and B. Dekoven, Macromolecules, 1995, 28, 1999–2008 CrossRef CAS.
- P. Mansky, Y. Liu, E. Huang, T. P. Russell and C. Hawker, Science, 1997, 275, 1458–1460 CrossRef CAS.
- K. S. Iyer, B. Zdyrko, H. Malz, J. Pionteck and I. Luzinov, Macromolecules, 2003, 36, 6519–6526 CrossRef CAS.
- K. Viswanathan, T. E. Long and T. C. Ward, J. Polym. Sci., Part A: Polym. Chem., 2005, 43, 3655–3666 CrossRef CAS.
- K. Sparnacci, D. Antonioli, V. Gianotti, M. Laus, F. F. Lupi, T. J. Giammaria, G. Seguini and M. Perego, ACS Appl. Mater. Interfaces, 2015, 7, 10944–10951 CrossRef CAS PubMed.
- M. Perego, G. Seguini, E. Arduca, A. Nomellini, K. Sparnacci, D. Antonioli, V. Gianotti and M. Laus, ACS Nano, 2018, 12, 178–186 CrossRef CAS PubMed.
- V. M. Ospina, R. Chiarcos, D. Antonioli, V. Gianotti, M. Laus, S. Kuschlan, C. Wiemer and M. Perego, ACS Appl. Electron. Mater., 2022, 4, 6029–6037 CrossRef CAS.
- M. Laus, R. Chiarcos, V. Gianotti, D. Antonioli, K. Sparnacci, G. Munaò, G. Milano, A. De Nicola and M. Perego, Macromolecules, 2021, 54, 499–508 CrossRef CAS.
- R. Chiarcos, D. Antonioli, A. Baldanza, C. Brondi, G. Munaò, G. Milano, M. Laus and M. Perego, Macromolecules, 2025, 58, 1935–1949 CrossRef CAS.
- C. Ivaldi, V. O. M. Guarin, D. Antonioli, G. Zuccheri, K. Sparnacci, V. Gianotti, M. Perego, R. Chiarcos and M. Laus, Macromol. Rapid Commun., 2024, 45, 2400288 CrossRef CAS.
- Y. Zhang, J. He, Y. Zhu, H. Chen and H. Ma, Chem. Commun., 2011, 47, 1190–1192 RSC.
- Y. Zhu, B. Lv, P. Zhang and H. Ma, Chem. Commun., 2011, 47, 9855–9857 RSC.
- H. A. Klok and J. Genzer, ACS Macro Lett., 2015, 4, 636–639 CrossRef CAS PubMed.
- S. Tugulu and H. Klok, Biomacromolecules, 2008, 9, 906–912 CrossRef CAS.
- N. C. Ataman and H. Klok, Macromolecules, 2016, 49, 9035–9047 CrossRef CAS.
- Y. Ko and J. Genzer, Macromolecules, 2019, 52, 6192–6200 CrossRef CAS.
- E. D. Bain, K. Dawes, A. O. Evren, X. Hu, C. B. Gorman, J. Srogl and J. Genzer, Macromolecules, 2012, 45, 3802–3815 CrossRef CAS.
- D. Paripovic and H. Klok, Macromol. Chem. Phys., 2011, 212, 950–958 CrossRef CAS.
- R. Quintana, M. Gosa, D. Jan, E. Kutnyanszky and G. J. Vancso, Langmuir, 2013, 29, 10859–10867 CrossRef CAS PubMed.
- M. B. Perez, M. Cirelli and S. De Beer, ACS Appl. Polym. Mater., 2020, 2, 3039–3043 CrossRef.
- F. Wang, W. Liu, R. Lu, J. Huang, B. Zuo and X. Wang, ACS Macro Lett, 2022, 11, 1041–1048 CrossRef CAS.
- P. G. de Gennes, Macromolecules, 1980, 13, 1069–1075 CrossRef CAS.
- Z. Frakas, I. Derenyi and T. Vicsek, J. Phys.: Condens. Matter, 2003, 15, 1767–1777 CrossRef.
- S. Panyukov, E. B. Zhulina, S. S. Sheiko, G. C. Randall, J. Brock and M. Rubinstein, J. Phys. Chem. B, 2009, 113, 3750–3768 CrossRef CAS.
- S. S. Sheiko, S. Panyukov and M. Rubinstein, Macromolecules, 2011, 44, 4520–4529 CrossRef CAS.
- L. Léger, E. Raphaël and H. Hervet, Adv. Polym. Sci., 1999, 138, 186–225 CrossRef.
- J. Wang and H. Klok, Angew. Chem., Int. Ed., 2019, 58, 9989–9993 CrossRef CAS PubMed.
- K. A. Melzak, K. Yu, D. Bo, J. N. Kizhakkedathu and J. L. Toca-herrera, Langmuir, 2015, 31, 6463–6470 CrossRef CAS PubMed.
- S. Sant, K. Kaur and H. Klok, Langmuir, 2024, 40, 21656–21662 CrossRef CAS.
- W. Jakubowski, K. Min and K. Matyjaszewski, Macromolecules, 2006, 39, 39–45 CrossRef CAS.
- L. Michalek, L. Barner and C. Barner-Kowollik, Adv. Mater., 2018, 30, 1–18 CrossRef PubMed.
- D. Antonioli, R. Chiarcos, V. Gianotti, M. Terragno, M. Laus, G. Munaò, G. Milano, A. De Nicola and M. Perego, Polym. Chem., 2021, 12, 6538–6547 RSC.
- R. Chiarcos, D. Antonioli, V. Gianotti, M. Laus, G. Munaò, G. Milano, A. De Nicola and M. Perego, Polym. Chem., 2022, 13, 3904–3914 RSC.
- R. Chiarcos, D. Antonioli, V. Ospina, M. Laus, M. Perego and V. Gianotti, Analyst, 2021, 146, 6145–6155 RSC.
- D. Antonioli, K. Sparnacci, M. Laus, F. Ferrarese Lupi, T. J. Giammaria, G. Seguini, M. Ceresoli, M. Perego and V. Gianotti, Anal. Bioanal. Chem., 2016, 408, 3155–3163 CrossRef CAS PubMed.
- R. Chiarcos, D. Antonioli, E. Podda, G. Muna, G. Milano, M. Perego and M. Laus, Polymer, 2025, 335, 128804 CrossRef CAS.
- V. Gianotti, D. Antonioli, K. Sparnacci, M. Laus, J. T. Giammaria, M. Ceresoli, F. Ferrarese Lupi, G. Seguini and M. Perego, J. Chromatogr. A, 2014, 1368, 204–210 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |