Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Design of a thermally stable succinonitrile mechanophore featuring electron-withdrawing triazine rings for radical polymerization
Hajime
Sugita
a,
Akira
Kodaka
a,
Akira
Takahashi
 a,
Koichiro
Mikami
*b and
Hideyuki
Otsuka
a,
Koichiro
Mikami
*b and
Hideyuki
Otsuka
 *ac
*ac
aDepartment of Chemical Science and Engineering, Institute of Science Tokyo, Meguro-ku, Tokyo 152-8550, Japan. E-mail: otsuka@mct.isct.ac.jp
bEngineering Division, Process Device Innovation Center, Panasonic Industry Co., Ltd, 1006 Kadoma, Kadoma City, Osaka 571-8506, Japan. E-mail: mikami.koichiro@jp.panasonic.com
cResearch Center for Autonomous Systems Materialogy (ASMat), Institute of Integrated Research, Institute of Science Tokyo, 4259 Nagatsuta-cho, Midori-ku, Yokohama, Kanagawa 226-8501, Japan
First published on 2nd March 2026
Abstract
A novel, triazine ring-containing succinonitrile-based mechanophore was designed and synthesized through radical stabilization energy calculations and molecular simulations. This mechanophore exhibits higher thermal stability than a classical mechanophore and a clear mechanochromic response via C–C bond cleavage. This enables its incorporation into polymers via radical polymerization. This study advances the design of thermally stable mechanophores and provides new insights into the relationship between structures and properties in radical-based mechanochemistry, offering new opportunities in polymer chemistry.
Introduction
Over the past two decades, various materials containing mechanophores that respond to mechanical energy have been developed. These materials exhibit novel functional properties such as self-healing,1–3 self-reinforcing,4,5 and damage detection,6–10 as well as the ability to release small molecules.11–17 The development of mechanophores is a central avenue of research in the field of mechanoresponsive materials.18–20 For example, mechanophores can induce color changes or fluorescence in materials.21 They can also enable the release of small molecules such as acids,22,23 carbocations,8,24–26 and ketenes27 through chemical reactions in response to mechanical forces.We have developed a series of radical-type mechanophores (RMs) that undergo homolytic cleavage of the central C–C bond under mechanical force to generate radical species.28–34 These species significantly affect the properties of the resulting materials.5,35–44 One drawback of RMs is their high tendency to dissociate under ambient conditions, which hinders the application of living radical polymerization techniques and the further development of mechanochromic materials. However, we have demonstrated that substituent effects on the resulting radicals can control the stability of the central C–C bond.33,34 For example, introducing electron-withdrawing groups (EWGs) such as pyridine or triazine rings at the 9-position of fluorene-based mechanophores greatly improves thermal stability and prevents dissociation under ambient conditions, compared to a phenyl group.33
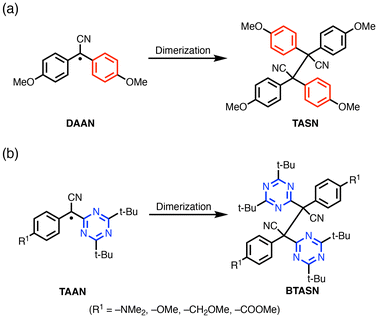
Herein, we report the synthesis and mechanochemical reactivity of a bistriazinyldiarylsuccinonitrile (BTASN)-type mechanophore, in which triazine rings serve as EWGs instead of phenyl groups in tetraarylsuccinonitrile (TASN), one of the most extensively studied RMs to date (Scheme 1). BTASN is a TASN derivative in which two of the aryl groups are triazine rings; it is a dimer of triazinylarylacetonitrile (TAAN) radicals, whereas typical TASN compounds are dimers of diarylacetonitrile (DAAN) derivatives. The presence of triazine rings as EWGs adjacent to the sp2-hybridized carbon radical center, flanked by two aromatic rings, is expected to significantly enhance the thermal stability of the RMs. This approach enables the design of new TASN-type mechanophores with high thermal tolerance and facilitates the synthesis of materials via living radical polymerization.
Results and discussion
Computational studies
Before synthesizing BTASN, we investigated the substitution effects on its dissociation behavior by calculating the radical stabilization energy (RSE), which reflects the thermodynamic stability of radicals, and by performing constrained geometry simulating external force (CoGEF) calculations,45,46 which can simulate the cleavage of the bond through mechanical force and allow theoretical evaluation of mechanochemical reactivity (Fig. 1). In our previous study, we demonstrated that the thermal tolerance of a series of RMs with the same molecular skeleton can be explained by the relationship between two key parameters: the RSE value and the Hammett or modified Swain–Lupton constant at the para position (σp).34 As expected, the RSE values decreased in the order –COOMe (−142.27 kJ mol−1), –CH2OMe (−145.46 kJ mol−1), –OMe (−153.40 kJ mol−1), and –NMe2 (−164.49 kJ mol−1), correlating with increasing σp values (Fig. 1a), showing a trend similar to that observed for TASNs.34 The values were clearly lower than those of TASNs (Table S3, please see also the section Revisit of RSE vs. Hammett constant and its scope and limitation). The CoGEF calculations further revealed that the mechanical elongation of BTASN leads to bond cleavage at the central C–C bond. The Fmax values obtained for BTASN-COOMe, BTASN-CH2OMe, BTASN-OMe and BTASN-NMe2 were 4.61, 4.56, 3.94 and 3.66 nN, respectively (Fig. 1b and S40), following the same trend as the RSE. Considering that the Fmax value of TASN is 4.54 nN,46BTASN-OMe is expected to exhibit greater thermal stability and mechanoresponsiveness. | ||
| Fig. 1 (a) σp values versus RSE and Fmax of BTASN-COOMe, BTASN-CH2OMe, BTASN-OMe, and BTASN-NMe2. (b) CoGEF calculation result of BTASN-OMe (unrestricted B3LYP/6-31G(d,p)). | ||
Molecular dynamics (MD) simulations at the single-molecular level supported this scenario. Canonical ensemble (NVT) calculations were performed for atactic PMMA pentamers grown from bifunctional atom transfer radical polymerization (ATRP) initiators containing BTASN-OMe and TASN, using a universal neural network potential.47,48 The simulations were performed by incrementally increasing the external force applied to the terminal carbon atoms in 0.01 nN steps, from 0.2 to 0.3 nN. For BTASN-OMe-PMMA5mers, homolytic cleavage of the central C–C bond occurred uniformly at 0.23 nN (Fig. S42 and Movies 1, 2). As we expected, the same bond in TASN-PMMA5mers was also cleaved at 0.23 nN (Fig. S43 and Movies 3, 4), indicating that BTASN-OMe exhibits not only enhanced thermal stability but also mechanoresponsiveness comparable to that of TASN. Based on these theoretical findings and the ease of synthetic accessibility, we proceeded to prepare BTASN-OMe-based RMs.
Synthesis and thermal tolerance study
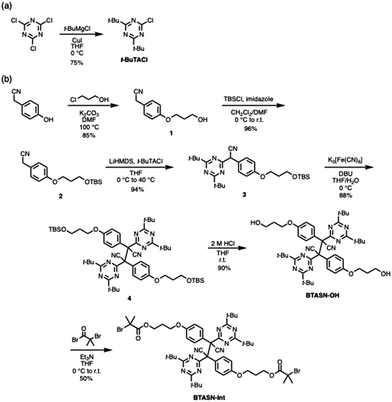
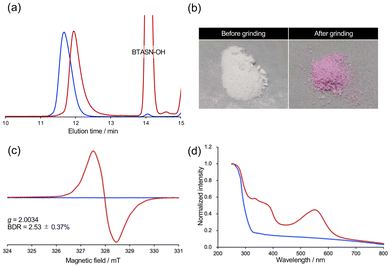
Scheme 2 shows the synthetic procedure for BTASN-OMe and its corresponding bifunctional ATRP initiator. A copper-catalyzed cross-coupling reaction was employed to introduce two tert-butyl groups onto the triazine ring (75%).49 Nucleophilic substitution of 1-chloropropanol afforded compound 1 (85%). Protection of the hydroxy group in 1 with a tert-butyldimethylsilyl (TBS) group gave compound 2 (96%). The introduction of the di-tert-butyltriazine ring to the TBS-protected intermediate afforded 3 (94%). Dimerization of 3 using K3[Fe(CN)6] and 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU) yielded 4, the BTASN mechanophore core (88%). Subsequent deprotection of TBS groups under acidic conditions (90%) furnished the diol (BTASN-OH), which was then esterified with 2-bromoisobutyryl bromide to afford the desired bifunctional ATRP initiator, BTASN-Int (50%).To evaluate the thermal tolerance of BTASN-OH, variable-temperature electron paramagnetic resonance spectroscopy (VT-EPR) measurement was performed in anisole solution (Fig. 2). Measurements at elevated temperatures revealed the formation of dissociated radical species with a g value of 2.003, which was absent at 25 °C (Fig. S35), consistent with the generation of carbon-centered radicals. The dissociation ratio of the central C–C bond increased with increasing temperature. The dissociation ratio of BTASN-OH at 100 °C was estimated to be 2.5 × 10−3%, whereas that of TASN under the same conditions was 0.16%,29,31 approximately 64 times higher than that of BTASN-OH. These results indicate that, as expected, the triazine ring substantially enhances the thermal stability of TASN-based RMs, similar to previous observations for fluorene-type RMs,32 due to its strong electron-withdrawing character.
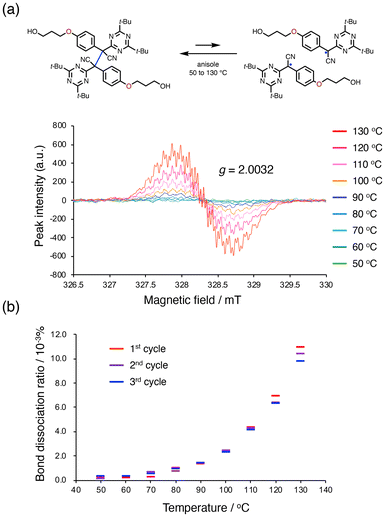 | ||
| Fig. 2 (a) VT-EPR spectra of BTASN-OH in anisole. (b) The plot of the temperature-dependent dissociation ratio. | ||
ATRP of MMA using the bifunctional initiator
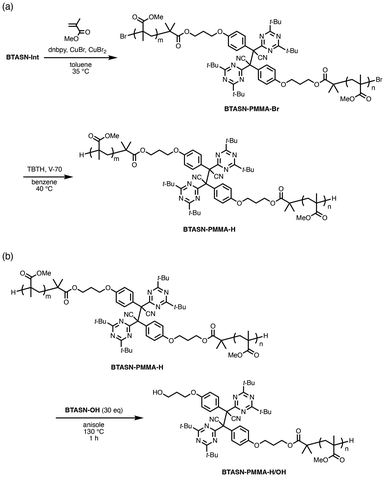
Due to the low thermal stability of the central C–C bonds in tetraaryl-type mechanophores, the TASN skeleton has been incorporated into polymeric materials through polyaddition, click reactions, sol–gel reactions, and cationic ring-opening polymerization to avoid radical processes.29,31,36,37,50–59 In contrast, the superior thermal stability of BTASN enables its introduction into polymer structures using conventional radical polymerization techniques. ATRP of methyl methacrylate (MMA) using BTASN-Int as the initiator was carried out in toluene at 35 °C (MMA/BTASN-Int/CuBr/CuBr2/dNbpy = 940/1/2/0.4/5). This was followed by dehalogenation of the polymer chain ends to prevent the terminal halogen atom from being extracted by the radicals generated by mechanical force (Scheme 3a). The number-average molecular weight (Mn) and dispersity (Mw/Mn) of the resulting PMMA, determined by gel permeation chromatography (GPC) in THF, were Mn (RI) = 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 g mol−1 and Mw/Mn = 1.08. To confirm whether ATRP initiated from BTASN-Int and proceeded in a bifunctional manner, a radical exchange reaction between the resulting polymer and excess BTASN-OH was performed at 130 °C for 1 h (Scheme 3b). After the exchange reaction, the Mn value of the polymer was reduced to nearly half of its original value, while the dispersity remained narrow (Mn = 9790, Mw/Mn = 1.10; Fig. 3a, red line). This result confirms that the polymerization proceeded as designed, affording the desired polymer product (BTASN-PMMA-H) with a BTASN unit located at the center of the main chain.
000 g mol−1 and Mw/Mn = 1.08. To confirm whether ATRP initiated from BTASN-Int and proceeded in a bifunctional manner, a radical exchange reaction between the resulting polymer and excess BTASN-OH was performed at 130 °C for 1 h (Scheme 3b). After the exchange reaction, the Mn value of the polymer was reduced to nearly half of its original value, while the dispersity remained narrow (Mn = 9790, Mw/Mn = 1.10; Fig. 3a, red line). This result confirms that the polymerization proceeded as designed, affording the desired polymer product (BTASN-PMMA-H) with a BTASN unit located at the center of the main chain.
Mechanoresponsive studies
To investigate the mechanical responsiveness of BTASN-PMMA-H, a grinding test was performed at room temperature using a ball mill (30 Hz, 10 min). The originally white powder turned pink after grinding (Fig. 3b). EPR measurement of the ground powder clearly showed a signal corresponding to the carbon-centered radicals (g = 2.003), attributed to the homolytic cleavage of the central C–C bond in the mechanophore (Fig. 3c). The mechanically induced bond dissociation ratio (BDR) was found to be 2.5% for BTASN, which is on the same order of magnitude as that of TASN (5.1%).29 In contrast, as shown in Fig. 2, the thermal dissociation ratio of BTASN-OH was approximately 64 times lower than that of TASN.31 Solid-state UV-vis spectroscopy of the ground sample revealed a new absorption peak at 550 nm, along with increased absorbance across the broad visible region (Fig. 3d), both attributed to the BTASN-derived radical species. These results are in good agreement with theoretical predictions, indicating that the triazine ring, acting as an electron-withdrawing group, imparts enhanced thermal stability to the mechanophore while retaining its mechanochemical reactivity.Excited state studies
Interestingly, TAAN radicals, generated by the homolytic cleavage of the central C–C bond in BTASN, did not exhibit fluorescence under UV irradiation, despite their structural similarity to fluorescent DAAN radicals.29 Both radicals consist of a methyl radical connected to two aromatic rings and a cyano group. To gain deeper insight into the fluorescence properties of these mechanophores, density functional theory (DFT) and time-dependent DFT (TD-DFT) calculations were performed for the corresponding DAAN and TAAN radicals (Fig. 4 and Fig. S44, 46, Tables S1, 2). For the DAAN radical, the D1 → D0 transition is primarily attributed to the β-SUMO(67) → β-HOMO(66) transition (96%) (Fig. 4a and Fig. S45, Table S2). In contrast, the D1 → D0 transition of the TAAN radical is mainly attributed to the β-SUMO(91) → β-HOMO(90) transition (77%) (Fig. 4b and Fig. S24, Table S1). The corresponding orbital transition in the DAAN radical is a π–π* transition, which is allowed, and there are no significant changes in the orbital characteristics between the D0 and D1 states. However, for the TAAN radical, the n-orbital (HOMO−1(89) in the D0 state) becomes the HOMO(90) in the D1 state. This transition is an n–π* transition, which is symmetry-forbidden, and its contribution to the D1 → D0 transition is substantial. This difference likely accounts for the absence of fluorescence in the TAAN radical. The study of fluorescence in radical species provides an important perspective on their electronic structures and excited-state dynamics,60 and the present findings contribute to a deeper understanding of the fundamental principles governing fluorescent radical molecules.Revisit of RSE vs. Hammett constant and its scope and limitation: thermal tolerance of RMs consisting of acyclic radicals
RMs composed of dimers of the same radical can be classified into two main radical types of parent skeletons (Fig. 5a). Type A comprises acyclic radicals, whereas type B comprises cyclic radicals. The corresponding radicals of TASN,28 BTASN and bisarylcyanoacetate (BiACA)34 as well as diaryltetracyanoethane (DATCE) reported by Sakamaki and Winter et al.61,62 are classified as type A (Fig. 5a). Type B features a methyl radical incorporated into a cyclic skeleton (Fig. 5a), and the corresponding radicals of difluorenylsuccinonitrile (DFSN),31 9,9′-bis(4,6-diphenyl-2-triazyl)-9,9′-bifluorene (BTAF), 9,9′-bis(5-methyl-2-pyridyl)-9,9′-bifluorene (BPyF)33 and diarylbibenzofuranone (DABBF)28 are classified as type B.Previously, we reported the prediction of the thermal tolerance of RMs with the same molecular skeleton, based on RSE and the Hammett constant.34 Building on this approach, this study demonstrates the potential for predicting the thermal tolerance of type A of RMs with different molecular skeletons. One of the parent skeletons in type A features a methyl radical that is sandwiched between one cyano group and one aryl group (Fig. 5b). The corresponding radicals of TASN, BTASN, BiACA and DATCE are classified as the parent skeleton among the type A radicals (Fig. 5b). As shown in Fig. 6, the RSE values of the corresponding radicals of the parent skeletons become lower as the Hammett constant increases. Furthermore, as shown in Fig. 7a, the logarithm values of the dissociation constants (Kd) of the RMs including DAANs, TAAN and ACAs linearly increase with increasing RSE values (please see the synthetic part of SI, Fig. S36–S39 and Table S3). The coefficient of determination (R2) of the linear fit is 0.8406. The dissociation of the central C–C bond of aryl dicyanomethyl (ADCM) dimers classified as the same parent skeleton is fast, and the corresponding radicals are stable under ambient conditions.62Fig. 7b shows the plots in which the ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Kd values calculated from the reported binding constants and the RSE values of ADCMs were added to the plots in Fig. 7a. As we expected, the values of ln
Kd values calculated from the reported binding constants and the RSE values of ADCMs were added to the plots in Fig. 7a. As we expected, the values of ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Kd linearly increased with the RSE values as in the case of Fig. 7a, and the R2 value of the linear fit was relatively good (R2 = 0.6476). The decrease in the R2 value is likely due to the fact that the values of ADCMs were measured in methanol or toluene at 50 °C, which is different from our experimental conditions. These results indicate that, within the classification of type A RMs, the thermal stability of RMs increases in the order BiACA > BTASN > TASN > ADCM structures owing to the generation of the corresponding radicals having lower RSE values, and that the introduction of electron-withdrawing groups enhances the thermal stability of RMs across different molecular skeletons.
Kd linearly increased with the RSE values as in the case of Fig. 7a, and the R2 value of the linear fit was relatively good (R2 = 0.6476). The decrease in the R2 value is likely due to the fact that the values of ADCMs were measured in methanol or toluene at 50 °C, which is different from our experimental conditions. These results indicate that, within the classification of type A RMs, the thermal stability of RMs increases in the order BiACA > BTASN > TASN > ADCM structures owing to the generation of the corresponding radicals having lower RSE values, and that the introduction of electron-withdrawing groups enhances the thermal stability of RMs across different molecular skeletons.
 | ||
| Fig. 6 Plot of RSE against the Hammett constant. Blue circle: ADCM radicals,34 green circle: DAAN radicals,34 pink circle: TAAN radicals, and orange circle: ACA radicals.34 DFT calculations were performed at the unrestricted B3LYP/6-31++G(d,p) level. | ||
 | ||
Fig. 7 Plot of RSE values and ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Kd of RMs. The dotted line and R2 indicate an approximated curve and coefficient of determination. Blue circle: ADCM radicals, green circle: DAAN radicals, pink circle: TAAN radicals, and orange circle: ACA radicals. These chemical structures are shown in Fig. 5b, in which the color corresponds to the filled circles. Kd of RMs. The dotted line and R2 indicate an approximated curve and coefficient of determination. Blue circle: ADCM radicals, green circle: DAAN radicals, pink circle: TAAN radicals, and orange circle: ACA radicals. These chemical structures are shown in Fig. 5b, in which the color corresponds to the filled circles. | ||
We reported that the relationship between RSE/thermodynamic stability and the thermal tolerance of RMs can be explained by the Hammond–Leffler postulate, which describes the transition-state (TS) geometry in an elementary step related to reactants, intermediates, and products.34 However, at present, it is still challenging to provide a consistent explanation for acyclic and cyclic radical systems as well as non-dimer type RMs based solely on RSE and substituent effects. For example, DFSN and BTAF classified as type B show enough thermal tolerance for application to ATRP, although the values of RSEs are almost the same as DABBF derivatives, whose central C–C bond undergoes homolysis even under ambient conditions.31,33 We believe that this discrepancy is likely due to the fact that the transition state structures are entirely different between cyclic radicals, which can adopt highly planar structures, and acyclic radicals, which cannot. Furthermore, diarylacetonitrile-α-carboxylic ester (DAANAC) is also an RM consisting of a DAAN radical and an alkoxycarbonyl radical, which shows exceptional thermal stability.63 DAANAC is not a dimer of radicals and the driving force for the generation of radicals might be decarboxylation generating carbon dioxide gas. Further research into the inherent understanding of the origin of thermal stability of radical mechanophores is underway in our group.
Conclusions
We demonstrated the synthesis and mechanochemical reactivity of BTASN-OMe, a triazine-substituted TASN-type mechanophore with enhanced thermal stability. Computational analyses, including RSE, CoGEF calculations, and MD simulations, revealed that the introduction of electron-withdrawing triazine rings significantly improved thermal resistance compared with conventional TASN while maintaining mechanical responsiveness. Experimental thermal tolerance tests supported these findings, showing that BTASN-OMe exhibited a dissociation ratio approximately 64 times lower than TASN. The high thermal stability enabled the successful ATRP of MMA using a bifunctional ATRP initiator derived from BTASN-OMe. The resulting polymers exhibited sufficient mechanical response while maintaining thermal stability. The difference in fluorescence behavior between TAAN and DAAN radicals was attributed to the nature of their electronic transitions: TAAN undergoes a forbidden n–π* transition due to orbital changes involving the nitrogen atom of the triazine ring in the D1 state, while DAAN undergoes an allowed π–π* transition. Furthermore, these results allowed us to predict the thermal tolerance of RMs consisting of the dimer of acyclic radicals through RSE values, even with different molecular skeletons. Overall, BTASN-OMe represents a significant advancement in the design of thermally stable mechanophores, containing enhanced thermal tolerance with robust mechanical responsiveness. This dual functionality broadens the potential applications of responsive polymer materials in fields requiring both mechanical reactivity and thermal endurance.Conflicts of interest
There are no conflicts of interest to declare.Data availability
Data supporting this article have been included in the supplementary information (SI). Supplementary information: synthetic procedures of compounds and polymers and their NMR, IR, GPC, and EPR data as well as details of the computations. See DOI: https://doi.org/10.1039/d5py01088h.Acknowledgements
This work was supported by JST CREST (grant no. JPMJCR1991) from the Japan Science and Technology Agency (JST). Computational calculations were performed at the Research Center for Computational Science, Okazaki, Japan (project no. 24-IMS-C111). We are grateful to Dr Yi Lu and Dr Takumi Yamamoto for the preparation and analysis of the TASN series.References
- Y. Yang and M. W. Urban, Chem. Soc. Rev., 2013, 42, 7446 RSC.
- S. Shahi, H. Roghani-Mamaqani, R. Hoogenboom, S. Talebi and H. Mardani, Chem. Mater., 2022, 34, 468 CrossRef CAS.
- K. Imato, M. Nishihara, T. Kanehara, Y. Amamoto, A. Takahara and H. Otsuka, Angew. Chem., Int. Ed., 2012, 51, 1138 CrossRef CAS PubMed.
- T. Matsuda, R. Kawakami, R. Namba, T. Nakajima and J. P. Gong, Science, 2019, 363, 504 CrossRef CAS PubMed.
- K. Seshimo, H. Sakai, T. Watabe, D. Aoki, H. Sugita, K. Mikami, Y. Mao, A. Ishigami, S. Nishitsuji, T. Kurose, H. Ito and H. Otsuka, Angew. Chem., Int. Ed., 2021, 60, 8406 CrossRef PubMed.
- D. A. Davis, A. Hamilton, J. Yang, L. D. Cremar, D. Van Gough, S. L. Potisek, M. T. Ong, P. V. Braun, T. J. Martínez, S. R. White, J. S. Moore and N. R. Sottos, Nature, 2009, 459, 68 CrossRef CAS PubMed.
- Y. Chen, A. J. H. Spiering, S. Karthikeyan, G. W. M. Peters, E. W. Meijer and R. P. Sijbesma, Nat. Chem., 2012, 4, 559 CrossRef CAS PubMed.
- J. R. Hemmer, C. Rader, B. D. Wilts, C. Weder and J. A. Berrocal, J. Am. Chem. Soc., 2021, 143, 18859 CrossRef CAS PubMed.
- T. Wang, N. Zhang, J. Dai, Z. Li, W. Bai and R. Bai, ACS Appl. Mater. Interfaces, 2017, 9, 11874 CrossRef CAS PubMed.
- Y. Chen, G. Mellot, D. Van Luijk, C. Creton and R. P. Sijbesma, Chem. Soc. Rev., 2021, 50, 4100 RSC.
- M. B. Larsen and A. J. Boydston, J. Am. Chem. Soc., 2013, 135, 8189 CrossRef CAS PubMed.
- G. R. Gossweiler, G. B. Hewage, G. Soriano, Q. Wang, G. W. Welshofer, X. Zhao and S. L. Craig, ACS Macro Lett., 2014, 3, 216 CrossRef CAS PubMed.
- X. Hu, T. Zeng, C. C. Husic and M. J. Robb, J. Am. Chem. Soc., 2019, 141, 15018 CrossRef CAS PubMed.
- H. Shen, M. B. Larsen, A. G. Roessler, P. M. Zimmerman and A. J. Boydston, Angew. Chem., Int. Ed., 2021, 60, 13559 CrossRef CAS PubMed.
- Y. Lu, H. Sugita, K. Mikami, D. Aoki and H. Otsuka, J. Am. Chem. Soc., 2021, 143, 17744 CrossRef CAS PubMed.
- K. Suwada, A. W. Ieong, H. L. H. Lo and G. De Bo, J. Am. Chem. Soc., 2023, 145, 20782 CrossRef CAS PubMed.
- P. Liu, Y.-L. Tseng, L. Ge, T. Zeng, D. Shabat and M. J. Robb, J. Am. Chem. Soc., 2024, 146, 22151 CrossRef CAS PubMed.
- J. Li, C. Nagamani and J. S. Moore, Acc. Chem. Res., 2015, 48, 2181 CrossRef CAS PubMed.
- Q. Mu and J. Hu, Phys. Chem. Chem. Phys., 2024, 26, 679 RSC.
- X. Chen, H. Shen and Z. Zhang, Chin. J. Chem., 2024, 42, 1418 CrossRef CAS.
- D. Xu, X. Cheng, W. Liu, Y. Sun, Y. Niu, M. Wang and H. Qian, Chem. Rev., 2026, 126 DOI:10.1021/acs.chemrev.5c00789.
- C. E. Diesendruck, B. D. Steinberg, N. Sugai, M. N. Silberstein, N. R. Sottos, S. R. White, P. V. Braun and J. S. Moore, J. Am. Chem. Soc., 2012, 134, 12446 CrossRef CAS PubMed.
- C. Nagamani, H. Liu and J. S. Moore, J. Am. Chem. Soc., 2016, 138, 2540 CrossRef CAS PubMed.
- T. Shiraki, C. E. Diesendruck and J. S. Moore, Faraday Discuss., 2014, 170, 385 RSC.
- C. E. Diesendruck, G. I. Peterson, H. J. Kulik, J. A. Kaitz, B. D. Mar, P. A. May, S. R. White, T. J. Martínez, A. J. Boydston and J. S. Moore, Nat. Chem., 2014, 6, 623 CrossRef CAS PubMed.
- L. Chen, R. Nixon and G. De Bo, Nature, 2024, 628, 320 CrossRef CAS PubMed.
- M. J. Robb and J. S. Moore, J. Am. Chem. Soc., 2015, 137, 10946 CrossRef CAS PubMed.
- K. Imato, A. Irie, T. Kosuge, T. Ohishi, M. Nishihara, A. Takahara and H. Otsuka, Angew. Chem., Int. Ed., 2015, 54, 6168 CrossRef CAS PubMed.
- T. Sumi, R. Goseki and H. Otsuka, Chem. Commun., 2017, 53, 11885 RSC.
- K. Ishizuki, H. Oka, D. Aoki, R. Goseki and H. Otsuka, Chem. – Eur. J., 2018, 24, 3170 CrossRef CAS PubMed.
- H. Sakai, T. Sumi, D. Aoki, R. Goseki and H. Otsuka, ACS Macro Lett., 2018, 7, 1359 CrossRef CAS PubMed.
- K. Kawasaki, D. Aoki and H. Otsuka, Macromol. Rapid Commun., 2020, 41, 1900460 CrossRef CAS PubMed.
- H. Sugita, Y. Lu, D. Aoki, H. Otsuka and K. Mikami, Chem. – Eur. J., 2023, 29, e202203249 CrossRef CAS PubMed.
- Y. Lu, H. Sugita, K. Mikami, D. Aoki and H. Otsuka, Chem. Sci., 2023, 14, 8792 RSC.
- K. Imato, T. Kanehara, S. Nojima, T. Ohishi, Y. Higaki, A. Takahara and H. Otsuka, Chem. Commun., 2016, 52, 10482 RSC.
- S. Kato, K. Ishizuki, D. Aoki, R. Goseki and H. Otsuka, ACS Macro Lett., 2018, 7, 1087 CrossRef CAS PubMed.
- F. Hoshino, T. Kosuge, D. Aoki and H. Otsuka, Mater. Chem. Front., 2019, 3, 2681 Search PubMed.
- T. Kosuge, X. Zhu, V. M. Lau, D. Aoki, T. J. Martinez, J. S. Moore and H. Otsuka, J. Am. Chem. Soc., 2019, 141, 1898 CrossRef CAS PubMed.
- H. Sakai, D. Aoki, K. Seshimo, K. Mayumi, S. Nishitsuji, T. Kurose, H. Ito and H. Otsuka, ACS Macro Lett., 2020, 9, 1108 CrossRef CAS PubMed.
- Y. Mao, Y. Kubota, T. Kurose, A. Ishigami, K. Seshimo, D. Aoki, H. Otsuka and H. Ito, Macromolecules, 2020, 53, 9313 CrossRef CAS.
- Y. Mao, Y. Kubota, J. Gong, T. Kurose, A. Ishigami, K. Seshimo, T. Watabe, D. Aoki, H. Otsuka and H. Ito, Macromolecules, 2021, 54, 8664 CrossRef CAS.
- Y. Mao, Y. Kubota, R. Feng, J. Gong, A. Ishigami, Y. Kobayashi, T. Watabe, D. Aoki, H. Otsuka and H. Ito, Macromolecules, 2022, 55, 3948 CrossRef CAS.
- T. Watabe, D. Aoki and H. Otsuka, Macromolecules, 2022, 55, 5795 CrossRef CAS.
- K. Ishizuki, A. Takahashi and H. Otsuka, Macromol. Rapid Commun., 2025, 46, 2400812 CrossRef CAS PubMed.
- M. K. Beyer, J. Chem. Phys., 2000, 112, 7307 CrossRef CAS.
- M. Klein, C. C. Husic, D. P. Kovács, N. J. Choquette and M. J. Robb, J. Am. Chem. Soc., 2020, 142, 16364 CrossRef PubMed.
- S. Yoon, J. H. Choi, B. J. Sung, J. Bang and T. A. Kim, NPG Asia Mater., 2022, 14, 61 CrossRef CAS.
- S. Takamoto, C. Shinagawa, D. Motoki, K. Nakago, W. Li, I. Kurata, T. Watanabe, Y. Yayama, H. Iriguchi, Y. Asano, T. Onodera, T. Ishii, T. Kudo, H. Ono, R. Sawada, R. Ishitani, M. Ong, T. Yamaguchi, T. Kataoka, A. Hayashi, N. Charoenphakdee and T. Ibuka, Nat. Commun., 2022, 13, 2991 CrossRef CAS PubMed.
- L. Hintermann, L. Xiao and A. Labonne, Angew. Chem., Int. Ed., 2008, 47, 8246 CrossRef CAS PubMed.
- S. Kato, D. Aoki and H. Otsuka, Polym. Chem., 2019, 10, 2636 RSC.
- S. Kato, S. Furukawa, D. Aoki, R. Goseki, K. Oikawa, K. Tsuchiya, N. Shimada, A. Maruyama, K. Numata and H. Otsuka, Nat. Commun., 2021, 12, 126 CrossRef CAS PubMed.
- S. Kato, D. Aoki and H. Otsuka, ACS Appl. Polym. Mater., 2021, 3, 594 CrossRef CAS.
- S. Kato, D. Aoki, K. Oikawa, K. Tsuchiya, K. Numata and H. Otsuka, ACS Macro Lett., 2021, 10, 623 CrossRef CAS PubMed.
- S. Kato, D. Aoki and H. Otsuka, Chem. Lett., 2021, 50, 1223 CrossRef CAS.
- M. Van Galen, J. P. Kaniraj, B. Albada and J. Sprakel, J. Phys. Chem. C, 2022, 126, 1215 CrossRef CAS PubMed.
- N. Li, Z. Cui, X. Yue, Y. Zhang, Z.-H. Ren and Z.-H. Guan, Sens. Actuators, B, 2024, 404, 135266 CrossRef CAS.
- Y. Zhu, M. Wang, J. Huang, H. Mi, Z. Xu, F. Wu, L. Chen, H. Yang and Y. Chen, Macromolecules, 2024, 57, 8329 CrossRef CAS.
- C. Zhang, N. Li, J. Ren, H. Ouyang, X. Dong, Z. Cui, Z.-H. Ren and Z.-H. Guan, Chem. Mater., 2024, 36, 10474 CrossRef CAS.
- Y. Xu, Y. Huang, J. Wang, S. Huang, H. Yang and Q. Li, Angew. Chem., Int. Ed., 2025, 64, e202423584 CrossRef CAS PubMed.
- A. Mizuno, R. Matsuoka, T. Mibu and T. Kusamoto, Chem. Rev., 2024, 124, 1034 CrossRef CAS PubMed.
- T. Kobashi, D. Sakamaki and S. Seki, Angew. Chem., Int. Ed., 2016, 55, 8634 CrossRef CAS PubMed.
- J. P. Peterson, M. R. Geraskina, R. Zhang and A. H. Winter, J. Org. Chem., 2017, 82, 6497 CrossRef CAS PubMed.
- Y. Uchida, H. Sugita, A. Takahashi and H. Otsuka, J. Am. Chem. Soc., 2025, 147, 44686 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |