Open Access Article
Open Access ArticleStructure–melt viscosity relationship of discrete sequence-specific linear oligo(dimethylsiloxane-co-diphenylsiloxane)s
Hiroyuki
Minamikawa
 *,
Takahiro
Kawatsu
*,
Takahiro
Kawatsu
 and
Kazuhiro
Matsumoto
and
Kazuhiro
Matsumoto
 *
*
Research Institute for Chemical Process Technology, National Institute of Advanced Industrial Science and Technology (AIST), Tsukuba Central 5, 1-1-1 Higashi, Tsukuba, Ibaraki 305-8565, Japan. E-mail: hiroyuki.minamikawa@aist.go.jp; kazuhiro.matsumoto@aist.go.jp
First published on 11th February 2026
Abstract
Polysiloxanes are widely used in various household, industrial, and biomedical products; however, the effects of their composition and sequence on the melt viscosity have not been investigated in detail. A series of discrete linear oligosiloxanes with precisely controlled compositions and sequences was prepared in this study, and the relationship between their molecular structures and melt viscosities was examined. The oligomers possessed linear structures composed of dimethylsiloxy (D) and diphenylsiloxy (P) units with trimethylsiloxy (M) ends. P-repeat oligomers contained consecutive P blocks including exact numbers of P units. The prepared oligomers were employed to explore the specific effects of block structures. The 26-meric sequence isomers exhibited identical M/D/P compositions and four sequence-repeating blocks: (PD)12, (PPDD)6, (PPPDDD)4, and (PPPPDDDD)3. This facile synthetic methodology enabled the preparation of well-defined oligomers on the gram scale and estimation of their melt viscosities through actual measurements. The oligodimethylsiloxanes were also compared using previously reported viscosity values. Each oligosiloxane was uniform in terms of the M/D/P composition, sequence, and molecular weight. These oligosiloxanes were distinguished by their viscosity–molecular weight relationships; a steep enhancement in viscosity with an increasing number of consecutively repeated P units and a distinct influence of block sequence patterns on the viscosity of the 26-mers were observed. To gain molecular insights, the oligosiloxane viscosities were analyzed using the Rouse model and its modifications, and the effects of structural factors on the melt viscosity of the oligosiloxanes were discussed. The findings of this work demonstrate that appropriate Rouse model variants can effectively explain how the composition and sequence influence the melt viscosity of oligosiloxanes.
1 Introduction
Siloxanes are widely utilized in personal care, household, industrial, and biomedical products.1–3 Taking various physical forms, such as oils, greases, waxes, rubbers, and resins, oligodimethylsiloxanes and polydimethylsiloxanes (PDMS) have been formulated into lubricants, antifoams, mold releases, adhesives, coatings, sealants, thermal and electrical insulators, and contact lens materials. Notably, attaching phenyl substituents to silicon atoms in PDMS drastically changes the macroscopic properties of the resulting copolymers. While PDMS undergoes crystallization between −50 and −40 °C according to its heating thermograms, the crystallization process is completely suppressed after 4–6 mol% substitution with methylphenylsiloxane4 and diphenylsiloxane5,6 in PDMS chains. For poly(dimethylsiloxane-co-diphenylsiloxane), substitution in the 6–60 mol% range monotonically elevates the glass transition temperature5,7,8 and enhances the thermal stability.8 These structural effects are usually described in terms of the average molecular weight, content, and sequence.4–13Siloxane copolymers with phenyl substitution have been prepared via the condensation of silanolates and chlorosilanes,5,9 condensation of alkoxysilanes and hydrosilanes (the Piers–Rubinsztajn reaction),11 ring-opening polymerization of cyclotrisiloxanes,4–6,8,10,12–14 and convergent preparation of block copolymers.15 In these polymerization reactions, random copolymerization to a phenyl content of 64 mol%8 and preparation of strictly alternating poly(dimethyl-co-diphenylsiloxane)9 and phenyl-rich block copolymers11 have been reported. Nevertheless, the fine control of the copolymer structure remains a difficult task. The copolymers inherently exhibit distributions of molecular weights, phenyl substitution contents, and sequence. Additionally, preparing diverse sequence patterns during polymerization is difficult. For example, a sequence distribution analysis demonstrated that increasing the diphenylsiloxane feed during copolymerization resulted in two and three consecutive diphenylsiloxane sequences;8,10 however, four or more consecutive sequences were difficult to obtain, likely due to steric hindrance.13 Thus, preparing well-defined siloxanes with uniform molecular weights and contents and various sequence patterns remains challenging.
Recently, one-pot sequence-controlled syntheses have been developed for linear, branched, and cyclic oligosiloxanes.16–19 This approach allows precise control of the siloxane composition, molecular weight, sequence, and topology through one-pot iteration of the B(C6F5)3-catalyzed dehydrocarbonative cross-coupling of alkoxysilanes with hydrosilanes and B(C6F5)3-catalyzed hydrosilylation of carbonyl compounds. In addition, the incorporation of siloxane building blocks into the iteration steps enabled the preparation of block siloxanes up to 26-mers.19 As a facile methodology for the gram-scale preparation process, it can potentially be used to prepare and characterize molecularly precise oligosiloxanes systematically.
The melt viscosities of siloxanes have attracted considerable attention from researchers because they reflect the characteristic chain flexibilities of oligomers, homopolymers, and copolymers. For linear oligodimethylsiloxanes and polydimethylsiloxanes (PDMS),20–27 the zero-shear viscosity η0 at 25 °C increases with increasing molecular weight M.27 The power law relationship η0 ∼ M1.2–1.4 holds below the critical molecular weight Mc, as typically observed for unentangled chains.28–30Mc values have been evaluated at around 34![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 g mol−1,27 corresponding to a remarkably high degree of polymerization (∼465 dimethylsiloxane units). Therefore, PDMS is considered one of the most flexible polymers.27,31 For phenyl-substituted random copolymers, the viscosity increases monotonically with the phenyl content.4,6,13 Poly(dimethylsiloxane-co-diphenylsiloxane) exhibits a more pronounced viscosity increase than poly(dimethylsiloxane-co-methylphenylsiloxane);13 the observed difference is explained in terms of chain mobility. Although these results indicate the flexibility of copolymer chains, specific effects of the polysiloxane composition and sequence on the melt viscosity have not been sufficiently investigated.
500 g mol−1,27 corresponding to a remarkably high degree of polymerization (∼465 dimethylsiloxane units). Therefore, PDMS is considered one of the most flexible polymers.27,31 For phenyl-substituted random copolymers, the viscosity increases monotonically with the phenyl content.4,6,13 Poly(dimethylsiloxane-co-diphenylsiloxane) exhibits a more pronounced viscosity increase than poly(dimethylsiloxane-co-methylphenylsiloxane);13 the observed difference is explained in terms of chain mobility. Although these results indicate the flexibility of copolymer chains, specific effects of the polysiloxane composition and sequence on the melt viscosity have not been sufficiently investigated.
The present study aims to examine the relationship between the structure and melt viscosity of discrete linear oligosiloxanes with precisely controlled compositions and sequences. Fig. 1 shows the linear oligosiloxanes investigated in this work. These oligomers possess linear structures composed of dimethylsiloxy (D) and diphenylsiloxy (P) units with trimethylsiloxy (M) ends. P-repeat oligomers 2, 3, 4, 5, and 6 contain consecutive P blocks with exact numbers of P units. The synthesized oligomers were used to explore the specific effects of the block structures. The 26-meric sequence isomers 7, 8, 9, and 10 exhibit the identical M/D/P composition and four sequence-repeating blocks (PD)12, (PPDD)6, (PPPDDD)4, and (PPPPDDDD)3, respectively.19 Our facile synthetic methodology enabled the preparation of well-defined oligomers 2 to 10 on the gram scale and evaluation of their melt viscosities by performing actual measurements. Oligodimethylsiloxanes 1n were also analyzed using previously reported viscosity values.20–22 Notably, each oligosiloxane has a uniform M/D/P composition, sequence, and molecular weight. The discrete sequence-specific molecular structures are a prerequisite for a close comparative examination. These oligosiloxanes were classified into three types based on their viscosity–molecular weight relationships. To gain some molecular insights, we estimated the viscosities of the studied oligosiloxanes by employing the Rouse model29,30,32 and its modifications and discussed the effect of structural factors on the melt viscosity.
2 Results and discussion
2.1 Sequence-controlled preparation of linear oligo(dimethylsiloxane-co-diphenylsiloxane)s
Discrete P-repeat oligomers 2–6 with 3–8 consecutive P units were successfully prepared following the synthetic methodology described in ref. 18. Notably, four or more consecutive P units are practically inaccessible as pure compounds by conventional methods. The 26-meric siloxane oligomers 7–10 possessing specific block sequences were prepared using the method described in ref. 19.2.2 Viscosity measurements
Viscosity measurements showed that oligomers 2–10 in the melt were viscous liquids. For each oligosiloxane, the shear viscosity η was nearly constant below the shear rate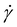 of 100 s−1 (Fig. S1). We utilized the viscosity at
of 100 s−1 (Fig. S1). We utilized the viscosity at 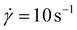 as the zero-shear viscosity η0.
as the zero-shear viscosity η0.
Fig. 2 shows the zero-shear viscosities η0 of the liquid siloxane oligomers plotted as functions of temperature T. The viscosity of each melt yields a separate curve, which monotonously decreases with temperature. Among the P-repeat oligomers 2–6, the temperature–viscosity curves are uniformly shifted upwards in the sequence 2 < 3 < 4 < 5 < 6 (curves a–e) for 3–8 consecutive P units. For the 26-meric sequence isomers 7–10, the curves are shifted upwards in the sequence 7 < 8 < 9 < 10 (curves f–i).
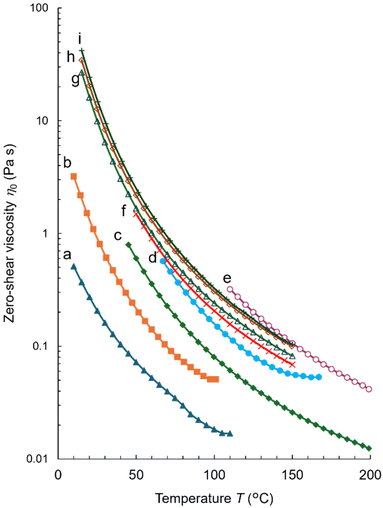 | ||
Fig. 2 Zero-shear viscosities η0 of the liquid siloxane oligomers plotted as functions of temperature T (shear rate 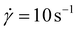 ). P-repeat oligomers: (a) 2 (blue filled triangles, ▲), (b) 3 (orange filled squares, ■), (c) 4 (green filled diamonds, ◆), (d) 5 (light blue filled circles, ●), and (e) 6 (purple open circles, ○). 26-mers: (f) 7 (red crosses, ×), (g) 8 (green open triangles, Δ), (h) 9 (brown open diamonds, ◊), and (i) 10 (green pluses, +). The curves f–i are redrawn from ref. 19. ). P-repeat oligomers: (a) 2 (blue filled triangles, ▲), (b) 3 (orange filled squares, ■), (c) 4 (green filled diamonds, ◆), (d) 5 (light blue filled circles, ●), and (e) 6 (purple open circles, ○). 26-mers: (f) 7 (red crosses, ×), (g) 8 (green open triangles, Δ), (h) 9 (brown open diamonds, ◊), and (i) 10 (green pluses, +). The curves f–i are redrawn from ref. 19. | ||
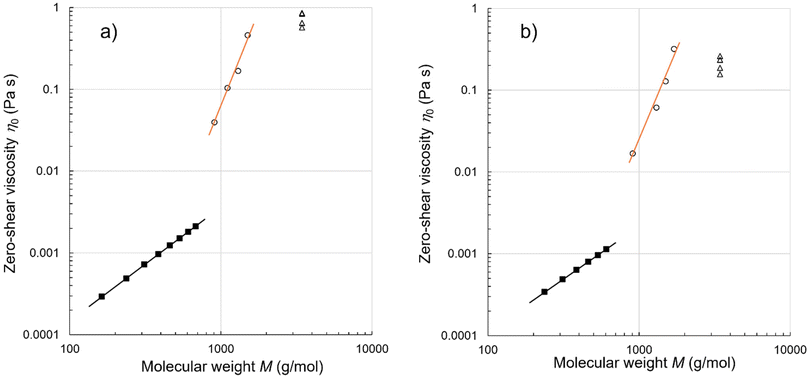
Fig. 3 compares the zero-shear viscosities η0 of the oligosiloxanes at 70 and 110 °C plotted as functions of molecular weight M. Here, the molecular weights are precise and not average values. For the dimethylsiloxane oligomers, the viscosity values are taken from the literature.21,22 The plot distinguishes three types of oligomers using the η0 − M power law relationship. In a series of dimethylsiloxane oligomers 1n (filled squares), the relationships η0 ∼ M1.4and η0 ∼ M1.3 can be observed at 70 and 110 °C (the black lines), respectively, and the exponents are typical ones for oligomeric compounds.28–30
At the above-mentioned temperatures, the viscosities of the P-repeat oligomers 2–6 (open circles) were between 0.01 and 1 Pa s, which were as large as those of PDMS with molecular weights above 104 g mol−1. Moreover, the viscosities steeply increased with M as η0 ∼ M4.8 at 70 °C and η0 ∼ M4.0 at 110 °C (the orange lines). It is a striking observation because the exponents of 4.0 and 4.8 obtained for the oligomers are exceptionally large as compared with the values of 3.2–3.6 determined for the previously reported entangled polymers.28–30,33–35 These results revealed that the consecutive P-repeats exhibited highly cumulative viscosity enhancements.
The viscosity values of 7–10 at 70 and 110 °C are plotted as the open triangles in Fig. 3. Despite the identical composition and molecular weight (3431.7 g mol−1), these isomers exhibit relatively small but distinct differences in viscosity, as mentioned previously.19 More specifically, their viscosities increased in the sequence 7 < 8 < 9 < 10 in the measured temperature range of 50–150 °C (Fig. 2). At each temperature, 10 produced 1.5–1.7 times higher viscosities than isomer 7; the observed differences were significant during viscosity measurements. These results demonstrate that the sequence patterns strongly influence the melt viscosity of the isomers.
In this section, the zero-shear viscosities of the oligosiloxanes with precise structures were compared. Their viscosity was significantly influenced by structural factors such as molecular weight, composition, and sequence. In particular, our precise synthesis allowed measuring and comparing the viscosities of P-repeat oligomers and 26-mers to demonstrate that the obtained values were highly sensitive to the compositions and sequences of the P and D groups. In section 2.3, we analyze these measurement results using a discrete Rouse model and its modifications.
2.3 Rouse model estimation
We examined the 7 to 12-mers 2–6 and the 26-meric isomers 7–10. Being significantly shorter than entangled PDMS chains (465-mers or longer),27 the chains were analyzed as unentangled ones. The viscosity of oligosiloxane liquids is sensitive to the M/D/P composition and sequence as well as to their molecular weight. In this section, the viscosities are estimated using four models: the Rouse model32 and its modifications, Models A, B, and C. In these bead-spring models, M, D, and P units are treated as beads that cause intermolecular friction, and Si–O–Si linkages are considered harmonic spring bonds relaxing to equilibration lengths. The specific sequence of each oligosiloxane is represented in a matrix with one-to-one correspondence. Throughout this study, the matrices were processed in a discrete form without transforming the bead-spring structure into a continuous, smooth string object. The discrete Rouse model36 provides a direct and effective method for analyzing the fine effects of defined sequences (details of the calculation procedure are described in the SI). The model parameters and measured and estimated viscosities are listed in Tables S1–S4.For the oligodimethylsiloxanes 1n, the density values were taken from ref. 20–22. In estimating the oligomer viscosities, the parameters σM and σD were optimized. For the P-containing oligomers 2–10, we assumed a molecular volume as the sum of the included unit volumes. From the specific volume of the MPM trimer, denoted as MD*M in ref. 20, the P unit volume was estimated by interpolation at 70 °C and by extrapolation at 110 °C.
To discuss the observed effects of the P-consecutive blocks, we here take intermolecular π–π or CH–π interaction into consideration. P units can interact intermolecularly with each other through relatively weak interactions, but the pair may easily dissociate. For the P-consecutive blocks, however, the dissociated P unit can find another P unit nearby in the counterpart block and immediately associate. Hence, the block structures may effectively prolong the original intermolecular pair, especially for the long P-consecutive blocks. This picture is speculative but may suggest some aspects of the observed increase in melt viscosity. It should also be noted that the reassociation process between associating units can play a critical role in the rheological behavior of weakly associative copolymers.37 We should check the plausible picture by simulation in the future.
In general, the larger friction coefficients result in longer relaxation times, leading to higher viscosity. To figure out the cumulative effect of the P-consecutive blocks in the matrix-based formulation, Model A introduces a phenomenological parameter (<1). To reproduce the observed exponential increase in viscosity for the P-consecutive oligomers, we replaced the matrix components σP with αnσP. It has been found that the factors αn increase the viscosity estimates in an exponential manner. Taking PPPPPP for instance, the series ασP, α2σP, α2σP, α2σP, α2σP, and ασP did not give the observed steep increase in viscosity; in contrast, the series ασP, α2σP, α3σP, α3σP, α2σP, and ασP successfully reproduced the observation. Parameters σM and σD were taken from the 1n estimation described above. The model details are provided in the SI.
Fig. 4b and 5b show the Model A estimations for the P-repeat oligomers 2, 3, 4, and 5 at 70 °C and 2, 4, 5, and 6 at 110 °C, respectively. The estimation (+) strongly agreed with the measured relations η0 ∼ M4.8 at 70 °C and η0 ∼ M4.0 at 110 °C. This model functioned effectively for the P-repeat oligomers.
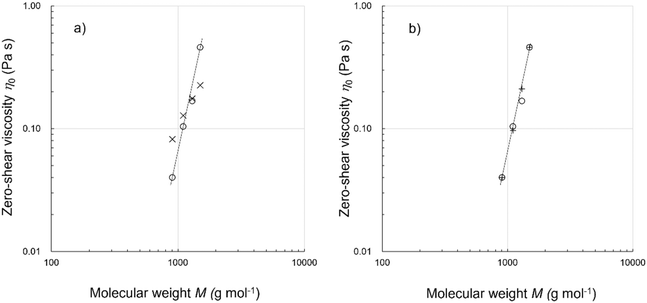
Fig. 6a compares the logarithmic values of the measured viscosities at 70 °C and Model A estimations. The dashed line is an eye guide. From left to right, the open circles indicate the P-repeat oligomers 2, 3, 4, and 5, and the open triangles denote the 26-mers 7, 8, 9, and 10. For the four P-repeat oligomers and 26-mers 9 and 10, the plots are located on the line. In contrast, the estimates obtained for 7 and 8 are significantly lower than the measured values. The coefficient of determination in Fig. 6a is 0.902. Model A was not sufficiently accurate for oligomers 7 and 8, which contained the repeating blocks of PD and PPDD, respectively. In the next section, we discuss Model B to improve our estimation accuracy.
Fig. 6b compares the Model B estimations with the measurement data obtained at 70 °C. The estimations for 7 and 8 are particularly improved using this model. The plot constructed for the eight oligosiloxanes yields a coefficient of determination of 0.948. These results demonstrate that the bond–bond correlation or local chain rigidity can strongly influence the chain relaxation responsible for liquid viscosity.
In Model B, the effects produced by the consecutive P units and local chain rigidity are simultaneously examined. When parameter α was set to 1 (no enhancement by the consecutive P units), the optimization resulted in a value of e equal to unity, reflecting the perfect rigidity in the PPP sequence. However, the estimated exponent in the η0 − M relationship for the consecutive P oligomers was 3.4 for 2–5 at 70 °C and 3.2 for 2–6 at 110 °C, which were considerably lower than the experimental values (4.8 at 70 °C and 4.0 at 110 °C). The α values of 0.183 at 70 °C and 0.301 at 110 °C, along with a zero e value, produced the estimates that were in good agreement with the measurement results (Fig. 6b and Fig. S5b, Table S1). This result indicates that a small α value works effectively not only for reproducing the consecutive P-sequence enhancement but also for expressing the PPP chain rigidity in place of e. Meanwhile, the estimated non-zero f and g values effectively improved the viscosity estimations for the 26-mers 7–10.
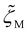 ,
, 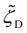 , and
, and 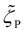 , which vary with the oligomer composition. First, the area fractions of M, D, and P units were estimated for each oligosiloxane. Second, the friction coefficients
, which vary with the oligomer composition. First, the area fractions of M, D, and P units were estimated for each oligosiloxane. Second, the friction coefficients 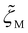 ,
, 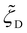 , and
, and 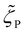 were calculated from the area fractions. Finally, a semiflexible chain estimation was performed using a procedure similar to that used in Model B (computational details are provided in the SI). Fig. 6c shows the Model C estimations obtained for the P-repeat isomers and 26-mers at 70 °C. Some improvements were achieved in the estimation for 8, as indicated by its higher coefficient of determination equal to 0.962.
were calculated from the area fractions. Finally, a semiflexible chain estimation was performed using a procedure similar to that used in Model B (computational details are provided in the SI). Fig. 6c shows the Model C estimations obtained for the P-repeat isomers and 26-mers at 70 °C. Some improvements were achieved in the estimation for 8, as indicated by its higher coefficient of determination equal to 0.962.
The present models only consider the relaxation modes of Si–O–Si bond stretching of linear siloxane chains. For branched or cyclic siloxane structures, we should develop relaxation-based models with bending or ring flipping modes.
3 Conclusions
In the present work, we synthesized discrete sequence-specific oligosiloxanes 2–10 containing M, D, and P units. Using our synthetic methodology, we successfully prepared oligosiloxanes with precise molecular weights, M/P/D compositions, and sequences on the gram scale. This efficient preparation procedure allowed us to investigate the effects of structural factors on the melt viscosity. In particular, we observed a steep enhancement in viscosity by the consecutive P repeats and a distinct influence of the block sequence patterns on the viscosity of the 26-mers.We applied the Rouse model and its modifications to estimate the viscosities of the sequence-specific oligomers synthesized in this work and oligodimethylsiloxanes reported in the literature. Representing the specific sequences with matrices, Model A successfully simulated the cumulative viscosity enhancement in the successive P-repeat blocks. This model is useful for applications involving P-repeat siloxanes. By considering bond–bond correlations (local chain rigidity), Model B improved the viscosity estimations for the 26-mers. The semiflexible chain approach particularly improved the estimates for the oligosiloxanes with PPD and PDP sequences. Model C considered the friction coefficients to be composition-dependent and further increased the estimation accuracy. This study demonstrates that appropriate Rouse model variants can effectively explain how the composition and sequence influence the melt viscosity of oligosiloxanes.
The gram-scale synthesis procedure employed in the present work can be used to prepare different types of discrete sequence-specific siloxanes possessing various sequence patterns, cyclic or branched topologies, and other functionalities.16–18 These varieties include telechelic oligosiloxanes with definite functionalities, which exhibit a high application potential in functional copolymerization.43 In addition, further characterization of these oligosiloxanes will be beneficial for elucidating the influence of siloxane structures on their physicochemical properties and designing and developing siloxanes for future applications. For discrete sequence-specific oligosiloxanes, our group is currently studying their structure–property relationships by investigating phase transitions, thermal stability, miscibility, and single-crystal properties, as well as their applications as novel materials.
Author contributions
T. K. and K. M. conducted the design and synthesis of oligosiloxanes. H. M. carried out shear viscosity measurement and analysis. H. M. and K. M. discussed the results and wrote the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5py00958h.Acknowledgements
This work was supported by JST PRESTO (Grant Number: JPMJPR21AE), Japan. The authors thank Ms Mariko Okada for her help in the preparation of siloxane materials.References
- R. G. Jones, W. Ando and J. Chojnowski, Silicon-Containing Polymers, Kluwer Academic Publishers, Dordrecht, the Netherlands, 2000 Search PubMed.
- M. A. Brook, Silicon in Organic, Organometallic, and Polymer Chemistry, John Wiley, New York, 2000 Search PubMed.
- S. J. Clarson, M. J. Owen, S. D. Smith and M. E. Van Dyke, Advances in Silicones and Silicone-Modified Materials, ACS Publication, Washington, D. C., 2010 Search PubMed.
- A. Zlatanic, D. Radojcic, X. Wan, J. M. Messman, D. E. Bowen and P. R. Dvornic, J. Polym. Sci., Part A: Polym. Chem., 2019, 57, 1122 CrossRef CAS.
- G. N. Babu, S. S. Christopher and R. A. Newmark, Macromolecules, 1987, 20, 2654 CrossRef CAS.
- A. Zlatanic, D. Radojcic, X. Wan, J. M. Messman and P. R. Dvornic, Macromolecules, 2017, 50, 3532 Search PubMed.
- I. Yilgör and J. E. MacGrath, Adv. Polym. Sci., 1988, 86, 1 CrossRef.
- J. Shi, N. Zhao, S. Xia, S. Liu and Z. Li, Polym. Chem., 2019, 10, 2126 Search PubMed.
- K.-A. T. Nguyen, S. R. Clarke, J. Matisons, B. W. Skelton, A. H. White and E. Markovic, Silicon, 2014, 6, 21 CrossRef CAS.
- L. Zhu, X. Cheng, W. Su, J. Zhao and C. Zhou, Polymers, 2019, 11, 1989 CrossRef CAS PubMed.
- A. F. Schneider, E. K. Lu, G. Lu and M. A. Brook, J. Polym. Sci., 2020, 58, 3095 CrossRef CAS.
- Y. Yu, Y. Zhao, B. Huang, Y. Li, Y. Zhao, Z. Zhang and H.-F. Fei, Polymer, 2022, 249, 124865 CrossRef CAS.
- S. Wang, Z. Peng, Y. Huang, M. Zhu, Y. Liu and Y. Zhang, Polymer, 2024, 311, 127554 CrossRef CAS.
- B. R. Harkness, M. Tachikawa, H. Yu and I. Mita, Chem. Mater., 1998, 10, 1700 CrossRef CAS.
- T. M. Gädda and W. P. Weber, J. Polym. Sci., Part A:Polym. Chem., 2006, 44, 3629 CrossRef.
- K. Matsumoto, Y. Oba, Y. Nakajima, S. Shimada and K. Sato, Angew. Chem., Int. Ed., 2018, 57, 4637 Search PubMed.
- K. Matsumoto, S. Shimada and K. Sato, Chem. – Eur. J., 2019, 25, 920 CrossRef CAS PubMed.
- T. Kawatsu, J.-C. Choi, K. Sato and K. Matsumoto, Macromol. Rapid Commun., 2021, 42, 20000593 CrossRef PubMed.
- T. Kawatsu, H. Minamikawa, K. Sato and K. Matsumoto, Polym. Chem., 2024, 15, 2740 Search PubMed.
- C. B. Hurd, J. Am. Chem. Soc., 1946, 68, 364 Search PubMed.
- D. F. Wilcock, J. Am. Chem. Soc., 1946, 68, 691 Search PubMed.
- E. B. Baker, A. J. Barry and M. J. Hunter, Ind. Eng. Chem., 1946, 38, 1117 CrossRef CAS.
- A. J. Barry, J. Appl. Phys., 1946, 17, 1020 CrossRef CAS.
- H. I. Waterman, W. E. R. van Herwijnen and H. W. den Hartog, J. Appl. Chem., 1958, 8, 62 CrossRef.
- T. G. Fox and V. R. Allen, J. Chem. Phys., 1964, 41, 344 CrossRef CAS.
- T. Kataoka and S. Ueda, J. Polym. Sci., Part B:Polym. Lett., 1966, 4, 317 CrossRef CAS.
- P. R. Dvornic, J. D. Jovanovic and M. N. Govedarica, J. Appl. Polym. Sci., 1993, 49, 1497 CrossRef CAS.
- G. C. Berry and T. G. Fox, Adv. Polym. Sci., 1968, 5, 261 CrossRef.
- M. Doi and S. F. Edwards, The Theory of Polymer Dynamics, Clarendon Press, Oxford, 1986 Search PubMed.
- G. Strobl, The Physics of Polymers, Springer, Berlin Heidelberg New York, 2007 Search PubMed.
- S. M. Aharoni, Macromolecules, 1983, 16, 1722 CrossRef CAS.
- P. E. Rouse, J. Chem. Phys., 1953, 21, 1272 CrossRef CAS.
- A. Casale, R. S. Porter and J. F. Johnson, J. Macromol. Sci., Part C:Polym. Rev., 1971, 5, 387 Search PubMed.
- M. Abdel-Goad, W. Pyckhout-Hintzen, S. Kahle, J. Allgaier, D. Richter and L. J. Fetters, Macromolecules, 2004, 37, 8135 CrossRef CAS.
- L. Yang, T. Uneyama, Y. Masubuchi and Y. Doi, J. Soc. Rheol., Jpn., 2021, 49, 267 CrossRef CAS.
- G. Tsolou, N. Stratikis, C. Baig, P. S. Stephanou and V. G. Marvrantzas, Macromolecules, 2010, 43, 10692 CrossRef CAS . The discrete model is presented in the Appendix.
- X. Cui, Y. Luo, Y. Yang and P. Tang, Macromolecules, 2025, 58, 1898 CrossRef CAS.
- M. Bixon and R. Zwanzig, J. Chem. Phys., 1978, 68, 1896 CrossRef CAS.
- R. G. Winkler, P. Reineker and L. Harnau, J. Chem. Phys., 1994, 101, 811 CrossRef.
- M. G. Guenza, J. Phys.:Condens. Matter, 2008, 20, 033101 CrossRef.
- M. Dolgushev and A. Blumen, Macromolecules, 2009, 42, 5378 Search PubMed.
- A. R. Tejedor, J. R. Tejedor and J. Ramírez, J. Chem. Phys., 2022, 157, 164904 CrossRef CAS PubMed.
- E. Yilgör and I. Yilgör, Prog. Polym. Sci., 2014, 39, 1165 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |