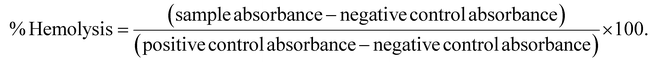Open Access Article
Open Access ArticleChitosan-coated cobalt ferrite nanocomplexes with dual antimicrobial activity and hemocompatibility
Md Salman Shakila,
Shakil Ahmed Polashb,
Md. Monir Hossainb,
Mahruba Sultana Niloyc,
Mohammad Mahfuz Ali Khan Shawanc,
Sheikh Manjura Hoque *d,
Md. Ashraful Hasan*c and
Satya Ranjan Sarker
*d,
Md. Ashraful Hasan*c and
Satya Ranjan Sarker b
b
aDepartment of Microbiology, Brac University, Kha 224 Pragati Sarani, Merul Badda, Dhaka 1212, Bangladesh
bDepartment of Biotechnology and Genetic Engineering, Jahangirnagar University, Savar, Dhaka 1342, Bangladesh. E-mail: satya.sarker@bgeju.edu.bd
cDepartment of Biochemistry and Molecular Biology, Jahangirnagar University, Savar, Dhaka 1342, Bangladesh. E-mail: ashrafulhasan@juniv.edu
dMaterial Science Division, Atomic Energy Centre, Dhaka 1000, Bangladesh
First published on 1st May 2026
Abstract
The escalating prevalence of antibiotic-resistant bacterial infections represents a critical global health challenge, necessitating the development of alternative antimicrobial strategies that are both effective and biocompatible. Nanomaterials offer a versatile platform in this regard, as their physicochemical properties can be tailored to enhance antibacterial performance. Among these, cobalt ferrite nanoparticles (CFs) exhibit attractive magnetic properties, chemical stability, and multifunctionality for biomedical applications; however, their clinical translation is hindered by aggregation, limited colloidal stability, and potential biocompatibility concerns. Building on our previous work demonstrating the biocompatibility and imaging potential of chitosan (CH)-coated CFs (CCFs), this study investigates their antibacterial efficacy against a diverse panel of Gram-positive (Bacillus subtilis RBW and Staphylococcus aureus) and Gram-negative bacteria (enteropathogenic Escherichia coli EPEC, E. coli DH5α, E. coli K-12, Salmonella typhi AF-4500, Shigella flexneri 3C, and Vibrio cholerae). Antibacterial activity was evaluated using the zone of inhibition (ZOI), minimum inhibitory concentration (MIC), and CellTox™ Green assays. CCFs demonstrated significantly enhanced antibacterial activity compared to CH alone or uncoated CFs, highlighting the synergistic contribution of the chitosan coating. Notably, S. aureus exhibited the highest susceptibility, with a maximum ZOI of ∼26.45 mm at 500 µg of CCFs. Elevated levels of malondialdehyde–thiobarbituric acid adducts following treatment suggest that membrane oxidative damage is a key mechanism underlying bacterial killing. Importantly, hemocompatibility assessments using rat and human erythrocytes revealed concentration-dependent cytotoxicity, indicating that CCFs maintain acceptable blood compatibility within a defined concentration range. Collectively, this study establishes CCFs as a promising dual-functional platform combining potent broad-spectrum antimicrobial activity with tunable hemocompatibility, underscoring their potential for future biomedical applications, provided that an optimal therapeutic window is carefully defined.
1. Introduction
The global rise of infectious diseases, compounded by the emergence of antibiotic-resistant Gram-positive and Gram-negative bacteria, poses a major public health challenge.1,2 This threat is further aggravated by the decline in new antibiotic approvals over the past decade,3 leaving common pathogens such as Staphylococcus aureus, Enterococcus faecalis, Escherichia coli EPEC, Salmonella typhi AF-4500, Shigella flexneri 3C, and Vibrio cholerae increasingly difficult to treat.4–9 The growing antibiotic resistance of these bacteria underscores the urgent need for safe and effective alternative therapeutics.2,10 Metal nanoparticles have emerged as promising antimicrobial agents due to their ability to disrupt bacterial membranes and limit resistance development pathways.11,12Among the magnetic nanoparticles, cobalt ferrite nanoparticles (CFs), a class of spinel-type ferrites, possess unique magnetic and physicochemical properties that enable applications in magnetic resonance imaging (MRI) contrast enhancement, hyperthermia, targeted drug delivery, and antimicrobial therapy.13–15 However, their biomedical application is often limited by particle agglomeration, poor aqueous dispersibility, and potential cytotoxicity. To address these limitations, surface modification has been widely explored as an effective strategy to improve their biological performance.
Chitosan (CH), a natural, biocompatible polymer,16,17 is particularly attractive for this purpose due to its broad applications in gene delivery, wound healing, tissue engineering and antimicrobial formulations.18 Beyond serving as a stabilizing coating, CH can improve nanoparticle dispersion, target specificity, and biological interactions while reducing aggregation and clearance by the reticuloendothelial system.12,19,20 Previous studies have also shown that CH may enhance the antimicrobial activity of CF-based nanomaterials.21,22 Moreover, CFs, CH, and CH-coated CFs (CCFs) have each been reported to possess antimicrobial potential.19,22–25 However, a comparative evaluation of their antimicrobial performance against commercially available FDA-approved antibiotics (e.g. ampicillin, tetracycline, ciprofloxacin) or disinfectant (e.g. povidone iodine) remains limited. The effects of CCFs on bacterial membrane lipid peroxidation and their hemocompatibility toward human red blood cells (HRBCs) have also not been investigated thoroughly. Such investigation is important for understanding both the antimicrobial potential and the future translational relevance of CCFs.
In the present study, we report the synthesis of CH-coated CFs by a co-precipitation method26 and systematically evaluate their antibacterial activity against Gram-positive (B. subtilis, S. aureus) and Gram-negative (E. coli EPEC, E. coli DH5α, S. typhi AF-4500, S. flexneri 3C, V. cholerae, and E. coli K-12) bacterial strains. Their antimicrobial efficacy was compared with commercially available antibiotics by measuring the zone of inhibition (ZOI), the clear circular region surrounding an antimicrobial agent where bacterial growth is suppressed. Additionally, we examined the bacterial membrane damage through lipid peroxidation analysis and evaluated the hemocompatibility of CFs and CCFs toward HRBCs. Taken together, this study explores the antimicrobial potential of CCFs while also assessing their hemocompatibility in a concentration-dependent manner, thereby providing a more balanced evaluation of their biomedical applicability.
2. Materials and methods
2.1 Chemicals and materials
Peptone, yeast extract, sodium chloride, and EDTA were collected from UNI-CHEM, China. Mueller Hinton Agar was obtained from Merk, Germany. Enterococcus faecalis, Staphylococcus aureus, Enteropathogenic Escherichia coli (EPEC), Salmonella typhi AF-4500, Shigella flexneri 3C, Vibrio cholera, and Escherichia coli K-12 were obtained from the department of Biochemistry and Molecular Biology, Jahangirnagar University, Savar, Dhaka 1342, Bangladesh. Escherichia coli DH5α and Bacillus subtilis RBW were obtained from the department of Biotechnology and Genetic Engineering, Jahangirnagar University, Savar, Dhaka 1342, Bangladesh.2.2 Methods
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm, 20 min), washed ten times, and annealed at 90 °C for 72 h to obtain the CFs.
000 rpm, 20 min), washed ten times, and annealed at 90 °C for 72 h to obtain the CFs.| CoCl2·6H2O + 2NaOH → Co(OH)2 + 2NaCl + 6H2O |
| 2FeCl3·6H2O + 6NaOH → 2Fe(OH)3 + 6NaCl + 12H2O |
| Co(OH)2 + 2Fe(OH)3 → CoFe2O4 + 4H2O |
Overall reaction:
| CoCl2·6H2O + 8NaOH + 2FeCl3·6H2O → CoFe2O4 + 8NaCl + 22H2O |
CFs were then coated using a 2% CH solution to prepare CCFs. 2% CH (w/v) was prepared by adding CH (Sigma-Aldrich, low molecular weight, quality level 100, 75–85% deacetylated) into a mixture of water (94 mL) and an acetic acid solution (6 mL, Sigma-Aldrich, 2 N). After 48 h of stirring, the CH solution was centrifuged for 20 minutes at 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm. 1 mL of the clear supernatant was then used to coat 20 mg CFs via repeated vortexing and ultrasonication.
000 rpm. 1 mL of the clear supernatant was then used to coat 20 mg CFs via repeated vortexing and ultrasonication.
Minimum inhibitory concentration (MIC) value was determined using the broth dilution method.27 Overnight bacterial cultures (10 μL) were diluted in 990 μL fresh LB broth and incubated at 37 °C with shaking (120 rpm) for 4 h. The cultures were then treated with a range of CH, CF, and CCF concentrations and incubated overnight. Bacterial growth was assessed by measuring OD600 using a UV-vis spectrophotometer to determine MIC values.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 35 min. The supernatant was collected and centrifuged again at the same speed for 20 min to remove residual protein precipitates and dead cells. The resulting supernatant, containing malondialdehyde (MDA), was mixed with 4 mL of freshly prepared 0.67% thiobarbituric acid (TBA) solution and incubated in a hot water bath for 10 min to allow the formation of the MDA–TBA adduct. After cooling to room temperature, the absorbance of the MDA–TBA complex was measured at 532 nm using a UV-vis spectrophotometer (Specord® 205, Analytik Jena, Germany).
000 rpm for 35 min. The supernatant was collected and centrifuged again at the same speed for 20 min to remove residual protein precipitates and dead cells. The resulting supernatant, containing malondialdehyde (MDA), was mixed with 4 mL of freshly prepared 0.67% thiobarbituric acid (TBA) solution and incubated in a hot water bath for 10 min to allow the formation of the MDA–TBA adduct. After cooling to room temperature, the absorbance of the MDA–TBA complex was measured at 532 nm using a UV-vis spectrophotometer (Specord® 205, Analytik Jena, Germany).3. Results and discussion
3.1 Characterization
Previously synthesized CFs (diameter ∼10 nm) were used in this study.26 The surface of the CFs was modified with CH to improve their biocompatibility for potential biomedical applications. Briefly, CFs were prepared via the co-precipitation method and subsequently coated with CH. The resulting CCFs were characterized using scanning transmission electron microscopy (STEM), Raman spectroscopy, Fourier-transform infrared (FTIR) spectroscopy, differential scanning calorimetry (DSC), thermogravimetric analysis (TGA), dynamic light scattering (DLS), and zeta (ζ) potential measurements. CCFs exhibited higher positive surface charges than CH or CFs alone (Table S1).26 In the present study, we evaluated the antimicrobial activity of CH, CFs, and CCFs against two Gram-positive and seven Gram-negative bacterial strains.3.2 Antimicrobial activity
The disc diffusion assay was performed to evaluate the antibacterial activity of CH, CFs, and CCFs. CFs alone exhibited poor antimicrobial activity (Table 1), whereas CH coating substantially enhanced their efficacy. The highest activity of CCFs was observed against the Gram-positive Staphylococcus aureus strain (Table 1), outperforming even commercially available FDA-approved antimicrobial agents, including ampicillin, tetracycline, ciprofloxacin and povidone iodine. In contrast, CCFs were less effective against Bacillus subtilis RBW. Povidone iodine demonstrated nearly uniform antibacterial activity across all tested strains. Interestingly, CCFs also showed activity against amoxicillin-resistant Gram-negative Escherichia coli EPEC. Consistent with the zones of inhibition, MIC measurements indicated that CCFs (0.000391–0.000781 µg) were more potent than CH or CFs, while CFs and CH displayed similar MIC values (0.0125 µg) against E. coli DH5α, E. faecalis, S. aureus, and E. coli K12 (Table S1). Previously, Kajani et al.34 reported that CCFs had higher antibacterial activity against E. coli than CFs or amoxicillin. The enhanced antimicrobial activity of CCFs is likely attributable to increased ROS production,19 while CH coating promotes the attachment of CCFs to the bacterial cell wall or membrane, facilitating cell lysis.35| Drug/Drug candidates | Bacterial strain | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Bacillus subtilis RBW | Escherichia coli DH5α | Enteropathogenic Escherichia coli (EPEC) | Salmonella typhi AF-4500 | Shigella flexneri 3C | Vibrio cholerae | Escherichia coli K-12 | Enterococcus faecalis | Staphylococcus aureus | |
| CFs: cobalt ferrite nanoparticles, CH: chitosan, CCFs: chitosan-coated cobalt ferrite nanoparticles. | |||||||||
| CH (300 µg) | — | 7.45 ± 0.05 | — | 7 ± 0 | 7.05 ± 0 | 7.1 ± 0.1 | 7.45 ± 0.05 | 18.7 ± 0.2 | 20.5 ± 0.2 |
| CH (400 µg) | 6.9 ± 0.1 | 7.85 ± 0.05 | — | 7.45 ± 0.05 | 7.45 ± 0.05 | 8.05 ± 0.05 | 7.55 ± 0.05 | 20.55 ± 0.25 | 22.4 ± 0.1 |
| CH (500 µg) | 7.05 ± 0.05 | 8.15 ± 0.05 | 7.45 ± 0.05 | 8.05 ± 0.05 | 8.1 ± 0.05 | 9.9 ± 0.1 | 10.05 ± 0.05 | 21.7 ± 0.4 | 24.25 ± 0.15 |
| CFs (300 µg) | 6.9 ± 0.1 | — | — | — | — | — | — | — | — |
| CFs (400 µg) | 7 ± 0 | — | — | — | — | — | — | — | — |
| CFs (500 µg) | 7.1 ± 0.1 | — | — | — | 7.45 ± 0.05 | — | — | 8.05 ± 0.15 | — |
| CCFs (300 µg) | 7.45 ± 0.05 | 15.4 ± 0.1 | 8.55 ± 0.05 | 9.05 ± 0.05 | 6.9 ± 0.1 | 9.05 ± 0.05 | 8.05 ± 0.05 | 18.15 ± 0.05 | 21.1 ± 0.3 |
| CCFs (400 µg) | 9.05 ± 0.05 | 17.1 ± 0.1 | 10.05 ± 0.05 | 10.1 ± 0.1 | 9.45 ± 0.05 | 9.95 ± 0.05 | 8.95 ± 0.05 | 19.55 ± 0.15 | 23.45 ± 0.65 |
| CCFs (500 µg) | 9.1 ± 0.1 | 20.05 ± 0.05 | 12.015 ± 0.015 | 11.1 ± 0.1 | 11.05 ± 0.05 | 11.4 ± 0.1 | 11.1 ± 0.1 | 23.55 ± 0.05 | 26.45 ± 0.05 |
| Ampicillin (10 µg) | 10.95 ± 0.05 | 12.9 ± 0.1 | — | 23.95 ± 0.05 | 23.1 ± 0.1 | 14.2 ± 0.1 | 15.9 ± 0.1 | 7.95 ± 0.15 | 8.35 ± 0.15 |
| Tetracycline (10 µg) | 17.15 ± 0.15 | 17.1 ± 0.1 | 20.05 ± 0.05 | 15.1 ± 0.1 | 19.95 ± 0.05 | 8.1 ± 0.1 | 17.1 ± 0.1 | 12.3 ± 0.1 | 11.45 ± 0.05 |
| Ciprofloxacin (5 µg) | 24.9 ± 0.1 | 34.2 ± 0.1 | 36.9 ± 0.1 | 38.9 ± 0.1 | 32.05 ± 0.05 | 22.05 ± 0.05 | 29.4 ± 0.05 | 19.35 ± 0.15 | 19.35 ± 0.15 |
| Povidone iodine (2000 µg) | 10.1 ± 0.1 | 10.95 ± 0.05 | 9.55 ± 0.05 | 10.45 ± 0.05 | 10.5 ± 0 | 10.05 ± 0.05 | 12.05 ± 0.05 | 19.55 ± 0.15 | 17.4 ± 0.2 |
3.3 Compromised bacterial cell membrane
CellTox™ Green is a DNA-binding dye that penetrates bacteria with compromised cell walls, while intact bacterial membranes exclude the dye.36 Among the Gram-positive bacteria, S. aureus treated with CCFs exhibited the highest fluorescence intensity, followed by E. faecalis (Fig. 1), with intensities approximately sevenfold higher than those of untreated controls. All Gram-negative strains showed nearly sixfold increases in fluorescence compared with their respective untreated controls. A significant increase in fluorescence was also observed in CCF-treated Gram-positive and Gram-negative bacteria compared with the untreated group and CF- and CH-treated groups. These results are consistent with the zones of inhibition data (Fig. 2). Green fluorescence of dead bacteria treated with CH, CFs, and CCFs was further confirmed under fluorescence microscopy (Fig. 2), with the highest level of cell death observed following CCF treatment. Previously, it has been reported that treatment with biogenic metal nanoparticles compromised the bacterial membrane, leading to an increase in the uptake of CellTox™ Green and fluorescence.27,37The trypan blue dye exclusion assay confirmed bacterial cell wall damage. Hydrophobic interactions between the nanoparticles and the bacterial cell wall caused structural disruption, allowing trypan blue to penetrate the cytosol of damaged or dead bacteria.38 Cells with compromised walls appeared blue under a phase-contrast microscope (Fig. S1).
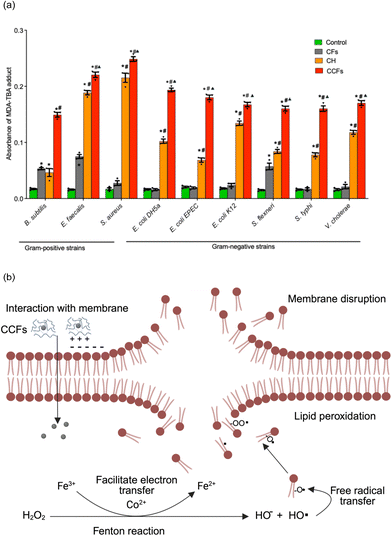
3.4 Lipid peroxidation of the bacterial membrane
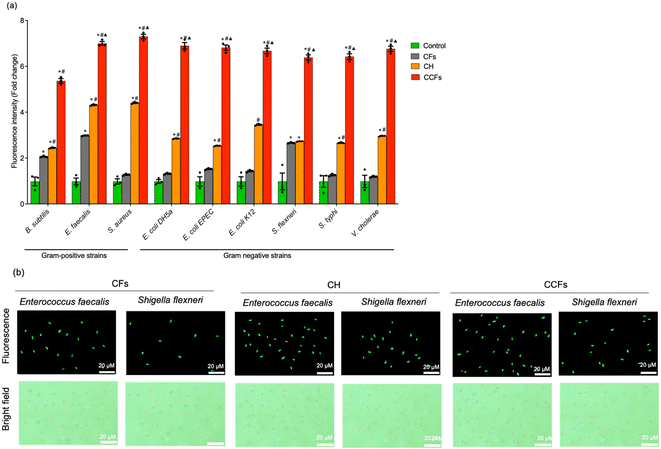
The LPO assay was performed to assess the oxidative damage to bacterial cell membrane fatty acids. The formation of the MDA–TBA adduct indicates cell wall damage induced by the therapeutic agents. Among the tested strains, treatment of S. aureus with CCFs resulted in the highest MDA–TBA adduct formation (Fig. 3a). Notably, E. faecalis also exhibited substantial MDA–TBA levels, indicating significant oxidative damage among the Gram-positive bacteria with CCFs (Fig. 3a). Previously, it has been reported that cobalt or iron doping in silver nanoparticles increased MDA adduct formation in both Gram-positive bacteria (E. faecalis and B. subtilis) and Gram-negative bacteria (E. cloacae and Pseudomonas putida).39The composition of bacterial cell walls varies between species, resulting in different interactions with the test samples.40 Consequently, different bacterial strains produced varying amounts of MDA–TBA adducts following treatment. LPO occurred due to the oxidation of bacterial cell membrane fatty acids induced by CH, CFs, or CCFs.
Positive surface charge of CH, CFs or CCFs could facilitate their interaction with negatively charged bacterial cell membranes. After CF internalization, ROS can be generated via the Fenton reaction and oxidize membrane polyunsaturated fatty acids.41 Membrane LPO can disrupt the integrity of the cell membrane (Fig. 3b).27 Additionally, ROS-mediated oxidative stress disrupts the electron transport chain and alters bacterial metabolic processes. Such disruptions can trigger the expression of apoptotic genes and oxidative stress-related proteins, ultimately leading to bacterial cell apoptosis.42,43
3.5 In vitro hemolytic activity
In vivo application of nanocomplexes requires excellent blood compatibility, including minimal hemolytic activity.14 Due to ethical concerns surrounding the direct human exposure without prior animal studies, we evaluated the blood compatibility of CH, CFs, and CCFs using HRBCs and RRBCs to predict their potential effects in vivo (Fig. 4). Lee et al.44 reported that acetylated CH nanoparticles exhibit negligible hemolytic activity, which can vary with their chemical modifications. In our study, CCFs induced higher hemolytic activity in both HRBCs and RRBCs compared to pristine CFs, likely due to differences in particle distribution and interaction with RBCs. While CFs tended to aggregate and precipitate at the bottom of the tube, CCFs remained homogeneously dispersed with altered particle size and surface area. RRBC membrane integrity was affected differently from that of HRBC with increasing concentrations of the test samples. These observations are consistent with the model proposed by Thomassen et al.,45 in which RBC membranes locally induce curvature and exhibit reduced adhesion energy upon interaction with aggregated nanoparticles, resulting in lower hemolytic activity compared with monodispersed particles of the same size. Additionally, particle size and surface area contribute significantly to hemolysis.46 Interestingly, CCFs showed higher cytotoxicity toward RRBCs than HRBCs in terms of HC50 values, which is less than the amount (500 μg) that exhibits the highest antibacterial activity against S. aureus (Fig. 4). In vivo studies also indicated that CCFs are well tolerated by RRBCs at lower concentrations.26 Given the observed in vitro toxicity at higher concentrations, establishing a safe therapeutic window for CCFs is essential before considering direct human applications.4. Conclusion
CCFs exhibited potent antimicrobial activity against both Gram-positive and Gram-negative bacteria, outperforming CH and CFs alone, as shown by larger zones of inhibition, lower MICs, and enhanced cytotoxicity in the CellTox™ Green assay. Among other bacterial strains, Staphylococcus aureus was the most susceptible strain, while LPO studies suggested that oxidative membrane damage is involved in the bactericidal mechanism. Overall, CH functionalization is an effective strategy for improving the antimicrobial performance of CFs. However, the hemolytic activity of CCFs is concentration-dependent, underscoring the need for careful dose optimization. Further work should, therefore, focus on defining a safe therapeutic window, establishing the behavior of these nanocomplexes in physiologically relevant systems, and validating their efficacy in vivo.Author contributions
Conceptualization: M. S. S., M. A. H, and S. R. S; nanoparticle synthesis: M. S. S. and S. M. H.; antimicrobial and hemolysis activity: M. S. S., S. A. P., M. M. H, and M. S. N.; microscopic analysis: M. M. H.; data analysis and diagram: M. S. S., S. A. P., and M. M. H.; manuscript writing: M. S. S. and S. A. P.; manuscript revision and approval: M. S. S., M. A. H., and S. R. S.Ethics statement
All animal procedures were performed in accordance with the Guidelines for Care and Use of Laboratory Animals of Jahangirnagar University and experiments were approved by the Animal Ethics Committee of the Biosafety, Biosecurity & Ethical Committee, Faculty of Biological Sciences, Jahangirnagar (Ref. No: BBEC, JU/M 2018 (11) 1).All experiments were performed in accordance with the Guidelines of Jahangirnagar University, and Experiments were approved by the ethics committee at the Biosafety, Biosecurity & Ethical Committee, Faculty of Biological Sciences, Jahangirnagar University (Ref. No: BBEC, JU/M 2019 (4) 1). Informed consent was obtained from the human participants of this study.
Conflicts of interest
There are no conflicts to declare.Data availability
Data are included in the article or in the supplementary information (SI). Supplementary information: Table S1: Characterization parameters of CFs, CH and CCFs. Table S2: MIC of CCFs, CH and CFs against bacteria. Fig. S1: Trypan blue dye exclusion assay showing CH, CF, and CCF induced bacterial cell wall damage. See DOI: https://doi.org/10.1039/d6pm00045b.Acknowledgements
Jahangirnagar University Research Grant 2019, and University Grants Commission (UGC)-Jahangirnagar University Joint Research Grant 2019, Government of Bangladesh, provided partial funding for this research project. The authors are thankful to Wazed Mia Science Research Centre, Jahangirnagar University, for allowing the use of their microscope facilities.References
- A. Azam, A. S. Ahmed, M. Oves, M. S. Khan, S. S. Habib and A. Memic, Antimicrobial activity of metal oxide nanoparticles against Gram-positive and Gram-negative bacteria: a comparative study, Int. J. Nanomed., 2012, 7, 6003–6009 CrossRef CAS PubMed.
- B. Aslam, W. Wang, M. I. Arshad, M. Khurshid, S. Muzammil, M. H. Rasool, M. A. Nisar, R. F. Alvi, M. A. Aslam, M. U. Qamar, M. K. F. Salamat and Z. Baloch, Antibiotic resistance: A rundown of a global crisis, Infect. Drug Resist., 2018, 11, 1645–1658 CrossRef CAS PubMed.
- X. Li, S. M. Robinson, A. Gupta, K. Saha, Z. Jiang, D. F. Moyano, A. Sahar, M. A. Riley and V. M. Rotello, Functional gold nanoparticles as potent antimicrobial agents against multi-drug-resistant bacteria, ACS Nano, 2014, 8(10), 10682–10686 CrossRef CAS PubMed.
- R. Barnett, Typhoid fever, Lancet, 2016, 388(10059), 2467 CrossRef PubMed.
- K. V. Singh, S. R. Nallapareddy and B. E. Murray, Importance of the ebp (endocarditis- and biofilm-associated pilus) locus in the pathogenesis of Enterococcus faecalis ascending urinary tract infection, J. Infect. Dis., 2007, 195(11), 1671–1677 CrossRef CAS PubMed.
- T. J. Ochoa and C. A. Contreras, Enteropathogenic Escherichia coli infection in children, Curr. Opin. Infect. Dis., 2011, 24(5), 478–483 CrossRef PubMed.
- S. Y. Tong, J. S. Davis, E. Eichenberger, T. L. Holland and V. G. Fowler, Jr., Staphylococcus aureus infections: Epidemiology, pathophysiology, clinical manifestations, and management, Clin. Microbiol. Rev., 2015, 28(3), 603–661 CrossRef CAS PubMed.
- A. V. Jennison and N. K. Verma, Shigella flexneri infection: Pathogenesis and vaccine development, FEMS Microbiol. Rev., 2004, 28(1), 43–58 CrossRef CAS.
- E. J. Nelson, J. B. Harris, J. G. Morris, Jr., S. B. Calderwood and A. Camilli, Cholera transmission: The host, pathogen and bacteriophage dynamic, Nat. Rev. Microbiol., 2009, 7(10), 693–702 CrossRef CAS PubMed.
- A. C. Rios, C. G. Moutinho, F. C. Pinto, F. S. Del Fiol, A. Jozala, M. V. Chaud, M. M. Vila, J. A. Teixeira and V. M. Balcão, Alternatives to overcoming bacterial resistances: State-of-the-art, Microbiol. Res., 2016, 191, 51–80 CrossRef CAS PubMed.
- Y. N. Slavin, J. Asnis, U. O. Häfeli and H. Bach, Metal nanoparticles: Understanding the mechanisms behind antibacterial activity, J. Nanobiotechnol., 2017, 15(1), 65 CrossRef PubMed.
- S. H. Hussein-Al-Ali, M. E. El Zowalaty, M. Z. Hussein, B. M. Geilich and T. J. Webster, Synthesis, characterization, and antimicrobial activity of an ampicillin-conjugated magnetic nanoantibiotic for medical applications, Int. J. Nanomed., 2014, 9, 3801–3814 CrossRef CAS PubMed.
- A. Ashour, A. I. El-Batal, M. A. Maksoud, G. S. El-Sayyad, S. Labib, E. Abdeltwab and M. El-Okr, Antimicrobial activity of metal-substituted cobalt ferrite nanoparticles synthesized by sol–gel technique, Particuology, 2018, 40, 141–151 CrossRef CAS.
- H. Wu, G. Liu, X. Wang, J. Zhang, Y. Chen, J. Shi, H. Yang, H. Hu and S. Yang, Solvothermal synthesis of cobalt ferrite nanoparticles loaded on multiwalled carbon nanotubes for magnetic resonance imaging and drug delivery, Acta Biomater., 2011, 7(9), 3496–3504 CrossRef CAS PubMed.
- F. Ahmad and Y. Zhou, Pitfalls and challenges in nanotoxicology: A case of cobalt ferrite (CoFe2O4) nanocomposites, Chem. Res. Toxicol., 2017, 30(2), 492–507 Search PubMed.
- S. K. Shukla, A. K. Mishra, O. A. Arotiba and B. B. Mamba, Chitosan-based nanomaterials: A state-of-the-art review, Int. J. Biol. Macromol., 2013, 59, 46–58 CrossRef CAS PubMed.
- A. Rafique, K. Mahmood Zia, M. Zuber, S. Tabasum and S. Rehman, Chitosan functionalized poly(vinyl alcohol) for prospects biomedical and industrial applications: A review, Int. J. Biol. Macromol., 2016, 87, 141–154 CrossRef CAS PubMed.
- W. Rao, H. Wang, J. Han, S. Zhao, J. Dumbleton, P. Agarwal, W. Zhang, G. Zhao, J. Yu, D. L. Zynger, X. Lu and X. He, Chitosan-decorated doxorubicin-encapsulated nanoparticle targets and eliminates tumor reinitiating cancer stem-like cells, ACS Nano, 2015, 9(6), 5725–5740 CrossRef CAS PubMed.
- D. Gingasu, I. Mindru, L. Patron, A. Ianculescu, E. Vasile, G. Marinescu, S. Preda, L. Diamandescu, O. Oprea and M. Popa, Synthesis and characterization of chitosan-coated cobalt ferrite nanoparticles and their antimicrobial activity, J. Inorg. Organomet. Polym. Mater., 2018, 28(5), 1932–1941 CrossRef CAS.
- C. I. Covaliu, I. Jitaru, G. Paraschiv, E. Vasile, S.-Ş. Biriş, L. Diamandescu, V. Ionita and H. Iovu, Core–shell hybrid nanomaterials based on CoFe2O4 particles coated with PVP or PEG biopolymers for applications in biomedicine, Powder Technol., 2013, 237, 415–426 CrossRef CAS.
- V. Umaiya Bharathi and S. Thambidurai, Bio-fabrication of chitosan-coated cobalt oxide nanocomposite for enhanced bacterial inhibition and oxidative stress mitigation, Chem. Phys. Lett., 2024, 855, 141561 CrossRef CAS.
- D. G. Guedes, G. G. Guedes, J. d. O. d. Silva, A. L. d. Silva, C. B. B. Luna, B. P. G. d. L. Damasceno and A. C. F. d. M. Costa, Development of scaffolds with chitosan magnetically activated with cobalt nanoferrite: A study on physical–chemical, mechanical, cytotoxic and antimicrobial behavior, Pharmaceuticals, 2024, 17(10), 1332 CrossRef CAS PubMed.
- L. Doan, A. H. Huynh, K. Tran, Q. N. Le and K. G. Huynh, Surface modifications of cobalt ferrites nanoparticles with chitosan, polyethylene glycol, polyvinyl alcohol, and polyvinylpyrrolidone as antibacterial agents against Staphylococcus aureus, Pseudomonas aeruginosa, and Salmonella enterica, Polyhedron, 2025, 267, 117354 CrossRef CAS.
- M. Ganan, A. V. Carrascosa and A. J. Martínez-Rodríguez, Antimicrobial activity of chitosan against Campylobacter spp. and other microorganisms and its mechanism of action, J. Food Prot., 2009, 72(8), 1735–1738 CrossRef CAS PubMed.
- Y. J. Jing, Y. J. Hao, H. Qu, Y. Shan, D. S. Li and R. Q. Du, Studies on the antibacterial activities and mechanisms of chitosan obtained from cuticles of housefly larvae, Acta Biol. Hung., 2007, 58(1), 75–86 CrossRef CAS PubMed.
- M. S. Shakil, M. A. Hasan, M. F. Uddin, A. Islam, A. Nahar, H. Das, M. N. I. Khan, B. P. Dey, B. Rokeya and S. M. Hoque, In vivo toxicity studies of chitosan-coated cobalt ferrite nanocomplex for its application as MRI contrast dye, ACS Appl. Bio Mater., 2020, 3(11), 7952–7964 CrossRef CAS PubMed.
- M. S. Niloy, M. M. Hossain, M. Takikawa, M. S. Shakil, S. A. Polash, K. M. Mahmud, M. F. Uddin, M. Alam, R. D. Shubhra, M. M. A. K. Shawan, T. Saha, S. Takeoka, M. A. Hasan and S. Ranjan Sarker, Synthesis of biogenic silver nanoparticles using Caesalpinia digyna and investigation of their antimicrobial activity and in vivo biocompatibility, ACS Appl. Bio Mater., 2020, 3(11), 7722–7733 CrossRef CAS PubMed.
- R. Mondal, S. A. Polash, T. Saha, Z. Islam, M. M. Sikder, N. Alam, M. S. Hossain and S. R. Sarker, Investigation of the phytoconstituents and bioactivity of various parts of wild type and cultivated Phyllanthus emblica L, Adv. Biosci. Biotechnol., 2017, 8, 211–227 CrossRef CAS.
- W. Strober, Trypan blue exclusion test of cell viability, Curr. Protoc. Immunol., 2001 Search PubMed , Appendix 3, Appendix 3B.
- M. M. Hossain, S. A. Polash, M. Takikawa, R. D. Shubhra, T. Saha, Z. Islam, S. Hossain, M. A. Hasan, S. Takeoka and S. R. Sarker, Investigation of the antibacterial activity and in vivo cytotoxicity of biogenic silver nanoparticles as potent therapeutics, Front. Bioeng. Biotechnol., 2019, 7, 239 CrossRef PubMed.
- S. Ranjan Sarker, S. A. Polash, J. Boath, A. E. Kandjani, A. Poddar, C. Dekiwadia, R. Shukla, Y. Sabri and S. K. Bhargava, Functionalization of elongated tetrahexahedral au nanoparticles and their antimicrobial activity assay, ACS Appl. Mater. Interfaces, 2019, 11(14), 13450–13459 CrossRef CAS PubMed.
- J. H. Zhang, T. D. Chung and K. R. Oldenburg, A simple statistical parameter for use in evaluation and validation of high throughput screening assays, J. Biomol. Screening, 1999, 4(2), 67–73 CrossRef PubMed.
- M. Hossain, S. A. Polash, M. Takikawa, R. D. Shubhra, T. Saha, Z. Islam, S. Hossain, M. Hasan, S. Takeoka and S. R. Sarker, Investigation of the antibacterial activity and in vivo cytotoxicity of biogenic silver nanoparticles as potent therapeutics, Front. Bioeng. Biotechnol., 2019, 7, 239 CrossRef PubMed.
- A. A. Kajani, A. Pouresmaeili and M. Kamali, Facile one-pot synthesis of the mesoporous chitosan-coated cobalt ferrite nanozyme as an antibacterial and MRI contrast agent, RSC Adv., 2024, 14(24), 16801–16808 RSC.
- D. Şen Karaman, S. Sarwar, D. Desai, E. M. Björk, M. Odén, P. Chakrabarti, J. M. Rosenholm and S. Chakraborti, Shape engineering boosts antibacterial activity of chitosan coated mesoporous silica nanoparticle doped with silver: a mechanistic investigation, J. Mater. Chem. B, 2016, 4(19), 3292–3304 RSC.
- M. Bittremieux, K. Mikoshiba and G. Bultynck, Data on cytotoxicity in HeLa and SU-DHL-4 cells exposed to DPB162-AE compound, Data Brief, 2017, 12, 91–96 CrossRef PubMed.
- S. A. Polash, M. M. Hossain, T. Saha and S. R. Sarker, Biogenic silver nanoparticles: A potent therapeutic agent, in Emerging Trends in Nanomedicine, ed. S. Singh, Springer Singapore, Singapore, 2021, pp. 81–127 Search PubMed.
- S. Ranjan Sarker, S. A. Polash, J. Boath, A. E. Kandjani, A. Poddar, C. Dekiwadia, R. Shukla, Y. Sabri and S. K. Bhargava, Functionalization of elongated tetrahexahedral Au nanoparticles and their antimicrobial activity assay, ACS Appl. Mater. Interfaces, 2019, 11(14), 13450–13459 CrossRef CAS PubMed.
- R. Torres-Mendieta, N. H. A. Nguyen, A. Guadagnini, J. Semerad, D. Łukowiec, P. Parma, J. Yang, S. Agnoli, A. Sevcu, T. Cajthaml, M. Cernik and V. Amendola, Growth suppression of bacteria by biofilm deterioration using silver nanoparticles with magnetic doping, Nanoscale, 2022, 14(48), 18143 RSC.
- T. Dörr, P. J. Moynihan and C. Mayer, Editorial: Bacterial cell wall structure and dynamics, Front. Microbiol., 2019, 10, 2051 CrossRef.
- N. T. Tasnim, N. Ferdous, M. M. H. Rumon and M. S. Shakil, The promise of metal-doped iron oxide nanoparticles as antimicrobial agent, ACS Omega, 2024, 9(1), 16–32 CrossRef CAS PubMed.
- L. Wang, C. Hu and L. Shao, The antimicrobial activity of nanoparticles: present situation and prospects for the future, Int. J. Nanomed., 2017, 12, 1227 CrossRef CAS PubMed.
- H.-J. Park, J. Y. Kim, J. Kim, J.-H. Lee, J.-S. Hahn, M. B. Gu and J. Yoon, Silver-ion-mediated reactive oxygen species generation affecting bactericidal activity, Water Res., 2009, 43(4), 1027–1032 CrossRef CAS PubMed.
- D.-W. Lee, K. Powers and R. Baney, Physicochemical properties and blood compatibility of acylated chitosan nanoparticles, Carbohydr. Polym., 2004, 58(4), 371–377 CrossRef CAS.
- L. C. Thomassen, V. Rabolli, K. Masschaele, G. Alberto, M. Tomatis, M. Ghiazza, F. Turci, E. Breynaert, G. Martra, C. E. Kirschhock, J. A. Martens, D. Lison and B. Fubini, Model system to study the influence of aggregation on the hemolytic potential of silica nanoparticles, Chem. Res. Toxicol., 2011, 24(11), 1869–1875 Search PubMed.
- Y. Zhao, X. Sun, G. Zhang, B. G. Trewyn, I. I. Slowing and V. S. Lin, Interaction of mesoporous silica nanoparticles with human red blood cell membranes: size and surface effects, ACS Nano, 2011, 5(2), 1366–1375 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |