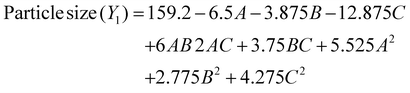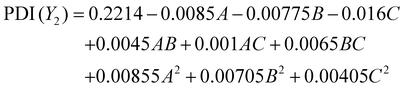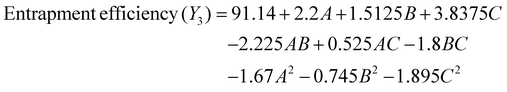Open Access Article
Open Access ArticleQbD-optimized HA–Pluronic nanomicelles for the targeted repurposing of tofacitinib in breast cancer
Suchita
Waghmare
 *a,
Rohini
Palekar
*a,
Rohini
Palekar
 a,
Pratiksha
Bramhe
a,
Pratiksha
Bramhe
 a,
Nilesh
Rarokar
a,
Nilesh
Rarokar
 b and
Pramod
Khedekar
b and
Pramod
Khedekar
 ac
ac
aDepartment of Pharmaceutical Sciences, Rashtrasant Tukadoji Maharaj Nagpur University, Nagpur, Maharashtra 440033, India. E-mail: suchitawaghmare26@gmail.com
bSharda Institute of Pharmaceutical Science and Research Centre, Goregaon, Gondia, India
cNagpur College of Pharmacy, Wanadongari, Hingna, India
First published on 6th March 2026
Abstract
Tofacitinib, a Janus kinase (JAK) inhibitor clinically approved for rheumatoid arthritis, holds significant potential for repurposing in breast cancer therapy due to its ability to suppress the oncogenic STAT3 signaling pathway; however, its therapeutic application is hindered by poor aqueous solubility, rapid systemic clearance, and non-specific distribution. To address these limitations, this study aimed to develop hyaluronic acid (HA)-decorated Pluronic F127 (PF127) micelles to enhance the solubility, bioavailability, and targeted delivery of Tofacitinib to CD44-overexpressing breast cancer cells. A Quality by Design (QbD) approach utilizing a Box–Behnken design (BBD) was employed to systematically optimize critical process parameters—drug-to-polymer ratio, polymer concentration, and stirring temperature—to minimize particle size and polydispersity index (PDI) while maximizing entrapment efficiency. The optimized micelles exhibited a uniform particle size of 159.2 ± 3.8 nm, a narrow PDI of 0.221, a high entrapment efficiency of 91.14%, and a negative zeta potential of −24.6 mV, ensuring colloidal stability. In vitro evaluation demonstrated a sustained, diffusion-controlled drug release profile and significantly enhanced cytotoxicity against MCF-7 breast cancer cells (IC50 14.2 µg mL−1) compared to the free drug (IC50 46.8 µg mL−1), attributed to CD44-mediated cellular uptake. Furthermore, in vivo pharmacokinetic analysis in Wistar rats revealed a 4.3-fold increase in oral bioavailability and a prolonged elimination half-life compared to free Tofacitinib, while histopathological studies confirmed the formulation's biocompatibility and safety in major organs. These findings collectively support the viability of the QbD-optimized HA–PF127 micellar system as a promising nanocarrier platform for the effective oral repurposing of Tofacitinib in breast cancer treatment.
1. Introduction
It is considered that breast cancer is the most frequently diagnosed malignancy and a leading cause of cancer-related mortality among women worldwide, accounting for an estimated 2.3 million new cases and 670![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 deaths in 2022.1 Although much progress has been made in molecular diagnostics and targeted therapies, systemic toxicity, multidrug resistance, and poor solubility of anticancer drugs remain major obstacles to the therapeutic management of breast cancer. The challenges mentioned above necessitate nanotechnology-based delivery systems that can improve solubility and pharmacokinetic profiles while achieving tumor-specific targeting with reduced adverse effects.2
000 deaths in 2022.1 Although much progress has been made in molecular diagnostics and targeted therapies, systemic toxicity, multidrug resistance, and poor solubility of anticancer drugs remain major obstacles to the therapeutic management of breast cancer. The challenges mentioned above necessitate nanotechnology-based delivery systems that can improve solubility and pharmacokinetic profiles while achieving tumor-specific targeting with reduced adverse effects.2
Among the nanocarrier systems, PMs have emerged as one of the versatile platforms for delivering anticancer drugs owing to their amphiphilic structure, biocompatibility, and high drug-loading ability. They self-assemble in aqueous media, forming a hydrophobic core that allows the encapsulation of poorly water-soluble drugs and a hydrophilic corona that provides colloidal stability.3 Pluronic F127, an FDA-approved triblock copolymer of PEO and PPO, has been widely used for the delivery of drugs because it is considered safe and promotes the formation of stable micellar systems. However, passive delivery only is usually not sufficient for selective tumor targeting.4
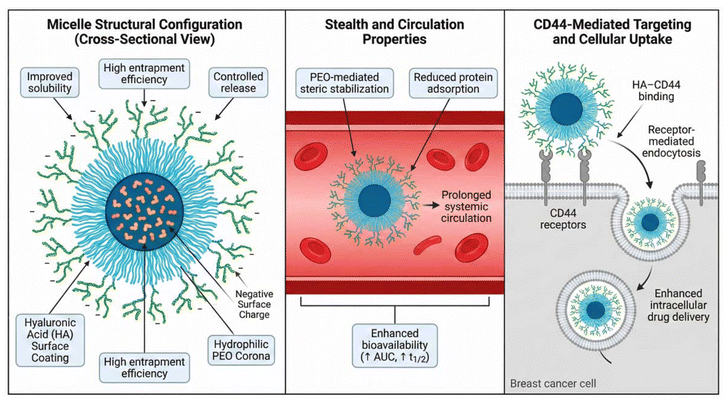
To overcome this limitation, hyaluronic acid (HA), a naturally occurring glycosaminoglycan, has been broadly introduced into nanocarriers with the purpose of increasing their biological recognition and cellular internalization via CD44 receptor-mediated endocytosis.5 CD44 is overexpressed on many breast cancer cells, and HA-based nanocarriers have demonstrated superior tumor penetration and accumulation when compared with non-targeted systems.6 As an example, HA-decorated doxorubicin nanoplatforms significantly enhanced cytotoxicity and reduced resistance in models of breast carcinoma, confirming the potential of HA-assisted targeting in nanomedicine.7 Along the same line, HA-functionalized polymeric micelles increased the therapeutic index of paclitaxel through an improved biodistribution and sustained drug release.8
The JAK/STAT pathway, especially STAT3, is considered a key oncogenic driver in the progression of breast cancer, regulating cell survival, proliferation, and metastasis. Poor prognosis and chemoresistance have been associated with sustained activation of STAT3. Targeting this pathway thus represents an effective therapeutic approach.9 Tofacitinib, a selective JAK inhibitor approved for rheumatoid arthritis, has recently received attention because of its ability to suppress STAT3 phosphorylation and its downstream oncogenic signaling.10 Several recent studies have proposed its repurposing for solid tumors, including breast and pancreatic cancers, given its dual action on the inflammatory and proliferative signaling pathways. However, poor aqueous solubility and low bioavailability of the drug require a nanocarrier-based approach to achieve efficient tumor-specific delivery.11
In the present study, hyaluronic acid (HA)–assisted Pluronic F127 micelles were rationally developed and optimized for the encapsulation and delivery of tofacitinib as a repurposed STAT3 inhibitor for breast cancer therapy. A Quality by Design (QbD)–based optimization strategy employing a Box–Behnken experimental design was implemented to systematically investigate the influence of critical formulation and process variables, including drug-to-polymer ratio, polymer concentration, and stirring temperature, on key quality attributes such as particle size and entrapment efficiency. This statistically driven approach enables predictive control of formulation performance, ensures batch-to-batch reproducibility, and aligns the development process with ICH Q8–Q11 regulatory guidelines.
The optimized micellar system was comprehensively characterized for physicochemical properties, morphology, and in vitro drug release behaviour, followed by evaluation of its cytotoxic potential against breast cancer cell lines and in vivo pharmacokinetic performance. Importantly, the selection of the oral route was guided by the clinically established oral administration of tofacitinib and the increasing reliance on chronic oral targeted therapies in breast cancer management. However, the application of tofacitinib in oncology is limited by poor aqueous solubility, low oral bioavailability, and rapid systemic clearance, which necessitate a nanocarrier-based delivery strategy.
Although HA-decorated Pluronic micellar systems have been reported previously, most studies rely on empirical formulation approaches, focus on conventional cytotoxic agents, and are primarily designed for parenteral administration. In contrast, the present work uniquely integrates a regulatory-aligned QbD framework, HA-mediated active targeting, and oral nanodelivery to enable the repurposing of tofacitinib, a clinically approved JAK inhibitor, for breast cancer therapy. By establishing a validated design space and control strategy and demonstrating enhanced oral bioavailability and sustained systemic exposure, this study advances HA–Pluronic micelles from a proof-of-concept nanocarrier toward a rational, scalable, and translational oral nanomedicine platform for targeted breast cancer treatment.
2. Materials
Hyaluronic acid (molecular weight 1.5–1.8 × 106 Da) was procured from BLD Pharma Pvt. Ltd, India. Pluronic F127 (poloxamer 407) was obtained from Himedia Laboratories Pvt. Ltd, Mumbai, India. Tofacitinib was provided as a gift sample by Dr Reddy's Laboratories, Hyderabad, India. Analytical-grade dimethyl sulfoxide (DMSO) and N,N-dimethylaminopyridine (DMAP) were purchased from Merck Specialties Pvt. Ltd, India, while carbonyldiimidazole (CDI) was obtained from SRL Pharma, Mumbai, India. All other solvents and reagents used in this study were of analytical grade and used without further purification. Milli-Q ultrapure water was used for all experimental procedures.3. Materials and methods
3.1. Preformulation studies
These studies were conducted to understand the physical and chemical properties of Tofacitinib, its interaction with HA, and the properties of Pluronic F127 excipients. Such studies form an important part of ensuring chemical stability, identifying potential interactions, and screening for suitable polymers toward micellar formulation.113.2. Design space of QbD for development of HA–Pluronic–tofacitinib micelles
A QbD framework was systematically executed to rationally develop Hyaluronic acid–Pluronic F127–Tofacitinib (HA–PF127–TF) micellar formulation. In the context of this study, QbD is a scientific approach with the purpose of ensuring the robustness of the formulation, improving process understanding, and assuring regulatory compliance by the systematic evaluation of formulation and process variables. This framework consisted of defining quality target product profile, identification of critical quality attributes, critical material attributes, and critical process parameters, structured risk analysis using failure mode and effects analysis, and determination of a statistically validated design space and control strategy.14| QbD element | Attribute/parameter | Target/range | Scientific rationale | Criticality/control strategy |
|---|---|---|---|---|
| QTPP | Intended use | Targeted delivery of tofacitinib for STAT3 inhibition in breast cancer | Enables repurposing of a clinically approved JAK inhibitor for oncology via nanocarrier-based delivery | High |
| Dosage form | Nanosized polymeric micelles (HA–Pluronic system) | Improves solubility, stability, and controlled release of poorly soluble drug | High | |
| Route of administration | Oral | Non-invasive, patient-friendly route aligned with existing clinical use of tofacitinib | Medium | |
| Stability | ≥6 months at 25 ± 2 °C | Ensures reproducibility and translational feasibility | High | |
| CQA | Particle size (nm) | 100–200 nm | Governs oral absorption, biodistribution, and tumor accumulation | High; controlled via design space |
| Polydispersity index (PDI) | <0.30 | Ensures formulation homogeneity and consistent pharmacokinetics | High; controlled by mixing and polymer concentration | |
| Zeta potential (mV) | −25 to −35 | Provides colloidal stability and reduces aggregation | High; controlled by HA concentration and pH | |
| Entrapment efficiency (%) | >80 | Ensures adequate drug incorporation and dose delivery | High; optimized via polymer ratio and temperature | |
| Drug loading (%) | 10–20 | Balances dose strength and micelle stability | Medium; controlled by drug-to-polymer ratio | |
| In vitro drug release | Sustained, up to 48 h | Maintains prolonged systemic exposure and reduces dosing frequency | High; governed by core–shell architecture | |
| CMA | Drug-to-polymer ratio | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10–1 10–1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 (w/w) 20 (w/w) |
Controls micellization, encapsulation efficiency, and size | High impact on CQAs |
| PF127 concentration (% w/v) | 0.5–1.5 | Influences micelle stability and size distribution | Moderate to high | |
| HA concentration (% w/v) | 0.1 | Enables CD44 targeting and surface charge modulation | Moderate | |
| CPP | Stirring temperature (°C) | 30–50 | Affects polymer self-assembly and drug entrapment | High; selected via FMEA and BBD |
| Stirring speed (rpm) | Fixed | Ensures uniform film formation and hydration | Low | |
| Sonication time | Fixed (3 min) | Reduces particle size and polydispersity | Low | |
| Hydration medium & pH | PBS, pH 6.8 | Mimics physiological conditions for oral delivery | Moderate |
Results of the FMEA (SI Table S1) identified the three most critical parameters that can affect micellar size and encapsulation efficiency: drug-to-polymer ratio, Pluronic F127 concentration, and stirring temperature. Thus, these variables were selected for experimental modeling via a Box–Behnken design based on this analysis.17
In the present study, an RPN threshold of ≥150 was selected to classify formulation and process variables as high-risk. This cut-off was chosen based on established pharmaceutical QbD practices, where parameters exhibiting a combination of high severity, high probability of occurrence, and low detectability are considered most likely to impact critical quality attributes. The selected threshold ensured focused prioritization of variables with the greatest potential to influence micellar size distribution and entrapment efficiency, while avoiding unnecessary inclusion of low-risk parameters that could compromise model robustness and interpretability. Variables exceeding this threshold were therefore subjected to further statistical optimization using the Box–Behnken design.
Relationships among the variables were established by fitting a polynomial regression model, and the adequacy of the fitted model was then checked using ANOVA. Contour plots and 3D response surfaces were used to visualize variable interactions and predict optimum conditions. Design space was defined as a region that would provide robust CQAs within the target range. The desirability function approach was used to choose the optimal formulation based on the criteria of minimum particle size and PDI, and maximum entrapment efficiency.18
Thus, the established QbD framework provided a structured, risk-based, and statistically driven methodology for the development of HA–Pluronic micelles of TF, ensuring product reproducibility with consistent performance.
Beyond laboratory-scale validation, the established control strategy provides a framework for scalable and reproducible manufacturing of HA–Pluronic–Tofacitinib micelles. The critical process parameters identified through QbD analysis—namely drug-to-polymer ratio, Pluronic F127 concentration, and stirring temperature—are defined within a validated design space that can be directly translated to larger-scale processing. During scale-up, these parameters may be controlled through predefined operating ranges, real-time monitoring of process temperature and mixing conditions, and in-process particle size assessment as part of process analytical technology (PAT) tools. Adherence to the defined design space allows for flexibility in scale without compromising critical quality attributes, in accordance with ICH Q8 and Q10 guidelines, thereby ensuring consistent product quality and regulatory compliance.
3.3 Formulation and optimization of tofacitinib-loaded HA–Pluronic micelles
The HA–PF127–TF micelles were then prepared by the solvent evaporation and thin-film hydration technique, followed by QbD-based optimization, as established in the preceding section. This approach allowed the particle size, entrapment efficiency, and homogeneity of the micelles to be precisely controlled via a systematic variation of key formulation parameters.19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 v/v ratio to obtain a clear organic solution. The organic solvent mixture was removed by evaporation under reduced pressure using a Buchi R-300 rotary evaporator at 40 °C and 120 rpm until a uniform thin film formed on the wall of the flask. The film was further dried under vacuum for 2 hours to remove residual solvent. The dried film was subsequently hydrated with 10 mL of phosphate-buffered saline, PBS, pH 6.8, at 37 °C, followed by gentle stirring at 300 rpm for 30 min to obtain self-assembled Pluronic micelles encapsulating TF.20
1 v/v ratio to obtain a clear organic solution. The organic solvent mixture was removed by evaporation under reduced pressure using a Buchi R-300 rotary evaporator at 40 °C and 120 rpm until a uniform thin film formed on the wall of the flask. The film was further dried under vacuum for 2 hours to remove residual solvent. The dried film was subsequently hydrated with 10 mL of phosphate-buffered saline, PBS, pH 6.8, at 37 °C, followed by gentle stirring at 300 rpm for 30 min to obtain self-assembled Pluronic micelles encapsulating TF.20
 | ||
| Fig. 1 Schematic illustration of the preparation of Hyaluronic Acid (HA)–modified Pluronic F127 micelles loaded with Tofacitinib. | ||
To investigate the interactive effects between the independent factors and the responses, seventeen experimental runs were generated. Table 2 illustrates the experimental matrix. The responses obtained were fitted to a second order polynomial equation, and adequacy of the model was checked through ANOVA.
| Category | Parameter | Code | Low level (−1) | Medium level (0) | High level (+1) | Experimental range/units | Optimization goal | Scientific justification |
|---|---|---|---|---|---|---|---|---|
| Independent variable |
A: drug-to-polymer ratio (Tofacitinib![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PF127) PF127) |
A | 01![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10 |
01![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 15 |
01![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 20 |
0.1 to 0.3 (w/w) equivalent | — | Higher polymer proportion improves micellization and EE up to an optimal limit (beyond 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 viscosity increases). 20 viscosity increases). |
| B: PF127 concentration (% w/v) | B | 0.5 | 1 | 1.5 | 0.5 to 1.5% | — | Determines micelle density and stability; excess polymer causes high viscosity and potential aggregation. | |
| C: stirring temperature (°C) | C | 30 | 40 | 50 | 30 to 50 °C | — | Temperature modulates micellization; optimal assembly occurs near 40 °C, above which polymer degradation risk increases. | |
| Dependent response | Y 1: particle size (nm) | Y 1 | — | — | — | — | Minimize | Nanometer-scale micelles (<200 nm) ensure enhanced permeation and retention (EPR) effect in tumors. |
| Y 2: polydispersity index (PDI) | Y 2 | — | — | — | — | Minimize | Reflects micelle uniformity and formulation stability. | |
| Y 3: entrapment efficiency (%) | Y 3 | — | — | — | — | Maximize | Ensures sufficient Tofacitinib incorporation for therapeutic efficacy. | |
| Fixed parameter | Hydration volume | — | — | — | — | 10 mL | — | Maintains consistent micelle concentration and reproducibility. |
| Stirring speed | — | — | — | — | 400 rpm | — | Ensures uniform film formation during solvent evaporation. | |
| Stirring duration | — | — | — | — | 30 min | — | Provides adequate polymer and drug dispersion. | |
| Sonication time | — | — | — | — | 3 min (30% amplitude) | — | Reduces micelle size while avoiding rupture. | |
| Hydration medium | — | — | — | — | PBS (pH 6.8) | — | Mimics physiological conditions for oral delivery. | |
| HA concentration | — | — | — | — | 0.1% w/v | — | Enables effective surface association for CD44 targeting. | |
| Storage temperature | — | — | — | — | 4 °C | — | Preserves micelle integrity prior to characterization. |
The response surface and contour plots were analyzed to identify the region of optimal formulation. The desirability function approach was employed to find the optimum conditions which would provide minimum particle size and PDI, along with maximum entrapment efficiency. Under such predicted conditions, the optimized formulation was prepared and experimental results were compared with the predicted values to verify model predictability.21
3.4. Characterization of optimized micelles
The optimized HA–PF127–TF micelles were characterized comprehensively using physicochemical and morphological techniques to confirm successful formulation, stability, and suitability for biological evaluation. All analyses were performed in triplicate under controlled laboratory conditions unless stated otherwise.22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 30 min at 4 °C using a Remi C-24 BL centrifuge in order to separate the unentrapped TF. Then, the supernatant collected, sutaibly diluted and analyzed for free drug content by HPLC under sutaible conditions. Total and entrapped drug content were calculated, and EE% and DL% were determined using standard mathematical relationships. All the experiments were performed in triplicate to ensure accuracy.
000 rpm for 30 min at 4 °C using a Remi C-24 BL centrifuge in order to separate the unentrapped TF. Then, the supernatant collected, sutaibly diluted and analyzed for free drug content by HPLC under sutaible conditions. Total and entrapped drug content were calculated, and EE% and DL% were determined using standard mathematical relationships. All the experiments were performed in triplicate to ensure accuracy.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000–14
000–14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 Da) containing 2 mL of micellar dispersion (equivalent to 2 mg of Tofacitinib) was inserted into 100 mL of phosphate buffer (pH 7.4), maintained at 37 ± 0.5 °C under continuous magnetic stirring at 100 rpm. At pre-calculated time intervals, 2 mL samples were withdrawn and replaced with an equivalent amount of fresh buffer to maintain sink conditions. The samples were assayed by HPLC at 287 nm. Zero-order, first-order, Higuchi, and Korsmeyer-Peppas equations were employed for the release kinetics modelling, and regression analysis was utilized to identify the most appropriate mechanism that best describes the release.25
000 Da) containing 2 mL of micellar dispersion (equivalent to 2 mg of Tofacitinib) was inserted into 100 mL of phosphate buffer (pH 7.4), maintained at 37 ± 0.5 °C under continuous magnetic stirring at 100 rpm. At pre-calculated time intervals, 2 mL samples were withdrawn and replaced with an equivalent amount of fresh buffer to maintain sink conditions. The samples were assayed by HPLC at 287 nm. Zero-order, first-order, Higuchi, and Korsmeyer-Peppas equations were employed for the release kinetics modelling, and regression analysis was utilized to identify the most appropriate mechanism that best describes the release.25
3.5. In vitro cytotoxicity (MTT assay)
The MTT colorimetric assay was conducted to assess the cytotoxic potential of the optimized HA–PF127–TF micelles against the MCF-7 human breast cancer cell line. In this experiment, the antiproliferative activity of the optimized micellar formulation was compared with that of free Tofacitinib and non-HA-modified micelles. All procedures were performed under aseptic conditions, and each experiment was carried out in triplicate to ensure reliability.273.6. In Vivo pharmacokinetic study
The optimized HA–PF127–TF micelles were subjected to pharmacokinetic evaluation with the view of assessing the impact of micellar encapsulation and hyaluronic acid modification on its systemic bioavailability after oral administration. The study was conducted in accordance with the guidelines for the care and use of laboratory animals and was approved by the Institutional Animal Ethics Committee (IAEC), Department of Pharmaceutical Sciences, Rashtrasant Tukadoji Maharaj Nagpur University, Nagpur, Maharashtra, India (Approval No. IAEC/UDPS/2024/02/23).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 10 min at 4 °C in a Remi C-24 BL refrigerated centrifuge. The separated plasma layer was collected and stored at −20 °C until further analysis.28
000 rpm for 10 min at 4 °C in a Remi C-24 BL refrigerated centrifuge. The separated plasma layer was collected and stored at −20 °C until further analysis.28
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 10 minutes. The resultant clear supernatant was separated, filtered through a 0.22 μm syringe filter, and transferred into HPLC vials for quantification.29
000 rpm for 10 minutes. The resultant clear supernatant was separated, filtered through a 0.22 μm syringe filter, and transferred into HPLC vials for quantification.29
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40 v/v), which was pumped through the column at a flow rate of 1.0 mL min−1. The analytes were then detected at a wavelength of 287 nm using an injection volume of 20 μL. The developed analytical method was further validated for selectivity, accuracy, linearity, precision, and recovery according to ICH Q2 (R2) guidelines.30
40 v/v), which was pumped through the column at a flow rate of 1.0 mL min−1. The analytes were then detected at a wavelength of 287 nm using an injection volume of 20 μL. The developed analytical method was further validated for selectivity, accuracy, linearity, precision, and recovery according to ICH Q2 (R2) guidelines.30
3.7. Histopathological examination
Histopathological examination of tissues was carried out to check for any signs of tissue change or toxic manifestation upon oral administration of the optimized HA–Pluronic–Tofacitinib micelles compared to plain drug and non-HA micellar formulation. The animal study was performed following the CPCSEA guidelines, which deal with the purpose of controlling and supervising the conduct of experiments on animals for ensuring animal welfare and ethical research practices.3.8. Statistical analysis
All quantitative data are presented as mean ± standard deviation (SD). Statistical analysis for in vitro cytotoxicity (MTT assay) was performed using one-way analysis of variance (ANOVA) followed by Tukey's multiple comparison post hoc test to compare differences among treatment groups. Pharmacokinetic parameters were calculated using non-compartmental analysis with Phoenix WinNonlin® software (Version 8.3, Certara, USA), and statistical comparisons between formulations were conducted using unpaired Student's t-test where applicable. GraphPad Prism® software (Version 9.0, GraphPad Software Inc., USA) was used for data plotting and statistical evaluation. Differences were considered statistically significant at p < 0.05.4 Results and discussion
4.1. Preformulation and compatibility studies
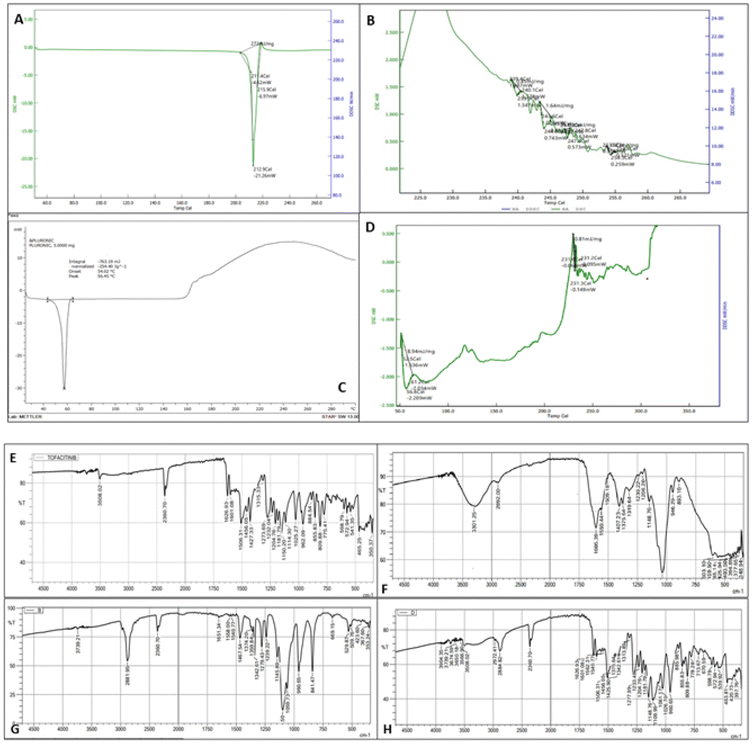
Preformulation studies were conducted to understand the physicochemical behavior of Tofacitinib and its compatibility with selected excipients, hyaluronic acid and Pluronic F127, for the micellar formulation.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching, and 1556 cm−1 due to aromatic C–N stretching (Fig. 3E). The spectra of Pluronic F127 and HA showed a broad band around 3400 cm−1 and 1100–1150 cm−1 due to O–H stretching and C–O–C ether linkage, respectively (Fig. 3F and G). All the major peaks were present in the physical mixture without any appearance of new absorption bands or disappearance of existing absorption bands, proving that no chemical interaction took place. In the HA–PF127–TF micelles, small shifts of the N–H and C
O stretching, and 1556 cm−1 due to aromatic C–N stretching (Fig. 3E). The spectra of Pluronic F127 and HA showed a broad band around 3400 cm−1 and 1100–1150 cm−1 due to O–H stretching and C–O–C ether linkage, respectively (Fig. 3F and G). All the major peaks were present in the physical mixture without any appearance of new absorption bands or disappearance of existing absorption bands, proving that no chemical interaction took place. In the HA–PF127–TF micelles, small shifts of the N–H and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bands were evident (Fig. 3H).14
O bands were evident (Fig. 3H).14
4.2. Formulation and optimization of HA–Pluronic–Tofacitinib micelles (QbD approach)
The formulation and optimization of HA–Pluronic–Tofacitinib micelles were performed using a systematic QbD framework based on BBD. The aim was to investigate the impact of formulation and process variables on critical micellar attributes. Three independent variables selected were drug-to-polymer ratio (A), PF127 concentration (B), and stirring temperature (C), while dependent responses were particle size (Y1), polydispersity index (Y2), and entrapment efficiency (Y3).The findings of the preformulation studies played a pivotal role in defining the critical quality attributes (CQAs) of the HA–Pluronic–Tofacitinib micellar system. DSC analysis confirmed the crystalline nature of pure tofacitinib and its conversion to an amorphous state upon micellar encapsulation, highlighting the importance of entrapment efficiency and physical stability as key CQAs. Similarly, FTIR studies demonstrated the absence of chemical incompatibility between tofacitinib and formulation excipients, supporting a formulation strategy based on physical encapsulation rather than chemical interaction. These observations underscored the necessity of controlling particle size and polydispersity index to ensure uniform drug distribution, colloidal stability, and reproducible release behavior. Consequently, particle size, PDI, entrapment efficiency, and drug release profile were selected as CQAs within the QbD framework, directly linking preformulation insights with formulation optimization and quality risk management.
| Response | Source | Sum of squares | df | Mean square | F-value | p-Value | Significance |
|---|---|---|---|---|---|---|---|
| Particle size | Model | 2263.71 | 9 | 251.52 | 10.12 | 0.0029 | Significant |
| A: drug-to-polymer ratio | 338 | 1 | 338 | 13.59 | 0.0078 | Significant | |
| B: PF127 concentration | 120.13 | 1 | 120.13 | 4.83 | 0.0639 | Moderate | |
| C: stirring temperature | 1326.13 | 1 | 1326.13 | 53.33 | 0.00016 | Highly significant | |
| Residual | Residual | 174.05 | 7 | 24.86 | — | — | — |
| PDI | Model | 0.00401 | 9 | 0.00045 | 9.18 | 0.0039 | Significant |
| A: drug-to-polymer ratio | 0.00058 | 1 | 0.00058 | 11.91 | 0.0107 | Significant | |
| B: PF127 concentration | 0.00048 | 1 | 0.00048 | 9.9 | 0.0162 | Significant | |
| C: stirring temperature | 0.00205 | 1 | 0.00205 | 42.2 | 0.00034 | Highly significant | |
| Residual | Residual | 0.00034 | 7 | 5.00 × 10−5 | — | — | — |
| Entrapment efficiency | Model | 240.81 | 9 | 26.76 | 11.24 | 0.0021 | Significant |
| A: drug-to-polymer ratio | 38.72 | 1 | 38.72 | 16.26 | 0.00498 | Significant | |
| B: PF127 concentration | 18.3 | 1 | 18.3 | 7.69 | 0.0276 | Significant | |
| C: stirring temperature | 117.81 | 1 | 117.81 | 49.47 | 0.0002 | Highly significant | |
| Residual | Residual | 16.67 | 7 | 2.38 | — | — | — |
The regression coefficients showed that factor C (stirring temperature) had the most pronounced effect throughout all the responses, followed by factor A (drug-to-polymer ratio) and factor B (PF127 concentration).
The Box–Behnken experimental design matrix, detailing the seventeen formulations generated and their corresponding observed values for particle size, PDI, and entrapment efficiency, is presented in Table 4.
| Run | Factors | Responses | ||||
|---|---|---|---|---|---|---|
A: drug-to-polymer ratio (tofacitinib![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PF127) (%w/w) equivalent PF127) (%w/w) equivalent |
B: PF127 concentration (% w/v) | C: stirring temperature (°C) | Particle size (nm) | PDI | Entrapment efficiency (%) | |
| 1 | 0.2 | 1.5 | 50 | 155 | 0.218 | 91.4 |
| 2 | 0.1 | 1 | 30 | 182 | 0.255 | 83.2 |
| 3 | 0.1 | 0.5 | 40 | 190 | 0.265 | 81 |
| 4 | 0.1 | 1.5 | 40 | 165 | 0.23 | 89.6 |
| 5 | 0.2 | 1 | 40 | 160 | 0.225 | 90.8 |
| 6 | 0.3 | 0.5 | 40 | 158 | 0.235 | 92.3 |
| 7 | 0.2 | 0.5 | 50 | 150 | 0.21 | 93.1 |
| 8 | 0.1 | 1 | 50 | 162 | 0.226 | 90 |
| 9 | 0.2 | 1 | 40 | 159 | 0.223 | 91 |
| 10 | 0.3 | 1 | 30 | 180 | 0.24 | 84.1 |
| 11 | 0.2 | 1 | 40 | 160 | 0.222 | 90.6 |
| 12 | 0.2 | 1 | 40 | 161 | 0.22 | 90.9 |
| 13 | 0.2 | 1.5 | 30 | 175 | 0.242 | 87.5 |
| 14 | 0.3 | 1.5 | 40 | 157 | 0.218 | 92 |
| 15 | 0.3 | 1 | 50 | 152 | 0.215 | 93 |
| 16 | 0.2 | 0.5 | 30 | 185 | 0.26 | 82 |
| 17 | 0.2 | 1 | 40 | 156 | 0.217 | 92.4 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15, while higher polymer content favored micellization due to better packing of the hydrophobic core. However, higher polymer resulted in slight aggregation that increased the size. On the other hand, with the increase in the temperature of stirring in C, the particle size decreased drastically due to high kinetic energy, which favored effective self-assembly as well as evaporation of the solvent to form micelles that were more compact. Fig. 4A presents the 2D contour plots and 3D response surface plots illustrating the interactive effects of drug-to-polymer ratio, PF127 concentration, and stirring temperature on the particle size of the micelles.
15, while higher polymer content favored micellization due to better packing of the hydrophobic core. However, higher polymer resulted in slight aggregation that increased the size. On the other hand, with the increase in the temperature of stirring in C, the particle size decreased drastically due to high kinetic energy, which favored effective self-assembly as well as evaporation of the solvent to form micelles that were more compact. Fig. 4A presents the 2D contour plots and 3D response surface plots illustrating the interactive effects of drug-to-polymer ratio, PF127 concentration, and stirring temperature on the particle size of the micelles.
PDI values of 0.21–0.26 indicated that all the formulations have a narrow size distribution. High polymer concentration with a moderate stirring temperature lowered PDI, as this improved the mobility of polymer chains. This would ensure uniform micellar nucleation. At very high concentrations, the polymers showed higher PDI values due to enhanced viscosity that interfered with proper dispersion. Fig. 4B illustrates the 2D contour plots and 3D response surface plots that depict the interaction effects of drug-to-polymer ratio, PF127 concentration, and stirring temperature on the Polydispersity Index (PDI).
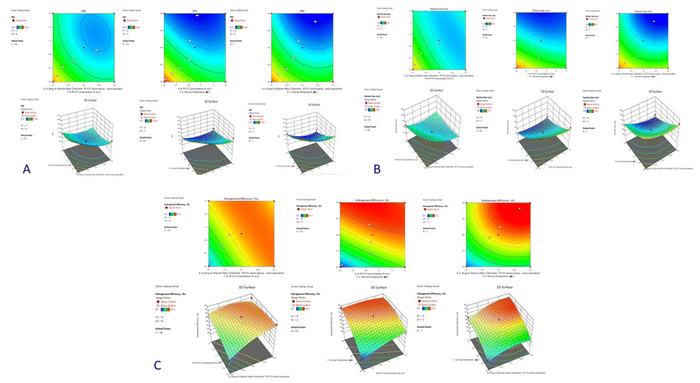
The entrapment efficiency was within the range of 81–93%, and with increased drug-to-polymer ratio and higher concentration of Pluronic, there was an enhancement in EE%. This may be because of increased availability of hydrophobic domains inside the micellar core which efficiently solubilized Tofacitinib. In addition, a mild positive effect of temperature was observed probably due to improved flexibility of the polymer and swelling of micelles at elevated temperatures, thus allowing deeper incorporation of the drug.34Fig. 4C displays the 2D contour plots and 3D response surface plots illustrating the combined effects of drug-to-polymer ratio, surfactant concentration, and stirring temperature on the entrapment efficiency.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 drug to polymer (w/w) ratio, PF127 concentration of 1.0% w/v, and a stirring temperature of 40 °C, the responses for a particle size of 159.2 nm, a PDI of 0.221, and an EE% of 91.14% were predicted. Experimental validation of these showed less than a 2% prediction error, thus confirming the reliability and robustness of the established model.
15 drug to polymer (w/w) ratio, PF127 concentration of 1.0% w/v, and a stirring temperature of 40 °C, the responses for a particle size of 159.2 nm, a PDI of 0.221, and an EE% of 91.14% were predicted. Experimental validation of these showed less than a 2% prediction error, thus confirming the reliability and robustness of the established model.
In this regard, QbD-based optimization of the nanocarrier formulation showed that a well-conducted experimental design is indeed able to provide straightforward insight into the relationships among parameters and responses in such complex nanosystems. Stirring temperature (C) represents the most influential variable, which means that thermodynamic control during micellization is the predominant factor affecting particle size and entrapment behaviour. The negative correlation of polymer concentration with both particle size and PDI suggests that micellar compaction is virtually driven by the amphiphilic re-organization of Pluronic, while HA adsorption has an ancillary role in colloidal stability. Integration of statistical modelling with mechanistic reasoning established reproducibility, reliability, and robustness as three important features for biological translation.15
4.3 Characterization of optimized micelles
Consequently, a QbD design-derived HA–Pluronic–Tofacitinib micellar formulation was further characterized with respect to physicochemical properties, structural morphology, thermal behaviour, and in vitro release performance. Characterization results showed the development of nanosized, stable, and uniform micelles suitable for further biological evaluation.The resultant particle size of less than 200 nm supported the suitability of the formulation for passive tumor targeting via the enhanced permeability and retention effect. The narrow PDI confirmed homogeneity in micellar formation, one of the critical quality attributes that are important for reproducibility and stability.36
In addition to their targeting functionality, the HA–Pluronic nanomicelles are expected to exhibit stealth-like properties. Pluronic F127 forms a hydrophilic poly(ethylene oxide) corona around the micellar core, which is widely reported to reduce nonspecific protein adsorption and reticuloendothelial system uptake. The presence of hyaluronic acid further contributes to surface hydration and a moderately negative zeta potential, minimizing nonspecific electrostatic interactions with plasma proteins. Moreover, the optimized micelles possessed a nanoscale size below 200 nm and demonstrated prolonged systemic circulation with increased half-life and AUC, indirectly supporting reduced clearance and stealth behavior. Collectively, these attributes suggest that the developed HA–Pluronic micelles combine passive stealth characteristics with active CD44-mediated targeting, contributing to enhanced in vivo performance.
The extensive physicochemical characterization conducted proved the successful fabrication of HA–PF127–Tofacitinib micelles with optimum properties for oral and targeted drug delivery. The nanosized, monodisperse micelles proved to be colloidally stable, with high drug loading, and FTIR and DSC data confirmed compatibility and amorphous conversion. Sustained release behaviour showed effective encapsulation with diffusion-controlled release, matching the very objectives of enhancing solubility and prolonging bioavailability. The resultant properties, in particular, of size (∼160 nm) and a negative surface charge are favourable for passive accumulation at tumor sites via the EPR effect. In addition, HA provides potential active targeting to CD44-overexpressing breast cancer cells. All the findings together justify the potential of the formulation as a repurposed nano-delivery platform for Tofacitinib in breast cancer therapy.38
4.4. In vitro cytotoxicity (MTT assay)
The MTT assay was performed with the optimized HA–Pluronic–Tofacitinib micelles against MCF-7 human breast cancer cells to investigate cytotoxic potential, as compared to free Tofacitinib and non-HA–modified Pluronic micelles. This study has been performed in order to determine the effect of nanoencapsulation and HA modification on the antiproliferative efficacy of Tofacitinib.The active targeting of CD44 receptors overexpressed on MCF-7 cells by HA decoration on micelles explains its enhanced cytotoxicity. HA-receptor interactions thus facilitate receptor-mediated endocytosis, which increases the internalization of drug-loaded micelles. Furthermore, improved solubilization and sustained release from the micellar core ensured higher intracellular retention of the drug and its prolonged exposure to Tofacitinib.
The reduction in IC50 observed for HA–Pluronic–Tofacitinib micelles was statistically significant compared with free tofacitinib and non-HA micelles (p < 0.05).
These findings underscore the importance of nanocarrier design for improving drug bioefficacy, as reflected by the substantial improvement in cytotoxic response following encapsulation in HA–Pluronic micelles. Besides facilitating selective uptake through CD44-mediated endocytosis, HA modification also promoted better solubility and intracellular retention of Tofacitinib. In this regard, similar observations have been made while delivering hydrophobic anticancer agents such as paclitaxel and curcumin using HA-functionalized micelles. A three-fold decrease in IC50 compared to free drug suggests that micellar encapsulation may potentially overcome the solubility and permeability barriers to ensure targeted cytotoxicity in models of breast cancer.
Collectively, these in vitro cytotoxicity results establish the superiority of HA–Pluronic–Tofacitinib micelles over free Tofacitinib and non-HA micelles in anticancer potential, thus validating the strategy of drug repurposing through targeted nanodelivery.39
4.5. In vivo pharmacokinetic evaluation
Finally, the optimized HA–Pluronic–Tofacitinib micelles were subjected to pharmacokinetic profiling in Wistar rats after oral administration, compared with free Tofacitinib and non-HA–modified Pluronic micelles, to investigate the effect of micellar encapsulation and HA surface modification on the systemic bioavailability and longevity of circulation.| Parameter | Free tofacitinib | Pluronic micelles | HA–Pluronic micelles |
|---|---|---|---|
| C max (μg mL−1) | 1.82 ± 0.14 | 3.46 ± 0.22 | 5.12 ± 0.28 |
| T max (h) | 1.0 ± 0.2 | 2.0 ± 0.3 | 4.0 ± 0.4 |
| AUC0−∞ (μg h mL−1) | 22.34 ± 2.85 | 54.71 ± 3.96 | 96.82 ± 5.31 |
| t 1/2 (h) | 3.2 ± 0.4 | 6.5 ± 0.6 | 8.9 ± 0.7 |
| Kel (h−1) | 0.216 ± 0.02 | 0.106 ± 0.01 | 0.078 ± 0.01 |
| MRT (h) | 4.1 ± 0.3 | 7.9 ± 0.5 | 10.7 ± 0.8 |
| Relative bioavailability (%) | 100 | 245 | 434 |
The pharmacokinetic results indicated that both the extent and duration of systemic exposure for the HA–Pluronic–Tofacitinib micelles were significantly improved compared with the free drug. Enhanced oral bioavailability is a result of multiple synergistic mechanisms: (i) increased solubilization of Tofacitinib within the hydrophobic micellar core, (ii) protection from enzymatic degradation due to the HA coating, (iii) mucoadhesive interaction of HA with the intestinal epithelium, thus facilitating prolonged absorption, and (iv) possible lymphatic transport, thereby reducing hepatic first-pass metabolism.
Such sustained-release behavior was further confirmed by the delayed Tmax and extended elimination half-life observed in vitro, which agreed with the controlled diffusion mechanism. The higher pharmacokinetic performances of HA-coated micelles reflect the trends described for HA-functionalized systems of drugs such as doxorubicin and curcumin, which all showed enhanced systemic retention and tumor accumulation.
Overall, these results substantiated the success of the QbD-optimized HA–Pluronic micellar system in enhancing the oral bioavailability and sustaining the therapeutic plasma concentration of Tofacitinib, hence advocating for its repurposing potential in the treatment of breast cancer.40
Statistical comparison of pharmacokinetic parameters revealed a significant increase in AUC and half-life for HA–Pluronic–Tofacitinib micelles compared to free drug (p < 0.05).
4.6. Histopathological examination
Histopathological examinations were carried out to investigate the safety and toxicity at the tissue level of the HA–Pluronic–Tofacitinib micelles compared with the free drug and blank micellar formulations. Organs such as the liver, kidney, heart, and lungs were collected and examined microscopically after 14 days of repeated oral administration for potential systemic toxicity or histoarchitectural changes. To verify the systemic safety of the optimized formulation, histological examination of the lungs, liver, and kidneys was performed, as shown in Fig. 6.Histological sections of the liver in the control and micellar-treated groups revealed normal hepatic lobular architecture with well-defined central veins and hepatic cords. Hepatocytes were polygonal with centrally located nuclei without evidence of necrosis or fatty degeneration. Histological sections of liver specimens from the free Tofacitinib suspension-treated animals, however, exhibited mild hepatocellular vacuolation together with slight infiltration of inflammatory cells, pointing to transient hepatic stress and probably related to drug accumulation and low solubility.
Renal sections from the HA–Pluronic micelle group revealed intact glomeruli and well-preserved tubular epithelium without necrosis and interstitial inflammation. However, the free drug group evinced mild tubular dilatation with epithelial desquamation, indicating irritation of the nephrons. All these findings confirmed the renal safety of this micellar formulation and also the possibility of reducing its nephrotoxic potential by controlled drug release and enhanced bioavailability.
The lung sections showed normal alveolar structures with intact septa and no inflammatory infiltrates. There was also no pulmonary edema or congestion in the micellar-treated group, further confirming the systemic biocompatibility of the formulation.
Histopathological evaluation was performed to assess the systemic safety of the optimized HA–Pluronic–Tofacitinib micelles following oral administration. Microscopic examination of hematoxylin and eosin–stained sections of liver, kidney, lung, and heart from micelle-treated animals revealed well-preserved tissue architecture comparable to the control group. Hepatic sections exhibited intact lobular organization with clearly defined central veins and hepatocyte cords, without evidence of necrosis, fatty degeneration, or inflammatory infiltration.
Renal tissues displayed normal glomerular morphology and intact tubular epithelium, with no signs of tubular dilation, epithelial desquamation, or interstitial inflammation. Pulmonary sections showed preserved alveolar architecture with thin septa and absence of edema or inflammatory cell infiltration, while cardiac tissues exhibited normal myocardial fiber arrangement without degeneration or necrosis. In contrast, animals treated with free tofacitinib showed mild, reversible histological alterations, including hepatocellular vacuolation and slight tubular changes, indicative of transient tissue stress.
Overall, these findings demonstrate that encapsulation of tofacitinib within HA–Pluronic micelles mitigates organ-level toxicity and supports the biocompatibility and systemic safety of the optimized oral nanocarrier system.41
4.7. Stability studies
Therefore, the optimized HA–Pluronic–Tofacitinib micelles were subjected to stability studies under accelerated and long-term conditions to determine their physicochemical integrity as well as storage suitability, according to the ICH Q1A(R2) guidelines.• Long-term stability: 4 ± 2 °C (refrigerated condition)
• Accelerated stability: 40 ± 2 °C/75 ± 5% RH (environmental chamber)
During this time, samples were periodically withdrawn at 0, 1, 2, and 3 months and analyzed for particle size, PDI, zeta potential, drug content, and EE%. The physical stability of the NPs was also inspected visually for color changes, precipitation, or aggregation.
Under accelerated conditions, particle size increased more 159.2 → 175.4 nm with a moderate increase in PDI 0.221 → 0.287 and slight reduction in EE 91.14 → 83.26% and zeta potential −24.6 → −20.9 mV. Yet, no significant aggregation and color change were observed, which revealed that at least for a period of 3 months the micellar formulation was stable under both conditions (Table 6).
| Parameter | Initial (0 month) | 1 month | 2 months | 3 months |
|---|---|---|---|---|
| Condition 4 °C | ||||
| Particle size (nm) | 159.2 ± 3.8 | 160.3 ± 3.9 | 161.8 ± 4.0 | 162.6 ± 4.1 |
| PDI | 0.221 ± 0.015 | 0.224 ± 0.018 | 0.230 ± 0.019 | 0.238 ± 0.020 |
| Zeta potential (mV) | −24.6 ± 1.2 | −24.1 ± 1.3 | −23.4 ± 1.4 | −22.8 ± 1.5 |
| Entrapment efficiency (%) | 91.14 ± 1.8 | 90.47 ± 1.9 | 89.63 ± 2.0 | 88.72 ± 2.1 |
| Condition 40 °C/75% RH | ||||
| Particle size (nm) | 159.2 ± 3.8 | 166.5 ± 4.0 | 170.8 ± 4.3 | 175.4 ± 4.6 |
| PDI | 0.221 ± 0.015 | 0.246 ± 0.020 | 0.267 ± 0.022 | 0.287 ± 0.025 |
| Zeta potential (mV) | −24.6 ± 1.2 | −23.6 ± 1.4 | −22.1 ± 1.6 | −20.9 ± 1.8 |
| Entrapment efficiency (%) | 91.14 ± 1.8 | 88.56 ± 2.0 | 85.93 ± 2.1 | 83.26 ± 2.4 |
The stability profile indeed showed that HA–Pluronic–Tofacitinib micelles exhibited excellent stability in storage at refrigerated conditions, and acceptable quality attributes were maintained under accelerated conditions. Minor increases in particle size and PDI would be expected as a function of polymer relaxation and hydration with time. Slight losses in zeta potential and EE could also arise from surface rearrangement and limited diffusion of drug through the hydrated polymeric shell.
These observations are in agreement with the previously reported stability data of Pluronic-based micellar systems, where storage at 4 °C effectively preserved the micellar integrity and drug content therein. The results confirm that the developed micelles are physically and chemically stable for extended storage, supporting their translational feasibility as a repurposed nanocarrier for Tofacitinib in breast cancer therapy.42
5. Conclusion
The present study demonstrates rational development, optimization, and evaluation of HA–Pluronic–Tofacitinib micelles as a repurposed nanocarrier system for targeted therapy against breast cancer. By using the Quality by Design approach, critical formulation and process parameters were identified and optimized in a systematic way for the formulation of nanomicelles with desirable attributes, uniform size (∼159 nm), narrow PDI (0.22), high entrapment efficiency (∼91%), and strong colloidal stability (−24 mV).A wide range of characterizations confirmed the successful formation of micelles, amorphous dispersion of drugs, and hydrogen bond-based stabilization within the hydrophobic core of Pluronic. The HA coating conferred additional surface stability and active targeting capability via uptake mediated by CD44 receptors. In vitro release studies evidenced a biphasic, diffusion-controlled profile, thus warranting prolonged drug availability.
The in vitro biological evaluation demonstrated a threefold increase in cytotoxicity against MCF-7 breast cancer cells compared to the free drug, confirming improved intracellular uptake and retention. A pharmacokinetic study confirmed 4.3-fold increased bioavailability but also extended half-life and delayed Tmax, proving the potential of the formulation for prolonging systemic circulation and improving therapeutic exposure. Besides that, histopathological examination confirmed its biocompatibility and safety to organs, establishing that HA–Pluronic micelles may mitigate systemic toxicity associated with Tofacitinib.
However, from a translational perspective, this study also offers drug repurposing through nanocarrier-based delivery as a means to extend the range of applications for clinically approved molecules, such as Tofacitinib. The dual-functional approach for enhanced solubility and active targeting positions HA–Pluronic micelles as a promising oral nanoplatform in the management of breast cancer.
It should be noted that the pharmacokinetic study was conducted with a limited sample size (n = 3), which represents a limitation of the present work. Although the observed improvements in oral bioavailability and systemic exposure were substantial, future studies involving larger animal cohorts are required to confirm these findings and support further translational development.
Author contributions
Suchita Waghmare: writing original draft; Rohini Palekar: editing original manuscript; Pratiksha Bramhe: diagram preparation; Nilesh Rarokar: results interpretation; Pramod Khedekar: supervision.Conflicts of interest
There is no conflict to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). The supplementary file consists of information regarding design expert parameters for optimization of micelles. Supplementary information is available. See DOI: https://doi.org/10.1039/d5pm00391a.Acknowledgements
I would like to thank Department of Pharmaceutical Sciences, Rashtrasant Tukadoji Maharaj Nagpur University, Nagpur for providing research facilities for my research work. This work was funded by Government of Maharashtra under the scheme of Mahatma Jyotiba Fuley Research Fellowship-2022 for Other Backword Class, VJ-NT and Special Backword Class [Letter No. MAHAJYOTI/2022/Ph.D. Fellow/1002 (1038) dated 13/12/2022].References
- J. Kim, A. Harper, V. McCormack, H. Sung, N. Houssami and E. Morgan, et al., Global patterns and trends in breast cancer incidence and mortality across 185 countries, Nat. Med., 2025, 31(4), 1154–1162 CrossRef CAS PubMed . Available from: https://www.nature.com/articles/s41591-025-03502-3.
- A. Jadhav and K. Nagaraj, Surfactant-Enabled Nanocarriers in Breast Cancer Therapy: Targeted Delivery and Multidrug Resistance Reversal, Pharmaceutics, 2025, 17(6), 779 CrossRef CAS PubMed . Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC12197182/.
- X. Meng, Y. Shen, H. Zhao, X. Lu, Z. Wang and Y. Zhao, Redox-manipulating nanocarriers for anticancer drug delivery: a systematic review, J. Nanobiotechnol., 2024, 22(1), 587 CrossRef PubMed . Available from: https://link.springer.com/article/10.1186/s12951-024-02859-w.
- K. Bhalodi, C. Kothari and S. Butani, Next-generation cancer nanotherapeutics: Pluronic® F127 based mixed micelles for enhanced drug delivery, Naunyn Schmiedebergs Arch. Pharmacol., 2024, 398(4), 3241–3270 CrossRef PubMed . Available from: https://link.springer.com/article/10.1007/s00210-024-03582-x.
- G. Mattheolabakis, L. Milane, A. Singh and M. M. Amiji, Hyaluronic acid targeting of CD44 for cancer therapy: From receptor biology to nanomedicine, J. Drug Targeting, 2015, 23(7–8), 605–618 CrossRef CAS PubMed . Available from: https://www.tandfonline.com/doi/pdf/10.3109/1061186X.2015.1052072.
- P. Kesharwani, R. Chadar, A. Sheikh, W. Y. Rizg and A. Y. Safhi, CD44-Targeted Nanocarrier for Cancer Therapy, Front. Pharmacol., 2022, 12, 800481 CrossRef PubMed . Available from: https://www.frontiersin.org.
- H. Zhao, Y. Wang, J. Qian, M. Yang, C. Liu and W. Xu, et al., Stimulus-responsive doxorubicin-conjugated hyaluronic acid/polypyrrole nanoplatform for targeted photothermal chemotherapy of breast cancer, Int. J. Biol. Macromol., 2025, 322, 146945 CrossRef CAS PubMed . Available from: https://www.sciencedirect.com/science/article/pii/S0141813025075026.
- B. Xiao, M. K. Han, E. Viennois, L. Wang, M. Zhang and X. Si, et al., Hyaluronic acid-functionalized polymeric nanoparticles for colon cancer-targeted combination chemotherapy, Nanoscale, 2015, 7(42), 17745–17755 RSC . Available from: https://pubs.rsc.org/en/content/articlehtml/2015/nr/c5nr04831a.
- W. Jin, Role of JAK/STAT3 Signaling in the Regulation of Metastasis, the Transition of Cancer Stem Cells, and Chemoresistance of Cancer by Epithelial–Mesenchymal Transition, Cells, 2020, 9(1), 217 CrossRef CAS PubMed . Available from: https://www.mdpi.com/2073-4409/9/1/217/htm.
- M. Palmroth, K. Kuuliala, R. Peltomaa, A. Virtanen, A. Kuuliala and A. Kurttila, et al., Tofacitinib Suppresses Several JAK-STAT Pathways in Rheumatoid Arthritis In Vivo and Baseline Signaling Profile Associates With Treatment Response, Front. Immunol., 2021, 12, 738481 CrossRef CAS PubMed . Available from: https://www.frontiersin.org.
- L. De Lellis, S. Veschi, N. Tinari, Z. Mokini, S. Carradori and D. Brocco, et al., Drug Repurposing, an Attractive Strategy in Pancreatic Cancer Treatment: Preclinical and Clinical Updates, Cancers, 2021, 13(16), 3946 CrossRef CAS PubMed . Available from: https://www.mdpi.com/2072-6694/13/16/3946/htm.
- U. S. Younis, E. Vallorz, K. J. Addison, J. G. Ledford and P. B. Myrdal, Preformulation and Evaluation of Tofacitinib as a Therapeutic Treatment for Asthma, AAPS PharmSciTech, 2019, 20(5), 167 CrossRef PubMed . Available from: https://link.springer.com/article/10.1208/s12249-019-1377-0.
- S. Bashir, M. Aamir, R. M. Sarfaraz, Z. Hussain, M. U. Sarwer and A. Mahmood, et al., Fabrication, characterization and in vitro release kinetics of tofacitinib-encapsulated polymeric nanoparticles: a promising implication in the treatment of rheumatoid arthritis, Int. J. Polym. Mater. Polym. Biomater., 2021, 70(7), 449–458 CrossRef CAS . Available from: https://www.tandfonline.com/doi/pdf/10.1080/00914037.2020.1725760.
- M. Kuchukuntla, V. Palanivel and A. Madhubabu, Tofacitinib Citrate-loaded Nanoparticle Gel for the Treatment of Alopecia Areata: Response Surface Design, Formulation and In vitro-In vivo Characterization, Recent Adv. Drug Delivery Formulation, 2023, 17(4), 314–331 CrossRef CAS PubMed . Available from: https://www.benthamdirect.com/content/journals/raddf/10.2174/0126673878264814231106094853.
- D. Tripathi, J. Kumari, K. Rathour, P. Yadav, V. Shukla and A. K. Rai, A Review on the Progress of QbD Approach in Nanosystems Optimization: Current Updates and Strategic Applications, Lett. Drug Des. Discovery, 2023, 21(13), 2545–2566 CrossRef . Available from: https://www.benthamdirect.com/content/journals/lddd/10.2174/0115701808256947231004110357.
- S. Namjoshi, M. Dabbaghi, M. S. Roberts, J. E. Grice and Y. Mohammed, Quality by Design: Development of the Quality Target Product Profile (QTPP) for Semisolid Topical Products, Pharmaceutics, 2020, 12(3), 287 CrossRef PubMed . Available from: https://www.mdpi.com/1999-4923/12/3/287/htm.
- L. E. Kristensen, S. Danese, A. Yndestad, C. Wang, E. Nagy and I. Modesto, et al., Identification of two tofacitinib subpopulations with different relative risk versus TNF inhibitors: an analysis of the open label, randomised controlled study ORAL Surveillance, Ann. Rheum. Dis., 2023, 82(7), 901–910 CrossRef CAS PubMed . Available from: https://ard.bmj.com/content/82/7/901.
- S. Nishal, V. Jhawat, P. Phaugat and R. Dutt, In vitro characterization of tofacitinib loaded novel nanoemulgel for topical delivery for the management of rheumatic arthritis, Drug Dev. Ind. Pharm., 2022, 48(8), 374–383 CrossRef CAS PubMed . Available from: https://www.tandfonline.com/doi/pdf/10.1080/03639045.2022.2119572.
- T. S. Anirudhan, S. Varghese and V. Manjusha, Hyaluronic acid coated Pluronic F127/Pluronic P123 mixed micelle for targeted delivery of Paclitaxel and Curcumin, Int. J. Biol. Macromol., 2021, 192, 950–957 CrossRef CAS PubMed . Available from: https://www.sciencedirect.com/science/article/pii/S0141813021022121.
- L. Zhao, J. Du, Y. Duan, Y. Zang, H. Zhang and C. Yang, et al., Curcumin loaded mixed micelles composed of Pluronic P123 and F68: Preparation, optimization and in vitro characterization, Colloids Surf., B, 2012, 97, 101–108 CrossRef CAS PubMed . Available from: https://www.sciencedirect.com/science/article/pii/S0927776512002226.
- H. Roy, S. P. Panda, S. K. Panda, A. K. Tripathi, S. K. Srivastava and B. S. Nayak, et al., N-trimethyl chitosan and tripalmitin loaded solid lipid nanoparticles of tofacitinib citrate: Characterization and in vivo anti-inflammatory assessment, J. Drug Delivery Sci. Technol., 2023, 87, 104789 CrossRef CAS . Available from: https://www.sciencedirect.com/science/article/abs/pii/S177322472300641X.
- Z. Daman, S. Ostad, M. Amini and K. Gilani, Preparation, optimization and in vitro characterization of stearoyl-gemcitabine polymeric micelles: A comparison with its self-assembled nanoparticles, Int. J. Pharm., 2014, 468(1–2), 142–151 CrossRef CAS PubMed . Available from: https://www.sciencedirect.com/science/article/abs/pii/S037851731400249X.
- Z. Sezgin, N. Yüksel and T. Baykara, Preparation and characterization of polymeric micelles for solubilization of poorly soluble anticancer drugs, Eur. J. Pharm. Biopharm., 2006, 64(3), 261–268 CrossRef CAS PubMed . Available from: https://www.sciencedirect.com/science/article/abs/pii/S0939641106001548.
- F. Rehan, N. Ahemad, R. A. Islam, M. Gupta, S. H. Gan and E. H. Chowdhury, Optimization and Formulation of Nanostructured and Self-Assembled Caseinate Micelles for Enhanced Cytotoxic Effects of Paclitaxel on Breast Cancer Cells, Pharmaceutics, 2020, 12(10), 984 CrossRef CAS PubMed . Available from: https://www.mdpi.com/1999-4923/12/10/984/htm.
- A. Salimi, B. S. Makhmal Zadeh and M. Kazemi, Preparation and optimization of polymeric micelles as an oral drug delivery system for deferoxamine mesylate: In vitro and ex vivo studies, Res. Pharm. Sci., 2019, 14(4), 293–307 CrossRef PubMed . Available from: https://journals.lww.com/rips/fulltext/2019/14040/preparation_and_optimization_of_polymeric_micelles.2.aspx.
- S. Kaewpaiboon and T. Srichana, Formulation Optimization and Stability of Polymyxin B Based on Sodium Deoxycholate Sulfate Micelles, J. Pharm. Sci., 2022, 111(8), 2249–2257 CrossRef CAS PubMed . Available from: https://www.sciencedirect.com/science/article/abs/pii/S0022354922000806.
- H. Zhang, L. Zhao, L. Chu, X. Han and G. Zhai, Preparation, optimization, characterization and cytotoxicity in vitro of Baicalin-loaded mixed micelles, J. Colloid Interface Sci., 2014, 434, 40–47 CrossRef CAS PubMed . Available from: https://www.sciencedirect.com/science/article/abs/pii/S0021979714005438.
- Z. Y. Fang, Z. X. Jun, J. Tian, S. Hu, B. X. Hong and X. Chen, Determination of teicoplanin in human plasma by reverse micelle mediated dispersive liquid-liquid microextraction with high performance liquid chromatography, J. Chromatogr. A, 2021, 1643, 462058 CrossRef PubMed , Available from: https://www.sciencedirect.com/science/article/abs/pii/S0021967321001825.
- K. D. Bharwad, P. A. Shah, P. S. Shrivastav and P. Singhal, Development and validation of a rapid and sensitive UPLC–MS/MS assay for the quantification of tofacitinib in human plasma, Biomed. Chromatogr., 2019, 33(4), e4458 CrossRef PubMed . Available from: https://doi/pdf/10.1002/bmc.4458.
- J. E. Kim, M. Y. Park and S. H. Kim, Simple determination and quantification of tofacitinib, a JAK inhibitor, in rat plasma, urine and tissue homogenates by HPLC and its application to a pharmacokinetic study, J. Pharm. Invest., 2020, 50(6), 603–612 CrossRef CAS . Available from: https://link.springer.com/article/10.1007/s40005-020-00490-z.
- S. Vieujean, D. Laharie, A. Buisson, X. Roblin, M. Fumery and S. Nancey, et al., Histological healing induced by tofacitinib in ulcerative colitis: A multicentre study, Dig. Liver Dis., 2024, 56(4), 613–621 CrossRef CAS PubMed . Available from: https://www.sciencedirect.com/science/article/abs/pii/S159086582301040X.
- S. Bashir, M. Aamir, R. M. Sarfaraz, Z. Hussain, M. U. Sarwer and A. Mahmood, et al., Fabrication, characterization and in vitro release kinetics of tofacitinib-encapsulated polymeric nanoparticles: a promising implication in the treatment of rheumatoid arthritis, Int. J. Polym. Mater. Polym. Biomater., 2021, 70(7), 449–458 CrossRef CAS . Available from: https://scholar.google.com/scholar_url?url=https://www.tandfonline.com/doi/pdf/10.1080/00914037.2020.1725760%3Fcasa_token%3DuTLx-im3_-gAAAAA:EOPIhrkn3tB24oTDSMGlKFMe9zLFRwQFOm_GUXL1_93TnMmurtKD-EV5LetaQFQXVA3CRf-CgF3Sig&hl=en&sa=T&oi=ucasa&ct=ucasa&ei=HoApafDkF6qy6rQP1qz4iAU&scisig=ABGrvjKWaQXptxj70J61kpP-fnFW.
- Z. Ansarypour, J. Emami, F. Hassanzadeh, M. Aghaei, M. Minaiyan and N. M. Davies, et al., Synthesis and in vitro evaluation of novel pH-triggered biocompatible folate-chondroitin sulfate-dexamethasone copolymers for delivery of tofacitinib in rheumatoid arthritis, Res. Pharm. Sci., 2025, 20(3), 316–342 CrossRef PubMed . Available from: https://journals.lww.com/rips/fulltext/2025/05000/synthesis_and_in_vitro_evaluation_of_novel.1.aspx.
- Z. Eskandari, F. Kazdal, F. Bahadori and N. Ebrahimi, Quality-by-design model in optimization of PEG-PLGA nano micelles for targeted cancer therapy, J. Drug Delivery Sci. Technol., 2018, 48, 393–402 CrossRef CAS . Available from: https://www.sciencedirect.com/science/article/abs/pii/S1773224718309055.
- S. Kim, Y. Shi, J. Y. Kim, K. Park and J. X. Cheng, Overcoming the barriers in micellar drug delivery: Loading efficiency, in vivo stability, and micelle-cell interaction, Expert Opin. Drug Delivery, 2010, 7(1), 49–62 CrossRef CAS PubMed . Available from: https://www.tandfonline.com/doi/pdf/10.1517/17425240903380446.
- T. K. Wei and S. Manickam, Response Surface Methodology, an effective strategy in the optimization of the generation of curcumin-loaded micelles, Asia–Pac. J. Chem. Eng., 2012, 7(Suppl. 1), S125–S133 CAS . Available from: https://doi/pdf/10.1002/apj.661.
- R. Kanade, M. Boche and V. Pokharkar, Self-Assembling Raloxifene Loaded Mixed Micelles: Formulation Optimization, In Vitro Cytotoxicity and In Vivo Pharmacokinetics, AAPS PharmSciTech, 2017, 19(3), 1105–1115 CrossRef PubMed . Available from: https://link.springer.com/article/10.1208/s12249-017-0919-6.
- A. Raval, A. Parmar, A. Raval and P. Bahadur, Preparation and optimization of media using Pluronic® micelles for solubilization of sirolimus and release from the drug eluting stents, Colloids Surf., B, 2012, 93, 180–187 CrossRef CAS PubMed . Available from: https://www.sciencedirect.com/science/article/abs/pii/S0927776512000021.
- Y. Duan, J. Wang, X. Yang, H. Du, Y. Xi and G. Zhai, Curcumin-loaded mixed micelles: Preparation, optimization, physicochemical properties and cytotoxicity in vitro, Drug Delivery, 2015, 22(1), 50–57 CrossRef CAS PubMed . Available from: https://www.tandfonline.com/doi/pdf/10.3109/10717544.2013.873501.
- J. Zhang, J. Liu, Y. Zhao, G. Wang and F. Zhou, Plasma and cellular pharmacokinetic considerations for the development and optimization of antitumor block copolymer micelles, Expert Opin. Drug Delivery, 2015, 12(2), 263–281 CrossRef CAS PubMed . Available from: https://www.tandfonline.com/doi/pdf/10.1517/17425247.2014.945417.
- H. M. Aboud, S. F. El Menshawe, N. H. Mohammed, A. S. Tulbah and A. A. Ali, Optimization and Appraisal of Nintedanib-Loaded Mixed Polymeric Micelles as a Potential Nanovector for Non-Invasive Pulmonary Fibrosis Mitigation, Pharmaceuticals, 2024, 17(10), 1275 CrossRef CAS PubMed . Available from: https://www.mdpi.com/1424-8247/17/10/1275/htm.
- C. M. L. Carvalho, J. M. S. Cabral and M. R. Aires-Barros, Cutinase stability in AOT reversed micelles: system optimization using the factorial design methodology, Enzyme Microb. Technol., 1999, 24(8–9), 569–576 CrossRef CAS . Available from: https://www.sciencedirect.com/science/article/abs/pii/S014102299800163X.
| This journal is © The Royal Society of Chemistry 2026 |