Open Access Article
Open Access Articlenor-MC3 and nor-KC2: cationic ionizable lipids for the delivery of therapeutic nucleic acids
Deaglan Arnolda,
Nagavenkata Durga Prasad Atmuria,
Fariba Saadatia,
Ardalan Nabia,
Daniel Z. Kureka,
Anthony Tama,
Taniya Adakb,
Dominik Witzigmanna,
Glenn Sammis b,
Pieter R. Cullis
b,
Pieter R. Cullis ac,
Jayesh Kulkarni*a and
Marco A. Ciufolini
ac,
Jayesh Kulkarni*a and
Marco A. Ciufolini *a
*a
aNanoVation Therapeutics, Inc., 2665 East Mall 2nd floor, Vancouver, BC V6T 1Z4, Canada. E-mail: j.kulkarni@nanovationtx.com; m.ciufolini@nanovationtx.com
bDepartment of Chemistry, University of British Columbia, 2036 Main Mall, Vancouver, BC V6T 1Z1, Canada
cDepartment of Biochemistry and Molecular Biology, University of British Columbia, Life Sciences Center, 2350 Health Sciences Mall, Vancouver, BC V6T 1Z3, Canada
First published on 4th March 2026
Abstract
nor-MC3 and nor-KC2, analogues of D-Lin-MC3-DMA (cationic ionizable lipid in Onpattro®) and D-Lin-KC2-DMA (valuable research tool) wherein C17 lipophilic chains replace C18 ones, are at least as efficacious as the originals, but more economical and safer to produce.
A recent paper describes a new type of lipid nanoparticles (LNPs) for the extrahepatic delivery of mRNA.1 A key component of such LNPs is a cationic ionizable lipid termed nor-MC3, 1. The significance of the above paper and the intense interest in LNP technology for the delivery of therapeutic nucleic acids2 prompt us to disclose details of the synthesis and properties of 1 and its congener, nor-KC2, 2 (Fig. 1).
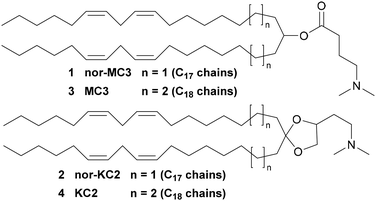 | ||
| Fig. 1 Structures of nor-MC3, 1, nor-KC2, 2, and of the parent lipids, MC3, 3, and KC2, 4. See SI for details. | ||
The new compounds are lower homologues of D-Lin-MC3-DMA,3 or more simply MC3 (3, the ionizable lipid component of Onpattro®),4 and D-Lin-KC2-DMA, or more simply KC2, 4.5 While the latter is not a component of any yet-approved medication, it is a valuable research tool that is especially effective, inter alia, for the delivery of plasmid DNA.6 Notice that in 1 and 2 C17 lipophilic chains replace the C18 chains present in 3 and 4, hence the designation nor-MC3/nor-KC2.
The synthesis of 1 and 2 (ref. 7) started with a Claisen condensation of methyl linoleate, 5, under Tanabe–Mukaiyama conditions,8 resulting in the formation of beta-ketoester 6 in 96% yield (Fig. 2).9 Notice that 5 (ca. USD 3 per g) is only 60% of the cost of linoleyl alcohol (ca. USD 5 per g) and considerably cheaper that linoleyl bromide (ca. USD 81 per g): the starting materials for the original syntheses of 3 and 4. Crude 6 exists as a mixture of keto- (major) and enol tautomers in variable proportions, typically about 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. The compound can be purified by normal phase medium pressure liquid chromatography (MPLC) for full characterization, in which case the keto and enol forms are separable. However, it is expedient to convert crude 6 directly into ketone 7 by ester saponification (aq. NaOH) followed by acidification and rotary evaporation of all volatiles at a bath temperature of 60 °C (decarboxylation). Crude 7 was thus obtained in just over 90% overall yield from 5. Very pure 7 can be obtained by MPLC (see SI).
1. The compound can be purified by normal phase medium pressure liquid chromatography (MPLC) for full characterization, in which case the keto and enol forms are separable. However, it is expedient to convert crude 6 directly into ketone 7 by ester saponification (aq. NaOH) followed by acidification and rotary evaporation of all volatiles at a bath temperature of 60 °C (decarboxylation). Crude 7 was thus obtained in just over 90% overall yield from 5. Very pure 7 can be obtained by MPLC (see SI).
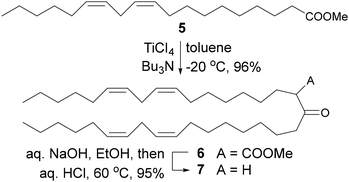 | ||
| Fig. 2 Preparation of ketone 7 by Claisen condensation of methyl linoleate under Tanabe–Mukaiyama conditions. See SI for details. | ||
It should be noted that the Claisen condensation of 5 carried out under customary basic conditions, e.g., with NaH in refluxing xylenes, as reported in a patent,10 promotes variable degrees of double bond isomerization, as inferred from the appearance of new signals in the olefinic region of NMR spectra.11 There seems to be no mention of the problem in said patent. We were unable to separate double bond isomers of 6, 7, or derived lipids, precluding the use of the latter in biological experiments. In contrast, no evidence of isomerization was apparent from the NMR spectra of crude 6 prepared by the Tanabe–Mukaiyama method, or from spectra of derived products.
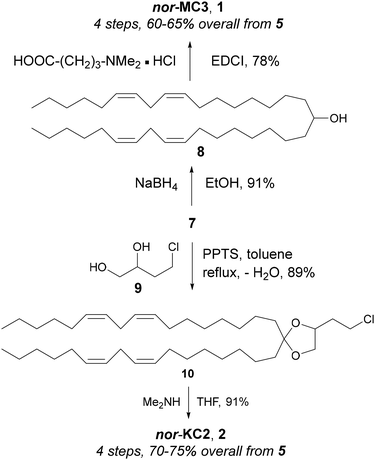
Ketone 7 was advanced to nor-MC3 by NaBH4 reduction to 8 and esterification thereof with 4-(dimethylamino)butanoic acid hydrochloride (78% yield over 2 steps), and to nor-KC2 by ketalization with chlorodiol 911 followed by halide displacement with dimethylamine (80% over 2 steps; Fig. 3). Lipids 1 and 2 are thus available from economical 5 in only 4 steps.
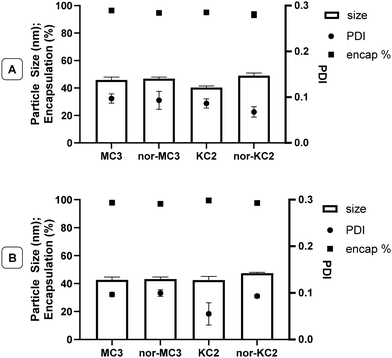
Key physical properties of LNP formulations of anti-firefly luciferase siRNA and firefly luciferase mRNA based on 1 and 2 were virtually identical to those of LNPs produced from MC3 and KC2 (Fig. 4), with a particle size of ca. 40–45 nm, high encapsulation (>90%), low polydispersity (<0.1), and identical apparent pKa in the LNPs (∼6.4).
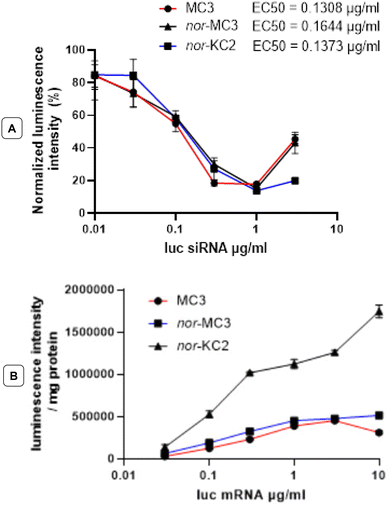
Lipids 1 and 2 proved to be at least as efficacious as the benchmark MC3 in their ability to deliver siRNA and mRNA both in vitro and in vivo. An in vitro siRNA luciferase suppression assay with LNPs based on either nor-lipid revealed an EC50 of about 0.1 μg siRNA per mL: equivalent to that of particles based on MC3. Likewise, the in vitro efficacy of nor-MC3-based LNP formulations of firefly luciferase mRNA was practically identical to that of MC3-containing ones, but interestingly, nor-KC2-centered formulations were significantly more efficacious, consistent with previous reports of delivery with KC2-containing formulations in vitro (Fig. 5).12
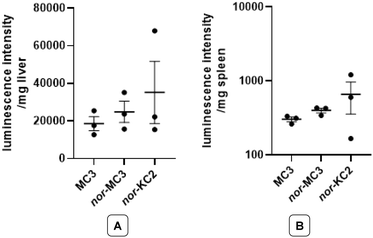
The difference in efficacy among the three lipids was attenuated in vivo (mice, Fig. 6). In all cases, no obvious adverse effects were observed in mice receiving formulations containing nor-MC3 or nor-KC2, suggesting that 1 and 2 probably are as safe as MC3. Formulations containing KC2 itself were excluded from in vivo studies, as MC3 is considered the gold standard for intravenous mRNA-LNP delivery and a clinically approved lipid.
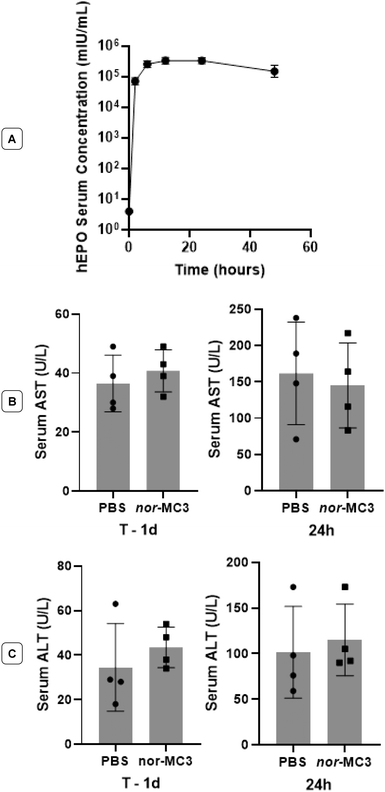
The efficacy and safety of nor-MC3 in non-human primates was evaluated in Macaca fascicularis (cynomolgus monkeys) following injection of human erythropoietin (hEPO) mRNA-LNP at a dose of 0.4 mg kg−1. High levels of plasma hEPO and no changes in hematology, clinical chemistry, or pro-inflammatory cytokine induction were observed compared to untreated control animals (Fig. 7). Due to the scale and ethical considerations of running studies in non-human primates, we chose to assess only nor-MC3, as MC3 has been reported on extensively.13
In summary, nor-MC3 and nor-KC2 show favorable in vivo delivery of nucleic acids relative to the benchmark MC3 and KC2 LNPs. Their chemical synthesis (4 steps from methyl linoleate in either case) is more concise than that of MC3 (5 steps from linoleyl alcohol3 or 6 from methyl linoleate3,11) or KC2 (8 (ref. 5) or 5 (ref. 11) steps from linoleyl alcohol), relative to which it bypasses hazardous Grignard14 and PCC oxidation15 reactions, which are best avoided in pharmaceutical manufacturing. Furthermore, the present route to 1 and 2 affords synthetic intermediates and final compounds that are easier to purify. All this translates into significant economies in terms of reagents, solvents, chromatographic supports, operator time, and waste disposal costs.
This research was supported by NanoVation Therapeutics, Inc., and MITACS (postdoctoral fellowship to T. Adak, award no. IT34879).
Author contributions
MAC conceived the nor-lipids. DW, PRC, JK conceived all biological work. DA, NDPA, FS, AN, TA, synthesized the compounds described herein and/or optimized the synthetic routes. DZK designed LNP formulations based on 1 and 2 and directed their preparation. AT designed and directed the biological studies. GS acted as TA's academic supervisor.Conflicts of interest
DW, PRC, JK, MAC are co-founders of NanoVation Therapeutics, Inc., and hold a financial interest in the company. DA was a full-time employee of NanoVation Therapeutics, Inc., during the conduct of the research described herein. NDPA, FS, AN, DZK, AT, DW, JK, MAC are full-time employees of NanoVation Therapeutics, Inc. TA, GS have no conflicts to declare.Ethical statement
All murine procedures were performed in accordance with the Guidelines for Care and Use of Laboratory Animals approved by the University of British Columbia Animal Care Committee. The cynomolgus monkey study was run by JOINN Laboratories Co. in an Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC) approved facility using non-naïve cynomolgus monkeys of Asian origin. Animal Care was compliant with the SOPs of JOINN Laboratories (Suzhou) Co., Ltd, the Guide for the Care and Use of Laboratory Animals (Institute of Laboratory Animal Resources, Commission on Life Sciences, National Research Council; National Academy Press; Washington, D.C., 2010), and the U.S. Department of Agriculture through the Animal Welfare Act (Public Law 99-198). The sample sizes for experimental groups were chosen based on ethical principles to use the fewest number of animals possible, consistent with the study objective.Data availability
Supplementary information (SI): procedures for the preparation, characterization, and evaluation of LNP formulations of nucleic acids; synthetic procedure, characterization data, and NMR spectra (1H and 13C) for all compounds. See DOI: https://doi.org/10.1039/d5pm00350d.Additional data may be requested from the authors.
References
- M. H. Y. Cheng, Y. Zhang, K. Fox, J. Leung, C. Strong, E. Kang, Y. Chen, M. Tong, H. Bommadevara, E. Jan, O. Y. L. Ip, C. Rodriguez-Rodriguez, K. Saatchi, U. O. Hafeli, A. Abdolahzadeh, D. Witzigmann and P. R. Cullis, Nat. Commun., 2025, 16, 4135 CrossRef CAS PubMed.
- Reviews: (a) P. R. Cullis and P. L. Felgner, Nat. Rev. Drug Discovery, 2024, 23, 709 CrossRef CAS PubMed; (b) Y. Wu, S. Yua and I. de Lázaro, Nanoscale, 2024, 16, 6820 RSC; (c) A. S. Alfutaimani, N. K. Alharbi, A. S. Alahmari, A. A. Alqabbani and A. M. Aldayel, Int. J. Pharm., 2024, 8, 100305 CAS.
- M. Jayaraman, S. M. Ansell, B. I. Mui, Y. K. Tam, J. Chen, X. Du, D. Butler, L. Eltepu, S. Matsuda, J. K. Narayanannair, K. G. Rajeev, I. M. Hafez, A. Akinc, M. A. Maier, M. A. Tracy, P. R. Cullis, T. D. Madden, M. Manoharan and M. J. Hope, Angew. Chem., Int. Ed., 2012, 51, 8529 CrossRef CAS PubMed.
- A. Akinc, M. A. Maier, M. Manoharan, K. Fitzgerald, M. Jayaraman, S. Barros, S. Ansell, X. Du, M. J. Hope, T. D. Madden, B. L. Mui, S. C. Semple, Y. K. Tam, M. A. Ciufolini, D. Witzigmann, J. A. Kulkarni, R. van der Meel and P. R. Cullis, Nat. Nanotechnol., 2019, 14, 1084 CrossRef CAS PubMed.
- S. C. Semple, A. Akinc, X. Chen, A. P. Sandhu, B. L. Mui, C. K. Cho, D. W. Y. Sah, D. Stebbing, E. J. Crosley, E. Yaworski, I. M. Hafez, J. R. Dorkin, J. Qin, Q. Lam, K. G. Rajeev, K. F. Wong, L. B. Jeffs, L. Nechev, M. L. Eisenhardt, M. Jayaraman, M. Kazem, M. A. Maier, M. Srinivasulu, M. J. Weinstein, Q. Chen, R. Alvarez, S. A. Barros, S. De, S. K. Klimuk, T. Borland, V. Kosovrasti, W. L. Cantley, Y. K. Tam, M. Manoharan, M. A. Ciufolini, M. A. Tracy, A. de Fougerolles, I. MacLachlan, P. R. Cullis, T. D. Madden and M. J. Hope, Nat. Biotechnol., 2010, 28, 172 CrossRef CAS PubMed.
- A. Algarni, E. H. Pilkington, E. J. A. Suys, H. Al-Wassiti, C. W. Pouton and N. P. Truong, Biomater. Sci., 2022, 10, 2940 RSC.
- nor-KC2, nor-MC3, and the chemical technology for their synthesis are protected by the following patent applications (a) M. A. Ciufolini, F. Saadati, A. Tam, D. Kurek, D. Witzigmann and J. Kulkarni, WO2022246555, 2022; (b) M. A. Ciufolini, F. Saadati, A. C. Tam, D. Kurek, D. Witzigmann and J. Kulkarni, WO2022246571, 2022; (c) M. A. Ciufolini, F. Saadati, A. C. Tam, D. Kurek, D. Witzigmann and J. Kulkarni, WO2022246568, 2022.
- (a) Y. Tanabe and T. Mukaiyama, Chem. Lett., 1984, 1867 CrossRef CAS; (b) Y. Tanabe and T. Mukaiyama, Chem. Lett., 1986, 1813 CrossRef CAS; (c) Y. Yoshida, R. Hayashi, R. Sumihara and Y. Tanabe, Tetrahedron Lett., 1997, 38, 8727 CrossRef CAS; (d) T. Misaki, R. Nagase, K. Matsumoto and Y. Tanabe, J. Am. Chem. Soc., 2005, 127, 2854 CrossRef CAS PubMed.
- Related reaction of methyl oleate: (a) A. Sytniczuk, M. Dabrowski, L. Banach, M. Urban, S. Czarnocka-Sniadala, M. Milewski, A. Kajetanowicz and K. Grela, J. Am. Chem. Soc., 2018, 140, 8895 CrossRef CAS PubMed; (b) M. Dabrowski, P. Wyrebek, D. Trzybinski, K. Wozniak and K. Grela, Chem. – Eur. J., 2020, 26, 3782 CrossRef CAS PubMed.
- M. Koizumi, Y. Onishi, T. Niwa, M. Tamura and Y. Kasuya, EP3020701B1, 2014.
- A. Nabi, H. Tran, F. Saadati, N. D. Prasad Atmuri, G. Dake and M. A. Ciufolini, Eur. J. Org. Chem., 2025, e202500592 CrossRef CAS.
- J. A. Kulkarni, J. L. Myhre, S. Chen, Y. Y. C. Tam, A. Danescu, J. M. Richman and P. R. Cullis, Nanomedicine, 2017, 13, 1377 CrossRef CAS PubMed.
- M. Sedic, J. J. Senn, A. Lynn, M. Laska, M. Smith, S. J. Platz, J. Bolen, S. Hoge, A. Bulychev, E. Jacquinet, V. Bartlett and P. F. Smith, Vet. Pathol., 2018, 55, 341 CrossRef CAS PubMed.
- D. J. am Ende and P. F. Vogt, Org. Process Res. Dev., 2003, 7, 1029 CrossRef CAS.
- (a) R. W. Dugger, J. A. Ragan and D. H. Brown Ripin, Org. Process Res. Dev., 2005, 9, 253 CrossRef CAS; (b) S. Caron, R. W. Dugger, S. Gut Ruggeri, J. A. Ragan and D. H. Brown Ripin, Chem. Rev., 2006, 106, 2943 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |