 Open Access Article
Open Access ArticleDevelopment and evaluation of fenticonazole nitrate-loaded, fucoidan-coated lecithin–chitosan nanoparticles for the treatment of vaginitis
Valamla Bhavanaa,
Padakanti Sandeep Charya,
Urati Anuradhab,
Kishan Kumar Paridab,
Nitin Pal Kalia b and
Neelesh Kumar Mehra
b and
Neelesh Kumar Mehra *a
*a
aPharmaceutical Nanotechnology Research Laboratory, Department of Pharmaceutics, National Institute of Pharmaceutical Education and Research (NIPER), Ministry of Chemicals and Fertilizers, Government of India, Hyderabad, Telangana, 500037 India. E-mail: neelesh@niperhyd.ac.in; neelesh81mph@gmail.com; Tel: +91-8839030437 Tel: +91-40-230747750
bDepartment of Biological Sciences, National Institute of Pharmaceutical Education and Research (NIPER), Hyderabad, Telangana, 500037 India
First published on 16th January 2026
Abstract
In this investigation, self-assembling fenticonazole-loaded lecithin–chitosan nanoparticles (FZNP) with cationic zeta potential were altered by coating them with an anionic fucoidan polymer (Fu-FZNP) through an ionic gelation method to overcome the vaginal mucosal barrier. FZNP and Fu-FZNP possessed particle sizes of 129.20 ± 0.25 nm and 227.10 ± 1.54 nm, polydispersity indexes of 0.21 ± 0.00 and 0.26 ± 0.01, and zeta potentials of 30.96 ± 1.15 mV and −26.75 ± 0.3 mV, respectively. The entrapment efficiency and drug loading were 65.47% ± 2.32% and 11.69% ± 0.414% for FZNP and 71.13% ± 5.74% and 7.41% ± 0.60% for Fu-FZNP, respectively. The nanoparticles exhibited spherical and smooth morphology under TEM imaging. An excised goat vagina was used for the ex vivo permeation studies, which showed drug permeations of 61.74% ± 2.07% for FZNP and 72.11% ± 1.4% for Fu-FZNP. FZNP and Fu-FZNP demonstrated antibacterial and antifungal properties against Staphylococcus aureus and Candida albicans, respectively, in vitro. Therefore, FZN and Fu-FZNP may be developed further for the safe, practical, and efficient treatment of mixed vaginal infections.
1. Introduction
Vaginal infection is a chronic gynecological issue in women of reproductive age that can lead to various health complications. Mixed vaginitis, which occurs rarely, is caused by the presence of two vaginal pathogens (bacterium and fungus (Candida species)), necessitating dual therapy with a combination of antifungal and antibacterial drugs or broad-spectrum monotherapy to eliminate concurrent symptoms.1–3Fenticonazole is an imidazole derivative with antifungal properties that was created about thirty years ago to treat dermatomycosis and Candida vaginitis topically. Fenticonazole nitrate (FZN) has demonstrated extensive antimycotic action against yeasts and dermatophytes in both in vitro and clinical investigations.4,5 Three distinct mechanisms allow fenticonazole to perform its specific antimycotic activity: (i) reduction of the protease acid secretion of Candida albicans, (ii) cytoplasmic membrane damage, and (iii) blockage of cytochrome oxidase and peroxidases.3,6–9 Fenticonazole nitrate is commercially available in conventional dosage forms, such as vaginal creams, ovules, and pessaries (e.g., 2% vaginal cream and 600 mg vaginal ovules), which are commonly prescribed for vulvovaginal candidiasis and mixed infections. However, these formulations suffer from limitations such as poor retention at the vaginal site and require frequent dosing, leading to reduced patient compliance.
Two additional pharmacological properties of fenticonazole are noteworthy. The drug persists in the skin's stratum corneum for a prolonged period, and it has unique pharmacokinetic properties that enable the accumulation of the active drug in mucosal tissue for up to 72 h. This accumulation forms a reservoir of FZN, delaying the need for consecutive administrations. Moreover, the assessment of fenticonazole plasma levels has validated the drug's inadequate systemic absorption. Consequently, for mixed infections comprising mycotic, bacterial, dermatophyte, and/or Trichomonas species, fenticonazole may be the perfect topical substitute for combination therapy.10–12
The vaginal route is an essential mode of drug delivery to treat local and systemic ailments. The vaginal route offers several benefits, including large surface area, adequate blood supply, lack of first-pass effect, slightly greater drug permeability, and self-insertion simplicity.13 However, conventional vaginal dosage forms are limited by short residence time, mucus turnover, and variable drug retention, which can reduce therapeutic efficacy. The literature suggests using inventions that attach to vaginal mucosa by forming chemical and physical bonds with the mucus as one way to address these challenges. However, mucosal drug delivery is intrinsically challenging since vaginal medication distribution is constrained by the presence of anatomical and physiological factors.14–16
Many medical conditions, such as cancer and fungal and bacterial infections, can be treated and prevented through vaginal drug delivery.17 The high porosity, negative charge, interconnectivity, and the hydrophilic and hydrophobic regions of the vaginal microenvironment contribute to the barrier properties of vaginal drug delivery. In order to enhance the vaginal delivery of active substances, mucoadhesive and mucus-penetrating particles have been designed by adjusting their chemical constituents, surface characteristics, size, and amount of surface functionalization.18,19
The advancement of nanotechnology in recent times has created novel pathways, i.e., using muco-adhesive and mucus-penetrating polymers and zeta-potential-modifying polymers to overcome the vaginal mucosal barriers.20–22 A naturally occurring polycationic polymer, chitosan, is created when chitin undergoes partial deacetylation. Chitosan has the added benefit of being naturally derived, which makes it biodegradable and biocompatible, with good bioadhesive properties.23 Chitosan is being utilized to produce nanoparticles via ionotropic gelation with tripolyphosphate.24,25 Furthermore, chitosan has been studied for the stabilization of microemulsions in combination with lecithin as an emulsifying agent.26 Lecithin is a phospholipid combination that is commonly utilized to form liposomes and micelles.27,28 Interestingly, encapsulating these lipid-based nanostructures with chitosan has been shown to improve their stability and give mucoadhesive properties.29–31 In recent years, films and gels built around the bonding between negatively charged phospholipids and chitosan have been considered for the transport of lipophilic drugs.32,33 Fucoidan is a sulfated polysaccharide with a high density of negatively charged sulfate groups, which plays a crucial role in modulating the surface charge (zeta potential) of lecithin–chitosan nanoparticles. This modulation reduces excessive mucoadhesion while promoting mucus penetration by minimizing the electrostatic interactions with negatively charged mucins. Additionally, fucoidan's bioactive properties, including anti-inflammatory and antimicrobial effects, further enhance its suitability for vaginal drug delivery.
The lecithin–chitosan nanoparticles are promising drug carriers formed by the supramolecular self-organizing electrostatic interactions between negatively charged lecithin and positively charged chitosan.34–36 Chitosan/lecithin nanoparticles loaded with melatonin, quercetin, and diflucortolone valerate are just a few examples in this study.37–40
Mucus is digested, excreted, regenerated, and secreted continually. Mucus has a brief turnover time, frequently measured in minutes to hours, especially for the poorly adhering mucus layer. Effective particle clearance is achieved by the vaginal mucus output rate of about 6 mL day−1 for vaginal medication delivery. Therefore, there is a need to develop smart delivery schemes that do not initially interact with the mucus but later adhere to the membrane and mucus to overcome the continually cleared mucus.41–43 To circumvent the drawbacks of available vaginal drug delivery systems while preserving and even refining the safety profiles, a novel vaginal drug delivery system was developed by surface modification of positively charged lecithin–chitosan nanoparticles with negatively charged fucoidan to alter the zeta potential of the system.
2. Materials and methods
2.1. Materials
Fenticonazole was obtained as a gift sample from Optimus Pharma, Hyderabad. Fucoidan and chitosan were purchased from Sigma-Aldrich, St Louis, Missouri, USA. The essential oils (lemongrass oil, lavender oil) were procured from Botanical Healthcare (Bangalore, India). Solvents such as methanol (HPLC analytical grade) were purchased from SRL Chemicals, India. Polyvinyl alcohol (98–99% hydrolysis) was purchased from Loba Chemie (Mumbai, India) and hyaluronic acid from PC CHEM, Mumbai, India. Deionized water was used for the studies, and other chemicals were used as received from the distributors.2.2. Methodology
 | ||
| Fig. 1 Schematic representation of the formation of fenticonazole nitrate-loaded, fucoidan-coated lecithin–chitosan nanoparticles by the ethanol injection method. | ||
| Batches | Lecithin (mg) | Chitosan (mg) | Fucoidan (mg mL−1) | Stirring speed (rpm) | FZN (mg) | Size (nm) | PDI | Zeta potential (mV) |
|---|---|---|---|---|---|---|---|---|
| FZNP-1 | 37.5 | 3 | 0 | 400 | 10 | 140.7 | 0.22 | +37.57 |
| FZNP-2 | 50 | 3 | 0 | 300 | 5 | 125.3 | 0.24 | +34.78 |
| FZNP-3 | 50 | 2.25 | 0 | 400 | 10 | 202.3 | 0.28 | +37.12 |
| FZNP-4 | 25 | 3 | 0 | 400 | 5 | 129.2 | 0.20 | +34.78 |
| FZNP-5 | 50 | 1.5 | 0 | 200 | 10 | 169.7 | 0.27 | +37.03 |
| FZNP-6 | 25 | 2.25 | 0 | 200 | 5 | 117.5 | 0.20 | +36.67 |
| FZNP-7 | 25 | 1.5 | 0 | 300 | 10 | 135.8 | 0.24 | +31.42 |
| FZNP-8 | 37.5 | 2.25 | 0 | 300 | 7.5 | 146.0 | 0.26 | +25.70 |
| FZNP-9 | 50 | 3 | 0 | 200 | 7.5 | 156.3 | 0.25 | +41.19 |
| FZNP-10 | 50 | 1.5 | 0 | 400 | 5 | 215.5 | 0.22 | +10.35 |
| FZNP-11 | 25 | 3 | 0 | 200 | 10 | 179.3 | 0.23 | +41.27 |
| FZNP-12 | 25 | 1.5 | 0 | 400 | 7.5 | 221.4 | 0.38 | +45.10 |
| FZNP-13 | 25 | 1.5 | 0 | 500 | 5 | 186.2 | 0.23 | +45.46 |
| FZNP-14 | 50 | 1.5 | 0 | 500 | 10 | 230.4 | 0.34 | +61.88 |
| FZNP-15 | 50 | 3 | 0 | 300 | 5 | 264.2 | 0.37 | +56.19 |
| FZNP-16 | 25 | 1.5 | 0 | 300 | 5 | 297.3 | 0.28 | +41.98 |
| FZNP-17 | 50 | 1.5 | 0 | 300 | 10 | 271.8 | 0.33 | +55.14 |
| FZNP-18 | 25 | 3 | 0 | 500 | 10 | 178.7 | 0.20 | +58.73 |
| FZNP-19 | 50 | 3 | 0 | 500 | 5 | 325.9 | 0.38 | +53.22 |
| Fu-FZNP-1 | 25 | 1.5 | 1.5 | 500 | 5 | 288.2 | 0.28 | −38.20 |
| Fu-FZNP-2 | 50 | 1.5 | 1 | 500 | 10 | 302.0 | 0.30 | −49.91 |
| Fu-FZNP-3 | 50 | 3 | 1 | 300 | 5 | 387.8 | 0.36 | −37.61 |
| Fu-FZNP-4 | 25 | 1.5 | 1 | 300 | 5 | 313.3 | 0.30 | −37.79 |
| Fu-FZNP-5 | 50 | 1.5 | 1.5 | 300 | 10 | 299.8 | 0.34 | −46.07 |
| Fu-FZNP-6 | 25 | 3 | 1 | 500 | 10 | 210.0 | 0.21 | −21.32 |
| Fu-FZNP-7 | 25 | 3 | 1.5 | 300 | 10 | 254.4 | 0.27 | −45.19 |
| Fu-FZNP-8 | 50 | 3 | 1.5 | 500 | 5 | 331.4 | 0.34 | −46.55 |
| Fu-FZNP-9 | 37.5 | 3 | 1 | 400 | 10 | 216.4 | 0.20 | −34.63 |
| Fu-FZNP-10 | 25 | 3 | 0.5 | 400 | 5 | 220.2 | 0.15 | −16.16 |
| Fu-FZNP-11 | 50 | 1.5 | 0.5 | 200 | 10 | 241.9 | 0.23 | −19.18 |
| Fu-FZNP-12 | 25 | 2.25 | 1 | 200 | 5 | 227.1 | 0.26 | −26.75 |
| Fu-FZNP-13 | 25 | 1.5 | 1 | 300 | 10 | 197.8 | 0.22 | −36.65 |
| Fu-FZNP-14 | 37.5 | 2.25 | 0.5 | 300 | 7.5 | 238.7 | 0.43 | −19.11 |
| Fu-FZNP-15 | 50 | 3 | 1 | 200 | 7.5 | 191.0 | 0.15 | −40.89 |
| Fu-FZNP-16 | 50 | 1.5 | 1 | 400 | 5 | 260.6 | 0.22 | −29.75 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 w/w). The mixture was incubated under gentle stirring at room temperature for 2 h to allow electrostatic interaction and adsorption of fucoidan onto the nanoparticle surface.
2 w/w). The mixture was incubated under gentle stirring at room temperature for 2 h to allow electrostatic interaction and adsorption of fucoidan onto the nanoparticle surface.Fucoidan and chitosan are multifunctional polymers that are capable of being ionized in an aqueous medium. Fucoidan can be expressed as FU–COOH–SO3H/FU–COO–SO3−, and chitosan as CHI–NH3+/CHI–NH2 in their protonated and deprotonated forms, respectively. The ionic cross-linking relies on favorable chitosan and fucoidan electrostatic contact, and the quantity of ionizable locations on the surface of the polymers will determine the outcome. Table 1 illustrates how fucoidan was added to the previously optimized batch. The particle size and zeta potential of the resultant nanoparticles were further examined. Rhodamine B was incorporated into the nanoparticles for ex vivo permeation studies by replacing fenticonazole nitrate with rhodamine B during the nanoparticle preparation process, following the same formulation protocol. The dye was dissolved in the aqueous phase prior to nanoparticle formation, allowing its entrapment within the nanoparticulate matrix. The nanoparticles were freeze-dried for further analysis.
2.2.3.1. Particle size, PDI and zeta potential. The particle size, PDI and zeta potential of FZNP and Fu-FZNP were examined with a Zetasizer (NanoZS Malvern, UK). The polystyrene cuvettes were filled with nanoparticle solutions and analyzed at 173° scattering angle at 25 ± 0.5 °C. All the samples were examined in triplicate, and the results are reported as mean ± SD (n = 3).44,45
2.2.3.2. ATR-FTIR analysis of nanoparticles. Chitosan (CH), fucoidan (Fu), FZN, PM (a physical mixture of lecithin, chitosan, fucoidan, drug in a 1
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio), freeze-dried blank chitosan lecithin nanoparticles (BNP), FZNP and Fu-FZNP were examined spectroscopically using ATR-FTIR (Spectrum RX-1, PerkinElmer, US). Separately, 5 mg of freeze-dried samples were placed on the holder and scanned at a resolution of 4 cm−1, covering a wavenumber spectrum of 400–4000 cm−1.46,47
1 ratio), freeze-dried blank chitosan lecithin nanoparticles (BNP), FZNP and Fu-FZNP were examined spectroscopically using ATR-FTIR (Spectrum RX-1, PerkinElmer, US). Separately, 5 mg of freeze-dried samples were placed on the holder and scanned at a resolution of 4 cm−1, covering a wavenumber spectrum of 400–4000 cm−1.46,47
2.2.3.3. Differential scanning calorimetry (DSC) analysis of nanoparticles. DSC was used to measure the thermodynamic events of excipients and drug (STARe system, Metler Toledo, Switzerland). After weighing the samples (chitosan, fucoidan, FZN, PM, BNP, FZNP and Fu-FZNP), they were positioned in an aluminium pan and heated to an elevated temperature, in compliance with the protocol, while flowing 50 mL min−1 of liquid nitrogen through it. The samples were analysed over a temperature range of 25 °C to 300 °C for 10 minutes.48
2.2.3.4. P-XRD analysis of nanoparticles. PXRD (Empyrean, Malvern Panalytical, Worcestershire, United Kingdom) analyses of chitosan, fucoidan, FZN, PM, BNP, FZNP and Fu-FZNP were performed. The freeze-dried samples were examined at a speed of 0.0436°/sec and at 2θ angles ranging from 2 to 40°.49,50
2.2.3.5. Surface morphology by transmission electron microscopy (TEM). The morphology and structure of FZNP and Fu-FZNP were evaluated by TEM (JEM1400 TEM; JEOL, Tokyo, Japan) with 500 k magnification and 120 kV illumination (installed at the National Institute of Animal Biology (NIAB), Hyderabad). The nanoparticles were allowed to dry on a small carbon-coated grid and viewed with the microscope at a suitable magnification. The samples were prepared on a carbon grid and negatively stained with a 1% uranyl acetate. Microphotographs of FZNP and Fu-FZNP were recorded at appropriate magnification.51
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ± 100 rpm for 2 h at 4 ± 0.5 °C. Following centrifugation, the clear supernatant containing the unentrapped drug was carefully collected without disturbing the nanoparticle pellet. To ensure complete removal of surface-associated drug, the nanoparticle pellet was washed twice with buffer, followed by centrifugation under the same conditions. The wash supernatants were pooled with the initial supernatant. The supernatant was collected, diluted with methanol, and further analyzed by RP-HPLC, based on a previously constructed calibration curve. The % entrapment efficiency and drug loading were estimated using eqn (1) and (2), respectively.
000 ± 100 rpm for 2 h at 4 ± 0.5 °C. Following centrifugation, the clear supernatant containing the unentrapped drug was carefully collected without disturbing the nanoparticle pellet. To ensure complete removal of surface-associated drug, the nanoparticle pellet was washed twice with buffer, followed by centrifugation under the same conditions. The wash supernatants were pooled with the initial supernatant. The supernatant was collected, diluted with methanol, and further analyzed by RP-HPLC, based on a previously constructed calibration curve. The % entrapment efficiency and drug loading were estimated using eqn (1) and (2), respectively.
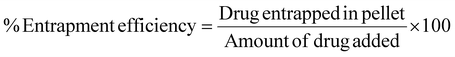 | (1) |
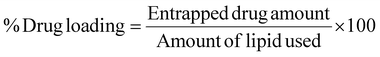 | (2) |
 | (3) |
3. Results and discussion
3.1. Optimization of FZNP and Fu-FZNP
A total of 36 batches were designed, developed, and characterized by varying the process and excipient-related attributes. The batches were optimized based on physical appearance, particle size, PDI and zeta potential, as shown in Table 1. In the current work, ethanolic lecithin/FZN solution was injected into an aqueous chitosan solution to produce lecithin–chitosan nanoparticles. As illustrated in Fig. 1, positively charged chitosan and negatively charged lecithin interacted through self-assembly. The resulting nanoparticles were clear and blue in appearance. As indicated in Table 1, their characteristics were determined by measuring the mean particle size, zeta potential and PDI. It is believed that the positively charged chitosan covering of the nanoparticles’ surface is the cause of their higher positive zeta potential.The material attributes utilized in the optimization of the formulation were the concentrations of drug, lecithin, chitosan, and fucoidan, followed by process parameters, including stirring speed and rate of addition. The amount of lecithin, chitosan and fucoidan had positive effects on the size and PDI. The stirring speed and the rate of addition had negative effects on the size. The concentration of lecithin was fixed at 25 mg, and chitosan at 3 mg, and the amount of drug was varied from 5 mg to 10 mg as the optimized batch, with a stirring speed of 500 rpm. The zeta potential increased with an increase in the chitosan amount and decreased with an increase in the lecithin amount. The fucoidan addition for altering the zeta potential resulted in increased particle size and a negative potential for Fu-FZNP. The optimized batch was further characterized and evaluated.
3.2. Physicochemical characterization of nanoparticles
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N of the imidazole group (1578 cm−1), C–H (1465 cm−1), ν(C
N of the imidazole group (1578 cm−1), C–H (1465 cm−1), ν(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C) (1403 cm−1), C–N (1295 cm−1), C–O–C (1118 cm−1), C–S (733 cm−1), and C–Cl (626 cm−1) were identified for FZN. The CH spectra exhibited peaks for C
C) (1403 cm−1), C–N (1295 cm−1), C–O–C (1118 cm−1), C–S (733 cm−1), and C–Cl (626 cm−1) were identified for FZN. The CH spectra exhibited peaks for C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N (1577 cm−1), C
N (1577 cm−1), C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O (1653 cm−1), C–O–C (1176 cm−1), and C–O (1054 cm−1). Fu exhibited peaks for S
O (1653 cm−1), C–O–C (1176 cm−1), and C–O (1054 cm−1). Fu exhibited peaks for S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O (1165–1220 cm−1), and C–O–S (812 cm−1), and the C–H (2922 cm−1 and 2853 cm−1) stretching band of the long fatty acid chain was observed for lecithin. PM illustrated all the characteristic peaks pertaining to the drug and excipients, without the formation of any new bonds, indicating the absence of interaction between the drug and excipients. The FZNP and Fu-FZNP exhibited characteristic peaks of CH, Fu, and Le with a redshift of the peak positions. The characteristic peaks of the drug were not observed in FZNP and Fu-FZNP, indicating the encapsulation of FZN in the nanoparticles.
O (1165–1220 cm−1), and C–O–S (812 cm−1), and the C–H (2922 cm−1 and 2853 cm−1) stretching band of the long fatty acid chain was observed for lecithin. PM illustrated all the characteristic peaks pertaining to the drug and excipients, without the formation of any new bonds, indicating the absence of interaction between the drug and excipients. The FZNP and Fu-FZNP exhibited characteristic peaks of CH, Fu, and Le with a redshift of the peak positions. The characteristic peaks of the drug were not observed in FZNP and Fu-FZNP, indicating the encapsulation of FZN in the nanoparticles.
| Batch | Sample interval | Stability conditions | Physical appearance | Particle size (nm) | PDI | Zeta potential | Entrapment efficiency (%) |
|---|---|---|---|---|---|---|---|
| FZNP | Initial | — | Bluish transparent | 129.20 ± 0.25 | 0.21 ± 0.003 | 30.96 ± 1.15 | 65.46 ± 2.32 |
| 1 month | 5 ± 3 °C | 130.20 ± 0.62 | 0.20 ± 0.004 | 32.74 ± 1.70 | 62.01 ± 1.63 | ||
| 25 ± 3 °C | 135.15 ± 0.73 | 0.24 ± 0.01 | 34.35 ± 1.06 | 61.97 ± 1.92 | |||
| Fu-FZNP | Initial | — | Milky white | 227.10 ± 0.26 | 0.26 ± 0.01 | 26.75 ± 0.32 | 73.14 ± 1.23 |
| 1 month | 5 ± 3 °C | 228.83 ± 0.85 | 0.27 ± 0.008 | 29.89 ± 0.38 | 70.09 ± 2.39 | ||
| 25 ± 3 °C | 224.13 ± 0.82 | 0.30 ± 0.036 | 26.92 ± 0.606 | 68.42 ± 2.64 |
4. Conclusion
In the present investigation, lecithin–chitosan nanoparticles and fucoidan-coated nanoparticles loaded with FZN for the management of mixed vaginal infections were formulated and evaluated. The FZNP and Fu-FZNP were developed by using the ethanol injection technique, and the nanoparticles displayed a mean particle size of 129.20 ± 0.25 nm, a PDI of 0.21 ± 0.003 and a zeta potential of +30.96 ± 1.15 mV. Furthermore, the fucoidan-coated zeta potential modulated nanoparticles were prepared by incubating FZNP with fucoidan. The optimized zeta potential modulating nanoparticles displayed a mean particle size of 227.10 ± 1.54 nm, a PDI of 0.26 ± 0.01 and a zeta potential of −26.75 ± 0.3 mV. The physicochemical characterization proved the encapsulation of FZN in the nanoparticles and that the coating of fucoidan resulted in zeta potential modified nanoparticles that can cross the mucus barrier and prevent back diffusion. The entrapment efficiency of FZNP and Fu-FZNP was 65.47% ± 2.32% and 71.13% ± 5.74%, with 11.69% ± 0.414% (FZNP) and 7.41% ± 0.60% (Fu-FZNP) drug loading. The spherical morphologies of the nanoparticles were confirmed by TEM imaging. The amount of drug permeated through the excised goat vagina was found to be 61.74% ± 2.07% and 72.11% ± 1.4% for FZNP and Fu-FZNP, respectively. The in vitro antimicrobial activity of FZNP and Fu-FZNP showed antibacterial action against Staphylococcus aureus and antifungal activity against Candida albicans species. These results indicate that FZN and Fu-FZNP lecithin–chitosan nanoparticles could be further developed for safe, convenient, and effective treatment of mixed vaginal infections.Conflicts of interest
The authors report no conflict of interest related to the manuscript.Data availability
Data will be available upon request.Acknowledgements
The authors would like to thank all individuals who contributed to this work. Their support and insights were invaluable to the completion of this study.References
- J. D. Sobel, C. Subramanian, B. Foxman, M. Fairfax and S. E. Gygax, Mixed vaginitis - More than coinfection and with therapeutic implications, Curr. Infect. Dis. Rep., 2013, 15(2), 104–108, DOI:10.1007/S11908-013-0325-5/METRICS.
- V. Goel, P. Bhalla, A. Sharma and Y. M. Mala, Lower genital tract infections in HIV-seropositive women in India, Indian J. Sex. Transm. Dis. AIDS, 2011, 32(2), 103–107 CrossRef PubMed.
- J. Bornstein and D. Zarfati, A Universal Combination Treatment for Vaginitis, Gynecol. Obstet. Invest., 2008, 65(3), 195–200, DOI:10.1159/000111946.
- M. Sanguinetti, E. Cantón, R. Torelli, F. Tumietto, A. Espinel-Ingroff and B. Posteraro, In vitro activity of fenticonazole against Candida and bacterial vaginitis isolates determined by mono- Or dual-species testing assays, Antimicrob. Agents Chemother., 2019, 63(7), 10–128, DOI:10.1128/AAC.02693-18.
- R. Albash, A. M. Al-Mahallawi, M. Hassan and A. A. Alaa-Eldin, Development and Optimization of Terpene-Enriched Vesicles (Terpesomes) for Effective Ocular Delivery of Fenticonazole Nitrate: In vitro Characterization and in vivo Assessment, Int. J. Nanomed., 2021, 16, 609–621, DOI:10.2147/IJN.S274290.
- S. Veraldi and R. Milani, Topical fenticonazole in dermatology and gynaecology: Current role in therapy, Drugs, 2008, 68(15), 2183–2194, DOI:10.2165/00003495-200868150-00007.
- A. L. Costa, A. Valenti and M. Veronese, Study of the Morphofunctional Alterations Produced by Fenticonazole on Strains of Candida albicans, using the Scanning Electron Microscope (S. E. M.), Mycoses, 1984, 27(1), 29–35, DOI:10.1111/J.1439-0507.1984.TB01979.X.
- A. Costa, M. Veronese, P. Puggeri and A. Valenti, Ultrastructural findings of Candida albicans blastoconidia submitted to the action of fenticonazole, Arzneimittelforschung, 1989, 39(2), 230–233 CAS . Accessed: Jun. 24, 2025. [Online]. Available: https://europepmc.org/article/med/2659001.
- J. Fernández-Alba, et al., Fenticonazole Nitrate for Treatment of Vulvovaginitis: Efficacy, Safety, and Tolerability of 1-Gram Ovules, Administered as Ultra-Short 2-Day Regimen, J. Chemother., 2004, 16(2), 179–186, DOI:10.1179/JOC.2004.16.2.179.
- F. Gorlero, A. Sartani, C. I. Cordaro, D. Bertin, C. Reschiotto and L. De Cecco, Fenticonazole tissue levels after the application of 3 different dosage forms of vaginal ovules, Int. J. Clin. Pharmacol. Ther. Toxicol., 1988, 26(10), 479–481 CAS.
- R. Albash, C. Yousry, A. M. Al-Mahallawi and A. A. Alaa-Eldin, Utilization of PEGylated cerosomes for effective topical delivery of fenticonazole nitrate: in vitro characterization, statistical optimization, and in vivo assessment, Drug Delivery, 2021, 28(1), 1–9, DOI:10.1080/10717544.2020.1859000.
- S. Veraldi and R. Milani, Topical fenticonazole in dermatology and gynaecology: Current role in therapy, Drugs, 2008, 68(15), 2183–2194, DOI:10.2165/00003495-200868150-00007.
- S. Srikrishna and L. Cardozo, The vagina as a route for drug delivery: A review, Int. Urogynecol. J. Pelvic. Floor Dysfunct., 2013, 24(4), 537–543, DOI:10.1007/S00192-012-2009-3.
- C. M. Caramella, S. Rossi, F. Ferrari, M. C. Bonferoni and G. Sandri, Mucoadhesive and thermogelling systems for vaginal drug delivery, Adv. Drug Delivery Rev., 2015, 92, 39–52, DOI:10.1016/j.addr.2015.02.001.
- R. Palmeira-de-Oliveira, A. Palmeira-de-Oliveira and J. Martinez-de-Oliveira, New strategies for local treatment of vaginal infections, Adv. Drug Delivery Rev., 2015, 92, 105–122, DOI:10.1016/j.addr.2015.06.008.
- J. das Neves, R. Nunes, A. Machado and B. Sarmento, Polymer-based nanocarriers for vaginal drug delivery, Adv. Drug Delivery Rev., 2015, 92, 53–70, DOI:10.1016/j.addr.2014.12.004.
- G. Chindamo, S. Sapino, E. Peira, D. Chirio and M. Gallarate, Recent advances in nanosystems and strategies for vaginal delivery of antimicrobials, Nanomaterials, 2021, 11(2), 1–29, DOI:10.3390/NANO11020311.
- B. Valamla, et al., Engineering drug delivery systems to overcome the vaginal mucosal barrier: Current understanding and research agenda of mucoadhesive formulations of vaginal delivery, J. Drug Delivery Sci. Technol, 2022, 70, 103162, DOI:10.1016/J.JDDST.2022.103162.
- J. das Neves, M. Amiji and B. Sarmento, Mucoadhesive nanosystems for vaginal microbicide development: Friend or foe?, Wiley Interdiscip. Rev.: Nanomed. Nanobiotechnol., 2011, 3(4), 389–399, DOI:10.1002/WNAN.144.
- R. Bansil and B. S. Turner, Mucin structure, aggregation, physiological functions and biomedical applications, Curr. Opin. Colloid Interface Sci., 2006, 11(2–3), 164–170, DOI:10.1016/J.COCIS.2005.11.001.
- K. Netsomboon and A. Bernkop-Schnürch, Mucoadhesive vs. mucopenetrating particulate drug delivery, Eur. J. Pharm. Biopharm., 2016, 98, 76–89, DOI:10.1016/J.EJPB.2015.11.003.
- B. Le-Vinh, N. M. N. Le, I. Nazir, B. Matuszczak and A. Bernkop-Schnürch, Chitosan based micelle with zeta potential changing property for effective mucosal drug delivery, Int. J. Biol. Macromol., 2019, 133, 647–655, DOI:10.1016/J.IJBIOMAC.2019.04.081.
- L. M. Hemmingsen, N. Škalko-Basnet and M. W. Jøraholmen, The expanded role of chitosan in localized antimicrobial therapy, Mar. Drugs, 2021, 19(12), 697, DOI:10.3390/MD19120697.
- K. A. Janes, P. Calvo and M. J. Alonso, Polysaccharide colloidal particles as delivery systems for macromolecules, Adv. Drug Delivery Rev., 2001, 47(1), 83–97, DOI:10.1016/S0169-409X(00)00123-X.
- S. A. Agnihotri, N. N. Mallikarjuna and T. M. Aminabhavi, Recent advances on chitosan-based micro- and nanoparticles in drug delivery, J. Controlled Release, 2004, 100(1), 5–28, DOI:10.1016/J.JCONREL.2004.08.010.
- Stabilization of colloidal systems by the formation of ionic lipid-polysaccharide complexes, May 1996.
- S. Batzri and E. D. Korn, Single bilayer liposomes prepared without sonication, Biochim. Biophys. Acta, Biomembr., 1973, 298(4), 1015–1019, DOI:10.1016/0005-2736(73)90408-2.
- Liposome Drug Delivery Systems - Guru V. Betageri, Scott Allen Jenkins, Daniel Parsons - Google Books. Accessed: Jun. 27, 2025. [Online]. Available: https://books.google.co.in/books?hl=en&lr=&id=RI8sEQAAQBAJ&oi=fnd&pg=PP1&dq=G.+V.+Betageri,+S.+A.+Jenkins,+and+D.+L.+Parsons,+Liposome+drug+delivery+systems.+Technomic+Pub,+1993.+Accessed:+Mar.+07,+2024.+&per;5BOnline&per;5D. +Available: https://www.routledge.com/Liposome-Drug-Delivery-Systems/Betageri-Jenkins-Parsons/p/book/9781566760300&ots=0pe8hiVm4f&sig=XJJljDOyh35YwuaoqGbRGIbumNE&redir_esc=y&hash;v=onepage&q&f=false.
- H. Takeuchi, Y. Matsui, H. Yamamoto and Y. Kawashima, Mucoadhesive properties of carbopol or chitosan-coated liposomes and their effectiveness in the oral administration of calcitonin to rats, J. Controlled Release, 2003, 86(2–3), 235–242, DOI:10.1016/S0168-3659(02)00411-X.
- I. Henriksen, S. R. Vågen, S. A. Sande, G. Smistad and J. Karlsen, Interactions between liposomes and chitosan II: Effect of selected parameters on aggregation and leakage, Int. J. Pharm., 1997, 146(2), 193–203, DOI:10.1016/S0378-5173(96)04801-6.
- I. Henriksen, G. Smistad and J. Karlsen, Interactions between liposomes and chitosan, Int. J. Pharm., 1994, 101(3), 227–236, DOI:10.1016/0378-5173(94)90218-6.
- E. A. Ho, V. Vassileva, C. Allen and M. Piquette-Miller, In vitro and in vivo characterization of a novel biocompatible polymer–lipid implant system for the sustained delivery of paclitaxel, J. Controlled Release, 2005, 104(1), 181–191, DOI:10.1016/J.JCONREL.2005.02.008.
- J. Grant, M. Blicker, M. Piquette-Miller and C. Allen, Hybrid films from blends of chitosan and egg phosphatidylcholine for localized delivery of paclitaxel, J. Pharm. Sci., 2005, 94(7), 1512–1527, DOI:10.1002/JPS.20379.
- F. Sonvico, et al., Formation of self-organized nanoparticles by lecithin/chitosan ionic interaction, Int. J. Pharm., 2006, 324(1), 67–73, DOI:10.1016/J.IJPHARM.2006.06.036.
- I. A. Walbi, et al., Development of a Curcumin-Loaded Lecithin/Chitosan Nanoparticle Utilizing a Box-Behnken Design of Experiment: Formulation Design and Influence of Process Parameters, Polymers, 2022, 14(18), 3758, DOI:10.3390/POLYM14183758.
- L. M. Hemmingsen, V. Panzacchi, L. M. Kangu, B. Giordani, B. Luppi and N. Škalko-Basnet, Lecithin and Chitosan as Building Blocks in Anti-Candida Clotrimazole Nanoparticles, Pharmaceuticals, 2023, 16(6), 790, DOI:10.3390/PH16060790.
- A. Hafner, J. Lovrić, D. Voinovich and J. Filipović-Grčić, Melatonin-loaded lecithin/chitosan nanoparticles: Physicochemical characterisation and permeability through Caco-2 cell monolayers, Int. J. Pharm., 2009, 381(2), 205–213, DOI:10.1016/J.IJPHARM.2009.07.001.
- Q. Tan, W. Liu, C. Guo and G. Zhai, Preparation and evaluation of quercetin-loaded lecithin-chitosan nanoparticles for topical delivery, Int. J. Nanomed, 2011, 10, 1621–1630 Search PubMed.
- I. Özcan, E. Azizoǧlu, T. Senyiǧit, M. Özyazici and Ö. Özer, Enhanced dermal delivery of diflucortolone valerate using lecithin/chitosan nanoparticles: in vitro and in vivo evaluations, Int. J. Nanomed., 2013, 8, 461–475, DOI:10.2147/IJN.S40519.
- M. Saha, et al., QbD based development of resveratrol-loaded mucoadhesive lecithin/chitosan nanoparticles for prolonged ocular drug delivery, J. Drug Delivery Sci. Technol., 2021, 63, 102480, DOI:10.1016/J.JDDST.2021.102480.
- A. Sosnik, J. Das Neves and B. Sarmento, Mucoadhesive polymers in the design of nano-drug delivery systems for administration by non-parenteral routes: A review, Prog. Polym. Sci., 2014, 39(12), 2030–2075, DOI:10.1016/J.PROGPOLYMSCI.2014.07.010.
- K. Netsomboon and A. Bernkop-Schnürch, Mucoadhesive vs. mucopenetrating particulate drug delivery, Eur. J. Pharm. Biopharm., 2016, 98, 76–89, DOI:10.1016/J.EJPB.2015.11.003.
- T. M. M. Ways, et al., Mucus-penetrating nanoparticles based on chitosan grafted with various non-ionic polymers: Synthesis, structural characterisation and diffusion studies, J. Colloid Interface Sci., 2022, 626, 251–264, DOI:10.1016/J.JCIS.2022.06.126.
- N. Rajana, et al., Targeted delivery and apoptosis induction of CDK-4/6 inhibitor loaded folic acid decorated lipid-polymer hybrid nanoparticles in breast cancer cells, Int. J. Pharm., 2024, 651, 123787, DOI:10.1016/J.IJPHARM.2024.123787.
- N. Rajana, et al., Fabrication and characterization of teriflunomide-loaded chondroitin sulfate hybridized zein nanoparticles for the management of triple negative breast cancer, Int. J. Biol. Macromol., 2025, 300, 140316, DOI:10.1016/J.IJBIOMAC.2025.140316.
- Y. S. Pooja, N. Rajana, R. Yadav, L. T. Naraharisetti, C. Godugu and N. K. Mehra, Design, development, and evaluation of CDK-4/6 inhibitor loaded 4-carboxy phenyl boronic acid conjugated pH-sensitive chitosan lecithin nanoparticles in the management of breast cancer, Int. J. Biol. Macromol., 2024, 258, 128821, DOI:10.1016/J.IJBIOMAC.2023.128821.
- P. S. Chary, et al., Enhancing breast cancer treatment: Comprehensive study of gefitinib-loaded poloxamer 407/TPGS mixed micelles through design, development, in-silico modelling, In vitro testing, and Ex vivo characterization, Int. J. Pharm., 2024, 657, 124109, DOI:10.1016/J.IJPHARM.2024.124109.
- S. Shaikh, P. S. Chary, O. Khan and N. K. Mehra, Development and evaluation of nilotinib-loaded Bovine Serum Albumin nanoparticles: In vitro and in-silico insights, Int. J. Biol. Macromol., 2025, 308, 142185, DOI:10.1016/J.IJBIOMAC.2025.142185.
- A. Mammella, V. Bhavana, P. S. Chary, U. Anuradha and N. K. Mehra, Modulation of chondroprotective hyaluronic acid and poloxamer gel with Ketoprofen loaded transethosomes: Quality by design-based optimization, characterization, and preclinical investigations in osteoarthritis, Int. J. Biol. Macromol., 2024, 280, 135919, DOI:10.1016/J.IJBIOMAC.2024.135919.
- N. U. Kumari, P. S. Chary, E. Pardhi and N. K. Mehra, Tailoring micellar nanocarriers for pemetrexed in breast cancer: design, fabrication and in vitro evaluation, Nanomedicine, 2024, 19, 1145–1166, DOI:10.2217/NNM-2024-0013.
- P. S. Chary, et al., Enhancing breast cancer treatment: Comprehensive study of gefitinib-loaded poloxamer 407/TPGS mixed micelles through design, development, in-silico modelling, In vitro testing, and Ex vivo characterization, Int. J. Pharm., 2024, 657, 124109, DOI:10.1016/J.IJPHARM.2024.124109.
- W. Zhou, et al., Mucus-penetrating dendritic mesoporous silica nanoparticle loading drug nanocrystal clusters to enhance permeation and intestinal absorption, Biomater. Sci., 2023, 11(3), 1013–1030, 10.1039/D2BM01404A.
- R. Albash, M. A. El-Nabarawi, H. Refai and A. A. Abdelbary, Tailoring of PEGylated bilosomes for promoting the transdermal delivery of olmesartan medoxomil: in vitro characterization, ex vivo permeation and in vivo assessment, Int. J. Nanomed., 2019, 14, 6555–6574, DOI:10.2147/IJN.S213613.
| This journal is © The Royal Society of Chemistry 2026 |






