Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Advancing sustainability through digitalization: from drug development to patient care
Remo
Eugster
 and
Paola
Luciani
and
Paola
Luciani
 *
*
Department of Chemistry, Biochemistry and Pharmaceutical Sciences, University of Bern, Bern, Switzerland. E-mail: paola.luciani@unibe.ch
First published on 19th February 2026
Abstract
Digitalization is transforming the pharmaceutical industry, offering innovative solutions to enhance sustainability across the pharmaceutical value chain. From drug discovery to patient care, digital tools such as artificial intelligence (AI), machine learning (ML), blockchain, and digital twins optimize processes, reduce resource use, and mitigate environmental and social impacts. In drug discovery, AI accelerates and optimizes research while green chemistry initiatives prevent toxic waste. During manufacturing, predictive maintenance and quality control boost efficiency, and digital tools ensure secure, traceable distribution and enhance supply chain transparency. Within clinical care, AI-driven personalized medicine improves patient outcomes and reduces inefficiencies, while digital health records, diagnostics, and therapies promote healthcare equity and expand access. Collectively, these innovations align the industry with global sustainability goals, advancing a pharmaceutical ecosystem that is more efficient, environmentally responsible, and socially just. Yet challenges in energy demand, data equity, and regulatory adaptation must be addressed to tap into the full potential of a digital transformation.
1. Introduction
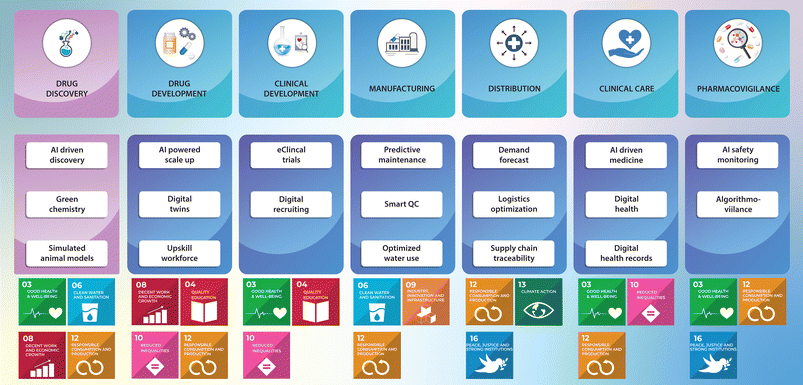
Sustainability has emerged as a critical priority across industries, with the pharmaceutical sector being no exception.1–3 As environmental concerns grow, the pharmaceutical industry faces unique challenges, from high energy use and resource consumption to toxic waste generation and the environmental impact of drug disposal.4 With increasing pressure to meet the United Nations’ Sustainable Development Goals (SDGs) and a growing focus on the Environmental, Social, and Governance (ESG) framework, pharmaceutical companies are rethinking their strategies and exploring new ways to minimize their ecological footprint and enhance their social impact.2,5 In this context, sustainability encompasses not only environmental stewardship but also economic viability and social equity. Digitalization, the integration of digital technologies to transform processes, offers significant potential in this regard, promising improvements in efficiency, cost reduction, and reduced resource consumption across the entire value chain of drug development, manufacturing, and patient care.6 A single digitalization effort can often address several SDGs at once, while also presenting new challenges.6Pharmaceutical digitalization encompasses various technologies, such as artificial intelligence (AI), machine learning (ML), blockchain, digital twins, and automation, among others.7 However, digitalization is not just about data handling, but also re-shapes chemical processes themselves, such as reaction design or solvent choice.8,9 Hence, these tools have a profound impact, driving sustainability through optimizing processes, minimizing waste, and enhancing environmental and social stewardship.7 Consequently, this review aims to first introduce and contextualize key digital concepts for a broad audience in the pharmaceutical sciences. Building on this foundation, we then provide tangible examples and evidence of how these specific tools are currently advancing established sustainability goals across the entire value chain – from drug development to clinical care and pharmacovigilance – creating a more environmentally friendly, efficient, and equitable pharmaceutical ecosystem (Fig. 1). The primary focus will be on the journey from drug development onward, while readers are referred to other recent publications for more detailed discussions on digitalization in drug discovery.10–15 Beyond showcasing opportunities, this article critically examines challenges such as energy demand, algorithmic bias, regulatory lag, and digital inequities, which together determine whether digitalization in pharma can truly advance sustainable development.
2. From drug development to e-development
2.1. Digitalization for accelerated and green drug discovery
Traditionally, drug discovery is time-consuming and resource-intensive. Digital technologies are transforming this process through computational methods such as ML, quantum computing, and in silico modelling.16,17 Specifically, the use of these computational tools, such as Virtual Screening, accelerates candidate identification while significantly reducing the need for physical screenings. This acceleration is substantial: bioinformatics has the potential to reduce the time from target identification to preclinical trials from 6 years to 3–4.5 years, dramatically speeding up the overall discovery timeline while saving resources.15,18,19 Furthermore, AI-driven approaches are showing promising early results, with some analyses indicating they achieve higher clinical success rates compared to conventionally discovered drugs.18 While data for phase III are limited, AI-discovered molecules tend to perform twice as well in phase I reaching success rates of 80%–90%, aligning with traditional success rates from phase II onward.18 These advances contribute to SDG 3 (Good Health and Well-being) by speeding drug discovery, SDG 8 (Decent Work and Economic Growth) by improving efficiency, and SDG 12 (Responsible Consumption and Production) by lowering chemical use and minimizing lab waste.Digitalization also advances green chemistry by identifying reaction pathways that minimize energy use and toxic waste.10 For example, Kumar et al. have demonstrated how bioinformatics can be leveraged to optimize enzymes and achieve 3-fold improvements.14 Computational optimization aligns discovery with sustainability goals by reducing environmental impacts, with some deep learning tools needing as little as 10 min and 25 Wh power on a consumer laptop to train and predict a candidate,20 directly supporting SDG 12. ML models further assess environmental impact early, evaluating biodegradability and predicting ecological consequences to enable preventive action.21,22 embedding sustainability into drug discovery. For example, Gbadago et al. developed a model, enhanced by reinforcement learning, to predict better and discover new biodegradable materials.22 Said model was able to generate biodegradable materials, with a 95% success rate and achieving an 87% area under the curve for the receiver operating characteristic, a key metric for evaluating sensitivity and failure rates.22
Challenges remain: SDG 5 (Gender Equality) could be undermined if training datasets carry historical biases,23,24 and AI tools risk misuse in designing toxic compounds.25 For one, gender bias in big data can originate from various sources, including biases in data gathering, treatment, or model training, according to statements by the United Nations.24 This issue is exacerbated by a lack of diversity in the workforce developing these tools; only about 6% of software developers at machine learning companies and 18% of authors of AI content are female, adding to the issue of historical biases in training datasets.26 This issue is reflected in the outcomes of the technology itself: gender biases appear in approximately 20% of instances in large language model-based approaches alone, according to UNESCO.26 Further, the dual use of AI-powered drug discovery models presents a significant risk: they have demonstrated the capability to design and predict new compounds potentially more toxic than current nerve agents, solely based on public databases and without being trained on any war agents.25 Moreover, the environmental burden shifts from experiments to the significant energy demands of large-scale computation.27 Nonetheless, the benefits outweigh the risks, as AI-driven approaches already show higher clinical success rates.18,27
2.2. From eDevelopment to eClinical trials
Beyond the initial screening of drug candidates, digital technologies are now being applied throughout the entire drug development journey, leading to more sustainable and efficient processes. ML models, for example, can simulate animal models or predict toxicity, significantly reducing the number of in vivo tests required.28,29 Zhu et al. have used computational tools to predict in vitro and in vivo behavior of amorphous solid dispersions and thereby optimizing drug delivery systems.30 This not only cuts down on costs and time but also addresses ethical concerns related to animal welfare, contributing to SDG 10 (Reduced Inequalities).28 Similarly, computational modeling and AI are being used to optimize manufacturing scale-up, predicting the behavior of a drug substance during production and helping to identify the most effective excipients.31,32 For instance, our laboratory leveraged molecular dynamics (MD) simulations to model and simulate liposomal depot formation and drug release within a physiological context, specifically simulating the subcutaneous space.33 This in silico analysis was completed within approximately four weeks of simulation time, compared to months of work in the lab traditionally.33 This approach provided a clear, proven understanding of system functionality in silico, which de-risked the subsequent physical testing.33 By validating the system virtually, we were able to conduct effective in vitro and in vivo tests and successfully advance the system through different species in vivo with increased confidence and efficiency.33 Further, we leveraged AI to predict critical quality attributes and process parameters during microfluidic production of liposomes to accelerate scale-up.31 Also, via explainable AI, we explored lipid behavior and effects of process parameters on microfluidic liposome production.31 These examples minimize material waste and the number of physical trials needed to find the proper formulation, reinforcing SDG 12 (Responsible Consumption and Production).2,31,32The application of digital tools extends even to clinical trials. ML models can simulate patient responses, helping to identify the most suitable patient cohorts and predict the efficacy and safety of a drug, thereby streamlining trial design.34–36 Patient recruiting is often the most time-consuming and labor-intensive part of a clinical trial, with up to 20% of trials failing to enroll a sufficient number of participants.36 This challenge presents a significant opportunity for acceleration using digital tools. Digital solutions, such as the Pathfinder tool, can instantly adjust participation criteria without affecting the clinical trial's hazard ratio.36 A case study demonstrated the power of such tools by showing their capability to double the number of eligible patients for lung cancer trials, among others, without increasing the hazard ratio.36 On the other hand, digital twins of patients – digitally cloning a patient – are capable of predicting how the same patient would have progressed in the control group and have the potential to reduce said patient groups by 20%–50%.36 In addition, such tools also benefit patients enrolled in trials, as they have a higher chance of receiving the new treatment rather than the placebo.36 These are powerful drivers for SDG 3 (Good Health and Well-being). A crucial element of this digital shift is the necessity for validation and a constant supply of high-quality, reliable data to train these models.36 Without robust data and rigorous validation of the models, the insights generated are unreliable, which could compromise both patient safety and the efficiency gains sought.36,37 However, the ethical aspects of patient data used from databases also need to be addressed.38 For example, issues with the sharing of de-identified data have been highlighted, which calls for stronger data access committees.38
The digitalization across the pharma value chain also has a social impact on the pharmaceutical workforce.7 As computational methods become more central to the process, there is a growing need for new skills in data science, bioinformatics, and AI.16 This is especially pressing with the AI market projected to rise by 26% over the next 5 years and 31% of R&D workforce nearing retirement, as shown by Kearney.16 Companies are using digital training tools to upskill existing employees and address the potential for job displacement due to automation, ensuring that the workforce can adapt to this new paradigm and continue to contribute to innovation.7,16 This is a direct contribution to SDG 4 (Quality Education) and SDG 8 (Decent Work and Economic Growth).
3. Manufacturing
3.1. Pharma 4.0: the digital revolution in production
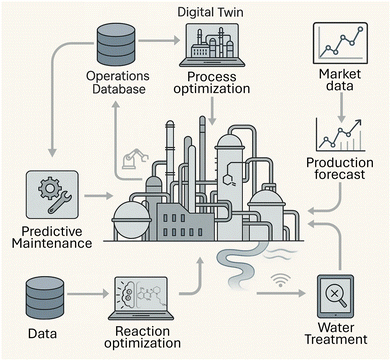
The pharmaceutical industry's production processes are undergoing a revolution with the advent of Industry 4.0, often referred to as Pharma 4.0.39,40 This transition is marked by the adoption of automation, data exchange, and smart manufacturing technologies.41 AI-driven process optimization enables companies to identify bottlenecks and inefficiencies in production, optimizing resource utilization and reducing environmental waste as illustrated in Fig. 2.39–41 For instance, chemical reactions can be optimized rapidly through automation and machine intelligence, saving precious resources, as demonstrated by the Schwaller Lab.8,42 Here, Vaucher et al. introduce a data driven model predicting entire sequences of synthesis steps, for application in batch organic chemistry, adequate for direct execution by either a human or automation hardware in more than 50% of the cases.42 This progress is a clear example of SDG 9 (Industry, Innovation, and Infrastructure) and SDG 12 (Responsible Consumption and Production) in action.Another key advantage of digitalizing pharmaceutical production is predictive maintenance.40 Maintenance costs typically range from 15% to 60% of total operating costs in manufacturing, offering a significant opportunity for savings when handled efficiently.43 AI tools can provide early warnings of equipment deterioration, helping manufacturers anticipate repair needs and prevent unexpected downtime.40,43,44 According to industry reports, predictive maintenance can prevent approximately 42% of production line errors, resulting in significant reduction of production waste.43 Thereby, it enhances line availability by up to 15% and results in a reduction of maintenance costs of almost 25% due to service activities being aligned with actual equipment needs.43 This not only saves energy but also reduces material waste and improves production efficiency. This also improves production efficiency but also allows workforce to plan interventions better.44
3.2. Quality control and digital twins
Quality control is another area where digitalization is making a significant impact.40,44 Historically, quality assurance of drug products relied on manual inspections, which suffered from drawbacks related to inconsistencies, cost, quality, and objectivity. Automated and AI-optimized monitoring systems offer particularly promising opportunities for improvement in pharmaceutical sciences.44,45 To address these challenges, the industry has applied digital tools such as deep learning models and image segmentation to analyze product videos in real-time digitally.44 These systems enable consistent product quality while reducing human error. In reported use cases, these digital systems are capable of inspecting 1000 capsules per minute with a remarkable 95% accuracy.44 This real-time monitoring identifies potential issues and improves batch standardization.44 Digital twins, virtual replicas of physical production processes, are emerging as powerful tools for sustainability.46,47 By simulating and optimizing manufacturing processes, digital twins allow companies to test different scenarios, optimize energy use, and minimize material waste without disrupting real-world operations.46,47 A digital twin is a virtual replica (or model) of a physical production process, equipment, or system that is continually fed real-time data from its physical counterpart.47 By simulating and optimizing manufacturing processes, digital twins allow companies to test different scenarios, optimize energy use, and minimize material waste without disrupting real-world operations.47 This aligns with both SDG 9 (Industry, Innovation, and Infrastructure) and SDG 12 (Responsible Consumption and Production). In vaccine production, for instance, digital twins have helped to identify bottlenecks and optimize the use of raw materials, ultimately reducing the overall environmental footprint of manufacturing.48 The application of a digital twin in such complex operations has, in fact, been shown to lead to 20% additional productivity gains at a 99.9% reliability rate.48 This showcases the immediate, tangible benefits of using virtual replicas to enhance efficiency and resource stewardship.48 However, it needs to be pointed out that a major barrier to sustainable digital transformation remains regulatory adaptation: while digital twins and AI-assisted quality control show clear potential, their integration into Good Manufacturing Practice (GMP) frameworks is still limited.493.3. Water stewardship
Digitalization is also critical for managing water, a decisive resource in pharmaceutical manufacturing.50 Chemical production steps are often water-intensive, yet only 42% of wastewater is currently treated and reused, making it crucial to reuse specific wastewater streams and efficiently recover solvents.51,52 Further, IoT (Internet of Things) sensors and digital twins can monitor and optimize water usage in real-time, identifying inefficiencies in cleaning, cooling, and solvent recovery systems.52 The IoT forms a network of physical devices and objects equipped with sensors and software that interact and perform tasks on the wastewater based on data-driven decisions.52 Optimization based on such tools has demonstrated the capability to save 4%–37% of energy while doubling recovery yield.50,52 Hence, this data-driven approach helps manufacturers reduce their water consumption and improve overall water stewardship, directly supporting SDG 6 (Clean Water and Sanitation).50,524. Distribution
4.1. Digital supply chains & circular economy
Pharmaceutical supply chains are notoriously complex, and digital tools are now being leveraged to improve sustainability in this domain. AI and ML models can predict fluctuations in demand with greater accuracy, reducing overproduction and minimizing transportation waste.44 Predictive analytics allow for better resource planning, which in turn decreases energy consumption and material use.44,53,54 These efforts contribute to SDG 12 (Responsible Consumption and Production). Further, automated dispensing systems in pharmacies optimize stock levels, reducing expired medications and associated waste.55The concepts of waste reduction and traceability in the supply chain are central to the Circular Economy. In the context of sustainability, the Circular Economy is an alternative to the traditional linear ‘take-make-dispose’ model. Instead, it aims to keep resources in use for as long as possible, recovering and regenerating products and materials at the end of each service life.56 Digitalization, with technologies like blockchain, facilitates this model by enabling reverse logistics, take-back programs for medical devices, and the tracking of material life cycles.54 Blockchain technology, which is essentially a secure, decentralized digital ledger, allows for transparent and tamper-proof tracking of goods.54 This is particularly critical for enhancing supply chain traceability.54 For example, blockchain can track products from production to distribution and disposal, helping to reduce fraud, waste, and counterfeit drugs while supporting sustainable recycling practices.54 However, pharmaceutical supply chains lag behind other industries in terms of performance across service, cost, and capital efficiency.57 This inefficiency is quantifiable: the pharma sector is estimated to use only half its capacity compared to consumer goods and has 13 times longer lead times, requiring it to rely on four times larger inventory.57 These inefficiencies make the supply chain a resource-intensive step due to labor, energy used for transport, and temperature-controlled storage. Digital technologies are increasingly being applied to enhance sustainability and operational efficiency across the pharmaceutical value chain. For example, artificial intelligence can support sustainable procurement by identifying suppliers with lower carbon footprints, which has been shown to reduce procurement-related emissions by up to 20%58 In logistics, machine learning–based route optimization contributes to more efficient last-mile delivery, decreasing annual fuel consumption by approximately 18%.58 Similarly, data-driven inventory management models that forecast material and product demand have demonstrated up to a 10% reduction in stock imbalances, thereby minimizing waste and improving resource utilization.59
Industry leaders in supply chain excellence suggest that by implementing digital technology and adopting best practices from other sectors, pharma companies can significantly improve their supply chains, delivering an improved customer experience and patient promise.57,60 It is estimated that agent-based AI could help with 75%–85% of workflows, reducing the time required for key tasks in the supply chain by 25%–35%.61 The overall financial potential in pharma supply chains is substantial: a potential rise in margins of 15% and a reduction in working capital by more than 25%.57 It must be noted that this effect and potential are approximately eight times more pronounced in generics compared to branded drugs, as production and R&D costs are smaller in relation to distribution within the generic market.60
Besides ensuring the product arrives at the patient, such tools can help track packaging material, active pharmaceutical ingredients (APIs), solvents, and excipients, ensuring they are disposed of or appropriately recovered. From a chemical perspective, digital traceability can support solvent recovery, polymer recycling, and API degradation studies, thereby integrating green chemistry principles directly into supply-chain management. For medical devices and packaging, digitalization is key to unlocking the growing potential for recycling. This is a critical gap to address, as currently only 5% of medical devices are recycled, despite many being designed for reusability.62 The increasing use of sophisticated, digitally tracked medical devices necessitates robust take-back and recycling programs.62 By tracking these complex products from production to disposal, companies can identify opportunities for reuse, refurbishment, and recycling, thereby moving beyond the current low recovery rates for mixed-material packaging.62 This is crucial given that the pharmaceutical industry generates approximately 300 million tons of plastic waste each year, and that digital oversight is required to unlock the potential for high-value materials, such as enabling techniques that can potentially achieve up to 80% recovery of components in difficult-to-recycle products.53,54,63 This is another direct contribution to SDG 12 (Responsible Consumption and Production).
Furthermore, digitalization is crucial for tackling Scope 3 emissions – the indirect emissions from the supply chain, which can account for a significant portion of a company's total carbon footprint.64 AI-driven logistics optimization is vital for reducing these emissions by finding more energy-efficient transportation routes and minimizing the need for unnecessary shipments.53,54,65 In addition, chemistry-driven digital models (e.g., life-cycle assessments of polymeric packaging or solvent inventories) can help quantify embedded carbon in materials, linking chemical processes to broader emission-reduction strategies. These measures directly support SDG 13 (Climate Action).
4.2. Governance and regulatory compliance
Despite rapid progress in drug development, the biopharmaceutical industry has lagged in digitalizing regulatory submissions, where manual processes create inefficiencies, errors, and delays in patient access.66 Digital solutions such as SCDM, AI, cloud platforms, and standardizing data formats have the potential to streamline submissions, enhance collaboration, and accelerate approvals while reducing waste – ultimately ensuring faster patient access to new therapies.66 The emergence of agentic AI, which shifts the technology's role from a mere tool to a collaborative coworker, offers significant benefits for complex functions such as regulatory affairs and compliance. Early industry analyses suggest that agentic AI systems could potentially reduce workloads in medical and regulatory affairs by approximately 23–38%; however, these values are projections based on modelling and expert assessment, and robust empirical validation at scale is still pending.61 Harmonized regulatory guidance, such as the European Medicines Agency's reflection papers on AI in drug development, will be essential to ensure both safety and sustainability without stifling innovation.49 Transparent reporting standards and validation protocols for digital tools could help build trust among regulators, industry, and patients alike, thereby accelerating responsible adoption.49 Further, incorporating blockchain technology or other digital tools into supply chains enhances transparency and traceability.67–69 By tracking products from production to distribution, tracking tools help reduce fraud, waste, and counterfeit drugs, while supporting sustainable sourcing practices.53,67,68 This is particularly important for ensuring that raw materials are responsibly sourced and that waste is minimized throughout the supply chain.53,70 This real-time visibility also helps companies ensure compliance with regulations like Good Distribution Practice (GDP), which ensures product integrity and patient safety, aligning with SDG 16 (Peace, Justice, and Strong Institutions) among others.16,53,655. Clinical care
5.1. Patient centric medicine
In clinical care, digitalization offers solutions that enhance sustainability while improving patient outcomes. AI-driven tools for personalized medicine help optimize drug dosing, administration times, and even formulation choice (e.g., controlled-release vs. immediate-release), all of which vary with patient-specific pharmacokinetics and metabolism.71,72 Implementing variability in dosing, as shown in clinical trials, can improve efficacy in chronic conditions where resistance develops.71,72 AI systems also help identify the best treatment regimens, reducing trial-and-error approaches that waste resources.71,72 This focus on individual patient needs, known as patient centricity, is a key social pillar of sustainability, addressing SDG 3 (Good Health and Well-being), SDG 12 (Responsible Consumption and Production) through reduced waste, and SDG 10 (Reduced Inequalities) by tailoring care to diverse populations. By integrating patient-reported outcomes into the development and care process, digital tools help ensure that treatments are not only effective but also aligned with patient values and needs.5.2. Digital health, therapies, and diagnostics
The emergence of digital health tools, including therapeutics and molecular-level diagnostics, is a crucial social dimension of sustainability.73,74 Currently, more than 500 digital therapeutics (DTx) are approved globally, with roughly two-thirds classified as treating diseases (particularly neurological and ophthalmic conditions), while 29% manage diseases and 5% focus on prevention.75 Regional focuses vary significantly: China predominantly targets disease treatment (88%), particularly for neurological and ophthalmic conditions, while Western countries focus more on monitoring.75 These technologies can significantly improve access to care, particularly in low- and middle-income countries and remote areas where traditional healthcare infrastructure is limited.74,76 For example, mobile apps for disease management and smartphone-based biosensors for analytes such as glucose or lactate can overcome geographic and economic barriers.71,72 The growing shift toward preventive and personalized medicine further increases the demand for reliable diagnostics to detect disease earlier and guide therapy.77 DTx such as AI-enabled speech analysis for early signs of Alzheimer's disease, or image-based algorithms for cancer detection, exemplify how advanced diagnostics can transform care delivery.78 This expanded access helps address health inequities and promotes a more inclusive healthcare system, which is vital for achieving SDG 3 (Good Health and Well-being) and SDG 10 (Reduced Inequalities). However, the effective implementation of these solutions relies on a robust digital infrastructure.79 Unlike conventional drugs, DTx often rely on changing long-term patient behaviors, requiring substantial physician engagement.75 While insurance policies in Europe and the US are adapting to provide economic compensation, and physician acceptance is increasing, widespread adoption remains contingent on robust clinical evidence and data security.75 Furthermore, a significant risk lies in the “digital fracture” – the widening of health inequalities where internet access, reliable electricity, or affordable devices are lacking.79 Addressing this requires inclusive design and investment in infrastructure and training.5.3. Healthcare equity
The shift to digital health records and telehealth is another key aspect of sustainability in clinical care. Digital records streamline patient management, reducing duplication of diagnostic assays and chemical tests, and improving the coordination of care.80 Telehealth tools and chatbots can deliver expertise to remote locations while freeing healthcare personnel to focus on core competencies.81,82 This leads to more efficient use of resources, reduces paper waste, and lowers the environmental footprint of healthcare.82 While several initiatives are ongoing, data privacy and ownership risks remain.83 Furthermore, locations that could benefit most from telehealth tools often lack basic digital infrastructure and access.79 Strikingly, in only 11% of African countries does the whole population have access to electricity, and even in the most advanced African nation, only about one-third of the population has broadband internet.79 While comparable challenges exist in parts of South America and Asia, particularly in rural or low-income regions, Africa remains the most severely affected continent, where the potential impact of improved digital health infrastructure is correspondingly the greatest. Therefore, successful adoption requires not just the technology itself, but also significant investment in infrastructure, accessible devices, and training on these emerging technologies.79 However, such measures would directly contribute to SDG 3 (Good Health and Well-being), SDG 10 (Reduced Inequalities), and SDG 12 (Responsible Consumption and Production).5.4. Cybersecurity
To ensure that digitalization in healthcare and pharmaceutical value chains remains sustainable and trustworthy, cybersecurity must be treated as a core governance and patient-safety requirement rather than a purely technical IT function. Since, modern care delivery relies on interconnected electronic health records, networked medical devices, telehealth platforms, and cloud infrastructures, all of which expand the potential attack surface and create direct risks to patients when compromised.7 Large-scale ransomware and system outages have been shown to delay diagnostics and treatments, force diversion of emergency cases, and in extreme cases threaten life by disrupting access to records and critical devices.84 Cyber incidents therefore represent not only data-privacy breaches but also tangible clinical safety events, requiring preparedness measures such as offline downtime protocols, regular drills, segmented networks, and secure backups to maintain continuity of care.84 From a governance perspective, a patient-centric cybersecurity approach is essential, placing the protection of individual wellbeing and data integrity at the centre of regulatory, organisational, and technical decisions.85 This includes systematic risk assessments, adequate budgeting for security capabilities, staff training against phishing and social engineering, and lifecycle security management of IoT-enabled medical devices.86,87 Embedding such measures aligns digital health innovation with ethical and regulatory responsibilities, safeguarding both operational resilience as well as patient trust and safety in increasingly digital healthcare ecosystems.6. Surveillance
6.1 Pharmacovigilance and digital surveillance systems
Pharmacovigilance, or the monitoring of drug safety after they have entered the market, is essential for ensuring that drugs do not cause harm to patients or the environment.88,89 Traditional pharmacovigilance systems are often slow to detect adverse effects.90 For one, the median underreporting rate is alarmingly high, around 94%, which severely affects the ability to detect safety signals early.90 Digital tools like machine learning models are transforming this process.89,90 By analyzing large datasets from clinical systems, call centers, and online sources, AI has the potential to identify patterns and safety signals faster than human operators.88,90 These systems contribute to sustainability by reducing the need for extensive human resources in drug monitoring, enabling earlier interventions that can prevent widespread harm.90,91 In fact, industry experts estimate that safety and pharmacovigilance functions can benefit significantly from AI agents, resulting in up to 45%–50% time savings compared to conventional work practices.61 However, it needs to be pointed out that also here historical biases in data can put certain populations at risk, especially women.24 Hence it is important to be aware of sex differences, among other aspects, and incorporate them into the digital approach. Chandak et al. for example, have demonstrated this with an approach to predicting sex differences in drug response, successfully mitigating 79% of underlaying sex differences.92 Additionally, the use of AI for surveillance minimizes the environmental costs associated with drug recalls, as unsafe drugs can be identified and removed from circulation more quickly.90,91 This also reinforces the governance aspect of sustainability by ensuring companies meet their regulatory and ethical obligations for public health and safety, supporting SDG 3 (Good Health and Well-being) and SDG 12 (Responsible Consumption and Production).6.2. Algorithmovigilance: monitoring AI in healthcare
Whereas pharmacovigilance focuses on detecting, assessing, and preventing adverse effects of medicinal products, algorithmovigilance specifically addresses explicitly the risks that arise from AI-driven systems themselves, such as biased decision support or unexpected model drift in clinical practice. As AI systems become more integrated into healthcare and drug development, there is a growing need for “algorithmovigilance” – the monitoring of AI systems themselves.93,94 Just as pharmacovigilance tracks drug safety, algorithmovigilance ensures that AI tools are functioning as intended, avoiding biases or errors that could compromise patient safety or lead to inefficiencies in drug development and clinical care.93 This new layer of surveillance is essential to ensure that AI-driven tools contribute to sustainability without introducing new risks and to uphold the governance standards of the industry, contributing to both SDG 3 (Good Health and Well-being) and SDG 16 (Peace, Justice, and Strong Institutions).93,94 It needs to be pointed out however, that given that this is a nascent field, empirical data and established frameworks are currently scarce. A search of PubMed reveals only 10 publications mentioning “algorithmovigilance”, with just four focusing primarily on the topic, all from very recent years. Crucially, none of the existing research has yet delivered quantitative comparisons against the current status quo to robustly measure actual efficiencies or improvements. Therefore, future research must urgently prioritize the development of robust standards and monitoring methodologies to safeguard the integrity of digital healthcare.7. The environmental footprint of digitalization
While digitalization offers significant environmental benefits, the ecological footprint of digital tools should also be acknowledged. The immense computational power required for AI, ML, and blockchain technologies comes with a substantial environmental cost.27,95 The energy consumption of data centres, for instance, is a growing concern, with the digital sector contributing to global carbon emissions.95 This challenges progress towards SDG 7 (Affordable and Clean Energy) and SDG 13 (Climate Action). However, from a global perspective, AI's impact on future electricity demand is currently relatively small with 1%–1.3% of world demand – the challenge however lies in its concentrated energy consumption and projection to grow to 4.5% by 2028.27 Further, this high energy demand has the potential to promote the adoption of sustainable and renewable energy sources, thereby driving innovation in the energy sector.96 Nevertheless, it is important to point out that computational tools must be used in an environmentally responsible manner.97 Schilter et al. provide several best practices to strike balance between computational power and sustainability for digital tools in chemistry and pharmaceutical sciences.97 In brief, to minimize the environmental footprint of digital chemistry, researchers should adopt best practices focused on transparency and efficiency.97 Crucially, they should implement carbon tracking (e.g., via CodeCarbon) to estimate and publish emissions alongside specific hardware details, creating benchmarks for future comparison.97 For instance, the preparation of this article is estimated to have a carbon footprint of 2–5 kg CO2, despite not relying on energy-intensive computational modeling. For perspective, computational carbon footprints directly correlate with the carbon intensity of the electricity source, which varies heavily by location.98 While Norway averages 0.01 kg CO2 kWh−1, South Africa averages 1.1 kg CO2 kWh−1, making it 110 times more carbon-efficient to train computational models in Norway.98 However, while it is good practice to shift workloads to renewable energy sources, incentivizing only established green hubs conflicts directly with the pressing need for AI adaptation and infrastructure development in regions like Africa. Efficiency can be further improved by utilizing machine learning surrogate models and optimization techniques to reduce costly simulations and physical experiments, while avoiding exhaustive hyperparameter screening.97 Furthermore, adhering to FAIR principles by sharing source code prevents duplicate efforts. Notably, AI primarily yields net CO2 reductions when deployed at scale, while potentially having a negative net impact with only nominal usage.98Beyond energy, the material footprint of digitalization, stems from the entire lifecycle of electronic components. The material impact of digitalization includes the unsustainable extraction of minerals (e.g., cobalt, lithium) for technology components and the growing problem of electronic waste (e-waste), which is contrary to the spirit of SDG 12 (Responsible Consumption and Production).99 E-waste contains a complex mixture of valuable materials, including precious metals, alongside hazardous substances such as lead, mercury, cadmium, and brominated flame retardants, necessitating selective redox reactions, precipitation chemistry, and solvent extraction to separate valuable fractions from toxic residues.100 Currently, due to this complexity, only 15%–20% of e-waste is recycled globally, despite containing materials with an estimated worth of 65 billion by 2026.100 These extraction processes are often highly energy-intensive and can lead to significant environmental degradation, including habitat loss, water contamination, and soil pollution due to the use of strong acids and other chemicals in separation processes.101 The principles of sustainable photochemistry, thermochemistry, mechanochemistry, electrochemistry and sonochemistry in e-waste recycling are illustrated by Niu et al.100 For example, mechanochemical activation can enhance the leaching efficiency of precious metals without corrosive solvents, while electrochemical methods allow selective recovery of Cu, Au, and rare earth elements under milder conditions.100 However, challenges remain, particularly in preliminary sorting steps where manual methods are highly inefficient.100 By implementing algorithms and automated spectral recognition, sorting precision could potentially reach over 90%.100
The concept of “rebound effects” is also critical to consider at this point. While digitalization may make individual processes more efficient (e.g., faster drug discovery), this efficiency could lead to increased consumption or scale (e.g., more research projects being initiated), which could ultimately offset the initial environmental savings.102 For example, while large-scale in silico screening reduces the need for early-stage animal testing, the vast increase in generated drug candidates could however paradoxically raise the overall experimental burden downstream – a classic rebound effect. So far, however, given the generally positive attitude towards AI, no empirical data has yet been published on this specific matter within the pharmaceutical industry, to our knowledge. Therefore, it is crucial to analyze the net impact of digital technologies and ensure that the benefits truly outweigh the ecological costs of their implementation and operation.
8. Concluding perspectives
The digital transformation of the pharmaceutical industry holds immense potential for advancing sustainability across all phases of the drug lifecycle.7 From AI-driven drug discovery and green chemistry to predictive maintenance in production, efficient distribution, and enhanced clinical care, digital tools are driving a more sustainable, efficient, and equitable pharmaceutical ecosystem. An overview of how specific digital tools translate into measurable environmental, operational, and societal benefits across the pharmaceutical value chain, and their alignment with the UN SDGs, is summarized in Table 1. By embracing these technologies, the industry can significantly reduce its environmental impact, lower costs, and improve patient outcomes, while aligning with global sustainability goals.| Digital tool/approach | Main sustainability impact in pharma | SDGs affected |
|---|---|---|
| AI/ML-based virtual screening and in silico drug design | Reduces need for physical high-throughput screening, lowering chemical consumption, lab waste, energy use, and development time | SDG 3 (Good Health and Well-being); SDG 8 (Decent Work & Economic Growth); SDG 12 (Responsible Consumption & Production) |
| Bioinformatics and computational target identification | Shortens discovery timelines and reduces experimental resource use | SDG 3; SDG 9 (Industry, Innovation & Infrastructure); SDG 12 |
| ML-guided green reaction and enzyme optimisation | Enables higher reaction efficiency and fewer toxic reagents and by-products | SDG 9; SDG 12; SDG 13 (Climate Action) |
| Predictive ADME/PK modelling | Reduces late-stage failure and redundant animal and lab studies | SDG 3; SDG 12 |
| Molecular dynamics and digital formulation modelling | De-risks formulation development and reduces iterative wet-lab experiments | SDG 9; SDG 12 |
| Automated and AI-assisted synthesis planning | Improves yield and reduces failed or wasteful synthesis attempts | SDG 9; SDG 12 |
| Digital twins of manufacturing processes | Optimises energy and material use without interrupting production | SDG 9; SDG 12; SDG 13 |
| AI-driven predictive maintenance | Prevents equipment failure, reducing scrap, downtime, and unnecessary energy use | SDG 9; SDG 12 |
| AI-based real-time quality control (vision systems) | Minimises batch rejection and variability, lowering material and energy waste | SDG 9; SDG 12 |
| AI-optimised wastewater treatment and IoT water management | Cuts energy demand and increases water and solvent recovery | SDG 6 (Clean Water & Sanitation); SDG 12; SDG 13 |
| Digital supply-chain optimisation (procurement, routing, inventory ML) | Lowers transport emissions, overproduction, and stock wastage | SDG 9; SDG 12; SDG 13 |
| Blockchain for supply-chain traceability and circularity | Reduces counterfeits and enables take-back, reuse, and recycling of products and devices | SDG 12; SDG 16 (Peace, Justice & Strong Institutions) |
| Digital tracking for device and packaging recycling | Increases recovery of high-value materials and reduces plastic waste | SDG 12; SDG 13 |
| Decentralised/virtual clinical trials and telehealth | Reduces patient travel and site resource use while improving access to care | SDG 3; SDG 10 (Reduced Inequalities); SDG 13 |
| AI-assisted patient recruitment and eligibility matching | Shortens trial duration and reduces redundant trial activity | SDG 3; SDG 9; SDG 12 |
| Patient digital twins in trials | Potentially reduces control-group sizes and associated resource use | SDG 3; SDG 12 |
| Digital therapeutics and remote monitoring | Substitutes or complements resource-intensive in-person care | SDG 3; SDG 10; SDG 12 |
| AI-supported pharmacovigilance and signal detection | Earlier detection of adverse effects, reducing harm and inefficient drug use | SDG 3; SDG 9 |
| Federated and interoperable electronic health records | Improves data use without duplicating storage and transfers | SDG 9; SDG 12 |
| Carbon tracking and efficient ML practices in research | Makes computational emissions transparent and reduces unnecessary compute | SDG 7 (Affordable & Clean Energy); SDG 13; SDG 12 |
However, the path to a fully sustainable digital pharmaceutical industry is not without its challenges. Non-technical barriers such as the time-consuming regulatory approval process for changes to manufacturing and supply chains, cultural resistance from employees to new workflows, and the difficulty of integrating new digital solutions with legacy systems must be overcome. Adaptation is crucial, requiring not only a shift in mindset but also a rethinking of the skills and talents needed in the workforce. Digital tools must be applied where they have measurable impact, while carefully monitoring for rebound effects and unintended social and ethical consequences, such as algorithmic bias. At the same time, regulatory frameworks need to evolve to keep pace with digital innovation, ensuring both compliance and sustainability progress. Future research is needed to quantify the net environmental and social impact of digitalization, weighing its benefits against the ecological costs of the technology itself. This includes developing robust, standardized metrics and establishing new oversight frameworks for nascent fields like algorithmovigilance. By addressing these challenges and continuing to innovate responsibly, the pharmaceutical industry can harness the full potential of digitalization to build a truly sustainable future.
Author contributions
R. E.: conceptualization, investigation, visualization, writing – original draft. P. L.: conceptualization, funding acquisition, supervision, writing – review and editing.Conflicts of interest
No private study sponsors had any involvement in the study design, data collection, or interpretation of data presented in this manuscript. P. L. declares the following competing interests: she has consulted for Lipoid GmbH, Sanofi-Aventis Deutschland, and DSM Nutritional Products Ltd; received research grants on unrelated projects from Lipoid GmbH, Sanofi-Aventis Deutschland, DSM Nutritional Products Ltd, and PPM Services S.A.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
Fig. 2 was partially generated using artificial intelligence (Sora; https://openai.com/sora/ Accessed: 20.Aug.2025) and all scientific content and technical details were reviewed and validated by the authors.References
- J. Tucker and M. Faul, Industrial research: Drug companies must adopt green chemistry, Nature, 2016, 534, 27–29 CrossRef CAS PubMed.
- G. Fuhrmann, Drug delivery as a sustainable avenue to future therapies, J. Controlled Release, 2023, 354, 746–754 CrossRef CAS PubMed.
- R. G. Eccles, I. Ioannou and G. Serafeim, The impact of corporate sustainability on organizational processes and performance, Manage. Sci., 2014, 60, 2835–2857 CrossRef.
- S. Riikonen, J. Timonen and T. Sikanen, Environmental considerations along the life cycle of pharmaceuticals: Interview study on views regarding environmental challenges, concerns, strategies, and prospects within the pharmaceutical industry, Eur. J. Pharm. Sci., 2024, 196, 106743 CrossRef CAS PubMed.
- United Nations, Sustainable Development Goals: THE 17 GOALS, https://sdgs.un.org/goals, (accessed 4 August 2025).
- M. E. Mondejar, R. Avtar, H. L. B. Diaz, R. K. Dubey, J. Esteban, A. Gómez-Morales, B. Hallam, N. T. Mbungu, C. C. Okolo, K. A. Prasad, Q. She and S. Garcia-Segura, Digitalization to achieve sustainable development goals: Steps towards a Smart Green Planet, Sci. Total Environ., 2021, 794, 148539 CrossRef CAS PubMed.
- G. Hole, A. S. Hole and I. McFalone-Shaw, Digitalization in pharmaceutical industry: What to focus on under the digital implementation process?, Int. J. Pharm.: X, 2021, 3, 100095 CAS.
- J. W. Sin, S. L. Chau, R. P. Burwood, K. Püntener, R. Bigler and P. Schwaller, Highly parallel optimisation of chemical reactions through automation and machine intelligence, Nat. Commun., 2025, 16, 6464 CrossRef CAS PubMed.
- C. Duan, G. H. Liu, Y. Du, T. Chen, Q. Zhao, H. Jia, C. P. Gomes, E. A. Theodorou and H. J. Kulik, Optimal transport for generating transition states in chemical reactions, Nat. Mach. Intell., 2025, 7, 615–626 CrossRef.
- A. I. Benítez-Mateos, D. Roura Padrosa and F. Paradisi, Multistep enzyme cascades as a route towards green and sustainable pharmaceutical syntheses, Nat. Chem., 2022, 14, 489–499 CrossRef PubMed.
- I. N. Derbenev, J. Dowden, J. Twycross and J. D. Hirst, Software tools for green and sustainable chemistry, Curr. Opin. Green Sustainable Chem., 2022, 35, 100623 CrossRef CAS.
- D. Kreutter, P. Schwaller and J. L. Reymond, Predicting enzymatic reactions with a molecular transformer, Chem. Sci., 2021, 12, 8648–8659 RSC.
- D. Kreutter and J. L. Reymond, Multistep retrosynthesis combining a disconnection aware triple transformer loop with a route penalty score guided tree search, Chem. Sci., 2023, 14, 9959–9969 RSC.
- A. Kumar, J. Taubitz, F. Meyer, N. Imstepf, J. Peng, E. Tassano, C. Moore, T. Lochmann, R. Snajdrova and R. Buller, Streamlining enzyme discovery and development through data analysis and computation, Chem. Catal., 2025, 5, 101445 CAS.
- R. Buller, S. Lutz, R. J. Kazlauskas, R. Snajdrova, J. C. Moore and U. T. Bornscheuer, From nature to industry: Harnessing enzymes for biocatalysis, Science, 2023, 382, 899 CrossRef PubMed.
- M. Philipp, M. Piccarreta and A. Pagani, The 2025 biopharma CEO strategy agenda, Kearney, 2025, 1–11 Search PubMed.
- A. V. Sadybekov and V. Katritch, Computational approaches streamlining drug discovery, Nature, 2023, 616, 673–685 CrossRef CAS PubMed.
- M. K. P. Jayatunga, M. Ayers, L. Bruens, D. Jayanth and C. Meier, How successful are AI-discovered drugs in clinical trials? A first analysis and emerging lessons, Drug Discovery Today, 2024, 29, 104009 CrossRef.
- S. Guan and G. Wang, Drug discovery and development in the era of artificial intelligence: From machine learning to large language models, Artif. Intell. Chem., 2024, 2, 100070 CrossRef.
- D. Probst, An explainability framework for deep learning on chemical reactions exemplified by enzyme-catalysed reaction classification, J. Cheminf., 2023, 15, 113 Search PubMed.
- S. Zhu, B. H. Nguyen, Y. Xia, K. Frost, S. Xie, V. Viswanathan and J. A. Smith, Improved environmental chemistry property prediction of molecules with graph machine learning, Green Chem., 2023, 25, 6612–6617 RSC.
- D. Q. Gbadago, G. Hwang, K. Lee and S. Hwang, Deep Learning for Green Chemistry: An AI-Enabled Pathway for Biodegradability Prediction and Organic Material Discovery, Korean J. Chem. Eng., 2024, 41, 2511–2524 CrossRef CAS.
- A. Joshi, Big data and AI for gender equality in health: bias is a big challenge, Front. Big Data, 2024, 7, 1436019 CrossRef PubMed.
- How AI reinforces gender bias—and what we can do about it, https://www.unwomen.org/en/news-stories/interview/2025/02/how-ai-reinforces-gender-bias-and-what-we-can-do-about-it, (accessed 9 October 2025).
- F. Urbina, F. Lentzos, C. Invernizzi and S. Ekins, Dual use of artificial-intelligence-powered drug discovery, Nat. Mach. Intell., 2022, 4, 189–191 CrossRef PubMed.
- Unesco, Challenging Systematic Prejudices An Investigation into Gender Bias in Large Language Models, 2024 Search PubMed.
- S. Chen, How much energy will AI really consume? The good, the bad and the unknown, Nature, 2025, 639, 22–24 CrossRef CAS PubMed.
- R. Stoyanova, P. M. Katzberger, L. Komissarov, A. Khadhraoui, L. Sach-Peltason, K. Groebke Zbinden, T. Schindler and N. Manevski, Computational Predictions of Nonclinical Pharmacokinetics at the Drug Design Stage, J. Chem. Inf. Model., 2023, 63, 442–458 CrossRef CAS.
- R. Satheeskumar, Enhancing drug discovery with AI: Predictive modeling of pharmacokinetics using Graph Neural Networks and ensemble learning, Intell. Pharm., 2025, 3, 127–140 Search PubMed.
- J. Zhu, P. Xiong, W. Wang, T. Lu and D. Ouyang, Integrating artificial intelligence and physiologically based pharmacokinetic modeling to predict in vitro and in vivo fate of amorphous solid dispersions, J. Controlled Release, 2025, 114123 CrossRef CAS PubMed.
- R. Eugster, M. Orsi, G. Buttitta, N. Serafini, M. Tiboni, L. Casettari, J.-L. Reymond, S. Aleandri and P. Luciani, Leveraging machine learning to streamline the development of liposomal drug delivery systems, J. Controlled Release, 2024, 376, 1025–1038 CrossRef CAS PubMed.
- R. Gazzi, R. Gelli, R. Eugster, C. Bühr, S. Schürch, R. Mezzenga, P. Luciani and S. Aleandri, Transitioning the production of lipidic mesophase-based delivery systems from lab-scale to robust industrial manufacturing following a risk-based quality by design approach augmented by artificial intelligence, J. Colloid Interface Sci., 2025, 678, 595–607 CrossRef CAS PubMed.
- R. Eugster, S. Aleandri, J. Bassila, D. Bochicchio, L. Baraldi, B. Hämmerle, S. Schürch, M. Vermathen, P. Vermathen, G. Rossi, A. Bergadano and P. Luciani, Hot cue: Physiologically controlled release from an in situ forming liposomal depot, J. Control. Release, 2026, 114739 CrossRef PubMed.
- Y. Santa-Ana-Tellez, B. Lagerwaard, A. J. de Jong, H. Gardarsdottir, D. E. Grobbee, K. Hawkins, M. Heath and M. Zuidgeest, Decentralised, patient-centric, site-less, virtual, and digital clinical trials? From confusion to consensus, Drug Discovery Today, 2023, 28, 103520 CrossRef PubMed.
- O. T. Inan, P. Tenaerts, S. A. Prindiville, H. R. Reynolds, D. S. Dizon, K. Cooper-Arnold, M. Turakhia, M. J. Pletcher, K. L. Preston, H. M. Krumholz, B. M. Marlin, K. D. Mandl, P. Klasnja, B. Spring, E. Iturriaga, R. Campo, P. Desvigne-Nickens, Y. Rosenberg, S. R. Steinhubl and R. M. Califf, Digitizing clinical trials, npj Digit. Med., 2020, 3, s41746 Search PubMed.
- M. Hutson, How AI is being used to accelerate clinical trials, Nature, 2024, 627, S2–S5 CrossRef CAS PubMed.
- A. Bender and I. Cortés-Ciriano, Artificial intelligence in drug discovery: what is realistic, what are illusions? Part 1: Ways to make an impact, and why we are not there yet, Drug Discovery Today, 2021, 26, 511–524 CrossRef CAS.
- F. McKay, B. J. Williams, G. Prestwich, D. Bansal, D. Treanor and N. Hallowell, Artificial intelligence and medical research databases: ethical review by data access committees, BMC Med. Ethics, 2023, 24, 49 CrossRef PubMed.
- V. J. Phiri, I. Battas, A. Semmar, H. Medromi and F. Moutaouakkil, Towards enterprise-wide pharma 4.0 adoption, Sci. Afr., 2025, 28, e02771 Search PubMed.
- A. Mastrantonas, P. Kokkas, A. Chatzopoulos, M. Papoutsidakis, C. Stergiou, A. Vairis and Z. Kanetaki, Identifying the effects of Industry 4.0 in the pharmaceutical sector: achieving the sustainable development goals, Discov. Sustain., 2024, 5, 460 CrossRef.
- D. Sharma, P. Patel and M. Shah, A comprehensive study on Industry 4.0 in the pharmaceutical industry for sustainable development, Environ. Sci. Pollut. Res., 2023, 30, 90088–90098 CrossRef CAS PubMed.
- A. C. Vaucher, P. Schwaller, J. Geluykens, V. H. Nair, A. Iuliano and T. Laino, Inferring experimental procedures from text-based representations of chemical reactions, Nat. Commun., 2021, 12, 2573 CrossRef CAS PubMed.
- A. Benhanifia, Z. Ben Cheikh, P. M. Oliveira, A. Valente and J. Lima, Systematic review of predictive maintenance practices in the manufacturing sector, Intell. Sys. Appl., 2025, 26, 200501 Search PubMed.
- A. Fournier, The Promise of AI in Pharmaceutical Manufacturing, Zurich, 2024 Search PubMed.
- P. Ullagaddi, Digital Transformation Strategies to Strengthen Quality and Data Integrity in Pharma, Int. J. Bus. Manag., 2024, 19, 16 CrossRef.
- W. Kritzinger, M. Karner, G. Traar, J. Henjes and W. Sihn, in IFAC-PapersOnLine, Elsevier B.V., 2018, vol. 51, pp. 1016–1022 Search PubMed.
- E. Katsoulakis, Q. Wang, H. Wu, L. Shahriyari, R. Fletcher, J. Liu, L. Achenie, H. Liu, P. Jackson, Y. Xiao, T. Syeda-Mahmood, R. Tuli and J. Deng, Digital twins for health: a scoping review, npj Digit. Med., 2024, 7, 77 CrossRef PubMed.
- A. Hengelbrock, F. Probst, S. Baukmann, A. Uhl, N. Tschorn, J. Stitz, A. Schmidt and J. Strube, Digital Twin for Continuous Production of Virus-like Particles toward Autonomous Operation, ACS Omega, 2024, 9, 34990–35013 CrossRef CAS PubMed.
- H. J. Warraich, T. Tazbaz and R. M. Califf, FDA Perspective on the Regulation of Artificial Intelligence in Health Care and Biomedicine, J. Am. Med. Assoc., 2025, 333, 241–247 CrossRef CAS PubMed.
- V. Ganthavee and A. P. Trzcinski, Artificial intelligence and machine learning for the optimization of pharmaceutical wastewater treatment systems: a review, Environ. Chem. Lett., 2024, 22, 2293–2318 CrossRef CAS.
- E. Strade, D. Kalnina and J. Kulczycka, Water efficiency and safe re-use of different grades of water-Topical issues for the pharmaceutical industry, Water Resour. Ind., 2020, 24, 100132 CrossRef.
- X. Chen, Z. Lei, J. S. Chang and D. J. Lee, Navigating future wastewater treatment plants with artificial intelligence: Applications, challenges, and innovations, J. Clean. Prod., 2025, 504, 145467 CrossRef.
- M. Shashi, Sustainable Digitalization in Pharmaceutical Supply Chains Using Theory of Constraints: A Qualitative Study, Sustainability, 2023, 15, 8752 CrossRef.
- J. Y. Ma, L. Shi and T. W. Kang, The Effect of Digital Transformation on the Pharmaceutical Sustainable Supply Chain Performance: The Mediating Role of Information Sharing and Traceability Using Structural Equation Modeling, Sustainability, 2023, 15, 649 CrossRef.
- A. M. Boyd and B. W. Chaffee, Critical Evaluation of Pharmacy Automation and Robotic Systems: A Call to Action, Hosp. Pharm., 2019, 54, 4–11 CrossRef.
- S. Castro de Hallgren, A. Julca, J. Palacin Lucio, R. Ferrufino and H. Silva Pacini Costa, Circular Economy, United Nations Economist Network, 2020 Search PubMed.
- M. Pablo and S. Ingo, Pivoting Pharma Supply Chains in a Digital World, 2019 Search PubMed.
- A. K. Vishwakarma, P. K. Patro and A. Acquaye, Applications of AI to low carbon decision support system for global supply chains, Clean Logist. Supply C., 2025, 17, 100261 CrossRef.
- Z. N. Jawad and Dr V. B. János, A comprehensive review of AI-enhanced decision making: An empirical analysis for optimizing medication market business, Mach. Learn. Appl., 2025, 20, 100676 Search PubMed.
- N. Sood, T. Shih, K. Van Nuys and D. Goldman, The Flow of Money Through the Pharmaceutical Distribution System Schaeffer Center White Paper Series, 2017 Search PubMed.
- D. Tinkoff, D. Zurkiya, E. Leydon and J. Lewis, Reimagining life science enterprises with agentic AI, McKinsey & Company, 2025, 1–19 Search PubMed.
- T. Hoveling, A. Svindland Nijdam, M. Monincx, J. Faludi and C. Bakker, Circular economy for medical devices: Barriers, opportunities and best practices from a design perspective, Conserv. Recycl., 2024, 208, 107719 CrossRef.
- R. Quelch, The role of pharmaceutical packaging in improving the bottom line for big pharma, European pharmaceutical review, 2021 Search PubMed.
- R. H. Hagenaars, R. Heijungs, A. de Koning, A. Tukker and R. Wang, The greenhouse gas emissions of pharmaceutical consumption and production: an input–output analysis over time and across global supply chains, Lancet Planet. Health, 2025, 9, e196–e206 CrossRef PubMed.
- W. P. Wong, P. S. Saw, S. Jomthanachai, L. S. Wang, H. F. Ong and C. P. Lim, Digitalization enhancement in the pharmaceutical supply network using a supply chain risk management approach, Sci. Rep., 2023, 13, 22287 CrossRef CAS PubMed.
- K. Ahluwalia, M. J. Abernathy, M. Algorri, N. S. Cauchon, N. M. Perico-Norred and R. Y. A. Youssef, The future of regulatory filings: digitalization, AAPS Open, 2025, 11, 9 CrossRef.
- A. J. Feeney, J. A. Goad and G. T. Flaherty, Global perspective of the risks of falsified and counterfeit medicines: A critical review of the literature, Travel Med. Infect. Dis., 2024, 61, 102758 CrossRef.
- S. S. Gomasta, A. Dhali, T. Tahlil, M. M. Anwar and A. B. M. S. Ali, PharmaChain: Blockchain-based drug supply chain provenance verification system, Heliyon, 2023, 9, e17957 CrossRef PubMed.
- S. M. Hosseini Bamakan, S. Ghasemzadeh Moghaddam and S. Dehghan Manshadi, Blockchain-enabled pharmaceutical cold chain: Applications, key challenges, and future trends, J. Cleaner Prod., 2021, 302, 127021 Search PubMed.
- H. Omidian and Y. Omidi, Blockchain in pharmaceutical life cycle management, Drug Discovery Today, 2022, 27, 935–938 Search PubMed.
- Y. Kolben, H. Azmanov, R. Gelman, D. Dror and Y. Ilan, Using chronobiology-based second-generation artificial intelligence digital system for overcoming antimicrobial drug resistance in chronic infections, Ann. Med., 2023, 55, 311–318 CrossRef PubMed.
- R. Gelman, N. Hurvitz, R. Nesserat, Y. Kolben, D. Nachman, K. Jamil, S. Agus, R. Asleh, O. Amir, M. Berg and Y. Ilan, A second-generation artificial intelligence-based therapeutic regimen improves diuretic resistance in heart failure: Results of a feasibility open-labeled clinical trial, Biomed. Pharmacother., 2023, 161, 114334 CrossRef CAS PubMed.
- C. McGenity, E. L. Clarke, C. Jennings, G. Matthews, C. Cartlidge, H. Freduah-Agyemang, D. D. Stocken and D. Treanor, Artificial intelligence in digital pathology: a systematic review and meta-analysis of diagnostic test accuracy, npj Digit. Med., 2024, 7, 114 CrossRef PubMed.
- I. J. Borges do Nascimento, H. M. Abdulazeem, L. T. Vasanthan, E. Z. Martinez, M. L. Zucolato, L. Østengaard, N. Azzopardi-Muscata, T. Zapata and D. Novillo-Ortiz, The global effect of digital health technologies on health workers’ competencies and health workplace: an umbrella review of systematic reviews and lexical-based and sentence-based meta-analysis, Lancet Digit. Health, 2023, 5, 534–578 Search PubMed.
- J. Liang, Q. Fang, X. Jiao, P. Xiang, J. Ma, Z. Zhang, Y. Liu, Y. He, Y. Li, Z. He and J. Lei, Approved trends and product characteristics of digital therapeutics in four countries, npj Digit. Med., 2025, 8, 308 CrossRef PubMed.
- D. A. Simon, S. Raza, C. Shachar and I. Glenn Cohen, Using digital technologies to diagnose in the home: recommendations from a Delphi panel, npj Digit. Med., 2024, 7, 18 CrossRef PubMed.
- S. A. Waldman and A. Terzic, Health Care Evolves From Reactive to Proactive, Clin. Pharmacol. Ther., 2019, 105, 10–13 CrossRef PubMed.
- H. Takeshige-Amano, G. Oyama, M. Ogawa, K. Fusegi, T. Kambe, K. Shiina, S. I. Ueno, A. Okuzumi, T. Hatano, Y. Motoi, I. Kawakami, M. Ando, S. Nakayama, Y. Ishida, S. Maei, X. Lu, T. Kobayashi, R. Wooden, S. Ota, K. Morito, Y. Ito, Y. Nakajima, A. Yoritaka, T. Kato and N. Hattori, Digital detection of Alzheimer's disease using smiles and conversations with a chatbot, Sci. Rep., 2024, 14, 26309 CrossRef CAS PubMed.
- T. Manyazewal, M. K. Ali, T. Kebede, M. J. Magee, T. Getinet, S. A. Patel, D. Hailemariam, C. Escoffery, Y. Woldeamanuel, N. Makonnen, S. Solomon, W. Amogne, V. C. Marconi and A. Fekadu, Mapping digital health ecosystems in Africa in the context of endemic infectious and non-communicable diseases, npj Digit. Med., 2023, 6, 97 CrossRef PubMed.
- A. Gedikci Ondogan, M. Sargin and K. Canoz, Use of electronic medical records in the digital healthcare system and its role in communication and medical information sharing among healthcare professionals, Inform. Med. Unlocked, 2023, 42, 101373 CrossRef.
- Y. Albogami, A. Alfakhri, A. Alaqil, A. Alkoraishi, H. Alshammari, Y. Elsharawy, A. Alhammad and A. Alhossan, Safety and quality of AI chatbots for drug-related inquiries: A real-world comparison with licensed pharmacists, Digital Health, 2024, 10, 11 CrossRef.
- J. A. Greer, J. S. Temel, A. El-Jawahri, S. Rinaldi, M. Kamdar, E. R. Park, N. K. Horick, K. Pintro, D. J. Rabideau, L. Schwamm, J. Feliciano, I. Chua, K. Leventakos, S. M. Fischer, T. C. Campbell, M. W. Rabow, F. Zachariah, L. C. Hanson, S. F. Martin, M. Silveira, L. Shoemaker, M. Bakitas, J. Bauman, L. Spoozak, C. Grey, L. Blackhall, K. Curseen, S. O'Mahony, M. M. Smith, R. Rhodes, A. Cullinan and V. Jackson, Telehealth vs In-Person Early Palliative Care for Patients with Advanced Lung Cancer: A Multisite Randomized Clinical Trial, J. Am. Med. Assoc., 2024, 332, 1153–1164 Search PubMed.
- R. Raab, A. Küderle, A. Zakreuskaya, A. D. Stern, J. Klucken, G. Kaissis, D. Rueckert, S. Boll, R. Eils, H. Wagener and B. M. Eskofier, Federated electronic health records for the European Health Data Space, Lancet Digit. Health, 2023, 5, e840–e847 Search PubMed.
- S. Li, K. Surineni and N. Prabhakaran, Cyber-Attacks on Hospital Systems: A Narrative Review, Am. J. Geriatr. Psychiatry: Open Sci. Educ. Pract., 2025, 7, 30–39 Search PubMed.
- A. F. Brantly and N. D. Brantly, Patient-centric cybersecurity, J. Cyber Policy, 2020, 5, 372–391 Search PubMed.
- S. Navya and R. Babu, Cyber Security for Healthcare-The Need of The Hour?, J. Med. Dent. Front., 2024, 1, 48–54 Search PubMed.
- B. Aldosari, Cybersecurity in Healthcare: New Threat to Patient Safety, Cureus, 2025, 17, e83614 Search PubMed.
- F. Salvo, J. Micallef, A. Lahouegue, L. Chouchana, L. Létinier, J. L. Faillie and A. Pariente, Will the future of pharmacovigilance be more automated?, Expert Opin. Drug Saf., 2023, 22, 541–548 CrossRef CAS PubMed.
- R. De Abreu Ferreira, S. Zhong, C. Moureaud, M. T. Le, A. Rothstein, X. Li, L. Wang and M. Patwardhan, A Pilot, Predictive Surveillance Model in Pharmacovigilance Using Machine Learning Approaches, Adv. Ther., 2024, 41, 2435–2445 Search PubMed.
- M. A. Shamim, M. A. Shamim, P. Arora and P. Dwivedi, Artificial intelligence and big data for pharmacovigilance and patient safety, J. Med. Surg. Public Health, 2024, 3, 100139 CrossRef.
- J. Warner, A. Prada Jardim and C. Albera, Artificial Intelligence: Applications in Pharmacovigilance Signal Management, Pharm. Med., 2025, 39, 183–198 CrossRef.
- P. Chandak and N. P. Tatonetti, Using Machine Learning to Identify Adverse Drug Effects Posing Increased Risk to Women, Patterns, 2020, 1, 100108 CrossRef PubMed.
- A. Balendran, M. Benchoufi, T. Evgeniou and P. Ravaud, Algorithmovigilance, lessons from pharmacovigilance, npj Digit. Med., 2024, 7, 270 CrossRef PubMed.
- P. J. Embi, Algorithmovigilance - Advancing Methods to Analyze and Monitor Artificial Intelligence-Driven Health Care for Effectiveness and Equity, JAMA Netw. Open, 2021, 4, 4622 Search PubMed.
- C. C. Lee, J. Zou and P. F. Chen, The impact of artificial intelligence on the energy consumption of corporations: The role of human capital, Energy Econ., 2025, 143, 108231 Search PubMed.
- J. Zeng and T. Wang, The impact of China's artificial intelligence development on urban energy efficiency, Sci. Rep., 2025, 15, 24129 CrossRef CAS PubMed.
- O. Schilter, P. Schwaller and T. Laino, Balancing computational chemistry's potential with its environmental impact, Green Chem., 2024, 26, 8669–8679 RSC.
- P. Delanoë, D. Tchuente and G. Colin, Method and evaluations of the effective gain of artificial intelligence models for reducing CO2 emissions, J. Environ. Manage., 2023, 331, 117261 CrossRef PubMed.
- J. Blumenthal and M. L. Diamond, Sustainability of the Internet of Things Requires Understanding of Mineral Demands and Supplies, Environ. Sci. Technol., 2022, 56, 9835–9837 CrossRef CAS PubMed.
- B. Niu, S. E, Q. Song, Z. Xu, B. Han and Y. Qin, Physicochemical reactions in e-waste recycling, Nat. Rev. Chem., 2024, 8, 569–586 CrossRef CAS PubMed.
- B. Liang, J. Gu, X. Zeng, W. Yuan, M. Rao, B. Xiao and H. Hu, A Review of the Occurrence and Recovery of Rare Earth Elements from Electronic Waste, Molecules, 2024, 29, 4624 CrossRef CAS PubMed.
- E. Andrew and D. C. A. Pigosso, Multidisciplinary perspectives on rebound effects in sustainability: A systematic review, J. Cleaner Prod., 2024, 470, 143366 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |