Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Redesigning valproic acid therapy in pregnancy: intranasal liposomes for targeted maternal treatment
Bruna Corradetti *ab,
Luigi C. Zangaric,
Francesco Mainardi
*ab,
Luigi C. Zangaric,
Francesco Mainardi c,
Tamara Pezzotta
c,
Tamara Pezzotta c,
Richard H. Finnellad and
Francesca Taraballief
c,
Richard H. Finnellad and
Francesca Taraballief
aCenter for Precision Environmental Health, Baylor College of Medicine, Houston, Texas, USA. E-mail: bruna.corradetti@bcm.edu
bDepartment of Medicine, Section Hematology/Oncology Baylor College of Medicine, Houston, Texas, USA
cDepartment of Pharmacy and Biotechnology (FaBIT), University of Bologna, Bologna, Italy
dDepartments of Molecular and Human Genetics, Molecular and Cellular Biology, and Medicine, Baylor College of Medicine, Houston, Texas, USA
eCenter for Musculoskeletal Regeneration, Houston Methodist Academic Institute, Houston, Texas, USA
fOrthopedics and Sports Medicine, Houston Methodist Academic Institute, Houston, Texas, USA
First published on 17th February 2026
Abstract
Valproic acid (VPA) is a potent antiseizure medication and mood stabilizer, yet its teratogenicity severely limits safe use during pregnancy. Despite guidelines advising against VPA use in women of reproductive age, it remains indispensable for certain drug-resistant epilepsies, highlighting the urgent need for safer and more targeted delivery strategies. Intranasal (IN) administration via nanocarriers represents a promising approach to enhance brain uptake while minimizing systemic exposure and placental transfer. In this review, we evaluate the scientific rationale and translational potential of IN nanoformulations of VPA specifically designed for use during pregnancy. We discuss strategies to engineer nanocarriers that achieve effective maternal brain delivery while reducing fetal risk, and we analyze preclinical data on biodistribution, placental passage, and therapeutic efficacy. Importantly, we highlight how gestational changes in maternal physiology and placental architecture can inform the rational design of pregnancy-adapted nanocarriers. By integrating insights from nanotechnology, pharmacology, and maternal-fetal medicine, this review outlines a paradigm shift from drug avoidance to precision delivery that maximizes reproductive safety. This strategy not only addresses the unmet need for safer VPA use in pregnancy, but also establishes a versatile framework for broader applications of nanomedicine in neurological and systemic disorders in women of childbearing age.
1. Introduction
Valproic acid (VPA) is a cornerstone in the management of epilepsy, particularly for patients with genetic generalized epilepsy (GGE) and juvenile myoclonic epilepsy (JME), where few alternative antiseizure medications (ASMs) match its efficacy.1 However, its use during pregnancy is associated with a markedly increased risk of congenital malformations, including a tenfold higher incidence of neural tube defects, as well as a range of neurodevelopmental disorders.2,3 These risks have prompted stringent regulatory recommendations to avoid VPA use in women of childbearing potential and to encourage the adoption of alternative therapies whenever possible.4 Nonetheless, in certain patients, switching to alternative ASMs fails to achieve adequate seizure control, making VPA an essential option despite its well-established teratogenic risk. This clinical dilemma highlights the urgent need for innovative strategies that preserve the therapeutic benefits of VPA while minimizing systemic and fetal exposure.In this review, we examine advanced strategies to enhance the targeted brain delivery of VPA while minimizing systemic exposure and placental transfer. Emphasis is placed on the importance of tailoring nanoparticle (NP) design to the physiological and pharmacokinetic changes that characterize pregnancy. We focus on intranasal (IN) nanoformulated VPA because this route offers a dual advantage: maintaining seizure control in patients for whom VPA is indispensable while reducing fetal exposure. Special focus is given to nanocarrier features that support efficient IN brain delivery and restrict placental passage. Finally, we review current nanocarrier platforms used for IN administration and evaluate physicochemical parameters associated with transplacental transport or exclusion, providing guidance for the development of safer VPA formulations during pregnancy.
2. Valproic acid: metabolism, teratogenicity, and emerging nanomedicine strategies
VPA is a broad-spectrum ASM with proven efficacy in the treatment of various epilepsy syndromes, including generalized and focal seizures.5,6 Its clinical utility also extends to bipolar disorder and migraine prophylaxis.7,8 However, therapeutic use of VPA is complicated by its narrow therapeutic index and significant risks of systemic toxicity, hepatotoxicity, and teratogenicity.9 These adverse effects are closely linked to the metabolic fate of VPA, which involves complex biotransformation pathways producing both pharmacologically active and toxic metabolites. Understanding these metabolic pathways is therefore critical to developing safer therapeutic strategies, particularly for vulnerable populations such as pregnant women, where fetal safety is paramount.2.1 Metabolic complexity and teratogenic risk of VPA
VPA is almost entirely metabolized in the liver, with only minimal amounts of the unmetabolized drug excreted renally. Its metabolism involves multiple enzymatic pathways, including oxidation, glucuronidation, β-oxidation, and conjugation with carnitine or glycine.10,11 This metabolic complexity yields a spectrum of pharmacologically active and toxic metabolites, linking systemic exposure to hepatotoxicity, neurotoxicity and teratogenic risk.12–18 Among these, cytochrome P450 (CYP)-dependent metabolites such as 2-propyl-4-pentenoic acid (4-ene-VPA) and 2-propyl-2-pentenoic acid (2-ene-VPA) have been most strongly associated with hepatotoxic and neurotoxic effects.12,19–21 The 4-ene-VPA metabolite is highly teratogenic, while the 2-ene-VPA metabolite is much less so.22,23 While glucuronidation via UGT enzymes produces valproate glucuronide, the major excretory product.10,12,21VPA-associated teratogenicity has been linked to metabolite-driven disruption of embryonic gene regulation and cellular homeostasis, affecting pathways involved in neuroepithelial integrity and neurulation.12,24–26 Toxic metabolites contribute to teratogenicity through the downregulation of genes, including IGF2R, RGS4, COL6A3, EDNRB, and KLF6. Genetic and environmental modifiers of VPA sensitivity converge on these same metabolic pathways, as demonstrated by strain-dependent metabolomic alterations correlating with susceptibility to VPA-induced neural tube defects in murine models.27,28 In parallel, in vitro neurulation models have shown that both genetic variants and VPA exposure impair neuroepithelial junctional organization, providing mechanistic insight into how systemic metabolic perturbations translate into failed neural tube closure.24
Given these risks, the WHO recommends avoiding VPA therapy in women of childbearing potential, emphasizing effective contraception and switching to alternative ASMs when pregnancy is planned.4 Nonetheless, VPA remains the most effective treatment for specific epilepsy syndromes, including primary generalized tonic-clonic seizures (PGTCS), GGE, and JME.1,9 Clinical data illustrate the difficulty of discontinuing VPA: while substantial proportion of women of reproductive age can withdraw from treatment, seizure relapse rates remain high, particularly in patients with GGE.29,30 These findings highlight a persistent clinical dilemma in which seizure control must be balanced against fetal safety.
Collectively, these data indicate that VPA-associated teratogenicity is primarily driven by systemic maternal exposure and efficient placental transfer of the parent compound and its metabolites. Strategies capable of preserving central nervous system efficacy while reducing maternal plasma concentrations therefore represent a rational approach to mitigating fetal risk, particularly during pregnancy when physiological adaptations further complicate systemic PK.
2.2 The transformative potential of nanoformulated VPA
Given the central role of VPA metabolism in driving systemic toxicity and teratogenicity, strategies capable of modulating biodistribution and limiting off-target exposure are urgently needed. Nanoformulated delivery systems offer a means to decouple central nervous system (CNS) efficacy from systemic exposure, thereby potentially reducing hepatic metabolism and the formation of toxic metabolites while preserving antiseizure activity. This approach is particularly relevant in pregnancy, where fetal risk is tightly linked to maternal plasma exposure. Several VPA-containing nanoformulations have been reported, employing both lipid-based and polymeric carriers (Table 1). Among these, IN formulations are of particular interest because they enable direct nose-to-brain transport and achieve high CNS exposure with reduced systemic distribution.| Type of formulation | Components | Formulation | Physical characterization | in vitro/ex vivo testing | in vivo testing (animal model/outcome) |
|---|---|---|---|---|---|
| Intranasal administration | |||||
| Liposomes31 | Phosphatidylcholine, cholesterol | [Lipo-VPA-4] | Size: 92.01 ± 1.87 nm | Permeation on sheep nasal mucosa; plasma and brain tissue HPLC analysis | Male Wistar rats |
| Thin-film hydration | PDI: 0.21 ± 0.01 | Outcome: four-fold increase in penetration compared to control | Outcome: increased Cmax and AUC in plasma and brain tissue with respect to free VPA | ||
| ZP: −43.47 ± 2.59 mV | |||||
| EE: 85.5 ± 1.07% | |||||
| NLCs32 | Cetyl palmitate, lecithin, octyldodecanol, poloxamer 188 | Emulsion-solvent diffusion, evaporation, ultrasonication | Size: 154 ± 16 nm | Plasma and brain tissue GC analysis | Male Wistar rats MES model |
| PDI: 0.2 ± 0.1 | Outcome: brain/plasma concentration ratio 20 times higher compared with VPA IP | Outcome: IN NLC (4 mg kg−1) achieves the same protection as free VPA IP (150 mg kg−1) with a 37-times lower dose | |||
| ZP: −10 ± 0.5 mV | |||||
| DL: 47 ± 0.8% | |||||
| DR: 75 ± 1.9% over 21 days | |||||
| Intraperitoneal administration | |||||
| VANE33 | Lecithin, safflower seed oil, medium chain triglyceride, alpha-tocopherol, Tween-80 | [F3] ultrasonication | Size: 142.4 ± 2.45 nm | BBB permeation model (hCMEC/D3 and CC-2565) and cytotoxicity | Sprague–Dawley rats |
| Homogenization | PDI: 0.089 ± 0.015 | Outcome: higher viability with F3, same permeation compared to free VPA | Outcome: higher plasma and brain concentration, half-life and decreased clearance with respect to free VPA | ||
| Drug concentration: 158.26 ± 3.48 μg mL−1 | |||||
| EE: 99.29 ± 4.57% | |||||
| PLGA-NPs34 | Poly-lactic glycolic-acid, Tween 80, span 20 | Double emulsion-solvent evaporation | Size: 220 ± 78 nm | Plasma level, biochemical evaluation of brain samples | PTZ-induced male Wistar rats |
| ZP: −32.9 mV | Outcome: reached therapeutic levels | Outcome: protection against seizures at 6h | |||
| DL: 13.96% | |||||
| EE: 30% | |||||
| Amphiphilic block polymer NPs35 | Methoxy-ended-poly(ethylene glycol)-b-poly(vinyl valproate block copolymer) | [PEG-b-PV(VPA)] | Size: 38nm | Spleen weight, hematological parameters, and liver function were evaluated by ALT and AST | PTZ-induced mice |
| dissolution in N,N-dimethylformamide and dialysis | PDI: 0.1 | Outcome: no toxicity compared to free VPA administration | Outcome: seizure protection even with weekly administration | ||
| Intravenous administration | |||||
| NBCA- NPs36 | Butylcyanoacrylate, Tween 85, Tween 80 dextran 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
Polymerization in acidic medium | N/A | Brain and plasma level GC analysis | NMRI mice |
| Outcome: comparable levels compared to free VPA, slower metabolism and lower metabolites concentrations | |||||
| Other studies | |||||
| Cationic nanoemulsion gel37 | Emulsion: Safflower seed oil, Tween 80, + lecithin (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), Phosphatidylglycerol, stearylamine 1), Phosphatidylglycerol, stearylamine |
[CVE6] | Size: 113 nm | Drug release with dialysis machine | N/A |
Gel![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Carbopol 934, triethanolamine Carbopol 934, triethanolamine |
Nanoemulsion via slow and spontaneous titration. | PDI: 0.26 | Outcome: complete release at pH 6.8, drug deposition in goat nasal mucosa, 60–70% DD at 6 h | ||
| [CVE6 gel 0.35% w/w VPA] | ZP: + 34.7 mV | ||||
| Gel obtained by mixing carbopol gel and lyophilized formulation | Size: 129 nm | ||||
| ZP: + 21.9 mV | |||||
| Viscosity: 1837 cP | |||||
| NLCs38 | Compritol® ATO 888, Miglyol® 812, Gelucire® 59/14, Tween®80, cetrimide | Emulsion and ultrasonication | Size: 76.1 ± 2.8pH: 6.8 nm | Biocompatibility study on SH-SY5Y, RPMI 2650, HepG2 cells; Mucin binding efficiency test | N/A |
| PDI: 0.190 ± 0.027 | Outcome: <75 μg mL−1 is deemed safe; estimated mucin binding efficiency: 58.66 ± 2.26% | ||||
| ZP: 28.1 ± 2.0 mV | |||||
| EE: 85.4 ± 0.8% | |||||
Nanostructured lipid carriers (NLCs) represent another lipid-based strategy. Eskandari et al. (2011) and Correia et al. (2024) reported VPA-loaded NLCs. Eskandari et al. (2011) reported the first IN VPA formulation using nanostructured lipid carriers (NLC), demonstrating in a male rat Maximum Electroshock Seizure model comparable efficacy to intraperitoneal administration of free VPA, while achieving a >20-fold higher brain-to-plasma concentration ratio at a 37-fold lower dose (4 mg kg−1).32 These findings highlight the ability of IN nanoformulations to markedly enhance CNS targeting while minimizing systemic exposure. More recently, Correia et al. (2024) optimized VPA-loaded NLCs for IN delivery using a design-of-experiments approach, although in vivo validation was not performed.38
IN liposomal delivery of VPA has also been explored. Yuwanda et al. (2022) evaluated IN-administered VPA-loaded liposomes in male Wistar rats and reported a two-fold increase in plasma concentration compared with free VPA, confirming enhanced bioavailability and CNS delivery.31 While systemic levels were not eliminated, the disproportionate increase in brain exposure relative to plasma supports partial pharmacokinetics (PK) uncoupling between CNS efficacy and systemic distribution.
Although fetal exposure was not directly assessed in these studies, the consistently elevated brain-to-plasma ratios achieved via IN delivery strongly suggests a reduction in systemic (and by extension fetal) exposure compared with oral or IV administration. This inference is supported by the broader principle that placental transfer is driven primarily by free drug concentrations in maternal plasma, which are expected to be lower following IN administration than after systemic dosing.39 Other nanoformulation strategies have been developed using systemic routes of administration. Lipid-based nanoemulsions reported by Tan et al. (2017) and Hussain et al. (2023) employed safflower seed oil, lecitin, and Tween80 to enhance BBB penetration.33,37 Polymeric nanocarriers, including PLGA-based NPs and covalent VPA conjugates, have likewise demonstrated improved seizure control and, in some cases, reduced levels of toxic metabolites following IP or IV administration.34–36 While these approaches improve therapeutic indices, their reliance on systemic delivery limits their capacity to restrict maternal-fetal transfer during pregnancy.
Together, these findings support the potential of IN nanoformulations to achieve effective CNS delivery of VPA while reducing systemic exposure, providing a mechanistic rationale for fetal sparing delivery strategies in the context of pregnancy. These considerations motivate deeper examination of the mechanistic basis of IN drug transport and the pregnancy-specific physiological factors that influence PK and maternal-fetal transfer.
3. Targeting the brain via the nasal route: mechanistic insights and liposome-enhanced drug delivery
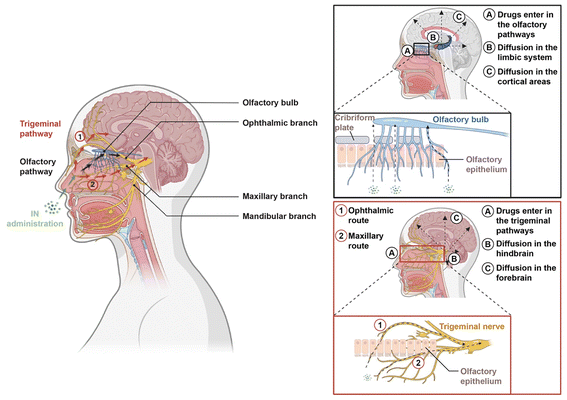
Building on the metabolic and systemic determinants of fetal risk discussed in Section 2, this section examines the IN route as a strategy for CNS drug delivery that may alter maternal systemic exposure and, by extension, placental drug transfer. Unlike oral or IV administration, where CNS efficacy is achieved through sustained maternal plasma concentrations, IN delivery enables partial direct transport to the brain with important implications for systemic exposure, maternal PK, and fetal risk.3.1 IN delivery as a non-invasive route for CNS targeting and systemic exposure modulation
The IN route, first described over a century ago, offers a non-invasive and promising strategy for delivering CNS-directed therapeutics by bypassing the BBB.40 Conventional oral or parenteral drug delivery relies on systemic circulation to reach the CNS but is constrained by hepatic first-pass metabolism, enzymatic degradation, systemic clearance, and physiological barriers such as the intestinal epithelium and the BBB,41,42 which excludes the majority of small molecules.43 Alternative approaches, including intrathecal administration or intracerebral infusion, can bypass the BBB but are invasive, unsuitable for chronic use, and associated with procedural risks.44 IN delivery provides direct access to the brain through two principal neural pathways: the olfactory and trigeminal nerves41 (Fig. 1). The olfactory nerve (cranial nerve I) enables transport via axonal pathways and perineuronal channels,45,46 while the trigeminal nerve (cranial nerve V) facilitates drug delivery to the hindbrain and forebrain through intracellular and extracellular mechanisms such as bulk flow and perivascular diffusion.47–50 Because these pathways allow CNS exposure without requiring high or sustained maternal systemic plasma concentrations, IN delivery can reduce peripheral exposure and off-target effects while improving central bioavailability, making it particularly attractive for CNS indications requiring rapid onset and dose sparing.Several IN formulations have already reached clinical use, including Spravato® (esketamine, Johnson & Johnson) for depression,41 NAYZILAM® (midazolam) and VALTOCO® (diazepam) for acute seizures,40 and Zavegepant, the first GCRP receptor antagonist nasal spray approved by the FDA for migraine.51,52 Additional candidates under clinical investigation include IN ketamine for major depressive disorder (Seelos Therapeutics, NCT04669665) and IN insulin combined with empagliflozin for early Alzheimer's disease patients (Wake Forest University, NCT05081219). Notably, while these agents demonstrate the feasibility of IN delivery for CNS disorders, systematic evaluations of maternal systemic and fetal exposure remain extremely limited.
3.2. Determinants of nose-to-brain transport and systemic spillover
Despite its advantages, IN delivery does not completely eliminate systemic absorption. The fraction of drug reaching the brain versus entering the systemic circulation is governed by both patient-dependent and drug-dependent factors. Patient-dependent variables include mucin secretion, mucociliary clearance, enzymatic degradation, P-glycoprotein (P-gp) efflux activity, and vascular absorption, all of which reduce the fraction of drug reaching the brain.41,53 Mucin, which constitutes ∼2.5–3% of the nasal mucosa,54 acts as a physical barrier,55,56 particularly impeding hydrophobic or charged hydrophilic molecules.46,57 Mucociliary clearance further limits residence time and absorption,41,58 with clearance occurring more rapidly in the respiratory epithelium than in the olfactory region.46,59Enzymatic degradation in the nasal cavity can function as a pseudo-first-pass effect,41,60 while P-gp, highly expressed in the olfactory epithelium, actively effluxes drugs and contributes to variability in absorption and systemic distribution.41,61,62 Drug-dependent factors, including molecular weight, lipophilicity, solubility, pH, viscosity, and osmolality,41,46 further shape the balance between CNS uptake and systemic spillover.41,46 From a maternal-fetal pharmacology perspective, these parameters are critical, as placental transfer is driven primarily by free drug concentrations in maternal plasma. Consequently, IN formulations that maximize nose-to-brain transport while limiting vascular absorption are expected to yield lower maternal plasma Cmax and area under the curve (AUC) compared with oral or IV administration, thus reducing fetal exposure.
3.3 Liposomes as modulators of brain targeting and maternal plasma exposure
A promising strategy to enhance nose-to-brain transport is the use of nanoformulated drugs, which can improve permeability, extend residence time, and increase brain concentrations.41,63 Among these, liposomes have emerged as versatile nanocarriers due to their biocompatibility, structural adaptability, and ability to encapsulate both hydrophilic and lipophilic drugs.64 Structurally, liposomes consist of one or more phospholipid bilayers surrounding an aqueous core and are commonly classified as unilamellar vesicles (single bilayer), multilamellar vesicles (multiple concentric bilayers), or multivesicular vesicles (non-concentric aqueous compartments).65,66Liposomes offer several advantages, including protection of labile drugs, controlled or targeted release, and reduced systemic toxicity.64,67 Their clinical utility, however, can be constrained by limited loading capacity, physicochemical instability leading to leakage or degradation, and rapid clearance by the mononuclear phagocyte system (MPS), primarily via the liver and spleen.68 Early approaches to improve systemic stability focused on optimizing lipid composition, including cholesterol content, to increase membrane rigidity.69 A transformative step was polyethylene glycol (PEG) functionalization (PEGylation), which generates a hydrophilic, neutrally charged steric barrier that reduces opsonization and MPS uptake and can prolong circulation time.70–73 While systemic half-life is not the primary goal for IN formulations, these surface modifications remain relevant because they influence mucus interactions, epithelial transport, and downstream biodistribution after any systemic absorption.
The functional performance of liposomes depends on key physicochemical parameters. Particle size, typically ranging from 50 nm to several micrometers, influences biodistribution, and cellular uptake and can be modulated by lipid composition (e.g., cholesterol-to-phospholipid ratio).74,75 A polydispersity index (PDI) ≤ 0.3 is generally used as an indicator of formulation uniformity and stability,76 while zeta potential (ZP) values above ±30 mV can enhance colloidal stability by reducing aggregation.74 Encapsulation efficiency (EE) reflects the fraction of drug successfully incorporated, with hydrophobic molecules partitioning into the lipid bilayer and hydrophilic compounds localizing within the aqueous core.77 Collectively, these parameters can be tuned to favor nose-to-brain transport while minimizing systemic spillover, an especially relevant consideration in pregnancy, where fetal exposure is strongly influenced by maternal plasma concentrations.
In the context of IN delivery, liposomes have shown considerable promise. Preclinical studies in male rats demonstrated that liposome-encapsulated drugs (e.g., DSPC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CHOL
CHOL![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PEG, 1
PEG, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5 mol/mol/mol; and EPC
0.5 mol/mol/mol; and EPC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DSPE-PEG2000
DSPE-PEG2000![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CHOL, 20
CHOL, 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 mol/mol/mol) achieved significantly higher brain exposure than free drug delivered intranasally or orally.78,79 In addition, Yuwanda et al. reported that phosphatidylcholine-rich liposomes (∼92 nm) enhanced VPA brain bioavailability following IN administration.31 Importantly, while systemic levels were not eliminated, disproportionate increases in brain exposure relative to plasma support partial PK uncoupling between CNS and efficacy and systemic distribution, consistent with the goal of reducing maternal plasma exposure while maintaining CNS delivery.
5 mol/mol/mol) achieved significantly higher brain exposure than free drug delivered intranasally or orally.78,79 In addition, Yuwanda et al. reported that phosphatidylcholine-rich liposomes (∼92 nm) enhanced VPA brain bioavailability following IN administration.31 Importantly, while systemic levels were not eliminated, disproportionate increases in brain exposure relative to plasma support partial PK uncoupling between CNS and efficacy and systemic distribution, consistent with the goal of reducing maternal plasma exposure while maintaining CNS delivery.
Surface charge and PEGylation also critically shape regional brain distribution and mucus interactions. Kurano et al. evaluated liposomes with different surface charges across multiple brain regions (including olfactory bulb, forebrain, hindbrain, bulbospinal tract, and trigeminal nerve) and found that neutral PEGylated liposomes (DSPE-PEG2000-coated) enabled broader distribution, whereas positively charged liposomes tended to accumulate in the olfactory bulb via electrostatic interactions with mucin.80 PEGylation may further facilitate parenchymal penetration. Narayan et al. reported that PEGylated liposomes (LP-16, SPC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CHOL
CHOL![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MPEG-DSPE, 8
MPEG-DSPE, 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.20) achieved higher brain AUC compared to IV administration or non-PEGylated liposomes.81 Moreover, PEGylation of larger NP (200–500 nm) markedly improved mucus penetration, countering earlier assumptions that PEG necessarily increases mucoadhesion. Low molecular weight PEG chains (∼2 kDa) improved particle mobility by reducing hydrophobic interactions and hydrogen bonding, yielding diffusion rates up to 100–1000 times higher.82,83 Interestingly, 200 nm polystyrene-PEG (PS-PEG) NP diffused more efficiently than 100 nm particles, likely due to greater PEG surface coverage.84 Collectively, these formulation-dependent parameters can be leveraged to favor nose-to-brain transport while further limiting systemic spill-over, thereby indirectly reducing the potential for transplacental diffusion.
0.20) achieved higher brain AUC compared to IV administration or non-PEGylated liposomes.81 Moreover, PEGylation of larger NP (200–500 nm) markedly improved mucus penetration, countering earlier assumptions that PEG necessarily increases mucoadhesion. Low molecular weight PEG chains (∼2 kDa) improved particle mobility by reducing hydrophobic interactions and hydrogen bonding, yielding diffusion rates up to 100–1000 times higher.82,83 Interestingly, 200 nm polystyrene-PEG (PS-PEG) NP diffused more efficiently than 100 nm particles, likely due to greater PEG surface coverage.84 Collectively, these formulation-dependent parameters can be leveraged to favor nose-to-brain transport while further limiting systemic spill-over, thereby indirectly reducing the potential for transplacental diffusion.
From a translational perspective, liposomes are supported by a strong clinical precedent, with more than 20 lipid-based formulations approved by the FDA or EMA across multiple indications.85 While current delivery routes include parenteral, intrathecal, epidural, and inhalation, the IN pathway remains comparatively underexplored yet highly promising for CNS-targeting. Notably, although pregnancy-specific pharmacokinetic data for nano-enabled nose-to-brain delivery remain extremely limited, a recent preclinical study showed that this approach improved seizure control and prevented congenital malformations in pregnant epileptic rats compared with systemic administration, providing initial functional support for its relevance during pregnancy.86 Accordingly, conclusions regarding maternal-fetal exposure following IN liposomal delivery must currently rely on mechanistic principles and preclinical brain-to-plasma exposure data, motivating the pregnancy-specific discussion of placental remodeling and the limited functional and clinical observations presented below. Table 2 summarizes key studies focused on liposome-based IN delivery.
| Encapsulated Drug | Liposome composition | Physical Characterization | Testing approach | Administration route | Outcome |
|---|---|---|---|---|---|
| Donepezil78 | DSPC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CHE CHE![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PEG PEG |
Size: 102 ± 3.3 nm | Wistar rats | IN | ↑ Brain bioavailability compared with the free drug |
Ratio:1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5 0.5 |
PDI: 0.28 ± 0.03 | ||||
| ZP: 28.31 ± 0.85 mV | |||||
| EE: 84.91% ± 3.31% | |||||
| H102 peptide79 | EPC, DSPE-PEG2000 | Size: 12.2 ± 6.4 nm | Male Sprague-Dawley rats | IN | ↑ Cerebral absorption, particularly in the hippocampus, vs. nasal solution |
Ratio: 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5 |
PDI: 0.185 ± 0.012 | ||||
| ZP: −2.96 ± 0.38 mV | |||||
| EE: 71.35 ± 0.87% | |||||
| VPA31 | [Lipo-VPA-4 (F4)] | Size: 92.01 ± 1.87 nm | Sheep nasal mucosa (ex vivo); Wistar rats | IN | ↑ BBB penetration and brain levels vs. free drug (in vivo); |
DRUG![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CHOL CHOL![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PC PC |
PDI: 0.21 ± 0.01 | ↑ Penetration (ex vivo) | |||
Ratio: 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 75 75 |
ZP: −43.47 ± 2.59 mV | ↑ Phosphatidylcholine → ↓ particle size | |||
| EE: 85.50% ± 1.07% | |||||
| N/A80 | Positively charged | Positively charged | Rat brain tissue slices, human brain cortex (ex vivo) | IN | Neutral PEGylated |
DOTAP![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DOPC DOPC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CHOL CHOL![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DSPE-PEG2000 DSPE-PEG2000 |
Size: 80.1 ± 0.4 nm | ↑ AUC in the forebrain, hindbrain, spinal cord, and trigeminal nerve vs. PEGylated positive and negative liposomes | |||
Ratio: 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5 0.5 |
PDI: 0.234 ± 0.009 | ↑ AUC vs. to non-PEGylated neutral liposomes in all tissues examined | |||
| ZP: + 28.8 ± 0.50 mV | ↓ AUC in plasma compared to other formulations | ||||
| Neutral liposome (PEGylated) | Neutral liposome (PEGylated) | Male mice (of the Deutschland, Denken, and Yoken (ddY) strains) | PEGylated and charged | ||
DOPC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CHOL CHOL![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DSPE-PEG2000 DSPE-PEG2000 |
Size: 89.7 ± 2.3 nm | +Charge | |||
Ratio: 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5 0.5 |
PDI: 0.256 ± 0.004 | ↑ AUC in the olfactory bulb compared to other liposomes. | |||
| Neutral (non-PEGylated) | ZP: −18.8 ± 0.38 mV | − Charge | |||
DOPC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CHOL CHOL![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DSPE-PEG2000 DSPE-PEG2000 |
Neutral (non-PEGylated) | ↑ AUC compared to positive liposomes in the trigeminal nerve and a slight ↑ in plasma | |||
Ratio: 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 0 |
Size: 84.0 ± 0.9 nm | ||||
| PDI: 0.222 ± 0.028 | |||||
| ZP: −7.8 ± 0.17 mV | |||||
| Negative liposome | Negative liposome | ||||
DOPC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DOPS DOPS![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CHOL CHOL![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DSPE-PEG2000 DSPE-PEG2000 |
Size: 86.4 ± 1.1 nm | ||||
Ratio:1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5 0.5 |
PDI: 0.260 ± 0.010 | ||||
| ZP: −39.3 ± 1.14 mV | |||||
| Risperidone81 | [LP-16] | Size: 98.51 ± 6.82 nm | Wistar Albino rats | IN vs. IV | Liposomal formulations |
SPC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CHOL CHOL![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MPEG-DSPE MPEG-DSPE |
ZP: -28.60 ± 3.62 mV | ↑ Brain exposure | |||
Ratio: 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.20 0.20 |
EE: 58.86% ± 1.38% | ↓ Plasma levels vs. IV drug | |||
| PEGylated LP-16 | |||||
| ↑ Risperidone absorption vs. conventional liposomes |
4. Physiological changes in pregnancy: structural remodeling, implications for maternal-fetal transfer, and drug pharmacokinetics
To understand how IN drug delivery may reduce fetal exposure compared to oral or IV administration, it is essential to consider two interacting processes: (i) gestation-dependent placental barrier remodeling and (ii) pregnancy-induced alterations in systemic PK that govern maternal-fetal drug transfer. In the context of CNS-targeted therapies, these processes determine how route of administration influences maternal plasma exposure, placental transfer, and ultimately fetal drug burden. Pregnancy triggers a cascade of anatomical and physiological adaptations that evolve across gestation and affect therapeutic performance, particularly for drugs with narrow therapeutic indices such as VPA. In this section, we examine placental barrier remodeling and systemic pharmacokinetics, with specific emphasis on how these factors differentially impact systemic (oral/IV) versus IN delivery strategies.4.1 Placental permeability across gestation
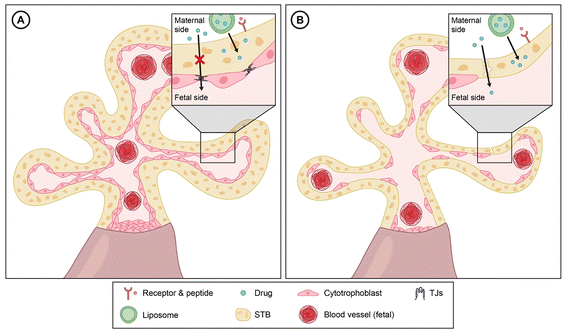
The human placenta is a transient, multifunctional organ that develops early in gestation to mediate the bidirectional exchange of gases, nutrients, metabolic waste, hormones, and immune factors between maternal and fetal circulations. Beyond its endocrine functions, it acts as a dynamic barrier regulating fetal exposure to exogenous compounds, including therapeutics. Maternal-fetal transfer occurs via passive (transcellular or paracellular) diffusion, facilitated diffusion, active transport by membrane-bound transporters, and endocytosis or transcytosis.87 Structurally, the human hemochorial placenta consists of chorionic villi lined by a continuous syncytiotrophoblast (STB) layer bathed in maternal blood. Beneath the STB lie cytotrophoblasts and fetal capillary endothelial cells. In early gestation, the trophoblast barrier is multilayered and reinforced by tight junctions (TJs), including proteins such as occludin and ZO-1, that confer high paracellular resistance.88,89 As gestation progresses, the placenta undergoes extensive morphological remodeling: the STB thins into a monolayer, fetal capillaries approach the villous surface, and TJ localization shifts primarily to the fetal endothelium (Fig. 2).Despite this anatomical thinning, passive diffusion remains restricted. Perfusion studies show that mid-sized molecules (40–70 kDa), such as horseradish peroxidase and dextrans, can cross the term placenta, likely via structural discontinuities in the STB, providing syncytial breaks and fibrin deposits.87,90 Recent imaging studies using three-dimensional electron microscopy have identified millions of trans-syncytial nanopores (TSNs) spanning the STB, providing a size-selective pathway for solute passage.91 Importantly, these placental transfer mechanisms are primarily engaged by drugs present in the maternal systemic circulation; delivery strategies that limit maternal plasma exposure, such as IN administration, are therefore expected to reduce interaction with placental transport pathways. Animal studies further illustrate the dynamic nature of placental permeability. In rats, paracellular pore diameters are wider at mid-gestation and narrows near term, indicating a gestation-dependent modulation of barrier properties.88,91
Collectively, these findings highlight that fetal drug exposure is closely tied to maternal plasma concentrations and placental engagement, reinforcing the importance of delivery strategies that minimize systemic exposure when fetal sparing is desired.
4.2 Pharmacokinetics alterations during pregnancy and implications for VPA exposure
Understanding placental remodeling is essential for the rational design of therapeutics aimed at modulating maternal-fetal transfer. However, successful implementation also requires consideration of broader PK changes during pregnancy, which significantly affect drug absorption, distribution, metabolism, and excretion. Pregnancy induces profound physiological adaptations that reshape the PK and pharmacodynamics (PD), altering drug efficacy and fetal exposure.One of the most pronounced changes is plasma volume expansion, which increases progressively across gestation, resulting in hemodilution and reduced total plasma drug concentrations.92 Concurrently, serum albumin levels decline,93 reducing protein-binding of highly bound drugs, such as VPA. Hepatic metabolism is also altered, with upregulation of uridine glucuronyl transferases (UGTs), the primary enzymes mediating VPA clearance, thereby accelerating elimination.94,95 Clinical data indicate that while total VPA plasma concentrations decline during pregnancy, free VPA levels remain relatively stable or may even increase near term due to reduced protein binding.96–99 As a result, free VPA concentrations are considered a more reliable indicator of fetal exposure than total levels, although they are infrequently monitored in clinical practice.100,101
VPA also displays substantial inter-individual variability in plasma concentrations, with variability more pronounced at lower doses.100,102,103 Importantly, higher cord blood VPA concentrations have been inversely associated with neonatal birth length, independent of maternal dose,104,105 highlighting the clinical relevance of maternal PK variability for fetal outcomes. These findings emphasize the need for individualized therapeutic drug monitoring during pregnancy.
Crucially, the impact of these physiological changes differs depending on the route of administration. In the context of oral or IV delivery, pregnancy-associated alternations in plasma volume, protein binding, hepatic metabolism, and placental permeability directly influence maternal systemic exposure and fetal drug transfer. By contrast, IN delivery is expected to partially decouple CNS exposure from systemic PK by enabling direct nose-to-brain transport. As a result, maternal plasma concentrations, and consequently, placental transfer, may be substantially lower following IN administration compared with oral or IV routes, even when therapeutically relevant CNS concentrations are maintained.
Despite these insights, almost nothing is known about the PK of IN-administered drugs during pregnancy, and no systematic studies have evaluated trimester-specific effects on absorption, distribution, or metabolism. The most widely used IN therapies in pregnancy are corticosteroids, whose safety evaluations have focused on congenital malformations rather than PK, with no increased risk compared to non-exposed pregnancies.106 Limited data from IN-administered fentanyl during labor suggests lower maternal plasma concentrations compared with non-pregnant adults, with detectable but reduced fetal exposure.99,107 These observations support the premise that IN delivery may confer PK advantages during pregnancy, but dedicated studies are urgently needed to define gestation-specific effects and optimize dosing strategies for fetal-sparing CNS therapies.
Altogether, these adaptations complicate prediction of maternal and fetal drug exposure, particularly for drugs with narrow therapeutic indices such as VPA or those delivered via alternative routes like IN administration. Dedicated studies are urgently needed to define gestation-specific effects on the PK of IN therapeutics, to enable optimized dosing that ensures maternal efficacy while safeguarding fetal development.
5. Nanocarriers for drug delivery during pregnancy
Building on the placental physiology and pregnancy-specific PK considerations outlined in Section 4, this section focuses on systemically administered nanocarriers, primarily liposomes, that have been empirically evaluated in pregnancy models. The aim is to identify formulation features associated with restricted placental and fetal exposure once a nanocarrier is present in maternal circulation, and to define the limitations of systemic nanomedicine when fetal protection is the primary therapeutic objective. IN delivery strategies, which rely on distinct transport mechanisms and bypass systemic circulation, are discussed separately in Section 3.To evaluate which liposomal parameters restrict or permit placental transfer following systemic administration, we reviewed selected lipid-based nanoformulations tested in pregnancy models. Although several liposomal systems have been reported, many lack critical physiochemical or biological characterization (e.g., ZP, EE, or release profiles), limiting meaningful comparison. Therefore, only formulations with sufficient detail were included. Two reports describing placental or fetal localization were excluded due to inadequate validation or unclear biological significance. The intent of this analysis is not to provide an exhaustive catalog, but to highlight formulation trends and persistent knowledge gaps.
The formulations summarized in Table 3 were evaluated in pregnant mice, most commonly at gestational day 16. Across these studies, none of the listed nanoformulations crossed the placental barrier or localized within the fetus, despite differences in lipid composition or sterol content. Reported formulations generally shared a narrow physicochemical range, with particle sizes between 68 and 102 nm, high EE (≥90%), and near-neutral surface charge. While these characteristics are associated with limited fetal localization in the reported models, the absence of direct comparative studies constrains broader interpretation. Notably, variations in sterol type or abundance within formulations did not alter the lack of transplacental passage observed under these conditions.108 A small number of systemically administered liposomal formulations have been engineered to preferentially localize to the placenta following intravenous delivery in pregnancy models. While informative, these placenta-targeting approaches fall outside the primary scope of this review, which focuses on strategies, particularly IN delivery, that minimize systemic exposure and avoid direct placental engagement.109 In a complementary approach, liposomal encapsulation has been used to prevent fetal exposure to drugs known to be harmful during pregnancy. Refuerzo et al. demonstrated that indomethacin encapsulated in liposomes and administered intravenously to pregnant mice on gestational day 18 remained restricted to the maternal compartment without detectable fetal transfer.110 Such strategies are particularly relevant when treatment discontinuation is neither feasible nor safe, as in the case of valproic acid, where withdrawal during pregnancy is generally discouraged despite teratogenic risk.66 Importantly, systemically administered nanocarriers do not eliminate maternal plasma exposure, even when fetal accumulation is prevented. Following intravenous delivery, nanoparticles remain subject to maternal circulation, hepatic uptake, and interaction with the placental barrier. As a result, placental exposure may still occur, particularly for drugs with narrow therapeutic indices. This limitation distinguishes systemic nanocarrier approaches from IN delivery, which offers a mechanistically distinct strategy by enabling direct nose-to-brain transport with reduced reliance on sustained maternal plasma concentrations. For this reason, IN liposomal delivery represents a complementary approach when minimizing fetal exposure is a primary therapeutic goal.
| Ionizable lipid | Phospholipid | Sterol | Lipid-PEG | Molar ratio | Size (nm) | Zeta potential (mV) | Encapsulation Efficiency (%) | PDI | Encapsulated molecule |
|---|---|---|---|---|---|---|---|---|---|
C12-494![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 111 111 |
DOPE | Cholesterol | C14-PEG2K | 35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 46.5 46.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 2.5 |
101.6 ± 0.3 | −7.0 ± 2.1 | 95.9 ± 0.6 | 0.19 ± 0.02 | Indomethacin |
C12-494![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 111 111 |
DOPE | Campesterol | C14-PEG2K | 35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 46.5 46.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 2.5 |
91.7 ± 0.4 | −8.4 ± 1.9 | 94.6 ± 0.2 | 0.24 ± 0.02 | Indomethacin |
C12-494![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 111 111 |
DOPE | β-Sitosterol | C14-PEG2K | 35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 46.5 46.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 2.5 |
93.0 ± 0.6 | −7.6 ± 0.7 | 94.2 ± 0.6 | 0.18 ± 0.02 | Indomethacin |
C12-494![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 111 111 |
DOPE | Stigmastanol | C14-PEG2K | 35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 46.5 46.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 2.5 |
92.1 ± 0.8 | −7.4 ± 1.8 | 94.7 ± 0.1 | 0.13 ± 0.02 | Indomethacin |
C12-494![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 111 111 |
DOPE | Cholesterol | C14-PEG2K | 35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 31.5 31.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 2.5 |
68.1 ± 1.3 | N/A | 90.0 ± 3.5 | 0.13 ± 0.02 | Indomethacin |
C12-494![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 111 111 |
DOPE | Cholesterol | C14-PEG2K | 35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 61.5 61.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 2.5 |
77.0 ± 0.3 | N/A | 90.0 ± 1.5 | 0.14 ± 0.02 | Indomethacin |
C12-494![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 112 112 |
DOPE | Cholesterol | C14-PEG2K | 35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 46.5 46.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 2.5 |
72.9 ± 3.4 | 4.07 ± 0.95 | 94.9 ± 0.3 | 0.36 ± 0.01 | mCherry mRNA |
C12-200![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 113 113 |
DOPE | Cholesterol | C14-PEG2K | 35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 46.5 46.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 2.5 |
∼80 | — | — | — | Luciferase mRNA |
Notably, until recently, no IN administered nanoformulations had been evaluated in pregnancy models. A newly published study has now examined IN delivery of a teratogenic dose (1250 mg kg−1 day−1) of niosome-formulated pregabalin in epileptic PTZ-treated pregnant rats, comparing this approach with oral pregabalin and standard diazepam administration. IN nano-delivery resulted in enhanced antiepileptic efficacy and, importantly, prevented congenital malformations relative to systemic treatment modalities.86 However, this study did not directly assess maternal plasma concentrations, placental transfer, or fetal drug levels. Accordingly, while these findings provide the first functional evidence supporting the potential of IN nanotherapeutic delivery during pregnancy, all formulations summarized in Table 3 remain grounded in systemic pharmacokinetic paradigms. The lack of quantitative maternal-fetal exposure data following IN administration remains a key limitation and motivates future pregnancy-specific pharmacokinetic studies. Given the structural and functional similarities between niosomes and liposomes,114 these observations nevertheless provide indirect support for the translational relevance of IN liposomal platforms in pregnancy.
6. Conclusions and future directions
The widespread use of ASMs among women of reproductive age continues to raise critical concerns regarding fetal safety. VPA remains indispensable for specific refractory epilepsy syndromes, yet its clinical utility is constrained by well-established teratogenic risks that are closely linked to systemic maternal exposure and placental transfer. Pregnancy-associated physiological changes further complicate PK, highlighting the need for delivery strategies that preserve maternal efficacy while limiting fetal exposure. In this context, IN liposomal delivery offers a compelling alternative to systemic or oral administration. By enabling direct brain targeting while reducing dependence on sustained maternal plasma concentrations, IN delivery is expected, based on established PK principles, to lower maternal Cmax and systemic AUC. Given that the fetal exposure is primarily driven by free drug levels in maternal plasma, this shift in exposure profile provides a mechanistic rationale for reduced placental transfer, even in the absence of direct fetal PK measurements. From a translational perspective, liposomes represent a mature and scalable drug-delivery platform. Established manufacturing strategies, including ethanol or solvent injection, high-pressure homogenization, extrusion-based size control, and continuous-flow or microfluidic mixing, are already implemented at clinical and commercial scale under GMP conditions for approved liposomal formulations.115–118 These paradigms support the feasibility of scaling IN liposomal systems for clinical translation.Importantly, the relevance of safer VPA delivery strategies extends well beyond epilepsy. VPA is widely prescribed for additional neurological and psychiatric indications, including bipolar disorder, migraine prophylaxis, and mood stabilization, substantially expanding potential exposure across the general population, including women who may become pregnant unintentionally. This broader clinical use further heightens the public health imperative for delivery approaches that decouple central nervous system efficacy from systemic and fetal exposure. More broadly, IN liposomal delivery may provide a generalizable framework for maternal-restricted CNS targeting, with potential applicability to other ASM and neuropsychiatric therapies used during pregnancy. By shifting maternal-fetal care from risk avoidance toward precision-guided delivery, such strategies may ultimately reduce fetal exposure while ensuring access to effective, evidence-based neurological treatment. Versatile platform with far-reaching implications. From a public health perspective, reducing fetal exposure through precision delivery has the potential to lower the incidence of congenital malformations, decrease long-term healthcare costs, and support regulatory pathways for the safer use of essential but teratogenic medications. More broadly, this approach reflects a shift in maternal-fetal therapeutics towards precision-guided delivery strategies that preserve maternal efficacy while minimizing fetal risks.
Author contributions
B. C. conceived the review, led the manuscript preparation, and wrote the main text. L. C. Z., F. M., and T. P. contributed equally to the literature review, manuscript drafting, and the preparation of tables and figures, each providing deep input throughout the process. R. H. F. contributed to the discussion. F. T. contributed equally with B. C. in conceptual guidance and manuscript development. All authors reviewed and approved the final version of the manuscript.Conflicts of interest
The authors declare no conflict of interest.Abbreviations
| ASM | Antiseizure medication |
| BBB | Blood-brain barrier |
| CNS | Central nervous system |
| GGE | Genetic generalized epilepsy |
| HDAC | Histone deacetylase |
| IN | Intranasal |
| IV | Intravenous |
| IP | Intraperitoneal |
| JME | Juvenile myoclonic epilepsy |
| MES | Maximum electroshock seizure |
| NLC | Nanostructured lipid carrier |
| NP | Nanoparticle |
| PD | Pharmacodynamics |
| PEG | Polyethylene glycol |
| P-gp | P-glycoprotein |
| PGTCS | Primary generalized tonic-clonic seizures |
| PK | Pharmacokinetics |
| STB | Syncytiotrophoblast |
| TDM | Therapeutic drug monitoring |
| MPS | Mononuclear phagocyte system |
| TJ | Tight junction |
| TSN | trans-Syncytial nanopores |
| VPA | Valproic acid |
| VANE | VPA nanoemulsion |
| PDI | Polydispersity index |
| MPEG-DSPE | Methoxy polyethylene glycol-distearoyl phosphatidylethanolamine |
Data availability
Data sharing is not applicable to this article as no new data were generated or analyzed. The data supporting the findings of this review are available within the article and cited in the references.Acknowledgements
The authors gratefully acknowledge Joanne Park for her valuable contribution to the design and preparation of the graphical elements included in this review. Her expertise in scientific visualization greatly enhanced the clarity and impact of the figures.References
- T. Tomson, D. Battino and E. Perucca, Valproic acid after five decades of use in epilepsy: time to reconsider the indications of a time-honoured drug, Lancet Neurol., 2016, 15, 210–218 CrossRef CAS PubMed.
- F. J. E. Vajda, S. R. L. Vajda and M. J. Eadie, Avoiding the use of valproate, Discovery Med., 2024, 1, 153 Search PubMed.
- C. T. Schmidt, et al., Transfer of anticonvulsants and lithium into amniotic fluid, umbilical cord blood & breast milk: A systematic review & combined analysis, Prog. Neuropsychopharmacol. Biol. Psychiatry, 2023, 124, 110733 CrossRef CAS PubMed.
- Statement on the risks associated with use of valproic acid (sodium valproate) in women and girls of childbearing potential. https://www.who.int/news/item/02-05-2023-use-of-valproic-acid-in-women-and-girls-of-childbearing-potential.
- C. Rathore, K. Y. Patel and P. Satishchandra, Current Concepts in the Management of Idiopathic Generalized Epilepsies, Ann. Indian Acad. Neurol., 2022, 25, 35 CrossRef PubMed.
- M. Romoli, et al., Valproic Acid and Epilepsy: From Molecular Mechanisms to Clinical Evidences, Curr. Neuropharmacol., 2019, 17, 926–946 CrossRef CAS PubMed.
- D. J. Bond, R. W. Lam and L. N. Yatham, Divalproex sodium versus placebo in the treatment of acute bipolar depression: a systematic review and meta-analysis, J. Affect. Disord., 2010, 124, 228–234 CrossRef CAS PubMed.
- M. Linde, W. M. Mulleners, E. P. Chronicle and D. C. McCrory, Valproate (valproic acid or sodium valproate or a combination of the two) for the prophylaxis of episodic migraine in adults, Cochrane Database Syst. Rev., 2013, 2013, CD010611 Search PubMed.
- A. Wahab and A. Iqbal, Black-Box Warnings of Antiseizure Medications: What is Inside the Box?, Pharm. Med., 2023, 37, 233–250 CrossRef PubMed.
- M. Rahman, A. O. Awosika and H. Nguyen, Valproic Acid, in StatPearls, StatPearls Publishing, Treasure Island (FL), 2025 Search PubMed.
- R. G. Dickinson, W. D. Hooper, P. R. Dunstan and M. J. Eadie, Urinary Excretion of Valproate and Some Metabolites in Chronically Treated Patients, Ther. Drug Monit., 1989, 11, 127 CrossRef CAS PubMed.
- N. A. Shnayder, et al., Therapeutic and Toxic Effects of Valproic Acid Metabolites, Metabolites, 2023, 13, 134 CrossRef CAS PubMed.
- M. F. B. Silva, et al., Valproic acid metabolism and its effects on mitochondrial fatty acid oxidation: A review, J. Inherited Metab. Dis., 2008, 31, 205–216 CrossRef CAS PubMed.
- S. Chateauvieux, et al., Valproic acid perturbs hematopoietic homeostasis by inhibition of erythroid differentiation and activation of the myelo-monocytic pathway, Biochem. Pharmacol., 2011, 81, 498–509 Search PubMed.
- W. Tang and F. S. Abbott, Bioactivation of a Toxic Metabolite of Valproic Acid, (E)-2-Propyl-2,4-pentadienoic Acid, via Glucuronidation. LC/MS/MS Characterization of the GSH−Glucuronide Diconjugates, Chem. Res. Toxicol., 1996, 9, 517–526 Search PubMed.
- S. Sepahi, B. Riahi-Zanjani and A. Ghorani-Azam, Effect of valproic acid on metabolic status and endocrine system in pediatric patients with epilepsy: systematic literature review, Rev. Clin. Med., 2017, 4, 7–13 Search PubMed.
- B. R. Jacob, T. B. Piorczynski and J. M. Hansen, 285 - VPA Inhibits P19 Neural Differentiation through Redox Dysregulation and Oxidative Stress, Free Radicals Biol. Med., 2017, 112, 190 CrossRef.
- A. P. Kudin, et al., Mitochondrial Liver Toxicity of Valproic Acid and Its Acid Derivatives Is Related to Inhibition of α-Lipoamide Dehydrogenase, Int. J. Mol. Sci., 2017, 18, 1912 CrossRef PubMed.
- T. N. Ferraro and R. J. Buono, The relationship between the pharmacology of antiepileptic drugs and human gene variation: An overview, Epilepsy Behav., 2005, 7, 18–36 CrossRef PubMed.
- P. C. Ho, F. S. Abbott, U. M. Zanger and T. K. H. Chang, Influence of CYP2C9 genotypes on the formation of a hepatotoxic metabolite of valproic acid in human liver microsomes, Pharmacogenomics J., 2003, 3, 335–342 CrossRef CAS PubMed.
- M.-M. Zhu, et al., The pharmacogenomics of valproic acid, J. Hum. Genet., 2017, 62, 1009–1014 CrossRef CAS PubMed.
- F. Gofflot, et al., In vitro neuroteratogenicity of valproic acid and 4-en-VPA, Neurotoxicol. Teratol., 1995, 17, 425–435 CrossRef CAS PubMed.
- P. B. M. Luís, et al., Valproyl-CoA inhibits the activity of ATP- and GTP-dependent succinate
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CoA ligases, J. Inherited Metab. Dis., 2014, 37, 353–357 CrossRef PubMed.
CoA ligases, J. Inherited Metab. Dis., 2014, 37, 353–357 CrossRef PubMed. - A. J. Copp and N. D. E. Greene, Neural tube defects–disorders of neurulation and related embryonic processes, Wiley Interdiscip. Rev.:Dev. Biol., 2013, 2, 213–227 CrossRef CAS PubMed.
- G. Pietrogrande, et al., Valproic acid-induced teratogenicity is driven by senescence and prevented by Rapamycin in human spinal cord and animal models, Mol. Psychiatry, 2025, 30, 986–998 CrossRef CAS PubMed.
- J. W. Steele, et al., Embryonic Hypotaurine Levels Contribute to Strain-Dependent Susceptibility in Mouse Models of Valproate-Induced Neural Tube Defects, Front. Cell Dev. Biol., 2022, 10, 832492 CrossRef PubMed.
- A. Hansler, et al., Untargeted metabolite profiling of murine embryos to reveal metabolic perturbations associated with neural tube closure defects, Birth Defects Res. A Clin. Mol. Teratol., 2014, 100, 623–632 Search PubMed.
- D. Akimova, et al., Metabolite profiling of whole murine embryos reveals metabolic perturbations associated with maternal valproate-induced neural tube closure defects, Birth Defects Res., 2017, 109, 106–119 Search PubMed.
- T. Tomson, et al., Withdrawal of valproic acid treatment during pregnancy and seizure outcome: Observations from EURAP, Epilepsia, 2016, 57, e173–e177 Search PubMed.
- A.Ç Atalar, N. G. Şirin, N. Bebek and B. Baykan, Predictors of successful valproate withdrawal in women with epilepsy, Epilepsy Behav., 2021, 119, 107980 CrossRef PubMed.
- A. Yuwanda, S. Surini, Y. Harahap and M. Jufri, Study of valproic acid liposomes for delivery into the brain through an intranasal route, Heliyon, 2022, 8, e09030 CrossRef CAS PubMed.
- S. Eskandari, J. Varshosaz, M. Minaiyan and M. Tabbakhian, Brain delivery of valproic acid via intranasal administration of nanostructured lipid carriers: in vivo pharmacodynamic studies using rat electroshock model, Int. J. Nanomed., 2011, 6, 363–371 CAS.
- S. F. Tan, B. P. Kirby, J. Stanslas and H. B. Basri, Characterisation, in vitro and in vivo evaluation of valproic acid-loaded nanoemulsion for improved brain bioavailability, J. Pharm. Pharmacol., 2017, 69, 1447–1457 CrossRef CAS PubMed.
- M. Meenu, K. H. Reeta, A. K. Dinda, S. K. Kottarath and Y. K. Gupta, Evaluation of sodium valproate loaded nanoparticles in acute and chronic pentylenetetrazole induced seizure models, Epilepsy Res., 2019, 158, 106219 CrossRef CAS PubMed.
- Y. Ikeda, Y. Tajika and Y. Nagasaki, Design of self-assembling anti-epileptic drug for long-acting drug delivery in vivo, Biomater. Sci., 2022, 10, 6307–6314 RSC.
- J. Darius, F. P. Meyer, B. A. Sabel and U. Schroeder, Influence of Nanoparticles on the Brain-to-serum Distribution and the Metabolism of Valproic Acid in Mice, J. Pharm. Pharmacol., 2000, 52, 1043–1047 CrossRef CAS PubMed.
- A. Hussain, M. A. Altamimi, M. Ramzan, M. A. Mirza and T. Khuroo, GastroPlus- and HSPiP-Oriented Predictive Parameters as the Basis of Valproic Acid-Loaded Mucoadhesive Cationic Nanoemulsion Gel for Improved Nose-to-Brain Delivery to Control Convulsion in Humans, Gels, 2023, 9, 603 CrossRef CAS PubMed.
- A. C. Correia, et al., Design of experiment (DoE) of mucoadhesive valproic acid-loaded nanostructured lipid carriers (NLC) for potential nose-to-brain application, Int. J. Pharm., 2024, 664, 124631 CrossRef CAS PubMed.
- S. Al-Enazy, S. Ali, N. Albekairi, M. El-Tawil and E. Rytting, Placental control of drug delivery, Adv. Drug Delivery Rev., 2017, 116, 63–72 CrossRef CAS PubMed.
- I. Drath, F. Richter and M. Feja, Nose-to-brain drug delivery: from bench to bedside, Transl. Neurodegener., 2025, 14, 23 CrossRef PubMed.
- J. Koo, C. Lim and K. T. Oh, Recent Advances in Intranasal Administration for Brain-Targeting Delivery: A Comprehensive Review of Lipid-Based Nanoparticles and Stimuli-Responsive Gel Formulations, Int. J. Nanomed., 2024, 19, 1767–1807 CrossRef CAS PubMed.
- M. Agrawal, Nose-to-brain drug delivery_ An update on clinical challenges and progress towards approval of anti-Alzheimer drugs, J. Controlled Release, 2018, 281, 139–177 Search PubMed.
- W. M. Pardridge, Treatment of Alzheimer's Disease and Blood–Brain Barrier Drug Delivery, Pharmaceuticals, 2020, 13, 394 CrossRef CAS PubMed.
- E. M. Delhaas and F. J. P. M. Huygen, Complications associated with intrathecal drug delivery systems, BJA Educ., 2020, 20, 51–57 Search PubMed.
- M. Helwany and B. Bordoni, Neuroanatomy, Cranial Nerve 1 (Olfactory), in StatPearls, StatPearls Publishing, Treasure Island (FL), 2025 Search PubMed.
- T. P. Crowe and W. H. Hsu, Evaluation of Recent Intranasal Drug Delivery Systems to the Central Nervous System, Pharmaceutics, 2022, 14, 629 Search PubMed.
- S. Cunha, B. Forbes, J. M. S. Lobo and A. C. Silva, Improving Drug Delivery for Alzheimer's Disease Through Nose-to-Brain Delivery Using Nanoemulsions, Nanostructured Lipid Carriers (NLC) and in situ Hydrogels, Int. J. Nanomed., 2021, 16, 4373–4390 CrossRef PubMed.
- A. C. Ottaiano, T. d. A. L. Freddi and L. L. Lucio, The Olfactory Nerve: Anatomy and Pathology, Semin. Ultrasound CT MRI, 2022, 43, 371–377 CrossRef PubMed.
- M. L. Formica, et al., On a highway to the brain: A review on nose-to-brain drug delivery using nanoparticles, Appl. Mater. Today, 2022, 29, 101631 CrossRef.
- T. Huff, L. J. Weisbrod and D. T. Daly, Neuroanatomy, Cranial Nerve 5 (Trigeminal), in StatPearls, StatPearls Publishing, Treasure Island (FL), 2025 Search PubMed.
- Pfizer. Double-Blind, Randomized, Placebo-Controlled, Safety and Efficacy Trial of BHV-3500 (Zavegepant) Intranasal for the Acute Treatment of Migraine. https://clinicaltrials.gov/study/NCT04571060 ( 2023).
- L. Powell, et al., Reduction of pain and functional disability over time in patients treated with zavegepant: a post-hoc analysis of the BHV3500-301 phase 3 randomized controlled trial, J. Headache Pain, 2025, 26, 1 Search PubMed.
- Q. Huang, X. Chen, S. Yu, G. Gong and H. Shu, Research progress in brain-targeted nasal drug delivery, Front. Aging Neurosci., 2024, 15, 1341295 CrossRef PubMed.
- N. E. Davidovich, Y. Kloog, M. Wolf and D. Elad, Mechanophysical Stimulations of Mucin Secretion in Cultures of Nasal Epithelial Cells, Biophys. J., 2011, 100, 2855–2864 CrossRef PubMed.
- N. N. Porfiryeva, I. I. Semina, I. A. Salakhov, R. I. Moustafine and V. V. Khutoryanskiy, Mucoadhesive and mucus-penetrating interpolyelectrolyte complexes for nose-to-brain drug delivery, Nanomedicine, 2021, 37, 102432 Search PubMed.
- C. Costa, J. N. Moreira, M. H. Amaral, J. M. Sousa Lobo and A. C. Silva, Nose-to-brain delivery of lipid-based nanosystems for epileptic seizures and anxiety crisis, J. Controlled Release, 2019, 295, 187–200 CrossRef CAS PubMed.
- H. H. Sigurdsson, J. Kirch and C.-M. Lehr, Mucus as a barrier to lipophilic drugs, Int. J. Pharm., 2013, 453, 56–64 CrossRef CAS PubMed.
- D. Roth, et al., Structure and function relationships of mucociliary clearance in human and rat airways, Nat. Commun., 2025, 16, 2446 Search PubMed.
- D. Inoue, et al., La relazione tra la clearance nasale del farmaco in vivo e la clearance mucociliare nasale in vitro : applicazione alla previsione dell'assorbimento nasale del farmaco, Eur. J. Pharm. Sci., 2018, 117, 21–26 CrossRef CAS PubMed.
- C. Rigaut, L. Deruyver, J. Goole, P. Lambert and B. Haut, A comprehensive analytical model for predicting drug absorption in the olfactory region: Application to nose-to-brain delivery, Int. J. Pharm., 2025, 674, 125392 CrossRef CAS PubMed.
- L. A. Bors, et al., Modulation of nose-to-brain delivery of a P-glycoprotein (MDR1) substrate model drug (quinidine) in rats, Brain Res. Bull., 2020, 160, 65–73 CrossRef CAS PubMed.
- I. I. Ahmed Juvale, A. A. Abdul Hamid, K. B. Abd Halim and A. T. Che Has, P-glycoprotein: new insights into structure, physiological function, regulation and alterations in disease, Heliyon, 2022, 8, e09777 Search PubMed.
- H. Nsairat, et al., Liposomes: structure, composition, types, and clinical applications, Heliyon, 2022, 8, e09394 CrossRef CAS PubMed.
- G. H. Shin, S. K. Chung, J. T. Kim, H. J. Joung and H. J. Park, Preparation of Chitosan-Coated Nanoliposomes for Improving the Mucoadhesive Property of Curcumin Using the Ethanol Injection Method, J. Agric. Food Chem., 2013, 61, 11119–11126 Search PubMed.
- M. Chamundeeswari, J. Jeslin and M. L. Verma, Nanocarriers for drug delivery applications, Environ. Chem. Lett., 2019, 17, 849–865 CrossRef CAS.
- L. J. Renshall, et al., Targeted Delivery of Epidermal Growth Factor to the Human Placenta to Treat Fetal Growth Restriction, Pharmaceutics, 2021, 13, 1778 CrossRef CAS PubMed.
- M. Niu, et al., Hypoglycemic activity and oral bioavailability of insulin-loaded liposomes containing bile salts in rats: The effect of cholate type, particle size and administered dose, Eur. J. Pharm. Biopharm., 2012, 81, 265–272 CrossRef CAS PubMed.
- S. Basak and T. K. Das, Liposome-Based Drug Delivery Systems: From Laboratory Research to Industrial Production—Instruments and Challenges, ChemEngineering, 2025, 9, 56 Search PubMed.
- H. M. Patel, N. S. Tu
![[z with combining umlaut]](https://www.rsc.org/images/entities/char_007a_0308.gif) el and B. E. Ryman, Inhibitory effect of cholesterol on the uptake of liposomes by liver and spleen, Biochim. Biophys. Acta, Gen. Subj., 1983, 761, 142–151 CrossRef CAS PubMed.
el and B. E. Ryman, Inhibitory effect of cholesterol on the uptake of liposomes by liver and spleen, Biochim. Biophys. Acta, Gen. Subj., 1983, 761, 142–151 CrossRef CAS PubMed. - A. L. Klibanov, K. Maruyama, V. P. Torchilin and L. Huang, Amphipathic polyethyleneglycols effectively prolong the circulation time of liposomes, FEBS Lett., 1990, 268, 235–237 CrossRef CAS PubMed.
- G. Blume and G. Cevc, Molecular mechanism of the lipid vesicle longevity in vivo, Biochim. Biophys. Acta, Biomembr., 1993, 1146, 157–168 CrossRef CAS PubMed.
- R. Gref, et al., Biodegradable Long-Circulating Polymeric Nanospheres, Science, 1994, 263, 1600–1603 CrossRef CAS.
- V. P. Torchilin and V. S. Trubetskoy, Which polymers can make nanoparticulate drug carriers long-circulating?, Adv. Drug Delivery Rev., 1995, 16, 141–155 Search PubMed.
- Q. Shu, J. Wu and Q. Chen, Synthesis, Characterization of Liposomes Modified with Biosurfactant MEL-A Loading Betulinic Acid and Its Anticancer Effect in HepG2 Cell, Molecules, 2019, 24, 3939 CrossRef CAS PubMed.
- E. D. Castañeda-Reyes, M. D. J. Perea-Flores, G. Davila-Ortiz, Y. Lee and E. Gonzalez De Mejia, Development, Characterization and Use of Liposomes as Amphipathic Transporters of Bioactive Compounds for Melanoma Treatment and Reduction of Skin Inflammation, A Review, Int. J. Nanomed., 2020, 15, 7627–7650 CrossRef PubMed.
- Z. Németh, et al., Quality by Design-Driven Zeta Potential Optimisation Study of Liposomes with Charge Imparting Membrane Additives, Pharmaceutics, 2022, 14, 1798 CrossRef PubMed.
- S. Maritim, P. Boulas and Y. Lin, Comprehensive analysis of liposome formulation parameters and their influence on encapsulation, stability and drug release in glibenclamide liposomes, Int. J. Pharm., 2021, 592, 120051 Search PubMed.
- A. K. Al Asmari, Z. Ullah, M. Tariq and A. Fatani, Preparation, characterization, and in vivo evaluation of intranasally administered liposomal formulation of donepezil, Drug Des., Dev. Ther., 2016, 10, 205–215 Search PubMed.
- X. Zheng, et al., Intranasal H102 Peptide-Loaded Liposomes for Brain Delivery to Treat Alzheimer's Disease, Pharm. Res., 2015, 32, 3837–3849 Search PubMed.
- T. Kurano, et al., Nose-to-brain/spinal cord delivery kinetics of liposomes with different surface properties, J. Controlled Release, 2022, 344, 225–234 CrossRef CAS PubMed.
- R. Narayan, et al., Development of risperidone liposomes for brain targeting through intranasal route, Life Sci., 2016, 163, 38–45 CrossRef CAS PubMed.
- S. K. Lai, et al., Rapid transport of large polymeric nanoparticles in fresh undiluted human mucus, Proc. Natl. Acad. Sci. U. S. A., 2007, 104, 1482–1487 CrossRef CAS PubMed.
- Y. Wang, et al., Addressing the PEG Mucoadhesivity Paradox to Engineer Nanoparticles that “Slip” through the Human Mucus Barrier, Angew. Chem., Int. Ed., 2008, 47, 9726–9729 CrossRef CAS PubMed.
- S. K. Lai, et al., Drug carrier nanoparticles that penetrate human chronic rhinosinusitis mucus, Biomaterials, 2011, 32, 6285–6290 CrossRef CAS PubMed.
- S. Giordani, V. Marassi, A. Zattoni, B. Roda and P. Reschiglian, Liposomes characterization for market approval as pharmaceutical products: Analytical methods, guidelines and standardized protocols, J. Pharm. Biomed. Anal., 2023, 236, 115751 CrossRef CAS PubMed.
- A. Gaber, et al., Brain-targeted intranasal pregabalin niosomes for the treatment and prevention of teratogenic congenital malformations in PTZ-induced seizures in pregnant rats without passing placental barrier, Eur. J. Pharmacol., 2025, 1007, 178279 CrossRef CAS PubMed.
- D. Edwards, C. J. P. Jones, C. P. Sibley and D. M. Nelson, Paracellular permeability pathways in the human placenta: A quantitative and morphological study of maternal-fetal transfer of horseradish peroxidase, Placenta, 1993, 14, 63–73 CrossRef CAS PubMed.
- D. Marzioni, Expression of ZO-1 and occludin in normal human placenta and in hydatidiform moles, Mol. Hum. Reprod., 2001, 7, 279–285 CrossRef CAS PubMed.
- J. C. Challier, et al., Junctions and adhesion molecules in first trimester and term human placentas, Cell. Mol. Biol., 2005, 51(Suppl), OL713–OL722 CAS.
- P. Brownbill, et al., Mechanisms of alphafetoprotein transfer in the perfused human placental cotyledon from uncomplicated pregnancy, J. Clin. Invest., 1995, 96, 2220–2226 CrossRef CAS PubMed.
- R. M. Lewis, et al., 3D visualization of trans-syncytial nanopores provides a pathway for paracellular diffusion across the human placental syncytiotrophoblast, iScience, 2022, 25, 105453 CrossRef PubMed.
- S. Aguree and A. D. Gernand, Plasma volume expansion across healthy pregnancy: a systematic review and meta-analysis of longitudinal studies, BMC Pregnancy Childbirth, 2019, 19, 508 CrossRef PubMed.
- J. E. Maher, et al., Albumin levels in pregnancy: a hypothesis—decreased levels of albumin are related to increased levels of alpha-fetoprotein, Early Hum. Dev., 1993, 34, 209–215 CrossRef CAS PubMed.
- Y. Ghodke-Puranik, et al., Valproic acid pathway: pharmacokinetics and pharmacodynamics, Pharmacogenet. Genomics, 2013, 23, 236–241 CrossRef CAS PubMed.
- E. A. Pinheiro and C. S. Stika, Drugs in pregnancy: Pharmacologic and physiologic changes that affect clinical care, Semin. Perinatol., 2020, 44, 151221 CrossRef PubMed.
- M. Koerner, M. Yerby, P. Friel and K. McCormick, Valproic Acid Disposition and Protein Binding in Pregnancy, Ther. Drug Monit., 1989, 11, 228 CrossRef CAS PubMed.
- A. Philbert, B. Pedersen and M. Dam, Concentration of valproate during pregnancy, in the newborn and in breast milk, Acta Neurol. Scand., 1985, 72, 460–463 Search PubMed.
- M. S. Yerby, P. N. Friel and K. McCormick, Antiepileptic drug disposition during pregnancy, Neurology, 1992, 42, 12–16 CAS.
- M. Kokki, et al., Neonatal safety of maternal fentanyl during labour, Br. J. Anaesth., 2015, 115, 636–638 CrossRef CAS PubMed.
- C. Johannessen Landmark, et al., Pharmacokinetic variability of valproate during pregnancy – Implications for the use of therapeutic drug monitoring, Epilepsy Res., 2018, 141, 31–37 CrossRef CAS PubMed.
- T. Tomson, C. J. Landmark and D. Battino, Antiepileptic drug treatment in pregnancy: Changes in drug disposition and their clinical implications, Epilepsia, 2013, 54, 405–414 CrossRef CAS PubMed.
- E. Perucca, Pharmacological and therapeutic properties of valproate: a summary after 35 years of clinical experience, CNS Drugs, 2002, 16, 695–714 CrossRef CAS PubMed.
- C. Johannessen Landmark, et al., Pharmacokinetic variability of valproate in women of childbearing age, Epilepsia, 2017, 58, e142–e146 CrossRef CAS PubMed.
- S. I. Patel and P. B. Pennell, Management of epilepsy during pregnancy: an update, Ther. Adv. Neurol. Disord., 2016, 9, 118–129 CrossRef CAS PubMed.
- I. Kacirova, M. Grundmann and H. Brozmanova, Serum levels of valproic acid during delivery in mothers and in umbilical cord - correlation with birth length and weight, Biomed. Pap., 2015, 159, 569–575 Search PubMed.
- A. H. Alhussien, R. A. Alhedaithy and S. A. Alsaleh, Safety of intranasal corticosteroid sprays during pregnancy: an updated review, Eur. Arch. Otorhinolaryngol., 2018, 275, 325–333 Search PubMed.
- M. Kokki, et al., Pharmacokinetics of intranasal fentanyl in parturient, Br. J. Anaesth., 2015, 115, 635–636 CrossRef CAS PubMed.
- E. Haber, et al., Route of administration-dependent anti-inflammatory effect of liposomal alendronate, J. Contr. Release, 2010, 148, 226–233 Search PubMed.
- A. King, et al., Tumor-homing peptides as tools for targeted delivery of payloads to the placenta, Sci. Adv., 2016, 2, e1600349 CrossRef PubMed.
- J. S. Refuerzo, et al., Liposomes: a nanoscale drug carrying system to prevent indomethacin passage to the fetus in a pregnant mouse model, Am. J. Obstet. Gynecol., 2015, 212, 508.e1–508.e7 CrossRef CAS PubMed.
- H. C. Safford, et al., Probing the Role of Lipid Nanoparticle Elasticity on mRNA Delivery to the Placenta, Nano Lett., 2025, 25, 4800–4808 Search PubMed.
- H. C. Geisler, et al., EGFR-targeted ionizable lipid nanoparticles enhance in vivo mRNA delivery to the placenta, J. Controlled Release, 2024, 371, 455–469 Search PubMed.
- K. L. Swingle, et al., Ionizable Lipid Nanoparticles for In Vivo mRNA Delivery to the Placenta during Pregnancy, J. Am. Chem. Soc., 2023, 145, 4691–4706 CrossRef CAS PubMed.
- H. d. A. C. Kodel, et al., Liposomes and Niosomes: New trends and applications in the delivery of bioactive agents for cancer therapy, Int. J. Pharm., 2025, 668, 124994 CrossRef CAS PubMed.
- J. A. Kulkarni, P. R. Cullis and R. van der Meel, Lipid Nanoparticles Enabling Gene Therapies: From Concepts to Clinical Utility, Nucleic Acid Ther, 2018, 28, 146–157 Search PubMed.
- M. Maeki, et al., A strategy for synthesis of lipid nanoparticles using microfluidic devices with a mixer structure, RSC Adv., 2015, 5, 46181–46185 Search PubMed.
- R. Tenchov, R. Bird, A. E. Curtze and Q. Zhou, Lipid Nanoparticles—From Liposomes to mRNA Vaccine Delivery, a Landscape of Research Diversity and Advancement, ACS Nano, 2021, 15, 16982–17015 Search PubMed.
- B. S. Pattni, V. V. Chupin and V. P. Torchilin, New Developments in Liposomal Drug Delivery, Chem. Rev., 2015, 115, 10938–10966 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |