 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
SideroGuard: overview of siderophore-enhanced nanofiber systems for biofilm prevention and control
Sitalakshmi Thyagarajan *a and
Senthamil Selvi Poongavanam*b
*a and
Senthamil Selvi Poongavanam*b
aDepartment of Bioinformatics, Saveetha School of Engineering, Saveetha Institute of Medical and Technical Sciences (SIMATS), Thandalam, Chennai-602105, Tamil Nadu, India. E-mail: sitalakshmit@gmail.com
bNano biomedicine Lab, Saveetha Medical College and Hospital, Saveetha Institute of Medical and Technical Sciences, Chennai-602105, Tamil Nadu, India
First published on 10th March 2026
Abstract
In recent years, there has been a considerable increase in scientific innovation regarding the prevention of biofilm formation on chronic wounds. Microbial biofilms are aggregates of microbial cells surrounded by a polymer matrix that may or may not adhere to surfaces and can also be present in the tissues or secretions. Free iron involved in biofilm formation delays wound healing and may even be responsible for the destruction of connective tissue. To reduce the availability of free iron at wound sites and thereby prevent biofilm formation, siderophores and their derivatives have become an area of focus. Researchers have developed various techniques for the formation of nanofibers. Electrospinning is an excellent method that has attracted significant interest for biomedical applications. It involves the use of electric fields to produce fine fibers with diameters that can be reduced to the nanometer scale, and it has wide application potential in the field of medicine. This review aims to discuss the importance of electrospun siderophore-based nanofibers as nanohealers for wound biofilm formation.
1. Introduction
A wound is defined as damage to the body's tissues, involving the breaking of the skin or its underlying structures. Wounds vary in severity from minor scrapes to more serious injuries like lacerations, puncture wounds, or burns. The causes of wounds can include accidents, trauma, surgical procedures, or medical conditions.1,2 Over the past few decades, despite advancements in therapies and wound dressing, the prevalence of chronic wounds has continued to rise, particularly due to biofilm formation in the wound regions, especially among populations with conditions like diabetes mellitus or those undergoing immunosuppressive treatments. Chronic wounds fail to heal in the expected time and might take a couple of weeks in some cases.3–6 Alterations in cytokines, growth factors and ECM components at the wound site may also lead to the formation of chronic wounds. Factors like oxidative damage by free radicals, infection, ischemia and the accumulation of necrotic tissue can also lead to wounds that do not heal.7,8 Several studies have demonstrated that chronic wounds have increased levels of microbial load and microbial biofilm formation.9 Biofilm-related issues pose significant challenges in wound healing, and connecting the gap between laboratory research and clinical practice is crucial for improving patient outcomes.10–14 While the field lacks a clear standard of clinical practice, several promising strategies are being explored for biofilm management.15 These strategies can help to study the advantages and limitations of electrospun nanofibers.11,16 This paper highlights the importance of microbial siderophore nanofibers as effective nanohealers for biofilm formation.2. Importance of biofilm in wound healing
Biofilms were first reported as animalcules by Anton von Leeuwenhoek, but were later observed as suitable surfaces that facilitate microbial growth, wherein biological activities were greatly accelerated. Studies on microbial varieties revealed the physical arrangement of the shiny layer that was later named “slime”, has paved the way for biofilm interaction. In 2002, Bill Costerton was the first to coin the term “biofilm”, which he described as a group of microorganisms covered within a self-produced matrix of extracellular polymeric substances (EPSs).17 Extracellular polymeric substances are produced by microorganisms to form a defensive and supportive structure around themselves and are made up of polysaccharides, proteins, nucleic acids, lipids, and other macromolecules. Their composition also varies for microbial species in relation to environmental conditions and the stage of biofilm development. Biofilm mainly protects bacteria from the host's immune response and environmental factors like antibiotics, disinfectants, and dehydration, thus providing a habitat for microorganisms. Its resistance makes it challenging to eradicate bacteria that cause chronic wounds. Microbes that exist in biofilms are more resistant to antibiotics because of gene transfer, which provides intercellular communication and encourages growth. Biofilm bacteria release the toxins and enzymes that damage host tissues and interfere with cellular signaling pathways involved in wound repair. Chronic inflammation caused by persistent infection further delays healing. Biofilm-associated infections are often recalcitrant to conventional treatments. Traditional antibiotics are less effective against biofilm bacteria due to their limited penetration into the biofilm matrix and the presence of dormant cells within the biofilm.17–19Iron plays a vital role in multiple stages of biofilm formation by acting as an important regulator of microbial metabolism, adhesion, quorum sensing, and extracellular polymeric substance (EPS) production. During the initial stages (stages 1–2), iron exists predominantly as ferric ions (Fe3+) bound to host proteins such as transferrin, ferritin, and heme-containing molecules released from damaged tissue. Surface-associated Fe3+ increases microbial attachment, thereby increasing electrostatic interactions between bacterial cell surfaces and host extracellular matrix components. Several pathogens, including Pseudomonas aeruginosa and Staphylococcus aureus, sense iron availability via iron-responsive regulatory systems (e.g., Fur regulon), which upregulate adhesins and surface proteins essential for irreversible attachment.20,21 During stage 3, microcolony formation occurs, which requires iron as a metabolic cofactor for DNA synthesis, respiration, and redox homeostasis. At this stage, bacteria actively secrete siderophores, which are high-affinity chelators that specifically bind Fe3+ from host sources. The iron–siderophore complexes are transported into the bacterial cell through specific outer membrane receptors, supporting rapid cell division and also facilitating interspecies competition since siderophore production limits iron availability to competing microbes, thereby shaping the biofilm community structure.22–24 At stage 4–5, iron availability directly acts on EPS synthesis and biofilm architecture, wherein the elevated levels of Fe3+ stimulate the production of polysaccharides such as alginate, Psl, and Pel, which increase biofilm thickness and mechanical stability. Iron also promotes quorum sensing–mediated gene expression and increases coordinated biofilm behaviors, including virulence factor secretion and stress tolerance. In mature biofilms, iron undergoes redox cycling between Fe3+ and ferrous iron (Fe2+), particularly in chronic wounds. Fe2+ generates reactive oxygen species (ROS), which cause host tissue damage and further impair wound healing, while biofilm bacteria remain protected within the EPS matrix.24 In stage 6, iron limitation is often induced by host sequestration mechanisms or therapeutic chelation, which downregulates EPS synthesis and promotes motility and planktonic behaviour, facilitating dissemination to new sites. Microbial siderophores such as pyoverdine, enterobactin, and desferrioxamine form stable complexes with Fe3+ by removing iron from host proteins. The incorporation of siderophore-based systems into wound dressings exploits this mechanism by sequestering free ferric iron at the wound site, thereby reducing the biofilm-forming bacteria of an essential micronutrient. This iron restriction disrupts biofilm establishment, reduces EPS production, attenuates virulence, and increases the bacterial susceptibility to host immune defence and antimicrobial agents.
The stages of biofilm formation are summarized in Fig. 1. The more mature biofilms are heterogeneous, having characteristics similar to those of multicellular organisms. Studies have reported that the formation of biofilms is mainly attributed to the presence of free iron, which leads to connective tissue destruction. Iron is an essential micronutrient for cellular processes; however, an excess of free iron can pose unique challenges in delaying wound healing.12,13
 | ||
| Fig. 1 Stages of biofilm formation highlighting iron-dependent maturation relevant to chronic wound environments. Reproduced from ref. 9 with permission from Frontiers, copyright 2022. | ||
The incorporation of iron chelators into wound dressing materials may hasten wound healing. Studies have revealed that lactoferrin, an iron chelator in mammals, inhibits the formation of biofilms. Similar reports were obtained with synthetic iron chelators.13 The use of microbial siderophores as iron chelators regulates biofilm formation, and reports have suggested that the incorporation of siderophore-based nanofibers for wound dressings controls bacterial growth and biofilm formation on chronic wounds.25
3. Consensus for management of biofilms
The control of biofilms on the wound sites is complex due to their flexibility and resistance to traditional treatments. The prevention of biofilm is often more effective than treating established biofilms, and involves maintaining good hygiene practices, proper sterilization procedures, and regular cleaning of surfaces to remove biofilm precursors.25 The mechanical methods, such as brushing, scraping, or flushing, physically disrupt biofilms and are effective for biofilms on surfaces like teeth, medical implants, or industrial equipment.15 Chemical agents that are used to break down the biofilm matrix and kill the embedded bacteria include enzymes, surfactants, chelating agents, and disinfectants. However, the selection of an appropriate agent is crucial as these biofilms are resistant to many antimicrobial compounds. Antibiotics or antimicrobial agents are used to target bacteria within the biofilm.13,16 However, the penetration of these agents into the biofilm matrix is limited, and bacteria within biofilms often exhibit increased resistance to antibiotics compared to planktonic (free-floating) bacteria. Combining different approaches, such as physical removal with chemical disruption or antimicrobial therapy, can reduce the risk of resistance development. In some cases, using materials that resist biofilm formation can be beneficial.2,16,17 For example, certain coatings or surface modifications discourage bacterial attachment and biofilm formation. One such modification includes the use of microbial siderophore nanofibers as biofilm inhibitors.17 The microbial siderophores hunt the iron components and disrupt the biofilm formation on the wounds. This biofilm wound care is an effective approach to biofilm management, which enhances the reduction of microbial load and biofilm burden at the wound sites.18,194. Microbial siderophore types and their chemistry
The types of siderophores are based on the chemical group utilization for iron coordination, leading to three primary classes: catecholates, hydroxamates, and (α-hydroxy-) carboxylates, with a fourth “mixed type” incorporating combinations of these groups (Fig. 2). Catecholate siderophores are a class of siderophores mainly produced by bacteria, which contain either mono or dihydroxybenzoic acid residues that are utilized to chelate iron. These residues are obtained from dihydroxybenzoic acid.20,21 The two main types of catecholate siderophores include monocatecholates, where these siderophores contain a single dihydroxybenzoic acid residue used for ferric iron chelation, and dicarboxylate siderophores that feature two dihydroxybenzoic acid residues for ferric iron binding. Enterobactin is the best-known example of catecholate siderophores and belongs to the dicarboxylate class.20,22 The hydroxamate siderophores are another class involved in iron acquisition, characterized by hydroxamic acid groups used for ferric iron chelation. These hydroxamic acids were typically derived from amines like lysine or ornithine. There are two main types of hydroxamate siderophores, namely monohydroxamate and dihydroxamate.23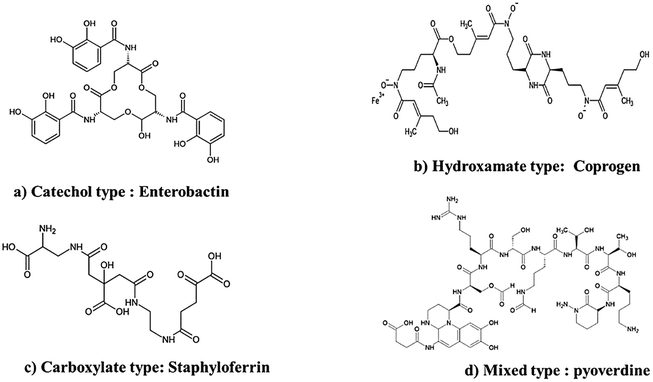 | ||
| Fig. 2 Structural classification of siderophores and their iron-chelating application in biofilm inhibition. | ||
The monohydroxamate siderophores mainly contain a single hydroxamic acid group that is used for iron chelation. These siderophores naturally contain one hydroxamic acid moiety derivative from amino acids such as lysine or ornithine. Monohydroxamate siderophores include ferrichrome, which is produced by various fungi and certain bacteria; for example, the monohydroxamate rhizobactin 1021 was produced by rhizobia bacteria. These siderophores play a vital role in iron acquisition and are vital for the existence and virulence of many microorganisms. The dihydroxamate siderophores contain two hydroxamic acid groups used for iron chelation, which are derived from amino acids such as lysine or ornithine.23 The examples of dihydroxamate siderophores include aerobactin, schizokinen, and alcaligin. Aerobactin is produced by various Gram-negative bacteria and is associated with virulence in certain pathogens. Schizokinen-type siderophores are produced by soil-dwelling plant symbionts, while alcaligin-type siderophores are produced by certain bacteria and are involved in iron acquisition in both host-associated and environmental contexts. The carboxylate type is characterized by the presence of carboxyl functional groups, which act as oxygen donor atoms to bind iron.
The examples of carboxylate siderophores include rhizobactin DM415, staphyloferrin A and B, vibrioferrin, fungal rhizoferrin, and bacterial rhizoferrin.24 The mixed-type siderophores are a different class of siderophores that incorporate different chemical groups for iron chelation. Unlike single-class siderophores, such as catecholates, hydroxamates, or carboxylates, mixed-type siderophores utilize a mixture of these groups in their structures.22,23 This class of siderophores determines the versatility of iron coordination, potentially enhancing their efficacy in iron acquisition. However, specific examples and features of mixed-type siderophores vary widely due to their diverse chemical compositions and structural variations.24
5. Siderophores: natural iron chelators as biofilm inhibitors
Siderophores have been fascinating for several decades of scientific inquiry and discovery. Siderophores were first discovered in the mid-20th century when researchers observed that microorganisms, particularly bacteria and fungi, secreted compounds to scavenge iron from their environment. The term siderophore was coined from “sidero”, meaning iron, and “phore”, meaning carrier, and describes small organic molecules with a high affinity for iron and the ability to acquire iron from their environment. The siderophores bind tightly to iron ions, forming stable complexes that can be observed by the microbes through the specific transport system on the cell membranes, effectively “stealing” iron from their environment.26 The research work by Jacques Monod and Andre Lwoff in the 1950s revealed that the siderophores were produced by microbes to scavenge iron from their surroundings and help them to transport it into the cell, which is very important for their survival and growth.27 As research progressed, the biological significance of siderophores became increasingly evident and was found to play crucial roles in microbial iron acquisition and also in various ecological processes, including competition between microorganisms for limited iron resources.Siderophores have drawn attention to their function in microbial physiology because of their uses in environmental remediation, biotechnology, and medicine. Siderophore-based strategies have been found to improve wound healing applications and function as innovative antimicrobial agents that target the iron metabolism of bacteria.28 Siderophore research has continued for more than a decade with a focus on uncovering novel siderophores, explaining their roles in microbial communities, and discovering their latent therapeutic applications, including as antimicrobial agents or as carriers for controlling biofilm formation. Enterobactin (siderophore) produced by E.coli has been subjected to extensive investigation due to its remarkable ability to bind iron. Its structure features three catecholate units arranged in C3 symmetry, branching from a chiral triserine lactone backbone. The metal-binding units of siderophores play pivotal roles in forming metal-binding complexes, as highlighted by the research studies of Pollack et al. in 1970. The macrocyclic structure of the siderophore backbone was elucidated, and it displays a preorganization that enhances its metal-binding ability, which lowers the pH and reduces proton competition, thereby facilitating stable complex formation. The ability of siderophores to form stable complexes with metals has encouraged researchers to explore their potential in wound applications.28 Certain studies have revealed that these stable complexes deplete the availability of iron in the wound area, paving the way to reducing biofilm formation. By harnessing their metal-binding capabilities, siderophores hold promise for novel approaches in eliminating the microbial load in wound regions and also eradicating biofilm formation.29 Combining electrospinning with siderophores could involve incorporating siderophores into the polymer solution or melting before electrospinning. This would result in the production of nanofibers with siderophores immobilized within the polymer matrix. These nanofibers could be used to capture metal ions from aqueous solutions or as components of wound dressings or tissue-engineering scaffolds with enhanced biofilm-inhibitor properties due to the iron-chelating ability of siderophores. This approach needs a careful analysis of the compatibility between the siderophores and the polymer used for electrospinning, as well as optimization of the electrospinning process to ensure uniform distribution of the siderophores.30
6. Microbial siderophore focus and the targeting of biofilm formation
As iron-chelating molecules, siderophores play an important role in biofilm formation, affecting microbial survival. These molecules scavenge iron from the environment, thus depriving the bacteria in the biofilm of this essential nutrients. By limiting iron availability, siderophores can delay the initial attachment of bacteria to surfaces and consequent biofilm development. At lower concentrations, siderophores primarily inhibit initial bacterial attachment and early biofilm development, while higher concentrations disrupt biofilm maturation, EPS production, and quorum sensing-regulated pathways. Their antibiofilm efficacy is most pronounced under iron-limited conditions, closely resembling host environments. Siderophores also influence bacterial communication systems like quorum sensing, which direct the biofilm formation.31–34 Quorum sensing depends on signaling molecule production and detection to coordinate microbial activities such as biofilm growth. Siderophores disrupt the quorum sensing pathways, affecting the biofilm formation or structure.34By reducing iron availability, siderophores can delay the initial attachment of bacteria to surfaces and consequent biofilm development. At lower concentrations, siderophore inhibit initial bacterial attachment while at higher concentrations they exhibit the disruption of biofilm maturation, EPS production, and quorum sensing–regulated pathways. Their antibiofilm efficacy is most pronounced under iron-limited conditions, closely resembling host environments. Siderophores also influence bacterial communication systems like quorum sensing, which directs the biofilm formation.31–33 Quorum sensing depends on signaling molecule production and detection to co-ordinate microbial activities such as biofilm growth. Siderophore disrupts the quorum sensing pathways, affecting biofilm formation or structure.34
In the host environment, iron is tightly controlled as a part of nutritional immunity to restrict microbial growth. Siderophores produced by pathogenic bacteria can compete with host iron-binding proteins for iron uptake. The spectrum of activity of siderophores is broad but variable. In bacterial biofilm, siderophores can alter the microbial composition and dynamics, causing changes in the biofilm structure and stability. Various reports have indicated that siderophore-mediated iron uptake is mostly responsible for the antibiofilm activities against Gram-negative bacteria such as Pseudomonas aeruginosa, Acinetobacter baumannii, and Escherichia coli. Siderophores affect competition and biofilm stability in polymicrobial biofilms by changing the availability of iron. Bacteria manage the host defenses and promote the production of biofilms in host tissues through siderophore-mediated iron uptake. Interactions among the different microbial species are important for biofilm formation, influencing interspecies interactions by modulating the iron availability and microbial competition within the biofilm community.35
Compared with traditional antibiotics, these siderophores function as anti-virulence agents by limiting iron, thereby killing bacteria and lowering the selective pressure for resistance while exhibiting limited effectiveness against mature biofilms. Siderophores exhibit better biocompatibility and higher iron selectivity than metal chelators like EDTA, which may unintentionally promote the growth of infections that use siderophores. The quorum-sensing inhibitors have a dual effect on iron metabolism and cell–cell signaling, and their activity is influenced by environmental iron levels.
Electrospinning nanofibers incorporated with siderophore compounds have the potential to provide numerous benefits, wherein these nanofibers physically disrupt the structure of biofilms, making them more susceptible to treatment.36 The siderophores present in the nanofibers chelate iron ions, depriving the biofilm-forming bacteria of this essential nutrient and inhibiting their growth and biofilm formation.37,38 By targeting iron chelation, these nanofibers have the potential to interfere with bacterial virulence, reducing their pathogenicity and the severity of infections.38
These siderophores demonstrate a promising strategy for antibiofilm control, particularly when incorporated into nanofiber-based delivery systems and also when used in combination with antibiotics or enzymatic agents. Their ability to target iron-dependent virulence pathways shows a biologically relevant approach to managing biofilm-associated infections and also recognizing the need for optimized dosing and combination therapies. This approach is more valuable in biofilm-related infections, in which the focus is often on conventional antimicrobial therapies due to the protective nature of the biofilm matrix.39
7. Medicated nanofibers for biofilm healing
Medicated nanofibers provide an excellent platform for studying biofilm formation. Their properties make them ideal substrates for mimicking various surfaces encountered in medical settings.40,41 Researchers use these nanofibers to investigate the initial attachment of microbes, kinetics, and the influence of surface topography on biofilm architecture. The advancements in wound dressing strategies and the availability of a wide variety of biomaterials provide the impetus for developing wound dressing materials for our desired needs. Such ideal wound dressing strategies include electrospun nanofibers. Nanofibers have gathered significant interest and found numerous applications in medicine and healthcare due to their unique versatility.41,42Nanofibers offer an innovative solution for all medical diagnostic tools. Their unique, versatile applications hold great promise for addressing unmet medical needs and improving patient outcomes in various healthcare settings.32 The history of nanofibers started in the early 17th–19th centuries, where the formation of fibers was observed when certain materials were subjected to mechanical or electrical forces. However, in the mid-20th century, significant progress was made in the development of polymer fibers, including synthetic polymers such as nylon and polyester.43,44 Techniques for producing macro-scale fibers were refined, laying the groundwork for nanoscale fiber production. Nanofibers can be prepared from a variety of substances, depending on the desired properties and application. Synthetic polymers, like polyacrylonitrile (PAN), polyvinyl alcohol (PVA), polyethylene oxide (PEO), polycaprolactone (PCL), and polylactic acid (PLA), are frequently used due to their flexibility and tunable properties.45,46 Proteins like collagen and gelatin serve as fundamental building blocks in the human body, making up a significant portion of connective tissues such as skin, bones, tendons, and ligaments. They provide structural support, elasticity, and strength to various tissues and organs.47,48 In recent years, collagen has gained significant attention in the field of nanofiber research and applications. Nanofibers made from collagen offer several advantages, including biocompatibility and biomimetic properties, providing an ideal environment for cell adhesion, proliferation, and cell differentiation.49 Certain biopolymers like cellulose, chitosan, and silk fibroin are used for their sustainability.50,51 The production of nanofibers can be achieved through various techniques, and one such recent technique is the application of electrospinning, which offers unique advantages and is suitable for different applications in the field of medicine Fig. 3.51,52
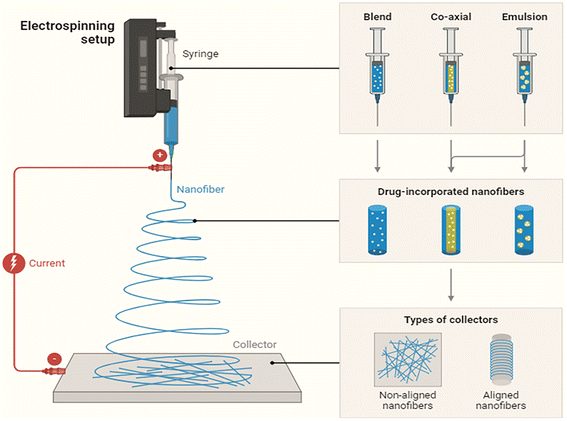 | ||
| Fig. 3 Schematic of the electrospinning-based fabrication of the siderophore-loaded nanofibers for the wound-biofilm control. | ||
8. Fabrication of siderophore–loaded nanofibers
The fabrication of siderophore-loaded nanofibers involves several key steps, which are important in ensuring the successful integration of siderophores into the nanofiber matrix and the production of fibers with desirable properties.53 The choice of polymer is important for the fabrication of siderophore-loaded nanofibers. PVA, PCL, and PEO are commonly used polymers for the fabrication of siderophore-loaded nanofibers through electrospinning, which is a promising technique for biofilm management.32 These fibers are used in medical, industrial, and environmental applications, offering a novel method to prevent biofilm formation.48 The fabrication of siderophore-loaded nanofibers using PCL by electrospinning is a suitable technique for biofilm management. PCL is a biodegradable polymer known for its excellent mechanical properties and biocompatibility, making it suitable for fabricating siderophore-loaded nanofibers for biofilm applications. These fibers have good potential in the field of medical, industrial, and environmental applications and offer a novel method to prevent biofilm formation.41,42Polyethylene oxide (PEO) is a water-soluble polymer that is known for biocompatibility and non-toxicity, making it an excellent candidate for fabricating siderophore-loaded nanofibers for biofilm applications. The fabrication of siderophore-loaded nanofibers using PEO via electrospinning is a promising technique for biofilm management.42
9. Electrospun nanofibers as a nanohealer
Electrospinning has become progressively fascinating in recent decades, by attracting much attention to its versatility and extensive applicability, particularly within disciplines such as biomaterials science, and nanotechnology.54 Electrospinning has its roots in the early 20th century.39,47 In 1902, British engineer Sir William Thomson (also known as Lord Kelvin) patented a method for the electrostatic spinning of fibers. However, it wasn't until the 1930s that further progress was made.35,49 In the 1930s, Anton Formhals, an American physicist, made an important contribution to the progress of electrospinning technology.55,56 He developed a method for producing ultra-fine fibers using electrostatic forces. The research work of Formhal et al. also laid a good foundation for modern electrospinning techniques in the mid-20th century.57,58 During this period, researchers discovered various aspects of the process, including the effects of different parameters such as voltage, polymer solution properties, and collector configurations on fiber morphology.39,60 This development gained importance in the late 1990s and early 2000s in the field of materials science and nanotechnology for various applications.59,61,62 Researchers began to explore the potential of electrospinning to produce nanoscale fibers with detailed control over their properties. In the past few decades, electrospinning has shown rapid progress by developing coaxial electrospinning and near-field electrospinning to enhance the versatility and capabilities of the process.39,63,64 The history of electrospinning shows (Table 1) the journey of investigation, innovation, and technological advancement with each milestone by contributing to our understanding of the process and expanding its potential applications.| Types | Inventors and year | Applications | Ref. |
|---|---|---|---|
| Mono-axial | Cooley and Eastman (1902) | Drug delivery | 52 |
| Co-axial | Sun et al. (2003) | Modelling drug release | 65 |
| Multi-jet | Ding et al. (2004) | Nanosensors | 66 |
| Magnetic field | Yarin et al. (2004) | Textile industries | 67 |
| Roller | Jirsák et al. (2005) | Skin tissue engineering | 68 |
| Porous tube | Dosunmu et al. (2006) | Waste water treatment | 69 |
| Bubble | Liu et al. (2008) | Therapeutic and diagnostic tools | 70 |
| Tri-axial | Kalra et al. (2009) | Loading of multiple Drugs | 71 |
| Ball | Miloh et al. (2009) | Water purification | 72 |
| Disk | Niu et al. (2009) | Drug delivery | 73 |
| Wet 3D | Yokoyama et al. (2009) | Waste water treatment | 74 |
| Cone | Lu et al. (2010) | Biomedical and food applications | 75 |
| Spiral coil | Wang et al. (2012) | Electrodes for batteries | 76 |
| Beaded chain | Liu et al. (2008) | Energy catalysis | 70 |
| Cold plate3D | Sheikh et al. (2015) | Skin damage and diabetic ulcers | 50 |
| 3-Dimensional | Vong et al. (2018) | Cell infiltration and wound healing | 77 |
The electrospinning setup comprises key components such as a syringe pump, a high-voltage power supply, and a metal collecting plate.77,78 The syringe pump directs a consistent flow of polymer solution, while a high voltage generates an electrically charged polymer jet from the nozzle. The setup generates an electric field at the nozzle, typically 1 to 50 kV per meter, initiating the process, and also leads to electrostatic repulsion forces to overcome the liquid's surface tension, collecting high-voltage electrical charges, and the liquid also reforms into a cone formation known as the Taylor cone, expelling a charged jet from its tip.60,77,78 The trajectory of the jet experiences bending variabilities, leading to whipping and oscillation as it progresses. As the jet stretches into thinner diameters, it swiftly solidifies, depositing solid fibers onto a grounded collector plate. Various collector designs, like plates, drums, mandrels, and discs, can be utilized. In some cases, residual solvent may remain in the nanofibrous mat, imposing post-processing adjustments.54,60
The electrospinning process includes charging a liquid droplet with electricity, forming a Taylor cone or cone-shaped jet, extending the jet in a straight path, thinning the jet using an electric field, increasing electrical bending instability (also known as whipping instability), and ultimately solidifying and collecting the jet as solid fibers on a grounded collector The electrospinning process involves charging a liquid droplet with electricity, forming a Taylor cone or cone-shaped jet, extending the jet in a straight path, thinning the jet using an electric field, increasing electrical bending instability (also known as whipping instability), and ultimately solidifying and collecting the jet as solid fibers on a grounded collector Among the various morphologies shown in Fig. 4, porous and core–shell nanofibers are particularly suitable for siderophore delivery and sustained iron chelation in wound environments.59 Different polymer matrices exhibit distinct physicochemical and biological properties that influence the siderophore loading efficiency, release behavior, and antibiofilm performance.59,63,79–85
 | ||
| Fig. 4 Various structures of electrospun fiber morphologies: (a) aligned, (b) randomly oriented, (c) core/shell, (d) hollow, (e) multi-channel microtubes, (f) colloidal nanoparticle-decorated, (g) shish-kebab, (h) helical, (i) porous, (j) necklace-like, and (k) island-like. (a–c and i) Reproduced from ref. 79 with permission from Wiley-VCH GmbH, copyright 2023, (d) reproduced from ref. 85 with permission from Wiley-VCH GmbH, copyright 2023, (e) reproduced from ref. 59 with permission from the American Chemical Society, copyright 2017, (f) reproduced from ref. 80 with permission from the American Chemical Society, copyright 2012, (g) reproduced from ref. 81 with permission from the American Chemical Society, copyright 2013, (h) reproduced from ref. 82 with permission from Elsevier, copyright 2008, (j) reproduced from ref. 84 with permission from the Royal Society of Chemistry, copyright 2021, (k) reproduced from ref. 83 with permission from the American Chemical Society, copyright 2017, and (l) reproduced from ref. 63 with permission from Springer Nature, copyright 2020. | ||
10. Current investigations and future directions
The preparation of nanofibers from microbial siderophores by the electrospinning process is a positive approach to address the challenges related to biofilms. Their ability to interrupt the biofilm and also the combined benefits of electrospinning technology make them suitable for various applications like healing medical wounds and also preventing industrial biofilm.23,86 According to previous reports, the stability of nanofibers leads to increased degradation of biofilms at wound sites. Since siderophores occur naturally, the biocompatibility of the entire nanofiber system confirms their safety for medical use. It is important to develop electrospinning processes that are cost-effective and scalable for the commercialization of nanofibers loaded with siderophores.Biphasic scaffolds prepared at the University of Nebraska are a fascinating method in drug delivery and antimicrobial therapy.74,87 The researchers combined nanofiber mats with microneedle arrays and developed a delivery system to target bacterial biofilms. The coaxial type used to make the scaffolds had good control over the structure, thereby enhancing the transfer of antimicrobial agents. The use of polyvinylpyrrolidone (PVP) microneedle arrays and Pluronic F-127-poly(ε-caprolactone) nanofiber mats also enhanced the function of the scaffold. The distribution of antimicrobial agents through a scaffold helps in the killing mechanism and results in synergistic efficacy against the biofilm. This approach demonstrates significant promise for treating infections associated with biofilm formation, which are often challenging to eradicate with conventional therapies. The successful demonstration of the antimicrobial delivery on human skin highlights its potential for clinical application in treating wounds infected with bacterial biofilm. The research and development in this area could lead to the development of novel therapies for combating antibiotic-resistant infections and improving patient outcomes.88
The current invention of core-shell nanofibers incorporated with PEO, chitosan (CS), PVP, and gelatin has made progress in the study of dual drug delivery of Vancomycin and Primaxin (imipenem/cilastatin).89 These nanofibers, with an average diameter ranging from 218 to 342 nm, were used in diabetic foot ulcer infections, particularly in methicillin-resistant Staphylococcus aureus (MRSA). The incorporation of antibiotics into these nanofibers allowed the controlled release of drugs that target bacterial strains like MRSA, Escherichia coli and Pseudomonas aeruginosa.90 This approach minimizes the risk associated with traditional drug delivery methods, such as nephrotoxicity and cytotoxicity.91 The work also focused on fabricating the electrospun nanofibers with anti-inflammatory and antipyretic properties. This study also highlights the importance of nanofiber-based therapies to address complex medical challenges while minimizing systemic side effects,73,92 as shown in Fig. 5. The comparative studies of polymers are summarized in Table 2.
 | ||
| Fig. 5 The schematic representation of electrospun nanofiber-based from siderophore for enhanced healing and biofilm resistant. | ||
| S.No. | Parameter | PCL | PVA | PEO |
|---|---|---|---|---|
| 1 | Polymer type | Hydrophobic aliphatic polyester93 | Hydrophilic water-soluble polymer | Hydrophilic non-ionic polymer |
| 2 | Fabrication methods | Electrospinning, solvent casting, nanoparticles, core–shell fibers | Electrospinning, hydrogels, freeze-drying | Hydrophilic matrix/tablets, hydrogels |
| 3 | Typical siderophore type studied | Few direct reports (inferred from drug/antibiotic models) | Few direct reports (hydrogel matrices)94 | Not commonly reported for siderophores, used in controlled release |
| 4 | Loading efficiency/mechanism | Compatible with hydrophobic drugs or hydrophobic modifications of siderophores when encapsulated in particles95,96 | Good entrapment with hydrophilic drugs; rapid release for water-soluble siderophores96 | Facilitates diffusion-controlled release due to swelling/erosion97 |
| 5 | Release profile | Sustained, slow release over weeks/months due to hydrophobicity and crystallinity98 | Burst or rapid release depending on gel/swelling; tunable via crosslinking | Fast release with hydrophilic swelling; slower with high MW grades |
| 6 | Mechanical/physical properties | Good mechanical strength; slow biodegradation suitable for implants99 | Flexible hydrogel; water solubility can limit mechanical integrity unless crosslinked | High water uptake; matrix can erode rapidly, weaker mechanical strength |
| 7 | Advantages of siderophore delivery | Sustained release ideal for prolonged antimicrobial action; strong network for protective encapsulation100 | High loading for hydrophilic siderophores; tunable porosity and swelling6 | Enhanced diffusivity; rapid release useful for acute siderophore payloads101 |
| 8 | Limitations | Hydrophobicity may impede the loading of hydrophilic siderophores; slow degradation | Rapid initial release (“burst”) may limit duration; water solubility may lead to fast clearance | Rapid erosion/swelling may undermine controlled, long-term delivery |
| 9 | Best use cases | Long-term localized delivery (e.g., implanted coatings, nanofiber mats) | Hydrogel pads/films for wound or topical delivery | Oral/implant hydrophilic matrix where rapid release is acceptable |
The combination of copper sulfide with PVP and gelatin nanofibers provides several beneficial medical properties, where copper sulfide is recognized for its antimicrobial properties that help to kill microbes, while PVP and gelatin offer structural support and biocompatibility. PVP is often used in the medical field for its ability to form films and fibers, making it an excellent substance for creating nanofibers. Gelatin obtained from collagen acts as a biodegradable material and is widely used in wound healing applications. By blending these materials into composite nanofibers, Liu et al. achieved a cooperative effect, where each compound contributes its unique properties and enhances the material's overall performance. This approach demonstrates the innovation and has gained importance for various biomedical applications, including wound dressings and antimicrobial coatings.102,103
These innovations and studies related to nanofibers have paved the way for a solution to the problem of biofilm formation. Electrospun nanofibers can mimic the architecture of natural extracellular matrices, making them suitable for medical applications. These nanofibers provide a good environment for cell adhesion, proliferation, and differentiation.99,104 They are used in the regeneration of various tissues and organs, including skin, bone, cartilage, nerve, and blood vessels.53,105 Electrospun nanofibers also serve as carriers for controlled and targeted drug delivery. Researchers have attained sustained release profiles, improved drug stability, and enhanced therapeutic efficacy by incorporating drugs, growth factors, or other bioactive molecules into the polymer matrix during electrospinning. Studies have also proven that electrospun nanofiber-based drug delivery systems are effective for applications in cancer therapy, wound healing, and the treatment of infections.106–108
11. Conclusion
Electrospun microbial siderophore-based nanofibers are an innovative strategy for addressing the challenges in biofilm-associated chronic wounds. The use of iron sequestration in nano-engineered dressings to disrupt microbial metabolism, quorum sensing, and biofilm maturation offers advantages over traditional antimicrobials, including a reduced risk of resistance development and multifunctional wound support. The ability of electrospinning to generate nanofibrous matrices with tunable architecture, high surface area, and controlled release profiles further strengthens the potential of siderophore-loaded systems as advanced wound care platforms that simultaneously promote antimicrobial activity, modulate inflammation, facilitate granulation tissue formation, and support cell adhesion and proliferation. Despite these promising attributes, the translation of siderophore-based nanofibers from the bench to the bedside presents several challenges. The current studies are limited to in vitro or short-term in vivo models due to the inefficiencies of long-term biocompatibility, immunogenicity, iron homeostasis disruption, and host–microbiome interactions. The variability in siderophore sources, stability under physiological wound conditions, and controlled release kinetics within complex wound environments require optimization. Regulatory and clinical considerations further complicate translation, as siderophore-based dressings may be classified as combination products, necessitating rigorous safety, toxicity, and efficacy evaluations. Future research should therefore prioritize standardized in vivo wound models, long-term safety assessments, and comparative studies against existing clinical dressings. Integrating smart or stimuli-responsive nanofiber systems, synergistic combinations with growth factors or probiotics, and advanced delivery strategies may further enhance therapeutic outcomes.The microbial siderophore-based electrospun nanofibers hold significant promise as next-generation “nanohealers” for biofilm-infected chronic wounds, and their clinical realization will depend on interdisciplinary efforts that bridge materials science, microbiology, pharmacology, and regulatory science. In addressing these translational gaps, it would be essential to harness their therapeutic potential and advance these systems toward safe, effective, and clinically viable wound care solutions.
Author contributions
Sitalakshmi Thyagarajan: Conceptualization, investigation, methodology, formal analysis, writing – original draft, writing – review & editing. Senthamil Selvi Poongavanam: Formal analysis, writing – review & editing.Conflicts of interest
The authors declared no conflicts of interest.Data availability
This review is based on previously published data, which are cited throughout the manuscript. No new data were generated or analyzed in the course of this study. All data supporting the findings of this review are available from the cited literature.Acknowledgements
The author gratefully acknowledges financial support for this work through the grants awarded by the Tamil Nadu State Council for Science and Technology, Chennai [IPD:772/11.12.25]. We extend our heartfelt gratitude to the Principal/Dean, Department of Bioengineering & Department of Nano Biomedicine Lab, Saveetha Medical College and Hospital, Saveetha Institute of Medical and Technical Sciences (SIMATS), Chennai, Tamil Nadu, India for their valuable support and encouragement.References
- N. J. Percival, Classification of wounds and their management, Surgery, 2002, 20(5), 114–117, DOI:10.1383/surg.20.5.114.14626.
- G. S. Lazarus, D. M. Cooper, D. R. Knighton, D. J. Margolis, R. E. Percoraro and G. Rodeheaver, et al., Definitions and guidelines for assessment of wounds and evaluation of healing, Wound Repair Regen., 1994, 2(3), 165–170, DOI:10.1046/j.1524-475x.1994.20305.x.
- S. L. Thyagarajan, S. Kandhasamy, G. Ramanathan, U. T. Sivagnanam and P. T. Perumal, Evaluation of stress-induced microbial siderophore from Pseudomonas aeruginosa strain S1 as a potential matrix metalloproteinase inhibitor in wound healing applications, Curr. Microbiol., 2016, 72(5), 583–588, DOI:10.1007/s00284-016-0988-x.
- M. Mir, M. N. Ali, A. Barakullah, A. Gulzar, M. Arshad and S. Fatima, et al., Synthetic polymeric biomaterials for wound healing: a review, Prog. Biomater., 2018, 7(1), 1–21, DOI:10.1007/s40204-018-0083-4.
- S. L. Thyagarajan, G. Ramanathan, S. Singaravelu, S. Kandhasamy, P. T. Perumal and U. T. Sivagnanam, Microbial Siderophore as MMP inhibitor:An interactive approach on wound healing application, Wound Med., 2017, 16, 7–14, DOI:10.1016/j.wndm.2016.12.002.
- S. L. Thyagarajan, G. Ramanathan, S. Singaravelu, S. Kandhasamy, P. T. Perumal and U. T. Sivagnanam, Characterization and evaluation of siderophore-loaded gelatin microspheres: a potent tool for wound-dressing material, Polym. Bull., 2017, 74(6), 2349–2363, DOI:10.1007/s00289-016-1840-y.
- G. Ramanathan, S. Thyagarajan and U. T. Sivagnanam, Accelerated wound healing and its promoting effects of biomimetic collagen matrices with siderophore loaded gelatin microspheres in tissue engineering, Mater. Sci. Eng., C, 2018, 93, 455–464, DOI:10.1016/j.msec.2018.08.026.
- A. D. Sezer and E. Cevher, Biopolymers as wound healing materials: challenges and new strategies, in Biomaterials applications for nanomedicine, 2011, p. 383 Search PubMed.
- S. Shaffique, M. Imran, S. H. Wani, M. A. Khan, S.-M. Kang and A. Adhikari, et al., Evaluating the adhesive potential of the newly isolated bacterial strains in research exploitation of plant microbial interaction, Front. Plant Sci., 2022, 13, 1004331, DOI:10.3389/fpls.2022.1004331.
- H. Lappin-Scott, S. Burton and P. Stoodley, Revealing a world of biofilms—the pioneering research of Bill Costerton, Nat. Rev. Microbiol., 2014, 12(11), 781–787, DOI:10.1038/nrmicro3343.
- R. J. C. McLean, J. S. Lam and L. L. Graham, Training the biofilm generation—a tribute to J. w. costerton, J. Bacteriol., 2012, 194(24), 6706–6711, DOI:10.1128/jb.01252-12.
- P. Stoodley, L. Hall-Stoodley, B. Costerton, P. DeMeo, M. Shirtliff and E. Gawalt, et al., Biofilms, biomaterials, and device-related infections, in Handbook of Polymer Applications in Medicine and Medical Devices, Elsevier, 2013, pp. 77–101. DOI:10.1016/b978-0-323-22805-3.00005-0.
- M. Miethke and M. A. Marahiel, Siderophore-based iron acquisition and pathogen control, Microbiol. Mol. Biol. Rev., 2007, 71(3), 413–451, DOI:10.1128/mmbr.00012-07.
- M. Hofmann, T. Heine, L. Malik, S. Hofmann, K. Joffroy and C. H. R. Senges, et al., Screening for microbial metal-chelating siderophores for the removal of metal ions from solutions, Microorganisms, 2021, 9(1), 111, DOI:10.3390/microorganisms9010111.
- E. Haesler, T. Swanson, K. Ousey and K. Carville, Clinical indicators of wound infection and biofilm: reaching international consensus, J. Wound Care, 2019, 28(Sup3b), s4–12, DOI:10.12968/jowc.2019.28.sup3b.s4.
- B. Szomolay, I. Klapper, J. Dockery and P. S. Stewart, Adaptive responses to antimicrobial agents in biofilms, Environ. Microbiol., 2005, 7(8), 1186–1191, DOI:10.1111/j.1462-2920.2005.00797.x.
- R. M. Donlan and J. W. Costerton, Biofilms: survival mechanisms of clinically relevant microorganisms, Clin. Microbiol. Rev., 2002, 15(2), 167–193 CrossRef CAS PubMed.
- S. Ishida, M. Arai, H. Niikawa and M. Kobayashi, Inhibitory effect of cyclic trihydroxamate siderophore, desferrioxamine E, on the biofilm formation of Mycobacterium species, Biol. Pharm. Bull., 2011, 34(6), 917–920, DOI:10.1248/bpb.34.917.
- C. Schaudinn, B. Costerton, P. Stoodley, D. Robinson, P. Webster and M. Baum, et al., Bacterial biofilms, other structures seen as mainstream concepts, Microbe, 2007, 2(5), 231–237, DOI:10.1128/microbe.2.231.1.
- G. J. Sharman, D. H. Williams, D. F. Ewing and C. Ratledge, Isolation, purification and structure of exochelin MS, the extracellular siderophore from Mycobacterium smegmatis, Biochem. J., 1995, 305(1), 187–196, DOI:10.1042/bj3050187.
- A. K. Patel, M. K. Deshattiwar, B. L. Chaudhari and S. B. Chincholkar, Production, purification and chemical characterization of the catecholate siderophore from potent probiotic strains of Bacillus spp, Bioresour. Technol., 2009, 100(1), 368–373, DOI:10.1016/j.biortech.2008.05.008.
- S. Sah and R. Singh, Siderophore: Structural and functional characterisation – A comprehensive review, Agriculture, 2015, 61(3), 97–114, DOI:10.1515/agri-2015-0015.
- A. Khan, P. Singh and A. Srivastava, Synthesis, nature and utility of universal iron chelator – Siderophore: A review, Microbiol. Res., 2018, 212–213, 103–111, DOI:10.1016/j.micres.2017.10.012.
- M. Gledhill, P. McCormack, S. Ussher, E. P. Achterberg, R. F. C. Mantoura and P. J. Worsfold, Production of siderophore type chelates by mixed bacterioplankton populations in nutrient enriched seawater incubations, Mar. Chem., 2004, 88(1–2), 75–83, DOI:10.1016/j.marchem.2004.03.003.
- T. Bianchi, R. D. Wolcott, A. Peghetti, D. Leaper, K. Cutting and R. Polignano, et al., Recommendations for the management of biofilm: a consensus document, J. Wound Care, 2016, 25(6), 305–317, DOI:10.12968/jowc.2016.25.6.305.
- R. Niehus, A. Picot, N. M. Oliveira, S. Mitri and K. R. Foster, The evolution of siderophore production as a competitive trait, Evolution, 2017, 71(6), 1443–1455, DOI:10.1111/evo.13230.
- B. Schwyn and J. B. Neilands, Universal chemical assay for the detection and determination of siderophores, Anal. Biochem., 1987, 160(1), 47–56, DOI:10.1016/0003-2697(87)90612-9.
- S. Ali and N. N. Vidhale, Bacterial Siderohore and their Application: A review, Int. J. Curr. Microbiol. Appl. Sci., 2013, 2, 303–312 Search PubMed.
- F. Sadaf, R. Saleem, M. Ahmed, S. I. Ahmad and Navaid-ul-Zafar, Healing potential of cream containing extract of Sphaeranthus indicus on dermal wounds in Guinea pigs, J. Ethnopharmacol., 2006, 107(2), 161–163, DOI:10.1016/j.jep.2006.02.022.
- A. Ojha and G. F. Hatfull, The role of iron in Mycobacterium smegmatis biofilm formation: the exochelin siderophore is essential in limiting iron conditions for biofilm formation but not for planktonic growth, Mol. Microbiol., 2007, 66(2), 468–483, DOI:10.1111/j.1365-2958.2007.05935.x.
- Y. Su, A. McCarthy, S. L. Wong, R. R. Hollins, G. Wang and J. Xie, Simultaneous delivery of multiple antimicrobial agents by biphasic scaffolds for effective treatment of wound biofilms, Adv. Healthcare Mater., 2021, 10(12) DOI:10.1002/adhm.202100135.
- Y. Su, J. T. Yrastorza, M. Matis, J. Cusick, S. Zhao and G. Wang, et al., Biofilms: Formation, research models, potential targets, and methods for prevention and treatment, Adv. Sci., 2022, 9(29) DOI:10.1002/advs.202203291.
- Y. Su, V. L. Mainardi, H. Wang, A. McCarthy, Y. S. Zhang and S. Chen, et al., Dissolvable microneedles coupled with nanofiber dressings eradicate biofilms via effectively delivering a database-designed antimicrobial peptide, ACS Nano, 2020, 14(9), 11775–11786, DOI:10.1021/acsnano.0c04527.
- A. Rajput, K. Kaur and M. Kumar, SigMol: repertoire of quorum sensing signaling molecules in prokaryotes, Nucleic Acids Res., 2016, 44(D1), D634–D639, DOI:10.1093/nar/gkv1076.
- J. Chen, Z. Sun, J. Jin, F. Wang, Q. Yang and H. Yu, et al., Role of siderophore in Pseudomonas fluorescens biofilm formation and spoilage potential function, Food Microbiol., 2023, 109(104151), 104151, DOI:10.1016/j.fm.2022.104151.
- J. J. Ahire and L. M. T. Dicks, Nisin incorporated with 2,3-dihydroxybenzoic acid in nanofibers inhibits biofilm formation by a methicillin-resistant strain of staphylococcus aureus, Probiotics Antimicrob. Proteins, 2015, 7(1), 52–59, DOI:10.1007/s12602-014-9171-5.
- M. Adeli-Sardou, M. Torkzadeh-Mahani, M. Yaghoobi and M. Dodel, Antibacterial and Anti-biofilm Investigation of Electrospun PCL/gelatin/Lawsone Nano Fiber Scaffolds against Biofilm Producing Bacteria, Biomacromol. J., 2018, 4(1), 46–57 Search PubMed . Available from: https://www.bmmj.org/article_34368.html.
- E. A. Kamoun, S. A. Loutfy, Y. Hussein and E.-R. S. Kenawy, Recent advances in PVA-polysaccharide based hydrogels and electrospun nanofibers in biomedical applications: A review, Int. J. Biol. Macromol., 2021, 187, 755–768, DOI:10.1016/j.ijbiomac.2021.08.002.
- C.-H. Xue, X.-J. Guo, M.-M. Zhang, J.-Z. Ma and S.-T. Jia, Fabrication of robust superhydrophobic surfaces by modification of chemically roughened fibers via thiol–ene click chemistry, J. Mater. Chem. A, 2015, 3(43), 21797–21804, 10.1039/c5ta04802h.
- Q. Ye, W. Chen, H. Huang, Y. Tang, W. Wang and F. Meng, et al., Iron and zinc ions, potent weapons against multidrug-resistant bacteria, Appl. Microbiol. Biotechnol., 2020, 104(12), 5213–5227, DOI:10.1007/s00253-020-10600-4.
- N. Hamdan, A. Yamin, S. A. Hamid, W. K. W. A. Khodir and V. Guarino, Functionalized antimicrobial nanofibers: Design criteria and recent advances, J. Funct. Biomater., 2021, 12(4), 59, DOI:10.3390/jfb12040059.
- S. Daneshmand, O. Shahraki, H. Hosseynipour, F. Roshan and M. A. Miri, Piperine-loaded Zein electrospun nanofibers: Development, characterization and antibacterial application, Bionanoscience, 2024, 14(1), 11–26, DOI:10.1007/s12668-023-01246-3.
- B. Ghafoor, A. Aleem, M. Najabat Ali and M. Mir, Review of the fabrication techniques and applications of polymeric electrospun nanofibers for drug delivery systems, J. Drug Delivery Sci. Technol., 2018, 48, 82–87, DOI:10.1016/j.jddst.2018.09.005.
- A. Mukherjee, S. Bose, A. Shaoo and S. K. Das, Nanotechnology based therapeutic approaches: an advanced strategy to target the biofilm of ESKAPE pathogens, Mater. Adv., 2023, 4(12), 2544–2572, 10.1039/d2ma00846g.
- S. Torres-Giner, J. V. Gimeno-Alcañiz, M. J. Ocio and J. M. Lagaron, Comparative performance of electrospun collagen nanofibers cross-linked by means of different methods, ACS Appl. Mater. Interfaces, 2009, 1(1), 218–223, DOI:10.1021/am800063x.
- J. X. Law, L. L. Liau, A. Saim, Y. Yang and R. Idrus, Electrospun collagen nanofibers and their applications in skin tissue engineering, Tissue Eng. Regener. Med., 2017, 14(6), 699–718, DOI:10.1007/s13770-017-0075-9.
- Z. Mbese, S. Alven and B. A. Aderibigbe, Collagen-based nanofibers for skin regeneration and wound dressing applications, Polymers, 2021, 13(24), 4368, DOI:10.3390/polym13244368.
- E. S. Place, J. H. George, C. K. Williams and M. M. Stevens, Synthetic polymer scaffolds for tissue engineering, Chem. Soc. Rev., 2009, 38(4), 1139, 10.1039/b811392k.
- J. Luo, H. Han, J. Wu, X. Wang, J. Feng and S. Toan, et al., Excellent photocatalytic activity of MoO3-adorned g-C3N4 systems: Construction of S-scheme heterojunction, Appl. Surf. Sci., 2022, 604(154512), 154512, DOI:10.1016/j.apsusc.2022.154512.
- F. A. Sheikh, H. W. Ju, J. M. Lee, B. M. Moon, H. J. Park and O. J. Lee, et al., 3D electrospun silk fibroin nanofibers for fabrication of artificial skin, Nanomedicine, 2015, 11(3), 681–691, DOI:10.1016/j.nano.2014.11.007.
- Z.-M. Huang, Y.-Z. Zhang, M. Kotaki and S. Ramakrishna, A review on polymer nanofibers by electrospinning and their applications in nanocomposites, Compos. Sci. Technol., 2003, 63(15), 2223–2253, DOI:10.1016/s0266-3538(03)00178-7.
- J. F. Cooley and C. Eastman, Apparatus for electrically dispersing fluids, United States patent, US692631, 1902 Search PubMed.
- C.-K. Huang, K. Zhang, Q. Gong, D.-G. Yu, J. Wang and X. Tan, et al., Ethylcellulose-based drug nano depots fabricated using a modified triaxial electrospinning, Int. J. Biol. Macromol., 2020, 152, 68–76, DOI:10.1016/j.ijbiomac.2020.02.239.
- Y. Zhou, H. Yang, X. Liu, J. Mao, S. Gu and W. Xu, Electrospinning of carboxyethyl chitosan/poly(vinyl alcohol)/silk fibroin nanoparticles for wound dressings, Int. J. Biol. Macromol., 2013, 53, 88–92, DOI:10.1016/j.ijbiomac.2012.11.013.
- A. Formhals, Process and apparatus for preparing artificial threads, US Pat., 1975504, 1934 Search PubMed.
- A. Formhals, Artificial fiber construction, US Pat., 2109333, 1938 Search PubMed.
- A. Formhals, Method of producing artificial fibers, US Pat., 2158415, 1939 Search PubMed.
- A. Formhals, Method and apparatus for the production of artificial fibers, US Pat., 2158416, 1939 Search PubMed.
- Y. Zhao, X. Cao and L. Jiang, Bio-mimic multichannel microtubes by a facile method, J. Am. Chem. Soc., 2007, 129(4), 764–765, DOI:10.1021/ja068165g.
- Y. Zhang, J. Luo, Q. Zhang and T. Deng, Growth factors, as biological macromolecules in bioactivity enhancing of electrospun wound dressings for diabetic wound healing: A review, Int. J. Biol. Macromol., 2021, 193, 205–218, DOI:10.1016/j.ijbiomac.2021.09.210.
- Z. Long, R. Li, Z. Dai, C. Shi, C. Wu and Q. Wei, et al., Necklace-like NiCo2O4@carbon composite nanofibers derived from metal–organic framework compounds for high-rate lithium storage, Mater. Chem. Front., 2021, 5(15), 5726–5737, 10.1039/d1qm00398d.
- T. Xu, H. Yang, D. Yang and Z.-Z. Yu, Polylactic acid nanofiber scaffold decorated with chitosan islandlike topography for bone tissue engineering, ACS Appl. Mater. Interfaces, 2017, 9(25), 21094–21104, DOI:10.1021/acsami.7b01176.
- A. Keirouz, N. Radacsi, Q. Ren, A. Dommann, G. Beldi and K. Maniura-Weber, et al., Nylon-6/chitosan core/shell antimicrobial nanofibers for the prevention of mesh-associated surgical site infection, J. Nanobiotechnol., 2020, 18(1) DOI:10.1186/s12951-020-00602-9.
- C. V. Boys, On the Production, Properties, and some suggested Uses of the Finest Threads, Proc. Phys. Soc., London, 1887, 9(1), 8–19 CrossRef . Available from: https://api.semanticscholar.org/CorpusID:138887096.
- Z. Sun, E. Zussman, A. L. Yarin, J. H. Wendorff and A. Greiner, Compound core–shell polymer nanofibers by co–electrospinning, Adv. Mater., 2003, 15(22), 1929–1932, DOI:10.1002/adma.200305136.
- B. Ding, E. Kimura, T. Sato, S. Fujita and S. Shiratori, Fabrication of blend biodegradable nanofibrous nonwoven mats via multi-jet electrospinning, Polymer, 2004, 45(6), 1895–1902, DOI:10.1016/j.polymer.2004.01.026.
- A. L. Yarin and E. Zussman, Upward needleless electrospinning of multiple nanofibers, Polymer, 2004, 45(9), 2977–2980, DOI:10.1016/j.polymer.2004.02.066.
- A. Trokhymchuk, I. Nezbeda, J. Jirsák and D. Henderson, Hard-sphere radial distribution function again, J. Chem. Phys., 2005, 123(2), 024501, DOI:10.1063/1.1979488.
- O. O. Dosunmu, G. G. Chase, W. Kataphinan and D. H. Reneker, Electrospinning of polymer nanofibres from multiple jets on a porous tubular surface, Nanotechnology, 2006, 17(4), 1123–1127, DOI:10.1088/0957-4484/17/4/046.
- Y. Liu, J.-H. He and J.-Y. Yu, Bubble-electrospinning: a novel method for making nanofibers, J. Phys.:Conf. Ser., 2008, 96, 012001, DOI:10.1088/1742-6596/96/1/012001.
- V. Kalra, J. H. Lee, J. H. Park, M. Marquez and Y. L. Joo, Confined assembly of asymmetric block–copolymer nanofibers via multiaxial jet electrospinning, Small, 2009, 5(20), 2323–2332, DOI:10.1002/smll.200900157.
- T. Miloh, B. Spivak and A. L. Yarin, Needleless electrospinning: Electrically driven instability and multiple jetting from the free surface of a spherical liquid layer, J. Appl. Phys., 2009, 106(11) DOI:10.1063/1.3264884.
- H. Niu and T. Lin, Fiber generators in needleless electrospinning, J. Nanomater., 2012, 2012(1) DOI:10.1155/2012/725950.
- Y. Yokoyama, S. Hattori, C. Yoshikawa, Y. Yasuda, H. Koyama and T. Takato, et al., Novel wet electrospinning system for fabrication of spongiform nanofiber 3-dimensional fabric, Mater. Lett., 2009, 63(9–10), 754–756, DOI:10.1016/j.matlet.2008.12.042.
- B. Lu, Y. Wang, Y. Liu, H. Duan, J. Zhou and Z. Zhang, et al., Superhigh–throughput needleless electrospinning using a rotary cone as spinneret, Small, 2010, 6(15), 1612–1616, DOI:10.1002/smll.201000454.
- X. Wang, H. Niu, X. Wang and T. Lin, Needleless electrospinning of uniform nanofibers using spiral coil spinnerets, J. Nanomater., 2012, 2012(1) DOI:10.1155/2012/785920.
- M. Vong, E. Speirs, C. Klomkliang, I. Akinwumi, W. Nuansing and N. Radacsi, Controlled three-dimensional polystyrene micro- and nano-structures fabricated by three-dimensional electrospinning, RSC Adv., 2018, 8(28), 15501–15512, 10.1039/c7ra13278f.
- G. I. Talyor, Disintegration of water drops in an electric field, Proceedings of the Royal Society of London. Series A, Mathematical and Physical Sciences, 1964, 280(1382), 383–397, DOI:10.1098/rspa.1964.0151.
- D. Li and Y. Xia, Direct fabrication of composite and ceramic hollow nanofibers by electrospinning, Nano Lett., 2004, 4, 933–938 CrossRef CAS.
- W. Yuan and K.-Q. Zhang, Structural evolution of electrospun composite fibers from the blend of polyvinyl alcohol and polymer nanoparticles, Langmuir, 2012, 28(43), 15418–15424, DOI:10.1021/la303312q.
- Z. L. Wang, Triboelectric nanogenerators as new energy technology for self-powered systems and as active mechanical and chemical sensors, ACS Nano, 2013, 7(11), 9533–9557, DOI:10.1021/nn404614z.
- J. Yu, Y. Qiu, X. Zha, M. Yu, J. Yu and J. Rafique, et al., Production of aligned helical polymer nanofibers by electrospinning, Eur. Polym. J., 2008, 44(9), 2838–2844, DOI:10.1016/j.eurpolymj.2008.05.020.
- T. Xu, H. Yang, D. Yang and Z. Z. Yu, Polylactic acid nanofiber scaffold decorated with chitosan islandlike topography for bone tissue engineering, ACS Appl. Mater. Interfaces, 2017, 9, 21094–21104 CrossRef CAS PubMed.
- Z. Long, R. Li, Z. Dai, C. Shi, C. Wu and Q. Wei, et al., Necklace-like NiCo 2 O 4@ carbon composite nanofibers derived from metal-organic framework compounds for high-rate lithium storage, Mater. Chem. Front., 2021, 5(15), 5726–5737 RSC.
- D. Li and Y. Xia, Direct fabrication of composite and ceramic hollow nanofibers by electrospinning, Nano Lett., 2004, 4(5), 933–938, DOI:10.1021/nl049590f.
- S. K. Ghosh, T. Bera and A. M. Chakrabarty, Microbial siderophore – A boon to agricultural sciences, Biol. Control, 2020, 144, 104214, DOI:10.1016/j.biocontrol.2020.104214.
- D. Gutschmidt, R. S. Hazra, X. Zhou, X. Xu, M. Sabzi and L. Jiang, Electrospun, sepiolite-loaded poly(vinyl alcohol)/soy protein isolate nanofibers: Preparation, characterization, and their drug release behavior, Int. J. Pharm., 2021, 594, 120172, DOI:10.1016/j.ijpharm.2020.120172.
- F. M. Guadagno and P. Revellino, Debris avalanches and debris flows of the Campania Region (southern Italy), in Debris-flow Hazards and Related Phenomena, Springer Berlin Heidelberg, Berlin, Heidelberg, 2007, pp. 489–518. (Springer Praxis Books). DOI:10.1007/3-540-27129-5_19.
- H. Adeli, M. T. Khorasani and M. Parvazinia, Wound dressing based on electrospun PVA/chitosan/starch nanofibrous mats: Fabrication, antibacterial and cytocompatibility evaluation and in vitro healing assay, Int. J. Biol. Macromol., 2019, 122, 238–254, DOI:10.1016/j.ijbiomac.2018.10.115.
- R. Sahay, V. Thavasi and S. Ramakrishna, Design modifications in electrospinning setup for advanced applications, J. Nanomater., 2011, 2011, 1–17, DOI:10.1155/2011/317673.
- Z.-Y. Qin, X.-W. Jia, Q. Liu, B.-H. Kong and H. Wang, Fast dissolving oral films for drug delivery prepared from chitosan/pullulan electrospinning nanofibers, Int. J. Biol. Macromol., 2019, 137, 224–231, DOI:10.1016/j.ijbiomac.2019.06.224.
- F. Davani, M. Alishahi, M. Sabzi, M. Khorram, A. Arastehfar and K. Zomorodian, Dual drug delivery of vancomycin and imipenem/cilastatin by coaxial nanofibers for treatment of diabetic foot ulcer infections, Mater. Sci. Eng., C, 2021, 123, 111975, DOI:10.1016/j.msec.2021.111975.
- B. Begines, T. Ortiz, M. Pérez-Aranda, G. Martínez, M. Merinero and F. Argüelles-Arias, et al., Polymeric nanoparticles for drug delivery: Recent developments and future prospects, Nanomaterials, 2020, 10(7), 1403 CrossRef CAS PubMed.
- E. M. Hetrick and M. H. Schoenfisch, Reducing implant–related infections: Active release strategies, J. Cheminf., 2006, 37(48) DOI:10.1002/chin.200648272.
- X. Liang, H.-J. Zhong, H. Ding, B. Yu, X. Ma and X. Liu, et al., Polyvinyl alcohol (PVA)-based hydrogels: Recent progress in fabrication, properties, and multifunctional applications, Polymers, 2024, 16(19), 2755, DOI:10.3390/polym16192755.
- N. A. Peppas and E. W. Merrill, Poly (vinyl alcohol) hydrogels: reinforcement of radiation-crosslinked networks by crystallization, J. Polym. Sci., Polym. Chem. Ed., 1976, 14(2), 441–457 CrossRef CAS.
- T. K. Dash and V. B. Konkimalla, Poly-ε-caprolactone based formulations for drug delivery and tissue engineering: A review, J. Controlled Release, 2012, 158(1), 15–33, DOI:10.1016/j.jconrel.2011.09.064.
- E. J. Bolívar-Monsalve, M. M. Alvarez, S. Hosseini, M. A. Espinosa-Hernandez, C. F. Ceballos-González and M. Sanchez-Dominguez, et al., Engineering bioactive synthetic polymers for biomedical applications: a review with emphasis on tissue engineering and controlled release, Mater. Adv., 2021, 2(14), 4447–4478, 10.1039/d1ma00092f.
- T. J. Sill and H. A. von Recum, Electrospinning: Applications in drug delivery and tissue engineering, Biomaterials, 2008, 29(13), 1989–2006, DOI:10.1016/j.biomaterials.2008.01.011.
- Í. Soares, J. Faria, A. Marques, I. A. C. Ribeiro, C. Baleizão and A. Bettencourt, et al., Drug delivery from PCL/chitosan multilayer coatings for metallic implants, ACS Omega, 2022, 7(27), 23096–23106, DOI:10.1021/acsomega.2c00504.
- S. B. Mayegowda and M. N. Gadilingappa, Microbial siderophores: A new insight on healthcare applications, BME Front., 2025, 6, 0112, DOI:10.34133/bmef.0112.
- Y. Bai, Y. Dou, L.-H. Xie, W. Rutledge, J.-R. Li and H.-C. Zhou, Zr-based metal–organic frameworks: design, synthesis, structure, and applications, Chem. Soc. Rev., 2016, 45(8), 2327–2367, 10.1039/c5cs00837a.
- Y. Bai, D. Wang, Z. Zhang, J. Pan, Z. Cui and D.-G. Yu, et al., Testing of fast dissolution of ibuprofen from its electrospun hydrophilic polymer nanocomposites, Polym. Test., 2021, 93, 106872, DOI:10.1016/j.polymertesting.2020.106872.
- X.-F. Liu, J. Zhang, J.-J. Liu, Q.-H. Zhou, Z. Liu and P.-Y. Hu, et al., Bifunctional CuS composite nanofibers via in situ electrospinning for outdoor rapid hemostasis and simultaneous ablating superbug, Chem. Eng. J., 2020, 401, 126096, DOI:10.1016/j.cej.2020.126096.
- A. Khalf and S. V. Madihally, Recent advances in multiaxial electrospinning for drug delivery, Eur. J. Pharm. Biopharm., 2017, 112, 1–17, DOI:10.1016/j.ejpb.2016.11.010.
- T. Hemamalini and V. R. Giri Dev, Comprehensive review on electrospinning of starch polymer for biomedical applications, Int. J. Biol. Macromol., 2018, 106, 712–718, DOI:10.1016/j.ijbiomac.2017.08.079.
- M. A. Jadidi Kouhbanani, S. Mosleh-Shirazi, N. Beheshtkhoo, S. R. Kasaee, S. Nekouian and S. Alshehery, et al., Investigation through the antimicrobial activity of electrospun PCL nanofiber mats with green synthesized Ag–Fe nanoparticles, J. Drug Delivery Sci. Technol., 2023, 85, 104541, DOI:10.1016/j.jddst.2023.104541.
- M. A. Asl, S. Karbasi, S. Beigi-Boroujeni, S. Zamanlui Benisi and M. Saeed, Evaluation of the effects of starch on polyhydroxybutyrate electrospun scaffolds for bone tissue engineering applications, Int. J. Biol. Macromol., 2021, 191, 500–513, DOI:10.1016/j.ijbiomac.2021.09.078.
| This journal is © The Royal Society of Chemistry 2026 |
