Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
The role of topical nano-based drug delivery systems for acne treatment: a systematic review and translational insights
Hamidreza Kelidari†
abc,
Anna Paula Azevedo de Carvalho† *abde and
Carlos Adam Conte-Junior
*abde and
Carlos Adam Conte-Junior abde
abde
aCenter for Food Analysis (NAL), Technological Development Support Laboratory (LADETEC), Chemistry Institute, Federal University of Rio de Janeiro (UFRJ), Rio de Janeiro, RJ 21941598, Brazil. E-mail: annacarvalho@iq.ufrj.br
bDepartment of Biochemistry, Chemistry Institute, Federal University of Rio de Janeiro, Rio de Janeiro, RJ 21941909, Brazil
cResearch Support Group on Nanomaterials, Polymers, and Interaction with Biosystems (BioNano), Chemistry Institute, Federal University of Rio de Janeiro, Rio de Janeiro, RJ 21941909, Brazil
dCenter for Food Analysis (NAL), Technological Development Support Laboratory (LADETEC), Federal University of Rio de Janeiro, Rio de Janeiro, RJ 21941598, Brazil
eAnalytical and Molecular Laboratorial Center (CLAn), Chemistry Institute, Federal University of Rio de Janeiro, Cidade Universitária, Rio de Janeiro, RJ 21941909, Brazil
First published on 8th April 2026
Abstract
Acne vulgaris predominantly affects sebaceous gland-rich areas, including the face, neck, upper chest, shoulders, and back. Its pathogenesis involves increased sebum production, abnormal follicular keratinization, Cutibacterium acnes-mediated inflammation, and immune responses. Topical therapy—such as benzoyl peroxide, retinoids, and antimicrobials—remains the first-line treatment for mild-to-moderate acne due to localized action and reduced systemic effects. However, antibiotic resistance, skin irritation, and poor adherence are limiting. This systematic review critically analyzes clinical evidence on topical nano-based drug delivery systems for acne, assessing clinical performance, safety, and their potential to address unmet therapeutic needs. Following PRISMA guidelines, searches of four databases retrieved 1303 records, of which 23 met inclusion criteria (2012–2025; 1341 participants). Primary outcomes included lesion count reduction (inflammatory, non-inflammatory, total), acne severity, sebum production, and safety/tolerability. Mechanistic pathways of nanomedicines in acne pathogenesis and translational barriers were also evaluated. Nanocarriers—as solid lipid nanoparticles, nanostructured lipid carriers, polymeric nanoparticles, nanocrystals, liposomes, and nanoemulsions—loaded with active pharmaceutical ingredients (APIs) or natural compounds demonstrated superior lesion reduction and faster onset of action compared to conventional formulations. Benefits were linked to enhanced follicular targeting, controlled release, and anti-inflammatory or sebostatic effects. Formulations containing synthetic APIs (e.g., tretinoin, adapalene, clindamycin) and natural actives (e.g., quercetin, mangostin, syringic acid, linoleic acid) showed comparable efficacy, with some natural compounds offering superior safety and patient acceptability. Topical nanomedicines present promising clinical advantages in acne management, potentially improving adherence and outcomes. Further large-scale, head-to-head trials are needed to confirm their role alongside standard therapies and support regulatory adoption.
Introduction
Acne vulgaris is a chronic, multifactorial inflammatory skin disorder of the pilosebaceous unit, affecting approximately 85% of adolescents and a growing proportion of adults worldwide.1 With an estimated prevalence of 9.4% worldwide and an annual economic burden exceeding 3 billion dollars in the United States alone, acne transcends mere cosmetic concerns, ranking among the top cutaneous diseases in terms of disability-adjusted life years.2,3 It is characterized by the formation of inflammatory and non-inflammatory lesions predominantly on the face and upper trunk, acne represents a chronic disorder of the pilosebaceous unit.4 Generally, non-inflammatory lesions (open and closed comedones), inflammatory papules and pustules, nodules, and potential scarring occur.Key pathogenesis driving acne development involves follicular hyperkeratinization, increased sebum production, Cutibacterium acnes (formerly Propionibacterium acnes) colonization, and inflammation,5 within the pilosebaceous unit, collectively contributing to the severity and persistence of the condition.6 While not life-threatening, acne significantly affects quality of life and mental health, often leading to psychological distress, including depression and anxiety.3 The treatment strategies for acne vary based on the type and severity of lesions, with current guidelines advocating for topical agents as first-line therapy for mild to moderate cases.7,8 However, concerns regarding adverse effects, antibiotic resistance, and treatment costs underscore the need for alternative and complementary therapies.9
The clinical management of acne has evolved over decades, transitioning from sulfur and salicylic acid applications in the early 20th century to the introduction of topical antibiotics and retinoids in the 1970s and 1980s.8,10 More recently, concerns over bacterial resistance,11 drug tolerability, and treatment adherence7 have spurred the search for advanced topical delivery systems capable of overcoming skin barrier limitations and enhancing site-specific drug delivery.
In this context, nanotechnology-based formulations have emerged as a transformative in dermatology, such as approach for acne management. Nanoformulations offer improved drug solubility, stability, controlled release, and better permeation through the stratum corneum while potentially reducing systemic exposure and adverse effects.12 Among these, solid lipid nanoparticles (SLNs),13 nanostructured lipid carriers (NLCs),14,15 nanoemulsions,16–18 liposomes,19 and niosomes,20 nanogels have been extensively investigated to carrier common acne drugs (i.e., clindamycin, spironolactone, benzoyl peroxide, azelaic acid, and topical retinoids).
While several preclinical studies have demonstrated the benefits of nanocarriers in enhancing drug delivery through the skin, there remains a gap in systematically assessing their clinical effectiveness and safety in human trials. Moreover, as the field increasingly incorporates natural compounds with anti-inflammatory and antimicrobial activities (e.g., quercetin, alpha-mangostin, linoleic acid, syringic acid), evaluating their performance in nanostructured vehicles becomes essential to support evidence-based integration into dermatologic practice.21 Some examples include tea tree oil, neem oil, mangosteen extract as main sources of bioactive compounds. Prior reviews have broadly described nano-delivery systems for acne,22–26 however a systematic synthesis of clinical trial data is lacking. Moreover, these reviews rarely compare synthetic versus natural active ingredients, or assess real-world tolerability and patient outcomes. This study fills this critical gap by focusing exclusively on human clinical trials and integrating quantitative outcomes such as total and inflammatory lesion count reduction, time to 50% improvement, porphyrin production, sebum levels, and patient satisfaction.
Therefore, this systematic review aims to synthesize and critically evaluate evidence of clinical trials regarding efficacy and safety of nanoformulated topical interventions for acne vulgaris, commenting gaps and emerging trends. Moreover, this review critically discusses translational challenges, integrating preclinical, regulatory, and market perspectives to propose a conceptual framework for clinical advancement. Finally, through a comprehensive synthesis of existing clinical evidence, this review seeks to inform clinical practice and guide future research endeavors to pursue optimized acne management strategies.
Search methods
Search strategy and eligibility criteria
A systematic search was conducted in four scientific databases (Science Direct, PubMed, Web of Science, and Scopus) from their inception to July 2025. The PubMed search strategy was used as a reference to develop search strategies for the other databases. English filtered the language of articles. The search terms included MeSH terms combined using Boolean operators to form the main search string, that was then adapted for each database style: (Nano OR Nanomedicine) AND (Acne OR “Acne vulgaris”) AND (Topical OR “Topical acne agent”) AND (Efficacy OR “Therapeutic efficacy”) AND (“Clinical trials” OR “Randomized Clinical trial” OR “Double-blind clinical trial”).As inclusion criteria, studies were included if they were clinical trials evaluating the effectiveness of topical nanomedicine, with outcome measures including lesion count, time to reduce lesions by 50%, lesion area, sebum production, acne severity, porphyrin production, global clinical assessment, participant evaluation, and quality of life. Studies involving participants who used oral or mechanical therapies for acne vulgaris, those lacking a description of therapeutic composition or nano products, studies with participants having other dermatological conditions interfering with treatment evaluation, and studies conducted on animals were excluded.
Data extraction and synthesis
According to PRISMA methodology,27 two authors independently reviewed and extracted data using a predefined protocol. In cases of discrepancy, a third reviewer was consulted. Data extracted from each study included author details, publication year, study design and duration, study population, intervention details (nano medicine, pharmaceutical form, dose/frequency, and administration route), controls, outcome measures, and adverse effects.Beyond systematically reviewing clinical trials on nanomedicines for acne, we critically analyzed translational challenges and regulatory gaps identified across the included studies and complementary literature. This dual approach enables both evidence synthesis and contextual insight into barriers for clinical implementation.
Assessment of risk of bias
Two independent reviewers assessed the risk of bias in included randomized controlled trials (RCTs) using the Cochrane Guide for Review Authors on Assessing Study Quality using the Cochrane Collaboration tool. Studies were classified as having “low risk”, “unclear risk”, or “high risk” of bias based on criteria including random sequence generation, allocation concealment, blinding, incomplete outcome data, selective reporting, and other sources of bias.Included studies
Database searches yielded a total of 1303 records up to July 2025. After removing duplicates, 1253 records were screened based on titles and abstracts, resulting in 115 articles selected for full-text reading. Of these, 92 articles did not meet inclusion criteria, leaving 23 studies for inclusion in the systematic review (Fig. 1). | ||
| Fig. 1 PRISMA flow diagram of the database search, trial selection, and articles included in this systematic review. | ||
Main findings and characteristics of the studies
The 23 included studies were summarized (Table 1). Among them, 11 were split-face studies, and 2 were before-and-after trials. Of all trials, 16 compared intervention with conventional treatment, 2 with another approved therapy, 3 with placebo, and 2 had no control. Study durations ranged from 4 to 12 weeks, involving 1341 participants. Various acne severity classifications were used, with lesion count being the most common. Interventions mostly included single nanomedicines or combinations of nanomedicine and antibiotic administered topically. Outcome measures included lesion count, time to reduce lesions by 50%, lesion area, sebum production, acne severity, porphyrin production, global clinical assessment, participant evaluation, and quality of life. Seventeen studies reported adverse effects.| Study design, duration | Participants N (intervention group/control group) | Acne classification (severity degree; classification system) | Intervention (nano medicine; pharmaceutical form(s); dose/frequency; route(s) of administration) | Control | Outcomes | Adverse effects | Ref. |
|---|---|---|---|---|---|---|---|
| GAGS: global acne grading system; EOs: essential oils; NLC: nanostructured lipid carriers; SLN: solid lipid nanocarriers; NR: not reported; ASI: acne severity index; TLC: total lesion count; RCT: randomized controlled trial; AgNP: silver nanoparticle. | |||||||
| RCT-8 weeks | 36/40 | Mild to moderate acne vulgaris; GAGS | Spironolactone-NLC 1%; gel | Spironolactone alcoholic gels 1% | Non-inflammatory, inflammatory, TLC, ASI | Dryness and itching most frequent symptoms in the control group | 14 |
| RCT-8 weeks | 20/20 | Mild to moderate acne; GAGS | Chitosan-coated spironolactone-NLC gel 1% + clindamycin 2% solution | Chitosan-coated NLC gel + clindamycin 2% solution | Non-inflammatory, inflammatory, TLC, ASI | NR | 15 |
| Before–after study-8 weeks | 20 | Mild to moderate acne vulgaris | Bergamot essential oil-nano-phytosomes-spironolactone | — | Non-inflammatory, inflammatory, TLC | Without adverse effects in both groups | 28 |
| RCT-12 weeks | 109/98 | Mild, moderate or severe; GAGS | Tretinoin nanoemulsion; gel: 0.025% (Nioret® Nanogel™); cutaneous | Tretinoin 0.025% gel (Retino-A® Gel) | Non-inflammatory, inflammatory, TLC, ASI | Dryness was the most common adverse event significantly less in the nano gel group | 17 |
| Split face study-6 weeks | 10 | Mild-to-moderate acne vulgaris | Tretinoin-nano emulsion 0.05% | Tretinoin emulsion 0.05% | Non-inflammatory and inflammatory lesions | NR | 29 |
| Split face study-8 weeks | 16 | Mild to moderate facial acne vulgaris | Tretinoin-NLC emulgel 0.05% | Tretinoin cream 0.05% | Non-inflammatory and inflammatory lesions | NR | 30 |
| Split face study-4 weeks | 6/6 | Mild to moderate acne vulgaris | Tretinoin-liposomal 0.025% gel | Tretinoin 0.025% gel | Non-inflammatory, inflammatory, and total lesion count | NR | 19 |
| Split face study-4 weeks | 12 | Facial acne vulgaris | Tretinoin-proniosomes 0.025% gel | Acretin 0.025% tretinoin | Non-inflammatory and inflammatory lesions | Very slight erythema (score ¼ 0.143 ± 0.377) was observed for TRT proniosomal gel | 31 |
| RCT-12 weeks | 119/93 | Mild, moderate, or severe acne vulgaris | Adapalene 0.1% and clindamycin 1% combination nano-emulsion gel (Adalene® NanogelTM) | Deriva-CMS® gel | Non-inflammatory, inflammatory, TLC, ASI | Lower local irritation and erythema were recorded with the nano group | 32 |
| Before–after study-12 weeks | 13 | Mild to moderate acne | Adapalene-NLC 0.1% | No | Non-inflammatory, inflammatory, TLC, ASI | Local adverse effects (burning, scaling, and erythema) occurred in three | 33 |
| RCT-12 weeks | 53/47 | Mild to moderate acne vulgaris-NR | Tea tree oil nanoemulsion containing adapalene 0.1%; gel | Adapalene 0.1% gel | Non-inflammatory, inflammatory, TLC, ASI | Dryness was the most common adverse effect in both groups | 34 |
| RCT-8 weeks | 15/15 | Mild to moderate facial acne | Isotretinoin-SLN 0.05% gel + clindamycin 2% solution | Isotrex® 0.05% gel + clindamycin 2% solution | Non-inflammatory, inflammatory, TLC, ASI | No skin irritancy in intervention while three patients in the Isotrex® group were omitted | 13 |
| Split face study-8 weeks | 30 | Mild (6%) to moderate (80%) facial acne vulgaris | Retinol nano liposomal solution (lipoceutical nanosome sesderma) | Topical retinoid cream | Number of total acne lesions, non-inflammatory, and inflammatory lesions | Local adverse events were significantly less in the nano formulation | 35 |
| Split face study-6 weeks | 20 | Mild to moderate acne facial acne vulgaris | Retinyl palmitate-ethosomal 0.05%; gel | Tretinoin cream (AcretinTM® 0.05%) | Non-inflammatory, inflammatory, TLC | All patients had no, or slight skin irritation on the nano group | 36 |
| Split face study-4 weeks | 23 | Mild to moderate acne | Retinaldehyde-loaded niosomes 0.05% Solution | — | Non-inflammatory lesions | Without adverse effects | 37 |
| RCT-12 weeks | 100/100 | Mild, moderate or severe facial acne vulgaris | Clindamycin nano-emulsion gel 1% (Zyclin® Nanogel™) | Clindamycin gel 1% (Clindac-A® Gel) | Non-inflammatory, inflammatory, TLC, ASI | Local adverse events were less in the nano formulation | 18 |
| RCT-12 weeks | 50/50 | Mild to moderate acne vulgaris; GAGS | Niosomal benzoyl peroxide 1% and clindamycin 1%; lotion | Niosomal clindamycin lotion 1% | TLC; % reduction of acne lesions; lesions, ASI, GAGS | Without significant adverse effects, pruritus at week 2 was higher in control group | 38 |
| RCT-8 weeks | 33/35 | Mild to moderate facial acne | Azelaic acid nanocrystal hydrogel 10% | Azelaic acid cream 20% (Skinoren®) | Non-inflammatory, inflammatory, TLC | NR | 39 |
| RCT-8 weeks | 32/32 | Moderate severity of acne vulgaris | AgNP gel 1% (ASAP®) + 2.5% benzoyl peroxide | 1% clindamycin gel + 2.5% benzoyl peroxide | Non-inflammatory, inflammatory, TLC, ASI | No adverse effects in silver nanoparticle group | 40 |
| Split-face study-8 weeks | 20 | Mild to moderate acne vulgaris; leeds system | Quercetin + EOs nanofibers | Placebo (Panthenol® Cream) | Non-inflammatory, inflammatory, TLC | Without adverse effects | 41 |
| Split-face study-8 weeks | 20 | Mild to moderate acne vulgaris; leeds system | Quercetin + EOs aspasomes thin film 1% | Placebo (Panthenol® Cream) | Non-inflammatory, inflammatory, TLC | Without adverse effects | 42 |
| Split face-before–after study-4 weeks | 20 | Mild to moderate; leeds system | Syringic acid loaded linoleic acid 1% transferosomes | Adapalene 0.1% Gel | Non-inflammatory, inflammatory, TLC | NR | 43 |
| Split-face study-12 weeks | 28 | Mild to moderate acne vulgaris; GAGS | Mangosteen extract nano gel 0.5% + 2.5% benzoyl peroxide cream | 1% clindamycin gel + 2.5% benzoyl peroxide cream | Comedones, inflammatory lesions | No severe side effects in intervention group | 44 |
Outcomes from clinical trials involving nano-based drug delivery systems for acne treatment
In the studies overviewed, a wide range of carriers were identified, including lipid-based vesicles (niosomes, liposomes, ethosomes, transferosomes), solid matrices (SLN, NLC), polymeric nanocarriers (electrospun fibers, polymeric nanoparticles), and hybrid systems such as phytosomes and aspasomes. This diversity reflects the tailored strategies adopted to improve solubility, penetration, and local tolerability depending on the chemical nature of each active compound.Efficacy by lesion type: inflammatory, non-inflammatory, and total lesions
The number of non-inflammatory lesions was reduced from baseline in the intervention groups in all studies that used it as an outcome. Of these studies, only 15 reported that the reduction was statistically significant, 1 achieved considerable reduction, and 7 without significant reduction compared to control groups (Table 2).| Intervention group | Inflammatory lesions | Non-inflammatory lesions | Total lesions | Study design, duration | Ref. |
|---|---|---|---|---|---|
| NLC: nanostructured lipid carriers; SLN: solid lipid nanocarriers; RCT: randomized controlled trial; AgNP: silver nanoparticle. | |||||
| Adapalene 0.1% and clindamycin 1% combination nano-emulsion gel (Adalene® NanogelTM) | Significant reduction | Significant reduction | Significant reduction | Split face study-8 weeks | 32 |
| Clindamycin nano-emulsion gel 1% (Zyclin® Nanogel™) | Significant reduction | Significant reduction | Significant reduction | RCT-12 weeks | 18 |
| Isotretinoin-SLN 0.05% gel + clindamycin 2% solution | Significant reduction | Significant reduction | — | RCT-8 weeks | 13 |
| Tretinoin nanoemulsion; gel: 0.025% (Nioret® Nanogel™) | Significant reduction | Significant reduction | — | RCT-12 weeks | 17 |
| Tretinoin-proniosomes 0.025% gel | Significant reduction | Significant reduction | Significant reduction | Split face study-4 weeks | 31 |
| Spironolactone-NLC 1%; gel | Significant reduction | No significant | Significant reduction | RCT-8 weeks | 14 |
| Tretinoin-liposomal 0.025% gel | Significant superior efficacy | Significant superior efficacy | — | Split face study-4 weeks | 19 |
| Retinol nano liposomal solution (lipoceutical nanosome sesderma) | Significant reduction | Significant reduction | Significant reduction | Split face study-8 weeks | 35 |
| Silver nanoparticle (AgNP) gel 1% (ASAP®) + 2.5% benzoyl peroxide | Slightly greater reduction | No significant difference | — | RCT-8 weeks | 40 |
| Tretinoin-nano emulsion 0.05% | Significant reduction | Significant reduction | — | Split face study-6 weeks | 29 |
| Mangosteen extract nano gel 0.5% + 2.5% benzoyl peroxide cream | Significant reduction | Significant reduction | — | Split-face study-12 weeks | 45 |
| Niosomal benzoyl peroxide 1% and clindamycin 1%; lotion | No significant difference | No significant difference | No significant difference | RCT-12 weeks | 38 |
| Quercetin-tea tree oil aspasomes thin film 1% | Significant reduction | No Significant reduction | Significant reduction | Split-face study-8 weeks | 42 |
| Azelaic acid nanocrystal hydrogel 10% | Significant reduction | Significant reduction | Significant reduction | RCT-8 weeks | 39 |
| Retinyl palmitate-ethosomal 0.05%; gel | Significant reduction | Significant reduction | Significant reduction | Split face study-6 weeks | 46 |
| Retinaldehyde-loaded niosomes 0.05% | Significant reduction | No significant difference | — | Split face study-4 weeks | 37 |
| Tea tree oil nanoemulsion containing adapalene 0.1%; gel | Significant reduction | No Significant reduction | Significant reduction | RCT-12 weeks | 34 |
| Tretinoin-NLC emulgel 0.05% | Significant reduction | Significant reduction | significantly higher | Split face study-8 weeks | 30 |
| Quercetin + tea tree oil-nanofibers; patch | Significant reduction | No significant difference | Significant reduction | Split-face study-8 weeks | 41 |
| Chitosan-coated spironolactone-NLC gel 1% + clindamycin 2% solution | No significant between groups | Significant reduction | Significant reduction | RCT-8 weeks | 15 |
| Bergamot essential oil-nano-phytosomes-spironolactone | Significant reduction | Significant reduction | Significant reduction | Before–after study-8 weeks | 28 |
| Syringic acid loaded linoleic acid 1% transferosomes | Considerable reduction | Significant reduction | Significant reduction | Split face-before–after study-4 weeks | 43 |
The studies reviewed used conventional synthetic drugs commonly used in acne treatment – such as tretinoin, adapalene, isotretinoin, clindamycin, and spironolactone – and naturally derived bioactives like quercetin, linoleic acid, syringic acid, and α-mangostin. These natural compounds have attracted increasing attention in dermatological nanomedicine due to their biocompatibility, anti-inflammatory and antioxidant properties, and lower potential for irritation.47 However, inherent limitations such as poor water solubility, chemical instability, and limited skin permeation have prompted the use of nanocarriers – such as transferosomes, nanofibers, lipid nanocrystals, phytosomes, and aspasomes – to enhance their delivery and therapeutic performance.
The presence of these bioactives reflects a current trend in nanomedicine: the pursuit of safer, more effective, and sustainable therapeutic alternatives, often combining anti-inflammatory, bacteriostatic, and skin-repair mechanisms.48 Thus, Fig. 2 summarizes the variety of nanoscale strategies available for acne management but also highlights the chemical and functional diversity of the active ingredients, offering a more integrated perspective between traditional pharmacology and nature-inspired innovation.
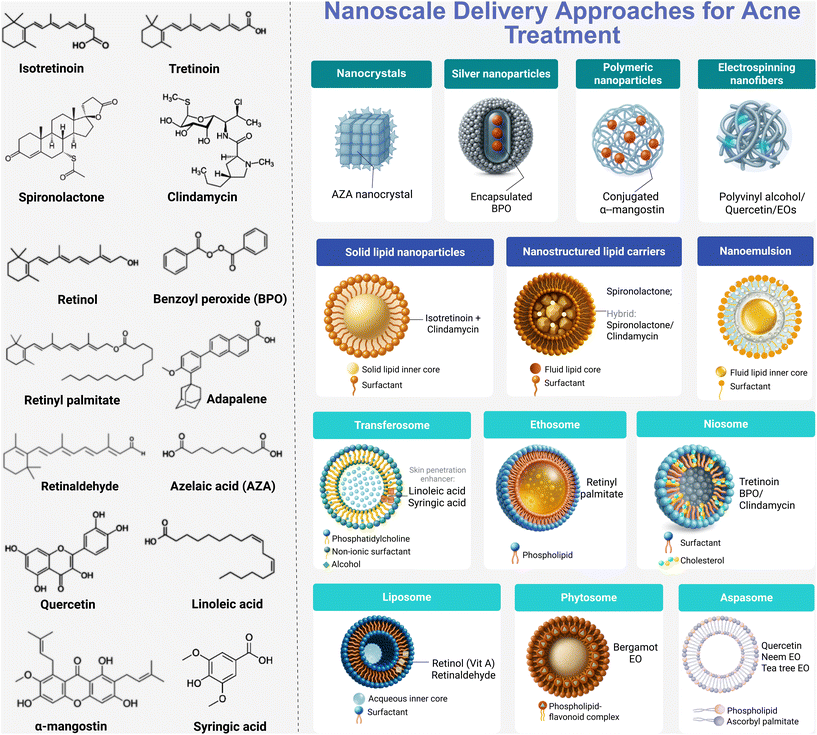 | ||
| Fig. 2 Main active pharmaceutical ingredients (APIs) used in topical nanomedicine formulated at the nanoscale for improved dermal delivery and anti-acne performance. | ||
Comparative efficacy by active ingredient delivered via nano-based drug delivery systems
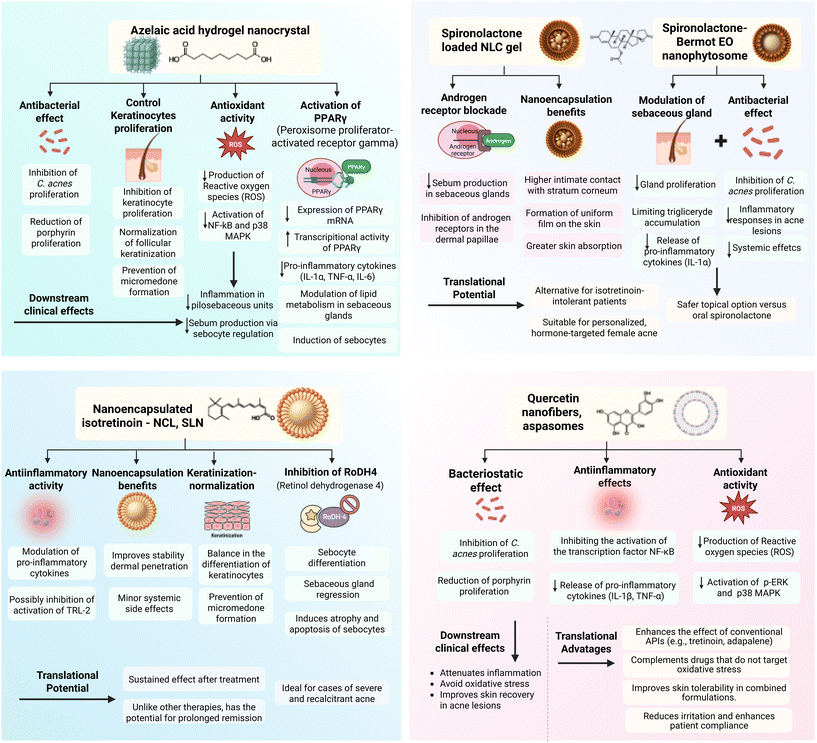
In this sense, stood outs NLCs, composed of a blend of solid and liquid lipids, offer enhanced drug loading efficiency and controlled release profiles. Their biocompatible lipid composition makes them particularly suitable for application on inflamed or sensitive skin.53 For example, literature reports the sebostatic activity NLCs for spironolactone in the management of alopecia, mediated by androgen receptor blockade. This mechanism involves the reduction of sebum production in sebaceous glands and the inhibition of androgen receptors in the dermal papillae.54
NLCs spironolactone allows for intimate contact with the stratum corneum, promoting greater skin absorption of the encapsulated spironolactone through the formation of a uniform film on the skin surface after application.14,55 Clinical evidence supports the efficacy of these systems, as an 8-week randomized clinical trial comparing a 1% spironolactone NLC gel with a 5% alcoholic spironolactone gel demonstrated significant reductions in both non-inflammatory and inflammatory lesions in both groups, although no statistically significant difference was observed between formulations.14 The NLC formulation reduced non-inflammatory lesions by approximately 47.6% and inflammatory lesions by 29.7%, whereas the alcoholic gel achieved reductions of 52.3% and 22.7%, respectively. Both treatments significantly decreased total lesion count from baseline (P < 0.001) (21).
Similarly, a study evaluating a chitosan-coated spironolactone NLC gel combined with 2% clindamycin solution demonstrated a more pronounced reduction in non-inflammatory lesions (from 26.45 ± 6.66 to 10.00 ± 3.9) compared to placebo (from 28.15 ± 6.97 to 20.25 ± 6.29) after 8 weeks (P < 0.0001), along with greater decreases in total lesion counts in the intervention group.15
Additional evidence from a before–after study indicated that spironolactone-loaded nanophytosomes combined with bergamot (Citrus medica L.) essential oil (BEO) achieved complete reduction of comedonal lesions (100%) and substantial improvements in inflammatory lesions and total lesion count, with reductions exceeding 90% after 8 weeks of treatment.28
These outcomes suggest that the combined phytosome system may enhance therapeutic efficacy through a potential antibacterial and anti-inflammatory synergy between BEO and spironolactone. This effect can be attributed to two proposed antiacne mechanisms of BEO reported in the literature:56 (i) modulation of sebaceous gland activity by reducing gland proliferation, limiting triglyceride accumulation, and decreasing the release of pro-inflammatory cytokines such as IL-1α; and (ii) induction of apoptosis in sebaceous glands coupled with antibacterial activity against C. acnes, both contributing to the attenuation of inflammatory responses in acne lesions.
Although the topical nanoformulations containing spironolactone—such as NLCs, phytosomes, and chitosan-coated carriers—demonstrated favorable outcomes in reducing acne lesion counts and minimizing adverse effects, the clinical significance of these differences remains uncertain. In some cases, the magnitude of improvement between formulations (e.g., phytosomes vs. NLCs) was modest and statistically overlapping, raising the question of whether these differences translate into meaningful clinical benefits for patients. Moreover, most studies reviewed were conducted with small sample sizes (ranging from 20 to 40 participants), which may limit the generalizability and statistical power to detect nuanced efficacy or safety differences. Another critical gap is the lack of long-term follow-up to assess sustained effectiveness and the safety of chronic use, especially given spironolactone's known systemic hormonal effects. Future clinical trials with larger cohorts, standardized protocols, and extended monitoring periods are warranted to validate these findings and to support regulatory and clinical adoption of topical spironolactone nanocarriers in acne management.
Among retinoids, adapalene is generally considered more stable and better tolerated due to its selective affinity for retinoic acid receptors, whereas tretinoin and isotretinoin are more photosensitive and often more irritating. Recent advances in nanotechnology have sought to overcome these limitations. Nanocarrier-based delivery systems—such as SLNs, nanoemulsions, and liposomes—have been explored to enhance drug stability, reduce irritation, and improve dermal penetration. These systems offer the potential to maintain therapeutic efficacy while mitigating adverse effects, thereby improving patient compliance.
Beyond its action on keratinocytes, tretinoin may stimulate collagen synthesis and influence sebaceous gland activity, although the clinical relevance of these effects in acne remains under investigation.15 The drug is associated with local adverse effects such as dryness, peeling, burning, and itching, which can lead to reduced patient adherence or discontinuation of therapy.15 Tretinoin's low aqueous solubility and instability when exposed to air, light, or heat necessitate higher concentrations or more frequent application, potentially exacerbating these side effects.28,57 Its irritation potential is known to depend on both concentration and formulation vehicle.28
Conventional topical delivery (e.g., creams, gels, lotions) often faces challenges such as poor skin penetration and stability, as the active compound is susceptible to degradation by ultraviolet light and environmental exposure.58 Nanotechnology-based delivery systems—including liposomes, niosomes, SLNs, and NLCs —have been investigated to improve tretinoin's stability, penetration, and therapeutic index.7,17,28,29
In a randomized clinical trial involving 207 patients, both tretinoin nanoemulsion gel (0.025%) and conventional tretinoin gel (0.025%) were shown to significantly reduce inflammatory and non-inflammatory lesions over a 12-week period.17 The nanoemulsion group showed a greater reduction in inflammatory lesions (78.1% ± 25.2%) compared to the conventional gel (66.9% ± 35.2%, p = 0.02), while the difference for non-inflammatory lesions was not statistically significant (p = 0.1).
A split-face study reported that a 0.05% tretinoin nanoemulsion resulted in significant reductions in blackheads, whiteheads, and papules at 6 weeks, whereas a conventional tretinoin emulsion did not achieve significant changes.29 Similarly, a 0.05% tretinoin NLC emulgel was shown to produce significantly greater reductions in blackheads and whiteheads than a conventional tretinoin cream at both 4 and 8 weeks (p < 0.001 for both lesions).30
On the other hand, a split-face study with 12 patients demonstrated that a 0.025% liposomal tretinoin gel achieved a significantly greater reduction in total lesion count compared to a conventional 0.025% gel.19 In an earlier study,31 the same research group also observed superior performance of a tretinoin proniosomal gel over marketed tretinoin formulations in improving various acne lesion types throughout the treatment period.
Although the clinical data reviewed suggest improved lesion reduction with tretinoin-loaded nanocarriers—particularly nanoemulsions, NLCs, liposomes, and proniosomes—comparative effectiveness varies among formulations. Differences in carrier composition, skin penetration depth, and drug-release profiles may explain the heterogeneity of clinical outcomes.
Importantly, although some trials indicate enhanced tolerability of tretinoin nanoformulations, this is not consistently reported across studies, and direct comparisons often lack standardized scoring systems for adverse effects. The presence of side effects such as erythema, peeling, and dryness remains a concern in both conventional and nano-based formulations.
Overall, while nanoformulations appear to offer improved pharmacokinetic profiles and moderate clinical advantages over conventional tretinoin gels—particularly in inflammatory lesion reduction—the clinical relevance of these differences remains to be fully established. Future randomized trials with larger sample sizes, longer follow-up periods, and harmonized reporting of both efficacy and tolerability outcomes are needed to better position tretinoin-based nanomedicine in acne therapy.
To overcome these limitations, various nanocarrier systems—such as nanoemulsions and NLCs—have been explored for adapalene delivery. These platforms offer potential advantages, including improved solubility, targeted delivery, controlled release, and enhanced stability.7,14
In a randomized clinical trial involving 212 participants, a nanoemulsion gel containing adapalene 0.1% and clindamycin 1% (Adalene® Nanogel™) was compared to a conventional gel formulation (Deriva-CMS® Gel).32 The nanoemulsion group demonstrated significantly greater reductions in total, inflammatory, and non-inflammatory lesions at week 12 (P < 0.001 for all endpoints). These improvements were already apparent by week 4 and persisted throughout the study duration.
Similarly, a randomized trial involving 100 patients (53 in the nanoemulsion group and 47 in the control group) evaluated a tea tree oil nanoemulsion combined with adapalene 0.1% compared to a standard adapalene 0.1% gel.34 The nanoemulsion formulation achieved superior reductions in total, inflammatory, and non-inflammatory lesion counts across all time points (P < 0.001 for all). The nanoemulsion group showed pronounced decreases in inflammatory lesions (β = −3.282 ± 0.5989) and non-inflammatory lesions (β = −5.473 ± 1.1429) after 12 weeks compared to controls. The overall difference in total lesion reduction between groups was statistically significant (β = −8.832 ± 1.5189; P < 0.001).
While both studies show statistically significant reductions in lesion counts with nanoemulsion-based adapalene formulations, the contribution of adjunct compounds—clindamycin32 and tea tree oil34— should be considered when interpreting efficacy outcomes. These actives possess known antibacterial and anti-inflammatory effects that may independently contribute to lesion reduction. Therefore, the enhanced results cannot be solely attributed to the nanocarrier or to adapalene alone. Additionally, while statistical differences were significant, the magnitude of lesion reduction should be evaluated in the context of clinical relevance—i.e., whether these changes translate to perceptible patient improvement or higher adherence. Future studies employing head-to-head designs with monotherapy nanoformulations would help clarify the specific role of nanocarriers in enhancing adapalene efficacy.
To address these challenges, the use of nanocarriers such as SLNs has been investigated to enhance cutaneous delivery, improve photostability, and reduce both systemic exposure and irritation.13
In a randomized clinical trial involving 30 participants, a formulation containing isotretinoin 0.05% SLN gel combined with clindamycin 2% solution was compared to a conventional isotretinoin 0.05% gel (Isotrex®) plus clindamycin.13 After 8 weeks of treatment, the SLN formulation demonstrated significantly greater improvements in both non-inflammatory and inflammatory lesions. Specifically, the recovery rate for non-inflammatory lesions at week 4 was 41.49% ± 6.24% in the SLN group compared to 32.25% ± 6.61% in the control group. By week 8, these rates increased to 77.05% ± 4.79% and 53.69% ± 7.34%, respectively (P < 0.01). Similarly, inflammatory lesion recovery was higher in the SLN group at week 8 (83.59% ± 3.79%) compared to controls (61.15% ± 5.34%; P < 0.01).
This clinical trial demonstrated statistically significant improvements in both inflammatory and non-inflammatory lesions with an SLN-based isotretinoin formulation.13 However, the overcome of some limitations must be considered. The study included a small sample size (n = 30), which may affect the generalizability of the findings. Furthermore, while lesion count reductions were notable, it remains unclear whether these translate into meaningful clinical benefits—such as improved patient satisfaction, long-term remission, or reduced relapse rates. Additionally, the combined use of clindamycin in both groups complicates the attribution of therapeutic efficacy solely to the nanocarrier system. Further large-scale, controlled trials could validate these results and assess the long-term safety, tolerability, and pharmacokinetic profile of topical SLN-based isotretinoin formulations.
Regarding the anti-acne mechanism, isotretinoin (13-cis-retinoic acid) exhibits a unique sebo-suppressive effect that distinguishes it from other retinoids. This activity has been linked to its ability to competitively inhibit the oxidative 3α-hydroxysteroid dehydrogenase (3α-HSD) function of retinol dehydrogenase-4 (RoDH-4), an enzyme expressed in both human liver and skin. RoDH-4 catalyzes the oxidation of steroid precursors such as 3α-androstanediol and androsterone into the potent androgens dihydrotestosterone (DHT) and androstanedione, which stimulate sebum production.59 Isotretinoin, as well as 3,4-didehydroretinoic acid, significantly inhibits this conversion in vitro, thereby reducing local androgenic stimulation of sebaceous glands. In contrast, other retinoids such as all-trans-retinoic acid, acitretin, and adapalene do not inhibit this pathway. These findings suggest that isotretinoin's inhibition of RoDH-4 may underlie its superior ability to suppress sebum secretion in acne therapy.
Retinyl palmitate, retinol, and retinaldehyde. Retinyl palmitate (RP), recognized as one of the most stable derivatives of retinoic acid, has gained attention for its potential in acne management when incorporated into nanocarrier systems. These formulations aim to improve RP's stability and enhance its cutaneous penetration, offering a promising alternative to conventional topical therapies.36
In a split-face study involving 20 patients, retinyl palmitate ethosomal hydrogel (0.05%) was compared to a marketed tretinoin cream (Acretin™ 0.05%) over six weeks.46 Both treatments resulted in significant reductions in total, inflammatory, and non-inflammatory lesion counts from baseline (P < 0.001 for both formulations). However, the RP ethosomal hydrogel was associated with a significantly greater reduction in non-inflammatory lesions (P = 0.008) and total lesion count (P = 0.016) at week 6 compared to the tretinoin cream. Importantly, the RP formulation also demonstrated superior tolerability, with minimal reports of skin irritation relative to the tretinoin-treated side.
An eight-week split-face study with 30 participants compared a retinol nano-liposomal solution (Lipoceutical Nanosome Sesderma) to a conventional topical retinoid cream.35 The nano-liposomal formulation achieved significantly greater reductions in total and inflammatory lesion counts compared to the conventional treatment (P < 0.001). While both formulations improved non-inflammatory lesions, the difference between them was not statistically significant. Notably, the nano-liposomal retinol formulation was more effective in reducing inflammatory lesions (P < 0.001).35
In another split-face study, retinaldehyde-loaded niosomal solution (0.05%) was evaluated in 23 patients with facial acne.37 The treatment resulted in significant reductions in both closed and open comedones compared to baseline, with closed comedones decreasing by 8.58% at week 2 (P = 0.036) and by 24.01% at week 4 (P < 0.001).37
The use of nanocarriers—such as ethosomes, liposomes, and niosomes—has shown promise in enhancing the cutaneous delivery, stability, and tolerability of vitamin A derivatives. However, their incremental clinical benefit over standard formulations remains to be fully established. Although several studies report superior reduction of lesions (particularly non-inflammatory or comedonal types) and improved tolerability, the cost-effectiveness and scalability of these advanced systems must be critically weighed. Additionally, variations in study design, small sample sizes, and short treatment durations may limit the generalizability of the findings. Further large-scale, head-to-head clinical trials are needed to determine whether these nanocarrier-based formulations offer meaningful advantages that justify their added formulation complexity and cost in real-world clinical settings.
Topical retinoids, including retinol derivatives, play a central role in acne treatment by targeting several key mechanisms of the disease. Once applied to the skin, retinol undergoes enzymatic conversion to retinoic acid, which exerts its biological activity by binding to nuclear retinoic acid receptors (RARs).60 This interaction leads to the regulation of gene transcription pathways involved in keratinocyte proliferation, differentiation, inflammation, and immune response.61 Among the three RAR subtypes (α, β, and γ), the γ isoform is predominant in the epidermis, and receptor selectivity may influence clinical outcomes.60 Retinoids promote normalization of desquamation within the follicular epithelium, reducing microcomedone formation, and they modulate inflammatory processes at the genetic level. Recent transcriptomic analyses have revealed that certain retinoids, such as trifarotene, can uniquely regulate gene networks associated with immune activation, cell migration, and extracellular matrix remodeling in acne lesions, offering mechanistic insights into their therapeutic effects.62
Its topical formulations typically require twice-daily application due to an approximate half-life of 12 hours. AZA is generally well tolerated, with a favorable safety profile and low potential for causing irritation.39
A randomized, double-blind clinical trial involving 70 patients (35 per group) compared a 10% azelaic acid nanocrystal hydrogel (AZA-NC) with a 20% azelaic acid cream (Skinoren®).39 Both treatments demonstrated significant reductions in inflammatory and non-inflammatory lesions by week 8, with noticeable improvements already observed at week 4. At week 8, the AZA-NC hydrogel achieved a 39.15% reduction in inflammatory lesions and a 34.58% reduction in non-inflammatory lesions (both P < 0.001), while the conventional cream produced reductions of 33.76% and 27.96%, respectively (P < 0.001 for both).39 The hydrogel formulation appeared to offer slightly greater efficacy, particularly for non-inflammatory lesions, and showed high effect sizes (partial η2 values of 0.794 for inflammatory, 0.848 for non-inflammatory, and 0.846 for total lesion reduction). Although both treatments were effective, these results suggest that the nanocrystal hydrogel may provide an incremental benefit in lesion reduction and skin tolerability over standard AZA cream.39 Further studies with larger sample sizes and longer follow-up periods may be needed to determine whether the nanocrystal formulation offers tangible benefits in real-world settings beyond improved cosmetic acceptability.
AZA exerts its anti-acne effects through multiple complementary mechanisms. It displays potent bacteriostatic activity against P. acnes by penetrating bacterial cells via ion transporters, disrupting cytoplasmic pH balance, and inhibiting thioredoxin reductase, a key enzyme in redox regulation.63 AZA also attenuates follicular hyperkeratinization by reversibly inhibiting keratinocyte proliferation and altering terminal differentiation, leading to reduced microcomedone formation.64 Its anti-inflammatory effects are mediated through the downregulation of the TLR2–KLK5–LL37 pathway and suppression of pro-inflammatory cytokines (IL-1β, IL-6, TNF-α) via NF-κB and MAPK signaling inhibition.65 Additionally, AZA reduces oxidative stress by limiting NADPH oxidase activity and enhancing peroxisome proliferator-activated receptor gamma (PPAR-γ) expression.66 Clinically, it has been shown to decrease sebum secretion and improve post-inflammatory hyperpigmentation and erythema, contributing to overall acne improvement and skin tone normalization.67
When used in conjunction with clindamycin68 or topical retinoids such as tretinoin,69 BPO enhances anti-inflammatory effects and lesion reduction without significantly increasing local irritation. Although it does not possess intrinsic anti-inflammatory properties, its ability to reduce bacterial load indirectly contributes to the mitigation of inflammation.70 On the other hand, this combination often increases the risk of local adverse effects—including irritation, peeling, dryness, and burning—which tend to be dose-dependent.38
When encapsulated in niosomes, BPO exhibits a controlled release profile that enhances its antibacterial and keratolytic activity. This delivery system improves BPO's stability and cutaneous penetration while minimizing irritation.38 The sustained release facilitates prolonged action against C. acnes colonization and reduces local inflammation, making it a promising strategy for enhancing efficacy and tolerability in acne treatment.
In a 12-week randomized clinical trial involving 100 patients, a niosomal formulation containing benzoyl peroxide 1% combined with clindamycin 1% lotion was compared to a niosomal clindamycin 1% lotion without BPO.38 The group receiving the BPO combination showed a greater mean reduction in acne lesions (64.21%) compared to the clindamycin-only group (59.04%). Nonetheless, this difference was not statistically significant. Baseline total lesion counts were similar between groups (28.16 ± 2.15 in the BPO group and 30.56 ± 2.76 in the control group; P = 0.49), and at the end of the study, both groups had comparable lesion counts (11.42 ± 0.91 vs. 10.36 ± 1.03; P = 0.44). The study suggests a numerical improvement with the addition of BPO, however, the lack of statistical significance invites further exploration into factors such as BPO concentration, vehicle, and duration of treatment. Future research may help elucidate whether nanoencapsulation strategies can optimize BPO's efficacy-tolerability balance and enhance clinical relevance in acne management.
The concentration of clindamycin that reaches the pilosebaceous ducts plays a critical role in its effectiveness, as insufficient or variable drug levels are associated with reduced efficacy and a higher likelihood of resistance development. Consequently, advanced delivery systems, as nanoemulsions, designed to improve penetration into the pilosebaceous unit, allowing more efficient delivery, and may enhance both therapeutic outcomes and resistance prevention.18 Clindamycin inhibits bacterial protein synthesis and downregulates pro-inflammatory cytokines, leading to a reduction in papules and pustules. When delivered via nanoemulsion, the formulation enhances skin penetration and may help mitigate bacterial resistance by maintaining effective local drug concentrations.18
A randomized clinical trial, involving 200 participants, compared a clindamycin 1% nano-emulsion gel (Zyclin® Nanogel™) with a conventional clindamycin 1% gel (Clindac-A® Gel).18 The nano-emulsion gel demonstrated significantly greater reductions in total acne lesions (69.3% vs. 51.9%; P < 0.001), inflammatory lesions (73.4% vs. 60.6%; P < 0.005), and non-inflammatory lesions (65.1% vs. 43.7%; P < 0.001). These differences were apparent as early as week 4 and persisted throughout the 12-week study. The nano-emulsion formulation also showed a trend toward improved tolerability compared to the conventional gel. These findings suggest that nano-emulsion formulations may offer meaningful clinical advantages in terms of both efficacy and tolerability. Future studies could explore their utility in broader patient populations and assess cost-effectiveness to inform decisions about their adoption in routine practice.
The advent of nanotechnology has enabled the development of silver nanoparticles (AgNPs), which offer improved antimicrobial efficacy due to enhanced skin penetration and increased surface area contacts with bacterial cell walls.72 Importantly, these nanoparticles require lower concentrations to achieve bactericidal effects, reducing the risk of side effects compared to conventional silver preparations.
In vitro studies have demonstrated that silver nanoparticles can inhibit C. acnes growth and may also provide anti-inflammatory benefits.40 A randomized controlled trial with 64 participants, compared 1% silver nanoparticle gel combined with 2.5% benzoyl peroxide (ASAP®) to 1% clindamycin gel plus 2.5% benzoyl peroxide over 8 weeks.40 Both groups showed substantial reductions in inflammatory and non-inflammatory lesion counts. By week 8, the reduction in inflammatory lesions was slightly greater in the AgNP group (79.7%) compared to the clindamycin group (72.6%), though the difference was not statistically significant (P = 0.18). Similarly, non-inflammatory lesion reduction was comparable between the groups (61.1% for AgNP vs. 66.8% for clindamycin; P = 0.22). Total lesion counts decreased in both groups by approximately 69–71%, again without significant differences (P = 0.71).
Silver nanoparticles exhibit promising antimicrobial and anti-inflammatory properties. It is crucial to mention that their clinical advantage over conventional antibiotics like clindamycin remains unclear based on current data. Further studies exploring long-term safety, formulation cost, and comparative tolerability could help clarify the potential role of AgNP-based treatments in routine acne management.
However, the clinical use of many natural actives is limited by intrinsic challenges such as poor aqueous solubility, instability under physiological conditions, and limited penetration across the stratum corneum.75 Nanotechnology-based delivery platforms, including NLC,78 liposomes,22 transferosomes,43 nanoparticles,45 nanofibers,41 and phytosomes,28 have been employed to overcome these limitations and to optimize the dermal bioavailability and therapeutic outcomes of natural compounds.
This section explores the clinical application of nanoformulations containing natural active ingredients evaluated in human trials. Key phytocompounds reviewed include quercetin, syringic acid, α-mangostin, and tea tree oil—each studied in combination with advanced nanostructured delivery systems. The available evidence highlights promising efficacy, particularly in the reduction of inflammatory lesions, and favorable tolerability profiles, reinforcing the potential of these agents in modern acne therapeutics.
Despite these promising properties, direct topical application of quercetin is limited by its poor water solubility, instability, and low epidermal permeability, which hinder its bioavailability and clinical effectiveness. As a result, nano-formulations have been developed to enhance its cutaneous delivery. Quercetin and tea tree oil nanofiber patch were studied in a split-face study involving 20 patients with mild to moderate acne vulgaris.41 After 8 weeks, the nanofiber patch achieved significant reductions in inflammatory lesions (61.2% ± 10.2) and total lesion count (52.9% ± 9.9), compared to a Panthenol® cream placebo (12.5% ± 15.2 and 15.3% ± 10.7, respectively; P < 0.05). However, the reduction in comedonal lesions was not significant (14.7% ± 16.5 vs. 22.5% ± 19.9; P > 0.05). In an earlier published split-face study, the same research group investigated a 1% quercetin-tea tree oil based-nanosicles (aspasomes) in 20 acne patients.42 The aspasome formulation led to significant reductions in inflammatory lesions (77.9% ± value not specified) and total lesion count (55.3% ± value not specified) compared to the placebo side (6.7% for inflammatory lesions and 10.1% for total lesions; P < 0.05). The reduction in comedones was minimal (11.8% vs. 19.1%; P > 0.05). These results suggest that quercetin-based nanoformulations are more effective against inflammatory lesions than comedonal lesions.
These findings suggest that quercetin-loaded nanoformulations may be especially effective in targeting inflammatory components of acne. While encouraging, further studies with larger sample sizes are needed to confirm the generalizability of these results. Additionally, the inclusion of tea tree oil—a compound with its own anti-inflammatory and antimicrobial effects—certainly contributed for outcomes observed.
Linoleic acid (LA), an omega-6 essential fatty acid obtained from dietary sources, was selected as a complementary component in nanoplatforms due to its antimicrobial action against C. acnes.85 LA not only exhibits antibacterial activity but also helps correct essential fatty acid imbalances in sebum that contribute to follicular plugging and acne progression.86 Furthermore, LA is known to enhance the skin penetration of nanocarriers, potentially boosting the therapeutic efficacy of encapsulated agents.87 Studies have also suggested that unsaturated fatty acid-based polymeric micelles can exert a synergistic effect when combined with active compounds, improving treatment outcomes.88
In a split-face, before-and-after study, the efficacy of syringic acid-loaded linoleic acid 1% transferosomes were evaluated in comparison to adapalene 0.1% gel.43 After one month of treatment, the side treated with syringic acid transferosomes exhibited a substantial reduction in total acne lesions (79.5% ± 7), whereas the adapalene-treated side showed only an 18.7% ± 6.1 reduction (P < 0.001). The nanocarrier formulation also appeared to provide superior anti-inflammatory benefits, as reflected in the greater overall lesion reduction. These results are promising, particularly in terms of rapid lesion reduction. Since they are derived from a single short-duration trial, larger and longer-term studies could confirm the durability and generalizability of these effects. Additionally, linoleic acid itself exhibits anti-acne properties, adding a synergistic contribution to the observed therapeutic outcomes.
In a split-face study involving 28 participants with mild to moderate acne vulgaris, the efficacy of 0.5% mangosteen extract nano gel combined with 2.5% benzoyl peroxide cream (MNLG) was compared to 1% clindamycin gel plus 2.5% benzoyl peroxide cream.45 Both treatments resulted in significant reductions in comedone counts from baseline by week 12: 66.86% reduction on the MNLG-treated side and 55.33% on the clindamycin-treated side. Although the MNLG formulation achieved a greater mean reduction in comedones, the difference between groups was not statistically significant. Regarding inflammatory lesions, both groups showed substantial improvement from baseline to week 12. The MNLG side achieved reductions of 18.32 ± 13.45 to 6.8 ± 6.34 (P < 0.001), and the clindamycin side showed reductions of 17.5 ± 12.87 to 8.04 ± 9.52 (P < 0.001), with no significant difference between treatments. These findings highlight the potential of mangosteen-based nano gels as a promising alternative or adjunct in acne treatment, particularly for patients seeking phytotherapeutic options. While the current evidence shows comparable efficacy to standard antibiotic-based regimens, larger and longer-term trials are warranted to clarify their role in acne management and to evaluate sustained benefits and safety profiles.
Mechanisms of action of nanoformulated actives in acne treatment
Beyond the individual outcomes observed across nanoformulated actives, several overlapping biological mechanisms contribute to the therapeutic effects of nanomedicines in acne treatment. These include antimicrobial activity against C. acnes, modulation of inflammation, regulation of sebum production, and normalization of follicular keratinization. The main mechanisms vary depending on the active compound and nanocarrier employed, as summarized in Fig. 3.Among the various mechanisms investigated in nanoformulated antiacne therapies, the modulation of inflammatory pathways via PPARγ activation by AZA emerges as particularly promising. Unlike other strategies focused primarily on sebostatic effects or keratinization normalization, the activation of PPARγ integrates multiple therapeutic benefits: suppression of NF-κB and p38 MAPK pathways, reduction of pro-inflammatory cytokine release, control of keratinocyte proliferation, and potential modulation of lipid metabolism in the skin. These effects position AZA not only as an anti-inflammatory agent but also as a modulator of cutaneous homeostasis. Considering that PPARγ is also implicated in the regulation of the skin barrier and microbiota, leveraging this pathway may open avenues for combined or adjunctive therapies, especially when paired with antioxidants or other PPARγ agonists. Thus, PPARγ-targeted interventions could extend benefits beyond lesion reduction to broader improvements in skin health and resilience.
Both isotretinoin and spironolactone exhibit distinct yet complementary mechanisms in acne management, each with translational relevance enhanced by nanotechnology. Isotretinoin, particularly in nanoformulations, induces sebaceous gland regression through sebocyte apoptosis and de-differentiation, alongside normalization of follicular keratinization and anti-inflammatory effects. This multimodal action results in a profound and often sustained therapeutic response, especially in severe or recalcitrant acne. In contrast, spironolactone operates via androgen receptor blockade, specifically mitigating hormonally driven sebum production with minimal systemic absorption when applied topically in NLCs. While isotretinoin offers a broad, systemic reset of acne pathophysiology, spironolactone provides a targeted, safer option for hormonal acne and isotretinoin-intolerant patients. These complementary mechanisms underscore the potential for personalized and combined therapeutic strategies, optimizing acne treatment through mechanistic precision.
Quercetin, a flavonoid widely distributed in various fruits and vegetables, demonstrates significant potential in acne management due to its antioxidant, anti-inflammatory, and antimicrobial properties. Its ability to scavenge reactive oxygen species (ROS) contributes to the reduction of oxidative stress, a key factor in acne pathophysiology. Additionally, quercetin modulates the NF-κB signaling pathway, leading to decreased production of pro-inflammatory cytokines such as TNF-α and IL-6. Importantly, quercetin also exhibits antibacterial effects against C. acnes and has been associated with the inhibition of porphyrin production, which further attenuates inflammation mediated by bacterial activity. When incorporated into nanocarrier systems, quercetin enhances the efficacy of conventional acne treatments by complementing their mechanisms of action, particularly in formulations that do not directly address oxidative stress. Moreover, its inclusion may improve cutaneous tolerability, reducing the irritation commonly associated with synthetic APIs, thereby contributing to better patient compliance and therapeutic outcomes.
Furthermore, the incorporation of natural bioactive compounds into nanomedicines alongside conventional APIs offers a promising complementary strategy for sustainable and innovative in acne treatment. Natural compounds such as quercetin, α-mangostin, and syringic acid contribute multifunctional mechanisms—including antioxidant, anti-inflammatory, and antibacterial effects—that can synergize with the targeted actions of synthetic drugs. This synergy not only enhances therapeutic efficacy but may also mitigate adverse effects commonly associated with conventional APIs, such as irritation or antibiotic resistance. Moreover, natural actives can modulate pathways not typically addressed by standard treatments, such as oxidative stress or skin microbiota balance, thus offering a more holistic approach to acne pathophysiology. A comprehensive overview of the multiple mechanisms by which nanoformulated actives exert therapeutic effects on acne is illustrated in Fig. 4.
 | ||
| Fig. 4 Schematic representation of the mechanisms of action of nanoformulated actives for acne treatment. | ||
Clinical severity outcomes (ASI and GAS)
In a randomized clinical trial involving 212 participants, nanoemulsion gel containing adapalene 0.1% and clindamycin 1% (Adalene® Nanogel™) was compared to conventional adapalene-clindamycin gel (Deriva-CMS®).32 The nanoemulsion group exhibited a greater mean reduction in ASI (1.9 ± 0.9 [95% CI: 1.71–2.02]) compared to the control group (1.4 ± 1.0 [95% CI: 1.17–1.58]; P < 0.001). This significant improvement in the nano group was observed as early as 4 weeks and persisted throughout the study (P < 0.05 at all assessments). A before–after study with 13 patients, applyed adapalene 0.1% NLC gel to one side of the face.33 After 12 weeks, the acne severity grade decreased significantly from 2.31 at baseline to 1.38 (P = 0.02), with 77% of patients achieving “clear” or “almost clear” status.
Similarly, a study involving 100 patients evaluated a tea tree oil nanoemulsion combined with adapalene 0.1% compared to a marketed adapalene 0.1% gel.16 The nanoemulsion group demonstrated significantly greater reductions in ASI over 12 weeks (P < 0.001). The trend analysis confirmed a larger decrease in the nano group (β = −5.79 ± 1.02; P < 0.001), with 71.69% of participants reaching successful outcomes (clear or almost clear), compared to 6.38% in the control group. Notably, no worsening of acne severity was reported in either group. Similar findings were reported in a 12-week randomized trial of 200 patients, comparing a clindamycin 1% nano-emulsion gel (Zyclin® Nanogel™) to conventional clindamycin 1% gel (Clindac-A®).18 The nano formulation led to a significantly greater reduction in acne severity (mean change: −1.6 ± 0.9 vs. −1.0 ± 0.8; P < 0.001). Approximately 55% of patients in the nano group achieved a two-grade improvement in severity, compared to 27% in the conventional group (P < 0.001).
In a randomized clinical trial with 212 participants, nano-emulsion gel combining adapalene 0.1% and clindamycin 1% (Adalene® Nanogel™) was compared to conventional adapalene-clindamycin formulation (Deriva-CMS® Gel).32 The nano-emulsion group showed a greater mean reduction in acne severity score (1.9 ± 0.9 [95% CI: 1.71–2.02]) than the control group (1.4 ± 1.0 [95% CI: 1.17–1.58]; P < 0.001). Notably, this significant improvement in severity was evident by week 4 and persisted throughout the treatment period (P < 0.05 at all time points).
Similarly, a before–after study with 13 patients, applying adapalene 0.1% NLC gel to one side of the face (right side), with the other side left untreated was conducted.33 After 12 weeks, the treated side showed a significant reduction in acne severity grade, decreasing from 2.31 at baseline to 1.38 (P = 0.02). Approximately 77% of participants achieved outcomes categorized as “clear” or “almost clear”.
These studies suggest that nanocarrier-based formulations may offer more rapid and sustained improvements in clinical acne severity. Most available data are derived from small sample sizes or split-face designs, which may limit generalizability. Future research involving larger, multicenter trials may help to determine whether these GAS outcomes translate into consistent and meaningful clinical benefits across broader and more diverse patient populations.
Time to 50% lesion reduction
In a before–after study, 13 patients with mild to moderate acne were treated with adapalene 0.1% NLC gel.33 After 12 weeks, significant reductions were observed in blackheads, whiteheads, and papules compared to baseline (P = 0.01, 0.03, and 0.04, respectively). The percentage reduction rates at 12 weeks were 40% for overall acne grading, 72% for papules, 64% for whiteheads, and 49% for blackheads. A split-face trial with 20 participants, compared 0.05% retinyl palmitate ethosomal hydrogel to a 0.05% tretinoin cream (Acretin™) in mild to moderate facial acne.46 By week 6, the nano-formulation side showed greater mean reductions in lesion counts: 46.69% for non-inflammatory lesions, 39.32% for inflammatory lesions, and 39.95% for total lesions, versus 34.32%, 28.35%, and 28.49% for the tretinoin-treated side. These differences, however, did not reach statistical significance (P = 0.071, 0.432, and 0.179, respectively).In a split-face study involving 28 patients, the clinical efficacy of 0.5% mangosteen extracts nano gel combined with 2.5% benzoyl peroxide cream was compared to a combination of 1% clindamycin gel and 2.5% benzoyl peroxide cream.45 The percent reduction in inflammatory lesions at week 12 was similar between groups (67.05% for the nano gel vs. 64.16% for the clindamycin group). However, clinical severity assessments by an expert panel showed a significantly greater improvement in the nano-treated side, with median severity grading improving from 2.33 to 1.33 (P < 0.001), compared to 2.33 to 1.67 (P < 0.001) for the clindamycin side. Furthermore, the final expert grading at week 12 favored the nano gel group (1.33 vs. 1.67; P = 0.004).
These findings suggest that certain nanoformulations—such as adapalene-NLC, retinyl palmitate ethosomes, and mangosteen extract nano gels—may accelerate lesion reduction when compared to conventional formulations. However, given the small sample sizes and split-face designs of these studies, it remains unclear whether these timeframes for achieving a 50% reduction in lesion counts can be generalized to broader clinical settings. Larger randomized trials with standardized endpoints are warranted to confirm these preliminary outcomes and inform evidence-based recommendations for acne management using nanocarriers.
Fig. 5 provides a comparative overview of the clinical performance of various nanoformulations in acne vulgaris, integrating both lesion reduction rates and time to achieve ∼50% reduction. Notably, adapalene-NLC and mangosteen nano gel achieved some of the highest reductions in inflammatory lesions (72% and 67%, respectively), yet differed substantially in their time to response (12 weeks for both). On the other hand, tretinoin nanoemulsion and retinyl palmitate ethosomal gel showed moderate efficacy (50% and 39% inflammatory lesion reduction, respectively) but reached this improvement more rapidly, in around 6 weeks. This dual-panel visualization underscores a key trend: while certain formulations demonstrate high efficacy, their onset of action may be slower, and vice versa. The comparison also highlights the relative consistency between inflammatory and non-inflammatory lesion responses within most nanoformulations, suggesting that these systems may offer balanced therapeutic benefits. Such insights are critical when selecting topical nanocarriers, as they enable clinicians to balance rapid clinical improvement against overall lesion clearance. The heterogeneity observed across formulations and lesion types also points to the need for further head-to-head trials and standardized outcome reporting in future clinical studies.
Porphyrin production as a biomarker
Porphyrin production, an indirect marker of C. acnes presence in the skin, was evaluated as an outcome in four studies involving a total of 67 participants. Different methodologies were employed for its quantification, including Wood's lamp examination, UV photography image analysis, e VISIA® system assessments.In a split-face study with 10 patients, the treatment with a 0.05% tretinoin nanoemulsion significantly reduced porphyrin-related parameters (size and quantity of fluorescent spots) in pilosebaceous follicles, whereas no significant change was observed with a conventional 0.05% tretinoin emulsion.29 Although both formulations reduced inflammatory and non-inflammatory lesion counts, the difference between treatments was not statistically significant, likely due to the small sample size. The nanoemulsion group demonstrated significantly greater reductions in porphyrin production compared to the conventional emulsion side. In a split-face study of 16 patients with mild to moderate acne, tretinoin 0.05% NLC emulgel significantly decreased both the size and intensity of porphyrin fluorescence after 8 weeks of treatment (P = 0.007 and P = 0.006, respectively).30 No significant reductions were seen with conventional tretinoin cream. Similarly, significant reductions in porphyrin size and intensity (P = 0.03) after 12 weeks of applying adapalene 0.1% NLC gel in a before–after study with 13 patients was observed in other study.33 Finally, in a split-face trial with 28 patients, reported significant reductions in porphyrin severity scores as assessed by Wood's lamp, were reported in both the mangosteen extract nano gel combined with 2.5% benzoyl peroxide cream group and the 1% clindamycin gel plus 2.5% benzoyl peroxide cream group.45 Both treatments significantly improved porphyrin grading scores from baseline to week 12, with no statistically significant difference between groups (P < 0.001 for both). While promising as a biomarker, evaluating the reliability of porphyrin suppression as a surrogate outcome could enhance the interpretability of acne treatment trials and support its integration into routine dermatological assessments.
Sebum production and desquamation
Sebum production was evaluated as an outcome in a single split-face study involving 23 participants.20 The trial investigated the effects of a 0.05% retinaldehyde-loaded niosomal solution on cutaneous sebum levels. Sebum secretion was measured using a Sebumeter®, showing a significant reduction over the treatment period. The mean baseline sebum level was 327.95 ± 90.20 μg cm−2, which decreased to 282.60 ± 99.70 μg cm−2 at week 2 and 250.65 ± 97.60 μg cm−2 at week 4 (P < 0.001). In addition to sebum levels, desquamation was assessed to evaluate improvement in pilosebaceous hyperkeratosis, using Visioscan® imaging. The desquamation index (DI) showed a significant decline from 10.99 ± 1.69 at baseline to 9.81 ± 1.10 at week 2 and 8.89 ± 1.32 at week 4 (P < 0.001). Thus, the reductions in sebum secretion and desquamation are statistically significant.Global clinical evaluation
Global clinical evaluations offer a comprehensive perspective on treatment response and tolerability. In the studies reviewed, global clinical evaluation was used as an outcome measure in three studies involving a total of 337 participants. Treatment response was categorized by specialist assessments based on study-specific scales. In general, “excellent” indicated complete or near-complete resolution of lesions; “good” indicated significant improvement; “fair” reflected partial improvement; and “poor” referred to no improvement or worsening of lesions. Tolerability was similarly graded according to the severity of adverse events, ranging from mild (minimal discomfort) to severe (interfering with daily activities).A 12-week randomized clinical trial involving 207 participants demonstrated that a tretinoin nano gel was significantly better tolerated than a conventional tretinoin gel (P = 0.003), with 86.7% of patients in the nano group rated as “excellent”, 7.6% as “good”, and 5.7% as “fair”, and no “poor” ratings.17 In contrast, the conventional group showed lower tolerability, with 75.3% rated as “excellent”, 6.2% as “good”, 14.4% as “fair”, and 4.1% as “poor”.17
Similarly, an 8-week split-face study involving 30 patients reported favorable clinical responses for a nano-liposomal retinol formulation, with lesion reduction classified as “excellent” in 8 patients, “good” in 12, and “fair” in 10.35 Tolerability was markedly improved in the nanoformulation group, with 90% of patients rated as “excellent” and 10% as “good”, and no reports of “poor” tolerability. In contrast, the conventional retinoid group showed lower tolerability, with 6.7% rated as “good” and 93.7% as “fair”, and no “excellent” ratings.35
In addition, a 12-week randomized trial involving 100 patients evaluating a niosomal formulation combining benzoyl peroxide 1% and clindamycin 1% reported that 80% of participants achieved “excellent” or “good” outcomes, compared to 76.1% in the niosomal clindamycin-only group, with no statistically significant difference between groups (P = 0.377).38 Final response distributions in the combination group were 18% “excellent”, 62% “good”, 20% “fair”, and 0% “poor”, whereas the control group showed 28.3% “excellent”, 47.8% “good”, 21.7% “fair”, and 2.2% “poor” (P = 0.37).38
Participants’ evaluation
Participants’ perspectives on treatment efficacy and formulation characteristics were reported in 12 studies, involving a total of 1011 patients (six split-face studies and six randomized controlled trials). Overall, patient satisfaction with nanoformulations appears consistently high.High levels of patient satisfaction and favorable product acceptability have been consistently reported for nano-based formulations. In an 8-week randomized clinical trial involving 40 patients with mild to moderate acne, high satisfaction was observed regarding product attributes such as smell, appearance, spreadability, and consistency with spironolactone-chitosan coated NLCs.15 Similarly, an 8-week before–after study with 20 patients reported good skin adherence and overall acceptance of a bergamot EO–spironolactone nano-phytosome formulation.28 Clinical efficacy outcomes further support these findings, as a 12-week randomized trial demonstrated a higher treatment success rate for a tretinoin nanoemulsion (57.6%) compared to a conventional gel (44.4%), although this difference did not reach statistical significance (P = 0.07), while complete lesion clearance was significantly more frequent in the nano group (9% vs. 1.1%; P = 0.01).17 In addition, a split-face study evaluating a tretinoin NLC emulgel reported higher patient satisfaction scores on the nanoformulation side (mean VAS scores of 7 and 7.4 at weeks 4 and 8) compared to the conventional cream (5.94 and 6.00; P = 0.016 and 0.001), with most participants reporting improved skin smoothness and lesion reduction.30
Higher treatment success rates have been consistently reported for nano-based formulations compared to conventional therapies. A nanoemulsion containing adapalene and clindamycin achieved a significantly greater success rate (58.5%) than a conventional formulation (26.4%; P < 0.001),32 while clindamycin nanoemulsions also demonstrated superior efficacy (53% vs. 28%; P < 0.001).18 Patient-reported outcomes further supported these findings, with 92% of participants reporting clearer skin and reduced lesion counts following treatment with adapalene-loaded NLCs,33 and high satisfaction rates observed with retinaldehyde-loaded niosomes, where 85% of participants were “very satisfied” or “satisfied”, and 80% perceived improved skin texture.37 Visible clinical improvement was also reported with a retinyl palmitate ethosomal gel.46 A niosomal formulation combining benzoyl peroxide and clindamycin showed higher improvement rates (64.21% vs. 59.04%), although without statistical significance.38 Similarly, a 10% azelaic acid nanocrystal hydrogel demonstrated a higher success rate (36.5%) compared to a 20% azelaic acid cream (30.4%; P < 0.001).39 Improved patient satisfaction was also observed with silver nanoparticles compared to clindamycin at week 6 (P = 0.01), although this difference was not sustained at week 8.40 Finally, both mangosteen nano gel and clindamycin treatments resulted in high satisfaction and significant improvements over time, with no statistically significant difference between groups.45
Adverse effects and tolerability
Nanomedicine-based formulations generally demonstrated favorable safety profiles, with fewer or milder adverse effects compared to conventional treatments. In an 8-week randomized clinical trial involving 76 patients with mild to moderate acne, no serious adverse events were reported, with dryness and itching occurring in 2.7% of patients treated with spironolactone-NLC 1% gel compared to 15% in those using an alcoholic spironolactone gel.14 Similarly, no significant differences in adverse reactions were observed between groups in an 8-week study involving 40 patients, with a chitosan-coated spironolactone NLC gel combined with clindamycin 2% solution showing no notable redness, itching, burning, or inflammation compared to placebo.15 Consistent with these findings, a before–after study involving 20 patients reported no cases of irritation or erythema during two months of treatment with a bergamot essential oil–spironolactone nano-phytosome formulation.28Improved tolerability was also observed in retinoid-based nanoformulations. A 12-week randomized trial involving 207 patients reported significantly fewer adverse events in the tretinoin nanoemulsion group (13.3%) compared to a conventional gel (24.7%; P = 0.04), with dryness being the most frequent event in both groups, while peeling, burning, and photosensitivity were observed only in the conventional formulation.17 Adverse events in the nano group were predominantly mild, whereas moderate events were more frequent in the control group.17 Similarly, in a 12-week randomized clinical trial with 212 patients, no serious adverse events were reported, and the nanoemulsion containing adapalene 0.1% and clindamycin 1% showed significantly lower adverse event rates (13.6%) compared to the control group (36.3%; P < 0.001), with reduced irritation and erythema (P = 0.025 and 0.045).32
Other nanostructured systems also demonstrated improved local tolerability. No irritation was reported in patients treated with isotretinoin-loaded solid lipid nanoparticles, whereas three patients in the conventional Isotrex® group discontinued treatment due to local side effects.13 In a split-face study, local reactions were more frequent and more severe with tretinoin cream compared to a retinyl palmitate ethosomal gel, which was better tolerated.46 Likewise, a retinol nano-liposomal formulation showed significantly fewer local adverse effects compared to a conventional retinoid cream.35 Retinaldehyde-loaded niosomes were also well tolerated in a 4-week split-face study involving 23 patients, with no significant irritation or adverse events reported.37
A trend toward improved tolerability was additionally observed for antimicrobial nanoformulations. In a 12-week randomized clinical trial, clindamycin nano-emulsion gel showed a tendency toward better tolerability compared to a conventional gel (P = 0.12), with all adverse events in the nano group classified as mild, whereas approximately one-third of events in the control group were moderate.18
A comparative analysis of adverse effects associated with topical nanoformulations in acne treatment reveals a clear distinction between formulations containing synthetic and natural actives. As shown in Fig. 6, the majority of natural-based nanoformulations were well tolerated, with no adverse events reported in most studies. In contrast, synthetic-based nanoformulations were more frequently associated with mild to moderate effects, including erythema, dryness, peeling, and local irritation. Notably, only one formulation containing natural actives (tea tree oil combined with adapalene) led to a high severity adverse reaction, same as for control group. These findings suggest that nanoformulations based on natural compounds may offer a safer tolerability profile, potentially enhancing patient adherence and comfort during acne therapy.
 | ||
| Fig. 6 Tolerability profile of topical nanoformulations in acne treatment – comparing adverse effects of conventional active pharmaceutic ingredients (APIs) and natural compounds. | ||
Natural bioactives in nano-based drug delivery systems for acne: clinical insights
The incorporation of naturally derived bioactive compounds in nanomedicine formulations for acne reflects a growing interest in leveraging phytochemicals’ anti-inflammatory, antimicrobial, and antioxidant properties. Clinical evidence on nanoformulated natural actives remains limited compared to synthetic drugs, yet preliminary trials suggest promising efficacy, particularly in reducing inflammatory lesions with favorable tolerability profiles.Among the phytocompounds evaluated in this review, Quercetin, a flavonoid with potent anti-inflammatory and antibacterial activities, was evaluated in two split-face studies using nanofiber patches41 and aspasome thin films,42 both combined with tea tree oil. These studies reported significant reductions in inflammatory and total lesion counts compared to placebo, although effects on comedonal lesions were modest. Quercetin's mechanism likely involves TNF-α inhibition and oxidative stress reduction, but poor solubility and low permeability necessitate nanoencapsulation for therapeutic viability.
Syringic Acid, a phenolic compound, was tested in a linoleic acid-loaded transferosome system.43 A clinical trial demonstrated a remarkable ∼80% reduction in total lesion counts within just one month of treatment, outperforming adapalene gel. The synergism between syringic acid's anti-inflammatory properties and linoleic acid's antimicrobial and barrier-restorative effects may underpin these outcomes, though larger and longer-term studies are needed.
α-Mangostin, derived from mangosteen extract, was investigated in a nano gel formulation combined with benzoyl peroxide.45 In a split-face trial, the mangosteen nano gel achieved comparable comedone and inflammatory lesion reductions to clindamycin-based therapies, with a trend toward superior tolerability.
Tea Tree Oil, with known antimicrobial effects against C. acnes, was incorporated into a nanoemulsion with adapalene.34 This combination achieved significant lesion reductions compared to adapalene alone, though disentangling the specific contribution of the essential oil versus the carrier remains challenging.
Collectively, these findings underscore that nanoformulations of natural compounds can enhance bioavailability, skin penetration, and clinical efficacy, while often exhibiting improved safety profiles compared to synthetic counterparts. However, the evidence base is still emergent, with studies typically limited by small sample sizes, short durations, and combination with other actives, which complicates efficacy attribution.
Future clinical research should:
• Explore head-to-head comparisons between nanoformulated natural and synthetic actives.
• Investigate the mechanistic pathways of natural compounds within nanocarriers, particularly their influence on skin microbiota and inflammatory mediators.
• Address pharmacokinetic profiling of natural bioactives delivered via nanocarriers.
• This growing domain suggests that natural-synthetic hybrid strategies, particularly within smart nanocarrier systems, may represent a sustainable and patient-preferred pathway in acne therapeutics.
Integrated framework to overcome translational barriers in nano-based drug delivery for acne
Although this review focuses on clinical trials, the clinical success of nano-based drug delivery systems relies on robust preclinical design and manufacturing reproducibility. Many limitations in efficacy or safety observed in trials can be traced to formulation or characterization gaps.Therefore, an integrated pharmaceutical framework is proposed, encompassing key stages of translation—from formulation design and predictive modeling to preclinical validation and adaptive clinical evaluation.
Technological and manufacturing innovations
Nanocarrier development for dermatological applications faces scalability challenges, batch-to-batch variability, and limitations in characterization. Implementing Design of Experiments (DoE) and Quality by Design (QbD) approaches can systematically optimize formulation parameters to ensure robustness and reproducibility.89 This is particularly relevant to acne clinical trials, where inconsistent formulation properties have been associated with variability in therapeutic outcomes and reduced reproducibility across studies.QbD paradigm is a systematic approach that links lifecycle stages to predefined quality targets while enabling control of critical attributes and process parameters.90 The implementation of QbD goes beyond formulation optimization, enabling the identification of critical quality attributes (CQAs) such as particle size, zeta potential, and drug release kinetics, and their linkage to critical process parameters (CPPs).91 This systematic approach facilitates regulatory alignment and enhances reproducibility across manufacturing scales. In this context, DoE serves as a key operational tool within QbD, allowing multivariate evaluation of formulation variables (e.g., surfactant concentration, lipid composition, homogenization conditions) and their interactions.92 This reduces empirical trial-and-error approaches and supports robust process design.
Although the implementation of QbD may require substantial initial investment, its capacity to enhance reproducibility and reduce long-term manufacturing costs supports its economic viability. Furthermore, when integrated with real-time analytical tools (Process Analytical Technology, PAT), QbD enables real-time monitoring of critical quality attributes, allowing early detection of deviations, minimizing process variability, accelerating scale-up, and reducing the risk of costly batch failures or recalls.93
The superiority of integrating QbD principles with nanotechnology in drug development to ensure product quality, consistency, and efficiency was recently highlighted.94 This paradigm aligns with current regulatory expectations for advanced drug delivery systems, reinforcing the role of QbD as a bridge between laboratory innovation and industrial translation.90
Furthermore, advanced analytical techniques, such as asymmetric flow field-flow fractionation (AF4-MALS)95 and cryo-TEM,96 provide critical insights into particle morphology, size distribution, and stability. Computational predictive models, leveraging machine learning, can be employed to forecast formulation behavior, predict stability profiles, and optimize process parameters for scale-up manufacturing.97
Advanced preclinical models
Conventional animal models inadequately reproduce human skin physiology and pathology, limiting the predictability of preclinical outcomes. Emerging in vitro and ex vivo systems—such as 3D human skin equivalents, organ-on-chip models, and in silico simulations of follicular targeting—offer improved human relevance and mechanistic understanding. This limitation is reflected in the weak correlation often observed between preclinical efficacy and clinical performance in acne studies, highlighting the need for more predictive human-relevant models.Advanced 3D human skin equivalents have emerged as highly relevant preclinical platforms to bridge the translational gap between conventional in vitro assays and clinical outcomes. Unlike traditional monolayer cell cultures or animal models, these systems better recapitulate the structural, biochemical, and barrier properties of human skin, including stratified epidermal layers, functional lipid organization, and follicular architecture.98 As demonstrated in recent studies, 3D reconstructed skin models enable more accurate assessment of nanocarrier penetration, retention, and follicular targeting, as well as local toxicity and inflammatory responses under physiologically relevant conditions.98 Moreover, these platforms allow the evaluation of formulation performance in diseased-like environments, including acne-associated alterations such as sebaceous activity and microbial interactions.99 Consequently, the integration of 3D human skin equivalents into preclinical pipelines enhances the predictive power of efficacy and safety data, reducing the reliance on animal testing and improving the likelihood of successful clinical translation of topical nanomedicines.
Therefore, recent advances in organ-on-a-chip technology have enabled the development of skin-on-a-chip platforms that better reproduce the physiological complexity of human skin. Most current models focus on integrating microvascular channels into full-thickness skin constructs,100 comprising both epidermal and dermal layers, or on co-culturing skin with other organ models to simulate inter-organ interactions. Unlike static in vitro systems, skin-on-a-chip platforms incorporate dynamic fluid flow, enabling the simulation of blood circulation, nutrient transport, and waste removal. These features allow more realistic evaluation of nanocarrier behavior, including permeation kinetics, distribution profiles, and time-dependent cellular responses under physiologically relevant conditions. In the context of acne, these systems offer unique opportunities to model key pathophysiological aspects, such as sebaceous gland activity, microbial colonization by C. acnes, and localized inflammatory responses.101 The ability to control microenvironmental conditions in real time makes these platforms particularly suitable for investigating nano-based therapeutic strategies targeting complex skin disorders.
In silico approaches have also been applied to identify bioactive compounds targeting acne-related pathways. For instance, molecular docking studies have demonstrated that phytochemicals derived from Ocimum sanctum exhibit potential anti-inflammatory activity through inhibition of the c-Jun N-terminal kinase (JNK) pathway, with favorable binding energies and interaction profiles comparable to reference ligands. Moreover, the incorporation of in silico-identified compounds into nanocarriers designed for follicular targeting may further enhance therapeutic efficacy by ensuring localized delivery at the primary site of acne pathogenesis.
In parallel, adopting standardized Core Outcome Sets (COS), such as those from the Acne Core Outcomes Research Network (ACORN), promotes harmonization between preclinical and clinical data collection and enhances reproducibility.102 ACORN emphasizes the importance of integrating multiple outcome domains, including clinical severity, patient-reported outcomes, long-term disease control, and safety profiles.103 This is particularly relevant for nano-based systems, where improved physicochemical performance must ultimately translate into clinically meaningful benefits captured by standardized outcome measures.
Adaptive clinical trial designs
Clinical trials investigating acne nano-drug delivery systems often face heterogeneous endpoints, small sample sizes, and limited stratification. Such limitations have been evident in the analysed trials, where heterogeneous endpoints and insufficient stratification contributed to inconclusive or modest efficacy results. Adaptive clinical designs—allowing interim data analysis and protocol adjustment—can improve trial efficiency and data quality.104Adaptive clinical trial designs have emerged as a flexible alternative to conventional RCTs, allowing for prospectively planned modifications to key study parameters based on interim analyses of accumulating data. These modifications may include adjustments to sample size, treatment arms, patient population, or endpoint selection, while maintaining control of statistical error rates. Several adaptive strategies have been described in dermatology, including group sequential designs enabling early stopping, response-adaptive randomization to favor more effective treatments, and seamless phase 2/3 designs that integrate dose-finding and confirmatory phases within a single protocol.105 Such approaches can improve trial efficiency, reduce development time, and minimize patient exposure to ineffective or potentially harmful treatments.
This flexibility is particularly relevant for nanomedicine-based therapies, where multiple formulation variables—such as particle size, surface properties, and drug release profiles—may influence clinical outcomes. Adaptive trial designs allow real-time optimization of these parameters at the clinical level, facilitating a more efficient translation of nanoformulations into clinically effective treatments.
Stratified trial designs involve the classification of patients into predefined subgroups based on baseline characteristics, allowing for more precise evaluation of treatment effects within clinically relevant populations.106 Stratified trial designs represent a key strategy to address interpatient variability by categorizing participants into clinically or biologically relevant subgroups. In dermatology, such stratification may consider factors such as disease severity, lesion type, skin microbiome composition, or inflammatory status.107 When integrated within adaptive trial frameworks, stratified designs can be implemented as adaptive enrichment strategies, in which interim analyses guide the selection or restriction of patient subpopulations more likely to benefit from the intervention.108 This not only could improve statistical efficiency but also facilitates a more personalized approach to treatment evaluation. Perhaps this is particularly relevant for nanomedicine-based therapies, as the performance of nanoformulations may vary significantly depending on patient-specific factors such as sebum production, follicular density, and skin barrier integrity. Stratified and enrichment-based designs therefore provide a valuable framework to align formulation performance with patient-specific responses.107
Regulatory and science alignment
The absence of harmonized regulatory guidelines for topical nanomedicines remains a critical translational bottleneck. Differences in regulatory expectations between agencies, particularly regarding physicochemical characterization and safety assessment, complicate the development and approval process. In this context, early alignment with regulatory science principles, including the identification of critical quality attributes (CQAs), such as particle size, surface charge, encapsulation efficiency, and release behavior—can be essential to ensure reproducibility and product consistency for regulatory approval.109Furthermore, the lack of standardized and validated analytical methodologies for nanomaterial characterization continues to hinder comparability across studies and limits regulatory confidence. Addressing these challenges requires integrated efforts combining robust analytical validation, standardized outcome measures, and translationally relevant models to bridge the gap between preclinical development and clinical application. Collaboration among academic, industrial, and regulatory partners can accelerate the development of standardized frameworks for safety and efficacy evaluation of dermatological nanomedicines.
An integrated pathway for pharmaceutical translation
Despite significant advances in nano-based drug delivery systems for acne treatment, the successful clinical translation of these technologies remains limited by fragmentation across the development pipeline. This review highlights the need for integrated frameworks that connect early-stage discovery, formulation optimization, preclinical validation, and clinical evaluation.Approaches such as in silico screening, advanced in vitro models (e.g., 3D human skin equivalents and organ-on-a-chip systems), and adaptive and stratified clinical trial designs collectively contribute to a more predictive and efficient development process. In particular, the incorporation of standardized outcome measures, such as those proposed by ACORN, and the QbD principles enable alignment between formulation performance and clinically meaningful endpoints.
However, regulatory and scientific misalignment remains a critical bottleneck, especially due to the lack of harmonized guidelines for topical nanomedicines and the absence of standardized analytical methodologies. Addressing these challenges requires early integration of regulatory science principles, including the identification of critical quality attributes and the adoption of validated characterization strategies.
Moving forward, the convergence of nanotechnology, regulatory science, and translational methodologies is essential to enable the development of more effective, reproducible, and patient-centered therapies. Such integration may ultimately support the emergence of precision nanomedicine approaches tailored to the complex and heterogeneous nature of acne.
Fig. 7 outlines the proposed integrated pharmaceutical framework, linking formulation design, preclinical validation, and adaptive clinical assessment into a cohesive translational continuum. This approach emphasizes predictive modelling and quality-by-design principles to strengthen the connection between formulation performance and clinical outcomes. By embedding these stages into a unified process, the framework supports the rational development and clinical translation of nanomedicines for acne therapy.
Conclusions and outlook
Topical nanoformulations for acne treatment have demonstrated promising therapeutic outcomes, particularly in reducing total, inflammatory, and non-inflammatory lesion counts while improving tolerability and patient satisfaction. Across the 23 included clinical trials, lipid-based nanocarriers – such as SLNs, NLCs, and nanoemulsions – proved especially effective for enhancing drug stability, skin penetration, and local bioavailability. Formulations containing both synthetic actives (e.g., tretinoin, adapalene, clindamycin) and natural compounds (e.g., quercetin, mangostin, syringic acid, linoleic acid) showed comparable clinical performance, with some natural-based systems offering superior safety profiles and patient acceptability.However, the heterogeneity of clinical endpoints, variation in outcome reporting, and limited standardization of evaluation tools (e.g., lesion counts, tolerability scales, porphyrin levels) hamper cross-study comparisons were main gaps to be filled. Moreover, most trials have small sample sizes and short durations, restricting the generalizability and long-term assessment of safety and efficacy. Emerging trends include the integration of biocompatible and stimuli-responsive nanocarriers, the co-delivery of multiple actives, and the development of hybrid formulations (e.g., chitosan-coated lipid carriers, phytosomes) that aim to enhance therapeutic synergy while minimizing adverse effects. The use of nanotechnology also aligns with the growing demand for plant-based and sustainable actives, offering an alternative to conventional retinoids and antibiotics, particularly in the context of rising antimicrobial resistance. Future research should focus on optimizing these combinatorial formulations to harness the full potential of natural-synthetic interactions within nanocarriers. This combinatorial strategy aligns with emerging trends in dermatology that seek holistic, safer, and more patient-centered solutions.
Moreover, this review identified critical translational barriers that hinder the broader clinical adoption of acne nanomedicines, including challenges in scalable manufacturing, limited predictability of preclinical models, heterogeneous clinical endpoints, and persistent regulatory uncertainties. Addressing regulatory and scientific alignment emerges as a central requirement to overcome these barriers. The absence of harmonized regulatory guidelines and standardized characterization methodologies compromises reproducibility and regulatory confidence. In this context, early integration of regulatory science principles—such as the identification and control of critical quality attributes (CQAs), including particle size, surface charge, encapsulation efficiency, and release kinetics—can provide a structured pathway to ensure product consistency, safety, and efficacy. Coupled with validated analytical methods and standardized clinical endpoints, this approach may facilitate more robust evidence generation and streamline the regulatory approval process.
Importantly, regulatory alignment should be complemented by advances in clinical trial design. Adaptive and stratified clinical approaches offer a powerful framework to address interpatient variability and optimize therapeutic evaluation. By enabling interim analyses, population enrichment, and subgroup-specific assessments, these strategies can improve statistical efficiency, reduce development timelines, and support more personalized treatment paradigms—particularly relevant for nanomedicine systems whose performance depends on patient-specific factors such as skin barrier integrity, sebum production, and microbiome composition.
To advance the clinical impact of acne nanomedicines, future research must prioritize: (i) well-powered, multicenter randomized controlled trials with standardized outcomes; (ii) long-term follow-up to assess sustained efficacy, relapse, and safety; (iii) comparative studies benchmarking nanoformulations against standard therapies and among different nanocarriers; (iv) mechanistic studies elucidating nanoparticle–skin and nanoparticle–microbiome interactions; and (v) early regulatory dialogue and economic modelling to support clinical translation.
Altogether, this review highlights the evolving potential of nanomedicine in dermatology, not only as a scientific innovation but as a viable clinical and commercial strategy. Strategic translational efforts will be essential to move beyond laboratory success and establish nanoformulations as a standard-of-care in acne management.
Author contributions
Hamidreza Kelidari: conceptualization, methodology, data curation, formal analysis, investigation, methodology, writing – original draft, validation. Anna Paula Azevedo de Carvalho: formal analysis, investigation, methodology, writing – original draft. Carlos Adam Conte-Junior: resources, writing – review & editing, funding acquisition, supervision.Conflicts of interest
There are no conflicts to declare.Data availability
There are no data to report; all information enclosed is in the public domain.Acknowledgements
Authors thank the Fundação Carlos Chagas de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ) [grant numbers E-26/201.636/2024, E-26/200.621/2022, E-26/210.385/2022, and E-26/200.891/2021]; and the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) [grant number 313119/2020-1].References
- H. C. Williams, R. P. Dellavalle and S. Garner, Acne vulgaris, Lancet, 2012, 379, 361–372 CrossRef PubMed.
- C.-Y. Huang, I.-J. Chang, N. Bolick, W.-T. Hsu, C.-H. Su, T.-S. Hsieh, I.-H. Huang and C.-C. Lee, Comparative Efficacy of Pharmacological Treatments for Acne Vulgaris: A Network Meta-Analysis of 221 Randomized Controlled Trials, Ann. Fam. Med., 2023, 21, 358–369 CrossRef PubMed.
- J. K. L. Tan and K. Bhate, A global perspective on the epidemiology of acne, Br. J. Dermatol., 2015, 172, 3–12 CrossRef PubMed.
- D. Li, Q. Chen, Y. Liu, T. Liu, W. Tang and S. Li, The prevalence of acne in Mainland China: a systematic review and meta-analysis, BMJ Open, 2017, 7, e015354 CrossRef PubMed.
- B. Dréno, What is new in the pathophysiology of acne, an overview, J. Eur. Acad. Dermatol. Venereol., 2017, 31, 8–12 CrossRef PubMed.
- L. Lu, M. Shi and Z. Chen, Efficacy of IPL therapy for the treatment of acne vulgaris: A meta–analysis, J. Cosmet. Dermatol., 2020, 19, 2596–2605 CrossRef PubMed.
- M. Vasam, S. Korutla and R. A. Bohara, Acne vulgaris: A review of the pathophysiology, treatment, and recent nanotechnology based advances, Biochem. Biophys. Rep., 2023, 36, 101578 CAS.
- A. L. Zaenglein, A. L. Pathy, B. J. Schlosser, A. Alikhan, H. E. Baldwin, D. S. Berson, W. P. Bowe, E. M. Graber, J. C. Harper, S. Kang, J. E. Keri, J. J. Leyden, R. V. Reynolds, N. B. Silverberg, L. F. Stein Gold, M. M. Tollefson, J. S. Weiss, N. C. Dolan, A. A. Sagan, M. Stern, K. M. Boyer and R. Bhushan, Guidelines of care for the management of acne vulgaris, J. Am. Acad. Dermatol., 2016, 74, 945–973 CrossRef PubMed.
- R. Patel and P. Prabhu, Nanocarriers as versatile delivery systems for effective management of acne, Int. J. Pharm., 2020, 579, 119140 CrossRef CAS PubMed.
- G. Plewig and A. M. Kligman, ACNE and ROSACEA, Springer Berlin Heidelberg, Berlin, Heidelberg, 2000 Search PubMed.
- A. S. Karadag, M. Aslan Kayıran, C.-Y. Wu, W. Chen and L. C. Parish, Antibiotic resistance in acne: changes, consequences and concerns, J. Eur. Acad. Dermatol. Venereol., 2021, 35, 73–78 CrossRef CAS PubMed.
- V. Gupta, S. Mohapatra, H. Mishra, U. Farooq, K. Kumar, M. Ansari, M. Aldawsari, A. Alalaiwe, M. Mirza and Z. Iqbal, Nanotechnology in Cosmetics and Cosmeceuticals—A Review of Latest Advancements, Gels, 2022, 8, 173 CrossRef CAS PubMed.
- P. Layegh, N. Mosallaei, D. Bagheri, M. R. Jaafari and S. Golmohammadzadeh, The efficacy of Isotretinoin-loaded solid lipid nanoparticles in comparison to Isotrex® on acne treatment, Nanomed. J., 2013, 1, 38–47 Search PubMed.
- H. R. Kelidari, M. Saeedi, Z. Hajheydari, J. Akbari, K. Morteza-Semnani, J. Akhtari, H. Valizadeh, K. Asare-Addo and A. Nokhodchi, Spironolactone loaded nanostructured lipid carrier gel for effective treatment of mild and moderate acne vulgaris: A randomized, double-blind, prospective trial, Colloids Surf., B, 2016, 146, 47–53 CrossRef CAS PubMed.
- M. Saeedi, K. Morteza-Semnani, J. Akbari, Z. Hajheydari, A. Goodarzi, S. S. Rostamkalaei, S. M. H. Hashemi and S. M. Rahimnia, Green formulation of spironolactone loaded chitosan coated nano lipid carrier for treatment of acne vulgaris: a randomized double-blind clinical trial, Adv. Pharm. Bull., 2023, 14(1), 161–175 Search PubMed.
- R. Najafi-Taher, A. Jafarzadeh kohneloo, V. Eslami Farsani, N. Mehdizade Rayeni, H. R. Moghimi, A. Ehsani and A. Amani, A topical gel of tea tree oil nanoemulsion containing adapalene versus adapalene marketed gel in patients with acne vulgaris: a randomized clinical trial, Arch. Dermatol. Res., 2022, 314, 673–679 CrossRef CAS PubMed.
- B. S. Chandrashekhar, Tretinoin Nanogel 0.025% Versus Conventional Gel 0.025% in Patients with Acne Vulgaris: A Randomized, Active Controlled, Multicentre, Parallel Group, Phase IV Clinical Trial, J. Clin. Diagn. Res., 2015, 9(1), 4–9 Search PubMed.
- B. Bhavsar, B. Choksi, J. Sanmukhani, A. Dogra, R. Haq, S. Mehta, S. Mukherjee, V. Subramanian, S. Sheikh and R. Mittal, Clindamycin 1% Nano-emulsion Gel Formulation for the Treatment of Acne Vulgaris: Results of a Randomized, Active Controlled, Multicentre, Phase IV Clinical Trial, J. Clin. Diagn. Res., 2014, 8 DOI:10.7860/JCDR/2014/9111.4769.
- S. A. Rahman, N. S. Abdelmalak, A. Badawi, T. Elbayoumy, N. Sabry and A. El Ramly, Tretinoin-loaded liposomal formulations: from lab to comparative clinical study in acne patients, Drug Delivery, 2016, 23, 1184–1193 CrossRef PubMed.
- J. Kim, J. Kim, Y. I. Lee, J. Suk, D. Lee and J. H. Lee, A pilot study evaluating the efficacy and safety of retinaldehyde-loaded niosomes against mild-to-moderate acne, J. Cosmet. Dermatol., 2021, 20, 3586–3592 CrossRef PubMed.
- H. Nasri, M. Bahmani, N. Shahinfard, A. Moradi Nafchi, S. Saberianpour and M. Rafieian Kopaei, Medicinal Plants for the Treatment of Acne Vulgaris: A Review of Recent Evidences, Jundishapur J. Microbiol., 2015, 8, 189 Search PubMed.
- N. Dragicevic and H. I. Maibach, Liposomes and Other Nanocarriers for the Treatment of Acne Vulgaris: Improved Therapeutic Efficacy and Skin Tolerability, Pharmaceutics, 2024, 16, 309 CrossRef CAS PubMed.
- L. D. Mahure, P. R. Hatwar, R. L. Bakal and Chetan C. Turankar, Nanotechnology-based approaches for acne treatment: A comprehensive review, GSC Biol. Pharm. Sci., 2025, 31, 156–162 CrossRef CAS.
- P. Rajak, N. Kimneiniang, S. Sarma, B. Bhuyan and H. Pathak, A Comprehensive Overview of Nanoparticles in Acne Management: Current Trends and Future Prospects, Curr. Cosmet. Sci., 2025, 4, e26667797362809 CrossRef.
- V. Singh, R. Redhu, R. Verma, V. Mittal and D. Kaushik, Anti-acne Treatment using Nanotechnology based on Novel Drug Delivery System and Patents on Acne Formulations: A Review, Recent Pat. Nanotechnol., 2021, 15, 331–350 CrossRef CAS PubMed.
- J. Lalrengpuii, K. Raza, A. Mishra and R. Shukla, Retinoid nanoparticulates: Approachable gateway for acne treatment, Health Sci. Rev., 2022, 4, 100042 Search PubMed.
- M. J. Page, J. E. McKenzie, P. M. Bossuyt, I. Boutron, T. C. Hoffmann, C. D. Mulrow, L. Shamseer, J. M. Tetzlaff, E. A. Akl, S. E. Brennan, R. Chou, J. Glanville, J. M. Grimshaw, A. Hróbjartsson, M. M. Lalu, T. Li, E. W. Loder, E. Mayo-Wilson, S. McDonald, L. A. McGuinness, L. A. Stewart, J. Thomas, A. C. Tricco, V. A. Welch, P. Whiting and D. Moher, The PRISMA 2020 statement: an updated guideline for reporting systematic reviews, Br. Med. J., 2021, n71 CrossRef PubMed.
- R. Albash, N. M. Badawi, M. I. A. Hamed, M. H. Ragaie, S. S. Mohammed, R. M. Elbesh, K. M. Darwish, M. O. Lashkar, S. S. Elhady and S. Mosallam, Exploring the Synergistic Effect of Bergamot Essential Oil with Spironolactone Loaded Nano-Phytosomes for Treatment of Acne Vulgaris: In Vitro Optimization, In Silico Studies, and Clinical Evaluation, Pharmaceuticals, 2023, 16, 128 CrossRef CAS PubMed.
- M. Sabouri, A. Samadi, S. Ahmad Nasrollahi, E. S. Farboud, B. Mirrahimi, H. Hassanzadeh, M. Nassiri Kashani, R. Dinarvand and A. Firooz, Tretinoin Loaded Nanoemulsion for Acne Vulgaris: Fabrication, Physicochemical and Clinical Efficacy Assessments, Skin Pharmacol. Physiol., 2018, 31, 316–323 CrossRef CAS PubMed.
- A. Samadi, Z. Sartipi, S. Ahmad Nasrollahi, B. Sheikholeslami, M. Nassiri Kashani, M. R. Rouini, R. Dinarvand and A. Firooz, Efficacy assessments of tretinoin-loaded nano lipid carriers in acne vulgaris: a double blind, split-face randomized clinical study, Arch. Dermatol. Res., 2022, 314, 553–561 CrossRef CAS PubMed.
- S. A. Rahman, N. S. Abdelmalak, A. Badawi, T. Elbayoumy, N. Sabry and A. El Ramly, Formulation of tretinoin-loaded topical proniosomes for treatment of acne: in vitro characterization, skin irritation test and comparative clinical study, Drug Delivery, 2015, 22, 731–739 CrossRef PubMed.
- S. Prasad, A. Mukhopadhyay, A. Kubavat, A. Kelkar, A. Modi, B. Swarnkar, B. Bajaj, M. Vedamurthy, S. Sheikh and R. Mittal, Efficacy and safety of a nano-emulsion gel formulation of adapalene 0.1% and clindamycin 1% combination in acne vulgaris: A randomized, open label, active-controlled, multicentric, phase IV clinical trial, Indian J. Dermatol. Venereol. Leprol., 2012, 78, 459 CrossRef PubMed.
- S. Ahmad Nasrollahi, F. Koohestani, A. Naeimifar, A. Samadi, A. Vatanara and A. Firooz, Preparation and evaluation of adapalene nanostructured lipid carriers for targeted drug delivery in acne, Dermatol. Ther., 2021, 34, e14777 CrossRef CAS PubMed.
- R. Najafi-Taher, A. Jafarzadeh kohneloo, V. Eslami Farsani, N. Mehdizade Rayeni, H. R. Moghimi, A. Ehsani and A. Amani, A topical gel of tea tree oil nanoemulsion containing adapalene versus adapalene marketed gel in patients with acne vulgaris: a randomized clinical trial, Arch. Dermatol. Res., 2022, 314, 673–679 CrossRef CAS PubMed.
- M. El-Samahy, M. A. Sharara and S. S. Abd Elaziz, Effect of Topical Application of Nano Retinol on Mild to Moderate Acne Vulgaris, Egypt. J. Hosp. Med., 2017, 68, 1049–1058 CrossRef.
- H. F. Salem, R. M. Kharshoum, S. M. Awad, M. Ahmed Mostafa and H. A. Abou-Taleb, Tailoring of Retinyl Palmitate-Based Ethosomal Hydrogel as a Novel Nanoplatform for Acne Vulgaris Management: Fabrication, Optimization, and Clinical Evaluation Employing a Split-Face Comparative Study, Int. J. Nanomed., 2021, 16, 4251–4276 CrossRef CAS PubMed.
- J. Kim, J. Kim, Y. I. Lee, J. Suk, D. Lee and J. H. Lee, A pilot study evaluating the efficacy and safety of retinaldehyde–loaded niosomes against mild–to–moderate acne, J. Cosmet. Dermatol., 2021, 20, 3586–3592 CrossRef PubMed.
- S. Mohammadi, A. Pardakhty, M. Khalili, R. Fathi, M. Rezaeizadeh, S. Farajzadeh, A. Mohebbi and M. Aflatoonian, Niosomal Benzoyl Peroxide and Clindamycin Lotion Versus NiosomalClindamycin Lotion in Treatment of Acne Vulgaris: A RandomizedClinical Trial, Adv. Pharm. Bull., 2019, 9, 578–583 CrossRef CAS PubMed.
- I. Tomić, S. Miočić, I. Pepić, D. Šimić and J. Filipović-Grčić, Efficacy and Safety of Azelaic Acid Nanocrystal-Loaded In Situ Hydrogel in the Treatment of Acne Vulgaris, Pharmaceutics, 2021, 13, 567 CrossRef PubMed.
- N. Jurairattanaporn, T. Chalermchai, S. Ophaswongse and M. Udompataikul, Comparative Trial of Silver Nanoparticle Gel and 1% Clindamycin Gel when Use in Combination with 2.5% Benzoyl Peroxide in Patients with Moderate Acne Vulgaris, J. Med. Assoc. Thai., 2017, 100, 78–85 Search PubMed.
- S. S. Amer, W. Mamdouh, M. Nasr, A. ElShaer, E. Polycarpou, R. T. A. Abdel-Aziz and O. A. Sammour, Quercetin loaded cosm-nutraceutical electrospun composite nanofibers for acne alleviation: Preparation, characterization and experimental clinical appraisal, Int. J. Pharm., 2022, 612, 121309 CrossRef CAS PubMed.
- S. S. Amer, M. Nasr, R. T. A. Abdel-Aziz, N. H. Moftah, A. El Shaer, E. Polycarpou, W. Mamdouh and O. Sammour, Cosm-nutraceutical nanovesicles for acne treatment: Physicochemical characterization and exploratory clinical experimentation, Int. J. Pharm., 2020, 577, 119092 CrossRef CAS PubMed.
- H. Abd-allah, M. H. Ragaie and E. Elmowafy, Unraveling the pharmaceutical and clinical relevance of the influence of syringic acid loaded linoleic acid transferosomes on acne, Int. J. Pharm., 2023, 639, 122940 CrossRef CAS PubMed.
- S. Lueangarun, K. Sriviriyakul, T. Tempark, C. Managit and P. Sithisarn, Clinical efficacy of 0.5% topical mangosteen extract in nanoparticle loaded gel in treatment of mild–to–moderate acne vulgaris: A 12−week, split–face, double–blinded, randomized, controlled trial, J. Cosmet. Dermatol., 2019, 18, 1395–1403 CrossRef PubMed.
- S. Lueangarun, K. Sriviriyakul, T. Tempark, C. Managit and P. Sithisarn, Clinical efficacy of 0.5% topical mangosteen extract in nanoparticle loaded gel in treatment of mild-to-moderate acne vulgaris: A 12-week, split-face, double-blinded, randomized, controlled trial, J. Cosmet. Dermatol., 2019, 18, 1395–1403 CrossRef PubMed.
- H. F. Salem, R. M. Kharshoum, S. M. Awad, M. A. Mostafa and H. A. Abou-Taleb, Tailoring of retinyl palmitate-based ethosomal hydrogel as a novel nanoplatform for acne vulgaris management: Fabrication, optimization, and clinical evaluation employing a split-face comparative study, Int. J. Nanomed., 2021, 16, 4251–4276 CrossRef CAS PubMed.
- Y. Huang, X. Guo, Y. Wu, X. Chen, L. Feng, N. Xie and G. Shen, Nanotechnology's frontier in combatting infectious and inflammatory diseases: prevention and treatment, Signal Transduction Targeted Ther., 2024, 9, 34 CrossRef PubMed.
- A. P. A. de Carvalho, R. É. P. de D. Santos and C. A. Conte-Junior, Nanocellulose-based platforms as a multipurpose carrier for drug and bioactive compounds: from active packaging to transdermal and anticancer applications, Int. J. Pharm., 2024, 123851 CrossRef PubMed.
- R. C. de Resende, O. M. M. S. Viana, J. T. J. Freitas, R. Bonfilio, A. L. M. Ruela and M. B. de Araújo, Analysis of spironolactone polymorphs in active pharmaceutical ingredients and their effect on tablet dissolution profiles, Braz. J. Pharm. Sci., 2016, 52, 613–621 CrossRef.
- A. Goodfellow, J. Alaghband-Zadeh, G. Carter, J. J. Cream, S. Holland, J. Scully and P. Wise, Oral spironolactone improves acne vulgaris and reduces sebum excretion, Br. J. Dermatol., 1984, 111, 209–214 CrossRef CAS PubMed.
- M. Santer and A. Layton, What do we know about prescribing spironolactone for acne?, Br. Med. J., 2023, 1114 CrossRef PubMed.
- E. M. Al-Edani, M. Abo Zeid, K. Khalefa, A. W. Abbas, A. A. Aboali, H. A. S. Raslan, Z. Shakhatreh, I. A. Yakout and A. Elrosasy, Oral and topical spironolactone in acne treatment: A meta-analysis of effectiveness and safety, Naunyn. Schmiedebergs, Arch. Pharmacol., 2025, 398, 9005–9019 CrossRef CAS PubMed.
- R. Muller, R. Petersen, A. Hommoss and J. Pardeike, Nanostructured lipid carriers (NLC) in cosmetic dermal products, Adv. Drug Delivery Rev., 2007, 59, 522–530 CrossRef CAS PubMed.
- R. Shamma and M. Aburahma, Follicular delivery of spironolactone via nanostructured lipid carriers for management of alopecia, Int. J. Nanomed., 2014, 5449 CrossRef CAS PubMed.
- H. R. Kelidari, M. Saeedi, J. Akbari, K. Morteza-Semnani, P. Gill, H. Valizadeh and A. Nokhodchi, Formulation optimization and in vitro skin penetration of spironolactone loaded solid lipid nanoparticles, Colloids Surf., B, 2015, 128, 473–479 CrossRef CAS PubMed.
- P. Sun, L. Zhao, N. Zhang, C. Wang, W. Wu, A. Mehmood, L. Zhang, B. Ji and F. Zhou, Essential Oil and Juice from Bergamot and Sweet Orange Improve Acne Vulgaris Caused by Excessive Androgen Secretion, Mediators Inflammation, 2020, 2020, 1–10 Search PubMed.
- J. Varani, R. L. Warner, M. Gharaee-Kermani, S. H. Phan, S. Kang, J. Chung, Z. Wang, S. C. Datta, G. J. Fisher and J. J. Voorhees, Vitamin A Antagonizes Decreased Cell Growth and Elevated Collagen-Degrading Matrix Metalloproteinases and Stimulates Collagen Accumulation in Naturally Aged Human Skin1, J. Invest. Dermatol., 2000, 114, 480–486 CrossRef CAS PubMed.
- N. Akombaetwa, A. B. Ilangala, L. Thom, P. B. Memvanga, B. A. Witika and A. B. Buya, Current Advances in Lipid Nanosystems Intended for Topical and Transdermal Drug Delivery Applications, Pharmaceutics, 2023, 15, 656 CrossRef CAS PubMed.
- T. Karlsson, A. Vahlquist, N. Kedishvili and H. Törmä, 13-cis-Retinoic acid competitively inhibits 3α-hydroxysteroid oxidation by retinol dehydrogenase RoDH-4: a mechanism for its anti-androgenic effects in sebaceous glands?, Biochem. Biophys. Res. Commun., 2003, 303, 273–278 CrossRef CAS PubMed.
- C. E. Orfanos, C. C. Zouboulis, B. Almond-Roesler and C. C. Geilen, Current Use and Future Potential Role of Retinoids in Dermatology, Drugs, 1997, 53, 358–388 CrossRef CAS PubMed.
- A. Thielitz, A. Krautheim and H. Gollnick, Update in retinoid therapy of acne, Dermatol. Ther., 2006, 19, 272–279 CrossRef PubMed.
- B. Dreno, S. Kang, J. Leyden and J. York, Update: Mechanisms of Topical Retinoids in Acne, J. Drugs Dermatol., 2022, 21, 734–740 Search PubMed.
- R. A. Bojar, W. J. Cunliffe and K. T. Holland, Disruption of the transmembrane pH gradient—a possible mechanism for the antibacterial action of azelaic acid in Propionibucterium acnes and Staphylococcus epidermidis, J. Antimicrob. Chemother., 1994, 34, 321–330 CrossRef CAS PubMed.
- M. Detmar, A. Mayer-da-Silva, R. Stadler and C. E. Orfanos, Effects of Azelaic Acid on Proliferation and Ultrastructure of Mouse Keratinocytes in Vitro, J. Invest. Dermatol., 1989, 93, 70–74 CrossRef CAS PubMed.
- A. Mastrofrancesco, M. Ottaviani, N. Aspite, G. Cardinali, E. Izzo, K. Graupe, C. C. Zouboulis, E. Camera and M. Picardo, Azelaic acid modulates the inflammatory response in normal human keratinocytes through PPARγ activation, Exp. Dermatol., 2010, 19, 813–820 CrossRef CAS PubMed.
- S. Briganti, E. Flori, A. Mastrofrancesco, D. Kovacs, E. Camera, M. Ludovici, G. Cardinali and M. Picardo, Azelaic acid reduced senescence–like phenotype in photo–irradiated human dermal fibroblasts: possible implication of PPARγ, Exp. Dermatol., 2013, 22, 41–47 CrossRef CAS PubMed.
- X. Feng, J. Shang, Z. Gu, J. Gong, Y. Chen and Y. Liu, Azelaic Acid: Mechanisms of Action and Clinical Applications, Clin., Cosmet. Invest. Dermatol., 2024, 17, 2359–2371 CrossRef CAS PubMed.
- W. J. Cunliffe, K. T. Holland, R. Bojar and S. F. Levy, A randomized, double-blind comparison of a clindamycin phosphate/benzoyl peroxide gel formulation and a matching clindamycin gel with respect to microbiologic activity and clinical efficacy in the topical treatment of acne vulgaris, Clin. Ther., 2002, 24, 1117–1133 CrossRef CAS PubMed.
- J. Del Rosso, J. Sugarman, L. Green, T. Lain, O. Levy-Hacham, R. Mizrahi and L. S. Gold, Efficacy and safety of microencapsulated benzoyl peroxide and microencapsulated tretinoin for the treatment of acne vulgaris: Results from two phase 3 double-blind, randomized, vehicle-controlled studies, J. Am. Acad. Dermatol., 2023, 89, 719–727 CrossRef CAS PubMed.
- J. Del Rosso, What is the Role of Benzoyl Peroxide Cleansers in Acne Management?, J. Clin. Aesthet. Dermatol., 2008, 1, 48–51 Search PubMed.
- J. Del Rosso, M. K. Armillei, I. B. Lomakin, A. Grada and C. G. Bunick, Clindamycin: A Comprehensive Status Report with Emphasis on Use in Dermatology, J. Clin. Aesthet. Dermatol., 2024, 17, 29–40 Search PubMed.
- P. R. More, S. Pandit, A. De Filippis, G. Franci, I. Mijakovic and M. Galdiero, Silver Nanoparticles: Bactericidal and Mechanistic Approach against Drug Resistant Pathogens, Microorganisms, 2023, 11, 369 CrossRef CAS PubMed.
- P. Sinha, S. Srivastava, N. Mishra and N. P. Yadav, New Perspectives on Antiacne Plant Drugs: Contribution to Modern Therapeutics, Biomed Res. Int., 2014, 2014, 1–19 CrossRef PubMed.
- R. Puspadewi, T. Milanda, M. Muhaimin and A. Y. Chaerunisaa, Nanoparticle-Encapsulated Plant Polyphenols and Flavonoids as an Enhanced Delivery System for Anti-Acne Therapy, Pharmaceuticals, 2025, 18, 209 CrossRef CAS PubMed.
- A. Lewińska, M. Domżał-Kędzia, E. Maciejczyk, M. Łukaszewicz and U. Bazylińska, Design and Engineering of “Green” Nanoemulsions for Enhanced Topical Delivery of Bakuchiol Achieved in a Sustainable Manner: A Novel Eco-Friendly Approach to Bioretinol, Int. J. Mol. Sci., 2021, 22, 10091 CrossRef PubMed.
- M. H. Siard, K. E. McMurry, D. W. Horohov and A. A. Adams, Effects of polyphenolic bioactive compounds (pterostilbene, resveratrol, curcuminoids, quercetin, and hydroxypterostilbene) on pro-inflammatory cytokine production in vitro, J. Equine Vet. Sci., 2013, 33, 343 CrossRef.
- S.-J. Choi, B. H. Tai, N. M. Cuong, Y.-H. Kim and H.-D. Jang, Antioxidative and anti-inflammatory effect of quercetin and its glycosides isolated from mampat (Cratoxylum formosum), Food Sci. Biotechnol., 2012, 21, 587–595 CrossRef CAS.
- G. Chen-yu, Y. Chun-fen, L. Qi-lu, T. Qi, X. Yan-wei, L. Wei-na and Z. Guang-xi, Development of a Quercetin-loaded nanostructured lipid carrier formulation for topical delivery, Int. J. Pharm., 2012, 430, 292–298 CrossRef PubMed.
- D. Aggarwal, M. Chaudhary, S. K. Mandotra, H. S. Tuli, R. Chauhan, N. C. Joshi, D. Kaur, L. Dufossé and A. Chauhan, Anti-inflammatory potential of quercetin: From chemistry and mechanistic insight to nanoformulations, Curr. Res. Pharmacol. Drug Discovery, 2025, 8, 100217 CrossRef CAS PubMed.
- Q. Tan, W. Liu, C. Guo and G. Zhai, Preparation and evaluation of quercetin-loaded lecithin-chitosan nanoparticles for topical delivery, Int. J. Nanomed., 2011, 6, 1621–1630 CAS.
- S.-M. Tang, X.-T. Deng, J. Zhou, Q.-P. Li, X.-X. Ge and L. Miao, Pharmacological basis and new insights of quercetin action in respect to its anti-cancer effects, Biomed. Pharmacother., 2020, 121, 109604 CrossRef CAS PubMed.
- B. H. Havsteen, The biochemistry and medical significance of the flavonoids, Pharmacol. Ther., 2002, 96, 67–202 CrossRef CAS PubMed.
- I. Bartel, I. Mandryk, J. O. Horbańczuk, A. Wierzbicka and M. Koszarska, Nutraceutical Properties of Syringic Acid in Civilization Diseases—Review, Nutrients, 2023, 16, 10 CrossRef PubMed.
- N. Assalem, H. Abd-allah, M. H. Ragaie, S. S. Ahmed and E. Elmowafy, Therapeutic potential of limonene-based syringic acid nanoemulsion: Enhanced ex vivo cutaneous deposition and clinical anti-psoriatic efficacy, Int. J. Pharm., 2024, 660, 124376 CrossRef CAS PubMed.
- F. Rouvier, L. Abou, E. Wafo and J. M. Brunel, Linoleic Fatty Acid from Rwandan Propolis: A Potential Antimicrobial Agent Against Cutibacterium acnes, Curr. Issues Mol. Biol., 2025, 47, 162 CrossRef CAS PubMed.
- E. Jourdan, S. Trompezinski, S. Weber, B. Cadars, F. Larue, N. Ardiet, M. Chavagnac-Bonneville and M. Sayag, Assessment of a new biological complex efficacy on dysseborrhea, inflammation, and Propionibacterium acnes proliferation, Clin., Cosmet. Invest. Dermatol., 2016, 9, 233–239 CrossRef PubMed.
- S. Mosallam, M. H. Ragaie, N. H. Moftah, A. H. Elshafeey and A. A. Abdelbary, Use of Novasomes as a Vesicular Carrier for Improving the Topical Delivery of Terconazole: In Vitro Characterization, In Vivo Assessment and Exploratory Clinical Experimentation, Int. J. Nanomed., 2021, 16, 119–132 CrossRef PubMed.
- R. Kumar, A. Sirvi, S. Kaur, S. K. Samal, S. Roy and A. T. Sangamwar, Polymeric micelles based on amphiphilic oleic acid modified carboxymethyl chitosan for oral drug delivery of bcs class iv compound: Intestinal permeability and pharmacokinetic evaluation, Eur. J. Pharm. Sci., 2020, 153, 105466 CrossRef CAS PubMed.
- T. Wang, N. S. Cauchon, J. P. Kirwan, M. K. Joubert, M. Algorri, B. Bell, R. J. Soto and D. J. Semin, Advancing the implementation of innovative analytical technologies in pharmaceutical manufacturing—Some regulatory considerations, J. Pharm. Sci., 2025, 114, 816–828 CrossRef CAS PubMed.
- D. Chiforisin and H. B. Grangeia, Quality by Design in nanotechnology applied to cosmetics: A systematic review from conception to production, J. Dermatol. Sci. Cosmet. Technol., 2026, 3, 100145 CrossRef.
- G. Amasya, C. Ozturk, B. Aksu and N. Tarimci, QbD based formulation optimization of semi-solid lipid nanoparticles as nano-cosmeceuticals, J. Drug Delivery Sci. Technol., 2021, 66, 102737 CrossRef CAS.
- A. C. Correia, J. N. Moreira, J. M. Sousa Lobo and A. C. Silva, Design of experiment (DoE) as a quality by design (QbD) tool to optimise formulations of lipid nanoparticles for nose-to-brain drug delivery, Expert Opin. Drug Delivery, 2023, 20, 1731–1748 CrossRef CAS PubMed.
- Y. Ijjeh, N. Alsarayreh, A. Rifai, H. Abdelnabi, S. Al-Mahamid, D. A. Alqudah, H. Nsairat, Z. Lafi, M. A. Mousa, Y. Al-Hiari and W. Alshaer, Quality by digital design for accelerated sustainable nanomedicine development, Eur. J. Pharm. Sci., 2025, 213, 107239 CrossRef CAS PubMed.
- R. Maheshwari, D. Kapoor, S. Polaka, S. Bhattacharya and B. Prajapati, Roadmap for Commercial Nanomedicine Development: Integrating Quality by Design Principles with Pharmaceutical Nanotechnology, Mol. Pharm., 2025, 22, 4337–4372 CrossRef CAS PubMed.
- J. Bian, N. Gobalasingham, A. Purchel and J. Lin, The Power of Field-Flow Fractionation in Characterization of Nanoparticles in Drug Delivery, Molecules, 2023, 28, 4169 CrossRef CAS PubMed.
- J. Kuntsche, J. C. Horst and H. Bunjes, Cryogenic transmission electron microscopy (cryo-TEM) for studying the morphology of colloidal drug delivery systems, Int. J. Pharm., 2011, 417, 120–137 CrossRef CAS PubMed.
- Z. Wei, S. Zhuo, Y. Zhang, L. Wu, X. Gao, S. He, X. Bo and W. Zhou, Machine learning reshapes the paradigm of nanomedicine research, Acta Pharm. Sin. B, 2025 DOI:10.1016/j.apsb.2025.05.014.
- C. F. Amantino, S. R. do Amaral, M. Aires-Fernandes, S. M. Oliani, A. C. Tedesco and F. L. Primo, Development of 3D skin equivalents for application in photodynamic biostimulation therapy assays using curcumin nanocapsules, Heliyon, 2024, 10, e32808 CrossRef CAS PubMed.
- I. Negut, C. Mazzanti, R. Laurano, G. Ciardelli, S. Bronco and C. S. Oliveira, From 1D microbiological assays to 3D advanced skin models: enhancing preclinical strategies to unravel the impact of bioactive textiles on the human skin microbiome, Front. Cell. Infect. Microbiol., 2025, 15, 1676663 CrossRef PubMed.
- B. S. Kwak, S. Jin, S. J. Kim, E. J. Kim, J. H. Chung and J. H. Sung, Microfluidic skin chip with vasculature for recapitulating the immune response of the skin tissue, Biotechnol. Bioeng., 2020, 117, 1853–1863 CrossRef CAS PubMed.
- Y. Huang, X. Wu, Y. Xu, N. Yang, P. Xi, Y. Wang, Y. Zhu and X. Chen, Organoids/organs-on-chips towards biomimetic human artificial skin, Burns Trauma, 2025, 13, tkaf029 CrossRef PubMed.
- D. M. Thiboutot, A. M. Layton, M.-M. Chren, E. A. Eady and J. Tan, Assessing effectiveness in acne clinical trials: steps towards a core outcome measure set, Br. J. Dermatol., 2019, 181, 700–706 CrossRef CAS PubMed.
- A. M. Layton, E. A. Eady, D. M. Thiboutot, J. Tan, F. Abad-Casintahan, K. Bhate, F. Collier, F. Cowdell, C. Donkor, L. Kircik, M. Landis, S. Marchbein, S. Nijland, F. Ochsendorf, N. Silverberg, G. Webster and J. Weiss, Identifying What to Measure in Acne Clinical Trials: First Steps towards Development of a Core Outcome Set, J. Invest. Dermatol., 2017, 137, 1784–1786 CrossRef CAS PubMed.
- W. N. Kernan, C. M. Viscoli, R. W. Makuch, L. M. Brass and R. I. Horwitz, Stratified Randomization for Clinical Trials, J. Clin. Epidemiol., 1999, 52, 19–26 CrossRef CAS PubMed.
- D. Potvin, P. D'Angelo, S. Bennett, J. Jankicevic and R. Bissonnette, Adaptive designs in dermatology clinical trials: Current status and future perspectives, J. Eur. Acad. Dermatol. Venereol., 2024, 38, 1694–1703 CrossRef CAS PubMed.
- T. Torres Moral, A. Sanchez-Niubo, A. Monistrol-Mula, C. Gerardi, R. Banzi, P. Garcia, J. Demotes-Mainard and J. Haro, Methods for Stratification and Validation Cohorts: A Scoping Review, J. Pers. Med., 2022, 12, 688 CrossRef PubMed.
- T. Evrenoglou, Stratification according to disease severity can better reveal the relative effectiveness of treatments for acne vulgaris, Br. J. Dermatol., 2022, 187, 637–638 CrossRef PubMed.
- Y. Tu and L. A. Renfro, Latest Developments in “Adaptive Enrichment” Clinical Trial Designs in Oncology, Ther. Innov. Regul. Sci., 2024, 58, 1201–1213 CrossRef PubMed.
- S. Đorđević, M. M. Gonzalez, I. Conejos-Sánchez, B. Carreira, S. Pozzi, R. C. Acúrcio, R. Satchi-Fainaro, H. F. Florindo and M. J. Vicent, Current hurdles to the translation of nanomedicines from bench to the clinic, Drug Delivery Transl. Res., 2022, 12, 500–525 CrossRef PubMed.
Footnote |
| † Co-first authors. These authors contributed equally to this manuscript, and each has the right to list themselves first in author order on their CVs. |
| This journal is © The Royal Society of Chemistry 2026 |