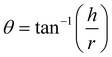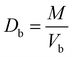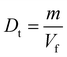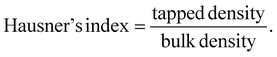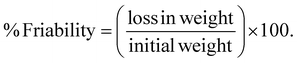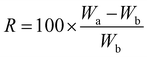Open Access Article
Open Access ArticleEnhancing the performance of granisetron HCl orodispersible tablets using co-processed MCC-mannitol excipients: a direct compression approach
Shazia Naseem Chaudhry and
Atiq Ur Rahman *
*
Riphah Institute of Pharmaceutical Sciences (RIPS), Riphah International University, Lahore Campus, Lahore, Pakistan. E-mail: atiq.wellness@hotmail.com; Tel: +92-321-4242781
First published on 20th March 2026
Abstract
Orodispersible tablets (ODTs) are favored for their rapid disintegration in the mouth without the need for water, thereby improving patient compliance, particularly in dysphagic, geriatric, and oncology patients. Advanced manufacturing technologies like Orasolv®, Durasolv®, and Zydis® offer unique benefits but are generally associated with high costs and intricate processes. Common manufacturing approaches include molding, freeze-drying, and direct compression, with the latter being the most economical and industrially practical method. The present study aimed to develop granisetron HCl ODTs using a laboratory-prepared co-processed excipient system composed of microcrystalline cellulose (MCC) and mannitol. The system was developed by combining the excipients at the sub-particle level to enhance flowability and compressibility, rather than relying on commercially available multifunctional excipients. The performance of this co-processed MCC–mannitol system was systematically compared with that of the corresponding physical mixtures. Granisetron HCl is a 5-HT3 receptor antagonist commonly prescribed to manage nausea and vomiting induced by chemotherapy. It is well-suited for ODTs due to its low dosage requirements and water solubility. In the formulations, microcrystalline cellulose and mannitol were used as bulking agents and sodium starch glycolate (SSG) and crospovidone (CP) as superdisintegrants. The performance of co-processed excipients was compared with that of physical mixtures. Precompression parameters, including the angle of repose and compressibility index, along with post-compression characteristics such as wetting time, water absorption ratio, disintegration time, and in vitro drug release, were evaluated. Among the tested formulations, formulation F7 containing 4% crospovidone and co-processed MCC–mannitol exhibited the most favorable performance, with a rapid disintegration time of 10 ± 0.12 s and 98.14% ± 0.25% drug release within 180 s. Comparative evaluation demonstrated that the co-processed excipient system provided improved powder flow and tablet performance relative to the physical mixtures. These findings indicate that at the sub-particle level, the co-processing of conventional excipients can be an effective and practical strategy to enhance the ODT performance using a cost-effective direct compression approach.
1. Introduction
Patient compliance supports effective therapy, and advances in science have led to the development of novel drug delivery systems (NDDSs).1 Among these innovations, orodispersible tablets (ODTs) have emerged as a promising solution to the issues associated with traditional solid dosage forms, particularly for individuals experiencing dysphagia, which affects around 35% of the general population and 30%–40% of older adults. Dysphagia, marked by difficulties in swallowing, can result from various conditions such as age-related physiological changes and neurological disorders.2 Conventional tablets often require water for administration, posing challenges for patients with limited fluid intake. In contrast, ODTs are formulated to dissolve swiftly in the oral cavity without the need for water, offering convenient drug delivery, especially in geriatric and pediatric populations. The quick disintegration upon contact with saliva promotes rapid absorption, thereby improving bioavailability.3 However, formulating ODTs involves several challenges including achieving a pleasant taste and rapid disintegration. Various techniques are employed in the preparation of ODTs tailored to different drug compounds.4 Freeze-drying is effective for heat-sensitive drugs.5 Sublimation techniques form pores in tablets by incorporating volatile substances, which decrease the disintegration time.6 Spray drying produces fine powders that dissolve quickly. Molding methods, including compression and heat molding, produce tablets that disintegrate rapidly through moistening and solidification with water-soluble sugars.7 Mass extrusion softens drug blends into a tablet form. Hot melt granulation employs molten binders to form granules without solvents and direct compression is a blending approach that utilizes modern disintegrates.8 The performance of ODTs prepared by direct compression is strongly influenced by the physicomechanical properties of excipients, particularly their flowability and compressibility. Co-processing is an established approach in which two or more excipients are combined at the sub-particle level to produce a composite material with improved functional properties compared to simple physical mixtures. Unlike commercially available multifunctional co-processed excipients, laboratory-prepared co-processed systems allow targeted modification of excipient behavior while retaining the regulatory familiarity of individual components. In this study, microcrystalline cellulose and mannitol were co-processed to improve powder flow and compaction behavior, and their performance was systematically compared with the corresponding physical mixtures for the development of granisetron HCl orodispersible tablets.Granisetron is a white crystalline powder that is freely soluble in water, sparingly soluble in methylene chloride, and slightly soluble in methanol. Chemically, it is 1-methyl-N-[(3-endo)-9-methyl-9-azabicyclo[3.3.1]non-3-yl]-1H-indazole-3-carboxamide (Fig. 1) with a low molecular weight of 348.87 g mol−1.9,10 Its mild bitter taste makes it an ideal candidate for formulation as an orodispersible tablet (ODT).
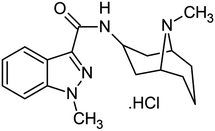 | ||
| Fig. 1 Structure of granisetron HCl.10 | ||
2. Materials and methodology
2.1. Materials
Granisetron HCl (Wuhan Biocause, China) was generously provided by CCL Pharmaceuticals, Lahore, Pakistan. Microcrystalline cellulose (JRS Pharma, Germany), sodium starch glycolate (JRS Pharma, Germany), bubble gum flavor (SMC, Pakistan), sucralose (Algemix, India), xanthan gum (Fengchen, China), colloidal silicon dioxide (Evonik, Germany), magnesium stearate (Peter Greven Asia, Singapore), Mannitol-D (Qingdo Bright Moon Seawood, China), and crospovidone (Trendchems, China) were supplied by Tagma Pharma (Pvt) Ltd, Lahore, Pakistan. All chemicals, solvents, and reagents used were of analytical grade.2.2. Instruments used
Disintegration apparatus (Pharma Test Germany), dissolution apparatus (Pharma Test Germany), double-beam UV/Visible spectrophotometer (UV1800 Shimadzu Corporation, Kyoto, Japan), electronic weighing balance (Panther BM-320, USA), Fourier transform infrared spectrometer (Agilent Cary 630, USA), pH meter (FP20 Mettler Toledo China), Friabilator (2020-Curio Pak), digital hardness tester (PTB-511 Pharma Test Germany), stability chamber (China), digital Vernier calliper (1112-150 INSIZE China), Zp-19 tablet press machine (China), mortar & pestle, Petri dishes, graduated cylinders, glass beakers, measuring flasks, and pipettes (Pyrex).2.3. Methodology
To develop optimal orodispersible tablets (ODTs), seven formulations were prepared by adjusting the excipients and their concentrations to achieve the desired orodispersibility. The excipients used included microcrystalline cellulose and mannitol as fillers, sodium starch glycolate and crospovidone as superdisintegrants, sucralose as sweetener and bubble gum flavor as flavorant, xanthan gum to improve mouthfeel, colloidal silicon dioxide as a glidant, and magnesium stearate as a lubricant. Two forms of excipients were evaluated: physical mixtures and co-processed blends of microcrystalline cellulose and mannitol. For the co-processing method, equal amounts of mannitol and microcrystalline cellulose were combined. The microcrystalline cellulose was dispersed in distilled water, and the mannitol was dissolved in the same water. These two mixtures were then thoroughly combined to form a uniform slurry. This slurry was spread onto trays and initially dried in a tray dryer at 50–60 °C for six hours. After semi-drying, the mixture was passed through mesh sieve no 20, to break up larger particles and then dried further at 40–50 °C for an additional two hours. The mix was sieved through mesh sieve no 30, to obtain fine uniform granules, which were then used in the formulation of the ODTs. Granisetron HCl orodispersible tablets were subsequently formulated using the direct compression technique. The excipients were precisely weighed according to the designated formulations (Table 1). The drug and excipients, excluding bubble gum flavor and magnesium stearate, were mixed geometrically. The bubble gum flavor was blended with a portion of the powder mix before being combined with the remaining blend. Magnesium stearate was then added lastly to the final mix. The powder blend was then compressed into tablets using a ZP-19 rotary punch machine with 7 mm-round punches.| Ingredientsb | F1 | F2 | F3 | F4 | F5 | F6 | F7 |
|---|---|---|---|---|---|---|---|
| PMa + DC | PMa + DC | PMa + DC | PMa + DC | PMa + DC | CP + DC | CP + DC | |
| a PM = physical mixture, DC = direct compression, and CP = co-processed.b Ingredient quantities are given as mg per tab. | |||||||
| Granisetron HCl (mg) | 1.1 | 1.1 | 1.1 | 1.1 | 1.1 | 1.1 | 1.1 |
| Microcrystalline cellulose (mg) | 60 | 58 | 58 | 58 | 58 | 58 | 58 |
| Mannitol (mg) | 60 | 58 | 58 | 58 | 58 | 58 | 58 |
| Sodium starch glycolate (mg) | 5.08 | 5.08 | 5.08 | 5.08 | — | 5.08 | — |
| Crospovidone (mg) | — | — | — | — | 5.08 | — | 5.08 |
| Sucralose (mg) | — | 2 | 2 | 2 | 2 | 2 | 2 |
| Xanthan gum (mg) | — | 0.3175 | 0.3175 | 0.3175 | 0.3175 | 0.3175 | |
| Colloidal silicon dioxide (mg) | — | — | — | 1.27 | 1.27 | 1.27 | 1.27 |
| Bubble gum flavor (ml) | — | 0.005 | 0.003 | 0.003 | 0.003 | 0.003 | 0.003 |
| Magnesium stearate (mg) | 1.905 | 1.905 | 1.905 | 1.27 | 1.27 | 1.27 | 1.27 |
| Weight/tablet (mg) | 127 | 127 | 127 | 127 | 127 | 127 | 127 |
2.4. Method development
2.5. Pre-compression evaluation of orodispersible tablets
The Angle of Repose was calculated using the following formula:11
where M is the mass of the powder and Vb is the bulk volume, expressed in g ml−1.
where m is the weight of the powder or granules (g) and Vf is the final tapped volume.
2.6. Post-compression evaluation
where R is the water absorption ratio, Wa is the weight after absorption, and Wb is the weight before absorption.19
The USP Paddle apparatus 2, commonly used for ODTs, was operated at 50 rpm with 500 ml phosphate buffer (pH 6.8) as the dissolution medium. The samples were withdrawn at specified time intervals, filtered, and analyzed spectrophotometrically at 302 nm.
The goodness of fit for each model was assessed by the coefficients of regression (r2) using the DD solver Excel add-in program.23
2.7. Drug excipient compatibility study (FTIR spectroscopy)
Fourier transform infrared (FTIR) spectral measurements were performed using an Agilent Technologies Fourier Transform Infrared Spectrophotometer (Agilent Cary 630, USA). The scans were recorded in the range of 4000 to 400 cm−1.24The FTIR spectra of the physical, co-processed mix of microcrystalline cellulose with mannitol, granisetron, and promising formulation F7 were recorded to analyze the interactions between components.
2.8. Taste evaluation study
Taste and mouthfeel are important factors of ODTs that impact patient acceptance and compliance. It was conducted with a panel of seven healthy volunteers of both gender, ages between 18 and 60 years, assessing various sensory attributes for each formulation before incorporating the active ingredient. A fasting period of 60 minutes was given to the volunteers before starting the evaluation. Then each member was given 250 ml of water for flushing of mouth and taste buds. After 10 minutes, one tablet from each formulation (without any active ingredient) was placed on the tongue of each volunteer, and the sensory responses were noted for various attributes including mouthfeel (gritty or smooth), residue (left or no residue), chalkiness (present or absent), and taste (unacceptable, acceptable, good, or excellent). The process was repeated for all formulations at 30-minute intervals. Each attribute was scored as follows: taste (0–30, where unacceptable = 0, acceptable = 1–10, good = 11–20, excellent = 21–30); chalkiness (yes = 0, minimal to no = 8–10); grittiness (gritty = 0, minimal grit to smooth = 8–10); and residue (yes = 0, minimal to no = 8–10). A piece of bread and water were provided after each evaluation to clear the taste buds before the next round.2.9. Stability study
A six-month stability study was conducted on F5 and F7, chosen for their rapid disintegration times compared to other formulations. Following the ICH Zone IV guidelines (40 °C ± 2 °C/75% RH ± 5%), the study assessed weight variations, hardness, friability, disintegration, and drug content to confirm the physical and chemical stability.252.10. Statistical analysis
All experimental data are expressed as mean ± standard deviation (SD) and analyzed using GraphPad Prism version 8.0.1. Each experiment was performed in triplicate unless otherwise stated. Statistical comparisons between formulations were carried out using Student's t-test or one-way analysis of variance (ANOVA). For stability studies, results obtained at different time points were statistically compared with initial values in accordance with ICH Q1A guidelines. Differences were considered statistically significant at p < 0.05. Statistical significance is indicated in the respective tables and figures.3. Results and discussion
3.1. Precompression evaluations
The flow properties of physical and co-processed mixtures of mannitol and microcrystalline cellulose (MCC) in a 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
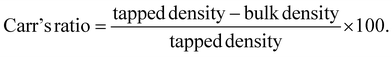
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 ratio were evaluated prior to incorporation into the powder blends (Table 1). The physical mix showed good flow, with an Angle of Repose between 30 and 35°, a Hausner's ratio of 1.15, and a Carr's index of 13.3. In contrast, the co-processed mix exhibited superior flow behavior, with a lower angle of repose (29.88°), reduced Hausner's ratio (1.10), and lower Carr's index (9.42) (Fig. 3), indicating excellent flow properties suitable for direct compression.
50 ratio were evaluated prior to incorporation into the powder blends (Table 1). The physical mix showed good flow, with an Angle of Repose between 30 and 35°, a Hausner's ratio of 1.15, and a Carr's index of 13.3. In contrast, the co-processed mix exhibited superior flow behavior, with a lower angle of repose (29.88°), reduced Hausner's ratio (1.10), and lower Carr's index (9.42) (Fig. 3), indicating excellent flow properties suitable for direct compression.
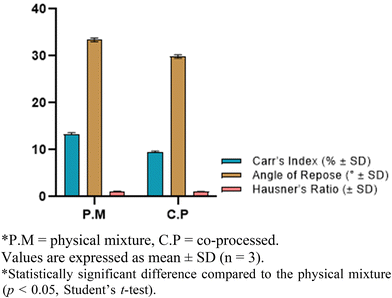 | ||
| Fig. 3 Angle of repose, Carr's index and Hausner's ratio of the physical mix and co-processed mix of mannitol and MCC. Values are expressed as mean ± SD (n = 3). | ||
The enhanced flow of the co-processed system can be attributed to the sub-particle level integration of MCC and mannitol, which improves particle packing efficiency and reduces inter-particle friction compared to simple physical mixtures. This modification is particularly advantageous for direct compression processes, where uniform die filling is critical for low-dose drugs such as granisetron HCl.
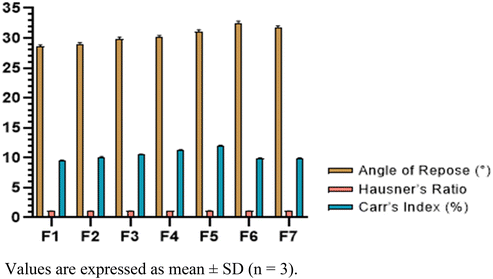
The physico-mechanical properties of all formulations (F1–F7) were also evaluated after blending with active ingredients and excipients. The angle of repose ranged from 28.63 ± 0.31° to 32.46 ± 0.37°, Hausner's ratio from 1.10 ± 0.23 to 1.14 ± 0.22, and Carr's index from 9.5 ± 0.32 to 12.4 ± 0.21, indicating good flowability across all blends. Notably, formulations incorporating co-processed fillers consistently showed Hausner's ratios below 1.12 and Carr's indices below 10 (Fig. 4), confirming the functional advantage of co-processing over physical blending.
3.2. Post-compression evaluations
Post-compression evaluation confirmed that all orodispersible tablet formulations complied with pharmacopeial specifications. The tablets exhibited thicknesses ranging from 3.0 to 3.1 mm, hardness between 3.7 and 4.2 kg cm−2, and friability values between 0.38% and 0.52%, indicating satisfactory mechanical integrity (Table 2). Formulations F6 and F7, which contained co-processed bulking agents, demonstrated improved hardness, minimal weight variation, and reduced friability. This improvement is attributed to enhanced inter-particle bonding and compressibility resulting from co-processing, which promotes uniform stress distribution during compression. All formulations complied with the British Pharmacopoeia weight variation limit of ±7.5% (Table 2). Wetting time and water absorption studies revealed clear differences between superdisintegrants. Formulations containing sodium starch glycolate (SSG) exhibited higher water absorption ratios (up to 144.0%) and longer wetting times (27–30 s), consistent with its swelling-based disintegration mechanism. In contrast, crospovidone-containing formulations showed lower water uptake (82.7%–84.5%) and significantly shorter wetting times (11–18 s), reflecting its capillary-driven action. Formulation F7 exhibited a wetting time of 18 s (Fig. 6).| Code | Thickness (mm ± SD) | Weight variation (mg ± SD) | Hardness (kg cm−2 ± SD) | Friability (% ± SD) |
|---|---|---|---|---|
| Values are expressed as mean ± SD (n = 3). | ||||
| F1 | 3.1 ± 0.12 | 129 ± 0.51 | 3.7 ± 0.16 | 0.52 ± 0.41 |
| F2 | 3.1 ± 0.10 | 127 ± 0.49 | 3.8 ± 0.29 | 0.51 ± 0.37 |
| F3 | 3.0 ± 0.08 | 131 ± 0.46 | 4.0 ± 0.24 | 0.43 ± 0.21 |
| F4 | 3.1 ± 0.07 | 129 ± 0.34 | 3.9 ± 0.27 | 0.40 ± 0.42 |
| F5 | 3.0 ± 0.09 | 128 ± 0.31 | 3.8 ± 0.23 | 0.42 ± 0.39 |
| F6 | 3.0 ± 0.06 | 127 ± 0.36 | 4.2 ± 0.16 | 0.38 ± 0.36 |
| F7 | 3.1 ± 0.05 | 127 ± 0.28 | 4.1 ± 0.14 | 0.39 ± 0.32 |
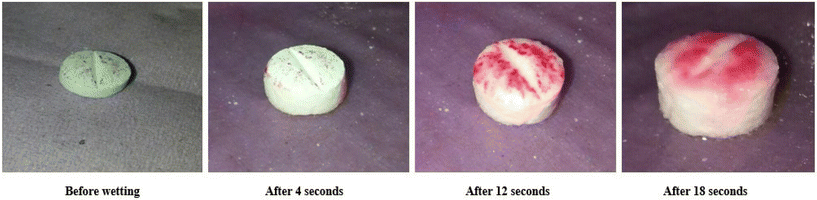
The modified in vitro disintegration test (Table 3) revealed disintegration times ranging from 10 to 28 seconds, within the acceptable range for ODTs, with F7 (Fig. 5) achieving the fastest disintegration time of 10 seconds, indicating a synergistic effect between co-processed fillers and crospovidone. Drug content ranged from 97.34% to 99.14%, confirming uniform drug distribution and formulation accuracy.
| Code | Wetting time (sec ± SD) | Water absorption ratio (% ± SD) | Modified disintegration time (sec ± SD) | Drug content (% ± SD) |
|---|---|---|---|---|
| Values are expressed as mean ± SD (n = 3).a Statistically significant compared to SSG-based formulations (p < 0.05, one-way ANOVA). | ||||
| F1 | 29 ± 0.23 | 134.0 ± 0.27 | 28 ± 0.16 | 99.12 ± 0.28 |
| F2 | 28 ± 0.14 | 132.0 ± 0.18 | 27 ± 0.21 | 98.06 ± 0.26 |
| F3 | 29 ± 0.24 | 113.0 ± 0.24 | 27 ± 0.24 | 98.57 ± 0.31 |
| F4 | 27 ± 0.27 | 144.0 ± 0.21 | 25 ± 0.19 | 97.34 ± 0.23 |
| F5 | 11 ± 0.21 | 82.7 ± 0.23 | 14 ± 0.22 | 98.55 ± 0.27 |
| F6 | 30 ± 0.19 | 123.82 ± 0.20 | 20 ± 0.16 | 99.14 ± 0.24 |
| F7 | 18 ± 0.21a | 84.51 ± 0.19a | 10 ± 0.12a | 98.87 ± 0.25 |
Collectively, these results confirm that the formulations, especially those incorporating co-processed excipients, possess the necessary properties for efficient ODT performance.
3.3. In vitro dissolution studies
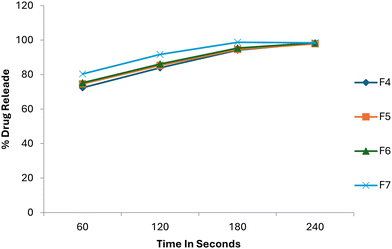
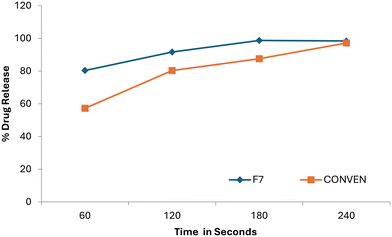
Dissolution studies were performed on formulations F4, F5, F6, and F7 selected based on their rapid behavior, and were compared with a market-sourced conventional tablet. All ODT formulations released more than 70% of drug within the first 60 seconds and exceeded 90% release by 180 seconds (Fig. 7). Formulation F7 demonstrated superior dissolution performance, releasing 80.38% of granisetron HCl within 60 seconds and achieving 98.74% cumulative release at 180 seconds. In contrast, the conventional tablet released only 57.24% of the drug in the first 60 seconds and 87.54% after 180 seconds, (Fig. 8). The initial drug release rate of the optimized ODT formulation was approximately 1.4-fold higher than that of the conventional tablet.The enhanced dissolution behavior of co-processed formulations can be attributed to rapid tablet disintegration and improved liquid penetration, facilitated by the intimate association of MCC and mannitol at the sub-particle level. This confirms that dissolution enhancement arises from excipient engineering rather than compositional changes.
3.4. Drug release kinetics
Zero-order and Higuchi models showed poor correlation for all formulations, indicating that drug release from developed orodispersible tablets was neither concentration-independent nor solely diffusion-controlled. Similarly, the Hixson–Crowell model did not adequately describe the release behavior, suggesting that changes in tablet surface area and geometry during dissolution were not the primary determinants of drug release. These models were therefore considered unsuitable for describing the dissolution behavior of the formulated ODTs.In contrast, formulations F5 and F7 demonstrated strong correlation with the first-order, Korsmeyer–Peppas, and Weibull models, as evidenced by higher r2 values (Table 4). The improved fit to the first-order model indicates concentration-dependent drug release, which is typical for rapidly disintegrating dosage forms. The Korsmeyer–Peppas model further suggested a combined mechanism of drug release involving diffusion and matrix relaxation/erosion, consistent with the fast tablet disintegration observed for these formulations.
| Parameters (r2) | Dissolution data modeling (r2 ≥ 0.5) | |
|---|---|---|
| F5 | F7 | |
| Models with r2 ≥ 0.5 are shown; models with poor correlation (r2 < 0.5) were considered unsuitable for mechanistic interpretation. | ||
| First order | 0.810 | 0.913 |
| Korsmeyer–Peppas | 0.991 | 0.940 |
| Weibull | 0.993 | 0.987 |
The Weibull model provided the best fit for formulations F5 and F7, indicating flexible and rapid release behavior characteristics. The superior kinetic fit observed for formulation F7 correlates well with its faster disintegration time and enhanced dissolution performance, highlighting the contribution of co-processed MCC–mannitol in facilitating rapid drug availability.
Overall, kinetic modeling confirms that drug release from the optimized ODT formulations is governed by rapid tablet disintegration followed by concentration-dependent release, rather than by diffusion-controlled or erosion-limited mechanisms. These findings further support the suitability of the co-processed excipient system for efficient immediate-release ODT formulations.
3.5. Drug excipient compatibility study (FTIR spectroscopy)
Fourier-transform infrared (FTIR) spectroscopy was employed to evaluate the potential chemical interactions between granisetron HCl and formulation excipients and to assess the impact of excipient co-processing. The spectra of pure granisetron HCl, physical mixtures of MCC and mannitol, co-processed MCC–mannitol, and the optimized formulation F7 were recorded individually and compared based on characteristic peak positions and band profiles (Fig. 9).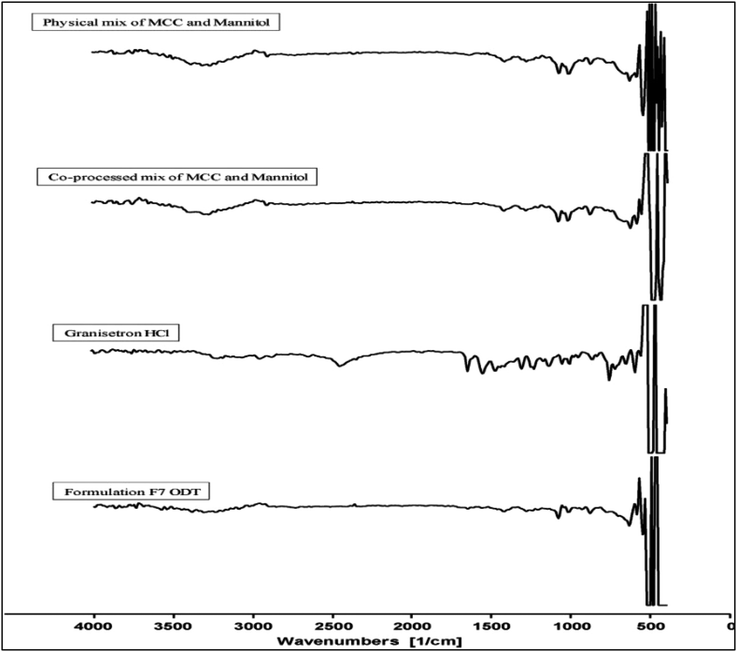 | ||
| Fig. 9 FTIR spectra of the physical mix of MCC and mannitol, co-processed mix of MCC and mannitol, granisetron HCl and ODT formulation F7. | ||
The FTIR spectrum for the physical mix of MCC and mannitol showed peaks around 3400–3300 cm−1 (O–H stretching), 2900 cm−1 (C–H stretching), and 1050–1150 cm−1 (C–O stretching), these peaks remained sharp and well-defined, indicating the absence of chemical interaction in the physically blended system.
In the co-processed MCC–mannitol blend, the O–H stretching region showed noticeable band broadening with a slight shift toward lower wavenumbers than those of the physical mixture. Such spectral changes are indicative of intermolecular hydrogen bonding between hydroxyl groups of MCC and mannitol, arising from closer molecular association during the co-processing step. These interactions are physical rather than chemical and are commonly associated with improved powder compactability and tablet strength.
Granisetron HCl spectrum displays characteristics peaks around 3100–3000 cm−1 (N–H stretching), 1600–1500 cm−1 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching), and 1400–1300 cm−1 (C–N stretching). These characteristic peaks were clearly retained in the FTIR spectrum of formulation F7, with no significant shifts or disappearance, confirming the chemical compatibility of the drug with co-processed excipients.
C stretching), and 1400–1300 cm−1 (C–N stretching). These characteristic peaks were clearly retained in the FTIR spectrum of formulation F7, with no significant shifts or disappearance, confirming the chemical compatibility of the drug with co-processed excipients.
Although spectral overlay analysis was not performed, comparative evaluation of individual spectra confirms that co-processing induces beneficial physical interactions without altering the chemical integrity. These findings support the improved mechanical and disintegration behavior observed for co-processed formulations.
3.6. Taste and mouthfeel evaluation
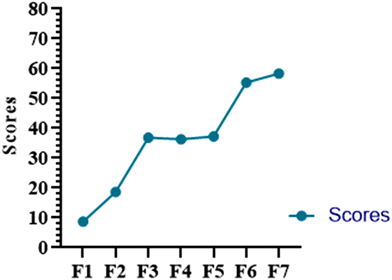
The taste evaluation of the formulations (Fig. 10) revealed notable differences in sensory attributes such as taste, chalkiness, residue, and grittiness across the seven formulations (F1–F7). Formulation F1 exhibited the lowest total score of 8.7, with unacceptable taste and no chalkiness, residue, or grittiness reported. This indicates that the formulation was not favorable in terms of taste and mouthfeel, which may impact patient acceptance. Formulation F2 showed a slight improvement with a total score of 18.6, again with an acceptable taste but still lacking any chalkiness. Formulations F3, F4, and F5, which contained varying concentrations of excipients, showed progressively higher total scores of 36.8, 36.3, and 37.2, respectively. These formulations had acceptable taste profiles, with F3 exhibiting some chalkiness, while F4 and F5 had minimal residues. The presence of superdisintegrants like crospovidone likely enhanced the disintegration characteristics influencing the overall taste and mouthfeel scores. Formulation F6, with a total score of 55.3, showed an improved taste profile, with a reduction in grittiness and minimal chalkiness. Formulation F7, achieved a total score of 58.3. This formulation demonstrated an excellent taste, with minimal to no residue, no grittiness, and an acceptable level of chalkiness, making it the most favorable formulation for patient acceptance.3.7. Stability studies
Statistical comparison of stability data revealed no significant differences (p > 0.05) in weight variation, hardness, friability, disintegration time, or drug content of formulations F5 and F7 over the six-month accelerated stability period when compared with initial values (Table 5). These findings comply with ICH Q1A stability requirements and confirm the physical and chemical stability of the optimized formulation. The absence of statistically significant changes further supports the robustness of the co-processed MCC–mannitol system under accelerated storage conditions.| Code | Time | Weight variation (mg ± SD) | Hardness (kg cm−2 ± SD) | Friability (% ± SD) | Disintegration time (sec ± SD) | Drug content (% ± SD) |
|---|---|---|---|---|---|---|
| Values are expressed as mean ± SD (n = 3).a No statistically significant difference compared to initial (0-month) values (p > 0.05, Student's t-test), in accordance with the ICH Q1A stability guidelines. | ||||||
| F5 | 0 month | 128 ± 0.31 | 3.8 ± 0.23 | 0.42 ± 0.39 | 14 ± 0.22 | 98.55 ± 0.27 |
| 1 month | 129 ± 0.37a | 3.5 ± 0.23a | 0.39 ± 0.21a | 15 ± 0.19a | 99.10 ± 0.10a | |
| 2 month | 128 ± 0.11a | 3.7 ± 0.15a | 0.41 ± 0.11a | 15 ± 0.21a | 98.98 ± 0.16a | |
| 3 month | 128 ± 0.15a | 4.0 ± 0.17a | 0.40 ± 0.17a | 15 ± 0.10a | 99.00 ± 0.16a | |
| 6 month | 129 ± 0.11a | 3.9 ± 0.11a | 0.41 ± 0.16a | 15 ± 0.31a | 98.89 ± 0.11a | |
| F7 | 0 month | 127 ± 0.28 | 4.1 ± 0.14 | 0.39 ± 0.32 | 10 ± 0.12 | 98.87 ± 0.25 |
| 1 month | 127 ± 0.31a | 4.0 ± 0.14a | 0.38 ± 0.23a | 10 ± 0.15a | 98.82 ± 0.19a | |
| 2 month | 126 ± 0.25a | 4.1 ± 0.15a | 0.38 ± 0.13a | 11 ± 0.11a | 98.88 ± 0.21a | |
| 3 month | 127 ± 0.23a | 4.2 ± 0.12a | 0.39 ± 0.15a | 10 ± 0.09a | 99.01 ± 0.21a | |
| 6 month | 127 ± 0.10a | 4.1 ± 0.13a | 0.40 ± 0.12a | 10 ± 0.10a | 99.00 ± 0.17a | |
4. Conclusion
Orodispersible tablets (ODTs) of granisetron HCl were successfully formulated, using a laboratory-prepared co-processed excipient system composed of microcrystalline cellulose and mannitol and manufactured via a direct compression approach. Comparative evaluation demonstrated that co-processing at the sub-particle level significantly improved powder flow and compressibility compared with the corresponding physical mixtures, facilitating robust tablet production by direct compression.Among the evaluated formulations, F7 containing 4% crospovidone and co-processed MCC–mannitol exhibited the most favorable performance, achieving rapid disintegration (10 ± 0.12 s), acceptable mechanical strength, and efficient drug release (98.14% ± 0.25% within 180 s). Stability testing under accelerated ICH conditions confirmed the physical and chemical stability of the optimized formulation over six months, with no statistically significant changes in critical quality attributes.
Overall, the study demonstrates that sub-particle level co-processing of conventional excipients, without dependence on proprietary multifunctional products, can enhance the performance of orodispersible tablets prepared by direct compression.
This approach offers a practical, cost-effective formulation strategy for developing fast-disintegrating oral dosage forms of low-dose, water-soluble drugs such as granisetron HCl, while maintaining industrial relevance and regulatory acceptability.
Author contributions
Shazia Naseem Chaudhry contributed to conceptualization, study design, experimental work, and ensuring methodological accuracy. Atiq Ur Rahman contributed to data analysis, interpretation of results, and preparation of the original manuscript draft.Conflicts of interest
The authors declare that they have no competing financial or personal interests that could have influenced the work reported in this manuscript.Ethical statement
Ethics approval and consent to participate: The sensory evaluation (taste and mouthfeel assessment) was conducted exclusively in healthy adult volunteers (≥18 years). No minors were included in the study. All participants were informed about the nature and purpose of the evaluation, and they provided verbal informed consent prior to participation. The assessment involved only placebo formulations (without any active drug) and posed no risk to participants. The study was conducted in accordance with institutional guidelines.Data availability
All data supporting the findings of this study are included within the article.Acknowledgements
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. The authors acknowledge the support provided by CCL Pharma (Pvt) Ltd and Tagma Pharma (Pvt) Ltd., Lahore, Pakistan, for supplying the materials used in this study.References
- K. Roshan, et al., A review on novel drug delivery system, Int. J. Res. Anal. Rev., 2021, 8(1), 183–199 Search PubMed.
- J. Shery, et al., Orodispersible films: current innovations and emerging trends, Pharmaceutics, 2023, 15(12), 2753 CrossRef PubMed.
- A. M. Irfan, D. D. Akila and G. Karthick, Review of Fast-dissolving Tablets-A New Era in Brand-new Drug Delivery Systems, J. Pharm. Res. Int., 2022, 34(20B), 41–49 Search PubMed.
- G. Honey, et al., Orally disintegrating systems: innovations in formulation and technology, Recent Pat. Drug Delivery Formulation, 2008, 2(3), 258–274 CrossRef PubMed.
- S. C. R. Shan, W. S. H. Paul and C. L. Wah, Application of freeze-drying in the development of oral drug delivery systems, Expert Opin. Drug Delivery, 2016, 13(11), 1595–1608 CrossRef PubMed.
- A. Pooja and S. V. Arora, Orodispersible tablets: A comprehensive review, 2013 Search PubMed.
- G. Darsh and T. Poonam, Formulation Aspects and Manufacturing Technology–A Review on Fast Disintegrating Tablets, YMER, 2023, 22(1), 1265–1288 Search PubMed.
- A. Yagmur, et al., Evaluation of preparation methods for orally disintegrating tablets, Medicine, 2020, 9(1), 265–269 Search PubMed.
- Granisetron hydrochloride, 5-HT3 antagonist. Available from:https://www.abcam.com/en-us/products/biochemicals/granisetron-hydrochloride-5-ht3-antagonist-ab120125.
- Granisetron hydrochloride. Available from:https://www.sigmaaldrich.com/PK/en/product/sigma/g3796.
- I. Širola, et al., Effect of crystal size and shape on bulk density of pharmaceutical powders, J. Cryst. Growth, 1997, 181(4), 403–409 CrossRef.
- S. Ravendra, et al., Real time monitoring of powder blend bulk density for coupled feed-forward/feed-back control of a continuous direct compaction tablet manufacturing process, Int. J. Pharm., 2015, 495(1), 612–625 CrossRef PubMed.
- A. Saker, et al., Powders flowability assessment in granular compaction: What about the consistency of Hausner ratio?, Powder Technol., 2019, 354, 52–63 CrossRef CAS.
- T. S. Patel and M. Sengupta, Fast dissolving tablet technology, World J. Pharm. Sci., 2013, 2(2), 485–508 CAS.
- A. Sharmin, et al., In vitro dissolution study of glimepiride from binary and ternary solid dispersion formulation, Univers. J. Pharm. Res., 2019, 7–11 Search PubMed.
- A. S. Kanwal, et al., Pharmaceutical quality evaluation of different glimepiride brands marketed in Karachi (Pakistan): In pursuance to global issue of availability and affordability of quality medicines, Pak. J. Pharm. Sci., 2019, 32(6), 2709–2715 Search PubMed.
- J. A. Seitz and G. M. Flessland, Evaluation of the physical properties of compressed tablets I: Tablet hardness and friability, J. Pharm. Sci., 1965, 54(9), 1353–1357 CrossRef CAS PubMed.
- W. N. French, et al., Pharmacopeial standards and specifications for bulk drugs and solid oral dosage forms. Similarities and differences, J. Pharm. Sci., 1967, 56(12), 1622–1641 CrossRef CAS PubMed.
- G. M. Poursharifi, et al., Formulation and quality control of orally disintegrating tablets (ODTs): recent advances and perspectives, BioMed Res. Int., 2021, 2021(1), 6618934 CrossRef PubMed.
- P. K. Lakshmi, K. D. Vinod and H. K, Formulation and evaluation of oro-dispersible tablets using modified polysaccharides, Saudi J. Med. Pharm. Sci., 2017, 3, 13–22 Search PubMed.
- M. Disha, et al., Chapter 9 - Dissolution Profile Consideration in Pharmaceutical Product Development, in Dosage Form Design Considerations, ed. R. K. Tekade, Academic Press, 2018, pp. 287–336 Search PubMed.
- B. M. Luciano, Strategies to modify the drug release from pharmaceutical systems, Woodhead Publishing, 2025 Search PubMed.
- P. M. Padmaa, et al., Release kinetics–concepts and applications, Int. J. Pharm. Res. Technol., 2018, 8(1), 12–20 Search PubMed.
- S. Da-Wen, Infrared spectroscopy for food quality analysis and control, Academic Press, 2009 Search PubMed.
- ICH, Stability testing of new drug substances and products. Q1A (R2), Current Step, 2003, 1–24.
| This journal is © The Royal Society of Chemistry 2026 |