 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Advanced bispecific antibody design and inhaled biologics; emerging strategies for asthma
Zeina Kassemab,
Jenny K. W. Lam b and
Hanieh Khalili
b and
Hanieh Khalili *ab
*ab
aSchool of Medicine and Biosciences, University of West London, W5 5RF, UK. E-mail: hanieh.khalili@uwl.ac.uk; hanieh.khalili@ucl.ac.uk
bUCL School of Pharmacy, University College London, WC1N 1AX, UK
First published on 22nd January 2026
Abstract
Asthma is a chronic inflammatory respiratory disease affecting over 300 million individuals worldwide. While current therapies, including inhaled corticosteroids and monoclonal antibodies, have significantly improved management of Type 2 (T2)-high asthma, treatment options for T2-low, steroid-resistant, and severe asthma remain limited. Recent advances in biologic therapeutics have introduced bispecific antibodies (bsAbs) as a promising next-generation strategy. BsAbs are engineered to simultaneously target two distinct inflammatory pathways, offering broader immunomodulatory effects and the potential to improve disease control in heterogeneous asthma phenotypes. Concurrently, pulmonary delivery systems such as nebulisers, dry powder inhalers (DPIs) and soft mist inhalers (SMIs) have emerged as attractive non-invasive alternatives to injections for biologic administration, providing localised treatment directly to the lungs, reduced systemic side effects, and enhanced patient adherence. This review examines the immunological basis of asthma endotypes, evaluates current monoclonal antibody therapies and their limitations, and explores the design, mechanisms, and clinical progress of bispecific antibodies in the treatment of asthma. Additionally, we analyse the feasibility and challenges of inhaled biologic delivery, including formulation strategies and device optimisation. We conclude by highlighting future research priorities, including the development of inhalable bsAbs for T2-low asthma and the need for scalable, stable, and patient-friendly formulations. This dual-focused approach, combining novel antibody engineering with targeted delivery, represents a critical step towards personalised, effective asthma care.
Introduction
Asthma is a chronic respiratory and inflammatory disorder of the airways characterised by airflow limitation and bronchial constriction. Over 300 million individuals are affected by asthma globally.1 The prevalence of asthma is expected to continue rising, reaching 400 million by 2025 because of population growth, aging, and elevated urbanisation.2 Asthma prevalence ranges from 1 to 18% depending on the country. For instance, higher prevalence rates are estimated in high-income countries such as the UK (15.6%), the USA (8.3%), and Portugal (7.1%).2 Although the prevalence is considered lower in low- and middle-income countries due to a lack of diagnosis and resources, asthma-related mortality is significantly higher. Low- and middle-income countries account for 90% of the asthma burden.3 The Global Burden of Disease Study reported that in 2019, 21.6 million lost healthy life years (DALYs) and 461![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 069 deaths were caused by asthma, particularly due to severe exacerbations and inadequate disease control.4 Globally, asthma ranks second after chronic obstructive pulmonary disease (COPD) as the most common chronic respiratory disease in adults and first as the most common chronic disease in children.2
069 deaths were caused by asthma, particularly due to severe exacerbations and inadequate disease control.4 Globally, asthma ranks second after chronic obstructive pulmonary disease (COPD) as the most common chronic respiratory disease in adults and first as the most common chronic disease in children.2
Symptoms of asthma, because of bronchoconstriction, include shortness of breath, wheezing, and coughing (Fig. 1). The pathogenesis of asthma is driven by airway inflammation, airway hyperresponsiveness (AHR), and airway remodeling.1 The inflammation in asthma is heterogeneous and can be divided into two endotypes: Type 2 (T2)-high and T2-low inflammation, each characterised by distinct immunopathological features.5 While corticosteroids and monoclonal antibodies (mAbs) have significantly improved outcomes in T2-high asthma, treatment options for T2-low asthma remain limited.6 mAbs target a single cytokine or pathway, which may not be adequate for patients with mixed or overlapping endotypes, and they fail to suppress the complex network of inflammatory mediators present in severe disease.7 In this context, bispecific antibodies (bsAbs) and novel delivery strategies such as inhalable biologics represent a new frontier in asthma management.8 Bispecific antibodies (bsAbs), which can bind to two distinct antigens or epitopes simultaneously, present an innovative strategy for achieving synergistic pathway modulation and more comprehensive immunological control, thereby overcoming the redundant cytokine pathways and crosstalk mechanisms that reduce the efficacy of monotherapy.9 This strategy holds significant promise as a novel intervention in asthma, particularly resistant and severe asthma unresponsive to the conventional treatments.10 Similarly, there is a need for inhalable biologics to enhance local delivery of the drugs to the airways in a non-invasive manner, reduce systemic toxicity, and improve patient compliance.11 Given the rising interest in bsAbs and the rapid expansion of clinical research in this field, this review aims to provide an in-depth evaluation of current asthma biologics, highlight the mechanistic rationale for bispecific targeting, and discuss the translational challenges and future opportunities for bsAbs development in respiratory medicine by summarising the ongoing clinical trials and highlighting preclinical proof of concept on bispecific antibodies in the management of asthma.
 | ||
| Fig. 1 Structural differences between normal and asthmatic airways. (a) Normal airway with relaxed smooth muscles and an open lumen. (b) The changes during an asthma attack include bronchoconstriction (narrowing of the airway), inflammation, and excessive mucus production. These changes significantly reduce airflow, causing symptoms such as coughing, wheezing, and shortness of breath.1 (Scheme drawn by biorender.) | ||
Asthma pathogenesis and endotypes
T2-high inflammation is more common, accounting for 80–85% of all asthma cases.12 It is induced by allergic and non-allergic factors such as irritants and infectious agents. T2-high eosinophilic asthma is strongly associated with atopy and allergic asthma.13 The T2-high endotype is characterised by the production of cytokines such as IL-4, IL-5, and IL-13 in response to the expression of GATA-binding protein 3 (GATA3) in type 2 helper T (Th2) cells and type 2 innate lymphoid cells (ILC2).14 IL-5 promotes eosinophil maturation, activation, and recruitment, whereas IL-4 and IL-13 contribute to goblet cell metaplasia, increased airway smooth muscle contractility, and airway hyperresponsiveness (AHR).15 Inflammatory cells, including eosinophils, mast cells, basophils, and IgE-producing plasma cells, are also involved in the pathogenesis of T2-high inflammation.16 On the contrary, T2-low inflammation is thought to be mediated by endotoxins, bacterial and viral infections, smoking, and other occupational agents.16 Unlike T2-high inflammation, T2-low inflammation is not associated with high eosinophil count and cannot be diagnosed by typical T2 biomarkers such as elevated IgE and FeNO.12 The pathogenesis of T2-low inflammation is less well understood. It is believed to be associated with Th1 and Th17 cytokines, which primarily trigger neutrophilic inflammation and oxidative damage.16 Th17 cells, which release IL-17 family cytokines, play a central role in neutrophilic inflammation and airway remodeling. They are implicated in poor responsiveness to corticosteroids.14 Th1 cells, through the production of interferon-γ (IFN-γ) and tumor necrosis factor-α (TNF-α), also promote neutrophil-driven inflammation.12 Th1 cells activate the JAK-STAT1 signaling pathway through the secretion of IFN-γ, which binds to IFN-γ receptor (IFNGR) present on several airway immune and structural cells, enhancing inflammation-associated genes transcription, aggravating antigen presentation via MHC class II, and facilitating further immune cells recruitment to the inflamed area through increasing the expression of adhesion molecules like ICAM-1 and VCAM-1.12 Similarly, TNF-α, upon binding to its receptors (TNFR1 and TNFR2), promotes further production of chemokines such as IL-8 and pro-inflammatory cytokines such as IL-6 through activation of downstream pathways (NF-kB and MAPK pathways).14 These chemokines attract and recruit neutrophils to the airways, resulting in excessive mucous secretion, epithelial damage, and airway remodeling through the release of harmful mediators such as reactive oxygen species (ROS), proteases like neutrophil elastase, and neutrophil extracellular traps (NETs).12Similarly, Th17 cells, which are activated by environmental factors such as pollutants and airway infections, mainly viral, play a key role in the pathogenesis of T2-low asthma and neutrophilic inflammation through the production of IL-17 significantly.14 IL-17 triggers airway inflammation, hyperresponsivity, and tissue damage by facilitating the recruitment and activation of airway neutrophils and enhancing the release of pro-inflammatory mediators such as IL-8 and granulocyte-macrophage colony-stimulating factors (GM-CSF).12 In addition to Th1, Th17, and neutrophils, macrophages also play a role in T2-low inflammation. Macrophages differentiate into the pro-inflammatory phenotype under the influence of certain mediators like IFN-γ and bacterial components such as lipopolysaccharide (LPS), producing high quantities of ROS, TNF-alpha, IL-12, and IL-6, which results in enhanced local inflammation and tissue damage.12 Moreover, the activation of toll-like receptors (TLRs) on airway epithelial and immune cells further elevates airway hyperresponsiveness and neutrophilic infiltration, through increased production of pro-inflammatory cytokines and chemokines.12 Patients with T2-low inflammation are often less responsive to corticosteroid therapy as they tend to have a more severe form at a late onset, with almost irreversible airway obstruction.17 The poor response to corticosteroid therapy, lack of clearly defined biomarkers, and neutrophil predominance in T2-low inflammation associated with Th17 and Th1 cells pose a significant challenge in the management of severe asthma.6 These highlight the need for a broader treatment to target non-inflammatory pathways and to overcome the limitations of existing treatments, including biologics.18 T2-low endotypes may also include mixed and pauci-granulocytic inflammation, but to a lesser extent.1 Recently, studies have shown that thymic stromal lymphopoietin (TSLP), a key epithelial-derived cytokine, might be involved in both endotypes.19 TSLP drives T2-high asthma by activating dendritic cells and ILC2s, leading to eosinophilic inflammation via the production of IL-4, IL-5, and IL-13.19 In contrast, it promotes neutrophilic inflammation and steroid resistance in T2-low asthma by inducing the production of IL-8 and IL-17, as well as epithelial remodeling.19 Table 1 shows the different cytokines involved in T2-high and T2-low asthma.
Current treatment of asthma
Non-biologic approach
Asthma is primarily treated with glucocorticoids. Glucocorticoids are anti-inflammatory drugs that act by inhibiting the infiltration and activation of the immune cells, such as eosinophils and Th2 cells.1 Inhaled corticosteroids (ICSs) are preferred and are generally well tolerated. Patients with mild to moderate asthma who remain uncontrolled on ICSs may benefit from increasing the corticosteroid dose and/or the addition of one or more controller medications, such as long-acting beta-adrenergic receptor agonists (LABAs) and/or leukotriene receptor antagonists (LTRAs).21 LTRAs are administered orally and act by blocking leukotriene receptors, thereby preventing leukotriene-induced inflammation in the airways. On the other hand, LABAs are administered via inhalation and act by inhibiting bronchoconstriction by binding to the beta-2 receptors for a prolonged duration of 12 to 24 hours. LABA and LTRAs are often used as adjunctive therapies to ICSs rather than as alternatives.1 Studies have shown that the use of LTRAs alone was associated with increased risk of exacerbation and poor asthma control. Similarly, LABAs carry a black box warning, issued by the FDA, for increased mortality when used as monotherapy.22 Hence, patients should use the daily controller regimen based on ICSs plus a reliever on an as-needed basis. The relievers used in asthma are short-acting beta-adrenoceptor agonists (SABAs), which are also administered by inhalation. They offer a fast onset of action within 15 to 30 minutes but with a shorter duration, typically up to 4 hours.1 Because SABAs lack anti-inflammatory properties, they should not be relied upon for long-term control. Excessive SABA use is a marker of poor asthma control, associated with increased risk of exacerbation and mortality. In such cases, patient adherence and symptom management should be reassessed.23 However, in more severe cases, even high doses of oral glucocorticoids may still be ineffective, a condition known as steroid-resistant asthma. Steroid resistance is generally defined as an improvement of less than 15% in baseline forced expiratory volume in one second (FEV1) following 14 days of daily oral prednisolone treatment at a dose of 40 mg. However, a response greater than 15% in FEV1 is observed after treatment with beta-2 receptor agonists.24 For these patients, novel therapies and recent advances in asthma management offer promising alternatives.1Biological approach
Biologic therapies target specific inflammatory mediators involved in asthma pathogenesis, particularly in patients with T2-high asthma. These treatments aim to reduce exacerbations, improve lung function, decrease dependence on oral corticosteroids, and enhance the quality of life in patients who remain uncontrolled despite receiving the maximum tolerated doses of standard treatment.25 As of December 2025, six monoclonal antibodies are approved by the FDA for the treatment of asthma.26Omalizumab was the first biologic approved by the FDA and EMA for treating allergic asthma. Omalizumab, a humanized IgG1 κ monoclonal anti-IgE antibody, works by binding to IgE at the Cε3 domain and preventing its interaction with the high-affinity FcεRI receptor on mast cells and basophils, thereby reducing the release of proinflammatory mediators, limiting the activation of basophils/mast cells, and attenuating the downstream allergic response. However, omalizumab reduces asthma exacerbations by only 25%, exerts a limited effect on lung function, and carries a black box warning for the risk of anaphylaxis.26 The next two approved mAbs for treating asthma are mepolizumab and reslizumab, which are humanised IgG1 and humanised IgG4/κ, respectively. Both target the IL-5 ligand, preventing it from binding to its receptor on eosinophils, thereby reducing eosinophilic airway inflammation. Reslizumab has demonstrated superior efficacy to mepolizumab, improving lung function (FEV1) and reducing exacerbations by 60%, compared to 50% with mepolizumab.26 Benralizumab, an afucosylated IgG1 antibody, acts as an anti-IL-5 by binding to the alpha-subunit of the IL-5 receptor, thereby blocking IL-5 binding and subsequently the recruitment and activation of eosinophils, and uniquely inducing antibody-dependent cell-mediated cytotoxicity. Benralizumab is as efficacious as reslizumab in reducing exacerbations by 25–60%, but unlike reslizumab, benralizumab has no significant positive effect on FEV1 and lung function.25 Dupilumab, a fully human IgG4 antibody, binds to the alpha-subunit of the IL-4 receptor, inhibiting both the IL-4 and IL-13 signaling pathways. It significantly reduces exacerbation rates by 50–70%, improves lung function, and is particularly effective in patients with coexisting atopic dermatitis.27
Tezepelumab, a fully human IgG2λ antibody, is the most recently approved mAb that functions by blocking TSLP. TSLP activates dendritic cells and ILC2s, which in turn promote the production of IL-4, IL-5, and IL-13. This cascade induces the recruitment of immune cells, IgE production, bronchoconstriction, mucus secretion, and airway remodeling.28 Tezepelumab reduces serum IL-5 and IL-13 levels, as well as blood and airway eosinophil levels.25 It significantly improves lung function and FEV1, reduces asthma exacerbations, alleviates symptoms, and enhances the quality of life in patients. TSLP facilitates interactions between airway structural cells and immune cells, which are not related solely to T2 inflammation. Thus, targeting TSLP may treat a broader type of asthma, including eosinophilic and non-eosinophilic asthma.29 In support of this, tezepelumab decreases annual asthma exacerbation rates compared to placebo by 63–71% in patients with severe eosinophilic asthma, 58–68% in those with severe allergic asthma, 67–71% in patients with both allergic and eosinophilic severe asthma, 34–49% in T2-low asthma, and 31–41% in individuals dependent on oral corticosteroids.30
The structural and mechanistic differences between each mAb result in varying efficacy across patient populations. For example, anti-IL-5 therapies are most effective in eosinophilic asthma, while dupilumab benefits patients with elevated FeNO or comorbid atopic disease.31 Dupilumab is also indicated for moderate to severe eosinophilic or oral steroid-dependent asthma in children from 6 to 11 years old and is administered subcutaneously (SC).32 Mepolizumab, reslizumab, and benralizumab are all anti-IL-5 therapies, and they are indicated in the treatment of severe eosinophilic asthma. However, they differ in the route of administration and the minimum approved age. For instance, mepolizumab is given subcutaneously (SC) from the age of 6 years. Benralizumab is also administered SC, but from the age of 12 years. On the other hand, reslizumab is not approved for the treatment of children with asthma. It is given via intravenous injection (IV) starting from the age of 18 years.33 Similarly, omalizumab is indicated for patients aged 6 years or older with allergic asthma marked by a high IgE count. Furthermore, its interaction with the FC domain of IgE may be the reason for the black box warning of anaphylaxis mentioned before.34 Moreover, tezepelumab offers the broadest therapeutic range due to its upstream target. It is reserved for children and adults aged 12 years and older with severe asthma, with no biomarker restriction, and given SC.30 However, the single-pathway design of these mAbs limits their effectiveness in patients with overlapping inflammatory pathways, illustrating the need for more versatile bispecific strategies.25
The pathogenesis of asthma and the mechanism of the mAbs discussed are summarised in Fig. 2.35 Several monoclonal antibodies are currently being tested in clinical trials for the treatment of asthma. Clazakizumab, Tocilizumab, and FB704A target IL-6. CSJ117 is being developed as an inhaled anti-TSLP therapy. FB825, CM310, and FB704A target the CemX domain on mIgE-expressing B cells, alpha-subunit of IL-4, and IL-5, respectively. Furthermore, MEDI3506 is an anti-IL-33 mAb. These agents are in various stages of clinical development, offering the potential of more targeted and personalised approaches to asthma management. The six mAbs currently approved in the market and those under investigation in clinical trials are summarised in Tables 2 and 3, respectively.
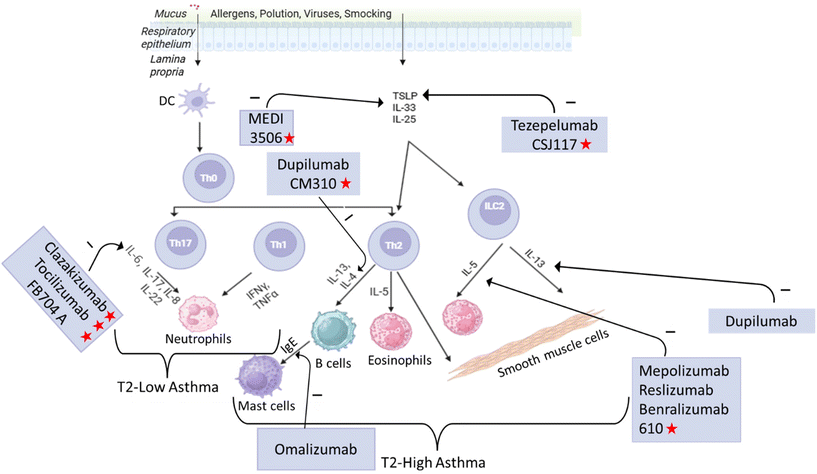 | ||
| Fig. 2 Inflammatory pathways in asthma and targeted biologics. Asthma involves both T2 and non-T2 immune responses. T2-high asthma includes both IgE-mediated and eosinophilic subtypes. IgE-driven asthma is treated with an anti-IgE mAb, omalizumab. Eosinophilic asthma involves key T2 cytokines, such as IL-5 and IL-4/IL-13 which are targeted by biologics including mepolizumab, reslizumab, and benralizumab (which block IL-5 or its receptor), as well as dupilumab and the investigational agent CM310★ (stapokibart), which inhibits IL-4Rα. Upstream cytokines such as thymic stromal lymphopoietin (TSLP) are targeted by tezepelumab and the investigational inhaled biologic CSJ117★ (ecleralimab), offering broader suppression of T2 inflammation. Other agents under development in this category include MEDI3506★ (tozorakimab), which inhibits IL-33. In contrast, T2-low asthma encompasses neutrophilic and paucigranulocytic phenotypes and is typically resistant to corticosteroids. While no approved biologics currently exist for this endotype, several investigational therapies are being explored, particularly those targeting the IL-6 pathway, such as clazakizumab★, tocilizumab★, and FB704A★. Drugs in development are marked with red stars.35 (Scheme drawn by biorender.) | ||
| Antibody | Target | Indications | Dose & frequency | Route | Effect on exacerbation | Effect on lung function (FEV1) | Notable features/limitations |
|---|---|---|---|---|---|---|---|
| Abbreviations: ADCC, antibody-dependent cellular cytotoxicity; IV, intravenous; SC, subcutaneous; TSLP, thymic stromal lymphopoietin. | |||||||
| Omalizumab | IgE | Moderate to severe allergic asthma (≥6 years) | 75–375 mg every 2–4 weeks (based on initial IgE total & weight) | SC | ↓ ∼25% | Minimal improvement | Risk of anaphylaxis (black box warning) |
| Mepolizumab | IL-5 | Severe eosinophilic asthma (≥6 years) | 100 mg every 4 weeks | SC | ↓ ∼50% | Moderate improvement | Well-tolerated; home or clinic admin |
| Reslizumab | IL-5 | Severe eosinophilic asthma (≥18 years) | 3 mg kg−1 every 4 weeks | IV | ↓ ∼60% | Greater improvement than mepolizumab | Requires IV infusion in the clinic |
| Benralizumab | IL-5Rα | Severe eosinophilic asthma (≥12 years) | 30 mg every 4 weeks (first 3 doses), then every 8 weeks | SC | ↓ 25–60% | Minimal effect | Eosinophil depletion via ADCC |
| Dupilumab | IL-4Rα (IL-4/13 blocker) | Moderate-to-severe eosinophilic or oral steroid-dependent asthma (≥6 years) | 400–600 mg first dose, then 200–300 mg every 2 weeks in adults and children from 6–11 years, 100 mg to 300 mg every 2 weeks | SC | ↓ 50–70% | Significant improvement | Also approved for eczema and nasal polyps |
| Tezepelumab | TSLP | Severe asthma with no biomarker restriction (≥12 years) | 210 mg every 4 weeks | SC | ↓ 31–71% (varies by phenotype) | Significant improvement | Broadest efficacy; works in T2-high & T2-low |
| Antibody | Target | Study stage | Route; dose |
|---|---|---|---|
| Abbreviations: IV, intravenous; SC, subcutaneous; TSLP, thymic stromal lymphopoietin. | |||
| Clazakizumab | IL-6 | Phase 2 | SC; 12.5 mg once every 4 weeks for 16 weeks |
| CSJ117 (eclarimab) | TSLP | Phase 2 | Inhaled via a dry powder inhaler; 4 mg inhaled once daily (morning)36 |
| FB825 | CemX domain on mIgE-expressing B cells | Phase 2 | IV; one dose of 8 mg kg−1 followed by 5 doses of 4 mg kg−1 every 4 weeks, subsequently infused over an hour duration |
| CM310 (stapokibart) | IL-4Rα | Phase 2 | SC; loading dose of 300 mg initially and then 150 mg every 2 weeks for a total of 12 doses. |
| 610 | IL-5 | Phase 2 | SC; 100 or 300 mg every 4 weeks |
| FB704A | IL-6 | Phase 2 | IV: 4 mg kg−1 infused every 2 weeks over an hour duration for a total of 4 doses |
| MEDI3506 (tozorakimab) | IL-33 | Phase 2 | SC: 300 or 600 mg every 4 weeks for a total of 4 doses |
Emerging bispecific antibody therapies
Bispecific antibody development
Asthma is a complex, heterogeneous disease involving multiple immunological and inflammatory pathways.37 Consequently, combining therapeutic strategies to target different mechanisms simultaneously has emerged as a promising approach. In this context, bsAbs have gained increasing attention for their potential to enhance treatment efficacy and improve disease control. BsAbs are engineered molecules designed to bind to two distinct antigens or receptors within a single structure, allowing simultaneous blockade of multiple signaling pathways involved in asthma pathophysiology.38 This dual targeting strategy can enhance therapeutic precision, improve drug delivery to relevant immune cells, and provide broader disease coverage compared to monospecific therapies. In contrast to combination therapies with two separate mAbs, bsAbs offer advantages such as simpler dosing regimens, improved patient compliance, and lower development costs.25Bispecific antibody formats
BsAbs are hybrid constructs classified into two structural categories, IgG-like and non-IgG-like (single-chain variable fragment (scFv)-based) (Fig. 3).39 IgG-like bsAbs retain the Fc region, providing advantages such as prolonged half-life, improved stability, and the ability to mediate immune functions, including complement-dependent and antibody-dependent cytotoxicity.39 IgG-like bsAbs are composed of 2 major regions, Fab and Fc. The Fab region is composed of both heavy and light chain components and is essential for antigen binding while providing high affinity and specificity. On the other hand, the Fc region is composed of hinge pairs, CH2, and CH3 constant domains, and plays key roles in inducing effector functions, cell killing mechanisms, as well as extending the plasma half-life of the antibody. The hinge region provides the antibody flexibility, allowing it to adopt various conformations.40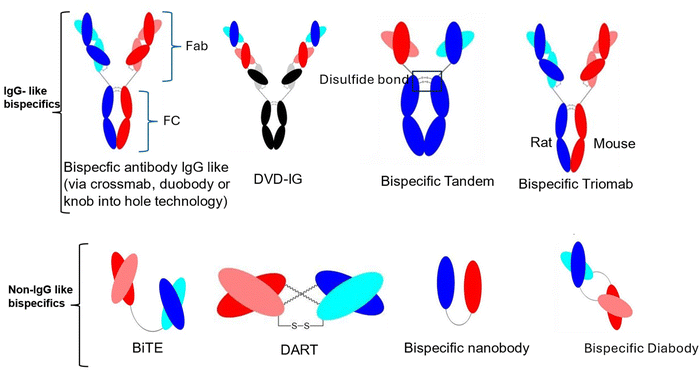 | ||
| Fig. 3 Representative formats of bispecific antibodies (bsAbs) and antibody fragments. This figure illustrates a range of bispecific antibody formats designed to bind two different antigens or epitopes. IgG-like formats (top row) include knobs-into-holes (KiH) for Fc heterodimerisation, dual variable domain (DVD-Ig) for tetravalent binding, two-in-one IgG for overlapping epitope recognition, triomab (rat/mouse quadroma-derived) for natural chain pairing, and CrossMAb for engineered light chain pairing via domain swapping. Fragment-based formats (bottom row) include diabodies (dimerised scFvs with bivalent binding), tandem scFvs (BiTEs) for dual targeting, DARTs with disulfide-stabilised diabody structure, and bispecific nanobody These formats offer distinct advantages in size, valency, and function, and are being developed for therapeutic applications such as cancer immunotherapy and targeted delivery.42 | ||
Techniques such as Knobs-into-Holes, CrossMab, and DuoBody facilitate the heterodimerisation of heavy chains, each with unique mechanisms to ensure structural integrity and functional specificity.41 For instance, Knobs-into-Holes induces mutations in the CH3 domain of each heavy chain in the Fc region, creating a knob in one CH3 domain to fit into the complementary holes in the opposite CH3 domain, ensuring the correct matching of the heavy chains and decreasing the risk of mispairing (Fig. 4A).25 Similarly, CrossMab technology ensures accurate chain pairing by interchanging domains at the Fab level, while DuoBody relies on controlled redox reactions for Fab-arm exchange.39 The DuoBody platform uses a precise method to independently and individually engineer two IgG molecules with a specific mutation in the CH3 domain, enabling correct chain pairing. The two IgG molecules, after production and purification, undergo a controlled exchange of Fab-arm to form the bispecific antibody.42 One drawback of DuoBody technology compared to Knobs-into-holes is that it relies on flexible chain exchange, which may result in off-target effects and less controlled pairings. CrossMab is also precisely engineered to ensure that the heavy and light chains are correctly paired and aligned, further augmenting antibody stability and functionality. This precession mitigates the risk of unintended interactions and enhances the therapeutic efficacy of these antibodies as well as their dual targeting function (Fig. 4B).42
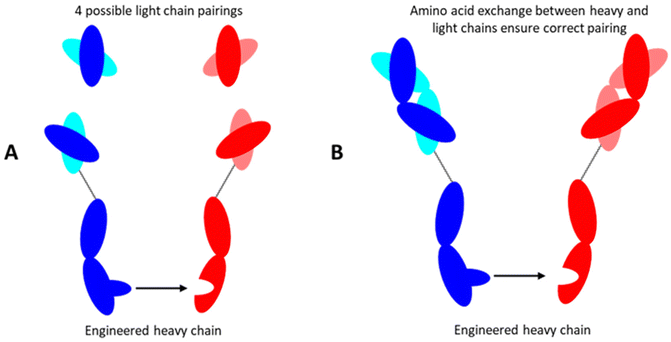 | ||
| Fig. 4 Illustration represents the bispecific antibodies production by (A) Knob into holes technology, and (B) CrossMAb technology. Knob-into-hole technology addresses heavy chain mispairing by engineering bulky amino acid residues into the CH3 domain, reducing the possible heavy chain combinations to four (panel A). CrossMAb technology further resolves light chain mispairing by exchanging amino acid blocks between the heavy and light chains, ensuring exclusive formation of the desired antibody (panel B). Together, these engineering strategies enhance bispecific antibody assembly by facilitating precise pairing of heavy and light chains.42 | ||
Other formats such as Triomab (produced via quadroma technology) and DVD-Ig (dual-variable domains) further expand the IgG-like bsAb repertoire, with each structure tailored to overcome specific design challenges, such as light-chain mismatches or antigen-binding limitations. For example, DVD-Ig is engineered to incorporate two different antigen-binding domains within a single IgG molecule. This is made possible by modifying the light and heavy chains, resulting in their different arrangement compared with the endogenous IgG, and enabling dual antigen targeting.43 Typically, the heavy chain of a conventional IgG antibody consists of one variable region (VH) and three constant regions (CH1, CH2, CH3). In contrast, the light chain includes a variable region (VL) and a constant region (CL). The VH and VL regions together form a single antigen-binding site. In contrast, a DVD-Ig is engineered to include two distinct VH regions within the heavy chain, each derived from different antibodies, while retaining the standard constant chains. These VH regions are paired with corresponding light chain regions, allowing the DVD-Ig to recognize and bind two separate antigens simultaneously. This dual-targeting capability provides potential therapeutic advantages by enabling a single antibody molecule to engage multiple disease pathways or antigens.39 On the other hand, Fc-free bsAbs lack the Fc region. They are generally smaller, allowing better tissue penetration and reduced immunogenicity. However, they suffer from shorter half-lives and structural instability.
The non-IgG-like bispecific antibodies have shorter half-lives due to the absence of the Fc region, which normally extends the half-life through interaction with neonatal Fc receptor for IgG (FcRn) and reduced immune-mediated clearance.42 Efforts have been made to increase the half-life of the antibody fragments (non-IgG-like) and to improve their stability through conjugation to proteins and/or polymers such as albumin and polyethylene glycol (PEG), respectively.44 PEGylation increases the size of the drug to slow kidney filtration, thereby reducing the drug clearance rate from the body. PEGylation also avoids opsonisation and recognition of the proteins by the immune system, prevents their rapid clearance by macrophages, and protects against proteolytic degradation. Similarly, fusing them to other proteins, such as albumin, improves the pharmacokinetic profile of the drugs. It reduces kidney filtration by increasing their size and protects them from degradation by in vivo proteases.45 Dual-affinity re-targeting molecules (DARTs) are stabilised through disulfide bonds to enhance their structural integrity. DART molecules are composed of two Fv fragments, each incorporating a VH domain from one antibody and a VL domain from another, enabling the formation of two unique antigen-binding sites.42 Unlike Bispecific T Cell Engagers (BiTEs), they are designed to mimic the natural interactions within the IgG molecules. BiTEs are composed of two single-chain variable fragments (scFvs) that are linked by a flexible linker and are able, for example, to target CD3 on T cells and tumor-specific antigens simultaneously, as in the field of cancer.39 BiTEs are characterized by their small size and flexibility, which facilitates the formation of an effective immune synapse and potent T cell-mediated cytotoxicity, which results in improved therapeutic index by allowing rapid tumor penetration and clearance from non-target tissues.42 TandAbs (Tandem Diabodies) are tetravalent bsAbs that can simultaneously bind to two sites on each antigen and are capable of recruiting both T and NK cells, thereby activating both innate and adaptive immune response.39 TandAbs are molecules with two antigen-binding sites for each targeted antigen, resulting from the polymerization of two diabodies joined by polypeptide chain molecules. In the context of cancer, for example, it creates a strong association between tumor cells and immune cells by connecting two scFvs to form a bispecific.42 Bispecific Nanobodies (BsNbs), composed solely of heavy-chain domains, preserve the VH region so that they can bind to multiple specific targets through the connection of the VH region of two or more antibodies, leading to enhanced tissue penetration with decreased molecular weight.39 The molecular weights of the different formats are summarised in Table 4.
| Bispecific antibody format | Molecular weight (KDa) | Mechanism of action | Advantages | Disadvantages | |
|---|---|---|---|---|---|
| IgG-like | Via CrossMAb technology | 150 | Ensures accurate chain pairing by interchanging domains at the Fab level | Better stability and versatility; precise pairing | Expensive |
| Via Knob into hole technology | 150 | Heavy chain heterodimerization achieved through complementary mutations in CH3 domains | High production efficiency; reduces heavy chain mispairing; cost-effective | By products formation and purification challenges | |
| Via duobody technology | 150 | Relies on controlled redox reactions for Fab-arm exchange | Large-scale production; minimal FC mutation sites; lower immunogenicity | Rely on flexible chain exchange which may result in off-target effects; less controlled pairings | |
| Triomab | 150 | Binds two antigens and Fcγ receptors simultaneously | Strong immune activation and multiple immune mechanisms | Retain immune effector functions; higher immunogenicity | |
| DVD-IgG | 200 | Incorporate two different antigen-binding domains within a single IgG molecule | Dual specificity; long half-life and enhanced stability | Complex design and manufacturing | |
| Non-IgG like | BiTE | 50–60 | Composed of two single-chain variable fragments (scFvs) that are linked by a flexible linker | High potency; small size with high penetration efficiency; able to link T cells to immune cells | Rapid clearance and short half-life |
| DART | 50–55 | Composed of two Fv fragments, each incorporating a VH domain from one antibody and a VL domain from another, enabling the formation of two unique antigen-binding sites | Mimic the natural interaction within the IgG molecule; enhanced stability and potency | Short half-life | |
| TandAB | 105–110 | Molecules with two antigen-binding sites for each targeted antigen | High specificity and efficacy; dual targeting; high flexibility | Short half-life; manufacturing complexity | |
| Diabody | 60 | Two scFv fragments with short linkers pair intermolecularly to form a compact bispecific dimer | Small size with rapid tissue penetration | Rapid clearance and short half-life | |
| Bispecific nanobody | 12–15 | Composed solely of heavy-chain domains, preserve the VH region so that they can bind to multiple specific targets through the connection of the VH region of two or more antibodies | Smaller size with higher penetration; ability to be engineered for high antigen-binding affinity; high stability and solubility; ease of production | Rapid clearance and short half-life | |
Clinical relevance of bispecifc antibody
Although concurrent administration of two biologics may provide additional benefits by targeting different inflammatory pathways involved in asthma pathogenesis, current guidelines do not recommend this approach for the management of severe asthma or type 2 comorbidities.46 Evidence from several real-world case studies and case reports show promising outcomes.47 For example, the combination of dupilumab with anti-IL-5 agents produced reductions of greater than 90% in sputum IL-5, IL-4, and IL-13, accompanied by clear clinical improvement.47 Similarly, another report demonstrated that co-administration of omalizumab and mepolizumab in a patient with severe uncontrolled asthma reduced blood eosinophils from 2330 to 200 cells per µL, increased FEV1 from 1.08 L to 1.32 L, and improved the 6-minute walk distance from 160 m to 280 m.47 However, the only randomized clinical trial to date—which allows a true head-to-head comparison—showed that adding itepekimab (anti-IL-33) to dupilumab (anti-IL-4/13) in 74 patients provided no additional benefit compared with placebo.47 The combination did not improve lung function (pre-BD FEV1) and did not reduce the rate of loss-of-control events (27% vs. placebo).47 Larger studies and more randomized trials are needed to understand the role of dual biologic therapy.48Special consideration is also required when selecting biologic combinations, as currently available biomarkers primarily characterise T2-high asthma. However, T2-low asthma remains largely unaddressed and cannot be accurately diagnosed due to the absence of reliable biomarkers.6 The latter limitations are also applicable to the design of bsAbs when considering epitopes and pathways to target.41 However, bsAbs offer some advantages over the combination of dual biologics. BsAbs can reduce treatment costs for both patients and healthcare systems.8 Additionally, bsAbs can improve patient adherence to treatment as patients will only receive one medication.8 Furthermore, bsAbs such as bispecific T cell engagers can redirect T cells to eliminate eosinophils, ILC2, and Th2 cells, a feature that is not possible with dual biologic combination therapy.49 For example, Kim et al. developed IL-5Rα × CD3 bispecific T cell engager to target simultaneously IL-5Rα, which is overexpressed on the surface of eosinophils, and CD3, which is present on the surface of T cells.50 The study has shown that the bispecific T cell engager can efficiently and selectively deplete eosinophils by redirecting T cells, with the strongest activity achieved by the scFv-Fc format targeting a membrane-proximal, high-affinity IL-5Rα epitope.50
BsAbs offer the advantage of creating a new pharmacology that is differentiated from monotherapy with biologics or combination therapy.49 They not only target two epitopes, or two different pathways simultaneously, but also can redirect T cells to adhere to inflammatory cells.49 BsAbs can also bring different cells or molecules, such as proteins, together, enhancing therapeutic efficacy.49 For instance, Chu et al. designed XmAb7195, a bsAb that suppressed the formation of IgE-secreting plasma cells and decreased the levels of both free and total IgE through IgE sequestration coupled with an FcgRIIb-mediated inhibitory mechanism.51 Compared with omalizumab, XmAb7195 binds human IgE with about five times greater affinity and engages FcγRIIb more than 400 times more strongly.51 Beyond simply trapping free IgE, XmAb7195 also suppresses the development of IgE-producing plasma cells by simultaneously binding the IgE B-cell receptor and FcγRIIb.51 In PBMC-humanised mice, treatment with XmAb7195 led to a dramatic reduction in total human IgE levels—up to a 40-fold decrease compared with omalizumab—while leaving IgG and IgM levels unchanged.51
A biologics combination might outweigh bsAbs in cases where health authorities have already approved individual components of the combination, as they have demonstrated proof of concept, since this lowers the threshold for development compared with bsAbs.49 However, more clinical trials are needed to evaluate the safety and efficacy of the combination of biologics compared to monotherapy with mAbs and versus bsAbs.47 BsAbs can open future opportunities for triplet design when considered for a combination therapy with a mAb or for quadrant design when combined with another bsAb, resulting in complete T cell activation and sustained response.49 Since the combination of biologic agents is based on combining independent agents to target complementary pathways, which can be directed at the same cell (different epitopes) or to different cells for additive or synergistic efficacy through distinct mechanisms without requiring physical linkage of targets, like the case with bsAbs, the pharmacokinetics are separate for each component.49 This allows for more flexibility in adjusting dose levels, ratios, and schedules to optimise exposure and therapeutic index.49 In the case of bsAbs, the pharmacokinetic profile is unified, influenced by both binding domains.42 In addition, the pharmacokinetics of bsAbs is often complicated by dual target-mediated drug disposition, as seen with short–half-life constructs like blinatumomab used in oncology, necessitating the need for continuous infusion.42
Immunogenicity also differs between combination and bsAbs approaches.49 The latter may induce domain-specific antidrug antibodies that interfere with one or both arms of the molecule, whereas the former requires evaluation of how coadministration alters each agent's immunogenicity profile compared to monotherapy.49 Safety challenges with BsAbs, especially T-cell engager constructs, include cytokine release, acute-phase reactions, and on-target but off-airway effects, such as excessive immune suppression and impaired host defense; mitigation often requires step-up dosing or prolonged infusions.52 In combinations, toxicities generally reflect pathway synergism but can be managed more easily by modulating the dose of each agent independently, something not possible in a fixed bispecific format.49 Dose optimisation for bsAbs must consider binding kinetics, dual-target occupancy, and the need to balance efficacy and safety within a single molecule.53 In contrast, biologic combinations typically begin with known monotherapy doses and evaluate multiple dose ratios to identify the optimal regimen.49
Regulatory expectations also diverge.54 BsAbs must demonstrate appropriate pharmacokinetics (PK) and pharmacodynamics (PD), as well as exposure–response relationships for both target interactions and often require complex bioanalytical assays capable of detecting functional, dual-active drugs.54 Combination products must show that each component contributes to the claimed effect and that their combined dosing is safe and effective, with attention to potential PK interactions and immunogenicity shifts.49 From a manufacturing perspective, bsAbs are more complex to engineer as they are prone to issues such as mispairing and aggregation, unlike biologic combinations, which leverage standard mAb production platforms but require manufacturing two distinct biologics.54 Overall, bsAbs are favored when spatial proximity or bridging of targets provides a clear biological advantage that cannot be achieved with separate agents.49 On the contrary, biologic combinations are preferred when maintaining independent control over each mechanism is critical for optimizing therapeutic index, safety, or dosing flexibility.49
From a mechanistic standpoint, bsAbs present an opportunity to simultaneously suppress upstream epithelial cytokines (TSLP, IL-33) and downstream effector pathways (IL-5, IL-13, IL-17).55 This dual modulation may be particularly beneficial in overlapping T2-high/T2-low phenotypes or in patients with persistent inflammation despite monotherapy.8 However, clinical trials are still limited, and evidence remains preliminary regarding whether bsAbs can consistently outperform optimised single-target therapies.56
Several bsAbs are currently under investigation for the treatment of asthma in preclinical studies (Table 5) and clinical trials (Table 6). For example, a bsAb targeting IL-4Rα and IL-5 is undergoing preclinical evaluation to inhibit Th2-driven pathways responsible for airway inflammation and eosinophilia.9 Additionally, monovalent and bivalent bsAbs, such as Zweimabs and Doppelmab, respectively, which target TSLP and IL-13, have been developed to modulate Th2 responses. These constructs exhibit a strong affinity for their human targets while maintaining efficacy comparable to their parental mAbs.57 Bivalent antibodies usually exhibit higher binding avidity, as they can bind with two Fab regions to the same antigen. In contrast, monovalent bsAbs bind each target antigen with only one Fab arm. While they can still target two different antigens simultaneously, they cannot cross-link the same antigen, unlike bivalents. However, monovalent bispecifics can still function by bridging two distinct targets, potentially facilitating interactions between cells. For instance, blinatumomab, a monovalent bispecific used in oncology, can bridge T cells and tumor cells by binding to CD3 on T cells and CD19 on tumor cells, each with a single binding arm (monovalent). This bridging brings the immune cells closer to the tumor cells, promoting targeted cellular cytotoxicity.58
| Bispecific | Target | Efficacy | Company |
|---|---|---|---|
| BBT002 | IL-4Rα and IL-5 | It provides more effective anti-inflammatory benefits and improved dosing convenience | Bambusa60 |
| Zweimabs and Doppelmabs | TSLP and IL-13 | They are highly potent with picomolar affinity to the targets | Boehringer Ingelheim57 |
| BiSpekDAb | IL-23 and TNF-α | It attenuated airway hyperresponsiveness (AHR), pulmonary inflammation, and other lung pathological changes in asthmatic rats | Protein Biopharmaceutical Lab led by Dr Abhay/H Pande61 |
| HXN-1013 | IL-33 and TSLP | It blocks TSLP and IL33 simultaneously, with each arm revealing activity comparable to the parent antibodies | Helixon Therapeutics62 |
| Trivalent bispecific nanobody | Two different epitopes of IL-5 and one epitope of albumin (HAS) | Excellent pharmacokinetic properties and pharmacodynamic effects in primates, supporting a 2–3-month dosing interval in future human studies | Shanghai Novamab Biopharmaceuticals63 |
| Bispecific | Target | Stage of development | Tested dose | Route | Efficacy | Sponsor/trial registration number |
|---|---|---|---|---|---|---|
| BITS7201A | IL-13 and IL-17 | Phase I | Single 30–750 mg and repeat 150–600 mg every 4 weeks for a total of 3 doses | IV and SC | Well tolerated, but a high incidence of anti-drug antibodies | Roche/NCT0274864259 |
| IBI3002 | IL-4α and TSLP | Phase I | 150–900 mg SQ (150, 300, 600, and 900 mg) and 600 mg IV | SC and IV | Actively recruiting, but in vitro assays have shown superiority over the marketed monoclonal antibodies for the respective targets | Innovent Biologics/NCT0621384464 |
| Lunsekimig | IL-13 and TSLP | Phase 2b | 400 mg single dose | SC | Well tolerated, suppressed T2 inflammation, and improved lung function in mild to moderate asthma | Sanofi/NCT0610200565 |
Furthermore, BITS7201A, a bsAb targeting IL-13 and IL-17, is currently being evaluated in a phase I clinical trial, aiming to address mixed eosinophilic and neutrophilic airway inflammation typically observed in more severe asthma phenotypes.59 While bsAbs hold considerable promise, challenges related to their molecular stability, immunogenicity, and scalable manufacturing still need to be carefully addressed to ensure their successful translation into clinical practice.54
Current challenges and future directions
Limitations of current treatments
Despite recent advances in asthma treatment, asthma remains a chronic condition that often requires lifelong management. Current treatments primarily aim to alleviate symptoms and prevent exacerbations.24 Approximately 10% of asthmatic patients remain uncontrolled despite using high doses of ICSs and oral corticosteroids.16 This is associated with increased mortality and risk of developing conditions such as diabetes, osteoporosis, hypertension, and cataracts.66 In addition, following the long-term usage of corticosteroids for over two weeks, even with low doses, around 15% of the patients develop steroid resistance. This resistance, characterised by a diminished anti-inflammatory response to corticosteroids, poses a significant challenge in asthma management and necessitates alternative therapeutic strategies.1Biological therapies have shown promise in overcoming steroid resistance by targeting specific receptors/inflammatory mediators with greater specificity, thereby reducing adverse side effects.1 While mAbs are effective, they are limited by their high cost and invasive route of administration. The currently approved mAbs are administered parenterally via intravenous or subcutaneous injections, necessitating the presence of medical personnel for administration.67 In addition, high dosages of mAbs are required to achieve effective concentration in the lung after parenteral administration due to the limited bioavailability at the target site. This increases systemic and off-target effects, further diminishing patients’ quality of life.24 Importantly, existing mAbs are effective only in T2-high asthma and eosinophilic asthma. No bsAbs have been approved or widely investigated for the treatment of chronic airway inflammatory diseases, particularly for T2-low asthma. Most of the ongoing bsAbs research focuses on the T2 asthma endotype by targeting common pathways involved.68 Therefore, future efforts should focus on advancing bsAbs as a novel biotherapeutic strategy targeting key mediators such as TSLP, alongside the development of inhalation formulation to enable localised, effective treatment for T2-low asthma.32
Although no bsAbs have yet been approved for the treatment of asthma, several candidates are currently being evaluated in early-phase clinical trials, including agents targeting IL-13/TSLP, IL-4Rα/IL-5, IL-4Rα/TSLP, IL-13/IL-17A, and IL-13/IL-4 as presented in Tables 4 and 5.57,59–62,64,65 Despite encouraging preclinical efficacy, multiple challenges have emerged that may limit their clinical translation.8 A major limitation is the heterogeneity of asthma, as not all patients exhibit concurrent activation of the dual inflammatory pathways targeted by bsAbs, underscoring the need for robust biomarker-driven patient stratification.1 In addition, most efficacy data are derived from animal models, which incompletely recapitulate the complexity and chronicity of human asthma, contributing to uncertainty regarding clinical efficacy.69 Long-term safety also remains a concern, as sustained dual cytokine blockade may impair host immune defenses and increase susceptibility to infections in a chronic disease setting.35 Furthermore, the structural complexity of bsAbs raises potential risks of immunogenicity and altered pharmacokinetics with repeated administration.41 Finally, manufacturing and scalability challenges associated with bispecific formats may hinder cost-effective development and widespread use.54 Collectively, these limitations underscore the need for optimised clinical trial design, long-term safety evaluation, and patient endotype-specific approaches to realize the therapeutic potential of bsAbs fully in asthma.49
Pulmonary route considerations and challenges
Pulmonary drug delivery has been integrated into the management of respiratory diseases such as asthma, chronic obstructive pulmonary disease (COPD), and cystic fibrosis for decades.24 This non-invasive route of administration allows the delivery of drugs directly to the site of action,27 while offering additional advantages such as rapid onset of action, enhanced bioavailability in the airways, lower required dose, and reduced systemic and off-target side effects. Moreover, administration by inhalation is considered more convenient and improves patient compliance, which is crucial for managing chronic conditions such as asthma.70Despite the benefits of pulmonary delivery, developing an inhalable biological drug is challenging.27 For a drug to be effectively deposited in the airways, several factors need to be considered, including particle size distribution, morphology, surface properties, and aerosol performance. For instance, a small aerodynamic particle diameter of below 5 µm is typically required to target the lower regions of the lungs. Another challenge when considering the inhalation route of administration is the mucociliary clearance and the alveolar macrophages, which serve as the natural barrier and clearance mechanism of the respiratory system.24 Hence, there is an emerging interest in developing inhalable biological drugs for targeted delivery to the site of inflammation, while addressing these challenges.
Existing inhalation devices, including nebulisers, dry powder inhalers (DPI), and metered dose inhalers (MDI), have been used in the management of asthma for several decades. Recently, the soft mist inhaler (SMI) has been introduced to the market in the past 10 to 15 years. MDIs are seldom investigated for biologics delivery due to compatibility issues with propellants and the limitation of the dose that can be delivered. Inhaled biologics are often explored with the use of nebulisers due to simpler formulation processes, faster development, and the capability of high-dose delivery. However, they are associated with stability in liquid formulations.24 Proteins in solutions are highly susceptible to hydrolysis-driven processes, including both physical and chemical degradation. Also, nebulisers are bulkier than other inhaler devices, and they require electricity to operate. SMI is a portable device that uses mechanical energy to generate aerosols. It delivers aerosol with a high fine-particle fraction at a slower speed, which enhances drug deposition in the lungs while reducing unwanted deposition in the mouth and throat.71 Additionally, the slower velocity and longer-lasting aerosol cloud make it easier for patients to coordinate the actuation and inhalation process. However, its use is limited by its higher cost and the requirement for basic assembly and priming before initial use, and protein stability in liquid form remains a problem.31 In contrast, biologics formulated in solid dosage forms are more stable, have longer shelf lives, and offer the potential to avoid cold-chain logistics. DPI formulation can be administered at higher doses to the airways. However, developing DPI formulations is complicated by the need for specialised drying processes and excipients to enhance stability and powder dispersion. Inhaled powder formulations of biologics can be produced by spray drying or spray freeze drying techniques. Spray drying is a highly efficient, cost-effective, and scalable process. It is capable of generating particles with a uniform size distribution, making it the preferred method for large-scale pharmaceutical production.72 In this one-step drying process, the liquid formulation is atomised into hot drying gas to allow the evaporation of solvent. But the structural integrity of the protein may be compromised due to thermal and shear stresses.72 Spray freeze drying, on the other hand, avoids thermal degradation by atomising formulation into a cryogenic liquid such as liquid nitrogen before lyophilisation. However, it still introduces shear stress during atomisation and amplifies protein aggregation due to the interfacial stresses. Hence, stabilising excipients should be carefully selected to preserve protein integrity throughout the drying process.
Conclusions
The production of bsAbs is often complex and faces plenty of challenges. Recombinant and genetic engineering methods are expensive and frequently yield issues with purity, immunogenicity, and stability.36 An alternative is chemical conjugation, which, while promising, still requires optimisation to transition from proof-of-concept studies to scalable production methods.37 Future research should also prioritise the use of single-chain variable fragments (scFvs) for bsAb design, as they are more suitable for large-scale conjugation and production.36 While current biologic treatments have transformed asthma management for some, there remains an urgent need for novel, accessible, and effective therapies, particularly for T2-low asthma. Inhalable bsAbs represent a promising frontier that warrants further preclinical and clinical investigation to address the limitations of systemic biologics and to meet the unmet needs in asthma care.Conflicts of interest
The authors declare no conflict of interest.Data availability
No primary research results, software or code have been included, and no new data were generated or analysed as part of this review.Acknowledgements
ZK is a PhD student at UWL who is grateful for the Vice-Chancellor Scholarship to support her PhD studies.References
- V. H. Patel, S. Thannir, M. Dhanani, I. Augustine, S. L. Sandeep and A. Mehadi, et al., Current Limitations and Recent Advances in the Management of Asthma, Disease-a-Month, 2023, 69(7), 101483 CrossRef PubMed . Available from: https://linkinghub.elsevier.com/retrieve/pii/S0011502922001675.
- S. M. Jayasooriya, G. Devereux, J. B. Soriano, N. Singh, R. Masekela and K. Mortimer, et al., Asthma: epidemiology, risk factors, and opportunities for prevention and treatment, Lancet Respir. Med., 2025, 13(8), 725–738 CrossRef PubMed . Available from: https://www.sciencedirect.com/science/article/pii/S2213260024003837.
- K. Mortimer, H. K. Reddel, P. M. Pitrez and E. D. Bateman, Asthma management in low and middle income countries: case for change, Eur. Respir. J., 2022, 60(3), 2103179 CrossRef PubMed . Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC9474897/.
- S. Momtazmanesh, S. S. Moghaddam, S. H. Ghamari, E. M. Rad, N. Rezaei and P. Shobeiri, et al., Global burden of chronic respiratory diseases and risk factors, 1990–2019: an update from the Global Burden of Disease Study 2019, eClinicalMedicine, 2023, 59, 101936 CrossRef PubMed . Available from: https://www.sciencedirect.com/science/article/pii/S258953702300113X.
- M. E. Kuruvilla, F. E. H. Lee and G. B. Lee, Understanding Asthma Phenotypes, Endotypes, and Mechanisms of Disease, Clin. Rev. Allergy Immunol., 2019, 56(2), 219–233 CrossRef . Available from: https://link.springer.com/10.1007/s12016-018-8712-1.
- D. Thomas, Y. Hamada, P. Gibson, C. E. Brightling, M. Castro and L. G. Heaney, Diagnosis and Treatment Options for T2-Low Asthma, J. Allergy Clin. Immunol.: Pract., 2025, 13(7), 1527–1539 CrossRef CAS . Available from: https://www.sciencedirect.com/science/article/pii/S2213219825004386.
- W. P. Alejandra, J. P. Miriam Irene, G. S. Fabio Antonio, R. G. R. Patricia, T. A. Elizabeth and J. P. Aleman-Aguilar, et al., Production of monoclonal antibodies for therapeutic purposes: A review, Int. Immunopharmacol., 2023, 120, 110376 CrossRef CAS . Available from: https://www.sciencedirect.com/science/article/pii/S1567576923006999.
- Y. Abe, Y. Suga, K. Fukushima, H. Ohata, T. Niitsu and H. Nabeshima, et al., Advances and Challenges of Antibody Therapeutics for Severe Bronchial Asthma, Int. J. Mol. Sci., 2021, 23(1), 83 CrossRef . Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC8744863/.
- M. Godar, K. Deswarte, K. Vergote, M. Saunders, H. De Haard and H. Hammad, et al., A bispecific antibody strategy to target multiple type 2 cytokines in asthma, J. Allergy Clin. Immunol., 2018, 142(4), 1185–1193 CrossRef CAS . Available from: https://linkinghub.elsevier.com/retrieve/pii/S0091674918308443.
- K. A. Eger and E. H. Bel, The emergence of new biologics for severe asthma, Curr. Opin. Pharmacol., 2019, 46, 108–115 CrossRef CAS PubMed . Available from: https://linkinghub.elsevier.com/retrieve/pii/S1471489219300153.
- N. A. Shaibie, N. D. F. Mohammad Faizal, F. Buang, T. Srichana and M. C. I. Mohd Amin, Inhaled biologics for respiratory diseases: clinical potential and emerging technologies, Drug Delivery Transl. Res., 2025, 15(11), 4098–4114, DOI:10.1007/s13346-025-01909-6.
- M. I. Reza and N. S. Ambhore, Inflammation in Asthma: Mechanistic Insights and the Role of Biologics in Therapeutic Frontiers, Biomedicines, 2025, 13(6), 1342 CrossRef CAS PubMed . Available from: https://www.mdpi.com/2227-9059/13/6/1342.
- A. Bakakos, N. Anagnostopoulos and P. Bakakos, Eosinophils and T2 inflammation in severe asthma, Explor. Asthma Allergy, 2024, 2(5), 399–409 CAS . Available from: https://www.explorationpub.com/Journals/eaa/Article/100953.
- A. Lindsley, N. Lugogo, K. Reeh, J. Spahn and J. Parnes, Asthma Biologics Across the T2 Spectrum of Inflammation in Severe Asthma: Biomarkers and Mechanism of Action, J. Asthma Allergy, 2025, 18, 33–57 CrossRef CAS PubMed . Available from: https://www.dovepress.com/asthma-biologics-across-the-t2-spectrum-of-inflammation-in-severe-asth-peer-reviewed-fulltext-article-JAA.
- W. J. Calhoun and G. L. Chupp, The new era of add-on asthma treatments: where do we stand?, Allergy, Asthma, Clin. Immunol., 2022, 18(1), 42 CrossRef PubMed . Available from: https://aacijournal.biomedcentral.com/articles/10.1186/s13223-022-00676-0.
- R. Tan, M. F. Liew, H. F. Lim, B. P. Leung and W. S. F. Wong, Promises and challenges of biologics for severe asthma, Biochem. Pharmacol., 2020, 179, 114012 CrossRef CAS PubMed . Available from: https://linkinghub.elsevier.com/retrieve/pii/S0006295220302409.
- I. Esteban-Gorgojo, D. Antolín-Amérigo, J. Domínguez-Ortega and S. Quirce, Non-eosinophilic asthma: current perspectives, J. Asthma Allergy, 2018, 11, 267–281 CrossRef CAS PubMed . Available from: https://www.dovepress.com/non-eosinophilic-asthma-current-perspectives-peer-reviewed-article-JAA.
- A. M. Fitzpatrick, B. E. Chipps, F. Holguin and P. G. Woodruff, T2-“Low” Asthma: Overview and Management Strategies, J. Allergy Clin. Immunol.: Pract., 2020, 8(2), 452–463 Search PubMed . Available from: https://linkinghub.elsevier.com/retrieve/pii/S2213219819309419.
- S. Smolinska, D. Antolín-Amérigo, F. D. Popescu and M. Jutel, Thymic Stromal Lymphopoietin (TSLP), Its Isoforms and the Interplay with the Epithelium in Allergy and Asthma, Int. J. Mol. Sci., 2023, 24(16), 12725 Search PubMed . Available from: https://www.mdpi.com/1422-0067/24/16/12725.
- B. N. Lambrecht, H. Hammad and J. V. Fahy, The Cytokines of Asthma, Immunity, 2019, 50(4), 975–991 Search PubMed . Available from: https://linkinghub.elsevier.com/retrieve/pii/S1074761319301359.
- F. M. Shahidi, Current recommendations for the treatment of mild asthma, J. Asthma Allergy, 2010, 2010(3), 169–176 Search PubMed . Available from: https://www.dovepress.com/current-recommendations-for-the-treatment-of-mild-asthma-peer-reviewed-article-JAA.
- O. O'Carroll, C. McCarthy and M. W. Butler, Treatments for poorly controlled asthma, Br. Med. J., 2021, 375, n2355 Search PubMed . Available from: https://www.bmj.com/lookup/doi/10.1136/bmj.n2355.
- R. H. Green, C. E. Brightling, I. D. Pavord and A. J. Wardlaw, Management of asthma in adults: current therapy and future directions, Postgrad. Med. J., 2003, 79(931), 259–267 Search PubMed . Available from: https://academic.oup.com/pmj/article/79/931/259/7045660.
- A. Boboltz, S. Kumar and G. A. Duncan, Inhaled drug delivery for the targeted treatment of asthma, Adv. Drug Delivery Rev., 2023, 198, 114858 Search PubMed . Available from: https://www.sciencedirect.com/science/article/pii/S0169409X23001734.
- G. Kardas, M. Panek, P. Kuna, P. Damiański and M. Kupczyk, Monoclonal antibodies in the management of asthma: Dead ends, current status and future perspectives, Front. Immunol., 2022, 13, 983852 Search PubMed . Available from: https://www.frontiersin.org/articles/10.3389/fimmu.2022.983852/full.
- H. J. Jin, Biological treatments for severe asthma, Yeungnam Univ. J. Med., 2020, 37(4), 262–268 Search PubMed . Available from: https://e-yujm.org/journal/view.php?doi=https://doi.org/10.12701/yujm.2020.00647.
- M. C. McGregor, J. G. Krings, P. Nair and M. Castro, Role of Biologics in Asthma, Am. J. Respir. Crit. Care Med., 2019, 199(4), 433–445 Search PubMed . Available from: https://www.atsjournals.org/doi/10.1164/rccm.201810-1944CI.
- E. E. West, M. Kashyap and W. J. Leonard, TSLP: a key regulator of asthma pathogenesis, Drug Discovery Today: Dis. Mech., 2012, 9(3–4), e83–e88 Search PubMed . Available from: https://linkinghub.elsevier.com/retrieve/pii/S1740676512000144.
- J. Parnes, N. A. Molfino, G. Colice, U. Martin, J. Corren and A. Menzies-Gow, Targeting TSLP in Asthma, J. Asthma Allergy, 2022, 15, 749–765 Search PubMed . Available from: https://www.dovepress.com/targeting-tslp-in-asthma-peer-reviewed-fulltext-article-JAA.
- R. Panettieri Jr., N. Lugogo, J. Corren and C. Ambrose, Tezepelumab for Severe Asthma: One Drug Targeting Multiple Disease Pathways and Patient Types, J. Asthma Allergy, 2024, 17, 219–236 CrossRef PubMed . Available from: https://www.dovepress.com/tezepelumab-for-severe-asthma-one-drug-targeting-multiple-disease-path-peer-reviewed-fulltext-article-JAA.
- M. R. Elliott, C. E. Grogan and G. D. Marshall, An Update on Monoclonal Antibody Therapy to Treat Moderate-to-Severe Asthma: Benefits, Choices, and Limitations, Am. J. Med., 2023, 136(8), 738–744 Search PubMed . Available from: https://linkinghub.elsevier.com/retrieve/pii/S0002934323003273.
- L. Rogers, M. Jesenak, L. Bjermer, N. A. Hanania, S. F. Seys and Z. Diamant, Biologics in severe asthma: A pragmatic approach for choosing the right treatment for the right patient, Respir. Med., 2023, 218, 107414 CrossRef PubMed . Available from: https://linkinghub.elsevier.com/retrieve/pii/S0954611123003025.
- E. Ridolo, F. Pucciarini, M. C. Nizi, E. Makri, P. Kihlgren and L. Panella, et al., Mabs for treating asthma: omalizumab, mepolizumab, reslizumab, benralizumab, dupilumab, Hum. Vaccines Immunother., 2020, 16(10), 2349–2356 CrossRef CAS PubMed . Available from: https://www.tandfonline.com/doi/full/10.1080/21645515.2020.1753440.
- N. A. Hanania, R. Niven, P. Chanez, D. Antoine, P. Pfister and L. Garcia Conde, et al., Long-term effectiveness and safety of omalizumab in pediatric and adult patients with moderate-to-severe inadequately controlled allergic asthma, World Allergy Organ. J., 2022, 15(10), 100695 CrossRef CAS PubMed . Available from: https://linkinghub.elsevier.com/retrieve/pii/S1939455122000710.
- A. N. Sitek, J. T. Li and T. Pongdee, Risks and safety of biologics: A practical guide for allergists, World Allergy Organ. J., 2023, 16(1), 100737 CrossRef PubMed . Available from: https://linkinghub.elsevier.com/retrieve/pii/S1939455122001132.
- P. M. O'Byrne, R. A. Panettieri, C. Taube, C. Brindicci, M. Fleming and P. Altman, Development of an inhaled anti-TSLP therapy for asthma, Pulm. Pharmacol. Ther., 2023, 78, 102184 CrossRef PubMed.
- L. Borish and J. A. Culp, Asthma: a syndrome composed of heterogeneous diseases, Ann. Allergy, Asthma, Immunol., 2008, 101(1), 1–9 CrossRef CAS PubMed . Available from: https://linkinghub.elsevier.com/retrieve/pii/S1081120610608265.
- U. Brinkmann and R. E. Kontermann, The making of bispecific antibodies, mAbs, 2017, 9(2), 182–212 CrossRef CAS PubMed . Available from: https://www.tandfonline.com/doi/full/10.1080/19420862.2016.1268307.
- A. A. Khosla, K. Jatwani, R. Singh, A. Reddy, I. Jaiyesimi and A. Desai, Bispecific Antibodies in Lung Cancer: A State-of-the-Art Review, Pharmaceuticals, 2023, 16(10), 1461 CrossRef CAS PubMed.
- Antibody structure–function relationships, in Therapeutic Antibody Engineering, Elsevier, 2012, pp. 37–595. Available from: https://linkinghub.elsevier.com/retrieve/pii/B9781907568374500038. Search PubMed.
- Q. Wang, Y. Chen, J. Park, X. Liu, Y. Hu and T. Wang, et al., Design and Production of Bispecific Antibodies, Antibodies, 2019, 8(3), 43 Search PubMed . Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6783844/.
- S. M. Choi, J. H. Lee, S. Ko, S. S. Hong and H. E. Jin, Mechanism of Action and Pharmacokinetics of Approved Bispecific Antibodies, Biomol. Ther., 2024, 32(6), 708–722 CrossRef CAS PubMed . Available from: https://www.biomolther.org/journal/view.html?doi=https://doi.org/10.4062/biomolther.2024.146.
- M. Collins and H. Khalili, Soluble Papain to Digest Monoclonal Antibodies; Time and Cost-Effective Method to Obtain Fab Fragment, Bioengineering, 2022, 9(5), 209 CrossRef CAS PubMed . Available from: https://www.mdpi.com/2306-5354/9/5/209.
- 12 - Antibody fragments as therapeutics, in Therapeutic Antibody Engineering, ed. W.R Strohl and L.M Strohl, Woodhead Publishing, Woodhead Publishing Series in Biomedicine, 2012, pp. 265–595. Available from: https://www.sciencedirect.com/science/article/pii/B9781907568374500129. Search PubMed.
- S. B. Ebrahimi and D. Samanta, Engineering protein-based therapeutics through structural and chemical design, Nat. Commun., 2023, 14(1), 2411 CrossRef CAS PubMed . Available from: https://www.nature.com/articles/s41467-023-38039-x.
- E. O. Okwuofu, A. C. H. Yong, J. C. W. Lim and J. Stanslas, The era of multiple biologics: Is combination and switching an option in the management of severe asthma?, Pulm. Pharmacol. Ther., 2025, 90, 102375 CrossRef CAS PubMed . Available from: https://linkinghub.elsevier.com/retrieve/pii/S109455392500032X.
- L. Carriera, M. Fantò, A. Martini, A. D'Abramo, G. Puzio and M. U. Scaramozzino, et al., Combination of Biological Therapy in Severe Asthma: Where We Are?, J. Pers. Med., 2023, 13(11), 1594 CrossRef PubMed . Available from: https://www.mdpi.com/2075-4426/13/11/1594.
- Y. Chen, L. Wang and J. Xie, Combining Dual Biologics Therapy for Severe Asthma: A Series of Ten Cases, J. Asthma Allergy, 2025, 18, 507–517 CrossRef PubMed . Available from: https://www.dovepress.com/combining-dual-biologics-therapy-for-severe-asthma-a-series-of-ten-cas-peer-reviewed-fulltext-article-JAA.
- S. Zheng, R. Prell, J. Sheng, Y. Wang and L. Hamuro, Changing the drug development and therapeutic paradigm with biologic drug combinations and bispecifics: How to choose between these two approaches?, Clin. Transl. Sci., 2022, 15(9), 2096–2104 CrossRef PubMed . Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC9468564/.
- J. H. Kim, D. S. Kim, H. S. Park and Y. S. Kim, Engineering bispecific T-cell engagers to deplete eosinophils for the treatment of severe eosinophilic asthma, Clin. Immunol., 2023, 255, 109755 CrossRef CAS PubMed . Available from: https://linkinghub.elsevier.com/retrieve/pii/S1521661623005181.
- S. Y. Chu, H. M. Horton, E. Pong, I. W. L. Leung, H. Chen and D. H. Nguyen, et al., Reduction of total IgE by targeted coengagement of IgE B-cell receptor and FcγRIIb with Fc-engineered antibody, J. Allergy Clin. Immunol., 2012, 129(4), 1102–1115 CrossRef CAS PubMed . Available from: https://linkinghub.elsevier.com/retrieve/pii/S0091674911018343.
- M. K. Wattana, J. Rowland, A. Qdaisat, H. Levavi, T. Anagnostou and L. Sanchez, et al., Diagnosis and management of bispecific T cell–engaging antibody toxicity: A primer for emergency physicians, Cancer Treat. Rev., 2025, 134, 102889 CrossRef CAS PubMed . Available from: https://www.sciencedirect.com/science/article/pii/S0305737225000118.
- A. Amash, G. Volkers, P. Farber, D. Griffin, K. S. Davison and A. Goodman, et al., Developability considerations for bispecific and multispecific antibodies, mAbs, 2024, 16(1), 2394229 CrossRef . Available from: https://www.tandfonline.com/doi/full/10.1080/19420862.2024.2394229.
- M. S. Karbyshev, I. V. Kalashnikova, V. V. Dubrovskaya, K. O. Baskakova, P. K. Kuzmichev and V. Sandig, Trends and challenges in bispecific antibody production, J. Chromatogr., A, 2025, 1744, 465722 Search PubMed . Available from: https://linkinghub.elsevier.com/retrieve/pii/S0021967325000718.
- E. Israel, M. E. Wechsler, D. J. Jackson and W. C. Moore, Anti-cytokine biologics for asthma in adults, Lancet, 2025, 406(10516), 2282–2294 CrossRef CAS PubMed . Available from: https://linkinghub.elsevier.com/retrieve/pii/S0140673625016253.
- Research C for DE and, Bispecific Antibodies: An Area of Research and Clinical Applications, FDA, 2024. Available from: https://www.fda.gov/drugs/spotlight-cder-science/bispecific-antibodies-area-research-and-clinical-applications Search PubMed.
- S. Venkataramani, S. Low, B. Weigle, D. Dutcher, K. Jerath and M. Menzenski, et al., Design and characterization of Zweimab and Doppelmab, high affinity dual antagonistic anti-TSLP/IL13 bispecific antibodies, Biochem. Biophys. Res. Commun., 2018, 504(1), 19–24 Search PubMed . Available from: https://www.sciencedirect.com/science/article/pii/S0006291X18317467.
- N. Dimasi, R. Fleming, K. F. Sachsenmeier, B. Bezabeh, C. Hay and J. Wu, et al., Guiding bispecific monovalent antibody formation through proteolysis of IgG1 single-chain, mAbs, 2017, 9(3), 438–454 CrossRef CAS PubMed . Available from: https://www.tandfonline.com/doi/full/10.1080/19420862.2016.1277301.
- T. L. Staton, K. Peng, R. Owen, D. F. Choy, C. R. Cabanski and A. Fong, et al., A phase I, randomized, observer-blinded, single and multiple ascending-dose study to investigate the safety, pharmacokinetics, and immunogenicity of BITS7201A, a bispecific antibody targeting IL-13 and IL-17, in healthy volunteers, BMC Pulm. Med., 2019, 19(1), 5 Search PubMed . Available from: https://bmcpulmmed.biomedcentral.com/articles/10.1186/s12890-018-0763-9.
- T. Ho, T. Zhai, Y. Chen, S. Dai, S. Zhou and Z. Zhang, et al., BBT002: A Novel Tetravalent Bispecific Antibody Targeting IL4Ra and IL5 for Enhanced Asthma Therapy, Am. J. Respir. Crit. Care Med., 2025, 211(Abstracts), A1302–A1302 CrossRef . Available from: https://www.atsjournals.org/doi/abs/10.1164/ajrccm.2025.211.Abstracts.A1302.
- C. K. Gala, P. A. Sandeep and S. S. Sharma, Bispecific domain antibody attenuates airway hyperresponsiveness and pulmonary inflammation in ovalbumin-lipopolysaccharide induced asthma model by inhibiting IL-23 and TNF-α, Pulm. Pharmacol. Ther., 2025, 88, 102347 CrossRef CAS PubMed . Available from: https://linkinghub.elsevier.com/retrieve/pii/S1094553925000045.
- H. Ran, J. Huang, H. Li, L. Dong, D. Liu and Y. Zhou, et al., Bispecific Antibody Targeting Both IL33 and TSLP for Asthma and COPD, Am. J. Respir. Crit. Care Med., 2025, 211(Abstracts), A1382–A1382 Search PubMed . Available from: https://www.atsjournals.org/doi/10.1164/ajrccm.2025.211.Abstracts.A1382.
- L. Ma, M. Zhu, G. Li, J. Gai, Y. Li and H. Gu, et al., Preclinical development of a long-acting trivalent bispecific nanobody targeting IL-5 for the treatment of eosinophilic asthma, Respir. Res., 2022, 23(1), 316 CrossRef CAS PubMed . Available from: https://respiratory-research.biomedcentral.com/articles/10.1186/s12931-022-02240-1.
- Y. Xiong, L. Li, S. Zhou, K. Tang, F. He and Y. Gao, et al., Preclinical Characterization of IBI3002, an Anti-IL-4Rα and Anti-TSLP Bispecific Antibody That Potently Dampens Inflammatory Response and Alleviates Asthma in Mice, Am. J. Respir. Crit. Care Med., 2025, 211(Abstracts), A7432–A7432 Search PubMed . Available from: https://www.atsjournals.org/doi/10.1164/ajrccm.2025.211.Abstracts.A7432.
- A. Deiteren, E. Krupka, L. Bontinck, K. Imberdis, G. Conickx and S. Bas, et al., A proof-of-mechanism trial in asthma with lunsekimig, a bispecific NANOBODY molecule, Eur. Respir. J., 2025, 65(4), 2401461 Search PubMed . Available from: https://publications.ersnet.org/lookup/doi/10.1183/13993003.01461-2024.
- T. Volmer, T. Effenberger, C. Trautner and R. Buhl, Consequences of long-term oral corticosteroid therapy and its side-effects in severe asthma in adults: a focused review of the impact data in the literature, Eur. Respir. J., 2018, 52(4), 1800703 CrossRef PubMed . Available from: https://publications.ersnet.org/content/erj/52/4/1800703.
- M. S. Castelli, P. McGonigle and P. J. Hornby, The pharmacology and therapeutic applications of monoclonal antibodies, Pharmacol. Res. Perspect., 2019, 7(6), e00535 CrossRef PubMed . Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6923804/.
- P. A. Shah and C. Brightling, Biologics for severe asthma—Which, when and why?, Respirology, 2023, 28(8), 709–721 CrossRef PubMed . Available from: https://onlinelibrary.wiley.com/doi/10.1111/resp.14520.
- T. Hartung, The (misleading) role of animal models in drug development, Front. Drug Discovery, 2024, 4, 1355044 CrossRef . Available from: https://www.frontiersin.org/journals/drug-discovery/articles/10.3389/fddsv.2024.1355044/full.
- H. W. Pan, J. Guo, L. Zhu, S. W. S. Leung, C. Zhang and J. K. W. Lam, Enhanced powder dispersion of dual-excipient spray-dried powder formulations of a monoclonal antibody and its fragment for local treatment of severe asthma, Int. J. Pharm., 2023, 644, 123272 CrossRef CAS PubMed . Available from: https://linkinghub.elsevier.com/retrieve/pii/S0378517323006920.
- T. Iwanaga, Y. Tohda, S. Nakamura and Y. Suga, The Respimat® Soft Mist Inhaler: Implications of Drug Delivery Characteristics for Patients, Clin. Drug Invest., 2019, 39(11), 1021–1030 Search PubMed . Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6800401/.
- H. W. Pan, H. C. Seow, J. C. K. Lo, J. Guo, L. Zhu and S. W. S. Leung, et al., Spray-Dried and Spray-Freeze-Dried Powder Formulations of an Anti-Interleukin-4Rα Antibody for Pulmonary Delivery, Pharm. Res., 2022, 39(9), 2291–2304, DOI:10.1007/s11095-022-03331-w.
| This journal is © The Royal Society of Chemistry 2026 |
