Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Photoactive chiral structures for light-driven enantioselective carbon–carbon bond-forming reactions
Vijay Kumar S
 ,
A. Hameed Ibrahim
,
A. Hameed Ibrahim
 and
Anjan Das
and
Anjan Das
 *
*
Department of Chemistry, SRM Institute of Science & Technology, Kattankulathur, Tamil Nadu 603203, India. E-mail: anjand@srmist.edu.in; Tel: +91 44 27417777
First published on 15th April 2026
Abstract
Asymmetric photocatalysis has emerged as a powerful strategy for forming enantioselective carbon–carbon bonds under mild conditions with high levels of enantioselectivity. Within this area, photoactive chiral systems represent an attractive and useful approach, as they eliminate the need for additional photosensitizers by combining the ability of light absorption and chiral induction within a single catalyst framework. Such catalysts act as chiral photosensitizers, enabling efficient harvesting of suitable light, generation of reactive excited states, and stereoselective bond formation. Recent studies demonstrate the effectiveness of this strategy in enantioselective C–C bond-forming reactions such as radical additions, cycloadditions, and cross-couplings. These methods improve efficiency and atom economy while broadening the scope of complex molecule synthesis. This review summarizes recent advances, mechanistic insights, and prospects of photocatalytic enantioselective C–C bond formation using self-sensitizing chiral systems, with relevance to drug discovery and fine chemical synthesis.
1. Introduction
Carbon–carbon (C–C) bond formation is one of the most fundamental and essential transformations in organic synthesis, serving as the cornerstone for constructing complex molecular architectures, including pharmaceuticals, agrochemicals, and materials.1 Traditional methods for asymmetric C–C bond formation rely on organocatalysis, transition metal catalysis, or enzymatic catalysis for the synthesis of enantiopure products.2Photocatalysis utilizes suitable light to generate highly reactive intermediates under mild conditions, enabling new reaction pathways that are otherwise challenging to achieve.3 The combination of photocatalysis with asymmetric catalysis, often referred to as asymmetric photocatalysis, has opened new pathways for achieving stereoselective transformations with excellent enantiocontrol as well as diastereocontrol.4 This approach takes advantage of excited-state intermediates, single-electron transfer (SET) processes, energy transfer, or radical-pairing mechanisms to enable bond formation with high selectivity.
As depicted in Fig. 1, key strategies developed for photocatalytic asymmetric C–C bond formation reactions can be mainly categorized into four routes. In the chiral organo-photocatalytic strategy, the photosensitization and chiral induction originate from a single photoactive chiral molecule, which includes chiral amines, phosphoric acids, and thioureas, preferably following the primary asymmetric photocatalytic routes (Fig. 2).5 Generally, substrates bind to the catalyst through hydrogen bonding or ionic interactions. In dual catalysis, photocatalysts (achiral transition metal complexes or organic dyes) and chiral organic molecules are separate entities that work synergistically to achieve asymmetric C–C bond formation via a secondary asymmetric photocatalytic route.6 This strategy successfully allows many enantioselective cross-coupling reactions, where light-driven SET initiates radical recombination and energy transfer (EnT) enables asymmetric cycloaddition and rearrangement reactions that are difficult to control under conventional conditions. In the field of discrete chiral photocatalysts for asymmetric organic reactions, photoactive chiral organic molecules and several metal complexes featuring both chiral and non-chiral ligands have been shown to achieve excellent stereocontrol following the primary asymmetric photocatalytic route.7 This success is likely due to the direct interaction between the substrates or reagents and the central transition metal. Two common strategies are documented to generate chiral metal complexes as discrete photocatalysts. The first strategy involves using a chiral organic ligand coordinated with an appropriate metal. The second, known as “chiral-at-metal”, involves coordinating achiral organic ligands with a metal to form an octahedral complex (Werner complexes), where the overall structure of the complex itself exhibits chirality. In the case of electron donor–acceptor (EDA) complexes, the formation of the EDA takes place in the ground state between the chiral ligand and the substrates for the formation of an active photocatalyst.8 Finally, bifunctional chiral photocatalysts are specialized catalysts that integrate two distinct functional components: a photocatalytic unit, which absorbs light and initiates photochemical transformations, typically through single-electron transfer (SET) or energy transfer (EnT) mechanisms via a secondary asymmetric photocatalytic route (Fig. 2), and a chiral-inducing unit, which provides a well-defined chiral environment to control the enantioselectivity of the reaction.9
While these published reviews provide important and comprehensive insights into enantioselective photocatalysis, they predominantly focus on broad catalytic concepts, catalyst development, or general mechanistic paradigms for dual catalytic systems. The purpose of this review article is to summarize advancements in homogeneous discrete chiral photocatalytic asymmetric C–C bond formation and its role in organic synthesis, specifically focusing on asymmetric C–C bond formation under visible-light irradiation, enabling a more targeted and practically useful understanding for synthetic chemists. We introduce a systematic classification based on various approaches to C–C bond construction, which allows direct comparison of diverse transformations across different catalytic systems. Moreover, this review compiles recent developments in this class of chiral photocatalysts and mechanistic insights, helping researchers gain a deeper understanding, strengthen fundamental knowledge and stay updated. Other photocatalytic strategies for photocatalytic enantioselective carbon–carbon bond formation reactions are not included here as they have already been reported.10 Additionally, this review addresses key challenges like low efficiency, limited substrate scope, and scalability for practical applications.
2. Chiral organo-photocatalysts
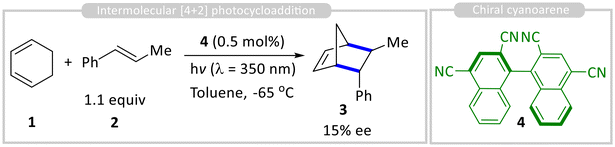
Chiral organo-photocatalysis has emerged as a powerful strategy for asymmetric carbon–carbon (C–C) bond formation, combining the advantages of organocatalysis and photocatalysis to achieve high enantioselectivity under mild conditions. Unlike traditional photocatalysis, which often relies on metal-based catalysts, organo-photocatalysis employs photoactive organic molecules, making it a sustainable and metal-free approach.11 Chiral organic chromophores have previously been reported for photosensitized asymmetric isomerization of cyclic alkenes, followed by thermal cycloaddition with a suitable dienophile to facilitate asymmetric carbon–carbon bond formation.12The photoactive chiral organic structure for enantioselective C–C bond formation was first reported by Schuster and coworkers. They developed a photocatalytic asymmetric [4 + 2] photocycloaddition of diene 1 and dienophile 2 using an axially chiral cyanoarene sensitizer 4 as an organo-photocatalyst. Their study revealed that, in polar solvents, the catalyst promotes radical cation Diels–Alder reactions, forming racemic products, while in nonpolar solvents, an enantioselective cycloadduct 3 (15% ee) is obtained (Scheme 1).13 Mechanistic investigations suggest that electron transfer in polar solvents generates a radical-ion pair, whereas in nonpolar solvents, an exciplex is formed. Time-resolved fluorescence studies indicated the presence of two diastereomeric catalyst–styrene exciplexes, with enantioselectivity attributed to differences in their excited-state lifetimes.
Bach and coworkers made a significant advancement in enantioselective photocatalytic C–C bond-forming reactions by integrating a suitable sensitizer into a chiral template. They accomplished this by incorporating an aromatic ketone sensitizer into the chiral Kemp's acid motif while maintaining its essential hydrogen-bonding functionality, enabling it to act as a discrete chiral organo-photocatalyst. Using this approach, they have reported various enantioselective carbon–carbon bond-forming reactions by either electron transfer or energy transfer from the attached organic photosensitizer 22 (Scheme 2). This photocatalytic process proceeds via the primary asymmetric photocatalytic cycle (Fig. 2a), with pre-association between the catalyst and substrate occurring in the ground state prior to photoexcitation. Using chiral template 22a, it was found to be optimal for an intramolecular enantioselective [2 + 2] photocycloaddition of 2-quinolone 5 via an energy transfer process (Scheme 2a).14 The rigid cyclohexyl backbone and sterically demanding benzoxazole moiety restricted conformational flexibility, effectively facilitating facial differentiation in the prochiral substrate 5 and producing enantioselective product 6 with excellent selectivity (88% yield with 88% ee). Since hydrogen-bonding interactions are stronger at low temperatures, the highest enantioselectivity was achieved under these conditions. Using the same strategy, they incorporated xanthone to develop organo-photocatalyst 22b, which was then applied to the intramolecular [2 + 2] cycloaddition of quinolones 7 & 8, forming cycloadducts 6 & 8. They have achieved excellent yield (70%) and ee (90%), with substrate 7 significantly higher than substrate 8 (Scheme 2b).15 As in both cases (7 and 5), catalyst 22b remained fully bound to the substrate at the beginning of the reaction, indicating that ground-state association was not responsible for the change in selectivity. Instead, the enhanced selectivity was attributed to the efficiency of photosensitization and the rate of the subsequent reaction. Although the substrates absorb light at the irradiation wavelength, the xanthone photocatalyst 22a has a much higher extinction coefficient than the substrates, enabling it to capture nearly all incident photons under the reaction conditions. Consequently, the racemic background reaction is significantly suppressed, resulting in high enantioselectivity.
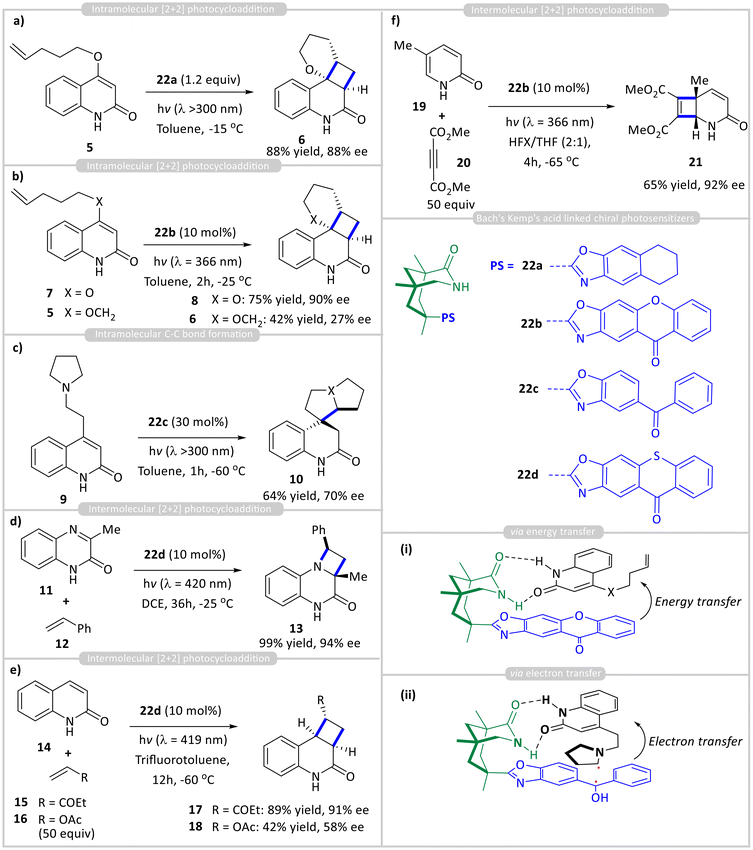 | ||
| Scheme 2 Chiral organo-photocatalysis for enantioselective (a) & (b) intramolecular cycloadditions; (c) intramolecular C–C bond formation; and (d)–(f) intermolecular cycloaddition reactions. | ||
In a previous study, using the electron transfer strategy, Bach et al. reported the intramolecular cyclization of pyrrolidine 9 to spirocycle 10 using 30 mol% of ketone catalyst 22c, achieving 70% enantiomeric excess (ee) through a hydrogen-bonding mechanism and photoinduced electron transfer (PET) (Scheme 2c).16 It is important to note that intermolecular photoreactions are significantly more challenging than their intramolecular counterparts due to the inherent competition between substrate dissociation and cyclization. Despite this difficulty, Bach successfully developed an intermolecular [2 + 2] photocycloaddition between cyclic imine 11 and styrene 12, photocatalyzed by 22d to produce the enantioselective cycloadduct 13 with 99% yield and 94% ee (Scheme 2d).17
Further studies on the intermolecular [2 + 2] photocycloaddition between quinolone 14 and various alkene coupling partners (15 and 16) revealed that the reaction, catalysed by 22d, required 50 equivalents of the alkene to ensure that cyclization occurred before dissociation from the catalyst (Scheme 2e).18 Based on this, it could be concluded that alkenes that react more slowly are expected to yield products with lower enantioselectivities. For example, vinyl acetate 16 reacts over an order of magnitude slower than ethyl vinyl ketone 15 when photosensitized by an achiral thioxanthone, resulting in significantly lower enantiomeric excess in the corresponding cycloadducts (91% ee vs. 58% ee).
In another study, they developed an intermolecular [2 + 2] photocycloaddition between pyridone 19 and acetylenedicarboxylates 20 that is catalyzed by 22b. Since pyridone 19 does not absorb the irradiated light under these reaction conditions, the enantioselectivity does not strongly correlate with catalyst loading. However, a clear relationship was observed between the alkyne concentration and enantioselectivity. It was proposed that a higher concentration of the coupling partner facilitates a faster bimolecular reaction, reducing the likelihood of substrate dissociation and leading to improved enantioselectivity (Scheme 2f).19
Xanthone and thioxanthone catalysts were initially limited to lactam-containing substrates, delivering good efficiency and enantioselectivity. To broaden the range of compatible binding motifs, Bach and co-workers developed new catalysts that integrate a thioxanthone sensitizer into alternative chiral hydrogen-bonding scaffolds, such as BINOL-derived phosphoric acids 29a and chiral thioureas 29b (Scheme 3). Using BINOL-derived phosphoric acid 29a as a chiral photocatalyst, an intermolecular [2 + 2] cycloaddition between carboxylic acid 23 and cyclopentene 24 was achieved with 61% yield and 86% ee (Scheme 3a).20 In contrast, the thiourea-linked thioxanthone 29b showed poor enantioselectivity (12% ee) in a sensitized 6π-electrocyclization. NMR studies confirmed that carboxylic acid 25 binds to catalyst 29a under reaction conditions, while computational analysis suggests that the low enantioselectivity arises from multiple, energetically similar binding modes of the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 complex. Additionally, thioxanthone catalyst 29a effectively promoted the enantioselective photocycloaddition of N,O-acetal 26 and alkene 27, affording 95% ee (Scheme 3b).21 NMR analysis indicated that 26 exists in equilibrium between a cyclic N,O-acetal and a ring-opened imine form. Emission studies showed that the triplet energy of the iminium ion (51 kcal mol−1) is lower than that of the thioxanthone catalyst (56 kcal mol−1), supporting an exothermic energy transfer process.
1 complex. Additionally, thioxanthone catalyst 29a effectively promoted the enantioselective photocycloaddition of N,O-acetal 26 and alkene 27, affording 95% ee (Scheme 3b).21 NMR analysis indicated that 26 exists in equilibrium between a cyclic N,O-acetal and a ring-opened imine form. Emission studies showed that the triplet energy of the iminium ion (51 kcal mol−1) is lower than that of the thioxanthone catalyst (56 kcal mol−1), supporting an exothermic energy transfer process.
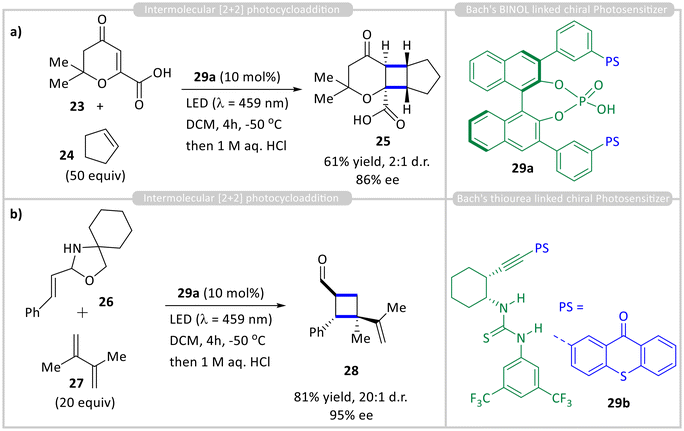 | ||
| Scheme 3 Chiral BINOL-derived phosphoric acid and thiourea-linked thioxanthone-sensitised enantioselective (a) & (b) intermolecular [2 + 2] cycloaddition reactions. | ||
Melchiorre and co-workers introduced an innovative approach to asymmetric photocatalysis based on electron donor–acceptor (EDA) complexes. In this method, an electron-rich donor and an electron-deficient acceptor interact in the ground state to form an EDA complex. This interaction results in the emergence of a new charge-transfer absorption band, which can be interpreted as an intra-complex electron transfer from the donor's HOMO to the acceptor's LUMO. Using this strategy, they reported enantioselective α-alkylation of aldehyde 30 with alkyl halide 31 to provide enantioselective 32 in 87% yield with 92% ee (Scheme 4a).22 A chiral secondary amine forms an enamine intermediate, which interacts with an alkyl bromide to create a visible-light-absorbing EDA complex. Excitation triggers electron transfer, generating radicals. The chiral catalyst facilitates both photochemical initiation and enantioselective radical addition (Scheme 4e). Melchiorre and co-workers showed that bromomalonate 34 enables the α-alkylation of aldehydes 30 using amine organocatalyst 36, achieving up to 94% ee and complete γ-selectivity for enals (Scheme 4b).23 While an EDA complex was initially suspected, UV–vis studies ruled it out, and Stern–Volmer quenching confirmed a SET mechanism. The excited enamine reduces bromomalonate 34, forming a malonyl radical that reacts with the ground-state enamine. Unlike EDA reactions, chain propagation occurs via bromine abstraction. The photochemical step serves only to initiate the radical chain (Scheme 4e). Alemán and co-workers reported a similar photo-organocatalytic reaction using a thioxanthone-substituted organocatalyst 39, which efficiently produces alkylated aldehyde 37 with high yield (up to 99%) and enantioselectivity (up to 99%) (Scheme 4c).24 In this reaction, rather than the enamine acting as the reducing agent, the thioxanthone catalyst 39 facilitates the reduction of bromomalonate 34, initiating the radical chain process. This mechanistic shift highlights the role of thioxanthone as a key photoredox mediator, broadening the scope of organocatalytic radical reactions and offering an alternative pathway for enantioselective α-alkylation of aldehydes. Other electron acceptors, like (phenylsulfonyl)-alkyl iodides, also worked well, yielding highly enantioselective products. A related photo-organocatalytic strategy was effectively extended to ketone substrate 40 using a cinchona-based primary amine catalyst 42 (Scheme 4d).25 Electron-deficient benzyl bromide 31 functioned as a suitable electron acceptor, though the reaction remained limited to cyclic ketones as chiral enamine electron-donor precursors.
 | ||
| Scheme 4 Chiral organo-photocatalyzed enantioselective (a)–(c) α-alkylation of aldehydes; (d) α-alkylation of ketones; and (e) general reaction mechanism. | ||
In 2017, Melchiorre and co-workers developed an asymmetric β-alkylation of enals using chiral iminium ions formed in situ from α,β-unsaturated aldehyde 43 and chiral amine catalysts 46 (Scheme 5a).26 Unlike enamine photocatalysis, which involves nucleophilic intermediates, iminium photocatalysis leverages electrophilic iminium ions that become strong oxidants upon excitation. The proposed mechanism involves visible-light absorption by the iminium ion, which oxidizes an alkyl trimethylsilane 44, generating radicals that undergo direct radical–radical coupling to form β-alkylated aldehyde 45 in up to 82% yield with 80% ee. This method provides selective 1,4-addition, improving upon traditional thermal iminium catalysis, which often yields a mixture of 1,2- and 1,4-addition products. Toluene 48 can reductively quench the excited-state iminium ion, enabling the formation of β-benzylated aldehyde 49 from enals generated from 47 and 50 (Scheme 5b).27 Upon oxidation, toluene's benzylic C–H bond becomes highly acidic, with an estimated pKa of −13 in CH3CN.28 Deprotonation generates a benzyl radical, which then undergoes radical–radical coupling with the β-enaminyl radical intermediate. Zn(OTf)2 was essential for this reaction, with Zn2+ facilitating iminium formation and the triflate counterion assisting in the deprotonation of the photogenerated toluene radical cation.
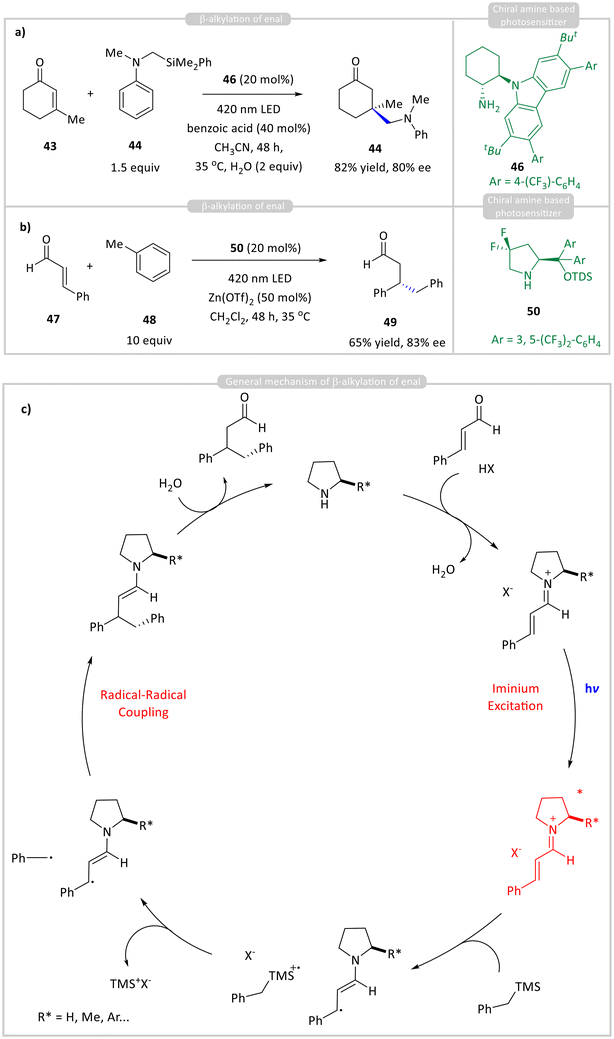 | ||
| Scheme 5 Chiral organo-photocatalyzed enantioselective (a) & (b) β-alkylation of enals; and (c) general mechanism. | ||
Scalability remains a key limitation of chiral organo-photocatalysts due to common photochemical challenges such as poor light penetration, non-uniform irradiation in batch systems, high catalyst loadings, and limited photostability under prolonged exposure. Continuous-flow photochemistry offers a promising solution by improving light distribution, heat dissipation, and reproducibility, making it more suitable for large-scale synthesis.
However, translating these reactions from batch to flow is still underexplored. Issues like catalyst solubility, adsorption or deactivation in flow channels, and the need to optimize residence time and light intensity must be addressed. Advancing robust, flow-compatible chiral organo-photocatalysts, alongside systematic reaction engineering, will be crucial for achieving scalable asymmetric synthesis.
3. Chiral metal complexes
Chiral metal complexes have emerged as powerful discrete photocatalysts for various enantioselective organic transformations, combining photocatalysis and asymmetric induction within a single catalytic framework.29,7a These complexes, typically based on transition metals such as Ru, Ir, or Cu, incorporate enantiopure ligands that create a well-defined chiral environment while simultaneously facilitating photoinduced electron transfer (SET) or energy transfer (EnT) processes. Additionally, these complexes can be generated from transition metals and achiral ligands (chiral-at-metal) as active chiral photocatalysts.30 Meggers pioneered the use of helically chiral iridium(III) polypyridyl complexes as enantioselective photocatalysts for a variety of synthetically important transformations. These catalysts can be easily prepared in stereochemically pure form using a chiral auxiliary approach and have been shown to be resistant to racemization.31 The metal centre (e.g., Ru or Rh) becomes a stereogenic centre due to the arrangement of achiral ligands in a chiral fashion. These complexes exist in Λ (lambda) or Δ (delta) configurations, and the metal's spatial arrangement imparts chirality (Fig. 3).32 These Ir/Rh complexes serve two roles simultaneously: as photocatalysts, they absorb visible light and enter an excited state; and as chiral Lewis acids, they coordinate and orient the substrate in a defined chiral environment, enabling stereocontrol.3.1 Rhodium/iridium incorporated chiral metal complexes
Meggers’ group has reported a series of photocatalytic enantioselective carbon–carbon bond formation reactions using these Ru- and Rh-based chiral-at-metal complexes with a pre-association mechanism (Fig. 2a), where the substrate binds to the chiral catalyst before the photochemical event (light excitation) (Scheme 6). This binding or pre-association plays a central role in controlling enantioselectivity. This strategy avoids issues with diffusion-controlled processes, where radicals formed freely in solution can react randomly, reducing enantioselectivity. This interaction often involves coordination to the metal centre (as a Lewis acid) or hydrogen bonding with the ligand framework (Yoon's strategy). Upon photoexcitation, the chiral metal complex bound with substrate becomes redox-active and initiates the reaction, either by single-electron transfer (SET) or energy transfer. Because the substrate is already held in a chiral pocket, the reactive intermediate (radical, cation, etc.) formed after excitation undergoes diastereoselective steps as the substrate–catalyst complex is already entity; hence, the subsequent reactions are diastereoselective, such as radical recombination, bond formation, or nucleophilic attack. Meggers’ pioneering studies revealed that Λ-51b efficiently catalyses the highly enantioselective α-alkylation of 2-acylimidazolyl ketone 53 with electron-deficient benzyl bromide 54 radical precursors, affording the α-alkylated product 55 with excellent enantioselectivity (99% ee) (Scheme 6a).33 In follow-up studies, they utilized a modified analogue, Λ-51c, to achieve the enantioselective α-trichloromethylation of N-phenyl-2-acylimidazoles 56, affording enantiopure α-trichloromethylated products 55 via the same mechanistic pathway (Scheme 6b).34 Using the same strategy, they reported the desilylative oxidation of α-silyl amines 60 using the substrate 59-bound chiral-at-metal Ir(III) complex Δ-51a as the active chiral photocatalyst (Scheme 6c).35 Chiral-at-metal Rh(III) photocatalysts exhibit superior versatility over first-generation iridium complexes. Using Λ-51a, N,N-dialkylaniline 62 was oxidized to a radical cation, which rapidly deprotonated to form an α-amino radical. Subsequent air oxidation generated an iminium ion that reacted asymmetrically with enolates from 59, affording product 63 in up to 80% yield and 95% ee (Scheme 6d).36 A photogenerated alkyl radical from Hantzsch ester 66 for asymmetric addition to α,β-unsaturated ketone derivatives 64, using the chiral-at-metal Rh(III) complex Λ-52a to obtain product 63, was successfully demonstrated by Meggers’ group (Scheme 6e).37 Furthermore, Meggers and co-workers reported the stereoselective asymmetric coupling of racemic α-chloro-imidazol-2-yl ketones 67 with N-arylglycines 68 using the chiral photocatalyst Δ-52b (Scheme 6f).38 Upon photoexcitation, the chiral-at-rhodium complex generates radicals from both substrates, which undergo enantioselective radical–radical coupling to afford β-amino ketone 69 with up to 93% ee. An asymmetric [3 + 2] cycloaddition was achieved using the chiral-at-Rh(III) photocatalyst Δ-52b, wherein cyclopropyl imidazolyl ketone 70 reacted with various alkenes and alkynes 71 to afford highly enantioselective cyclopentanes and cyclopentenes 72 (Scheme 6g).39 Through a series of experiments, the authors have proposed a plausible mechanism for the catalytic reaction. X-ray crystallographic analysis confirms that the substitutionally labile acetonitrile ligands of Λ/Δ-51/52 are replaced by the bidentate substrate while preserving the stereochemical integrity of the metal centre. In the presence of a base, deprotonation generates a chiral enolate complex. Furthermore, Stern–Volmer luminescence quenching studies, together with electrochemical measurements, suggest that this intermediate functions as the active photocatalyst, absorbing visible light to reduce the electron-deficient alkyl bromide. The resulting alkyl radical subsequently attacks the enolate within a chiral environment, affording the enantiopure product.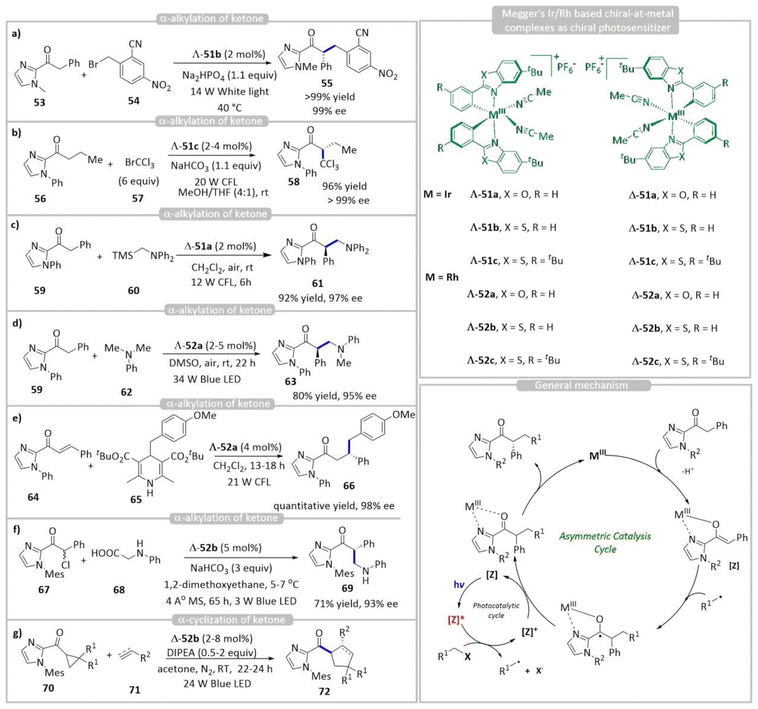 | ||
| Scheme 6 Chiral-at-metal complexes as discrete photocatalysts for asymmetric (a–g) α-alkylation of ketones. | ||
3.2 Iridium-incorporated chiral metal complexes with H bonding
Inspired by Meggers’ work, Yoon developed Ir-based chiral-at-metal complexes with hydrogen-bonding functionality through the ligand. They have followed the pre-association via hydrogen bonding. The Ir(III) complex contains ligands that can form hydrogen bonds with carbonyl or amide-containing substrates. This pre-association orients the substrate in a defined spatial arrangement within the chiral pocket of the catalyst. Upon irradiation with visible light (blue LED), the Ir(III) complex bound with the substrate is excited to its triplet MLCT (metal-to-ligand charge transfer) state. This excited state has a long lifetime and can transfer energy to the bound substrate. Unlike photoredox SET processes, Yoon's approach often uses energy transfer to excite the bound alkene substrate to its triplet state, initiating a [2 + 2] cycloaddition. The bound substrate undergoes triplet excitation while remaining hydrogen-bonded and geometrically restricted. The excited substrate reacts with another alkene or a conjugated system in a [2 + 2] fashion, forming a cyclobutane ring. Because the substrate is fixed in the chiral environment during excitation and reaction, the cycloaddition proceeds with high facial selectivity, affording enantioselective cyclobutanes (Scheme 7). Using the chiral iridium photosensitizer, Λ-73a, a photocatalytic intramolecular asymmetric [2 + 2] cycloaddition of quinolone substrate 74 generated enantioselective cycloadduct 75 with excellent yield (>99%) and enantioselectivity (90% ee) (Scheme 7a).40 Computational studies, including DFT and transient absorption spectroscopy, have revealed that π–π stacking interactions between the Ir complex and the substrate further stabilize the complex and influence enantioselectivity. They further applied this strategy to the photocatalytic asymmetric intermolecular [2 + 2] cycloaddition between 3-isopropoxyquinolone 76 and maleimide 77. Using a slightly modified chiral photocatalyst, Λ-73b, the reaction delivered cyclobutane 78 in high yield with enantioselectivity of up to 97% ee (Scheme 7b).41 Mechanistic studies revealed a rebound pathway for this intermolecular [2 + 2] photocycloaddition. While the chiral Ir complex preferentially coordinates to quinolone 76, the energy transfer is directed to the maleimide 77 partner. The resulting triplet-excited maleimide 79 then engages the bound quinolone 78 in a stereoselective fashion, affording cyclobutane 78 with excellent enantioselectivity (up to 97% ee). They next reported an enantioselective 6π photoelectrocyclization of substrates 79 bearing a C-acylimidazole unit, enabled by a single hydrogen-bonding chiral Ir(III) photosensitizer, Λ-73c (Scheme 7c).42 Key to this transformation was the rational design of a strong hydrogen-bonding interaction between the pyrazole moiety of photocatalyst Λ-73c and the imidazolyl ketone group of the substrate. This interaction effectively controlled stereoselectivity, affording indoline products 80 in good yields with excellent enantiomeric excesses (>90% ee).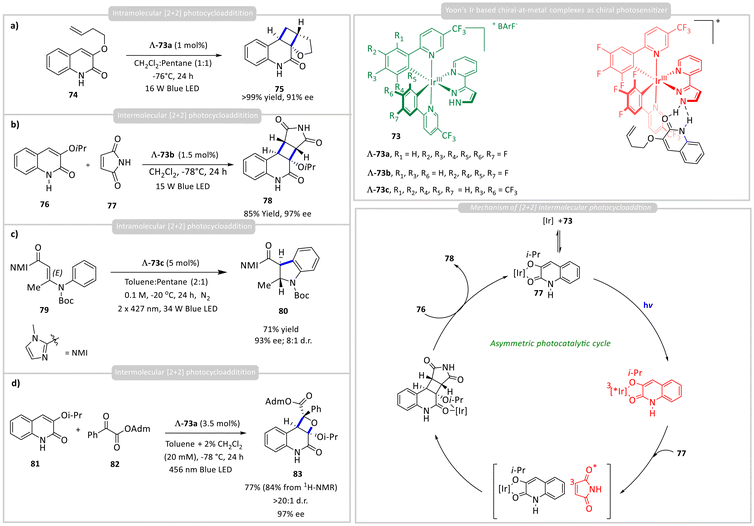 | ||
| Scheme 7 Enantioselective (a) & (c) intra- and (b) & (d) intermolecular [2 + 2] photocycloadditions with a chiral hydrogen-bonding iridium photosensitizer. | ||
3.3 Ruthenium/iridium-incorporated chiral metal complexes attached with chiral auxiliaries
A few groups have developed Ir(III)/Ru(II)-based photosensitizers integrated with chiral hydrogen-bonding ligands for asymmetric photocatalysis. For instance, Bach and co-workers introduced an iridium catalyst 86 bearing a chiral lactam-functionalized bipyridine ligand, which showed limited stereocontrol in the reductive cyclization of α-bromo carbonyls 84, yielding cyclized product 85 with low enantioselectivity (<10% ee) due to competing hydride bromination (Scheme 8a).43 However, better results were achieved in the energy-transfer-mediated rearrangement of spiroepoxide 87, producing ketone 88 with excellent yield and 29% ee (Scheme 8b). In a related strategy, Ceroni, Lombardo, and Cozzi developed a supramolecular photocatalyst 93 combining an Ir(III) sensitizer with a chiral imidazolidinone organocatalyst. This system enabled asymmetric alkylation of aldehydes 90 with bromo compound 36, achieving up to 84% yield and 70% ee of product 91 (Scheme 8c).44 Separate addition of the two components significantly reduced both the yield (57%) and enantioselectivity (59%), highlighting the importance of spatial proximity. The mechanism involves photoinduced reductive quenching of the Ir(III) complex, generating an alkyl radical that reacts with an enamine intermediate, though no definitive evidence supports a radical chain process. Ohkubo and co-workers introduced chiral substituents on bipyridine ligands to develop Ru(II) complex 95 with enhanced stereocontrol. They further demonstrated that Δ-95 catalyses the atroposelective oxidative coupling of 2-naphthol 93 to form BINOL 94 with 33% yield and 16% ee (Scheme 8d).45,46 The reaction proceeds via photoexcited Δ-95, which is oxidation by Co(acac)3, generating Ru(III), which in turn oxidizes 93 to initiate the coupling.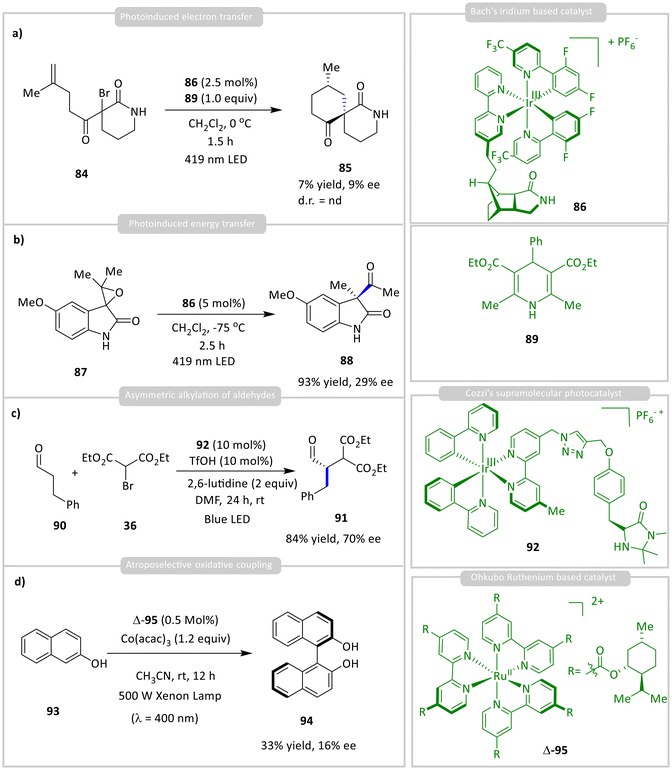 | ||
| Scheme 8 Asymmetric (a) PET, (b) energy-transfer, (c) asymmetric alkylation and (d) atroposelective oxidative coupling reactions using a chiral hydrogen-bonding ligand. | ||
3.4 Other iridium/rhodium-incorporated chiral metal complexes
A few groups have successfully applied chiral bifunctional photocatalysts that combine ionic interactions between a photosensitizer and a chiral catalyst for asymmetric C–C bond formation. Ooi and co-workers developed a system using a cationic Ir(III) photocatalyst 99 paired with a chiral hydrogen-bonding borate counteranion for an asymmetric [3 + 2] cycloaddition (Scheme 9a).47 The strained N-cyclopropylurea substrate 96 binds to the chiral borate 99, accelerating the reaction compared to the racemic Ir complex. Notably, the Ir centre's chirality had no impact on selectivity, as both Δ and Λ isomers gave similar results. The reaction proceeds via photoinduced oxidation of the borate-bound urea to form a radical-ion pair intermediate, which selectively reacts with α-substituted styrene 97 to yield cyclopentane products 98 with up to 98% yield and 93% ee. This strategy enables efficient access to α-quaternary β-amino acids, which are key intermediates for β-peptides and peptidomimetics.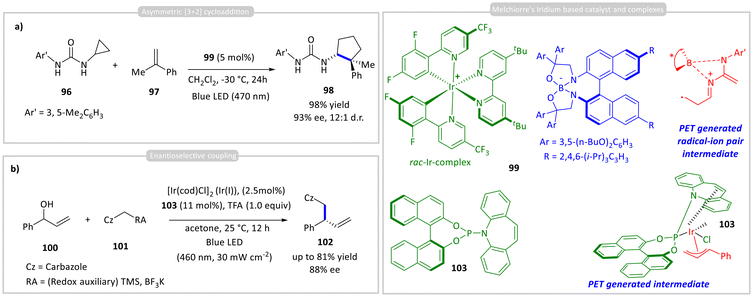 | ||
| Scheme 9 (a) Asymmetric [3 + 2] cycloaddition; (b) enantioselective coupling using iridium-chiral borate ion pairs as a chiral photocatalyst and an (η3-alyl)iridium(III) photocatalyst. | ||
Separately, Melchiorre and co-workers reported enantioselective allylic substitutions using iridium complex 103, which acts both as a privileged organometallic intermediate and as a photoredox catalyst (Scheme 9b).48 Upon excitation, it oxidizes carbazoles 101 bearing cleavable redox auxiliaries, generating stabilized radicals that undergo enantioselective coupling with an (η3-allyl)Ir(II) complex via reductive elimination, affording product 102 in high yield and enantioselectivity.
Kang and co-workers developed an asymmetric conjugate radical addition of photogenerated α-amino radicals from tetrahydroisoquinoline 105 to α,β-unsaturated 2-acylimidazoles 104 coordinated to a chiral rhodium catalyst (Λ-107), achieving excellent yields (up to 92%) and enantioselectivity (>99% ee) across various substrates (Scheme 10a).49 The reaction proceeds via ground-state complexation of 104 with Λ-107, followed by photoexcitation and single-electron reduction by 105 to generate the α-amino radical. This radical adds to the metal-bound substrate, forming a secondary radical that undergoes further reduction and protonation to yield product 106, completing the cycle. In a subsequent study, they applied Λ-107 to the asymmetric Giese addition of p-aminobenzyl radicals derived from carboxylic acids 109 to α,β-unsaturated 2-acylimidazoles 108, affording product 110 in up to 79% yield and 95% ee under visible light (Scheme 10b).50
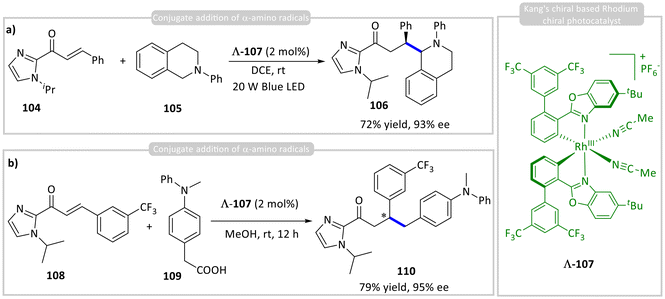 | ||
| Scheme 10 Asymmetric photocatalytic (a) & (b) conjugate addition of α-amino radicals with Michael acceptors using a photoactive rhodium-amine catalyst. | ||
3.5 Copper-incorporated chiral metal complexes
Copper-based photocatalysts have become popular materials for photoredox catalytic reactions due to their abundance, low cost, and ability to provide strong photoexcited reducing power. Key characteristics of Cu(I) photocatalysts involve the production of radical species through the reduction of organic substrates upon photoexcitation, followed by the capture of the resulting radical and/or anionic species in a rebound process. The transient Cu(III) intermediate formed can undergo reductive elimination to yield cross-coupled products. Alternatively, the Cu(II) intermediate can exchange ligands with the radical to form cross-coupled products.51In the case of chiral photocatalysis, the majority of reported photoactive chiral copper-based photocatalysts involve the use of chiral ligands for the stereocontrol of organic reactions. This is because tetrahedral copper complexes readily undergo fast ligand exchange, which prevents the exploitation of any metal-centred chirality.52 Bach and co-workers reported the photocatalytic electrocyclization of 6π system 111 using Cu(II)-chiral BOX ligand 113 (Scheme 11).53 As the substrate co-ordinates with the copper centre of the photocatalyst, a bathochromic shift is observed in the absorption spectra of the substrate. Using an in situ-generated photocatalyst with 50 mol% of Cu(ClO4)·6H2O and 60 mol% of chiral BOX ligand 113, they achieved up to 40% ee and 53% yield of the cyclized product 112.
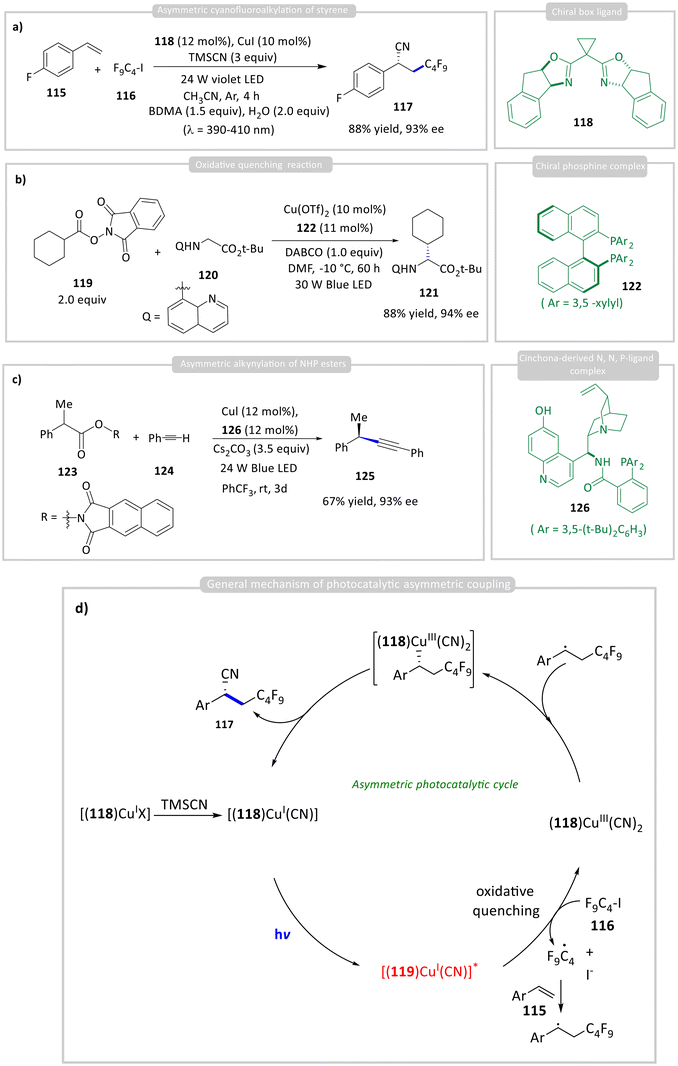
Xu, Wang, and co-workers later reported a chiral photocatalytic method for the asymmetric cyanofluoroalkylation of styrene 115 using a chiral BOX ligand 118 with CuI (Scheme 12a).54 The active photocatalyst, an in situ-generated complex of the chiral BOX ligand 118 and CuI, exhibited a high reduction potential in its excited state (−2.24 V vs. SCE). This allowed for the generation of an alkyl radical from perfluoroalkyl iodide 116 (−1.32 V vs. SCE), resulting in the formation of product 117 (Scheme 12d) in 88% yield with 93% enantiomeric excess (ee). Xu and Wang reported the enantioselective C(sp3)–H alkylation of quinolinyl-appended glycinate ester 119 using NHP ester 120 as the alkyl radical precursor (Scheme 12b).55 Coordination of 120 to a chiral phosphine-Cu(I) complex generates the photocatalyst in situ, while oxidative quenching by 119 furnishes the alkyl radical. The optimized reaction delivered the cross-coupled product 121 in 88% yield and 94% ee. Similarly, Liu developed an asymmetric alkynylation of NHP esters 123 with a Cu(I)-acetylide complex bearing cinchona-derived N,N,P-ligand 126 (Scheme 12c).56 The use of naphthyl NHP ester 123 was crucial, as it suppressed Glaser homocoupling of alkyne 124 and radical dimerization of 123, likely by slowing quenching of the excited Cu complex relative to standard NHP esters. Control experiments showed that without ligand 126, only 10% of product 125 was formed, highlighting the ligand's key role in enhancing photocatalytic efficiency. Unfortunately, the homobenzylic substrates delivered racemic coupling products 125a & 125b.
Zhang and co-workers developed another chiral photocatalyst, and this was applied for enantioselective dual carbofunctionalization of styrene derivative 14 (Scheme 13a).57 From the UV-vis experiments, they observed that the photocatalyst is generated through the aggregation of chiral t-Bu-BOPA ligand 130 and a nucleophile from alkyne 126 to form a Cu(I)-acetylide species, leading to the formation of enantioselective products 129 in up to 71% yield with 97% ee (Scheme 13b). The co-ordination of alkyl halide 128 was confirmed by steady-state luminescence quenching studies.
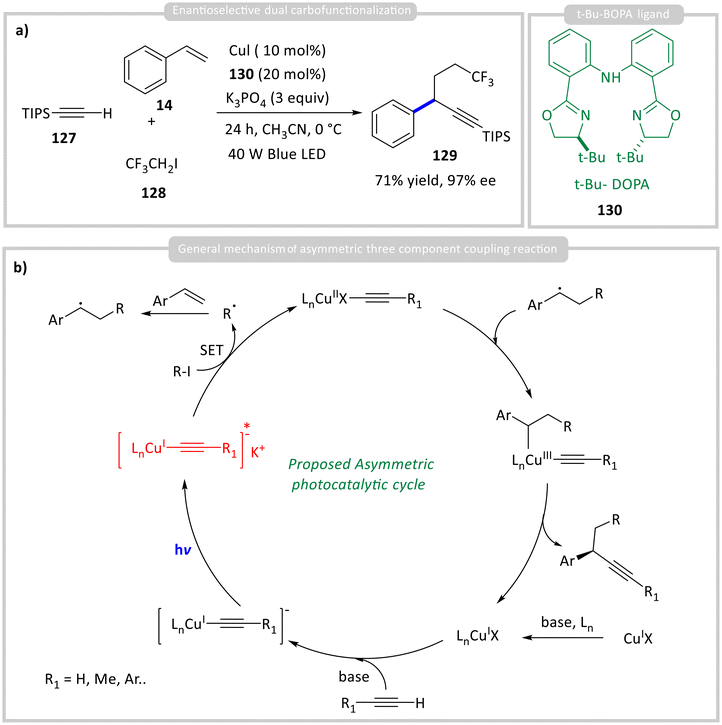 | ||
| Scheme 13 Photocatalytic asymmetric (a) dual carbofunctionalization reaction and (b) general mechanism. | ||
Gong and co-workers developed a Cu(II) complex with ligands 134/138 as chiral photocatalysts. This system was used for the photocatalytic asymmetric alkylation of imines 133/135 with 132/135, producing enantioselective products 133/147 containing tetrasubstituted carbon centres in yields of up to 96% with an enantiomeric excess (ee) of 98% of 134 and 77% yield and 93% ee of 137, respectively (Scheme 14).58 The mechanism involves photoexcited ligand-to-metal charge transfer (LMCT), which generates an alkyl radical. This radical then undergoes nucleophilic addition to the coordinated imine substrate, forming a nitrogen-centred radical stabilized by Cu(II), which is subsequently reduced by the Cu(I) complex. It is worth noting that the alkyl-substituted aldimine exhibited low reactivity under the standard reaction conditions. The desired product 137a was obtained in 73% yield with significantly reduced enantioselectivity (19% ee) at an elevated temperature of 25 °C over 40 h. Furthermore, a bioactive dihydroquinoxazoline 137b was synthesized in 74% yield with 91% ee via a Pd-catalyzed intramolecular cross-coupling reaction of the corresponding enantiopure intermediate.
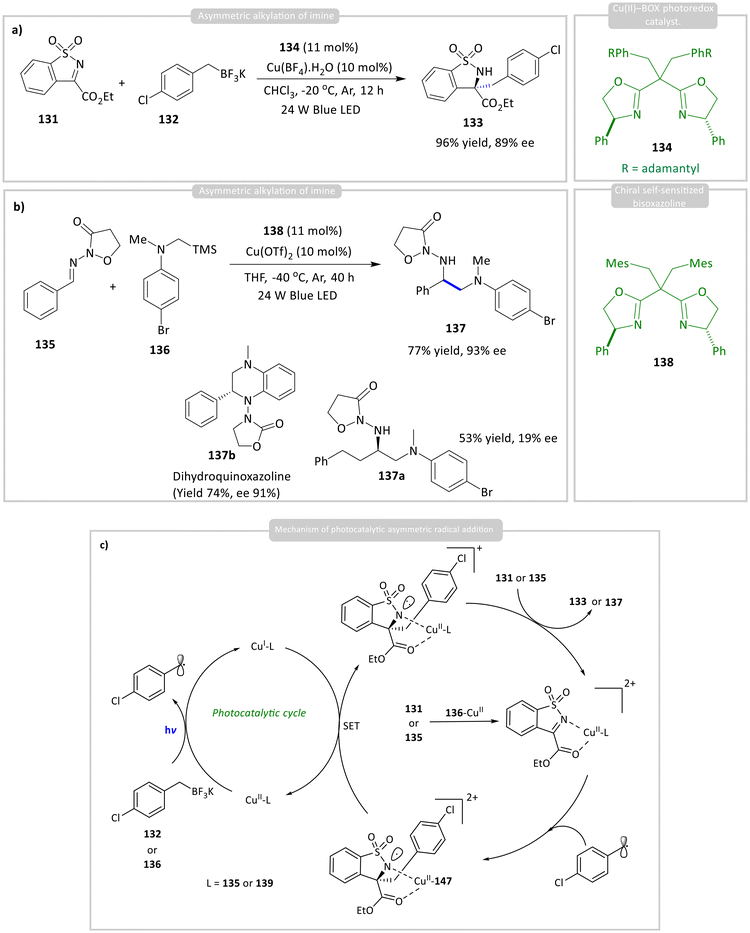 | ||
| Scheme 14 Photocatalytic asymmetric (a) & (b) alkylation of imines; (c) general mechanism of the reaction using a chiral Cu(I)-BOX photocatalyst. | ||
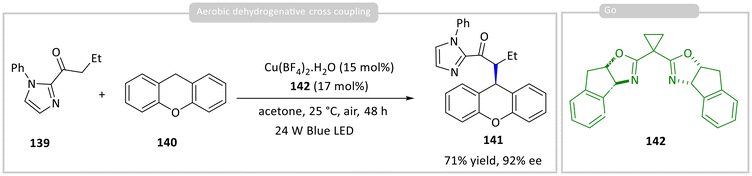
In a subsequent study, Gong and co-workers applied the same strategy to the asymmetric cross-coupling of 2-acylimidazole 139 with xanthene derivatives 140, using O2 as the terminal oxidant to afford product 141 in up to 71% yield and 92% ee (Scheme 15).59 Mechanistic insights from electrochemical and spectroscopic studies revealed that the chiral photocatalyst, a Cu(II)–BOX complex formed from Cu(BF4)2·H2O and chiral ligand 142, initiates the reaction through oxidation of xanthene 140. Moreover, binding of the prochiral substrate 139 by the Cu(II)–BOX complex provides the chiral environment necessary for enantioinduction for the synthesis of 141.
3.6 Nickel-incorporated chiral metal complexes
Shen and co-workers developed a bifunctional Ni(II)–DBFOX complex, prepared from Ni(ClO4)2·6H2O and the chiral DBFOX ligand 146. This complex acts simultaneously as a photoredox catalyst and a chiral Lewis acid, enabling the asymmetric addition of α-amino radicals derived from 144 to α,β-unsaturated compounds 143, a de-silylative photo-oxidation pathway that generates the key α-amino radical intermediate, affording product 145 (Scheme 16).60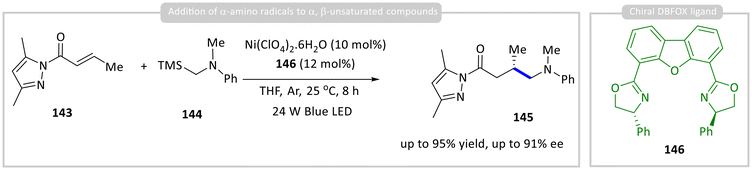 | ||
| Scheme 16 Photocatalytic asymmetric addition of α-amino radicals to α,β-unsaturated carbonyl compounds. | ||
3.7 Lanthanide-incorporated chiral metal complexes
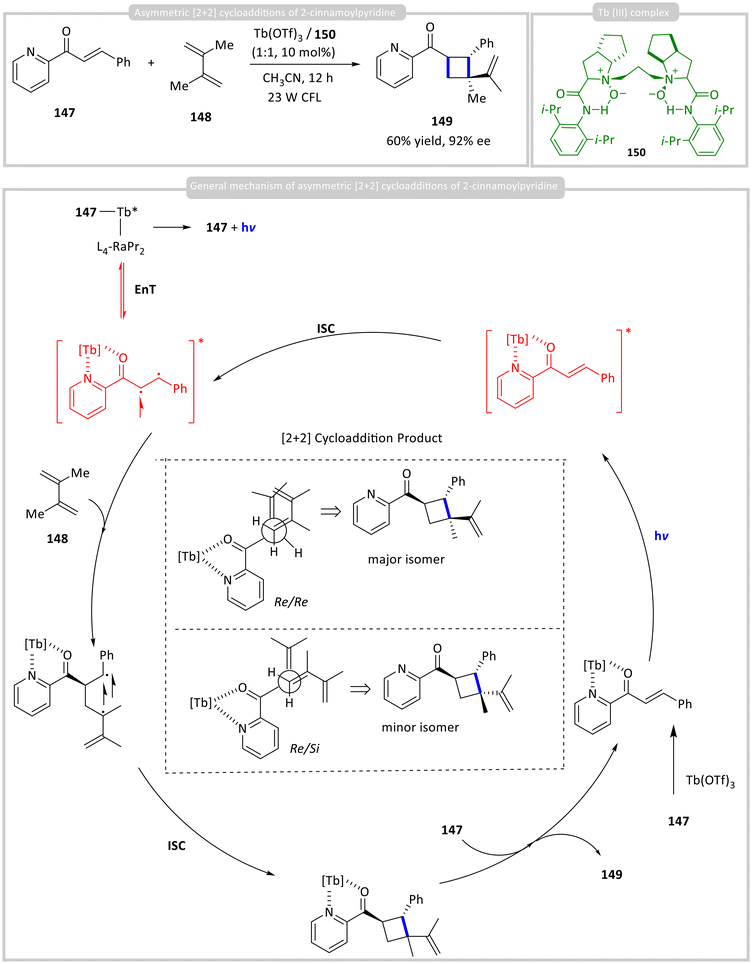
Only a limited number of chiral photocatalysts based on lanthanide coordination complexes have been reported for asymmetric photocatalytic carbon–carbon bond-forming reactions. Liu, Feng, and co-workers developed such a system using a chiral Tb(III) complex, prepared from Tb(OTf)3 and chiral ligand 150, as a chiral photocatalyst for the asymmetric [2 + 2] cycloaddition of 2-cinnamoylpyridine 147 with dialkene 148. This method delivered cycloadduct 149 in up to 92% enantiomeric excess (ee) and 60% yield (Scheme 17).61 The formation of the chiral Tb(III) complex was confirmed by UV-vis absorption spectroscopy. The heavy-atom effect of Tb(III) promotes intersystem crossing (ISC), enabling efficient harvesting of the triplet state and driving the bimolecular reaction under photoexcitation. Single-crystal X-ray analysis of the in situ-generated metal complex revealed an eight-coordinate, square-antiprismatic geometry. The proposed mechanism involves pre-association of the chiral Tb(III) complex with substrate 147 in the ground state, followed by excitation. After intersystem crossing, substrate 148 replaces product 149 in the coordination sphere, thereby completing the catalytic cycle.3.8 Cobalt-incorporated chiral metal complexes
Lu, Xiao, and co-workers reported the development of a chiral octahedral cobalt(II) complex Δ-154 from Earth-abundant cobalt(II) and bisoxazoline-type (BOX-like) N,N-bidentate chelating N4 ligands. The chiral Co(II) complex Δ-154 was demonstrated to function as a highly efficient catalyst for visible-light-induced conjugate addition reactions. Enone 151 undergoes asymmetric addition with acyl radicals that are generated in situ from the corresponding Hantzsch esters 152. The catalytic cycle proceeds under visible-light irradiation, where the Co(II) complex Δ-154 not only enables photoredox activation but also imparts stereocontrol through its chiral coordination environment to obtain enantioselective 1,4-dicarbonyl product 153 in >99% yields with 90% ee (Scheme 18).62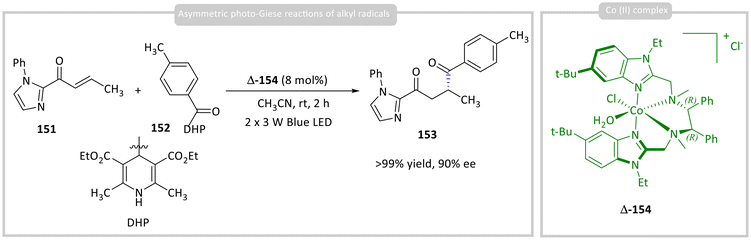 | ||
| Scheme 18 Photocatalytic asymmetric photo-Giese reactions of alkyl radicals using a chiral Co(II) photocatalyst. | ||
4. Summary and outlook
The field of chiral photocatalysis has witnessed remarkable growth in recent years, particularly in enabling asymmetric carbon–carbon (C–C) bond-forming reactions under mild and sustainable conditions. These catalysts control their well-defined chiral environments to promote stereo-controlled C–C bond formation under mild photochemical conditions, reducing dependence on harsh reagents or elaborate reaction setups. Although many chiral photocatalysts were initially designed for thermal asymmetric transformations, their successful application in excited-state photocatalytic reactions has demonstrated remarkable control over stereochemistry, particularly in C–C bond-forming processes.After these breakthroughs, a variety of chiral photocatalytic systems incorporating chromophores or chiral ligands have been developed specifically for reactions that are challenging or inaccessible with ground-state catalysis. Notably, Bach's and Meggers’ groups have made significant contributions through the development of chiral organo-photocatalysts and chiral-at-metal complexes, which act as photoactive chiral molecules in enantioselective C–C bond-forming reactions. In parallel, other strategies have employed dual catalysts, where a chiral catalyst is attached to a photocatalyst to guide asymmetric induction during C–C coupling. Yoon's group has also advanced the field with hydrogen-bonding chiral-at-metal photocatalysts, enabling highly stereoselective cycloadditions and related C–C bond formations. Despite differences in mechanistic pathways and structural designs, a key aspect underlying successful enantioselective C–C bond formation is the pre-association of substrates with the chiral catalyst in the ground state, which facilitates chiral induction upon photoexcitation and helps suppress racemic background reactions. The continued evolution of photoactive chiral systems has massively expanded the synthetic approaches for constructing complex, enantioselective carbon frameworks. As the field advances, chiral photocatalysts are expected to remain central to the development of selective and scalable synthetic methods for asymmetric C–C bond formation.
In terms of atom economy and reaction efficiency, both organic and metal-based photocatalysts effectively promote bond formation under light irradiation. Organic photocatalysts often provide high selectivity with minimal side reactions. In contrast, metal-based photocatalysts, particularly transition-metal complexes, though relatively expensive, possess well-defined redox properties and efficiently mediate single-electron transfer processes, enabling a broad substrate scope and high enantioselectivity in asymmetric transformations. From a sustainability perspective, organic photocatalysts are generally considered more environmentally benign due to their metal-free nature, lower toxicity, and compatibility with visible light and green solvents. Regarding practical implementation and scalability, organic photocatalysts typically operate under mild conditions, improving energy efficiency and reducing operational costs. While metal-based systems offer superior catalytic control, challenges associated with catalyst recovery and recycling may limit their industrial applicability. In this context, metal-free and immobilized catalytic systems, such as those based on chiral COFs, offer advantages in recyclability and simplified purification, making them attractive for scalable processes.
Future efforts will focus on enhancing yields, enantiomeric excess (ee), and operational stability, with particular emphasis on designing robust, tuneable photocatalysts capable of long-term use under visible light. These developments will be critical for enabling industrial-scale applications, especially in pharmaceutical synthesis, where precise control over stereochemistry is essential. Moreover, chiral photocatalysts are anticipated to impact materials science, facilitating the creation of enantioselective polymers and chiral surfaces for use in sensors, optoelectronics, and other advanced technologies. Improving photocatalyst stability and resistance to photodegradation will be crucial for unlocking the full potential of asymmetric C–C bond formation in photocatalytic systems.
Author contributions
AD planned, designed and overall supervised this review article. VKS and AHI gathered references and prepared the schemes and structures.Conflicts of interest
The authors declare no conflict of interest.Data availability
No primary research results, software or code have been included, and no new data were generated or analysed as part of this review.Acknowledgements
VKS and AHI gratefully acknowledge the Department of Chemistry, SRM Institute of Science and Technology, Kattankulathur, Tamil Nadu, India, for financial support through an Institute Doctoral Fellowship. AD sincerely acknowledges SRMIST for the seed grant (no. SRMIST/R/AR(A/SERI2024/174/14-342)), the Anusandhan National Research Foundation (ANRF-ECRG, Grant No. ANRF/ECRG/2024/003299/CS), and the Royal Society-Newton International Alumni Fellowship (AL\24100051) for financial support.References
- (a) E. J. Corey and X.-M. Cheng, The Logic of Chemical Synthesis, John Wiley & Sons, New York, 1989 Search PubMed; (b) D. Ravelli, S. Protti and M. Fagnoni, Chem. Rev., 2016, 116, 9850–9913 CrossRef CAS PubMed; (c) V. Saini, B. J. Stokes and M. S. Sigman, Angew. Chem., Int. Ed., 2013, 52, 11206–11220 CrossRef CAS PubMed; (d) N. G. Schmidt, E. Eger and W. Kroutil, ACS Catal., 2016, 6, 4286–4311 CrossRef CAS PubMed; (e) S. Van de Vyver and Y. Roman-Leshkov, Angew. Chem., Int. Ed., 2015, 54, 12554–12561 CrossRef CAS PubMed; (f) Q. Liu, R. Jackstell and M. Beller, Angew. Chem., Int. Ed., 2013, 52, 13871–13873 CrossRef CAS PubMed; (g) T. Tsubogo, T. Ishiwata and S. Kobayashi, Angew. Chem., Int. Ed., 2013, 52, 6590–6604 CrossRef CAS PubMed; (h) H. Schönherr and T. Cernak, Angew. Chem., Int. Ed., 2013, 52, 12256–12267 CrossRef PubMed; (i) S. H. Cho, J. Y. Kim, J. Kwak and S. Chang, Chem. Soc. Rev., 2011, 40, 5068–5083 RSC; (j) F. Monnier and M. Taillefer, Angew. Chem., Int. Ed., 2009, 48, 6954–6971 CrossRef CAS PubMed; (k) A. Alexakis, J.-E. Bäckvall, N. Krause, O. Pàmies and M. Diéguez, Chem. Rev., 2008, 108, 2796–2823 CrossRef CAS PubMed; (l) B. M. Trost, F. D. Toste and A. B. Pinkerton, Chem. Rev., 2001, 101, 2067–2096 CrossRef CAS PubMed; (m) N. Miyaura and A. Suzuki, Chem. Rev., 1995, 95, 2457–2483 CrossRef CAS; (n) R. Franzén, Can. J. Chem., 2000, 78, 957–962 CrossRef; (o) R. Franzén and Y. Xu, Can. J. Chem., 2005, 83, 266–272 CrossRef; (p) L. Yin and J. Liebscher, Chem. Rev., 2006, 107, 133–173 CrossRef; (q) J. Barluenga, M. Tomás-Gamasa, F. Aznar and C. Valdés, Nat. Chem., 2009, 1, 494–499 CrossRef CAS PubMed; (r) P. Clapés, W.-D. Fessner, G. A. Sprenger and A. K. Samland, Curr. Opin. Chem. Biol., 2010, 14, 154–167 CrossRef PubMed; (s) D. G. Gillingham, P. Stallforth, A. Adibekian, P. H. Seeberger and D. Hilvert, Nat. Chem., 2010, 2, 102–105 CrossRef CAS PubMed; (t) J. Soloducho, J. Cabaj, K. Idzik, A. Nowakowska-Oleksy, A. Swist and M. Lapkowski, Curr. Org. Chem., 2010, 14, 1234–1245 CrossRef CAS; (u) V. Resch, J. H. Schrittwieser, E. Siirola and W. Kroutil, Curr. Opin. Biotechnol., 2011, 22, 793–799 CrossRef CAS PubMed; (v) M. Pérez, M. Fañanás-Mastral, P. H. Bos, A. Rudolph, S. R. Harutyunyan and B. L. Feringa, Nat. Chem., 2011, 3, 377–381 CrossRef PubMed; (w) A. Bhunia, R. S. Yetra and A. T. Biju, Chem. Soc. Rev., 2012, 41, 3140–3152 RSC; (x) G. Brahmachari, Chem. Rec., 2016, 16, 98–123 CrossRef CAS PubMed.
- (a) E. Negishi, Angew. Chem., Int. Ed., 2011, 50, 6738–6764 CrossRef CAS PubMed; (b) C. C. C. Johansson Seechurn, M. O. Kitching, T. J. Colacot and V. Snieckus, Angew. Chem., Int. Ed., 2012, 51, 5062–5085 CrossRef CAS PubMed; (c) A. H. Cherney, N. T. Kadunce and S. E. Reisman, Chem. Rev., 2015, 115, 9587–9652 CrossRef CAS PubMed; (d) K. Faber, W.-D. Fessner and N. J. Turner, Biocatalysis in Organic Synthesis, in Science of Synthesis, Georg Thieme, Stuttgart, 2015, vol. 1–3 Search PubMed; (e) H. Lechner, D. Pressnitz and W. Kroutil, Biotechnol. Adv., 2015, 33, 457–480 CrossRef CAS PubMed; (f) Y.-F. Miao, M. Rahimi, E. M. Geertsema and G. J. Poelarends, Curr. Opin. Chem. Biol., 2015, 25, 115–123 CrossRef CAS PubMed; (g) L. A. Wessjohann, J. Keim, B. Weigel and M. Dippe, Curr. Opin. Chem. Biol., 2013, 17, 229–235 CrossRef CAS PubMed; (h) K. Fesko and M. Gruber-Khadjawi, ChemCatChem, 2013, 5, 1248–1272 CrossRef CAS; (i) M. Müller, Adv. Synth. Catal., 2012, 354, 3161–3174 CrossRef; (j) K. Faber, Biotransformations in Organic Chemistry, Springer, Heidelberg, 6th edn, 2011 CrossRef; (k) M. Brovetto, D. Gamenara, P. S. Mendez and G. A. Seoane, Chem. Rev., 2011, 111, 4346–4403 CrossRef CAS PubMed; (l) G. Brahmachari, RSC Adv., 2016, 6, 64676–64725 RSC.
- (a) C. K. Prier, D. A. Rankic and D. W. C. MacMillan, Chem. Rev., 2013, 113, 5322–5363 CrossRef CAS PubMed; (b) D. Staveness, I. Bosque and C. R. J. Stephenson, Acc. Chem. Res., 2016, 49, 2295–2306 CrossRef CAS PubMed; (c) F. Strieth-Kalthoff, M. J. James, M. Teders, L. Pitzera and F. Glorius, Chem. Soc. Rev., 2018, 47, 7190–7202 RSC; (d) J. Xuan and W.-J. Xiao, Angew. Chem., Int. Ed., 2012, 51, 6828–6838 CrossRef CAS; (e) L. Shi and W. Xia, Chem. Soc. Rev., 2012, 41, 7687–7697 RSC; (f) G. Ciamician, Science, 1912, 36, 385–394 CrossRef CAS PubMed; (g) H. D. Roth, Angew. Chem., Int. Ed. Engl., 1989, 28, 1193–1207 CrossRef; (h) Y. Inoue and V. Ramamurthy, Chiral Photochemistry, Marcel Dekker, 2004, vol. 11 CrossRef; (i) A. Das, T. Banerjee and K. Hanson, Chem. Commun., 2016, 52, 1350–1353 RSC; (j) Visible Light Photocatalysis in Organic Chemistry, ed. C. R. J. Stephenson, D. W. C. MacMillan and T. P. Yoon, Wiley-VCH, Weinheim, 2018 Search PubMed; (k) M. H. Shaw, J. Twilton and D. W. C. MacMillan, J. Org. Chem., 2016, 81, 6898–6926 CrossRef CAS; (l) V. Ramamurthy and J. Sivaguru, Chem. Rev., 2016, 116, 9914–9993 CrossRef CAS PubMed; (m) F. Mohamadpour and A. M. Amani, RSC Adv., 2024, 14, 20609–20615 RSC; (n) A. Albini and M. Fagnoni, Green Chem., 2004, 6, 1–6 RSC; (o) N. Hoffmann, Chem. Rev., 2008, 108, 1052–1103 CrossRef CAS PubMed; (p) A. Houmam, Chem. Rev., 2008, 108, 2180–2237 CrossRef CAS PubMed; (q) J. M. R. Narayanam and C. R. J. Stephenson, Chem. Soc. Rev., 2011, 40, 102–113 RSC; (r) D. M. Schultz and T. P. Yoon, Science, 2014, 343, 1239176 CrossRef PubMed; (s) N. A. Romero and D. A. Nicewicz, Chem. Rev., 2016, 116, 10075–10166 CrossRef CAS PubMed; (t) K. L. Skubi, T. R. Blum and T. P. Yoon, Chem. Rev., 2016, 116, 10035–10074 CrossRef CAS PubMed; (u) J. Twilton, C. Le, P. Zhang, M. H. Shaw, R. W. Evans and D. W. C. MacMillan, Nat. Rev. Chem., 2017, 1, 0052 CrossRef CAS; (v) J. K. Matsui, S. B. Lang, D. R. Heitz and G. A. Molander, ACS Catal., 2017, 7, 2563–2575 CrossRef CAS PubMed; (w) P. Chuentragool, D. Kurandina and V. Gevorgyan, Angew. Chem., Int. Ed., 2019, 58, 11586–11590 CrossRef CAS PubMed; (x) R. Kancherla, K. Muralirajan, S. Arunachalam and M. Rueping, Trends Chem., 2019, 1, 510–523 CrossRef CAS; (y) D. Kurandina, P. Chuentragool and V. Gevorgyan, Synthesis, 2019, 985–999 CAS; (z) W.-J. Zhou, G.-M. Cao, Z.-P. Zhang and D.-G. Yu, Chem. Lett., 2019, 48, 181–184 CrossRef CAS.
- (a) Y. Inoue, Chem. Rev., 1992, 92, 741–770 CrossRef CAS; (b) R. Brimioulle, D. Lenhart, M. M. Maturi and T. Bach, Angew. Chem., Int. Ed., 2015, 54, 3872–3890 CrossRef CAS PubMed; (c) B. S. Green, A. T. Hagler, Y. Rabinsohn and M. Rejtő, Isr. J. Chem., 1976, 15, 124–130 CrossRef; (d) L. M. Tolbert and M. B. Ali, J. Am. Chem. Soc., 1982, 104, 1742–1744 CrossRef CAS; (e) A. Joy, J. R. Scheffer and V. Ramamurthy, Org. Lett., 2000, 2, 119–121 CrossRef CAS PubMed; (f) M. Nishijima, T. Wada, T. Mori, T. C. S. Pace, C. Bohne and Y. Inoue, J. Am. Chem. Soc., 2007, 129, 3478–3479 CrossRef CAS; (g) L. Addadi, J. V. Mil and M. Lahav, J. Am. Chem. Soc., 1982, 104, 3422–3429 CrossRef CAS; (h) S. V. Evans, M. G. Garibay, N. Omkaram, J. R. Scheffer, J. Trotter and F. Wireko, J. Am. Chem. Soc., 1986, 108, 5648–5650 CrossRef CAS; (i) A. Das, S. Ayad and K. Hanson, Org. Lett., 2016, 18, 5416–5419 CrossRef CAS PubMed; (j) Y. Li, Z. Huangfu, Y. Li, X. Song, Y. Duan, Y. Zhang and X. Li, ACS Catal., 2025, 15, 7543–7577 CrossRef CAS.
- (a) Y. Wang, E. A. Bazan-Bergamino and M.-Y. Ngai, ChemCatChem, 2022, 14, e202101292 CrossRef PubMed; (b) C. Wang and Z. Lu, Org. Chem. Front., 2015, 2, 179–190 RSC; (c) A. F. Garrido-Castro, M. C. Maestro and J. Alemán, Tetrahedron Lett., 2018, 59, 1286–1289 CrossRef CAS; (d) M. Silvi and P. Melchiorre, Nature, 2018, 554, 41–49 CrossRef CAS PubMed; (e) Y.-Q. Zou, F. M. Hoermann and T. Bach, Chem. Soc. Rev., 2018, 47, 278–290 RSC; (f) X. Jiang, X. Han, F.-D. Lu, L.-Q. Lu, Z. Zuo and W.-J. Xiao, CCS Chem., 2025, 7, 1567–1602 CrossRef CAS.
- (a) S. J. Chapman, W. B. Swords, C. M. Le, I. A. Guzei, F. D. Toste and T. P. Yoon, J. Am. Chem. Soc., 2022, 144, 4206–4213 CrossRef CAS PubMed; (b) Z. Zuo, H. Cong, W. Li, J. Choi, G. C. Fu and D. W. C. MacMillan, J. Am. Chem. Soc., 2016, 138, 1832–1835 CrossRef CAS; (c) E. R. Welin, A. A. Warkentin, J. C. Conrad and D. W. C. MacMillan, Angew. Chem., Int. Ed., 2015, 54, 9668–9672 CrossRef CAS PubMed; (d) J. A. Terrett, M. D. Clift and D. W. C. MacMillan, J. Am. Chem. Soc., 2014, 136, 6858–6861 CrossRef CAS PubMed; (e) S. Qian, T. M. Lazarus and D. A. Nicewicz, J. Am. Chem. Soc., 2023, 145, 18247–18252 CrossRef CAS PubMed; (f) J. B. McManus, J. D. Griffin, A. R. White and D. A. Nicewicz, J. Am. Chem. Soc., 2020, 142, 103 CrossRef PubMed; (g) C. Prentice, J. Morrisson, A. D. Smith and E. Zysman-Colman, Beilstein J. Org. Chem., 2020, 16, 2363–2441 CrossRef PubMed; (h) Y. Li, Z. Huangfu, Y. Li, X. Song, Y. Duan, Y. Zhang and X. Li, ACS Catal., 2025, 15, 7543–7577 CrossRef CAS; (i) B.-C. Hong, Org. Biomol. Chem., 2020, 18, 4298–4353 RSC; (j) C. Jiang, W. Chen, W.-H. Zheng and H. Lu, Org. Biomol. Chem., 2019, 17, 8673–8689 RSC.
- (a) A. J. David, R. Balaji and A. Das, Adv. Synth. Catal., 2025, 367, e202401543 CrossRef; (b) E. Studer, S. Mandal, T. Stünkel and R. Gilmour, Angew. Chem., Int. Ed., 2025, 64, e202513320 CrossRef CAS; (c) M. J. Genzink, J. B. Kidd, W. B. Swords and T. P. Yoon, Chem. Rev., 2022, 122, 1654–1716 CrossRef CAS PubMed; (d) J. Soika, C. Onneken, T. Morack and R. Gilmour, Acc. Chem. Res., 2025, 58, 1710–1723 CrossRef CAS PubMed.
- (a) G. E. M. Crisenza, D. Mazzarella and P. Melchiorre, J. Am. Chem. Soc., 2020, 142, 5461–5476 CrossRef CAS PubMed; (b) C. G. S. Lima, T. M. Lima, M. Duarte, I. D. Jurberg and M. W. Paixão, ACS Catal., 2016, 6, 1389–1407 CrossRef CAS.
- (a) W. Ding, L.-Q. Lu, Q.-Q. Zhou, Y. Wei, J.-R. Chen and W.-J. Xiao, J. Am. Chem. Soc., 2017, 139, 63–66 CrossRef CAS PubMed; (b) A. B. Rolka, N. Archipowa, R. J. Kutta, B. König and F. D. Toste, J. Org. Chem., 2023, 88, 6509–6522 CrossRef CAS PubMed; (c) Y. Zhang, Y. Sun, X. Ren, J. Hu, H. Yu, J. Liu, H. Huang and J. Han, Angew. Chem., Int. Ed., 2025, 64, e202416221 CrossRef CAS PubMed; (d) B.-J. Bian, L. Yang, L.-X. Qiao, Q. Zhang and W. He, RSC Adv., 2025, 15, 2874–2880 RSC; (e) J. Lyu, M. Leone, A. Claraz, C. Allain, L. Neuville and G. Masson, RSC Adv., 2021, 11, 36663–36669 RSC; (f) A. B. Rolka and B. König, Nat. Synth., 2023, 2, 913–925 CrossRef CAS.
- C. Jiang, W. Chen, W.-H. Zheng and H. Lu, Org. Biomol. Chem., 2019, 17, 8673–8689 RSC.
- (a) N. Holmberg-Douglas, Y. Choi, B. Aquila, H. Huynh and D. A. Nicewicz, ACS Catal., 2021, 11, 3153–3158 CrossRef CAS PubMed; (b) V. A. Pistritto, M. E. Schutzbach-Horton and D. A. Nicewicz, J. Am. Chem. Soc., 2020, 142, 17187–17194 CrossRef CAS PubMed.
- (a) R. Maeda, T. Wada, T. Mori, S. Kono, N. Kanomata and Y. Inoue, J. Am. Chem. Soc., 2011, 133, 10379–10381 CrossRef CAS PubMed; (b) R. Hoffmann and Y. Inoue, J. Am. Chem. Soc., 1999, 121, 10702–10710 CrossRef CAS; (c) Y. Inoue, H. Shimoyama, N. Yamasaki and A. Tai, Chem. Lett., 1991, 20, 593–596 CrossRef CAS; (d) Y. Inoue, N. Yamasaki, H. Shimoyama and A. Tai, J. Org. Chem., 1993, 58, 1785–1793 CrossRef CAS.
- J.-I. Kim and G. B. Schuster, J. Am. Chem. Soc., 1990, 112, 9635–9637 CrossRef CAS.
- (a) T. Bach, H. Bergmann and K. Harms, Angew. Chem., Int. Ed., 2000, 39, 2302–2304 CrossRef CAS; (b) T. Bach, H. Bergmann, B. Grosch, K. Harms and E. Herdtweck, Synthesis, 2001, 1395–1405 CrossRef CAS.
- C. Müller, A. Bauer and T. Bach, Angew. Chem., Int. Ed., 2009, 48, 6640–6642 CrossRef PubMed.
- (a) A. Bauer, F. Westkämper, S. Grimme and T. Bach, Nature, 2005, 436, 1139–1140 CrossRef CAS PubMed; (b) Y. Inoue, Nature, 2005, 436, 1099–1100 CrossRef CAS PubMed.
- X. Li, J. Großkopf, C. Jandl and T. Bach, Angew. Chem., Int. Ed., 2021, 60, 2684–2688 CrossRef CAS PubMed.
- A. Tröster, R. Alonso, A. Bauer and T. Bach, J. Am. Chem. Soc., 2016, 138, 7808–7811 CrossRef PubMed.
- M. M. Maturi and T. Bach, Angew. Chem., Int. Ed., 2014, 53, 7661–7664 CrossRef CAS PubMed.
- F. Pecho, Y.-Q. Zou, J. Gramüller, T. Mori, S. M. Huber, A. Bauer, R. M. Gschwind and T. Bach, Chem. – Eur. J., 2020, 26, 5190–5194 CrossRef CAS PubMed.
- F. Pecho, Y. Sempere, J. Gramüller, F. M. Hörmann, R. M. Gschwind and T. Bach, J. Am. Chem. Soc., 2021, 143, 9350–9354 CrossRef CAS PubMed.
- E. Arceo, I. D. Jurberg, Á. Álvarez-Fernández and P. Melchiorre, Nat. Chem., 2013, 5, 750–756 CrossRef CAS PubMed.
- M. Silvi, E. Arceo, I. D. Jurberg, C. Cassani and P. Melchiorre, J. Am. Chem. Soc., 2015, 137, 6120–6123 CrossRef CAS PubMed.
- T. Rigotti, A. Casado-Sánchez, S. Cabrera, R. Pérez-Ruiz, M. Liras, V. A. de la Peña O'Shea and J. Alemán, ACS Catal., 2018, 8, 5928–5940 CrossRef CAS.
- E. Arceo, A. Bahamonde, G. Bergonzini and P. Melchiorre, Chem. Sci., 2014, 5, 2438–2442 RSC.
- Z.-Y. Cao, T. Ghosh and P. Melchiorre, Nat. Commun., 2018, 9, 3274 CrossRef PubMed.
- D. Mazzarella, G. E. M. Crisenza and P. Melchiorre, J. Am. Chem. Soc., 2018, 140, 8439–8443 CrossRef CAS PubMed.
- A. M. Nicholas and D. R. Arnold, Can. J. Chem., 1982, 60, 2165–2179 CrossRef CAS.
- (a) A. G. Amador and T. P. Yoon, Angew. Chem., Int. Ed., 2016, 55, 2304–2306 CrossRef CAS PubMed.
- (a) L. Gong, L.-A. Chen and E. Meggers, Angew. Chem., Int. Ed., 2014, 53, 10865–10874 CrossRef PubMed; (b) L. Zhang and E. Meggers, Chem. – Asian J., 2017, 12, 2335–2342 CrossRef CAS PubMed.
- (a) E. Meggers, Chem. – Eur. J., 2010, 16, 752–758 CrossRef CAS PubMed; (b) J. Ma, X. Zhang, X. Huang, S. Luo and E. Meggers, Nat. Protoc., 2018, 13, 605–632 CrossRef CAS PubMed.
- (a) A. Werner, Ber. Dtsch. Chem. Ges., 1911, 44, 1887–1898 CrossRef CAS; (b) S. Herrero and M. A. Usón, J. Chem. Educ., 1995, 72, 1065 CrossRef CAS; (c) Y. Zhang, H. Han, Z. Wei, M. Zulqarnain, T. Chang, Y.-S. Chen and E. V. Dikarev, Inorg. Chem., 2025, 64, 13318–13328 CrossRef CAS PubMed.
- H. Huo, X. Shen, C. Wang, L. Zhang, P. Röse, L. A. Chen, K. Harms, M. Marsch, G. Hilt and E. Meggers, Nature, 2014, 515, 100–103 CrossRef CAS PubMed.
- H. Huo, X. Huang, X. Shen, K. Harms and E. Meggers, Synlett, 2016, 749–753 CAS.
- C. Wang, Y. Zheng, H. Huo, P. Röse, L. Zhang, K. Harms, G. Hilt and E. Meggers, Chem. – Eur. J., 2015, 21, 7355–7359 CrossRef CAS PubMed.
- Y. Tan, W. Yuan, L. Gong and E. Meggers, Angew. Chem., Int. Ed., 2015, 54, 13045–13048 CrossRef CAS PubMed.
- F. F. de Assis, X. Huang, M. Akiyama, R. A. Pilli and E. Meggers, J. Org. Chem., 2018, 83, 10922–10932 CrossRef CAS PubMed.
- Z. Zhou, X. Nie, K. Harms, R. Riedel, L. Zhang and E. Meggers, Sci. China: Chem., 2019, 62, 1512–1518 CrossRef CAS.
- X. Huang, J. Lin, T. Shen, K. Harms, M. Marchini, P. Ceroni and E. Meggers, Angew. Chem., Int. Ed., 2018, 57, 5454–5458 CrossRef CAS PubMed.
- K. L. Skubi, J. B. Kidd, H. Jung, I. A. Guzei, M. H. Baik and T. P. Yoon, J. Am. Chem. Soc., 2017, 139, 17186–17192 CrossRef CAS PubMed.
- J. Zheng, W. B. Swords, H. Jung, K. L. Skubi, J. B. Kidd, G. J. Meyer, M. H. Baik and T. P. Yoon, J. Am. Chem. Soc., 2019, 141, 13625–13634 CrossRef CAS PubMed.
- W. B. Swords, H. Lee, Y. Park, F. Llamas, K. L. Skubi, J. Park, I. A. Guzei, M.-H. Baik and T. P. Yoon, J. Am. Chem. Soc., 2023, 145, 27045–27053 CrossRef CAS PubMed.
- A. Böhm and T. Bach, Synlett, 2016, 1056–1060 Search PubMed.
- A. Gualandi, F. Calogero, A. Martinelli, A. Quintavalla, M. Marchini, P. Ceroni, M. Lombardo and P. G. Cozzi, Dalton Trans., 2020, 49, 14497–14505 RSC.
- T. Hamada, H. Ishida, S. Usui, Y. Watanabe, K. Tsumura and K. Ohkubo, J. Chem. Soc., Chem. Commun., 1993, 909–911 RSC.
- T. Hamada, H. Ishida, S. Usui, K. Tsumura and K. Ohkubo, J. Mol. Catal., 1994, 88, L1–L5 CrossRef CAS.
- Y. Kimura, D. Uraguchi and T. Ooi, Org. Biomol. Chem., 2021, 19, 1744–1747 RSC.
- G. E. M. Crisenza, A. Faraone, E. Gandolfo, D. Mazzarella and P. Melchiorre, Nat. Chem., 2021, 13, 575–580 CrossRef CAS PubMed.
- S. X. Lin, G. J. Sun and Q. A. Kang, Chem. Commun., 2017, 53, 7665–7668 RSC.
- L. Chen, H. Liangjian, D. Yu, S. Weiping and K. Qiang, Chin. J. Org. Chem., 2020, 40, 3944–3952 CrossRef CAS.
- (a) A. Reichle and O. Reiser, Chem. Sci., 2023, 14, 4449 RSC; (b) A. Hossain, A. Bhattacharyya and O. Reiser, Science, 2019, 364, eaav9713 CrossRef PubMed; (c) M. Zhong, X. Pannecoucke, P. Jubault and T. Poisson, Beilstein J. Org. Chem., 2020, 16, 451–481 CrossRef CAS PubMed; (d) Y. Zhang, Q. Wang, Z. Yan, D. Ma and Y. Zheng, Beilstein J. Org. Chem., 2021, 17, 2520–2542 CrossRef CAS PubMed.
- K. Endo, Y. Liu, H. Ube, K. Nagata and M. Shionoya, Nat. Commun., 2020, 11, 6263 CrossRef CAS PubMed.
- V. Edtmüller, A. Pöthig and T. Bach, Tetrahedron, 2017, 73, 5038–5047 CrossRef.
- Q. Guo, M. Wang, Q. Peng, Y. Huo, Q. Liu, R. Wang and Z. Xu, ACS Catal., 2019, 9, 4470–4476 CrossRef CAS.
- R. Qi, C. Wang, Y. Huo, H. Chai, H. Wang, Z. Ma, L. Liu, R. Wang and Z. Xu, J. Am. Chem. Soc., 2021, 143, 12777–12783 CrossRef CAS PubMed.
- H. D. Xia, Z. L. Li, Q. S. Gu, X. Y. Dong, J. H. Fang, X. Y. Du, L. L. Wang and X. Y. Liu, Angew. Chem., Int. Ed., 2020, 59, 16926–16932 CrossRef CAS PubMed.
- Y. Zhang, Y. Sun, B. Chen, M. Xu, C. Li, D. Zhang and G. Zhang, Org. Lett., 2020, 22, 1490–1494 CrossRef CAS PubMed.
- Y. Li, K. Zhou, Z. Wen, S. Cao, X. Shen, M. Lei and L. Gong, J. Am. Chem. Soc., 2018, 140, 15850–15858 CrossRef CAS PubMed.
- K. Zhou, Y. Yu, Y.-M. Lin, Y. Li and L. Gong, Green Chem., 2020, 22, 4597–4603 RSC.
- X. Shen, Y. Li, Z. Wen, S. Cao, X. Hou and L. Gong, Chem. Sci., 2018, 9, 4562–4568 RSC.
- H. Yu, S. Dong, Q. Yao, L. Chen, D. Zhang, X. Liu and X. Feng, Chem. – Eur. J., 2018, 24, 19361–19367 CrossRef CAS PubMed.
- K. Zhang, L.-Q. Lu, Y. Jia, Y. Wang, F.-D. Lu, F. Pan and W.-J. Xiao, Angew. Chem., Int. Ed., 2024, 58, 13375–13379 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2026 |