Open Access Article
Open Access ArticleMild PIFA-mediated synthesis of benzo[4,5]thiazolo[3,2-a]indoles via oxidative cyclization
Xinya Liua,
Christine Tran*a,
Olivier Provota,
Pascal Retailleaub and
Abdallah Hamze *a
*a
aCNRS, BioCIS, Université Paris-Saclay, 91400 Orsay, France. E-mail: abdallah.hamze@universite-paris-saclay.fr
bICSN, Université Paris-Saclay, UPR 2301, 91198, Gif-sur-Yvette, France
First published on 24th April 2026
Abstract
We report a novel synthetic approach for the synthesis of benzo[4,5]thiazolo[3,2-a]indoles. This strategy relies on a straightforward cyclization of 1-(2-(methylthio)phenyl)-1H-indoles using (bis(trifluoroacetoxy)iodo)benzene (PIFA) as an oxidant. The reaction proceeds smoothly in MeCN at room temperature within 15 minutes, affording 28 diversely substituted benzo[4,5]thiazolo[3,2-a]indoles in good to excellent yields. The viability of the method was confirmed with the preparation of melatonin and tryptophan derivatives, along with a late-stage diversification via Barluenga–Valdés cross-coupling. Detailed mechanistic investigations support PIFA-mediated cyclization via a thionium ion intermediate.
Introduction
Fused heterocyclic systems combining sulfur atoms and indole frameworks have emerged as valuable scaffolds in modern medicinal chemistry, owing to their rich chemical versatility and broad pharmacological scope.1–4 Among them, indole-annulated sulfur heterocycles are particularly attractive, as they display a wide range of biological activities, including effects on the central nervous system,5 as well as anticancer,6 analgesic,7 and antipsychotic properties.8 For instance, thiazolo[3,2-a]indoles constitute potential 5HT4-receptor antagonists,9 and the 3-methylthiazolo[3,2-a]indole moiety is a potential anticancer scaffold10 (Fig. 1). | ||
| Fig. 1 Representative examples of biologically active indole-fused sulfur-containing tricyclic heterocycles. | ||
Beyond their biomedical relevance, these frameworks have also proven useful in the development of donor–acceptor chromophores with promising optoelectronic properties.11,12 Moreover, such structures are not solely synthetic, as they also occur in nature, notably among phytoalexins produced by cruciferous plants as part of their defense mechanisms.13
Despite the growing interest in sulfur-fused indole derivatives, most synthetic methodologies have focused on 2,3-thiophene-fused indoles.14 In contrast, synthetic access to the less-explored 1,2-fused isomers, particularly thiazolo[3,2-a]indoles remains limited. This disparity is primarily attributed to the lower nucleophilicity of the indole nitrogen compared to the more reactive C3 carbon, thereby rendering conventional annulation strategies less efficient.
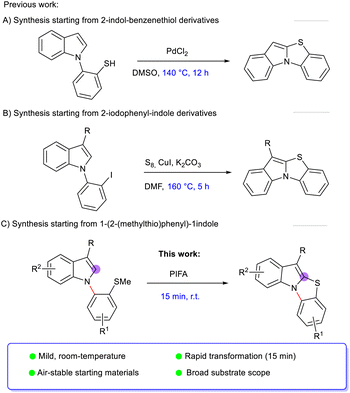
To the best of our knowledge, only a limited number of methods have been reported for the synthesis of benzo[4,5]thiazolo[3,2-a]indoles. In 2022, Lee et al. described a palladium-catalyzed coupling method utilizing 2-(1H-indol-1-yl)benzenethiol derivatives (Scheme 1A).15 More recently, a copper-catalyzed protocol was developed using 1-(2-iodophenyl)-1H-indoles and elemental sulfur, requiring elevated temperatures (up to 160 °C) to afford the desired products (Scheme 1B).16
Although these methods provide access to the target scaffold, they typically rely on transition metal catalysis, harsh reaction conditions, and prolonged reaction times, which may limit their practical applicability and functional group tolerance. In particular, the requirement for high temperatures and specific prefunctionalized substrates can restrict substrate diversity.
These limitations highlight the need for more efficient and mild synthetic approaches, ideally operating under metal-free conditions for the cyclization step, with shorter reaction times and improved functional group compatibility.
Over the past few decades, bis(trifluoroacetoxy)iodobenzene (PIFA) has emerged as an environmentally friendly alternative to toxic organometallic catalysts and hazardous heavy-metal oxidants, gaining increasing prominence in organic synthesis.17,18
PIFA has demonstrated remarkable versatility across a wide range of transformations, including sulfenylation, oxidative coupling, amination, and alkyl arylation reactions.19,20 In addition to its broad reactivity, PIFA offers practical advantages such as commercial availability, low cost, low toxicity, and convenient handling.21 Of particular interest is the PIFA-mediated aminotrifluoroacetoxylation of alkenes, which has drawn our attention. In this transformation, the alkene is oxidized by PIFA to form an iodonium ion intermediate, which is then be intramolecularly attacked by a nucleophile, leading to the formation of cyclic compounds.22
This development builds upon our laboratory's recent achievements, which include the successfully development of a PIFA-mediated C–S bond oxidative cyclization system for constructing benzothiophene compounds at room temperature.23
Herein, we report a PIFA-mediated C2-selective cyclization of indoles leading to benzo[4,5]thiazolo[3,2-a]indole derivatives from readily accessible 1-(2-(methylthio)phenyl)indole substrates (Scheme 1C). This method provides a concise and environmentally friendly route to structurally complex and highly functionalized benzo-thiazoloindole frameworks.
Results and discussion
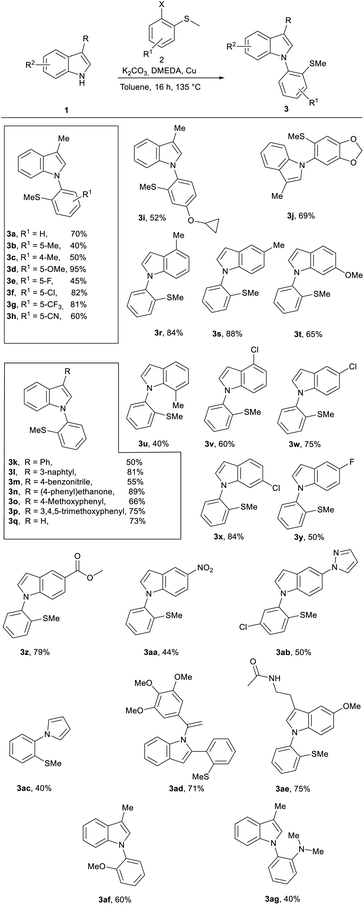
The synthesis of 1-(2-(methylthio)phenyl)-1H-indole compounds 3 was achieved via a copper-mediated Ullmann aryl amination using commercially available (2-haloaryl)(methyl)sulfanes 2 (Scheme 2).The coupling exhibited a broad substrate scope and good tolerance toward both electron-rich and electron-deficient aryl halides, affording the desired products 3a–3j in generally high isolated yields.
The methodology was also applicable to a range of substituted indoles, providing derivatives 3k–3aa in synthetically useful yields. More challenging heteroaryl partners, such as the 5-pyrazolyl-indole derivative 3ab and, the electron-rich pyrrole derivative 3ac, were obtained in moderate yields. In addition, the protocol enabled access to vinyl-substituted indole 3ad and allowed late-stage functionalization toward bio-relevant motifs, including the melatonin precursor 3ae, further supporting the utility of the system for diversification of sulfur-containing indole scaffolds.
The introduction of additional functional groups was also investigated. Substrates bearing functional groups such as aldehyde, carboxylic acid, or amino substituents (e.g., indole-3-carboxaldehyde, indole-3-carboxylic acid, and 5-aminoindole) did not afford the desired products, possibly due to interference with the PIFA-mediated activation or competing side reactions. In addition, halogenated substrates (e.g., 5-iodo- and 5-bromoindole) were also unsuccessful, which may be attributed to altered electronic properties or reduced stability of key intermediates under the reaction conditions.
To further probe the role of the sulfur substituent, compounds 3af and 3ag, bearing OMe and NMe2 groups, respectively, were synthesized in acceptable yields to enable comparison with the SMe-containing analogs in the subsequent cyclization step.
Subsequently, compound 3a was subjected to cyclization, and the results of the preliminary optimization studies are detailed in Table 1. Initial experiments using 1.0 equivalent of PIFA in MeCN and DCM (0.3 M) afforded moderate to low yields of 40% and 25%, respectively (entries 1 and 2). When PIFA was replaced with iodobenzene diacetate (PIDA), no formation of 4a was observed, and the starting material 3a was fully recovered (data not shown in Table 1).
| Entry | Solvent | PIFA (equiv.) | Conc. (M) | Yield of 4a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b (%) b (%) |
|---|---|---|---|---|
| a Cyclization conditions: 3a (0.3 mmol, 1.0 equiv.), PIFA (as indicated), solvent, rt, under air, 15 min.b Isolated yield.c n.d. = not detected, reaction performed without solvent.d The yield is the average of 3 reaction runs.e Reaction was performed at 0 °C.f Reaction was performed at 50 °C.g Reaction was performed under nitrogen. | ||||
| 1 | MeCN | 1 | 0.3 | 40 |
| 2 | DCM | 1 | 0.3 | 25 |
| 3 | MeCN | 1.5 | 0.3 | 56 |
| 4 | MeCN | 2.0 | 0.3 | 74 |
| 5 | MeCN | 3.0 | 0.3 | 75 |
| 6 | MeCN | 2.0 | 1.5 | 71 |
| 7 | MeCN | 2.0 | 3.0 | 76 |
| 8 | — | 2.0 | Neat | n.d.c |
| 9 | MeCN | 1.1 | 3.0 | 63 |
| 10 | MeCN | 1.3 | 3.0 | 74d |
| 11 | MeCN | 1.3 | 3.0 | 74e |
| 12 | MeCN | 1.3 | 3.0 | 40f |
| 13 | MeCN | 1.3 | 3.0 | 74g |
| 14 | EtOH | 1.3 | 3.0 | 23 |
| 15 | MeOH | 1.3 | 3.0 | 10 |
| 16 | DCE | 1.3 | 3.0 | 20 |
| 17 | Dioxane | 1.3 | 3.0 | 0 |
| 18 | THF | 1.3 | 3.0 | 0 |
Increasing the PIFA loading to 1.5 equivalents in MeCN improved the yield to 56% (entry 3), while a further increase to 2 equivalents led to a significant increase to 74% (entry 4). A slight increase to 3 equivalents of PIFA provided only a marginal improvement in yield (75%, entry 5). Reducing the solvent volume to 0.2 mL (1.5 M, entry 6) and 0.1 mL (3.0 M, entry 7) maintained high yields of 71% and 76%, respectively, demonstrating the robustness of the transformation under more concentrated conditions. In contrast, performing the reaction under neat conditions resulted in no conversion (entry 8), underscoring the importance of solvent in the coupling process. To improve reagent economy, the reaction was then evaluated at high concentration (3.0 M) with reduced PIFA loading. Lowering the amount of PIFA to 1.1 equivalents led to a decrease in yield (63%, entry 9), whereas 1.3 equivalents restored the yield to 74% (entry 10), representing an optimal balance between efficiency and reagent usage.
Additional fine-tuning optimizations showed that there is no major variation in the reaction efficiency by lowering the temperature to 0 °C (entry 11) or under nitrogen atmosphere (entry 13). Increasing the reaction temperature contributed to the yield reduction, probably due to the thermal decomposition of 4a. It should be also noted that longer reaction times led to the decomposition of product 4a, which could be observed on TLC after 20 min-reaction (data not shown).
Finally, solvent screening revealed that polar protic solvents such as EtOH or MeOH significantly decreased the reaction efficiency (entries 14 and 15).
The same trend was observed with apolar solvents (entries 16 and 17). Likewise, no product was noticed by using THF (entry 18).
Using the standard condition, indole substrates bearing OMe (3af) or NMe2 (3ag) substituents in place of SMe were evaluated; however, no cyclization was observed. This result underscores the essential role of the sulfur substituent, likely due to its ability to form a reactive thionium intermediate under PIFA-mediated conditions.
In the case of the NMe2-substituted derivative, only the starting material was recovered, indicating no productive reactivity under these conditions. For the OMe-substituted indole, LC–MS analysis of the crude reaction mixture revealed degradation products, with no trace of the expected cyclized compound, suggesting poor stability of this substrate in the oxidative cyclization environment.
Finally, the optimal conditions were identified as 1.3 equivalents of PIFA in MeCN (3.0 M), at room temperature for 15 min, consistently affording 4a in 74% yield across three independent experiments (entry 10).
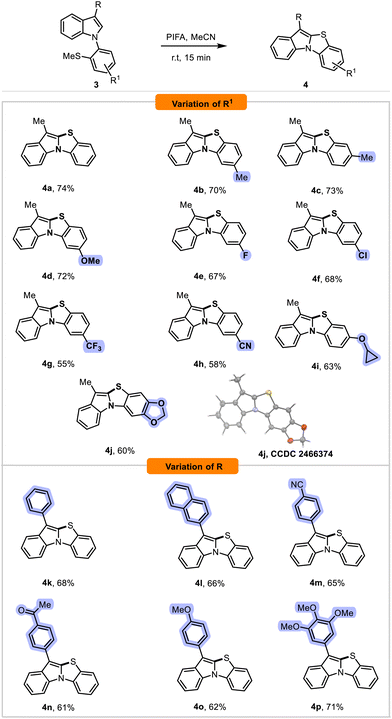
Under the optimized conditions, we initially investigated the reactivity scope of 3-methyl-1-(2-(methylthio)phenyl)-1H-indole 3, bearing various substituents on the phenyl ring (R1), (Scheme 3). All indole derivatives 3a–3j underwent cyclization to afford the corresponding target products 4a–4j in moderate to good yields (Scheme 3). Slightly higher yields were observed for compounds 4a–4d bearing electron-donating groups (EDGs), compared to those with strong electron-withdrawing groups (EWGs) such as trifluoromethyl 4g and cyano 4h. Moreover, this transformation was compatible with a cyclopropoxy group on the phenyl part, affording compound 4i in good yield. The presence of a 1,3-dioxole moiety on the phenyl ring of indole was well tolerated, delivering the pentacyclic compound 4j in a good 60% yield. It is worth noting that the structure of 4j was fully confirmed through X-ray diffraction analysis (see SI).
Subsequently, we expanded our investigation to explore the scope of indole derivatives by modifying the 3-aryl substituent (R). Notably, this transformation exhibited remarkable versatility for functionalizing the C3 position of the indole ring. The reaction proved to be effective for a variety of 3-arylindoles bearing various substituents, including phenyl, naphthyl groups, and compounds 4k and 4l were obtained in good yield (Scheme 3). Next, we examined the influence of electronic effects on this transformation. Substrates bearing EWGs, such as cyano 4m or acetyl 4n, as well as those with EDGs, such as methoxy 4o, or trimethoxyphenyl 4p substituent, all afforded the desired products in good yields. These results suggest that the electronic nature of the 3-aryl substituent has little influence on the efficiency of the transformation.
Next, to further explore the scope of this cyclization, we examined indole derivatives with an unsubstituted C3 position, in combination with variations of the R2 substituent on the indole ring (Scheme 4). Overall, no clear dependency on electronic effects was observed, as both EDGs, such as in compounds 4r, 4s, 4t, and EWGs, such as in compounds 4v, 4w, 4x, 4y, and 4z, led to good yields. However, a decrease in yield was noted when using 7-methylindole as the starting material, with compound 4u obtained in a moderate yield of 50%, likely due to increased steric hindrance from the 7-position substituent, which negatively affected the reaction efficiency.
 | ||
Scheme 4 Substrate scope of C3-unsubstituted indole. Same conditions as in Scheme 1. a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Not detected. Not detected. | ||
Cyclization was not observed for the nitro-substituted indole 3aa, and the starting material was fully recovered, likely due to the strong electron-withdrawing effect reducing the nucleophilicity of the indole core. It should be noted that the structure of compound 4y was unambiguously determined by single-crystal X-ray diffraction.
Finally, the incorporation of heteroaryl motifs was briefly explored. The pyrazole ring, a well-known pharmacophore in enzyme inhibitors and receptor modulators due to its unique hydrogen bond donor and acceptor properties, was successfully introduced, highlighting the potential of this methodology for expanding molecular diversity and accessing biologically relevant scaffolds.24
As an illustrative example, the 5-(1H-pyrazol-1-yl)-1H-indole substrate underwent PIFA-mediated cyclization to afford the cyclized product 4ab in a good yield.
Interestingly, replacing the indole core with a 1H-pyrrole scaffold enabled the synthesis of benzo[d]pyrrolo[2,1-b]thiazole 4ac in good, isolated yield, further demonstrating the versatility and adaptability of this methodology.
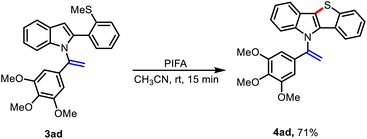
To expand the scope of this method and investigate selectivity during the cyclization process, we prepared vinyl-indole 3ad, which presents two possible sites for cyclization: one at the C3 position of the indole and the other one at the terminal alkene (Scheme 5).
When vinyl-indole 3ad was subjected to the standard condition, cyclisation occurred selectively between the C3 position of the indole and the sulfur atom, affording the desired (1-phenylvinyl)-10H-benzo[4,5]thieno[3,2-b]indole derivative 4ad in 71% yield. Collectively, these results highlight the broad applicability of this method for constructing structurally diverse molecular architectures.
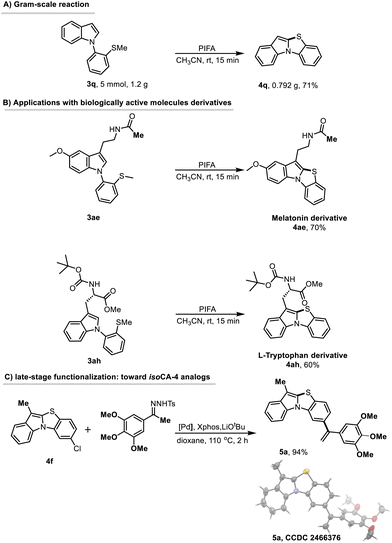
To evaluate the synthetic practicality of the developed method, we carried out the cyclization of 1-(2-(methylthio)phenyl)-1H-indole (3q) under standard conditions on a gram scale (Scheme 6). The desired product 4q was obtained in 71% yield, demonstrating that the protocol can be effectively scaled up without significant loss in yield.
To further demonstrate the broad applicability of this PIFA-mediated cyclization method, we prepared two bioactive molecules derivatives following our synthetic methodology.
Melatonin, a multifunctional molecule, plays a critical role in regulating circadian rhythm, exerting antioxidant effect, protecting the cardiovascular system, and exhibiting anti-aging properties.25
Its diverse mechanisms of action and therapeutic potential make it a prominent target in biomedical research. As anticipated, the PIFA-mediated cyclization of 3ae proceeded smoothly, affording the target product 4ae in a good 70% yield (Scheme 6).
Additionally, L-tryptophan derivatives, known for their unique structures and biological activities, are of significant importance in drug development, biochemical research, nutrition, health, and material science.26–29 Using 3ah as a substrate, we carried out the cyclization under standard conditions to obtain the target compound 4ah in a good 60% yield. These examples further underscore the potential of this method for the synthesis of biologically active compounds derivatives.
Furthermore, we exploited the C–Cl bond in substrate 4f to achieve further late-stage functionalization. Compound 4f was subjected to a Barluenga–Valdés cross-coupling reaction with an N-tosylhydrazone derivative having a trimethoxyphenyl group, affording compound 5a in an excellent 94% yield. Single-crystal X-ray diffraction analysis confirmed the molecular structure of 5a (see SI). This compound can be regarded as an analog of isoCA-4, a potent antitubulin agent with notable antitumor activity.30,31 This synthetic route highlights the versatility of the approach and demonstrates its potential as a valuable tool for drug development and synthetic organic chemistry.
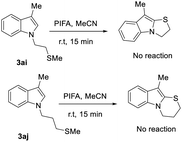
Next, we investigated N-alkylated indole derivatives for oxidative cyclization in place of N-aryl substrates (Scheme 7). Using linkers R = CH2CH2SMe (3ai) and R = CH2CH2CH2SMe (3aj) gave no cyclized product upon PIFA treatment, returning either starting material for 3ai or undefined degradation by LC–MS for 3aj. These results show that N-alkylated thio-tethered indoles are not competent substrates under the current oxidative cyclization conditions, owing to limited oxidative stability and precursor accessibility.
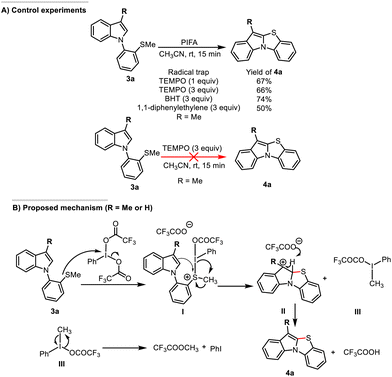
To investigate the reaction mechanism of this transformation, substrate 3a was treated with 1 to 3 equivalents of TEMPO. The desired product 4a was obtained in 66 to 67% yield, indicating that radical pathways are unlikely to be involved (Scheme 8A).
Consistently, the addition of 3 equivalents of BHT did not significantly affect the reaction outcome, while only a slight decrease in yield was observed in the presence of 1,1-diphenylethylene.
Furthermore, no reaction occurred when 3a was treated with TEMPO alone, and no TEMPO adducts were detected, with complete recovery of the starting material. These observations collectively argue against a radical mechanism.
On the basis of these results of previous reports on PIFA-mediated reactions,32 a plausible ionic mechanism is proposed (Scheme 8B). Initial oxidation of substrate 3a by PIFA generates a thionium intermediate I, likely via activation of the sulfur atom accompanied by the loss of a trifluoroacetate anion. The enhanced electrophilicity at sulfur facilitates intramolecular nucleophilic attack by the indole moiety.
Cyclization preferentially occurs at the C2 position of the indole, despite the intrinsic nucleophilicity of C3. This selectivity can be rationalized by both electronic and structural factors: (i) in C3-substituted indoles, steric hindrance disfavors attack at C3; and (ii) C2-attack leads to the formation of a more favorable fused bicyclic system, whereas C3-attack would generate a less stable, non-fused intermediate. Notably, even in C3-unsubstituted indoles (R = H), the reaction proceeds selectively via C2 cyclization, suggesting that thermodynamic control and ring annulation stability dominate over intrinsic nucleophilicity.
Next, the C2 position of the indole moiety attacks the sulfur atom of the thionium salt I, leading to the formation of carbocation intermediate II, along with the formation of a trivalent iodine species III. Deprotonation of intermediate II by trifluoroacetate anion then affords the final product, benzo[4,5]thiazolo[3,2-a]indole 4a. Meanwhile, intermediate III decomposes into methyl trifluoroacetate and iodobenzene, the latter being detected and identified by 1H NMR.
Conclusions
In summary, we have developed a mild and facile procedure for the construction of benzo[4,5]thiazolo[3,2-a]indole derivatives. This reaction is based on a PIFA-mediated cyclization of 1-(2-(methylthio)phenyl)-1H-indoles at room temperature. This methodology efficiently accommodates a wide substrate scope, with 28 examples of variously functionalized benzo[4,5]thiazolo[3,2-a]indoles synthesized. The reliability of our strategy was demonstrated with a gram-scale reaction and the synthesis of bioactive molecules derivatives. Late-stage functionalization through a Barluenga–Valdés cross-coupling highlights the added value of benzo[4,5]thiazolo[3,2-a]indoles as key intermediates in the synthesis of bioactive compounds.Conflicts of interest
There are no conflicts to declare.Data availability
The data related to this work are available in the supplementary information (SI). Supplementary information: 1H, 13C and 19F NMR spectra of all new compounds. See DOI: https://doi.org/10.1039/d6ob00393a.CCDC 2466374–2466376 contain the supplementary crystallographic data for this paper.33a–c
Acknowledgements
The authors acknowledge the support of this project by CNRS, Paris-Saclay University, and La Fondation ARC pour la recherche sur le cancer ARCPJA2022060005209. The authors also thank the China Scholarship Council (CSC) for a fellowship to X. L.References
- W. Zeng, C. Han, S. Mohammed, S. Li, Y. Song, F. Sun and Y. Du, RSC Med. Chem., 2024, 15, 788–808 RSC.
- H. T. Hamad, Results Chem., 2025, 15, 102182 CrossRef CAS.
- S. Pathania, R. K. Narang and R. K. Rawal, Eur. J. Med. Chem., 2019, 180, 486–508 CrossRef CAS PubMed.
- S. B. Seen, Y. Gong and M. Ashton, in Advances in Heterocyclic Chemistry, ed. E. F. V. Scriven and C. A. Ramsden, Academic Press, 2023, vol. 139, pp. 1–85 Search PubMed.
- A. Kumari and R. K. Singh, Bioorg. Chem., 2019, 89, 103021 CrossRef CAS.
- Y. Sugita, H. Hosoya, K. Terasawa, I. Yokoe, S. Fujisawa and H. Sakagami, Anticancer Res., 2001, 21, 2629–2632 CAS.
- S. Takada and Y. Makisumi, Chem. Pharm. Bull., 1984, 32, 872–876 CrossRef CAS.
- L. A. van Vliet, N. Rodenhuis, D. Dijkstra, H. Wikström, T. A. Pugsley, K. A. Serpa, L. T. Meltzer, T. G. Heffner, L. D. Wise, M. E. Lajiness, R. M. Huff, K. Svensson, S. Sundell and M. Lundmark, J. Med. Chem., 2000, 43, 2871–2882 CrossRef CAS PubMed.
- L. Gaster and P.-A. Wyman, WO9318036A1, World Intellectual Property Organization (WIPO), 1993.
- S. Short, S. Rhodes, V. S. Bhave, R. Hojo and M. Jha, Synthesis, 2019, 4263–4270 CAS.
- T. Qi, W. Qiu, Y. Liu, H. Zhang, X. Gao, Y. Liu, K. Lu, C. Du, G. Yu and D. Zhu, J. Org. Chem., 2008, 73, 4638–4643 CrossRef CAS PubMed.
- J. Kim, S. Chae, A. Yi, S. Hong, H. J. Kim and H. Suh, J. Appl. Polym. Sci., 2019, 136, 47624 CrossRef.
- M. S. C. Pedras and D. P. O. Okinyo, Org. Biomol. Chem., 2008, 6, 51–54 RSC.
- G. Mari, L. De Crescentini, G. Favi, S. Santeusanio and F. Mantellini, Org. Biomol. Chem., 2022, 20, 4167–4175 RSC.
- N.-J. Lee, Y.-J. Heo, W.-J. Jeong and D.-J. Kim, WO2021045469, 2021.
- Z. Wang, Y. Yuan, S. Jin, Y. Wang, Y. Gu, C. Wang and Y. Li, New J. Chem., 2023, 47, 1055–1058 RSC.
- R. D. Richardson and T. Wirth, Angew. Chem., Int. Ed., 2006, 45, 4402–4404 CrossRef CAS PubMed.
- T. Wirth, Angew. Chem., Int. Ed., 2005, 44, 3656–3665 CrossRef CAS PubMed.
- V. V. Zhdankin, in Hypervalent Iodine Chemistry: Modern Developments in Organic Synthesis, ed. T. Wirth, Springer Berlin Heidelberg, Berlin, Heidelberg, 2003, pp. 99–136, DOI:10.1007/3-540-46114-0_4.
- F. F. Fleming, L. Funk, R. Altundas and Y. Tu, J. Org. Chem., 2001, 66, 6502–6504 CrossRef CAS PubMed.
- S. Kumar, A. Arora, S. K. Singh, R. Kumar, B. Shankar and B. K. Singh, Org. Biomol. Chem., 2024, 22, 3109–3185 RSC.
- H. M. Lovick and F. E. Michael, J. Am. Chem. Soc., 2010, 132, 1249–1251 CrossRef CAS PubMed.
- X. Liu, O. Provot, C. Tran, J. F. Soule and A. Hamze, Org. Chem. Front., 2025, 12, 24–32 RSC.
- S. Fustero, M. Sánchez-Roselló, P. Barrio and A. Simón-Fuentes, Chem. Rev., 2011, 111, 6984–7034 CrossRef CAS PubMed.
- H. J. Kim, H. J. Kim, M. K. Bae and Y. D. Kim, Int. J. Mol. Sci., 2017, 18, 1142 CrossRef PubMed.
- T. Santos, J. Carvalho, M. C. Corvo, E. J. Cabrita, J. A. Queiroz and C. Cruz, Int. J. Biol. Macromol., 2016, 87, 385–396 CrossRef CAS PubMed.
- M. C. Almeida, D. Resende, P. M. da Costa, M. M. M. Pinto and E. Sousa, Eur. J. Med. Chem., 2021, 209, 112945 CrossRef CAS PubMed.
- S. Xiao, Z. Wang, B. Wang, B. Hou, J. Cheng, T. Bai, Y. Zhang, W. Wang, L. Yan and J. Zhang, Front. Microbiol., 2023, 14, 1099098 CrossRef PubMed.
- J. Graff, J. Müller, A. Sadurní, M. Rubin, I. A. Canivete Cuissa, C. Keller, M. Hartmann, S. Singer, J. Gertsch and K.-H. Altmann, ChemMedChem, 2022, 17, e202200308 CrossRef CAS PubMed.
- S. Messaoudi, B. Treguier, A. Hamze, O. Provot, J.-F. Peyrat, J. R. De Losada, J.-M. Liu, J. Bignon, J. Wdzieczak-Bakala, S. Thoret, J. Dubois, J.-D. Brion and M. Alami, J. Med. Chem., 2009, 52, 4538–4542 CrossRef CAS PubMed.
- A. Hamze, A. Giraud, S. Messaoudi, O. Provot, J.-F. Peyrat, J. Bignon, J.-M. Liu, J. Wdzieczak-Bakala, S. Thoret, J. Dubois, J.-D. Brion and M. Alami, ChemMedChem, 2009, 4, 1912–1924 CrossRef CAS PubMed.
- S. Spyroudis and A. Varvoglis, J. Chem. Soc., Chem. Commun., 1979, 615–616 RSC.
- (a) CCDC 2466374: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2nsggd; (b) CCDC 2466375: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2nsghf; (c) CCDC 2466376: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2nsgjg.
| This journal is © The Royal Society of Chemistry 2026 |