Open Access Article
Open Access ArticleNon-catalytic low-cost and sustainable amidation using a twisted amide
Takefumi Kuranaga*,
Ai Koyama,
Kosuke Shimizu,
Tensei Tokuda and
Hideaki Kakeya *
*
Department of System Chemotherapy and Molecular Sciences, Division of Medicinal Frontier Sciences, Graduate School of Pharmaceutical Sciences, Kyoto University, Yoshida, Kyoto 606-8501, Japan. E-mail: tkuranaga@pharm.kyoto-u.ac.jp; scseigyo-hisyo@pharm.kyoto-u.ac.jp
First published on 24th March 2026
Abstract
Cost reductions and environmental friendliness have been strongly desired in amide synthesis. In this study, carboxylic acid was converted to a twisted amide using inexpensive tosyl isocyanate and bromoacetate. The twisted amide was then reacted with an amine to give the desired amide and commercially valuable tosylglycine esters. Epimerization-suppressed amidation, application to peptide synthesis, column chromatography-free amide synthesis, and solid-phase amide synthesis were established. Moreover, the effective use of the co-product tosylglycine ester was demonstrated by sustainable peptoid synthesis. The established method can become a practical amidation approach that solves the cost and sustainability issues in amide synthesis.
Introduction
Amides are important structural motifs in small molecule pharmaceuticals and chemical products, and form the main chains of peptides and synthetic polymers such as nylon. Therefore, amidation is in high demand industrially. In recent years, cost reductions and environmental friendliness have been a focus for large-scale amidation.1 In fact, “amide bond formation avoiding poor atom economy reagents” was identified as the top challenge by the American Chemical Society Green Chemistry Institute Pharmaceutical Roundtable (ACS GCIPR) in 2007.2Catalysis is an important technology for achieving cost reduction and environmental friendliness in industrial synthesis.3 However, while many catalytic amidation reactions have been reported in the past decade,4 they are not optimal in terms of synthetic efficiency, cost, or sustainability. One of the major obstacles in catalytic amidation is that the catalysts typically have more complex structures and are more difficult to synthesize than the target amide, which makes the amidation unprofitable. Furthermore, if the catalyst is synthesized by multi-step non-catalytic reactions, then the amidation is not truly sustainable.
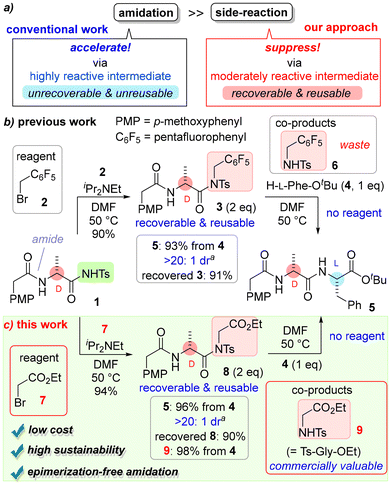
To solve this issue, we established a concept for non-catalytic but low-cost and highly sustainable amide bond formation (Scheme 1a). To achieve ideal amidation without side reactions, the amidation reaction rate must be faster than any side reactions.5 The main focus of recently developed coupling reagents/catalysts is to promote amidation over side reactions (Scheme 1a).6 By contrast, our concept uses a moderately reactive intermediate to reduce the rates of side reactions more than that of amidation (Scheme 1b).7 The amidation of 3 with amine 4 yields stereochemically pure amide 5 without any additional reagent/additive. Although the reaction rate of amidation via 3 is slower than that of amidation via conventional coupling reagents, an advantage of using a moderately reactive intermediate is that excess 3 can be recovered and reused after the amidation. The moderate stability of the intermediate also allows for all reactions to be performed under air, which is an important factor for low-cost organic synthesis. If the catalyst/reagent is unstable in the presence of air and/or moisture, it will require special handling, which will increase the cost.
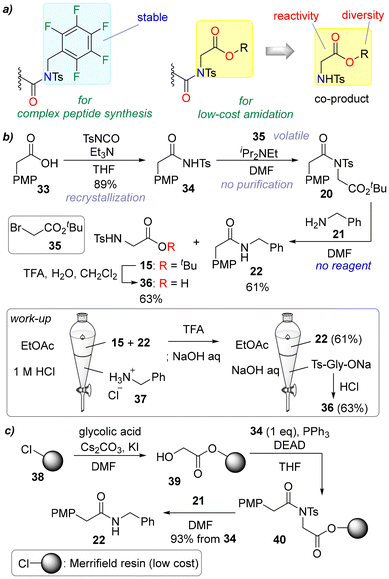
Alkylated acyl sulfonamides such as 3 are recognized as acyclic twisted amides,8 and we have continued our research using these acyclic twisted amides.9 Recently, we found that 8 had comparable reactivity to 3 for amidation and was advantageous in terms of cost and sustainability (Scheme 1c). This crutial modification solved a significant problem in the amidation reaction, the details of which are reported in this manuscript.
Results and discussion
Amidation via 3 is very effective for isomerization-suppressed peptide synthesis compared with the conventional method but yields 1 eq. of 6 as a co-product (see Scheme S1 in the SI for details). We have not found an effective use for 6 and it is considered a waste of the amidation. We anticipated that if 7 was used instead of 2 to activate sulfonamide 1, the twisted amide 8 would be obtained and the co-product in the amidation of 8 would be the protected α-amino acid 9. As previously reported, acyl sulfonamide 1 was readily prepared from the corresponding carboxylic acid by the action of tosyl isocyanate, and twisted amide 8 was obtained by alkylation of 1 with ethyl bromoacetate (7). Tosyl isocyanate is produced industrially and used as a stabilizer in paints and adhesives, while bromoacetates are ubiquitous reagents used in industrial organic synthesis, and both of these reagents are very inexpensive (Scheme S2). Although pentafluorobenzyl bromide (2) is not expensive, bromoacetates are less expensive. Furthermore, protected α-amino acid 9, which is generated as a co-product of amide 5, is a valuable compound that is used commercially as a material to produce peptides and secondary amines.10 This means that the co-product in the amidation will not be classed as waste.As proof of our hypothesis, twisted amide 8 was synthesized from 1 using 7 (Scheme 1c). Our preliminary experiments on amidation with 8 showed that 5 was obtained in good yield with stereochemical purity, and 9 was isolated as a co-product as expected. Excess twisted amides could be recovered for reuse (Scheme S3).
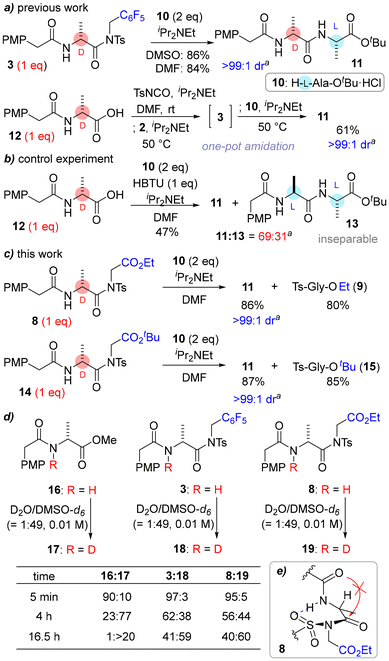
Encouraged by the results, we then examined this low-cost and sustainable amidation in detail. Initially, the ability for isomerization suppression was tested using ethyl ester 8 and t-butyl ester 14 (Scheme 2). In these amidations, an excess of 10 (2 eq.) over 8/14 was used, which meant that Cα-epimerization was problematic (Scheme 2b) because the final concentration of 8/14 reached zero. This has been discussed in our previous studies on inverse peptide synthesis.7b Under these conditions, N-capped by-products11 would also be generated from excess 10 when using a conventional coupling reagent such as HBTU.12
The results of the amidation are summarized in Scheme 2c and the details are shown in Scheme S4. As expected, no epimerized peptide was generated, and the reaction rates were not significantly different between ethyl ester and t-butyl ester. Because the mechanism of isomerization suppression was assumed to be similar to that of 3, we performed the same NMR experiments (Scheme 2d). The results suggested there was an intramolecular hydrogen bond that prevented cyclization to azlactone13 (Scheme 2e) like with 3.
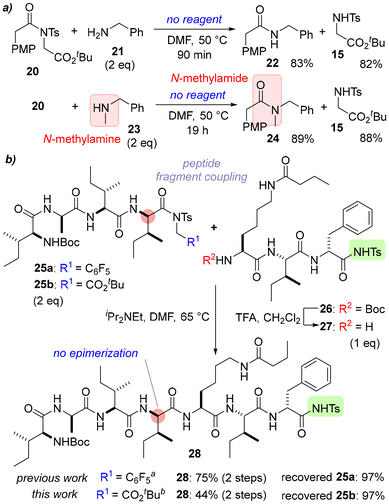
Next, the scope and limitations of the amidation were investigated (Scheme 3). Because the demand for low-cost amidation is not limited to peptide synthesis, non-peptidic compounds were first synthesized (Scheme 3a). Amidation of 20 with the sterically less hindered amine 21 gave 22 and 15 within 90 min at 50 °C. Additionally, N-methyl amide 24 was obtained by the reaction of 20 and 23 without any additional reagents/additives. While no significant differences in reactivity were observed in the synthesis of simple amides, acetate derivatives were less reactive than pentafluorobenzyl derivatives in the synthesis of complex peptides. Recently, we reported using pentafluorobenzyl derivatives for isomerization-suppressed peptide fragment couplings.7b The amidation of tetrapeptide 25a with tripeptide 27 gave heptapeptide 28 in 75% yield, whereas 28 was obtained in only 44% yield from 25b with a reaction time of over 2.5 days (Scheme 3b). Although acetate derivatives are cost effective for the synthesis of simple amides, pentafluorobenzyl derivatives are still useful for the synthesis of complex peptides (see also Scheme S7).
Intermolecular reactions can be accelerated by excess building blocks, while intramolecular amidations cannot be performed with excess reactants. Next, the efficiency of the present method in the amidation using equimolar amounts of twisted amides and amines was demonstrated by several intramolecular amidations (Schemes 4, 5, and S6).
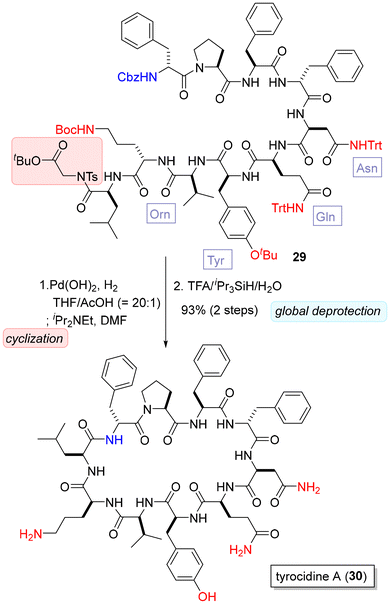
The applicability of the present method in the synthesis of a complex medium-sized cyclic peptide was showcased in the synthesis of tyrocidine A (30, Scheme 4).14 This total synthesis also confirmed the functional group tolerance of the amidation in peptide synthesis. The established method can be applied to the synthesis of complex peptides with various protected side chains.
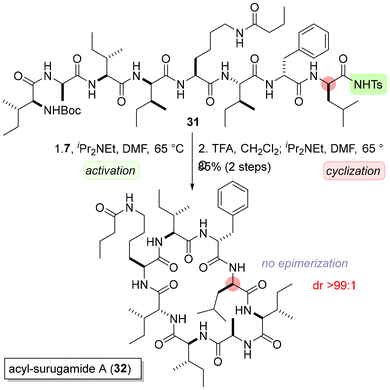
Total synthesis of an antimicrobial cyclic peptide acyl-surugamide A (32)15 from 28 was also demonstrated (Scheme 5). Linear octapeptide 31 could be readily obtained from 28 by inverse peptide synthesis in our previous work. First, the C-terminus of 31 was activated with inexpensive ethyl bromoacetate (7).
Second, the N-terminal Boc group was removed by TFA, and the resulting TFA salt was dissolved in DMF with a base to provide stereochemically pure 32. A highly isomerizable cyclic peptide 32![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7b could be synthesized without the use of high-cost coupling reagents such as HATU.16 Isomerization suppression by the intramolecular hydrogen bond (vide supra, Scheme 2e) is expected to be effective under high dilution conditions, making the present method a useful tool for the synthesis of macrolactams.
7b could be synthesized without the use of high-cost coupling reagents such as HATU.16 Isomerization suppression by the intramolecular hydrogen bond (vide supra, Scheme 2e) is expected to be effective under high dilution conditions, making the present method a useful tool for the synthesis of macrolactams.
Another major difference between pentafluorobenzyl derivatives and acetate derivatives is the structural diversity and reactivity (Scheme 6a). The structural diversity/reactivity of acetate derivatives in their ester moiety makes purification easier. Initially, solution-phase amidation without column chromatography was demonstrated (Scheme 6b). Carboxylic acid 33 was treated with TsNCO to give 34, which was then alkylated by 35 in the presence of i-Pr2NEt to afford 20. Excess 35 and i-Pr2NEt were removed in vacuo, and then 20 was reacted with 21 to generate 22 and co-product 15. After removal of excess 21 by acidic aqueous work-up, the mixture of 22 and 15 was treated with TFA. The resulting 22 and 36 could be easily separated by basic aqueous work-up, and 22 and 36 could be obtained from 33 without chromatographic purification.
 | ||
| Scheme 6 Column chromatography-free amidation. All yields are isolated yields after recrystallization. | ||
Because no significant differences in reactivity were observed among the different ester structures (vide supra, Scheme 2c), we then examined the solid-phase amidation using resin-bound glycolate 39 (Scheme 6c). The advantages of this method compared with the conventional on-resin amidation17 are summarized in Scheme S8. In the on-resin amide synthesis using relatively inexpensive resins such as Merrifield resin,18 harsh conditions are often required to cleave the amide from the resins.19 By contrast, our method does not require a cleavage reaction because the amidation releases the amide into the solution. In addition to resins used for general peptide synthesis, several specialized resins such as resin-bound HOBt20 have been reported (Scheme S8c), but they are expensive compared with the amidation in the present study. Furthermore, amidation via these resins requires the activation of carboxylic acids with coupling reagents, and the excess carboxylic acid and coupling reagents cannot be recovered and reused, resulting in a large variety and volume of waste. The solid-phase synthesis commenced with the loading of glycolic acid onto Merrifield resin (38) with Cs2CO3/KI to furnish 39. Sulfonamide 34 was then loaded onto the resin by an on-resin Mitsunobu reaction21 to afford 40. The resin-bound twisted amide 40 was then treated with 21, releasing amide 22 into the solution. Excess 21 was removed by acidic aqueous work-up to obtain 22 without the need for chromatographic purification.
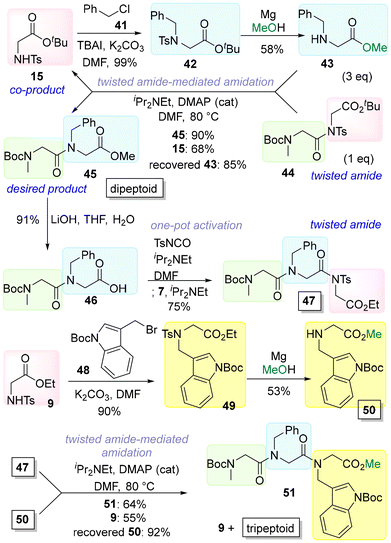
The effective use of the co-product tosylglycine esters 9/15 in the amidations was also demonstrated in this study (Scheme 7). The tosyl group is used as a protecting group for amines and functions to increase the acidity of the tosylamide and promote N-alkylation. Therefore, tosylglycine esters are widely used as starting materials for the synthesis of various secondary amines. In this study, 15 was readily alkylated by 41 to give 42. Subsequently, 42 was treated with magnesium in MeOH,22 realizing simultaneous cleavage of the tosyl group and transesterification to deliver secondary amine 43. Amidation between 43 and 44 gave both the di-N-alkyl glycine (dipeptoid)23 45 and the starting material 15 as a co-product. C-terminal deprotection of 45 by LiOH afforded acid 46, which was then transformed into twisted amide 47 via a one-pot activation reaction. Amidation between 47 and 50 delivered tripeptoid 51 and co-product 9. Alkylation of the co-product 9 with 48 followed by treatment with magnesium in MeOH regenerated the starting material 50. Low-cost and highly sustainable peptoid synthesis was realized by our twisted amide-mediated amidation.
Experimental
NMR spectra of new compounds, HPLC charts, and detailed synthetic procedures are provided in the supplementary information (SI).Conclusions
In summary, a robust and practical method for low-cost and sustainable amide bond formation was established. Carboxylic acid was converted to a twisted amide using tosyl isocyanate and bromoacetate. The resulting twisted amide was then reacted with an amine to give the corresponding amide along with the valuable co-product tosylglycine ester. Excess twisted amide was recovered and reused after the amidation. Epimerization-suppressed amidation, application to medium-sized cyclic peptide synthesis, column chromatography-free amide synthesis, and solid-phase amide synthesis were established. Furthermore, the effective use of the co-product tosylglycine ester was demonstrated.The amidation was performed under air, and all the reagents used in this study are inexpensive and readily available. Therefore, the developed method can immediately become a practical amidation strategy that can solve the cost and sustainability issues in amide synthesis.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6ob00340k.Acknowledgements
This work was supported in part by a Grant-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology (MEXT), Japan [grant numbers 17H06401, 19H02840, 22H04901, 23H04882 and 24H00493 (all awarded to H. K.), and 22K05112 and 25K08639 (both awarded to T. K.)] and the Project for Promotion of Cancer Research and Therapeutic Evolution [JP24ama221540, JP25ama221540, and JP26ama221540 (H. K.)] and the Platform Project for Supporting Drug Discovery and Life Science Research [JP24ama121034, JP25ama121034, and JP26ama121034 (H. K.)] from the Japan Agency for Medical Research and Development (AMED), Japan. The support from Tokyo Chemical Industry to T. K. is also greatly appreciated.References
- (a) J. Magano, Org. Process Res. Dev., 2022, 26, 1562 CrossRef CAS; (b) A. Isidro-Llobet, M. N. Kenworthy, S. Mukherjee, M. E. Kopach, K. Wegner, F. Gallou, A. G. Smith and F. Roschangar, J. Org. Chem., 2019, 84, 4615 CrossRef CAS PubMed.
- M. T. Sabatini, L. T. Boulton, H. F. Sneddon and T. D. Sheppard, Nat. Catal., 2019, 2, 10 CrossRef CAS.
- I. T. Horváth, Chem. Rev., 2018, 118, 369 CrossRef PubMed.
- M. Todorovic and D. M. Perrin, Pept. Sci., 2020, 112, e24210 CrossRef CAS.
- W. Muramatsu, T. Hattori and H. Yamamoto, Chem. Commun., 2021, 57, 6346 RSC.
- P. Acosta-Guzmán, A. Ojeda-Porras and D. Gamba-Sánchez, Adv. Synth. Catal., 2023, 365, 4359 CrossRef.
- (a) A. Koyama, T. Kuranaga, T. Suo, R. Morimoto, T. Matsumoto and H. Kakeya, Chem. – Eur. J., 2024, 30, e202403288 CrossRef CAS PubMed; (b) T. Kuranaga, A. Koyama, J. Asano, T. Matsumoto and H. Kakeya, Chem. – Eur. J., 2025, 31, e202500331 CrossRef CAS PubMed.
- G. Meng, J. Zhang and M. Szostak, Chem. Rev., 2021, 121, 12746 CrossRef CAS PubMed.
- T. Tokuda, T. Kuranaga, M. Minote, T. Suo, H. Ikeda, C. Pan and H. Kakeya, Chem. – Eur. J., 2025, 31, e202500357 CrossRef CAS PubMed.
- E. Fischer and W. Lipschitz, Ber. Dtsch. Chem. Ges., 1915, 48, 360 CrossRef CAS.
- S. C. Story and J. V. Aldrich, Int. J. Pept. Protein Res., 1994, 43, 292 CrossRef CAS PubMed.
- R. Knorr, A. Trzeciak, W. Bannwarth and D. Gillessen, Tetrahedron Lett., 1989, 30, 1927 CrossRef CAS.
- P. P. De Castro, G. M. F. Batista, H. F. Dos Santos and G. W. Amarante, ACS Omega, 2018, 3, 3507 CrossRef CAS PubMed.
- L. A. Carpino, J. Am. Chem. Soc., 1993, 115, 4397 CrossRef CAS.
- R. D. Hotchkiss and R. J. Dubos, J. Biol. Chem., 1941, 141, 155 CrossRef CAS.
- F. Xu, B. Nazari, K. Moon, L. B. Bushin and M. R. Seyedsayamdost, J. Am. Chem. Soc., 2017, 139, 9203 CrossRef CAS PubMed.
- R. Behrendt, P. White and J. Offer, J. Pept. Sci., 2016, 22, 4 CrossRef CAS PubMed.
- R. B. Merrifield, J. Am. Chem. Soc., 1963, 85, 2149 CrossRef CAS.
- J. Lenard and A. B. Robinson, J. Am. Chem. Soc., 1967, 89, 181 CrossRef CAS PubMed.
- R. Kalir, A. Warshawsky, M. Fridkin and A. Patchornic, Eur. J. Biochem., 1975, 59, 55 CrossRef CAS PubMed.
- O. Mitsunobu, Synthesis, 1981, 1 CrossRef CAS.
- B. Nyasse, L. Grehn and U. Ragnarsson, Chem. Commun., 1997, 1017 RSC.
- R. J. Simon, R. S. Kania, R. N. Zuckermann, V. D. Huebner, D. A. Jewell, S. Banville, S. Ng, L. Wang, S. Rosenberg and C. K. Marlowe, Proc. Natl. Acad. Sci. U. S. A., 1992, 89, 9367 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |