Open Access Article
Open Access ArticleDesign and synthesis of thiophene-based C2-symmetric chiral phosphoric acids on a decahydroquinoxaline scaffold for stereoselective transformations
Margherita Gazzotti†
 a,
Vincenzo Mirco Abbinante†
a,
Vincenzo Mirco Abbinante† b,
Fabrizio Medici
b,
Fabrizio Medici a,
Sara Ghirardib,
Sergio Rossi
a,
Sara Ghirardib,
Sergio Rossi *a,
Tiziana Benincori
*a,
Tiziana Benincori b,
Roberto Cirilli
b,
Roberto Cirilli c and
Maurizio Benaglia
c and
Maurizio Benaglia *a
*a
aDipartimento di Chimica, Università degli Studi di Milano, Via Golgi 19, 20133 Milano, Italy. E-mail: maurizio.benaglia@unimi.it; sergio.rossi@unimi.it
bDipartimento di Scienza ed Alta Tecnologia dell'Università degli Studi dell'Insubria, Via Valleggio 11, 22100 Como, Italy
cDipartimento del Farmaco, Istituto Superiore di Sanità, Viale Regina Elena 299, 00161 Roma, Italy
First published on 1st April 2026
Abstract
In this study, the design and the synthesis of a thiophene-based phosphoric acid based on a chiral decahydroquinoxaline scaffold derived from enantiopure trans-1,2-diaminocyclohexane was reported. This catalyst was then employed in stereoselective transformations such as the enantioselective Friedel–Crafts reaction of indoles with imines to afford 3-indolyl methanamines. High yields (up to 98%) and high enantioselectivities (up to 98% ee) were obtained. DFT calculations were performed to investigate the key transition states, providing mechanistic insight and confirming the origin and sense of the observed stereochemical outcome.
Introduction
Chiral phosphoric acids (CPAs), an important class of chiral Brønsted acids, have been widely employed as organocatalysts in many stereoselective reactions, facilitating the formation of C–C and C–N bonds.1,2 The remarkable development of CPAs in organic synthesis is primarily attributed to their structural characteristics, which provide precise control over the three-dimensional arrangement of reactants. Chemical activation is enabled by the presence of both a Brønsted acid and Lewis basic sites, while stereochemical control arises from the geometry of the chiral backbone. Such control occurs through the formation of non-covalent interactions with the substrates, including hydrogen bonding, π-stacking, and ion pairing.3Since the early works by Terada4 and Akiyama,5 who reported the use of BINOL-derived CPAs in enantioselective reactions, the application of this class of compounds has expanded rapidly in recent years, with numerous studies exploring the various activation models they can adopt: mono activation, dual activation, and bifunctional activation.6,7 This exceptional adaptability and broad range of activation modes make CPAs a versatile class of catalysts, which has driven the development of numerous chiral phosphoric acid derivatives. Examples of CPAs derived from various chiral backbones, such as BINOL, SPINOL,8,9 VAPOL,10 and TADDOL,11,12 were reported so far, each designed to enhance specific steric or electronic properties, thereby enabling more efficient catalytic reactions.
Despite the success of these CPA derivatives, challenges remain in optimizing their catalytic performance, particularly regarding substrate scope and reaction conditions. Achieving consistent results often demand careful and precise fine-tuning of reaction parameters, even when expanding the substrate scope for the same transformation.13
To address some of these challenges, we recently reported a simple synthetic strategy for the synthesis of new CPAs based on a decahydroquinoxaline scaffold where the catalytic site is incorporated into a 9-membered ring (Fig. 1).14 The exceptional chemical and stereochemical efficiency of this novel class of CPAs was demonstrated in the Friedel–Crafts alkylation of indole with N-tosylimines,15 resulting in the formation of the corresponding adducts in high yields and with enantiomeric excesses often exceeding 90%.
To further advance the development of novel CPA structures, we aimed to improve the electron-donating properties of decahydroquinoxaline-based CPAs. Specifically, we hypothesized that replacing the phenyl groups at the 2- and 3-positions of the decahydroquinoxaline core with thiophene rings would affect the stereoelectronic properties of the catalyst, due to the different electronic character of thiophene compared to benzene ring.
Furthermore, the introduction of two phenyl groups at the ortho positions relative to the catalytic site would generate the necessary steric hindrance to define the chiral pocket of the catalyst.
Results and discussion
A recently reported methodology efficiently synthesizes decahydroquinoxaline-based CPAs14 by formation of the chiral backbone by condensation of an aromatic aldehyde with enantiopure trans-1,2-diaminocyclohexane, followed by a stereoselective imino–pinacol coupling.16 Inspired by this strategy, we proposed a comparable approach to synthesize catalyst 2. The complete synthetic pathway is illustrated in Scheme 1. The synthesis starts from 2,3,5-tribromothiophene 3 which was subjected to the regioselective Suzuki reaction mediated by Pd(PPh3)4 in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of THF
1 mixture of THF![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O in the presence of Na2CO3. The reaction was performed for 24 h and the desired 3-bromo-2,5-diphenylthiophene 4 was obtained in 59% yield.17 Compound 4 was reacted in a copper catalysed nucleophilic substitution with an excess of sodium methoxide,18,19 leading to the formation of 3-methoxy-2,5-diphenyl-thiophene 5 in 62% yield. Reaction of 5 with n-BuLi in the presence of N,N,N′,N’-tetramethyl ethylenediamine (TMEDA), afforded aldehyde (6) in 57% yield. Finally, after a treatment of compound 6 with BBr3 in CH2Cl2, aldehyde 7 was formed in quantitative yield. As expected, since hydroxythiophenes are prone to tautomeric equilibrium, the thermodynamically stable ketonic form 7a of 7 was also observed. The synthesis of the decahydroquinoxaline scaffold was then achieved through a manganese-mediated reductive cyclization of the intermediate 8 obtained by condensation of two molecules of aldehyde 7 with enantiomerically pure (1S,2S)-trans-1,2-diaminocyclohexane yielding (2R,2R,4aS,8aS)-9 in 87% yield.20
H2O in the presence of Na2CO3. The reaction was performed for 24 h and the desired 3-bromo-2,5-diphenylthiophene 4 was obtained in 59% yield.17 Compound 4 was reacted in a copper catalysed nucleophilic substitution with an excess of sodium methoxide,18,19 leading to the formation of 3-methoxy-2,5-diphenyl-thiophene 5 in 62% yield. Reaction of 5 with n-BuLi in the presence of N,N,N′,N’-tetramethyl ethylenediamine (TMEDA), afforded aldehyde (6) in 57% yield. Finally, after a treatment of compound 6 with BBr3 in CH2Cl2, aldehyde 7 was formed in quantitative yield. As expected, since hydroxythiophenes are prone to tautomeric equilibrium, the thermodynamically stable ketonic form 7a of 7 was also observed. The synthesis of the decahydroquinoxaline scaffold was then achieved through a manganese-mediated reductive cyclization of the intermediate 8 obtained by condensation of two molecules of aldehyde 7 with enantiomerically pure (1S,2S)-trans-1,2-diaminocyclohexane yielding (2R,2R,4aS,8aS)-9 in 87% yield.20
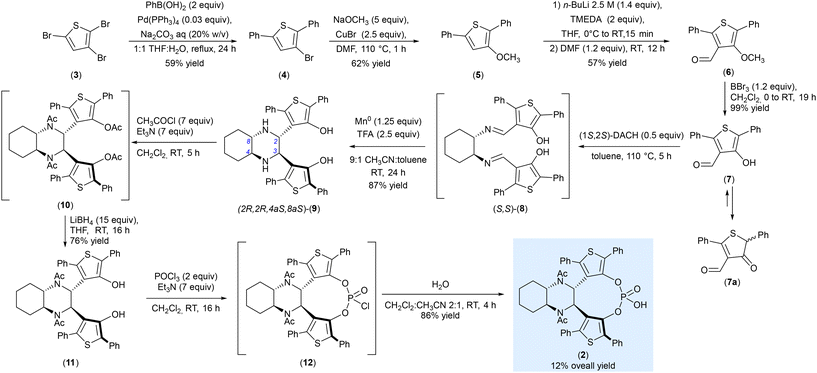 | ||
| Scheme 1 Synthesis of thiophene-based C2-symmetric chiral phosphoric acid (2) based on a decahydroquinoxaline scaffold. | ||
The same synthetic transformation was performed using (1R,2R)-trans-1,2-diaminocyclohexane and the resulting products were then analyzed and compared using enantioselective HPLC coupled with a circular dichroism detector. This analysis confirmed that the two products, (2R,2R,4aS,8aS)-9 derived from (1S,2S)-DACH and (2S,2S,4aR,8aR)-9 derived from (1R,2R)-DACH, are enantiomers, as evidenced by their HPLC traces, which display identical but opposite profiles (Fig. 2). These results clearly highlight that the configuration of the two new stereocenters formed during the pinacol coupling are dictated by the initial configuration of DACH used in the reaction.
In the next step, compound (2R,2R,4aS,8aS)-9 was efficiently protected at the nitrogen atoms as acetyl amides by treating the substrate with an excess of acetyl chloride in the presence of triethylamine. Under these conditions, both NH and OH groups reacted, yielding the tetra-acylated intermediate 10 which was not isolated and directly treated with an excess of LiBH4 in THF to afford desired compound 11 in 76% yield.
The synthesis of the phosphoric acid 2 was finally achieved by reaction of diol 11 with POCl3 in the presence of Et3N following a one-pot-two-steps procedure. The intermediate phosphoryl chloride 12, which was not isolated, was then hydrolysed with water allowing the formation of desired compound 2 in 86% yield. A further treatment with aqueous HCl after chromatographic purification ensures the complete removal of phosphate salts which may be formed during the purification process.21
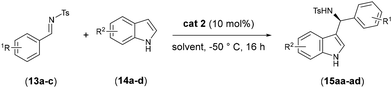
The chemical and stereochemical efficiency of the new chiral Brønsted acid 2 was then investigated in the stereoselective Friedel–Crafts alkylation of indole with N-tosyl imines for the synthesis of enantiopure 3-indolyl methanamine derivatives. In a typical experiment, benzaldehyde-derived N-tosylimines 13a–c (1 equiv.) were reacted with indole 14 (5 equiv.) in the presence of 10 mol% of catalyst, operating at −50 °C. Results are reported in Table 1.
When the reaction was performed in toluene, product 15aa was obtained in 90% yield with 72% ee (entry 1). Notably, replacing toluene with dichloromethane afforded compound 15aa in almost quantitative yield and 92% ee. With the solvent effect established, the scope of the reaction was subsequently examined. N-Tosylimines bearing either electron-withdrawing or electron-donating substituents on the aromatic ring were well tolerated: products 15ba and 15ca were obtained in comparable yields with enantioselectivities of 70% ee and 86% ee, respectively (entries 3 and 4). While the difference in enantioselectivity between compound 15aa and 15ca is modest (92% vs. 86% ee), the more pronounced drop observed for the 4-chloro analogue 15ba suggests that the aryl substituent on the imine plays a role in determining the stereochemical outcome of the reaction, probably due to some electronic interactions with the aromatic rings of the catalysts.
The substrate scope of the phosphoric acid-catalyzed Friedel–Crafts reaction of indoles with N-tosylimines was then further evaluated using substituted indoles bearing either electron-withdrawing or electron-donating groups at the 5-position (entries 5–7). In all cases, the reactions proceeded smoothly to give the corresponding products in high yields and good to excellent enantioselectivities, reaching up to 98% ee when 5-methoxy-1H-Indole 14d was employed (entry 6). In agreement with the trend observed for the imine substituents, electron-donating groups on the indole ring afforded the desired products with higher enantioselectivities, whereas the presence of an electron-withdrawing substituent (as in the case of indole 14a) resulted in the formation of 15ba with only 70% ee.
DFT investigation
DFT calculations were employed in order to investigate the steric properties, the Brønsted acidity, and transition-state geometries of the thiophene-based C2-symmetric chiral phosphoric acid 2 when involved in the Friedel–Crafts alkylation of indole with N-tosylimines. Initial conformational geometries were generated through Monte Carlo conformational analysis using molecular mechanics calculations with the OPLS2005 force field,22 as implemented in the MacroModel package of the Schrödinger suite.23 These structures were subsequently fully optimized at the DFT level using the M06-2X functional with the 6-31G(d) basis set in Gaussian.24 Harmonic vibrational frequency calculations were performed at the same level of theory to confirm the nature of the stationary points, ensuring the absence of imaginary frequencies for local minima and the presence of a single imaginary frequency for transition states. The M06-2X functional was selected due to its proven reliability in describing long-range dispersion interactions compared to the B3LYP functional, and its successful application in previous computational studies on chiral phosphoric acids.14The steric properties of the chiral pocket associated to catalyst 2 were evaluated in terms of buried volume (%Vbur), a steric parameter that has been successfully employed as a key descriptor for quantifying the fraction of space occupied around the catalytic site.25
Compared to the decahydroquinoxaline-based CPA 1, which has a %Vbur of 45.1%, compound 2 exhibits a slightly lower %Vbur value of 40.1% (Fig. 3). Although this corresponds to a slightly larger and more open chiral pocket compared to chiral phosphoric acid 1, the steric environment of catalyst 2 remains within the range characteristic of high-performing phosphoric acids, providing a spatial confinement able to influence the substrate orientation and to promote enantioselective control; as demonstrated by the stereochemical outcome observed in the Friedel–Crafts alkylation of indole with N-tosylimines. Notably, this value is comparable to that of the SPINOL-derived phosphoric acid (%Vbur = 41.1%) and significantly higher than that of the BINOL-derived analogue, which displays a %Vbur of 33%.14
Moreover, dedicated computational studies performed to determine the acidity of catalyst 2. Two complementary approaches were employed: the isodesmic method,26 computed at M06-2X/6-31G(d) level of theory, and the linear free energy solvation relationship (LFESR) model,27 performed at the M06-2X/6-311++G(d,p) level with the SMD(DMSO) solvation model. The results indicate a corrected pKa(isodesmic) in DMSO of 1.00, whereas the LFESR approach yielded slightly higher values of 1.36 in DMSO (pKa(LFESR) CH3CN = 10.99). Overall, the two methods provide consistent trends, confirming the relatively strong Brønsted acidity of catalyst 2 compared to catalyst 1 (pKa(isodesmic) DMSO = 3.97). At the present it is premature to offer a rationalization of this observation, since several factors are likely contributing to the pKa value. Although thiophene is generally considered as an electron-rich heterocycle, it must be noted that sulfur atom is more polarizable than carbon atom, and the 5-membered thiophene rings lead to a larger chiral pocket (see Fig. 3), where the conjugate base of catalyst 2 will be accommodated, and may experience stereoelectronic interactions with other aryl rings that may affect the negative charge stabilization.
After the steric properties of the chiral pocket and the Brønsted acidity of the catalyst were characterized, the origin of stereoselectivity in the enantioselective addition of indoles to N-tosylimines catalyzed by chiral phosphoric acid 2 was investigated. In this transformation, CPAs are known to operate through a bifunctional activation mechanism, in which the imine is activated via hydrogen bonding to the Lewis basic site, thereby polarizing the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N bond and increasing its electrophilicity, while the NH group of the indole forms a hydrogen bond with the Brønsted site of the catalyst, aligning the nucleophilic C3 of the indole for enantioselective attack.8,28
N bond and increasing its electrophilicity, while the NH group of the indole forms a hydrogen bond with the Brønsted site of the catalyst, aligning the nucleophilic C3 of the indole for enantioselective attack.8,28
Although decahydroquinoxaline-based CPAs feature an additional amidic functionality, allowing several possible coordination modes, the classical model has been shown to be energetically preferred.14 For this reason, only this coordination mode was considered in this computational study. In Fig. 4 the transition states corresponding to eight possible conformers are reported.
DFT calculations indicate that transition states in which the imine adopts the E configuration are consistently more stable than those involving the Z configuration. In particular, TS3-E-endo-(S), leading to the experimentally observed (S)-15aa product, is calculated to be 1.35 kcal mol−1 lower in energy than TS4-E-endo-(R). Interestingly, in contrast to traditional CPAs based on the decahydroquinoxaline scaffold, the new C–C bond formation occurs according to an endo orientation, in which the imine C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N bond superimposes with the five-membered ring of the indole, likely as a result of improved accommodation within a less sterically congested chiral pocket of the catalyst.
N bond superimposes with the five-membered ring of the indole, likely as a result of improved accommodation within a less sterically congested chiral pocket of the catalyst.
Gibbs free energies computed a M06-2X/6-311G(++)(2d,2p) PCM (dichloromethane)//M06-2X/6-31G(d) were used to estimate an enantiomeric excess of 91% in favour of the (S)-15aa enantiomer for catalyst 2 at −50 °C in DCM. The ee was calculated using the Eyring equation, relating the relative free energies of the transition states to the corresponding rate constants. This theoretical prediction is in perfect agreement with the experimentally observed enantioselectivity of 92% obtained under the same conditions.
Conclusions
In this work, we have developed a new example of chiral phosphoric acid based on a decahydroquinoxaline scaffold29 which incorporates two thiophene rings.The chiral backbone was synthesized through the condensation of aldehyde 7 with enantiopure trans-1,2-diaminocyclohexane 6, followed by a stereoselective intramolecular pinacol coupling. The optical purity of a key intermediate 9 was confirmed through HPLC analysis with a CD detector.
The catalytic performance of the resulting CPA was evaluated in the stereoselective Friedel–Crafts alkylation of indole with N-tosylimines, resulting in the formation of the desired products with yields up to 96% and up to 98% enantiomeric excesses. DFT calculations further showed that the transition state leading to the experimentally observed (S)-product is lower in energy than that of the competing (R)-pathway, in line with the observed experimental data. Gibbs free energies computed for all relevant transition states were used to estimate the enantiomeric excess, yielding a value in good agreement with the experimental results.
These findings demonstrate that the new CPA exhibits both high catalytic activity and predictable stereocontrol. Ongoing studies are focused on further optimizing catalyst structure and exploring the application of these catalysts in additional stereoselective transformations to expand their utility in synthetic organic chemistry.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Computational data presented in the study are openly available in Dataverse UNIMI at: https://doi.org/10.13130/RD_UNIMI/SVQMPB.Supplementary information is available. See DOI: https://doi.org/10.1039/d6ob00309e.
Acknowledgements
S. Rossi and M. Benaglia thank MUSA – Multilayered Urban Sustainability Action – project, funded by the European Union – NextGenerationEU, under the National Recovery and Resilience Plan (NRRP) Mission 4 Component 2 Investment Line 1.5: Strengthening of research structures and creation of R&D “innovation ecosystems”, set up of “territorial leaders in R&D”. S. Rossi thanks MUR for the project PRIN 2022 “Enabling technologies for sustainable and innovative catalytic transformations – BEST-CAT” (CUP G53D23003260006) and University of Milan grant PSR2025 “Catalytic approaches to the sustainable synthesis of high added-value fine chemicals. M. Gazzotti thanks University of Milan for a PhD fellowship.S. Rossi, M. Benaglia and T. Benincori thank Project 6154644 – GREEN-TECH of Regione Lombardia (Bando collabora e Innova).
Computational studies were performed using INDACO Platform, which is a project of High Performance Computing at the University of Milan. Mass spectrometry analyses were performed at the MS facility of the Unitech COSPECT at the University of Milan (Italy). Scientific support from CRIETT centre of University of Insubria (instrument code: MAC01, MAC15) is greatly acknowledged.
References
- A. G. Woldegiorgis, Z. Han and X. Lin, J. Mol. Struct., 2024, 1297, 136919 CrossRef CAS.
- E. I. Jimenez, Org. Biomol. Chem., 2023, 21, 3477–3502 RSC.
- X. del Corte, E. Martínez de Marigorta, F. Palacios, J. Vicario and A. Maestro, Org. Chem. Front., 2022, 9, 6331–6399 RSC.
- D. Uraguchi and M. Terada, J. Am. Chem. Soc., 2004, 126, 5356–5357 CrossRef CAS PubMed.
- T. Akiyama, J. Itoh, K. Yokota and K. Fuchibe, Angew. Chem., Int. Ed., 2004, 43, 1566–1568 CrossRef CAS PubMed.
- R. Maji, S. C. Mallojjala and S. E. Wheeler, Chem. Soc. Rev., 2018, 47, 1142–1158 RSC.
- D. Parmar, E. Sugiono, S. Raja and M. Rueping, Chem. Rev., 2014, 114, 9047–9153 CrossRef CAS PubMed.
- F. Xu, D. Huang, C. Han, W. Shen, X. Lin and Y. Wang, J. Org. Chem., 2010, 75, 8677–8680 CrossRef CAS PubMed.
- A. Rahman and X. Lin, Org. Biomol. Chem., 2018, 16, 4753–4777 RSC.
- W. Wulff and A. Desai, Synthesis, 2010, 3670–3680 CrossRef.
- T. Akiyama, Y. Saitoh, H. Morita and K. Fuchibe, Adv. Synth. Catal., 2005, 347, 1523–1526 CrossRef CAS.
- A. Voituriez and A. B. Charette, Adv. Synth. Catal., 2006, 348, 2363–2370 CrossRef CAS.
- J. P. Reid and J. M. Goodman, Chemistry, 2017, 23, 14248–14260 CrossRef CAS PubMed.
- M. Gazzotti, M. Orlandi, S. Rossi, L. Raimondi and M. Benaglia, ChemistryEurope, 2025, 202500234 CrossRef.
- Q. Kang, Z-A. Zhao and S-L. You, J. Am. Chem. Soc., 2007, 129(6), 1484–1485 CrossRef CAS PubMed.
- For a electrochemical stereoselective imino pinacol coupling see: M. Gazzotti, F. Medici, V. Chiroli, L. Raimondi, S. Rossi and M. Benaglia, Beilstein J. Org. Chem., 2025, 21, 1897–1908 CrossRef CAS PubMed; See also CrossRef PubMed; F. Medici, S. Resta, A. Puglisi, S. Rossi, L. Raimondi and M. Benaglia, Molecules, 2021, 26(22), 6968 CrossRef PubMed.
- S. Gabrieli, R. Cirilli, T. Benincori, M. Pierini, S. Rizzo and S. Rossi, Eur. J. Org. Chem., 2017, 861–870 CrossRef CAS.
- M. A. Keegstra, T. H. A. Peters and L. Brandsma, Tetrahedron, 1992, 48, 3633–3652 CrossRef CAS.
- L. D. Peeters, S. G. Jacobs, W. Eevers and H. J. Geise, Tetrahedron, 1994, 50, 11533–11540 CrossRef CAS.
- G. J. Mercer and M. S. Sigman, Org. Lett., 2003, 5, 1591–1594 CrossRef CAS PubMed.
- B. List, M. Klussmann, L. Ratjen, S. Hoffmann, V. Wakchaure and R. Goddard, Synlett, 2010, 2189–2192 Search PubMed.
- C. Lu, C. Wu, D. Ghoreishi, W. Chen, L. Wang, W. Damm, G. A. Ross, M. K. Dahlgren, E. Russell, C. D. Von Bargen, R. Abel, R. A. Friesner and E. D. Harder, J. Chem. Theory Comput., 2021, 17, 4291–4300 CrossRef CAS PubMed.
- Schrödinger Release 2024-1: MacroModel, Schrödinger, LLC, New York, NY, 2024 Search PubMed.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, G. A. Petersson, H. Nakatsuji, X. Li, M. Caricato, A. V. Marenich, J. Bloino, B. G. Janesko, R. Gomperts, B. Mennucci, H. P. Hratchian, J. V. Ortiz, A. F. Izmaylov, J. L. Sonnenberg, D. Williams-Young, F. Ding, F. Lipparini, F. Egidi, J. Goings, B. Peng, A. Petrone, T. Henderson, D. Ranasinghe, V. G. Zakrzewski, J. Gao, N. Rega, G. Zheng, W. Liang, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, K. Throssell, J. A. Montgomery, Jr., J. E. Peralta, F. Ogliaro, M. J. Bearpark, J. J. Heyd, E. N. Brothers, K. N. Kudin, V. N. Staroverov, T. A. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. P. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, J. M. Millam, M. Klene, C. Adamo, R. Cammi, J. W. Ochterski, R. L. Martin, K. Morokuma, O. Farkas, J. B. Foresman and D. J. Fox, Gaussian 16, Revision C.01, Gaussian, Inc., Wallingford CT, 2019 Search PubMed.
- A. Poater, B. Cosenza, A. Correa, S. Giudice, F. Ragone, V. Scarano and L. Cavallo, Eur. J. Inorg. Chem., 2009, 1759–1766 CrossRef CAS.
- C. Yang, X. S. Xue, J. L. Jin, X. Li and J. P. Cheng, J. Org. Chem., 2013, 78, 7076–7085 CrossRef CAS PubMed.
- M. Busch, E. Ahlberg, E. Ahlberg and K. Laasonen, ACS Omega, 2022, 7, 17369–17383 CrossRef CAS PubMed.
- L. Simon and J. M. Goodman, J. Org. Chem., 2010, 75, 589–597 CrossRef CAS PubMed.
- For the use of chiral decahydroquinoxaline-based phosphoroamidite see: B. Zhao, Z. Wang and K. Ding, Adv. Synth. Catal., 2006, 348, 1049–1057 CrossRef CAS.
Footnote |
| † The two authors have equally contributed to the work. |
| This journal is © The Royal Society of Chemistry 2026 |