Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
An asymmetric tandem reaction of dicyanoalkenes with conjugated sulfinyl imino esters
Francesc Ferrer-Péreza,
Marcos Escolanoa,
Daniel Gaviñaa,
Santiago Díaz-Oltra a,
Amparo Sanz-Marco
a,
Amparo Sanz-Marco b,
María Sánchez-Roselló
b,
María Sánchez-Roselló *a and
Carlos del Pozo
*a and
Carlos del Pozo *a
*a
aDepartment of Organic Chemistry, University of Valencia, Avda. Vicent Andrés Estellés 22, 46100-Burjasot-Valencia, Spain. E-mail: carlos.pozo@uv.es; maria.sanchez-rosello@uv.es
bDepartment of Organic Chemistry, University of Valencia, Doctor Moliner 50, 46100-Burjasot-Valencia, Spain
First published on 16th March 2026
Abstract
An asymmetric tandem reaction of α,α-dicyanoalkenes and enantiomerically pure α,β-unsaturated N-sulfinyl imino esters is reported herein. It involves a four-step protocol that ends up in the generation of a new family of tetracyclic products in a diastereoselective fashion. The overall process takes place with good yields and moderate to good enantioselectivities, and entails the formation of three bonds and four stereocenters. A plausible reaction mechanism has also been proposed.
Introduction
Tandem reactions, categorized as a series of chemical transformations that follow one another in a defined order, have revolutionized the field of organic chemistry. They enable us to create molecular complexity in a single chemical operation, thereby avoiding time-consuming purification and intermediate isolation processes and approaching the concept of ideal synthesis introduced by Hendrickson several decades ago.1 It is known that nature uses this principle in the highly efficient synthesis of biomolecules with extraordinary selectivity. Moreover, the concept of tandem reactions has been around for more than a century, the Robinson annulation being one of the earliest and more emblematic examples.2 However, it was not until the end of the last century that tandem reactions gained exposure in organic synthesis, standing nowadays at the forefront of chemical synthesis. Additionally, the interest in combining asymmetric processes with tandem reactions is obvious, since multiple stereogenic centers can be created in a single synthetic step.3The ambident character of 1,1-dicyanoalkenes bearing enolizable sites converts them into ideal reagents to engage in domino processes, since they can react as nucleophiles through the α- and γ-positions and as electrophiles through the β-position.4 Reactions that take advantage of the excellent vinylogous donor properties of dicyanoalkenes start with their reaction with an electrophile, ending up with a remaining conjugated malononitrile moiety, suitable for further transformations in a tandem fashion. This reactivity shows the usefulness of the vinylogy principle for the design of tandem protocols. A wide variety of electrophiles have been combined with 1,1-dicyanoalkenes in tandem protocols such as 2-hydroxycinnamaldehydes,5 enynals,6 2,2-disubstituted cyclopentene-1,3-diones,7 5-vinyloxazolidine-2,4-diones,8 nitroalkenes,9 α-succinimide-substituted allenoates,10 3-ethynyl-2-oxoindolin-3-yl acetates,11 4-unsaturated isoxazol-5-ones,12 α-vinyl enals,13 2-mercaptoquinoline-3-carbaldehydes,14 N-protected methyleneindolinones,15 2-pyrrole benzaldehydes,16 3-alkenyl-oxindoles,17 methylene cyclopropanes,18 δ-sulfonamido substituted enones,19 allenyllic alcohols,20 2-nitrobenzofuranes21 and trifluoromethyl aryl ketones,22 rendering new families of heterocycles and carbocycles.
Despite the widespread usage of 1,1-dicyanoalkenes in tandem protocols, their reactivity with conjugated sulfinyl imines as electrophilic partners has only been evaluated by our research group. On the one hand, we found that the reaction of 1,1-dicyanoalkenes with fluorinated conjugated N-tert-butylsulfinyl imines in the presence of DBU gave rise to polycyclic trifluoromethyl arenes by means of a new cycloaromatization cascade process. This took place in isopropanol with concomitant elimination of both cyano groups and the sulfinyl amine moiety (Scheme 1A).23 On the other hand, the use of fluorinated conjugated N-p-tolylsulfinyl imines in dichloromethane triggered a divergent reactivity pathway that led to tetracyclic compounds through an azetidinimine rearrangement, with elimination of the sulfinyl amine moiety while retaining in this case both cyano groups (Scheme 1B).24
In this work, we found that the reaction of 1,1-dicyanoalkenes with conjugated N-sulfinyl imino esters follows, again, a different reactivity pathway, rendering a new family of polycyclic heterocycles and increasing the structural diversity of the overall process just by changing the substitution pattern of the starting conjugated sulfinyl imines (Scheme 1C). The optimization of this asymmetric tandem process and the evaluation of its scope are reported herein. A plausible mechanistic explanation of the divergent reactivity observed is also discussed.
Results and discussion
Our study was initiated with dicyanoalkene 1a, derived from 1-tetralone, and enantiomerically pure conjugated N-tert-butyl- and N-p-tolylsulfinyl α-imino esters 2a, b as model substrates. Previous work from our laboratory demonstrated that DBU was the base of choice to perform the reaction of 1,1-dicyanoalkenes with fluorinated conjugated sulfinyl imines. Therefore, a mixture of compounds 1a and 2a was treated with DBU in dichloromethane at room temperature. After 16 hours, the novel tetracyclic derivative 3a was isolated in 57% yield and 86![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 14 enantiomeric ratio (Table 1, entry 1). The unexpected formation of compound 3a reveals a new divergent pathway in the reaction of dicyanoalkenes with conjugated N-sulfinyl imino esters, ending up with the creation of three new bonds and four stereocenters in a very selective manner. Other organic and inorganic bases, such as Et3N, Na2CO3 and NaH, were also tested in this tandem process with little success (Table 1, entries 2–4). The reaction with N-p-tolylsulfinyl imino ester 2b took place less efficiently, affording product 3a in 36% yield and 77
14 enantiomeric ratio (Table 1, entry 1). The unexpected formation of compound 3a reveals a new divergent pathway in the reaction of dicyanoalkenes with conjugated N-sulfinyl imino esters, ending up with the creation of three new bonds and four stereocenters in a very selective manner. Other organic and inorganic bases, such as Et3N, Na2CO3 and NaH, were also tested in this tandem process with little success (Table 1, entries 2–4). The reaction with N-p-tolylsulfinyl imino ester 2b took place less efficiently, affording product 3a in 36% yield and 77![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 23 enantiomeric ratio (Table 1, entry 5). The influence of the solvent was examined next. However, the efficiency of the tandem protocol did not improve with any of the different types of solvents tested (Table 1, entries 6–9), when compared with the reaction in dichloromethane.
23 enantiomeric ratio (Table 1, entry 5). The influence of the solvent was examined next. However, the efficiency of the tandem protocol did not improve with any of the different types of solvents tested (Table 1, entries 6–9), when compared with the reaction in dichloromethane.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a
a
| Entry | 2 | Solvent | Base | T (°C) | Time (h) | 3ab,c (%) | erd |
|---|---|---|---|---|---|---|---|
| a Reactions were performed with 1a (0.4 mmol), 2a/b (0.2 mmol) and base (0.4 mmol) in the corresponding solvent (0.03 M), temperature and time.b Isolated yields after flash column chromatography.c In all cases, compound 3a was obtained with excellent diastereoselectivity.d Enantiomeric ratios were determined by HPLC analysis on a chiral stationary phase (see the SI for details). | |||||||
| 1 | 2a | CH2Cl2 | DBU | 25 | 16 | 57 | 86![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 14 14 |
| 2 | 2a | CH2Cl2 | Et3N | 25 | 16 | — | — |
| 3 | 2a | CH2Cl2 | Na2CO3 | 25 | 16 | — | — |
| 4 | 2a | CH2Cl2 | NaH | 25 | 16 | 18 | 76![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 24 24 |
| 5 | 2b | CH2Cl2 | DBU | 25 | 16 | 36 | 77![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 23 23 |
| 6 | 2a | i-PrOH | DBU | 25 | 16 | 21 | 79![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 21 21 |
| 7 | 2a | Toluene | DBU | 25 | 16 | 29 | 82![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18 18 |
| 8 | 2a | THF | DBU | 25 | 16 | 25 | 78![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 22 22 |
| 9 | 2a | MeCN | DBU | 25 | 16 | 22 | 69![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 31 31 |
| 10 | 2a | CH2Cl2 | DBU | −78 to rt | 16 | 15 | 84![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16 16 |
| 11 | 2a | CH2Cl2 | DBU | 0 | 16 | 39 | 92![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8 8 |
| 12 | 2a | CH2Cl2 | DBU | 0 | 48 | 68 | 93![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7 7 |
Moreover, in order to enhance the enantioselectivity, the reaction was performed at low temperature. In this context, when starting the process at −78 °C and allowing the reaction mixture to reach room temperature, the enantiomeric ratio of the final product (84![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16 er) was comparable to that obtained at room temperature, although with a noticeable drop in yield (Table 1, entry 10). When the reaction was performed at 0 °C, the enantioselectivity of the process increased until 92
16 er) was comparable to that obtained at room temperature, although with a noticeable drop in yield (Table 1, entry 10). When the reaction was performed at 0 °C, the enantioselectivity of the process increased until 92![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8 er, albeit with a poor 39% chemical yield (Table 1, entry 11). Finally, by extending the reaction time until 48 h, it was possible to increase the final yield to 68%, with an excellent enantiomeric ratio of 93
8 er, albeit with a poor 39% chemical yield (Table 1, entry 11). Finally, by extending the reaction time until 48 h, it was possible to increase the final yield to 68%, with an excellent enantiomeric ratio of 93![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
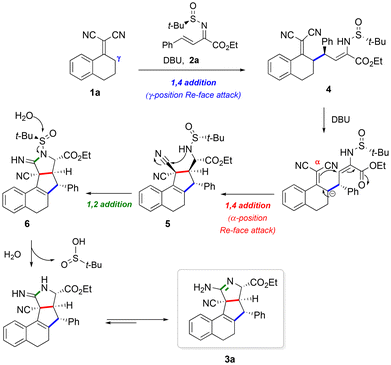
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7 (Table 1, entry 12). In light of this study, we concluded that the optimal conditions for the new asymmetric tandem reaction involved the use of tert-butylsulfinyl imine 2a and DBU as a base, in dichloromethane at 0 °C for 48 h (Table 1, entry 12). These reaction conditions were further applied to other dicyanoalkenes 1 and conjugated sulfinyl imino esters 2 in order to evaluate the scope of our tandem process. The results of this study are summarized in Table 2.
7 (Table 1, entry 12). In light of this study, we concluded that the optimal conditions for the new asymmetric tandem reaction involved the use of tert-butylsulfinyl imine 2a and DBU as a base, in dichloromethane at 0 °C for 48 h (Table 1, entry 12). These reaction conditions were further applied to other dicyanoalkenes 1 and conjugated sulfinyl imino esters 2 in order to evaluate the scope of our tandem process. The results of this study are summarized in Table 2.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a,b,c,d
a,b,c,d
| a Unless otherwise noted, reactions were carried out with 1 (0.4–1.0 mmol), 2 (0.2–0.5 mmol), and DBU (2 equiv.) in dichloromethane (2 mL) at 0 °C for 48 h.b Isolated yields after flash column chromatography.c Enantiomeric ratios were determined by HPLC analysis on a chiral stationary phase (see the SI for details).d Diastereoisomeric ratios were determined by 1H-NMR of the crude reaction mixtures. |
|---|
 |
Initially, we evaluated the asymmetric tandem reaction with conjugated sulfinylimine 2a (R2 = Ph, R3 = Et) and several substituted bicyclic 1,1-dicyanoalkenes 1 derived from 1-tetralone (X = CH2). Compared to the unsubstituted compound 3a (R1 = H, 93![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7 er), the electron-donating methoxy group at the 6 position produced a slightly lower er value (3b, 90
7 er), the electron-donating methoxy group at the 6 position produced a slightly lower er value (3b, 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 er); however, the bromine electron-withdrawing group provided the best enantiomeric ratio (3c, 97
10 er); however, the bromine electron-withdrawing group provided the best enantiomeric ratio (3c, 97![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 er), and the methoxy group at the 8 position led to comparable results (3d, 92
3 er), and the methoxy group at the 8 position led to comparable results (3d, 92![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8 er). The presence of two methoxy substituents at the 6 and 7 positions of the starting dicyanoalkene led to a significant drop in enantioselectivity (3e, 84
8 er). The presence of two methoxy substituents at the 6 and 7 positions of the starting dicyanoalkene led to a significant drop in enantioselectivity (3e, 84![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16 er), while substitution at the 5 position completely prevented the tandem reaction, probably due to steric issues. 1,1-Dicyanoalkenes derived from 4-chromanone (X = O) and 4-thiochromanone (X = S) were also compatible with the reaction, giving rise to the corresponding tetracycles 3g (95
16 er), while substitution at the 5 position completely prevented the tandem reaction, probably due to steric issues. 1,1-Dicyanoalkenes derived from 4-chromanone (X = O) and 4-thiochromanone (X = S) were also compatible with the reaction, giving rise to the corresponding tetracycles 3g (95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 er) and 3h (78
5 er) and 3h (78![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 22 er), respectively, the latter being quite less efficient in terms of enantiocontrol. In all cases, the tandem reaction was highly diastereoselective (up to 20
22 er), respectively, the latter being quite less efficient in terms of enantiocontrol. In all cases, the tandem reaction was highly diastereoselective (up to 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr) and the final products were obtained in good yields (49–68%) (Table 2), considering that the tandem process comprises four chemical transformations.
1 dr) and the final products were obtained in good yields (49–68%) (Table 2), considering that the tandem process comprises four chemical transformations.
Starting dicyanoalkenes 1 bearing heterocyclic five-membered rings were also examined. In this context, substrates containing a furane and a thiophene moiety behaved as expected, giving rise to tetracycles 3i and 3j with moderate er values (79![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 21 and 89
21 and 89![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 11, respectively). However, the dicyanoalkene containing a pyrrole moiety did not react with the conjugated sulfinyl imine 2a (Table 2).
11, respectively). However, the dicyanoalkene containing a pyrrole moiety did not react with the conjugated sulfinyl imine 2a (Table 2).
The scope of our tandem protocol with respect to the sulfinyl imine counterparts (2) in their reaction with bicyclic dicyanoalkenes 1a (derived from 1-tetralone) and 1g (derived from 4-chromanone) was examined next. Regarding the ester substitution (R3), isopropyl and methyl esters were good partners for the tandem reaction, albeit providing lower er values for the tetracyclic products 3l (85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 er) and 3m (90
15 er) and 3m (90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 er) than the ethyl ester derivative (3a, 93
10 er) than the ethyl ester derivative (3a, 93![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7 er). With respect to the β-position (R2), the tandem protocol was compatible with aromatic substituents bearing both electron-withdrawing (3n) and electron-donating (3o) substituents, as well as naphthyl groups (3p, q) and heteroaromatic substituents such as the 2-thienyl group (3r). The process was also tolerated with an aliphatic substituent (3s) at the R2 position of the conjugated sulfinyl imino esters 2. In these cases, the expected tetracyclic products were obtained in good yields (45–75%) and enantioselectivity ranging from 78
7 er). With respect to the β-position (R2), the tandem protocol was compatible with aromatic substituents bearing both electron-withdrawing (3n) and electron-donating (3o) substituents, as well as naphthyl groups (3p, q) and heteroaromatic substituents such as the 2-thienyl group (3r). The process was also tolerated with an aliphatic substituent (3s) at the R2 position of the conjugated sulfinyl imino esters 2. In these cases, the expected tetracyclic products were obtained in good yields (45–75%) and enantioselectivity ranging from 78![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 22 to 92
22 to 92![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8 er (Table 2).
8 er (Table 2).
We also tested a monocyclic 1,1-dicyanoalkene derived from propiophenone (1l). This reacted with sulfinyl imino ester 2a, affording the expected product 3t in good yield but with a dramatic drop in enantioselectivity (61![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 39 er), compared with the bicyclic dicyanoalkenes. This difference was probably due to the lower conformational flexibility of the bicyclic substrates that confers rigidity to the transition state, allowing for a better enantiocontrol. Finally, dicyanoalkene 1m (derived from 2-tetralone), which possesses two reactive sites, also provided the desired product 3u, by means of its reaction, through the most acidic benzylic position, with conjugated sulfinyl imino ester 2a (Table 2).25
39 er), compared with the bicyclic dicyanoalkenes. This difference was probably due to the lower conformational flexibility of the bicyclic substrates that confers rigidity to the transition state, allowing for a better enantiocontrol. Finally, dicyanoalkene 1m (derived from 2-tetralone), which possesses two reactive sites, also provided the desired product 3u, by means of its reaction, through the most acidic benzylic position, with conjugated sulfinyl imino ester 2a (Table 2).25
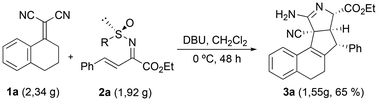
To assess the utility of this tandem protocol, a gram-scale experiment was carried out. Thus, starting from 2.34 g (12 mmol) of 1,1-dicyanoalkene 1a and 1.92 g (6 mmol) of conjugated N-tert-butylsulfinyl imino ester 2a, 1.55 g of tetracyclic derivative 3a were obtained, i.e. this product was isolated in 65% overall yield after four reaction steps that occurred in a tandem fashion (Scheme 2). Moreover, no erosion of the enantioselectivity was observed.
The structure of final products 3 was determined by means of an X-ray diffraction analysis of compound (±)-3m.26 Furthermore, the absolute configuration of the four newly created stereocenters was unequivocally assigned by electronic circular dichroism analysis of compound 3c, assuming identical stereochemical outcome for all other tetracycles 3, as displayed in Table 2 (see the SI for details).
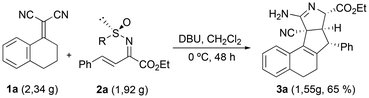
Once the unexpected molecular skeleton of products 3 was confirmed, a mechanistic proposal to explain their formation was established, as outlined in Scheme 3. The tandem reaction would start with the base-mediated conjugate addition of the 1,1-dicyanoalkene 1 through its γ-position to the sulfinyl imino ester 2, rendering enamino ester 4. This 1,4-addition reaction would take place at the opposite face of the bulky tert-butyl substituent at the sulfinyl group, providing the configuration of intermediate 4 at the Ph-containing stereocenter. This stereochemical outcome is based on theoretical calculations performed in our previous work with fluorinated conjugated sulfinyl imines.23 Then, deprotonation of intermediate 4 would generate an anionic species that, this time, would undergo intramolecular conjugate addition through the α-position of the dicyanoalkene to the conjugated ester moiety, affording the fused five-membered ring 5. In this case, the nucleophilic addition would take place at the upper face (Re-face attack) since the bottom one would be shielded by the phenyl group at the adjacent carbon. Subsequently, the N-sulfinyl amine would preferentially react with the upwards nitrile moiety, giving rise to amidine 6, with four stereocenters. Finally, hydrolysis of the sulfinyl group would deliver the final tetracyclic heterocycle 3 after tautomeric equilibrium (Scheme 3).
Conclusions
In conclusion, the reaction of 1,1-dicyanoalkenes 1 with enantiomerically pure conjugated N-sulfinyl α-imino esters 2 has been studied in this work. Thus, in the presence of DBU as a base, enantiomerically enriched tetracyclic compounds 3 were formed in good yields by means of a tandem process involving four chemical steps, with concomitant elimination of the sulfinyl group that acted as a chiral inducer. The conformational constraint imposed by the bicyclic dicyanoalkene counterpart allowed good stereochemical control of the process, rendering the final products with moderate to good enantiomeric ratios and high diastereoselectivity.This work complements our previous studies regarding the divergent reactivity showed by 1,1-dicyanoalkenes and conjugated N-sulfinyl ketimines, which allowed us to synthesize structurally diverse collections of compounds, and represents an example of diversity-oriented synthesis.
Experimental procedure
General procedure for the synthesis of rearranged polycycles 3
To a stirred solution of imine 2 (1 equiv.) and dicyanoalkene 1 (2 equiv.) in DCM (0.03 M), 2 equivalents of DBU were added. After stirring for 48 h at 0 °C, the solvents were evaporated under vacuum and the crude product purified by means of flash column chromatography on silica gel using mixtures of n-hexane and ethyl acetate as eluents.Ethyl (7R,7aR,8S,10aS)-10-amino-10a-cyano-7-phenyl-5,6,7,7a,8,10a-hexahydrobenzo[6,7]indeno[1,2-c]pyrrole-8-carboxylate (3a)
Starting from imine 2a (61 mg, 0.2 mmol) and dicyanoalkene 1a (78 mg, 0.4 mmol), following the general procedure indicated before, 3a was obtained as inseparable mixture of diastereoisomers as a brown solid (54 mg, 68%, 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr, 93
1 dr, 93![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7 er) after purification by column chromatography with Hex
7 er) after purification by column chromatography with Hex![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc (1
EtOAc (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) as an eluent. The er value was determined by HPLC analysis using a Chiralcel OD-H column (hexane
1) as an eluent. The er value was determined by HPLC analysis using a Chiralcel OD-H column (hexane![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) isopropanol 90
isopropanol 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10); flow rate = 1.0 mL min−1, tmajor = 29.5 min, tminor = 52.0 min. M.p. = 194.3–195 °C. [α]25D = +50.6 (c 0.1, CHCl3). 1H NMR (300 MHz, CDCl3) δ 7.85 (d, J = 7.5 Hz, 1H), 7.37–7.27 (m, 4H), 7.25–7.18 (m, 4H), 4.52 (d, J = 4.5 Hz, 1H), 4.26–4.19 (m, 2H), 4.03 (d, J = 3.1 Hz, 1H), 3.63 (dd, J = 4.5, 3.1 Hz, 1H), 2.95–2.76 (m, 2H), 2.17 (dd, J = 10.4, 5.5 Hz, 2H), 1.29 (t, J = 7.1 Hz, 3H). 13C NMR (75 MHz, CDCl3) δ 172.0, 162.4, 147.4, 140.2, 136.6, 130.3, 129.6, 129.2 (2C), 128.8, 128.2, 127.7, 127.7 (2C), 127.1, 122.2, 118.9, 75.4, 62.8, 61.6, 60.6, 58.4, 28.3, 23.7, 14.2. HRMS (ESI/Q-TOF) m/z calculated for C25H24N3O2+ [M + H]+: 398.1863 found 398.1859.
10); flow rate = 1.0 mL min−1, tmajor = 29.5 min, tminor = 52.0 min. M.p. = 194.3–195 °C. [α]25D = +50.6 (c 0.1, CHCl3). 1H NMR (300 MHz, CDCl3) δ 7.85 (d, J = 7.5 Hz, 1H), 7.37–7.27 (m, 4H), 7.25–7.18 (m, 4H), 4.52 (d, J = 4.5 Hz, 1H), 4.26–4.19 (m, 2H), 4.03 (d, J = 3.1 Hz, 1H), 3.63 (dd, J = 4.5, 3.1 Hz, 1H), 2.95–2.76 (m, 2H), 2.17 (dd, J = 10.4, 5.5 Hz, 2H), 1.29 (t, J = 7.1 Hz, 3H). 13C NMR (75 MHz, CDCl3) δ 172.0, 162.4, 147.4, 140.2, 136.6, 130.3, 129.6, 129.2 (2C), 128.8, 128.2, 127.7, 127.7 (2C), 127.1, 122.2, 118.9, 75.4, 62.8, 61.6, 60.6, 58.4, 28.3, 23.7, 14.2. HRMS (ESI/Q-TOF) m/z calculated for C25H24N3O2+ [M + H]+: 398.1863 found 398.1859.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: detailed experimental procedures, compound characterization, NMR spectra and copies of HPLC chromatograms. See DOI: https://doi.org/10.1039/d6ob00277c.CCDC 2513043 (3m) contains the supplementary crystallographic data for this paper.26
Acknowledgements
We gratefully thank the Spanish Ministerio de Ciencia e Innovación (PID2023-152270NB–I00) and Conselleria d'Innovació, Universitats, Ciència i Societat Digital of the Generalitat Valenciana (CIAICO/2022/216) for financial support. SCSIE and ICTS NANBIOSIS U26 (Universidad de Valencia) are gratefully acknowledged for the equipment employed.References
- J. B. Hendrikson, J. Am. Chem. Soc., 1975, 97, 5784 CrossRef.
- W. S. Rapson and R. Robinson, J. Chem. Soc., 1935, 1285 RSC.
- For recent reviews regarding asymmetric tandem reactions, see: (a) H. Pellissier, Adv. Synth. Catal., 2023, 365, 620 CrossRef CAS; (b) H. Pellissier, Adv. Synth. Catal., 2023, 365, 768 CrossRef CAS; (c) L. Bai and X. Jiang, Chem Catal., 2023, 3, 100752 CAS; (d) L. Zou, Y. Gao, Q. Zhang, X.-Y. Ye, T. Xie, L.-W. Ye and Y. Wang, Chem. – Asian J., 2023, 18, e202300617 CrossRef CAS PubMed; (e) R. Westphal, E. V. Filho, F. Medici, M. Benaglia and S. J. Greco, Synthesis, 2022, 2927 CAS; (f) R.-X. Liang and Y.-X. Jia, Acc. Chem. Res., 2022, 55, 734 CrossRef CAS PubMed; (g) R. K. Maurya, D. Sharma, S. Kumari, R. Chatterjee, M. Khatravath and R. Dandela, ChemistrySelect, 2022, 7, e202201408 CrossRef CAS; (h) Y. Hussain, Tamanna, M. Sharma, A. Kumar and P. Chauan, Org. Chem. Front., 2022, 9, 572 RSC; (i) B. Delayre, Q. Wang and J. Zhu, ACS Cent. Sci., 2021, 7, 559 CrossRef CAS PubMed; (j) M. Zhang, Y. Gong, W. Zhou, Y. Zhou and X.-L. Liu, Org. Chem. Front., 2021, 8, 3968 RSC; (k) Y.-X. Song and D.-M. Du, Adv. Synth. Catal., 2021, 363, 4667 CrossRef CAS; (l) F.-L. Hong and L.-W. Ye, Acc. Chem. Res., 2020, 53, 2003 CrossRef CAS PubMed; (m) H. Pellissier, Adv. Synth. Catal., 2020, 362, 2289 CrossRef CAS; (n) H. Pellissier, Adv. Synth. Catal., 2019, 361, 173 CrossRef; (o) H. A. Döndas, M. de Retamosa and J. M. Sansano, Organometallics, 2019, 38, 1828 CrossRef; (p) C. S. Evans and L. O. Davis, Molecules, 2018, 23, 33 CrossRef PubMed; (q) P. Chauan, S. Mahajan and D. Enders, Acc. Chem. Res., 2017, 50, 2809 CrossRef PubMed.
- For reviews on the chemistry of dicyanoalkenes, see: (a) B. Farajpour and A. Alizadeh, Org. Biomol. Chem., 2022, 20, 8366 RSC; (b) C. Curti, A. Sartori, L. Battistini and F. Zanardi, Synlett, 2018, 266 CAS; (c) L. Battistini, C. Curti, G. Rassu, A. Sartori and F. Zanardi, Synthesis, 2017, 2297 CAS; (d) J. Kaur, P. Chauhan and S. S. Chimni, Org. Biomol. Chem., 2016, 14, 7832 RSC; (e) M. Sánchez-Roselló, C. del Pozo and S. Fustero, Synthesis, 2016, 2553 CrossRef; (f) H.-L. Cui and Y.-C. Chen, Chem. Commun., 2009, 4479 RSC.
- H.-C. Wu, C. Wang, Y.-H. Chen and Y.-K. Liu, Chem. Commun., 2021, 57, 1762 RSC.
- Y. Hayashi, X. Han and N. Mori, Chem. – Eur. J., 2023, 29, e202301093 CrossRef CAS PubMed.
- K. Choudhary, H. Joshi, S. Rohilla and V. K. Singh, Chem. – Eur. J., 2024, 30, e202304078 CrossRef CAS PubMed.
- K. Li, S. Zhen, W. Wang, J. Du, S. Yu, Y. Wu and H. Guo, Chem. Sci., 2023, 14, 3024 RSC.
- L.-F. Cao, Y. Yin, Y.-L. Xie and J.-W. Xie, ChemistrySelect, 2024, 9, e202401711 CrossRef CAS.
- Z. Gao, X. Zhou, L. Xie, X. Wang, S. Wang, H. Liu and H. Guo, J. Org. Chem., 2024, 89, 7169 CrossRef CAS PubMed.
- S. Lu, Y. Zhao, S. Xie, W. Li and S.-W. Li, New J. Chem., 2023, 47, 21155 RSC.
- Y. Wang, C. Niu, D.-H. Xie and D.-M. Du, Org. Biomol. Chem., 2021, 19, 8572 RSC.
- Y. Gao, X. Song, R.-J. Yan, W. Du and Y.-C. Chen, Org. Biomol. Chem., 2021, 19, 151 RSC.
- W.-B. Yao, X.-S. Xie, J.-N. Liu and J.-W. Xie, Org. Biomol. Chem., 2022, 20, 1982 RSC.
- H. Mei, L. Lin, B. Shen, X. Liu and X. Feng, Org. Chem. Front., 2018, 5, 2505 RSC.
- M. Guo, F. Dong, X. Yin, L. Xu, L. Wang and S.-S. Li, Org. Chem. Front., 2021, 8, 2224 RSC.
- S. Lu, Z. Zhao, Y. Ren, G. Du, J. Zhao and S.-W. Li, Org. Chem. Front., 2022, 9, 3446 RSC.
- I. Suzuki, S. Tsunoi and Y. Shibata, Org. Lett., 2017, 19, 2690 CrossRef CAS PubMed.
- M. Liu, L. Zhou, W. Shi, Y. Hu, J. Liao, Z. Duan, W. Wang, Y. Wu, B. Zheng and H. Guo, Org. Lett., 2021, 23, 7703 CrossRef CAS PubMed.
- Z. Duan, M. Liu, B. Zheng, Y. Tang, J. Du, L. Wang, S. Yu, Y. Wu and H. Guo, Org. Lett., 2023, 25, 3298 CrossRef CAS PubMed.
- J.-R. Zhuo, B.-X. Quan, J.-Q. Zhao, M.-L. Zhang, Y.-Z. Chen, X.-M. Zhang and W.-C. Yuan, Tetrahedron, 2020, 76, 131115 CrossRef CAS.
- J. Wang, C.-F. Gao, H. Zhou, F. Li, J. Nie, J.-A. Ma and F. G. Zhang, Adv. Synth. Catal., 2025, 367, e70090 CrossRef CAS.
- A. Sanz-Vidal, D. Gaviña, L. Sotorríos, E. Gómez-Bengoa, F. López-Ortiz, M. Sánchez-Roselló and C. del Pozo, Chem. Commun., 2021, 57, 8023 RSC.
- D. Gaviña, M. Escolano, L. Sotorríos, E. Gómez-Bengoa, F. López-Ortiz, M. Sánchez-Roselló and C. del Pozo, Adv. Synth. Catal., 2024, 366, 970 CrossRef.
- Compounds 3 were obtained as inseparable mixtures of diastereoisomers.
- CCDC 2513043: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qc0x1.
| This journal is © The Royal Society of Chemistry 2026 |