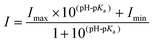Open Access Article
Open Access ArticleUltrafast fluorescent detection of mercury(II) ions in aqueous and biological systems using a boronic acid-based reaction probe
Kishor
Khadka
a,
Sumita
Subedi
a,
Eun-Taex
Oh
 b and
Keun-Hyeung
Lee
b and
Keun-Hyeung
Lee
 *a
*a
aEducation and Research Center for Smart Energy Materials and Process, Department of Chemistry and Chemical Engineering, Inha University, Incheon 402-751, South Korea. E-mail: leekh@inha.ac.kr
bDepartment in Biomedical Sciences, College of Medicine, Inha University, Incheon 22212, South Korea
First published on 26th March 2026
Abstract
A new reaction-based fluorescent chemodosimeter for Hg2+ ions was developed based on an irreversible substitution reaction between Hg2+ and a phenylboronic acid moiety. The probe exhibits a rapid and highly selective fluorescence turn-on response toward Hg2+ with dual green and red emissions under visible-light excitation. Notably, the sensing reaction is completed within 2–3 min in aqueous solution at pH 7.4 containing only 1% organic solvent, demonstrating excellent reactivity under near-physiological conditions. The probe (1) forms a well-defined 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 covalent adduct with Hg2+, avoiding complicated multi-adduct formation and enabling reliable quantitative detection. In addition, the boronic acid-based design effectively suppresses interference from competing metal ions. The probe shows efficient cellular uptake and allows real-time imaging of intracellular Hg2+ through simultaneous enhancement of green and red fluorescence signals. These results demonstrate that the present probe provides a robust sulfur-free platform for rapid, selective, and sensitive detection of mercury ions in aqueous environments and living cells, offering significant potential for environmental monitoring and biological mercury imaging.
1 covalent adduct with Hg2+, avoiding complicated multi-adduct formation and enabling reliable quantitative detection. In addition, the boronic acid-based design effectively suppresses interference from competing metal ions. The probe shows efficient cellular uptake and allows real-time imaging of intracellular Hg2+ through simultaneous enhancement of green and red fluorescence signals. These results demonstrate that the present probe provides a robust sulfur-free platform for rapid, selective, and sensitive detection of mercury ions in aqueous environments and living cells, offering significant potential for environmental monitoring and biological mercury imaging.
1. Introduction
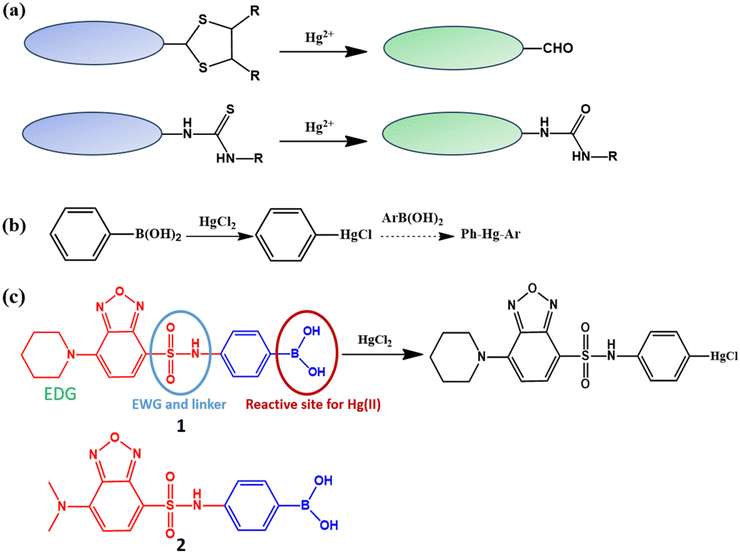
Among mercury species, mercury ions (Hg2+) pose a significant public health risk due to their persistence and high toxicity, because Hg2+ ions readily accumulate in the human body and can cause severe diseases.1–3 Hg2+ ions are generated both naturally and as a result of increasing human activity,1–3 leading to the contamination of aquatic environments and raising serious concerns about environmental and biological hazards. Because Hg2+ is bioavailable and can easily enter organisms via water, there is a critical need for methods capable of detecting Hg2+ sensitively and selectively in aqueous environments and live cells.Among various analytical techniques, fluorescence-based sensors have emerged as powerful tools for mercury detection owing to their high sensitivity, operational simplicity, real-time response, and potential applicability to living systems.4–14 In particular, reaction-based fluorescent probes (chemodosimeters) have been intensively investigated for the detection of Hg2+ ions in environmental and bio-samples because they offer high selectivity through irreversible chemical transformations induced by Hg2+.13–24 Unlike reversible coordination-based probes, chemodosimeters rely on irreversible chemical transformations, which provide significant fluorescence signal changes, generate accumulated fluorescence signals, and enable more reliable and unambiguous signal transduction.5–8
Among them, most Hg2+ chemodosimeters reported to date rely on selective interactions between Hg2+ ions and sulfur-containing functional groups (Scheme 1a).13–31 However, sulfur-based chemodosimeters often suffer from limited specificity and practical applicability due to cross-reactivity with other soft heavy metal ions, slow reactions in the presence of soft metal ions, and complicated reaction pathways. In addition, their sensing performance is frequently affected by oxidative conditions, which can suppress or distort the mercury-triggered desulfurization process, particularly in complex biological environments.
To overcome these intrinsic limitations, alternative sulfur-free reaction-based strategies have been actively explored.5–8 Among them, boronic acid-based chemodosimeters have emerged as a promising platform for mercury sensing, as they rely on the irreversible substitution reaction of arylboronic acids with Hg2+ (Scheme 1b).31–36 This reaction proceeds with high selectivity toward Hg2+ and is largely unaffected by competing soft metal ions, thereby offering improved specificity, reproducibility, and reliability in aqueous and biological environments.
In our earlier studies, we introduced a fluorescent chemodosimeter based on the substitution reaction between Hg2+ ions and phenylboronic acid moieties. Although this approach successfully demonstrated the feasibility of sulfur-free mercury sensing, several intrinsic limitations remained. In aqueous media, the boronic acid–mercury reaction proceeded relatively slowly (>5 min) or, in some cases, resulted in the formation of a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 covalent adduct, which complicated signal interpretation and quantitative analysis.31–36 In addition, boronic acid groups are known to strongly interact with diol-containing saccharides on cell surfaces, significantly impeding cellular uptake and thereby limiting the applicability of these probes for intracellular mercury detection.33,34
2 covalent adduct, which complicated signal interpretation and quantitative analysis.31–36 In addition, boronic acid groups are known to strongly interact with diol-containing saccharides on cell surfaces, significantly impeding cellular uptake and thereby limiting the applicability of these probes for intracellular mercury detection.33,34
In the present work, we report a new boronic acid-based fluorescent chemodosimeter (1) that overcomes these limitations (Scheme 1c). The probe is excitable with visible light and exhibits a pronounced turn-on fluorescence response, generating dual red and green emissions upon exposure to Hg2+ ions. Notably, the highly reactive probe displays an instantaneous fluorescence turn-on response (<2 min) in aqueous media containing only 1% organic solvent and forms a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 covalent adduct with Hg2+. Importantly, the probe readily penetrates living cells and enables intracellular Hg2+ detection, as evidenced by a significant enhancement of both red and green fluorescence signals. Collectively, these results represent a substantial advance in boronic acid-based mercury chemodosimeters and highlight their strong potential for real-time mercury sensing in complex biological and aqueous environments.
1 covalent adduct with Hg2+. Importantly, the probe readily penetrates living cells and enables intracellular Hg2+ detection, as evidenced by a significant enhancement of both red and green fluorescence signals. Collectively, these results represent a substantial advance in boronic acid-based mercury chemodosimeters and highlight their strong potential for real-time mercury sensing in complex biological and aqueous environments.
2. Experiments
2.1. Materials
4-Chloro-7-chlorosulfonyl-2,1,3-benzoxadiazole and 4-dimethylaminopyridine (DMAP) were purchased from TCI. 4-Aminophenylboronic acid pinacol ester, piperidine, and dimethylamine (in THF) were obtained from Sigma-Aldrich. Other reagents used for synthesis, including dichloromethane (DCM) and trifluoroacetic acid (TFA), were purchased from Acros Organics.2.2. Synthesis of fluorescent probes
4-Chloro-7-chlorosulfonyl-2,1,3-benzoxadiazole (253 mg, 1.0 mmol) and 4-aminophenylboronic acid pinacol ester (219 mg, 1.0 mmol) were dissolved in dry dichloromethane (20 mL). Triethylamine (279 µL, 2.0 mmol) was added dropwise, and the reaction mixture was stirred at 25 °C for 4 h. After removal of the solvent under reduced pressure, the crude product was purified by silica gel column chromatography (particle size 0.063–0.200 mm) to afford the sulfonamide intermediate 3.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20, v/v, 20 mL) at room temperature for approximately 6 h to deprotect the pinacol boronate ester. The solvent was removed under reduced pressure, and the crude product was purified by silica gel column chromatography. The final product (probe 1) was further purified by preparative HPLC using a C18 column, and its purity was confirmed by analytical HPLC (Fig. S1).
20, v/v, 20 mL) at room temperature for approximately 6 h to deprotect the pinacol boronate ester. The solvent was removed under reduced pressure, and the crude product was purified by silica gel column chromatography. The final product (probe 1) was further purified by preparative HPLC using a C18 column, and its purity was confirmed by analytical HPLC (Fig. S1).
The structures of probes 1 and 2 and intermediate 3 were characterized by high-resolution mass spectrometry (HRMS) and 1H and 13C NMR spectroscopy (Fig. S3–S11).
Probe 1: orange solid; mp 118–120 °C; overall yield 54.6%.
1H NMR (400 MHz, DMSO-d6) δ 10.57 (s, 1H), 7.90 (d, J = 8.0 Hz, 1H), 7.52 (d, J = 8.4 Hz, 2H), 6.99 (d, J = 8.0 Hz, 2H), 6.46 (d, J = 8.4 Hz, 1H), 3.82 (s, 4H), 1.61 (s, 6H).
13C NMR (101 MHz, DMSO-d6) δ 146.4, 145.2, 142.9, 139.9, 139.8, 135.5, 117.7, 109.6, 104.0, 50.3, 25.7, 24.1.
HRMS (m/z): [M + Na+]+ calcd for C17H19BN4O5SNa, 425.10669; found 425.10645.
Probe 2: orange solid; mp 117 °C; overall yield 52.5%.
1H NMR (400 MHz, DMSO-d6) δ 10.51 (s, 1H), 7.90 (d, J = 8.8 Hz, 1H), 7.51 (d, J = 8.8 Hz, 2H), 6.97 (d, J = 8.8 Hz, 2H), 6.16 (d, J = 8.4 Hz, 1H), 3.40 (s, 6H).
13C NMR (101 MHz, DMSO-d6) δ 146.3, 145.2, 143.8, 140.0, 139.9, 135.5, 117.6, 107.8, 101.8, 42.9.
HRMS (m/z): [M + Na+]+ calcd for C14H18BN4O5SNa, 385.07539; found 385.07526.
Intermediate 3: yellow solid; mp 152–153 °C; yield 76.2%.
1H NMR (400 MHz, DMSO-d6) δ 11.07 (s, 1H), 8.10 (d, J = 8.8 Hz, 1H), 7.84 (d, J = 8.8 Hz, 1H), 7.59 (d, J = 8.8 Hz, 2H), 7.03 (d, J = 8.4 Hz, 2H).
13C NMR (101 MHz, DMSO-d6) δ 149.3, 145.3, 140.1, 136.6, 136.5, 136.1, 131.2, 126.9, 126.8, 118.9, 114.7, 84.1, 83.4, 25.2, 25.1.
HRMS (m/z): [M]+ calcd for C18H19BClN3O5S, 435.08270; found 435.0830.
2.3. UV-visible and fluorescence measurements
A stock solution of the probe (0.5 mM) was prepared in a mixed solvent of dimethylformamide (DMF) and distilled water (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9, v/v). The stock solution of the probe was stable at 4 °C for at least four weeks, as verified by UV–visible absorption and fluorescence spectra. This stock solution was used for subsequent dilutions. A stock solution of HgCl2 (0.5 mM) was prepared in distilled water. Fluorescence emission spectra were recorded in aqueous buffered solution (10 mM HEPES, pH 7.4) containing 1% DMF using a fluorescence spectrometer (Jasco FP-8500). The emission spectra were obtained with an excitation wavelength of 440 nm. UV–visible absorption spectra were recorded using a Jasco UV–visible spectrophotometer (V-750).
9, v/v). The stock solution of the probe was stable at 4 °C for at least four weeks, as verified by UV–visible absorption and fluorescence spectra. This stock solution was used for subsequent dilutions. A stock solution of HgCl2 (0.5 mM) was prepared in distilled water. Fluorescence emission spectra were recorded in aqueous buffered solution (10 mM HEPES, pH 7.4) containing 1% DMF using a fluorescence spectrometer (Jasco FP-8500). The emission spectra were obtained with an excitation wavelength of 440 nm. UV–visible absorption spectra were recorded using a Jasco UV–visible spectrophotometer (V-750).
2.4. Determination of the limit of detection (LOD), quantum yield, and pKa
The limit of detection (LOD) was determined by fluorescence titration experiments according to a reported method.37 To evaluate the signal-to-noise (S/N) ratio, the fluorescence emission intensity of the blank sample was measured six times, and the standard deviation (σ) was calculated. The average fluorescence intensity was plotted as a function of analyte concentration to obtain the slope of the calibration curve (k). The LOD value was calculated using the equation LOD = 3σ/k.The fluorescence quantum yields were determined using fluorescein as a reference standard (Φ = 0.79).
The pKa value was measured based on the fluorescence intensity of probe 1 at different pH levels. The pKa values were obtained by nonlinear least-squares fitting of the data using the Henderson–Hasselbalch equation:
2.5. Fluorescent detection of Hg2+ in live cells by confocal microscopy
Cell viability assays and fluorescence imaging experiments were performed according to previously reported methods.38 The fluorescence imaging procedure is briefly described as follows. A549 cells plated on chamber slides were incubated with probe 1 in DMEM medium containing 1% DMF at 37 °C for 8 h. After incubation, the probe-loaded cells were washed four times with phosphate-buffered saline (PBS) to remove excess probe. Subsequently, Hg2+ ions were added to the preloaded A549 cells and incubated for 30 min. Fluorescence images were then acquired using an inverted confocal laser scanning microscope (TE2000E, Nikon, Tokyo, Japan).2.6. Quantification of Hg2+ ions in real-world samples
To evaluate the applicability of the probe for detecting Hg2+ ions in real water samples, tap water and groundwater samples were analyzed. The sample solutions were prepared by adding 10% (v/v) tap water or groundwater to an aqueous buffered solution (10 mM HEPES, pH 7.4) containing 1% DMF. The fluorescence intensity at 585 nm was recorded in the presence of different concentrations of Hg2+ ions using standard solutions ranging from 0 to 1000 nM. The fluorescence intensities obtained from the sample solutions were then converted to Hg2+ concentrations using a calibration curve.3. Results and discussion
3.1. Design of fluorescent probe 1 based on phenylboronic acid as a reactive site for Hg2+
Most fluorescent probes for Hg2+ exhibit a turn-off response due to the strong fluorescence-quenching ability of Hg2+ ions.5–8 However, turn-off-type detection suffers from critical limitations in practical applications, as fluorescence decreases can also arise from probe precipitation, nonspecific environmental quenching, or absorption by unknown impurities, leading to ambiguous signal interpretation. To overcome these drawbacks, we designed turn-on fluorescent probes in which an intramolecular charge transfer (ICT) fluorophore, a benzofurazan moiety, is conjugated to a phenylboronic acid moiety through a thioamide linker (Scheme 1c). In this design, the phenylboronic acid functions as the reactive site for Hg2+, while the benzofurazan moiety is used as a fluorophore.39 The thioamide group serves as a linker between the fluorophore and the reactive site and an electron-withdrawing group. Piperidine was introduced as the electron-donating group in the ICT fluorophore part of probe 1, whereas dimethylamine was conjugated as the electron-donating group in the ICT fluorophore part of probe 2 because piperidine provides improved quantum yield and photostability compared to commonly used dimethylamine donors.40 Both fluorescent probes were designed to be excitable with visible light and to exhibit a broad, long-wavelength emission profile, including green and red emissions. These optical features effectively minimize background interference from complex biological environments and are advantageous for biological and environmental mercury sensing.3.2. Synthesis and photophysical properties of fluorescent probes 1 and 2
Probes 1 and 2 were synthesized through a two-step sequence via the sulfonamide intermediate 3 (Scheme S1). The sulfonyl chloride group of 4-chloro-7-chlorosulfonyl-2,1,3-benzoxadiazole was first coupled with 4-aminophenylboronic acid pinacol ester in the presence of triethylamine to afford intermediate 3. Subsequent nucleophilic substitution of the chloro group with piperidine or dimethylamine provided the corresponding intermediates, which were finally converted to probe 1 and probe 2, respectively, through deprotection of the pinacol boronate ester using TFA/DCM. The successful synthesis and high purity of the probes were confirmed by 1H NMR, 13C NMR, mass spectrometry, and HPLC analyses.According to our design concept, both fluorescent probes are excited with visible light and show green and red emissions ranging from 550 nm to 650 nm in purely aqueous solutions. As the poor water solubility of many fluorescent probes limits their practical applications in biological and aqueous environments, we evaluated the solubility of probes 1 and 2 in aqueous solution (pH 7.4) containing 1% DMF. Both probes exhibited good solubility under these conditions, as evidenced by a linear correlation between absorbance and concentration over the range of 0–25 μM (Fig. 1a–c). Upon excitation at 440 nm, probes 1 and 2 displayed broad emission bands spanning 530–730 nm, and the fluorescence intensity increased linearly with concentration over the same range (0–25 μM). In addition, the relative fluorescence intensities of both probes were monitored for 180 min under continuous visible-light irradiation, demonstrating sufficient photostability in aqueous solution containing 1% DMF.
3.3. Fluorescent responses of chemodosimeters to Hg2+ and characterization of the reaction product
Fluorescence titration experiments of the fluorescent chemodosimeters with Hg2+ were conducted in aqueous solution (10 mM HEPES, pH 7.4) containing 1% DMF (Fig. 2a and e). Upon gradual addition of Hg2+, pronounced fluorescence enhancement was observed for both probes over the emission range of 450–650 nm. Specifically, the emission intensity at 585 nm increased by approximately fourfold for probe 1, whereas a twofold increase was observed for probe 2.Notably, probe 1 exhibited mainly fluorescence intensity enhancement with little change in the emission wavelength, whereas probe 2 showed a slight red shift in the emission spectrum upon reaction with Hg2+. This difference can be attributed to the stronger electron-donating ability of the piperidine group in probe 1 compared with the dimethylamine group in probe 2, which results in a stronger ICT character and thus minimizes spectral shifts while enhancing fluorescence intensity. In contrast, the weaker donor in probe 2 leads to a slightly greater redistribution of the ICT state upon formation of the arylmercury(II) chloride species, giving rise to the observed bathochromic shift.
Interestingly, both probes exhibited exceptionally rapid responses toward Hg2+ (Fig. 2b and f). The fluorescence enhancement reached completion within 2–3 min after Hg2+ addition, and even at a low Hg2+ concentration (1 μM), the fluorescence response was completed within 120 s. These results demonstrate the high reactivity and rapid sensing capability of the probes toward Hg2+ in aqueous media.
To evaluate the influence of counteranions, the fluorescence responses of the probes to different mercury salts, including HgCl2, Hg(ClO4)2, Hg(NO3)2, and Hg(OAc)2, were examined (Fig. 2c, d and Fig. S12). All mercury salts produced nearly identical final fluorescence intensities within 3 min, regardless of the anionic species. This observation confirms that the reaction-based sensing process of the probes toward Hg2+ proceeds efficiently and is essentially independent of the counteranion, indicating negligible anion effects on both detection sensitivity and response kinetics.
As probe 1 exhibited a more pronounced fluorescence enhancement in response to Hg2+ than probe 2, we further investigated the effect of pH on the Hg2+ sensing performance of probe 1 (Fig. 2g). As the pH increased above 8.0, the fluorescence enhancement induced by Hg2+ gradually decreased, which can be attributed to the formation of insoluble mercury species such as Hg(OH)2, leading to reduced availability of free Hg2+ ions. When the pH decreased from 8.0 to 5.0, the fluorescence enhancement remained significant. However, a pronounced decrease in emission intensity was observed at pH 4.5, which is attributed to the protonation of the cyclohexylamine group in the fluorophore moiety of the probe (pKa ≈ 4.5),12,41 resulting in disruption of the intramolecular charge transfer process. These results indicate that the probe exhibits optimal sensing performance toward Hg2+ over a wide pH range (5–9), including physiological pH, demonstrating its suitability for both biological and environmental applications.
To verify the irreversible nature of the reaction between 1 and Hg2+, EDTA, a strong reversible chelator for Hg2+, was employed (Fig. 3a). After completion of the fluorescence enhancement induced by Hg2+, the subsequent addition of EDTA caused no noticeable change in the fluorescence spectrum. This result indicates that Hg2+ was irreversibly consumed during the sensing reaction.
In addition, EDTA was added simultaneously with Hg2+ to examine whether EDTA interferes with the sensing process of 1. Under these conditions, the fluorescence intensity change of the reaction-based probe was almost identical to that observed in the absence of EDTA, demonstrating that EDTA does not inhibit the Hg2+-triggered irreversible reaction. Overall, the results confirm that probe 1 detects Hg2+ through an irreversible reaction mechanism and that this reaction is not affected by the presence of EDTA.
The reaction product of 1 with Hg2+ was further analyzed by C18-TLC, HPLC, and mass spectrometry (Fig. 3b, c, and Fig. S13). Probe 1 was incubated with Hg2+ in distilled water containing 20% CH3CN for 5 min, and the resulting major product was analyzed by HPLC–ESI mass spectrometry. A new spot/peak with lower polarity (higher hydrophobicity) than that of probe 1 was observed in both the TLC and HPLC chromatograms. ESI mass spectrometric analysis revealed a molecular ion peak at m/z 559.3, corresponding to an arylmercury species (Scheme 1), supporting the replacement reaction of the phenylboronic acid moiety with Hg2+. Further analysis of the product by FAB mass spectrometry confirmed this transformation, resulting in the formation of an arylmercury(II) chloride (ArHgCl) species (Fig. S14).
3.4. Selectivity study of the fluorescent chemodosimeter (1)
The selectivity of probe 1 toward Hg2+ was evaluated by measuring its fluorescence response to various metal ions, including Na+, Mg2+, Ca2+, Al3+, Cr3+, Co2+, Ni2+, Mn2+, Cu2+, Zn2+, Cd2+, Ag+, Pb2+, K+, and Hg2+, in aqueous buffered solution (10 mM HEPES, pH 7.4) containing 1% DMF (Fig. 4a). Among all tested metal ions, only Hg2+ induced a significant fluorescence enhancement, while the other metal ions produced negligible changes. This result confirms that the phenylboronic acid-based displacement reaction is highly selective for Hg2+ in aqueous solutions.Encouraged by these results, we next investigated the fluorescence response of probe 1 to Hg2+ in the presence of competing metal ions (Fig. S15). Even in the presence of Group I and Group II metal ions (1 mM) and other heavy metal ions (10 μM), the fluorescence enhancement induced by Hg2+ was not significantly affected. However, a noticeable decrease in the Hg2+-induced fluorescence enhancement was observed in the presence of coexisting Cu2+ ions. The suppressed fluorescence response toward Hg2+ in the presence of Cu2+ is not attributed to a direct fluorescence quenching effect, because the addition of Cu2+ after completion of the Hg2+-triggered reaction did not affect the emission intensity. Instead, Cu2+ is likely to interfere with the Hg2+-induced reaction by forming a coordination complex with the boronic acid moiety, thereby reducing the reactivity of the probe toward Hg2+. Thus, the inhibitory effect of Cu2+ on the Hg2+-induced fluorescence response was completely removed by EDTA. In the presence of EDTA, the probe exhibited the same fluorescence enhancement upon the simultaneous addition of Hg2+ and Cu2+ as that observed with Hg2+ alone (Fig. 4b), confirming that Cu2+ interferes with the sensing process through reversible coordination rather than fluorescence quenching. These results demonstrate that EDTA effectively masks Cu2+, thereby preventing its inhibitory interaction with the boronic acid moiety and restoring the intrinsic Hg2+-triggered fluorescence response.
This observation indicates that probe 1 can selectively detect Hg2+ without significant interference from coexisting metal ions. The selectivity was further visualized by naked-eye observation under UV illumination (Fig. 4c). Only the Hg2+-treated solution exhibited a distinct red emission, whereas no noticeable fluorescence color change was observed in the presence of other metal ions. These results clearly demonstrate the excellent visual selectivity of probe 1 for Hg2+.
The detection limit of probe 1 for Hg2+ was determined based on the fluorescence intensity at 585 nm (Fig. 4d). The limit of detection was calculated to be 7.56 nM (R2 = 0.99) in aqueous buffered solution (10 mM HEPES, pH 7.4). This detection limit is comparable to the maximum allowable concentration of mercury ions in drinking water (10 nM, 2 ppb) established by the U.S. Environmental Protection Agency (EPA).
The low limit of detection (LOD) and the negligible interference from other metal ions demonstrate the practical applicability of probe 1 for environmental mercury monitoring. To further evaluate its practical utility, probe 1 was applied to quantify Hg2+ in real water samples, including groundwater (GW) and tap water (TW). To validate the analytical performance, known amounts of Hg2+ were spiked into GW and TW samples, and the concentrations were determined using the fluorescence emission intensity of probe 1 (Table S1). The recovery values for Hg2+ ranged from 92.9% to 99.9% in GW and from 94.3% to 101.1% in TW. These satisfactory recovery results indicate that probe 1 can be reliably applied for the quantitative determination of Hg2+ in real aqueous samples.
The fluorescent probe based on the reaction of phenylboronic acid with Hg2+ exhibited higher selectivity for Hg2+ than most previously reported fluorescent chemodosimeters that rely on sulfur-based reactions with Hg2+. Considering the detection characteristics of reported fluorescent chemodosimeters (Table S2), probe 1 displayed several notable sensing properties, including visible-light excitation, enhancement of red fluorescence upon exposure to Hg2+, operation in a purely aqueous solution containing only 1% organic solvent, and a fast response to low concentrations of Hg2+.
3.5. Fluorescence imaging of Hg2+ in live cells using chemodosimeter 1
Since probe 1 exhibited a sensitive turn-on fluorescence response to Hg2+ in aqueous buffered solution at pH 7.4, we further investigated its applicability for Hg2+ detection in live human cells.The cytotoxicity of 1 toward A549 cells was evaluated using an ATP-Glo assay. As shown in Fig. S16, incubation with 1 at concentrations ranging from 0 to 30 μM for 24 h did not cause any significant reduction in cell viability, indicating good biocompatibility of the probe within this concentration range.
For fluorescence imaging experiments, A549 cells were incubated with probe 1 (10 μM) at 37 °C for 8 h, followed by treatment with various concentrations of Hg2+. As shown in Fig. 5a, cells treated with 1 alone exhibited only weak background fluorescence in both the green and red channels. In contrast, upon the addition of Hg2+, a gradual and concentration-dependent enhancement of both green and red fluorescence emissions was clearly observed. Consequently, the merged fluorescence images displayed an increasing yellow signal with increasing Hg2+ concentration. These results demonstrate that probe 1 efficiently penetrates cell membranes and sensitively detects intracellular Hg2+ through a fluorescence turn-on response.
To further examine the irreversibility of the fluorescence response, cell-permeable EDTA was added to the cells containing probe 1 and Hg2+, which exhibited strong red and green emissions. As shown in Fig. 5b, the addition of EDTA did not induce a significant decrease in either green or red fluorescence intensities. This observation confirms that the fluorescence enhancement does not arise from reversible Hg2+ coordination but rather from an irreversible chemical reaction between chemodosimeter 1 and Hg2+. These results clearly demonstrate that chemodosimeter 1 enables highly sensitive turn-on fluorescence detection of Hg2+ in living cells. More importantly, the irreversible reaction between 1 and Hg2+ proceeds efficiently even within the highly complex intracellular environment.
3.6. Binding mode and self-activation mechanism of chemodosimeter 1 toward Hg2+
UV–vis absorption titration experiments were performed to investigate the binding mode between probe 1 and Hg2+. Upon gradual addition of Hg2+, the absorption band of 1 centered at 460 nm exhibited a distinct red shift (Fig. 6a). This spectral change can be attributed to the formation of a covalent aryl–Hg+ adduct (Fig. 3c), in which the Hg+ moiety acts as a stronger electron-withdrawing group than the original boronic acid functionality. This substitution enhances the intramolecular charge transfer character of the fluorophore part, leading to a bathochromic shift and increased fluorescence emission. Consistently, the fluorescence quantum yield (ϕ = 0.022) of the reaction product measured in aqueous solution was significantly higher than that of the parent probe (ϕ = 0.0083), confirming that replacement of the boronic acid group by Hg2+ substantially improves the fluorescence efficiency.To understand the origin of the ultrafast response of 1 toward Hg2+, we considered the reaction mechanism between phenylboronic acid and Hg2+.42,43 This transformation is generally described as an electrophilic substitution-assisted transmetalation process (Fig. 6b), in which the boronate form is more reactive than the neutral boronic acid species. Accordingly, the pKa value of the phenylboronic acid moiety in probe 1 was determined (Fig. S17). The obtained pKa value of 8.3 indicates that the neutral boronic acid form predominates at physiological pH (7.4), which alone cannot explain the exceptionally fast reaction rate.
We therefore propose that the ultrafast response arises from an intermolecular self-activation mechanism mediated by supramolecular dimer formation (Fig. 6b). In aqueous media containing a low fraction of organic solvent, the amphiphilic nature of probe 1 promotes the formation of supramolecular dimeric assemblies. In these assemblies, the aromatic moieties stack through hydrophobic interactions, while the thioamide groups further stabilize the structure via intermolecular hydrogen bonding. Within this dimeric arrangement, the piperidine unit of one molecule is positioned in close proximity to the boronic acid group of the neighboring molecule. This spatial organization enables an intermolecular acid–base interaction, in which the basic piperidine nitrogen partially deprotonates the boronic acid moiety (pKa ≈ 8.3), generating a boronate-like activated species even under physiological pH conditions. Consequently, the intermolecular acid–base interaction may enhance the aromatic substitution reaction with Hg2+, leading to a dramatically accelerated reaction rate.
Three experimental observations strongly support this self-activation model. First, dimeric species were directly observed in the ESI mass spectrum (Fig. 6c). Although the ionization process generally induces dissociation of noncovalent aggregates due to charge repulsion, a distinct peak at m/z 805.4 was detected, which corresponds to the protonated dimeric species (2 × 403.2 + H+). The persistence of this dimeric ion under high-energy ionization conditions strongly indicates that probe 1 has an intrinsic propensity to form dimeric assemblies in solution, thereby supporting the feasibility of dimer formation in aqueous environments.
Second, an organic solvent was introduced to disrupt the dimeric assembly of 1 and to examine its effect on the reaction kinetics with Hg2+ (Fig. 6d). As the DMF content increased from 1% to 20%, the fluorescence response became markedly slower. This pronounced retardation of the reaction clearly indicates that the dimeric assemblies in aqueous media play a critical role in facilitating the self-activation process toward Hg2+. Third, the reaction kinetics of the probe were investigated under different solvent conditions. In the presence of 10% and 20% DMF, the reaction rate between the probe and Hg2+ could be reliably monitored (Fig. S18). To determine the reaction order with respect to the probe, the concentration of Hg2+ was fixed at ten times that of the probe to maintain pseudo-first-order conditions. The reaction rates were measured at various probe concentrations, and a plot of ln(rate) versus ln[probe] was used to determine the reaction order (Fig. 6e and f).
Under 20% DMF conditions, the reaction order with respect to the probe concentration was determined to be 0.92, indicating an approximately first-order dependence. In contrast, under 10% DMF conditions, the reaction order increased to 1.76. This higher reaction order strongly suggests the involvement of probe dimerization and supports the proposed dimeric self-activation mechanism of the fluorescent probe. In 1% DMF, the reaction proceeded too rapidly to accurately determine the reaction order with respect to the probe concentration. Overall, this dimeric self-activation mechanism effectively converts the boronic acid moiety into a highly reactive boronate-like species without requiring alkaline conditions.
4. Conclusions
In summary, we developed a new boronic acid-based reaction-type fluorescent chemodosimeter for the selective and rapid detection of Hg2+ ions. The probe (1) operates through an irreversible substitution reaction between the phenylboronic acid moiety and Hg2+, producing a stable 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 covalent adduct accompanied by a pronounced dual-channel fluorescence turn-on response. The present probe exhibits excellent selectivity, minimal interference from competing metal ions, and a remarkably fast response time within 2–3 min in nearly pure aqueous solutions. Furthermore, the probe readily penetrates living cells and enables real-time visualization of intracellular Hg2+ through both green and red fluorescence channels.
1 covalent adduct accompanied by a pronounced dual-channel fluorescence turn-on response. The present probe exhibits excellent selectivity, minimal interference from competing metal ions, and a remarkably fast response time within 2–3 min in nearly pure aqueous solutions. Furthermore, the probe readily penetrates living cells and enables real-time visualization of intracellular Hg2+ through both green and red fluorescence channels.
These features collectively overcome key limitations of previous boronic acid-based mercury probes, including slow reaction kinetics, poor aqueous reactivity, and limited cellular uptake. Therefore, this work provides a significant advancement in sulfur-free mercury chemodosimeter design and offers a promising molecular platform for practical mercury detection in complex environmental and biological systems.
Conflicts of interest
There are no conflicts to declare.Data availability
All experimental and spectroscopic data supporting this study are available in the supplementary information (SI). Supplementary information: NMR spectra, HRMS data and fluorescence measurements. See DOI: https://doi.org/10.1039/d6ob00170j.Acknowledgements
This work was supported by a grant from Inha University.References
- U.S. Environmental Protection Agency, Mercury Update: Impact on Fish Advisories, EPA Fact Sheet EPA-823-F-01-011, Office of Water, Washington, DC, 2001.
- W. F. Fitzgerald, C. H. Lamborg and C. R. Hammerschmidt, Chem. Rev., 2007, 107, 641–662 CrossRef CAS PubMed.
- Y.-S. Hong, Y.-M. Kim and K.-E. Lee, J. Prev. Med. Public Health, 2012, 45, 353–363 CrossRef PubMed.
- B. Valeur and M. N. Berberan-Santos, Molecular Fluorescence: Principles and Applications, Wiley-VCH, Weinheim, 2012, 2nd edn, pp. 479–531 Search PubMed.
- H. Shuai, C. Xiang, L. Qian, F. Bin, L. Xiaohui, D. Jipeng, Z. Chang, L. Jiahui and Z. Wenbin, Dyes Pigm., 2021, 187, 109125 CrossRef CAS.
- M. Saleem, M. Rafiq and M. Hanif, J. Fluoresc., 2017, 27, 31–58 CrossRef CAS PubMed.
- S. H. Park, N. Kwon, J. H. Lee, J. Yoon and I. Shin, Chem. Soc. Rev., 2020, 49, 143–179 RSC.
- Y. Wang, L. Zhang, X. Han, L. Zhang, X. Wang and L. Chen, Chem. Eng. J., 2021, 406, 127166 CrossRef CAS.
- S. Oh, J. Jeon, J. Jeong, J. Park, E.-T. Oh, H. J. Park and K.-H. Lee, Anal. Chem., 2020, 92, 4917–4925 CrossRef CAS PubMed.
- H. Park, E.-T. Oh, J. Park, S. Subedi, H. J. Park and K.-H. Lee, Anal. Chem., 2025, 97, 5982–5991 CrossRef CAS PubMed.
- H. Park, S. Subedi, E.-T. Oh, H. J. Park and K.-H. Lee, Microchem. J., 2024, 200, 110375 CrossRef CAS.
- P. K. Mehta, H. Park, E.-T. Oh, H. J. Park and K.-H. Lee, Sens. Actuators, B, 2023, 385, 133670 CrossRef CAS.
- S. K. Ko, Y. K. Yang, J. Tae and I. Shin, J. Am. Chem. Soc., 2006, 128, 14150–14155 CrossRef CAS PubMed.
- J. Hu, Z. Hu, S. Liu, Q. Zhang, H. W. Gao and K. Uvdal, Sens. Actuators, B, 2016, 230, 639–644 CrossRef CAS.
- M. Deng, D. Gong, S.-C. Han, X. Zhu, A. Iqbal, W. Liu, W. Qin and H. Guo, Sens. Actuators, B, 2017, 243, 195–202 CrossRef CAS.
- Z. Zhang, Y. Li, H. Zuo, C. Wang and Y. Shen, J. Photochem. Photobiol., A, 2017, 332, 293–298 CrossRef.
- Q. Zou and H. Tian, Sens. Actuators, B, 2010, 149, 20–27 CrossRef CAS.
- L. Lan, Q. Niu and T. Li, Anal. Chim. Acta, 2018, 1023, 105–114 CrossRef CAS PubMed.
- S.-L. Pan, K. Li, L.-L. Li, M.-Y. Li, L. Shi, Y.-H. Liu and X.-Q. Yu, Chem. Commun., 2018, 54, 4955–4958 RSC.
- Q. Zhang, Y. Li, H. Zuo, C. Wang and Y. Shen, Analyst, 2015, 140, 2778–2784 RSC.
- C. Kan, X. Shao, F. Song, J. Xu, J. Zhu and L. Du, Microchem. J., 2019, 150, 104142 CrossRef CAS.
- M. Zhao, Y. S. Guo, G. D. Fu, Q. Wang, W. L. Sheng and D. S. Guo, Microchem. J., 2021, 171, 106855 CrossRef CAS.
- Y. K. Yang, S. K. Ko, I. Shin and J. Tae, Nat. Protoc., 2007, 2, 1740–1745 CrossRef CAS PubMed.
- M. Tian, C. Wang, Q. Ma, Y. Bai, J. Sun and C. Ding, ACS Omega, 2020, 5, 18176–18184 CrossRef CAS.
- M. Matsushita, M. M. Meijler, P. Wirsching, R. A. Lerner and K. D. Janda, Org. Lett., 2005, 7, 4943–4946 CrossRef CAS PubMed.
- L. Li, H. Ouyang, Z. Long, Q. Zhang, Y. Jiang, M. Cai, S. Xiong, S. Peng, G. Xu and Q. He, Org. Biomol. Chem., 2023, 21, 5560–5566 RSC.
- S. Fang, L. Zhang, Y. Zhao, X. Zhang, L. Zhang, L. Chen, J. Yoon and S. Liu, Sens. Actuators, B, 2024, 411, 135768 CrossRef CAS.
- D. B. C. Leslee, U. Venkatachalam, J. Gunasekaran, S. Karuppannan and S. B. Kuppannan, Org. Biomol. Chem., 2023, 21, 4130–4143 RSC.
- Z. Ruan, X. Dong, T. Long, S. Liu, Y. Chen and J. Lin, J. Mater. Chem. C, 2024, 12, 7572–7579 RSC.
- X. Zhang, Y. Zhang, J. Zhao, H. Li and L. Chen, J. Mater. Chem. C, 2018, 6, 773–780 RSC.
- Z. Ruan, C. Fan, X. Wang, D. Shao, X. Yang, W. He, T. Xu, J. Lin and Z. Tian, Dyes Pigm., 2024, 222, 111835 CrossRef CAS.
- S. W. Lee, S. Y. Lee and S. H. Lee, Tetrahedron Lett., 2019, 60, 151048 CrossRef CAS.
- S. Subedi, L. N. Neupane, H. Yu and K.-H. Lee, Sens. Actuators, B, 2021, 338, 129814 CrossRef CAS.
- S. Subedi, L. N. Neupane, P. K. Mehta and K.-H. Lee, Dyes Pigm., 2021, 191, 109374 CrossRef CAS.
- X. Guo, J. Huang, Y. Wei, Q. Zeng and L. Wang, J. Hazard. Mater., 2020, 381, 120969 CrossRef CAS PubMed.
- L. N. Neupane, J. Park, P. K. Mehta, E.-T. Oh, H. J. Park and K.-H. Lee, Chem. Commun., 2020, 56, 2941–2944 RSC.
- G. L. Long and J. D. Winefordner, Anal. Chem., 1983, 55, 712A–724A CrossRef CAS.
- H. Park, E.-T. Oh, J. Park, S. Subedi, H. J. Park and K.-H. Lee, Anal. Chem., 2025, 97, 5982–5990 CrossRef CAS PubMed.
- S. Uchiyama, T. Santa, T. Fukushima, H. Homma and K. Imai, J. Chem. Soc., Perkin Trans., 1998, 2, 2165–2174 RSC.
- P. K. Mehta, K. Ryu, C. K. Kim and K.-H. Lee, New J. Chem., 2022, 46, 7003–7013 RSC.
- W. Zhai, X. Sun, T. D. James and J. S. Fossey, Chem. – Asian J., 2015, 10, 1836–1848 CrossRef CAS PubMed.
- R. C. Larock, Organomercury Compounds in Organic Synthesis, Springer-Verlag, Berlin, 1985, pp. 15–16 Search PubMed.
- K. Torssell, Acta Chem. Scand., 1959, 13, 115–119 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |