Open Access Article
Open Access ArticleLate-stage chirality generation strategies for the total synthesis of macrocyclic natural products
Haruhiko
Fuwa
 * and
Mina
Tateya
* and
Mina
Tateya

Department of Applied Chemistry, Faculty of Science and Engineering, Chuo University, 1-13-27 Kasuga, Bunkyo-ku, Tokyo 112-8551, Japan. E-mail: hfuwa.50m@g.chuo-u.ac.jp
First published on 2nd April 2026
Abstract
Macrocyclic natural products, including macrolactones, macrolactams, macrocyclic (depsi)peptides, and macrocyclic cyclophanes, occupy a chemical space that does not overlap significantly with that of traditional low molecular weight and sp2-carbon rich pharmaceuticals. Traditionally, total synthesis toward macrocyclic natural products has been typically based on installation of backbone stereogenic centers at early- to mid-stage and closure of the macrocyclic backbone at late stage. However, these synthesis strategies suffer from multiple concession steps, making them less attractive in terms of step-economy. In this review, we provide an overview of late-stage chirality generation strategies in macrocyclic natural product synthesis, embodying stereoselective functional group and/or skeletal transformations that take advantage of macrocyclic conformational constraints. Expanding our repertoire of transformations amenable to late-stage chirality generation as well as advancing controllability over the conformational property of macrocycles will facilitate future developments in the total synthesis of macrocyclic natural products.
Introduction
Chemical space that has been less explored because of synthetic hurdles may represent a promising source of new chemotherapeutics for the treatment and cure of intractable human diseases.1 It has been argued that the chemical space occupied by macrocyclic natural products, such as macrolactones, macrolactams, macrocyclic (depsi)peptides, and macrocyclic cyclophanes, is considerably different from that occupied by traditional low-molecular-weight, sp2-carbon-rich pharmaceuticals.2,3 Typical macrocyclic natural products have molecular weights of greater than 500 Dalton, large molecular surface areas, multiple functional groups on the edge of their macrocyclic skeletons, and the unique ability to bind with the shallow or flat surface of proteins. Importantly, such protein binding sites are often involved in ‘undruggable’ protein–protein interactions that are difficult to be accessed by traditional small molecules.While macrocyclic (depsi)peptides are becoming accessible by in vitro ribosomal synthesis,4 total synthesis is a gold standard for the practical supply of macrocyclic natural products and their analogues for detailed biological testing and applications in medicinal chemistry and chemical biology. However, at the same time, it is still a challenging task for chemists to synthesize macrocyclic natural products, especially those having a stereochemically complex backbone structure. For example, the total synthesis of macrocycles often requires more than 20 steps from commercially available inexpensive materials, though a body of reliable methods are currently available for the stereocontrolled synthesis of substructures and efficient macrocyclization. Total synthesis of macrocycles has been typically achieved in the way in which backbone stereogenic centers are installed precisely at early- to mid-stage and macrocyclic skeleton is closed at late stage.5 It appears reasonable to circumvent late-stage chirality-generating transformations because of the ambiguity in anticipating the conformation and reactivity of macrocycles with varying degrees of conformational flexibility. At the same time, the traditional synthesis strategies toward macrocycles tend to require many concession steps such as protecting group manipulations and oxidation state adjustments due to the early to mid-stage installation of multiple functional groups. For example, in polyketide macrocycle synthesis, substructures are accessible in a solid manner through iterative asymmetric aldol or crotylation reactions.6 However, such iterative strategies require multiple transformations for oxidation/reduction and protection/deprotection, which make them less attractive in terms of step-economy (Scheme 1A). A more step-economical approach can be envisioned by considering an aldol coupling of aldehydes and metal enolates, although it is still not free from concession steps and its stereochemical outcome is substantially affected by substrate bias (Scheme 1B). In contrast, it may be even more step-economical if we could install the requisite stereogenic center(s) at the late stage by making use of prochiral functional groups, such as olefins and ketones, under macrocyclic conformational constraints (Scheme 1C).
Late-stage functionalization of structurally intricate natural products enables the generation of an array of structural analogues that are previously difficult to access by traditional target-oriented total synthesis, thereby facilitating natural product chemical biology and drug discovery.7 The past two decades have seen significant progress in late-stage functionalization with the advent of new synthetic methods such as C–H functionalization, photocatalysis, and biocatalysis. Nevertheless, it appears that the application of these new synthetic methods has been mostly limited to late-stage functionalization of natural products with rigid scaffolds so far.
Now that a body of efficient macrocyclization reactions are made available to the synthetic community, an emerging frontier in the total synthesis of macrocyclic natural products in the age of synthetic efficiency is late-stage chirality generation by taking advantage of macrocyclic conformational constraint. Stereoselective functional group transformations at a late stage should increase the flexibility of synthesis design and facilitate derivatization and even stereodivergent synthesis. Meanwhile, stereoselective skeletal transformations including macrocyclizations,8 transannular reactions,9 and skeletal reorganizations would likely help increase the molecular complexity with high step-economy10 but are largely underexplored in macrocycle synthesis. This review provides an overview of late-stage chirality generation strategies for the total synthesis of macrocyclic natural products by highlighting selected examples.
Early development
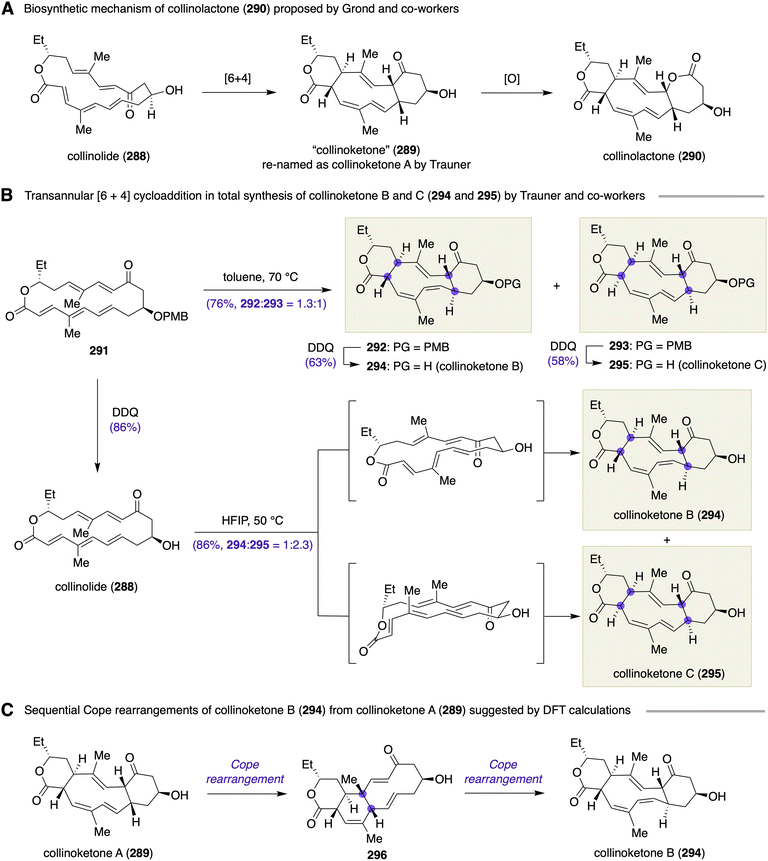
Alkylation of carbonyls, conjugate addition to α,β-unsaturated carbonyls, oxidation of olefins, and reduction of ketones and olefins embedded within macrocycles are known to proceed diastereoselectively to create a new stereogenic center(s) when the two faces of the double bond of these functional groups are differentiable by the virtue of the intrinsic conformational constraints of macrocycles, i.e., “macrocyclic stereocontrol”.11 Pioneering works on such chirality-generating functional group transformations on macrocycles were published during the late 1970s to the 1980s.Significant contributions were made by Still and co-workers to formulating the concept “macrocyclic stereocontrol” during the late 1970s to the early 1980s. The Still group investigated chirality-generating functionalization on macrocycles of various ring sizes.12 In 1979, Still described that chirality-generating oxidations were very effective for installing two epoxides onto the macrocyclic skeleton of (±)-periplanone B (1) with good to excellent diastereoselectivities at the late stage of the synthesis (Scheme 2A).13
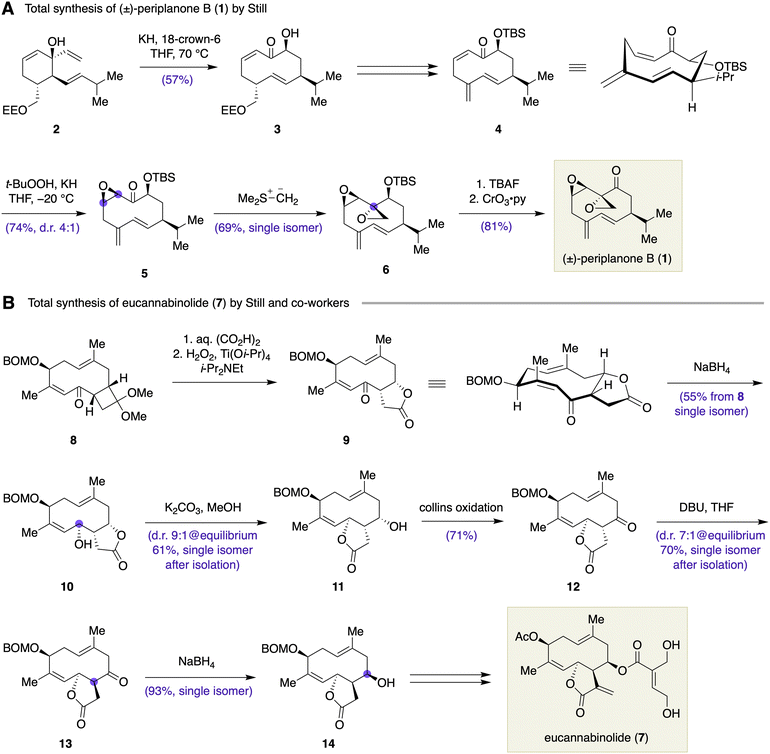 | ||
| Scheme 2 (A) Total synthesis of periplanone B (1) by Still. (B) Total synthesis of eucannabinolide (7) by Still and co-workers. | ||
Thus, macrocyclic α,β-unsaturated ketone 4, derived from a simple cyclohexanone derivative 2via an oxy–Cope rearrangement, was epoxidized chemo- and stereo-selectively with t-BuOOH/KH to give epoxy ketone 5 in 74% yield with 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 diastereoselectivity, which was then subjected to Corey–Chaykovsky epoxidation using dimethylsulfonium methylide to afford diepoxide 6 in 69% yield as a single isomer. The exomethylene group was installed to the macrocycle prior to the epoxidations for controlling the conformation. Desilylation and oxidation completed the synthesis of 1. Two additional diastereomers of periplanone B were also synthesized by modifying the peripheral substituents and reaction conditions to control the conformation of the macrocyclic backbone. In the total synthesis of eucannabinolide (7) by the Still group, a four-step sequence of functional group transformations, i.e., ketone reduction (9 → 10), acyl migration (10 → 11), epimerization (12 → 13), and ketone reduction (13 → 14), were performed on late-stage macrocyclic intermediates to establish the three contiguous stereogenic centers of the target natural product (Scheme 2B).14
1 diastereoselectivity, which was then subjected to Corey–Chaykovsky epoxidation using dimethylsulfonium methylide to afford diepoxide 6 in 69% yield as a single isomer. The exomethylene group was installed to the macrocycle prior to the epoxidations for controlling the conformation. Desilylation and oxidation completed the synthesis of 1. Two additional diastereomers of periplanone B were also synthesized by modifying the peripheral substituents and reaction conditions to control the conformation of the macrocyclic backbone. In the total synthesis of eucannabinolide (7) by the Still group, a four-step sequence of functional group transformations, i.e., ketone reduction (9 → 10), acyl migration (10 → 11), epimerization (12 → 13), and ketone reduction (13 → 14), were performed on late-stage macrocyclic intermediates to establish the three contiguous stereogenic centers of the target natural product (Scheme 2B).14
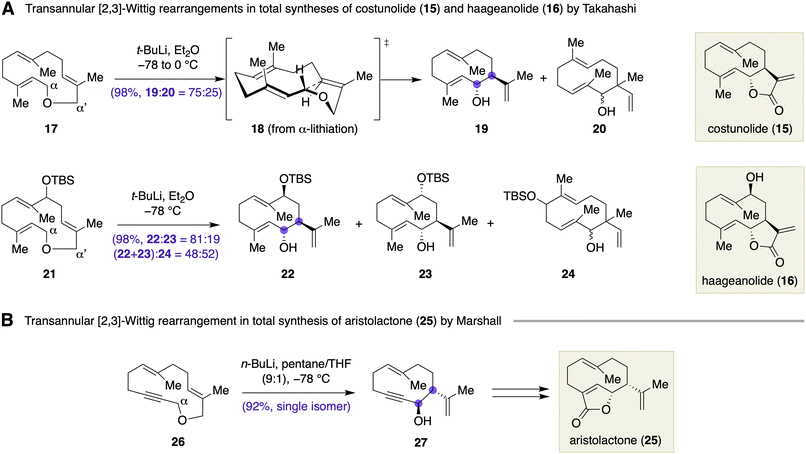
Takahashi and colleagues demonstrated total syntheses of sesquiterpene lactones, costunolide (15) and haageanolide (16), empowered by transannular [2,3]-Wittig rearrangement of macrocyclic diallyl ethers (Scheme 3A).15 Thus, treatment of macrocyclic diallyl ether 17, prepared from farnesyl acetate, with t-BuLi in Et2O at −78 to 0 °C provided a 75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 25 mixture of ring-contracted product 19 and its isomer 20. The lithiation of 17 preferred the α position relative to the α′ position. The stereochemical course of the rearrangement, giving the desired product 19 exclusively as trans isomer, was reasoned by transition state 18 that was generated by MM2 transition structure models. A regioselective lithiation/oxidation of 19 followed by MnO2 oxidation afforded costunolide (15). Total synthesis of haageanolide (16) was achieved in a similar fashion. Transannular [2,3]-Wittig rearrangement of macrocyclic diallyl ether 21 led to a mixture of ring-contracted products 22, 23, and 24. In this case, lithiation of 21 occurred with no regioselectivity between the α and α′ positions. Marshall et al. reported the total synthesis of aristolactone (25), in which a transannular [2,3]-Wittig rearrangement of macrocyclic propargylic ether 26 was used for obtaining ring-contracted product 27 in 92% yield as a single trans isomer (Scheme 3B).16 It appears that the lithiation of 26 with n-BuLi occurred exclusively at the α position to secure the desired product 27 in a high yield. These works represent an early example of late-stage skeletal reorganization in the total synthesis of macrocyclic natural products.
25 mixture of ring-contracted product 19 and its isomer 20. The lithiation of 17 preferred the α position relative to the α′ position. The stereochemical course of the rearrangement, giving the desired product 19 exclusively as trans isomer, was reasoned by transition state 18 that was generated by MM2 transition structure models. A regioselective lithiation/oxidation of 19 followed by MnO2 oxidation afforded costunolide (15). Total synthesis of haageanolide (16) was achieved in a similar fashion. Transannular [2,3]-Wittig rearrangement of macrocyclic diallyl ether 21 led to a mixture of ring-contracted products 22, 23, and 24. In this case, lithiation of 21 occurred with no regioselectivity between the α and α′ positions. Marshall et al. reported the total synthesis of aristolactone (25), in which a transannular [2,3]-Wittig rearrangement of macrocyclic propargylic ether 26 was used for obtaining ring-contracted product 27 in 92% yield as a single trans isomer (Scheme 3B).16 It appears that the lithiation of 26 with n-BuLi occurred exclusively at the α position to secure the desired product 27 in a high yield. These works represent an early example of late-stage skeletal reorganization in the total synthesis of macrocyclic natural products.
Because of their stereochemically rich, complex macrocyclic structures, macrolide antibiotics spurred the interest of the synthetic community during the late 20th century and stimulated investigations into macrocyclic stereocontrol. In 1978, the Corey group completed the total synthesis of erythronolide B (28) for the first time, in which they exploited macrocyclic stereocontrol to establish the C10 and C11 stereogenic centers at the final stage of the synthesis (Scheme 4A).17 Thus, stereoselective epoxidation of an α,β-unsaturated ketone derived from 29 by MnO2 oxidation, followed by the hydrogenolysis of the resultant epoxy ketone 30 gave alcohol 31, which underwent thermodynamic epimerization at C10 upon treatment with K2CO3 in methanol. The removal of acetonide completed the total synthesis of 28. A similar tactic was used in the Corey synthesis of erythronolide A.18
Thereafter, Still and Novack demonstrated an elegant synthesis of 3-deoxyrosaranolide (32) from macrolactone 33 through an extensive use of chirality-generating transformations on the macrocyclic backbone (Scheme 4B).11 Regioselective deprotonation of 33 at C8 with KHMDS and alkylation of the derived potassium enolate with MeI provided C8 methylated product 34 in 70% yield with 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 diastereoselection. Removal of the dithioacetal (>95%), followed by regio- and stereo-selective alkylation with LHMDS/BrCH2CO2t-Bu, gave C6 alkylated product 35 in 73% yield with ca. 6–10
1 diastereoselection. Removal of the dithioacetal (>95%), followed by regio- and stereo-selective alkylation with LHMDS/BrCH2CO2t-Bu, gave C6 alkylated product 35 in 73% yield with ca. 6–10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr. After transformation of 35 into α,β-unsaturated ketone 36, conjugate addition of PhSH and Raney Nickel desulfurization afforded C4 methylated product 37 in 44% overall yield with greater than 25
1 dr. After transformation of 35 into α,β-unsaturated ketone 36, conjugate addition of PhSH and Raney Nickel desulfurization afforded C4 methylated product 37 in 44% overall yield with greater than 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 diastereomer ratio. Cleavage of the t-butyl ester, acylation with ClCO2Et, and NaBH4 reduction provided alcohol 38 with 5
1 diastereomer ratio. Cleavage of the t-butyl ester, acylation with ClCO2Et, and NaBH4 reduction provided alcohol 38 with 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 diastereoselectivity at C5. After MnO2 oxidation of the C9 hydroxy group (75%), regio- and stereo-selective epoxidation of the Δ12,13 olefin with mCPBA gave rise to the corresponding epoxide in 88% yield with greater than 15
1 diastereoselectivity at C5. After MnO2 oxidation of the C9 hydroxy group (75%), regio- and stereo-selective epoxidation of the Δ12,13 olefin with mCPBA gave rise to the corresponding epoxide in 88% yield with greater than 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 diastereoselectivity. Finally, oxidation of the 1,4-diol moiety with (Ph3P)3RuCl2 furnished 3-deoxyrosaranolide (32) in 47% yield at 56% conversion. It is remarkable that all but two stereogenic centers along the macrocyclic backbone of 32 were set up at the post-macrocyclization stage.
1 diastereoselectivity. Finally, oxidation of the 1,4-diol moiety with (Ph3P)3RuCl2 furnished 3-deoxyrosaranolide (32) in 47% yield at 56% conversion. It is remarkable that all but two stereogenic centers along the macrocyclic backbone of 32 were set up at the post-macrocyclization stage.
Paterson and Rawson described an elegant synthesis of (+)-(9S)-dihydroerythronolide A (39), which involved chemo- and stereo-selective dihydroxylation of macrocyclic enol silyl ether 40, chelate-controlled zinc borohydride reduction of ketone 41, desilylation of 42, and stereoselective dihydroxylation of 43 (Scheme 4C).19 Thus, four out of eleven stereogenic centers of 39 were established at the final stage of the synthesis. Empowered by a late-stage chirality generation strategy, the present synthesis was completed in 20 steps from a simple starting material, (±)-2-methyl-3-phenylthiopropanal, and is remarkably concise given the complexity of the target molecule.
Late-stage chirality-generating functional group transformations
Succeeding examples
The scope of late-stage chirality-generating functional group transformations has now been extended to various macrocyclic natural products, demonstrating the conceptual generality of macrocyclic stereocontrol.Li, Yue, and co-workers described the total synthesis of (−)-ivorenolide A (57, Scheme 6), the unnatural enantiomer of an immunosuppressive 18-membered macrolide.21 This natural product is structurally unusual in that it contains a conjugated diyne in the macrocyclic skeleton. The Li/Yue synthesis of 57 involved stereoselective epoxidation of macrocyclic olefin 58, prepared via a Yamaguchi macrolactonization. Specifically, treatment of 58 with mCPBA in CH2Cl2 at room temperature furnished ivorenolide A (57) in 84% yield as the sole product. The stereochemical consequence may be counterintuitive but can be reasoned by the conformation of the precursor 58; it was assumed that mCPBA approached from the less hindered face of 58.22
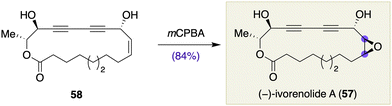 | ||
| Scheme 6 Stereoselective epoxidation in the total synthesis of (−)-ivorenolide A (57) by Li, Yue, and co-workers. | ||
Clark and Romiti reported the total synthesis of amphidinolide T1, T3, and T4 (59, 60, and 61, respectively, Scheme 7) from a common advanced intermediate by late-stage chirality-generating functional group transformations.23 Macrocyclic alkyne 62 was synthesized via a Yamaguchi macrolactonization. Ruthenium-catalyzed hydrosilylation of 62 under the catalysis of [Cp*Ru(MeCN)3]PF6 in the presence of (EtO)2MeSiH24 provided an approximately 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 separable mixture of vinylsilane 63 (44%) and 64 (45%). mCPBA epoxidation of 63 proceeded in a regio- and stereo-selective manner, and subsequent Tamao–Fleming oxidation of the derived silyl epoxide furnished amphidinolide T1 (59) and its 13-epimer 13-epi-59 in 73% and 7% yields, respectively. When the regioisomeric vinylsilane 64 was subjected to the same reaction sequence, however, amphidinolide T3 (60) and T4 (61) were afforded in 30% and 49% yields, respectively. This result indicated that only a moderate level of conformational bias was operating in the epoxidation of 64. In contrast, Shi asymmetric epoxidation25 of 64 with D-fructose-derived ketone 65 and Oxone, followed by Tamao–Fleming oxidation, gave rise to amphidinolide T3 (60) in 61% yield as a single diastereomer. Using L-fructose-derived ketone, ent-65, the same reaction sequence afforded amphidinolide T4 (61) in 57% yield as a single stereoisomer. This is a nice piece of work that illustrates substrate- and reagent-controlled epoxidations of macrocyclic olefins, which resulted in a collective synthesis of amphidinolides T1, T3, and T4.
1 separable mixture of vinylsilane 63 (44%) and 64 (45%). mCPBA epoxidation of 63 proceeded in a regio- and stereo-selective manner, and subsequent Tamao–Fleming oxidation of the derived silyl epoxide furnished amphidinolide T1 (59) and its 13-epimer 13-epi-59 in 73% and 7% yields, respectively. When the regioisomeric vinylsilane 64 was subjected to the same reaction sequence, however, amphidinolide T3 (60) and T4 (61) were afforded in 30% and 49% yields, respectively. This result indicated that only a moderate level of conformational bias was operating in the epoxidation of 64. In contrast, Shi asymmetric epoxidation25 of 64 with D-fructose-derived ketone 65 and Oxone, followed by Tamao–Fleming oxidation, gave rise to amphidinolide T3 (60) in 61% yield as a single diastereomer. Using L-fructose-derived ketone, ent-65, the same reaction sequence afforded amphidinolide T4 (61) in 57% yield as a single stereoisomer. This is a nice piece of work that illustrates substrate- and reagent-controlled epoxidations of macrocyclic olefins, which resulted in a collective synthesis of amphidinolides T1, T3, and T4.
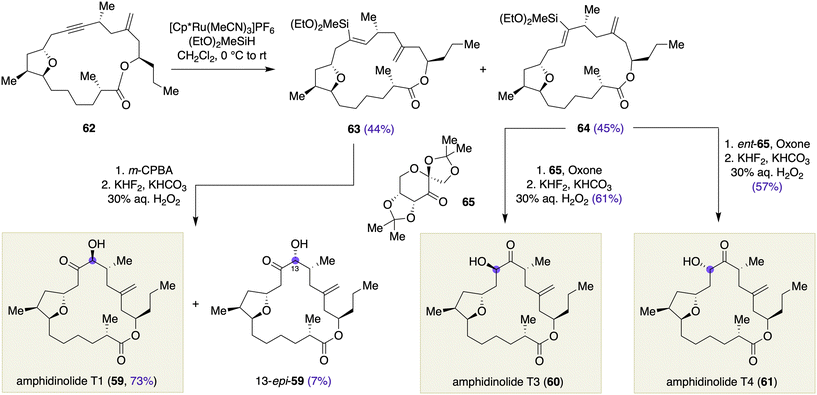 | ||
| Scheme 7 Substrate- and reagent-controlled epoxidations in the collective total synthesis of amphidinolides T1, T3, and T4 (59, 60, and 61) by Clark and Romiti. | ||
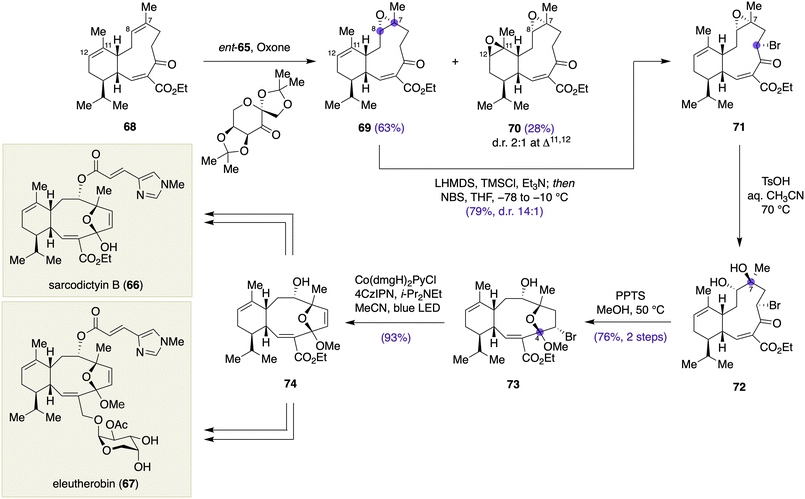
An expedient synthesis of sarcodictyin B (66) and eleutherobin (67) has been recently disclosed by Britton and colleagues, wherein late-stage oxidations on the carbon skeleton of eunicellin diterpenoid 68 was exploited for installing stereogenic centers at the C4, C7, and C8 positions (Scheme 8).26 Based on the X-ray structure and solution conformation of 68, Britton et al. envisioned that epoxidation of the Δ7,8 olefin would proceed on the Si face to give the corresponding epoxide with the correct configuration at C8. However, standard epoxidation conditions were problematic due to a competing side reaction at the Δ11,12 olefin. After screening a variety of reaction conditions, Shi asymmetric epoxidation of 68 using L-fructose-derived ketone ent-65 was found to provide the desired Δ7,8 epoxide 69 in 63% yield, along with diepoxide 70 in 28% yield (dr 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 at Δ11,12 epoxide). Treatment of 69 with LHMDS in the presence of TMSCl/Et3N, followed by NBS, gave bromide 71 in 79% yield with 14
1 at Δ11,12 epoxide). Treatment of 69 with LHMDS in the presence of TMSCl/Et3N, followed by NBS, gave bromide 71 in 79% yield with 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 diastereoselection. Acidic hydrolysis of the epoxide moiety of 71 occurred with an inversion of the configuration at C7 to deliver diol 72, which was then transformed to methyl ketal 73 upon exposure to PPTS in MeOH (76%, two steps), thereby establishing the stereogenic center at the C4 position. The subsequent photocatalytic halogen-atom transfer reaction under Leonori conditions27 cleanly eliminated the superfluous bromide to furnish dihydrofuran 74 (93%). Additional two- to five-step sequences of transformations completed the synthesis of sarcodictyin B (66) and eleutherobin (67).
1 diastereoselection. Acidic hydrolysis of the epoxide moiety of 71 occurred with an inversion of the configuration at C7 to deliver diol 72, which was then transformed to methyl ketal 73 upon exposure to PPTS in MeOH (76%, two steps), thereby establishing the stereogenic center at the C4 position. The subsequent photocatalytic halogen-atom transfer reaction under Leonori conditions27 cleanly eliminated the superfluous bromide to furnish dihydrofuran 74 (93%). Additional two- to five-step sequences of transformations completed the synthesis of sarcodictyin B (66) and eleutherobin (67).
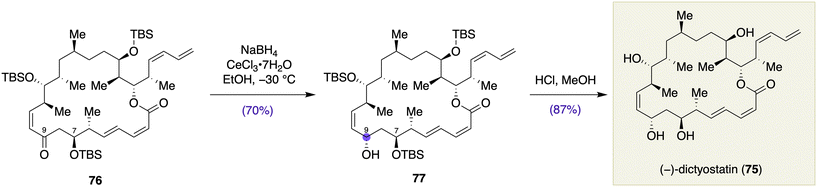 | ||
| Scheme 9 Stereoselective ketone reduction in the total synthesis of (−)-dictyostatin (75) by Paterson and co-workers. | ||
Our group has recently reported the total synthesis of (+)-neopeltolide (78) and its C9-epimer, 9-epi-78 (Scheme 10).29 Earlier studies by Kozmin,30 Floreancig,31 and us32 suggested that catalytic hydrogenation of exo-olefin 79 would proceed favorably on the sterically less encumbered β-face to deliver 80. Exo-olefin 79 could be synthesized by our tandem macrocyclization/pyran cyclization strategy (vide infra). As anticipated, hydrogenation of 79 provided 80 quantitatively with 84![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16 diastereoselection. Meanwhile, Mn-catalyzed hydrogen-atom transfer conditions (Mn(dpm)3, PhSiH3, t-BuOOH, i-PrOH)33 led to 9-epi-80 in 72% yield with 85
16 diastereoselection. Meanwhile, Mn-catalyzed hydrogen-atom transfer conditions (Mn(dpm)3, PhSiH3, t-BuOOH, i-PrOH)33 led to 9-epi-80 in 72% yield with 85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 diastereoselection. The stereochemical consequence could be reasoned by a late transition state model involving a tertiary alkyl radical intermediate with the C9 methyl group equatorially disposed to favor the ‘thermodynamic’ product 9-epi-80. Thus, late-stage stereodivergent reduction of 79, making use of its conformational bias, allowed an expedient access to (+)-neopeltolide (78) and 9-epi-neopeltolide (9-epi-78), both of which were potent antiproliferative agents against various human cancer cell lines.
15 diastereoselection. The stereochemical consequence could be reasoned by a late transition state model involving a tertiary alkyl radical intermediate with the C9 methyl group equatorially disposed to favor the ‘thermodynamic’ product 9-epi-80. Thus, late-stage stereodivergent reduction of 79, making use of its conformational bias, allowed an expedient access to (+)-neopeltolide (78) and 9-epi-neopeltolide (9-epi-78), both of which were potent antiproliferative agents against various human cancer cell lines.
 | ||
| Scheme 11 Stereoselective conjugate addition and Sharpless asymmetric epoxidation in the synthesis of the macrocyclic core 81 of laulimalide (82) by Paterson and co-workers. | ||
Ichikawa and co-workers described the total synthesis of (+)-acaulide (86) and (+)-acaulone A (87), which was highlighted by a late-stage double Michael addition inspired by a plausible biosynthetic mechanism (Scheme 12).35 Specifically, Michael addition of 6-methyldihydropyran-2,4-dione (88) to macrodiolide 89 under the influence of Et3N and AcOH in pyridine at 40 °C provided double adduct 90 in 41% yield, along with single adduct 91 in 34% yield. Presumably, 91 was produced via a hydrolysis of the dihydropyran-2,4-dione moiety and a subsequent decarboxylation. The stereoselectivity of the Michael addition was accounted for by conformational analysis based on the X-ray structure and NOE correlations of 89. Finally, the total synthesis of (+)-acaulide (86) and (+)-acaulone A (87) was completed by the deprotection of the benzyl groups.
 | ||
| Scheme 12 Stereoselective Michael addition in the total synthesis of (+)-acaulide (86) and (+)-acaulone A (87) by Ichikawa and co-workers. | ||
Myers and co-workers reported the synthesis of a >300-membered library of macrolide antibiotic candidates, including solithromycin (92),36 based on a convergent synthetic approach (Scheme 13). In the synthesis of solithromycin (92), Myers et al. forged the 14-membered macrolactone skeleton through acylketene macrocyclization of dioxinone 93. Thus, heating a solution of 93 in chlorobenzene at 132 °C resulted in macrolactone 94 in 66% yield as an approximately 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of diastereomers at the C2 position. Stereoselective fluorination of 94 at C2 was brought about using KOt-Bu/FN(SO2Ph)2 to give fluoride 95 (85%). Treatment of 95 with carbonyldiimidazole (CDI)/DBU provided the corresponding acyl imidazole at the C12 position. Finally, amide condensation with amine 96 and concomitant stereoselective intramolecular aza-Michael addition of transient intermediate 97 gave rise to solithromycin (92, 87% for two steps).
1 mixture of diastereomers at the C2 position. Stereoselective fluorination of 94 at C2 was brought about using KOt-Bu/FN(SO2Ph)2 to give fluoride 95 (85%). Treatment of 95 with carbonyldiimidazole (CDI)/DBU provided the corresponding acyl imidazole at the C12 position. Finally, amide condensation with amine 96 and concomitant stereoselective intramolecular aza-Michael addition of transient intermediate 97 gave rise to solithromycin (92, 87% for two steps).
Late-stage chirality-generating skeletal transformations: macrocyclizations
Late-stage skeletal transformations in the total synthesis of macrocyclic natural products may involve: (1) macrocyclizations for closing the macrocyclic skeleton; (2) transannular reactions for constructing embedded ring system(s) within the macrocyclic skeleton; and (3) transannular reactions for skeletal reorganizations. Concurrent generation of a stereogenic center(s) in these late-stage skeletal transformations should bring about step-economical synthesis of stereochemically rich macrocyclic natural products. In this section, selected examples of chirality-generating macrocyclizations will be discussed.Chirality-generating macrocyclizations
Macrocyclizations with concomitant generation of a stereogenic center(s) should expedite the synthesis of naturally occurring macrocycles having multiple stereogenic centers along the macrocyclic backbone. This section will only show selected examples of chirality-generating macrocyclizations. Additional examples may be found in recent reviews on this specific topic by Zheng and Hong.37![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 diastereoselectivity. In this reaction, the diethyl acetal functional group served as an equivalent to an aldehyde. Complementarily, olefinic aldehyde 100 also underwent Prins-type macrocyclization under the same reaction conditions to afford alcohol 99 in 68% yield with >20
1 diastereoselectivity. In this reaction, the diethyl acetal functional group served as an equivalent to an aldehyde. Complementarily, olefinic aldehyde 100 also underwent Prins-type macrocyclization under the same reaction conditions to afford alcohol 99 in 68% yield with >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 diastereoselectivity, after methanolysis. Thus, 100 served as a better precursor than 98 in terms of product yield and diastereoselectivity. The Mitsunobu reaction of 99 with α,β-unsaturated carboxylic acid 101 furnished (+)-neopeltolide (78). In these examples, the 14-membered macrolactone skeleton and its engrafted tetrahydropyran ring of (+)-neopeltolide were forged with spontaneous generation of two stereogenic centers in a single transformation.
1 diastereoselectivity, after methanolysis. Thus, 100 served as a better precursor than 98 in terms of product yield and diastereoselectivity. The Mitsunobu reaction of 99 with α,β-unsaturated carboxylic acid 101 furnished (+)-neopeltolide (78). In these examples, the 14-membered macrolactone skeleton and its engrafted tetrahydropyran ring of (+)-neopeltolide were forged with spontaneous generation of two stereogenic centers in a single transformation.
Due to the high stereochemical fidelity and mild reaction conditions, Prins-type macrocyclizations have been extensively applied to the synthesis of macrolide natural products embedded with tetrahydropyran ring(s), such as bryostatins,40 kendomycin,41 and polycavernoside A.42 In these remarkable applications, allylsilanes,40b–d allylbis(silane)s,40a and dioxinones43 were used as nucleophilic functional groups, thereby expanding the scope of Prins-type macrocyclizations.
Pattenden and Maleczka independently reported NHK macrocyclization strategies for the construction of the phomactin macrocyclic backbone (Scheme 15). The Pattenden group subjected iodoolefin-tethered aldehyde 115 to NHK macrocyclization (25 mol% NiCl2, 6 equiv. of CrCl2, DMSO) to obtain macrocyclic alcohol 116, representing the carbon skeleton of phomactins C and D, in 52% yield albeit as a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of diastereomers.46 Meanwhile, the Maleczka group found that the treatment of iodoolefin-tethered aldehyde 117 (E/Z 1.3
1 mixture of diastereomers.46 Meanwhile, the Maleczka group found that the treatment of iodoolefin-tethered aldehyde 117 (E/Z 1.3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) with 2 mol% Ni(acac)2 and 10 equiv. of CrCl2 in 1
1) with 2 mol% Ni(acac)2 and 10 equiv. of CrCl2 in 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 THF/DMSO (9 mM) at room temperature delivered macrocyclic alcohols 119E and 119Z in 33 and 27% yields, respectively, along with unreacted 117 in 25% yield.47 Importantly, the NHK macrocyclization was highly stereoselective and gave 119E and 119Z as single stereoisomers with respect to the newly generated stereogenic center. Maleczka et al. ascribed this stereochemical consequence to the steric and/or dipole interaction(s) between the formyl group and the silyloxy group in 118. Thus, the local structure around the cyclohexane ring was crucial to the success of the chirality generation.
3 THF/DMSO (9 mM) at room temperature delivered macrocyclic alcohols 119E and 119Z in 33 and 27% yields, respectively, along with unreacted 117 in 25% yield.47 Importantly, the NHK macrocyclization was highly stereoselective and gave 119E and 119Z as single stereoisomers with respect to the newly generated stereogenic center. Maleczka et al. ascribed this stereochemical consequence to the steric and/or dipole interaction(s) between the formyl group and the silyloxy group in 118. Thus, the local structure around the cyclohexane ring was crucial to the success of the chirality generation.
Marshall and Eidam disclosed the formal synthesis of callipeltoside A aglycone, in which they attempted to construct the 14-membered macrolactone skeleton with the concomitant generation of the C9 stereogenic center (Scheme 16).48 The exposure of aldehyde iodoolefin 121 to 6 mol% NiCl2(dppp) and 7 equiv. of CrCl2 in DMSO/Me2S (50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) at room temperature delivered macrocyclic alcohol 122 in 71% yield as a single stereoisomer, although the configuration at the C9 position was opposite to the desired one. The stereochemical course of the NHK macrocyclization was reasoned by a torsional interaction model. Because TS-A suffers from the torsional strain between the existing C8 methyl group and the generating C9 alkoxy group, the reaction proceeds viaTS-B to avoid such an unfavorable interaction and gives the undesired diastereomer 122. While this example was not entirely successful, it illustrates the difficulties in predicting the stereochemistry of chirality-generating macrocyclizations.
1, v/v) at room temperature delivered macrocyclic alcohol 122 in 71% yield as a single stereoisomer, although the configuration at the C9 position was opposite to the desired one. The stereochemical course of the NHK macrocyclization was reasoned by a torsional interaction model. Because TS-A suffers from the torsional strain between the existing C8 methyl group and the generating C9 alkoxy group, the reaction proceeds viaTS-B to avoid such an unfavorable interaction and gives the undesired diastereomer 122. While this example was not entirely successful, it illustrates the difficulties in predicting the stereochemistry of chirality-generating macrocyclizations.
 | ||
| Scheme 16 NHK macrocyclization in the total synthesis of callipeltoside A (120) by Eidam and Marshall. | ||
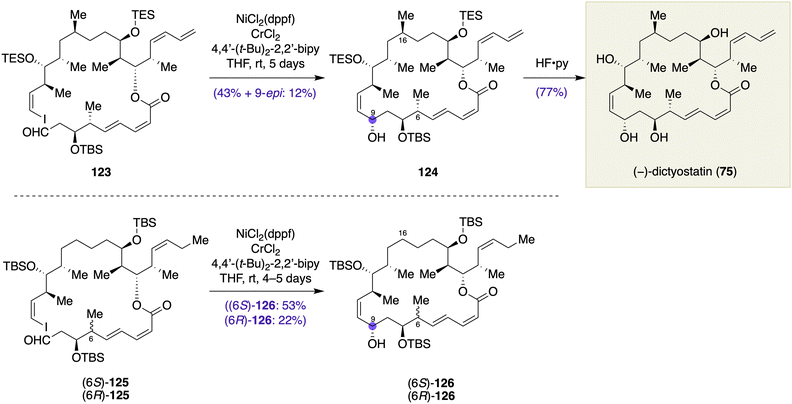
Curran and co-workers described in their total synthesis of (−)-dictyostatin (75) and its stereoisomers the NHK macrocyclization of aldehyde iodoolefin 123 to close the 22-membered macrocyclic skeleton with concomitant formation of the stereogenic center at the C9 position, giving macrocyclic alcohol 124 in 43% yield along with its C9 epimer in 12% yield (Scheme 17).49 The macrocyclization was executed at the final stage of the synthesis. Importantly, Curran et al. observed in their preliminary optimization studies that subtle structural changes in the macrocyclization precursor had significant influence on the stereochemical consequence of the macrocyclization. Thus, NHK macrocyclization of aldehyde iodoolefin 6S-125 provided macrocyclic alcohol 126 with desired C9 configuration in 53% yield as a single stereoisomer, whereas that of 6R-125 resulted in a 75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 25 mixture of diastereomers at C9 (22% isolated yield). It appears that the remote stereogenic centers at C6 and even at C16 would have an impact on the transition state of the macrocyclization.
25 mixture of diastereomers at C9 (22% isolated yield). It appears that the remote stereogenic centers at C6 and even at C16 would have an impact on the transition state of the macrocyclization.
Total syntheses of structurally complex cyclic imine neurotoxins by Kishi and co-workers were based on a late-stage Diels–Alder macrocyclization strategy (Scheme 18).53 In the first total synthesis of (−)-pinnatoxin A (127), the unnatural enantiomer, SN2′ displacement of mesylate 128 with DABCO, followed by elimination of the derived adduct with Et3N, resulted in the generation of the transient diene 129, which underwent Diels–Alder macrocyclization in dodecane (0.2 mM) at 70 °C to give the desired exo-product 130a, along with the undesired exo-product 130b and endo-product 130c in 78% combined yield with 1.0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.9
0.9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.4 selectivity. Kishi et al. noted that the exo-selectivity of the Diels–Alder macrocyclization was dependent on the local structure of the C25–C32 moiety. Subsequently, in their total synthesis of pteriatoxins, the Kishi group investigated in detail into the stereochemistry of Diels–Alder macrocyclization using a series of substrates.54 Kishi et al. found that the reactivity and exo/endo-selectivity of Diels–Alder macrocyclization of dienes 132 depended on the configuration at the C34 position and the protecting group ‘X’ of the C34 and C35 hydroxy groups. Diels–Alder macrocyclization of bis(p-methoxybenzoate) 132 gave the best result and provided the desired product 133 in 51% isolated yield.
0.4 selectivity. Kishi et al. noted that the exo-selectivity of the Diels–Alder macrocyclization was dependent on the local structure of the C25–C32 moiety. Subsequently, in their total synthesis of pteriatoxins, the Kishi group investigated in detail into the stereochemistry of Diels–Alder macrocyclization using a series of substrates.54 Kishi et al. found that the reactivity and exo/endo-selectivity of Diels–Alder macrocyclization of dienes 132 depended on the configuration at the C34 position and the protecting group ‘X’ of the C34 and C35 hydroxy groups. Diels–Alder macrocyclization of bis(p-methoxybenzoate) 132 gave the best result and provided the desired product 133 in 51% isolated yield.
 | ||
| Scheme 18 Diels–Alder macrocyclizations in the total syntheses of (−)-pinnatoxin A (127) and pteriatoxin A (131) by Kishi and co-workers. | ||
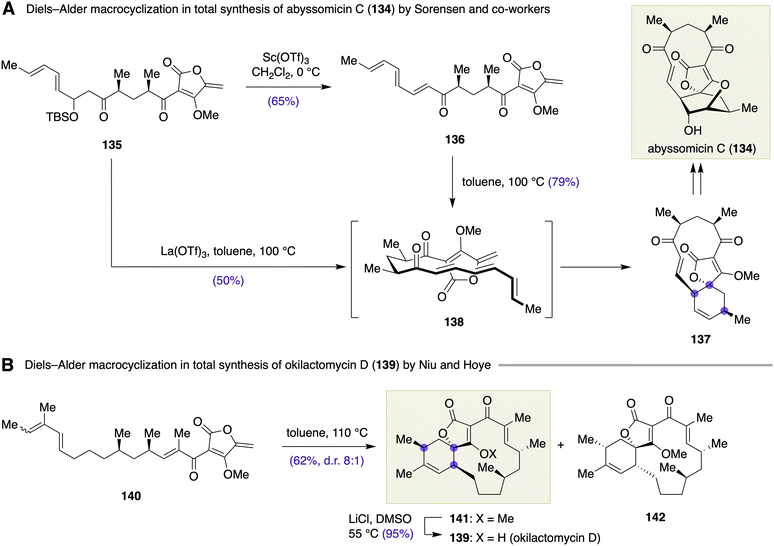
Sorensen and co-workers demonstrated that a Diels–Alder macrocyclization was remarkably effective for constructing the carbon skeleton of abyssomicin C (134) (Scheme 19A).55 The elimination of the β-silyloxy group from ketone 135 led to sensitive trienone 136 (65%), which underwent Diels–Alder macrocyclization to deliver macrocycle 137 upon heating in toluene at 100 °C (79%). The reaction was supposed to proceed via transition state 138, which accounted for the observed stereochemical consequence. Tandem elimination/Diels–Alder macrocyclization from 135 was also possible by treatment with La(OTf)3 in toluene at 100 °C, providing 137 in 50% yield. A similar Diels–Alder macrocyclization strategy was successfully implemented in the total synthesis of okilactomycin D (139) by Niu and Hoye (Scheme 19B).56
The Danishefsky group employed an intramolecular aldol reaction for the total synthesis of an immunosuppressive macrocyclic natural product, rapamycin (143) (Scheme 20).57 The macrocyclization of 144 was achieved through an intramolecular aldol reaction via a titanium enolate generated by treatment with TiCl3(Oi-Pr), furnishing macrocycle 145 in 33% yield as a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.3 diastereomeric mixture. Subsequent desilylation of 145 provided rapamycin (143) in 85% yield. Importantly, this titanium enolate-mediated aldol macrocyclization enabled the simultaneous construction of the 31-membered ring and installation of the two stereogenic centers, although the product yield and the diastereoselectivity were moderate. By merging macrocycle formation and chirality generation into a single transformation, this strategy provided an efficient solution to the total synthesis of a large and architecturally complex macrolide natural product, rapamycin.
2.3 diastereomeric mixture. Subsequent desilylation of 145 provided rapamycin (143) in 85% yield. Importantly, this titanium enolate-mediated aldol macrocyclization enabled the simultaneous construction of the 31-membered ring and installation of the two stereogenic centers, although the product yield and the diastereoselectivity were moderate. By merging macrocycle formation and chirality generation into a single transformation, this strategy provided an efficient solution to the total synthesis of a large and architecturally complex macrolide natural product, rapamycin.
 | ||
| Scheme 20 Aldol macrocyclization in the total synthesis of rapamycin (143) by Danishefsky and co-workers. | ||
The Sammakia group assembled the macrocyclic skeleton of (+)-peloruside A (146) through an intramolecular vinylogous aldol reaction58,59 (Scheme 21). The exposure of 147 to 2.0 equiv. of lithium 2,2,6,6-tetramethylpiperidine (LTMP) and 2.2 equiv. of aluminum tris(2,6-diphenylphenoxide) (ATPH)60 in toluene/THF at −48 °C delivered macrocyclic alcohol 148 in 86% yield as a 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 diastereomeric mixture; however, the configuration at the C5 position of the major diastereomer was opposite to the desired configuration. Sammakia et al. noted that the stereochemistry of the cyclization could be dictated by the conformation of the forming macrocycle and that a different conformation could provide the desired stereochemical outcome. Therefore, the vinylogous aldol macrocyclization of silylene 149 was investigated. Subjection of 149 to the intramolecular vinylogous aldol reaction conditions using LTMP/ATPH provided macrocyclic alcohol 150 in 84% yield as a 6
1 diastereomeric mixture; however, the configuration at the C5 position of the major diastereomer was opposite to the desired configuration. Sammakia et al. noted that the stereochemistry of the cyclization could be dictated by the conformation of the forming macrocycle and that a different conformation could provide the desired stereochemical outcome. Therefore, the vinylogous aldol macrocyclization of silylene 149 was investigated. Subjection of 149 to the intramolecular vinylogous aldol reaction conditions using LTMP/ATPH provided macrocyclic alcohol 150 in 84% yield as a 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 diastereomeric mixture, where the major diastereomer had the desired absolute configuration at C5. Subsequent transformations of 150, including an Achmatowicz reaction, afforded peloruside A macrolactone core 151. Thus, the silylene group within 149 served successfully as a ring constraint for directing the vinylogous aldol macrocyclization to give macrocyclic alcohol 150 with correct configuration, although it is still difficult to rationalize the stereochemical consequence of this specific case.
1 diastereomeric mixture, where the major diastereomer had the desired absolute configuration at C5. Subsequent transformations of 150, including an Achmatowicz reaction, afforded peloruside A macrolactone core 151. Thus, the silylene group within 149 served successfully as a ring constraint for directing the vinylogous aldol macrocyclization to give macrocyclic alcohol 150 with correct configuration, although it is still difficult to rationalize the stereochemical consequence of this specific case.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
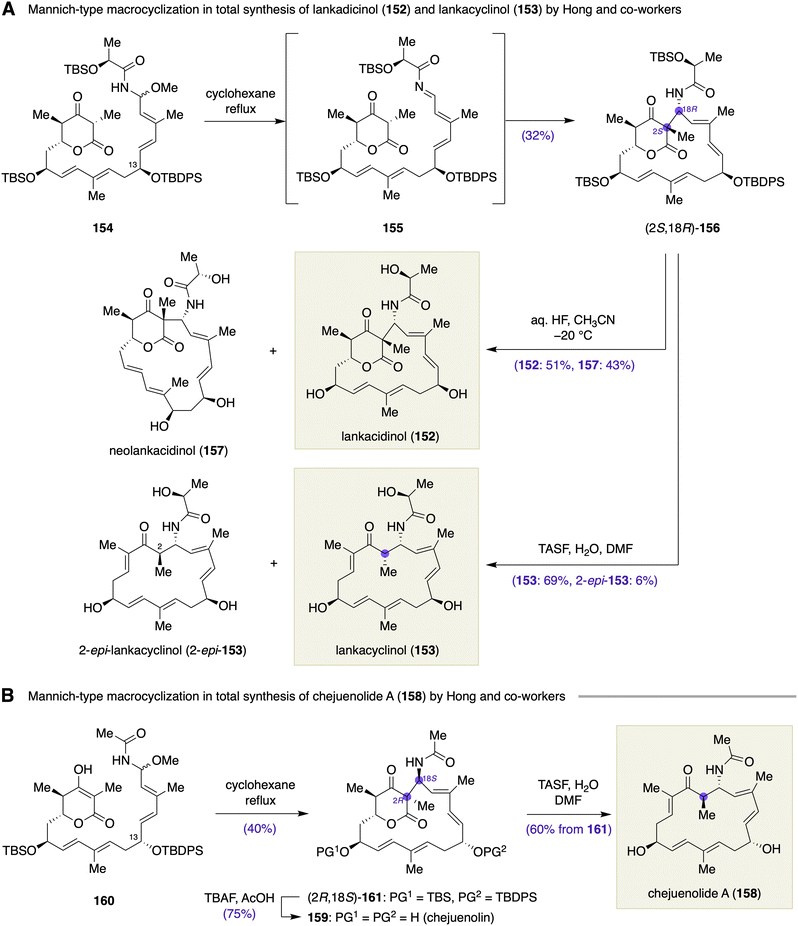
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of 152 and 157 (38%, 88% based on recovered starting material). Meanwhile, treatment of (2S,18R)-156 with TASF and H2O in DMF resulted in a decarboxylative collapse of the δ-lactone with concomitant desilylation to deliver lankacyclinol (153) in 69% yield and its 2-epimer, 2-epi-153, in 6% yield. Hong et al. further demonstrated that (2S,18R)-156 was a versatile intermediate for the total synthesis of other members of the lankacidin antibiotics including lankacidin A and C, lankacidinol A, and lankacyclinol A.
1 mixture of 152 and 157 (38%, 88% based on recovered starting material). Meanwhile, treatment of (2S,18R)-156 with TASF and H2O in DMF resulted in a decarboxylative collapse of the δ-lactone with concomitant desilylation to deliver lankacyclinol (153) in 69% yield and its 2-epimer, 2-epi-153, in 6% yield. Hong et al. further demonstrated that (2S,18R)-156 was a versatile intermediate for the total synthesis of other members of the lankacidin antibiotics including lankacidin A and C, lankacidinol A, and lankacyclinol A.
Hong and co-workers applied their biomimetic Mannich-type macrocyclization to the total synthesis of chejuenolide A (158) by assuming a hypothetical biosynthetic precursor, chejuenolin (159) (Scheme 22B).62 A solution of N,O-acetal 160 in cyclohexane was refluxed to give (2R,18S)-161 in 40% yield as the major diastereomer, along with (2S,18S)-161 and (2S,18R)-161 (structures not shown) in 7 and 13% yields, respectively. The major diastereomer (2R,18S)-161 represents a protected form of chejuenolin (159). In comparison with the lankacidinol case, the stereochemical outcome of the Mannich-type macrocyclization of 160 was likely dependent on the configuration at the C13 position. Thus, it seems that the C13 stereogenic center has a significant impact on the conformation of N,O-acetal 160, the macrocyclization precursor, and consequently, it affects the transition state of the Mannich-type macrocyclization. Deprotection of the silyl groups from (2R,18S)-161 with TBAF buffered with AcOH afforded chejuenolin (159). Meanwhile, the desilylation of (2R,18S)-161 with TASF and H2O in DMF triggered concomitant decarboxylation to afford chejuenolide A (158) in 60% yield.
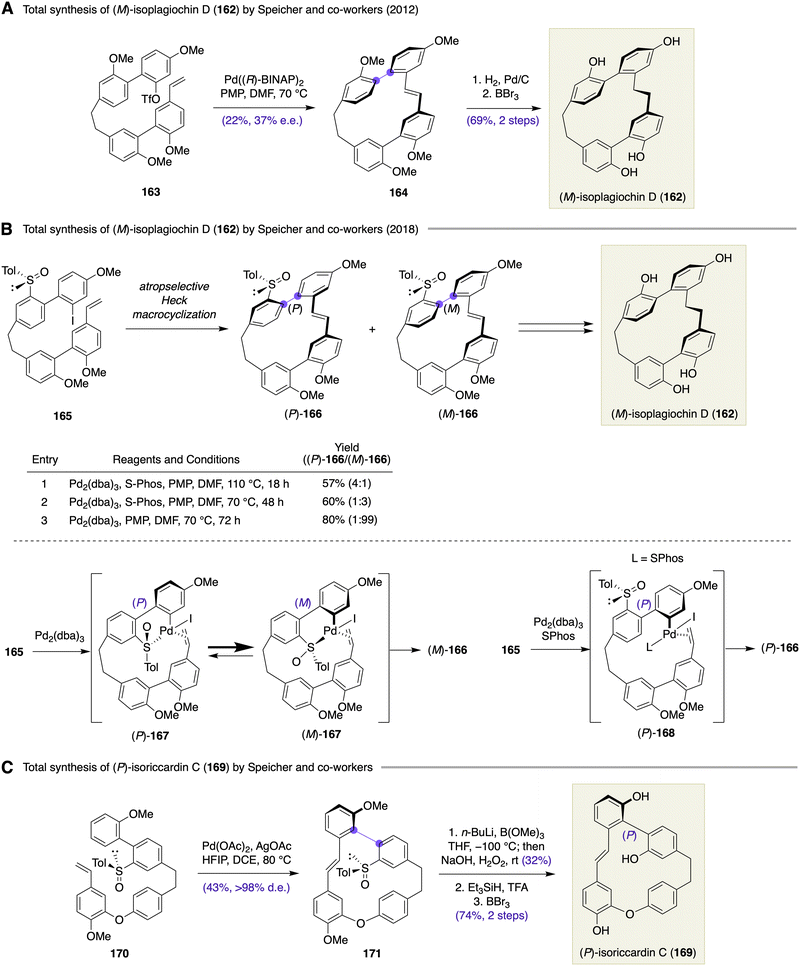
Speicher and co-workers reported an atropselective Heck macrocyclization64 for enantioselective total synthesis of (M)-isoplagiochin D (162), a bis(bibenzyl) natural product (Scheme 23A).65 Heck macrocyclization of olefinic triflate 163 was examined under various conditions. When the reaction was performed in the presence of the preformed Pd((R)-BINAP)2 complex as the catalyst and PMP as the base in DMF at 70 °C, (M)-164 could be obtained in 22% yield with 37% ee. Although the product yield and optical purity were moderate, atropselective Heck macrocyclization was certainly a viable approach for the construction of the strained macrocyclic skeleton of 162.
Subsequently, Speicher et al. described atropselective Heck macrocyclization of iodide 165 as an improved approach toward isoplagiochin D (Scheme 23B).66 The macrocyclization precursor 165 was substituted with a chiral sulfinyl group, which served as an atropselectivity inducer. Interestingly, the atropselectivity of the macrocyclization of 165 was found to be dependent on the reaction conditions. Treatment of 165 with Pd2(dba)3/S-Phos and PMP in DMF at 110 °C provided (P)-166 in 57% yield with 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 atropselectivity. Interestingly, the atropselectivity reversed upon lowering the reaction temperature to 70 °C and gave (M)-166 in 60% yield with 3
1 atropselectivity. Interestingly, the atropselectivity reversed upon lowering the reaction temperature to 70 °C and gave (M)-166 in 60% yield with 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 atropselectivity. The reaction of 165 under the catalysis of Pd2(dba)3 resulted in further enhancement of the atropselectivity toward (M)-166, which was isolated in 80% yield with 99
1 atropselectivity. The reaction of 165 under the catalysis of Pd2(dba)3 resulted in further enhancement of the atropselectivity toward (M)-166, which was isolated in 80% yield with 99![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 selectivity. The observed atropselectivity switching between (P)-166 and (M)-166 could be reasoned by the following scenario. Under kinetic control (Pd2(dba)3, 1,2,2,6,6-pentamethylpiperidine (PMP), DMF, 70 °C), oxidative addition of 165 to the ‘ligandless’ palladium complex results in the formation of an equilibrating mixture of palladacycles (P)-167 and (M)-167. Through energetically more favored (M)-167, insertion into the proximal olefin occurs to afford (M)-166. Meanwhile, oxidative addition of 165 to Pd(S-Phos) complex under thermodynamic conditions (Pd2(dba)3/S-Phos, PMP, DMF, 110 °C) gives organopalladium intermediate (P)-168, which undergoes insertion into the olefin to provide (P)-166 as the major isomer. As a relevant example, Speicher et al. achieved an enantioselective total synthesis of (P)-isoriccardin C (169) through a chiral sulfinyl group-directed atropselective macrocyclic C–H activation (Scheme 23C).67
1 selectivity. The observed atropselectivity switching between (P)-166 and (M)-166 could be reasoned by the following scenario. Under kinetic control (Pd2(dba)3, 1,2,2,6,6-pentamethylpiperidine (PMP), DMF, 70 °C), oxidative addition of 165 to the ‘ligandless’ palladium complex results in the formation of an equilibrating mixture of palladacycles (P)-167 and (M)-167. Through energetically more favored (M)-167, insertion into the proximal olefin occurs to afford (M)-166. Meanwhile, oxidative addition of 165 to Pd(S-Phos) complex under thermodynamic conditions (Pd2(dba)3/S-Phos, PMP, DMF, 110 °C) gives organopalladium intermediate (P)-168, which undergoes insertion into the olefin to provide (P)-166 as the major isomer. As a relevant example, Speicher et al. achieved an enantioselective total synthesis of (P)-isoriccardin C (169) through a chiral sulfinyl group-directed atropselective macrocyclic C–H activation (Scheme 23C).67
A pioneering example was described by Stang and White in their total synthesis of 6-deoxyerythronolide B (172) via a late-stage palladium-catalyzed allylic C–H oxidation in a macrocyclization format (Scheme 24A).68 Thus, olefin-tethered carboxylic acid 173, synthesized through standard aldol chemistry, underwent allylic C–H oxidative macrocyclization under the catalysis of Pd(OAc)2·(PhS(O)CH2)2 in the presence of benzoquinone to afford macrolactone 175 with the correct configuration at C13 (56% yield plus 8% r.s.m. after two recycles). The reaction was thought to involve a π-allyl–Pd–carboxylate intermediate 174 and proceed through a product-like transition state. The actual product 175 was calculated to be 3 kcal mol−1 more stable than 13-epi-175 (structure not shown).
Haydl and Breit demonstrated in their total synthesis of epothilone D (176), a rhodium-catalyzed macrocyclization of an advanced alkyne carboxylic acid (Scheme 24B).69 Specifically, the reaction of 177 in the presence of [Rh(cod)Cl]2/DPEPhos and benzoic acid in DCE at 70 °C afforded macrolactone 178 in 43% yield with dr 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.
1.
Because of the unique ability to inhibit bacterial type 1 signal peptidase, arylomycins have caught significant attention from the synthetic community as a promising lead for the development of new therapeutics against drug-resistant bacterial infections. Arylomycins constitute a family of lipopeptide antibiotics having a characteristic biaryl-containing 14-membered macrocycle and exist as a mixture of two atropisomers at room temperature, as indicated by NMR spectroscopy. The first total synthesis of arylomycin A2 (179) was achieved by the group of Romesberg, who demonstrated a Suzuki–Miyaura macrocyclization for atropselective closure of the macrocycle of 179 (Scheme 25A).72 After detailed optimization experiments, they found that Suzuki–Miyaura macrocyclization73 of 180 in the presence of PdCl2(dppf) as the catalyst and K2CO3 as the base in CH3CN at 80 °C, followed by deprotection of the Boc group of 181, afforded macrocycle 182 in 49% yield for the two steps. NMR studies on model macrocycles indicated that the axial chirality depends on the presence or absence of the N-methyl group of the phenylglycine residue; the aS configuration was favored in the presence of the N-methyl group. X-ray crystallographic analysis of an arylomycin A2-signal peptidase complex showed that only aS configured arylomycin A2 was bound to signal peptidase.74
Shortly after the first total synthesis of arylomycin A2 (179) by the Romesberg group, Zhu and co-workers disclosed the second total synthesis of 179, which involved an atropselective Suzuki–Miyaura macrocyclization of 183, which corresponded to the N-methylated counterpart of 180 (Scheme 25B).75 Extensive optimization experiments showed that the reaction proceeded most efficiently under the catalysis of PdCl2(S-Phos)2 in the presence of NaHCO3 as the base in toluene/H2O (30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) at 90 °C, giving macrocycle 184 in 54% yield along with its epimer at the 4-hydroxyphenylglycine residue in 9% yield. Zhu et al. also completed the total synthesis of arylomycin B2 based on the Suzuki–Miyaura macrocyclization strategy.
1) at 90 °C, giving macrocycle 184 in 54% yield along with its epimer at the 4-hydroxyphenylglycine residue in 9% yield. Zhu et al. also completed the total synthesis of arylomycin B2 based on the Suzuki–Miyaura macrocyclization strategy.
Even more complex macrocyclic peptides with axial chirality are chloropeptin I (185) and complestatin (186, chloropeptin II) (Scheme 26). Both natural products have an aR configuration at the biaryl axis, and complestatin (186) isomerizes to chloropeptin I (185) under acidic conditions with the retention of the axial configuration. These non-ribosomal peptides have a couple of strained macrocycles, one of which is responsible for their atropisomeric chirality. Because of their highly complex structures and potential as an HIV therapeutic, these macrocyclic peptides have garnered significant interest from the synthetic community.
The first total synthesis of chloropeptin I (185) was accomplished by Hoveyda, Snapper, and co-workers, who made use of a Stille macrocyclization76 of aryl stannane 187 (Pd(Pt-Bu3)2, collidine, CsF, dioxane, 50 °C) to forge the ‘right-hand’ biaryl macrocycle with the desired aR configuration atropselectively (Scheme 26A).77 Hoveyda et al. later disclosed the total synthesis of isocomplestatin (189), an unnatural atropisomer of complestatin (186), through a Suzuki–Miyaura macrocyclization (Scheme 26B).78 Thus, the reaction of aryl boronate 190 under the influence of PdCl2(dppf)·CH2Cl2 as the catalyst and K2CO3 as the base in dioxane/H2O (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) at 80 °C provided macrocycle 191, i.e., isocomplestatin methyl ester, in 63% yield as a single stereoisomer, which had an aS configuration at the biaryl axis. Hydrolysis of the methyl ester completed the total synthesis of isocomplestatin (189). Quite interesting was the observation that the Stille macrocyclization of arylstannane 187 provided chloropeptin-type bis-macrocycle 188 with the natural aR configuration, whereas the Suzuki–Miyaura macrocyclization of aryl boronate 190 delivered complestatin-type bis-macrocycle 191 with the unnatural aS configuration. Moreover, a model compound lacking the ‘left-hand’ biaryl ether macrocycle also underwent Suzuki–Miyaura macrocyclization but delivered the corresponding macrocyclic product without atropselectivity. These results suggested the possibility that the stereochemical consequence of macrocyclizations may be highly substrate structure dependent.
1) at 80 °C provided macrocycle 191, i.e., isocomplestatin methyl ester, in 63% yield as a single stereoisomer, which had an aS configuration at the biaryl axis. Hydrolysis of the methyl ester completed the total synthesis of isocomplestatin (189). Quite interesting was the observation that the Stille macrocyclization of arylstannane 187 provided chloropeptin-type bis-macrocycle 188 with the natural aR configuration, whereas the Suzuki–Miyaura macrocyclization of aryl boronate 190 delivered complestatin-type bis-macrocycle 191 with the unnatural aS configuration. Moreover, a model compound lacking the ‘left-hand’ biaryl ether macrocycle also underwent Suzuki–Miyaura macrocyclization but delivered the corresponding macrocyclic product without atropselectivity. These results suggested the possibility that the stereochemical consequence of macrocyclizations may be highly substrate structure dependent.
Zhu et al. carried out more detailed investigations into the atropselectivity of Suzuki–Miyaura macrocyclizations toward the total synthesis of complestatin (186) (Scheme 27).79 Specifically, Zhu and co-workers found that Suzuki–Miyaura reaction of linear tripeptide 192 under the catalysis of PdCl2(dppf)·CH2Cl2 in the presence of K2CO3 in dioxane/H2O (15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) at 80 °C afforded macrocycle 193 in 66% yield with an aR configuration. The macrocycle 193 represents the DEFG-ring macrocycle of complestatin (186). In contrast, intramolecular Suzuki–Miyaura reaction of hexapeptide 194 with the biaryl ether macrocycle under essentially the same reaction conditions delivered macrocycle 195 in 52% yield with an aS configuration, which could be converted to isocomplestatin methyl ester (191) by desilylation.80 Similarly, intramolecular Suzuki–Miyaura reaction of hexapeptide 190, in which the D-ring phenol was unprotected, provided macrocycle 191 in 66% yield with an aS configuration. These results strongly indicated that the atropselectivity of Suzuki–Miyaura macrocyclization in the complestatin case was highly substrate dependent. This was further underscored by Suzuki–Miyaura macrocyclizations of 196 and 197, both of which are epimers at the aryl glycine residue C. These isomers showed a significant change in the peptide backbone conformation, and upon exposure to the optimized conditions, it led to macrocyclization products 198 and 199 with an aS configuration.
1) at 80 °C afforded macrocycle 193 in 66% yield with an aR configuration. The macrocycle 193 represents the DEFG-ring macrocycle of complestatin (186). In contrast, intramolecular Suzuki–Miyaura reaction of hexapeptide 194 with the biaryl ether macrocycle under essentially the same reaction conditions delivered macrocycle 195 in 52% yield with an aS configuration, which could be converted to isocomplestatin methyl ester (191) by desilylation.80 Similarly, intramolecular Suzuki–Miyaura reaction of hexapeptide 190, in which the D-ring phenol was unprotected, provided macrocycle 191 in 66% yield with an aS configuration. These results strongly indicated that the atropselectivity of Suzuki–Miyaura macrocyclization in the complestatin case was highly substrate dependent. This was further underscored by Suzuki–Miyaura macrocyclizations of 196 and 197, both of which are epimers at the aryl glycine residue C. These isomers showed a significant change in the peptide backbone conformation, and upon exposure to the optimized conditions, it led to macrocyclization products 198 and 199 with an aS configuration.
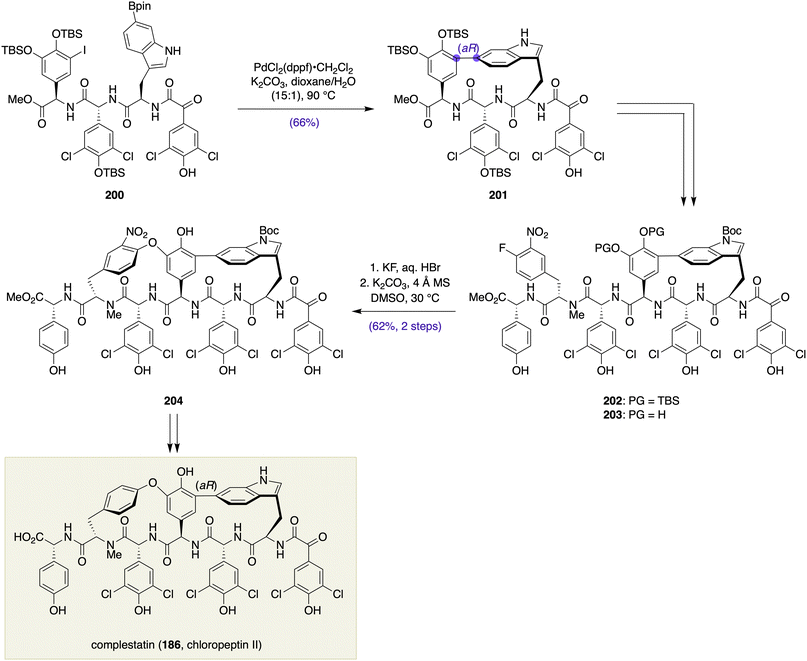
Accordingly, the Zhu group accomplished the total synthesis of complestatin (186) by initially forging the phenyl–indole linkage from a linear tripeptide precursor by a Suzuki–Miyaura macrocyclization to establish the desired aR configuration, followed by building the biaryl ether macrocycle via an intramolecular SNAr cyclization (Scheme 28).81 Thus, Suzuki–Miyaura macrocyclization of linear tripeptide 200 was achieved under the previously optimized conditions to give macrocycle 201 in 66% yield with the desired aR configuration. After coupling with a tripeptide fragment, the macrocyclization of the resultant hexapeptide 203 by the action of K2CO3 and 4 Å MS in DMSO afforded bis-macrocycle 204 in 62% yield from 202 with excellent regio- and atrop-selectivities. Although it was of no consequence, the atropselectivity of this base-mediated macrocyclization was also substrate dependent. The advanced intermediate 204 was finally transformed into complestatin (186) in four steps, including the removal of the superfluous nitro group and protecting groups. The Zhu synthesis of complestatin (186) clearly underscores the importance of substrate design in directing the stereochemical outcome of the Suzuki–Miyaura macrocyclization, although it was carried out at an early stage.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) at 110 °C provided Larock macrocyclization product 205 in 89% yield with 4
1) at 110 °C provided Larock macrocyclization product 205 in 89% yield with 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 atropselectivity, favoring the desired aR isomer. The ‘left-hand’ biaryl ether macrocycle was subsequently closed via an SNAr macrocyclization to complete the first total synthesis of complestatin (186). In the second-generation synthesis, Boger et al. demonstrated a late-stage Larock macrocyclization on advanced intermediate 207 in which the ‘left-hand’ biaryl ether macrocycle was already in place.84 Thus, exposure of 207 to the optimized reaction conditions (Pd(OAc)2, DtBPF, Et3N, toluene/CH3CN (10
1 atropselectivity, favoring the desired aR isomer. The ‘left-hand’ biaryl ether macrocycle was subsequently closed via an SNAr macrocyclization to complete the first total synthesis of complestatin (186). In the second-generation synthesis, Boger et al. demonstrated a late-stage Larock macrocyclization on advanced intermediate 207 in which the ‘left-hand’ biaryl ether macrocycle was already in place.84 Thus, exposure of 207 to the optimized reaction conditions (Pd(OAc)2, DtBPF, Et3N, toluene/CH3CN (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), 110 °C) furnished bis-macrocycle 208 in 56% yield with >20
1), 110 °C) furnished bis-macrocycle 208 in 56% yield with >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 atropselectivity. The stereochemical outcome of this Larock macrocyclization was in sharp contrast to those of relevant Suzuki–Miyaura macrocyclizations previously reported by the Hoveyda/Snapper group and the Zhu group. Boger et al. speculated that the atropselectivity of Larock macrocyclizations in the complestatin case appeared to be governed by a steric contact between the aniline protecting group and the peptide backbone. The Boger group also completed the total synthesis of complestatin A and B (neuroprotectin A and B), oxidized derivatives of complestatin, through a Larock macrocyclization.85
1 atropselectivity. The stereochemical outcome of this Larock macrocyclization was in sharp contrast to those of relevant Suzuki–Miyaura macrocyclizations previously reported by the Hoveyda/Snapper group and the Zhu group. Boger et al. speculated that the atropselectivity of Larock macrocyclizations in the complestatin case appeared to be governed by a steric contact between the aniline protecting group and the peptide backbone. The Boger group also completed the total synthesis of complestatin A and B (neuroprotectin A and B), oxidized derivatives of complestatin, through a Larock macrocyclization.85
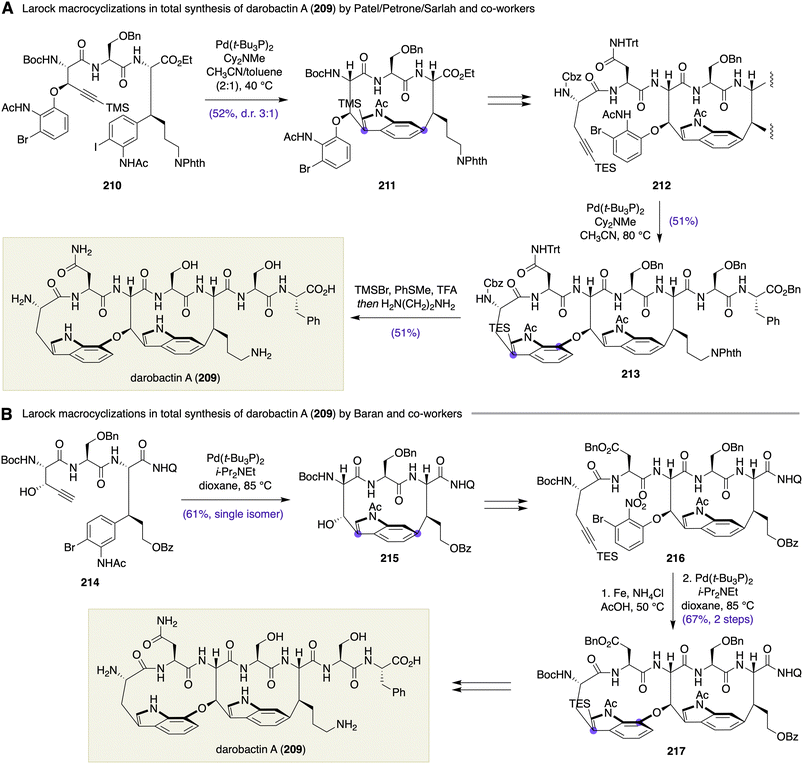
More recently, the Patel/Petrone/Sarlah group and the Baran group independently disclosed total synthesis of darobactin A (209) (Scheme 30). This natural product is a structurally exotic bis-macrocyclic heptapeptide that exhibits planar chirality and shows selective antibiotic activity against Gram-negative pathogens. Shared between the Patel/Petrone/Sarlah and Baran syntheses was a similar synthetic strategy that exploited two-fold Larock macrocyclization for the atropselective construction of the two tryptophan-containing strained macrocycles.
In the Patel/Petrone/Sarlah synthesis of darobactin A (209),86 exquisitely designed tripeptide 210, harboring a silyl-substituted alkyne, an ortho-bromo acetanilide, and an ortho-iodo acetanilide, was used as a substrate for the first Larock macrocyclization (Scheme 30A). Upon exposure of 210 to Pd(t-Bu3P)2 and Cy2NMe in CH3CN/toluene (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) at 40 °C, chemoselective Larock macrocyclization occurred between the alkyne and the ortho-iodo acetanilide moieties, giving rise to macrocycle 211 in 52% yield with 3
1) at 40 °C, chemoselective Larock macrocyclization occurred between the alkyne and the ortho-iodo acetanilide moieties, giving rise to macrocycle 211 in 52% yield with 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 atropselectivity without compromising the aryl bromide functionality. After completing the peptide backbone structure, heptapeptide 212 was subjected to the second Larock macrocyclization under the catalysis of Pd(t-Bu3P)2 (Cy2NMe, CH3CN, 80 °C) to furnish bis-macrocycle 213 in 51% yield. Finally, sequential removal of the protecting groups from 213 yielded darobactin A (209). The Patel/Petrone/Sarlah group described that the order of two Larock macrocyclizations was crucial for achieving the desired atropselectivity at the two indole rings and that an attempted double Larock macrocyclization resulted in a low product yield with undesired atropselectivity at the left-hand indole ring.
1 atropselectivity without compromising the aryl bromide functionality. After completing the peptide backbone structure, heptapeptide 212 was subjected to the second Larock macrocyclization under the catalysis of Pd(t-Bu3P)2 (Cy2NMe, CH3CN, 80 °C) to furnish bis-macrocycle 213 in 51% yield. Finally, sequential removal of the protecting groups from 213 yielded darobactin A (209). The Patel/Petrone/Sarlah group described that the order of two Larock macrocyclizations was crucial for achieving the desired atropselectivity at the two indole rings and that an attempted double Larock macrocyclization resulted in a low product yield with undesired atropselectivity at the left-hand indole ring.
In the Baran synthesis of darobactin A (209),87 tripeptide 214 decorated with a terminal alkyne and an ortho-bromo acetanilide underwent the first Larock macrocyclization under the influence of Pd(t-Bu3P)2 and i-Pr2NEt in dioxane at 85 °C to deliver macrocycle 215 in 61% yield as a single atropisomer on a gram scale (Scheme 30B). In this case, the regioselectivity was substrate-directed and did not require a terminal alkyne substituent; rather, the substitution of the terminal alkyne with a triethylsilyl group made this reaction unproductive. Notably, other methods including Suzuki–Miyaura and Heck macrocyclizations and various macrolactamizations were all unsuccessful. After extending the peptide backbone and installing an ortho-bromo acetanilide, the second Larock macrocyclization was executed on pentapeptide 216 (Fe, NH4Cl, AcOH, 50 °C; then Pd(t-Bu3P)2, i-Pr2NEt, dioxane, 85 °C) to afford bis-macrocycle 217 in 67% yield from 216 as a single atropisomer. The Baran synthesis of darobactin A (209) was highlighted by Larock macrocyclizations at the mid-stage of the synthesis. Aiming at medicinal chemistry applications, the end game involved a late-stage coupling of the C-terminus dipeptide and completion of the lysine side chain.
Nakamura and co-workers have recently disclosed their works on the total syntheses of cihunamide B and strecintide 839 based on Larock macrocyclizations.88,89
Chirality-generating tandem macrocyclization/transannular cyclization
Tandem (or domino, cascade) reactions are those that involve multiple transformations consecutively by taking advantage of the functional groups generated in the prior transformation.90 Because tandem reactions obviate the need for isolating intermediates, the implementation of tandem reactions in the total synthesis helps improving the synthetic efficiency. Complementary to chirality-generating macrocyclizations, tandem reactions that involve a macrocyclization and a chirality-generating transannular cyclization(s) are a promising means to synthesize complex natural macrocycles expediently.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5). The cycloadduct 221 was then transformed into the tetracyclic skeleton 222 of the spinosyn A pseudoaglycone via transannular vinylogous Morita–Baylis–Hillman reaction in a stereoselective manner. In contrast, Horner–Wadsworth–Emmons macrocyclization of precursor 223 lacking the C8 substituent under the same reaction conditions used for 219 provided transannular Diels–Alder cycloadduct 225 in 75% yield as a 73
5). The cycloadduct 221 was then transformed into the tetracyclic skeleton 222 of the spinosyn A pseudoaglycone via transannular vinylogous Morita–Baylis–Hillman reaction in a stereoselective manner. In contrast, Horner–Wadsworth–Emmons macrocyclization of precursor 223 lacking the C8 substituent under the same reaction conditions used for 219 provided transannular Diels–Alder cycloadduct 225 in 75% yield as a 73![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12
12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9
9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6 mixture of four diastereomers. Thus, it was clear that the C8 silyloxy group played a significant role in destabilizing competing transition states of the transannular Diels–Alder reaction of 220. The omission of the C6 steric directing group, i.e.226, resulted in a similar diastereoselectivity (dr 70
6 mixture of four diastereomers. Thus, it was clear that the C8 silyloxy group played a significant role in destabilizing competing transition states of the transannular Diels–Alder reaction of 220. The omission of the C6 steric directing group, i.e.226, resulted in a similar diastereoselectivity (dr 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18
18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12), indicating that the C6 bromine did not have a significant impact on the stereochemical course of the transannular Diels–Alder reaction of 219 and 223. As shown by the reaction of 229, further deletion of the C21 ethyl group did not significantly alter the stereochemical outcome, giving cycloadduct 231 as a mixture of four diastereomers in a ratio of 71
12), indicating that the C6 bromine did not have a significant impact on the stereochemical course of the transannular Diels–Alder reaction of 219 and 223. As shown by the reaction of 229, further deletion of the C21 ethyl group did not significantly alter the stereochemical outcome, giving cycloadduct 231 as a mixture of four diastereomers in a ratio of 71![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18
18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6
6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5. Nonetheless, transannular Diels–Alder reaction of 223, 226, and 229 was clearly better than the intramolecular Diels–Alder reaction of 232 (dr 53
5. Nonetheless, transannular Diels–Alder reaction of 223, 226, and 229 was clearly better than the intramolecular Diels–Alder reaction of 232 (dr 53![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 37
37![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5
5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5) with respect to the diastereoselectivity. Thus, the macrocyclic skeleton of 224, 227, and 230 must play a beneficial role in directing the stereochemical course of the transannular Diels–Alder reaction in a desired sense.
5) with respect to the diastereoselectivity. Thus, the macrocyclic skeleton of 224, 227, and 230 must play a beneficial role in directing the stereochemical course of the transannular Diels–Alder reaction in a desired sense.
 | ||
| Scheme 31 Tandem macrocyclization/transannular Diels–Alder cycloadditions in the total synthesis of (−)-spinosyn A pseudoaglycone (218) by Roush and co-workers. | ||
The examples shown above clearly indicate that the stereocontrol of transition states by designing appropriate precursors is vital for the successful stereochemical control of transannular Diels–Alder cycloadditions.
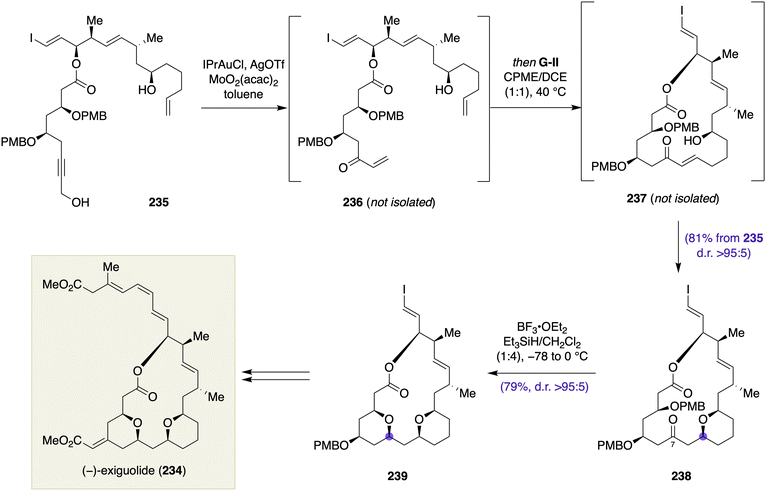
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) at 40 °C gave rise to 2,6-cis-configured tetrahydropyran 238 in 81% yield from 235 with >95
1) at 40 °C gave rise to 2,6-cis-configured tetrahydropyran 238 in 81% yield from 235 with >95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 diastereoselection. The stereochemical outcome of the transannular oxa-Michael addition (237 → 238) was reasoned by a chair-like transition-state model, which was in accordance with the previous work by Bates.100 The C7 ketone of 238 could be directly used in the subsequent transannular Kishi reduction to afford bis-tetrahydropyran 239 in 79% yield with >95
5 diastereoselection. The stereochemical outcome of the transannular oxa-Michael addition (237 → 238) was reasoned by a chair-like transition-state model, which was in accordance with the previous work by Bates.100 The C7 ketone of 238 could be directly used in the subsequent transannular Kishi reduction to afford bis-tetrahydropyran 239 in 79% yield with >95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 diastereoselection. The Kishi reduction in a transannular format (238 → 239) proceeded under stereoelectronic control, wherein an axial hydride delivery to the intermediate macrocyclic oxocarbenium ion occurred to deliver the desired 2,6-cis-configured tetrahydropyran. Noteworthy is that the 20-membered macrocyclic backbone and two embedded tetrahydropyran rings were constructed in only two steps from the macrocyclization precursor 235 with perfect stereocontrol. Thus, the total synthesis of (−)-exiguolide (234) was completed in only 13 steps from a commercially available material, representing the shortest synthesis of this complex macrolide natural product.101 Empowered by our tandem macrocyclization/transannular oxa-Michael addition strategy, we could make deep-seated stereochemical modifications on the macrocyclic core of (−)-exiguolide (234) for structure–activity relationship investigations.95d
5 diastereoselection. The Kishi reduction in a transannular format (238 → 239) proceeded under stereoelectronic control, wherein an axial hydride delivery to the intermediate macrocyclic oxocarbenium ion occurred to deliver the desired 2,6-cis-configured tetrahydropyran. Noteworthy is that the 20-membered macrocyclic backbone and two embedded tetrahydropyran rings were constructed in only two steps from the macrocyclization precursor 235 with perfect stereocontrol. Thus, the total synthesis of (−)-exiguolide (234) was completed in only 13 steps from a commercially available material, representing the shortest synthesis of this complex macrolide natural product.101 Empowered by our tandem macrocyclization/transannular oxa-Michael addition strategy, we could make deep-seated stereochemical modifications on the macrocyclic core of (−)-exiguolide (234) for structure–activity relationship investigations.95d
Late-stage chirality-generating skeletal transformations: transannular reactions
Transannular reactions offer late-stage opportunities for creating a new bond(s) across macrocycles with concurrent introduction of a stereogenic center(s).9 In general, the consequence of transannular reactions largely depends on the conformational constraints of the macrocycle of interest. Thus, it is important to design macrocyclic precursors with an appropriate substitution/unsaturation pattern for achieving the desired transition state in an energetically favored way.Transannular conjugate additions
While 10-membered rings are classified as medium-sized rings in a strict sense, there are several examples that exploit transannular Michael additions for the closure of 10-membered rings found in natural products. Shimizu and Nakagawa described the synthesis of jasmine ketolactone (240) in a racemic form through a transannular Michael addition (Scheme 33A).102 The precursor macrolactone 241 was synthesized via a macrolactonization of the corresponding seco-ester under Otera conditions. Upon exposure of 241 to KH (2 equiv.) in toluene at 90 °C, transannular Michael addition occurred to give trans-fused jasmine ketolactone (240) in 69% yield as a single diastereomer. Interestingly, an intramolecular Michael addition using a non-tethered counterpart was not successful, indicating the importance of the macrocyclic constraint in the present case.Snider and Zhou showed in their total synthesis of (−)-Sch 642305 (242) that transannular Michael addition of macrolactone 243, accessed through a Yamaguchi macrolactonization, proceeded by the action of NaH (THF, 0 °C) to deliver cis-fused transannular product 244 in 80% yield as a single stereoisomer (Scheme 33B).103 Thermodynamic epimerization under acidic conditions gave trans-fused isomer 245 in 23% yield (58% based on recovered starting material), which was desilylated with TBAF/AcOH to furnish (−)-Sch 642305 (242). The late-stage transannular Michael reaction enabled a quick access to the bicyclic skeleton of (−)-Sch 642305, although the configuration of the transannular Michael product 244 did not correspond to that of the natural product.
In their synthetic studies on dictyosphaeric acids, Taylor et al. examined transannular Michael addition of macrocycle 246 to construct the tricyclic core structure 247 (Scheme 33C).104 The precursor 246 was synthesized by means of a macrocyclic ring-closing metathesis. Treatment of 246 with NaH (1.2 equiv., THF, 0 °C to rt) resulted in a transannular Michael addition as anticipated. After hydrogenation of the double bond, an approximately 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of cis-247 and trans-247 was obtained in 78% yield for the two steps.
1 mixture of cis-247 and trans-247 was obtained in 78% yield for the two steps.
Transannular Michael additions have also found their use in the total synthesis of stereochemically complex polycyclic norcembranoids from macrocyclic precursors, which will be discussed later.
Transannular hetero-Michael additions are a powerful means to construct heterocyclic rings embedded within macrocyclic natural products. Fürstner and co-workers have been actively investigating various transannular skeletal transformations on macrocyclic alkynes in the context of total synthesis of macrocyclic natural products, by taking advantage of their ring-closing alkyne metathesis chemistry.105 A transannular aza-Michael addition106 was successfully implemented in the total synthesis of (−)-lythranidine (249), a macrocyclic piperidine alkaloid (Scheme 34A).107 Upon exposure of α,β-unsaturated ketone 250 to TsOH in DCE at 45 °C, transannular aza-Michael addition occurred to deliver 2,6-trans-configured piperidine 251 in 94% yield based on the recovered starting material with approximately 2.4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 diastereoselectivity. Attempts at improving the diastereomer ratio using chiral Brønsted acids were unsuccessful, suggesting that the stereochemical course of the transannular aza-Michael addition was strongly dependent on the macrocyclic conformation. The transannular product 251 was elaborated into (−)-lythranidine (249) through a stereoselective carbonyl reduction.
1 diastereoselectivity. Attempts at improving the diastereomer ratio using chiral Brønsted acids were unsuccessful, suggesting that the stereochemical course of the transannular aza-Michael addition was strongly dependent on the macrocyclic conformation. The transannular product 251 was elaborated into (−)-lythranidine (249) through a stereoselective carbonyl reduction.
More recently, an expedient synthesis of (−)-lythranidine (249) was disclosed by Sherburn and co-workers (Scheme 34B).108 Treatment of bis-α,β-unsaturated ketone 252 with benzylamine in MeOH/CH2Cl2 at ambient temperature triggered intermolecular/transannular aza-Michael additions to give piperidine 253 with trans/cis 85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 selectivity. In situ chelate controlled reduction with NaBH4 in the presence of MnCl2·4H2O provided diol 254 in 64% yield. The stereochemical course of the transannular cyclization was under thermodynamic control, as was evidenced by a thermodynamic equilibration experiment using a separately synthesized 2,6-cis-configured isomer. Importantly, the stereochemical outcome of the transannular aza-Michael addition could be reversed. Upon treatment of bis-α,β-unsaturated ketone 252 with CbzNH2 and HBF4 in CH3CN/H2O at ambient temperature, tandem intermolecular/transannular aza-Michael additions proceeded under Brønsted acid catalysis to give 2,6-cis-configured piperidine 255 with approximately 80
15 selectivity. In situ chelate controlled reduction with NaBH4 in the presence of MnCl2·4H2O provided diol 254 in 64% yield. The stereochemical course of the transannular cyclization was under thermodynamic control, as was evidenced by a thermodynamic equilibration experiment using a separately synthesized 2,6-cis-configured isomer. Importantly, the stereochemical outcome of the transannular aza-Michael addition could be reversed. Upon treatment of bis-α,β-unsaturated ketone 252 with CbzNH2 and HBF4 in CH3CN/H2O at ambient temperature, tandem intermolecular/transannular aza-Michael additions proceeded under Brønsted acid catalysis to give 2,6-cis-configured piperidine 255 with approximately 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 diastereoselectivity, and 255 was isolated in a diastereomerically pure form in 74% yield after flash column chromatography using silica gel. The diastereoselectivity of the transannular aza-Michael addition was thermodynamically controlled. This was confirmed by re-subjection of the minor trans-configured isomer to the reaction conditions.
20 diastereoselectivity, and 255 was isolated in a diastereomerically pure form in 74% yield after flash column chromatography using silica gel. The diastereoselectivity of the transannular aza-Michael addition was thermodynamically controlled. This was confirmed by re-subjection of the minor trans-configured isomer to the reaction conditions.
A stereodivergent transannular oxa-Michael addition was demonstrated by Shishido and co-workers in their total synthesis of aspergillides A and B (256 and 257, respectively) (Scheme 35).109 Transannular oxa-Michael addition of α,β-unsaturated ester 258 using DBU/LiCl in CH3CN at room temperature provided 2,6-cis-configured tetrahydropyran 259 in quantitative yield with complete diastereoselection. This stereochemical consequence could be reasoned by a transition state in which a lithium cation is coordinated to the ester carbonyl oxygen. In contrast, the reaction of 258 with KH in the presence of 18-crown-6 in THF at 0 °C afforded 2,6-trans-configured tetrahydropyran 260 in 96% yield as a single diastereomer. Control experiments demonstrated that 2,6-cis-configured tetrahydropyran 259 could be converted into 2,6-trans-configured tetrahydropyran 260 by treatment with KH/18-crown-6 (94%), whereas 2,6-trans-configured tetrahydropyran 260 remained unreactive upon treatment with DBU/LiCl in refluxing CH3CN (90% recovery).
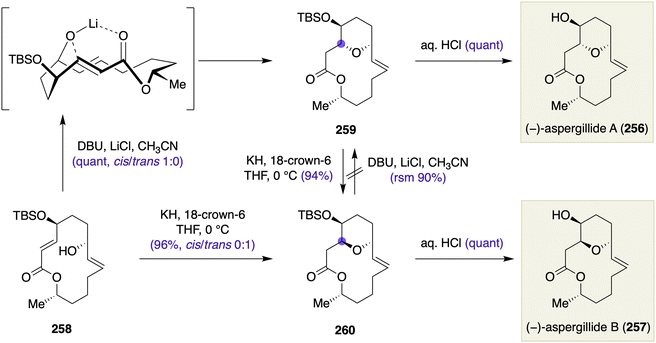 | ||
| Scheme 35 Transannular oxa-Michael additions in the stereodivergent total synthesis of (−)-aspergillide A (256) and (−)-aspergillide B (257) by Shishido and co-workers. | ||
Recently, our group has achieved a concise total synthesis of (−)-enigmazole A (261) based on our macrocyclization/transannular pyran cyclization strategy (Scheme 36).110 Macrocyclization precursor 262 was prepared through Yamaguchi esterification of carboxylic acid 263 and alcohol 264 followed by Nolan's α,β-unsaturated ketone synthesis111 of the derived ester. The 18-membered macrocyclic backbone of 261 was closed by ring-closing metathesis of 262 under the catalysis of G-II complex. Treatment of the resultant macrocyclic α,β-unsaturated ketone 265 with aq. HF in CH3CN/THF (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) at −20 to −3 °C triggered desilylative transannular oxa-Michael addition to afford 2,6-cis-configured tetrahydropyran 266 in 75% yield with >95
1, v/v) at −20 to −3 °C triggered desilylative transannular oxa-Michael addition to afford 2,6-cis-configured tetrahydropyran 266 in 75% yield with >95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 diastereoselection, in accordance with the Bates late-transition-state model. The stereochemical outcome of the transannular oxa-Michael addition could be reversed by using TBAF as a desilylation reagent.
5 diastereoselection, in accordance with the Bates late-transition-state model. The stereochemical outcome of the transannular oxa-Michael addition could be reversed by using TBAF as a desilylation reagent.
 | ||
| Scheme 36 Desilylative transannular oxa-Michael addition in the total synthesis of (−)-enigmazole A (261) by Fuwa and co-workers. | ||
Transannular cycloetherifications
In contrast to transannular conjugate additions involving macrocyclic α,β-unsaturated carbonyls, transannular cycloetherification proceeds via an electrophilic activation of non-activated macrocyclic olefins.The Maier synthesis of (−)-apicularen A (267) exploited a transannular oxymercuration of macrocyclic olefin 268 under the influence of Hg(OCOCF3)2 in CH2Cl2 at room temperature, giving 2,6-trans-configured tetrahydropyran 270 in 89% yield (from 268) after reduction with LiBH4/Et3B (Scheme 37A).112 Interestingly, treatment of 268 with N-(phenylseleno)phthalimide resulted in no reaction. Turning to more electrophilic PhSeOTf, transannular oxyselenylation of 268 did proceed but was accompanied by side reactions, giving the corresponding transannular product in only 34% yield (structure not shown). Meanwhile, the Tae synthesis of (−)-apicularen A (267) was based on a transannular oxyselenylation using PhSeCl as an electrophile (Scheme 37B).113 Thus, treatment of macrocyclic olefin 271 with PhSeCl, after radical reduction using Bu3SnH/AIBN, provided 2,6-trans-configured tetrahydropyran 273 in 87% overall yield from 271. Importantly, the C–O bond formed by the transannular cycloetherification in the Tae synthesis is different from that in the Maier synthesis. Tae et al. noted that they failed to promote transannular oxymercuration of macrocyclic olefin 271 using Hg(OAc)2 or Hg(OCOCF3)2.
Sharma and co-workers showed in their formal synthesis of (+)-neopeltolide (78), transannular cycloetherifications for the stereoselective closure of the embedded tetrahydropyran ring (Scheme 38).114 When macrocyclic olefin 274 was exposed to I2 in CH3CN at −40 to 0 °C, transannular iodoetherification occurred to give 2,6-trans-configured tetrahydropyran 275 in 60% yield as an 85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 mixture of diastereomers. In contrast, treatment of 274 with Hg(OCOCF3)2 in CH2Cl2 at 0 °C and then with aq. KBr at room temperature brought about transannular oxymercuration to deliver 2,6-cis-configured tetrahydropyran 276 in 84% yield as a single diastereomer. However, the exact reason for the observed stereodivergency in transannular cycloetherifications of 274 remains elusive.
15 mixture of diastereomers. In contrast, treatment of 274 with Hg(OCOCF3)2 in CH2Cl2 at 0 °C and then with aq. KBr at room temperature brought about transannular oxymercuration to deliver 2,6-cis-configured tetrahydropyran 276 in 84% yield as a single diastereomer. However, the exact reason for the observed stereodivergency in transannular cycloetherifications of 274 remains elusive.
 | ||
| Scheme 38 Transannular iodoetherification and oxymercuration in the formal synthesis of (+)-neopeltolide (78) by Sharma and co-workers. | ||
Transannular cycloadditions
In their total synthesis of (+)-superstolide A (277), the Roush group envisioned that the cis-fused octahydronaphthalene moiety embedded within 277 could be constructed through a late-stage, bioinspired transannular Diels–Alder cycloaddition (Scheme 39).115 The precursor octaene 278 was synthesized through Suzuki–Miyaura macrocyclization of ester 279. The crucial transannular Diels–Alder cycloaddition of 278 proceeded in CDCl3 at 23 °C for 5 days or in toluene at 80 °C for 2 h to afford cycloadduct 280 with the correct configuration in 30–35% yield over the two steps from 279 as the only isolable product. Notably, initial efforts that involved construction of the cis-fused octahydronaphthalene moiety at an early stage of the synthesis by means of an intramolecular Diels–Alder cycloaddition of 281 resulted in octahydronaphthalene 282 with only moderate diastereoselectivity (dr 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Furthermore, it eventually turned out that it was difficult to close the macrocycle at a later stage of the synthesis. Thus, the order of fragment assembly → macrocyclization → transannular cycloaddition was crucial for the success of the total synthesis of (+)-superstolide A (277).
1). Furthermore, it eventually turned out that it was difficult to close the macrocycle at a later stage of the synthesis. Thus, the order of fragment assembly → macrocyclization → transannular cycloaddition was crucial for the success of the total synthesis of (+)-superstolide A (277).
 | ||
| Scheme 39 Transannular Diels–Alder cycloaddition in the total synthesis of (+)-superstolide A (277) by Roush and co-workers. | ||
Transannular Diels–Alder cycloaddition has also been exploited in tandem with macrocyclizations, as discussed above.
Kanoh, Kakeya, and co-workers described the total synthesis of heronamides A–C (283–285), in which they transformed heronamide C (285) into heronamides A and B (283 and 284, respectively) through a transannular thermal [6 + 4] cycloaddition and a transannular photochemical [6 + 6] cycloaddition at the final stage of the synthesis (Scheme 40).116 These transannular transformations were inspired by a postulated biosynthesis mechanism. Placing 285 under an atmosphere of O2 in DMF/DMSO (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) at 50 °C for seven days provided heronamide A (283) in 27% yield (45% yield BORSM). Based on the proposed biosynthetic pathway, it was assumed that oxidation of the Δ16,17 double bond of 285 would trigger the transannular epoxide-opening reaction of 286 with the amide nitrogen to deliver pyrrolidine 287, which would then undergo transannular [6 + 4] cycloaddition to give heronamide A (283). Meanwhile, UV irradiation (365 nm) of heronamide C (285) in the presence of BHT in DMF at room temperature caused transannular [6 + 6] cycloaddition to afford heronamide B (284) in 27% yield.
1, v/v) at 50 °C for seven days provided heronamide A (283) in 27% yield (45% yield BORSM). Based on the proposed biosynthetic pathway, it was assumed that oxidation of the Δ16,17 double bond of 285 would trigger the transannular epoxide-opening reaction of 286 with the amide nitrogen to deliver pyrrolidine 287, which would then undergo transannular [6 + 4] cycloaddition to give heronamide A (283). Meanwhile, UV irradiation (365 nm) of heronamide C (285) in the presence of BHT in DMF at room temperature caused transannular [6 + 6] cycloaddition to afford heronamide B (284) in 27% yield.
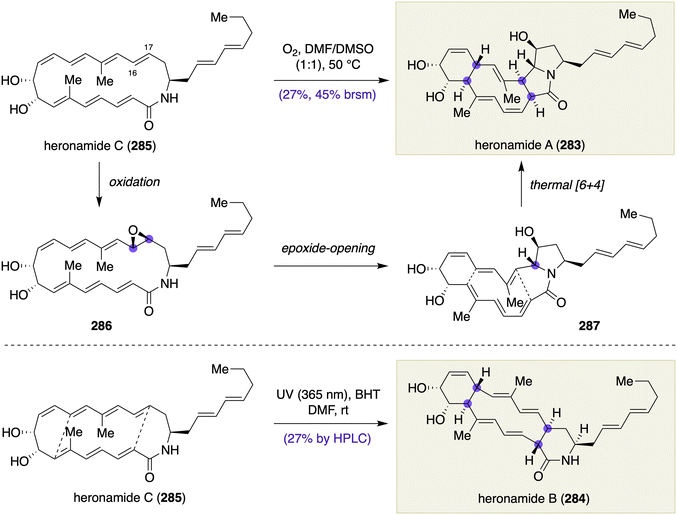 | ||
| Scheme 40 Transannular cycloadditions in the total syntheses of heronamides A and B (283 and 284) by Kanoh, Kakeya, and co-workers. | ||
Trauner, Houk, and co-workers reported the synthesis of a highly unsaturated macrolactone, collinolide (288), and its transannular [6 + 4] cycloaddition.117 Collinolide (288) has been proposed as a hypothetical biosynthetic precursor of the tricyclic cyclodecatriene “collinoketone” (re-named as collinoketone A by Trauner et al., 289) and a neuroprotective natural product, collinolactone (290) (Scheme 41A). According to the proposal by Grond et al.,118 a transannular [6 + 4] cycloaddition of collinolide (288) furnishes “collinoketone” (289), which would then be processed to collinolactone (290) through an enzymatic Baeyer–Villiger oxidation. The Trauner group synthesized the precursor macrolactone 291 through a Horner–Wadsworth–Emmons macrocyclization (Scheme 41B).
Upon heating in toluene at 70 °C, macrolactone 291 cleanly underwent a transannular [6 + 4] cycloaddition to afford a diastereomeric mixture of cyclodecatrienes 292 and 293 in a ratio of 1.3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. The removal of the PMB group from these transannular cycloaddition products 292 and 293 afforded the final products, named collinoketone B (294) and collinoketone C (295), respectively. Notably, the 1H and 13C NMR spectroscopic data of collinoketone B (294) fully matched those reported for natural “collinoketone”. Thus, the structure of “collinoketone” isolated from natural sources was revised to be that represented by the stereoisomer, collinoketone B (294). In contrast, the ratio was reversed to 1
1. The removal of the PMB group from these transannular cycloaddition products 292 and 293 afforded the final products, named collinoketone B (294) and collinoketone C (295), respectively. Notably, the 1H and 13C NMR spectroscopic data of collinoketone B (294) fully matched those reported for natural “collinoketone”. Thus, the structure of “collinoketone” isolated from natural sources was revised to be that represented by the stereoisomer, collinoketone B (294). In contrast, the ratio was reversed to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.3 when the transannular cycloaddition reaction was run using collinolide (288) in 1,1,1,3,3,3-hexafluoropropan-2-ol (HFIP) at 50 °C. This suggested that the (Z,Z,E) isomers are thermodynamic products whereas (Z,E,E) isomers are kinetically favored. Furthermore, DFT calculations suggested that the transannular cycloaddition proceeds through an ambimodal transition state, from which both [6 + 4] and [4 + 2] cycloadducts can arise. Finally, DFT calculations showed that collinoketone A (289) should readily undergo sequential Cope rearrangements to give energetically more stable collinoketone B (294) (Scheme 41C). Thus, it was proposed that the biosynthetic intermediate collinoketone A (289) would still be the most plausible precursor of collinolactone (290) and that an enzyme would be responsible for the preferential formation of collinoketone A (289) in the transannular [6 + 4] cycloaddition of collinolide (288).
2.3 when the transannular cycloaddition reaction was run using collinolide (288) in 1,1,1,3,3,3-hexafluoropropan-2-ol (HFIP) at 50 °C. This suggested that the (Z,Z,E) isomers are thermodynamic products whereas (Z,E,E) isomers are kinetically favored. Furthermore, DFT calculations suggested that the transannular cycloaddition proceeds through an ambimodal transition state, from which both [6 + 4] and [4 + 2] cycloadducts can arise. Finally, DFT calculations showed that collinoketone A (289) should readily undergo sequential Cope rearrangements to give energetically more stable collinoketone B (294) (Scheme 41C). Thus, it was proposed that the biosynthetic intermediate collinoketone A (289) would still be the most plausible precursor of collinolactone (290) and that an enzyme would be responsible for the preferential formation of collinoketone A (289) in the transannular [6 + 4] cycloaddition of collinolide (288).
Transannular electrocyclizations
Pericyclic reactions play a central role in skeletal rearrangements and ring formation and have been extensively utilized in biomimetic synthesis. As shown above, transannular cycloadditions offer an exceptionally efficient approach to the assembly of complex polycyclic molecules. In contrast, successful examples of transannular electrocyclizations are extremely rare due to their innate reversibility.119West et al. reported a bioinspired synthesis of briareolate ester B (297), featuring a transannular oxa-6π electrocyclization that was inspired by a biosynthetic hypothesis for briareolate esters (Scheme 42).120 The irradiation of a methanolic solution of briareolate ester L (298) with UVC light (λ < 260 nm) afforded only a mixture of 298 and 299 with no formation of 297 (298![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 299
299![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 297 = 37
297 = 37![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0). The formation of 297 was not observed under these conditions due to a rapid retro-oxa-6π electrocyclization from 297 to 299. In contrast, the irradiation of a methanolic solution of 298 with low-energy UVA light (λ > 350 nm) for 3 h afforded briareolate ester B (297) as the major product (298
0). The formation of 297 was not observed under these conditions due to a rapid retro-oxa-6π electrocyclization from 297 to 299. In contrast, the irradiation of a methanolic solution of 298 with low-energy UVA light (λ > 350 nm) for 3 h afforded briareolate ester B (297) as the major product (298![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 299
299![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2997 = 23
2997 = 23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12
12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 65). To follow up on these results, the irradiation of briareolate ester B (297) using UVA light resulted in no reaction over several hours. In contrast, the irradiation of briareolate ester B (297) using UVC light triggered a rapid and complete retro-6π electrocyclization to give a mixture of 298 and 299 (298
65). To follow up on these results, the irradiation of briareolate ester B (297) using UVA light resulted in no reaction over several hours. In contrast, the irradiation of briareolate ester B (297) using UVC light triggered a rapid and complete retro-6π electrocyclization to give a mixture of 298 and 299 (298![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 299
299![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 297 = 37
297 = 37![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0). Thus, the retro-6π electrocyclization was rapidly induced under UVC irradiation but not under UVA irradiation. These results established a unique photochemical switch in which selective irradiation with UVA or UVC light enables controlled interconversion between distinct skeletal frameworks. This study provides an insight into the transannular oxa-6π-electrocyclic reactions of macrocyclic conjugated dienone systems via a two-photon process and likely finds numerous applications in the synthesis of medium-sized macrocycles.
0). Thus, the retro-6π electrocyclization was rapidly induced under UVC irradiation but not under UVA irradiation. These results established a unique photochemical switch in which selective irradiation with UVA or UVC light enables controlled interconversion between distinct skeletal frameworks. This study provides an insight into the transannular oxa-6π-electrocyclic reactions of macrocyclic conjugated dienone systems via a two-photon process and likely finds numerous applications in the synthesis of medium-sized macrocycles.
 | ||
| Scheme 42 Transannular oxa-6π electrocyclization in the total synthesis of briareolate ester B (297) by West and co-workers. | ||
Transition metal-catalyzed transannular reactions
Despite their widespread general use in natural product synthesis, palladium-catalyzed cross-coupling reactions have been underexploited in a transannular format. A rare example of chirality-generating transannular Heck reaction was reported by Gu and Zakarian in their total synthesis of (−)-rhazinilam (302) (Scheme 43).121 Lactam 303 was synthesized through a macrolactamization of acyclic amino acid 305 using 2-chloro-1-methylpyridinium iodide. The sign of axial chirality in 303 came from its 1H NMR spectrum, wherein non-equivalent signals were observed for the methylene protons at the C3 position. Although enantioselective macrolactamization of amino acid 305 using several chiral reagents was unsuccessful, chiral HPLC resolution of 303 provided (aR)-303 and (aS)-303 with greater than 99% ee each. Transannular Heck reaction of (aR)-303 under the catalysis of Pd(PPh3)4 (CH3CN/Et3N, 100 °C) afforded rhazinilam skeleton 307 through alkylpalladium species 306 in 60% yield with >99% ee, representing a highly efficient axial-to-point chirality transfer.122 | ||
| Scheme 43 Transannular Heck reaction in the total synthesis of (−)-rhazinilam (302) by Gu and Zakarian. | ||
Complementary to traditional intramolecular keto-diol cyclization, intramolecular alkyne hydroalkoxylation catalyzed by transition metal complexes is an efficient means to access various spiroketal derivatives, although the regioselectivity issue may be problematic in cases where endo/exo cyclization modes are both plausible.123 In their total synthesis of spirastrellolide F methyl ester (308), Fürstner et al. closed the macrocyclic skeleton of the target by means of ring-closing alkyne metathesis (Scheme 44).124 Upon exposure of macrocyclic alkyne 309 to cationic Au complex 310 (4 Å MS, CH2Cl2),125 nucleophilic attack of the sterically less encumbered C13 hydroxy group onto the alkyne group occurred regioselectively, giving endocyclic enol ether 311 in 62% yield with 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 endo/exo. In contrast, AuCl or AuCl·SMe2 catalyzed the alkyne hydroalkoxylation of 309 in a highly exo selective fashion. Treatment of 311 with PPTS in toluene at 80 °C resulted in spiroketalization with a sterically more hindered C21 hydroxy group, affording 6,6-spiroketal 312 in 81% yield with the correct configuration at the C17 position.
1 endo/exo. In contrast, AuCl or AuCl·SMe2 catalyzed the alkyne hydroalkoxylation of 309 in a highly exo selective fashion. Treatment of 311 with PPTS in toluene at 80 °C resulted in spiroketalization with a sterically more hindered C21 hydroxy group, affording 6,6-spiroketal 312 in 81% yield with the correct configuration at the C17 position.
Hermann and Fürstner have recently disclosed a collective synthesis of cembrane diterpenoids, in which a transition metal-catalyzed transannular alkyne hydroalkoxylation was used for the stereoselective construction of the 5,5-spiroketal moiety of isochandonanthone, i.e., 314 → 313 (Scheme 45).126
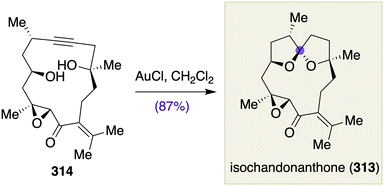 | ||
| Scheme 45 Au-catalyzed transannular alkyne hydroalkoxylation in the total synthesis of isochandonanthone (313) by Hermann and Fürstner. | ||
Transannular skeletal reorganizations
The power of late-stage chirality-generating transformations including transannular skeletal reorganizations was amply demonstrated in the total synthesis of polycyclic norfuranocembranoids, (+)-ineleganolide (315) and (−)-sinulochmodin C (316), by Wood and co-workers (Scheme 46)127 and in the total synthesis of cyclic imine toxins, (+)-portimine A (317) and (+)-portimine B (318), by Baran and co-workers (Scheme 47).128 Although these natural products do not have a macrocycle (a 12-membered or larger ring) in a strict sense, these outstanding synthetic works involve multiple chirality-generating transformations on complex macrocyclic intermediates, which are worth mentioning in this article.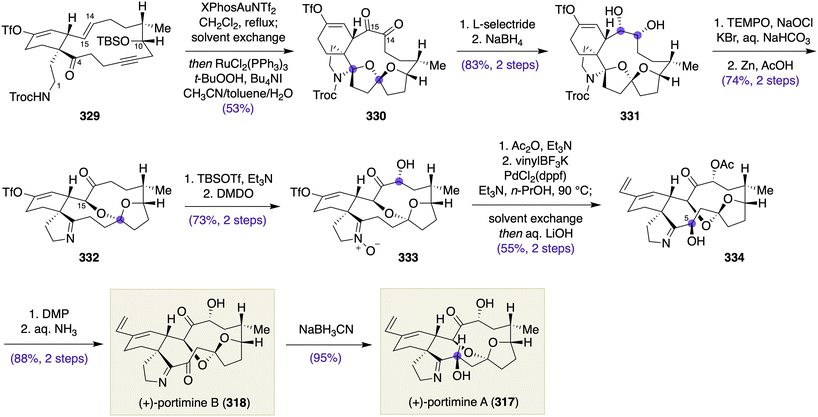 | ||
| Scheme 47 Transannular skeletal reorganizations in the total synthesis of (+)-portimine A (317) and (+)-portimine B (318) by Baran and co-workers. | ||
The polycyclic furanocembranoid family of natural products have attracted significant attention from the chemical community because of their densely functionalized, complicated structures and potentially useful anticancer activities. An earlier study by Li and Pattenden demonstrated a biomimetic transformation of the acetate derivative 320 of naturally occurring 5-epi-sinuleptolide into ineleganolide (315) and sinulochmodin C (316) through a transannular Michael addition (Scheme 46A).129 A relevant transannular Michael addition was implemented in the total synthesis of a norcembranoid, (−)-sinulariadiolide A, by Meng and Fürstner.130
Building on the Pattenden's contribution, Wood and co-workers envisioned a chirality-generating NHK macrocyclization for constructing a macrocycle intermediate at the mid-stage and transannular oxa-Michael and Michael additions for completing the polycyclic skeleton at the final stage (Scheme 46B). Thus, treatment of allylic bromide (13S)-321 with CrCl2 in the presence of 4 Å MS in THF at 23 °C resulted in macrocyclic homoallylic alcohol 322 in 82% yield as a single diastereomer (dr > 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). In contrast, allylic bromide (13R)-321 underwent the same reaction to give macrocyclic homoallylic alcohol 323 with wrong configuration at the C1 position in 80% yield as a single diastereomer (dr > 20
1). In contrast, allylic bromide (13R)-321 underwent the same reaction to give macrocyclic homoallylic alcohol 323 with wrong configuration at the C1 position in 80% yield as a single diastereomer (dr > 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Accordingly, NHK macrocyclization was carried out on (13S)-321 to generate the stereogenic center at C1 with the correct configuration. The configuration at C13 was subsequently reversed to achieve stereoselective elimination of the C13 hydroxy group at the late stage. The removal of the superfluous hydroxy group of the NHK macrocyclization product 322 with BF3·OEt2/Et3SiH, followed by in situ desilylation, gave alcohol 324. Dess–Martin oxidation of 324 and stereoselective reduction of the derived ketone with NaBH4/CaCl2 provided alcohol 325 with the correct configuration at C13. After acetylation of 325, the resultant acetate was treated with TBAF to bring about desilylation and concomitant E1cB elimination of the C13 acetate, giving rise to (Z)-α,β-unsaturated lactone 326. Chemoselective oxidation of the furan ring with 1O2 in the presence of Rose Bengal as the photosensitizer, followed by reductive work-up with Me2S, delivered conjugated ene dione 327, which without isolation was treated with TsOH to catalyze transannular oxa-Michael addition, affording tetrahydrofuran 328 in 88% yield from 326. Finally, exposure of 328 to excess LHMDS brought about deprotonation at C4 or C5 and transannular Michael addition of the derived lithium enolates, resulting in a ca. 3
1). Accordingly, NHK macrocyclization was carried out on (13S)-321 to generate the stereogenic center at C1 with the correct configuration. The configuration at C13 was subsequently reversed to achieve stereoselective elimination of the C13 hydroxy group at the late stage. The removal of the superfluous hydroxy group of the NHK macrocyclization product 322 with BF3·OEt2/Et3SiH, followed by in situ desilylation, gave alcohol 324. Dess–Martin oxidation of 324 and stereoselective reduction of the derived ketone with NaBH4/CaCl2 provided alcohol 325 with the correct configuration at C13. After acetylation of 325, the resultant acetate was treated with TBAF to bring about desilylation and concomitant E1cB elimination of the C13 acetate, giving rise to (Z)-α,β-unsaturated lactone 326. Chemoselective oxidation of the furan ring with 1O2 in the presence of Rose Bengal as the photosensitizer, followed by reductive work-up with Me2S, delivered conjugated ene dione 327, which without isolation was treated with TsOH to catalyze transannular oxa-Michael addition, affording tetrahydrofuran 328 in 88% yield from 326. Finally, exposure of 328 to excess LHMDS brought about deprotonation at C4 or C5 and transannular Michael addition of the derived lithium enolates, resulting in a ca. 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of (−)-sinulochmodin C (316) and (+)-ineleganolide (315) in 46% yield. Notably, the final steps of the present synthesis (328 → 316 + 315) enabled late-stage skeletal reorganizations with the concomitant generation of three stereogenic centers, thereby completing the complex polycyclic architectures of (+)-ineleganolide (315) and (−)-sinulochmodin C (316) in a concise manner.
1 mixture of (−)-sinulochmodin C (316) and (+)-ineleganolide (315) in 46% yield. Notably, the final steps of the present synthesis (328 → 316 + 315) enabled late-stage skeletal reorganizations with the concomitant generation of three stereogenic centers, thereby completing the complex polycyclic architectures of (+)-ineleganolide (315) and (−)-sinulochmodin C (316) in a concise manner.
The Baran group assembled the carbon macrocycle of portimines at the mid-stage of the synthesis through a ring-closing alkyne metathesis (Scheme 47). Prior to the oxidation of the Δ14,15 double bond of 329, the sensitive functional groups were temporarily masked as a unique tricyclic system by means of a transannular skeletal transformation. Thus, the treatment of 329 with a catalytic amount of XPhosAuNTf2 in refluxing CH2Cl2 enabled double spirocyclization by engaging the alkyne, C1 carbamate, C4 carbonyl, and C10 silyloxy groups. Subsequent oxidation of the double bond with RuCl2(PPh3)3 and t-BuOOH in the presence of Bu4NI afforded 1,2-diketone 330 in 53% overall yield. Next, the oxidation state at the C15 position was adjusted through a three-step sequence. Initially, the C14 carbonyl of 330 was reduced with L-selectride in a site- and stereo-selective fashion. The C15 carbonyl was then reduced with NaBH4 in a stereoselective manner. The diastereoselectivities of these peripheral ketone reductions would be ascribed to the steric congestion of the α-face of 330. The C14 hydroxy group of the derived diol 331 was oxidized selectively with TEMPO/NaOCl to give a ketone, from which the Troc group was removed with Zn/aq. AcOH at 40–75 °C. Under these conditions, the tricyclic system collapsed and transannular transketalization occurred with the C15 hydroxy group to deliver ketal 332 in 61% yield for the four steps (from 330). After the transformation of 332 into the corresponding enol silyl ether, oxidation with dimethyldioxirane (DMDO) from the sterically less encumbered α-face gave alcohol 333 with concomitant oxidation of the imine nitrogen (73%, two steps). Upon treatment of 333 with Ac2O/Et3N in CH2Cl2 at 35 °C, the rearrangement of the nitrone occurred to provide the corresponding diacetate. Suzuki–Miyaura cross-coupling with vinylBF3K, followed by the selective liberation of the C5 hydroxy group, furnished alcohol 334 in 55% yield in the two steps. The oxidation of 334 and acetate removal furnished (+)-portimine B (318) in 88% yield for the two steps, which was reduced with NaBH3CN to afford (+)-portimine A (317) in 95% yield. Note that the structure of (+)-portimine B (318) was revised through the present total synthesis. The Baran synthesis of portimines A and B is remarkable in that it followed the “two-phase strategy” originally developed for terpene synthesis and extensively exploited chirality-generating functional group transformations and transannular skeletal reorganizations in the oxidation phase.
Conclusions and prospects
Late-stage chirality generation strategies can be beneficial not only for realizing step-economy in target-oriented synthesis but also for structure diversification aiming at structure–activity relationship investigations. Nonetheless, it is still not easy to implement late-stage chirality-generating transformations in the total synthesis of macrocyclic natural products because of the difficulties in gaining an insight into the conformational landscape of complex macrocycles with moderate conformational flexibility.131 In fact, the key to success in many of the above-described examples was conformational design.132Local substituents often have a profound influence on the overall conformation of macrocycles. Steric interactions such as allylic strain and syn-pentane interaction, dipole interactions, and intramolecular hydrogen bonding are potentially useful structural elements for conformational design. The installation of substituents responsible for these interactions at the pre-macrocyclization stage likely result in confining the conformational flexibility of downstream intermediates at the post-macrocyclization stage, thereby making the stereochemical outcome of late-stage chirality-generating transformations easier to anticipate.
Biosynthetic mechanisms as well as their proposals133 have served as a potential source of inspirations for late-stage transannular reactions, regardless of whether they seem to be catalyzed by an enzyme. However, it is still important to design macrocyclic substrates with an appropriate substitution pattern and stereochemistry for the success of transannular reactions, as can be seen in the synthetic studies on spinosyn A by Roush and co-workers.
The stereoselectivity observed in some examples covered in this review was reasoned by their X-ray structures and/or NMR spectroscopic data. In addition to these analytical techniques, theoretical calculation is a promising approach for visualizing the conformational landscape of complex macrocycles.134 Recent advances in conformational search algorithms and machine learning technologies enable conformational sampling of moderately flexible macrocycles with greater accuracy than ever before.135 Coupled with appropriate statistical analysis, theoretical calculation has been shown to be useful for configurational assignment and conformational analysis of macrocyclic natural products.136–138 If available, NMR spectroscopic data such as J values or NOE correlations may be helpful for making constraints in calculation to reduce the computation costs.
Currently, late-stage chirality-generating transformations in macrocyclic natural product synthesis depend mainly on classical textbook reactions. Radical-mediated transformations, especially stereoinversions at specific tertiary chiral centers,139 are rapidly developing in recent years and should be a potentially useful strategy for the late-stage diversification of macrocyclic natural product structures. Enzymatic late-stage transformations represent a growing area of importance in organic synthesis but their application to the synthesis of macrocyclic natural products is still limited.140,141 Expanding our repertoire of transformations amenable to late-stage chirality generation as well as advancing our controllability and predictability of the conformational property of macrocycles will facilitate future developments in the total synthesis of macrocyclic natural products.
Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results have been included, and no new data were generated or analyzed as part of this review.Acknowledgements
This work was financially supported by KAKENHI, Grant No. 25K01906, from the Japan Society for the Promotion of Science (JSPS).References
- S. Dandapani and L. A. Marcaurrelle, Nat. Chem. Biol., 2010, 6, 861 CrossRef CAS PubMed.
- For selected reviews on the role of macrocycles in drug discovery, see: (a) T. Kim, E. Baek and J. Kim, Pharmaceuticals, 2025, 18, 617 CrossRef CAS PubMed; (b) D. Menche, ChemMedChem, 2021, 16, 2068 CrossRef CAS PubMed; (c) D. G. Jimenez, V. Poongavanam and J. Kihlberg, J. Med. Chem., 2023, 66, 5377 CrossRef PubMed; (d) T. Passioura, Biochemistry, 2020, 59, 139 CrossRef CAS PubMed; (e) F. Giordanetto and J. Kihlberg, J. Med. Chem., 2014, 57, 278 CrossRef CAS PubMed; (f) E. Marsault and M. L. Peterson, J. Med. Chem., 2011, 54, 1961 CrossRef CAS PubMed; (g) E. M. Driggers, S. P. Hale, J. Lee and N. K. Terrett, Nat. Rev. Drug Discovery, 2008, 7, 608 CrossRef CAS PubMed.
- L. A. Viarengo-Baker, L. E. Brown, A. A. Rzepiela and A. Whitty, Chem. Sci., 2021, 12, 4309 RSC.
- For example: (a) M. Nagano, Y. Huang, R. Obexer and H. Suga, J. Am. Chem. Soc., 2021, 143, 4741 CrossRef CAS PubMed; (b) R. Takatsuji, K. Shinbara, T. Katoh, Y. Goto, T. Passioura, R. Yajima, Y. Komatsu and H. Suga, J. Am. Chem. Soc., 2019, 141, 2279 CrossRef CAS PubMed.
- For selected reviews on total synthesis of macrocyclic natural products: (a) I. Ciss, M. Seck, B. Figadère and L. Ferrié, Chem. – Eur. J., 2024, 30, e202400471 CrossRef CAS PubMed; (b) C. P. Bold and K.-H. Altmann, Tetrahedron, 2024, 156, 133908 CrossRef CAS; (c) B. Chatterjee, D. Mondal and S. Bera, Asian J. Org. Chem., 2023, 12, e202200702 CrossRef CAS; (d) H. Fuwa, Org. Chem. Front., 2021, 8, 3990 RSC; (e) N. Y. S. Lam and I. Paterson, Eur. J. Org. Chem., 2020, 2310 CrossRef CAS; (f) M. S. Alehashem, A. B. Ariffin, P. B. Savage, W. A. Y. Dabdawb and N. F. Thomas, RSC Adv., 2020, 10, 10989 RSC; (g) K. Lee, M. L. Lanier, J.-H. Kwak, H. Kim and J. Hong, Nat. Prod. Rep., 2016, 33, 1393 RSC; (h) H. Fuwa, Mar. Drugs, 2016, 14, 65 CrossRef PubMed; (i) A. Lorente, J. Lamariano-Merketegi, F. Albericio and M. Álvarez, Chem. Rev., 2013, 113, 4567 CrossRef CAS PubMed; (j) S. Essig and D. Menche, Pure Appl. Chem., 2013, 85, 1103 CrossRef CAS; (k) K. J. Hale and S. Manaviazar, Chem. – Asian J., 2010, 5, 704 CrossRef CAS PubMed; (l) K.-S. Yeung and I. Paterson, Chem. Rev., 2005, 105, 4237 CrossRef CAS PubMed; (m) E. J. Kang and E. Lee, Chem. Rev., 2005, 105, 4348 CrossRef CAS PubMed; (n) J. Mulzer and H. J. Martin, Chem. Rec., 2004, 3, 258 CrossRef CAS PubMed.
- For selected reviews on aldol reaction, see: (a) Y. Hayashi, Chem. Rec., 2023, 23, e202200159 CrossRef CAS PubMed; (b) P. Bhamboo, S. Bera and D. Mondal, Asian J. Org. Chem., 2021, 10, 2763 CrossRef CAS; (c) Y. Yamashita, T. Yasukawa, W.-J. Yoo, T. Kitasono and S. Kobayashi, Chem. Soc. Rev., 2018, 47, 4388 RSC; (d) S. B. J. Kan, K. K.-H. Ng and I. Paterson, Angew. Chem., Int. Ed., 2013, 52, 9097 CrossRef CAS PubMed; (e) J. Matsuo and M. Murakami, Angew. Chem., Int. Ed., 2013, 52, 9109 CrossRef CAS PubMed.
- For reviews on late-stage functionalization, see: (a) S. Y. Shim, Chem. – Eur. J., 2023, 29, e202302620 CrossRef CAS PubMed; (b) J. Borgel and T. Ritter, Chem, 2020, 6, 1877 CrossRef; (c) B. Hong, T. Luo and X. Lei, ACS Cent. Sci., 2020, 6, 622 CrossRef CAS PubMed; (d) T. Cernak, K. D. Dykstra, S. Tyagarajan, P. Vachal and S. W. Krska, Chem. Soc. Rev., 2016, 45, 546 RSC.
- For selected reviews on macrocyclizations, see: (a) P. Fang, W.-K. Pang, S. Xuan, W.-L. Chan and K. C.-F. Leung, Chem. Soc. Rev., 2024, 53, 11725 RSC; (b) C. Bechtler and C. Lamer, RSC Med. Chem., 2021, 12, 1325 RSC; (c) I. Saridakis, D. Kaiser and N. Maulide, ACS Cent. Sci., 2020, 6, 1869 CrossRef CAS PubMed; (d) H. Itoh and M. Inoue, Chem. Rev., 2019, 119, 10002 CrossRef CAS PubMed; (e) K. T. Mortensen, T. J. Osberger, T. A. King, H. F. Sore and D. R. Spring, Chem. Rev., 2019, 119, 10288 CrossRef CAS PubMed; (f) V. Martí-Centelles, M. D. Pandey, M. I. Burguete and S. V. Luis, Chem. Rev., 2015, 115, 8736 CrossRef PubMed; (g) T. O. Ronson, R. J. K. Taylor and I. J. S. Fairlamb, Tetrahedron, 2015, 71, 989 CrossRef CAS; (h) V. Martí-Centelles, M. D. Pandey, M. I. Burguete and S. V. Luis, Chem. Rev., 2015, 115, 8736 CrossRef PubMed; (i) A. Parenty, X. Moreau, G. Niel and J.-M. Campagne, Chem. Rev., 2013, 113, PR1 CrossRef CAS PubMed; (j) C. J. White and A. K. Yudin, Nat. Chem., 2011, 3, 509 CrossRef CAS PubMed; (k) A. Parenty, X. Moreau and J.-M. Campagne, Chem. Rev., 2006, 106, 911 CrossRef CAS PubMed; (l) A. Gradillas and J. Pérez-Castells, Angew. Chem., Int. Ed., 2006, 45, 6086 CrossRef CAS PubMed.
- For selected reviews on transannular reactions, see: (a) E. Reyes, L. Prieto, L. Carrillo, U. Uria and J. L. Vicario, Synthesis, 2022, 4167 CrossRef CAS; (b) E. Reyes, U. Uria, L. Carrillo and J. L. Vicario, Tetrahedron, 2014, 70, 9461 CrossRef CAS; (c) P. A. Clarke, A. T. Reeder and J. Winn, Synthesis, 2009, 691 CrossRef CAS.
- P. A. Wender, V. A. Verma, T. J. Paxton and T. H. Pillow, Acc. Chem. Res., 2008, 41, 40 CrossRef CAS PubMed.
- W. C. Still and V. J. Novack, J. Am. Chem. Soc., 1984, 106, 1148 CrossRef CAS.
- (a) W. C. Still and I. Galynker, Tetrahedron, 1981, 37, 3981 CrossRef CAS; (b) W. C. Still, L. J. MacPherson, T. Harada, J. F. Callahan and A. L. Rheingold, Tetrahedron, 1984, 40, 2275 CrossRef CAS.
- W. C. Still, J. Am. Chem. Soc., 1979, 101, 2493 CrossRef CAS.
- W. C. Still, S. Murata, G. Revial and K. Yoshihara, J. Am. Chem. Soc., 1983, 105, 625 CrossRef CAS.
- T. Takahashi, H. Nemoto, Y. Kanda, J. Tsuji, Y. Fukazawa and T. Okajima, Tetrahedron, 1987, 43, 5499 CrossRef CAS.
- J. A. Marshall, J. Lebreton, B. S. DeHoff and T. M. Jenson, Tetrahedron Lett., 1987, 28, 723 CrossRef CAS.
- E. J. Corey, S. Kim, S.-E. Yoo, K. C. Nicolaou, L. S. Melvin Jr., D. J. Brunelle, J. R. Falck, E. J. Trybulski, R. Lett and P. W. Sheldrake, J. Am. Chem. Soc., 1978, 100, 4620 CrossRef CAS.
- E. J. Corey, P. B. Hopkins, S. Kim, S.-E. Yoo, K. P. Nambiar and J. R. Falck, J. Am. Chem. Soc., 1979, 101, 7131 CrossRef CAS.
- I. Paterson and D. J. Rawson, Tetrahedron Lett., 1989, 30, 7463 CrossRef CAS.
- G. Höfle, A. C. O'Sullivan, G. Rihs, M. Sutter and T. Winkler, Tetrahedron, 1995, 51, 3159 CrossRef.
- B. Zhang, Y. Wang, S.-P. Yang, Y. Zhou, W.-B. Wu, W. Tang, J.-P. Zuo, Y. Li and J.-M. Yue, J. Am. Chem. Soc., 2012, 134, 20605 CrossRef CAS PubMed.
- E. Salamci, Tetrahedron, 2010, 66, 4010 CrossRef CAS.
- J. S. Clark and F. Romiti, Angew. Chem., Int. Ed., 2013, 52, 10072 CrossRef CAS PubMed.
- (a) B. M. Trost and Z. T. Ball, J. Am. Chem. Soc., 2005, 127, 17644 CrossRef CAS PubMed; (b) F. Lacombe, K. Radkowski, G. Seidel and A. Fürstner, Tetrahedron, 2004, 60, 7315 CrossRef CAS.
- (a) Y. Tu, Z.-X. Wang and Y. Shi, J. Am. Chem. Soc., 1996, 118, 9806 CrossRef CAS; (b) J. D. Warren and Y. Shi, J. Org. Chem., 1999, 64, 7675 CrossRef CAS.
- D. Driedger, A. Fers-Lidou, M. Schroeder, I. Elisia, G. Krystal and R. Britton, J. Am. Chem. Soc., 2025, 147, 28117 CrossRef CAS PubMed.
- H. Zhao, A. J. McMillan, T. Constantin, R. C. Mykura, F. Juliá and D. Leonori, J. Am. Chem. Soc., 2021, 143, 14806 CrossRef CAS PubMed.
- I. Paterson, R. Britton, O. Delgado, A. Meyer and K. G. Poullennec, Angew. Chem., Int. Ed., 2004, 43, 4629 CrossRef CAS PubMed.
- K. Nakazato, M. Oda and H. Fuwa, Bull. Chem. Soc. Jpn., 2023, 96, 257 CrossRef CAS.
- O. A. Ulanovskaya, J. Janjic, M. Suzuki, S. S. Sabharwal, P. T. Schumacker, S. J. Kron and S. A. Kozmin, Nat. Chem. Biol., 2008, 4, 418 CrossRef CAS PubMed.
- (a) Y. Cui, W. Tu and P. E. Floreancig, Tetrahedron, 2010, 66, 4867 CrossRef CAS PubMed; (b) Y. Cui, R. Balachandran, B. W. Day and P. E. Floreancig, J. Org. Chem., 2012, 77, 2225 CrossRef CAS PubMed.
- (a) H. Fuwa, A. Saito and M. Sasaki, Angew. Chem., Int. Ed., 2010, 49, 3041 CrossRef CAS PubMed; (b) H. Fuwa, M. Kawakami, K. Noto, T. Muto, Y. Suga, K. Konoki, M. Yotsu-Yamashita and M. Sasaki, Chem. – Eur. J., 2013, 19, 8100 CrossRef CAS PubMed.
- K. Iwasaki, K. K. Wan, A. Oppedisano, S. W. M. Crossley and R. A. Shenvi, J. Am. Chem. Soc., 2014, 136, 1300 CrossRef CAS PubMed.
- I. Paterson, C. DeSavi and M. Tudge, Org. Lett., 2001, 3, 213 CrossRef CAS PubMed.
- J. Hirabayashi, F. Yakushiji, A. Katsuyama and S. Ichikawa, Org. Lett., 2020, 22, 5545 CrossRef CAS PubMed.
- I. B. Seiple, Z. Zhang, P. Jakubec, A. Langlois-Mercier, P. M. Wright, D. T. Hog, K. Yabu, S. R. Allu, T. Fukuzaki, P. N. Carlsen, Y. Kitamura, X. Zhou, M. L. Condakes, F. T. Szczypiński, W. D. Green and A. G. Myers, Nature, 2016, 533, 338 CrossRef CAS PubMed.
- For reviews on central chirality-generating macrocyclizations: (a) K. Zheng and R. Hong, J. Org. Chem., 2024, 89, 7355 CrossRef CAS PubMed; (b) K. Zheng and R. Hong, Nat. Prod. Rep., 2019, 36, 1546 RSC.
- For a review on Prins macrocyclization CrossRef CAS PubMed; E. A. Crane and K. A. Scheidt, Angew. Chem., Int. Ed., 2010, 49, 8316 CrossRef CAS PubMed.
- S. K. Woo, M. S. Kwon and E. Lee, Angew. Chem., Int. Ed., 2008, 47, 3242 CrossRef CAS PubMed.
- (a) Y. Zhang, Q. Guo, X. Sun, J. Lu, Y. Cao, Q. Pu, Z. Chu, L. Gao and Z. Song, Angew. Chem., Int. Ed., 2018, 57, 942 CrossRef CAS PubMed; (b) P. A. Wender, C. T. Hardman, S. Ho, M. S. Jeffreys, J. K. Maclaren, R. V. Quiroz, S. M. Ryckbosch, A. J. Shimizu, J. L. Sloane and M. C. Stevens, Science, 2017, 358, 218 CrossRef CAS PubMed; (c) Y. Lu, S. K. Woo and M. J. Krische, J. Am. Chem. Soc., 2011, 133, 13876 CrossRef CAS PubMed; (d) G. E. Keck, Y. B. Poudel, T. J. Cummins, A. Rudra and J. A. Covel, J. Am. Chem. Soc., 2011, 133, 744 CrossRef CAS PubMed.
- K. B. Bahnck and S. D. Rychnovsky, J. Am. Chem. Soc., 2008, 130, 13177 CrossRef CAS PubMed.
- S. K. Woo and E. Lee, J. Am. Chem. Soc., 2010, 132, 4564 CrossRef CAS PubMed.
- D. W. Custar, T. P. Zabawa and K. A. Scheidt, J. Am. Chem. Soc., 2008, 130, 804 CrossRef CAS PubMed.
- H. Jin, J. Uenishi, W. J. Christ and Y. Kishi, J. Am. Chem. Soc., 1986, 108, 5644 CrossRef CAS.
- A. Gil, F. Albericio and M. Álvarez, Chem. Rev., 2017, 117, 8402 CrossRef PubMed.
- K. M. Foote, M. John and G. Pattenden, Synlett, 2001, 365 CrossRef CAS.
- B. Mi and R. E. Maleczka Jr., Org. Lett., 2001, 3, 1491 CrossRef CAS PubMed.
- J. A. Marshall and P. M. Eidam, Org. Lett., 2008, 10, 93 CrossRef CAS PubMed.
- W. Zhu, M. Jiménez, W.-H. Jung, D. P. Camarco, R. Balachandran, A. Vogt, B. W. Day and D. P. Curran, J. Am. Chem. Soc., 2010, 132, 9175 CrossRef CAS PubMed.
- For reviews on Diels–Alder cycloadditions, see: (a) M. Juhl and D. Tanner, Chem. Soc. Rev., 2009, 38, 2983 RSC; (b) K. Takao, R. Munakata and K. Tadano, Chem. Rev., 2005, 105, 4779 CrossRef CAS PubMed; (c) K. C. Nicolaou, S. A. Snyder, T. Montagnon and G. Vassilikogiannakis, Angew. Chem., Int. Ed., 2002, 41, 1668 CrossRef CAS; (d) E. Marrsault, A. Toró, P. Nowak and P. Deslongchamps, Tetrahedron, 2001, 57, 4243 CrossRef.
- E. J. Corey and M. Petrzilka, Tetrahedron Lett., 1975, 2537 CrossRef CAS.
- G. Stork and E. Nakamura, J. Am. Chem. Soc., 1983, 105, 5510 CrossRef CAS.
- J. A. McCauley, K. Nagasawa, P. A. Lander, S. G. Mischke, M. A. Semones and Y. Kishi, J. Am. Chem. Soc., 1998, 120, 7647 CrossRef CAS.
- F. Matsuura, R. Peters, M. Anada, S. S. Harried, J. Hao and Y. Kishi, J. Am. Chem. Soc., 2006, 128, 7463 CrossRef CAS PubMed.
- C. W. Zapf, B. A. Harrison, C. Drahl and E. J. Sorensen, Angew. Chem., Int. Ed., 2005, 44, 6533 CrossRef CAS PubMed.
- D. Niu and T. R. Hoye, Org. Lett., 2012, 14, 828 CrossRef CAS PubMed.
- C. M. Hayward, D. Yohannes and S. J. Danishefsky, J. Am. Chem. Soc., 1993, 115, 9345 CrossRef CAS.
- J. A. Gazaille, J. A. Abramite and T. Sammakia, Org. Lett., 2012, 14, 178 CrossRef CAS PubMed.
- For selected reviews on vinylogous aldol reactions: (a) C. Curti, L. Battistini, A. Sartori and F. Zanardi, Chem. Rev., 2020, 120, 2448 CrossRef CAS PubMed; (b) M. Kalesse, M. Cordes, G. Symkenberg and H.-H. Lu, Nat. Prod. Rep., 2014, 31, 563 RSC; (c) S. E. Denmark, J. R. Heemstra Jr. and G. L. Beutner, Angew. Chem., Int. Ed., 2005, 44, 4682 CrossRef CAS PubMed.
- (a) S. Saito, M. Shiozawa, T. Nagahara, M. Nakadai and H. Yamamoto, J. Am. Chem. Soc., 2000, 122, 7847 CrossRef CAS; (b) S. Saito, T. Nagahara, M. Shiozawa, M. Nakadai and H. Yamamoto, J. Am. Chem. Soc., 2003, 125, 6200 CrossRef CAS PubMed.
- (a) K. Zheng, D. Shen and R. Hong, J. Am. Chem. Soc., 2017, 139, 12939 CrossRef CAS PubMed; (b) K. Zheng, D. Shen, B. Zhang and R. Hong, J. Org. Chem., 2020, 85, 13818 CrossRef CAS PubMed; (c) K. Zheng, D. Shen, B. Zhang and R. Hong, J. Org. Chem., 2021, 86, 10991 CrossRef CAS PubMed.
- B. Zhang, K. Zheng and R. Hong, ACS Cent. Sci., 2023, 9, 84 CrossRef CAS PubMed.
- For reviews on macrocyclic cyclophane natural products: (a) S. Kotha, M. E. Shirbhate and G. T. Waghule, Beilstein J. Org. Chem., 2015, 11, 1274 CrossRef CAS PubMed; (b) T. Gulder and P. S. Baran, Nat. Prod. Rep., 2012, 29, 899 RSC.
- For selected reviews on Heck reaction: (a) J. Le Bras and J. Muzart, Chem. Rev., 2011, 111, 1170 CrossRef CAS PubMed; (b) D. Mc Cartney and P. J. Guiry, Chem. Soc. Rev., 2011, 40, 5122 RSC; (c) A. B. Dounay and L. E. Overman, Chem. Rev., 2003, 103, 2945 CrossRef CAS PubMed; (d) I. P. Beletskaya and A. V. Cheprakov, Chem. Rev., 2000, 100, 3009 CrossRef CAS PubMed.
- M. Groh, D. Meidlinger, G. Bringmann and A. Speicher, Org. Lett., 2012, 14, 4548 CrossRef CAS PubMed.
- D. Meidlinger, L. Marx, C. Bordeianu, S. Choppin, F. Colobert and A. Speicher, Angew. Chem., Int. Ed., 2018, 57, 9160 CrossRef CAS PubMed.
- L. Marx, D. Lamberty, S. Choppin, F. Colobert and A. Speicher, Eur. J. Org. Chem., 2021, 1351 CrossRef CAS.
- E. M. Stang and M. C. White, Nat. Chem., 2009, 1, 547 CrossRef CAS PubMed.
- A. M. Haydl and B. Breit, Chem. – Eur. J., 2017, 23, 541 CrossRef CAS PubMed.
- For a review on macrocyclic glycopeptide antibiotics CrossRef CAS PubMed; M. S. Butler, K. A. Hansford, M. A. T. Blaskovich, R. Halai and M. A. Cooper, J. Antibiot., 2014, 67, 631 CrossRef CAS PubMed.
- For a review on total synthesis of vancomycin-related glycopeptide antibiotics CrossRef CAS PubMed; A. Okano, N. A. Isley and D. L. Boger, Chem. Rev., 2017, 117, 11952 CrossRef CAS PubMed.
- T. C. Roberts, P. A. Smith, R. T. Cirz and F. E. Romersberg, J. Am. Chem. Soc., 2007, 129, 15830 CrossRef CAS PubMed.
- For selected reviews on Suzuki–Miyaura reaction: (a) J. W. Meringdal and D. Menche, Chem. Soc. Rev., 2025, 54, 5746 RSC; (b) A. J. J. Lennox and G. C. Lloyd-Jones, Chem. Soc. Rev., 2014, 43, 412 RSC; (c) A. J. J. Lennox and G. C. Lloyd-Jones, Angew. Chem., Int. Ed., 2013, 52, 7362 CrossRef CAS PubMed; (d) G. Siedel and A. Fürstner, Chem. Commun., 2012, 48, 2055 RSC; (e) A. Suzuki, Angew. Chem., Int. Ed., 2011, 50, 6723 Search PubMed; (f) R. Martin and S. L. Buchwald, Acc. Chem. Res., 2008, 41, 1461 CrossRef CAS PubMed; (g) S. Kotha, K. Lahiri and D. Kashinath, Tetrahedron, 2002, 58, 9633 CrossRef CAS; (h) N. Miyaura and A. Suzuki, Chem. Rev., 1995, 95, 2457 CrossRef CAS.
- M. Paetzel, J. J. Goodall, M. Kania, R. E. Dalbey and M. G. Page, J. Biol. Chem., 2004, 279, 30781 CrossRef CAS PubMed.
- (a) J. Dufour, L. Neuville and J. Zhu, Chem. – Eur. J., 2010, 16, 10523 CrossRef CAS PubMed; (b) J. Dufour and J. Zhu, Synlett, 2008, 2355 CAS.
- For selected reviews on Stille reaction: (a) L. Anwar, S. Ahmad, K. G. Ali, B. Parveen, A. F. Zahoor, S. A. R. Naqvi, J. Ashraf and U. Nazeer, Tetrahedron, 2026, 191, 135074 CrossRef CAS; (b) C. Cordovilla, C. Bartolomé, J. M. Martínez-Ilarduya and P. Espinet, ACS Catal., 2015, 5, 3040 CrossRef CAS; (c) P. Espinet and A. M. Echavarren, Angew. Chem., Int. Ed., 2004, 43, 2 CrossRef PubMed; (d) J. K. Stille, Angew. Chem., Int. Ed. Engl., 1986, 25, 508 CrossRef.
- H. Deng, J.-K. Jung, T. Liu, K. W. Kuntz, M. L. Snapper and A. H. Hoveyda, J. Am. Chem. Soc., 2003, 125, 9032 CrossRef CAS PubMed.
- T. Shinohara, H. Deng, M. L. Snapper and A. H. Hoveyda, J. Am. Chem. Soc., 2005, 127, 7334 CrossRef CAS PubMed.
- Y. Jia, M. Bois-Choussy and J. Zhu, Org. Lett., 2007, 9, 2401 CrossRef CAS PubMed.
- Y. Jia, M. Bois-Choussy and J. Zhu, Angew. Chem., Int. Ed., 2008, 47, 4167 CrossRef CAS PubMed.
- Z. Wang, M. Bois-Choussy, Y. Jia and J. Zhu, Angew. Chem., Int. Ed., 2010, 49, 2018 CrossRef CAS PubMed.
- R. C. Larock and E. K. Yum, J. Am. Chem. Soc., 1991, 113, 6689 CrossRef CAS.
- J. Garfunkle, F. S. Kimball, J. D. Trzupek, S. Takizawa, H. Shimamura, M. Tomishima and D. L. Boger, J. Am. Chem. Soc., 2009, 131, 16036 CrossRef CAS PubMed.
- H. Shimamura, S. P. Breazzano, J. Garfunkle, F. S. Kimball, J. D. Trzupek and D. L. Boger, J. Am. Chem. Soc., 2010, 132, 7776 CrossRef CAS PubMed.
- S. P. Breazzano and D. L. Boger, J. Am. Chem. Soc., 2011, 133, 18945 CrossRef PubMed.
- M. Nesic, D. B. Ryffel, J. Maturano, M. Shevlin, S. R. Pollack, D. R. Gauthier Jr., P. Trigo-Mouriño, L.-K. Zhang, D. M. Schultz, J. M. M. Dunn, L.-C. Campeau, N. R. Patel, D. A. Petrone and D. Sarlah, J. Am. Chem. Soc., 2022, 144, 14026 CrossRef CAS PubMed.
- Y.-C. Lin, F. Schneider, K. J. Eberle, D. Chiodi, H. Nakamura, S. H. Reisberg, J. Chen, M. Saito and P. S. Baran, J. Am. Chem. Soc., 2022, 144, 14458 CrossRef CAS PubMed.
- L. Yu, Y. Nagata and H. Nakamura, J. Am. Chem. Soc., 2024, 146, 2549 CrossRef CAS PubMed.
- H. Ogawa and H. Nakamura, Tetrahedron Chem, 2024, 11, 100084 CrossRef CAS.
- For selected reviews on tandem reactions: (a) H. Pellissier, Curr. Org. Chem., 2021, 25, 1457 CrossRef CAS; (b) H. Pellissier, Chem. Rev., 2013, 113, 442 CrossRef CAS PubMed; (c) D. Enders, C. Grondal and M. R. M. Hüttl, Angew. Chem., Int. Ed., 2007, 46, 1570 CrossRef CAS PubMed; (d) P. J. Parsons, C. S. Penkett and A. J. Shell, Chem. Rev., 1996, 96, 195 CrossRef CAS PubMed; (e) R. A. Bunce, Tetrahedron, 1995, 51, 13103 CrossRef CAS.
- (a) S. M. Winbush, D. J. Mergott and W. R. Roush, J. Org. Chem., 2008, 73, 1818 CrossRef CAS PubMed; (b) D. J. Mergott, S. A. Frank and W. R. Roush, Proc. Natl. Acad. Sci. U. S. A., 2004, 101, 11955 CrossRef CAS PubMed.
- R. K. Boeckman Jr. and T. E. Barta, J. Org. Chem., 1985, 50, 3421 CrossRef.
- W. R. Roush and M. Kageyama, Tetrahedron Lett., 1985, 26, 4327 CrossRef CAS.
- D. Mizukami, K. Iio, M. Oda, Y. Onodera and H. Fuwa, Angew. Chem., Int. Ed., 2002, 61, e202202549 CrossRef PubMed.
- (a) K. Nakazato, M. Oda and H. Fuwa, Org. Lett., 2022, 24, 4003 CrossRef CAS PubMed; (b) Ref. 27; (c) H. Fuwa, Synlett, 2024, 1488 CrossRef CAS; (d) R. Murakami, T. Mori, K. Murata and H. Fuwa, J. Org. Chem., 2025, 90, 753 CrossRef CAS PubMed.
- For selected reviews on oxa-Michael addition: (a) T. Ahmed and N. Ullah, Org. Chem. Front., 2021, 8, 1329 RSC; (b) J. Hu, M. Bian and H. Ding, Tetrahedron Lett., 2016, 57, 5519 CrossRef CAS; (c) C. F. Nising and S. Bräse, Chem. Soc. Rev., 2012, 41, 988 RSC; (d) H. Fuwa, Heterocycles, 2012, 85, 1255 CrossRef CAS.
- (a) H. Fuwa, K. Noto and M. Sasaki, Org. Lett., 2010, 12, 1636 CrossRef CAS PubMed; (b) H. Fuwa, T. Noguchi, K. Noto and M. Sasaki, Org. Biomol. Chem., 2012, 10, 8108 RSC; (c) H. Fuwa and M. Sasaki, Bull. Chem. Soc. Jpn., 2016, 89, 1403 CrossRef CAS; (d) K. Sakurai, M. Sasaki and H. Fuwa, Angew. Chem., Int. Ed., 2018, 57, 5143 CrossRef CAS PubMed.
- M. Egi, Y. Yamaguchi, N. Fujiwara and S. Akai, Org. Lett., 2008, 10, 1867 CrossRef CAS PubMed.
- For selected reviews on Meyer–Schuster rearrangement: (a) F. Justaud, A. Hachem and R. Grée, Eur. J. Org. Chem., 2021, 514 CrossRef CAS; (b) V. Cadierno, P. Crochet, S. E. García-Garrido and J. Gimeno, Dalton Trans., 2010, 39, 4015 RSC; (c) D. A. Engel and G. B. Dudley, Org. Biomol. Chem., 2009, 7, 4149 RSC.
- D. Csókás, A. X. Y. Ho, R. O. Ramabhadran and R. W. Bates, Org. Biomol. Chem., 2019, 17, 6293 RSC.
- For a review CrossRef CAS; H. Fuwa, Heterocycles, 2022, 104, 1709 CrossRef CAS.
- I. Shimizu and H. Nakagawa, Tetrahedron Lett., 1992, 33, 4957 CrossRef CAS.
- B. B. Snider and J. Zhou, Org. Lett., 2006, 8, 1283 CrossRef CAS PubMed.
- C. W. Barfoot, A. R. Burns, M. G. Edwards, M. N. Kenworthy, M. Ahmed, S. E. Chanahan and R. J. K. Taylor, Org. Lett., 2008, 10, 353 CrossRef CAS PubMed.
- For a review CrossRef PubMed; A. Fürstner, Acc. Chem. Res., 2021, 54, 861 CrossRef PubMed.
- For selected reviews on aza-Michael addition: (a) A. Y. Rulev, Eur. J. Org. Chem., 2023, e202300451 CrossRef CAS; (b) A. Y. Rulev, Adv. Synth. Catal., 2023, 365, 1908 CrossRef CAS; (c) A. Y. Rulev and I. A. Tyumentsev, Adv. Synth. Catal., 2022, 364, 1622 CrossRef CAS; (d) M. Sánchez-Roselló, M. Escolano, D. Gaviña and C. del Pozo, Chem. Rec., 2022, 22, e202100161 CrossRef PubMed; (e) Y.-X. Song and D.-M. Du, Adv. Synth. Catal., 2021, 363, 4667 CrossRef CAS; (f) R. W. Bates, W. Ko and V. Barát, Org. Biomol. Chem., 2020, 18, 810 RSC.
- K. Gebauer and A. Fürstner, Angew. Chem., Int. Ed., 2014, 53, 6393 CrossRef CAS PubMed.
- N. L. Magann, M. T. Blyth and M. S. Sherburn, Angew. Chem., Int. Ed., 2021, 60, 18561 CrossRef CAS PubMed.
- M. Kanematsu, M. Yoshida and K. Shishido, Angew. Chem., Int. Ed., 2011, 50, 2618 CrossRef CAS PubMed.
- (a) T. Masuda, K. Ohyama, A. Yoshimura and H. Fuwa, Org. Lett., 2024, 26, 2045 CrossRef CAS PubMed; (b) K. Ohyama, T. Masuda, K. Sakamoto, A. Yoshimura, K. Tatsumoto, K. Murata and H. Fuwa, J. Org. Chem., 2025, 90, 10500 CrossRef CAS PubMed.
- N. Marion, P. Carlqvist, R. Gealageas, P. de Frémont, F. Maseras and S. P. Nolan, Chem. – Eur. J., 2007, 13, 6437 CrossRef CAS PubMed.
- A. F. Petri, A. Bayer and M. E. Maier, Angew. Chem., Int. Ed., 2004, 43, 5821 CrossRef CAS PubMed.
- Y.-H. Jung, Y.-J. Kim, J. Lee and J. Tae, Chem. - Asian J., 2007, 2, 656 CrossRef CAS PubMed.
- G. V. M. Sharma, S. V. Reddy and K. V. S. Ramakrishna, Org. Biomol. Chem., 2012, 10, 3689 RSC.
- (a) M. Tortosa, N. A. Yakelis and W. R. Roush, J. Am. Chem. Soc., 2008, 130, 2722 CrossRef CAS PubMed; (b) M. Tortosa, N. A. Yakelis and W. R. Roush, J. Org. Chem., 2008, 73, 9657 CrossRef CAS PubMed.
- N. Kanoh, S. Itoh, K. Fujita, K. Sakanishi, R. Sugiyama, Y. Terajima, Y. Iwabuchi, S. Nishimura and H. Kakeya, Chem. – Eur. J., 2016, 22, 8586 CrossRef CAS PubMed.
- H. J. Reiter, T. K. Mukhopadhyay, F. Zhao, Q. Zhou, F. Liu, K. N. Houk and D. H. Trauner, J. Am. Chem. Soc., 2025, 147, 43206 CrossRef CAS PubMed.
- J. C. Schmid, K. Frey, M. Scheiner, J. F. G. Garzón, L. Stafforst, J.-N. Fricke, M. Schuppe, H. Schiewe, A. Zeeck, T. Weber, I. Usón, R. Kemkemer, M. Decker and S. Grond, Angew. Chem., Int. Ed., 2021, 60, 23212 CrossRef CAS PubMed.
- B. S. Olson and D. Trauner, Synlett, 2005, 700 CAS.
- A. J. Hall, S. P. Roche and L. M. West, Org. Lett., 2017, 19, 576 CrossRef CAS PubMed.
- Z. Gu and A. Zakarian, Org. Lett., 2010, 12, 4224 CrossRef CAS PubMed.
- For a review on point-to-axial chirality transfer RSC; T. T. Nguyen, Org. Biomol. Chem., 2019, 17, 6952 RSC.
- For selected reviews on metal-catalyzed alkyne hydroalkoxylation: (a) S. K. Nanda and R. Mallik, Chem. – Eur. J., 2021, 27, 15571 CrossRef CAS PubMed; (b) R. Dorel and A. M. Echavarren, Chem. Rev., 2015, 115, 9028 CrossRef CAS PubMed.
- S. Benson, M.-P. Collin, A. Arlt, B. Gabor, R. Goddard and A. Fürstner, Angew. Chem., Int. Ed., 2011, 50, 8739 CrossRef CAS PubMed.
- For selected reviews on gold catalysis: (a) B. Lin, T. Liu and T. Luo, Nat. Prod. Rep., 2024, 41, 1091 RSC; (b) S. De, A. K. Dan, R. Sahu, S. Parida and D. Das, Chem. - Asian J., 2022, 17, e202200896 CrossRef CAS PubMed; (c) D. Pflästerer and A. S. K. Hashmi, Chem. Soc. Rev., 2016, 45, 1331 RSC.
- A. Hermann and A. Fürstner, Angew. Chem., Int. Ed., 2025, 64, e202518836 CrossRef CAS PubMed.
- J. P. Tuccinardi and J. L. Wood, J. Am. Chem. Soc., 2022, 144, 20539 CrossRef CAS PubMed.
- J. Tang, W. Li, T.-Y. Chiu, F. Martínez-Peña, Z. Luo, C. T. Chong, Q. Wei, N. Gazaniga, T. J. West, Y. Y. See, L. L. Lairson, C. G. Parker and P. S. Baran, Nature, 2023, 622, 507 CrossRef CAS PubMed.
- Y. Li and G. Pattenden, Tetrahedron, 2011, 67, 10045 CrossRef CAS.
- Z. Meng and A. Fürstner, J. Am. Chem. Soc., 2019, 141, 805 CrossRef CAS PubMed.
- For reviews on the conformational property of macrocycles: (a) S. D. Appavoo, S. Huh, D. B. Diaz and A. K. Yudin, Chem. Rev., 2019, 119, 9724 CrossRef CAS PubMed; (b) E. M. Larsen, M. R. Wilson and R. E. Taylor, Nat. Prod. Rep., 2015, 32, 1183 RSC.
- For reviews on conformational design: (a) R. Chen, Y. Shen, S. Yang and Y. Zhang, Angew. Chem., Int. Ed., 2020, 59, 14198 CrossRef CAS PubMed; (b) R. W. Hoffmann, Angew. Chem., Int. Ed., 2000, 39, 2054 CrossRef; (c) R. W. Hoffmann, Angew. Chem., Int. Ed. Engl., 1992, 31, 1124 CrossRef.
- For reviews on bioinspired or biomimetic synthesis: (a) L. Chen, P. Chen and Y. Jia, Acc. Chem. Res., 2024, 57, 3524 CrossRef CAS PubMed; (b) M. Razzak and J. K. De Brabander, Nat. Chem. Biol., 2011, 7, 865 CrossRef CAS PubMed; (c) J. Kim and M. Movassaghi, Chem. Soc. Rev., 2009, 38, 3035 RSC; (d) P. G. Bulger, S. K. Bagal and R. Marquez, Nat. Prod. Rep., 2008, 25, 254 RSC; (e) C. M. Beaudry, J. P. Malerich and D. Trauner, Chem. Rev., 2005, 105, 4757 CrossRef CAS PubMed; (f) M. C. de la Torre and M. A. Sierra, Angew. Chem., Int. Ed., 2003, 43, 160 CrossRef PubMed; (g) K. C. Nicolaou, T. Montagnon and S. A. Snyder, Chem. Commun., 2003, 551 RSC.
- For selected reviews: (a) M. O. Marcarino, S. Cicetti, M. M. Zanardi and A. M. Sarotti, Nat. Prod. Rep., 2022, 39, 58 RSC; (b) F. L. P. Costa, A. C. F. de Albuquerque, R. G. Fiorot, L. M. Lião, L. H. Martorano, G. V. S. Mota, A. L. Valverde, J. W. M. Carneiro and F. M. dos Santos Jr., Org. Chem. Front., 2021, 8, 2019 RSC; (c) M. O. Marcarino, M. M. Zanardi, S. Cicetti and A. M. Sarotti, Acc. Chem. Res., 2020, 53, 1922 CrossRef CAS PubMed; (d) G. Lauro and G. Bifulco, Eur. J. Org. Chem., 2020, 3929 CrossRef CAS.
- For selected examples: (a) P. Bonnet, D. K. Agrafiotis, F. Zhu and E. Martin, J. Chem. Inf. Model., 2009, 49, 2242 CrossRef CAS PubMed; (b) I.-J. Chen and N. Foloppe, Bioorg. Med. Chem., 2013, 21, 7898 CrossRef CAS PubMed; (c) V. Poongavanam, E. Danelius, S. Peintner, L. Alcaraz, G. Caron, M. D. Cummings, S. Wlodek, M. Erdelyi, P. C. D. Hawkins, G. Ermondi and J. Kihlberg, ACS Omega, 2018, 3, 11742 CrossRef CAS PubMed; (d) O. Gutten, D. Bím, J. Řezác and L. Rulíšek, J. Chem. Inf. Model., 2018, 58, 48 CrossRef CAS PubMed; (e) H. M. Hashim and J. N. Harvey, J. Chem. Theory Comput., 2025, 21, 10604 CrossRef CAS PubMed.
- (a) K. Murata, H. Mori and H. Fuwa, Bull. Chem. Soc. Jpn., 2022, 95, 1775 CrossRef CAS; (b) T. Obana, M. Nakajima, K. Nakazato, H. Nakagawa, K. Murata, M. Tsuda and H. Fuwa, J. Am. Chem. Soc., 2024, 146, 29836 CrossRef CAS PubMed.
- O. Iglesias-Menduiña, C. Martínez, B. Vaz, S. Alvarez, R. Alvarez and A. R. de Lera, J. Org. Chem., 2025, 90, 2429 CrossRef PubMed.
- C. Cuadrado, F. Cen-Pacheco and A. H. Daranas, Org. Lett., 2024, 26, 6529 CrossRef CAS PubMed.
- For selected reviews: (a) Y. Liu, B. Yang, H. He and S. Gao, Angew. Chem., Int. Ed., 2025, 64, e202516814 CrossRef CAS PubMed; (b) G. Occhialini and A. E. Wendlandt, Acc. Chem. Res., 2025, 58, 2255 CrossRef CAS PubMed; (c) M. Iglhaut and T. Bach, Acc. Chem. Res., 2025, 58, 777 CrossRef CAS PubMed; (d) M. Huang, T. Pan, X. Jiang and S. Luo, J. Am. Chem. Soc., 2023, 145, 10917 CrossRef CAS PubMed; (e) P.-Z. Wang, W.-J. Xiao and J.-R. Chen, Nat. Rev. Chem., 2023, 7, 35 CrossRef PubMed.
- For selected reviews: (a) E. Romero, B. S. Jones, B. N. Hogg, A. R. Casamajo, M. A. Hayes, S. L. Flitsch, N. J. Turner and C. Schnepel, Angew. Chem., Int. Ed., 2021, 60, 16824 CrossRef CAS PubMed; (b) F. Li, X. Zhang and H. Renata, Curr. Opin. Chem. Biol., 2019, 49, 25 CrossRef CAS PubMed.
- (a) B. J. Curtis, H. A. Boesger, J. J. Schmidt, M. L. Adrover-Castellano, A. N. Momin, C. A. Glasser and D. H. Sherman, JACS Au, 2026, 6, 801 CrossRef CAS PubMed; (b) M. M. Gilbert, M. D. DeMars II, S. Yang, J. M. Grandner, S. Wang, H. Wang, A. R. H. Narayan, D. H. Sherman, K. N. Houk and J. Montgomery, ACS Cent. Sci., 2017, 3, 1304 CrossRef CAS PubMed; (c) P. Le-Huu, T. Heidt, B. Claasen, S. Laschat and V. B. Urlacher, ACS Catal., 2015, 5, 1772 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |