Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Enantioselective copper(II) catalysed (4 + 1) cycloaddition of aza-o-quinone methides and bromomalonates. Facile access to enantioenriched indolines
Sergio
Torres-Oya
,
Manuel A.
Fernández-Rodríguez
 * and
Mercedes
Zurro
* and
Mercedes
Zurro
 *
*
Universidad de Alcalá (IRYCIS), Departamento de Química Orgánica y Química Inorgánica, Instituto de Investigación Química “Andrés M. del Río” (IQAR), 28805 Alcalá de Henares, Madrid, Spain. E-mail: mercedes.zurro@uah.es
First published on 23rd January 2026
Abstract
Optically active indolines are valuable structural motifs present in numerous naturally occurring and biologically active molecules. Although several methodologies have been reported in the literature for the synthesis of chiral indolines, many of them rely on the hydrogenation of indoles using expensive metal catalysts. In this report, a copper(II)-catalysed enantioselective (4 + 1) cycloaddition of aza-o-quinone methides (aza-o-QMs) with bromomalonates to access indolines is described. The reactive aza-o-QMs are generated in situ from simple and easily accessible 2-chloromethyl arylsulfonamides under basic conditions, and subsequently undergo cyclization with the in situ formed bromomalonate anion to deliver diverse chiral indoline derivatives in up to 69% yields and 96![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 er. Scale up and further derivatizations occurred without erosion of enantioselectivity, showing the robustness of this methodology.
4 er. Scale up and further derivatizations occurred without erosion of enantioselectivity, showing the robustness of this methodology.
Introduction
Chiral indolines are privileged scaffolds found in many alkaloids1–4 such as strychnine,5 as well as in pharmaceutical compounds such as physostigmine6 and pentopril7 (Fig. 1). Structurally, indolines consist of a partially saturated skeleton in which a benzene ring is fused to a pyrrolidine ring. The benzene ring can interact with the hydrophobic residues of proteins through van der Waals interactions, whereas the pyrrolidine NH is capable of forming hydrogen bonding interactions with amino acid residues. Notably, in the indoline structure, the two rings are non-coplanar, which increases water solubility relative to the corresponding indole scaffold.8Due to their exceptional bioactive properties, considerable attention has been devoted in recent years to the development of novel enantioselective methodologies for the synthesis of chiral indolines.9–14 Among these, asymmetric hydrogenation of indoles is one of the most explored approaches. These processes offer significant advantages such as operational simplicity and atom economy; however, in many cases, they require expensive rhodium,15–20 ruthenium21–23 and iridium24,25 catalysts. Other approaches to access optically active indolines involve their de novo synthesis from different precursors, thereby avoiding the use of indoles as starting materials. This strategy mainly relies on the construction of the pyrrolidine ring through cyclization.26–28
Asymmetric cycloaddition constitutes an efficient and atom-economical strategy for the synthesis of structurally diverse cyclic derivatives, which are often difficult to access via traditional synthetic methodologies utilizing readily available starting materials.29–31 In particular, the (4 + 1) cycloaddition of aza-o-QM with C1 synthons provides a highly efficient route to construct indolines with precise stereocontrol. In this context, decarboxylative cyclization strategies involving benzoxazinanones under palladium(0)32 or copper(II)33 catalysis have been developed in recent years for the synthesis of chiral indolines (Scheme 1a and b). In both methodologies, the enantioselectivity originates from the coordination of the metal catalyst with the vinyl and alkynyl benzoxazinanone, respectively, generating in situ a chiral polarized aza-o-quinone methide. This intermediate then reacts through (4 + 1) cycloaddition with sulfur ylides to afford 2,3-disubstituted indolines with high enantioselectivity.
On the other hand, in 2009, Stoltz and co-workers developed a stereoablative alkylation of 3-halooxindoles with malonates (Scheme 1c).34 The reaction proceeds via a pseudo aza-o-QM intermediate, with a chiral copper(II) complex serving as the catalyst. The complex not only increases the acidity of the malonate but also provides a chiral environment, leading to oxindoles with C3 all-carbon quaternary sterocenters in moderate to excellent enantioselectivities. More recently, Han and co-workers reported an asymmetric cycloaddition of stabilized o-quinone methides and bromomalonate in the presence of a phase transfer organocatalyst (PTC), yielding dihydrobenzofurans with enantioselectivities ranging from moderate to excellent (Scheme 1d).35 This study demonstrates that bromomalonate can serve as a C1 synthon, reacting with o-quinone methides in a (4 + 1) cycloaddition.
Drawing inspiration from these studies, and in line with our research program on the synthesis of aza-heterocycles utilizing aza-o-QMs as reactive intermediates,36 we envisioned that an enantioselective (4 + 1) cycloaddition catalysed by copper(II) salts could provide an efficient and straightforward strategy for the construction of optically active indolines, employing chloromethyl arylsulfonamides as aza-o-QM precursors and bromomalonate as the C1 synthon (Scheme 1e).
Results and discussion
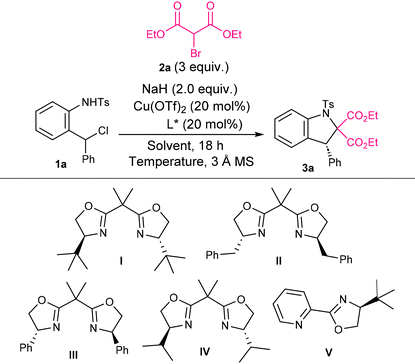
To initiate the investigation, and based on our experience in cycloadditions involving aza-o-QM intermediates, N-(2-(chloro(phenyl)methyl)phenyl)-4-methylbenzenesulfonamide 1a was selected as the model substrate. Diethyl 2-bromomalonate 2a was employed as the coupling partner, together with N,N-diisopropylethylamine (DIPEA) as the base, 20 mol% Cu(OTf)2 as the catalyst, and bis(oxazoline) I as the ligand, using dichloromethane (DCM) as the solvent. The resulting reaction mixture was stirred at room temperature for 18 h; however, analysis of the crude reaction mixture revealed a complex mixture of products in this initial experiment (Table 1, entry 1). We next examined different bases, such as Cs2CO3, K2CO3 or DBU, under the same reaction conditions. However, in all cases, complex mixtures were observed (see full optimization in the SI). To our delight, the use of sodium hydride as the base afforded the desired indoline 3a, albeit as a racemic mixture (Table 1, entry 2). Encouraged by this result, we then screened a series of bis(oxazoline) ligands II–IV and a pyridine–oxazoline ligand V (Table 1, entries 3–6), observing the appearance of enantioselectivity with ligands II, III and V. Among them, ligand III bearing a phenyl substituent provided the highest enantiomeric ratio (58![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 42 er). A subsequent temperature screening with ligand III revealed that lowering the temperature enhanced the enantioselectivity (Table 1, entries 7 and 8), with −20 °C giving the best result (68
42 er). A subsequent temperature screening with ligand III revealed that lowering the temperature enhanced the enantioselectivity (Table 1, entries 7 and 8), with −20 °C giving the best result (68![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 32 er). We then performed a solvent evaluation using a wide variety of solvents, including toluene, ethyl acetate and MeOH (see full optimization in the SI), which revealed that ethereal solvents were beneficial for the reaction (Table 1, entries 9 and 10). In particular, methyl tert-butyl ether (MTBE) provided the highest yield and enantioselectivity (70% yield, 89
32 er). We then performed a solvent evaluation using a wide variety of solvents, including toluene, ethyl acetate and MeOH (see full optimization in the SI), which revealed that ethereal solvents were beneficial for the reaction (Table 1, entries 9 and 10). In particular, methyl tert-butyl ether (MTBE) provided the highest yield and enantioselectivity (70% yield, 89![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 11 er). Reproducibility tests under the latter conditions initially showed variations in yield and enantioselectivity (see the SI). To circumvent this issue, commercially available sodium hydride (90% purity) was used instead of 60% NaH on paraffin. This subtle modification led to a reproducible reaction, affording indoline 3a in 69% yield and 95
11 er). Reproducibility tests under the latter conditions initially showed variations in yield and enantioselectivity (see the SI). To circumvent this issue, commercially available sodium hydride (90% purity) was used instead of 60% NaH on paraffin. This subtle modification led to a reproducible reaction, affording indoline 3a in 69% yield and 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 er (Table 1, entry 11). Accordingly, the optimal reaction conditions were established as follows: copper(II) triflate (20 mol%) and bis(oxazoline) ligand III (20 mol%) with substrate (1.0 equiv.), diethyl 2-bromomalonate (3.0 equiv.), and MTBE as solvent at −20 °C for 23 h.
5 er (Table 1, entry 11). Accordingly, the optimal reaction conditions were established as follows: copper(II) triflate (20 mol%) and bis(oxazoline) ligand III (20 mol%) with substrate (1.0 equiv.), diethyl 2-bromomalonate (3.0 equiv.), and MTBE as solvent at −20 °C for 23 h.
| Entry | Base | Ligand | Solvent | Temperature | Yielda | erb |
|---|---|---|---|---|---|---|
| Reaction conditions: Cu(OTf)2 (20 mol%), L (20 mol%) and 3 Å molecular sieves (32.0 mg) were stirred in the solvent (0.1 M) at r.t. for 30 min. Then 1a (0.1 mmol, 1.0 equiv.), diethyl 2-bromomalonate (0.3 mmol, 3.0 equiv.) and base (0.2 mmol, 2.0 equiv.) were added. The reaction mixture was stirred for 18 h.a Isolated yields.b The enantiomeric ratio was determined by analytical chiral HPLC.c 60% NaH on paraffin was used.d 90% NaH was used.e The reaction mixture was stirred for 23 h. | ||||||
| 1 | DIPEA | I | DCM | r.t. | n.d. | n.d. |
| 2 | NaHc | I | DCM | r.t. | 68 | 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 50 |
| 3 | NaHc | II | DCM | r.t. | 67 | 55![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45 45 |
| 4 | NaHc | III | DCM | r.t. | 66 | 58![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 42 42 |
| 5 | NaHc | IV | DCM | r.t. | 77 | 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 50 |
| 6 | NaHc | V | DCM | r.t. | 63 | 53![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 47 47 |
| 7 | NaHc | III | DCM | −50 °C | 31 | 67![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 33 33 |
| 8 | NaHc | III | DCM | −20 °C | 30 | 68![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 32 32 |
| 9 | NaHc | III | Et2O | −20 °C | 52 | 81![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 19 19 |
| 10 | NaHc | III | MTBE | −20 °C | 70 | 89![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 11 11 |
| 11e | NaHd | III | MTBE | −20 °C | 69 | 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5 |
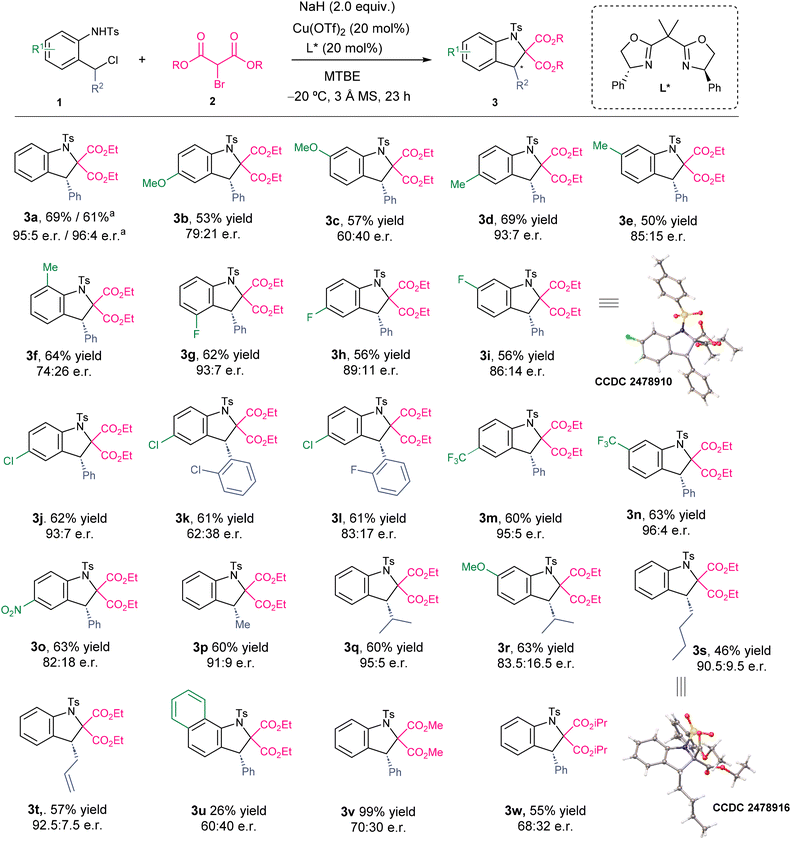
With the optimal conditions in hand, we explored the substrate scope of the transformation (Scheme 2). First, we examined the influence of an electron-donating substituent at the aromatic ring of aniline 1. A methoxy substituent at positions 4- and 3- led to indolines 3b and 3c, respectively, with diminished enantioselectivity. In contrast, the methyl substituted aza-quinone precursors 1d–f delivered the corresponding indolines with excellent enantioselectivities for 3d bearing a methyl substituent at position 5- and with slightly lower enantioselectivities for the 6- and 7-substituted derivatives 3e and 3f. Next, we evaluated the effect of a fluoro substituent at different positions of the aniline ring; in all cases, indolines 3g–i were obtained with excellent enantioselectivities. Similarly, the chloro substituted aza-o-QM precursor 1j led to indoline 3j in 93![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7 er and 62% yield. However, the analogous substrates 1k and 1l bearing chloro or fluoro substituents at the ortho position of the benzylic phenyl ring gave slightly lower enantioselectivities in 3k and 3l. We also investigated the influence of other electron-withdrawing groups on the benzene ring. Derivatives 3m and 3n bearing CF3 substituents at positions 5- and 6- were formed in excellent enantioselectivities, whereas the analogous 5-nitroindoline 3o was obtained in 63% yield and 82
7 er and 62% yield. However, the analogous substrates 1k and 1l bearing chloro or fluoro substituents at the ortho position of the benzylic phenyl ring gave slightly lower enantioselectivities in 3k and 3l. We also investigated the influence of other electron-withdrawing groups on the benzene ring. Derivatives 3m and 3n bearing CF3 substituents at positions 5- and 6- were formed in excellent enantioselectivities, whereas the analogous 5-nitroindoline 3o was obtained in 63% yield and 82![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18 er. The influence of the substituent at the benzylic position of the chloromethyl arylsulfonamide was also examined. The methyl substituted derivative 3p showed high yield and enantioselectivity. Expanding the alkyl scope at the benzylic position, an isopropyl-substituted aniline derivative led to indoline 3q in 60% yield and 95
18 er. The influence of the substituent at the benzylic position of the chloromethyl arylsulfonamide was also examined. The methyl substituted derivative 3p showed high yield and enantioselectivity. Expanding the alkyl scope at the benzylic position, an isopropyl-substituted aniline derivative led to indoline 3q in 60% yield and 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 er, while a substrate bearing both an isopropyl group at the benzylic position and a methoxy group on the aromatic ring produced 3r in 63% yield and 83.5
5 er, while a substrate bearing both an isopropyl group at the benzylic position and a methoxy group on the aromatic ring produced 3r in 63% yield and 83.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16.5 er. This result is particularly noteworthy when compared to that obtained for 1c, which bears a phenyl group at the benzylic position; in that case, the enantiomeric ratio of the corresponding indoline 3c is significantly lower (60
16.5 er. This result is particularly noteworthy when compared to that obtained for 1c, which bears a phenyl group at the benzylic position; in that case, the enantiomeric ratio of the corresponding indoline 3c is significantly lower (60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40 er). Moreover, a butyl-substituted substrate 1s furnished 3s in 90.5
40 er). Moreover, a butyl-substituted substrate 1s furnished 3s in 90.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9.5 er, while an allyl-substituted aza-o-QM precursor 1t gave 3t in 92.5
9.5 er, while an allyl-substituted aza-o-QM precursor 1t gave 3t in 92.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7.5 er. Finally, we performed the reaction with the extended-conjugation derivative 1u, which afforded indoline 3u in a low yield and enantioselectivity caused by the higher steric hindrance due to the extended conjugation of the aromatic ring.
7.5 er. Finally, we performed the reaction with the extended-conjugation derivative 1u, which afforded indoline 3u in a low yield and enantioselectivity caused by the higher steric hindrance due to the extended conjugation of the aromatic ring.
Furthermore, we also studied the effect of the 2-bromomalonate on the reaction. While the reaction of dimethyl 2-bromomalonate with the aza-o-QM precursor 1a gave indoline 3v in 99% yield and 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 er, diisopropyl 2-bromomalonate led to indoline 3w in 55% yield and 68
30 er, diisopropyl 2-bromomalonate led to indoline 3w in 55% yield and 68![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 32 er, indicating the reaction is sensitive to a higher or lower steric hindrance of the malonate.
32 er, indicating the reaction is sensitive to a higher or lower steric hindrance of the malonate.
The absolute configuration of the newly formed chiral center was determined by X-ray diffraction analysis of 3i and 3s, confirming the R-configuration in both cases. The configuration of the remaining indolines was assigned by analogy. Finally, a scale up of the model substrate 1a to the 1 mmol scale afforded 3a in 61% yield with a 96![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 er, demonstrating the robustness of the developed methodology.
4 er, demonstrating the robustness of the developed methodology.
It is worth noting that the synthesis of 2-substituted chiral indolines has been widely explored using various precursors.37 In contrast, installing a chiral centre at the 3-position is more challenging, and in the few cases where it has been successfully achieved, the scope of substituents at this position is limited. Furthermore, to the best of our knowledge, the synthesis of optically active indolines bearing only a chiral centre at the 3-position has not yet been reported using aza-o-QM,38,39 highlighting the novelty of the transformation reported herein.
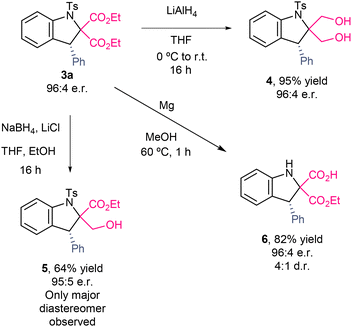
To demonstrate the synthetic applicability of the synthesized indolines, further derivatizations were carried out (Scheme 3). First, reduction with lithium aluminium hydride afforded diol 4 in 95% yield. Selective reduction of an ester group was achieved using sodium borohydride as the reducing agent in the presence of lithium chloride as the additive, yielding derivative 5 in 64% yield as a single diastereomer. Additionally, treatment with magnesium in methanol furnished indoline 6, bearing a free NH group and a carboxylic acid, in 82% yield with 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr. All transformations occurred with retention of enantioselectivity.
1 dr. All transformations occurred with retention of enantioselectivity.
A proposal for the mechanism is depicted in Scheme 4. First, the chloromethyl arylsulfonamide is deprotonated by NaH, generating the aza-o-QM intermediate A with the release of a molecule of hydrogen. On the other hand, based on Evans’ studies,40,41 we propose that the bromomalonate is activated by the copper complex, which coordinates in a bidentate manner, increasing its acidity. Furthermore, the copper complex may also coordinate with the nitrogen atom of the aza-o-QM intermediate, promoting an effective approach of the bromomalonate toward the reactive methylene moiety. Upon deprotonation of the bromomalonate by NaH, it is converted into a nucleophile that reacts with the aza-o-QM intermediate in an enantioselective fashion, leading to intermediate B, which subsequently undergoes intramolecular cyclization to afford indoline 3.
Conclusions
In this work, we report a copper(II)-catalysed enantioselective (4 + 1) cycloaddition between in situ-generated aza-o-QMs, derived from readily available chloromethyl arylsulfonamides and bromomalonates, providing facile access to chiral indolines under mild conditions. This method, which expands the use of copper bis(oxazoline) complexes in asymmetric catalysis for the synthesis of chiral indolines, features a broad substrate scope, high enantioselectivity, and operational simplicity, offering a practical and scalable route to indoline frameworks. The developed methodology enables the synthesis of 3-substituted optically active indolines bearing a wide variety of substituents at this position. However, some limitations remain, such as the use of electron rich substrates, which leads to lower enantioinductions. Further derivatizations show the synthetic applicability of the synthesized indolines, allowing access to various alcohol derivatives and N-unsubstituted indolines. Ongoing investigations in our laboratory aim to expand this methodology toward the synthesis of novel heterocycles.Author contributions
S. T.-O.: investigation. M. A. F.-R.: funding acquisition and writing – review and editing. M. Z.: conceptualization, supervision, writing – original draft, review and editing, and funding acquisition.Conflicts of interest
There are no conflicts to declare.Data availability
Supplementary information (SI): all experimental and characterization data, as well as NMR spectra. See DOI: https://doi.org/10.1039/d6ob00091f.CCDC 2478910 (3i) and and 2478916 (3s) contain the supplementary crystallographic data for this paper.42a,b
Acknowledgements
We are grateful to Ministerio de Ciencia, Innovación y Universidades (grant PID2023-146343NB-I00 financed by MCIU/AEI/10.13039/501100011033/FEDER, UE); RICORS2040 (RD24/0004/0008) funded by Instituto de Salud Carlos III (ISCIII) and co-funded by the European Union; and Comunidad de Madrid (grant CM/DEMG/2024-002).References
- Z. Gan, P. T. Reddy, S. Quevillon, S. Couve-Bonnaire and P. Arya, Stereocontrolled Solid-Phase Synthesis of a 90-Membered Library of Indoline-Alkaloid-like Polycycles from an Enantioenriched Aminoindoline Scaffold, Angew. Chem., Int. Ed., 2005, 44, 1366–1368, DOI:10.1002/anie.200462298
.
- K. C. Nicolaou, P. Bheema Rao, J. Hao, M. V. Reddy, G. Rassias, X. Huang, D. Y. Chen and S. A. Snyder, The Second Total Synthesis of Diazonamide A†, Angew. Chem., Int. Ed., 2003, 42, 1753–1758, DOI:10.1002/anie.200351112
.
- M. Ori, N. Toda, K. Takami, K. Tago and H. Kogen, Total Synthesis of (+)-Benzastatin E via Diastereoselective Grignard Addition to 2-Acylindoline, Angew. Chem., Int. Ed., 2003, 42, 2540–2543, DOI:10.1002/anie.200351069
.
- D. Zhang, H. Song and Y. Qin, Total Synthesis of Indoline Alkaloids: A Cyclopropanation Strategy, Acc. Chem. Res., 2011, 44, 447–457, DOI:10.1021/ar200004w
.
- H. Zhang, J. Boonsombat and A. Padwa, Total Synthesis of (±)-Strychnine via a [4 + 2]-Cycloaddition/Rearrangement Cascade, Org. Lett., 2007, 9, 279–282, DOI:10.1021/ol062728b
.
- T. Bui, S. Syed and C. F. I. Barbas II, Thiourea-Catalyzed Highly Enantio- and Diastereoselective Additions of Oxindoles to Nitroolefins: Application to the Formal Synthesis of (+)-Physostigmine, J. Am. Chem. Soc., 2009, 131, 8758–8759, DOI:10.1021/ja903520c
.
- A. Rakhit, G. M. Kochak, V. Tipnis, P. Radensky, M. E. Hurley and R. Williams, Pharmacokinetics of pentopril in the elderly, Br. J. Clin. Pharmacol., 1987, 24, 351–357, DOI:10.1111/j.1365-2125.1987.tb03180.x
.
- H. Wei, B. Li, N. Wang, Y. Ma, J. Yu, X. Wang, J. Su and D. Liu, Development and Application of Indolines in Pharmaceuticals, ChemistryOpen, 2023, 12, e202200235, DOI:10.1002/open.202200235
.
- L. Chen, C. Wang, L. Zhou and J. Sun, Chiral 2,3-Disubstituted Indolines from Indoles and Aldehydes by Organocatalyzed Tandem Synthesis Involving Reduction by Trichlorosilane, Adv. Synth. Catal., 2014, 356, 2224–2230, DOI:10.1002/adsc.201301133
.
- R. Borrmann, N. Knop and M. Rueping, Asymmetric Synthesis of Optically Active Spirocyclic Indoline Scaffolds through an Enantioselective Reduction of Indoles, Chem. – Eur. J., 2017, 23, 798–801, DOI:10.1002/chem.201605450
.
- Q. Gu and D. Yang, Enantioselective Synthesis of (+)-Mitomycin K by a Palladium-Catalyzed Oxidative Tandem Cyclization, Angew. Chem., Int. Ed., 2017, 56, 5886–5889, DOI:10.1002/anie.201701895
.
- M. Rueping, C. Brinkmann, A. P. Antonchick and I. Atodiresei, Org. Lett., 2010, 12, 4604–4607, DOI:10.1021/ol1019234
.
- K. Saito, Y. Shibata, M. Yamanaka and T. Akiyama, Chiral Phosphoric Acid-Catalyzed Oxidative Kinetic Resolution of Indolines Based on Transfer Hydrogenation to Imines, J. Am. Chem. Soc., 2013, 135, 11740–11743, DOI:10.1021/ja406004q
.
- F. O. Arp and G. C. Fu, Kinetic Resolutions of Indolines by a Nonenzymatic Acylation Catalyst, J. Am. Chem. Soc., 2006, 128, 14264–14265, DOI:10.1021/ja0657859
.
- R. Kuwano, K. Sato, T. Kurokawa, D. Karube and Y. Ito, Catalytic Asymmetric Hydrogenation of Heteroaromatic Compounds, Indoles, J. Am. Chem. Soc., 2000, 122, 7614–7615, DOI:10.1021/ja001271c
.
- R. Kuwano, K. Kaneda, T. Ito, K. Sato, T. Kurokawa and Y. Ito, Highly Enantioselective Synthesis of Chiral 3-Substituted Indolines by Catalytic Asymmetric Hydrogenation of Indoles, Org. Lett., 2004, 6, 2213–2215, DOI:10.1021/ol049317k
.
- R. Kuwano, M. Kashiwabara, K. Sato, T. Ito, K. Kaneda and Y. Ito, Catalytic asymmetric hydrogenation of indoles using a rhodium complex with a chiral bisphosphine ligand PhTRAP, Tetrahedron: Asymmetry, 2006, 17, 521–535, DOI:10.1016/j.tetasy.2006.01.016
.
- N. Mršić, T. Jerphagnon, A. J. Minnaard, B. L. Feringa and J. G. de Vries, Asymmetric hydrogenation of 2-substituted N-protected-indoles catalyzed by rhodium complexes of BINOL-derived phosphoramidites, Tetrahedron: Asymmetry, 2010, 21, 7–10, DOI:10.1016/j.tetasy.2009.11.017
.
- A. M. Maj, I. Suisse, C. Méliet and F. Agbossou-Niedercorn, Enantioselective hydrogenation of indoles derivatives catalyzed by Walphos/rhodium complexes, Tetrahedron: Asymmetry, 2010, 21, 2010–2014, DOI:10.1016/j.tetasy.2010.06.030
.
- J. Wen, X. Fan, R. Tan, H. Chien, Q. Zhou, L. W. Chung and X. Zhang, Brønsted-Acid-Promoted Rh-Catalyzed Asymmetric Hydrogenation of N-Unprotected Indoles: A Cocatalysis of Transition Metal and Anion Binding, Org. Lett., 2018, 20, 2143–2147, DOI:10.1021/acs.orglett.8b00312
.
- R. Kuwano and M. Kashiwabara, Ruthenium-Catalyzed Asymmetric Hydrogenation of N-Boc-Indoles, Org. Lett., 2006, 8, 2653–2655, DOI:10.1021/ol061039x
.
- Z. N. Yang, F. Chen, Y. He, N. Yang, Q. Fan, Z. Yang, F. Chen, Y. He, N. Yang and Q.-H. Fan, Highly Enantioselective Synthesis of Indolines: Asymmetric Hydrogenation at Ambient Temperature and Pressure with Cationic Ruthenium Diamine Catalysts, Angew. Chem., Int. Ed., 2016, 55, 13863–13866, DOI:10.1002/anie.201607890
.
- T. Touge and T. Arai, Asymmetric Hydrogenation of Unprotected Indoles Catalyzed by η6-Arene/N-Me-sulfonyldiamine–Ru(II) Complexes, J. Am. Chem. Soc., 2016, 138, 11299–11305, DOI:10.1021/jacs.6b06295
.
- A. Baeza and A. Pfaltz, Iridium-Catalyzed Asymmetric Hydrogenation of N-Protected Indoles, Chem. – Eur. J., 2010, 16, 2036–2039, DOI:10.1002/chem.200903105
.
- J. L. Núñez-Rico, H. Fernández-Pérez and A. Vidal-Ferran, Asymmetric hydrogenation of unprotected indoles using iridium complexes derived from P–OP ligands and (reusable) Brønsted acids†, Green Chem., 2014, 16, 1153–1157, 10.1039/C3GC42132E
.
- M. C. Paderes, J. B. Keister and S. R. Chemler, Mechanistic Analysis and Optimization of the Copper-Catalyzed Enantioselective Intramolecular Alkene Aminooxygenation, J. Org. Chem., 2013, 78, 506–515, DOI:10.1021/jo3023632
.
- T. W. Liwosz and S. R. Chemler, Copper-Catalyzed Enantioselective Intramolecular Alkene Amination/Intermolecular Heck-Type Coupling Cascade, J. Am. Chem. Soc., 2012, 134, 2020–2023, DOI:10.1021/ja211272v
.
- M. T. Bovino and S. R. Chemler, Catalytic Enantioselective Alkene Aminohalogenation/Cyclization Involving Atom Transfer, Angew. Chem., Int. Ed., 2012, 51, 3923–3927, DOI:10.1002/anie.201109044
.
- A. Moyano and R. Rios, Asymmetric Organocatalytic Cyclization and Cycloaddition Reactions, Chem. Rev., 2011, 111, 4703–4832, DOI:10.1021/cr100348t
.
- B. Xu, Q. Wang, C. Fang, Z. Zhang and J. Zhang, Recent advances in Pd-catalyzed asymmetric cyclization reactions, Chem. Soc. Rev., 2024, 53, 883–971, 10.1039/D3CS00489A
.
- S. Torres-Oya and M. Zurro, Non-covalent organocatalyzed enantioselective cyclization reactions of α,β-unsaturated imines, Beilstein J. Org. Chem., 2024, 20, 3221–3255, DOI:10.3762/bjoc.20.268
.
- T. Li, F. Tan, L. Lu, Y. Wei, Y. Wang, Y. Liu, Q. Yang, J. Chen, D. Shi and W. Xiao, Asymmetric trapping of zwitterionic intermediates by sulphur ylides in a palladium-catalysed decarboxylation-cycloaddition sequence, Nat. Commun., 2014, 5, 5500, DOI:10.1038/ncomms6500
.
- Q. Wang, T. Li, L. Lu, M. Li, K. Zhang and W. Xiao, Catalytic Asymmetric [4 + 1] Annulation of Sulfur Ylides with Copper–Allenylidene
Intermediates, J. Am. Chem. Soc., 2016, 138, 8360–8363, DOI:10.1021/jacs.6b04414
.
- S. Ma, X. Han, S. Krishnan, S. Virgil and B. Stoltz, Catalytic Enantioselective Stereoablative Alkylation of 3-Halooxindoles: Facile Access to Oxindoles with C3 All-Carbon Quaternary Stereocenters†, Angew. Chem., Int. Ed., 2009, 48, 8037–8041, DOI:10.1002/anie.200902943
.
- X. Lian, A. Adili, B. Liu, Z. Tao and Z. Han, Enantioselective [4 + 1] cycloaddition of ortho-quinone methides and bromomalonates under phase-transfer catalysis, Org. Biomol. Chem., 2017, 15, 3670–3673, 10.1039/C7OB00484B
.
- S. Torres-Oya, M. A. Fernández-Rodríguez and M. Zurro, Synthesis of 3,4-Dihydroquinazolinones via Base-Promoted Formal [4 + 2] Cycloadditions, Adv. Synth. Catal., 2025, 367, e70056, DOI:10.1002/adsc.70056
.
- T. Hua, C. Xiao, Q. Yang and J. Chen, Recent advances in asymmetric synthesis of 2-substituted indoline derivatives, Chin. Chem. Lett., 2020, 31, 311–323, DOI:10.1016/j.cclet.2019.07.015
.
- H. Liao, S. Miñoza, S. Lee and M. Rueping, Aza-Ortho-Quinone Methides as Reactive Intermediates: Generation and Utility in Contemporary Asymmetric Synthesis, Chem. – Eur. J., 2022, 28, e202201112, DOI:10.1002/chem.202201112
.
- M. Zurro and A. Maestro, Asymmetric Catalytic Transformations of Aza-ortho- and Aza-para-Quinone Methides, ChemCatChem, 2023, 15, e202300500, DOI:10.1002/cctc.202300500
.
- D. A. Evans, S. J. Miller, T. Lectka and P. von Matt, Chiral Bis(oxazoline)copper(II) Complexes as Lewis Acid Catalysts for the Enantioselective Diels–Alder Reaction, J. Am. Chem. Soc., 1999, 121, 7559–7573, DOI:10.1021/ja991190k
.
- J. S. Johnson and D. A. Evans, Chiral Bis(oxazoline) Copper(II) Complexes: Versatile Catalysts for Enantioselective Cycloaddition, Aldol, Michael, and Carbonyl Ene Reactions, Acc. Chem. Res., 2000, 33, 325–335, DOI:10.1021/ar960062n
.
-
(a)
CCDC 2478910: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2p6hv8
; (b) CCDC 2478916: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2p6j1h
.
| This journal is © The Royal Society of Chemistry 2026 |