Open Access Article
Open Access ArticleSelective functionalization of the 1,6-anhydro moiety and of the double bond of levoglucosenone
Marco
Rizzo
,
Maria-Jose
Calandri
 ,
Dmitrii
Kurnosov
,
Chiara
Lambruschini
,
Dmitrii
Kurnosov
,
Chiara
Lambruschini
 ,
Francesco
Raboni
,
Renata
Riva
,
Francesco
Raboni
,
Renata
Riva
 and
Luca
Banfi
and
Luca
Banfi
 *
*
Department of Chemistry and Industrial Chemistry, University of Genova, via Dodecaneso, 31, 16146 Genova, Italy. E-mail: luca.banfi@unige.it
First published on 25th February 2026
Abstract
Through a very efficient, step-economical (only 4 steps) and diastereoselective–chemoselective procedure, bio-based levoglucosenone has been converted into a versatile azide, which may be regarded as a synthetic equivalent of a 6-amino-6-deoxysugar. This procedure involves: (a) reduction of the ketone of LGO; (b) opening of the 1,6-anhydro moiety with acetic anhydride and a protic acid to give a triacetate; (c) biocatalytic deacetylation of the primary alcohol; (d) substitution of the alcohol with the azide. The overall yield from LGO is 59%. This azide has been employed in two diversity-generating protocols: Huisgen-Sharpless 1,3-dipolar cycloaddition with alkynes (also derived from bio-based phenols), and the Ugi multicomponent reaction. Furthermore, the double bond has been dihydroxylated with nearly complete diastereoselection both at the azide level or on the triazoles derived from Huisgen cycloadditions, furnishing compounds with the rare D-altro configuration. The chemistry described in this work may help in devising synthetic applications of levoglucosenone, a densely functionalised product of pyrolysis of lignocellulosic matter.
Introduction
Nowadays, oil is the main source of fine chemicals, but it is a non-renewable carbon source, destined to end by 50–100 years. Therefore, renewable biomass is entitled to become the oil of the future.1 However, until now, it has been mainly used for the production of high volume, low value chemicals (polymers, solvents),2 whereas its exploitation for the synthesis of more complex fine chemicals,3 also through a diversity-oriented approach,4 is still relatively underexplored. In this context, during the last years, our research group has studied the use of various bio-based derived components in multicomponent reactions such as the Passerini,5–7 the Ugi,8,9 the Ugi-Joullié,10 and the Hosomi-Sakurai11 reactions.Building blocks from biomass are often very challenging from the point of view of organic synthesis, being densely functionalized. On the other hand, they may offer the advantage to be chiral and enantiomerically pure. One such example is levoglucosenone 1 (LGO)12 (Scheme 1). LGO is the main product of acid catalysed pyrolysis of cellulose containing products, including low value materials such as waste or lignocellulosic matters.13–16 If one consider that 180 billion tons of non-food lignocellulosic biomass are produced annually, the potential of LGO as starting material is very high.
Several transformations (including total syntheses)17 have been accomplished starting from this building block,12,18 and its derivatives have been often employed in the field of drug discovery.19 Anyway, we think that there is still ample room for further investigation. In particular, to our knowledge, only one example of multicomponent reaction has been reported so far on LGO (or better on its saturated derivative).20 Thus, at the outset of this work, we wanted to study new possible applications of MCRs or other diversity generation processes, such as the azide–alkyne cycloaddition (“click” reaction),21,22 employing this bio-based building block.
Results and discussion
As shown in Scheme 1, our initial project involved ring-opening of the bicyclic acetal of 1 with isocyanides or other C- or O-nucleophiles, to give polyfunctionalised building-blocks 6 or 7. Before opening the bicyclic acetal, we chose to convert LGO 1 into the corresponding alcohol 2. Although this process, using Luche's reduction,23 was already described,24,25 in our hands the reported work-up conditions turned out to be not user-friendly. Because of the hydrophilicity of 2, the standard aqueous work-up involved indeed many extractions, leading anyway to a significative loss of product. We eventually found out that the best procedure involves quenching with solid citric acid followed by filtration through Celite, evaporation, and direct chromatography.26 In this way alcohol 2 could be obtained in 90% yield and 94![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6 diastereomeric ratio. The minor diastereomer (slower running in TLC) is slightly separated and thus we were able to obtain 2 in up to 98
6 diastereomeric ratio. The minor diastereomer (slower running in TLC) is slightly separated and thus we were able to obtain 2 in up to 98![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 dr by chromatography. This alcohol was protected to give known compounds 3,274,27 and 5.26
2 dr by chromatography. This alcohol was protected to give known compounds 3,274,27 and 5.26
On these protected compounds, we investigated the possible reaction with isocyanides, under the catalysis of Lewis acids or TMS-OTf (trimethylsilyl trifluoromethanesulphonate).
Although very few examples of reactions of isocyanides with acetals are reported in the literature,28–30 we hoped to be able to open the bicyclic system introducing a new C–C bond in order to obtain interesting C-glycosides containing a secondary amide function. A search in the literature revealed very few examples regarding opening of 2 or its protected derivatives with O-nucleophiles,31–33 and none with C-nucleophiles.
However, as better described in the SI, despite several attempts, we never succeeded in introducing isocyanides as nucleophiles onto protected derivatives 3–5. During these attempts, we reasoned that this failure might be due to reversible additions of the isocyanides and that trapping the liberated primary alcohol in 6 could be beneficial. Hoping to be able to obtain adducts 7, we thus added Ac2O to the reaction mixture. To our surprise, starting from 4, and using TMS-OTf or Lewis acids as catalysts, we isolated, with excellent diastereoselectivity (>10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), diacetate 9. The same result was obtained with protic acids. This was somehow unexpected, because, in standard Passerini reactions, it is always the isocyanide to act as the nucleophile, and not the carboxylate anion.
1), diacetate 9. The same result was obtained with protic acids. This was somehow unexpected, because, in standard Passerini reactions, it is always the isocyanide to act as the nucleophile, and not the carboxylate anion.
Scheme 2 show possible mechanisms of this opening reaction with Ac2O. For the sake of clarity, protic catalysis is shown, but similar mechanisms can be envisaged using Lewis acids or TMSOTf. In mechanism A, the acid promotes opening of the ring to give oxocarbenium ion 11, which may be attacked by a nucleophile. This mechanism does not seem very likely to us. First of all, we do not understand why 11 should not be attacked by the isocyanide, but by the carboxylate anion instead. Then, addition of acetate anion does not improve the process. Free alcohol 12 was never detected. Finally, according to this mechanism, the reaction should be poorly stereoselective or even give the other stereoisomer, as in other reported acid-catalysed opening of LGO with O-nucleophiles.33
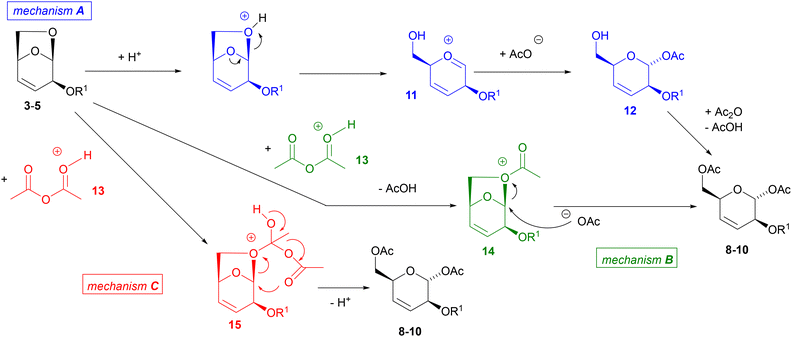 | ||
| Scheme 2 Possible mechanisms for conversion of 3–5 into 8–10. Similar mechanisms may be envisaged using a Lewis acid or TMSOTf. | ||
Other possible mechanisms are B or C, where acylation of the bridge oxygen precedes the opening of the ring. In mechanism B, an intermolecular substitution by the acetoxy anion either through a SN1 (via oxocarbenium ion) or SN2 process should then take place. Once again, it is not clear why other added nucleophiles, such as isocyanides, do not participate.
Thus, we think (although we do not have definitive proofs about that) that the reaction may proceed through the concerted mechanism C, with simultaneous entrance of both acetoxy groups starting from cation 15. This mechanism, involving a 6-membered cyclic T.S., may also explain the high diastereoselectivity observed, and the preferred diastereomer. Reactions passing through an oxocarbenium ion should be less stereoselective (or even favour the other isomer),33 and d.r.s should strongly depend on the nature of protecting group (because of anchimeric assistance), whereas in our case we did not observe significant differences.
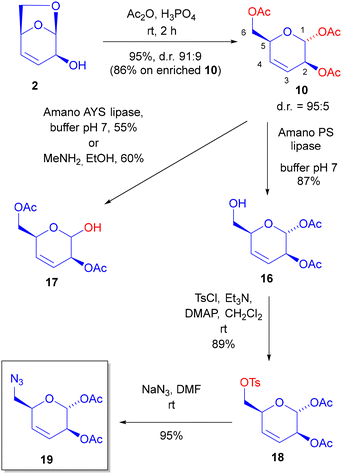
Apart from the mechanistic rationalisations, we reasoned that diacetates 8–9 or triacetate 10 could be useful building blocks for diversity-oriented processes using glycomimetic fragments. This opening reaction was thus optimized (see SI). To make a long story short, we eventually found that: (a) protic acids, especially phosphoric acid, behave better than Lewis acids or TMSOTf, being also more sustainable; (b) previous protection of the allylic alcohol was not really necessary, and the reaction could be carried out directly on alcohol 2, affording triacetate 10 in just 2 steps from LGO, with nearly complete stereocontrol for the stereocenter at position 1 (sugar numbering).
Although in this way we obtained a triacetate, we were confident to be able to differentiate the three groups by chemoenzymatic means. This was indeed the case. After screening various lipases, we found out that Amano P lipase was highly chemoselective, allowing hydrolysis of the primary acetate in 10 to give 16 (Scheme 3). Other enzymes (see SI) were less selective, whereas Amano AYS lipase preferred to hydrolyse the anomeric acetate to give 17. Selective hydrolysis of the latter can be also achieved chemically, using MeNH2 in EtOH.
Thus alcohol 16 could be obtained in just three steps from LGO 1, in overall 62% yield, and with an operationally simple and “green” procedure.
In order to get a nitrogen containing building block, we then converted 16 into azide 19, through the tosylate 18, by a classical SN2 substitution.
A comment on diastereomeric purity of alcohol 16, tosylate 18 and azide 19 is worthy. We started from nearly pure (dr: 98![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) alcohol 2 and performed diastereoselective opening to 10 (91
2) alcohol 2 and performed diastereoselective opening to 10 (91![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9) and finally chemoselective enzymatic hydrolysis to 16. These two last steps are highly, but not completely, selective and thus small amounts of isomers were present. These could be eliminated by careful chromatographies at the level of 10, 16 and 19. However, tosylate 18 is a white solid, and we found out that, working on a larger scale, it is also possible to be less accurate in chromatography of intermediates, since a single trituration at the level of 18 can afford a very pure compound.
9) and finally chemoselective enzymatic hydrolysis to 16. These two last steps are highly, but not completely, selective and thus small amounts of isomers were present. These could be eliminated by careful chromatographies at the level of 10, 16 and 19. However, tosylate 18 is a white solid, and we found out that, working on a larger scale, it is also possible to be less accurate in chromatography of intermediates, since a single trituration at the level of 18 can afford a very pure compound.
Azide 19 may be considered a synthetic equivalent of a 6-aminosugar, taking into account the possibility to convert the double bond into a diol, as described below. Although quite rare, 6-aminosugars are endowed with interesting biological activities.34–36
As a first application we employed azide 19 in a series of 1,3-dipolar cycloadditions with alkynes (“click” reactions).21 The alkynes used are shown in Scheme 4, whereas the results are listed in Table 1. As already previously observed by us in another work,37 when using very small amounts of catalysts (0.01 equivalents of CuSO4 and 0.1 equivalents of Na ascorbate) (see Table 1), these 1,3-dipolar cycloadditions tended to stop, probably because of inactivation of Cu(I). Therefore, initially we used to add a second or even a third portion of both catalysts when the reaction appeared to stop.37 In the last experiments, we preferred to add a higher catalyst quantity (especially ascorbate) from the beginning in order to have more complete reactions.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a
a
| Alkyne | Triazole | Cat. amount (ascorbate/CuSO4) | Reaction timeb | Yieldc (%) |
|---|---|---|---|---|
a Conditions: azide 19 and alkyne 20 (1.2 equiv.) were treated, in t-BuOH/H2O 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, at rt, with the indicated equiv. of Na ascorbate and of CuSO4. The same amount of catalysts was added after the first time indicated in column 4. In some cases a third addition of catalysts was necessary.
b For example (line 1), 4 h + 1 h means 4 h, followed by a second addition of catalyst and then further reaction for 1 h.
c Isolated yields. 1, at rt, with the indicated equiv. of Na ascorbate and of CuSO4. The same amount of catalysts was added after the first time indicated in column 4. In some cases a third addition of catalysts was necessary.
b For example (line 1), 4 h + 1 h means 4 h, followed by a second addition of catalyst and then further reaction for 1 h.
c Isolated yields.
|
||||
| 20a | 21a | 0.1/0.01 equiv. × 2 | 4 h + 1 h | 65 |
| 20b | 21b | 0.1/0.01 equiv. × 2 | 3 h + 2 h | 66 |
| 20c | 21c | 0.1/0.01 equiv. × 2 | 2 h + 1 h | 90 |
| 20d | 21d | 0.1/0.01 equiv. × 3 | 2 h + 2 h + 1 h | 84 |
| 20e | 21e | 0.5/0.2 equiv. | 4 h | 93 |
In these click reactions, we have also used some alkynes containing the catechol structure typical of natural polyphenols.
In order to gain access to hybrid peptidomimetics–glycomimetics, we also decided to employ azide 19 in Ugi multicomponent reactions.38,39 Our idea was to use azide 19 as a synthetic equivalent of a primary amine.
In the literature there are some examples40–42 of one-pot Staudinger-aza-Wittig-Ugi sequence, where an azidoaldehyde is first reduced by a phosphine (Staudinger reduction) to form a phosphazene, followed by an intramolecular aza-Wittig reaction affording a cyclic imine. Finally, the latter is the substrate of a 3-component Ugi-Joullié reaction.
However, this protocol was always applied to azidoaldehydes, in an intramolecular way. We tried to transfer the same approach to an intermolecular process, thus applying a true 4-component Ugi reaction. We were pleased to find out that this was indeed possible, by treating azide 19 with PPh3 in the presence of an aldehyde, followed by addition of an isocyanide and of a carboxylic acid. A series of Ugi products, shown in Scheme 5, were obtained. Unfortunately, diastereoselectivity was very low, as usual for this MCR, giving nearly 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of the possible diastereomers.
1 mixture of the possible diastereomers.
When using an aliphatic aldehyde (cyclohexanecarboxaldehyde to give 23d) we surprisingly observed at the end of the process, the presence of starting azide 19, notwithstanding the fact that PPh3 was used in excess. We reasoned that the aldehyde could somehow inhibit the reduction of the azide. Therefore, in this case, we repeated the reaction treating the azide with PPh3 before addition of the aldehyde and the yield was improved from 40% to 55%.
In all the products shown in Schemes 4 and 5, the double bond of starting levoglucosenone is still present. This double bond can be seen as a synthetic equivalent of a diol. In order to prove this synthetic equivalence, we studied dihydroxylation of azide 19 using catalytic OsO4 (3%) in the presence of stoichiometric N-methylmorpholine-N-oxide (NMO) (Scheme 6). With our surprise, this dihydroxylation was found to be highly diastereoselective and we could isolate diol 24 as a single diastereomer after chromatography. Careful examination of the crude and of some head fractions of chromatography allowed to detect two possible isomers, in quantities around 5% and 3%, relative to the major isomer. Osmium catalysed hydroxylation is known to be stereospecific and thus we were a little surprised by the presence of two isomers. Due to the very low amount of them it was not possible to understand their structure.
Anyway, the relative stereoinduction of the two cis diols is higher than 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, the yield is high and 24 can be easily isolated in pure form by chromatography. The relative configuration of this major product was easily assessed by examination of the coupling constants in 1H NMR, which were compatible only with the 4C1 conformation of the altro diastereomer, shown in Scheme 6 (sugar numbering). In particular, there is a J4–5 equal to 9.7 Hz, indicating that H-4 and H-5 are both axial and trans to each other. On the other hand, J3–4 and J2–3, respectively 3.5 and 3.3 Hertz are in agreement with a cis (3–4) or a trans diequatorial (2–3) relationship. Neither conformation of the talo isomer fit with these coupling constants, because H-4 and H-5 would be cis and thus J4–5 would be around 3 Hz. The preference for the 4C1 conformation may be ascribed to the α effect. Although highly diastereoselective dihydroxylation of levoglucosenone from the bottom face is known,24,43 in that case the bridge causes a much higher shield of the upper face than in the case of 19 and thus this very high diastereoselectivity was unexpected to us.
1, the yield is high and 24 can be easily isolated in pure form by chromatography. The relative configuration of this major product was easily assessed by examination of the coupling constants in 1H NMR, which were compatible only with the 4C1 conformation of the altro diastereomer, shown in Scheme 6 (sugar numbering). In particular, there is a J4–5 equal to 9.7 Hz, indicating that H-4 and H-5 are both axial and trans to each other. On the other hand, J3–4 and J2–3, respectively 3.5 and 3.3 Hertz are in agreement with a cis (3–4) or a trans diequatorial (2–3) relationship. Neither conformation of the talo isomer fit with these coupling constants, because H-4 and H-5 would be cis and thus J4–5 would be around 3 Hz. The preference for the 4C1 conformation may be ascribed to the α effect. Although highly diastereoselective dihydroxylation of levoglucosenone from the bottom face is known,24,43 in that case the bridge causes a much higher shield of the upper face than in the case of 19 and thus this very high diastereoselectivity was unexpected to us.
Compound 24 has the relative and absolute configuration of α-D-altropyranose, a very rare sugar. Therefore, this building block may be very useful for the “click” assembly of glycomimetics having this unusual relative configuration.
Hydroxylated azide 24 was used in 1,3-dipolar cycloaddition with alkynes 20c and 20e to give triazoles 25, 26, which were isolated as pure diastereomers. Alternatively, triazoles 21c and 21e may be dihydroxylated, again with high diastereoselection, to afford the same altro derivatives 25, 26. In the case of 21c, we obtained a dr of 98![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, whereas, with 21e, stereoinduction was slightly lower (93
2, whereas, with 21e, stereoinduction was slightly lower (93![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7). In these cases, a third isomer was again present, but in very little amount (<1%).
7). In these cases, a third isomer was again present, but in very little amount (<1%).
Conclusions
In conclusion, we have developed an efficient transformation of levoglucosenone into azide 19, which may be considered an useful building block for diversity-oriented synthesis of complex molecule containing a fragment corresponding to a 6-aminosugar. Azide 19 was synthesized thanks to a highly diastereoselective opening of the bridge with acetic anhydride and protic acids, followed by an enzymatic differentiation of the hydroxy group at carbon 6.The suitability of azide 19 in diversity-oriented synthesis was demonstrated by a series of click reactions or of Ugi reactions starting from it.
We also demonstrated the synthetic equivalence of the double bond of levoglucosenone with a diol, by an OsO4 catalysed dihydroxylation.
The use of azide 19 in Ugi and click reactions demonstrates its synthetic equivalence with rare 6-deoxy-6-aminosugars.34–36 There are indeed very few examples of this kind of sugars in the literature, and none with the particular D-altro configuration that can be achieved by osmylation. While 1-aminosugars have been previously used as component in the Ugi reactions,44 to our knowledge no example of use of 6-aminosugars in MCRs is known.
From the stereochemical point of view, it is worth noting that a series of 3 highly diastereoselective transformations has allowed to convert LGO (only 2 chirality centers) into stereopure compounds 24–26 (5 chirality centers).
Author contributions
Conceptualization: LB, RR. Funding acquisition: LB, RR. Resources: CL. Data analysis: CL, LB, RR. Investigation: MR, MJC, FR, DK. Supervision: CL, LB. Writing of draft: LB. Writing: editing and review: CL, RR, DK.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: procedures, optimizations, characterization data, NMR spectra, and discussion on the assessment of relative configuration. See DOI: https://doi.org/10.1039/d6ob00050a.Additional references were quoted within the SI.45
Acknowledgements
We acknowledge financial support under the National Recovery and Resilience Plan (NRRP), Mission 4, Component 2, Investment 1.1, Call for tender No. 104 published on 2.2.2022 by the Italian Ministry of University and Research (MUR), funded by the European Union – NextGenerationEU – Project Title SUSTCARB – CUP D53D23010360006, Grant Assignment Decree No. 1064 adopted on July 18, 2023, by the Italian Ministry of University and Research (MUR).We thank Compagnia San Paolo for financial support (Trapezio initiative).
We thank Amano for kind gift of its enzymes.
References
- N. Brun, P. Hesemann and D. Esposito, Expanding the biomass derived chemical space, Chem. Sci., 2017, 8, 4724–4738 RSC.
- F. H. Isikgor and C. R. Becer, Lignocellulosic biomass: a sustainable platform for the production of bio-based chemicals and polymers, Polym. Chem., 2015, 6, 4497–4559 RSC.
- T. Dekker, J. W. Harteveld, G. Wágner, M. C. M. de Vries, H. Custers, A. C. van de Stolpe, I. J. P. de Esch and M. Wijtmans, Green Drug Discovery: Novel Fragment Space from the Biomass-Derived Molecule Dihydrolevoglucosenone (Cyrene™), Molecules, 2023, 28, 1777 CrossRef CAS PubMed.
- L. Banfi, C. Lambruschini, L. Moni and R. Riva, in RSC Green Chemistry, ed. R. Ballini, Royal Society of Chemistry, 2019, vol. 2019-January, pp. 115–140 Search PubMed.
- G. Vitali Forconesi, A. Basso, L. Banfi, D. Gugliotta, C. Lambruschini, M. Nola, R. Riva, V. Rocca and L. Moni, Total Synthesis of 4-epi-Bengamide E, Molecules, 2024, 29, 1715 CrossRef PubMed.
- L. Moni, L. Banfi, D. Cartagenova, A. Cavalli, C. Lambruschini, E. Martino, R. V. A. Orru, E. Ruijter, J. M. Saya, J. Sgrignani and R. Riva, Zinc(II)-mediated diastereoselective Passerini reactions of biocatalytically desymmetrised renewable inputs, Org. Chem. Front., 2020, 7, 380–398 RSC.
- G. Vitali Forconesi, L. Banfi, A. Basso, C. Lambruschini, L. Moni and R. Riva, Synthesis of polyoxygenated heterocycles by diastereoselective functionalization of a bio-based chiral aldehyde exploiting the Passerini reaction, Molecules, 2020, 25, 3227 CrossRef PubMed.
- D. Galante, L. Banfi, G. Baruzzo, A. Basso, C. D’Arrigo, D. Lunaccio, L. Moni, R. Riva and C. Lambruschini, Multicomponent synthesis of polyphenols and their in vitro evaluation as potential β-amyloid aggregation inhibitors, Molecules, 2019, 24, 2636 CrossRef PubMed.
- C. Lambruschini, A. Basso, L. Moni, A. Pinna, R. Riva and L. Banfi, Bicyclic Heterocycles from Levulinic Acid through a Fast and Operationally Simple Diversity-Oriented Multicomponent Approach, Eur. J. Org. Chem., 2018, 5445–5455 CrossRef CAS.
- V. Cerulli, L. Banfi, A. Basso, V. Rocca and R. Riva, Diversity oriented and chemoenzymatic synthesis of densely functionalized pyrrolidines through a highly diastereoselective Ugi multicomponent reaction, Org. Biomol. Chem., 2012, 10, 1255–1274 RSC.
- C. Lambruschini, A. Barbero, A. Cherubin, L. Moni, L. Palio, R. Riva and L. Banfi, Multicomponent Hosomi-Sakurai reaction on chiral, bio-based, alcohols, RSC Adv., 2025, 15, 17897–17905 RSC.
- Z. J. Witczak, in Levoglucosenone and Levoglucosans, Chemistry and Applications: Proceedings of the Symposium on Levoglucosenone and Levoglucosans, Sponsored by the Division of Carbohydrate Chemistry at the 204th National Meeting of the American Chemical Society, Washington, D.C., 1992.
- S. Y. Feng, S. Li, W. Z. Huang, Q. Niu, D. Y. Chen, Z. M. Fang, L. J. Gao, K. Li and Q. Lu, Alkali pretreatment-assisted catalytic pyrolysis of waste cellulose acetate for selective preparation of levoglucosenone, J. Environ. Chem. Eng., 2024, 12, 113858 CrossRef CAS.
- A. M. Gonzalez-Rosario, O. Oyola-Rivera and N. Cardona-Martínez, Production of Levoglucosan and Levoglucosenone from Cellulose Using Brønsted Acid Catalysts in Polar Aprotic Solvents, Waste Biomass Valorization, 2024, 16, 787–803, DOI:10.1007/s12649-024-02708-9.
- H. Yang, S. Lei, K. Xu, Y. Fang, X. Chen, Y. Chen, X. Wang and H. Chen, Catalytic pyrolysis of cellulose with sulfonated carbon catalyst to produce levoglucosenone, Fuel Process. Technol., 2022, 234, 107323 CrossRef CAS.
- J. Klepp, W. Dillon, Y. Lin, P. Feng and B. W. Greatrex, Org. Synth., 2020, 97, 38–53 CrossRef CAS.
- M. B. Comba, Y. H. Tsai, A. M. Sarotti, M. I. Mangione, A. G. Suárez and R. A. Spanevello, Levoglucosenone and Its New Applications: Valorization of Cellulose Residues, Eur. J. Org. Chem., 2018, 590–604, DOI:10.1002/ejoc.201701227.
- Z. J. Witczak and K. Tatsuta, Carbohydrates Synthons in Natural Products Synthesis, Oxford University Press, Washington, 2002 Search PubMed.
- J. E. Camp and B. W. Greatrex, Levoglucosenone: Bio-Based Platform for Drug Discovery, Front. Chem., 2022, 10, 902239 CrossRef CAS PubMed.
- L. A. Martinho, T. P. F. Rosalba, G. G. Sousa, C. C. Gatto, J. R. S. Politi and C. K. Z. Andrade, Cyrene: a very reactive bio-based chiral ketone in diastereoselective Passerini reactions, Mol. Diversity, 2024, 28, 111–123 CrossRef CAS PubMed.
- P. Wu, A. K. Feldman, A. K. Nugent, C. J. Hawker, A. Scheel, B. Voit, J. Pyun, J. M. J. Fréchet, K. B. Sharpless and V. V. Fokin, Efficiency and Fidelity in a Click-Chemistry Route to Triazole Dendrimers by the Copper(I)-Catalyzed Ligation of Azides and Alkynes, Angew. Chem., Int. Ed., 2004, 43, 3928–3932 CrossRef CAS PubMed.
- Y. H. Tsai, C. M. B. Etichetti, C. Di Benedetto, J. E. Girardini, F. T. Martins, R. A. Spanevello, A. G. Suárez and A. M. Sarotti, Synthesis of Triazole Derivatives of Levoglucosenone As Promising Anticancer Agents: Effective Exploration of the Chemical Space through retro-aza-Michael//aza-Michael Isomerizations, J. Org. Chem., 2018, 83, 3516–3528 CrossRef CAS PubMed.
- J. L. Luche, Lanthanides in organic chemistry. 1. Selective 1,2 reductions of conjugated ketones, J. Am. Chem. Soc., 1978, 100, 2226–2227 CrossRef CAS.
- E. D. V. Giordano, A. Frinchaboy, A. G. Suárez and R. A. Spanevello, Synthesis of Tri-O-acetyl-D-allal from Levoglucosenone, Org. Lett., 2012, 14, 4602–4605 CrossRef CAS PubMed.
- B. T. Sharipov, A. N. Davidova, A. S. Ryabova, N. F. Galimzyanova and F. A. Valeev, Synthesis and fungicidal activity of methylsulfanylmethyl ether derivatives of levoglucosenone, Chem. Heterocycl. Compd., 2019, 55, 31–37 CrossRef CAS.
- X. H. Ma, X. Liu, P. Yates, W. Raverty, M. G. Banwell, C. X. Ma, A. C. Willis and P. D. Carr, Manipulating the enone moiety of levoglucosenone: 1,3-Transposition reactions including ones leading to isolevoglucosenone, Tetrahedron, 2018, 74, 5000–5011 CrossRef CAS.
- K. Matsumoto, T. Ebata and H. Matsushita, Novel Synthesis of 3-Acetamido-3-Deoxy-D-Altrose and 4-Acetamido-4-Deoxy-D-Altrose from Levoglucosenone Using Regioselective Cis-Oxyamination, Carbohydr. Res., 1995, 267, 187–201 CrossRef CAS.
- M. Tobisu, A. Kitajima, S. Yoshioka, I. Hyodo, M. Oshita and N. Chatani, Brønsted Acid Catalyzed Formal Insertion of Isocyanides into a C–O Bond of Acetals, J. Am. Chem. Soc., 2007, 129, 11431–11437 CrossRef CAS PubMed.
- L. Y. Lyu, M. Y. Jin, Q. J. He, H. Xie, Z. X. Bian and J. Wang, Bi(OTf)3-catalyzed addition of isocyanides to 2H-chromene acetals: an efficient pathway for accessing 2-carboxamide-2H-chromenes, Org. Biomol. Chem., 2016, 14, 8088–8091 RSC.
- N. E. Golantsov, H. M. Nguyen, A. V. Varlamov, A. V. Aksenov and L. G. Voskressensky, Three-component reaction of ketals, isonitriles, and trimethylsilyl azide, Chem. Heterocycl. Compd., 2017, 53, 446–450 CrossRef CAS.
- M. E. Jung and M. Kiankarimi, Synthesis of methylene-expanded 2′,3′-dideoxyribonucleosides, J. Org. Chem., 1998, 63, 8133–8144 CrossRef CAS.
- M. Ostermeier and R. Schobert, Total Synthesis of (+)-Chloriolide, J. Org. Chem., 2014, 79, 4038–4042 CrossRef CAS PubMed.
- F. A. Valeev, I. N. Gaisina, L. V. Spirikhin and M. S. Miftakhov, Reactions of 1,6-anhydro-3,4-dideoxy-2-O-methyl-beta-D-threo-hex-3-enopyranose with thiols and methanol, Russ. Chem. Bull., 1997, 46, 528–531 CrossRef CAS.
- D. Lafont, A. Wollny and P. Boullanger, Synthesis of 6-amino-1,6-anhydro-6-deoxysugar derivatives, Carbohydr. Res., 1998, 310, 9–16 CrossRef CAS.
- F. Kudo, Y. Kitayama, A. Miyanaga, M. Numakura and T. Eguchi, Stepwise Post-glycosylation Modification of Sugar Moieties in Kanamycin Biosynthesis, ChemBioChem, 2021, 22, 1668–1675 CrossRef CAS PubMed.
- T. Li, X. Gao, L. Yang, Y. Shi and Q. Gao, Methyl 6-Amino-6-deoxy-d-pyranoside-Conjugated Platinum(II) Complexes for Glucose Transporter (GLUT)-Mediated Tumor Targeting: Synthesis, Cytotoxicity, and Cellular Uptake Mechanism, ChemMedChem, 2016, 11, 1069–1077 CrossRef CAS PubMed.
- E. Tassano, A. Alama, A. Basso, G. Dondo, A. Galatini, R. Riva and L. Banfi, Conjugation of Hydroxytyrosol with Other Natural Phenolic Fragments: From Waste to Antioxidants and Antitumour Compounds, Eur. J. Org. Chem., 2015, 6710–6726 CrossRef CAS.
- M. A. Fouad, H. Abdel-Hamid and M. S. Ayoup, Two decades of recent advances of Ugi reactions: synthetic and pharmaceutical applications, RSC Adv., 2020, 10, 42644–42681 RSC.
- E. Fotopoulou, P. K. Anastasiou, C. Tomza and C. G. Neochoritis, The Ugi reaction as the green alternative towards active pharmaceutical ingredients, Tetrahedron Green Chem, 2024, 3, 100044 CrossRef CAS.
- T. Wennekes, K. M. Bonger, K. Vogel, R. J. B. H. N. van den Berg, A. Strijland, W. E. Donker-Koopman, J. M. F. G. Aerts, G. A. van der Marel and H. S. Overkleeft, The Development of an Aza-C-Glycoside Library Based on a Tandem Staudinger/Aza-Wittig/Ugi Three-Component Reaction, Eur. J. Org. Chem., 2012, 6420–6454, DOI:10.1002/ejoc.201200923.
- L. Moni, L. Banfi, A. Basso, A. Galatini, M. Spallarossa and R. Riva, Enantio- and diastereoselective synthesis of highly substituted benzazepines by a multicomponent strategy coupled with organocatalytic and enzymatic procedures, J. Org. Chem., 2014, 79, 339–351 CrossRef CAS PubMed.
- L. Banfi, A. Basso, G. Guanti and R. Riva, Enantio- and diastereoselective synthesis of 2,5-disubstituted pyrrolidines through a multicomponent Ugi reaction and their transformation into bicyclic scaffolds, Tetrahedron Lett., 2004, 45, 6637–6640 CrossRef CAS.
- K. Matsumoto, T. Ebata, K. Koseki, H. Kawakami and H. Matsushita, Synthesis of D-altrose via D-altrosan from levoglucosenone, Bull. Chem. Soc. Jpn., 1991, 64, 2309–2310 CrossRef CAS.
- O. Lockhoff, An Access to Glycoconjugate Libraries through Multicomponent Reactions, Angew. Chem., Int. Ed., 1998, 37, 3436–3439 CrossRef CAS PubMed.
- F. A. Valeev, L. K. Kalimullina, S. M. Salikhov, O. V. Shitikova, I. P. Tsypysheva and M. G. Safarov, Synthesis of 2-amino derivatives of levoglucosenone, Chem. Nat. Compd., 2004, 40, 521–525 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |