Open Access Article
Open Access ArticleDesign of multivalent pharmacological chaperones against Pompe disease via metal-free ligation
Marc
Borie-Guichot
a,
Abdelouahd
Oukhrib
cd,
My Lan
Tran
a,
Cédric-Olivier
Turrin
cd,
Yves
Génisson
 a,
Laure
Gibot
b,
Stéphanie
Ballereau
a,
Laure
Gibot
b,
Stéphanie
Ballereau
 a and
Cécile
Dehoux
a and
Cécile
Dehoux
 *a
*a
aUniversité de Toulouse, CNRS, SPCMIB, UMR5068, 118 Route de Narbonne, F-31062 Toulouse, France. E-mail: cecile.dehoux@utoulouse.fr
bLaboratoire Softmat, Université de Toulouse, CNRS UMR 5623, Université Toulouse III - Paul Sabatier, 31062 Toulouse, France
cIMD-Pharma, 205 Route de Narbonne, 31077 Toulouse Cedex 4, France
dLaboratoire de Chimie de Coordination du CNRS, 205 Route de Narbonne, BP 44099, 31077 Toulouse CEDEX 4, France
First published on 25th February 2026
Abstract
We report herein the design of multivalent pharmacological chaperones (PCs), based on D- and L-deoxynojirimycin (DNJ), to treat Pompe disease (PD), a lysosomal storage disorder caused by mutations in the acid alpha-glucosidase (GAA) enzyme. Multivalent DNJ platforms were built on a pentaerythritol core by Copper Azide–Alkyne Click (CuAAC) ligation. Moreover, new phosphorus-containing dendrimers were decorated with DNJ by Strain Promoted Azide–Alkyne Click (SPAAC) reaction. Our main objective was to systematically study how the chirality and the hydrophobicity of these constructs could modulate their ability to inhibit, stabilize, and restore GAA activity. Thermal stabilization assays identified several highly effective stabilizers. Most importantly, the phosphorus-containing dendrimer 15b, an L-N-undecanyl-DNJ derivative, was found to enhance GAA activity by 1.2 fold in fibroblasts from a Pompe patient at the dose of only 0.5 nM. This finding is a significant breakthrough as it represents the first report of a multivalent compound increasing GAA activity in patient cells. This novel multivalent PC, which is a very weak GAA inhibitor, offers a promising avenue for the development of effective treatments for Pompe disease.
Introduction
Lysosomal storage diseases (LSDs) are a group of metabolic disorders mostly caused by mutations of lysosomal hydrolases. The dysfunction of these proteins induces a lysosomal burden of the non-hydrolysed substrates.1,2 Enzyme replacement therapy (ERT), which is the main therapeutic response, consists in biweekly infusions of recombinant lysosomal glycosidase to empty the lysosomal overload. Its main limitations are its ineffectiveness in treating neurological forms, its high cost and the associated immune responses. Pharmacological chaperone (PC) therapy, introduced in the early 2000s, consists in the use of protein ligands that stabilize the correct conformation of the misfolded protein by selective binding. In doing so, PCs escort the mutant protein to the lysosome where it may express its residual level of function.3 This strategy historically took advantage of the available portfolio of high affinity competitive enzyme inhibitors. These active-site specific chaperones paradoxically allow an increase in hydrolase activity in patient cells whereas they were originally designed to inhibit their wild-type counterpart. To date, the only marketed PC is 1-deoxygalactonojirimycin (Galafold™, migalastat), which is a competitive inhibitor of α-galactosidase A used against Fabry disease.4 However, the extent of proteins inhibition by active-site specific PCs is not directly correlated to their level of chaperoning activity. It therefore remains clinically difficult to control the balance between these two opposite effects. Non-inhibitory PCs, typically binding to an allosteric site, present the advantage of potentially activating the misfolded protein without competing with the endogenous substrate.5 Moreover, PCs could be utilized in a combined PC-ERT therapy to reduce patient hospitalization, lower costs, and minimize the side effects associated with ERT.6In the search of potent glycosidase inhibitors, the concept of multivalency has been widely explored for a diversity of enzymes.7 Over the past decade, many multivalent iminosugars have demonstrated remarkable multivalent effects toward certain glycosidases.8,9 This highlights that the principle of multivalency can also be applied to enzymes with a single active site, broadening its potential for innovative therapeutic strategies. Among the reported multivalent glycosidase inhibitors, some addressed lysosomal enzymes.9,10 In 2012, a breakthrough was brought by Compain and Coll. with the first report of multivalent iminosugars targeting glucocerebrosidase (GCase, E.C.3.2.1.45), the lysosomal enzyme implied in the Gaucher disease, the highest prevalent LSD.11 An heptavalent deoxynojirimycin (DNJ) built on a cyclodextrin scaffold showed a high multivalent inhibitory effect (rp/n = 28, rp = relative potency and n = valency) towards GCase. For this glycosidase, several mechanisms may contribute to the multivalent effect,9 including clustering or cross-linking effects—since previous studies have suggested that GCase exists as a dimeric or tetrameric structure12—as well as subsite-binding effects. Moreover, Compain and Coll. demonstrated for the first time that this multivalent inhibitor also improved the enzyme activity in N370S human fibroblasts (1.5-fold increase at 10 µM). Thus, multivalency was not only valuable for developing glycosidase inhibitors but was also relevant for accessing pharmacological chaperones. Several examples of multivalent PCs of GCase have latter on been reported by Compain et al.13,14 and other research groups.15–20 To our knowledge, multivalent PCs have been described for only two other lysosomal enzymes: alpha-galactosidase19 and alpha-mannosidase,21 which are involved in Fabry disease and alpha-mannosidosis, respectively. Recently, we described original poly(phosphorhydrazone) dendrimers bearing monofluorocyclooctyne units on their periphery.15,16 Thanks to Strain Promoted Azide–Alkyne Click (SPAAC) reaction, we synthesized multivalent DNJs15 and multivalent hydroxypyrrolidines.16 Most of these constructs were found to act as potent GCase inhibitors and were able to increase the GCase activity of fibroblasts from Gaucher patients with N370S mutation. During this work, we noticed with interest that the synthesized multivalent DNJs were better inhibitors of acid alpha-glucosidase (GAA, EC 3.2.1.20) than of GCase. In particular, multivalent compounds 1 and 2 displayed sub-micromolar inhibition of GAA (Fig. 1).15 This finding caught our attention since GAA is the enzyme implied in Pompe disease (PD).
 | ||
| Fig. 1 D-DNJ based iminosugars reported by us in previous work.15 | ||
PD, also known as glycogen storage disease type II, is a LSD characterized by mutations in the gene encoding GAA. It results in an abnormal glycogen accumulation in the lysosomes of the heart, muscles, and other cells.22 PD displays a broad phenotypic spectrum ranging from severe infantile-onset to milder later-onset forms. Most PD patients suffer however from progressive muscle hypotonia and respiratory failure. To date, ERT is the first and only treatment approved (Alglucosidase marketed as Lumizyme in U.S.A. and Myozyme elsewhere, as well as Avalglucosidase Alfa, Cipaglucosidase Alfa) and is administered regularly in one- to two-week intervals for life to prevent the accumulation of glycogen. Several active site-specific PCs of GAA have been described,23 most often belonging to sugar24–26 or iminosugar families, such as DNJ27–29 or N-Butyl DNJ (NB-DNJ).30 In 2023, a combination therapy (Pombiliti), consisting in Cipaglucosidase Alfa plus the NB-DNJ PC, was approved by FDA to treat certain adults with late-onset Pompe disease. In this case, PC allowed the stabilization of the recombinant enzyme at neutral pH in circulating blood which led to an enhancement of the GAA activity in the patients lysosomes.31 Non-inhibitory PCs have also been reported,23 such as N-acetylcysteine and L-NB-DNJ.32,33 Allosteric binding sites of NAC have been identified by a co-crystal structure,34 whereas mechanism of action of L-NB-DNJ has not yet been identified.33 Regarding multivalent compounds, only Compain and C. have evaluated certain of their DNJ-cyclodextrin conjugates for GAA inhibition and observed a weak multivalent effect.11,15 To date, no multivalent PC of GAA has been identified.
Prompted by the preliminary results obtained with 1 and 2, we conducted an in-depth study of the effect of multivalent DNJs on GAA inhibition, enzyme thermal stabilization, and restoration of cellular enzyme activity. For this purpose, we relied on two multivalent scaffolds: pentaerythritol-based dendrimers and a new family of phosphorus-containing dendrimers in which the clickable monofluorocyclooctyne unit is replaced by an azadibenzocyclooctyne (ADIBO), known to have a greater reactivity toward SPAAC chemistry (compounds 3-G0 and 4-G1, Fig. 2).35L or DN-substituted DNJs were grafted on the surface of these dendrimers, either through an aliphatic or a pegylated spacer, in order to study the influence of hydrophobicity and chirality.
Results and discussion
Chemical synthesis
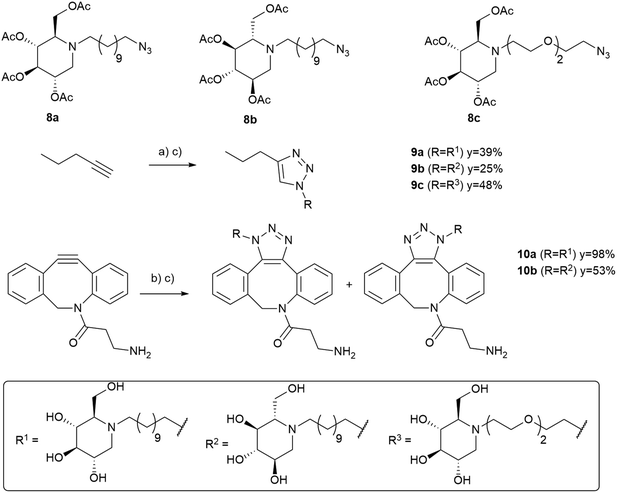
Stirring of DNJ derivatives 8a and 8b with commercial azadibenzocyclooctyne amine in a methanol/THF mixture at 45 °C followed by ammonia deprotection give SPAAC products 10a and 10b as an inseparable 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 mixture of regioisomeric cycloadducts with 98% and 53% yield, respectively. The same sequence was applied to obtain multivalent compounds by CuAAC and SPAAC reactions. Thus, the three clickable DNJ derivatives 8a–c were grafted under microwaves on tetravalent and hexavalent pentaerythritol-based dendrimers 11 and 13 prepared following reported procedures (Scheme 3).41,42 Deprotection with aqueous ammonia led to desired compounds 12a–c and 14a–c with high yields. DNJ derivatives 8a and 8b were grafted on hexavalent and dodecavalent phosphorus dendrimers 3-G0 and 4-G1 by means of a simple stirring in methanol/THF. After purification, deprotection led to the desired compounds 15a–b and 16a–b, isolated as a mixture of regioisomers with good yields over the two steps (Scheme 4).
3 mixture of regioisomeric cycloadducts with 98% and 53% yield, respectively. The same sequence was applied to obtain multivalent compounds by CuAAC and SPAAC reactions. Thus, the three clickable DNJ derivatives 8a–c were grafted under microwaves on tetravalent and hexavalent pentaerythritol-based dendrimers 11 and 13 prepared following reported procedures (Scheme 3).41,42 Deprotection with aqueous ammonia led to desired compounds 12a–c and 14a–c with high yields. DNJ derivatives 8a and 8b were grafted on hexavalent and dodecavalent phosphorus dendrimers 3-G0 and 4-G1 by means of a simple stirring in methanol/THF. After purification, deprotection led to the desired compounds 15a–b and 16a–b, isolated as a mixture of regioisomers with good yields over the two steps (Scheme 4).
 | ||
| Scheme 3 (a) Azido derivative 8a, 8b or 8c, CuSO4·5H2O, sodium ascorbate, DMF/H2O, MW, 80 °C, 30 min. (b) NH4OH, 2M, CH3OH. | ||
Enzymatic assays
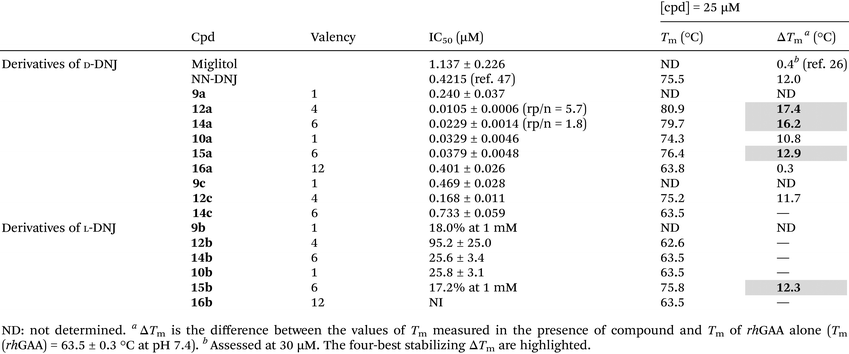
As expected, the dendrimers decorated with L-N-undecanyl-DNJ did not, or only weakly, inhibit GAA. Conversely, all the multivalent compounds bearing D-N-undecanyl-DNJ (compounds 9a, 10a, 12a, 14a, 15a and 16a) were GAA inhibitors with IC50 values between 0.0105 µM and 0.401 µM. This confirms that GAA inhibition is sensitive to chirality, as observed earlier.33 However, multivalent D-DNJs with a PEG spacer (compounds 9c, 12c and 14c) were poor inhibitors of GAA, suggesting that a hydrophilic chain is detrimental to the inhibition of GAA. Regarding the dendrimers 12a and 14a built on a pentaerythritol core, the elongation of the alkyl chain tended to have a slightly favourable effect on inhibition when compared to our previous results.15 Furthermore, moderate multivalent were observed for these compounds effects (1.8 and 5.7 rp/n values for 12a and 14a, respectively). The phosphorus-containing dendrimers 15a and 16a presenting D-N-undecanyl-DNJ were also inhibitors with IC50 values of the same order of magnitude as those previously described with monofluorocyclooctatriazole units (compounds 1 and 2, Fig. 1).15 However, no multivalent effect was observed with the phosphorus dendrimers and, more generally, it seems that the inhibition is less significant for dendrimers of this family.
At 25 µM, all the tested D-DNJ multivalent derivatives (except 16a and 14c) increased the global stability of the rhGAA with a ΔTm between 10.8 and 17.4 °C. For L-DNJ derivatives, only 15b induced an enhancement in Tm of 12.3 °C at 25 µM. However, when tested at 250 µM, the monovalent reference 10b produced a significant shift of 8.1 °C while 12b, 14b and 16b still did not induce any thermal stabilizing effect (see SI). These results indicate that no clear relationship exists between the thermal stabilizing effect and the inhibitory potency of rhGAA as observed by others before.29 Compounds 12a, 14a, 15a and 15b, which induced the best stabilising effect at 25 µM, were selected to be assessed as PC in cellulo. In the search of non-inhibitory PC, compound 15b, which only weakly inhibit GAA at up to 1 mM, was particularly promising.
Cell assays
The effect of the four compounds 12a, 14a, 15a and 15b was evaluated on mutant GAA activity in cultured fibroblasts derived from two adult PD patients (Table 2). The mutation c.-32-13T > G carried by these patients, frequently reported in cases of late-onset PD, occurs in 40% to 90% of the cases, with a predominance among Caucasian populations.44 Residual activity (RA) was assessed in these cell lines and amounts to 4.9% for patient 1 and 9.0% for patient 2 (Table 2). Miglitol at 1 µM was used as a positive control.26,34 Cells were incubated in the presence of 0.5 nM to 50 nM of tested compound for 48 h with six replicates and the GAA activity was compared to that obtained in untreated cells to determine the fold increase (Fig. 3). The choice of these very low concentrations was based on preliminary tests (not shown) at 400 nM where only decrease in enzyme activity was observed with D-undecanyl-DNJ dendrimers. In patient 1's cells, all the compounds induced a decrease in GAA activity, with the most significant decrease observed for compound 14a at 50 nM. Only miglitol led to a 1.2-fold increase in GAA activity. For 14a and 15b, the decrease in GAA activity revealed to be dose-dependent. This observation is surprising for 15b, since it is was shown to be a very weak inhibitor of the enzyme. This might be explained by the potential lysosomal accumulation of these multivalent lipophilic amines due to the density of positive charges at their surface once in the acidic lysosomal environment.45 In patient 2's cells, similar trends were observed, with a dose effect for 14a and 15b. Remarkably, at the very low concentration of 0.5 nM, 15b induces this time an enhancement in enzymatic activity by a factor of 1.2. Even though this effect is weak, it is however statistically significant and might be therapeutically relevant in cells with sufficient residual enzyme activity such as that of patient 2.46 Thus, compound 15b induces a decrease in GAA activity in patient 1's cells but an increase in patient 2's cells, indicating an allele-dependent pharmacological chaperone effect. This dependence is also noted with miglitol. These observations highlight that the balance between inhibitory and chaperone activity of iminosugars is highly sensitive to the nature of the second mutant GAA allele. It is thus the first time that a multivalent compound is shown to allow the increase of GAA activity in Pompe patient cells. This result is particularly interesting since 15b is a ligand of the GAA protein and a weak inhibitor of the enzyme (17.2% inhibition at 1 mM). However, these very encouraging results need to be confirmed in fibroblasts of patients carrying other mutations.| Patient ID | Mutation allele 1 | Mutation allele 2 | % GAA RAa |
|---|---|---|---|
| a RA: residual activity. The residual activity percentage corresponds to the ratio between the GAA activity measured in patient fibroblasts and that measured in healthy control fibroblasts. | |||
| Patient 1 | c.-32-13T > G | c.2237G > A | 4.9% |
| Patient 2 | c.-32-13T > G | c.1076-1G > C and c.2065G > A | 9.0% |
Conclusion
New polythiophosphoramidate dendrimers grafted with 6 or 12 copies of a clickable dibenzocyclooctyne motif have been prepared. A strain-promoted alkyne–azide cycloaddition (SPAAC) enabled the straightforward synthesis of multivalent phosphorus-containing dendrimers functionalized on the surface with D- or L-DNJ pharmacophores. Additionally, a copper-catalyzed click reaction allowed the preparation of tetra- and hexavalent L- or D-DNJ derivatives based on a pentaerythritol core. As expected, dendrimers decorated with L-N-undecanyl-DNJ showed little or no GAA inhibition, whereas those grafted with D-N-undecanyl-DNJ were inhibitors. Weak multivalent effects were observed with tetra- and hexavalent dendrimers 12a and 14a. Compounds 12a, 14a, 15a and 15b which exhibited the highest thermal stabilizing effect at 25 µM according to DSF were selected to be assessed as PCs in patient fibroblasts. The phosphorus-containing dendrimer 15b presenting L-N-undecanyl-DNJ induced a small but significant enhancement in GAA activity. This effect is remarkable because it is the first time that a multivalent ligand is observed to induce an increase in GAA activity in cellulo. Crucially, this compound displays only very weak enzyme inhibitory properties. Current investigations are underway to fully elucidate the mechanism of action of this dendrimer. Future work will involve extending these studies to include fibroblasts derived from patients carrying diverse GAA mutations to comprehensively assess its broad applicability.Experimental
Synthetic chemistry
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, [Alkyne] = 0.01 M) before adding water (1/3 of the final volume). The mixture was stirred at 40 °C at 1200 rpm.
1, [Alkyne] = 0.01 M) before adding water (1/3 of the final volume). The mixture was stirred at 40 °C at 1200 rpm.
Inhibition assays on rhGAA
Recombinant human alpha-Glucosidase acid/GAA (8329-GH), purchased from R&D was used in the inhibition studies. The used substrate 4-methylumbelliferyl-α-D-glucopyranoside47 was purchased from Sigma-Aldrich. Briefly, enzyme solutions (10 μL from a stock solution containing 2.2 μg mL−1) in the presence of 0.25% (w/v) sodium taurocholate and 0.1% (v/v) Triton X-100 in Mcllvaine buffer (100 mM sodium citrate and 200 mM sodium phosphate buffer, pH 5.2) were incubated at 37 °C without (control) or with inhibitor at a final volume of 50 μL for 30 min. After addition of 25 μL of 4-methylumbelliferyl-α-D-glucopyranoside (4.7 mM, Mcllvaine buffer pH 4.6), the samples were incubated at 37 °C for 15 min.Enzymatic reactions were stopped by the addition of aliquots (100 μL) of glycine/NaOH buffer (100 mM, pH 10.6). The amount of 4-methylumbelliferone formed was determined with a FLUOstar microplate reader (BMG Labtech) at 355 nm (excitation) and 460 nm (emission).
Assay of GAA activity on human fibroblasts
Primary cultured skin fibroblasts isolated from patients with Pompe disease were obtained from the Clinical Chemistry Department of Toulouse University Hospital and supplied by Prof. T. Levade (INSERM UMR1037, Cancer Research Center of Toulouse). All experiments were conducted in accordance with the French rules and institute policies on ethics and human material usage and were approved by the CODECOH Ethics Committee of the Softmat laboratory (approval number DC-2020-4322), recognized by the “Ministère de l'enseignement supérieur, de la recherche et de l'innovation”. Written informed consent was obtained from all participants for molecular genetic analyses and cell culture studies. The human fibroblasts were immortalized after transfection with a plasmid encoding the SV40 large T antigen. Cells were routinely cultured in DMEM medium supplemented with 10% inactivated fetal calf serum and 1% antibiotics. Protocol to assess cell GAA activity in 48-well plates was adapted from https://doi.org/10.1016/j.bmc.2022.117129. 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 fibroblasts were seeded in 48-well plates and incubated for 48 h with compounds to be tested. Then, cells were washed twice with cold PBS. Cell lysates were obtained directly in the well by adding 50 µl of 0.1% Triton X100, and stored for 48 h at −80 °C before protein quantification by BCA Assay (Pierce). GAA enzyme activity was determined on cell lysates in sodium acetate buffer pH 5.6 using 4-methylumbelliferyl-α-D-glucopyranoside (Sigma-Aldrich, St Louis, MO, USA) as substrate.
000 fibroblasts were seeded in 48-well plates and incubated for 48 h with compounds to be tested. Then, cells were washed twice with cold PBS. Cell lysates were obtained directly in the well by adding 50 µl of 0.1% Triton X100, and stored for 48 h at −80 °C before protein quantification by BCA Assay (Pierce). GAA enzyme activity was determined on cell lysates in sodium acetate buffer pH 5.6 using 4-methylumbelliferyl-α-D-glucopyranoside (Sigma-Aldrich, St Louis, MO, USA) as substrate.
Conflicts of interest
There are no conflicts of interest to declare.Data availability
The data that support the findings of this study are available in the published article and its supplementary information (SI). Supplementary information: synthesis and characterization, NMR spectra for new compounds, mass spectra for 15a and 16a, IC50 and DSF curves. See DOI: https://doi.org/10.1039/d6ob00021e.Acknowledgements
We thank Pr. T. Levade (Institut National de la Santé et de la Recherche Médicale (INSERM) UMR1037, Centre de Recherches en Cancérologie de Toulouse (CRCT)) for his help and his valuable guidance throughout this work. We also acknowledge the French Ministry of Higher Education and Research for funding Dr Guichot’s PhD fellowship.References
- G. Parenti, G. Andria and A. Ballabio, Annu. Rev. Med., 2015, 66, 471–486 CrossRef CAS PubMed.
- G. Parenti, D. L. Medina and A. Ballabio, EMBO Mol. Med., 2021, 13, 1–21 Search PubMed.
- G. Parenti, G. Andria and K. J. Valenzano, Mol. Ther., 2015, 23, 1138–1148 CrossRef CAS.
- L. Liguori, M. Monticelli, M. Allocca, B. Hay Mele, J. Lukas, M. V. Cubellis and G. Andreotti, Int. J. Mol. Sci., 2020, 21, 489 CrossRef CAS PubMed.
- M. L. Tran, Y. Génisson, S. Ballereau and C. Dehoux, Molecules, 2020, 25, 3145 CrossRef CAS PubMed.
- S. L. Macauley, Pediatr. Endocrinol. Rev., 2016, 13, 639–648 Search PubMed.
- S. G. Gouin, Chem. – Eur. J., 2014, 20, 11616–11628 CrossRef CAS PubMed.
- M. González-Cuesta, C. Ortiz Mellet and J. M. García Fernández, Chem. Commun., 2020, 56, 5207–5222 Search PubMed.
- C. Matassini, C. Parmeggiani, F. Cardona and A. Goti, Tetrahedron Lett., 2016, 57, 5407–5415 CrossRef CAS.
- P. Compain, Chem. Rec., 2020, 20, 10–22 CrossRef CAS.
- C. Decroocq, D. Rodríguez-Lucena, K. Ikeda, N. Asano and P. Compain, ChemBioChem, 2012, 13, 661–664 CrossRef CAS PubMed.
- L. Smith, S. Mullin and A. H. V. Schapira, Exp. Neurol., 2017, 298, 180–190 CrossRef CAS PubMed.
- E. Laigre, D. Hazelard, J. Casas, J. Serra-Vinardell, H. Michelakakis, I. Mavridou, J. M. F. G. Aerts, A. Delgado and P. Compain, Carbohydr. Res., 2016, 429, 98–104 CrossRef CAS PubMed.
- A. Joosten, C. Decroocq, J. de Sousa, J. P. Schneider, E. Etamé, A. Bodlenner, T. D. Butters and P. Compain, ChemBioChem, 2014, 15, 309–319 CrossRef CAS.
- M. L. Tran, M. Borie-Guichot, V. Garcia, A. Oukhrib, Y. Génisson, T. Levade, S. Ballereau, C. Turrin and C. Dehoux, Chem. – Eur. J., 2023, 29, e202301210 CrossRef CAS PubMed.
- M. Borie-Guichot, M. L. Tran, V. Garcia, A. Oukhrib, F. Rodriguez, C.-O. O. Turrin, T. Levade, Y. Génisson, S. Ballereau and C. Dehoux, Bioorg. Chem., 2024, 146, 107295 CrossRef CAS.
- F. Buco, C. Matassini, C. Vanni, F. Clemente, P. Paoli, C. Carozzini, A. Beni, F. Cardona, A. Goti, S. E. Moya, M. G. Ortore, P. Andreozzi, A. Morrone and M. Marradi, Org. Biomol. Chem., 2023, 21, 9362–9371 RSC.
- M. G. Davighi, F. Clemente, A. Morano, F. Mangiavacchi, F. Cardona, A. Goti, P. Paoli, A. Morrone, F. Nieto-Fabregat, R. Marchetti and C. Matassini, Chem. – Eur. J., 2026, 32, e02436 CrossRef CAS.
- M. Martínez-Bailén, A. T. Carmona, F. Cardona, C. Matassini, A. Goti, M. Kubo, A. Kato, I. Robina and A. J. Moreno-Vargas, Eur. J. Med. Chem., 2020, 192, 112173 CrossRef.
- C. Vanni, F. Clemente, P. Paoli, A. Morrone, C. Matassini, A. Goti and F. Cardona, ChemBioChem, 2022, 23, e202200077 Search PubMed.
- R. Risquez-Cuadro, R. Matsumoto, F. Ortega-Caballero, E. Nanba, K. Higaki, J. M. García Fernández and C. Ortiz Mellet, J. Med. Chem., 2019, 62, 5832–5843 CrossRef CAS PubMed.
- V. Sánchez-Porras, J. M. Guevara-Morales and O. Y. Echeverri-Peña, Int. J. Mol. Sci., 2023, 24, 12481 CrossRef PubMed.
- M. Borie-Guichot, M. L. Tran, Y. Génisson, S. Ballereau and C. Dehoux, Molecules, 2021, 26, 7223 CrossRef CAS PubMed.
- R. Kakavanos, J. J. Hopwood, D. Lang, P. J. Meikle and D. A. Brooks, FEBS Lett., 2006, 580, 4365–4370 CrossRef CAS PubMed.
- T. Okumiya, M. A. Kroos, L. Van Vliet, H. Takeuchi, A. T. Van der Ploeg and A. J. J. Reuser, Mol. Genet. Metab., 2007, 90, 49–57 CrossRef CAS PubMed.
- C. Bruckmann, H. Repo, E. Kuokkanen, H. Xhaard and P. Heikinheimo, ChemMedChem, 2012, 7, 1943–1953 CrossRef CAS PubMed.
- R. Khanna, R. Soska, Y. Lun, J. Feng, M. Frascella, B. Young, N. Brignol, L. Pellegrino, S. A. Sitaraman, R. J. Desnick, E. R. Benjamin, D. J. Lockhart and K. J. Valenzano, Mol. Ther., 2010, 18, 23–33 CrossRef CAS PubMed.
- R. Khanna, A. C. Powe, Y. Lun, R. Soska, J. Feng, R. Dhulipala, M. Frascella, A. Garcia, L. J. Pellegrino, S. Xu, N. Brignol, M. J. Toth, H. V. Do, D. J. Lockhart, B. A. Wustman and K. J. Valenzano, PLoS One, 2014, 9, e102092 CrossRef.
- A. Vieira Da Cruz, V. Perraudin, N. Minopoli, R. Iacono, V. Roig-Zamboni, A. Bossio, S. Tangara, M. Fayolle, A. Kanazawa, C. Philouze, A. Tarallo, J. J. A. Heming, M. Artola, J.-B. Behr, H. S. Overkleeft, M. Moracci, G. Sulzenbacher, G. Parenti and S. Py, J. Med. Chem., 2025, 68, 19269–19286 CrossRef CAS PubMed.
- D. D'Alonzo, M. De Fenza, C. Porto, R. Iacono, M. Huebecker, B. Cobucci-Ponzano, D. A. Priestman, F. Platt, G. Parenti, M. Moracci, G. Palumbo and A. Guaragna, J. Med. Chem., 2017, 60, 9462–9469 Search PubMed.
- C. Porto, M. Cardone, F. Fontana, B. Rossi, M. R. Tuzzi, A. Tarallo, M. V. Barone, G. Andria and G. Parenti, Mol. Ther., 2009, 17, 964–971 Search PubMed.
- C. Porto, M. C. Ferrara, M. Meli, E. Acampora, V. Avolio, M. Rosa, B. Cobucci-Ponzano, G. Colombo, M. Moracci, G. Andria and G. Parenti, Mol. Ther., 2012, 20, 2201–2211 CrossRef CAS PubMed.
- D. D'Alonzo, M. De Fenza, C. Porto, R. Iacono, M. Huebecker, B. Cobucci-Ponzano, D. A. Priestman, F. Platt, G. Parenti, M. Moracci, G. Palumbo and A. Guaragna, J. Med. Chem., 2017, 60, 9462–9469 CrossRef.
- V. Roig-Zamboni, B. Cobucci-Ponzano, R. Iacono, M. C. Ferrara, S. Germany, Y. Bourne, G. Parenti, M. Moracci and G. Sulzenbacher, Nat. Commun., 2017, 8, 1111 CrossRef PubMed.
- J. Dommerholt, F. P. J. T. Rutjes and F. L. van Delft, Top. Curr. Chem., 2016, 374, 1–20 CrossRef CAS PubMed.
- R. Jebbawi, A. Oukhrib, E. Clement, H. Labie, S. Cassel, S. Fruchon, A. Caminade, M. Simon, R. Poupot, C. Turrin and M. Blanzat, Adv. Healthcare Mater., 2025, e03282 CrossRef PubMed.
- M. Blanzat, R. Jebbawi, R. Poupot, C.-O. Turrin, S. Michel, E. Clément, H. Labie and A. Oukhrib, WO 2021130453-A1, 2019.
- P. Compain, C. Decroocq, J. Iehl, M. Holler, D. Hazelard, T. Mena Barragán, C. Ortiz Mellet and J. Nierengarten, Angew. Chem., 2010, 122, 5889–5892 Search PubMed.
- V. De Pasquale, A. Esposito, G. Scerra, M. Scarcella, M. Ciampa, A. Luongo, D. D'Alonzo, A. Guaragna, M. D'Agostino and L. M. Pavone, J. Med. Chem., 2023, 66, 1790–1808 CrossRef CAS PubMed.
- C. Decroocq, A. Joosten, R. Sergent, T. Mena Barragán, C. Ortiz Mellet and P. Compain, ChemBioChem, 2013, 14, 2038–2049 CrossRef CAS.
- I. Papp, J. Dernedde, S. Enders and R. Haag, Chem. Commun., 2008, 44, 5851 RSC.
- L. C. Moraes, G. P. de Souza, H. V. Fajardo, S. C. Luz, E. Álvarez, F. Lloret, R. M. Ribeiro-Viana, J. Rojo, H. O. Stumpf, R. C. Figueiredo and R. S. Corrêa, Inorg. Chim. Acta, 2019, 489, 93–99 Search PubMed.
- A. Kato, I. Nakagome, M. Kise, K. Yoshimura, N. Tanaka, R. J. Nash, G. W. J. Fleet, Y. Kobayashi, H. Ikeda, T. Okada and N. Toyooka, J. Med. Chem., 2023, 66, 9023–9039 Search PubMed.
- A. Ezzat, M. ElBagoury, S. Roushdy and Y. Aktham, Int. J. Med. Dev. Ctries., 2022, 6, 855–863 Search PubMed.
- S. D. B. Goldman, R. S. Funk, R. A. Rajewski and J. P. Krise, Bioanalysis, 2009, 1, 1445–1459 CrossRef CAS PubMed.
- J. Q. Fan, Biol. Chem., 2008, 389, 1–11 CrossRef CAS PubMed.
- U. Andersson, G. Reinkensmeier, T. D. Butters, R. A. Dwek and F. M. Platt, Biochem. Pharmacol., 2004, 67, 697–705 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |