Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
A tertiary amine synthesis via a hydroxylamine alkylation/catalytic reduction sequence
Daniel
Barriault
 a,
Nicholas M.
Halliday
a,
Nicholas M.
Halliday
 a,
Sterling
Renzoni
a,
Sterling
Renzoni
 a,
Huy M.
Ly
a,
Huy M.
Ly
 b,
Monica A.
Gill
b,
Monica A.
Gill
 a and
André M.
Beauchemin
a and
André M.
Beauchemin
 *a
*a
aCentre for Catalysis Research and Innovation, Department of Chemistry and Biomolecular Sciences, University of Ottawa, Ottawa, ON K1N 6N5, Canada. E-mail: andre.beauchemin@uottawa.ca
bDepartment of Organic Chemistry, School of Pharmacy, University of Medicine and Pharmacy at Ho Chi Minh City, Vietnam
First published on 10th February 2026
Abstract
Overalkylation is a common side reaction that typically prevents the synthesis of tertiary N-methylamines from secondary amines, using alkyl iodides and other reactive electrophiles. Herein, we present an indirect approach featuring hydroxylamines, using a reaction sequence involving alkylation, followed by an optimized, catalytic in situ reduction of the N-oxide intermediate to give the desired N-methyl tertiary amine. An optimized isolation of these products is also reported.
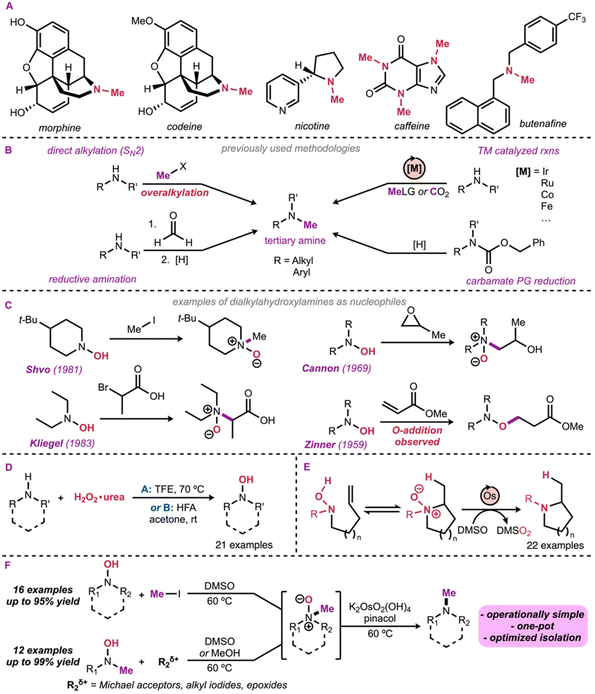
N-Methyl tertiary amines are a common motif in many pharmaceutical and natural products (Scheme 1A), making them important target compounds for synthetic chemists.1 Given the importance of N-methyl tertiary amines, many approaches have been developed for their synthesis (Scheme 1B). Although the formation of tertiary amines may seem conceptually simple, the direct alkylation of secondary amines via SN2 reactions is often plagued by overalkylation.2 This problem arises from the nucleophilicity of the amine increasing as it becomes more substituted, making the products more reactive than the starting materials.3 Overalkylation is a more common synthetic challenge for reactions with iodomethane, and related reactive alkylating agents that lack the steric hindrance that could help reduce overalkylation.4 A variety of systems have been developed to reduce the overalkylation using iodomethane, however these can be overall substrate dependent and/or moderate yielding.5 Selective N-methylation of secondary amines is often achieved in good yield using a reductive amination approach using formaldehyde and a range of reducing agents, including NaBH3CN,6a Zn(BH4)2,6b NaBH(OAc)3,6c and others.6d The Eschweiler–Clarke reaction of secondary amines with formaldehyde, using formic acid as reductant, has also been a common way to form N-methylamines.6e Recent advances in the Eschweiler–Clarke reaction have reported using H2 with heterogenous catalysts,6f the use an iridium catalyst to enhance the rate of the reduction step,6g as well as a simplified reaction using only formaldehyde and high heat.6h Metal catalyzed processes have also been developed, especially with the use of methanol in a borrowing hydrogen strategy.7 These processes tend to give overall very high mono-N-methylation selectivity, however, do require the use of more forcing conditions and the use of precious metal catalysts, such as rhodium and iridium.8 These strategies have been expanded to different C1-electrophiles, such as formic acid,9 carbon dioxide,10 and dialkyl phosphites.11 Carbamates have also been used as late-stage methyl group precursors in the synthesis of complex molecules.12 Indeed, carbamates may be reduced to the methylamine group, but also often act as a protecting group for the nitrogen atom throughout the synthesis. This protecting group strategy led us to wonder if hydroxylamines could be used in a similar fashion (Scheme 1B). The use of disubstituted hydroxylamines as nucleophiles in alkylation reactions has been reported sporadically,13 but this reactivity has not been thoroughly investigated, perhaps due to the fact that such reactions afford N-oxide products. An example of N-methylation of a dialkylhydroxylamine using iodomethane was reported by Shvo in 1981, giving the corresponding N-oxide in low yield while using forcing conditions.13c Alkyl bromides were also explored and showed moderate reactivity.13d–g Dialkylhydroxylamines demonstrated moderate reactivity with propylene oxide to form 2-hydroxypropyl N-oxides.13i The conjugate N-addition of mono-substituted hydroxylamines onto various Michael acceptors has been thoroughly reported in the literature,13j–l however, the same cannot be said with the dialkyl derivatives. In these cases, only the thermodynamic O-addition products have been observed, due to the fast Cope-elimination of the kinetic N-oxide product (the N-addition product).13l Another contributing factor to this gap in the literature may be inefficient synthetic methodology and challenging isolation for accessing the hydroxylamine starting materials. Our group has a long-standing interest in the synthesis and reactivity of hydroxylamines, notably in the context of Cope-type hydroamination reactions.14 We have recently published methodology enabling a direct, oxidative synthesis and isolation of N,N-disubstituted hydroxylamines from the parent amines (Scheme 1D).15 In parallel, we discovered that N-oxides can be chemoselectively reduced in the presence of hydroxylamines, allowing challenging hydroamination reactions and access to cyclic tertiary amines (Scheme 1E).16 These advances led us to explore the alkylation of hydroxylamines to give the corresponding N-oxides and subsequent catalytic reduction, thereby expanding the diversity of tertiary amines accessible with this strategy. Herein, we report the transformation of hydroxylamines to their corresponding tertiary amines via an N-alkylation/catalytic N-oxide reduction sequence, with the specific goal of synthesizing N-methyl tertiary amines (Scheme 1F).
Given the importance of N-methylamines, we sought the development of mild, but reliable conditions to alkylate various N,N-dialkylhydroxylamines to form the N-oxide intermediate, followed by efficient catalytic reduction conditions to form the desired products. Selected optimization data is presented in Tables 1 & 2. In early optimization toward a suitable reaction sequence, alkylation of secondary hydroxylamines proceeded rapidly to form N-oxide 2a quantitatively using a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio of hydroxylamine to methyl iodide13a,b in DMSO (entry 1). Less polar solvents showed mixed reactivity, with acetone and tetrahydrofuran showing the best reactivity, 26 and 27% respectively (entries 2 & 3). Dichloroethane gave a 3% yield (entry 4), while acetonitrile resulted in a complex, intractable mixture (entry 5).17 When solvent polarity was further increased, dimethylformamide showed an increase in the desired reactivity (75%; entry 6). Protic solvents were also evaluated. Methanol, ethanol and isopropanol gave similar results, with 53, 49 and 35% N-oxide formation being observed, respectively (entries 7–9). The use of TFE and HFIP was also evaluated, as they are shown to aid hydroamination reactions by stabilizing the formed N-oxides.18 With both these solvents, no reaction was observed, likely resulting from the stabilization of the hydroxylamines (entries 10 & 11). Thus, DMSO was used for the alkylation step, which added to the simplicity of the one-pot process, as it also acts as the stoichiometric reductant in the next step.
1 ratio of hydroxylamine to methyl iodide13a,b in DMSO (entry 1). Less polar solvents showed mixed reactivity, with acetone and tetrahydrofuran showing the best reactivity, 26 and 27% respectively (entries 2 & 3). Dichloroethane gave a 3% yield (entry 4), while acetonitrile resulted in a complex, intractable mixture (entry 5).17 When solvent polarity was further increased, dimethylformamide showed an increase in the desired reactivity (75%; entry 6). Protic solvents were also evaluated. Methanol, ethanol and isopropanol gave similar results, with 53, 49 and 35% N-oxide formation being observed, respectively (entries 7–9). The use of TFE and HFIP was also evaluated, as they are shown to aid hydroamination reactions by stabilizing the formed N-oxides.18 With both these solvents, no reaction was observed, likely resulting from the stabilization of the hydroxylamines (entries 10 & 11). Thus, DMSO was used for the alkylation step, which added to the simplicity of the one-pot process, as it also acts as the stoichiometric reductant in the next step.
| Entry | Solvent | Yieldb (%) |
|---|---|---|
a Hydroxylamine (1 equiv.) in the solvent (1.0 M), then MeI (1 equiv.) added, 60 °C, 0.75 h.
b ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1H NMR yields determined using 1,3,5-trimethoxybenzene as an internal standard. 1H NMR yields determined using 1,3,5-trimethoxybenzene as an internal standard.
|
||
| 1 | DMSO | >99 |
| 2 | Acetone | 26 |
| 3 | Tetrahydrofuran | 27 |
| 4 | 1,2-Dichloroethane | 3 |
| 5 | Acetonitrile | N.D. |
| 6 | N,N-Dimethylformamide | 75 |
| 7 | Methanol | 53 |
| 8 | Ethanol | 49 |
| 9 | Isopropanol | 35 |
| 10 | 2,2,2-Trifluoroethanol | 0 |
| 11 | 1,1,1,3,3,3-Hexafluoroisopropanol | 0 |
| Entry | Deviation from standard conditions | Yieldb (%) |
|---|---|---|
a Alkylation: hydroxylamine (1 equiv.) in DMSO (1.0 M), then MeI (1 equiv.) added, 60 °C, 0.75 h. Reduction: [Os] DMSO solution (1 mol%) was added, 60 °C, 2 h.
b ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1H NMR yields determined using 1,3,5-trimethoxybenzene as an internal standard.
c Isolated yields are shown parentheses. 1H NMR yields determined using 1,3,5-trimethoxybenzene as an internal standard.
c Isolated yields are shown parentheses.
|
||
| 1 | None | >99 |
| 2 | 1 or 2 equiv. of pinacol w/ cat. | 72 or 76 |
| 3 | Ethylene glycol or (S,S)-(–)-hydrobenzoin | 90 or 62 |
| 4 | B2pin2 (1 equiv.) | >99 |
| 5 | Morpholin-4-ol w/ EtI or EtBr | 18 or <5 |
| 6 | NaBr 1 equiv. w/ reductant | 59 |
| 7 | Bis-2-ethylhexyl-NOH (1 g) 6 h or 18 h | 14 or (97)c |
Building on our recent work for the subsequent catalytic reduction,16 it was found that modified conditions using 1 mol% of an osmium catalyst (prepared from K2OsO2(OH)4 with 3 equivalents of pinacol; see SI for additional details) for 2 h at 60 °C resulted in a quantitative reduction of the N-oxide by NMR, and oxidation of DMSO to dimethylsulfone (DMSO2). With this, the catalytic conditions previously developed for cyclic N-oxides16 were optimized to allow efficient reduction of acyclic N-oxides (Table 2, entry 1), making the desired reaction sequence possible. Lowering the equivalents of pinacol ligand in the osmium catalyst solution had a detrimental impact on the reduction, lowering the yield to 72% and 76% for 1 and 2 equivalents, respectively (entry 2). Varying the ligand from pinacol to ethylene glycol resulted in a slight decrease in yield to 90%, whereas the change to (S,S)-(–)-hydrobenzoin showed a significant decrease to 62% yield (entry 3). The use of different reductants was also explored. B2pin2 is a known N-oxide stoichiometric reducing agent,19 which led the desired amine in high yield (entry 4). B2pin2 was not used for the remainder of the development due to it not being amenable to catalytic reductions, as well as its lower chemoselectivity. Alkyl bromides were also explored as alkylating agents. Initially using morpholin-4-ol, it was found that the alkylation proceeded comparably to its iodide equivalent, however no reduction was observed (entry 5). We hypothesized that the bromide ion was poisoning the catalyst, preventing it from reducing the N-oxide.20 Thus a reaction with added NaBr present during the reduction step was performed, for which a lower yield of 59% was obtained (entry 6), lending support to this hypothesis. During the evaluation of the scope of the sequence (vide infra), it was found that the yield decreased for more hindered substrates (such as 1g); the alkylation was efficient, but the reduction of sterically bulky N-oxides was much slower. Fortunately, increasing the reaction time to 18 h allowed the reduction to go to completion (entry 7).
Although the formation of these model tertiary amines was optimized, the isolation was not trivial. The presence of iodide, DMSO, DMSO2 and osmium proved to be a purification challenge. The iodine was quenched post-reaction using an aqueous sodium thiosulfate solution. Column chromatography was ineffective, and while different additives and solvent systems were attempted, little success was obtained.21 Liquid–liquid extraction was found to be the most effective way to separate the amines from the undesired starting materials and by-products. Basification using aqueous NaOH or KOH, followed by extraction with dichloromethane and several washes with a saturated aqueous NaCl solution allowed for isolation of high purity tertiary amines. The removal of DMSO this way enabled the isolation of the more volatile amines.22
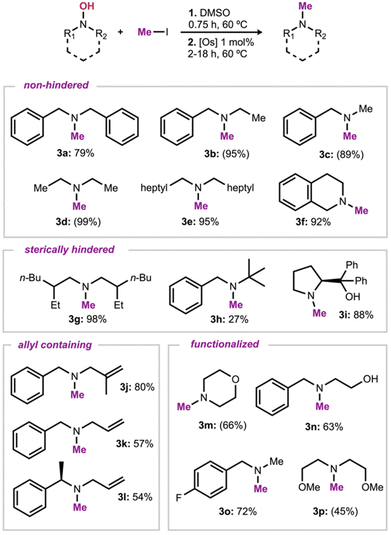
With suitable conditions and isolation procedures, evaluation of the substrate scope of the reaction sequence was conducted by reacting various hydroxylamines with iodomethane (Scheme 2). N,N-Dibenzylhydroxylamine gave a good isolated yield of 3a (79%). Variation of one of the benzyl groups for an ethyl or methyl group resulted in a consistently higher NMR yield of 95% & 89%, respectively. Less hindered products were obtained in higher yields (entries 3d & 3e 99% & 95%, respectively). This method also proved efficient with cyclic amine 3f (92%). A hydroxylamine with increased steric hinderance at the β-position reacted efficiently, allowing formation of the parent amine with a minimal decrease in yield relative to its less hindered counterparts (3g, 98% yield). A low isolated yield was obtained for 3h, 27%, which is not unexpected due to the higher steric hindrance on the α-carbon, decreasing the efficiency of the reduction reaction. When using a cyclic hydroxylamine with reduced steric hindrance at the α-position but higher on the β-carbon, we see this trend continue as high isolated yields were observed (3i, 88%). Allylic hydroxylamines were also evaluated and showed good reactivity, with isolated yields for 3j and 3k of 80 and 57% respectively. A lower yield was obtained for amine 3l (54%), which is, again, consistent with a more challenging reduction step. Functional group tolerance was also explored. A morpholine derivative 3m was obtained in a 66% NMR yield. The tolerance of substrates with pendant alcohols was evaluated, and good yields were obtained with the benzylethanolamine derivative 3n, and the diphenylprolinol derivative 3i (63% and 88%, respectively). These results show that products that could coordinate to the osmium catalyst23 can be tolerated under the reaction conditions. Fluorinated amine 3o was also obtained in good yield (72%), in line with the result obtained with the non-functionalized analogue 3a. The tolerance of ether groups was further evaluated, and acyclic amine 3p was prepared (45% yield).
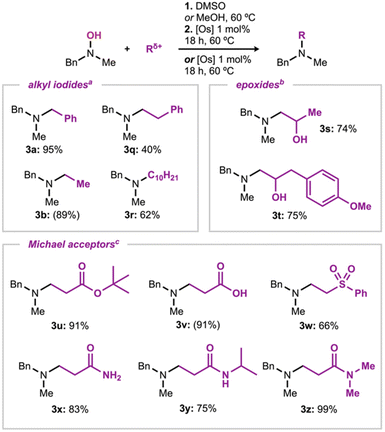
As previously mentioned, it was observed that the overall sequence suffered in yield during the reduction step when higher steric bulk was present around the N-oxide. In order to expand the scope of the reaction sequence forming N-methyl tertiary amines, the reaction of N-methylbenzylhydroxylamine with various alkylating agents, and subsequent reduction, was then studied (Scheme 3). This approach allowed us to expand the possible alkylating agents being used. The use of benzyl, ethyl and decyl iodide in the two-step process resulted in moderate to excellent overall yields of the corresponding amines, 95, 89 and 62%, respectively (3a, 3b & 3r). More notably, in the case of 3a and 3b, previous scope entries (from Scheme 2) were re-synthesized in similar or greater yields. Surprisingly, the reaction with phenethyl iodide with N-methylbenzylhydroxylamine to yield 3q, gave a lower-than-expected yield of the reduced product, 40%. This trend is consistent with other alkylation/reductions performed with phenethyl iodide (see 3ad & 3ai).24 Epoxides were also explored as electrophiles to form the corresponding amino alcohols. Using a modified alkylation procedure, in which methanol was used as a solvent,13g3s and 3t were obtained in high yields, 74 and 75%, respectively. Michael acceptors were of interest due to the reports of N-alkylation being the faster transformation, however due to its reversibility (via the Cope elimination), the thermodynamically favored O-alkylation was observed.13j The reducing catalyst was added to the reaction mixture from the beginning with the Michael acceptors to permit the chemoselective reduction of the N-oxide as it formed. This strategy was successful in obtaining the N-alkylated product as the sole product. The tert-butyl acrylate derivative 3u was isolated in high yield (91%). Acrylic acid also resulted in good reactivity, affording an amino acid derivative in high NMR yield of 91% (3v). The reaction with vinyl sulfone also resulted in good yields, 66% (3w). Acrylamide derivatives were also well tolerated. Acrylamide afforded the product in 83% isolated yield (3x). Mono- and di-substituted acrylamide derivatives were also obtained in high yields, 75 and 99% respectively (3y & 3z). These results demonstrate that the reaction sequence and catalytic reduction conditions are applicable to form a diverse group of cyclic and acyclic tertiary amines. The results also show the broad applicability of reactions with alkyl iodides, epoxides, and Michael acceptors to form N-oxides efficiently. Remarkably, the osmium catalyzed reductions described above proceeded with high chemoselectivity during the reduction step, avoiding the formation of oxidized byproducts derived from further oxidation of the N-methyl tertiary amines.25
We elected to study the steric effects on the reduction step in a systematic fashion. To delineate the reactivity of the alkylation/reduction sequence of tertiary amines for reagents with varying steric hindrance, the structurally analogous N-hydroxypyrrolidine and N,N-diethylhydroxylamine were tested using various alkylating agents (Scheme 4). Unsurprisingly, the methylation of N-hydroxypyrrolidine and subsequent reduction of the N-oxide intermediate resulted in high yield, 85% (3aa). The reaction sequence with ethyl iodide also worked in high yield, 93% (3ab), which was surprising as the ethylation of morpholin-1-ol was quite inefficient (Table 2, entry 5). Reaction sequences initiated by alkylation of N-hydroxypyrroline with benzyl iodide (3ac) and vinyl sulfone (3ae) gave the corresponding amines in decreased yields, 62 and 49%, respectively. The reaction sequence with phenethyl iodide (3ad) only gave a 6% yield, with the presence of 14% styrene from the competing Cope-elimination reaction, suggesting this is due to an unstable N-oxide intermediate.24 When methylating the acyclic variant N,N-diethylhydroxylamine, the reduction of the N-oxide worked comparably to that of the N-hydroxypyrrolidine analog (74%, 3af). However, a significantly lower yield was observed when ethyl iodide was used, giving only 23% of reduced acyclic amine 3ag, in contrast to the 93% observed with the N-hydroxypyrrolidine derivative (3ab). Benzylation of the N,N-diethylhydroxylamine resulted in a modest yield of 53% for the overall sequence (3ah), and the reduction of the phenethyl reaction sequence was equally as inefficient as for N-hydroxypyrrolidine, 8% (3ai). The reduction of the conjugate addition product of the N,N-diethylhydroxylamine onto the vinyl sulfone was observed in a moderate yield, 52% (3aj). This set of experiments provided additional insight into the steric limitations of the reduction of the N-oxide with the osmium catalyst, showing the impact of steric hindrance and proximal electron-withdrawing groups on the reduction step. These findings are aligned with previous observations,16 as well as what is reported in the literature.26
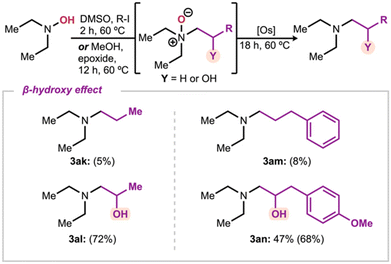
Interestingly, the systematic variation above also allowed comparison of the reactivity of relatively similar substrates (Scheme 5). For example, while products 3ak and 3al have similar substituents, it is the most hindered product containing an additional hydroxyl group that is formed more efficiently (5 & 72%). We see the same trend when comparing 3am to 3an, showing an improvement in yields from negligible to moderate (8 & 68%). These results suggest that the presence of an alcohol functionality in the β-position is beneficial for the reduction reactivity, possibly due to a directed reduction. While this warrants additional investigation, the ability of osmium to participate in directed catalytic oxidation reactions has been reported.27
In summary, a new approach for the N-methylation of dialkylhydroxylamines using iodomethane and the subsequent reduction of the corresponding N-oxides using an osmium catalyst and DMSO has been developed. A complementary alkylation approach of N-methyl-N-benzylhydroxylamine using alkyl iodides, epoxides and Michael acceptors was also developed to form N-methyl tertiary amines. Insight into the steric limitations for the reduction of the N-oxide intermediates has also been reported. The reported methods demonstrated compatibility with alkenes, alcohols, ethers, esters, amides and fluorinated functional groups. Limitations include the difficult isolation of very volatile amines as well as decreased yields in the reduction of highly sterically encumbered N-oxides. Overall, this methodology demonstrates the utility of various secondary hydroxylamines for the selective formation of tertiary amines in a one-pot reaction. It further shows that the recently developed conditions for osmium catalyzed N-oxide reduction are chemoselective, as seen with the tolerance of reactive functional groups (i.e. alkenes, secondary hydroxylamines, Michael acceptors). This method can facilitate the formation of complex nitrogen-containing molecules, amine N-oxides, and N-methyl tertiary amines, which have high relevance for the pharmaceutical industry.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article has been included as part of the supplementary information (SI). Supplementary information: Table S1, general procedures, full characterization data for all new compounds, 1H and 13C NMR spectra, and further experimental details. See DOI: https://doi.org/10.1039/d5ob01902h.Acknowledgements
The authors thank the University of Ottawa and NSERC for generous financial support, and Paraza Pharma, MITACS, and NSERC (Alliance grant) for support of related research. D. B. thanks the Province of Ontario, S. R. and N. M. H. thank NSERC, and S. R. and H. M. L. thank MITACS, for scholarships.References
- (a) J. A. Joule, Natural Products Containing Nitrogen Heterocycles—Some Highlights 1990–2015, in Heterocyclic Chemistry in the 21st Century—A Tribute to Alan Katritzky, Advances in Heterocyclic Chemistry, Elsevier, Amsterdam, Netherlands, 2016, ch. 4, vol. 119, pp. 81–106. DOI:10.1016/bs.aihch.2015.10.005; (b) C. M. Marshall, J. G. Federice, C. N. Bell, P. B. Cox and J. T. Njardarson, J. Med. Chem., 2024, 67, 11622–11655, DOI:10.1021/acs.jmedchem.4c01122; (c) E. J. Barreiro, A. E. Kümmerle and C. A. M. Fraga, Chem. Rev., 2011, 111, 5215–5246, DOI:10.1021/cr200060g; (d) A. Sharma, A. Kumar, S. A. H. A. Monaim, Y. E. Jad, A. El-Faham, B. G. de la Torre and F. Ablericio, Biopolymers, 2018, 109, e23110, DOI:10.1002/bip.23110.
- (a) A. Trowbridge, S. M. Walton and M. J. Gaunt, Chem. Rev., 2020, 120, 2613–2692, DOI:10.1021/acs.chemrev.9b00462; (b) R. N. Salvatore, C. H. Yoon and K. W. Jung, Tetrahedron, 2001, 57, 7785–7811, DOI:10.1016/S0040-4020(01)00722-0; (c) Y. Chen, Chem. – Eur. J., 2019, 25, 3405–3439, DOI:10.1002/chem.201803642.
- L. F. Resende and J. R. Pliego, Jr., J. Mol. Model., 2024, 30, 107, DOI:10.1007/s00894-024-05902-7.
- (a) J. K. Laerdahl and E. Uggerud, Int. J. Mass Spectrom., 2002, 214, 277–314, DOI:10.1016/S1387-3806(01)00575-9; (b) M. Gallegos, A. Costales and Á. M. Pendás, J. Comput. Chem., 2022, 43, 785–795, DOI:10.1002/jcc.26834; (c) M. Gallegos, A. Costales and Á. M. Pendás, J. Phys. Chem. A, 2022, 126, 1871–1880, DOI:10.1021/acs.jpca.2c00415; (d) A. G. Evans and M. Polanyi, Nature, 1942, 149, 608, DOI:10.1038/149608a0.
- Primary amines only: (a) G. Bar-Haim and M. Kol, Org. Lett., 2004, 6, 3549–3551, DOI:10.1021/ol048554k; (b) T. Lebleu, X. Ma, J. Maddaluno and J. Legros, Chem. Commun., 2014, 50, 1836–1838, 10.1039/C3CC48997C; (c) C. Chiappe, P. Piccioli and D. Pieraccini, Green Chem., 2006, 8, 277–281, 10.1039/B509851C.
- (a) R. F. Borch, M. D. Bernstein and H. D. Durst, J. Am. Chem. Soc., 1971, 93, 2897–2904, DOI:10.1021/ja00741a013; (b) S. Bhattacharyya, A. Chatterjee and S. K. Duttachowdhury, J. Chem Soc., Perkin Trans. 1, 1994, 1–2, 10.1039/P19940000001; (c) A. F. Abdel-Magid, K. G. Carson, B. D. Harris, C. A. Maryanoff and R. D. Shah, J. Org. Chem., 1996, 61, 3849–3862, DOI:10.1021/jo960057x; (d) E. Byun, B. Hong, K. A. De Castro, M. Lim and H. Rhee, J. Org. Chem., 2007, 72, 9815–9817, DOI:10.1021/jo701503q; (e) X. Zhou, X. Ni, X. Wu and L. Yin, Molecules, 2025, 30, 3504, DOI:10.3390/molecules30173504; (f) H. Wang, Y. Huang, X. Dai and F. Shi, Chem. Commun., 2017, 53, 5542–5545, 10.1039/C7CC02314F; (g) Y. Zhang, K. Zhang, J. Xu and Z. Yang, Green Chem., 2025, 27, 5136–5148, 10.1039/D4GC05652C; (h) K. O. Biriukov, E. Podyacheva, I. Tarabrin, O. I. Afanasyev and D. Chusov, J. Org. Chem., 2024, 89, 3580–3584, DOI:10.1021/acs.joc.3c02476.
- For reviews on alkylation, see: (a) X. Yu, X. Cui, H. Jing, B. Qian, H. Yuan and F. Shi, ChemCatChem, 2024, 16, e202400291, DOI:10.1002/cctc.202400291; (b) J. Templ and M. Schnürch, Chem. – Eur. J., 2024, 30, e202304205, DOI:10.1002/chem.202304205.
- For selected examples of metal catalyzed processes, see: (a) R. Grigg, T. R. B. Mitchell, S. Sutthivaiyakit and N. Tongpenyai, J. Chem. Soc., Chem. Commun., 1981, 611–612, 10.1039/C39810000611; (b) Y. Watanabe, Y. Morisaki, T. Kondo and T. Mitsudo, J. Org. Chem., 1996, 61, 4214–4218, DOI:10.1021/jo9516289; (c) G. Choi and S. H. Hong, Angew. Chem., Int. Ed., 2018, 57, 6166–6170, DOI:10.1002/anie.201801524; (d) R. Liang, S. Li, R. Wang, L. Lu and F. Li, Org. Lett., 2017, 19, 5790–5793, DOI:10.1021/acs.orglett.7b02723; (e) Primary amines only F. Valot, F. Fache, R. Jacquot, M. Spagnol and M. Lemaire, Tetrahedron Lett., 1999, 40, 3689–3692, DOI:10.1016/S0040-4039(99)00588-2.
- Secondary amines only S. Savourey, G. Lefèvre, J.-C. Berthet and T. Cantat, Chem. Commun., 2014, 50, 14033–14036, 10.1039/C4CC05908E.
- (a) X. Cui, X. Dai, Y. Zhang, Y. Deng and F. Shi, Chem. Sci., 2014, 5, 649–655, 10.1039/C3SC52676C; (b) H. Zhang, Y. Zhang and K. Gao, Org. Chem. Front., 2023, 10, 2491–2497, 10.1039/D3QO00058C.
- Primary amines only S. K. Kundu, K. Mitra and A. Majee, RSC Adv., 2013, 3, 8649–8651, 10.1039/C3RA40509E.
- (a) Secondary amines only M. Magre, M. Szewczyk and M. Rueping, Org. Lett., 2020, 22, 3209–3214, DOI:10.1021/acs.orglett.0c00988; (b) I. Paul, D. A. Kisan, S. Dey, A. Sau and T. K. Panda, J. Org. Chem., 2025, 90, 10539–10544, DOI:10.1021/acs.joc.5c01045.
- For methylation of hydroxylamines, see: (a) W. R. Dunstan and E. Goulding, J. Chem. Soc., Trans., 1899, 75, 792–807, 10.1039/CT8997500792; (b) O. Schnider, A. Brossi and K. Vogler, Helv. Chim. Acta, 1954, 37, 710–720, DOI:10.1002/hlca.19540370310; (c) Y. Shvo and E. D. Kaufman, J. Org. Chem., 1981, 46, 2148–2152, DOI:10.1021/jo00323a035. For other alkylation of hydroxylamines, see: (d) J. Chen, R. Properzi, D. P. Uccello, J. A. Young, R. G. Dushin and J. T. Starr, Org. Lett., 2014, 16, 4146–4149, DOI:10.1021/ol501842b; (e) W. Kliegel and E. Ahlenstiel, J. Organomet. Chem., 1984, 277, 173–188, DOI:10.1016/0022-328X(84)80698-1; (f) W. Kliegel and J. Graumann, Liebigs Ann. Chem., 1983, 6, 950–961, DOI:10.1002/jlac.198319830608; (g) J. B. Hester Jr, A. D. Rudzik and P. F. Von Voigtlander, J. Med. Chem., 1980, 23, 392–402, DOI:10.1021/jm00178a009; (h) A. Lachman, Ber. Dtsch. Chem. Ges., 1900, 33, 1022–1030, DOI:10.1002/cber.190003301180. For epoxide reactions with hydroxylamines, see: (i) J. G. Cannon, J. G. Rose, D. E. Nerland and L. L. Darko, J. Heterocycl. Chem., 1969, 6, 747–749, DOI:10.1002/jhet.5570060525. For conjugate additions with hydroxylamines, see: (j) S. D. Pastor and E. T. Hessell, J. Org. Chem., 1988, 53, 5776–5779, DOI:10.1021/jo00259a032; (k) G. Zinner, Angew. Chem., 1959, 71, 311, DOI:10.1002/ange.19590710907; (l) Y. Du, D. Mao, D. Zhang-Negrerie and K. Zhao, Conjugate Addition of Hydroxylamines, Oximes and Hydroxamic Acids, in PATAI'S Chemistry of Functional Groups, ed. Z. Rappoport, Wiley, 2010, pp. 1–50. DOI:10.1002/9780470682531.pat0501.
- (a) J. Moran, S. I. Gorelsky, E. Dimitrijevic, M.-E. Lebrun, A.-C. Bédard, C. Séguin and A. M. Beauchemin, J. Am. Chem. Soc., 2008, 130, 17893–17906, DOI:10.1021/ja806300r; (b) M. J. MacDonald, D. J. Schipper, P. J. Ng, J. Moran and A. M. Beauchemin, J. Am. Chem. Soc., 2011, 133, 20100–20103, DOI:10.1021/ja208867g; (c) M. A. Allen, H. M. Ly, G. F. O'Keefe and A. M. Beauchemin, Chem. Sci., 2022, 13, 7264–7268, 10.1039/D2SC00481J.
- D. Barriault, H. M. Ly, M. A. Allen, M. A. Gill and A. M. Beauchemin, J. Org. Chem., 2024, 89, 8767–8772, DOI:10.1021/acs.joc.4c00674.
- H. M. Ly, H. Almeneim, M. A. Gill, J. W. Keillor and A. M. Beauchemin, Org. Lett., 2024, 26, 9885–9890, DOI:10.1021/acs.orglett.4c03688.
- A. Vörös, Z. Mucsi, Z. Baán, G. Timári, I. Hermecz, P. Mizsey and Z. Finta, Org. Biomol. Chem., 2014, 12, 8036–8047, 10.1039/C4OB00854E.
- D. Kang, S. T. Cheung, A. Wong-Rolle and J. Kim, ACS Cent. Sci., 2021, 7, 631–640, DOI:10.1021/acscentsci.0c01586.
- (a) H. P. Kokatla, P. F. Thomson, S. Bae, V. R. Doddi and M. K. Lakshman, J. Org. Chem., 2011, 76, 7842–7848, DOI:10.1021/jo201192c; (b) J. Kim and C. R. Bertozzi, Angew. Chem., Int. Ed., 2015, 54, 15777–15781, DOI:10.1002/anie.201508861; (c) D. Kang, S. Lee and J. Kim, Chem, 2022, 8, 2260–2277, DOI:10.1016/j.chempr.2022.05.018.
- H. D. Kirschman and W. R. Crowell, J. Am. Chem. Soc., 1933, 55, 488–495, DOI:10.1021/ja01329a007.
- Amines and hydroxylamines are known to interact strongly in solution, making their separation challenging. For an entry in this literature, see ref. 15.
- NMR yields were taken for entries 3b, 3c, 3d, 3m and 3p due to high volatility.
- G. Chelucci, S. Baldino and W. Baratta, Acc. Chem. Res., 2015, 48, 363–379, DOI:10.1021/ar5003818.
- O. Acevedo and W. L. Jorgensen, J. Am. Chem. Soc., 2006, 128, 6141–6146, DOI:10.1021/ja057523x.
- D. Bernier, U. K. Wefelscheid and S. Woodward, Org. Prep. Proced. Int., 2009, 41, 173–210, DOI:10.1080/00304940902955756.
- J. Körber, S. Löffler, D. Schollmeyer and U. Nubbemeyer, Synthesis, 2013, 2875–2887, DOI:10.1055/s-0033-1338519.
- (a) T. J. Donohoe, Synlett, 2002, 1223–1232, DOI:10.1055/s-2002-32947; (b) T. J. Donohoe, K. Blades, M. Helliwell, P. R. Moore, J. J. G. Winter and G. Stemp, J. Org. Chem., 1999, 64, 2980–2981, DOI:10.1021/jo982468e; (c) T. J. Donohoe, C. J. R. Bataille and P. Innocenti, Hydrogen-Bonding-Mediated Directed Osmium Dihydroxylation, in Organic Reactions, ed. S. E. Denmark, Wiley, 2012, pp. 1–48, DOI:10.1002/0471264180.or076.01.
| This journal is © The Royal Society of Chemistry 2026 |