Open Access Article
Open Access ArticleUgi multicomponent reactions of optically active aldehydes and chiral aminoindanols: diastereoselective synthesis of bisamides relevant to SARS-CoV-2 Mpro inhibitors
Arun K.
Ghosh
 *ab,
Amlan
Bhattacharjee
a,
Uttara
Jayashankar
*ab,
Amlan
Bhattacharjee
a,
Uttara
Jayashankar
 c,
Sydney N.
Bogan
c,
Sydney N.
Bogan
 d and
Andrew D.
Mesecar
acd
d and
Andrew D.
Mesecar
acd
aDepartment of Chemistry, Purdue University, 560 Oval Drive, West Lafayette, IN 47907, USA. E-mail: akghosh@purdue.edu
bDepartment of Medicinal Chemistry and Molecular Pharmacology, Purdue University, 560 Oval Drive, West Lafayette, IN 47907, USA
cDepartment of Biological Sciences, Purdue University, West Lafayette, IN 47907, USA
dDepartment of Biochemistry, Purdue University, West Lafayette, IN 47907, USA
First published on 9th March 2026
Abstract
We have investigated diastereoselective Ugi-multicomponent reactions (Ugi-MCR) relevant to bis-amide scaffolds related to SARS-CoV-2 main protease (Mpro) inhibitors. The studies were mainly focussed on development of diastereoselective tools for generation of Ugi products. Ugi reactions using either chiral aldehyde or chiral amine provided bis-amide derivatives with marginal diastereoselectivity. However, the reaction with a matched combination of (R)-isopropylidene glyceraldehyde and (1S,2R)-aminoindanol derivatives provided good diastereoselectivity, up to a dr of 91![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9. The stereochemistry of 1,2-aminoindanol plays an important role in diastereoselectivity. The stereochemical outcome of the MCR reaction was rationalized using Felkin transition state models. The Ugi-MCR products generated from these studies did not show any appreciable SARS-CoV-2 Mpro inhibitory activity.
9. The stereochemistry of 1,2-aminoindanol plays an important role in diastereoselectivity. The stereochemical outcome of the MCR reaction was rationalized using Felkin transition state models. The Ugi-MCR products generated from these studies did not show any appreciable SARS-CoV-2 Mpro inhibitory activity.
Introduction
The Ugi multicomponent reaction (Ugi-MCR) is a versatile reaction that combines an aldehyde or ketone, a primary amine, an isocyanide, and a carboxylic acid to form products known as α-acylaminoamides or bis-amides.1,2 The Ugi-MCR sets up multiple carbon–carbon and carbon-heteroatom bonds, conveniently providing molecules with structural complexities and incorporating important functionalities in a one-pot operation.3–6 Not surprisingly, Ugi reactions are often utilized in the context of drug discovery and medicinal chemistry ventures.7,8 It is widely employed in the generation of peptidomimetic derivatives for potential pharmaceutical applications and also for the preparation of compound libraries for lead generation using screening.9,10 The Ugi-MCR reaction has also been extensively utilized in a variety of fields including in the total syntheses of bioactive natural products.11–13 It was utilized in achieving the total synthesis of Ecteinascidin 743 (1, Fig. 1), a potent antitumor agent isolated from the Caribbean tunicate Ecteinascidia turbinata.14,15 The Ugi-MCR was also used in synthesizing pseudoridimycin (2), an antimicrobial agent with potent activity against both Gram-positive and Gram-negative bacteria, including drug-resistant strains.16,17 The reaction was also employed in the synthesis of (−)-FR901483 (3), a potent immunosuppressive agent extracted from the fermentation broth of Clasdobotryum sp.18,19 Besides the applications in natural product syntheses, the Ugi reactions have been extensively utilized in drug discovery and medicinal chemistry.20–22 Variations of the Ugi reaction have also been used to provide alternative and more scalable routes to medicinally significant compounds.23,24 One such example includes nirmatrelvir (4), the SARS-CoV-2 Mpro inhibitor drug recently developed and approved for the treatment of SARS-CoV-2 infection.25,26 Interestingly, it is shown that it can be accessed in a highly diastereoselective manner via a three component Ugi strategy utilizing a cyclic imine precursor.27The Ugi-MCR reaction has been utilized in accessing bisamide derivatives for the development of both noncovalent and covalent SARS-CoV-2 Mpro inhibitors.28 Mesecar and coworkers first reported the bis-amide structural platform ML-188 (5) as a non-covalent inhibitor of the SARS-CoV-1 Mpro (IC50 value 1.5 µM). They have shown that this class of compounds bind to the SARS-CoV-1 Mpro active site.28 Subsequently, bisamide scaffolds have been explored to develop potent SARS-CoV-2 Mpro inhibitors in recent years. Song and coworkers reported the Ugi-MCR approach to synthesize various epoxide-containing covalent inhibitors, describing additional mechanistic insights of this enzyme pocket.29,30 Despite efforts in synthesizing SARS-CoV-2 Mpro inhibitors utilizing the Ugi-MCR reaction, there has yet to be practical methods for synthesizing these particular compounds using asymmetric synthetic methodologies.31,32 In general, diastereoselective Ugi-MCR reactions using optically active starting materials are seldom explored.33,34 Chiral aminoindanols are widely used in various asymmetric methodologies due to their constrained architecture as well as ready availability.35,36 However, to the best of our knowledge, their application in diastereoselective Ugi reactions has not been explored. Herein, we report on the diastereoselective syntheses of bisamide derivatives employing the Ugi-MCR reaction using optically active aminoindanols paired with optically active aldehydes. Our studies particularly focused on examining diastereoselectivity associated with chiral amine and aldehyde pairs as well as generation of α-acylaminoamides with (R)-configuration.
Results and discussion
Our initial optimization efforts involved the use of (R)-isopropylidene glyceraldehyde 6 as the only optically active starting material, which can be readily synthesized from D-mannitol in 2-steps.37,38 The rationale behind the selection of this aldehyde is the fact that the inherent α-chirality on this aldehyde has been shown to direct a variety of asymmetric reactions.39,40 As shown in Scheme 1, we examined the Ugi-MCR reaction with this aldehyde in combination with equimolar amounts of 1H-imidazole-5-carboxylic acid 7, t-butyl aniline 8, and t-butyl isocyanide 9 in methanol at 23 °C to provide MCR product 10ab as a diastereomeric mixture (44![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 56) in 33% yield. The ratio of the diastereomer was determined by 1H-NMR analysis of the crude mixture after workup. The reaction was then optimized under various conditions, and the results are shown in Table 1. The reaction at 0 °C for 48 h provided a lower yield of diastereomers 10ab (entry 2) with no appreciable increase in diastereoselectivity. Running the reaction at 40 °C for 45 h (entry 3) resulted in no overall improvement over the reaction at 23 °C. Solvent screening showed that methanol is the ideal solvent for the reaction, as shown by its higher yields across the board compared to other solvents (entries 3 and 7). We examined the reaction using 2 equiv. aldehyde and 1 equiv. all other reagents which resulted in an increase of reaction yield to 67%, however the diastereoselectivity remained unchanged (entry 7). The reason for the use of 2 equiv. of aldehyde is due to the tendency of isopropylidene glyceraldehyde 6 to polymerize over time.41 We also examined this optimized reaction in 1,1,1-trifluoro ethanol as the solvent. This condition provided lower yield of products, interestingly however it improved diastereoselectivity in favor of the isomer 10a (entry 8). The diastereomers can be separated by column chromatography. To confirm the stereochemical identity, the dominant diastereomer 10b from entry 1 was converted to its nitrobenzyl derivative 10c in 65% yield by treatment of 10b with K2CO3 and 4-nitrobenzylbromide in CH3CN at 60 °C for 1.5 h. The X-ray structural analysis of 10c showed that the α-stereocenter has (R)-configuration.42,43 The ORTEP picture is shown in Fig. 2.
56) in 33% yield. The ratio of the diastereomer was determined by 1H-NMR analysis of the crude mixture after workup. The reaction was then optimized under various conditions, and the results are shown in Table 1. The reaction at 0 °C for 48 h provided a lower yield of diastereomers 10ab (entry 2) with no appreciable increase in diastereoselectivity. Running the reaction at 40 °C for 45 h (entry 3) resulted in no overall improvement over the reaction at 23 °C. Solvent screening showed that methanol is the ideal solvent for the reaction, as shown by its higher yields across the board compared to other solvents (entries 3 and 7). We examined the reaction using 2 equiv. aldehyde and 1 equiv. all other reagents which resulted in an increase of reaction yield to 67%, however the diastereoselectivity remained unchanged (entry 7). The reason for the use of 2 equiv. of aldehyde is due to the tendency of isopropylidene glyceraldehyde 6 to polymerize over time.41 We also examined this optimized reaction in 1,1,1-trifluoro ethanol as the solvent. This condition provided lower yield of products, interestingly however it improved diastereoselectivity in favor of the isomer 10a (entry 8). The diastereomers can be separated by column chromatography. To confirm the stereochemical identity, the dominant diastereomer 10b from entry 1 was converted to its nitrobenzyl derivative 10c in 65% yield by treatment of 10b with K2CO3 and 4-nitrobenzylbromide in CH3CN at 60 °C for 1.5 h. The X-ray structural analysis of 10c showed that the α-stereocenter has (R)-configuration.42,43 The ORTEP picture is shown in Fig. 2.
| Entry | Solventa | Temp. (°C) | Time (h) | Yieldb (%) | dr (10a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10b)c 10b)c |
|---|---|---|---|---|---|
| a All reactions are performed in 0.2 M MeOH relative to aldehyde. b Isolated yield of both isomers after column chromatography. c The dr was determined by 1H-NMR of crude product after workup. d Reaction utilized two equivalents of aldehyde. e TFE = 1,1,1-trifluoroethanol. | |||||
| 1 | MeOH | 23 | 36 | 33 | 44![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 56 56 |
| 2 | MeOH | 0 | 48 | 17 | 42![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 58 58 |
| 3 | MeOH | 40 | 45 | 34 | 44![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 56 56 |
| 4 | MeCN | 0–23 | 24 | 9 | 35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 65 65 |
| 5 | DCM | 0–23 | 24 | NR | N/A |
| 6 | Toluene | 0–23 | 24 | NR | N/A |
| 7d | MeOH | 0–23 | 16 | 67 | 43![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 57 57 |
| 8d | TFEe | 0–23 | 16 | 39 | 58![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 42 42 |
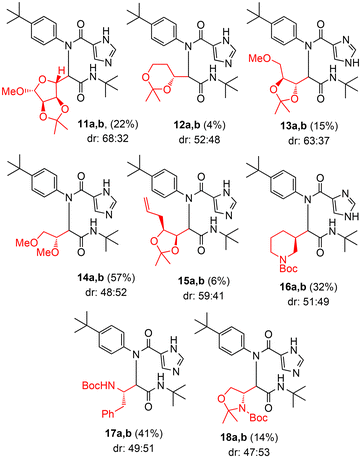
We then screened a variety of readily available cyclic and acyclic aldehydes containing an α-chiral center to see if the α-chiral center in different steric and electronic environments can influence the ratio of diastereomers. The results are shown in Scheme 2. Ugi-MCR was performed with 1 equiv. of aldehyde and equimolar amounts of amine, carboxylic acid, and isonitrile in dry methanol (0.2 M solution relative to aldehyde) from 0 °C to 23 °C for 24 h. The diastereoselectivity was determined by using 1H-NMR of crude mixture after workup, or by HPLC analysis for chromatographically inseparable mixtures. As can be seen, the diastereoselectivity of the reaction products 11–18 never breached far beyond 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, regardless of the structural complexity of the aldehyde. The yields of the Ugi-MCR products were particularly low for some hindered aldehydes. The modest yield for the Ugi adducts is possibly due to the bulky nature of the amine and aldehyde substrates.44 For compound 14, 2 equiv. of aldehyde was used. In general, the α-chiral center of these aldehydes did not show much influence on the diastereoselectivity.
1, regardless of the structural complexity of the aldehyde. The yields of the Ugi-MCR products were particularly low for some hindered aldehydes. The modest yield for the Ugi adducts is possibly due to the bulky nature of the amine and aldehyde substrates.44 For compound 14, 2 equiv. of aldehyde was used. In general, the α-chiral center of these aldehydes did not show much influence on the diastereoselectivity.
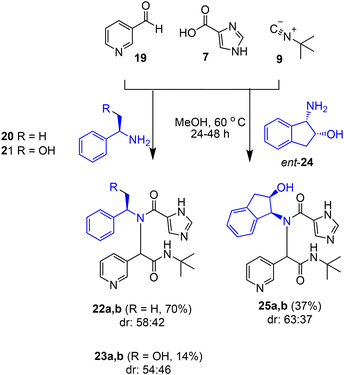
We briefly explored the diastereoselectivity associated with reactions with pyridine-3-carbaldehyde 19 and chiral acyclic amines 20 and 21 in combination with carboxylic acid 7 and isocyanide 9 as shown in Scheme 3. Ugi-MCR products 22 and 23 were obtained in 70% and 14% yields, respectively. However, diastereomeric ratios were moderate. We then investigated commercially available chiral cyclic aminoindanols. Reaction of (1S,2R)-1-amino-2-indanol ent-24 at 70 °C for 24 h resulted in product 25 as a 63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 37 mixture of diastereomers in 37% yield. The reaction was slow at 23 °C and it was necessary to heat up the reaction to 60 °C to improve product yield. This is particularly the case with less reactive aromatic aldehydes. However, the higher temperature predictably eroded the diastereoselectivity of the reaction.
37 mixture of diastereomers in 37% yield. The reaction was slow at 23 °C and it was necessary to heat up the reaction to 60 °C to improve product yield. This is particularly the case with less reactive aromatic aldehydes. However, the higher temperature predictably eroded the diastereoselectivity of the reaction.
We then explored the diastereoselectivity associated with reactions with (R)-isopropylidene glyceraldehyde 6 in combination with commercially available optically active cis-amino indanol derivatives. As shown in Scheme 4, reaction of 2 equiv. aldehyde 6, 1 equiv. (1R,2S)-1-amino-2-indanol 24, 1 equiv. imidazole carboxylic acid 7, and 2 equiv. isocyanide 9 in MeOH at 0 °C to 40 °C for 24 h resulted in MCR products 26ab as a mixture of diastereomers. In addition to distereomers 26ab, ester derivative 26c also formed in this reaction and provided 30% combined yield. The dominant diastereomer 26a was separated by chromatography, however diastereomer 26b and ester 26c were isolated as an inseparable mixture. The combined yield of 26a–c was 30%. HPLC analysis of crude reaction mixture showed the ratio of diastereomers 26a and 26b was marginal (68![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 32). The ratio of products 26a–c was 62
32). The ratio of products 26a–c was 62![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 28
28![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10. Reaction of the enantiomeric (1S,2R)-1-amino-2-indanol ent-24 under similar conditions also resulted in diastereomeric MCR products 27ab as well as ester derivative 27c in a combined isolated yield of 37%. The ratio of diastereomers 27a and 27b was determined to be 91
10. Reaction of the enantiomeric (1S,2R)-1-amino-2-indanol ent-24 under similar conditions also resulted in diastereomeric MCR products 27ab as well as ester derivative 27c in a combined isolated yield of 37%. The ratio of diastereomers 27a and 27b was determined to be 91![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9 by HPLC analysis of the crude mixture. The ratio of products 27a–c was 83
9 by HPLC analysis of the crude mixture. The ratio of products 27a–c was 83![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8
8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9. Diastereomer 27a was separated by silica gel chromatography using 5% MeOH in CH2Cl2 as the eluent. Compounds 27b and 27c could not be separated by chromatography. However, compound 27c was separated by HPLC using a TOSOH Bioscience CM-2SW column using a 10%-90% MeCN/H2O gradient. The stereochemical identity of the major diastereomer 27a and ester derivative 27c was determined by single crystal X-ray analysis.41,45,46 The ORTEP drawing is shown in Fig. 3. The Ugi-MCR reaction with (1S,2R)-1-amino-2-indanol created a new α-stereo center which was shown to have (R)-configuration for the dominant diatereomer 27a. However, the ester derivative 27c possessed (S)-configuration. It was previously shown that (R)-configuration is the biologically active configuration for SARS-CoV-2 Mpro inhibitors using this scaffold.27,28
9. Diastereomer 27a was separated by silica gel chromatography using 5% MeOH in CH2Cl2 as the eluent. Compounds 27b and 27c could not be separated by chromatography. However, compound 27c was separated by HPLC using a TOSOH Bioscience CM-2SW column using a 10%-90% MeCN/H2O gradient. The stereochemical identity of the major diastereomer 27a and ester derivative 27c was determined by single crystal X-ray analysis.41,45,46 The ORTEP drawing is shown in Fig. 3. The Ugi-MCR reaction with (1S,2R)-1-amino-2-indanol created a new α-stereo center which was shown to have (R)-configuration for the dominant diatereomer 27a. However, the ester derivative 27c possessed (S)-configuration. It was previously shown that (R)-configuration is the biologically active configuration for SARS-CoV-2 Mpro inhibitors using this scaffold.27,28
 | ||
| Fig. 3 The ORTEP diagram of compound 27a and 27c. Carbon = black, oxygen = red, and hydrogen = white. | ||
In an effort to improve yield and diastereoselectivity we further surveyed other reaction conditions as shown in Table 2. Reaction of 2 equiv. aldehyde 6 in combination with 1 equiv. amount of amine, acid and isocyanide at 0 °C to 23 °C for 24 h resulted in MCR with similar diastereomeric ratio, however the isolated yield depleted to 17% (entry 1). Modifying the reaction by increasing the amount of isocyanide to 1.5 equiv. under similar conditions led to a slight improvement of yield and diastereomeric ratio (entry 2). Further reactions with 2 equiv. and 3 equiv. of isocyanide did not improve ratio (entries 3 and 4). We also examined reactions with 2 equiv. isocyanide at 0 °C to 50 °C for 24 h and separately at 0 °C to 60 °C for 24 h. In both cases, product yields were unchanged and the ratio of diastereoselectivity was decreased (entries 6 and 7).
| Entry | Isocyanide (equiv.) | Temp. (°C) | Yielda,b (%) | drc (a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b) b) |
|---|---|---|---|---|
| a All reactions are done in 0.2 M MeOH relative to aldehyde. Aldehyde (2 equiv.), acid (1 equiv.), amine (1 equiv.) were used at specified temperature for 24 h. b Isolated yield after chromatography. c The dr obtained via HPLC analysis of mixture after chromatography. | ||||
| 1 | 1 | 0 °C to 23 °C | 17 | 89![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 11 11 |
| 2 | 1.5 | 0 °C to 23 °C | 25 | 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10 |
| 3 | 2 | 0 °C to 23 °C | 23 | 88![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12 12 |
| 4 | 3 | 0 °C to 23 °C | 30 | 87![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 13 13 |
| 5 | 2 | 0 °C to 40 °C | 37 | 91![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9 9 |
| 6 | 2 | 0 °C to 50 °C | 27 | 85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 15 |
| 7 | 2 | 0 °C to 60 °C | 28 | 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 20 |
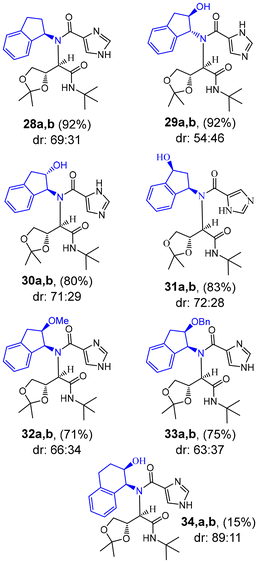
The above results show the importance of stereochemistry of chiral aminoindanols. To assess the origin of diastereoselectivity, we examined the effect of the hydroxyl functionality and the stereochemistry of the aminoindanols. The results are shown in Scheme 5. Reaction of optically active 1-(R)-aminoindane using reaction condition described in entry 5, Table 2, resulted in MCR product 28a,b in 92% yield. HPLC analysis of the crude products showed that the product contains as a (69![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 31) mixture of diastereomers. This result indicates the importance of alcohol functionality in (1S,2R)-1-amino-3-indanol ent-24. Reaction with enantiomerically pure trans-aminoindanols resulted in significant improvement of yields. However, diastereoselectivity in products 29a,b and 30a,b was reduced. Reaction of (1S,3R)-1-amino-3-indanol did not improve diastereoselectivity for the MCR product 31a,b. Interestingly, the yield of these MCR products was significantly better than the sterically hindered cis-1,2-aminoindanols. Ugi-MCR reactions with 2-methyl ether and benzyl ether also resulted in very good yield of MCR products 32a,b and 33a,b with reduction of diastereoselectivity. We also have investigated the effect of ring size using 1-(S)-amino-2(R)-hydroxy-tetrahydronaphthalene. The Ugi-MCR reaction resulted in products 34a,b in 17% yield, comparable to the reaction with cis-aminoindanol derivatives.
31) mixture of diastereomers. This result indicates the importance of alcohol functionality in (1S,2R)-1-amino-3-indanol ent-24. Reaction with enantiomerically pure trans-aminoindanols resulted in significant improvement of yields. However, diastereoselectivity in products 29a,b and 30a,b was reduced. Reaction of (1S,3R)-1-amino-3-indanol did not improve diastereoselectivity for the MCR product 31a,b. Interestingly, the yield of these MCR products was significantly better than the sterically hindered cis-1,2-aminoindanols. Ugi-MCR reactions with 2-methyl ether and benzyl ether also resulted in very good yield of MCR products 32a,b and 33a,b with reduction of diastereoselectivity. We also have investigated the effect of ring size using 1-(S)-amino-2(R)-hydroxy-tetrahydronaphthalene. The Ugi-MCR reaction resulted in products 34a,b in 17% yield, comparable to the reaction with cis-aminoindanol derivatives.
We further carried out reactions with (1S,2R) aminoindanol ent-24 in combination with various other aldehydes that have the same (R)-configuration as aldehyde 6. The results are shown in Scheme 6. As can be seen, reaction with a bulkier (R)-cyclohexylidene glyceraldehyde47 provided MCR products 35a,b in 23% yield and a diastereomeric ratio of 83![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 17. The reaction of (R)-2,2-dimethyl-1,3-dioxane-carbaldehyde48 furnished products 36a,b with good diasteroselectivity. HPLC analysis of compound mixture showed the presence of a very small amount unidentified, possibly an ester byproduct as seen previously. Reaction of (R)-2(2,2-dimethly-1,3-dioxolan-4yl) acetaldehyde49 resulted in products 37a,b in 21% yield and 2-carbaldehyde50 exhibited MCR products 38a,b with good diastereoselectivity (dr 87
17. The reaction of (R)-2,2-dimethyl-1,3-dioxane-carbaldehyde48 furnished products 36a,b with good diasteroselectivity. HPLC analysis of compound mixture showed the presence of a very small amount unidentified, possibly an ester byproduct as seen previously. Reaction of (R)-2(2,2-dimethly-1,3-dioxolan-4yl) acetaldehyde49 resulted in products 37a,b in 21% yield and 2-carbaldehyde50 exhibited MCR products 38a,b with good diastereoselectivity (dr 87![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 13). The corresponding reaction with (R)-tetrahydropyran carbaldehyde51 gave products 39a,b with similar diastereoselectivity and yield compared to tetrahydrofuranyl derivatives 38a,b. HPLC analysis of compound 39 showed the presence of a very small amount ester byproduct. Further reaction with (R)-1-benzylprrolidine-carbaldehyde52 provided MCR product 40a,b in 38% yield and showed lower diastereomeric ratio (65
13). The corresponding reaction with (R)-tetrahydropyran carbaldehyde51 gave products 39a,b with similar diastereoselectivity and yield compared to tetrahydrofuranyl derivatives 38a,b. HPLC analysis of compound 39 showed the presence of a very small amount ester byproduct. Further reaction with (R)-1-benzylprrolidine-carbaldehyde52 provided MCR product 40a,b in 38% yield and showed lower diastereomeric ratio (65![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 35) compared to reactions with tetrahydrofuran and tetrahydropyran derivatives in 38a,b and 39a,b. We also examined reaction of (R)-silyloxypropanal53 which resulted in major diastereomer 41a,b with a dr of 78
35) compared to reactions with tetrahydrofuran and tetrahydropyran derivatives in 38a,b and 39a,b. We also examined reaction of (R)-silyloxypropanal53 which resulted in major diastereomer 41a,b with a dr of 78![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 22, however the isolated yield was only 11%. The major diastereomer maintained (R) stereochemistry at the α-amino stereocenter, which was confirmed by X-ray crystallography. The ORTEP picture of the major diastereomer 41a is shown in Fig. 4.40,54 As can be seen, the newly created α-amino stereocenter possesses (R)-configuration which was shown previously as the required stereochemistry for SARS-CoV-1 Mpro inhibitory activity.27,28 Our current studies are directed towards the development of a diastereoselective Ugi-MCR reaction for the generation of biologically active SARS-CoV-2 Mpro inhibitors in a stereo predictable fashion. Due to structural similarities of many Ugi-MCR products with (R)-ML188 (5), we have evaluated all products in our SARS-CoV-2 Mpro inhibitory assays. However, none of these current derivatives show any appreciable SARS-CoV-2 Mpro inhibitory activity. The assay protocols and the results of SARS-CoV-2 Mpro inhibitory activity of these compounds are shown in the SI.
22, however the isolated yield was only 11%. The major diastereomer maintained (R) stereochemistry at the α-amino stereocenter, which was confirmed by X-ray crystallography. The ORTEP picture of the major diastereomer 41a is shown in Fig. 4.40,54 As can be seen, the newly created α-amino stereocenter possesses (R)-configuration which was shown previously as the required stereochemistry for SARS-CoV-1 Mpro inhibitory activity.27,28 Our current studies are directed towards the development of a diastereoselective Ugi-MCR reaction for the generation of biologically active SARS-CoV-2 Mpro inhibitors in a stereo predictable fashion. Due to structural similarities of many Ugi-MCR products with (R)-ML188 (5), we have evaluated all products in our SARS-CoV-2 Mpro inhibitory assays. However, none of these current derivatives show any appreciable SARS-CoV-2 Mpro inhibitory activity. The assay protocols and the results of SARS-CoV-2 Mpro inhibitory activity of these compounds are shown in the SI.
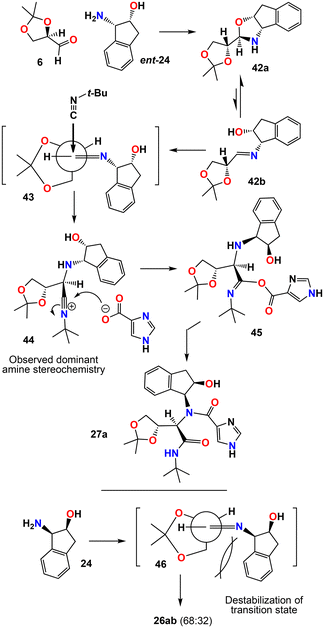
The stereochemical outcome and the reason for higher diastereoselectivity can be rationalized using models in Scheme 7. Our results show that the α-stereo center on the aldehyde and the structure and chirality on the amine play important roles in Ugi-MCR product diastereoselectivity. The degree of diastereoselection by using a chiral aldehyde or by a chiral amine alone did not result in high diastereoselectivity. However, matched combination of stereochemistry of the aldehyde and the amine resulted in Ugi-MCR products with significant diastereoselectivity. This is shown in the formation of the Ugi-MCR products between (R)-isopropylidene glyceraldehyde 6 and (1S,2R) aminoindanol ent-24. Reaction of aldehyde 6 and ent-24 showed a diastereomeric ratio of 91![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9 in comparison to reaction between aldehyde 6 and enantiomeric 24 which provided marginal diastereoselectivity. Reactions between aldehyde 6 and amine ent-24 would lead to oxazolidine and imine intermediates 42a and 42b, respectively. Presumably, the majority of the imine would exist as stable oxazolidine derivative 42a. Due to matched stereochemistry of aldehyde 6 and amine ent-24, the steric clashes are minimum for both intermediates. Presumably, subsequent reaction with isonitrile will proceed through the minor imine intermediate 42b. We postulate that the attack by the t-BuNC nucleophile proceeds through the Felkin transition state intermediate 43 where steric clashes are minimized due to complementary stereochemistry of the aldehyde and amine side chains.55,56 Subsequent reaction of imidazole carboxylate as shown in 44 leads to imine-ester intermediate 45. Subsequent Mumm rearrangement provides the major diastereomer 27a.57,58 However, the reaction of aldehyde 6 and amine 24 leads to the corresponding diastereomeric Felkin transition state 46 which is destabilized due to steric interactions between the bulky aminoindanol scaffold and the dioxolane ring of the aldehyde. This may explain the lack of diastereoselectivity for the formation of products 26ab (dr, 68
9 in comparison to reaction between aldehyde 6 and enantiomeric 24 which provided marginal diastereoselectivity. Reactions between aldehyde 6 and amine ent-24 would lead to oxazolidine and imine intermediates 42a and 42b, respectively. Presumably, the majority of the imine would exist as stable oxazolidine derivative 42a. Due to matched stereochemistry of aldehyde 6 and amine ent-24, the steric clashes are minimum for both intermediates. Presumably, subsequent reaction with isonitrile will proceed through the minor imine intermediate 42b. We postulate that the attack by the t-BuNC nucleophile proceeds through the Felkin transition state intermediate 43 where steric clashes are minimized due to complementary stereochemistry of the aldehyde and amine side chains.55,56 Subsequent reaction of imidazole carboxylate as shown in 44 leads to imine-ester intermediate 45. Subsequent Mumm rearrangement provides the major diastereomer 27a.57,58 However, the reaction of aldehyde 6 and amine 24 leads to the corresponding diastereomeric Felkin transition state 46 which is destabilized due to steric interactions between the bulky aminoindanol scaffold and the dioxolane ring of the aldehyde. This may explain the lack of diastereoselectivity for the formation of products 26ab (dr, 68![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 32). The formation of ester derivative 27c may result from the minor diastereomeric intermediate like 45. Presumably, the proximity of the hydroxyl group and its favorable stereochemistry allow a competing intramolecular attack by the free hydroxyl group on the imine-ester intermediate prior to Mumm rearrangement,57,58 resulting in the formation of ester derivative 27c.
32). The formation of ester derivative 27c may result from the minor diastereomeric intermediate like 45. Presumably, the proximity of the hydroxyl group and its favorable stereochemistry allow a competing intramolecular attack by the free hydroxyl group on the imine-ester intermediate prior to Mumm rearrangement,57,58 resulting in the formation of ester derivative 27c.
Conclusion
The Ugi reaction plays an important role in generating bis-amide derivatives for versatile applications in medicinal chemistry and drug discovery. Despite many advances in Ugi-multicomponent reactions in terms of efficiency and development of practical protocols, stereoselective generation of Ugi-MCR products is an area that is underdeveloped. We have investigated asymmetric Ugi-MCR reactions in an effort to generate bis-amide derivatives in a stereo predictable fashion. In our studies, we have demonstrated that the α-stereo center on the aldehyde or the chirality of the amine component on their own is not adequate to synthesize bis-amide derivatives diastereoselectively. However, an appropriate matched combination of chiral aldehyde and chiral amine can synergize reactions providing Ugi-MCR products with high diastereoselectivity. In particular, we showed that a combination of (R)-isopropylidene glyceraldehyde and (1S,2R)-aminoindanol provided diastereoselectivity up to a dr of 91![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9. The corresponding reaction with the (1R,2S)-aminoindanol enantiomer was less selective (dr: 68
9. The corresponding reaction with the (1R,2S)-aminoindanol enantiomer was less selective (dr: 68![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 32). While the yields were modest, the stereochemistry as well as the presence of alcohol functionality appear to play an important role in diastereoselection. We have proposed stereochemical models where the MCR may have progressed through a Felkin-type transition state that provided these diastereoselective Ugi-MCR products. All current Ugi-MCR products were evaluated in SARS-CoV-2 Mpro inhibitory assays. However, none of the current derivatives showed any appreciable SARS-CoV-2 Mpro activity. The current studies may serve to further the development of practical methods for highly diastereoselective Ugi-MCR reaction products in a stereopredictable manner. Further application of this chemistry in the development of novel SARS-CoV-2 Mpro inhibitors is in progress.
32). While the yields were modest, the stereochemistry as well as the presence of alcohol functionality appear to play an important role in diastereoselection. We have proposed stereochemical models where the MCR may have progressed through a Felkin-type transition state that provided these diastereoselective Ugi-MCR products. All current Ugi-MCR products were evaluated in SARS-CoV-2 Mpro inhibitory assays. However, none of the current derivatives showed any appreciable SARS-CoV-2 Mpro activity. The current studies may serve to further the development of practical methods for highly diastereoselective Ugi-MCR reaction products in a stereopredictable manner. Further application of this chemistry in the development of novel SARS-CoV-2 Mpro inhibitors is in progress.
Experimental section
General methods
All reagents were purchased commercially and used without further purification unless specified. Unless otherwise stated, all reactions were carried out under an Argon atmosphere. Tetrahydrofuran was dried over sodium metal and distilled before use. Anhydrous dichloromethane was prepared by distillation over calcium hydride. Anhydrous methanol was prepared by drying over 3A molecular sieves. TLC analysis was carried out using 60 A, 250 µm thick F-254 glass-backed plates. Flash column chromatography was done using 230–400 mesh silica gel. 1H and 13C NMR spectra was obtained on either a Bruker AV-III-400-HD or NEO-500. Chemical shifts are reported in ppm, J-values in Hz, and all peaks are referenced to the residual deuterated solvent peak. NMR data is reported as: δ value (chemical shift, J-value (Hz), integration, where s = singlet, d = doublet, t = triplet, q = quartet, brs = broad singlet, m = multiplet). Optical rotations were obtained using an automatic digital polarimeter with a sodium lamp and are reported as follows: [α]λ T °C (c = 0.1(mg mL−1), solvent). High-resolution mass spectrometry (HRMS) spectra were recorded under positive electron spray ionization (ESI+) at Agilent 6550 Q-TOF LC/MS instrument at the Purdue University Analytical Mass Spectrometry Facility.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 57 dr) as an amorphous solid. An analytical sample of pure diastereomers 10a and 10b was prepared after chromatographic separation.
57 dr) as an amorphous solid. An analytical sample of pure diastereomers 10a and 10b was prepared after chromatographic separation.
10a (minor isomer). White amorphous solid. Rf (5% MeOH/DCM) = 0.27. [α]23D −173.7 (c 0.27, CHCl3). 1H NMR (500 MHz, CDCl3) δ 7.66 (s, 1H), 7.46 (d, J = 8.1 Hz, 2H), 7.43–7.13 (m, 0H), 6.53 (s, 1H), 5.45 (s, 1H), 5.23 (d, J = 9.4 Hz, 1H), 4.26 (dt, J = 9.3, 6.0 Hz, 1H), 4.05 (dd, J = 9.0, 6.4 Hz, 1H), 3.72 (dd, J = 9.0, 5.7 Hz, 1H), 1.43 (s, 3H), 1.37–1.35 (m, 18H), 1.30 (s, 3H). 13C NMR (126 MHz, CDCl3) δ 168.1, 161.7, 153.7, 136.5, 135.0, 131.8, 130.0, 126.8, 125.8, 109.4, 77.4, 77.2, 76.9, 72.3, 67.6, 63.0, 51.6, 35.0, 31.5, 28.8, 27.1, 25.3. HRMS (ESI) m/z: [M + H]+ calcd for C25H37N4O4 457.2814. Found 457.2799.
10b (major isomer). White amorphous solid. Rf (5% MeOH/DCM) = 0.21 [α]23D +24.5 (c 0.49, CHCl3). 1H NMR (400 MHz, CDCl3) δ 7.59 (s, 1H), 7.42 (d, J = 8.3 Hz, 2H), 7.29 (d, J = 8.2 Hz, 2H), 6.82 (s, 1H), 5.47 (s, 1H), 4.79 (q, J = 6.2 Hz, 1H), 4.63–4.47 (m, 1H), 4.12 (dd, J = 8.6, 5.9 Hz, 1H), 3.99 (dd, J = 8.6, 5.5 Hz, 1H), 1.44 (s, 3H), 1.36–1.30 (m, 19H). 13C NMR (126 MHz, CDCl3) δ 167.0, 161.6, 153.1, 138.3, 136.8, 128.9, 127.0, 110.6, 77.4, 77.2, 76.9, 74.2, 68.0, 66.3, 51.5, 35.0, 31.5, 28.9, 27.1, 25.6. HRMS (ESI) m/z: [M + H]+ calcd for C25H37N4O4 457.2814. Found 457.2801.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 32). An analytical sample of pure diastereomers 11a and 11b was prepared after chromatographic separation.
32). An analytical sample of pure diastereomers 11a and 11b was prepared after chromatographic separation.
11a (major isomer). White amorphous solid. Rf (5% MeOH/DCM) = 0.3. [α]23D +178.6 (c 0.50, CHCl3). 1H NMR (400 MHz, CDCl3) δ 7.76–7.30 (m, 5H), 6.59 (s, 1H), 5.47 (s, 1H), 5.40 (d, J = 11.6 Hz, 1H), 5.01 (s, 1H), 4.69–4.61 (m, 2H), 4.34 (dd, J = 11.6, 0.9 Hz, 1H), 3.27 (s, 3H), 1.42 (s, 3H), 1.37 (s, 9H), 1.36 (s, 8H), 1.29 (s, 3H). 13C NMR (126 MHz, CDCl3) δ 167.8, 161.9, 153.6, 136.4, 134.1, 131.3, 126.8, 125.8, 112.5, 109.4, 84.9, 82.8, 82.7, 77.3, 77.0, 76.8, 60.9, 55.1, 51.4, 34.9, 31.4, 28.8, 26.4, 24.9. HRMS (ESI) m/z: [M + H]+ calcd for C28H41N4O6 529.3026. Found 529.3016.
11b (minor isomer). White amorphous solid. Rf (5% MeOH/DCM) = 0.27. [α]23D −84.0 (c 0.17, CHCl3). 1H NMR (400 MHz, CDCl3) δ 7.61 (s, 1H), 7.47 (d, J = 8.2 Hz, 2H), 7.36–7.26 (m, 2H), 6.52 (s, 1H), 5.39 (s, 1H), 5.20–5.15 (m, 1H), 5.03 (s, 1H), 4.88 (d, J = 6.0 Hz, 1H), 4.70–4.62 (m, 2H), 3.40 (s, 3H), 1.46 (s, 3H), 1.36 (s, 9H), 1.34 (s, 9H), 1.32 (s, 3H). 13C NMR (126 MHz, CDCl3) δ 166.6, 161.2, 153.7, 136.5, 129.9, 127.0, 113.1, 110.3, 85.2, 84.2, 82.1, 77.4, 77.2, 76.9, 56.0, 51.7, 35.1, 31.5, 28.8, 26.7, 25.3. HRMS (ESI) m/z: [M + H]+ calcd for C28H41N4O6 457.2814. Found 529.3013.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 48) as an off-white amorphous solid. Analytical samples of pure diastereomers 11a and 11b were prepared after chromatographic separation.
48) as an off-white amorphous solid. Analytical samples of pure diastereomers 11a and 11b were prepared after chromatographic separation.
12a (major isomer). White amorphous solid. Rf (5% MeOH/DCM) = 0.39. [α]23D −59.2 (c 0.60, CHCl3). 1H NMR (400 MHz, CDCl3) δ 7.62 (s, 1H), 7.44 (d, J = 7.9 Hz, 2H), 7.31 (d, J = 8.1 Hz, 2H), 6.65 (s, 1H), 5.42 (s, 1H), 4.93 (d, J = 9.4 Hz, 1H), 4.05 (t, J = 9.9 Hz, 1H), 3.91–3.73 (m, 2H), 1.70–1.52 (m, 2H), 1.35 (d, J = 1.7 Hz, 18H), 1.25 (s, 3H), 1.10 (s, 2H). 13C NMR (126 MHz, CDCl3) δ 167.7, 161.8, 153.3, 136.5, 135.4, 132.1, 130.5, 126.1, 98.7, 77.4, 77.2, 76.9, 65.4, 59.5, 51.5, 35.0, 31.5, 30.1, 28.7, 28.2, 18.6. HRMS (ESI) m/z: [M + H]+ calcd for C26H39N4O4 471.2971. Found 471.2956.
12b (minor isomer). White amorphous solid. Rf (5% MeOH/DCM) = 0.35. [α]23D +21.0 (c 0.62, CHCl3). 1H NMR (400 MHz, CDCl3) δ 7.57–7.29 (m, 4H), 7.09 (s, 1H), 5.54 (s, 1H), 4.96 (s, 1H), 4.13 (d, J = 6.8 Hz, 1H), 4.03 (t, J = 11.8 Hz, 1H), 3.90 (dd, J = 11.7, 5.1 Hz, 1H), 1.86 (d, J = 7.8 Hz, 1H), 1.75 (d, J = 12.7 Hz, 1H), 1.39 (s, 3H), 1.35 (s, 9H), 1.29 (s, 9H), 1.25 (s, 3H). 13C NMR (126 MHz, CDCl3) δ 166.5, 152.7, 140.0, 136.5, 128.8, 126.7, 98.9, 77.3, 77.0, 76.8, 69.6, 59.8, 51.1, 34.8, 31.4, 30.0, 29.7, 28.8, 28.6, 19.6. HRMS (ESI) m/z: [M + H]+ calcd for C26H39N4O4 471.2971. Found 471.2959
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 37) as a white amorphous solid. Analytical samples of pure diastereomers 13a and 13b were prepared after chromatographic separation.
37) as a white amorphous solid. Analytical samples of pure diastereomers 13a and 13b were prepared after chromatographic separation.
13a (major isomer). Off-white amorphous solid. Rf (5% MeOH/DCM) = 0.34. [α]23D −141.0 (c 0.5, CHCl3). 1H NMR (400 MHz, CDCl3) δ 7.68 (s, 1H), 7.52–7.42 (m, 4H), 6.60 (s, 1H), 5.40 (s, 1H), 5.27 (d, J = 9.4 Hz, 1H), 4.20–4.11 (m, 1H), 4.11–4.03 (m, 1H), 3.59 (dd, J = 10.4, 2.5 Hz, 1H), 3.48–3.36 (m, 1H), 3.34 (s, 3H), 1.45 (s, 3H), 1.41 (s, 3H), 1.38–1.33 (m, 18H). 13C NMR (126 MHz, CDCl3) δ 167.4, 161.6, 153.6, 136.5, 135.0, 131.5, 130.1, 126.8, 125.9, 110.3, 79.2, 77.4, 77.2, 76.9, 74.2, 73.8, 59.4, 51.7, 35.0, 31.5, 28.8, 28.7, 27.8, 27.7. HRMS (ESI) m/z: [M + H]+ calcd for C27H41N4O5 501. Found 501.3058.
13b (minor isomer). Off-white amorphous solid. Rf (5% MeOH/DCM) = 0.32. [α]23D +19.0 (c 0.40, CHCl3). 1H NMR (400 MHz, CDCl3) δ 7.68 (s, 1H), 7.61–7.30 (m, 4H), 6.94 (s, 1H), 5.53 (s, 1H), 4.80 (t, J = 7.7 Hz, 1H), 4.41–4.27 (m, 2H), 3.63–3.48 (m, 2H), 3.33 (s, 3H), 1.45 (s, 3H), 1.43 (s, 3H), 1.35 (s, 9H), 1.30 (s, 9H). 13C NMR (126 MHz, CDCl3) δ 166.2, 161.5, 153.1, 139.5, 136.7, 131.7, 129.0, 127.0, 126.0, 110.3, 79.4, 77.4, 77.2, 76.9, 76.0, 73.6, 68.2, 59.7, 51.5, 35.0, 31.5, 28.8, 27.4, 27.3. HRMS (ESI) m/z: [M + H]+ calcd for C27H41N4O5 501.3077. Found 501.3058.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 48). Rf (5% MeOH/DCM) = 0.22. The dr was determined from the mixture after column chromatography by HPLC (TOSOH BioScience CM-2SW 10–90% MeCN/H2O over 90 min). Retention times: 93.2 min, 100.2 min. Spectra of the major diastereomer: 1H NMR (500 MHz, CDCl3) δ 7.59 (d, J = 2.1 Hz, 1H), 7.44 (dd, J = 12.0, 8.4 Hz, 2H), 7.34 (d, J = 8.6 Hz, 2H), 6.74 (s, 1H), 5.49 (s, 1H), 4.56 (d, J = 8.3 Hz, 1H), 4.26 (d, J = 8.2 Hz, 1H), 3.73 (dd, J = 10.8, 2.9 Hz, 1H), 3.57 (dd, J = 10.8, 3.8 Hz, 1H), 3.43 (s, 3H), 3.40 (s, 3H), 1.34 (s, 10H), 1.31 (s, 10H). 13C NMR (126 MHz, CDCl3) δ 168.1, 167.0, 161.6, 152.8, 136.7, 136.5, 130.3, 129.2, 126.6, 79.2, 77.4, 77.2, 76.9, 76.4, 71.1, 59.5, 59.5, 51.2, 34.9, 31.5, 28.9. LRMS (ESI) m/z: [M + H]+ calcd for C24H37N4O4 445.3. Found 445.3.
48). Rf (5% MeOH/DCM) = 0.22. The dr was determined from the mixture after column chromatography by HPLC (TOSOH BioScience CM-2SW 10–90% MeCN/H2O over 90 min). Retention times: 93.2 min, 100.2 min. Spectra of the major diastereomer: 1H NMR (500 MHz, CDCl3) δ 7.59 (d, J = 2.1 Hz, 1H), 7.44 (dd, J = 12.0, 8.4 Hz, 2H), 7.34 (d, J = 8.6 Hz, 2H), 6.74 (s, 1H), 5.49 (s, 1H), 4.56 (d, J = 8.3 Hz, 1H), 4.26 (d, J = 8.2 Hz, 1H), 3.73 (dd, J = 10.8, 2.9 Hz, 1H), 3.57 (dd, J = 10.8, 3.8 Hz, 1H), 3.43 (s, 3H), 3.40 (s, 3H), 1.34 (s, 10H), 1.31 (s, 10H). 13C NMR (126 MHz, CDCl3) δ 168.1, 167.0, 161.6, 152.8, 136.7, 136.5, 130.3, 129.2, 126.6, 79.2, 77.4, 77.2, 76.9, 76.4, 71.1, 59.5, 59.5, 51.2, 34.9, 31.5, 28.9. LRMS (ESI) m/z: [M + H]+ calcd for C24H37N4O4 445.3. Found 445.3.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 41) as a yellow amorphous solid. Analytical samples of pure diastereomers 15a and 15b were prepared after chromatographic separation.
41) as a yellow amorphous solid. Analytical samples of pure diastereomers 15a and 15b were prepared after chromatographic separation.
15a (major isomer). Off-white amorphous solid. Rf (5% MeOH/DCM) = 0.44. [α]23D +131 (c 0.21, CHCl3). 1H NMR (400 MHz, CDCl3) δ 7.70–6.93 (m, 5H), 6.66 (s, 1H), 5.86 (ddt, J = 13.6, 10.1, 6.8 Hz, 1H), 5.47–5.37 (m, 2H), 5.16–5.06 (m, 2H), 4.38–4.22 (m, 1H), 4.17 (dd, J = 10.5, 5.5 Hz, 1H), 2.30 (q, J = 9.6 Hz, 1H), 2.19 (m, 1H), 1.46 (s, 3H), 1.37 (s, 9H), 1.35 (s, 9H), 1.22 (s, 3H). 13C NMR (126 MHz, CDCl3) δ 167.8, 161.7, 153.8, 136.6, 134.9, 134.2, 132.1, 127.0, 125.8, 117.3, 108.2, 77.4, 77.2, 76.9, 73.3, 58.8, 51.5, 35.5, 35.0, 31.5, 29.8, 28.7, 28.4, 25.7. HRMS (ESI) m/z: [M + H]+ calcd for C28H41N4O4 497.3128. Found 497.3110.
15b (minor isomer). Off-white amorphous solid. Rf (5% MeOH/DCM) = 0.36. [α]23D −39 (c 0.5, CHCl3). 1H NMR (500 MHz, CDCl3) δ 7.57 (s, 1H), 7.45 (d, J = 8.9 Hz, 2H), 7.41–7.02 (m, 3H), 6.72 (s, 1H), 5.92–5.80 (m, 1H), 5.40 (s, 1H), 5.21 (d, J = 9.5 Hz, 1H), 5.14–5.06 (m, 2H), 4.36 (dd, J = 9.6, 5.4 Hz, 1H), 4.13 (ddd, J = 10.8, 5.5, 3.1 Hz, 1H), 2.44–2.27 (m, 2H), 1.49 (s, 3H), 1.36 (s, 9H), 1.33 (s, 9H), 1.29 (s, 3H). 13C NMR (126 MHz, CDCl3) δ 167.1, 161.6, 153.5, 136.7, 136.2, 134.5, 129.6, 126.9, 117.7, 108.8, 77.5, 75.2, 59.8, 51.6, 35.0, 34.8, 31.5, 28.8, 28.4, 26.0. HRMS (ESI) m/z: [M + H]+ calcd for C28H41N4O4 497.3128. Found 497.3112.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 49). Rf (5% MeOH/DCM) = 0.41. The dr was determined after column chromatography by HPLC similar to 14ab. Retention times: 96.3 min, 99.3 min: spectra of dominant diastereomer: 1H NMR (400 MHz, DMSO) δ 7.69–7.07 (m, 8H), 4.97 (d, J = 10.7 Hz, 1H), 3.79 (m, 2H), 2.84–2.57 (m, 3H), 1.89 (m, 2H), 1.63 (m, 2H), 1.32 (s, 9H), 1.25 (s, 9H), 1.22 (s, 9H). 13C NMR (126 MHz, CDCl3) δ 168.2, 167.6, 161.7, 155.0, 153.2, 136.5, 129.5, 128.8, 126.9, 126.8, 79.7, 77.4, 77.2, 76.9, 53.6, 51.5, 51.5, 35.0, 34.7, 31.5, 29.8, 28.8, 28.6, 27.8, 24.2. LRMS (ESI) m/z: [M + H]+ calcd for C30H46N5O4 540.4. Found 540.3.
49). Rf (5% MeOH/DCM) = 0.41. The dr was determined after column chromatography by HPLC similar to 14ab. Retention times: 96.3 min, 99.3 min: spectra of dominant diastereomer: 1H NMR (400 MHz, DMSO) δ 7.69–7.07 (m, 8H), 4.97 (d, J = 10.7 Hz, 1H), 3.79 (m, 2H), 2.84–2.57 (m, 3H), 1.89 (m, 2H), 1.63 (m, 2H), 1.32 (s, 9H), 1.25 (s, 9H), 1.22 (s, 9H). 13C NMR (126 MHz, CDCl3) δ 168.2, 167.6, 161.7, 155.0, 153.2, 136.5, 129.5, 128.8, 126.9, 126.8, 79.7, 77.4, 77.2, 76.9, 53.6, 51.5, 51.5, 35.0, 34.7, 31.5, 29.8, 28.8, 28.6, 27.8, 24.2. LRMS (ESI) m/z: [M + H]+ calcd for C30H46N5O4 540.4. Found 540.3.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 49). Rf (5% MeOH/DCM) = 0.43. The dr was determined from the mixture after column chromatography by HPLC similar to 14. Retention times: 87.3 min, 89.8 min. Following spectra is reported where both diastereomers are normalized to one compound: 1H NMR (500 MHz, DMSO) δ 7.99–5.96 (m, 13H), 5.32–4.08 (m, 2H), 3.05–2.55 (m, 2H), 1.33 (s, 8H), 1.21 (s, 8H), 1.13 (s, 9H). 13C NMR (126 MHz, CDCl3) δ 168.2, 168.0, 166.9, 161.6, 157.0, 155.6, 153.3, 137.7, 137.1, 136.2, 130.3, 129.4, 128.6, 126.7, 79.2, 77.4, 77.2, 76.9, 61.4, 51.7, 50.9, 39.9, 35.0, 31.5, 28.5, 28.5. LRMS (ESI) m/z: [M + H]+ calcd for C33H46N5O4 576.4. Found 576.3.
49). Rf (5% MeOH/DCM) = 0.43. The dr was determined from the mixture after column chromatography by HPLC similar to 14. Retention times: 87.3 min, 89.8 min. Following spectra is reported where both diastereomers are normalized to one compound: 1H NMR (500 MHz, DMSO) δ 7.99–5.96 (m, 13H), 5.32–4.08 (m, 2H), 3.05–2.55 (m, 2H), 1.33 (s, 8H), 1.21 (s, 8H), 1.13 (s, 9H). 13C NMR (126 MHz, CDCl3) δ 168.2, 168.0, 166.9, 161.6, 157.0, 155.6, 153.3, 137.7, 137.1, 136.2, 130.3, 129.4, 128.6, 126.7, 79.2, 77.4, 77.2, 76.9, 61.4, 51.7, 50.9, 39.9, 35.0, 31.5, 28.5, 28.5. LRMS (ESI) m/z: [M + H]+ calcd for C33H46N5O4 576.4. Found 576.3.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 47) as an off-white amorphous solid. Analytical samples of pure diastereomers 18a and 18b were prepared after chromatographic separation.
47) as an off-white amorphous solid. Analytical samples of pure diastereomers 18a and 18b were prepared after chromatographic separation.
18a (major isomer). Off-white amorphous solid. Rf (5% MeOH/DCM) = 0.48. [α]23D −16.0 (c 0.5, CHCl3). 1H NMR (500 MHz, CDCl3) δ 7.55 (s, 1H), 7.43 (s, 4H), 6.39 (s, 1H), 5.61 (s, 1H), 4.85 (d, J = 5.6 Hz, 1H), 4.67 (s, 1H), 4.02 (dd, J = 9.9, 1.8 Hz, 1H), 3.76–3.68 (m, 1H), 1.55 (s, 3H), 1.44 (s, 3H), 1.40 (s, 9H), 1.34 (s, 9H), 1.32 (s, 9H). 13C NMR (126 MHz, CDCl3) δ 167.7, 161.2, 153.3, 153.0, 138.4, 136.5, 131.8, 129.4, 126.8, 126.2, 94.5, 81.0, 77.4, 77.4, 77.2, 76.9, 66.7, 65.4, 57.8, 51.8, 35.0, 31.5, 28.7, 28.4, 27.3. HRMS (ESI) m/z: [M + H]+ calcd for C30H46N5O5 556.3499. Found 556.3480.
18b (minor isomer). Off-white amorphous solid. Rf (5% MeOH/DCM) = 0.43. [α]23D −24.2 (c 0.173, CHCl3). 1H NMR (500 MHz, CDCl3) δ 7.66 (s, 1H), 7.44 (d, J = 8.4 Hz, 2H), 7.35–7.31 (m, 3H), 5.51 (s, 1H), 5.01–4.79 (m, 1H), 4.72–4.49 (m, 1H), 4.13 (d, J = 9.5 Hz, 1H), 3.90 (dd, J = 9.5, 5.2 Hz, 1H), 1.53 (s, 3H), 1.45 (sj, 9H), 1.39 (s, 3H), 1.35 (s, 9H), 1.32 (s, 9H). 13C NMR (126 MHz, CDCl3) δ 167.6, 167.3, 161.4, 153.7, 153.1, 136.9, 129.6, 128.9, 128.6, 127.0, 94.7, 81.3, 77.4, 77.2, 76.9, 66.1, 57.2, 51.6, 35.0, 31.5, 29.8, 28.8, 28.7, 28.6, 28.4. HRMS (ESI) m/z: [M + H]+ calcd for C30H46N5O5 556.3499. Found 556.3488.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 42) as a white amorphous solid. The dr was determined by HPLC of crude material after concentration using a YMC ODS-A C18 column ran at 20% MeCN/H2O (w/0.1% TFA v/v). Analytical samples of pure diastereomers 22a and 22b were prepared after chromatographic separation.
42) as a white amorphous solid. The dr was determined by HPLC of crude material after concentration using a YMC ODS-A C18 column ran at 20% MeCN/H2O (w/0.1% TFA v/v). Analytical samples of pure diastereomers 22a and 22b were prepared after chromatographic separation.
22a (major isomer). White amorphous solid. Rf (5% MeOH (NH3)/DCM) = 0.25. [α]23D +225.2 (c 0.46, CHCl3). Retention time: 9.2 min. 1H NMR (500 MHz, DMSO) δ 8.26 (dd, J = 4.7, 1.6 Hz, 1H), 8.20–7.17 (m, 10H), 7.13 (dd, J = 8.0, 4.7 Hz, 1H), 4.90–4.34 (m, 1H), 1.66 (d, J = 7.0 Hz, 3H), 1.31 (s, 9H). 13C NMR (126 MHz, DMSO) δ 169.0, 164.1, 147.7, 136.5, 135.3, 132.7, 128.2, 122.6, 61.8, 55.1, 54.9, 50.4, 40.0, 39.9, 39.9, 39.8, 39.7, 39.6, 39.5, 39.4, 39.2, 39.0, 28.2. HRMS (ESI) m/z: [M + H]+ calcd for C23H28N5O2 406.2243. Found 406.2243.
22b (minor isomer). White amorphous solid. Rf (5% MeOH (NH3)/DCM) = 0.13. [α]23D +38.0 (c 0.49, CHCl3). Retention time: 17.9 min. 1H NMR (500 MHz, DMSO) δ 8.56–8.50 (m, 1H), 8.44 (d, J = 4.8 Hz, 1H), 7.87 (s, 1H), 7.76 (s, 2H), 7.48 (d, J = 7.5 Hz, 3H), 7.38–7.32 (m, 4H), 5.27–4.56 (m, 1H), 1.60–1.53 (m, 3H), 1.07 (s, 9H). 13C NMR (126 MHz, DMSO) δ 167.3, 163.8, 149.5, 147.9, 136.4, 135.9, 135.4, 133.2, 128.5, 127.6, 123.2, 122.8, 60.7, 55.2, 50.0, 39.6, 39.5, 28.1, 17.5. HRMS (ESI) m/z: [M + H]+ calcd for C23H28N5O2 406.2243. Found 406.2247.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 46) as a white amorphous solid. Dr was obtained similar to procedure for 22ab. Trace amounts of ester byproduct are detected by HPLC. An analytical sample of pure diastereomer 23a was prepared after chromatographic separation.
46) as a white amorphous solid. Dr was obtained similar to procedure for 22ab. Trace amounts of ester byproduct are detected by HPLC. An analytical sample of pure diastereomer 23a was prepared after chromatographic separation.
23a (major isomer). White amorphous solid. Rf (10% MeOH (NH3)/DCM) = 0.41. Retention time: 15.4 min. [α]23D +2.6 (c 0.5, CHCl3). Note: NMR of this compound shows rotamerization. 1H NMR (500 MHz, DMSO) δ 8.51–8.42 (m, 2H), 7.83 (s, 1H), 7.78 (s, 1H), 7.69 (d, J = 8.1 Hz, 1H), 7.46–7.28 (m, 10H), 4.93–4.86 (m, 1H), 4.27 (dd, J = 11.3, 4.6 Hz, 1H), 4.22–4.15 (m, 1H), 4.04–3.94 (m, 2H), 3.63–3.50 (m, 1H), 1.23 (s, 9H). 13C NMR (126 MHz, DMSO) δ 170.5, 160.7, 148.4, 148.2, 140.8, 136.0, 134.2, 128.9, 128.5, 128.2, 128.1, 127.7, 126.9, 123.3, 64.6, 60.7, 59.9, 50.3, 40.0, 39.9, 39.9, 39.8, 39.7, 39.5, 39.4, 39.2, 39.0, 28.3. HRMS (ESI) m/z: [M + H]+ calcd for C23H28N5O3 422.2192. Found 422.2189.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 37) as a white amorphous solid. Dr was determined by crude 1H-NMR. An analytical sample of the pure dominant diastereomer 25a was prepared after chromatographic separation.
37) as a white amorphous solid. Dr was determined by crude 1H-NMR. An analytical sample of the pure dominant diastereomer 25a was prepared after chromatographic separation.
25a (major isomer). Amber amorphous solid. Rf (5% MeOH/DCM) = 0.19. 1H NMR (500 MHz, CDCl3) δ 8.69–8.65 (m, 1H), 8.55 (d, J = 1.7 Hz, 1H), 7.83 (d, J = 7.8 Hz, 1H), 7.70 (s, 1H), 7.64 (s, 1H), 7.39–7.35 (m, 1H), 7.31 (dd, J = 7.9, 4.9 Hz, 1H), 7.25–7.22 (m, 2H), 6.59 (s, 1H), 5.71 (q, J = 2.7 Hz, 1H), 4.54 (s, 1H), 4.28 (d, J = 5.4 Hz, 1H), 3.25 (qd, J = 17.1, 4.5 Hz, 2H), 1.30 (s, 9H), 1.27–1.24 (m, 2H). 13C NMR (126 MHz, CDCl3) δ 170.7, 149.3, 148.8, 141.9, 139.3, 135.4, 135.3, 129.7, 128.4, 127.8, 127.2, 126.4, 125.7, 125.2, 124.1, 123.9, 75.5, 64.1, 62.6, 51.4, 36.9, 28.6. HRMS (ESI) m/z: [M + H]+ calcd for C24H28N5O3 434.2192. Found 434.2178.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 32) as a white amorphous solid. HPLC analysis of crude mixture using a TOSOH BioScience CM-2SW column with a gradient of 10–90% MeCN/H2O over 90 min, flow rate 0.5 mL min−1 determined the dr of 26ab. Ratio of 26abc based on crude HPLC is 62
32) as a white amorphous solid. HPLC analysis of crude mixture using a TOSOH BioScience CM-2SW column with a gradient of 10–90% MeCN/H2O over 90 min, flow rate 0.5 mL min−1 determined the dr of 26ab. Ratio of 26abc based on crude HPLC is 62![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 28
28![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10. An analytical sample of 26a was prepared after chromatographic separation, whilst 26b and ester byproduct 26c were chromatographically inseparable.
10. An analytical sample of 26a was prepared after chromatographic separation, whilst 26b and ester byproduct 26c were chromatographically inseparable.
26a (major isomer). Retention time: 39.9 min. Rf (5% MeOH/DCM) = 0.45. [α]23D −145.9 (c 0.49, CHCl3) 1H NMR (500 MHz, DMSO) δ 10.06 (s, 1H), 8.04 (d, J = 1.2 Hz, 1H), 7.77 (s, 1H), 7.25 (d, J = 7.1 Hz, 2H), 7.13 (dt, J = 19.8, 7.3 Hz, 2H), 6.91 (d, J = 7.3 Hz, 1H), 5.88 (d, J = 6.6 Hz, 1H), 5.21 (d, J = 9.7 Hz, 1H), 4.98 (d, J = 5.1 Hz, 1H), 4.69 (ddd, J = 10.0, 6.2, 3.9 Hz, 1H), 4.60 (m, 1H), 4.09 (dd, J = 9.1, 6.3 Hz, 1H), 3.54 (dd, J = 9.1, 3.9 Hz, 1H), 3.05 (dd, J = 16.1, 4.8 Hz, 1H), 2.88 (d, J = 16.1 Hz, 1H), 1.32 (s, 9H), 1.26 (s, 3H), 1.00 (s, 3H). 13C NMR (126 MHz, DMSO) δ 168.2, 166.3, 139.2, 138.9, 135.6, 135.3, 126.9, 125.8, 125.5, 123.9, 122.3, 108.9, 75.9, 73.5, 66.6, 65.4, 64.1, 50.4, 41.5, 40.0, 39.9, 39.9, 39.7, 39.5, 39.4, 39.2, 39.0, 28.2, 26.6, 25.3. HRMS (ESI) m/z: [M + H]+ calcd for C24H33N4O5 457.2451. Found 457.2440.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9) as a white amorphous solid. Dr of 27ab was determined from the crude mixture following the same procedure for 26ab. Ester derivative 27c was separated by HPLC. An analytical sample of pure diastereomer 27a was prepared after chromatographic separation. Data for 27b is reported based on the mixture of 27bc.
9) as a white amorphous solid. Dr of 27ab was determined from the crude mixture following the same procedure for 26ab. Ester derivative 27c was separated by HPLC. An analytical sample of pure diastereomer 27a was prepared after chromatographic separation. Data for 27b is reported based on the mixture of 27bc.
27a (major isomer). White amorphous solid. Rf (5% MeOH/DCM) = 0.33. [α]23D +57.8 (c 0.49, CHCl3). Retention time: 29.7 min. 1H NMR (500 MHz, DMSO) δ 9.80 (s, 1H), 8.04 (s, 1H), 7.79 (s, 1H), 7.23 (d, J = 7.4 Hz, 1H), 7.11 (dt, J = 22.6, 7.4 Hz, 2H), 6.86 (d, J = 7.4 Hz, 1H), 5.38 (d, J = 9.2 Hz, 1H), 5.30 (d, J = 9.8 Hz, 1H), 5.09 (d, J = 5.9 Hz, 1H), 4.80 (ddd, J = 9.9, 6.0, 4.0 Hz, 1H), 4.46 (dt, J = 10.7, 5.6 Hz, 1H), 4.09 (dd, J = 9.2, 6.0 Hz, 1H), 3.72 (dd, J = 9.3, 3.9 Hz, 1H), 3.13 (dd, J = 16.6, 5.6 Hz, 1H), 2.92 (d, J = 16.5 Hz, 1H), 1.36 (s, 10H), 1.26 (s, 3H), 1.21 (s, 3H). 13C NMR (126 MHz, DMSO) δ 167.7, 165.9, 139.0, 135.8, 134.9, 127.0, 126.0, 125.3, 124.7, 122.4, 108.9, 75.0, 73.2, 65.6, 64.7, 61.9, 50.3, 41.7, 28.4, 26.9, 25.1. HRMS (ESI) m/z: [M + H]+ calcd for C24H33N4O5 457.2451. Found 457.2432.
27b (minor isomer). Retention time 52.7 min. NMR peaks based on mixture of 27bc: 1H NMR (500 MHz, DMSO) δ 7.86 (d, J = 23.6 Hz, 1H), 7.60 (d, J = 14.6 Hz, 1H), 7.35–7.26 (m, 3H), 7.22–7.09 (m, 2H), 4.85–4.69 (m, 1H), 4.55 (d, J = 28.3 Hz, 2H), 4.03–3.94 (m, 1H), 3.26–3.14 (m, 2H), 3.14–3.04 (m, 2H), 2.84 (dd, J = 15.8, 9.5 Hz, 1H), 1.34 (s, 3H), 1.27 (s, 3H), 1.23 (s, 9H). 13C NMR (126 MHz, DMSO) δ 168.1, 141.5, 136.7, 132.0, 129.5, 129.2, 126.8, 126.5, 124.8, 123.6, 106.7, 73.4, 69.5, 63.9, 63.2, 50.1, 37.5, 34.2, 28.4, 28.2, 26.2. LRMS (ESI) m/z: [M + H]+ calcd for C24H33N4O5 457.3 Found 457.2.
Compound 27c. White amorphous solid. [α]23D −76.3 (c 0.2, CHCl3) Retention time: 55.2 min. 1H NMR (500 MHz, DMSO) δ 7.76–7.70 (m, 2H), 7.56 (s, 1H), 7.46–7.40 (m, 1H), 7.25 (m, 4H), 5.66–5.60 (m, 1H), 4.19 (s, 1H), 4.09 (d, J = 6.7 Hz, 1H), 3.84 (dd, J = 8.5, 6.3 Hz, 1H), 3.79–3.72 (m, 1H), 3.26–3.12 (m, 3H), 3.05–2.89 (m, 1H), 2.64 (m, 1H), 1.22 (s, 9H), 1.09 (s, 3H), 0.98 (s, 3H). 13C NMR (126 MHz, DMSO) δ 169.7, 142.8, 139.6, 127.7, 126.7, 124.9, 123.9, 108.4, 76.3, 74.0, 65.9, 64.0, 63.0, 49.8, 36.9, 28.3, 25.7, 25.1. LRMS (ESI) m/z: [M + H]+ calcd for C24H33N4O5 457.3 Found 457.2.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 31), which was isolated as a chromatographically inseparable mixture of diastereomers as a white amorphous solid. Rf (5% MeOH/DCM) = 0.4. Retention times: 32.2 min, 46.4 min. Spectra values that follow are of the major diastereomer 28a. 1H NMR (500 MHz, DMSO) δ 9.50 (s, 1H), 7.93 (s, 1H), 7.59 (s, 1H), 7.15 (d, J = 7.2 Hz, 1H), 7.12–7.05 (m, 2H), 7.02 (t, J = 7.3 Hz, 1H), 6.96 (d, J = 7.5 Hz, 1H), 5.26 (d, J = 9.7 Hz, 1H), 5.05 (t, J = 8.4 Hz, 1H), 4.78 (ddd, J = 10.1, 6.2, 4.1 Hz, 1H), 4.05 (dd, J = 8.6, 6.2 Hz, 1H), 3.54 (dd, J = 8.8, 4.2 Hz, 1H), 3.06–2.98 (m, 1H), 2.87–2.77 (m, 2H), 2.41 (ddd, J = 17.7, 10.0, 5.9 Hz, 1H), 2.33–2.25 (m, 1H), 1.40 (s, 9H), 1.25 (s, 3H), 1.20 (s, 3H). 13C NMR (126 MHz, DMSO) δ 168.3, 162.7, 142.6, 142.2, 136.2, 135.0, 126.4, 125.5, 124.2, 122.8, 122.1, 109.0, 73.1, 65.4, 61.5, 60.7, 50.2, 40.1, 40.0, 39.9, 39.9, 39.8, 39.7, 39.5, 39.4, 39.2, 39.0, 30.3, 29.8, 28.5, 26.8, 25.0. LRMS (ESI) m/z: [M + H]+ calcd for C24H33N4O4 441.3. Found 441.2.
31), which was isolated as a chromatographically inseparable mixture of diastereomers as a white amorphous solid. Rf (5% MeOH/DCM) = 0.4. Retention times: 32.2 min, 46.4 min. Spectra values that follow are of the major diastereomer 28a. 1H NMR (500 MHz, DMSO) δ 9.50 (s, 1H), 7.93 (s, 1H), 7.59 (s, 1H), 7.15 (d, J = 7.2 Hz, 1H), 7.12–7.05 (m, 2H), 7.02 (t, J = 7.3 Hz, 1H), 6.96 (d, J = 7.5 Hz, 1H), 5.26 (d, J = 9.7 Hz, 1H), 5.05 (t, J = 8.4 Hz, 1H), 4.78 (ddd, J = 10.1, 6.2, 4.1 Hz, 1H), 4.05 (dd, J = 8.6, 6.2 Hz, 1H), 3.54 (dd, J = 8.8, 4.2 Hz, 1H), 3.06–2.98 (m, 1H), 2.87–2.77 (m, 2H), 2.41 (ddd, J = 17.7, 10.0, 5.9 Hz, 1H), 2.33–2.25 (m, 1H), 1.40 (s, 9H), 1.25 (s, 3H), 1.20 (s, 3H). 13C NMR (126 MHz, DMSO) δ 168.3, 162.7, 142.6, 142.2, 136.2, 135.0, 126.4, 125.5, 124.2, 122.8, 122.1, 109.0, 73.1, 65.4, 61.5, 60.7, 50.2, 40.1, 40.0, 39.9, 39.9, 39.8, 39.7, 39.5, 39.4, 39.2, 39.0, 30.3, 29.8, 28.5, 26.8, 25.0. LRMS (ESI) m/z: [M + H]+ calcd for C24H33N4O4 441.3. Found 441.2.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 46) as a white amorphous solid. Dr determined by HPLC of crude similar to 26ab. Analytical samples of pure diasteromers 29a and 29b were prepared after chromatographic separation.
46) as a white amorphous solid. Dr determined by HPLC of crude similar to 26ab. Analytical samples of pure diasteromers 29a and 29b were prepared after chromatographic separation.
29a (major isomer). White amorphous solid. Rf (5% MeOH/DCM) = 0.22. [α]23D +19.0 (c 0.50, CHCl3). Retention time: 47.1 min. 1H NMR (500 MHz, CDCl3) δ 7.54 (s, 1H), 7.49 (s, 1H), 7.37–7.15 (m, 4H), 6.85 (s, 1H), 6.28 (s, 1H), 5.17 (q, J = 6.5 Hz, 1H), 4.53 (q, J = 6.7 Hz, 1H), 4.10 (dd, J = 8.9, 6.2 Hz, 1H), 3.99 (dd, J = 8.8, 6.0 Hz, 1H), 3.47 (d, J = 7.5 Hz, 1H), 3.20 (dd, J = 15.7, 7.2 Hz, 1H), 2.85 (dd, J = 15.7, 7.1 Hz, 1H), 1.41–1.37 (m, 2H), 1.34 (s, 14H), 1.17 (s, 3H). 13C NMR (126 MHz, CDCl3) δ 169.8, 166.3, 141.2, 138.0, 135.5, 128.8, 127.5, 125.7, 125.6, 110.5, 76.6, 76.4, 71.9, 67.6, 62.6, 51.7, 37.3, 28.8, 27.0, 25.7. HRMS (ESI) m/z: [M + H]+ calcd for C24H33N4O5 457.2451. Found 457.2432.
29b (minor isomer). White amorphous solid. Rf (5% MeOH/DCM) = 0.32. [α]23D −276.8 (c 0.50, CHCl3). Retention time: 29.0 min 1H NMR (500 MHz, CDCl3) δ 11.78 (s, 1H), 9.78 (s, 1H), 7.34 (s, 1H), 7.29–7.24 (m, 1H), 7.20 (t, J = 7.5 Hz, 1H), 7.04 (t, J = 7.5 Hz, 1H), 6.93 (d, J = 7.5 Hz, 1H), 6.56 (s, 1H), 5.53 (q, J = 7.8 Hz, 1H), 5.31 (d, J = 10.1 Hz, 1H), 5.08 (d, J = 6.5 Hz, 1H), 4.99 (ddd, J = 9.8, 6.5, 3.0 Hz, 1H), 4.22 (dd, J = 9.4, 6.4 Hz, 1H), 4.01 (s, 1H), 3.78 (dd, J = 9.4, 2.9 Hz, 1H), 3.42 (dd, J = 16.0, 8.3 Hz, 1H), 2.95 (dd, J = 16.0, 8.1 Hz, 1H), 1.41 (s, 9H), 1.35 (s, 3H), 1.14 (s, 3H). 13C NMR (126 MHz, CDCl3) δ 168.4, 165.5, 140.8, 140.0, 136.6, 135.1, 127.2, 126.4, 125.2, 122.2, 122.2, 110.4, 76.6, 73.8, 70.1, 67.5, 63.2, 51.3, 38.3, 28.8, 27.0, 25.3. HRMS (ESI) m/z: [M + H]+ calcd for C24H33N4O5 457.2451. Found 457.2435.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 29) as an inseparable mixture of diastereomers as a white amorphous solid. Dr obtained after silica gel chromatography by HPLC, similar to that of 26ab. Rf (5% MeOH/DCM) = 0.20. Retention times: 24.0 min, 42.7 min. Spectra of dominant diastereomer 30a: 1H NMR (400 MHz, DMSO) δ 9.43 (s, 1H), 7.94 (s, 1H), 7.63–7.58 (m, 1H), 7.13–6.98 (m, 3H), 6.91 (d, J = 7.4 Hz, 1H), 5.36 (d, J = 9.8 Hz, 1H), 5.23 (d, J = 7.0 Hz, 1H), 4.89 (p, J = 6.8 Hz, 1H), 4.85–4.73 (m, 2H), 4.05 (dd, J = 8.9, 5.9 Hz, 1H), 3.59 (dd, J = 8.9, 3.6 Hz, 1H), 3.30–3.24 (m, 1H), 2.69 (dd, J = 16.0, 6.3 Hz, 1H), 1.39 (s, 9H), 1.22 (s, 3H), 1.20 (s, 3H). 13C NMR (126 MHz, DMSO) δ 168.3, 163.1, 140.8, 139.9, 136.2, 135.0, 126.7, 125.8, 124.2, 122.9, 122.2, 108.7, 75.4, 73.8, 68.8, 65.9, 60.9, 50.2, 40.3, 28.5, 27.0, 25.3. LRMS (ESI) m/z: [M + H]+ calcd for C24H33N4O5 457.3. Found 457.2.
29) as an inseparable mixture of diastereomers as a white amorphous solid. Dr obtained after silica gel chromatography by HPLC, similar to that of 26ab. Rf (5% MeOH/DCM) = 0.20. Retention times: 24.0 min, 42.7 min. Spectra of dominant diastereomer 30a: 1H NMR (400 MHz, DMSO) δ 9.43 (s, 1H), 7.94 (s, 1H), 7.63–7.58 (m, 1H), 7.13–6.98 (m, 3H), 6.91 (d, J = 7.4 Hz, 1H), 5.36 (d, J = 9.8 Hz, 1H), 5.23 (d, J = 7.0 Hz, 1H), 4.89 (p, J = 6.8 Hz, 1H), 4.85–4.73 (m, 2H), 4.05 (dd, J = 8.9, 5.9 Hz, 1H), 3.59 (dd, J = 8.9, 3.6 Hz, 1H), 3.30–3.24 (m, 1H), 2.69 (dd, J = 16.0, 6.3 Hz, 1H), 1.39 (s, 9H), 1.22 (s, 3H), 1.20 (s, 3H). 13C NMR (126 MHz, DMSO) δ 168.3, 163.1, 140.8, 139.9, 136.2, 135.0, 126.7, 125.8, 124.2, 122.9, 122.2, 108.7, 75.4, 73.8, 68.8, 65.9, 60.9, 50.2, 40.3, 28.5, 27.0, 25.3. LRMS (ESI) m/z: [M + H]+ calcd for C24H33N4O5 457.3. Found 457.2.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 28) as an chromatographically inseparable mixture of diastereomers as a white amorphous solid. Rf (5% MeOH/DCM) = 0.40. Retention times: 26.3 min, 40.9 min. Spectral values shown is that of the major diastereomer 31a: 1H NMR (400 MHz, DMSO) δ 9.53 (s, 1H), 7.95 (s, 1H), 7.62 (s, 1H), 7.32 (d, J = 7.1 Hz, 1H), 7.14 (dt, J = 20.4, 7.4 Hz, 3H), 6.95 (d, J = 7.3 Hz, 1H), 5.39 (d, J = 6.9 Hz, 1H), 5.25 (d, J = 9.7 Hz, 1H), 4.98–4.90 (m, 1H), 4.79 (t, J = 8.1 Hz, 2H), 4.08 (tt, J = 14.5, 7.5 Hz, 1H), 3.53 (dd, J = 8.8, 4.0 Hz, 1H), 2.67–2.55 (m, 1H), 2.48–2.38 (m, 1H), 1.38 (s, 9H), 1.25 (s, 3H), 1.20 (s, 3H). 13C NMR (126 MHz, DMSO) δ 168.2, 163.0, 144.5, 141.4, 136.1, 135.1, 126.9, 126.6, 123.9, 122.9, 121.8, 109.1, 73.1, 71.6, 65.3, 61.5, 57.0, 50.2, 41.2, 28.5, 26.8, 25.1. LRMS (ESI) m/z: [M + H]+ calcd for C24H33N4O5 457.3. Found 457.2.
28) as an chromatographically inseparable mixture of diastereomers as a white amorphous solid. Rf (5% MeOH/DCM) = 0.40. Retention times: 26.3 min, 40.9 min. Spectral values shown is that of the major diastereomer 31a: 1H NMR (400 MHz, DMSO) δ 9.53 (s, 1H), 7.95 (s, 1H), 7.62 (s, 1H), 7.32 (d, J = 7.1 Hz, 1H), 7.14 (dt, J = 20.4, 7.4 Hz, 3H), 6.95 (d, J = 7.3 Hz, 1H), 5.39 (d, J = 6.9 Hz, 1H), 5.25 (d, J = 9.7 Hz, 1H), 4.98–4.90 (m, 1H), 4.79 (t, J = 8.1 Hz, 2H), 4.08 (tt, J = 14.5, 7.5 Hz, 1H), 3.53 (dd, J = 8.8, 4.0 Hz, 1H), 2.67–2.55 (m, 1H), 2.48–2.38 (m, 1H), 1.38 (s, 9H), 1.25 (s, 3H), 1.20 (s, 3H). 13C NMR (126 MHz, DMSO) δ 168.2, 163.0, 144.5, 141.4, 136.1, 135.1, 126.9, 126.6, 123.9, 122.9, 121.8, 109.1, 73.1, 71.6, 65.3, 61.5, 57.0, 50.2, 41.2, 28.5, 26.8, 25.1. LRMS (ESI) m/z: [M + H]+ calcd for C24H33N4O5 457.3. Found 457.2.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 34) as a white amorphous solid. Dr determined by HPLC of crude similar to 26ab. Analytical samples of pure diastereomers 32a and 32b were prepared after chromatographic separation.
34) as a white amorphous solid. Dr determined by HPLC of crude similar to 26ab. Analytical samples of pure diastereomers 32a and 32b were prepared after chromatographic separation.
32a (major isomer). White amorphous solid. Rf (5% MeOH/DCM) = 0.45. [α]23D +208.8 (c 0.50, CHCl3). Retention time: 52.6 min. 1H NMR (500 MHz, DMSO) δ 9.31 (s, 1H), 7.86 (s, 1H), 7.58 (s, 1H), 7.15–7.08 (m, 2H), 7.08–7.02 (m, 2H), 5.42 (d, J = 9.9 Hz, 1H), 5.27 (d, J = 9.0 Hz, 1H), 4.53 (dd, J = 7.8, 5.6 Hz, 1H), 4.20 (q, J = 7.5 Hz, 1H), 3.87 (dd, J = 9.4, 5.7 Hz, 1H), 3.75 (d, J = 9.4 Hz, 1H), 3.19 (qd, J = 14.8, 7.5 Hz, 2H), 1.42 (s, 9H), 1.24 (s, 3H), 1.21 (s, 3H). 13C NMR (126 MHz, DMSO) δ 169.0, 162.1, 141.8, 140.0, 136.5, 134.6, 127.2, 126.1, 124.1, 123.4, 122.9, 108.4, 79.0, 74.1, 66.1, 61.1, 59.5, 56.9, 50.0, 40.1, 40.0, 39.9, 39.9, 39.8, 39.7, 39.5, 39.4, 39.2, 39.0, 38.0, 28.6, 27.4, 25.2. HRMS (ESI) m/z: [M + H]+ calcd for C25H35N4O5 471.2608. Found 471.2590.
32b (minor isomer). White amorphous solid. Exists as 2 equal rotamers at 25 °C, signals merge at 125 °C in DMSO-d6. 1H NMR. Rf (5% MeOH/DCM) = 0.4. [α]23D −69.8 (c 0.50, CHCl3). Retention time: 60.4 min. 1H NMR (400 MHz, DMSO, 125 °C) δ 7.69 (s, 1H), 7.52 (d, J = 7.5 Hz, 1H), 7.22 (s, 3H), 4.72 (q, J = 7.2 Hz, 1H), 4.21–4.15 (m, 1H), 4.06–4.02 (m, 1H), 3.49–3.03 (m, 6H), 2.97–2.88 (m, 2H), 1.32 (s, 9H), 1.23 (s, 2H), 1.09 (s, 3H). 13C NMR (126 MHz, DMSO, 25 °C) δ 168.64, 167.69, 164.63, 164.34, 140.82, 140.70, 140.64, 138.34, 137.19, 137.14, 134.99, 134.40, 128.68, 127.94, 126.79, 126.74, 125.95, 125.03, 124.86, 124.02, 123.57, 121.88, 108.61, 107.26, 81.93, 79.19, 73.90, 73.18, 68.21, 66.97, 64.18, 63.57, 61.97, 60.24, 57.32, 56.80, 50.10, 49.84, 38.23, 35.61, 28.40, 28.28, 26.05, 25.97, 25.38, 24.60. Please note that 13C at 25 °C shows a mixture of rotamers. HRMS (ESI) m/z: [M + H]+ calcd for C25H35N4O5 471.2608. Found 471.2594.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 37) as a chromatographically inseparable mixture of diastereomers as a white amorphous solid. Dr determined based on crude residue by HPLC similar to 26ab. Rf (5% MeOH/DCM) = 0.41. Retention times: 51.2 min, 61.3 min. Spectral values are that of the major diastereomer. 1H NMR (500 MHz, DMSO) δ 9.25 (s, 1H), 7.86 (s, 1H), 7.59 (s, 1H), 7.41–7.00 (m, 10H), 5.39 (d, J = 9.8 Hz, 1H), 5.29 (d, J = 8.9 Hz, 1H), 4.69–4.58 (m, 1H), 4.56–4.38 (m, 4H), 3.57–3.47 (m, 1H), 3.22–3.03 (m, 2H), 1.41 (s, 9H), 1.18 (s, 3H), 1.12 (s, 3H). 13C NMR (126 MHz, DMSO) δ 169.1, 162.3, 141.7, 140.0, 138.5, 137.8, 136.6, 134.6, 128.1, 127.7, 127.5, 127.4, 126.3, 126.0, 124.1, 123.4, 122.5, 108.2, 77.0, 73.8, 70.5, 66.1, 61.0, 59.7, 50.0, 39.8, 38.3, 28.6, 27.8, 27.2. LRMS (ESI) m/z: [M + H]+ calcd for C31H39N4O5 547.3. Found 547.3.
37) as a chromatographically inseparable mixture of diastereomers as a white amorphous solid. Dr determined based on crude residue by HPLC similar to 26ab. Rf (5% MeOH/DCM) = 0.41. Retention times: 51.2 min, 61.3 min. Spectral values are that of the major diastereomer. 1H NMR (500 MHz, DMSO) δ 9.25 (s, 1H), 7.86 (s, 1H), 7.59 (s, 1H), 7.41–7.00 (m, 10H), 5.39 (d, J = 9.8 Hz, 1H), 5.29 (d, J = 8.9 Hz, 1H), 4.69–4.58 (m, 1H), 4.56–4.38 (m, 4H), 3.57–3.47 (m, 1H), 3.22–3.03 (m, 2H), 1.41 (s, 9H), 1.18 (s, 3H), 1.12 (s, 3H). 13C NMR (126 MHz, DMSO) δ 169.1, 162.3, 141.7, 140.0, 138.5, 137.8, 136.6, 134.6, 128.1, 127.7, 127.5, 127.4, 126.3, 126.0, 124.1, 123.4, 122.5, 108.2, 77.0, 73.8, 70.5, 66.1, 61.0, 59.7, 50.0, 39.8, 38.3, 28.6, 27.8, 27.2. LRMS (ESI) m/z: [M + H]+ calcd for C31H39N4O5 547.3. Found 547.3.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 11) as a white amorphous solid. Dr determined by HPLC of crude similar to 26ab. The minor diastereomer coelutes with a chromatographically inseparable impurity.
11) as a white amorphous solid. Dr determined by HPLC of crude similar to 26ab. The minor diastereomer coelutes with a chromatographically inseparable impurity.
34a (major isomer). White amorphous solid. Rf (5% MeOH/DCM) = 0.39. [α]23D +163.8 (c 0.50, CHCl3). Retention time: 25.2 min. 1H NMR (400 MHz, DMSO) δ 9.73 (s, 1H), 8.06 (d, J = 1.2 Hz, 1H), 7.83 (d, J = 1.2 Hz, 1H), 7.24–6.90 (m, 5H), 6.14 (d, J = 3.2 Hz, 1H), 5.42 (d, J = 9.9 Hz, 1H), 5.00 (d, J = 3.5 Hz, 1H), 4.74 (ddd, J = 9.7, 5.9, 2.8 Hz, 1H), 4.15–3.99 (m, 2H), 3.72 (dd, J = 9.0, 2.9 Hz, 1H), 3.06 (td, J = 15.0, 5.4 Hz, 1H), 2.57 (dd, J = 16.6, 5.0 Hz, 1H), 1.95 (s, 1H), 1.84 (tq, J = 13.2, 5.8 Hz, 1H), 1.34 (s, 9H), 1.24 (s, 3H), 1.22 (s, 3H). 13C NMR (126 MHz, DMSO) δ 166.9, 166.5, 136.7, 135.9, 134.8, 133.2, 128.6, 126.1, 125.5, 125.2, 125.2, 109.1, 73.4, 68.0, 65.2, 61.7, 60.8, 50.3, 29.6, 28.4, 27.1, 25.1, 23.0. HRMS (ESI) m/z: [M + H]+ calcd for C25H35N4O5 471.2608. Found 471.2594.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 17) as a white amorphous solid. Dr was determined via HPLC of crude similar to 26ab. An analytical sample of pure diasteromer 35a was prepared after chromatographic separation.
17) as a white amorphous solid. Dr was determined via HPLC of crude similar to 26ab. An analytical sample of pure diasteromer 35a was prepared after chromatographic separation.
35a (major isomer). White amorphous solid. Rf (5% MeOH/DCM) = 0.48. [α]23D +120.2 (c 0.47, CHCl3). Retention time: 36.0 min. 1H NMR (500 MHz, DMSO) δ 9.70 (s, 1H), 8.03 (s, 1H), 7.77 (s, 1H), 7.22 (d, J = 7.0 Hz, 2H), 7.11 (dt, J = 23.6, 7.5 Hz, 2H), 6.86 (d, J = 7.4 Hz, 1H), 5.33 (dd, J = 9.8, 6.5 Hz, 2H), 5.12 (t, J = 4.8 Hz, 1H), 4.79 (ddd, J = 9.9, 5.9, 3.6 Hz, 1H), 4.49–4.39 (m, 1H), 4.06 (dd, J = 9.1, 5.9 Hz, 1H), 3.74 (dd, J = 9.7, 3.4 Hz, 1H), 3.13 (dd, J = 16.7, 5.8 Hz, 1H), 2.92 (d, J = 16.6 Hz, 1H), 1.53–1.39 (m, 10H), 1.36 (s, 9H). 13C NMR (126 MHz, DMSO) δ 167.8, 165.8, 139.1, 135.7, 134.9, 126.9, 126.0, 125.2, 124.7, 122.4, 109.3, 74.8, 73.0, 65.3, 64.5, 61.8, 50.2, 41.7, 40.0, 39.9, 39.9, 39.8, 39.7, 39.5, 39.4, 39.2, 39.0, 36.5, 34.3, 28.4, 28.3, 24.5, 23.6, 23.3. HRMS (ESI) m/z: [M + H]+ calcd for C27H37N4O5 497.2764. Found 497.2745.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16) as a white amorphous solid. Dr obtained by HPLC of crude similar to 26ab. Ratio of 36abc is 67
16) as a white amorphous solid. Dr obtained by HPLC of crude similar to 26ab. Ratio of 36abc is 67![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12
12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 21. An analytical isomer of major isomer 36a was prepared after chromatographic separation. Minor diastereomer 36b was not isolated.
21. An analytical isomer of major isomer 36a was prepared after chromatographic separation. Minor diastereomer 36b was not isolated.
36a (major isomer). White amorphous solid. Rf (5% MeOH/DCM) = 0.47. Retention time: 34.6 min. 1H NMR (500 MHz, DMSO) δ 9.61 (s, 1H), 8.06 (d, J = 1.2 Hz, 1H), 7.78 (d, J = 3.7 Hz, 1H), 7.29–7.20 (m, 2H), 7.12 (dt, J = 22.2, 7.3 Hz, 2H), 6.86 (d, J = 7.4 Hz, 1H), 5.87 (d, J = 8.7 Hz, 1H), 5.16–5.09 (m, 2H), 4.64–4.52 (m, 2H), 4.05–3.96 (m, 1H), 3.76–3.70 (m, 1H), 3.27–3.09 (m, 2H), 2.90 (d, J = 16.6 Hz, 1H), 2.05–1.98 (m, 1H), 1.33 (s, 9H), 1.19 (s, 3H), 1.18 (s, 3H). 13C NMR (126 MHz, DMSO) δ 167.1, 166.7, 139.0, 138.6, 135.9, 134.8, 126.9, 125.9, 125.4, 124.6, 122.4, 97.9, 75.4, 66.5, 65.4, 64.4, 58.7, 50.2, 41.6, 40.0, 39.9, 39.9, 39.8, 39.7, 39.5, 39.4, 39.2, 39.0, 29.7, 28.3, 27.4, 19.3. HRMS (ESI) m/z: [M + H]+ calcd for C25H35N4O5 471.2608. Found 471.2586.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30) as a white amorphous solid. Dr determined by HPLC of crude similar to 26ab. Analytical samples of pure diasteromers 27a and 27b were prepared after chromatographic separation.
30) as a white amorphous solid. Dr determined by HPLC of crude similar to 26ab. Analytical samples of pure diasteromers 27a and 27b were prepared after chromatographic separation.
37a (major isomer). White amorphous solid. Rf (5% MeOH/DCM) = 0.42. [α]23D +129.8 (c 0.45, CHCl3). Retention time: 30.6 min. 1H NMR (500 MHz, DMSO) δ 10.08 (s, 1H), 8.04 (s, 1H), 7.75 (s, 1H), 7.23 (d, J = 7.1 Hz, 1H), 7.12 (dt, J = 20.1, 7.2 Hz, 2H), 6.91 (t, J = 7.1 Hz, 1H), 5.50 (d, J = 9.1 Hz, 1H), 5.38 (dd, J = 9.0, 5.4 Hz, 1H), 4.88 (d, J = 6.0 Hz, 1H), 4.58–4.50 (m, 1H), 4.04 (p, J = 6.0 Hz, 1H), 3.81 (dd, J = 8.1, 5.9 Hz, 1H), 3.48 (dd, J = 8.1, 6.2 Hz, 1H), 3.16 (dd, J = 16.6, 5.6 Hz, 1H), 2.92 (d, J = 16.6 Hz, 1H), 2.32 (ddd, J = 13.9, 9.1, 5.3 Hz, 1H), 1.90 (ddd, J = 13.1, 7.6, 5.3 Hz, 1H), 1.36 (s, 9H), 1.18 (s, 3H), 1.15 (s, 3H). 13C NMR (126 MHz, DMSO) δ 168.5, 166.0, 139.3, 139.1, 135.6, 135.2, 126.9, 125.9, 125.2, 123.9, 122.5, 108.0, 75.6, 72.5, 68.3, 64.8, 57.7, 50.3, 41.8, 39.9, 39.9, 39.8, 39.7, 39.5, 39.4, 39.2, 39.0, 33.9, 28.3, 26.8, 25.6. HRMS (ESI) m/z: [M + H]+ calcd for C25H35N4O5 471.2608. Found 471.2586.
37b (minor isomer). White amorphous solid. Rf (5% MeOH/DCM) = 0.32. [α]23D −65.6 (c 0.31, CHCl3). Retention time: 50.2 min. 1H NMR (500 MHz, DMSO) δ 8.43 (s, 1H), 7.88 (s, 1H), 7.70 (s, 1H), 7.43–7.30 (m, 3H), 7.30–7.21 (m, 2H), 5.90 (d, J = 7.0 Hz, 1H), 4.53–4.46 (m, 1H), 3.77–3.72 (m, 1H), 3.20 (dd, J = 16.1, 8.0 Hz, 2H), 2.95–2.72 (m, 3H), 2.08 (t, J = 7.9 Hz, 1H), 1.28–1.22 (m, 2H), 1.18 (s, 9H), 1.14 (s, 3H), 1.10 (s, 3H). 13C NMR (126 MHz, DMSO) δ 169.2, 141.7, 138.0, 136.5, 135.1, 129.4, 127.4, 127.2, 125.6, 121.4, 107.2, 73.7, 72.2, 66.9, 63.6, 56.8, 49.8, 38.2, 35.3, 28.4, 26.8, 25.9. HRMS (ESI) m/z: [M + H]+ calcd for C25H35N4O5 471.2608. Found 471.2610.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 13) as a white amorphous solid. Dr determined via HPLC of crude similar to 26ab. An analytical sample of pure diastereomer 38a was prepared after chromatographic separation.
13) as a white amorphous solid. Dr determined via HPLC of crude similar to 26ab. An analytical sample of pure diastereomer 38a was prepared after chromatographic separation.
38a (major isomer). White amorphous solid. Rf (5% MeOH/DCM) = 0.24. [α]23D +174.8 (c 0.46, CHCl3). Retention time: 31.0 min. 1H NMR (500 MHz, DMSO) δ 9.72 (s, 1H), 8.04 (d, J = 1.2 Hz, 1H), 7.79 (d, J = 1.2 Hz, 1H), 7.25 (d, J = 7.3 Hz, 1H), 7.14 (t, J = 7.3 Hz, 1H), 7.09 (t, J = 7.4 Hz, 1H), 6.85 (d, J = 7.4 Hz, 1H), 5.75 (s, 1H), 5.14–5.08 (m, 2H), 4.57–4.45 (m, 2H), 3.64 (t, J = 6.9 Hz, 2H), 3.09 (dd, J = 16.6, 5.0 Hz, 1H), 2.89 (d, J = 16.5 Hz, 1H), 2.22–2.12 (m, 1H), 1.80 (dt, J = 12.2, 7.2 Hz, 1H), 1.62 (tq, J = 12.3, 6.6 Hz, 1H), 1.54–1.42 (m, 2H), 1.34 (s, 9H). 13C NMR (126 MHz, DMSO) δ 167.9, 166.2, 139.0, 138.7, 135.8, 134.9, 126.9, 125.9, 125.4, 124.6, 122.3, 76.0, 75.7, 67.6, 65.3, 63.5, 54.9, 50.2, 41.6, 28.4, 25.0. HRMS (ESI) m/z: [M + H]+ calcd for C23H31N4O4 427.2345. Found 427.2334.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 14) as a white amorphous solid. Ratio of 39ab determined via HPLC of crude similar to 26ab. Ratio of 39abc is 78
14) as a white amorphous solid. Ratio of 39ab determined via HPLC of crude similar to 26ab. Ratio of 39abc is 78![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 13
13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9. Analytical sample of 39a was prepared after chromaotgraphic separation.
9. Analytical sample of 39a was prepared after chromaotgraphic separation.
39a (major isomer). White amorphous solid. Rf (5% MeOH/DCM) = 0.46. [α]23D +138.4 (c 0.51, CHCl3). Retention time: 30.3 min. 1H NMR (500 MHz, MeOD) δ 9.99 (s, 1H), 7.90 (s, 1H), 7.74–7.70 (m, 1H), 7.30–7.21 (m, 2H), 7.17 (t, J = 7.4 Hz, 1H), 7.10 (t, J = 7.5 Hz, 1H), 6.95 (d, J = 7.5 Hz, 1H), 5.37 (d, J = 9.8 Hz, 1H), 5.30 (d, J = 5.3 Hz, 1H), 4.54 (t, J = 5.1 Hz, 1H), 4.01 (t, J = 10.2 Hz, 1H), 3.90 (dd, J = 11.9, 3.8 Hz, 1H), 3.52 (td, J = 10.8, 3.1 Hz, 1H), 3.16 (dd, J = 16.5, 4.7 Hz, 1H), 3.03 (d, J = 16.6 Hz, 1H), 2.24 (d, J = 13.3 Hz, 1H), 1.86 (d, J = 13.4 Hz, 1H), 1.67–1.56 (m, 2H), 1.56–1.47 (m, 3H). 13C NMR (126 MHz, DMSO) δ 167.6, 166.5, 138.9, 138.5, 135.8, 134.8, 126.9, 125.9, 125.5, 124.6, 122.3, 75.8, 74.4, 67.7, 65.4, 64.1, 50.2, 41.5, 39.9, 39.7, 39.5, 39.4, 39.2, 39.0, 28.4, 27.6, 25.6, 22.2. HRMS (ESI) m/z: [M + H]+ calcd for C24H33N4O4 441.2502. Found 441.2489.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 35) as a white amorphous solid. Dr determined via HPLC of crude similar to 26ab, but for solvent system, 0.1% (v/v) TFA in MilliQ H2O replaced that of solely MilliQ H2O for the polar mobile phase component. Analytical samples of pure diasteromers 40a and 40b were prepared after chromatographic separation.
35) as a white amorphous solid. Dr determined via HPLC of crude similar to 26ab, but for solvent system, 0.1% (v/v) TFA in MilliQ H2O replaced that of solely MilliQ H2O for the polar mobile phase component. Analytical samples of pure diasteromers 40a and 40b were prepared after chromatographic separation.
40a (major isomer). White amorphous solid. Rf (5% MeOH/DCM) = 0.48. [α]23D +198.8 (c 0.45, CHCl3). Retention time: 10.5 min 1H NMR (400 MHz, DMSO) δ 9.90 (s, 1H), 8.07 (d, J = 1.3 Hz, 1H), 7.79 (d, J = 1.2 Hz, 1H), 7.40–7.05 (m, 9H), 6.86 (d, J = 7.3 Hz, 1H), 5.88 (d, J = 8.5 Hz, 1H), 5.24 (d, J = 5.3 Hz, 1H), 5.18 (d, J = 10.1 Hz, 1H), 4.53 (dt, J = 9.5, 5.1 Hz, 1H), 4.38 (d, J = 12.8 Hz, 1H), 3.76–3.67 (m, 1H), 3.42 (d, J = 12.8 Hz, 1H), 3.12 (dd, J = 16.5, 4.9 Hz, 1H), 2.90 (d, J = 16.5 Hz, 1H), 2.60–2.55 (m, 1H), 2.23 (td, J = 9.8, 6.0 Hz, 1H), 1.99–1.85 (m, 1H), 1.82–1.73 (m, 1H), 1.68–1.56 (m, 2H), 1.38 (s, 9H). 13C NMR (126 MHz, DMSO) δ 169.2, 166.3, 140.8, 139.3, 138.6, 135.8, 135.1, 128.3, 127.9, 126.8, 126.4, 125.8, 125.4, 124.6, 122.2, 76.0, 65.2, 64.3, 62.7, 60.8, 54.2, 50.3, 41.6, 39.9, 39.7, 39.5, 39.4, 39.2, 39.0, 28.4, 27.2, 24.2. HRMS (ESI) m/z: [M + H]+ calcd for C30H38N5O3 516.2975. Found 516.2951.
40b (minor isomer). White amorphous solid. Rf (5% MeOH/DCM) = 0.43. [α]23D +3.6 (c 0.50, CHCl3). Retention time: 12.1 min. 1H NMR (400 MHz, DMSO) δ 7.88–6.95 (m, 13H), 5.60–5.52 (m, 1H), 4.25 (m, 1H), 3.88 (d, J = 13.4 Hz, 1H), 3.25 (dd, J = 16.4, 5.9 Hz, 0H), 3.20–3.09 (m, 2H), 3.05 (m, 2H), 2.56 (ddd, J = 9.6, 7.1, 2.8 Hz, 1H), 2.03 (q, J = 8.6 Hz, 1H), 1.92–1.78 (m, 1H), 1.66–1.53 (m, 1H), 1.47 (m, 1H), 1.41–1.31 (m, 1H), 1.28–1.20 (m, 1H), 1.05 (s, 9H). 13C NMR (126 MHz, DMSO) δ 172.0, 162.1, 143.0, 139.7, 139.3, 136.9, 131.9, 128.1, 128.1, 127.8, 126.7, 126.7, 124.9, 124.2, 123.4, 74.7, 64.8, 63.3, 62.0, 58.5, 53.7, 49.3, 36.2, 28.4, 28.0, 22.8. HRMS (ESI) m/z: [M + H]+ calcd for C30H38N5O3 516.2975. Found 516.2977.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 22) as a white amorphous solid. Dr determined by HPLC of crude similar to 26ab. An analytical sample of pure diastereomer 41a was prepared after chromatographic separation.
22) as a white amorphous solid. Dr determined by HPLC of crude similar to 26ab. An analytical sample of pure diastereomer 41a was prepared after chromatographic separation.
41a (major isomer). White amorphous solid. Rf (5% MeOH/DCM) = 0.36. [α]23D +200.0 (c 0.46, CHCl3). Retention time: 35.0 min. 1H NMR (400 MHz, DMSO) δ 9.76 (s, 1H), 8.07 (s, 1H), 7.79 (s, 1H), 7.25 (d, J = 7.3 Hz, 1H), 7.14 (t, J = 7.2 Hz, 1H), 7.08 (t, J = 7.2 Hz, 1H), 6.82 (d, J = 7.3 Hz, 1H), 5.95 (d, J = 8.6 Hz, 1H), 5.18 (d, J = 5.3 Hz, 1H), 5.03 (d, J = 9.4 Hz, 1H), 4.51–4.40 (m, 2H), 3.12 (dd, J = 16.5, 4.8 Hz, 1H), 2.90 (d, J = 16.6 Hz, 1H), 1.34 (s, 9H), 1.27–1.22 (m, 1H), 1.20 (d, J = 6.0 Hz, 3H), 0.81 (s, 9H), 0.09 (s, 3H), 0.07–0.04 (m, 3H). 13C NMR (126 MHz, DMSO) δ 168.0, 166.5, 139.0, 138.5, 135.8, 135.0, 126.9, 125.9, 125.4, 124.7, 122.2, 75.7, 66.5, 66.4, 65.4, 50.2, 41.6, 39.9, 39.9, 39.8, 39.7, 39.5, 39.4, 39.2, 39.0, 28.5, 25.7, 20.1, 17.6, −4.5, −5.0. HRMS (ESI) m/z: [M + H]+ calcd for C27H43N4O4Si 515.3054. Found 515.3041.
Conflicts of interest
The authors declare no conflict of interest.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: full characterisation data for all new compounds and crystallographic data collection and refinement statistics. See DOI: https://doi.org/10.1039/d5ob01897h.CCDC 2474023–2474025 and 2501300 (27a, 27c, 38 and 10c) contain the supplementary crystallographic data for this paper.43,45,46,54
Acknowledgements
The research reported here was supported in part by grants and a contract from the National Institute of Allergy and Infectious Diseases, National Institutes of Health (A. K. G. AI150466, and A. K. G. & A. D. M., AI158649 and contract HHSN272201700060C). UJ was supported by NIH NIAID T32 training grant AI 148103 (Drug Discovery in Infectious Disease Training) and SB were supported by NIH NIGMS T32 training grant GM132024 (Purdue University Molecular Biophysics Training Program). The authors also wish to acknowledge support from the Purdue Institute for Cancer Research, NIH grant P30 CA023168, for use of the shared NMR and Macromolecular Crystallization and X-ray diffraction facilities available in the Biomolecular Structure Shared Resource.References
- I. Ugi, R. Meyr, U. Fetzer and C. Steinbrückner, Angew. Chem., 1959, 71, 386 Search PubMed.
- C. de Graaff, E. Ruijter and R. V. Orru, Chem. Soc. Rev., 2012, 41, 3969–4009 RSC.
- P. S. G. Nunes, H. D. A. Vidal and A. G. Corrêa, Org. Biomol. Chem., 2020, 18, 7751–7773 RSC.
- E. Ruijter, R. Scheffelaar and R. V. Orru, Angew. Chem., Int. Ed., 2011, 50, 6234–6246 CrossRef CAS PubMed.
- S. Bhattacharjee and S. Gajurel, Tetrahedron Lett., 2026, 174, 155893 CrossRef CAS.
- W. Zhang, P. Tang, M. A. Abubaker, G.-H. Hu and F.-E. Chen, Green Synth. Catal., 2025, 6, 255–266 Search PubMed.
- J. D. Sunderhaus and S. F. Martin, Chemistry, 2009, 15, 1300–1308 CrossRef CAS PubMed.
- M. A. Fouad, H. Abdel-Hamid and M. S. Ayoup, RSC Adv., 2020, 10, 42644–42681 RSC.
- M. T. Nazeri, H. Farhid, R. Mohammadian and A. Shaabani, ACS Comb. Sci., 2020, 22, 361–400 Search PubMed.
- B. H. Rotstein, S. Zaretsky, V. Rai and A. K. Yudin, Chem. Rev., 2014, 114, 8323–8359 CrossRef CAS PubMed.
- A. Dömling, W. Wang and K. Wang, Chem. Rev., 2012, 112, 3083–3135 CrossRef PubMed.
- E. L. Larghi, A. B. J. Bracca, S. O. Simonetti and T. S. Kaufman, Org. Biomol. Chem., 2024, 22, 429–465 RSC.
- K. L. Rinehart, T. G. Holt, N. L. Fregeau, J. G. Stroh, P. A. Keifer, F. Sun, L. H. Li and D. G. Martin, J. Org. Chem., 1990, 55, 4512–4515 Search PubMed.
- A. Endo, A. Yanagisawa, M. Abe, S. Tohma, T. Kan and T. Fukuyama, J. Am. Chem. Soc., 2002, 124, 6552–6554 CrossRef CAS PubMed.
- S. I. Maffioli, Y. Zhang, D. Degen, T. Carzaniga, G. Del Gatto, S. Serina, P. Monciardini, C. Mazzetti, P. Guglierame, G. Candiani, A. I. Chiriac, G. Facchetti, P. Kaltofen, H. G. Sahl, G. Dehò, S. Donadio and R. H. Ebright, Cell, 2017, 169, 1240–1248 Search PubMed.
- R. Okawa, C. C. Aldrich and S. Ichikawa, Chem. Commun., 2022, 58, 7956–7959 Search PubMed.
- K. Sakamoto, E. Tsujii, F. Abe, T. Nakanishi, M. Yamashita, N. Shigematsu, S. Izumi and M. Okuhara, J. Antibiot., 1996, 49, 37–44 Search PubMed.
- S. Ieda, A. Masuda, M. Kariyama, T. Wakimoto, T. Asakawa, T. Fukuyama and T. Kan, Heterocycles, 2012, 86, 1071–1092 CrossRef CAS PubMed.
- H. A. Younus, M. Al-Rashida, A. Hameed, M. Uroos, U. Salar, S. Rana and K. M. Khan, Expert Opin. Ther. Pat., 2021, 31, 267–289 CrossRef CAS PubMed.
- S. E. John, S. Gulati and N. Shankaraiah, Org. Chem. Front., 2021, 8, 4237–4287 RSC.
- A. Dömling, W. Wang and K. Wang, Chem. Rev., 2012, 112, 3083–3135 CrossRef.
- D. Insuasty, J. Castillo, D. Becerra, H. Rojas and R. Abonia, Molecules, 2020, 25, 505 CrossRef PubMed.
- G. Graziano, A. Stefanachi, M. Contino, R. Prieto-Díaz, A. Ligresti, P. Kumar, A. Scilimati, E. Sotelo and F. Leonetti, Int. J. Mol. Sci., 2023, 24, 6581 CrossRef CAS PubMed.
- D. R. Owen, C. M. N. Allerton, A. S. Anderson, L. Aschenbrenner, M. Avery, S. Berritt, B. Boras, R. D. Cardin, A. Carlo, K. J. Coffman, A. Dantonio, L. Di, H. Eng, R. Ferre, K. S. Gajiwala, S. A. Gibson, S. E. Greasley, B. L. Hurst, E. P. Kadar, A. S. Kalgutkar, J. C. Lee, J. Lee, W. Liu, S. W. Mason, S. Noell, J. J. Novak, R. S. Obach, K. Ogilvie, N. C. Patel, M. Pettersson, D. K. Rai, M. R. Reese, M. F. Sammons, J. G. Sathish, R. S. P. Singh, C. M. Steppan, A. E. Stewart, J. B. Tuttle, L. Updyke, P. R. Verhoest, L. Wei, Q. Yang and Y. Zhu, Science, 2021, 374, 1586–1593 CrossRef CAS.
- Y. N. Lamb, Drugs, 2022, 82, 585–591 CrossRef CAS PubMed.
- H. D. Preschel, R. T. Otte, Y. Zhuo, R. E. Ruscoe, A. J. Burke, R. Kellerhals, B. Horst, S. Hennig, E. Janssen, A. P. Green, N. J. Turner and E. Ruijter, J. Org. Chem., 2023, 88, 2565–12571 Search PubMed.
- J. Jacobs, V. Grum-Tokars, Y. Zhou, M. Turlington, S. A. Saldanha, P. Chase, A. Eggler, E. S. Dawson, Y. M. Baez-Santos, S. Tomar, A. M. Mielech, S. C. Baker, C. W. Lindsley, P. Hodder, A. Mesecar and S. R. Stauffer, J. Med. Chem., 2013, 56, 534–546 CrossRef CAS PubMed.
- N. Kitamura, M. D. Sacco, C. Ma, Y. Hu, J. A. Townsend, X. Meng, F. Zhang, X. Zhang, M. Ba, T. Szeto, A. Kukuljac, M. T. Marty, D. Schultz, S. Cherry, Y. Xiang, Y. Chen and J. Wang, J. Med. Chem., 2022, 65, 2848–2865 CrossRef CAS PubMed.
- M. Ashraf-Uz-Zaman, T. K. Chua, X. Li, Y. Yao, B. K. Moku, C. B. Mishra, V. Avadhanula, P. A. Piedra and Y. Song, ACS Infect. Dis., 2024, 10, 715–731 CrossRef CAS PubMed.
- D. J. Ramón and M. Yus, Angew. Chem., Int. Ed., 2005, 44, 1602–1634 CrossRef PubMed.
- J. Zhang, P. Yu, S. Y. Li, H. Sun, S. H. Xiang, J. J. Wang, K. N. Houk and B. Tan, Science, 2018, 361, eaas8707 CrossRef PubMed.
- A. Katsuyama, A. Matsuda and S. Ichikawa, Org. Lett., 2016, 18, 2552–2555 CrossRef CAS PubMed.
- Q. Wang, D.-X. Wang, M.-X. Wang and J. Zhu, Acc. Chem. Res., 2018, 51, 1290–1300 CrossRef CAS PubMed.
- A. K. Ghosh, S. Fidanze and C. H. Senanayake, Synthesis, 2000, 937–961 Search PubMed.
- A. K. Ghosh, S. Fidanze and C. H. Senanayake, Synthesis, 1998, 937–961 Search PubMed.
- S. Ghosh, R. V. Rao and J. Shashidhar, Tetrahedron Lett., 2005, 46, 5479–5481 Search PubMed.
- C. R. Schmid, J. D. Bryant, M. Dowlatzedah, J. L. Phillips, D. E. Prather, R. D. Schantz, N. L. Sear and C. S. Vianco, J. Org. Chem., 1991, 56, 4056–4058 CrossRef CAS.
- E. Dibello, D. Gamenara and G. Seoane, Org. Prep. Proced. Int., 2015, 47, 415–442 Search PubMed.
- J. Jurczak, S. Pikul and T. Bauer, Tetrahedron, 1986, 42, 447–488 CrossRef CAS.
- J. Mulzer and A. Angerm, Tetrahedron Lett., 1983, 24, 2843–2846 Search PubMed.
- C. R. Schmid and J. D. Bryant, Org. Synth., 1995, 72, 6 Search PubMed.
- Single-Crystal X-Ray analysis was performed in our X-Ray Crystallography laboratory at Purdue University, Dr. Matthias Zeller, Department of Chemistry, Purdue University.
- CCDC 2501300: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pyt3l.
- A. Jakas, A. Višnjevac and I. Jerić, J. Org. Chem., 2020, 85, 3766–3787 Search PubMed.
- CCDC 2474023: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2p1f6d.
- CCDC 2474024: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2p1f7f.
- L. Caruso, A. Puglisi, E. Gillon and M. Benaglia, Molecules, 2019, 24, 4588 CrossRef CAS PubMed.
- Ł. Mucha, K. Parda, O. Staszewska-Krajewska, S. Stecko, A. Ulikowski, J. Frelek, A. Suszczyńska, M. Chmielewski and B. Furman, Tetrahedron: Asymmetry, 2016, 27, 12–21 CrossRef.
- M. J. Lilly, N. A. Miller, A. J. Edwards, A. C. Willis, P. Turner, M. N. Paddon-Row and M. S. Sherburn, Chem. – Eur. J., 2005, 11, 2525–2536 CrossRef CAS PubMed.
- M. S. W. Richardson, C. J. Tame, D. L. Poole and T. J. Donohoe, Chem. Sci., 2019, 10, 6336–6340 RSC.
- A. Bartolozzi, A. Berry, E. R. Hickey, M. Ostermeier, D. Riether, A. Sauer, D. S. Thomson, L. Wu, R. M. Zindell, P. Amouzegh, N. J. Blumire, S. P. East, M. Ermann, S. Khor and I. Mushi, US2012010184A1, 2012.
- L. Zhu, L. Cheng, Y. Zhang, R. Xie and J. You, J. Org. Chem., 2007, 72, 2737–2743 CrossRef CAS PubMed.
- S. Mandal, D. Mahananda, D. Paladugu and B. Thirupathi, J. Org. Chem., 2024, 89, 4165–4175 CrossRef CAS PubMed.
- CCDC 2474025: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2p1f8g.
- M. Chérest, H. Felkin and N. Prudent, Tetrahedron Lett., 1968, 9, 2199–2204 CrossRef.
- P. DeShong and J. M. Leginus, J. Am. Chem. Soc., 1983, 105, 1686–1688 CrossRef CAS.
- O. Mumm, H. Hesse and H. Volquartz, Ber. Dtsch. Chem. Ges., 1915, 48, 379 CrossRef CAS.
- M. A. Rouhani, J. Phys. Org. Chem., 2020, 33, e4106 CrossRef CAS.
- C. Moreau, T. Kirchberger, J. M. Swarbrick, S. J. Bartlett, R. Fliegert, T. Yorgan, A. Bauche, A. Harneit, A. H. Guse and B. V. L. Potter, J. Med. Chem., 2013, 56, 10079–10102 CrossRef CAS PubMed.
- B. Jäger, H. Lay, J. Lehmann and L. Ziser, Carbohydr. Res., 1991, 217, 99–106 CrossRef PubMed.
- X. Liu, J. Jia, Y. Jia, H. Gu, J. Luo and X. Chen, Org. Lett., 2018, 20, 1945–1948 CrossRef CAS PubMed.
- J. J. Baldwin, D. A. Claremon, C. M. Tice, S. Cacatian, L. W. Dillard, A. V. Ishchenko, J. Yuan, Z. Xu, G. Mcgeehan, W. Zhao, R. D. Simpson and S. B. Singh, WO2007117560A2, 2007.
- X. Wu, B. M. Aquila, L. Shao, H. Radeke, G. D. Cuny, J. R. Hauske and R. L. Xie, WO0222579A2, 2002.
- F. W. Foss, A. H. Snyder, M. D. Davis, M. Rouse, M. D. Okusa, K. R. Lynch and T. L. Macdonald, Bioorg. Med. Chem., 2007, 15, 663–677 CrossRef CAS PubMed.
- K. Saigo, K. Kinbara and Y. Katsumata, US6479702B1, 2002.
- J. Zhuo, T. P. Maduskuie, D.-Q. Qian and W. Yao, US20100240671A1, 2010.
- E. J. Hennessy, A. Adam, B. M. Aquila, L. M. Castriotta, D. Cook, M. Hattersley, A. W. Hird, C. Huntington, V. M. Kamhi, N. M. Laing, D. Li, T. MacIntyre, C. A. Omer, V. Oza, T. Patterson, G. Repik, M. T. Rooney, J. C. Saeh, L. Sha, M. M. Vasbinder, H. Wang and D. Whitston, J. Med. Chem., 2013, 56, 9897–9919 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |