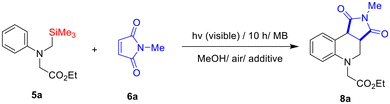Open Access Article
Open Access ArticleSET-oxidative photocyclization reactions of N-(α-trimethylsilyl)-N-phenylglycinates with N-substituted maleimides: a role of silyl group in photocyclization
Hayeon You,
Suk Hyun Lim* and
Dae Won Cho *
*
Department of Chemistry, Yeungnam University, Gyeongsan, Gyeongbuk 712-749, Republic of Korea. E-mail: dwcho00@yu.ac.kr
First published on 3rd February 2026
Abstract
SET-promoted photocyclization reactions of N-(α-trimethylsilyl)-N-phenylglycinates with N-substituted maleimides were explored using methylene blue (MB) as a photosensitizer, and mechanistic studies were performed. In contrast to the non-silylated N-phenylglycinate substrates, which showed no reactivity with maleimides, the MB-photosensitized reactions of N-(α-trimethylsilyl)-N-phenylglycinates with maleimides led to the formation of tetrahydroquinoline derivatives efficiently. These observations reveal that the silyl group adjacent to the nitrogen atom plays a crucial role in enabling a successful SET-oxidative cyclization reaction. Overall, this reaction protocol can expand the range of amine substrates in the diverse SET-oxidative photochemical reactions.
Introduction
Since the recognition of photoinduced single electron transfer (SET) process as a central tool in synthetic photochemistry, numerous SET-promoted photochemical reactions using amine electron donors have been explored so far, mainly due to the low oxidation potentials (Eox < 1 V vs. SCE) of aliphatic- and aromatic amines and unique chemical behaviors of in situ formed intermediates, amine radical cations.1–8 For instance, when suitable electron acceptors (i.e., either electron-accepting reactant or photosensitizer) with an excited-state reduction potential (*Ered) higher than the oxidation potential of amines (Eox) are used, photoirradiation can induce SET from amines to the acceptors, generating amine radical cations. These radical cations participate in diverse chemical pathways depending on the structure of amines and the reaction conditions.3–9 The most common reaction pathways open to these intermediates are either α-C(sp3)–H deprotonation to form α-amino radicals or a secondary SET process to form iminium ions.1,10–12 A hydrogen-atom-transfer (HAT) process can result in the formation of iminium ions, providing an alternative pathway. Then, either α-amino radicals or iminium ions react with electrophilic or nucleophilic substrates to form amine moiety-containing molecules.As one of the valuable synthetic applications of this chemistry, SET-oxidative photocyclization reactions between N,N-dialkylanilines and N-substituted maleimides to construct tetrahydroquinolines have drawn considerable attention due to a broad range of pharmacological and biological interest of the formed heterocycles.9–15 In the presence of a photosensitizer—such as a transition metal complex,13–15 metal oxide16 or organic dyes—17–20 irradiation with visible or UV light promotes the efficient conversion of N,N-dimethylanilines 1 to α-amino radicals 2 through a sequential SET- α-CH deprotonation (Scheme 1). These radicals were added to N-alkyl/aryl maleimides 3 to form intermediates 4a, which then underwent cyclization, followed by dehydrogenation, to form the tetrahydroquinolines 4b. Recently, a diverse set of photocatalyst-free photochemical methods was also developed for the preparation of tetrahydroquinolines.21–23 Alternatively, thermochemical reactions of N,N-dialkylanilines with N-substituted maleimides using either various oxidants or a combination of oxidant and metal ionic salts have also been explored to prepare tetrahydroquinolines.24–32
 | ||
| Scheme 1 SET-promoted photocyclization reactions of N,N-dialkylanilines with N-substituted maleimides. | ||
Despite the synthetic utility of these photo- and thermochemical methods, the substrate scope of amines remains largely restricted to N,N-dialkylanilines. In particular, Bian and coworkers showed that, unlike N,N-dialkylanilines, photocyclization reactions of anilines bearing electron-withdrawing groups, such as N-methyl-N-phenylglycinates (PhN(Me)CH2CO2Et), with N-phenylmaleimide proceeded inefficiently and produced unwanted byproducts, such as oxamates, competitively.13 Thus, to overcome these limitations and expand substrate applicability, the development of more general synthetic protocols is required. Building on our recent success with silyl group-containing amine substrates (i.e., N-α-silylamines) in SET-promoted reactions,33–36 we envisioned that the silyl group within amine substrates could facilitate efficient α-amino radical formation, resulting in enhancing radical addition to maleimides and cyclization efficiency in the preparation of N-heterocyclic tetrahydroquinolines. Thus, as part of our continuing research efforts, we explored SET-oxidative photocyclization reactions of N-α-trimethylsilyl-N-phenylglycinates, bearing various substituents on the phenyl ring, with N-substituted maleimides. The results showed that SET-oxidative photocyclization reactions proceeded efficiently to form tetrahydroquinolines, and the silyl group played a crucial role in the cyclization. Detailed results are described and discussed below.
Results and discussion
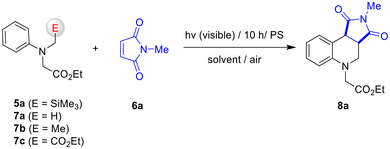
To explore SET-oxidative photocyclization reactions of electron-withdrawing group tethered amines, N-α-trimethylsilyl-N-phenylglycinate 5a and N-methylmaleimide 6a were chosen as a model substrate. For the photoreaction, the solutions (15 mL) containing glycinate 5a (0.38 mmol, 1 equiv.) and maleimide 6a (0.75 mmol, 2 equiv.) in the presence of photosensitizer (3 or 5 mol%) were irradiated with a 20 W compact fluorescent lamp (CFL) under air at room temperature. To figure out the role of the silyl group within the glycinate substrate, the photoreaction of non-silylated 7a–7c37 with 6a was also conducted under the same reaction conditions.As shown in Table 1, the methylene blue (MB) -photosensitized photoreactions of 5a with 6a in MeOH solvent led to the efficient and exclusive formation of tetrahydroquinoline 8a (entry 1). The stereochemistry of the formed tetrahydroquinoline 8a was determined by comparison with the previously reported tetrahydroquinoline analogs.38–44 In polar aprotic solvents, both conversion- and photoproduct yields dropped significantly (entries 2–5). These observations seem to result from silophilic MeOH-assisted fast desilylation (∼SiMe3+) rather than α-CH deprotonation, leading to the generation of an α-amino radical intermediate.7,8,33,34,36 Although other visible-light photosensitizers such as rose Bengal (RB), eosin-Y (EY), and Ru(bpy)3Cl2 could lead to the formation of 8a as well, these sensitizers were less efficient than MB (entries 6–11). From a further screening of reaction conditions, we found that 5 mol% MB is the optimal amount for this SET-oxidative cyclization reaction (entry 12). Control experiments revealed that both photosensitizer and visible light were essential to these photochemical transformation reactions (entries 13, 14). Noticeably, the MB-photosensitized reaction of non-silylated glycinate 7a–7c with maleimide 6a did not afford any detectable photoproducts (entries 15, 16), even in the much longer visible-light irradiation condition (up to 30 h). (The glycinates 7a–7c remained intact).
| Entry | Glycinate | Reaction conditions | Conversion yieldb (%) | Product yieldc (%) |
|---|---|---|---|---|
| a Reaction conditions: the solutions (15 mL) of glycinate (5a or 7a–7b, 0.38 mmol, 1 equiv.), N-methylmaleimide 6a (0.75 mmol, 2 equiv.), and photosensitizer (3 or 5 mol%) were irradiated using a 20 W CFL for 10 h in the air.b Determined by recovered glycinate 5a or 7a–7b.c Isolation yields.d 3 mol% PS was used.e 5 mol% PS was used.f Reaction in the dark (no light).g Not detected. The starting glycinates remained intact. | ||||
| 1 | 5a | MBd/MeOH | 100 | 79 |
| 2 | 5a | MBd/MeCN | 21 | 9 |
| 3 | 5a | MBd/DMF | 65 | 34 |
| 4 | 5a | MBd/DMSO | 49 | 20 |
| 5 | 5a | MBd/CH2Cl2 | 28 | 13 |
| 6 | 5a | RBd/MeCN | 54 | 28 |
| 7 | 5a | RBd/MeOH | 100 | 58 |
| 8 | 5a | EYd/MeCN | 74 | 24 |
| 9 | 5a | EYd/MeOH | 72 | 36 |
| 10 | 5a | Ru(bpy)3Cl2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) d/MeCN d/MeCN |
100 | 64 |
| 11 | 5a | Ru(bpy)3Cl2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) d/MeOH d/MeOH |
100 | 74 |
| 12 | 5a | MBe/MeOH | 100 | 85 |
| 13f | 5a | MBe/MeOH | 0 | n.dg |
| 14 | 5a | No/MeOH | 0 | n.dg |
| 15 | 7a | MBe/MeOH | 0 | n.dg |
| 16 | 7b | MBe/MeOH | 0 | n.dg |
| 17 | 7c | MBe/MeOH | 0 | n.dg |
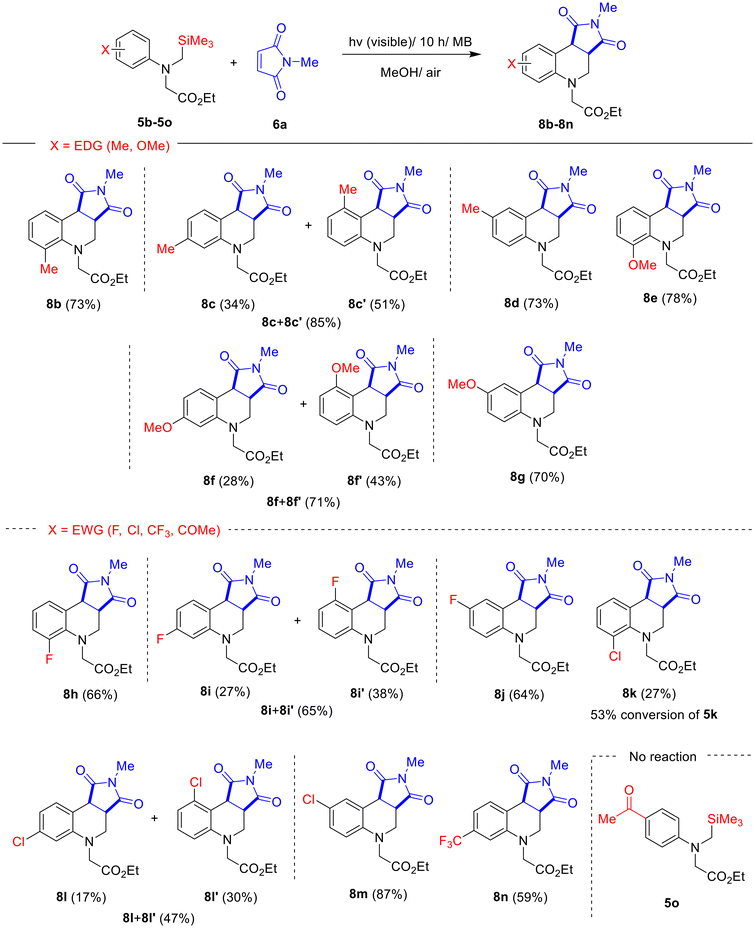
With optimized reaction conditions in hand (entry 12 in Table 1), we explored the substrate scope for this photochemical reaction. Initially, we assessed the performances of N-α-trimethylsilyl-N-phenylglycinates 5b–5o, bearing either various electron-donating (Me, OMe) or electron-withdrawing (F, Cl, CF3, COMe) groups at ortho, meta, or para position of the phenyl ring. As depicted in Scheme 2, 10 h visible-light irradiation of MeOH solutions containing either methyl (Me)- or methoxy (OMe) group-containing glycinates 5b–5g with maleimide 6a furnished the corresponding tetrahydroquinolines 8b–8g in moderate yields ranging from 70% to 85%. The MB-photosensitized reactions of fluoro- (5h–5j), chloro- (5k–5m), or trifluoromethyl (5n)-substituted glycinates provided the expected photoproducts 8h–8n, but the yields obtained from the electron-withdrawing substituents 5h–5l (except 5m) were generally lower than those of their electron-donating analogues 5b–5g. The photoreactions using acyl-substituted glycinate 5o failed to afford the corresponding products, with the starting materials remaining unchanged. Notably, the photoreaction of ortho-chlorophenyl-substituted glycinate 5k was sluggish, with only 53% conversion after 10 h of visible-light irradiation, yielding a low yield (27%) of photoproduct 8k. In contrast, the photoreaction of para-chlorophenyl substituted glycinate 5m with 6a produced the highest yield (87%) of photoproduct 8m. It is also noteworthy that the MB-photosensitized reactions of glycinates bearing meta-substituents (5c, 5f, 5i, 5l) consistently afforded regioisomeric mixtures, in which one isomer (8c′, 8f′, 8i′, and 8l′) predominated over the other (8c, 8f, 8i, and 8l).13,17,25,27
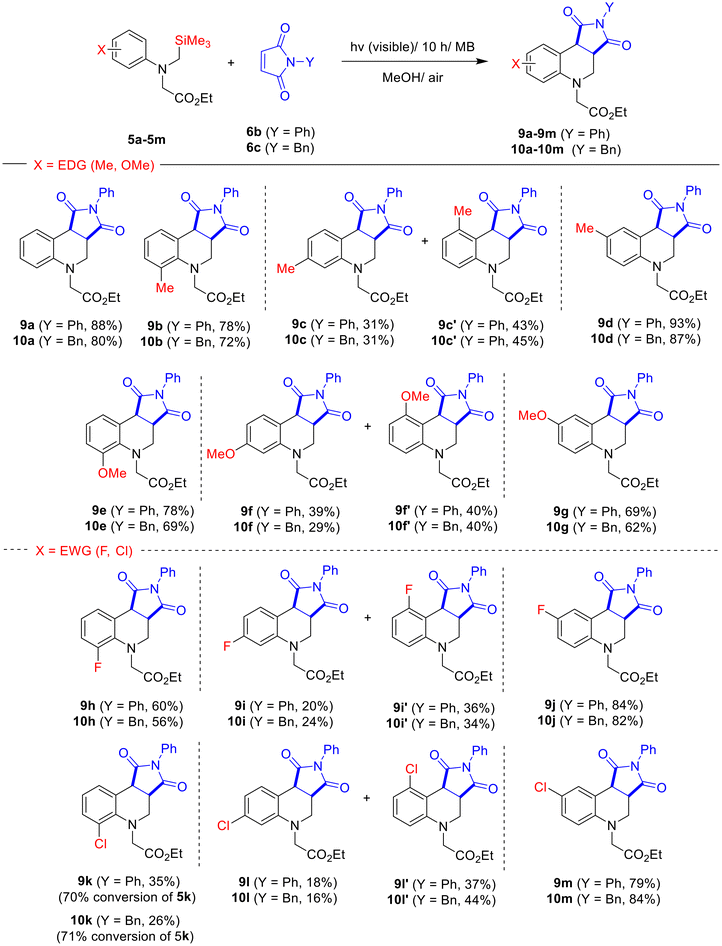
To examine the substrate scope of N-substituted maleimides, the MB-photosensitized reactions of N-α-trimethylsilyl-N-phenylglycinates 5a–5m with N-phenyl- (6b) and N-benzylmaleimide (6c) were performed under the optimized conditions. As illustrated in Scheme 3, photoreactions between glycinates 5a–5m and N-phenylmaleimide 6b proceeded efficiently, yielding tetrahydroquinolines 9a–9m. As with 5k and 6a, the photoreaction of glycinate 5k (X = o-Cl) with 6b was less reactive, affording only 70% conversion and 35% product yield under the optimized conditions. Likewise, similar trends were observed in the photochemical reactions of glycinates 5a–5m with 6c, where 5k produced 10k with 71% conversion and 26% product yield, underscoring its consistently lower reaction efficiency in the SET-oxidative photocyclization reactions (Scheme 3).
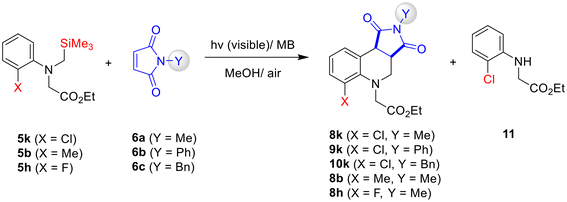
Because glycinate 5k exhibited low reaction efficiency (i.e., poor conversion and low product yields), we anticipated that extending the visible-light irradiation time for its reactions with 6a–6c might enable complete consumption of 5k, thereby improving product yields. However, even after extending the visible-light irradiation time to 25 h, no significant increase in the yields of 8k–10k was observed. Instead, substantial formation of a secondary glycinate 11 was detected (Table 2). Notably, no analogous secondary glycinate was observed in the photoreactions of 5b (X = o-Me) and 5h (X = o-F) (entries). Although mechanistic details remain unclear at this stage, it is plausible that, unlike other glycinates, the radical cation of 5k, generated via SET between 5k and MB, undergoes competitive secondary photochemical pathways (e.g., desilylation vs. H-atom abstraction followed by hydrolysis).
| Entry | Substrate | Irradiation time (h) | Conversion yieldb (%) | Product yieldc (%) |
|---|---|---|---|---|
| a Reaction condition: the MeOH solutions (15 mL) of glycinate (5b, 5h, and 5k, 0.38 mmol, 1 equiv.), maleimide (6a–6c, 0.75 mmol, 2 equiv.), and MB (5 mol%) were irradiated using a 20 W CFL for fixed irradiation time (10 and 25 h) in the air.b Determined by recovered glycinate 5b, 5h, and 5k.c Isolation yields. | ||||
| 1 | 5k + 6a | 10 | 53 | 8k (27) |
| 2 | 5k + 6a | 25 | 100 | 8k (35), 11 (23) |
| 3 | 5k + 6b | 10 | 70 | 9k (35) |
| 4 | 5k + 6b | 25 | 100 | 8k (41), 11 (22) |
| 5 | 5k + 6c | 10 | 71 | 10k (26) |
| 6 | 5k + 6c | 25 | 100 | 10k (40), 11 (33) |
| 7 | 5b + 6a | 10 | 100 | 8b (73) |
| 8 | 5b + 6a | 25 | 100 | 8b (71) |
| 9 | 5h + 6a | 10 | 100 | 8h (66) |
| 10 | 5h + 6a | 25 | 100 | 8h (65) |
To assess the generality of these photochemical reactions, a range of amine derivatives bearing electron-withdrawing groups—such as benzyloxycarbonyl (CO2Bn), cyano (CN), acetyl (COMe), and benzoyl (COPh) (12a–12h)—as well as chiral glycinates (13a–13b) were examined as substrates in MB-photosensitized reactions. As shown in Scheme 4a, silyl-free amine substrates 12e–12h did not undergo any conversion during the photochemical reaction and remained unchanged. In contrast, amine substrates bearing both silyl- and electron-withdrawing groups—namely 12a (EWG = CO2Bn), 12b (EWG = CN), 12c (EWG = COMe), and 12d (EWG = COPh)-reacted efficiently with 6a to produce the corresponding tetrahydroquinoline 14a–14d in good yields (65–83%). In the photoreactions using chiral glycinate derivatives 13a and 13b, two diastereomeric products (15a/15a′ and 15b/15b′) were formed, respectively, in approximately 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratios (Scheme 4b). Unfortunately, the stereochemistry of these isomeric photoproducts could not be determined due to their non-crystalline nature.
1 ratios (Scheme 4b). Unfortunately, the stereochemistry of these isomeric photoproducts could not be determined due to their non-crystalline nature.
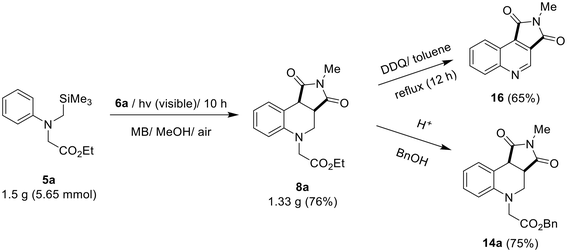
To verify the synthetic usefulness of this photochemical method, a gram-scale reaction using glycinate 5a and several synthetic transformations was performed. MB-photosensitized reaction of 5a (1.5 g, 5.65 mmol, 1 equiv.) with 6a (2 equiv.) afforded 8a in 76% yield (1.33 g). Reflux of 8a at 120 °C in the presence of DDQ led to the formation of quinoline 16,29 and transesterification of 8a with BnOH afforded 14a in high yield (75%) (Scheme 5).
To gain a better understanding of the mechanistic pathways involved in tetrahydroquinoline formation, several experiments were carried out. To clarify the initial involvement of the SET process between glycinate substrates and MB, the photoluminescence quenching experiment of MB in the presence of varying concentrations of either silyl- (5a, 5d, 5g, 5j) or silyl-free (7) glycinate substrates (0–3 mM) was monitored (Fig. S1 in SI). We envisioned that because the oxidation potentials of employed glycinate electron donors (Eox < 1 V vs. SCE) are considerably lower than the reduction potential of triplet state of MB (3Ered = 1.6 V vs. SCE), as a consequence, the SET process from glycinates to 3MB* would be thermodynamically favorable.33,34,45–47 The results showed that the photoluminescence of MB gradually decreased with increasing concentration of the employed glycinates. Notably, although the photoluminescence of MB was quenched by non-silyl glycinate 7, no reaction leading to photoproduct formation took place (Fig. S1 in SI). This suggests that the desilylation process (∼SiMe3+) of the initially generated amine radical cations plays a crucial role in efficiently forming α-amino radicals, which serve as a key precursor in the formation of tetrahydroquinoline derivatives.
Furthermore, the results from the MB-sensitized photoreactions of 5a with 6a in the presence of various additives provided valuable mechanistic insight (Table 3). First, when 2 equiv. of DABCO, a known singlet oxygen (1O2) quencher, were added, no significant changes in either the conversion or product yields were observed. This indicates that 1O2 is not involved in the reaction pathway, despite MB's ability to generate 1O2 (Φ(1O2) = 0.52). In contrast, the addition of TEMPO or BHT, well-known radical quenchers, led to a dramatic decrease in both conversion and product yields. These findings strongly suggest that radical intermediates are involved in the formation of the photoproducts.
| Additive | Conversion yieldb (%) | Yieldc (%) |
|---|---|---|
| a Reaction conditions: the solutions (15 mL) of 5a (0.38 mmol, 1 equiv.), 6a (0.75 mmol, 2 equiv.), and MB (5 mol%) were irradiated using a 20 W CFL for 10 h in the air.b Determined by recovered glycinate 5a.c Isolation yields. | ||
| No | 100 | 85 |
| DABCO (2 equiv. for 5a) | 100 | 83 |
| TEMPO (2 equiv. for 5a) | 52 | 38 |
| BHT (2 equiv. for 5a) | 43 | 26 |
Based on the above observations and the previous reports,17–20,48–51 a feasible mechanistic pathway responsible for the MB-sensitized photocyclization reactions was suggested (Scheme 6). Through visible-light irradiation, SET occurs from glycinates 17 to the photochemically formed triplet state of MB (3MB*) to produce glycinate radical cations and radical anions of MB (MB˙−). While MB˙− ions subsequently react w ith O2 to regenerate the ground-state MB and O2˙−, the generated glycinate radical cations 18 undergo solvent-assisted desilylation to produce α-amino radicals 19. These intermediates then add to the ene moiety of the maleimides to produce radical intermediates 20, which undergo intramolecular cyclization reactions, followed by dehydrogenative oxidation, to form tetrahydroquinolines 22. When the ortho-chlorophenyl substituted glycinate 17 (X = o-Cl) is employed, its radical cation 18 (X = o-Cl) can also undergo competitive H-atom abstraction to form iminium ion 23, which is subsequently hydrolyzed to yield secondary amine 11.
Conclusion
We investigated SET-oxidative photocyclization reactions of electron-deficient N-(α-trimethylsilyl)-N-phenylglycinates with N-substituted maleimides under visible light using the organic photosensitizer methylene blue (MB). The results revealed that, unlike their non-silylated N-alkyl-N-phenylglycinate counterparts, N-(α-trimethylsilyl)-N-phenylglycinates underwent efficient formation of α-amino radicals via sequential SET-desilylation processes, in which these radicals subsequently reacted with maleimides to afford tetrahydroquinoline products. Notably, the presence of the silyl group at the α-position to the nitrogen atom proved critical for the success of cyclization reactions. Overall, this photochemical strategy offers a valuable approach for expanding the scope of amine substrates in the diverse SET-promoted photochemical reactions.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article are included in the supplementary information (SI). Supplementary information: experimental section, 1H and 13C NMR spectroscopic data for compounds, and MB photoluminescence quenching data by glycinates. See DOI: https://doi.org/10.1039/d5ob01839k.Acknowledgements
This study was financially supported by grants from the Basic Science Research Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Education, Science, and Technology (RS-2023-00211633 for S. H. Lim), and a Korea Basic Science Institute (National Research Facilities and Equipment Center) grant funded by the Ministry of Education (2019R1A6C1010046).References
- N. A. Romero and D. A. Nicewicz, Chem. Rev., 2016, 116, 10075–10166 Search PubMed.
- J. W. Tucker, J. M. R. Narayanam, S. W. Krabbe and C. R. J. Stephenson, Org. Lett., 2010, 12, 368–371 Search PubMed.
- D. Ravelli, S. Protti and M. Fagnoni, Chem. Rev., 2016, 116, 9850–9913 CrossRef CAS PubMed.
- A. G. Griesbeck, H. Heckroth and H. Schmickler, Tetrahedron Lett., 1999, 40, 3137–3140 CrossRef CAS.
- H. Jiang, Y. Cheng, R. Wang, M. Zheng, Y. Zhang and S. Yu, Angew. Chem., Int. Ed., 2013, 52, 13289–13292 CrossRef CAS PubMed.
- J. Xuan, L. Lu, J. Chen and W. Xiao, Eur. J. Org. Chem., 2013, 6755–6770 Search PubMed.
- D. W. Cho, U. C. Yoon and P. S. Mariano, Acc. Chem. Res., 2011, 44, 204–215 Search PubMed.
- U. C. Yoon and P. S. Mariano, Acc. Chem. Res., 2001, 34, 523–533 CrossRef CAS PubMed.
- J. Hu, J. Wang, T. H. Nguyen and N. Zheng, Beilstein J. Org. Chem., 2013, 9, 1977–2001 CrossRef PubMed.
- J. W. Beatty and C. R. J. Stephenson, Acc. Chem. Res., 2015, 48, 1474–1484 Search PubMed.
- Y. Zou, F. M. Hörmann and T. Bach, Chem. Soc. Rev., 2018, 47, 278–290 RSC.
- D. B. Freeman, L. Furst, A. G. Condie and C. R. J. Stephenson, Org. Lett., 2012, 14, 94–97 CrossRef CAS PubMed.
- X. Ju, D. Li, W. Li, W. Yu and F. Bian, Adv. Synth. Catal., 2012, 354, 3561–3567 Search PubMed.
- T. P. Nicholls, G. E. Constable, J. C. Robertson, M. G. Gardiner and A. C. Bissember, ACS Catal., 2016, 6, 451–457 Search PubMed.
- X. Yang, J. Guo, T. Lei, B. Chen, C. Tung and L. Wu, Org. Lett., 2018, 20, 2916–2920 Search PubMed.
- J. Tang, G. Grampp, Y. Liu, B. Wang, F. Tao, L. Wang, X. Liang, H. Xiao and Y. Shen, J. Org. Chem., 2015, 80, 2724–2732 CrossRef CAS PubMed.
- Z. Liang, S. Xu, W. Tian and R. Zhang, Beilstein J. Org. Chem., 2015, 11, 425–430 CrossRef CAS PubMed.
- A. K. Yadav and L. D. S. Yadav, Tetrahedron Lett., 2017, 58, 552–555 CrossRef CAS.
- J. Guo, D. Yang, Z. Guan and Y. He, J. Org. Chem., 2017, 82, 1888–1894 CrossRef CAS PubMed.
- N. F. Nikitas, M. A. Theodoropoulou and C. G. Kokotos, Eur. J. Org. Chem., 2021, 1168–1173 CrossRef CAS.
- C. Hsu and H. Sundén, Org. Lett., 2018, 20, 2051–2054 CrossRef CAS PubMed.
- S. K. Hota, S. P. Panda, S. Das, S. K. Mahapatra, L. Roy, S. De Sarkar and S. Murarka, J. Org. Chem., 2023, 88, 2543–2549 CrossRef CAS PubMed.
- M. Tang, F. Draper, L. N. Pham, C. C. Ho, H. Huang, J. Sun, S. C. Thickett, M. L. Coote, T. U. Connell and A. C. Bissember, J. Org. Chem., 2024, 89, 2683–2690 CrossRef CAS PubMed.
- G. A. Swan, J. Chem. Soc. C, 1971, 2880–2888 Search PubMed.
- M. Nishino, K. Hirano, T. Satoh and M. Miura, J. Org. Chem., 2011, 76, 6447–6451 CrossRef CAS PubMed.
- S. Murata, K. Teramoto, M. Miura and M. Nomura, Heterocycles, 1993, 36, 2147–2153 Search PubMed.
- N. Sakai, S. Matsumoto and Y. Ogiwara, Tetrahedron Lett., 2016, 57, 5449–5452 Search PubMed.
- A. K. Yadav and L. D. S. Yadav, Tetrahedron Lett., 2016, 57, 1489–1491 CrossRef CAS.
- K. Sharma, B. Das and P. Gogoi, New J. Chem., 2018, 42, 18894–18905 Search PubMed.
- J. Y. Hwang, A. Y. Ji, S. H. Lee and E. J. Kang, Org. Lett., 2019, 22, 16–21 CrossRef PubMed.
- S. Prasanna Kumari, B. Naveen, P. Suresh Kumar and S. Selva Ganesan, Chem. Pap., 2023, 77, 151–158 Search PubMed.
- Z. Song and A. P. Antonchick, Tetrahedron, 2016, 72, 7715–7721 Search PubMed.
- S. H. Lim, M. Kim, K. Wee and D. W. Cho, J. Org. Chem., 2023, 88, 12294–12310 CrossRef CAS PubMed.
- S. H. Lim, M. Ahn, K. Wee, J. H. Shim, J. Choi, D. Ahn and D. W. Cho, J. Org. Chem., 2020, 85, 12882–12900 Search PubMed.
- S. H. Lim, A. B. Atar, G. Bae, K. Wee and D. W. Cho, RSC Adv., 2019, 9, 5639–5648 RSC.
- S. H. Lim, H. C. Jeong, Y. Sohn, Y. Kim, D. W. Cho, H. Woo, I. Shin, U. C. Yoon and P. S. Mariano, J. Org. Chem., 2016, 81, 2460–2473 Search PubMed.
- (a) S. O'Sullivan, E. Doni, T. Tuttle and J. A. Murphy, Angew. Chem., Int. Ed., 2014, 53, 474–478 CrossRef PubMed; (b) V. Pace, W. Holzer, G. Verniest, A. R. Alcantara and N. D. Kimpe, Adv. Synth. Catal., 2013, 355, 919–926 CrossRef CAS; (c) M. Shahid, S. S. Razi, P. Srivastava, R. Ali, B. Maiti and A. Misra, Tetrahedron, 2012, 68, 9076–9084 CrossRef CAS.
- X. Wang, S. Wang, E. Ohkoshi, L. Wang, E. Hamel, K. Qian, S. L. Morris-Natschke, K. Lee and L. Xie, Eur. J. Med. Chem., 2013, 67, 196–207 CrossRef CAS PubMed.
- P. D. Leeson, R. W. Carling, K. W. Moore, A. M. Moseley, J. D. Smith, G. Stevenson, T. Chan, R. Baker, A. C. Foster and S. Grimwood, J. Med. Chem., 2002, 35, 1954–1968 CrossRef PubMed.
- T. Asberom, T. A. Bara, J. W. Clader, W. J. Greenlee, H. S. Guzik, H. B. Josien, W. Li, E. M. Parker, D. A. Pissarnitski and L. Song, Bioorg. Med. Chem. Lett., 2007, 17, 205–207 Search PubMed.
- D. Su, J. J. Lim, E. Tinney, B. Wan, M. B. Young, K. D. Anderson, D. Rudd, V. Munshi, C. Bahnck and P. J. Felock, Bioorg. Med. Chem. Lett., 2009, 19, 5119–5123 Search PubMed.
- H. Steinhagen and E. J. Corey, Org. Lett., 1999, 1, 823–824 Search PubMed.
- M. Ori, N. Toda, K. Takami, K. Tago and H. Kogen, Tetrahedron, 2005, 61, 2075–2104 Search PubMed.
- G. M. Staub, J. B. Gloer, D. T. Wicklow and P. F. Dowd, J. Am. Chem. Soc., 1992, 114, 1015–1017 CrossRef CAS.
- S. P. Pitre, C. D. McTiernan, H. Ismaili and J. C. Scaiano, ACS Catal., 2014, 4, 2530–2535 CrossRef CAS.
- T. Shen, Z. Zhao, Q. Yu and H. A. Xu, J. Photochem. Photobiol., A, 1989, 47, 203–212 Search PubMed.
- R. H. Kayser and R. H. Young, Photochem. Photobiol., 1976, 24, 395–401 CrossRef CAS.
- M. Uygur, T. Danelzik and O. García Mancheño, Chem. Commun., 2019, 55, 2980–2983 RSC.
- D. W. Manley, A. Mills, C. O'Rourke, A. M. Z. Slawin and J. C. Walton, Chem. – Eur. J., 2014, 20, 5492–5500 Search PubMed.
- S. Cheng, J. Liao, Y. Lin and L. Gong, Org. Lett., 2023, 25, 6566–6570 Search PubMed.
- P. Kanti Roy, S. Okunaka, H. Tokudome and Y. Hitomi, Adv. Synth. Catal., 2023, 365, 4556–4561 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |