 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
tert-Butyl hydroperoxide mediated Wacker-type oxidations: taking advantage of hexafluoro-2-propanol for challenging substrates
Calum Maguirea,
Qun Caoab,
Selena C. L. Gilmer c,
Bill C. Hawkins
c,
Bill C. Hawkins *c,
Peter C. Knipe
*c,
Peter C. Knipe *a and
Mark J. Muldoon
*a and
Mark J. Muldoon *a
*a
aSchool of Chemistry and Chemical Engineering, Queen's University Belfast, David Keir Building, Stranmillis Road, Belfast, BT9 5AG, UK. E-mail: P.Knipe@qub.ac.uk; m.j.muldoon@qub.ac.uk
bCollege of Chemical Engineering, State Key Laboratory of Advanced Separation Membrane Materials, Zhejiang University of Technology, Hangzhou 310014, China
cDepartment of Chemistry, University of Otago, Dunedin 9054, New Zealand. E-mail: bhawkins@chemistry.otago.ac.nz
First published on 9th April 2026
Abstract
Wacker-type oxidation of alkene substrates containing heteroatoms, such as allylic ethers, is challenging. Herein we describe the use of a dicationic Pd(II) complex, with tert-butyl hydroperoxide (TBHP) as the oxidant and how hexafluoro-2-propanol (HFIP) can improve performance. A series of substituted O-allylphenols and more complex derivatives were studied.
In organic synthesis, the conversion of a terminal alkene to a methyl ketone is a highly valuable transformation. Such Wacker-type oxidation reactions have been studied for decades, but their utility means that there is continued interested in improving methods. Palladium based catalysts have been the most prevalent,1,2 although in recent years, methods using first-row transition metals have been emerging.3 A variety of methods have been reported for palladium, using different additives and different terminal oxidants. In comparison to other areas of catalysis, (e.g. Pd catalyzed cross coupling) Wacker-type oxidation reactions lag significantly behind in terms of performance. Overall, there is a need to improve on key issues such as general reaction conditions, catalyst loading and product selectivity. Allylic ethers are a good example of a challenging class of substrates; the presence of the nearby oxygen can result in hindered catalytic performance and mixtures of products. Wacker-type oxidations are often used in target-oriented syntheses, where allylic ether functionality is present in relatively complex substrates.4–8 Even for a relatively simple substrate such as allyl phenyl ether (1a), methods often require undesirable conditions, high catalyst loadings or result in low product yield.9–15 In Fig. 1, we illustrate some recent examples of aerobic methods16,17 and a tert-butyl hydroperoxide (TBHP) method.18 The TBHP method reported by Zou and colleagues,18 is arguably one of the best in terms of product yield and catalyst loading, although the use of elevated temperatures with peroxides such as TBHP is not ideal. Their method uses hexafluoro-2-propanol (HFIP) as the solvent. We have also been interested in solvent effects on Wacker-type oxidations and the work by Zou was reported while we were carrying out our own investigations with HFIP.
 | ||
| Fig. 1 Examples of the Wacker-type oxidation of allyl phenyl ether (1a) to phenoxy-2-propanone (1b). | ||
Our studies on Wacker-type oxidation reactions have explored the use of dicationic Pd(II) complexes. Sigman and co-workers were first to show that such complexes were highly active for TBHP mediated Wacker-type oxidations.19–23 The use of complexes with weakly coordinating anions enables more facile binding of substrates, and cationic complexes are known to improve the electrophilic activation of metal bound alkenes.24 Sigman's quinoline-2-oxazoline (Quinox) catalyst system (Fig. 2A) was able to operate effectively under mild conditions. Their method generated the dicationic complex in situ using silver hexafluoroantimonate to abstract the chloride ions from the palladium complex.
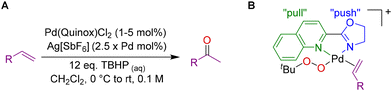 | ||
| Fig. 2 A. General conditions for TBHP Wacker with Sigman's Quinox catalyst. B. Illustration of the ligand's electronic asymmetry and a proposed intermediate. | ||
Sigman and co-workers carried out mechanistic studies,22 and found that the electronic asymmetry of the Quinox ligand was beneficial for these reactions. They examined a series of functionalised ligands and carried out a Hammett analysis which led to a “push–pull” hypothesis (Fig. 2B). It was found that a Quinox derivative with a CF3 functionalised quinoline led to faster rates. There were still some challenges though, as such ligands were more difficult to prepare than standard Quinox, and although they exhibited higher initial rates, this did not translate to lower catalyst loadings as the turnover number (TON) was similar. Nonetheless, these seminal studies laid the foundation for catalyst design and greatly increased the understanding of these systems. Since then, we25–29 and other groups have explored the use of dicationic and/or electronic asymmetric ligands for Wacker reactions.18,30–33 In addition to the mechanistic work by Sigman and co-workers,22 we (Muldoon) have carried out experimental mechanistic work with Zare and Waymouth26 and computational studies with Waymouth and García-Melchor.28 In the interest of brevity, we have highlighted some key elements in a simplified version of the catalytic cycle (Fig. 3A). For more detailed discussion and supporting evidence, please see the aforementioned studies.
In 2023, we reported a new dicationic complex which used 2-(5-(trifluoromethyl)pyridin-2-yl)benzo[d]oxazole (5-CF3-PBO) as a ligand. This ligand was shown to enable very effective catalysis for TBHP mediated Wacker-type oxidation reactions.27 An advantage of the PBO ligand scaffold versus Quinox is that a range of PBO derivatives can be readily prepared. In that study we examined 22 PBO derivatives and Quinox as a benchmark. In line with Sigman's observations on electronic asymmetry, the best results were obtained with the 5-CF3-PBO ligand. We also examined isolated dicationic complexes as well as those generated in situ with silver salts (with several different anions). Overall, isolated complexes gave better results, although for some substrates/conditions, there was little difference. The optimal catalyst is shown in Fig. 3B, which has triflate ([OTf]−) counterions, which are more stable than [SbF6]−, a species that readily undergoes hydrolysis.34
For cationic catalysts, it is important to use weakly coordinating solvents and methylene chloride had previously been used in the Sigman system. Methylene chloride is a popular choice for cationic catalysts, because it is polar but weakly coordinating.35–37 In our studies, we found that trifluorotoluene improved catalyst TON in comparison to methylene chloride. Additionally, we showed how the presence of HFIP could lead to improved catalytic performance for the oxidation of oct-1-en-3-yl acetate. This was a model substrate for a class of substrates (protected allylic alcohols) which had previously been shown to be challenging.19,22 HFIP is weakly coordinating but with strong hydrogen bond donor abilities. In our studies, it was concluded that hydrogen bonding between HFIP and the Lewis basic sites on this substrate (and product) enables the Pd(II) catalyst to oxidise the substrate more efficiently. HFIP was also found to improve catalyst stability. Control experiments and diffusion-ordered spectroscopy (DOSY) NMR studies indicated that HFIP slowed down catalyst deactivation. Water and TBHP were thought to be responsible for this aggregation, and the presence of HFIP was found to slow down the rate of aggregation.
We wanted to explore this method for other challenging substrates, such as allylic ethers, which as mentioned earlier, are known to be difficult for Wacker-type oxidation reactions. In addition, we (Hawkins) had explored the use of Wacker oxidation in studies towards the synthesis of oxyisocyclointegrin.8 In these studies, it was found that Sigman's Quinox/TBHP method,19 was superior to aerobic Tsuji–Wacker conditions. Nonetheless, a catalyst loading of 20 mol% was required to oxidize 16a (product 16b shown later in Fig. 5). Herein, we discuss the oxidation of model allylic ether substrates along with more complex substrates.
We began by examining allyl phenyl ether (1a), using a variety of conditions similar to those we had employed in our earlier studies.27 We tested the isolated [Pd(5-CF3-PBO)(S)2][OTf]2 complex, and the one prepared in situ from Pd(5-CF3-PBO)Cl2 and Ag[OTf]. We began with the in situ method and using anhydrous conditions; with a solution of TBHP in trifluorotoluene. It was clear that HFIP was beneficial to these reactions (Fig. S1), but it was found that using water-free conditions was not ideal for this substrate. Although high yields of product could be obtained, at longer reaction times the yield diminished as the catalyst began to decompose the product (Fig. S1–S3). We found that using aqueous TBHP prevented this decomposition (Fig. S4). In addition, the use of aqueous TBHP is advantageous as it is a commercial product. We found that the catalyst generated in situ gave similar results to the isolated complex (Fig. S9) and that 20 equivalents of HFIP delivered more consistent catalytic performance than 10 equivalents (Fig. S11 and S12). We then decided to use the isolated complex for the remainder of the studies as there are no concerns about substrate/product interactions with the silver salt and reactions are fully homogeneous. Fig. 4 shows that 1a is converted to 1b rapidly with 1 mol% of this complex at 27 °C.
A higher yield of product was obtained when we started the reaction at 0 °C, an approach previously used by Sigman and co-workers.19 It was found that when the reaction was held at 0 °C for 30 minutes, then transferred to a pre-warmed heating block at 27 °C, the reaction affords a higher final 1b yield (see also Fig. S13). We have observed that for substrates which react fast at room temperature there is considerable heat evolved. This is not ideal when peroxides are present; in addition, the heat likely leads to faster catalyst deactivation. Holding the reaction at 0 °C for the entire duration did not give any further improvement (Fig. 4). At 1 mol% catalyst loading, raising the HFIP content afforded little improvement (Fig. S14). However, with 0.5 mol% catalyst loading, a noticeably higher yield of 1b was obtained with 40 eq. HFIP than with 20 eq. (Fig. S15), though with even greater HFIP content, the yield of 1b became diminished, which correlates with our previous findings where the dicationic catalyst was unstable in higher (neat) HFIP content.27 The conditions were further studied with variations to the method (Fig. S16 and S17), but no further improvement was observed.
Another substrate tested, allyl benzyl ether (2a), could be converted to its methyl ketone in 85% yield (Fig. 5), but this was not improved by the initial lower temperature (Fig. S20). It was also found that extended reaction times led to reduced yields, even when using aqueous TBHP (Fig. S20). This is likely due to over-oxidation of the activated benzylic position in 2b which is absent in the previous substrates.
Next, the functional group tolerance of the catalyst system was inspected, by synthesizing and oxidizing derivatives of 1a (Fig. 6). Initial screening used 1 mol% catalyst loading but some substrates appeared to be less reactive (Fig. S21 and S22). Hence, the catalyst loading was raised slightly to 1.5 mol%. Good yields could be obtained for a number of substrates, and no significant electronic effect was observed. In the case of 13a, the yield of 13b is low and it appears that coordination of the nitrile group inhibits the catalyst, something which is supported by additional additive studies that we carried out. To investigate the system's ability to tolerate functionalities, we used using an approach similar to that developed by Glorius and co-workers.38 We carried out the oxidation of 1a in the presence of other additives (Fig. S23). Halogenated additives (iodobenzene and bromobenzene) had no impact on the reaction, whilst phenol, benzonitrile and benzoic acid markedly reduced product yield. When amines, N,N-diethylaniline and pyridine, N-allylaniline (15a) were screened as additives, no conversion occurred and only unreacted 1a was seen. This is consistent with the lack of conversion obtained for 14a and 15a. The results of the substrate and additive screening indicate substrates including basic nitrogen-containing functionalities are problematic, likely due to strong interactions with the Pd(II) centre under these mild conditions.
Wacker oxidation has important applications in complex organic synthesis, including the oxidation of allylic ethers.4–8 Hence, the system was applied to the oxidation several complex organic structures containing terminal alkenes (Fig. 7). Substrate 16a was converted to 16b in 68% yield, which is comparable to that obtained previously in work by Hawkins and Smith with this substrate using the [Pd(Quinox)Cl2]/Ag[SbF6] system (74%) but at a much lower catalyst loading (5 mol% vs. 20 mol%).8 Substrates 17a and 18a afforded the corresponding ketone products in 74% and 55% yields respectively, showing the method to be compatible with acid labile spirocyclic acetals, as well as tolerating the presence of ester, flavone, bromo and methoxy functional groups. Compatibility with substrates bearing an unprotected allylic alcohol was shown by oxidation of 19a to 19b in 57% yield. Conversion of the acetate-protected 20a to methyl ketone 20b was low, with most of the material recovered found to be the alkene substrate. It is possible that the presence of additional coordinating oxygen atoms hamper activity through coordination with the Pd(II) centre. Indeed, small changes to the structure of such compounds can have a significant effect and we found that for a number of substrates (21a–25a), we were unable to obtain the desired products (see Fig. S25).
We have demonstrated the application of the [Pd(5-CF3-PBO)(S)2][OTf]2 catalyst system for Wacker oxidation of allylic ethers. HFIP was found to play an essential role in enabling high levels of activity. The system generally exhibits excellent performance for a range of allylic ether substrates and has applicability to some complex substrates. Overall, we have demonstrated that our catalyst and conditions are superior to previously reported methods.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). The SI document contains data for additional catalytic conditions not shown in the main article. It also describes detailed experimental protocols along with analytical data for synthesised compounds. See DOI: https://doi.org/10.1039/d5ob01683e.Acknowledgements
We thank Conor McGrann, Donal Moran and Angela Brownlie of Queen's University's ASEP Analytical Services for ESI-MS, elemental analysis and FTIR. For funding, we are grateful to the Department for the Economy (DfE) Northern Ireland for a studentship (C. Maguire).References
- R. A. Fernandes, A. K. Jha and P. Kumar, Catal. Sci. Technol., 2020, 10, 7448–7470 RSC.
- O. Temkin, Kinet. Catal., 2020, 61, 663–720 CrossRef CAS.
- P. Rajeshwaran, J. Trouvé, K. Youssef and R. Gramage-Doria, Angew. Chem., Int. Ed., 2022, 61, e202211016 CrossRef CAS PubMed.
- B. Xu, Y. Feng, B. Lv, G. Xu, L. Zhang, J. Du, K. Peng, M. Xu, J. Dong, W. Zhang, T. Zhang, L. Zhu, H. Ding, Z. Sheng, A. Welihinda, B. Seed and Y. Chen, Bioorg. Med. Chem., 2010, 18, 4422–4432 CrossRef CAS PubMed.
- I. Kadota, H. Ueno, Y. Sato and Y. Yamamoto, Tetrahedron Lett., 2006, 47, 89–92 CrossRef CAS.
- S.-R. Wang, P. A. Sánchez-Murcia, F. Gago and W.-S. Fang, Org. Biomol. Chem., 2016, 14, 345–352 RSC.
- M. L. Lanier, H. Park, P. Mukherjee, J. C. Timmerman, A. A. Ribeiro, R. A. Widenhoefer and J. Hong, Chem. – Eur. J., 2017, 23, 7180–7184 CrossRef CAS PubMed.
- R. J. Smith and B. C. Hawkins, Eur. J. Org. Chem., 2019, 6847–6854 CrossRef CAS.
- J. E. Bäckvall, R. B. Hopkins, H. Grennberg, M. Mader and A. K. Awasthi, J. Am. Chem. Soc., 1990, 112, 5160–5166 CrossRef.
- B. Betzemeier, F. Lhermitte and P. Knochel, Tetrahedron Lett., 1998, 39, 6667–6670 CrossRef CAS.
- I. A. Ansari, S. Joyasawal, M. K. Gupta, J. S. Yadav and R. Gree, Tetrahedron Lett., 2005, 46, 7507–7510 CrossRef CAS.
- D. A. Chaudhari and R. A. Fernandes, J. Org. Chem., 2016, 81, 2113–2121 CrossRef CAS PubMed.
- R. A. Fernandes and V. Bethi, Tetrahedron, 2014, 70, 4760–4767 CrossRef CAS.
- S. Donck, E. Gravel, N. Shah, D. V. Jawale, E. Doris and I. N. N. Namboothiri, ChemCatChem, 2015, 7, 2318–2322 CrossRef CAS.
- M. G. Kulkarni, Y. B. Shaikh, A. S. Borhade, S. W. Chavhan, A. P. Dhondge, D. D. Gaikwad, M. P. Desai, D. R. Birhade and N. R. Dhatrak, Tetrahedron Lett., 2013, 54, 2293–2295 CrossRef CAS.
- S. Tamura, Y. Shimoyama, D. Hong and Y. Kon, Mol. Catal., 2020, 496, 111178 CAS.
- M. Miyazaki and Y. Ura, ACS Omega, 2023, 8, 41983–41990 CrossRef CAS PubMed.
- S. Zhang, J. Zhang and H. Zou, Org. Lett., 2023, 25, 1850–1855 CrossRef CAS PubMed.
- B. W. Michel, A. M. Camelio, C. N. Cornell and M. S. Sigman, J. Am. Chem. Soc., 2009, 131, 6076–6077 CrossRef CAS PubMed.
- B. W. Michel, J. R. McCombs, A. Winkler and M. S. Sigman, Angew. Chem., Int. Ed., 2010, 49, 7312–7315 CrossRef CAS PubMed.
- J. R. McCombs, B. W. Michel and M. S. Sigman, J. Org. Chem., 2011, 76, 3609–3613 CrossRef CAS PubMed.
- B. W. Michel, L. D. Steffens and M. S. Sigman, J. Am. Chem. Soc., 2011, 133, 8317–8325 CrossRef CAS PubMed.
- R. J. DeLuca, J. L. Edwards, L. D. Steffens, B. W. Michel, X. Qiao, C. Zhu, S. P. Cook and M. S. Sigman, J. Org. Chem., 2013, 78, 1682–1686 CrossRef CAS PubMed.
- C. Hahn, Chem. – Eur. J., 2004, 10, 5888–5899 CrossRef CAS PubMed.
- Q. Cao, D. S. Bailie, R. Fu and M. J. Muldoon, Green Chem., 2015, 17, 2750–2757 RSC.
- K. L. Walker, L. M. Dornan, R. N. Zare, R. M. Waymouth and M. J. Muldoon, J. Am. Chem. Soc., 2017, 139, 12495–12503 CrossRef CAS PubMed.
- M. N. Blair, M. Murray-Williams, C. Maguire, C. L. Brown, Q. Cao, H. Chai, Y. Li, R. L. O'Hagan, P. Dingwall, P. Manesiotis, C. L. Lyall, J. P. Lowe, U. Hintermair, P. C. Knipe and M. J. Muldoon, Catal. Sci. Technol., 2023, 13, 6224–6232 RSC.
- M. Mu, K. L. Walker, G. Sánchez-Sanz, R. M. Waymouth, C. Trujillo, M. J. Muldoon and M. García-Melchor, ACS Catal., 2024, 14, 1567–1574 CrossRef CAS PubMed.
- H. Chai, Q. Cao, L. M. Dornan, N. L. Hughes, C. L. Brown, P. Nockemann, J. Li and M. J. Muldoon, Eur. J. Inorg. Chem., 2017, 2017, 5604–5608 CrossRef CAS.
- S. Saha, S. Yadav, N. U. D. Reshi, I. Dutta, S. Kunnikuruvan and J. K. Bera, ACS Catal., 2020, 10, 11385–11393 CrossRef CAS.
- T. Jan, T. Jan, M. A. Bhat and N. U. D. Reshi, Phys. Chem. Chem. Phys., 2025, 27, 23386–23399 RSC.
- L. Dong, Z. Yu, X. Li, X. Huang, J. Fu, L. Chen, J. Zhang and H. Yang, New J. Chem., 2025, 49, 18886–18892 RSC.
- W. Fan, X. Zeng, M. Ying, H. Yang, Z.-G. She and J. Zhang, Tetrahedron Lett., 2026, 156031 CrossRef CAS.
- W. A. Mazeika and H. Neumann, Inorg. Chem., 1966, 5, 309–311 CrossRef CAS.
- P. Longo, L. Oliva, A. Grassi and C. Pellecchia, Makromol. Chem.: Makromol. Chem. Macromol. Chem. Phys., 1989, 190, 2357–2361 CrossRef CAS.
- P. Wasserscheid, C. M. Gordon, C. Hilgers, M. J. Muldoon and I. R. Dunkin, Chem. Commun., 2001, 1186–1187 RSC.
- S. R. Foley, R. A. Stockland, H. Shen and R. F. Jordan, J. Am. Chem. Soc., 2003, 125, 4350–4361 CrossRef CAS PubMed.
- K. D. Collins and F. Glorius, Nat. Chem., 2013, 5, 597–601 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |





![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Analysed by GC-FID with an internal standard.
Analysed by GC-FID with an internal standard. 